Abstract
Objective:
The prevalence of treatment-resistant schizophrenia (TRS) among people with first-episode schizophrenia (FES) has been sub-optimally researched in Australia and internationally. We evaluated the prevalence of TRS among a cohort of FES patients and compared their sociodemographic and clinical characteristics to those with FES who were treatment responsive.
Methods:
Over 2 years, we collated demographic, clinical and treatment-related data of all patients with ICD-10 (International Classification of Diseases, Tenth revision) diagnosis of schizophrenia who were active in October 2020 at four early psychosis intervention services (EPIS) in Western Australia. We used a modified version of Suzuki et al. criteria to diagnose TRS. The data were analysed utilising descriptive statistics, the Mann–Whitney U test, Student’s t-test and the False-Discovery Rate method.
Results:
The prevalence of TRS among the 167 patients diagnosed with FES was 41.3%, and the rates did not differ significantly between the services (p = 0.955). Those in the TRS group were less independent (p = 0.011), had more prolonged unemployment (p = 0.014) and were more likely to be on disability pension (p = 0.011) compared to the treatment responsive group. Furthermore, they had greater severity of symptoms (p = 0.002), longer duration of psychiatric symptoms (p = 0.019), more hospitalisations (p = 0.002) and longer cumulative admission durations (p = 0.002).
Conclusions:
Our study revealed that treatment resistance to antipsychotics is prevalent among people with FES managed at EPIS. Notably, it establishes an association between TRS and heightened clinical severity and psychosocial and treatment burden. These findings highlight the imperative for early detection of treatment resistance and timely and specialised interventions for this condition in mental health services.
Keywords
Introduction
Treatment-resistant schizophrenia (TRS) is both a clinical diagnosis and research descriptive and has been varyingly defined (Howes et al., 2017; Suzuki et al., 2012). Of late, attempts have been made to define the concept of TRS more precisely, incorporating parameters such as a validated diagnosis of schizophrenia and persistence of significant symptoms despite adequate pharmacological treatment (Howes et al., 2017). A recent meta-analysis reported a pooled prevalence of TRS among people with schizophrenia at various stages of their illness (mean illness duration 16.2 years) of 36.7%, with a range between 15.4% and 75.6% (Diniz et al., 2023). TRS is associated with a lower quality of life, higher unemployment rates, more significant impairment in achieving living milestones and greater reliance on supported accommodation in comparison to treatment responsive patients with schizophrenia (Demjaha et al., 2017; Iasevoli et al., 2016; Lally et al., 2016). From a clinical perspective, TRS is associated with an increased number of hospitalisations, more days spent in hospital, more severe positive and negative symptoms, worse cognitive function, more comorbidities, higher prevalence of substance abuse, poorer prognosis and significantly higher healthcare costs compared to treatment responders (Correll et al., 2019; Kane et al., 2019; Kennedy et al., 2014; Millgate et al., 2022).
Many clinicians conceptualise TRS as associated with chronicity and diagnosing TRS, particularly among those within the first episode of schizophrenia (FES), can often be missed or delayed (Demjaha et al., 2017; Lally et al., 2016; Wold et al., 2023). The prevalence of TRS among FES cohorts has been sub-optimally researched. A recent meta-analysis highlighted the research gap regarding the prevalence of TRS among FES patients, estimating the prevalence rate of 24.4% (Siskind et al., 2022). However, the generalisability of this finding is constrained by several factors. The meta-analysis identified a limited number of studies examining TRS prevalence in FES cohorts, revealing considerable heterogeneity among them with rates ranging from 8.97% (Kahn et al., 2018) to 44.6% (Yoshimura et al., 2019). Many studies also included individuals with other psychotic disorders, alongside FES patients (Demjaha et al., 2017; Doyle et al., 2017; Kahn et al., 2018; Lally et al., 2016; Malla et al., 2006; Smart et al., 2019; Yoshimura et al., 2019). Some of the included studies were short-term algorithm-based intervention research evaluating the efficacy of medications (Agid et al., 2011; Kahn et al., 2018; Yoshimura et al., 2019) or used proxy markers for TRS, such as the prescription of clozapine (Doyle et al., 2017; Wimberley et al., 2016).
Approximately 70–80% of patients diagnosed with TRS among those experiencing FES, have treatment resistance from the initiation of antipsychotic treatment. Conversely, an additional 20–30% develop resistance after 6 months following an initial period of good response to treatment (Demjaha et al., 2017; Lally et al., 2016). This latter group is likely to be missed in short-term studies on the prevalence of TRS. Similarly, while clozapine is considered the gold standard for the treatment of people with TRS (Wagner et al., 2021), its initiation is often delayed for many years (John et al., 2018), and it is under-prescribed (Bachmann et al., 2017). Thus, identifying TRS based on clozapine prescription could underestimate its rate. Moreover, very few studies have compared TRS patients’ clinical and sociodemographic characteristics to treatment responders in early schizophrenia (Crespo-Facorro et al., 2021; Nucifora et al., 2019). In addition, to our knowledge, the prevalence of TRS in FES has not been well-researched in Australian settings. Thien et al. (2018) conducted a study on 544 young people with first-episode psychosis at an early psychosis programme in Melbourne and reported that 65 met the criteria for TRS. However, only 13.1% of their sample had a diagnosis of schizophrenia, and the rest attracted other diagnoses. We evaluated the prevalence of treatment resistance in individuals with FES receiving clinical services at Early Psychosis Intervention Services (EPIS). In addition, we compared the sociodemographic and clinical characteristics of those with treatment-resistant and treatment-responsive FES.
Method
Ethics approval
This study was part of a larger study titled the clinical and economic benefits of early use of clozapine in first-episode schizophrenia. The Western Australian Health Human Research Ethics Committee and the governance committees of the sites involved approved the study. This component of the study was done under a waiver of consent approved by the ethics committee.
Setting
This study included participants from four EPIS outpatient programmes in metropolitan Western Australia, comprising a non-governmental organisation (headspace Early Psychosis Program – hEP), two public EPIS from the Southern Metropolitan Health Service (SMHS) and one from the East Metropolitan Health Service (EMHS). These services provided intensive clinical services to patients in the early stages of psychosis, and patients who were accepted to these services had a psychotic illness of less than 5 years duration. The services were clinically led by consultant psychiatrists, and the multidisciplinary clinical staff provided clinical and care coordination services to the patients. As part of the EPIS clinical protocol, the care coordinators made frequent clinical contacts (on average, weekly) with patients and their families, both within the clinic setting and in the community. The psychiatrists or psychiatry registrars of the team reviewed the patients’ mental state, medications and progress, on average, once a month. Detailed notes of the reviews were recorded in the patients’ medical records.
Participant selection
Lists of all patients who were active with the four EPIS in October 2020 were obtained. From these lists, we selected all patients diagnosed with schizophrenia according to the International Classification of Diseases 10th Revision. Diagnoses were made through a comprehensive review of medical notes and consultation with the team’s consultant psychiatrist.
Instruments, measures and sources of information
Before the study, the recruited research officers, who were experienced mental health clinicians, were trained by a senior psychiatrist (A.P.J.) in gathering relevant clinical and demographic data from the patients’ medical records. These trained research officers collected existing demographic, clinical and treatment-related data of the patients with a diagnosis of schizophrenia from these patients’ medical records. The research officers collected the data from October 2020 through August 2022. Data available until to the point of review of the individual patients’ medical records were collected. Collected sociodemographic details included age, gender, years of formal education, marital status, income and accommodation. Clinical details such as the primary diagnosis, comorbidities, substance abuse, age of first psychiatric symptoms, age of onset of psychosis, number of psychiatric admissions, total days in hospital for psychiatric illness, nature, dose and duration of antipsychotic treatments. The duration of psychiatric symptoms was calculated as documented age of first psychiatric symptoms subtracted from age at time of study. Similarly, the duration of psychosis was calculated as documented age of first psychotic symptoms subtracted from age at time of study. The response of the patients to each antipsychotic trial and the reasons for discontinuation of antipsychotics were obtained. The research officers also completed the Clinical Global Impression – Schizophrenia (CGI-SCH; Haro et al., 2003) scale for each patient, relying on clinical information extracted from the patients’ medical records of symptom profile and severity.
Defining treatment resistance
The research officers used a modified version of the Suzuki et al. (2012) criteria to identify TRS. They categorised patients into the TRS sub-category after reviewing all data and consulting with two senior consultant psychiatrists (A.P.J. and G.S.). Criteria included a confirmed diagnosis of schizophrenia, at least two past antipsychotic trials of 6 weeks each with suboptimal symptom response, a current CGI-SCH positive symptom score of 4 or more and functional impairment. Recent assessments of patients’ support level, accommodation, employment status, income status and social support were considered in determining functional impairment. We did not use a rating scale such as the Functional Assessment for Comprehensive Treatment of Schizophrenia (FACT-Sz) or the Global Assessment of Functioning (GAF), nor did we employ a cut-off score for functional impairment, as suggested by Suzuki et al. (2012). Medical records were carefully reviewed regarding information on non-adherence. If the persistence of positive symptoms was considered as due to non-adherence with antipsychotics, those patients were not rated as meeting the criteria of TRS of failed antipsychotic treatment.
In Western Australia, clozapine is indicated for TRS or for patients with schizophrenia who experience intolerable side effects from at least two other antipsychotics (Guidelines for the Safe and Quality Use of Clozapine Therapy in the WA health; www.health.wa.gov.au/guidelines). Consequently, all patients treated with clozapine were considered to have TRS, irrespective of their current CGI-SCH score or functional disability, unless documentation indicated that clozapine was prescribed for reasons other than TRS. In cases of uncertainty regarding TRS status, clarification was sought from the consultant psychiatrist associated with the respective treating team.
Statistical analysis
Data were analysed using the open-source statistical package Jamovi version 2.2.5. Descriptive statistics were used. For comparisons between the groups, the Mann–Whitney U test was applied for non-parametric continuous values, whereas the Student’s t-test was applied for continuous variables. Non-normal distributions are described using median and interquartile ranges (IQRs), while normal distributions are described using mean and standard deviation (SD). The chi-square test was applied to categorical values. The false-discovery rate method was utilised to adjust p-values, given the necessity for multiple comparisons within the same dataset (Menyhart et al., 2021). The threshold level of significance was set at α = 0.05.
Results
Composition of the study groups
A total of 394 patients were part of the EPIS services when the lists were obtained. Out of these, 167 had a diagnosis of schizophrenia, with 48% (80) originating from the hEP programme, 38% (64) from SMHS and 14% (23) from EMHS. These patients were considered as having FES. Almost three-quarters of the cohort with FES were male; their mean age was 24 years (SD 4.44 years; see Table 1). Most participants were single (81.44%) and resided with family or friends (76.05%) and half (50.3%) were classified as requiring support for daily living. Around 60% were unemployed and nearly half of the sample received regular government financial support. The cohort had an average education level of 12 years. Regarding clinical characteristics, the mean duration of psychosis was over 3 years (SD 1.88 years), and the mean age of onset of psychosis was 20.8 years (SD 4.55 years). About 35.3% were actively engaged in substance abuse, 65.9% had a concurrent psychiatric condition, and 25.7% had a comorbid medical condition.
Sociodemographic and clinical characteristics of the study sample (n = 167).
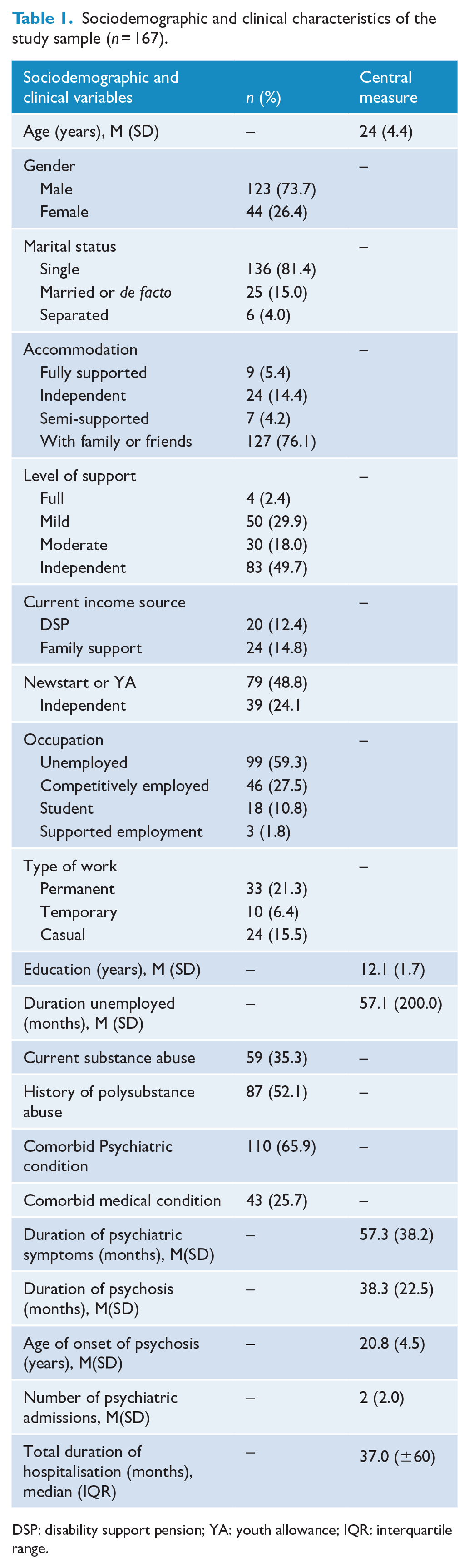
DSP: disability support pension; YA: youth allowance; IQR: interquartile range.
Prevalence of TRS
The overall prevalence rate of TRS was 41.3% among this cohort, with an insignificant variation across the service providers (hEP 42.5%, EMHS 43.5%, SMHS 39.1%, χ2 = 0.328, p = 0.955).
Sociodemographic differences between TRS and non-TRS patients
A significantly higher proportion of the TRS patients were recipients of the disability support pension (20.29%, p = 0.011) than the non-TRS patients (6.12%; see Table 2). Moreover, the TRS patients were deemed as significantly less independent in terms of the daily level of support required than the non-TRS cohort (p 0.011). Although a larger percentage of TRS patients were male, single and unemployed, these differences did not achieve statistical significance (p = 0.062, p = 0.055 and p = 0.068, respectively). Nevertheless, their unemployment duration was significantly longer by approximately 1.5 years compared to the non-TRS group (p = 0.014). The age, education level and type of accommodation were the same between the groups.
Sociodemographic characteristics of the TRS group and non-TRS group.
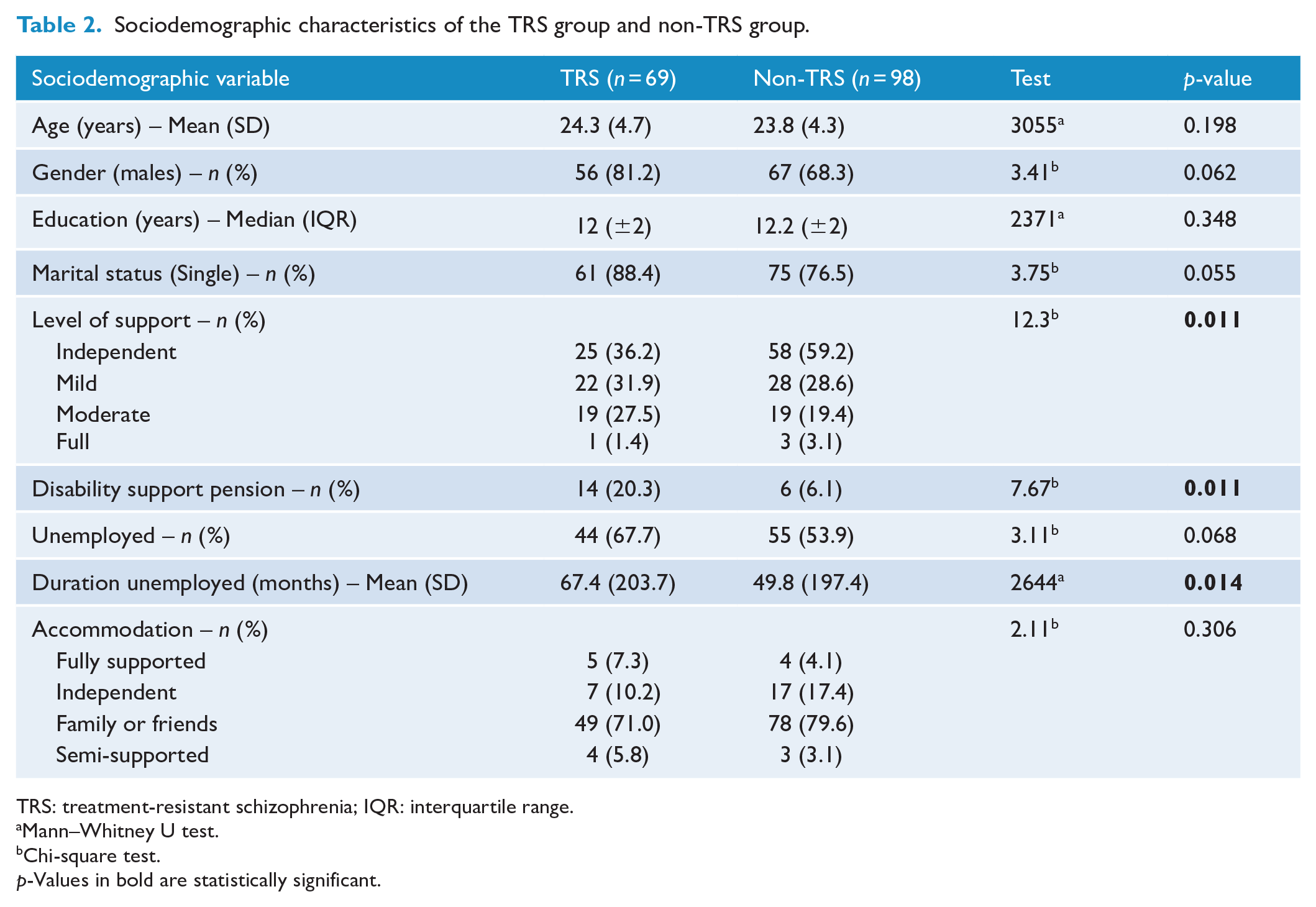
TRS: treatment-resistant schizophrenia; IQR: interquartile range.
Mann–Whitney U test.
Chi-square test.p-Values in bold are statistically significant.
Clinical differences between TRS and non-TRS patients
In the analysis of clinical factors (see Table 3), TRS patients endured psychiatric symptoms for an additional 12 months (p = 0.019) compared to the treatment responsive group. They also scored significantly higher across the positive, negative and cognitive subdomain and CGI total scores than the non-TRS patients (p = 0.002). Furthermore, the TRS patients had significantly more hospitalisations for psychiatric illness than non-TRS patients, with approximately 3.04 admissions (SD = 2.21) compared with 1.76 (SD = 1.41), respectively (p = 0.002). In addition, TRS patients had undergone significantly longer cumulative duration of psychiatric admissions, with a median of 52 months (IQR ±118) compared with 26 months (±44) for non-TRS patients (p = 0.002). The TRS group underwent significantly more antipsychotic trials, with a median of 4 (IQR ±2), compared to a median of 3 (IQR ±2) for non-TRS patients (p = 0.002). However, there were no differences observed in the age of onset of psychosis and psychiatric symptoms, as well as in the levels of comorbid psychiatric and general medical illnesses between the groups. In addition, the groups showed similarities in terms of substance abuse history and usage patterns.
Clinical characteristics of the TRS Group and non-TRS Group.
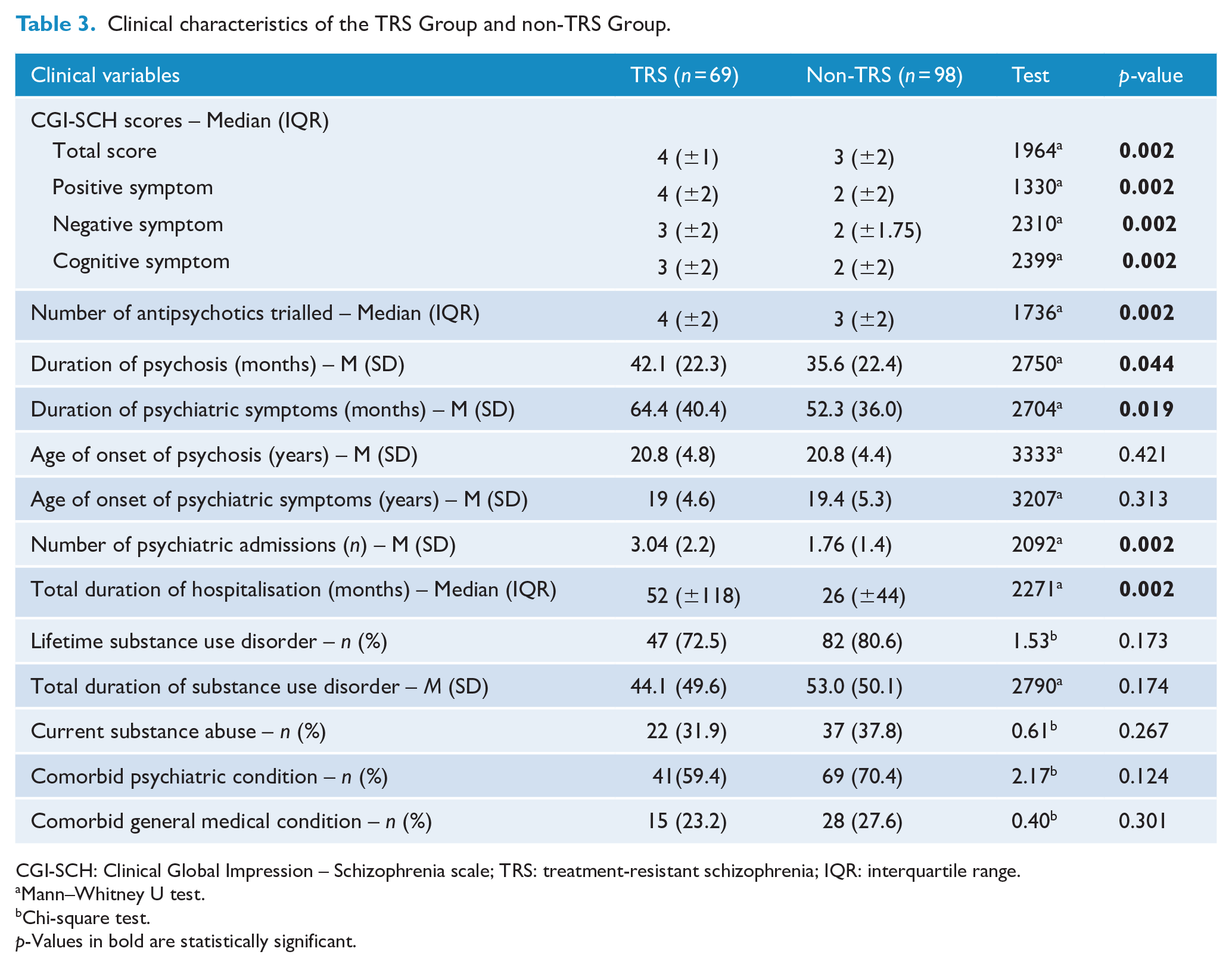
CGI-SCH: Clinical Global Impression – Schizophrenia scale; TRS: treatment-resistant schizophrenia; IQR: interquartile range.
Mann–Whitney U test.
Chi-square test.
p-Values in bold are statistically significant.
Discussion
Prevalence of TRS at early intervention services
Our study revealed a prevalence of TRS of 41.32% among a cohort of FES patients in Western Australia’s EPIS. The prevalence of TRS in our sample is higher than the figures of 22% and 24.4% reported in the metanalyses of TRS among FES (Diniz et al., 2023; Siskind et al., 2022) but close to the rate of 37.8% reported among 239 FES patients followed up for approximately 5 years after first presentation to psychiatric services (Ajnakina et al., 2020). However, other researchers have reported lower prevalence rates of 15–23% (Demjaha et al., 2017; Lally et al., 2016; Smart et al., 2019; Stokes et al., 2020) during 5 to 15 years of follow-up of patients with first-episode psychosis/schizophrenia. The authors of the meta-analysis on FES observed that there were only nine studies that evaluated the prevalence of TRS in FES, and the rates ranged from 8.97% (Kahn et al., 2018) to 44.6% (Yoshimura et al., 2019) with significant methodological differences between the studies. The length of follow-up periods among these studies varied between 2 and 120 months. A significant proportion of patients with FES, reported to be between 20% and 30% of those who have TRS, develop it at a later stage of illness, in the first 5 years following an initial period of treatment response (Demjaha et al., 2017; Lally et al., 2016). A considerable number of the previous studies had a short follow-up period (Agid et al., 2011; Kahn et al., 2018; Lieberman et al., 1993; Malla et al., 2006; Smart et al., 2019) and could have missed the patients with FES who later developed TRS. In our study, the TRS group exhibited a mean duration of psychosis over 42 months (SD 22.3 months), and it is possible that a subset of our TRS patients developed treatment resistance over the preceding period of psychosis, contributing to the overall higher TRS prevalence rate in our study. Furthermore, among the previous studies on TRS prevalence in FES, lost to follow-up ranged from 15% to 68% (Agid et al., 2011; Lieberman et al., 1993), while our data collection included all individuals originally included in the cohort. The low prevalence rate of TRS reported by some of the studies with high dropouts is likely to be an underestimate. It is feasible that those with TRS were more likely to have dropped out from follow-up, thereby artificially decreasing the rate of TRS (Siskind et al., 2022). Some authors have only included patients treated with clozapine in their definition of TRS (Doyle et al., 2017; Lally et al., 2016; Wimberley et al., 2016). However, clozapine is underutilised, and its commencement is often delayed for many years (John et al., 2018; Muñoz-Manchado et al., 2023), and thus it is quite likely that these studies have underreported the true prevalence of TRS. Furthermore, many studies on the prevalence of TRS in first-episode psychosis have included diagnoses other than schizophrenia, such as psychosis not otherwise specified (Demjaha et al., 2017; Doyle et al., 2017; Kahn et al., 2018; Lally et al., 2016; Malla et al., 2006; Smart et al., 2019; Thien et al., 2018; Yoshimura et al., 2019) and affective and substance-induced psychosis (Thien et al., 2018). The nature of the cohort, the setting from where they are drawn, the study design and criteria used to define TRS can all affect the prevalence rate. While the TRIPP criteria to define TRS has been broadly accepted, some authors have suggested that the prevalence of TRS as identified by the TRRIP or similar criteria in the early course of illness is likely to be an underestimate and should be interpreted with caution, as a large proportion of patients with primary antipsychotic resistance could remain unidentified (Dempster et al., 2021; Wold et al., 2023).
The prevalence rate of TRS in our study was similar across the services included. The specialised EPIS included in our study are likely to provide clinical services to individuals with more severe FES. In contrast, treatment-responsive FES patients may receive care through primary or other secondary services elsewhere, which were not captured in our study. While we carefully ascertained treatment responses to antipsychotic trials through a comprehensive review of the medical and multidisciplinary staff’s clinical notes in the medical records, it is possible that the treating teams would have missed some cases of non-adherence with antipsychotics or the psychotic symptoms were directly caused by substance abuse and not entered as such in the medical records. These patients could have been wrongly classified as treatment non-responders to antipsychotics and falsely classified as having TRS, inflating the prevalence of TRS in this cohort. Non-adherence with antipsychotics and substance abuse is very common in people with schizophrenia (Cahaya et al., 2022; Kennedy et al., 2014), and a significant proportion of those referred to specialised services for evaluation of TRS were found to be non-adherent (Howes et al., 2017). The TRRIP Working Group recommended determining treatment adherence through several sources, including serum levels, which may reduce false-positive treatment resistance by mitigating treatment nonresponse secondary to non-adherence (Howes et al., 2017).
TRS patients have more severe psychopathology, psychosocial burden and treatment needs compared with non-TRS patients
Our study revealed that patients with TRS had a higher global CGI score and scored higher in all the CGI sub-domains of positive, negative and cognitive symptoms than their non-TRS counterparts. These findings are in keeping with the existing literature that demonstrates the persistence of psychotic, negative and cognitive symptoms in TRS patients (Howes et al., 2017; Iasevoli et al., 2016; Millgate et al., 2022), contributing to more significant social and functional limitations (Correll et al., 2019; Kennedy et al., 2014). We also found that TRS patients were receiving the disability support pension at more significant proportions and had been unemployed for a longer mean duration of months. TRS patients also required higher levels of daily support than non-TRS patients. These findings are in keeping with earlier studies, which showed that individuals with TRS are more likely to experience a decrease in quality of life, increased medical costs and have lower employment rates (Iasevoli et al., 2016; Kane et al., 2019), a trend that was also observed in early schizophrenia (Crespo-Facorro et al., 2021). The greater clinical severity and functional impairments and nonresponse to traditional antipsychotics from illness onset have prompted some to postulate that TRS is a categorically distinct type of schizophrenia with changes in receptors such as glutamate, serotonin and gamma-aminobutyric acid (GABA) rather than dopamine, neuroimaging findings, greater genetic loading and neurocognitive impairments (Demjaha et al., 2012; Farooq et al., 2013; Mouchlianitis et al., 2016; Suzuki et al., 2015). However, the neurobiological mechanisms that lead to the development of treatment resistance are not distinctly demarcated and understood. An alternate theory posits that TRS exists at the more severe end of the continuum with treatment-responsive schizophrenia and non-affected first-degree relatives of patients with schizophrenia (Frank et al., 2015; Millgate et al., 2022). Others have suggested that dopamine supersensitivity due to long-term D2 receptor blockade by antipsychotics (Silvestri et al., 2000) or cumulative lifetime adversities and trauma (Hassan and De Luca, 2015) have a role in the genesis of TRS.
Our study revealed that TRS patients had a more significant total number of psychiatric admissions and had lengthier admissions compared with non-TRS patients, which implies higher direct costs to health systems. This is supported by a recent study investigating the economic implications of early treatment resistance in schizophrenia, which found that individuals who exhibited early nonresponse incurred more than double the healthcare costs compared to early responders (Ascher-Svanum et al., 2008). In addition, researchers who have reviewed the literature in this area suggested that the total health costs for TRS patients were approximately tenfold higher than those for non-TRS patients, with the major contributor being increased days spent in hospital by patients with TRS (Kennedy et al., 2014; Peng et al., 2011). Other research has highlighted that individuals in the early stages of schizophrenia exhibit a higher rate of psychiatric hospitalisation and more prolonged hospital admissions, resulting in increased direct medical costs (Nicholl et al., 2010). Moreover, healthcare expenditures have been shown to rise in tandem with the severity of the disease (Millier et al., 2012). Consequently, early schizophrenia patients who are resistant to treatment are likely to incur substantially higher healthcare costs compared to treatment responders and those with chronic TRS.
We observed that the proportion of males and those who were single were higher among the patients with TRS, but the figures narrowly failed to reach statistical significance. The role of gender and marital status in predicting TRS is still unsettled (Bozzatello et al., 2019), and a meta-analysis did not observe an increase in males among the TRS group (Diniz et al., 2023). We did not find significant differences between earlier onset of psychosis or psychiatric illness or level of education between the two groups. These findings are not in concordance with some of the recent research findings that younger age of onset and lower educational level are robust predictors of those likely to go on to develop TRS among people with FES (Bozzatello et al., 2019; Smart et al., 2022).
EPIS offers an ideal environment for the early diagnosis of treatment-resistant FES. Although this study did not examine treatments, early identification of TRS among people with FES within these settings may facilitate the timely initiation of appropriate interventions. Unfortunately, pharmacological guidelines for treating FES often provide varied and less-specific recommendations, with the selection of antipsychotics typically based on side-effect concerns rather than comparative efficacy benefits (Keating et al., 2017). In a recent study involving a cohort of 207 patients with FES, it was noted that, after 12 months of treatment, only 35 out of the 97 individuals who met the criteria for no early clinical recovery had undergone sufficient trials with two antipsychotics to satisfy the TRIPP criteria (Wold et al., 2023). Persisting with treatment with ineffective antipsychotics or trying augmentation with additional psychotropic agents are not uncommon practices among clinicians in managing treatment nonresponse (Dempster et al., 2021; Wold et al., 2023). A structured evaluation of treatment response in the early phases to identify non-responding FES patients and providing active interventions, including changing ineffective treatments and commencing clozapine early, should become part of the usual clinical practice of EPIS (Bittner et al., 2023; Stokes et al., 2020; Thien et al., 2018).
Strengths and limitations of the study
This study has notable strengths, including selecting the cohort from diverse EPIS from various metropolitan regions in WA. The data, meticulously compiled by trained research officers under senior psychiatric oversight, enhances the study’s robustness. Another strength is the comprehensive follow-up of all initially included FES patients throughout the study, covering a significant duration from their initiation with the early intervention teams.
However, the study has certain limitations. All data, including that of the determination of TRS status and rating of CGI-SCH was completed based on documented clinical notes raising the possibility of documentation and interpretation bias. It is possible that a proportion of patients who were treatment responsive were discharged from the service relatively early, which could affect the findings of the study. Moreover, the exclusive focus on actively engaged EPIS patients might overlook individuals utilising alternative psychiatric or primary care services, thus potentially introducing selection bias. The majority of male participants could limit the generalisability of the findings to broader populations. Furthermore, our study did not measure functional impairment using a validated scale, unlike Suzuki’s criteria (Suzuki et al., 2012), which requires both moderate symptom severity and functional impairment evidenced by a score of ⩽ 49 on FACT-Sz or ⩽ 50 on GAF to determine treatment failure. This may introduce measurement bias. In addition, we did not verify treatment adherence through multiple sources, as recommended by the TRRIP Working Group (Howes et al., 2017), which could result in misclassification and an overestimation of treatment resistance rates among FES patients.
Conclusion
In summary, our study sheds light on the high prevalence of TRS among people with FES and the more severe clinical manifestations and profound psychosocial disabilities of this group compared to the non-TRS group. The identified high prevalence and severity of illness emphasise the significant proportion of individuals facing challenges in treatment response early in their psychotic illness. Longitudinal long-term prospective studies of FES would provide a clearer picture of the prevalence and time course of development of TRS in FES. The high prevalence of TRS and the significant clinical severity and functional impairment of TRS in FES point towards the need for EPIS and other mental health services to be vigilant in detecting TRS early and providing appropriate interventions promptly, especially earlier consideration and use of clozapine.
Footnotes
Acknowledgements
We thank the contributions of Dr Susanne Stanley, Ms Cherry Acabo, Dr Alexandra Jaworska and Dr Ajay Velayudhan in collecting / accessing data for this study.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The study was funded by a SHARC RTP grant from the Department of Health, Western Australia.
Ethics Statement
The Western Australian Department of Health Human Research and Ethics Committee approved this study.
Data Availability Statement
The data supporting this study’s findings are available upon reasonable request from the corresponding author once permission from the ethics committee to share data is obtained.
