Abstract
Objectives:
This guideline provides recommendations for the clinical management of schizophrenia and related disorders for health professionals working in Australia and New Zealand. It aims to encourage all clinicians to adopt best practice principles. The recommendations represent the consensus of a group of Australian and New Zealand experts in the management of schizophrenia and related disorders. This guideline includes the management of ultra-high risk syndromes, first-episode psychoses and prolonged psychoses, including psychoses associated with substance use. It takes a holistic approach, addressing all aspects of the care of people with schizophrenia and related disorders, not only correct diagnosis and symptom relief but also optimal recovery of social function.
Methods:
The writing group planned the scope and individual members drafted sections according to their area of interest and expertise, with reference to existing systematic reviews and informal literature reviews undertaken for this guideline. In addition, experts in specific areas contributed to the relevant sections. All members of the writing group reviewed the entire document. The writing group also considered relevant international clinical practice guidelines. Evidence-based recommendations were formulated when the writing group judged that there was sufficient evidence on a topic. Where evidence was weak or lacking, consensus-based recommendations were formulated. Consensus-based recommendations are based on the consensus of a group of experts in the field and are informed by their agreement as a group, according to their collective clinical and research knowledge and experience. Key considerations were selected and reviewed by the writing group. To encourage wide community participation, the Royal Australian and New Zealand College of Psychiatrists invited review by its committees and members, an expert advisory committee and key stakeholders including professional bodies and special interest groups.
Results:
The clinical practice guideline for the management of schizophrenia and related disorders reflects an increasing emphasis on early intervention, physical health, psychosocial treatments, cultural considerations and improving vocational outcomes. The guideline uses a clinical staging model as a framework for recommendations regarding assessment, treatment and ongoing care. This guideline also refers its readers to selected published guidelines or statements directly relevant to Australian and New Zealand practice.
Conclusions:
This clinical practice guideline for the management of schizophrenia and related disorders aims to improve care for people with these disorders living in Australia and New Zealand. It advocates a respectful, collaborative approach; optimal evidence-based treatment; and consideration of the specific needs of those in adverse circumstances or facing additional challenges.
Off-label prescribing
In this guideline, evidence and expert opinion for the effectiveness of treatments for schizophrenia and related disorders have been reviewed and considered. In some instances, the therapies (e.g. medicines) identified as effective may not be specifically approved for such use in Australia and/or New Zealand.
The use of such therapeutic agents outside their approved indication(s) is sometimes referred to as ‘off-label’ use, and in practice, this may impact eligibility for third-party payer subsidy. We recommend careful documentation supporting your clinical use of specific therapeutic agents over alternatives that are approved in your country. It is also recommended that this issue is explained to patients, including informing them that they may personally have to meet added costs due to lack of third-party payer subsidy.
Introduction
This clinical practice guideline (CPG) was developed on behalf of the Royal Australian and New Zealand College of Psychiatrists (RANZCP). It updates the previous RANZCP CPG for the management of schizophrenia and related disorders (McGorry et al., 2005). It takes into account the findings of new research published since the previous edition and reflects an increasing emphasis on early intervention, physical health, psychosocial treatments and improving vocational outcomes.
It is important to acknowledge the extraordinary courage and perseverance shown by many people with schizophrenia, who often live with the burdens of symptoms, stigma, social exclusion and socioeconomic disadvantage. Their families and carers also face significant ongoing challenges.
Scope and purpose
This guideline provides recommendations for the clinical management of schizophrenia and related disorders for health professionals working in Australia and New Zealand. It aims to encourage all clinicians to adopt best practice principles. The recommendations represent the consensus of a group of Australian and New Zealand experts in the management of schizophrenia and related disorders.
It is intended mainly for psychiatrists, psychiatry trainees, resident medical officers and hospital interns in psychiatry. It may also be useful to general practitioners (GPs), mental health nurses, other clinicians who work with people with schizophrenia and related psychoses, and policy makers. It does not override the responsibility of clinicians to make appropriate decisions, taking into account the unique circumstances of the person they are treating.
The scope of this guideline is the schizophrenia spectrum which includes schizophrenia, schizoaffective disorder, schizotypal disorder, schizophreniform disorder and acute transient psychotic disorder with symptoms of schizophrenia. The spectrum notion is based on the presumption (and partial evidence) of a shared genetic background. Notably, persistent delusional disorder is not part of the spectrum. This guideline includes the management of ultra-high risk syndromes, first-episode psychoses (FEPs) and prolonged psychoses, including psychoses associated with substance use. Childhood onset schizophrenia, a very rare condition requiring specialist management, is not included. The prescription of antipsychotic drugs for conditions other than schizophrenia and related disorders is also beyond the scope of this CPG. Psychoses associated with affective disorders are covered in the RANZCP CPG on mood disorders (Malhi et al., 2015). This CPG does not include detailed information about mental state examination, diagnostic criteria and differential diagnosis – these matters are covered in textbooks and manuals of diagnostic criteria.
Method
Developing the recommendations and commentary
The working group planned the scope and individual members drafted sections according to their area of interest and expertise, with reference to existing research literature and reviews. In addition, experts in specific areas contributed to the relevant sections. The working group considered recent international CPGs, including the UK National Institute for Health and Care Excellence (NICE) (2014) clinical guideline on the treatment and management of psychosis and schizophrenia in adults and the World Federation of Societies of Biological Psychiatry guidelines for the biological treatment of schizophrenia (Hasan et al., 2012, 2013, 2015; Thibaut et al., 2015). The staging model (McGorry et al., 2006) provided a framework for assessment and clinical management and for the choice and timing of interventions.
For intervention studies, levels of evidence were assigned according to Australian National Health and Medical Research Council (NHMRC) (2009b) levels of evidence (Table 1).
NHMRC levels of evidence for intervention studies.
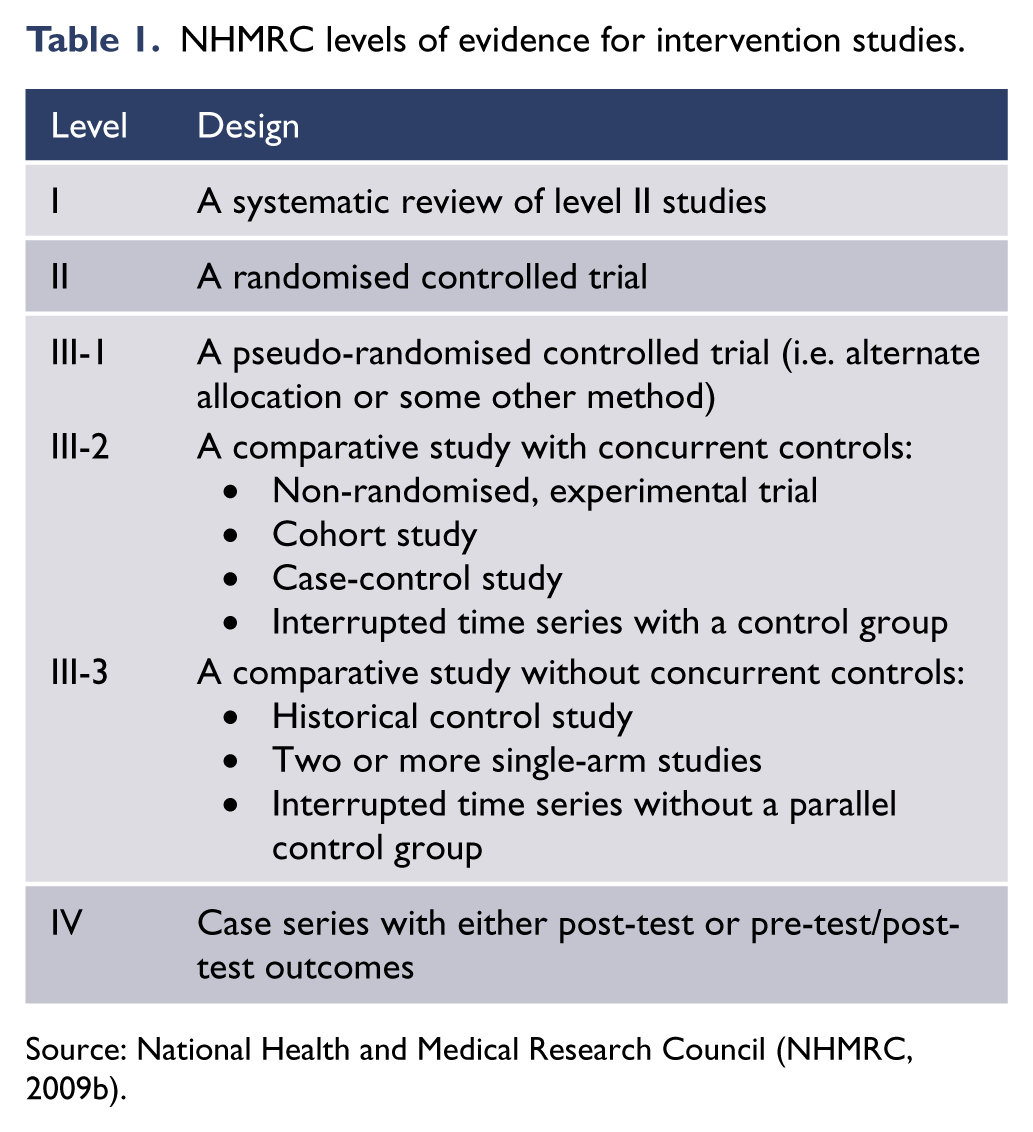
Source: National Health and Medical Research Council (NHMRC, 2009b).
Evidence-based recommendations (EBRs) were formulated when the working group judged that there was sufficient evidence on a topic. For each EBR, the number indicates the level of evidence on which the recommendation was based. Where evidence was weak or lacking, consensus-based recommendations (CBRs) were formulated. CBRs are based on the consensus of a group of experts in the field and are informed by their agreement as a group, according to their collective clinical and research knowledge and experience. Where applicable, key considerations were selected from the recommendations on each topic.
The whole working group reviewed the entire manuscript, and discussion occurred via a series of teleconferences. Where members disagreed about clinical advice or interpretation of evidence, the issue was discussed until consensus was reached.
This guideline also refers readers to selected current documents, statements or algorithms that have been published elsewhere. These were either developed in Australasia or by expert groups led by, or including, Australasian experts.
Expert review and public consultation
A draft version of this guideline was reviewed by experts in schizophrenia (clinical and academic) from Australia and New Zealand, between 1 July and 25 July 2014. A total of 11 expert advisers provided their feedback. The working group considered all reviewers’ comments and revised the manuscript in response to their suggestions.
A revised version of the guideline was released for public consultation between 2 February and 5 March 2015. During the consultation period, the draft guideline was publicly available on the RANZCP website. To encourage wide community participation, RANZCP invited review by key stakeholders including RANZCP Fellows and trainees, professional bodies, special interest groups and consumer and carer groups. The reviewers were asked to respond via an online survey, which asked the following four questions for each section of the guideline:
Are there any significant gaps (of topic, literature, other)?
Are there errors in the content?
Is the structure logical and easy to use?
Do you have any other comments?
Key stakeholders who provided feedback are listed in the ‘Acknowledgements’ section. The working group considered all feedback received from the expert advisers and broader consultation process and modified the draft where necessary. The revised draft was then reviewed by the following RANZCP committees:
Committee for Therapeutic Interventions and Evidence-Based Practice
Practice, Policy and Partnership Committee
Corporate Governance and Risk Committee.
Several amendments were made and final draft was sent to the RANZCP Board in October 2015 for approval. The approved draft was sent to the Australian & New Zealand Journal of Psychiatry (ANZJP) for peer review and publication.
Expert advisors, professional bodies and special interest groups are listed in the ‘Acknowledgements’ section.
Declaration of interests
Members of the schizophrenia CPG working group were required to sign a deed of undertaking at the time of appointment, in which they agreed to declare any conflict, whether actual, potential, perceived or likely to arise in matters considered by the working group.
The process for management of conflict of interest (COI) was detailed in the working group terms of reference: as part of the standing items of any meeting, declarations of COI were called for and minuted. Once a COI had been declared, the individual concerned could be excluded from the discussion at the discretion of the Chair.
The working group members signed an updated COI form at the time of submitting the guideline for publication. All members and their declaration of interests are listed in Appendix 1.
Background
What is schizophrenia?
Schizophrenia is a complex disorder of brain function with wide variation in symptoms and signs, and in the course of the illness. The experiential ‘core’ of schizophrenia has been described as a ‘disturbance involving the most basic functions that give the normal person a feeling of individuality, uniqueness and self-direction’ (World Health Organization [WHO], 1992). The deficits in neurological, psychological and social function that manifest in the various syndromes of schizophrenia appear to have a number of genetic and environmental causes.
Schizophrenia is the most common and most important disorder within a spectrum of clinically similar (and possibly genetically related) conditions, which include schizoaffective disorder, schizotypal disorder and acute transient psychotic disorders (Jablensky, 2006). The term ‘schizophrenia’ includes a range of clinical presentations and personal experiences that result from complex interactions between genes and the environment, and are influenced by the person’s reaction to their experience of the disorder.
Variation in the incidence and prevalence of schizophrenia between populations is greater than was once believed (Simeone et al., 2015). As many as 1% of people meet diagnostic criteria for the disorder over their lifetime. Schizophrenia often has profound effects on people with the disorder and their families. In terms of the global burden of disease and disability, schizophrenia ranks among the top 10 disorders worldwide (Mathers and Loncar, 2006).
Clinical presentation and diagnosis
There is currently no validated biological marker of schizophrenia. The diagnosis is made by identifying the symptoms and signs of the disorder, which include delusional beliefs, hallucinations, disorganised thinking and speech, cognitive impairment, abnormal motor behaviour and negative symptoms. While neuroimaging and cognitive testing may help to rule out alternatives, such as schizophrenia-like manifestations of other disorders affecting brain function, schizophrenia is essentially a clinical diagnosis. The syndrome of schizophrenia, as defined in the Diagnostic and Statistical Manual of Mental Disorders (DSM) and International Classification of Diseases (ICD) classification systems, can be diagnosed with a high degree of inter-rater reliability. However, the validity of the syndromal diagnosis is currently under examination due to the variation in the course and clinical presentation of the disorder.
Since the onset of psychotic symptoms is often preceded by a prolonged period of disturbance of cognition, affect and behaviour, the importance of a thorough exploration of the person’s developmental history and premorbid personality cannot be overestimated. Among the presenting symptoms, those of particular importance for the diagnosis include persistent delusions and hallucinations, incongruent or blunted affect, and interruptions in the train of thought. Characteristic ‘first-rank’ symptoms (thought insertion, withdrawal or broadcasting, hallucinatory voices commenting on the person’s behaviour and passivity experiences) may be present, but are not pathognomonic for schizophrenia. Depressive symptoms and anxiety are common. In differential diagnosis, one should consider other primary psychotic disorders as well as a range of neurological and systemic medical conditions (Cardinal and Bullmore, 2011; Freudenreich et al., 2009). The differentiation of schizophrenia from substance-induced psychotic disorders may be particularly difficult and should be based on a careful review of the temporal relationship between drug taking and the emergence and persistence of psychotic symptoms (see ‘Section 2. Comorbid substance use’).
Aetiology and pathogenesis
Schizophrenia is a complex, multifactorial disorder. Its aetiology has a major genetic component involving multiple genes of small effect, individual assortments of rare mutations (copy number variations) and molecular pathways that are likely to be heterogeneous, both within and across populations (Sullivan et al., 2012). Environmental factors, ranging from neurodevelopmental insults (e.g. maternal pregnancy complications and birth complications) to psychosocial adversity and substance misuse, interact with genetic susceptibility to produce widespread phenotypic variation (Demjaha et al., 2012; Gottesman and Bertelsen, 1989). The ‘social defeat’ hypothesis draws together various environmental risk factors to explain how they might lead to schizophrenia (Selten et al., 2013). Precursors of schizophrenia, including developmental delays, cognitive abnormalities, attenuated symptoms and odd behaviour, may appear very early in life. However, such developmental precursors – if they occur – are usually subtle and are not specific indicators of subsequent psychosis.
Most research into schizophrenia is based on the highly unlikely assumption that schizophrenia is a single, uniform disorder. Research into the various forms of schizophrenia has been assisted by the conceptual tool of endophenotypes, which are heritable, objectively measurable biological traits that co-segregate with clinical illness in pedigrees and may also be expressed in unaffected members. Endophenotypes include distinct patterns on neuropsychological tests of cognitive function, brain electrophysiological measures and neuroimaging variables (Thibaut et al., 2015). Measures of cognitive deficit, including deficits in memory and attention, executive function and sensory gating, have been shown to be particularly sensitive to the dysfunction that sets people with schizophrenia apart from healthy controls (Nuechterlein et al., 2012).
Prevalence, incidence and lifetime risk
Much has been learnt in recent years about the epidemiology of schizophrenia. As one would expect for a heterogeneous group of disorders, frequency estimates related to the incidence and prevalence of schizophrenia vary (McGrath et al., 2004; Saha et al., 2005). Some variation can be attributed to study methodology, but high-quality multicentre studies show significant variation in the incidence of schizophrenia between nations (Sartorius et al., 1986) and within nations (Kirkbride et al., 2006). The incidence of schizophrenia is higher in men than in women; the ratio of males to females is 1.4 to 1 (Aleman et al., 2003; McGrath et al., 2008). Some migrant groups have substantially higher risks of schizophrenia (Bourque et al., 2011; Cantor-Graae and Selten, 2005), and people born and raised in urban areas have an increased risk of schizophrenia, compared with those born and raised in rural settings (Vassos et al., 2012).
Two Australian nationally representative surveys produced very similar estimates of the treated annual prevalence of broadly defined psychotic disorders among people in contact with mental health services: 4.0 per 1000 in 1997–1998 (Jablensky et al., 2000) and 4.5 per 1000 in 2010 (Morgan et al., 2012). With respect to schizophrenia (a subgroup of the psychotic disorders), the estimated lifetime morbid risk (age range 18–45) is 2.37 per 1000 population (Morgan et al., 2014). The frequency of schizophrenia in Australian Indigenous communities is unknown, but higher rates of admission to psychiatric hospitals, as well as the effects of pervasive social disadvantage, suggest that the prevalence may be higher among Aboriginal people than in the wider society (Hunter, 2013). The prevalence of schizophrenia is thought to be elevated among Māori (Kake et al., 2008). An understanding of the social and cultural contexts of schizophrenia and related disorders among Aboriginal and Torres Strait Islander peoples and Māori is essential for planning clinical care (see ‘Section 5. Specific populations and circumstances’).
Course and outcome
The course of schizophrenia is variable. At least three domains that do not co-vary over time need to be independently assessed: symptom severity, functional impairment (including cognitive deficits) and social and occupational disability. The onset is usually in adolescence and early adult life, coinciding with a developmental stage of incomplete social maturation, educational attainment and acquisition of occupational skills (see ‘Section 1. The stages of schizophrenia: a framework for clinical care’). The intrusion of psychosis at this stage may result in a severely truncated repertoire of social skills, and disrupted vocational and developmental trajectory, which can create lifelong socioeconomic disadvantage and failure to achieve the person’s full potential. Both the positive and negative symptoms of the disease can interfere with the person’s capacity to cope with the expectations of daily life. People with schizophrenia experience particular cognitive difficulties in dealing with complex demands and environments, especially those that involve social interaction. These difficulties can be exacerbated by the societal reaction to individuals manifesting aberrant behaviours, which includes stigma and social exclusion. Interactions between these adverse factors can cause the ‘social breakdown syndrome’ (Gruenberg, 1974) – the cluster of secondary and tertiary impairments in schizophrenia that result in a loss of social support networks and greatly diminished quality of life.
A meta-analysis of 320 studies published between 1895 and 1992 with a total of 51,800 subjects (Hegarty et al., 1994) showed that approximately 40% of people with schizophrenia were reported as improved after a mean follow-up of 5.6 years. A significant increase in the rate of improvement has occurred since the 1960s, likely due to the introduction of antipsychotic medications. Two major, multicentre prospective follow-up studies by the WHO (Hopper et al., 2007; Jablensky et al., 1992) found striking differences between countries in the course and outcome of schizophrenia, but overall, more than 30% had relatively favourable clinical outcomes after 15 and 25 years of follow-up. A long-term follow-up study of people presenting with first-episode schizophrenia showed that about half had good outcomes, and 16% of people with early, unremitting symptoms achieved late-phase recovery (Harrison et al., 2001). There is emerging evidence that early detection and treatment are associated with a better long-term outcome (Craig et al., 2004; Ten Velden Hegelstad et al., 2012).
A 2010 Australian national survey (Morgan et al., 2014) revealed that 6% of participants with schizophrenia experienced a single episode of psychosis followed by good recovery, 54.8% experienced multiple episodes with good or partial recovery between the episodes, and 38.8% of participants showed an unremitting continuous course with deterioration. Despite the high risk of chronic disability, loss of developmental potential and diminished quality of life associated with schizophrenia, and despite widespread neglect of the care of people with schizophrenia around the world, about one in seven of those who meet the diagnostic criteria for schizophrenia ultimately attain nearly complete recovery (Jaaskelainen et al., 2013). Predictors of recovery include a higher level of premorbid occupational achievement and social competence, lower likelihood of substance use, better insight and a sense of an internal ‘locus of control’ (Hopper et al., 2007). The possibility of complete recovery justifies an optimistic approach, not only early in the course of illness but also in people with persistent, chronic symptoms and disability.
Comorbidity and mortality
Schizophrenia is associated with excess mortality, which has been well documented by epidemiological studies on large cohorts over extended periods. Standardised mortality ratios (SMRs) of 2.6 or higher have been reported, which correspond to a reduction in life expectancy of approximately 20% (Chwastiak and Tek, 2009; Saha et al., 2007). Laursen (2011) found a reduction in life expectancy of 18.7 years for men and 16.3 years for women with schizophrenia. The leading causes of premature death among people with schizophrenia are cardiometabolic diseases, suicide and accidents (Laursen, 2011).
The second Australian National Survey of Psychosis (Cooper et al., 2012) estimated the prevalence of tobacco smoking among people with schizophrenia at 71% in men and 59% in women – more than three times higher than in the general population. Substance use is the most common comorbid health problem (Moore et al., 2012); the addictive use of cannabis, stimulants and nicotine is disproportionately high among people with psychosis and may be related to the underlying neurobiology of the disorder.
Suicide accounts for 28% of the excess mortality in people with schizophrenia, with aggregated SMR of 9.6 for men and 6.8 for women (Saha et al., 2007). Several risk factors have been suggested that are relatively specific to schizophrenia: the combination of young age and male sex, high level of education, the presence of insight, family history of suicide, substance use and the presence of depressive symptoms, hallucinations and delusions (Hor and Taylor, 2010). In an Australian study (Lawrence et al., 2000), the highest suicide risk was found in the first 7 days after discharge from inpatient care.
Social and economic costs
The social and economic costs of schizophrenia are disproportionately high, relative to its incidence and prevalence. Schizophrenia is associated with a greater burden of long-term disability than any other mental disorder.
Recent evidence from epidemiological, clinical and neurobiological research reinforces the view that people affected by schizophrenia are highly responsive to the social environment and that progressive social exclusion is a major contributor to their disablement, low self-esteem and apathy. Deficits caused by the disorder become amplified by environmental factors, resulting in loss of peer networks and social support, loss of meaningful goals and role fulfilment, disuse of acquired skills and knowledge, and downgrading of attitudes and expectations. Keeping people with schizophrenia socially engaged, especially by facilitating their participation in the workforce through supported employment, is one of the most effective strategies for countering social exclusion.
In most developed countries, the direct costs of schizophrenia (incurred by hospital or community-based treatment, supervised accommodation and related services) amount to 1.4–2.8% of national health care expenditure and up to 20% of the direct costs of all mental health conditions. The indirect costs are likely to be comparable in scale to the direct costs, considering lost productivity and employment and the economically devastating long-term impact of the illness on families and other caregivers. A detailed analysis of the differences between the first (1997–1998) and second (2010) Australian National Survey of Psychosis revealed minimal change in the overall average annual costs, but a significant redistribution of specific expenditures (Neil et al., 2014). This change was broadly related to the reforms driven by the National Mental Health Plan (Australian Government Department of Health, 2009), which prioritised community-based outpatient care and services provided by the non-governmental organisation (NGO) sector. There were significant increases in the average annual cost for outpatient care (increased by AUD$7,380 per person): NGO services (increased by AUD$2,448 per person) and pharmaceuticals (increased by AUD$1,892 per person). These increases were offset by a significant reduction of inpatient costs (reduced from AUD$22,715 to AUD$10,925 per person) and a 6.5-fold decrease in NGO crisis accommodation costs (reduced by AUD$604 per person).
However, productivity losses (the greatest component cost) did not change, indicating the constancy of low employment levels of people with schizophrenia. The proportion of individuals receiving government income support remained at 85%. Given that most people with schizophrenia are smokers and regularly purchase tobacco products, there is little money available for clothing, food, transport and recreational activities. The lack of improvement in workforce participation is very disappointing, given that many people with schizophrenia express a desire to work, and most likely reflects a failure to develop effective vocational rehabilitation services. Improved vocational outcomes would have economic benefits, as well as improving social inclusion and quality of life.
Early intervention for psychotic disorders has been shown to be cost-effective (Hastrup et al., 2013; McCrone et al., 2009). Further research is needed in this area.
Legal, forensic and ethical issues
People with schizophrenia face higher rates of incarceration than the general population, sometimes for violence but also for trivial offences. They are also more likely to be victims of crime. The relationship between psychosis and violence is complex. Delay in the diagnosis and treatment of FEP and loss of continuity of care of people with disturbing symptoms and comorbid substance use have been shown to contribute to episodes of violence (Nielssen et al., 2012). The balance between personal rights, the propensity to harm others and recognition of the need for treatment are the main considerations in decisions to provide involuntary treatment (see ‘Involuntary treatment’ in ‘Section 3. Treatment: context, structure and content of interventions’).
Section 1. The stages of schizophrenia: a framework for clinical care
The concept of staging
Staging is routine for many medical conditions. Over the last decade, a clinical staging model for mental illnesses has been developed (McGorry et al., 2006), which proposes that the course of illness is a continuum (Table 2). Clinical staging models assume that treatments offered earlier in the course of an illness have the potential to be safer, more acceptable, more effective and more affordable than those offered later. Interventions can be evaluated in terms of their ability to prevent or delay progression from earlier to later stages of illness, and can be selected by the individual with schizophrenia and their clinicians on the basis of defined risk/benefit criteria.
Clinical stages of schizophrenia and related disorders.

Source: Adapted from McGorry et al. (2006).
SOFAS: social and occupational function assessment scale; GAF: global assessment of function.
At all stages, the therapeutic relationship is the foundation of clinical care. Time must be spent building trust and good communication. This is just as important for people with unremitting illness, as it is for those early in the course of the disorder. It is essential to take a respectful approach, provide accurate information and address the person’s questions and concerns. People with thought disorder, or other difficulties with conversation, are generally capable of meaningful communication and will often appreciate the opportunity to express their point of view and participate in clinical decision-making (Galletly and Crichton, 2011).
Comorbid substance abuse, which is very common among people with schizophrenia, can complicate the presentation and worsen outcomes (see ‘Section 2. Comorbid substance use’).
The pre-psychotic or prodromal stage (stages 1a, 1b)
Before the emergence of positive psychotic symptoms that are sufficiently severe and persistent to justify a diagnosis of schizophrenia or FEP, most people show a prolonged period of symptoms and increasing disability. This is commonly termed the ‘prodrome’ in retrospect, and the ‘ultra-high-risk mental state’ or ‘at-risk mental state’ prospectively. The pre-psychotic or prodromal stage is associated with evidence of changes in brain structure, probably reflecting active neurobiological processes (Tognin et al., 2014). The neuropathological basis of these changes remains unclear.
A 2012 meta-analysis (Fusar-Poli et al., 2012) showed that among people identified as being at high risk of psychosis, rates of transition to psychosis were 22% at 1 year and 36% at 3 years. However, prediction of transition to psychosis is unreliable and some recent studies show transition rates under 10%. Those who do not transition to psychosis mostly have mood/anxiety disorders, which need appropriate treatment (Fusar-Poli et al., 2013; Lin et al., 2012).
The potential benefits of identifying and proactively treating individuals at risk of psychosis are significant because much of the psychosocial disability that becomes entrenched in the subthreshold period, prior to FEP, is difficult to reverse even when the core symptoms remit with effective treatment. People are also at risk of suicidal behaviour during the pre-psychotic or prodromal stage (Kelleher et al., 2013).
The ultra-high risk or at-risk mental state typically affects young people, usually aged between 14 and 35 years. It is characterised by a change in subjective experience and behaviour that is persistent and often progressive (although it may fluctuate in severity), together with either of the following:
Subthreshold positive symptoms that are not severe or persistent enough to be regarded as evidence of sustained frank psychosis sufficient for a diagnosis of a psychotic disorder (other than brief psychotic episode) according to the Diagnostic and Statistical Manual of Mental Disorders–Fifth Edition (DSM-5) criteria (American Psychiatric Association, 2013).
A family history of psychotic disorder or schizotypal disorder in a first-degree relative, plus a significant yet non-specific decline in psychosocial function within the past year or so that is not resolving.
In addition, to meet ultra-high risk criteria, there must be a functional decline to caseness or chronic poor function (30% drop in the Social and Occupational Functioning Assessment Scale in previous 12 months or score < 50 for previous 12 months).
Intervention during the pre-psychotic stage has several potential benefits:
Intervention may prevent or lessen social disability, which can develop during this stage.
Engagement with mental health professionals can occur while the individual is not experiencing acute illness (Yung, 2003).
Trauma and stigma might be lessened because there is less risk of acute behaviour problems or embarrassing behaviour, and less need for hospitalisation. However, this consideration needs to be balanced against the potential risk of self-stigmatisation, especially when a false positive diagnosis is made (Malla and Norman, 2002).
The duration of untreated psychosis might be reduced; if FEP does occur, it can be recognised rapidly in well-engaged individuals.
Transition to FEP may be delayed or prevented (Van der Gaag et al., 2013).
These potential benefits need to be balanced against the potential risks of overtreatment and ‘labelling’.
There is a considerable body of research examining the effectiveness of treatments during the pre-psychotic phase. A number of randomised controlled trials (RCTs) have investigated whether interventions can prevent or delay transition to psychosis in a proportion of people at risk (Van der Gaag et al., 2013). Interventions that have been evaluated include cognitive behaviour therapy (CBT), antipsychotic medication, a combination of CBT and antipsychotic medication, an intensive treatment that included family intervention, and fish oil supplementation (Stafford et al., 2013). There is evidence that transition to psychosis can be delayed (Van der Gaag et al., 2013), and further clinical trials, with longer follow-up periods, are needed to ascertain whether transition can be completely averted. Meanwhile, early intervention for people who meet criteria for an ultra-high risk mental state should be supported. Besides the obvious benefits to the individual, their family, and the wider community, there are also economic advantages to preventing transition to psychosis (Ising et al., 2015).

CBT: cognitive behaviour therapy; EBR: evidence-based recommendation.
FEP (stage 2)
FEP is defined as 1 week or more of sustained positive symptoms above the psychosis threshold for delusions and hallucinations in particular. There are a range of diagnoses captured here with about 60% falling within the schizophrenia spectrum at this point although more ‘graduate’ later. The purposes of early intervention in FEP are to ensure the safety of the young person and others, to reduce the duration of untreated psychosis as much as possible and to preserve and restore function, thereby reducing the disability associated with psychotic illness. Management of FEP requires a holistic, systematic approach which involves a comprehensive range of pharmacological and psychosocial interventions. More comprehensive information is available at www.ranzcp.org/Publications/Guidelines-for-clinical-practice/Schizophrenia-early-psychosis-resources.aspx. There is evidence for the cost-effectiveness of specialised early psychosis services (Hastrup et al., 2013; Park et al., 2014).
A comprehensive assessment including physical health screening (Table 3) is essential (Freudenreich et al., 2009).
Physical assessment and investigations for people presenting with first-episode psychosis.

Source: Adapted from Freudenreich et al. (2009).
ESR: erythrocyte sedimentation rate; HIV: human immunodeficiency virus; NMDAR: N-methyl-
Liaise with neurology colleagues on the interpretation of test results.
Expert opinion is divided about whether MRI scan of the brain is necessary for all people with first-episode psychosis.
It may be necessary to wait until the mental state is stable before performing psychometric testing.
Duration of untreated psychosis
While there has been debate about the impact of the duration of untreated psychosis (Killackey and Yung, 2007), this has been shown to be an independent predictor of outcome (Penttilä et al., 2014). Reducing the duration of untreated psychosis is, therefore, an important aim. Research has shown that combining awareness-raising campaigns with a readily available FEP service leads to a significant reduction in the duration of untreated psychosis (Joa et al., 2008).
Managing the critical period
People experiencing a first episode of psychosis need comprehensive treatment that continues throughout the entire critical period. The critical period is defined as the first 5 years for a subset of people (Birchwood and Fiorillo, 2000). For all others, there is international consensus that treatment should continue for at least 2 years (Orygen Youth Health Research Centre, 2011).
The OPUS study in Denmark compared people accessing a FEP service with those receiving standard care. There were significant differences between groups in favour of intervention at the end of the 2-year treatment period (Thorup et al., 2005). After the 2 years of specialised first-episode treatment, participants transferred to standard care, and when these participants were followed up at 5 years, most of the gains made at the 2 year assessment had been lost (Bertelsen et al., 2008). It has been shown that longer term (5 years) continuity of care within a specialised early intervention programme is associated with continuing benefits (Norman et al., 2011) and that FEP services can achieve gains that are still measurable 10 years later (Ten Velden Hegelstad et al., 2012).
Prescribing considerations
A treatment algorithm for pharmacological treatment for first-episode non-affective psychosis is provided in Figure 1.
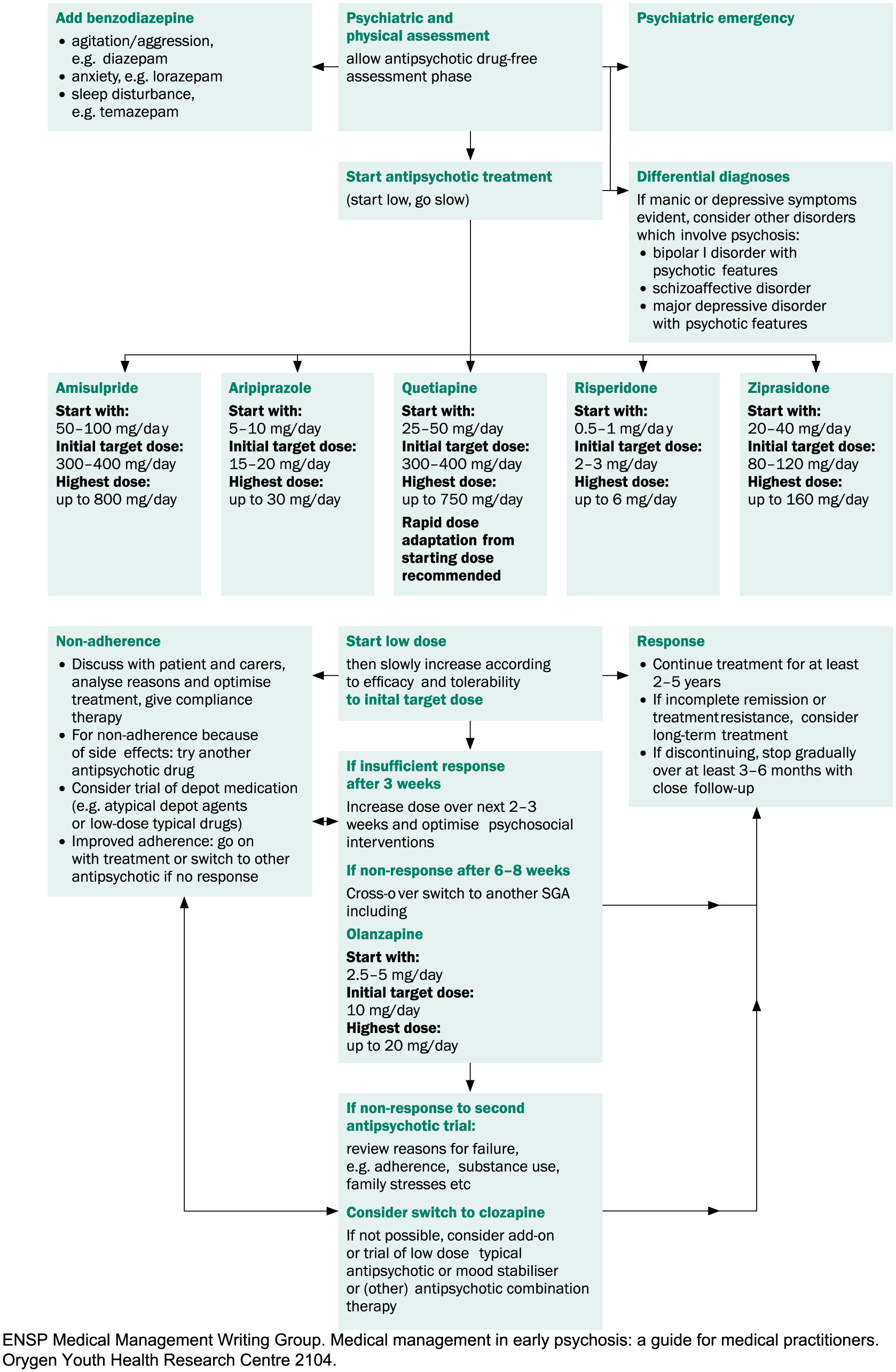
Pharmacological treatment for first-episode non-affective psychosis.
Young people are particularly sensitive to the side effects of psychotropic medicines. A low starting dose with gradual titration up to an effective level is recommended, unless the young person is acutely mentally ill and requires urgent treatment. Second-generation antipsychotic agents (SGAs) are recommended in preference to first-generation antipsychotic agents (FGAs) due to their better tolerability and extrapyramidal side-effect profile (Kahn et al., 2008) (see ‘Antipsychotic medication’ in ‘Section 3. Treatment: context, structure and content of interventions’). There is some evidence of improved relapse prevention with SGAs, compared with FGAs, although this meta-analysis included only oral medications, and the most common FGA comparator was haloperidol (Kishimoto et al., 2013).
There are high levels of non-adherence to medication among people with psychotic disorders. Adherence should be proactively and sensitively addressed. There is a need for careful ongoing monitoring of medication in this stage of illness, combined with a willingness to decrease dosages. This is likely to work better in the presence of a multi-dimensional psychosocial programme to assist recovery.
Attempting to avoid relapse has been a rationale for medication in the management of FEP. Many researchers have emphasised the need for ongoing treatment to prevent relapse (Emsley et al., 2012) and the negative impact of relapse on the long-term course of the disorder (Emsley et al., 2013a). However, a recent study (Wunderink et al., 2013) observed that compared with a standard maintenance treatment regimen, dose reduction or supervised discontinuation of antipsychotic medication during the early phases of FEP led to a higher relapse rate initially, but improved long-term outcomes. This study has been criticised for its unequal distribution across diagnostic groups, high attrition rate, failure to separate the dose reduction and discontinuation groups, and the fact that most patients in each arm of the study did receive medication. These findings have not been replicated. See also ‘Antipsychotic medicines for FEP’ in ‘Section 3. Interventions: context, structure and content’.
Non-pharmacological approaches
For people with FEP, as for all people with psychotic illness, medication is only a part of the required treatment. Studies have shown that an early functional recovery is more predictive of a long-term full recovery than early symptomatic recovery alone (Álvarez-Jiménez et al., 2012a). This finding also aligns with the goals of young people with FEP. The top five goals, in order, are employment, education, housing, relationships and health (Iyer et al., 2011; Ramsay et al., 2011). Therefore, it is imperative to provide psychosocial interventions designed to achieve these goals.
Compared with standard treatment, comprehensive FEP services have demonstrated greater family satisfaction and lower family burden (Jeppesen et al., 2005). In addition, the provision of family psychoeducation and family group therapy can help reduce relapse and readmission rates (McNab and Linszen, 2009).
Unemployment is a significant area of disability for people with psychosis (Killackey et al., 2006). Education, the foundation of career, is often disrupted through the onset of illness (Waghorn et al., 2012). Working is one of the defining normative activities of adults in all societies. It is also the main avenue by which individuals achieve independence and social and economic inclusion. It therefore makes sense to start rehabilitative efforts in this area during the first episode. By doing this, a track record of employment is developed, training is completed and the skills associated with job seeking and working are acquired. Supported employment is the most successful of the vocational interventions (Drake et al., 2012), and individual placement and support is the most effective form of supported employment. Two RCTs of individual placement and support in people with FEP have demonstrated vocational recovery in over 80% of participants receiving this intervention (Killackey et al., 2008; Nuechterlein et al., 2008b). A number of other non-randomised trials of individual placement and support in young people with FEP have found that about two-thirds of participants return to school and/or work, and this effect persists for at least 2 years (Rinaldi et al., 2010). See also ‘Vocational rehabilitation’ in ‘Section 3. Treatment: context, structure and content of interventions’.
Physical health should be addressed early in the course of illness (International Physical Health in Youth [iphYs] Working Group, 2013). Long-term physical health outcomes for people with schizophrenia and related disorders are poor (Galletly et al., 2012; Mitchell et al., 2013). Obesity and tobacco smoking are overwhelmingly responsible for the excess mortality (Olfson et al., 2015; Thornicroft, 2011). Weight gain due to side effects of medicines occurs disproportionately early in the course of illness, so monitoring and prevention at this point are obvious interventions (Foley et al., 2013). Monitoring needs to be systematic and regular, and monitoring protocols can assist in managing cardiometabolic health (Curtis et al., 2012; Stanley and Laugharne, 2014). However, no randomised trials assessing the effectiveness of physical health monitoring in people with serious mental illness have been completed (Tosh et al., 2014). There is some evidence that lifestyle interventions can prevent weight gain (Curtis et al., 2015; Daumit et al., 2013; Álvarez-Jiménez et al., 2006). Further research is needed, including replication of these studies and demonstration of enduring long-term benefit (Gates et al., 2015). Metformin has been found to limit weight gain in people with FEP (Jarskog et al., 2013). See also ‘Section 4. Physical health’.
There is currently no evidence to guide smoking cessation efforts in people with FEP. This is another area that requires further research. In the meantime, the recommendations of Mendelsohn et al. (2015) should be followed.
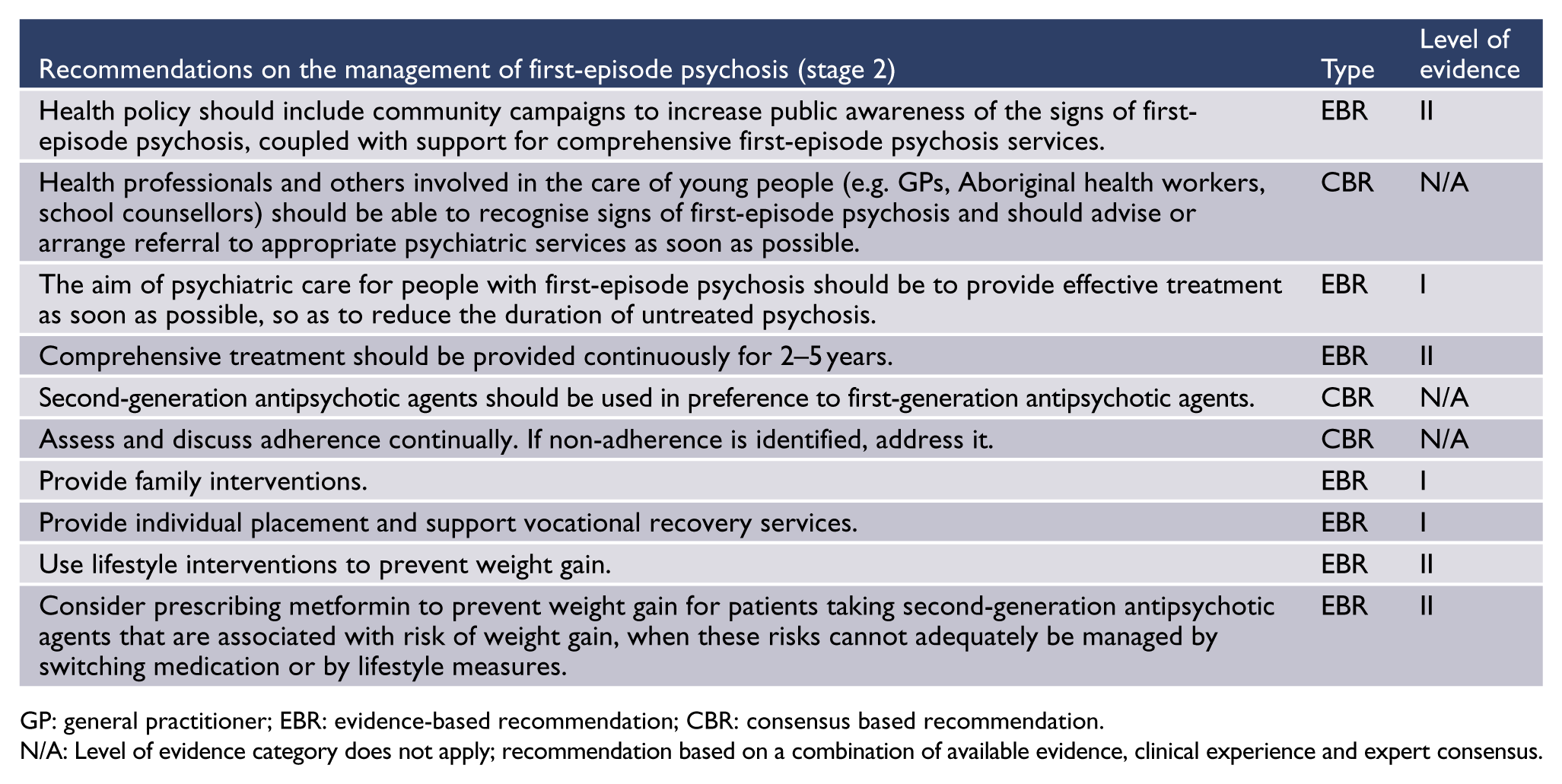
GP: general practitioner; EBR: evidence-based recommendation; CBR: consensus based recommendation.
N/A: Level of evidence category does not apply; recommendation based on a combination of available evidence, clinical experience and expert consensus.
Further guidance on the assessment and management of early psychosis is provided in the Australian Clinical Guidelines for Early Psychosis: www.ranzcp.org/Publications/Guidelines-for-clinical-practice/Schizophrenia-early-psychosis-resources.aspx. The importance of holistic preventive health care in people with FEP is described in the Healthy Active Lives (HeAL) consensus statement 2013; available at www.nationalelfservice.net/cms/wp-content/uploads/2013/06/HeAL-international-concensus-statement.pdf. Information about managing cardiometabolic effects of antipsychotic medication in people with FEP is found in Positive Cardiometabolic Health: an early intervention framework for patients on psychotropic medication, NSW Government Health Education Training Institute (2011) (available at www.heti.nsw.gov.au/cmalgorithm).
Persistent/established illness (stages 3a, 3b, 3c)
A significant proportion of people who have one episode of psychosis will go on to have more episodes or continuing disability. Naturalistic follow-up studies show that the early years after entry to treatment are often characterised by a turbulent early course, which can reduce ultimate levels of recovery (Birchwood and Fiorillo, 2000; Ho and Andreasen, 2001). Relapses are common, with about half of people with FEP relapsing during a 3-year follow-up period (Álvarez-Jiménez et al., 2012b; Robinson et al., 1999). Young people, in particular, find it difficult to accept the lifestyle change of taking medicines daily, especially if they have substantially recovered. Non-adherence to antipsychotic medication is a major risk factor for relapse during this period (Emsley et al., 2013b).
Relapses are disruptive and may contribute to an increased chance of treatment resistance. A substantial minority of people experience a ‘stormy’ early course of illness. Deaths from both suicide and natural causes occur at a much higher rate than in the general population (Power et al., 2003; Yuen et al., 2014). Secondary consequences such as persisting and worsening substance use and dependence, vocational failure, family stress and relationship breakdown are common. There is a serious risk of marginal lifestyles, homelessness and committing minor criminal offences. It is essential that high-quality, intensive care is continuously and assertively provided during this period. Recovery from an acute relapse can take longer than after the initial episode.
For some people, the move from specialised first-episode services to standard care results in loss of many of the gains made. This indicates the need for generic mental health services to provide the same level of support and assertive, individualised treatment as specialised FEP services. See also ‘Section 3. Treatment: context, structure and content of interventions’.
Management of acute relapse
It is important to determine the reason for relapse, carefully assessing adherence to medication and recent stressors. A family assessment can be helpful. Good adherence to antipsychotic medication and specific psychosocial interventions, particularly family interventions, may reduce the risk of relapse (Pharoah et al., 2010; Pilling et al., 2002; Schooler, 2005).
Poorly engaged individuals and those with frequent relapses benefit most from intensive case management (ICM) or assertive community treatment models of care (Marshall and Lockwood, 2011). All services should provide care based on this model of clinical intervention.
Comorbid substance use is another common contributing factor to relapse. A number of interventions, including CBT and motivational interviewing, have been developed to help people with severe mental illness manage substance abuse, but a recent Cochrane review (Hunt et al., 2013) found no evidence to support any one psychosocial treatment over another in terms of treatment engagement, reduction in substance abuse or improvement in mental state. See also ‘Section 2. Comorbid substance use’.


CBT: cognitive behaviour therapy; CBR: consensus-based recommendation; EBR: evidence-based recommendation.
N/A: Level of evidence category does not apply; recommendation based on a combination of available evidence, clinical experience and expert consensus.
Severe persistent or unremitted illness (stage 4)
Clinical remission in schizophrenia is not uncommon, based on the findings of studies that have applied objective criteria (Andreasen et al., 2005). However, a substantial minority of people with schizophrenia have persisting disabling and distressing symptoms. Apparent treatment resistance should be a trigger to reassess the treatment plan. For information on the management of treatment-resistant schizophrenia, see ‘Treatment resistance and clozapine’ in ‘Section 3. Treatment: context, structure and content of interventions’.
The recovery paradigm has reframed concepts of outcomes to include the subjective views of the person living with schizophrenia (Leamy et al., 2011; Slade, 2013). The therapeutic approach at this stage is guided by a person-centred value system of a ‘life worth living’. More information about recovery is provided in ‘The recovery paradigm’ in ‘Section 3. Treatment: context, structure and content of interventions’.
The clinician’s role is to establish a mutually respectful therapeutic relationship and optimise the management of potentially treatable factors such as unrecognised depression, inadequate psychosocial rehabilitation, poor adherence to prescribed medicines, substance use, medication side effects, differential responses to medicines, drug interactions and suboptimal drug therapy. The clinician also needs to work in partnership with primary care physicians and NGOs to ensure physical health and non-clinical needs are adequately addressed.


CBR: consensus-based recommendation; GP: general practitioner; EBR: evidence-based recommendation.
N/A: Level of evidence category does not apply; recommendation based on a combination of available evidence, clinical experience and expert consensus.
Section 2. Comorbid substance use
Psychosis and substance use
Comorbid substance use is a major complication of schizophrenia and an impediment to effective treatment and recovery. In the 2010 Australian National Survey of Psychotic Disorders, a diagnosis of harmful use of, or dependence on, psychoactive substances was made in 63.2% of men and 41.7% of women with psychosis (compared with 12.0% of men and 5.8% of women in the general population) (Moore et al., 2012).
Cannabis
Among people with psychosis, those with heavy cannabis use have a poorer prognosis. In addition, a systematic review of published data on cannabis exposure and the onset of schizophrenia (Large et al., 2011a) found that the use of cannabis brought forward the onset of schizophrenia by nearly 3 years. The increased risk appears to be dose-related and may be greater in those with a family history of schizophrenia.
Cannabis is arguably the most serious comorbidity with schizophrenia because of its widespread use among young people with mental illness (Schimmelmann et al., 2012). Cannabis use has a well-recognised role in triggering onset and exacerbations of schizophrenia (Gururajan et al., 2012; Stefanis et al., 2014) and is associated with a worse outcome and reduced efficacy of treatment (Gupta et al., 2013). Cannabis use can precipitate psychotic relapse in people with schizophrenia who had previously achieved remission.
Stimulants
Stimulants such as methamphetamine and cocaine can trigger psychotic states in people with schizophrenia and tend to exacerbate acute psychotic symptoms. Reported rates of stimulant use disorder in people with psychosis are much higher than in the general population (Sara et al., 2015). However, amphetamine use does not seem to have a measurable effect on the incidence of schizophrenia (Sara et al., 2015). People with schizophrenia are particularly sensitive to the psychotogenic effects of stimulant drugs, which act by releasing dopamine (Seeman and Seeman, 2014). This effect is evident in those experiencing FEP who have never been treated with antipsychotic medication and in those who have received treatment. Moreover, there appears to be a dose-related effect, with more frequent transient psychotic states and longer episodes of psychosis in more frequent and heavier users (McKetin et al., 2013).
Alcohol
Alcohol use disorders (AUD) are common in schizophrenia, with one Australian study finding as many as 57% of inpatients and 46% of outpatients meeting the criteria for the diagnosis of an AUD (Dawe et al., 2000). The prevalence of comorbid lifetime AUD worldwide is 20.6% (Koskinen et al., 2009). Alcohol abuse contributes to violence, imprisonment and homelessness of people with schizophrenia and is also associated with a worse outcome through non-adherence to treatment and poor physical health. Despite these problems, there has been relatively little research on the effects of alcohol in people with schizophrenia.
Managing substance use
There is often a separation between drug and alcohol and mental health services, with variable levels of collaboration between the two services. Integration of mental health services and drug treatment services would make treatment for alcohol and other drugs more accessible to people with psychotic illness, and this approach may be more effective (Brunette and Mueser, 2006).
CBT strategies have been used in the management of substance use in people with psychotic illness (Lubman et al., 2010). These approaches aim to examine the motivations for substance use, especially lifestyle factors, and teach more effective interpersonal and coping skills. Engaging the social network can also be helpful in managing substance use (Mueser and Gingerich, 2013).
Despite the lack of robust empirical evidence for the efficacy of these interventions (Hunt et al., 2013), motivational interviewing and integrated therapy (which combines CBT with lifestyle interventions and case management) are recommended approaches to the management of comorbid substance use in schizophrenia. An encouraging observation is that a high proportion of people do give up substance use after the first episode of illness (Wisdom et al., 2011).

CBR: consensus-based recommendation; CBT: cognitive behaviour therapy.
N/A: Level of evidence category does not apply; recommendation based on a combination of available evidence, clinical experience and expert consensus.
Drug-precipitated psychosis
Only a small proportion of people who use cannabis, stimulant drugs or hallucinogens become psychotic. Therefore, any person presenting with delusional beliefs apparently emerging as a result of having used one of those drugs should be considered, at a minimum, to be at ultra-high risk of developing psychotic illness.
It can be difficult to separate drug-induced psychotic states from relapsing mental illness. However, drug-precipitated psychoses, including the toxic delirium that follows persistent amphetamine use, are typically transient, and present with illusions rather than true hallucinations, plausible persecutory beliefs, affect that is appropriate to the content of the beliefs and an absence of thought disorder. People with symptoms that are persistent rather than transient or with auditory hallucinations, bizarre and enduring delusional beliefs, incongruous or blunted affect or thought disorder should be suspected of having a primary psychotic illness requiring longer term observation and treatment.
A significant proportion of people admitted to hospital with a diagnosis of drug-precipitated psychosis go on to develop chronic and relapsing psychotic illness (Crebbin et al., 2009; Sara et al., 2014). Therefore, close monitoring of such individuals (with drug-free periods, if possible) is important, as is counselling about the harms of drug use.

CBR: consensus-based recommendation.
N/A: Level of evidence category does not apply; recommendation based on a combination of available evidence, clinical experience and expert consensus.
Section 3. Treatment: context, structure and content of interventions
The recovery paradigm
Recovery-oriented practice is based on the central importance of the lived experience and the perspective of the person with mental illness. Distinctions can be made between clinical recovery (which includes lessening of symptoms), functional recovery and personal recovery (Slade, 2009). Personal recovery has been described as a unique process of changing one’s attitudes, values, feelings, goals, skills and/or roles (Anthony, 1993). It is a way of living a satisfying, hopeful and contributing life even within the limitations caused by illness. The concept of personal recovery draws on constructs of hope, self-identity, meaning and personal responsibility (Slade, 2009). For the individual, gaining knowledge about the disorder and the available treatment options, developing illness self-management skills and taking responsibility for his or her own treatment are all important.
Recovery-oriented services emphasise peer relationships, social networks, person-centred (strengths-based) assessment and recovery planning. People are encouraged to take up new challenges. Risk can be reduced through the use of relapse prevention plans and advanced health directives, along with the development of self-management skills to prevent unnecessary crises and minimise the loss of personal responsibility during periods of crisis.
Recovery is a concept relevant to all stages of illness, beginning as soon as there is a need for care (in the prodromal or ultra-high risk period) and continuing through the first episode and the critical period. Recovery is a key goal in real terms and not merely in the sense of acceptance of persistent illness or adapting around it to have a meaningful life (critical though this is for many people). Clinicians should aim for recovery for everyone, and across all stages, as there are often late remissions or improvements.
Interventions essential to community-supported recovery include supporting people in early prodromal or symptomatic stages, responding quickly to requests for assistance, providing meaningful support, planning together with individuals and carers, listening to family members, facilitating connections with peer support networks and professional advocates, ready access to suitable facilities and helping people stay in their own accommodation (Mental Health Council of Australia [MHCA], 2006). Assistance needs to be personalised to the individual. An essential component of care is coordination of complex systems and assistance in negotiating the common barriers of social and economic disadvantage.
Training for clinicians should include both the values and the practical aspects of the recovery model. The required recovery competencies include an awareness of recovery within a cultural context, understanding and accommodating diverse views on mental illness and treatment, respectful communication, appreciating the individual’s resourcefulness and understanding the impact of stigma and social exclusion (Mental Health Commission, 2001).
In Australia, the national framework for recovery-oriented mental health services (Australian Health Ministers’ Advisory Council, 2013) recommends provision of a respectful environment which allows people to be treated with dignity, and the implementation of recovery principles across multiple levels of service delivery. Standardised instruments have been developed to measure individual recovery (Shanks et al., 2013) and the recovery-orientation of services (Williams et al., 2012), which should enable more research and evaluation.


CBR: consensus-based recommendation; EBR: evidence-based recommendation.
N/A: Level of evidence category does not apply; recommendation based on a combination of available evidence, clinical experience and expert consensus.
The service system and models of intervention
Mental health care in Australia and New Zealand is centred on community-based care in both public and private sectors. Reliance on inpatient beds has been decreasing over recent years (Raudino et al., 2014). The National Mental Health plans of both countries (Australian Government Department of Health, 2009; New Zealand Ministry of Health, 2012) emphasise partnerships between mental health services, primary care providers, private providers, the non-government sector, peer operated services and family-based care.
For many, the GP is well positioned to coordinate care and attend to the physical health needs of people with mental illness. Cost and access barriers need to be acknowledged and addressed. Emerging methods of service delivery, such as telemedicine and online mental health education and therapies, may be especially helpful for people living in rural and remote communities.
Mental health services generally have the following elements:
Inpatient services acute beds subacute ‘step-up, step-down’ beds residential rehabilitation extended care beds consultation liaison services psychiatric emergency care, usually attached to the emergency departments of hospitals
Specialised ambulatory care mental health services crisis teams or community assessment and treatment teams which provide acute phase community-based assessment, treatment and, where appropriate, home-based care community mental health continuing care teams offering multidisciplinary extended mental health care in the community developmentally specific teams including early intervention teams and aged care teams assertive community treatment teams and intensive case management (ICM) providing mobile care to a more complex subset of people including both home-based treatment and hospital diversion community-based clinical care in residential rehabilitation units
Specialised mental health community support individual and group peer support often related to lifestyle or education and employment family and carer support respite services non-clinical residential services
Private sector mental health providers primary care physicians psychiatrists psychologists and other counsellors community and day programmes inpatient facilities.
The second Australian National Survey of Psychosis demonstrated the positive impact of some of the recent changes, which include increased community staffing and funding for services provided by the non-government sector (Neil et al., 2013). However, social and economic disadvantages remain significant challenges for people living with psychosis (Morgan et al., 2012).
Intensive case management (ICM)
ICM is a model of service delivery for people with severe mental illness, most commonly schizophrenia. ICM has evolved from assertive community treatment, a model that relies on team-based management rather than individual caseloads. Both models include frequent service contact, mobile active outreach and low staff–patient ratios. Better outcomes are achieved by community services that have higher levels of fidelity to the ICM model.
A Cochrane review of RCTs (Dieterich et al., 2010) compared ICM (in which case managers had a caseload of fewer than 20 patients) with non-intensive case management (where patients had the same package of care but case managers had a caseload of more than 20 patients), and with standard care. Compared with standard care, people receiving ICM were significantly more likely to stay in contact with the service and had a lower rate of hospitalisation, a higher rate of stable housing and better overall function. The only difference between ICM and non-intensive case management was that the ICM patients were more likely to remain engaged in care.
Non-government services
The Australian and New Zealand governments are making significant investments in the non-government sector. NGOs provide a diverse range of services to the mentally ill and are considered to be more flexible, responsive and innovative than public mental health services.
NGO services provide a range of housing options, including staffed residential facilities for people who require help with illness stabilisation, skill retrieval and development, and community integration over a longer period of time. People with schizophrenia who have disabling cognitive or negative symptoms can benefit from community support worker interventions. These can range from low-level support, such as a face-to-face contact once a week, to higher frequency support delivered after a comprehensive needs assessment in the form of a ‘package of care’. Packages of care can consist of both individualised hours of face-to-face support and assistance with the person’s physical environment and organising their daily activities. Finding the most suitable services and coordination of care can be difficult especially when there are numerous small NGOs, and improved links between NGO-based community care services and clinical services are needed. Innovative models exist where mental health and NGO clinicians are co-located and work as a virtual team providing integrated care for people with significant mental illness.
NGOs also provide specific services to people from diverse cultural groups such as Australian Aboriginal and Torres Strait Islander people, Māori, Pacific Peoples and refugees. For those who have had adverse experiences with clinical services or difficulty trusting clinicians, involvement with the NGO sector can be an effective way of engaging with treatment.
Primary care
There are several models for working with GPs. It is preferable that people with schizophrenia who have significant ongoing symptoms and disability and a history of serious severe psychotic relapses are followed up by specialist mental health services. These individuals will benefit from the input of a multidisciplinary team and regular assertive follow-up to ensure continuity of treatment. The GP may play an important role in managing physical health conditions. GPs should receive appropriate clinical information, including the treatment plan, and should have regular communication with mental health clinicians.
Sometimes people with schizophrenia who have had a period of stability under specialist mental health services are considered suitable to have further mental health care transferred to general practice. Before this happens, the following issues should be considered:
The individual and their family, carers and/or support people need to have participated in the development of a relapse recovery plan.
There should be a clear, shared understanding of the role of medication in relapse prevention and how medicines will be prescribed, dispensed and monitored.
There should be careful consideration of which GP would be providing the follow-up and how the person would access this service. Before a transfer to primary care, it is appropriate for specialist mental health to contact the primary care practice to discuss the follow-up needs of the individual and to provide clear written information regarding the treatment plan.
Rural and remote services
In rural and remote regions of Australia and New Zealand, resources for the care of people with mental illness are scarce, as for many areas of medicine. The number of psychiatrists per population is less than in urban regions, and distances from hospitals are greater.
In these settings, it may be the GP who will be the main medical contact for people with schizophrenia rather than a psychiatrist. If possible, psychiatric opinions should be sought especially when there is diagnostic uncertainty or complexity such as dual diagnosis or treatment resistance. Telemedicine services can be useful in enabling ongoing specialist assessment and advice.


EBR: evidence-based recommendation; CBR: consensus-based recommendation; GP: general practitioner.
N/A: Level of evidence category does not apply; recommendation based on a combination of available evidence, clinical experience and expert consensus.
Antipsychotic medication
Overview
Antipsychotic medicines treat the symptoms of schizophrenia but not its underlying causes. In the absence of new treatments, these medicines remain the cornerstone of both acute and maintenance therapy for schizophrenia. All antipsychotic medicines derive their effect on positive symptoms of psychosis from the blocking of dopamine receptors. These medications are described as FGA or SGA, but this distinction is not a neat one and the individual properties of each agent should be taken into account. The reason for including the distinction in this guideline is that much of the quoted literature uses the distinction. It can be expected that the classification into FGA/SGA will become less recognised in clinical practice over time.
Most FGAs are high-potency medicines that are tightly bound to dopamine receptors and often cause debilitating extrapyramidal side effects (see Table 6 for the approximate relative frequency of common side effects of individual antipsychotic drugs). There is little evidence that the SGAs are more effective than the FGAs in the acute treatment of positive symptoms. However, there is some evidence that oral SGAs are more effective than oral FGAs in relapse prevention (Kishimoto et al., 2013). Furthermore, there are data suggesting that some of the SGAs are more effective for managing negative and neurocognitive symptoms (Zhang et al., 2013a).
However, some of the SGAs have serious adverse effects, especially metabolic and cardiac side effects. Metabolic changes have been detected even in the young (Foley et al., 2013), so monitoring and intervention should begin early for people prescribed these medicines. Where possible, medicines with the least risk of weight gain should be selected (Bak et al., 2014). Extrapyramidal side effects, akathisia and tardive dyskinesia can occur with both FGAs and SGAs, although in general these tend to be more common with FGAs.
Neuroleptic malignant syndrome (NMS) is a rare and potentially life-threatening condition characterised by fever, rigidity, tremor, sympathetic nervous system dysregulation and creatine kinase (CK) elevation (Tse et al., 2015). Prompt diagnosis and treatment (usually in a general hospital) are essential. Both FGA and SGA can cause NMS. More information about side effects is available in ‘Management of antipsychotic side effects’ in this section.
Antipsychotic medicines are associated with substantial inter-individual variation in both efficacy and adverse effect profiles. It is essential that clinicians work with the individual to find the best possible medication for them in terms of both efficacy and tolerability, and develop a dosing regimen that will minimise the impact of side effects on daily function. The proposed choice of antipsychotic medicine should be discussed with the person who will be taking it, providing information about both expected benefit and side effects. Their previous experiences with medication should be taken into account.
There may be differences in tolerability between ethnic groups. For example, East Asian people may require lower doses and be more sensitive to extrapyramidal side effects (Ormerod et al., 2008). Clinicians prescribing anticholinergic agents should be aware of contraindications to these medicines and advise about possible anticholinergic side effects.
Aids to medication adherence such as dose administration aids, packed by the dispensing pharmacist or by the individual or carer, and programmed reminders (e.g. on a mobile phone) can be useful. Long-acting injectable (also known as depot) antipsychotic agents can be helpful if adherence is poor or uncertain (Bosanac and Castle, 2015); sometimes there is an impression of good adherence when in fact little medication is being taken. Depot medications bypass hepatic first-pass metabolism.

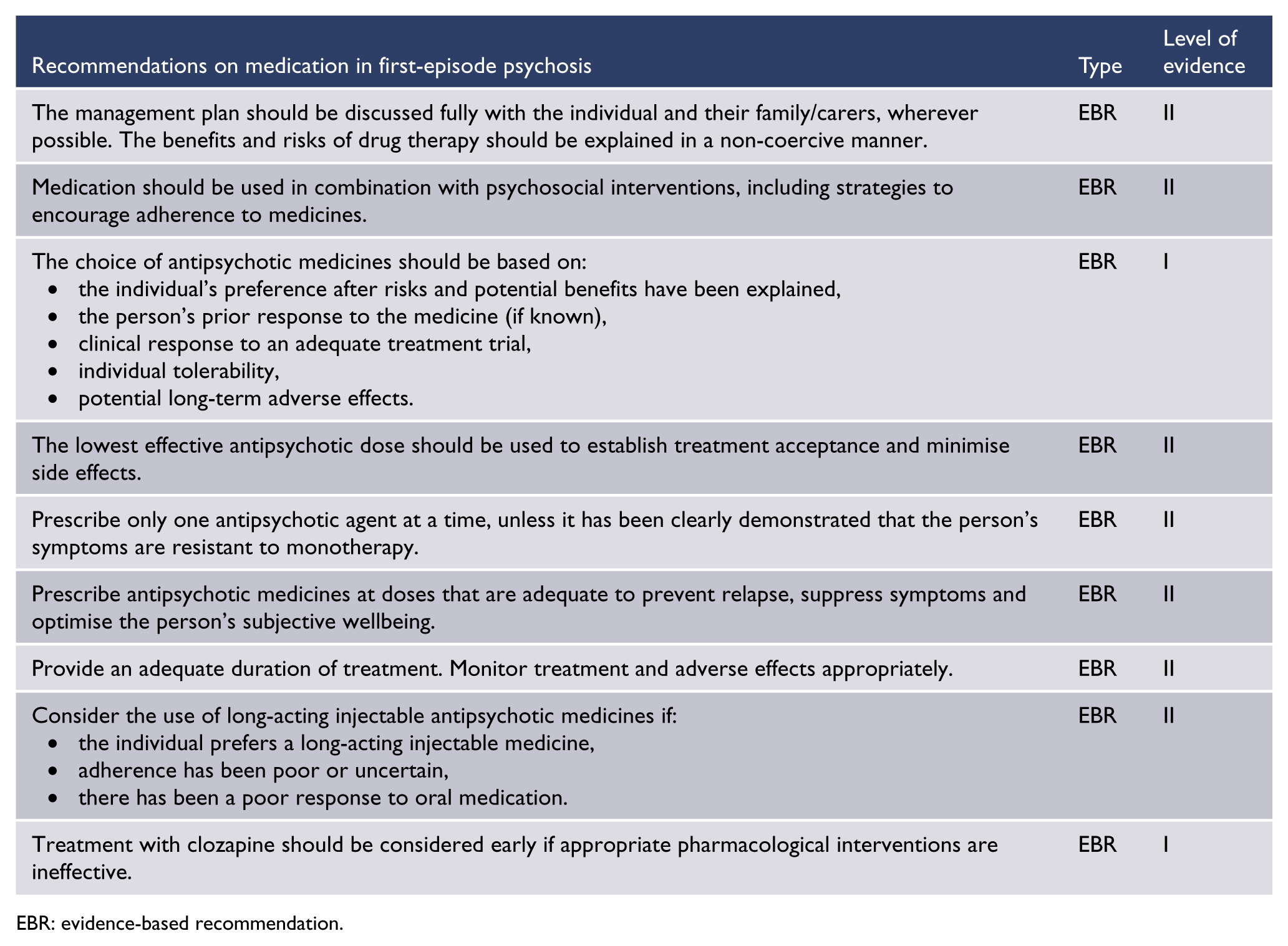
EBR: evidence-based recommendation.
Antipsychotic medicines for FEP
A treatment algorithm for pharmacological treatment for first-episode non-affective psychosis is provided in Figure 1 (above).
Oral SGAs should be prescribed as first- and second-line treatment for people with FEP. The choice of SGA depends on various factors such as side effects, tolerability/discontinuation rate and long-term outcomes. The initial dose should be low. If response is slow or incomplete, the dose should be increased slowly at suitable intervals.
Distress, insomnia and agitation can be treated initially with benzodiazepines. Other symptoms such as mood elevation and depression require specific treatment with mood stabilisers and antidepressants.
Antipsychotic medicines for acute relapse and maintenance therapy (stages 3a, 3b, 3c)
Acute phase
Where possible, the choice of antipsychotic medicine should take into consideration prior response to treatment, side effects and the person’s preference. Oral SGAs are usually the treatment of choice, in view of their generally lower risk of extrapyramidal side effects, compared with FGAs.
Long-acting injectable antipsychotic agents, particularly SGAs, provide an important treatment option in all phases of the disease for people whose adherence to oral treatment is poor. For some patients, the use of long-acting injections is a convenient option to overcome the need of taking medications every day and maintain remission. Thus, an appropriate and non-judgemental approach should be part of the pharmacological discussion. If it is clear that a long-acting injectable antipsychotic is the most suitable choice for maintenance therapy, selection of the appropriate oral antipsychotic that is available as a long-acting injectable will provide an option for later change (e.g. initiation of zuclopenthixol hydrochloride tablet prior to administration of zuclopenthixol decanoate long-acting injectable).
Parenteral administration of antipsychotics (along with benzodiazepines) may be required for the treatment of agitation in emergency situations when the person refuses oral medications (see ‘The management of acute behavioural disturbance in schizophrenia and related disorders’ in this section).
Maintenance phase
In established illness, it is generally considered advisable to continue maintenance treatment with the prescribed antipsychotic to which the person responded in the acute episode, as long as the efficacy and benefits outweigh the side effects. However, if the drug is causing adverse effects such as weight gain, then switching to a drug with less potential for causing these side effects should be considered (see ‘Switching medications’ in this section). If dose reduction is indicated, it should be performed gradually to avoid withdrawal effects and rebound psychoses. When planning long-term treatment, antipsychotic agents with the best balance between efficacy, side effects and adherence for the individual should be selected. Higher doses than those usually recommended (Table 4) may sometimes be required, but there is a lack of research about efficacy and safety. Higher doses should be used cautiously, and careful monitoring of safety and tolerability is essential. Clozapine should be considered when there is a poor response or significant side effects.
Dosing regimen for common oral antipsychotic agents.
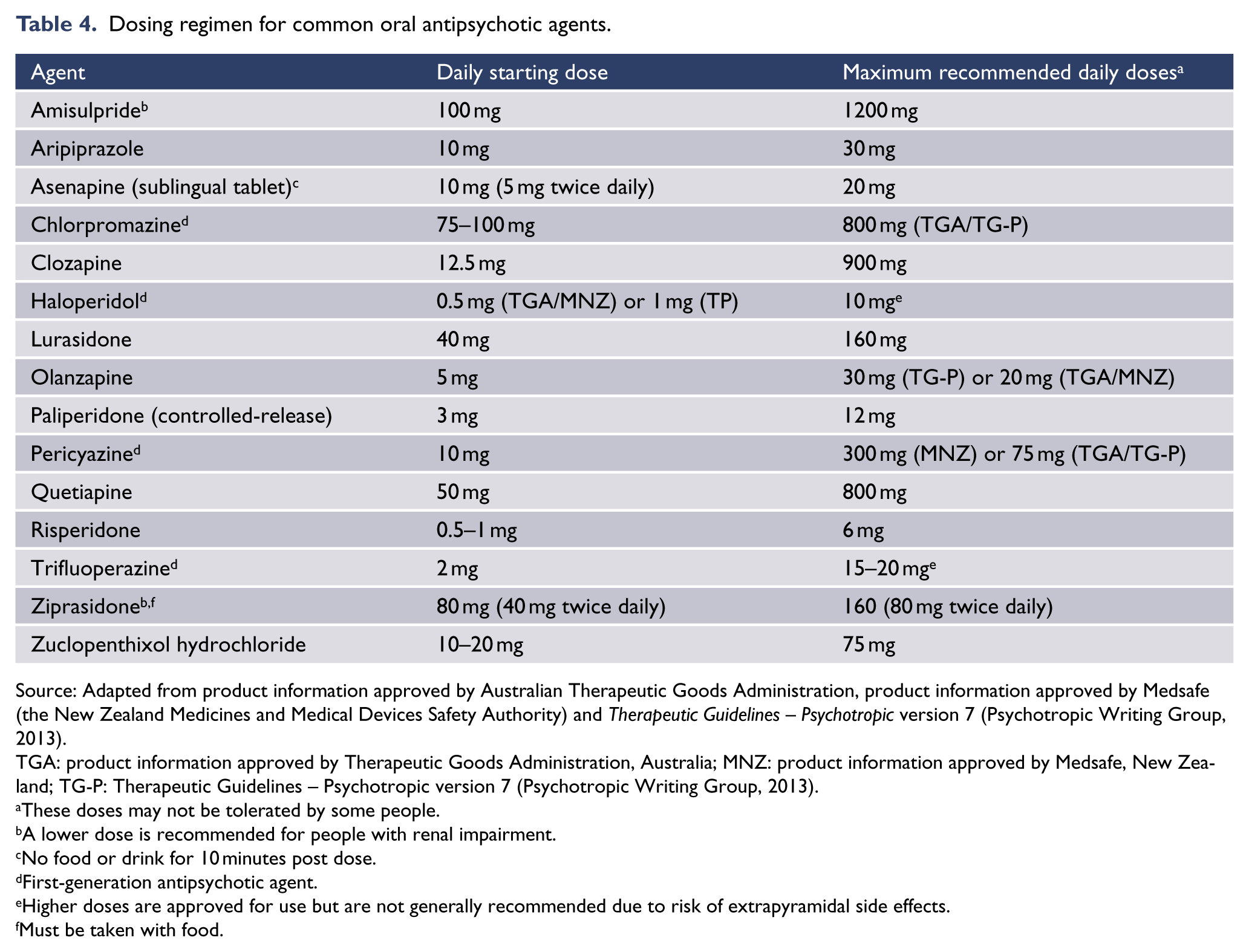
Source: Adapted from product information approved by Australian Therapeutic Goods Administration, product information approved by Medsafe (the New Zealand Medicines and Medical Devices Safety Authority) and Therapeutic Guidelines – Psychotropic version 7 (Psychotropic Writing Group, 2013).
TGA: product information approved by Therapeutic Goods Administration, Australia; MNZ: product information approved by Medsafe, New Zealand; TG-P: Therapeutic Guidelines – Psychotropic version 7 (Psychotropic Writing Group, 2013).
These doses may not be tolerated by some people.
A lower dose is recommended for people with renal impairment.
No food or drink for 10 minutes post dose.
First-generation antipsychotic agent.
Higher doses are approved for use but are not generally recommended due to risk of extrapyramidal side effects.
Must be taken with food.
If there are problems with adherence, the person’s reasons for non-adherence and strategies to address these should be explored. A long-acting injectable antipsychotic agent should be considered, and this option should be discussed with the person and their carers.
Monitoring and treatment of side effects
During both the acute and maintenance phases of treatment, it is essential to regularly monitor antipsychotic adverse effects, including extrapyramidal side effects, akathisia, weight gain, cardiovascular and metabolic side effects (Curtis et al., 2012; De Hert et al., 2011) and tardive dyskinesia. Severe medication side effects, such as akathisia (Lohr et al., 2015), can be a risk factor for non-adherence, exacerbation of symptoms, aggression or suicide, and appropriate interventions should be undertaken promptly (see ‘Management of antipsychotic side effects’, below). The management plan should include clear information about who is responsible for implementing physical health interventions (such as prescribing metformin, antihypertensive medication, organising dental care and/or consultation with a dietitian). Information about managing cardiometabolic effects of antipsychotic medication in people with psychosis is found in Positive Cardiometabolic Health: an early intervention framework for patients on psychotropic medication, NSW Government Health Education Training Institute (2011) (available at www.heti.nsw.gov.au/cmalgorithm).
Cessation of medication
The decision to reduce, or possibly cease, medication requires careful discussion and evaluation of risks and benefits for each individual. The person should have made a full recovery and been well for at least 12 months before cessation of medication is considered. Severity of illness, risk factors, individual circumstances and family history should be taken into account, and medical supervision during and after medication cessation is essential. The support of family and friends, and their assistance in monitoring changes in mental state or social function, is helpful. An advance directive rescue plan, developed in collaboration with the person, their family and friends, can be very helpful if relapse does occur.
Long-acting injectable antipsychotic agents
Long-acting injectable antipsychotic agents provide an important option, particularly in the management of non-adherence, and can reduce relapse rates and rehospitalisation in people with FEP or schizophrenia (Kishimoto et al., 2013). They should only be prescribed when this is the person’s preference or as part of a treatment plan in which adherence is a clinical priority.
Long-acting injectable antipsychotic agents can reduce the risk of accidental or deliberate overdose in high-risk individuals. However, adjustment to optimal dose may not be as flexible as oral antipsychotics and prolonged distressing side effects after discontinuation may occur. With some long-acting injectable antipsychotic agents, it may take 2–4 months to achieve the desired steady-state plasma concentrations (Table 5).
Long-acting injectable antipsychotic agents.
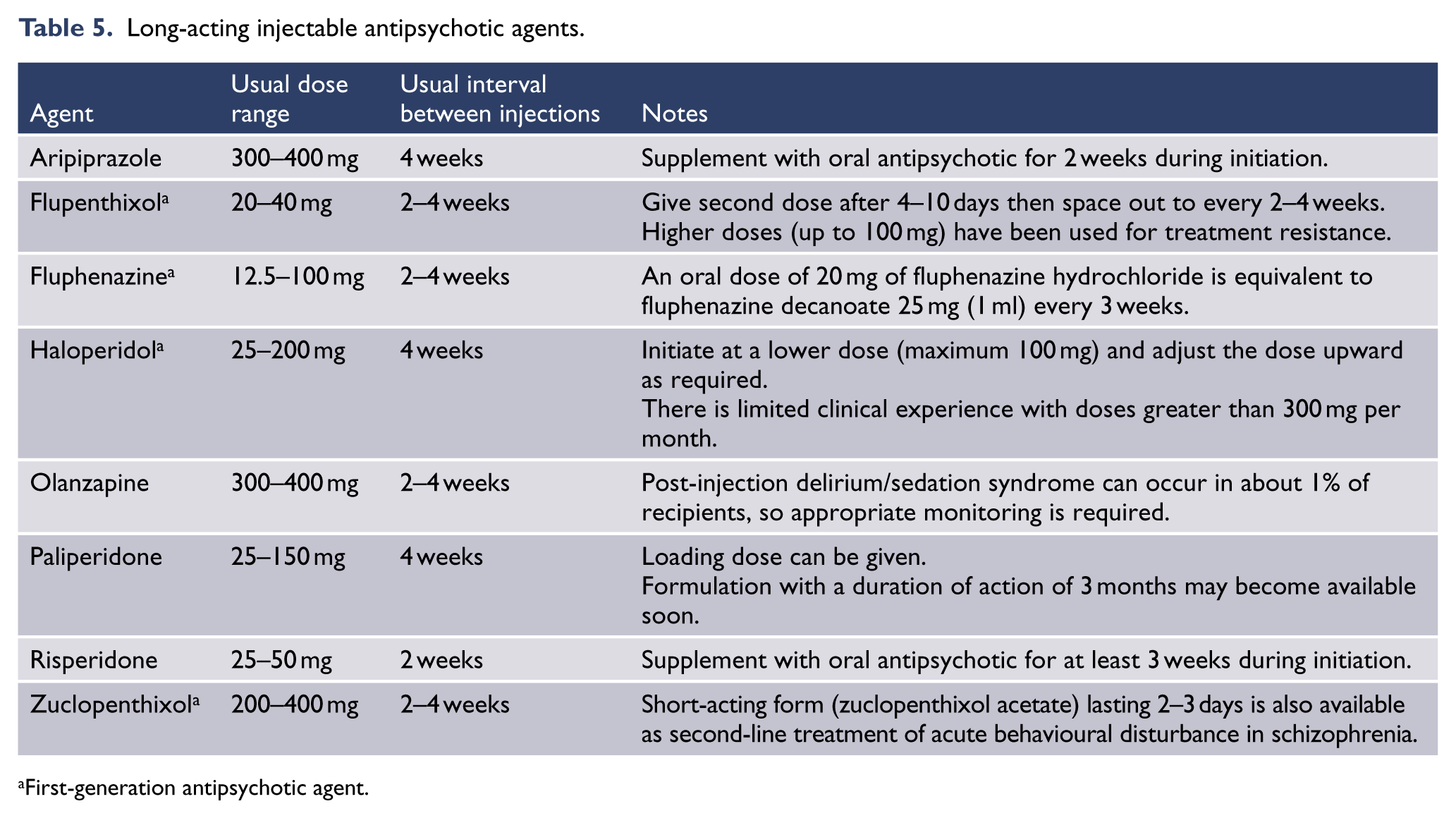
First-generation antipsychotic agent.
The side effects are essentially the same as the oral preparations; most are likely to cause extrapyramidal side effects, with the exception of long-acting injectable olanzapine, which is associated with weight gain.

EBR: evidence-based recommendation.
While SGAs are generally preferred, there is limited choice and occasionally there is a better result with a first-generation long-acting injectable antipsychotic agent. Careful monitoring for extrapyramidal side effects and tardive dyskinesia is needed.
It is vital to ensure that intended switching is completed appropriately:
For most antipsychotic agents, an initial oral test dose is required before starting the long-acting injectable preparation.
For olanzapine and paliperidone, a loading dose is recommended when switching from oral preparations to long-acting injectable preparations, so an initial oral supplement may not be required.
Switching medications
A slow crossover titration is preferred and close monitoring of mental state, side effects and rebound phenomena is essential during switching (Knox and Stimmel, 2004). Potential adverse events during crossover include the following:
Cholinergic rebound (flu-like symptoms with malaise, agitation, anxiety, insomnia, nausea and diarrhoea) can occur if switching from an agent that has a high affinity for the muscarinic M1 receptor to an agent with low M1 affinity (e.g. switching from clozapine to risperidone or aripiprazole). Slow crossover is advisable and short-term use of an anticholinergic such as benztropine is recommended.
Rebound psychosis or extrapyramidal side effects – these are more likely to occur if switching from an agent that is loosely bound to the dopamine 2 receptor (D2), such as clozapine or quetiapine, and normally occur earlier than would be expected from a true psychotic relapse. Cross-titrate slowly and use symptomatic treatment such as a beta-blocker or benzodiazepine if necessary.
Rebound insomnia – this may occur if changing from more sedating agents (clozapine, olanzapine) to less sedating agents (risperidone, paliperidone, amisulpride, aripiprazole). Give the new medication in the morning rather than the evening and prescribe a short-term hypnotic agent, such as temazepam or zopiclone, if needed.
Unwanted pregnancy – this is more likely to occur when switching from antipsychotics that cause hyperprolactinaemia to antipsychotics with minimal prolactin elevation. It is important to educate women about this risk, and contraception should be considered.


Treatment resistance and clozapine
Definition
Treatment resistance is usually defined as continued positive symptoms after trials of at least two different antipsychotics at moderate doses (usually at least 300 mg chlorpromazine equivalents per day) for a reasonable period (usually at least 6 weeks). Every effort must be made to ensure the person has actually taken the prescribed medication, including a trial of a long-acting injectable agent in people suspected of non-adherence. It is important to differentiate non-response from lack of tolerability. There is a tendency to see resolution of positive symptoms as the main goal of treatment, but negative symptoms can be more disabling. It is important to differentiate primary negative symptoms from negative symptoms secondary to factors such as depression, anxiety or medication.
Clozapine
Clozapine can be a highly effective agent for those who fail to respond to other antipsychotic medications. Ideally, treatment resistance should be identified early (within the first 6–12 months) and treatment with clozapine started without delay, to minimise the disability that develops with unremitting illness. Clozapine is the only antipsychotic shown to reduce suicidal behaviour in people with schizophrenia (Hennen and Baldessarini, 2005; Meltzer et al., 2003). The high side-effect burden means clozapine is reserved for people who have not responded to other treatments.
Mandated blood monitoring (weekly for the first 18 weeks, every 4 weeks thereafter) means that blood dyscrasia is likely to be detected, but cardiomyopathy and myocarditis are also potentially dangerous side effects. There is little evidence that the cardiac adverse events can be predicted, but baseline echocardiogram and troponin are advisable. Regular echocardiograms do not have a strong evidence base and it has been suggested that they be undertaken only if clinically indicated (Murch et al., 2013; Ronaldson et al., 2011). Metabolic monitoring and advice about diet and exercise are essential.
There is some evidence to support the use of metformin (500 mg bd) to minimise weight gain and insulin resistance for people taking clozapine (Chen et al., 2013). It is essential that the prescribing doctor is familiar with the unique profile of clozapine and can manage other less common side effects. If a person taking clozapine has a neutrophil count in the amber range, the addition of a low dose of lithium may be useful to protect against clozapine-induced neutropenia and maintain the person on clozapine (Paton and Esop, 2005).

EBR: evidence-based recommendation.
It can take up to 12 months to see the full benefits of clozapine. If clozapine is ineffective, the choice is whether to augment clozapine with another drug or electroconvulsive therapy (ECT), or switch to another agent. If there has been absolutely no response and/or side effects are severe, a switch is suggested. Care needs to be taken in down-titration. If clozapine has had some – even modest – benefit, augmentation is justifiable, although the evidence for this strategy is equivocal. On an individual level, the addition of a high affinity dopamine D2 blocking agent such as risperidone or amisulpride to clozapine can be effective for residual positive symptoms. The addition of aripiprazole to clozapine can effect significant weight reduction and improvement in metabolic parameters. Antidepressants and mood stabilisers cannot be recommended as adjuncts in the absence of mood or anxiety symptoms.

EBR: evidence-based recommendation.
N-acetyl cysteine (Berk et al., 2008) and ethyl-eicosapentaoic acid (EPA) (fish oil) (Porcelli et al., 2012) are undergoing evaluation as clozapine augmenters. ECT has a limited place in the management of people with treatment-resistant schizophrenia (see ‘Electroconvulsive therapy’ in this section), including those on clozapine (Petrides et al., 2015; Tharyan and Adams, 2005).
Combination of antipsychotic agents
The concurrent use of two or more antipsychotics for people with treatment-resistant or refractory schizophrenia is common practice, despite limited evidence to support this practice.
Compared with antipsychotic monotherapy, combined antipsychotic treatment has been associated with an increased side effect burden, high-dose prescribing, increased hospitalisation rates and length of stay, higher treatment costs and increased mortality (Gallego et al., 2012). Switching from polypharmacy back to monotherapy is possible, and strategies to reduce polypharmacy by educating clinicians to change their prescribing behaviour have had some success (Tani et al., 2013).

EBR: evidence-based recommendation.
Smoking and antipsychotic medication
Smoking may increase the metabolism of particular antipsychotic agents (e.g. clozapine, olanzapine, haloperidol) via the induction of certain hepatic enzymes, particularly CYP 1A2. Plasma levels of both clozapine and olanzapine may be reduced by up to 50% in people who smoke tobacco products. If people taking these drugs cease smoking abruptly, the plasma levels can increase substantially and very rapidly.
Accordingly, higher doses of these antipsychotic agents are required to achieve the same therapeutic effect in smokers, compared with non-smokers (Taylor et al., 2015). It is therefore vital to routinely discuss smoking status and adjust the antipsychotic dosage appropriately. Clozapine levels should be closely monitored if there is a change in smoking behaviour, and the dose should be adjusted if necessary.
Nicotine replacement therapy, on the contrary, has no effect on this induction pathway. For information on strategies to support smoking cessation, see ‘Smoking’ in ‘Section 4. Physical health’.
Management of antipsychotic side effects
Antipsychotic medications are associated with a range of side effects (Table 6) including neurological, metabolic, sexual, endocrine, sedative and cardiovascular side effects (Psychotropic Writing Group, 2013; Taylor et al., 2015). See also ‘Section 3. Antipsychotic medication: overview’. It is important to routinely assess people taking antipsychotic medications for side effects (Table 7) and consider appropriate intervention if necessary (Table 8) (Castle and Buckley, 2015; Lambert et al., 2010).
Approximate relative frequency of common side effects of antipsychotics.
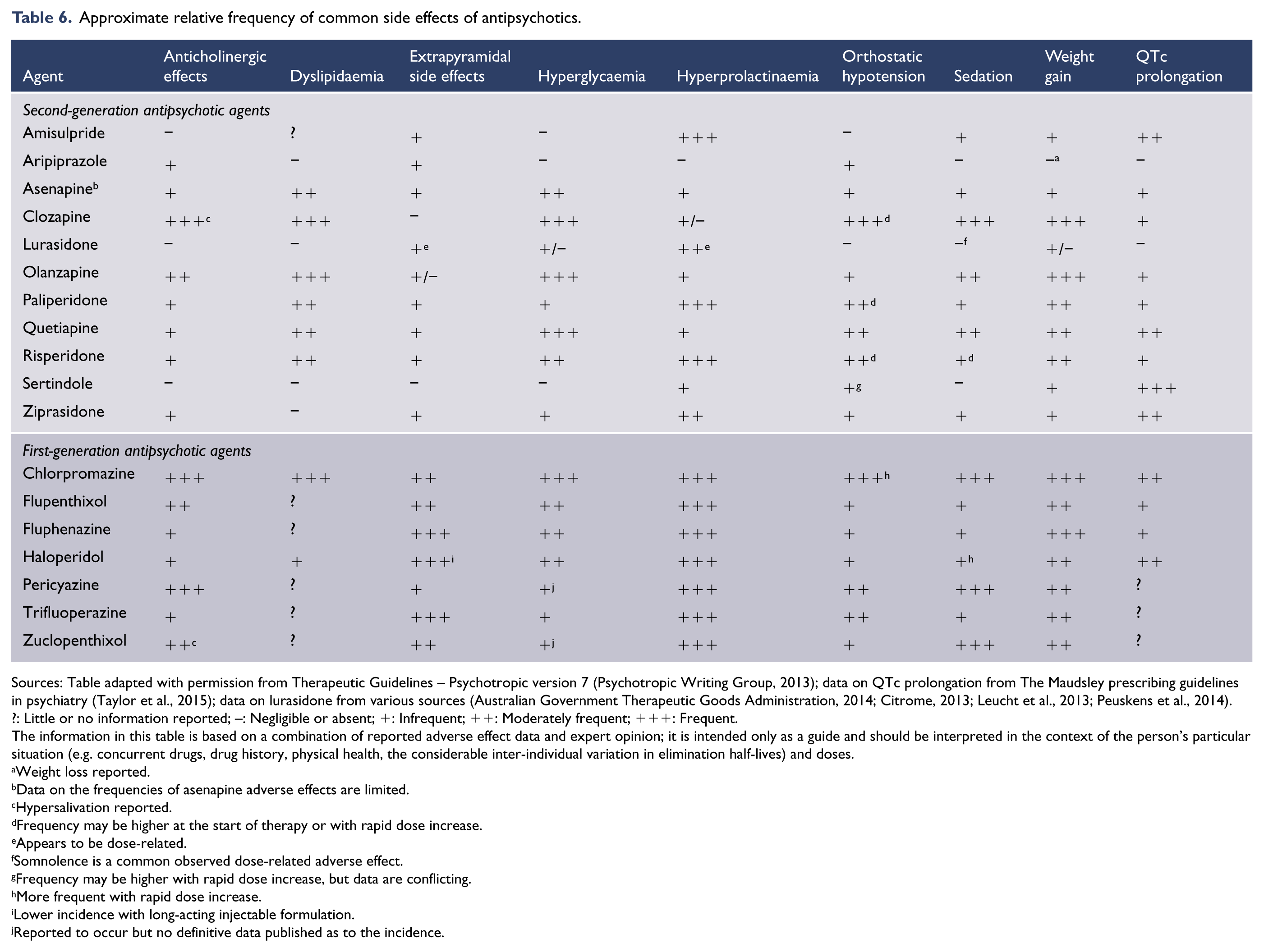
Sources: Table adapted with permission from Therapeutic Guidelines – Psychotropic version 7 (Psychotropic Writing Group, 2013); data on QTc prolongation from The Maudsley prescribing guidelines in psychiatry (Taylor et al., 2015); data on lurasidone from various sources (Australian Government Therapeutic Goods Administration, 2014; Citrome, 2013; Leucht et al., 2013; Peuskens et al., 2014).
?: Little or no information reported; –: Negligible or absent; +: Infrequent; ++: Moderately frequent; +++: Frequent.
The information in this table is based on a combination of reported adverse effect data and expert opinion; it is intended only as a guide and should be interpreted in the context of the person’s particular situation (e.g. concurrent drugs, drug history, physical health, the considerable inter-individual variation in elimination half-lives) and doses.
Weight loss reported.
Data on the frequencies of asenapine adverse effects are limited.
Hypersalivation reported.
Frequency may be higher at the start of therapy or with rapid dose increase.
Appears to be dose-related.
Somnolence is a common observed dose-related adverse effect.
Frequency may be higher with rapid dose increase, but data are conflicting.
More frequent with rapid dose increase.
Lower incidence with long-acting injectable formulation.
Reported to occur but no definitive data published as to the incidence.
Monitoring for people taking antipsychotic medication.
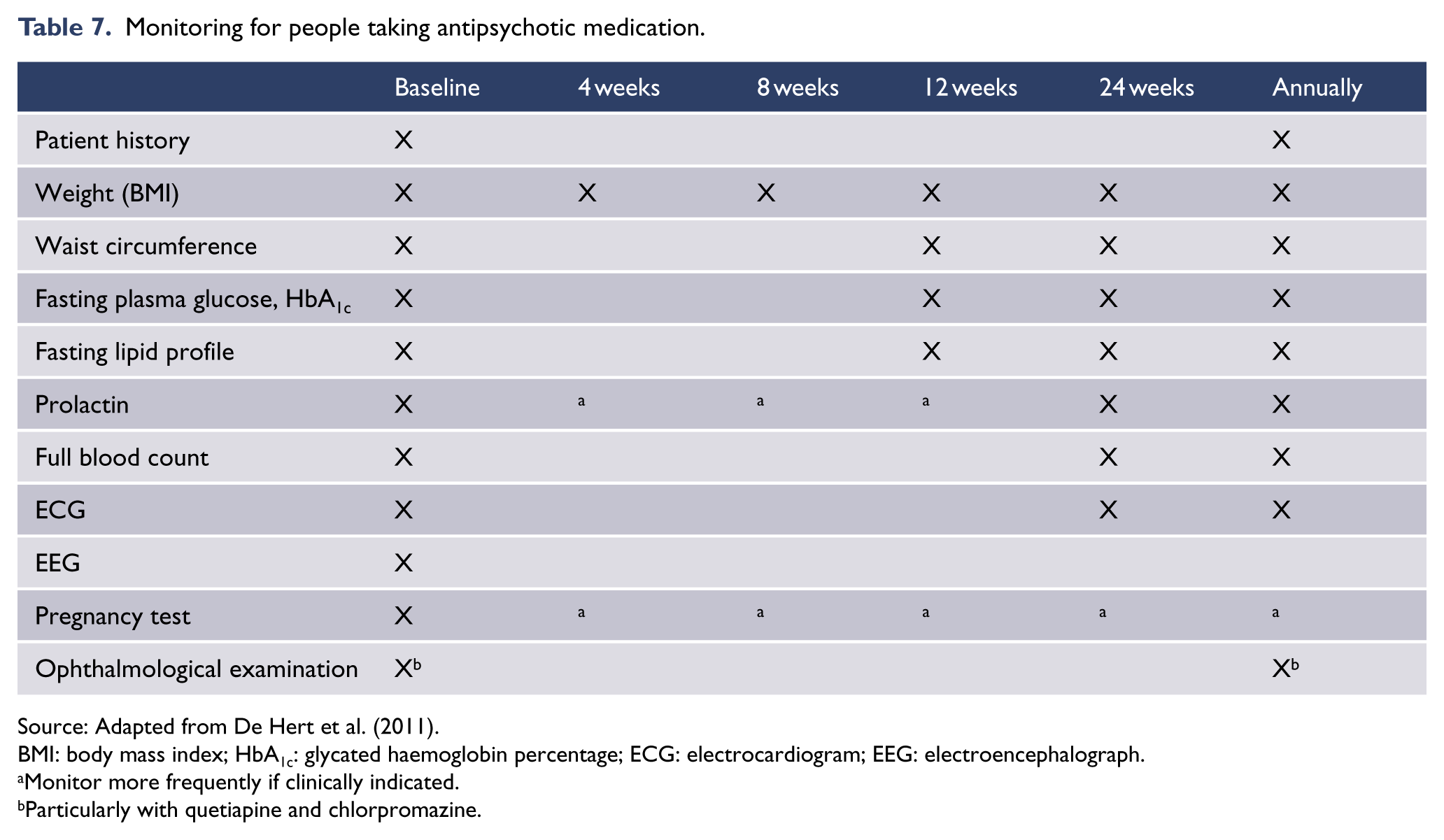
Source: Adapted from De Hert et al. (2011).
BMI: body mass index; HbA1c: glycated haemoglobin percentage; ECG: electrocardiogram; EEG: electroencephalograph.
Monitor more frequently if clinically indicated.
Particularly with quetiapine and chlorpromazine.
Management strategies for side effects of antipsychotic drugs.
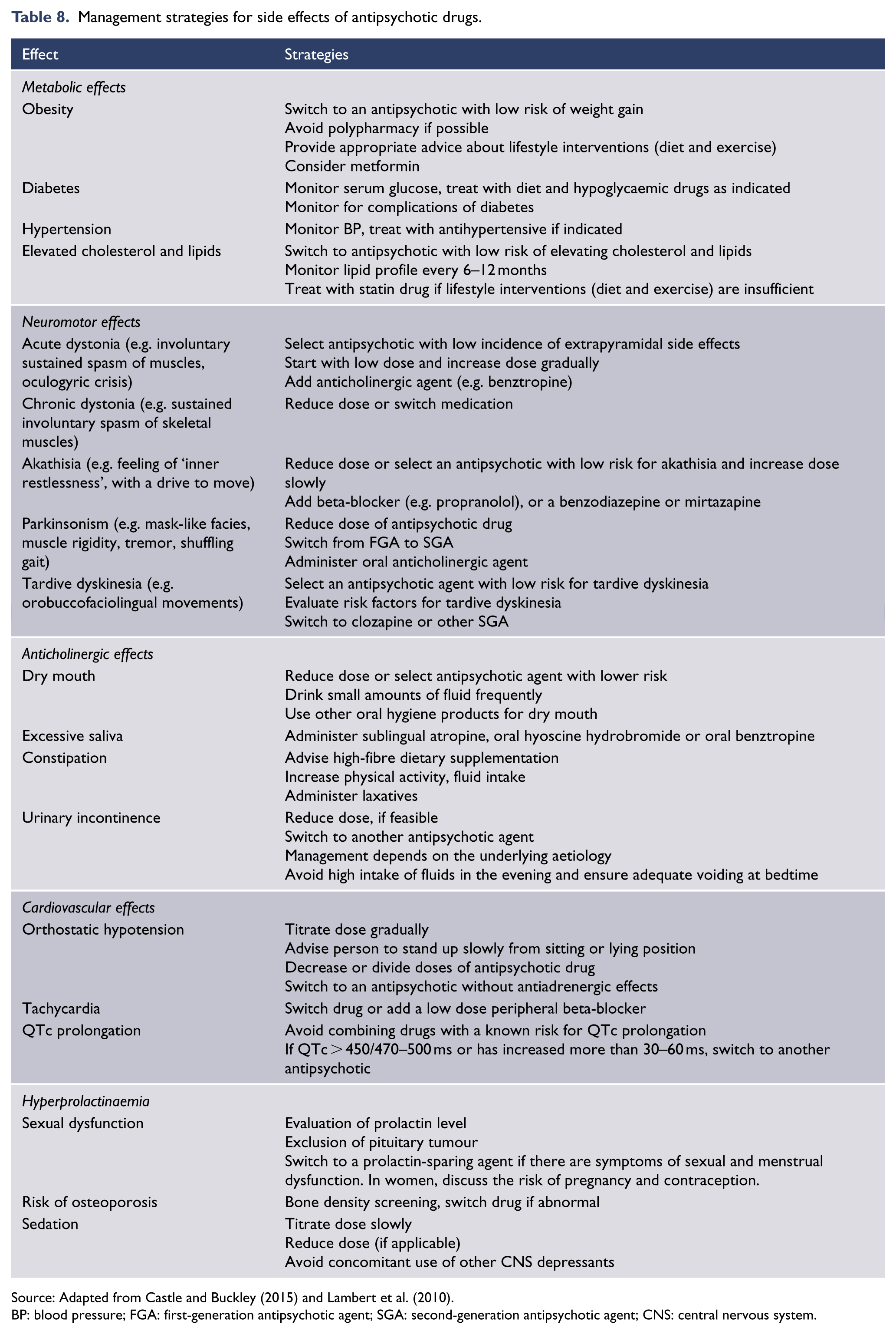
Source: Adapted from Castle and Buckley (2015) and Lambert et al. (2010).
BP: blood pressure; FGA: first-generation antipsychotic agent; SGA: second-generation antipsychotic agent; CNS: central nervous system.
Australians with psychotic disorders have high rates of obesity and metabolic syndrome (Galletly et al., 2012). Several algorithms for screening and managing these problems have been developed in Australia (Curtis et al., 2012; Stanley and Laugharne, 2014). See also ‘Metabolic syndrome and obesity’ in ‘Section 4. Physical health’.
If anticholinergic drugs are required to treat extrapyramidal side effects, their use should be reviewed after 3 months. It is sometimes possible to gradually withdraw the anticholinergics without changing the dose of the antipsychotic. Cessation of anticholinergics can be associated with improvement in cognition and in tardive dyskinesia (Desmarais et al., 2012).
Other classes of medicines commonly used in schizophrenia
Benzodiazepines
Benzodiazepines are commonly used for people with schizophrenia or related disorders, especially where there is a persistent high risk of aggression. While there is no good evidence for their use in the longer term (Dold et al., 2013), these agents can be useful in managing anxiety or brief exacerbations of symptoms.
Mood stabilisers
Mood stabilisers such as lithium and sodium valproate are commonly added to antipsychotic agents, especially in individuals who have an affective component to their illness, although there have been few methodologically sound RCTs evaluating the efficacy of these drugs in the management of schizophrenia (Leucht et al., 2007, 2014; Schwarz et al., 2008). Care needs to be taken with adding mood stabilisers, as an increase in mortality has been reported with antipsychotic–mood stabiliser combinations (Tenback et al., 2012).
Antidepressants
In the absence of comorbid depression, evidence for the use of antidepressants in people with psychotic illness is equivocal (Sommer et al., 2012) with limited evidence of benefit.
Omega-3 fatty acids
Fish oils contain EPA and docosahexanoic acid (DHA). Fish oil supplementation showed benefit for people with an at-risk mental state in a single study (Amminger et al., 2010), but this has yet to be replicated. An earlier Cochrane review (Irving et al., 2006) found that the evidence for using supplementary fish oil in people with schizophrenia was inconclusive.
Emerging treatments
It is important that clinicians independently review the evidence base for new treatments. New SGAs include lurasidone, which has recently become available, and iloperidone, which may be available soon. There are several promising emerging pharmacological approaches to the treatment of schizophrenia. A metabotropic glutamate receptor agonist showed efficacy for both positive and negative symptoms of schizophrenia (Marsman et al., 2013; Patil et al., 2007b), but these outcomes were not replicated and the agent has been withdrawn. Other glutamatergic agents are in various stages of development, including the glycine transport inhibitors bitopertin and sarcosine (Arango et al., 2013).
The management of acute behavioural disturbance in schizophrenia and related disorders
The management of the acutely disturbed psychotic person requires calm strategies that protect the safety and dignity of all concerned. The first step should be to try to engage the person and understand what is driving their agitation. Sometimes simple measures such as orienting and explaining what is happening can be enough to defuse the situation. The opportunity should be taken to perform a cognitive screen to exclude delirium/intoxication and also, where possible, physical examination, blood tests, urinary drug screen and an electrocardiogram (ECG).
Attention should be paid to the physical environment, such that stimulation is reduced and safety ensured. Objects that might be thrown or used as weapons should be removed, where possible. An escape route should be made available by ensuring that the exit doorway is not blocked. Back-up should be available and staff should know where duress alarms are situated.
If several staff are involved, ensure one person takes control of the situation and orchestrates matters, with clear communication as to each individual’s role. One person should communicate with the agitated individual in a clear even voice. The individual should be offered reassurance that they are safe and that the intent is to ensure the safety of all concerned. Interventions need to be carefully explained and a rationale provided.
Always try to defuse the situation by careful negotiation. The following techniques can be useful:
Ventilation: allowing the opportunity to express fears, frustration, anger
Redirection: exploring solutions to allow the person to gain control
Time out: offering the person a low stimulus environment, for example, their bedroom or a similar quiet environment to assist them to gain control.
If physical restraint is required, ensure sufficient trained staff are available. If medication is used, the person needs to be informed about what the medicine is and the rationale for its use. Seclusion is rarely required but, if employed, should be used for as short a time as possible and local procedures need to be followed. At a reasonable time interval after restraint or seclusion, the person should be offered the opportunity to debrief about the incident. Service protocols should be reviewed regularly in the light of both recent research and local experience.
If non-pharmacological approaches have been unsuccessful, pharmacological management may be needed (see Figure 2).
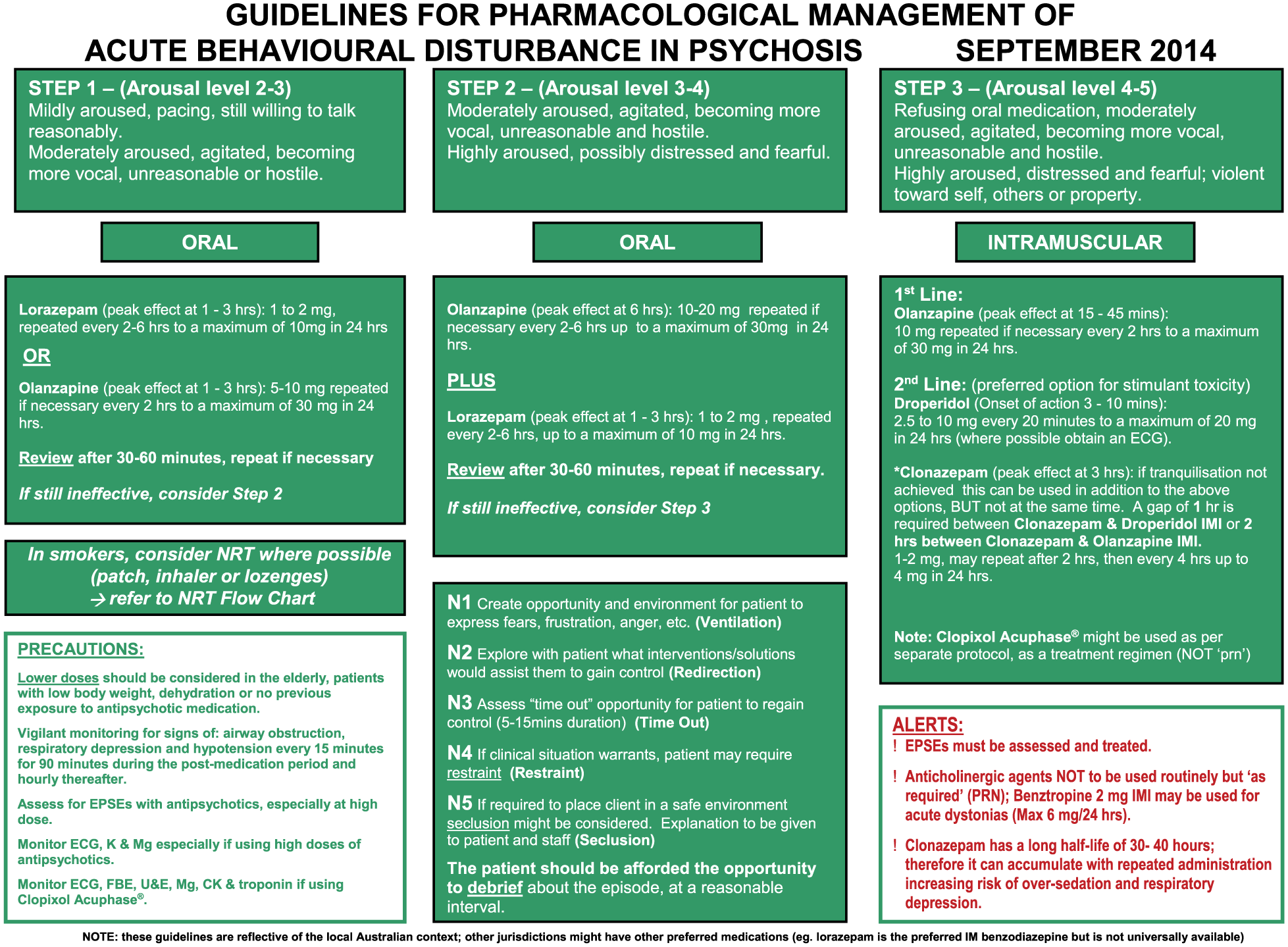
Flowchart for the pharmacological management of acute behavioural disturbance in psychosis. Barnes et al., (2002). Adapted from Castle et al., (2012).
A number of antipsychotic drugs, including olanzapine (10–20 mg), risperidone (2–6 mg), quetiapine (300–800 mg) and aripiprazole (5–30 mg), have been shown to be effective for acute agitation (Kinon et al., 2008; Zeller and Rhoades, 2010). Often, a benzodiazepine, such as lorazepam, is also given. Particular care should be taken with midazolam because of the risk of respiratory depression. Oral administration of medication is preferable, but if this is not possible, intramuscular preparations may be used. Intramuscular olanzapine and benzodiazepines should not be given together because there is a risk of respiratory depression with this combination. In selecting the type and dose of medications, individual factors such as the person’s ethnicity, whether they are drug naive and current medications and substance use must be taken into account.

CBR: consensus-based recommendation; EBR: evidence-based recommendation.
N/A: Level of evidence category does not apply; recommendation based on a combination of available evidence, clinical experience and expert consensus.
Nicotine transdermal patches may be helpful in reducing agitation and aggression in smokers with schizophrenia if they are prevented from smoking in hospital (Allen et al., 2011). This finding reinforces the importance of establishing whether people are smokers and considering whether some agitation might be due to nicotine withdrawal.
Catatonia is a rare condition that can occur in both schizophrenia and mood disorders (Grover et al., 2015). While retarded catatonia is more common, excited catatonia may occasionally present as an acute behavioural disturbance. Catatonia should be treated with IV lorazepam or emergency ECT (Sienaert et al., 2014).
Neurostimulation therapies
Electroconvulsive therapy (ECT)
A Cochrane review (Tharyan and Adams, 2005) concluded that ECT, either alone or added to antipsychotic medication, resulted in increased rates of global improvement and faster rates of symptomatic improvement than sham ECT or placebo in people with schizophrenia. ECT may have a particular role in the management of treatment-resistant schizophrenia, although there are few studies. A small randomised sham-controlled trial of ECT in treatment-resistant schizophrenia (Goswami et al., 2003) found no clear advantage for active ECT, but open studies suggest that adding ECT to antipsychotic agents is a safe and potentially useful strategy for people with treatment-resistant schizophrenia (Braga and Petrides, 2005; Chanpattana and Sackeim, 2010). The combination of ECT with clozapine may be of particular benefit for people who have an inadequate response to clozapine alone (Havaki-Kontaxaki et al., 2006; Petrides et al., 2015).
Any advantage of adding ECT to antipsychotic medication, compared with antipsychotic medication alone, is no longer evident within 6–8 weeks of ceasing ECT, despite ongoing antipsychotic treatment (Tharyan and Adams, 2005). In the only RCT of continuation ECT in people with treatment-resistant schizophrenia who had responded to the combination of ECT and antipsychotic medication, rates of relapse were much lower with the combination of ECT and antipsychotics, compared with either treatment alone (Chanpattana et al., 1999).
In a limited analysis of data from studies comparing bitemporal and unilateral ECT, Tharyan and Adams (2005) found no clear advantage for either electrode placement. Recent data suggest that bifrontal ECT may be more effective than bitemporal ECT in people with schizophrenia and schizoaffective disorder, with the added benefit of causing less cognitive impairment (Phutane et al., 2013). This finding is yet to be replicated. One randomised study observed a more rapid response in people treated with bitemporal ECT at doses two and four times seizure threshold, compared with treatment at the seizure threshold, but there was no difference in outcome at the end of the course (Chanpattana et al., 2000). Ultra-brief ECT is likely to have significant advantages in reducing cognitive side effects, although its efficacy in schizophrenia is unproven. There has been one case report of the successful use of right unilateral ultra-brief ECT in the treatment of catatonic schizophrenia (Cupina et al., 2013).
There has been a traditional view that prolonged courses of ECT (up to 20 index treatments) are required for people with schizophrenia. However, in RCTs where ECT has been demonstrated to be of benefit in combination with medication, 8–12 index treatments were efficacious (Abraham and Kulhara, 1987; Brandon et al., 1985; Taylor and Fleminger, 1980). Given the relationship between bitemporal ECT and increased cognitive adverse effects in the treatment of depression, careful consideration of the benefits and risks is warranted before prescribing prolonged index courses of bitemporal ECT (Dunne and McLoughlin, 2012; Phutane et al., 2013; Sackeim et al., 2006). The adverse cognitive effects of ECT in people with schizophrenia appear to be consistent with those observed in the treatment of depression. However, detailed studies in people with schizophrenia are lacking. Available evidence suggests that positive rather than negative symptoms predominantly respond to ECT (Chanpattana and Sackeim, 2010). The use of an objective rating scale to assess symptom severity and quantify response is strongly recommended.

ECT: electroconvulsive therapy.

EBR: evidence-based recommendation; CBR: consensus-based recommendation.
N/A: Level of evidence category does not apply; recommendation based on a combination of available evidence, clinical experience and expert consensus.
Repetitive transcranial magnetic stimulation
There is some evidence for the efficacy of active low frequency (⩽1 Hz) repetitive transcranial magnetic stimulation (rTMS) applied to the temporo-parietal cortex in treating auditory hallucinations (Zhang et al., 2013b). There is also limited evidence for the efficacy of rTMS applied to the left dorsolateral prefrontal cortex to treat negative symptoms of schizophrenia (Freitas et al., 2009; Prikryl et al., 2007). Given that rTMS is generally well tolerated, a trial of rTMS is a low risk strategy with some possibility of benefit.
Transcranial direct current stimulation
The use of transcranial direct current stimulation (tDCS) to treat schizophrenia is still in the research phase. There is preliminary evidence that tDCS may have benefits in treating auditory verbal hallucinations, and positive and negative symptoms of schizophrenia generally (Brunelin et al., 2012), although these results need further replication. Research is underway to evaluate the potential role of tDCS in cognitive remediation in schizophrenia, when given concurrently with cognitive training exercises (Tarur Padinjareveettil et al., 2014).
Psychological and psychosocial therapies
Overview
There is a growing evidence base to support the use of psychotherapy and psychosocial strategies for psychosis. These should be provided along with optimal antipsychotic medication. There is clear evidence for CBT for psychosis and cognitive remediation, with an emerging evidence base for other therapies (Lyman et al., 2014; Wykes et al., 2008, 2011). This trend is likely to continue, given clinicians’ appreciation of the limits of pharmacotherapy in addressing all the domains of schizophrenia and the current focus on person-centred, individualised care.
The therapeutic relationship is the cornerstone of effective treatment. All clinicians working with people with schizophrenia need psychological skills. Services should ensure that clinicians can spend time delivering psychological therapies. People living with psychosis may have experienced many losses, traumas and hardships, and rejection by others and by society. Clinicians must acknowledge these painful aspects of the person’s life and respond empathically. A promising new possibility is to draw on techniques from positive psychology to enhance positive mental health and wellbeing. Much of the work of the growing non-government sector has begun to focus on this aspect but more research, skill development and training is required. It is important for clinicians to expand their mindset and role to accommodate these new developments.
Potential barriers to offering psychological and psychosocial therapies include the limited tenure of admission to a specialist mental health service, the frequent rotation and turnover of clinicians, the possible limitations on time spent with individuals and families, the fragmentation of the clinical and non-government services, and the lack of training in psychological therapies.
Cognitive function
Cognitive deficits need to be taken into account in planning psychosocial treatments for the individual. Eight cognitive domains are commonly (but not uniformly) impaired in people living with schizophrenia:
Speed of processing
Attention/vigilance
Working memory (non-verbal)
Working memory (verbal)
Verbal learning
Visual learning
Reasoning and problem solving
Social cognition, defined as the combination of facial affect recognition, social perception and processing, theory of mind and social attributions (Nuechterlein et al., 2008a).
Impairment in these domains influences functional capacity and limits functional outcomes, either directly or by moderating other variables. For example, working memory and attention have direct effects on work, executive skills and interpersonal behaviour. A distinction can be made between functional capacity and performance, with some cognitive domains necessary to improve skills (verbal memory and executive functions), while other functions relate to competence (e.g. processing speed and attention/working memory associated with social competence) (Bowie et al., 2008). Processing speed underpins performance in other domains and appears to be the core impairment in skill acquisition and everyday functional performance.
Several tools for cognitive screening, which were developed for research purposes, have been considered for routine clinical use to enable clinicians to assess the cognitive dimension of the illness and to interpret and apply this knowledge to treatment plans (Velligan et al., 2004). The Brief Cognitive Assessment (BCA) (Velligan et al., 2004) and the Brief Cognitive Assessment Tool for Schizophrenia (B-CATS) (Hurford et al., 2011) have sound psychometric properties and good correlations with functional measures and can be completed in 15 minutes. The Brief Assessment of Cognition in Schizophrenia (BACS) battery has good psychometric properties, has alternate forms to minimise practice effects and the composite score relates strongly to functional measures (Keefe et al., 2006a). In the future, cognitive screens suitable for clinical use may be available via smart technology. Cognitive assessment should take place as part of the initial assessment (once the acute symptoms have settled) and before and after specific interventions.
Ideally, mental health services for people with schizophrenia would have access to a neuropsychologist experienced in psychosis who could provide comprehensive neurocognitive testing and sophisticated interpretation of results. However, this is uncommon in clinical practice. More routinely, multidisciplinary teams have access to occupational therapists skilled in administration and interpretation of the Allen’s Cognitive Level Scale, which correlates well with neuropsychological tests and can be used to guide care plans (Su et al., 2011).
Clinically, the focus should be on improving cognitive functioning in the service of improving functional competency in the ‘real world’. A number of interview-based cognitive measures have been developed with arguably more face validity with regard to cognitive function. The Schizophrenia Cognition Rating Scale (SCoRS) (Keefe et al., 2006b) and the Cognitive Assessment Interview (CAI) (Ventura et al., 2010) have been found to relate well to performance-based measures of cognition. These measures have also been found to explain variance in real-world functioning not explained by standard cognitive batteries.
Psychoeducation
Psychoeducation programmes for people with schizophrenia improve adherence to treatment and lead to better outcomes, better management of subsequent relapse, lower readmission rates and a greater sense of wellbeing (Xia et al., 2011). In the early phases, psychoeducation tends to focus on support and educating the person with schizophrenia and their family about the illness, using a biopsychosocial perspective. As the individual recovers, the subject matter may evolve to more general topics, such as life skills (Medalia and Revheim, 2002). Provided consent is given, information should also be given to others who are in contact with the person with schizophrenia. These may include staff of community agencies, NGOs and supported accommodation residences.
Illness self-management training
Illness self-management training is concordant with the recovery model in providing access to knowledge to allow informed decision-making as well as teaching strategies to assist in medication adherence and relapse prevention. Core components of training include psychoeducation, behavioural interventions to facilitate adherence, coping strategies for residual symptoms and a relapse prevention plan (Mueser et al., 2013).
There are many different evidence-based peer-delivered programmes. Wellness recovery action planning (WRAP) (Cook et al., 2012) and the Illness Management and Recovery (IMR) programme (Mueser and Gingerich, 2011) have the most evidence for effectiveness.
Family support and psychoeducation
Families of people with schizophrenia experience tremendous distress, grief and chronic day-to-day stress, which can be extreme and result in significant risks to their health and wellbeing. These issues have generally been neglected by services and by many health professionals – yet effective support for families is crucial, since for many people with schizophrenia, survival and recovery depend on their family relationships. Families deserve involvement and support from professionals wherever possible.
The link between family burden and distress and outcomes for the person with schizophrenia has led to the recognition that optimal care should include significant others in the individual’s recovery environment, particularly their family. Multiple programmes and modalities (individual versus group) have been empirically validated (McFarlane, 2002; Pilling et al., 2002). Common elements include education about the illness and coping strategies (Sin and Norman, 2013).

EBR: evidence-based recommendation.
Peer workers
Many services now employ peer workers as part of the clinical team, or engage them as clinical consultants (Nestor and Galletly, 2008). Peer workers can assist with motivation, building confidence and improving social interaction (Henderson and Kemp, 2013). There is a rise in peer-delivered therapies such as voice hearer groups and strengths-based interventions (Romme and Escher, 2012). Peers are also delivering lifestyle interventions, such as quit-smoking groups (Ashton et al., 2013). A recent Cochrane review concluded that involving peer workers (also called consumer-providers) in mental health teams results in psychosocial, mental health symptom and service use outcomes for clients that were no better or worse than those achieved by professionals employed in similar roles, particularly for case management services (Pitt et al., 2013). There is low-quality evidence that involving consumer-providers in mental health teams results in a small reduction in clients’ use of crisis or emergency services. These workers generally receive training through educational courses, often run by NGOs. Their lived experience can be very valuable, especially for young people with FEP.
CBT for psychosis
CBT for people with psychosis (CBTp) has emerged as an evidenced-based treatment usually used as an adjunct to pharmacotherapy to reduce the distress and disability associated with residual or persistent psychotic symptoms. CBTp is more effective than other talking therapies such as supportive counselling for hallucinations, and there may be long-term advantages for CBTp in dealing with emotional distress and depression, but advantages over other therapies for overall mental state and delusions are not established (Jauhar et al., 2014; Jones et al., 2012; Van der Gaag et al., 2014). The low risks and broad potential symptom targets of CBTp have resulted in its use in both FEP and persistent illness. A multicentre RCT of CBTp in people at risk of psychosis reported lowered symptom severity but no impact on rates of transition to psychosis (Morrison et al., 2004).
Although positive symptoms were the initial targets of CBTp, research has also been conducted using CBTp for negative symptoms and for comorbid depression, anxiety and substance use (Wykes et al., 2008).
Cognitive remediation therapy and compensation
Cognitive remediation aims to improve cognitive processes (attention, memory, executive function, social cognition or meta-cognition). There is emerging evidence of associated improved real-world function (Wykes et al., 2011), but the outstanding issues for further research include the mechanisms of change and ensuring real-world transfer. A number of cognitive remediation programmes are available online, although not all of these programmes have been evaluated. The evidence to date relies on programmes facilitated by clinicians trained in cognitive remediation therapy (CRT).
CRT has been used at all stages of illness including at-risk states, early psychosis and in people with established illness. When combined with vocational rehabilitation, CRT is associated with an increase in hours worked and job retention (McGurk and Wykes, 2008).
Compensatory strategies, as distinct from remediation, can also be useful (e.g. diaries, calendar, mobile phone reminders and environmental modifications). A manualised form of compensatory strategies, ‘cognitive adaptation therapy’, should be considered, particularly if there is no access to CRT or the individual prefers to utilise cognitive adaptation therapy (Velligan et al., 2008).
Emerging psychological and psychosocial therapies
Social cognition therapy
Social cognition is defined as ‘the ability to construct representations of the relations between oneself and others, and to use those representations flexibly to guide social behaviours’ (Adolphs, 2001). Components of social cognition include theory of mind (the ability to infer other’s mental states), emotional and social perception and social attribution biases. Impairment in social cognition is a separate domain of pathology, independent of positive symptoms and only partially accounted for by negative symptoms and impairment in neurocognition.
People with schizophrenia generally have impairments in social cognition. A number of social cognition remediation programmes have been developed, including the SoCog programme (Marsh et al., 2013) and Social Cognition and Interaction Training (Combs et al., 2007; Roberts et al., 2014). Evaluations of these programmes have been encouraging, especially in regards to improving emotional perception (Kurtz et al., 2012; Kurtz and Richardson, 2011). The association of both neurocognition and social cognition with community function has resulted in programmes integrating the two therapies, for example, cognitive enhancement therapy (Eack et al., 2013; Hogarty et al., 1997).
Acceptance and commitment therapy and mindfulness-based therapies for psychosis
Third-wave CBTs, including ACT and mindfulness groups, aim to modify the person’s relationship with their psychotic experiences (Khoury et al., 2013). A key premise is that attempting to suppress mental events can paradoxically bring these experiences into the foreground. These therapies promote an alternate stance of noticing the experiences, non-judgementally and without struggle, to reduce the extent to which they dominate the person’s experience and behaviour. Both involve teaching meta-cognitive awareness, meditation and mindfulness skills. ACT also focusses on the person’s goals and values. ACT is supported by some RCT evidence (Gaudiano and Herbert, 2006; Shawyer et al., 2012), but it remains unclear when or for whom these approaches might be particularly indicated. An Australian trial of ACT in psychosis is currently underway (Thomas et al., 2014).
Meta-cognitive training
Meta-cognitive training (MCT) is a therapy influenced by CBT and CRT approaches to psychosis (Moritz et al., 2010). MCT focusses more on the process of thinking, especially cognitive biases, rather than the content. Therapy is usually in a small group psychoeducation based format with additional individual sessions. Early studies have found improvement in positive symptoms and self-efficacy (Balzan et al., 2014; Moritz et al., 2014).
Other therapies
Other therapies requiring more research and evaluation include the following:
Art therapy is included in the UK NICE clinical guideline on the treatment and management of psychosis and schizophrenia in adults (NICE, 2014), but RCT evidence is inconclusive (Crawford, 2012).
Music therapy has a long history in schizophrenia and recent trials support its efficacy in social domains and quality of life (Grocke et al., 2009, 2014; Mössler et al., 2011).
Narrative therapy aims to promote the development of positive personal narratives along with competence and a sense of control over one’s life (Dickerson and Lehman, 2011). There are several forms of narrative therapy including some that integrate CBT techniques. This approach is congruent with the emphasis on the person and their subjective experience (Strauss, 2013).
Solution-focussed therapy is an approach that is not specific for any particular condition but has been used in a range of mental health conditions. The approach is concordant with a recovery approach, reducing the emphasis on problems and focussing on future opportunities.
Sensory modulation/integration therapy has been used in occupational therapy since the 1970s. More recent modifications have led to a renewed interest in its application to schizophrenia, especially to reduce seclusion and restraint (Chalmers et al., 2012).
Open Dialogue, developed in Finland by Seikkula (2002), focusses on the social network and on establishing a therapeutic dialogue. Further research would be needed to provide evidence to support this approach.


CBR: consensus-based recommendation; EBR: evidence-based recommendation; CBTp: CBT for people with psychosis.
N/A: Level of evidence category does not apply; recommendation based on a combination of available evidence, clinical experience and expert consensus.
Vocational rehabilitation
The desire to work in the open labour market is often a priority for people with psychotic illness (Morgan et al., 2011; Waghorn et al., 2012). Vocational rehabilitation aims to improve economic and social participation. There are two main models (Drake et al., 1999, 2003, 2012):
Prevocational training – training is provided during a period of preparation before the person seeks competitive employment
Supported employment – people are placed in competitive employment with on-the-job support.
A Cochrane review (Kinoshita et al., 2013) found that supported employment was more effective than other strategies (including prevocational training) in improving vocational outcomes relevant to people with severe mental illness. The most effective form of supported employment was individual placement and support. On average, 60% of people given individual placement and support returned to employment, compared with 25% in the various control groups (Bond et al., 2012).
Trials of individual placement and support in FEP have extended vocational outcomes to include education, which has boosted vocational outcomes, with approximately 85% returning to school or work (Killackey et al., 2008). The addition of targeted CRT can further enhance rates of attaining and retaining work among people with schizophrenia (McGurk and Wykes, 2008).
The lack of effective vocational rehabilitation services is a major deficit in many mental health services. Lack of employment has serious consequences in terms of self-esteem, income, personal growth and social inclusion. In general, job agencies, even those with an interest in rehabilitation of people with disabilities, are not effective in getting people with schizophrenia into the workforce. As with so many other aspects of care, specialised services for people with schizophrenia, integrated into the mental health services, are needed.
For a discussion of the right to work for people with psychosis, refer to ‘Meaningful lives. Supporting young people with psychosis in education, training and employment’ (International First Episode Vocational Recovery Group, 2008).


EBR: evidence-based recommendation; CBR: consensus-based recommendation.
N/A: Level of evidence category does not apply; recommendation based on a combination of available evidence, clinical experience and expert consensus.
Involuntary treatment
Schizophrenia is often associated with poor recognition of the origin of symptoms and the need for treatment. The main purpose of mental health laws is to allow for the involuntary treatment of people who do not recognise the need for that treatment, while safeguarding the rights of the mentally ill. Improving the person’s capacity to make sound treatment decisions is an objective of the recovery model of care, and an important function of mental health laws is to allow treatment in order to restore the capacity to make rational decisions in people who have temporarily lost that capacity.
The mental health laws of the states and territories of Australia and in New Zealand are mainly concerned with protecting individuals from harming themselves and others. A further consideration in some laws is to allow treatment of people who have serious impairment in their capacity for self-care. Never-treated psychosis is associated with a greatly increased rate of self-harm and violence (Nielssen and Large, 2010), and a long duration of untreated psychosis is associated with less complete recovery. Hence, involuntary treatment may be indicated for people in the first episode of psychosis who cannot be persuaded to have a trial of treatment with antipsychotic medication. Involuntary treatment may also be warranted for people who have previously responded to treatment but have experienced a relapse of illness and no longer recognise the need for treatment.
One of the challenges in clinical practice is the consideration of when or whether involuntary treatment, either in hospital or in a community setting, is helpful for a person’s recovery. Forming a therapeutic alliance can be difficult when working with people who have had adverse experiences of involuntary treatment and treatment with antipsychotic medication. Decisions to invoke involuntary treatment should consider the person’s best interests and their perception of the value of treatment when they are well, if possible, in the form of an advance directive to their carers.
Although community treatment orders (CTOs) have been associated with controversy regarding both ethics and efficacy, Australia has one of the highest rates of CTOs in the world, with as many as 10% of people with schizophrenia on a CTO (Light et al., 2012). On balance, there is some evidence to suggest that involuntary community treatment may be helpful for people with schizophrenia when there have been difficulties with voluntary engagement with services and there has been a pattern of harmful behaviour or serious functional impairment associated with exacerbations of illness after refusing treatment. Involuntary community treatment is not a substitute for effective person-centred care but is a way to facilitate engagement in treatment to prevent worse outcomes. Treatment plans that form the justification for any CTO should include information regarding the individual’s wishes in the form of previous advance directives and collaborative recovery plans.


CBR: consensus-based recommendation.
N/A: Level of evidence category does not apply; recommendation based on a combination of available evidence, clinical experience and expert consensus.
Section 4. Physical health
Metabolic syndrome and obesity
People with schizophrenia have significantly higher than expected rates of ischaemic heart disease, cerebrovascular disease and diabetes. In the second Australian National Survey of Psychosis (Morgan et al., 2011), 53% of participants aged 18–64 years met criteria for metabolic syndrome, including at-risk levels of abdominal obesity (82%), high-density lipoproteins (50%), triglycerides (48%) and hypertension (49%). About one-quarter were at high risk of a cardiovascular event in the next 5 years. Less than half of those with known hypertension, hyperglycaemia or elevated cholesterol were receiving medication for these conditions (Galletly et al., 2012).
Metformin may reduce the risk of obesity and diabetes (Correll et al., 2013), but can be associated with Vitamin B12 deficiency. Statins, antihypertensive medicines and other evidence-based treatments for metabolic conditions should be prescribed as indicated. Many mental health services now undertake cardiometabolic monitoring (Stanley and Laugharne, 2014). It is essential that this is linked to effective treatment, generally involving close liaison with primary care (which may be co-located with the community mental health service). Another option is for psychiatrists to take responsibility for at least some of the necessary investigations and prescribing of medications for physical conditions, with referral to specialised medical services as needed. This may require some up-skilling but is no more complicated than managing lithium or clozapine.
Lifestyle interventions must take into account the cognitive dysfunction, impaired motivation and energy, and low income of most people with schizophrenia. The second Australian National Survey of Psychosis (Morgan et al., 2012) found that the majority of participants reported very low levels of physical activity, so exercise needs to start at an appropriate level. There is evidence that an 18-month behavioural weight loss programme can assist people with schizophrenia to lose weight (Daumit et al., 2013). Baker et al. (2015) reported some improvements with an intervention targeting both smoking and cardiovascular risk factors in people with psychotic disorders, but much more research and evaluation is urgently needed to find effective interventions that can be widely adopted across mental health services (Gates et al., 2015).
The importance of holistic preventive health care in people with FEP is described in the HeAL consensus statement 2013 (available at: http://www.iphys.org.au). Information about managing cardiometabolic effects of antipsychotic medication in people with FEP is found in Positive Cardiometabolic Health: an early intervention framework for patients on psychotropic medication, NSW Government Health Education Training Institute (2011) (available at www.heti.nsw.gov.au/cmalgorithm).
Smoking
The second Australian National Survey of Psychosis found that two-thirds of participants were current smokers and 81% had a lifetime history of smoking (Cooper et al., 2012). There has been no change in the prevalence of smoking among people with schizophrenia between 1999 and 2010 (Morgan et al., 2012), indicating that general population measures such as media advertisements and telephone support lines are ineffective for this group of people.
However, both individual and group smoking cessation programmes that are specifically tailored to the needs of people with schizophrenia can be helpful (Bennett et al., 2013). Group treatment programmes involving peer workers have been shown to be effective in Australian mental health services (Ashton et al., 2015). Nicotine replacement therapy is often useful. Simple guidelines are available for the management of smoking in people with schizophrenia (Mendelson et al., 2015). Smoking increases the metabolism of some antipsychotic agents, and doses may need to be reduced when people quit smoking (see ‘Smoking and antipsychotic medication’ in ‘Section 3. Treatment: context, structure and content of interventions’).
Obstructive sleep apnoea
Obstructive sleep apnoea (OSA) is a breathing disorder characterised by repeated collapse and obstruction of the upper airway, causing nocturnal hypoxia and sleep arousals (Patil et al., 2007a). Symptoms include non-rejuvenating sleep, daytime hyper-somnolence, cognitive impairment, morning headaches, dry mouth, nocturia, snoring and breathing pauses during sleep (Lal et al., 2012; Patil et al., 2007a). People with schizophrenia have a high prevalence of OSA risk factors including obesity, smoking, alcohol consumption and hypnotic medication use (Al Lawati et al., 2009; Galletly et al., 2012). OSA is associated with an increased risk of stroke, cardiac arrhythmias, heart failure, diabetes, hypertension, cardiovascular disease and motor vehicle accidents (Al Lawati et al., 2009; Drager et al., 2013; Gami et al., 2013; Kendzerska et al., 2014).
OSA may be more likely, and less detectable, among people with schizophrenia in whom sleep disturbance, hyper-somnolence and cognitive impairment may be mistaken for negative symptoms of schizophrenia or medication side effects. A systematic review found that the prevalence of OSA varied from 1.6% to 52%; OSA was associated with male gender, age > 50 years and obesity (Myles et al., 2016). It is imperative to assess for OSA by questioning about daytime somnolence, snoring and breathing pauses. If OSA is suspected, the person should be referred to a respiratory physician or sleep physician.
The treatment of OSA is non-invasive nocturnal ventilation, usually via continuous positive airway pressure (CPAP). CPAP has been demonstrated in controlled trials to improve sleep symptoms and improve blood pressure and heart failure in people with sleep apnoea (Gami et al., 2013; Kendzerska et al., 2014), but as yet there are no published trials of CPAP in people with schizophrenia who have OSA.
Other physical disorders
People with psychotic disorders living in Australia have higher rates of virtually all common physical disorders including chronic pain, headaches, asthma and arthritis (Morgan et al., 2012). They also tend to have poor dental health. Good quality coordination, delivery and monitoring of physical health care are essential.


GP: general practitioner; EBR: evidence-based recommendation; CBR: consensus-based recommendation.
N/A: Level of evidence category does not apply; recommendation based on a combination of available evidence, clinical experience and expert consensus.
Section 5. Specific populations and circumstances
Aboriginal and Torres Strait Islander peoples
The Social and Emotional Wellbeing Framework 2004–2009 (Social Health Reference Group for National Aboriginal and Torres Strait Islander Health Council and National Mental Health Working Group, 2004) sets out a holistic approach to the health of Aboriginal and Torres Strait Islander people that is concordant with the cultural and spiritual beliefs of Australia’s first people. This paradigm recognises the complex interplay of factors determining health and mental health outcomes, while acknowledging resilience and strength in the face of adversity.
It is essential for mental health workers who provide care for Aboriginal and Torres Strait Islander people with psychotic illness to appreciate the psychosocial context set out in this framework and important related documents:
National strategic framework for Aboriginal and Torres Strait Islander health, 2003–2013 (National Aboriginal and Torres Strait Islander Health Council, 2004).
A Contributing Life, the 2012 National Report Card on Mental Health and Suicide Prevention (National Mental Health Commission, 2012) – acknowledges the historical context of colonisation, dispossession and displacement and calls for a more nuanced understanding of the diversity of the Aboriginal and Torres Strait Islander peoples with multiple language groups, cultures and traditions.
The mental health and social and emotional wellbeing of Aboriginal and Torres Strait Islander peoples, families and communities (Holland et al., 2013) – a supplementary paper to A Contributing Life.
There is a lack of comprehensive epidemiological data for Aboriginal and Torres Strait Islander populations for mental health generally and for psychosis specifically (Black et al., 2015). A number of issues have contributed to this lack of research, including methodological issues relating to the need for culturally appropriate tools and research approaches and the diversity of Aboriginal and Torres Strait Islander populations, which makes accurate generalisation difficult (Haswell-Elkins et al., 2007). A study of psychosis in the Indigenous populations of Cape York and the Torres Strait (Hunter et al., 2012) found an overall treated prevalence rate of 1.68%, with higher rates for males than females and higher rates for Aboriginal people than Torres Strait Islander people.
Aboriginal and Torres Strait Islander peoples have disproportionately high rates of suicide, substance use and incarceration. Aboriginal people are hospitalised for mental health and behavioural disorders at around 1.7 times the rate of other Australians (Dudgeon et al., 2014). The prevalence of mental disorder among Indigenous adults in custody across Australia is also likely to be disproportionately high, based on findings in a Queensland prison study (Heffernan et al., 2012). Rates of mental illness among Indigenous people in custody were found to very high compared with community estimates, including rates of psychotic illness of 8% for men and 23% for women.
Social disadvantage is an important contributing factor to mental disorders, including psychosis, in Aboriginal and Torres Strait Islander people (Hunter, 2007). Such disadvantage has a compounding effect on probable rates of psychotic illness, through increased rates of developmental disability and acquired cognitive impairment, secondary to lack of access to antenatal care and to affordable quality nutrition in more remote communities, increased rates of childhood adversity and accidental physical trauma in young people – often complicated by maternal substance abuse and exposure to drug and alcohol use earlier in development.
There is a high burden of physical health problems among Aboriginal and Torres Strait Islander people, even young people (Australian Institute of Health and Welfare [AIHW], 2015), which complicates the management of psychosis. There are increased risks for diabetes, heart disease and renal failure along with psychiatric comorbidities such as depression and substance abuse.
High rates of substance use, particularly in some rural and remote Aboriginal and Islander communities, are likely to contribute to higher rates of psychotic illness or a worsening course of illness, particularly combined with poor access to both community and hospital specialist mental health and drug and alcohol services. Although adequate prevalence data are not available regarding differential prevalence and incidence rates of psychotic illness among Aboriginal and Torres Strait Islander populations within metropolitan, regional, rural and remote communities, rates of psychotic illness are possibly inversely proportionate to community size, which can disproportionately affect smaller communities caring for extended family members living with severe and recurring illness, in settings poor in formal mental health services.
When working with Aboriginal and Torres Strait Islander people, it is important to be aware of the complex interplay of culture, context and clinical significance (Hunter, 2014). ‘Symptoms’ require particular consideration during assessment and diagnosis especially for somebody who is presenting for the first time with what appears to be psychotic symptomatology. The presence of culturally influenced symptoms similar to psychosis requires careful consideration in diagnosis. Cultural expressions of grief, trauma and loss may be informed by respect for elders and ancestors.
The use of traditional healers, cultural consultants, cultural treatments and Aboriginal health workers can all aid in a clearer formulation and more accurate diagnosis, or serve to provide a culturally informed second opinion. Elders can provide vital cultural insights to the treating team as well as comfort, support, mentoring and advocacy to the individual (Dudgeon et al., 2014). Non-Indigenous clinicians need to accept and incorporate the advice of Indigenous non-clinicians to ensure that schizophrenia is not diagnosed in error or erroneously attributed to cultural factors, and that the subtleties of the interplay between illness and culture are not overlooked.
When designing care plans for Aboriginal and Torres Strait Islander people, the provision of traditional healing treatments and ways, in partnership with mainstream Western medicine, can bring a sense of environmental familiarity and cultural safety, especially if the person is far from home and family. Establishing specialist Indigenous sub-teams within mainstream mental health services has been shown to be a successful model of care (Catts et al., 2013).
Non-Indigenous health professionals may need to modify their communication styles when working with Aboriginal and Torres Strait Islander patients (Dudgeon et al., 2014). All mental health clinicians working in Australia should have mandatory training in Aboriginal and Torres Strait Islander cultural awareness which should include understanding of the following:
The current relevance of post-colonisation history for Aboriginal and Torres Strait Islander peoples, particularly in regard to collective grief, trauma and loss
A rights-based approach with particular reference to self-determination and social justice
Psychosocial determinants of poor mental health in Aboriginal and Torres Strait Islander populations, particularly unstable poor quality accommodation, poor access to health care, unemployment, social exclusion, stress, trauma, violence and substance use
The principles of ‘A contributing life’ (National Mental Health Commission, 2012) involving ‘thriving not just surviving’, timely and effective care and treatment, meaningful activity and meaningful connections with family, friends, culture and community.
Although the evidence base is lacking, there are some practice principles that should be implemented in clinical care. The following recommendations are based on the current knowledge base and, for the most part, are not specific to schizophrenia or psychosis but reflect a culturally concordant approach.

CBR: consensus-based recommendation.
N/A: Level of evidence category does not apply; recommendation based on a combination of available evidence, clinical experience and expert consensus.
Māori
Māori mental health is an area of high priority due to the prevalence and pattern of mental health disorders among Māori. There has been an extensive body of work throughout New Zealand exploring various models of care specifically relevant to Māori. These developments include best practice principles that incorporate clinical expertise and cultural competency to enhance services to Māori along with the development of outcome measures that are more appropriate and acceptable to Māori. It is beyond the scope of this guideline to comprehensively review this extensive literature; however, it is imperative that the assessment, treatment and care of Māori who suffer with schizophrenia and their whānau (family) are considered within this broader context.
Māori health services
The development of Māori mental health services had a strong foundation in holistic models, such as the Whare Tapa Wha model described by Mason Durie (Durie, 1985), which includes four dimensions of health necessary for Māori wellbeing: spiritual, mental and emotional, physical, and family and community. Te Wheke is another framework of health from a Māori perspective, which has existed for many generations and captures eight dimensions of health (Durie, 1998). Kaupapa Māori services are now common throughout New Zealand and are constantly evolving in response to research and the changing needs of Māori. The key principle underpinning these services is culturally appropriate best practice which has been described as the ‘synthesis between Indigenous values and the highest international clinical standards’ (Te Rau Matatini and The Werry Centre, 2004). The overall aim of these services is to enhance recovery for Māori who access Mental Health Services by having a service run by Māori for Māori. Kaupapa Māori services operate in the community and in forensic services, inpatient settings and the non-government sector.
The three principles of a Kaupapa Māori service are:
Tino Rangatiratanga (right to self-determination)
He Tangata He Tangata (recognise the diverse realities of Māori)
Tautau Tautau (collective responsibility of Māori health).
These principles are outlined in Te ara ahu whakamua: Strategic Directions for Māori: A Discussion Document (Ministry of Māori Development, 1993). In Te puawaiwhero, the second Māori mental health and addiction framework, whānau ora (family health) is described as a guiding principle. This acknowledges that mental health for Māori is achieved through maximising the health and wellbeing of the whānau (family) (New Zealand Ministry of Health, 2008). A whānau ora approach means attending to the health and social needs of the whānau of the identified patient. When Māori families are supported to obtain maximum health and wellbeing, this also improves the recovery of the individual.
Providing appropriate care for Māori in mainstream services
Cultural competencies for the non-Māori workforce working with Māori are pivotal to successful service engagement and outcome. Support for the Māori mental health workforce and the appreciation that Māori staff offer a dual competency has been a focus for Te Rau Matatini (Baker and Levy, 2013).
Whether Māori are being assessed and treated in Kaupapa Māori services or in mainstream services, it is appropriate that they have access to cultural support and cultural assessment. Often Māori may have already sought advice from Kaumatua (elders) or tohunga (traditional healers) within their own networks. If this is the case, the person presenting with psychosis and their whānau may have excluded cultural or spiritual attributions for their experiences. However, offering the intervention of Kaumatua (elders, either from within or external to the service) may be a key component to improved engagement and outcome. The individual and whānau may also wish to integrate traditional aspects of health care such as mirimiri (massage) or rongoā (healing native plants) along with usual clinical care. Optimal recovery is often achieved by synthesising both clinical and traditional approaches.
Culturally appropriate outcome measures have also been developed. Hua Oranga (a Māori mental health outcome tool) assesses the effectiveness of mental health treatment and care from the basis of a Māori model of health and uses the perspective of a patient, clinician and whānau member (Kingi and Durie, 1999).
Schizophrenia in Māori
Despite the significant importance of this area, research specifically relating to Māori with schizophrenia is limited. In 2003–2005, Māori were over 3.5 times more likely to be hospitalised for schizophrenia than non-Māori. Māori men had a hospitalisation rate for schizophrenia of 416.7 per 100,000 (age standardised to the Māori population) compared with 222.4 for Māori women, 119.7 for non-Māori men and 62.3 per 100,000 for non-Māori women (Robson and Harris, 2007). One-year prevalence of schizophrenia was estimated at 1% for Māori, which was approximately three times that of non-Māori. Māori men had the highest prevalence rates overall of 1.27% in 2002–2003, followed by Māori women (0.7%), non-Māori men (0.41%), then non-Māori women (0.24%) (Kake et al., 2008). Kake (2008) used a capture–recapture method to estimate the population prevalence of schizophrenia. This involved the data from two large data sets recording the diagnosis of people discharged from public and private hospitals and the diagnosis of community mental health patients. This is the first published account of the prevalence of schizophrenia being calculated using this methodology.
Sociocultural factors of relevance for Māori that could contribute to increased risk of schizophrenia include colonisation, poverty and racism (Harris et al., 2006). Māori men have a higher rate and younger onset of cannabis use, which has been identified as an important risk factor for schizophrenia (Gururajan et al., 2012).
Research on Māori and Pakeha (European) people being treated for schizophrenia within clinical services did not demonstrate a difference in attitude towards medication (Sanders et al., 2011). Being aware of the importance of Māori culture should not come with assumptions that Māori will be necessarily opposed to or not interested in information about schizophrenia as a medical condition and the effective treatments available.
Māori have lower life expectancy and higher rates of heart disease and diabetes than Pakeha. They also have higher rates of poverty and cigarette use (New Zealand Ministry of Health, 2008). While a physical health-monitoring programme for people on SGAs is a key part of any mental health service, this needs to be particularly robust for Māori.

CBR: consensus-based recommendation.
N/A: Level of evidence category does not apply; recommendation based on a combination of available evidence, clinical experience and expert consensus.
New Zealand’s Pacific Islander peoples
Pacific Islander people living in New Zealand (Pasifika peoples) have higher admission rates to acute mental health units than European people (Nielssen et al., 2013). For the inpatient population, they are three times more likely to have a diagnosis of schizophrenia or a psychotic illness than European people (Gaines et al., 2003).
From the late 1990s, there have been national initiatives to improve the mental health outcomes of Pacific people in New Zealand. Pacific-specific services exist both in the non-government sector and the clinical provider sector. These services provide either direct care to clients or support to general mental health teams. A number of frameworks and models have been developed to assist service delivery for Pacific people. These are specific for Cook Islands, Samoan or Tongan people, but all share the concept of health care integrated with respect for spirituality, roles within the family and community and the use of language (Agnew et al., 2004).
The ‘Real skills plus Seitapu’ framework for working with Pacific Peoples recognises the integration of cultural and clinical competencies for staff as an expected standard of care (Te Pou o Te Whakaaro Nui, 2009). Core components of a Pacific mental health service include cultural assessments, use of Pacific language, inviting environments and recognition of spirituality. Assistance with recovery involves having access to culturally and clinically competent staff. Staff also need to be mindful of the importance of stigma for Pacific people with mental illness (Suaalii-Sauni et al., 2009).
Working with a Pacific person who has schizophrenia requires an understanding of family structure, dynamics and roles. Traditionally, Pacific families are extended family groups with the head of the group having responsibility for the wellbeing of all family members. In this case, the leader may make decisions involving the individual’s engagement with treatment and also have a role at mobilising supports. Not all Pacific people will be part of such a structure, as the impact of migration has affected families differently. However, developing rapport with the family as well as the client is an essential foundation of a treatment plan.
Understanding the role of language for a Pacific person with schizophrenia includes an awareness of not only verbal but also non-verbal communication. The use of cultural advisors or interpreters enables better rapport building and sharing of information. Another way to build rapport is to observe and demonstrate signs of respect in Pacific culture, for example, taking off shoes before entering a house and using the appropriate Pacific Island greetings.
Traditionally, Pacific people did not consider mental illness to originate from within an individual, but rather as a spiritual possession or the result of a transgression of tapu prohibitions. Spirituality for Pacific clients and family may be a blend of traditional, Christian and non-Christian beliefs.
Qualitative research has identified barriers to adherence to antipsychotic medication for Samoan people living in New Zealand (Ioasa-Martin and Moore, 2012). A significant barrier to medication adherence is poverty, which affects the ability to purchase medicines and also affects access to transportation for visits to the doctor or chemist. Another barrier to adherence can occur when family members feel shame about the stigma of mental illness and as a result discourage adherence to antipsychotic medication and related activities such as attending medical appointments and visiting the pharmacist. Additional factors thought to affect poor adherence include the attribution of spiritual causes to mental illness (as noted above) as well as, in some cases, the absence of traditional family support structures.

CBR: consensus-based recommendation.
N/A: Level of evidence category does not apply; recommendation based on a combination of available evidence, clinical experience and expert consensus.
Refugees and culturally and linguistically diverse people
International studies have found that both first- and second-generation migrants are at heightened risk of psychotic disorders (Coid et al., 2008). Visible minority status (related to ethnic group or race) is associated with higher risk. The persistence of risk into the second generation suggests that adverse social factors such as discrimination and social defeat play an important role. However, apart from migrants from Oceania, recent migrants to Australia do not have a greater risk of hospital admission for treatment of psychosis (Nielssen et al., 2013).
Social and cultural factors need to be taken into account in managing schizophrenia in people from refugee backgrounds and culturally and linguistically diverse backgrounds. In particular, cultural beliefs about the causes and treatment of psychotic symptoms will influence the attitude of the person and their family to the illness and to treatment. Refugees and migrants have higher rates of substance abuse and post-traumatic stress disorder than the general population, so assessment and treatment of these comorbidities is often needed.
Culture and explanatory models in mental health
Cultural factors will influence presenting features, diagnostic assessment, course and outcome of a range of mental illnesses (Bhui and Bhugra, 2007; Nguyen et al., 2012). Clinicians must be open to the explanatory model used by the person with psychosis. Based on the work of Kleinman and Seeman (2000), this means being open to the way individuals and family members explain, understand or interpret their own or another’s mental illness or mental wellbeing. Procter et al. (2014) explain that ‘a person’s explanatory model will influence the way in which thoughts and experiences are presented, the nature, scope and consequences of distress, behaviour pattern of help seeking, perception of as well as adherence or non-adherence to treatment’ (p. 204). While all consumers make interpretations of their health and wellbeing, cultural explanatory models attempt to understand the deeper meaning structures related to questions such as what something is called and why it started when it did, as well as the severity and likely treatment outcome.
A person’s cultural and linguistic background can greatly influence how Western concepts of illness and wellbeing are understood and, therefore, how conditions such as schizophrenia and related disorders should be treated (Procter et al., 2014). For example, in some cultures and religions, conditions like schizophrenia may be attributed to bad karma, spiritual possession or mind–body imbalance. The mind and associated spiritual aspects of being may be viewed as being inseparable from the body, and this can lead to differences in understanding and explaining a mental illness.
For others, there may be even greater challenges when deep inner emotional or spiritual conflicts emerge. This comes into play when certain religious or cultural beliefs or values, which are protective in other circumstances, break down and such events are attributed to the causes of mental illness. People who feel this sense of failure are often reticent to seek help. In other instances, personal hardship may be seen as a way of life with the conditions of mental distress accepted as fate or destiny.
The Framework for Mental Health in Multicultural Australia (2013) highlights the fact that compared with the general community, people from culturally and linguistically diverse communities (including refugees) receive poorer quality mental health care and are more likely to be exposed to adverse safety risks. Cultural awareness questions (Table 9) are designed to help practitioners understand the nature, scope and potential implications of the person’s explanatory model (Procter, 2007).
Cultural awareness questions for the assessment of people from refugee and immigrant backgrounds.

Source: Procter (2007).
People with Australian Temporary Protection Visas
In December 2014, the Australian Government passed legislation to reintroduce Temporary Protection Visas. These Visas could apply to approximately 30,000 asylum seekers currently living in Australia on Bridging Visas, and to asylum seekers currently held in Immigration Detention Centres on the Australian mainland and on Christmas Island.
Clinicians should be aware of:
Cultural beliefs that might present as psychotic symptoms, such as belief in demons
The variability between interpreters, and challenges in using telephone interpreters instead of face-to-face interpreters
Cultural factors that affect people’s trust in authority figures such as doctors and government health services
The potential call on mental health services to advocate for asylum seekers to remain in Australia
Challenges in obtaining informed consent from some people with schizophrenia, who are used to deferring treatment decisions to doctors
The potential to misinterpret symptoms of other conditions, particularly post-traumatic stress disorder, as symptoms of psychosis
The possible feigning of psychotic symptoms in an attempt to garner support from treating clinicians for asylum seekers to remain in Australia
The effect on asylum seekers of changes in Government policy regarding such issues as family reunification and obtaining permanent residency.
Women
Women with schizophrenia experience the illness differently from men (Seeman, 2013b). A tailored approach to the diagnosis and treatment of psychotic disorders in women may improve their outcomes.
Development of schizophrenia in women
Women tend to have a later age of first onset of schizophrenia (Angermeyer and Kühnz, 1988; Eranti et al., 2013), with better premorbid function and better outcomes (Grossman et al., 2006; Walker and Bollini, 2002). Women have been found to experience more paranoid symptoms (Beratis et al., 1994) and to be more likely to appear depressed than men (Morgan et al., 2008). The presence of affective symptoms is linked to more favourable outcomes (Lewine, 2004). Women with schizophrenia tend to have better insight than men (Gómez-de-Regil et al., 2010) which may also lead to a better outcome. Female incidence rates increase more slowly and show a peak between the ages of 15 and 30 years and then a second smaller peak in the 45–50 age group (Castle et al., 1993).
The oestrogen protection hypothesis
The sex differences noted in the age of onset of schizophrenia, in particular the bimodal distribution of the incidence of new schizophrenia cases in women, led researchers to propose that oestrogen may confer protection against the early onset of severe schizophrenia (Häfner et al., 1998; Seeman and Lang, 1990). During the perimenopausal reduction in oestrogen production, women may have a higher risk of developing psychosis for the first time, and women with schizophrenia may have a higher risk of relapse.
Building on evidence for oestrogen having a neuroprotective effect, clinical trials using oestrogen as a treatment adjunct have shown promising results (Kulkarni et al., 2008a, 2014a). More recently, clinical trials conducted with raloxifene (a selective oestrogen receptor modulator) have also shown promising results for women with schizophrenia (Kulkarni et al., 2010). Adjunctive raloxifene treatment improves attention and memory in men and women with schizophrenia (Weickert et al., 2015), adding further evidence to the hypothesis that oestrogen has a neuroprotective effect.
Pregnancy and schizophrenia
Women with schizophrenia are no different to other women in their desire to become parents (Sands, 1995). In the past, however, women with schizophrenia were either actively discouraged or, more likely, their treating clinicians largely ignored the topic. Discussions between women and their doctors rarely include planning pregnancies, since the control of psychosis symptoms often overwhelms the clinical interaction (Kulkarni, 2006). The preferred community-based treatment model provides greater socialisation and hence greater opportunities for sexual relationships. Clearly, there are many psychosocial issues facing pregnant women with schizophrenia, which clinicians need to address with their female patients (Seeman, 2013a).
Unlike FGAs, some SGAs (e.g. clozapine, quetiapine, aripiprazole) do not increase prolactin, and others (e.g. olanzapine) are less likely to increase prolactin, and hence are less likely to reduce women’s fertility. When changing from a drug that increases prolactin to one that does not cause prolactin elevation, clinicians should inform women about this normalisation of their fertility and the potential for unplanned pregnancies, and should discuss contraception (Psychotropic Writing Group, 2013). Risperidone is still associated with some diminution of fertility through its action of elevating release of prolactin by the pituitary gland (Haddad and Wieck, 2004).
Good antenatal care and the safe delivery of a healthy baby are important outcomes. There are two important goals: to ensure that the mother remains well during the pregnancy and the postnatal periods and to ensure that the developing baby does not suffer malformations or developmental problems (Raha et al., 2012).
There is a lack of evidence-based information about the safety of antipsychotic medicines during pregnancy. An evidence-based guideline for antipsychotic prescribing during pregnancy is needed. The Australian National Register of Antipsychotic Medication in Pregnancy (NRAMP) will provide local data towards developing such guidance (Kulkarni et al., 2008b, 2014b). An NRAMP prospective cohort study (Kulkarni et al., 2014b) found that cessation of antipsychotic medication during pregnancy was associated with relapse of psychosis, maternal hospitalisation and the subsequent separation of mother and baby, leading to attachment and bonding problems. A registry similar to NRAMP has been established in Boston in the United States. Results from the Boston sample of 214 live births to women taking SGAs in pregnancy (Cohen et al., 2015) replicate the NRAMP findings that it would be unlikely for SGAs to significantly raise the risk of major malformations beyond that observed in the general population or among control groups using other psychotropic medicines. If women are taking adjunctive mood stabilisers, then the potential teratogenicity of these drugs must also be considered. Sodium valproate is known to be associated with foetal malformation and must be ceased. Lithium can also cause foetal abnormalities but, despite the risk, is occasionally used during pregnancy – expert advice and antenatal care are essential.
Special antenatal care is needed for pregnant women with schizophrenia, particularly those with relapsing, severe illness. Education about nutrition and ceasing smoking, illicit drug use and alcohol intake during pregnancy are important issues for good antenatal care (NHMRC, 2009a). Unfortunately, most health systems do not provide comprehensive, integrated care for pregnant women with schizophrenia. The communication between obstetric services and mental health systems can be minimal, leading to difficult clinical situations and even postpartum relapse of psychosis. The management of postpartum psychosis needs a holistic approach with a special focus on keeping mother and baby together. Clinicians should aim to enable the woman to mother her child well because this is critical to optimise the mental health of the next generation.
The safety of antipsychotic agents during breastfeeding is also largely unknown and data are also required about this important area. Most clinicians negotiate the issue of breastfeeding with the new mother. The desire of the new mother to breastfeed her newborn, and to provide the best start in life for her baby, is a strong motivator for breastfeeding. However, there are concerns about antipsychotic drug ingestion by the neonate, and this clinical problem requires data to create evidence-based guidelines.
Women whose first experience of psychosis occurs postpartum require highly specialised care. The classification of postpartum psychosis is still unclear, although the symptoms that most women present with seem to share features of schizoaffective disorder and of bipolar 1 disorder, manic episode. Medication management involves a combination of antipsychotic agents and mood stabilisers such as lithium (Dodd and Berk, 2006; Sharma, 2008). Breastfeeding concerns include drug ingestion by the neonate and sleep deprivation for the mother, which can further exacerbate the illness.
Women with postpartum psychosis, either experiencing psychosis for the first time or presenting with relapse of previous psychosis, require management by a team of clinicians who care for the mother and baby together. There are very few public hospital bed-based services that cater to both the woman and her baby. Specialised mother–baby units are required to provide the best possible care, particularly in view of the need for good bonding between mother and child.
Menopause and women with schizophrenia
Many women with schizophrenia experience a worsening of their mental state at this stage of life, probably due to declining oestrogen levels. Perimenopausal women experience hot flushes, sleep disturbances, mood swings and poor cognitive function, as well as worsening of psychotic symptoms (Soares, 2008). Deterioration in a middle-aged woman’s mental state may not be recognised as being due to menopause transition hormone shifts, and treatment involving increased antipsychotic medication, hospitalisations and adding in other psychotropic medicines may only be partially effective.
Hormone Replacement Therapy (HRT) provides an alternative and perhaps more effective approach to the treatment of menopause symptoms, including worsening psychosis. Risks associated with HRT must be considered before prescribing (De Villiers et al., 2013). Good medical care and ongoing monitoring will be needed if HRT is used as a treatment adjunct for menopausal women with schizophrenia. Selective oestrogen receptor modulators, the so-called ‘brain oestrogens’ (Nickelsen et al., 1999), may have a role in the augmentation of antipsychotic agents in perimenopausal and postmenopausal women with schizophrenia (Kulkarni et al., 2010).
Other physical health issues associated with the menopause, such as weight gain, hypercholesterolaemia, diabetes, osteoporosis and increased risk of breast cancer, need ongoing medical monitoring. Many of these physical health issues can be exacerbated by antipsychotic medication; hence, there is a greater need for good physical health care and monitoring for women with schizophrenia as they enter the menopause transition (Gupta et al., 2012; Seeman, 2012).
Psychosocial issues related to being middle-aged, such as fears of ageing, loss of fertility, change in domestic situations and other mid-life events, also need assessment and appropriate action by treating clinicians.
Special prescribing considerations
Clinicians need to factor in the sex, weight and ethnicity of their patients in order to prescribe a correct, therapeutic dose of drug with minimal side effects. Because the pharmacokinetics and metabolism of antipsychotic medicines differ between women and men (Seeman, 2009), drug-related adverse effects can occur in women if they are prescribed doses calculated for men. Antipsychotic medicines are sequestered into adipose tissue differently in women, and different liver enzymatic actions can impact on the availability of the drug to the central nervous system (CNS) (Haack et al., 2009; Yonkers et al., 1992).
Gonadal steroid hormones act in the CNS to affect the drug response and are different in men and women (Kulkarni et al., 2008b). Tardive dyskinesia is seen more commonly in women than men (Usall et al., 2007; Yassa and Jeste, 1992). Weight gain is a troubling side effect of many antipsychotic drugs for men and women (Galletly et al., 2012; Haack et al., 2009). A Japanese retrospective investigation of BMI in patients who had been treated with antipsychotic agents over extended periods (Koga, 2003) observed that the risk of weight gain was significantly higher in women than in men (odds ratio 4.94). A review of antipsychotic adverse effects (Seeman, 2009) reported that women experienced worse effects than men in many domains, including weight gain, movement disorders, cardiac arrhythmias and fertility.
Antipsychotic-induced hyperprolactinaemia has been estimated to occur in up to 70% of patients with schizophrenia, depending on the choice of antipsychotic agent (Inder and Castle, 2011). Antipsychotic drugs that cause hyperprolactinaemia pose special problems for women. Prolactin has potent antagonist impact on oestrogen, progesterone and testosterone production. Long-term use of medicines associated with hyperprolactinaemia can cause osteoporosis in both women and men (Dursun et al., 2008; Petty, 1999). Elevated prolactin levels may also be associated with increased risk of developing rapidly progressing breast cancers (Clevenger and Plank, 1997). While a more recent cohort study has not shown an increase in breast cancers in women with serious mental illness (Osborn et al., 2013), it did not report data on the use of medicines associated with hyperprolactinaemia and the duration of treatment of women with schizophrenia. For good clinical care, it is advisable to recommend breast monitoring and bone density scanning for women older than 50 years who are taking antipsychotic agents that cause hyperprolactinaemia.
Hyperprolactinaemia is also associated with sexual dysfunction, lowered libido, poor vaginal lubrication and anorgasmia. Adding oestrogen or testosterone treatment to the antipsychotic medication may assist these sexual dysfunction problems in postmenopausal and premenopausal women (Davis et al., 2008), but the possible risks of these treatments must be considered. High prolactin levels can also cause anovulatory menstrual cycles and hence infertility.
Psychological treatment for women with schizophrenia
CBT is a useful adjunctive therapy for treatment of persistent symptoms. Specific empowerment issues may need to be addressed (Notman and Nadelson, 2006). Techniques aimed at coming to terms with the illness and the associated losses, and understanding the social context of femininity, are some of the newer approaches that address gender-specific issues.
Cognitive remediation techniques are useful for work skilling programmes for women. Women with longer term schizophrenia may have specific difficulties about entering/re-entering the workforce. It is rare for schizophrenia recovery programmes to focus on parenting skills but for women with schizophrenia who have lost custody of their children due to their illness, this is a key skill which may be needed to be taught in order to regain access to their children. In developing new recovery programmes, the inclusion of mothering skills training should have a high priority. Ongoing support for women with schizophrenia who are caring for children is essential (Campbell et al., 2012).
Service provision for women with schizophrenia
Managing schizophrenia in women requires new approaches that have a clear gender focus. In this way, more specific, better tailored treatments for women with schizophrenia can be developed and implemented to hopefully provide better outcomes for women suffering with the devastating mental illness of schizophrenia.
Over time, the public sector psychiatry wards have come to house more acutely and severely ill patients. In Western countries, most psychiatry wards contain men and women in the same ward setting. Women with schizophrenia can have histories of domestic abuse, and living in mixed gender wards with disinhibited male patients can create situations that may re-traumatise them.
Clinicians have a duty of care to manage their female patients safely and in an environment that promotes recovery. Sexual and other assaults have occurred on inpatient units, and women inpatients experience the majority of such assaults (Johnson, 2006). In 2006, the UK National Patient Safety Agency published a detailed analysis of mental health patient safety incidents that occurred between November 2003 and September 2005, and recommended a policy of gender segregation on psychiatric wards (Johnson, 2006). This is a new trend in psychiatry ward design and will hopefully improve the inpatient treatment experience for women with mental illness


CBR: consensus-based recommendation; EBR: evidence-based recommendation; HRT: Hormone Replacement Therapy.
N/A: Level of evidence category does not apply; recommendation based on a combination of available evidence, clinical experience and expert consensus.
Older people with schizophrenia
Approximately 85% of people with schizophrenia have an onset of illness before age 45, so their experience of ageing is complicated by serious mental illness (McAlpine, 2003). Others experience onset at an older age. There is consensus acceptance of the following diagnostic subtypes (Howard et al., 2000; Rodriguez-Ferrera et al., 2004):
Late-onset schizophrenia (onset between the ages of 40 and 60 years).
Very-late onset schizophrenia (onset after the age of 60 years).
Early- and late-onset schizophrenia share the fundamental clinical features of positive symptoms, negative symptoms and functional deficits (Maglione et al., 2014). A higher proportion of women than men experience late-onset illness. Compared with earlier onset, late-onset schizophrenia is associated with less severe positive symptoms and response to lower doses of antipsychotic medications (Maglione et al., 2014; Vahia et al., 2010).
Unmet needs
Older people with schizophrenia have been overlooked in both research and service delivery in favour of younger populations (Cohen et al., 2000; Green et al., 1997). Provision of care for older people with schizophrenia has been characterised by disputes between adult and older adult mental health services over who takes responsibility for coordinating care (McCleery et al., 2008). Older people with schizophrenia prematurely face challenges associated with ageing because of the earlier onset of medical comorbidities and poorer health outcomes. They are at risk of homelessness, inappropriate placement in acute wards, placement in settings primarily designed for management of dementia, poor medical and end-of-life care, or missing out on service provision altogether (McCleery et al., 2008; McGrath and Holewa, 2004). A collaborative approach is needed across service providers, including general adult and older adult mental health services, as well as multidisciplinary teams (including aged care assessors, geriatric medical and palliative care teams).
Outcomes for older people with schizophrenia
Evidence from studies reporting outcomes for people with schizophrenia in later life is limited, but suggests that older people with schizophrenia remain symptomatic and impaired, with full recovery being rare (Auslander and Jeste, 2004; Jeste et al., 2003). Cognitive impairment has a greater impact on function in daily living than the continued presence of positive and negative symptoms (Twamley et al., 2002).
There is no evidence of an increased incidence of Alzheimer’s disease in older people with schizophrenia, compared with the general population. It has been speculated that the progression of cognitive impairment in schizophrenia might be explained by the concept of decreased cognitive reserve, in which the impact of ageing on the brain is accentuated (Arnold, 2001; Arnold et al., 1998).
Older people with schizophrenia have elevated rates of cardiovascular, respiratory, gastrointestinal, neurological and endocrine diseases, leading to increased mortality and morbidity (Goff et al., 2005), and are more likely than the general population to have unhealthy lifestyle factors. Despite this, older people with schizophrenia are less likely to receive assistance with health interventions such as support with smoking cessation, less likely to obtain correctional hearing and visual aids and are overall less likely to receive best quality medical care (Druss et al., 2001). There has been very little research addressing the issue of end-of-life care for people with mental illnesses, but this population is disadvantaged in accessing quality palliative care (McGrath and Holewa, 2004).
Pharmacological treatments
On average, people with late-onset and very-late-onset schizophrenia respond to much lower doses of antipsychotic medicines than people with early-onset schizophrenia; doses may be reduced by half in the management of late-onset schizophrenia and by as much as 10 times lower in the management of very-late-onset schizophrenia (Howard et al., 2000).
Older people are more susceptible to side effects from antipsychotic medicines. There is an increased risk of death associated with the use of antipsychotics in older people, particularly in people with comorbid dementia (Maust et al., 2015). Older people are more sensitive to anticholinergic side effects, and psychotropic medicines may increase vulnerability to delirium. The metabolic side effects of antipsychotic medicines are particularly relevant. While some older people may receive benefit from clozapine, there is increased risk of clozapine-induced agranulocytosis (Essali et al., 2009). The incidence of tardive dyskinesia in people treated with antipsychotic medications has been found to be 60% in people aged over 65 years and as high as 93% in people aged over 75 years (Byne et al., 1998; Quinn et al., 2001). A correlation between tardive dyskinesia and cognitive impairment has been found (Quinn et al., 2001; Waddington, 1995).
Psychological and psychosocial therapies
Older people with schizophrenia may benefit from a range of psychological and psychosocial therapies. For instance, cognitive behavioural-based social skills training has been shown to improve coping, self-management of symptoms and social functioning (Granholm et al., 2005). Other therapies such as animal-assisted therapies have shown benefit (Barak et al., 2001).
Lifestyle and accommodation support
A wide range of accommodation options is required, ranging from appropriately supported independent community living to high-level care and residential aged care accommodation. Older people with schizophrenia are at risk of being placed in aged care environments that are primarily designed for dementia care, which have marked disadvantages for people with schizophrenia due to their different clinical needs (McCleery et al., 2008).
Coordinated service provision
Discussion of service provision for older people with schizophrenia has focussed on whether people growing older with early-onset schizophrenia should continue to receive services provided by general adult services or transition to services for older adults. Continuing with adult services may provide important continuity for individuals who have established relationships with care providers. Nevertheless, services for older adults may have more developed skills and knowledge relating to managing comorbid cognitive and medical issues associated with ageing and may have improved access to services for older people experiencing frailty and decreasing function. It is increasingly acknowledged that entry points to mental health services for older people should be determined by assessment of individual needs rather than age of onset of illness or previous relationship with an adult service (McCleery et al., 2008).

CBR: consensus-based recommendation; EBR: evidence-based recommendation.
N/A: Level of evidence category does not apply; recommendation based on a combination of available evidence, clinical experience and expert consensus.
Schizophrenia in forensic settings
Studies of mental disorder among prisoners consistently show that about 5% of prisoners have a diagnosis of schizophrenia (Nielssen and Misrachi, 2005). The rate of psychotic disorder appears to be even higher among Aboriginal and Torres Strait Islander people in prison, especially females (Heffernan et al., 2012). Prison is often a suboptimal setting to treat mental illness because of the loss of continuity of care through the frequent movement of prisoners between prisons and in and out of custody, and because the environment in prisons can be threatening and counter-therapeutic.
Schizophrenia is associated with an increased rate of recidivism and re-imprisonment (Fazel and Yu, 2011). Comorbid substance use and non-adherence to treatment are major factors contributing to the higher rate of imprisonment of people with schizophrenia (Yee et al., 2011) and also higher rates of recidivism after release (Fazel et al., 2014). Persistent aggression despite adequate treatment presents a challenge for both forensic and community mental health services, and may warrant a trial of treatment with clozapine, which is reported to reduce aggression and self-harm, even when there is no appreciable reduction in positive symptoms (Frogley et al., 2012).
The interface between prison and community services is often poorly managed. Prisoners released from remote low-security prisons are often distant from their family and friends and from the services likely to be involved in their ongoing care. When there is uncertainty about a released prisoner’s address, it is unclear which service will be responsible for providing care. Staff of community services can be anxious about engaging with people with a history of violence and offending. Finally, there is often a lack of coordination between parole and mental health services.

CBR: consensus-based recommendation; EBR: evidence-based recommendation.
N/A: Level of evidence category does not apply; recommendation based on a combination of available evidence, clinical experience and expert consensus.
Section 6. Other considerations
Housing and homelessness
The poverty and social disability associated with schizophrenia affect the ability to obtain and keep accommodation. As many as one-third of the homeless people in our cities have schizophrenia (Teesson et al., 2004). Moreover, the lack of stable housing has an adverse effect on continuity of care and recovery from episodes of illness. The lack of suitable housing also has an adverse effect on the mental health system, as many hospital beds are occupied by people who cannot be discharged because they do not have suitable accommodation. Improving housing improves many health indicators. The literature clearly supports the ‘housing first’ approach (Chilvers et al., 2006). The level of support needs to be tailored to the social skills and deficits of each individual.
Many people with schizophrenia live with parents or other relatives. The next most common model is public housing, in the form of subsidised single-person dwellings. A third model is private boarding house accommodation, either renting a room or with the addition of meals and supervised medication. A fourth model is purpose built shared accommodation, with a cluster of individual units set around shared facilities, with either a caretaker or concierge monitoring access or support staff providing assistance during the day. There is a clear need for more services in residential settings, including support for family members caring for a person with schizophrenia at home, supervision of adherence to medication, protection from exploitation and assistance to overcome deficits in social skills (e.g. financial management, cleaning and food preparation).
Treatment of people with schizophrenia who are homeless or choose to live in the open presents particular challenges. Common reasons for homelessness include the choice to save money for substance use, as well as the presence of persecutory beliefs involving services and institutions. The challenge is to design services that can attract and serve this group and persuade them to accept more secure forms of housing.

CBR: consensus-based recommendation; EBR: evidence-based recommendation.
N/A: Level of evidence category does not apply; recommendation based on a combination of available evidence, clinical experience and expert consensus.
Comorbid psychiatric conditions
Prevalence and risk factors
People with schizophrenia suffer from high rates of psychiatric comorbidities, including depression and anxiety disorders (Buckley et al., 2009). The lifetime modal rate for major depression in schizophrenia has been estimated at 25%, while rates of depression not meeting major depression criteria are very much higher (Siris, 2000). Overall, lifetime rates of 10–12% for generalised anxiety disorder, 4–20% for panic disorder, 5–20% for agoraphobia and 30% for social anxiety disorder have been reported in people with schizophrenia (Pokos and Castle, 2006). Obsessive compulsive disorder (OCD) appears to become more common as schizophrenia progresses, such that around 11–15% of people with early-onset schizophrenia might manifest the disorder, growing to 22–30% later in the course of the illness (Pokos and Castle, 2006).
Antipsychotic agents with antiserotonergic properties (notably clozapine and olanzapine) can be associated with exacerbation or de novo presentation of OCD (Schirmbeck et al., 2011). Psychiatric comorbidities are often under-treated due to several factors:
The prevailing hierarchical approach to psychiatric diagnoses can lead to clinicians labelling all behaviours and symptoms as part of the schizophrenia syndrome (e.g. assuming social withdrawal is due to negative symptoms rather than other factors such as social anxiety disorder).
Clinicians may fail to ask requisite questions to disentangle core psychotic symptoms from depressive and anxiety symptoms.
Clinicians may believe that psychological and pharmacological treatments for depression and anxiety disorders will not be effective in people with schizophrenia.
Management
There is little evidence to guide the management of comorbid psychiatric conditions in people with schizophrenia. Most studies have focussed on depression and have evaluated pharmacological treatments.
Comorbid depression
Antidepressant medicines appear to be only slightly effective for managing comorbid major depression in people with schizophrenia (Castle and Bosanac, 2012). The potential for drug–drug interactions and additional side effects should be considered before prescribing antidepressants in people already taking psychotropic medicines (Castle and Bosanac, 2012). For example, akathisia can be worsened by selective serotonin reuptake inhibitors (SSRIs) and serotonin–norepinephrine reuptake inhibitors (SNRIs), and weight gain and sedation can be exacerbated by mirtazapine.
It has been suggested that treatment with antidepressants be deferred in the acute phase of psychosis as symptoms may resolve as the psychosis settles, and that some antidepressants may exacerbate psychotic symptoms (Siris, 2000). There is controversy as to whether the SGAs have inherent antidepressant effects in schizophrenia (Castle and Bosanac, 2012). A specific effect of clozapine in reducing suicidality in people with schizophrenia has been demonstrated (Hennen and Baldessarini, 2005; Meltzer et al., 2003).
Psychological approaches have not been widely employed to target depression in schizophrenia. A number of studies using CBT, which have effectively targeted residual psychotic symptoms in schizophrenia, have reported associated improvements in depressive symptoms (Pilling et al., 2002). Social isolation, stigma and family stress are obvious targets for intervention in the depressed person with schizophrenia. The concepts of demoralisation (Clarke and Kissane, 2002) and social defeat (Selten et al., 2013) can assist in understanding the subjective experiences of many people with schizophrenia, especially those with depressive symptoms that may not qualify for a diagnosis of comorbid major depressive disorder. However, very few studies have evaluated the effects of social interventions.
Comorbid anxiety disorders
There is even less evidence to guide the management of anxiety disorders than depression in people with schizophrenia. Very few studies have evaluated treatments for comorbid anxiety disorders in people with schizophrenia. Anxiety symptoms often predominate in the prodromal period. In addition, many people with schizophrenia and related disorders experience symptoms of anxiety without necessarily meeting full criteria for an anxiety disorder.
In the absence of specific guidelines for managing comorbid anxiety disorders in people with schizophrenia, clinicians usually follow general treatment guidelines for anxiety disorders. While this seems a reasonable approach, standard approaches to the treatment of anxiety disorders in non-psychotic people have not been evaluated for people with schizophrenia. Potential adverse effects include drug–drug interactions (e.g. fluvoxamine increasing serum levels of clozapine) and cumulative side effect burden (e.g. sexual side effects with SSRIs and antipsychotics).
Psychological strategies might also need to be modified (e.g. by linking inter-session homework for social anxiety in schizophrenia to case-manager-aided exposure response prevention exercises) (Halperin et al., 2000; Kingsep et al., 2003).
EBR: evidence-based recommendation.
Trauma
Exposure to adversity in childhood, including physical, sexual and emotional abuse, neglect, parental death and bullying, is associated with almost a three-fold risk of developing a psychotic illness (Varese et al., 2012). Unsurprisingly, people with schizophrenia have high rates of comorbid post-traumatic stress disorder as well as other comorbidities known to be associated with childhood adversity, such as depression, anxiety and substance use disorders (Achim et al., 2011). In addition, the experience of psychosis can also cause post-traumatic symptoms.
Clinicians often avoid asking people with schizophrenia about exposure to adversity (Lommen and Restifo, 2009; Read et al., 2005). Perceived lack of knowledge and training, concern about deterioration and poor engagement and lack of institutional support have all been identified as barriers preventing treatment of post-traumatic symptoms in people with psychosis (Gairns et al., 2015). As part of the assessment of a person with psychosis, clinicians should ask about exposure to adversity, as this has a bearing on the clinical presentation, illness course and treatment.
Further research is needed to determine the best treatments for post-traumatic symptoms in people with schizophrenia. Psychoeducation and cognitive and behavioural strategies to reduce anxiety may be helpful (Frueh et al., 2009; Mueser et al., 2008). Exposure therapy and other specific therapies to address post-traumatic stress disorder in people with schizophrenia require further research before they can be recommended as treatments for schizophrenia. As trauma exposure and its sequelae are so common in those with schizophrenia, this should be a research priority.


CBR: consensus-based recommendation.
N/A: Level of evidence category does not apply; recommendation based on a combination of available evidence, clinical experience and expert consensus.
Violence, suicide and victimisation
Suicide is a significant cause of premature mortality in schizophrenia, and 5–6% of people with a diagnosis of schizophrenia end their lives by suicide (Hor and Taylor, 2010; Palmer et al., 2005). The risk of suicide and self-harm appears to be greatest prior to initial treatment and in the period immediately after initial treatment (Challis et al., 2013), but continues throughout life. The main risk factors for suicide in schizophrenia are the same as those identified in people with other psychiatric disorders, and include male sex, previous suicide attempts, depressed mood and substance use (Hor and Taylor, 2010). Lopez-Morinigo et al. (2012) found that there is little evidence that insight is a risk factor for suicide in people with schizophrenia.
Although most people with schizophrenia will never commit an act of violence, people with schizophrenia are over-represented among perpetrators of violence, especially serious violence. For example, meta-analysis has shown that worldwide, 6.5% of homicide offenders have schizophrenia (Large et al., 2009) compared to the community prevalence of schizophrenia of 0.44% (McGrath et al., 2004). The most common reasons for violence are the presence of frightening symptoms, comorbid substance abuse (which has negative impacts on lifestyle and self-control) and the loss of continuity of care.
The routine use of risk-assessment instruments is likely to be of little value in predicting which person with schizophrenia will go on to commit serious violence, because of the low rate of serious violence, the lack of specific risk factors associated with acts of violence and the influence of an individual’s particular circumstances on the timing of acts of violence (Large et al., 2011b). However, some acts of violence might be prevented by the universal precautions of treating FEP earlier, assertive treatment of comorbid substance use and providing urgent assessment and treatment to people presenting with symptoms in which they believe they are in danger.
People with psychotic disorders are also more likely to be victims of violence. Morgan et al. (2015) found that compared to the population rate of 3.4% per year, people with psychosis are 4.8 times more likely to be the victim of a violent offence. Reasons for the increased rate of being assaulted include poverty and living in deprived areas with higher rates of violence, living in the open, the increased risk of assault in custodial settings and the effect of alarming behaviour associated with untreated illness (Maniglio, 2009). However, the key predictor of a person with schizophrenia being the victim of violence is comorbid substance abuse (Dolan et al., 2012).


EBR: evidence-based recommendation; CBR: consensus-based recommendation.
N/A: Level of evidence category does not apply; recommendation based on a combination of available evidence, clinical experience and expert consensus.
Research and evaluation
Clinical measurement
Much of the treatment of people with schizophrenia involves heuristic decision-making that is based on the clinical experience of the person providing the care, and is not informed by any objective measurement of the results of changes in treatment. This is of particular concern for consistency of treatment when doctors change regularly (e.g. for people with long-term schizophrenia treated at community facilities by trainee psychiatrists, who rotate every few months). Moreover, the lack of experience or any developed habit of clinical measurement affects trainee psychiatrists’ ability to critically evaluate research based on commonly used instruments. Medical records are full of risk-assessment instruments, but contain very little in the way of clinical measurement. Ideally, all doctors treating people with schizophrenia would routinely use scales for measurement of symptoms, including positive, negative and depressive symptoms, as well as side effects of treatment and cognitive and social performance. Widely used scales that might improve the objective measurement of symptoms and side effects include the Brief Psychiatric Rating Scale (BPRS) (Overall and Gorham, 1962) and the Abnormal Involuntary Movement Scale (AIMS) (Guy, 1976) for side effects. Trainee psychiatrists should be familiar with the items in the Positive and Negative Symptom Scale (PANSS) (Kay et al., 1987) which forms the basis of assessing the efficacy of antipsychotic medication. Depression can be assessed using the Calgary Depression Scale for Schizophrenia (CDS) (Addington et al., 1992), the Montgomery–Asberg Depression Rating Scale (MADRS) (Montgomery and Asberg, 1979) or the Beck Depression Inventory (Beck et al., 1961).

CBR: consensus-based recommendation.
N/A: Level of evidence category does not apply; recommendation based on a combination of available evidence, clinical experience and expert consensus.
Cognition is not a unitary concept and there is no universal cognition scale that would be reliable and valid in any routine clinical assessment. The tool that is close to a practicable clinical assessment of cognitive functions is the Repeatable Battery for the Assessment of Neuropsychological Status (RBANS; Randolph et al., 1998). The use of the RBANS by non-psychologists requires a brief supervised training that can be provided by a neuropsychologist. Another option is the Brief Cognitive Assessment Tool for Schizophrenia (B-CATS; Hurford et al., 2011) which has not been widely validated but is made up of three tests selected from more comprehensive neuropsychological batteries. The Brief Cognitive Assessment (BCA; Velligan et al., 2004) is a brief battery designed to measure changes in cognition. See also ‘Cognitive function’ in ‘Section 3. Treatment: context, structure and content of interventions’.
There are numerous measures of social function used in research, but most are too long to be practical for clinical use. The Daily Activity Report (DAR) takes a new approach, measuring functional outcome by recording a person’s daily activity for 7 days, based on phone calls made three times a day (Velligan et al., 2015).
There is also a need for services to record information that allows the comparison of performance of services and the performance of a service over time, including measures of duration of untreated psychosis, duration from first contact to treatment, rates of seclusion, absconding, use of involuntary treatment, continuity of care, duration to first relapse, reliability of clinical communication and particularly measures of the individual’s participation in their treatment planning and their satisfaction with the care that they are receiving.
Footnotes
Appendix
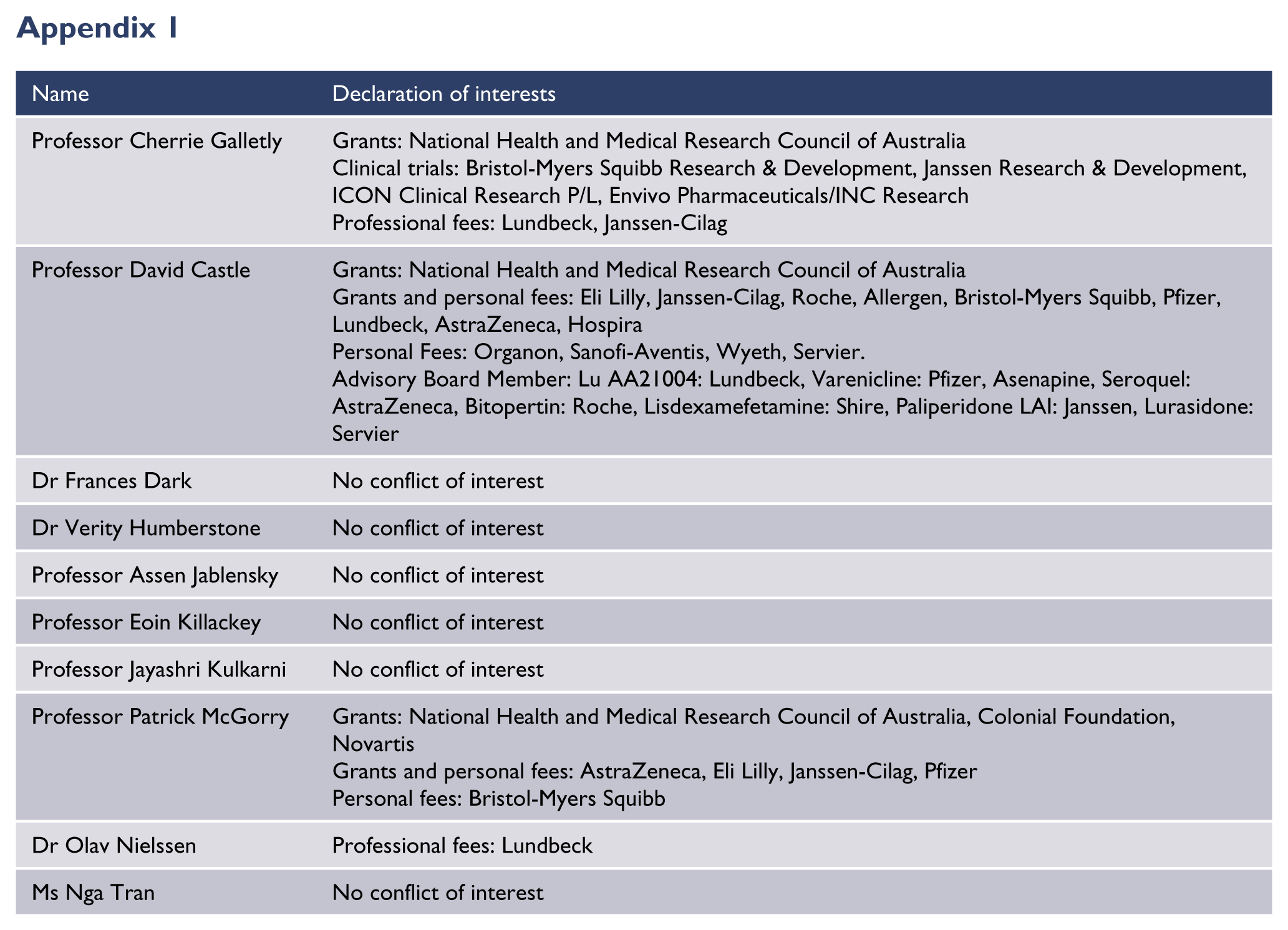
| Name | Declaration of interests |
|---|---|
| Professor Cherrie Galletly | Grants: National Health and Medical Research Council of Australia Clinical trials: Bristol-Myers Squibb Research & Development, Janssen Research & Development, ICON Clinical Research P/L, Envivo Pharmaceuticals/INC Research Professional fees: Lundbeck, Janssen-Cilag |
| Professor David Castle | Grants: National Health and Medical Research Council of Australia Grants and personal fees: Eli Lilly, Janssen-Cilag, Roche, Allergen, Bristol-Myers Squibb, Pfizer, Lundbeck, AstraZeneca, Hospira Personal Fees: Organon, Sanofi-Aventis, Wyeth, Servier. Advisory Board Member: Lu AA21004: Lundbeck, Varenicline: Pfizer, Asenapine, Seroquel: AstraZeneca, Bitopertin: Roche, Lisdexamefetamine: Shire, Paliperidone LAI: Janssen, Lurasidone: Servier |
| Dr Frances Dark | No conflict of interest |
| Dr Verity Humberstone | No conflict of interest |
| Professor Assen Jablensky | No conflict of interest |
| Professor Eoin Killackey | No conflict of interest |
| Professor Jayashri Kulkarni | No conflict of interest |
| Professor Patrick McGorry | Grants: National Health and Medical Research Council of Australia, Colonial Foundation, Novartis Grants and personal fees: AstraZeneca, Eli Lilly, Janssen-Cilag, Pfizer Personal fees: Bristol-Myers Squibb |
| Dr Olav Nielssen | Professional fees: Lundbeck |
| Ms Nga Tran | No conflict of interest |
Acknowledgements
The following people and organisations assisted with the writing of the RANZCP Clinical Practice Guideline for schizophrenia and related disorders: Expert contributors – Dr Bruce Gynther, Prof. Colleen Loo, Dr Duncan McKellar, Dr Hannah Newell, Dr Michael Paton, Prof. Nicholas Procter, A/Prof. James Scott and Dr Brett Simpson. Expert advisors – A/Prof. David Ash, Ms Joanne Baxter, Prof. Stanley Catts, Dr Jackie Curtis, A/Prof. Anthony Harris, Dr Harry Hustig, Prof. Dan Lubman, Dr Mark Taylor, Dr Neil Thomas, Dr Kipling Walker and Dr Douglas Wilson.
Respondents to public consultation:
Community Collaboration Committee
Aboriginal and Torres Strait Islander Mental Health Committee
Te Kaunihera
Te Pou o Te Whakaaro Nui (NGO, New Zealand)
SANE Australia
The New Zealand Psychological Society
Australian Psychological Society
RANZCP Fellows and trainees
Special acknowledgements:
Jenni Harman, Medical Writer, Meducation
RANZCP project team:
Ms Rosie Forster, Senior Department Manager, Practice, Policy and Partnerships; Dr Huseyin Mustafa, Project Manager, Policy Operations and Committees
Disclaimer
Compiled for the Royal Australian and New Zealand College of Psychiatrists (RANZCP), this information and advice is based on current medical knowledge and practice as at the date of publication. This clinical practice guideline is intended as a general guide only, not as a substitute for individual medical advice. The RANZCP and its employees accept no responsibility for any consequences arising from relying upon the information contained in this publication.
Declaration of Conflicting Interests
Funding
The development of this guideline was supported and funded by the RANZCP. The RANZCP acknowledges the substantial pro-bono input of RANZCP Fellows and other expert contributors in the development of this guideline. The RANZCP thanks those who have given of their time, experience and expertise.
