Abstract
Objective:
While there is now strong evidence that psychological therapies can alter the activity of individual brain regions, their impact on the functional integration between regions has not yet been systematically evaluated. This area is important given that brain dysconnectivity has been implicated across almost all psychiatric disorders. Accordingly, we sought to establish connectivity predictors and mechanisms of effective psychological therapies. We further establish whether connectivity changes represent normalisation of disorder pathophysiology or compensatory changes.
Method:
We reviewed studies examining structural and functional connectivity longitudinally as either a predictor or outcome variable of successful psychological therapies across psychiatric disorders.
Results:
Fifteen studies met our inclusion criteria. All but three related to cognitive behavioural therapy. Of these, five assessed resting state, nine probed affective processing and one probed cognitive processing. Twelve studies reported evidence of functional connectivity as a significant predictor or outcome of cognitive behavioural therapy, with prefronto-limbic circuitry most commonly implicated. Only six studies included healthy participants, limiting direct inferences about normalisation as opposed to compensatory changes. Anxiety disorders were overrepresented, totalling 13 of the studies reviewed. No studies examined structural connectivity or utilised analyses allowing the directionality of functional connectivity to be inferred.
Conclusion:
While the evidence base is still in its infancy for other therapy approaches, there was clearer evidence that functional connectivity both predicts and is altered by cognitive behavioural therapy. Connections from prefrontal cortex appear especially key, perhaps given their role in cognitive appraisal of lower order affective, motivational and cognitive processes. A number of recommendations are made for this rapidly developing literature.
Introduction
Psychological interventions have been shown to be effective for psychiatric disorders, and there has been considerable progress in characterising their psychoneurobiological mechanisms. While the predictors and mechanisms of therapeutic response have been examined by several recent reviews, to date these have all focused on treatment-related changes in brain activity. As a prominent axiom would, however, suggest, in clinically applied cognitive neuroscience, the sum is surely greater than its parts. There is increasing recognition that the way in which information is integrated between regions is crucial in the genesis and maintenance of psychiatric disorders (Buckholtz and Meyer-Lindenberg, 2012). This review sought to examine the functional connectivity predictors and mechanisms of response to psychological interventions across disorders.
Functional integration and psychological therapy
Brain functions show a high degree of spatial specificity, and therefore, we rely relies on connectivity to bind together information from numerous domains, including sensory, affective and (social) cognitive information (‘functional integration’, Friston et al., 1997). Given this need to constantly maintain and reconfigure these long-range connections, it is unsurprising that clinical symptoms may result from lapses or failures of connectivity at different points in these networks (Buckholtz and Meyer-Lindenberg, 2012) and may respond to interventions.
At the psychological level, it has been argued that a common mechanism of change across psychological therapies is the awareness of conflict amongst beliefs or goals and increasing flexibility of strategies to reduce this conflict to also reduce distress (e.g. Higginson et al., 2011). Cognitive behavioural therapy (CBT; Beck, 1976), for example, fosters change in conflicting and inflexible appraisals of affective, physiological, social and cognitive states by overcoming experiential and behavioural avoidance. At the neural level, therapeutic change is likely to involve reorganisation of the functional networks supporting these multiple processes. Connections from prefrontal cortical regions may be particularly important, given their importance for higher order cognitive and metacognitive skills, including appraisals and control over affect (Wager et al., 2008) and behaviour (see, for example, Holroyd and Yeung, 2012). The subjective experience of threat and distress has been linked to ventral and limbic regions, particularly amygdalo-hippocampal regions and insula cortex (e.g. Mobbs et al., 2009). An influential model proposes that emotion regulation involves the recruitment of dorsal prefrontal cortical regions which modulate ventral and limbic regions through top-down connectivity (Buhle et al., 2014; Wager et al., 2008). Another model derived from meta-analytical evidence conceptualises the dorsal and ventral portions of anterior cingulate cortex as important in the experience and regulation of affect, respectively (Etkin et al., 2011). In their model, the dorsal portion shows positive coupling with amygdala during experience of affect, whereas the ventral portion shows negative coupling when inhibiting affect. Accordingly, CBT and other psychological therapies may strengthen the top-down aspects of these mechanisms (Clark and Beck, 2010). Although no reviews have evaluated CBT in relation to functional connectivity, systematic reviews of brain activation studies consistently identify post-therapy increases in prefrontal and cingulate cortex activations, including dorsolateral, medial and ventral regions (for reviews see Barsaglini et al., 2014; Frewen et al., 2008; Quidé et al., 2012). Reduced insula activation has also been identified as an outcome of psychological therapy across affective, anxiety and psychotic disorders (e.g. Barsaglini et al., 2014). In addition to its role in signalling threat, this region is linked to self-focus and interoception (Zaki et al., 2012), social information processing (Carr et al., 2003) as well as salience and attention (Menon and Uddin, 2010).
Overview of brain connectivity and its measurement
In this section, we will provide a brief overview of the different types of structural and functional connectivity and how they are quantified (for a more in-depth discussion, see, for example, O’Reilly et al., 2012). Whereas structural connectivity refers to anatomical connections between brain regions, functional connectivity refers to the degree of co-activation between them (Figure 1). Structural connectivity is most commonly ascertained using diffusion-tensor weighted magnetic resonance imaging (MRI). This method quantifies the diffusion of water molecules, which can be used to infer the density and orientation of white matter tracts (see Le Bihan et al., 1988).
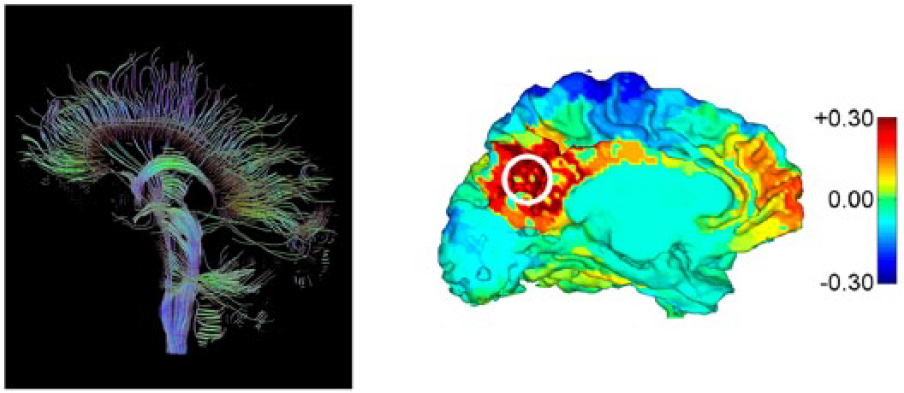
Illustration of structural (left) and functional (right) connectivity. Left: Whole-brain white matter tractography illustrates physical connections. Right: A map of correlations of the activity time courses of all voxels with that of a pre-defined seed region (circled). Note that the map is typically used to capture a snapshot of a specific time point or stimulus presentation and is not static over time.
While structural methods have proved invaluable for producing an atlas of the whole brain’s gross physical connections, they tell us little about how brain regions subgroup into functional networks (i.e. functional connectivity). Historically, studies have focused on functional connectivity supporting specific psychological processes by probing them experimentally (Figure 2). More recently, there has also been work examining the functional significance of the spontaneous fluctuations in neural activation that arise in the absence of specific task demands, which are highly synchronised (‘resting-state’ connectivity). Multiple ‘resting-state’ networks have been proposed, related to distinct aspects of consciousness and self-referential processing and as such are of clinical interest. For example, networks involved in attention, salience and executive control have been identified (see Raichle, 2011) and show alterations across numerous psychiatric disorders (e.g. Kühn and Gallinat, 2013).
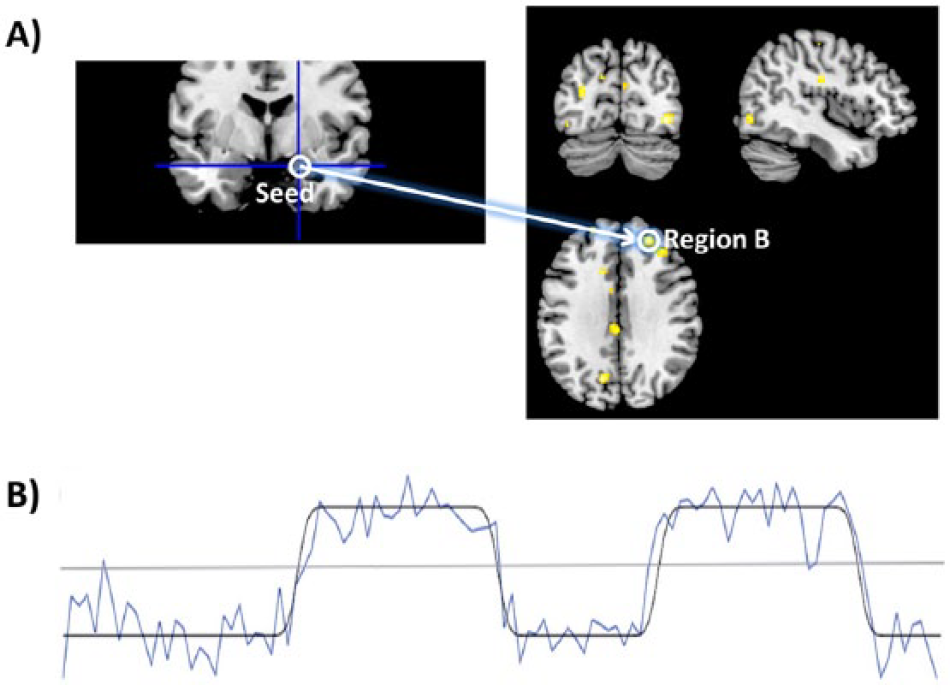
How functional connectivity is measured from a seed region. (A) After electing a probable seed region (based on theoretical or empirical grounds), correlations between its activity time course and that of all voxels across the whole brain are performed and thresholded for statistical significance. (B) Correlation time series between seed and a voxel in region B (blue) in relation to task condition (task regressor convolved with the haemodynamic respond function; black). Correlations above and below the grey line are positively and negatively correlated, respectively. We can conclude that the seed region and region B coactivate during task blocks, but that they are inversely correlated during rest blocks.
Task and resting-state connectivity can be inferred either by model-driven or data-driven methods. Model-driven methods require an a priori hypothesis about at least one region and frequently involve seed-based analyses, such as the psychophysiological interaction (Friston et al., 1997) approach, in which a map of functionally connected regions is inferred from a seed region. For task-related connectivity, this analysis approach includes information about task conditions to identify regions that modulate their connectivity coincident with specific psychological processes elicited experimentally (Figure 2; see O’Reilly et al., 2012). Where there is a task-specific increase in connectivity, this indicates that the regions are exchanging information to engage in that task. In contrast to these model-based approaches, data-driven methods such as independent component analysis (e.g. Svensén et al., 2002) blindly classify areas of covarying activation into different groups and so are well suited to exploratory analyses without well-formed hypotheses about implicated regions. More recently, analysis methods have been developed which allow the directionality of functional connections to be inferred (effective connectivity, Friston, 2011), and use of this approach is growing rapidly.
Methods
Eligibility criteria
We followed the criteria of another recently published systematic review of the neurobiological effects of psychological therapy (Barsaglini et al., 2014). Studies were included if they (1) included group-level analyses published in a peer-reviewed journal; (2) used structural or functional connectivity to predict response to, or evaluate changes following, psychological therapies; and (3) employed a within-subjects design in which the same patients were scanned before and, where applicable, after therapy. We included both therapist-led and self-help (e.g. Internet-delivered) forms of therapy. We excluded studies focusing on physical health (e.g. chronic pain) to reduce heterogeneity. We retrospectively restricted studies to CBT only, given the small number of studies evaluating other therapy approaches (n = 1 per therapy approach).
Information sources and search
Studies were primarily identified by searching electronic databases (PubMed and Web of Science). We also manually checked the reference lists of relevant articles and contacted the corresponding authors of studies included in a recent review of functional activation studies (Barsaglini et al., 2014). Where conference presentations or trial registrations were found, authors were contacted to check for reports of peer-reviewed journal articles. Studies were only included if they were available in English.
We combined the following three search criteria:
‘neuroimaging’ OR ‘tomography’ OR ‘magnetic resonance’ OR ‘FMRI’ OR ‘MRI’ OR ‘PET’ OR ‘EEG’ OR ‘MEG’ OR ‘electroencephalography’ OR ‘magnetoencephalography’.
‘connectivity’ OR ‘connectome’ OR ‘connections’ OR ‘coupling’ OR ‘granger’ OR ‘DCM’ OR ‘PPI’ OR ‘psychophysiological’.
‘CBT’ OR ‘psychotherapy’ OR ‘DBT’ OR ‘behavio* therapy’ OR ‘Eye movement desensiti*’ OR ‘EMDR’ OR ‘systematic desensiti*’ OR ‘remediation’ OR ‘behavio*ral activation’ OR ‘mindfulness’ OR ‘exposure therapy’ OR ‘commitment therapy’.
An asterisk after a term matched all terms that begin or end with that root. The search was run on 25 January 2015 with no time range specified for date of publication. An additional search was performed on 28 October 2015 to find new articles since this time.
Study selection
Eligibility assessment was performed separately by the first author and a second reviewer and compared. Study abstracts were screened using the eligibility criteria if the title referenced a psychological therapy and a neuroimaging technique or neurobiological mechanism. Any missing information was clarified with corresponding authors.
Data extraction
We extracted the following details from each study: disorder and treatment studied, design, inclusion criteria, sample size and demographics, neuroimaging modality, experimental task used (including resting state), connectivity measures and symptom measures (see Supplementary Materials for summary form used). Assessment of quality and risk of bias was guided by Cochrane Collaboration’s tool for assessing risk of bias in cohort studies (Lundh and Gøtzsche, 2008) and included sample size, random allocation to therapy, treatment response as well as, where appropriate, proportion of patients lost to follow-up and whether analyses followed the intention-to-treat principle.
Results
A total of 18 studies were identified (see Figure 3). The majority evaluated CBT, with very few for other therapy approaches (one cognitive remediation therapy, one mindfulness and one guided imagery therapy; Supplementary Table 1), and so these were discarded for this review. A total of 15 CBT studies were included (Table 1). Of these, all evaluated functional connectivity, with five examining predictors of therapy outcome, eight examining outcomes of therapy longitudinally and two examining both. There were no structural connectivity studies of CBT (one evaluated guided imagery therapy; Supplementary Table 1). The final studies identified were consistent across both reviewers.
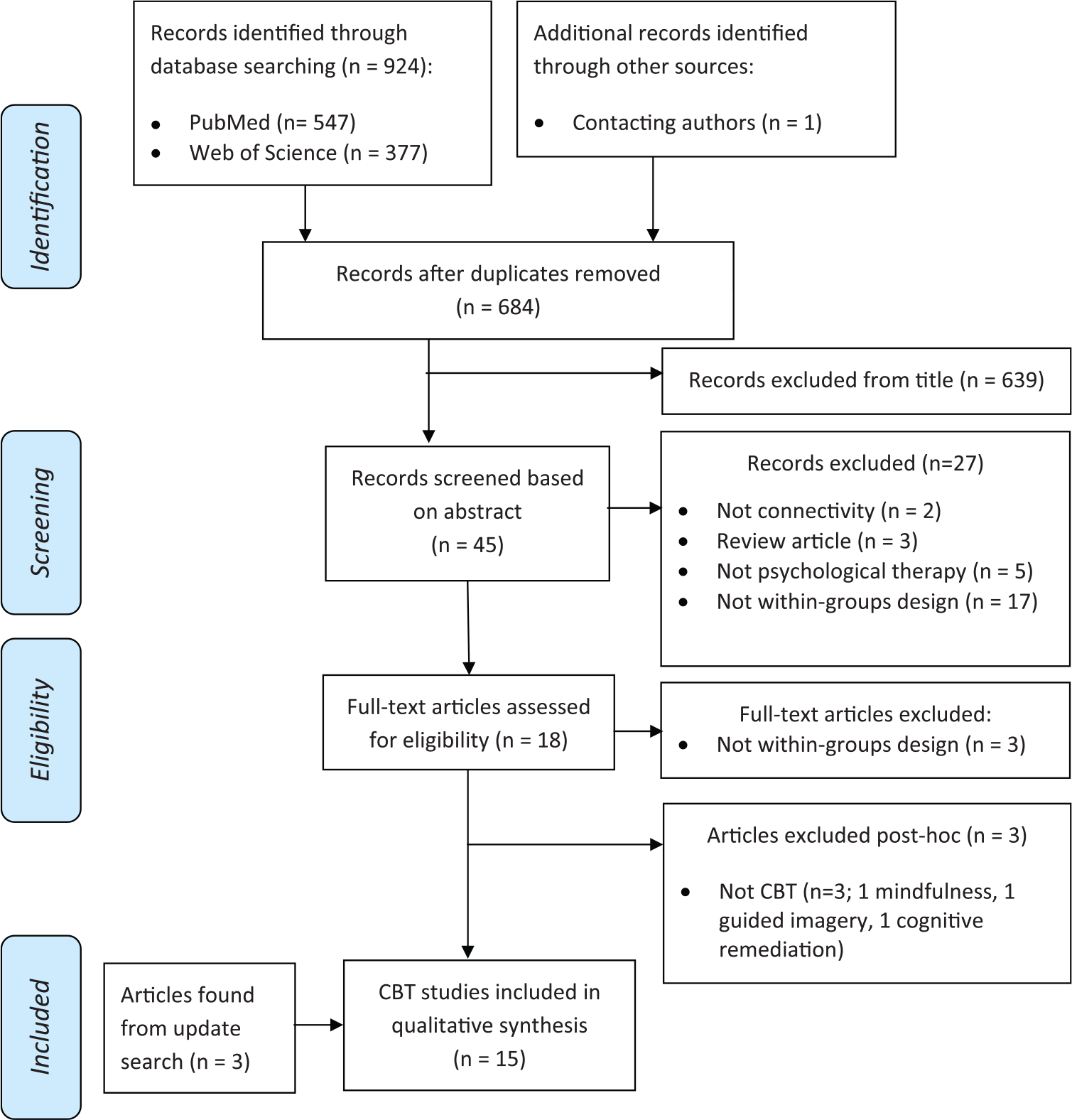
Flow chart depicting study inclusion.
Summary of studies examining neural connectivity as a predictor or target of CBT.
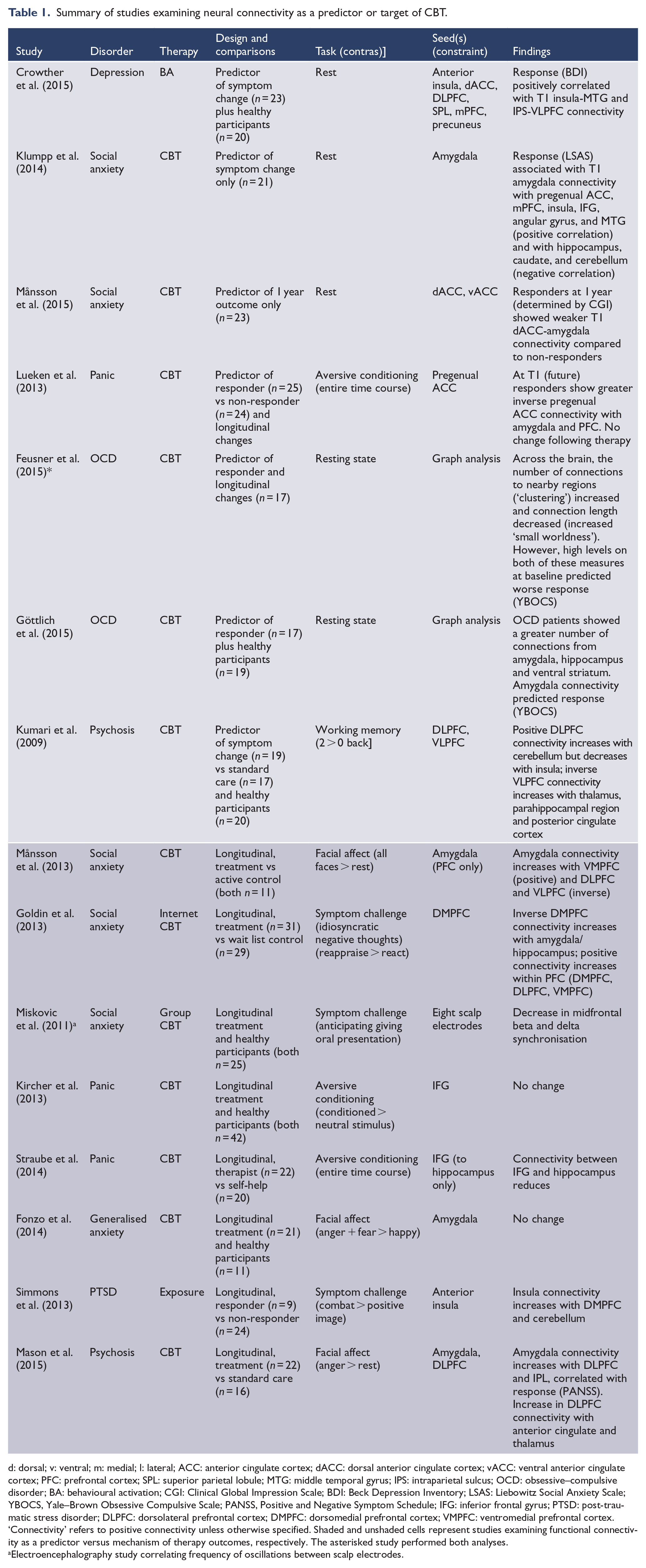
d: dorsal; v: ventral; m: medial; l: lateral; ACC: anterior cingulate cortex; dACC: dorsal anterior cingulate cortex; vACC: ventral anterior cingulate cortex; PFC: prefrontal cortex; SPL: superior parietal lobule; MTG: middle temporal gyrus; IPS: intraparietal sulcus; OCD: obsessive–compulsive disorder; BA: behavioural activation; CGI: Clinical Global Impression Scale; BDI: Beck Depression Inventory; LSAS: Liebowitz Social Anxiety Scale; YBOCS, Yale–Brown Obsessive Compulsive Scale; PANSS, Positive and Negative Symptom Schedule; IFG: inferior frontal gyrus; PTSD: post-traumatic stress disorder; DLPFC: dorsolateral prefrontal cortex; DMPFC: dorsomedial prefrontal cortex; VMPFC: ventromedial prefrontal cortex.
‘Connectivity’ refers to positive connectivity unless otherwise specified. Shaded and unshaded cells represent studies examining functional connectivity as a predictor versus mechanism of therapy outcomes, respectively. The asterisked study performed both analyses.
Electroencephalography study correlating frequency of oscillations between scalp electrodes.
Resting-state connectivity
Four studies examined whether resting-state connectivity could be used to predict response to CBT. Crowther et al. (2015) examined treatment predictors for behavioural activation therapy in 23 depressed patients (with all other comorbidities excluded). They examined connectivity strength from seeds in four canonical resting networks including the default mode (seeds were precuneus and dorsomedial prefrontal cortex [DMPFC]), salience (insula and dorsal anterior cingulate cortex [ACC]), dorsal attention (superior parietal lobule) and executive control (dorsolateral prefrontal cortex [DLPFC]) (for a description, see Raichle, 2011). Of nine connection pairs differentiating depressed from healthy participants, better outcome was predicted by milder deficits in the salience (insula seed connectivity with temporal cortex) and dorsal attention (superior parietal lobule connectivity with orbitofrontal cortex) networks. A limitation of this study is that by restricting analyses to the connections that differentiated depressed from healthy participants (i.e. the extent of pathophysiology present), they precluded the discovery of other treatment predictors (i.e. compensatory or protective connectivity). Klumpp et al. (2014) found that positive connectivity from amygdala predicted response to CBT for social anxiety disorder. They found that social anxiety symptom improvement positively correlated with the strength of amygdala coupling with frontal regions (medial prefrontal cortex, inferior frontal gyrus (IFG), and pregenual ACC) and also with angular gyrus, but was negatively correlated with amygdala coupling with other limbic regions (hippocampus and caudate) and with the cerebellum. The authors conclude that a greater ability to cognitively regulate emotional arousal is advantageous for therapy. Although they did not include healthy participants, it seems probable that the attributes that predicted desirable outcome may be milder pathophysiology, given that reduced prefronto-limbic resting connectivity differentiates social anxiety from healthy participants (Hahn et al., 2011; Prater et al., 2013). Two recent studies utilised graph analysis of resting-state data to predict outcomes of CBT in separate samples of 17 obsessive–compulsive disorder (OCD) patients. Göttlich et al. (2015) found that, compared to healthy participants, OCD patients exhibited reduced centrality (fewer connections, indicating reduced processing efficiency) from amygdala, hippocampus and ventral striatum, with amygdala connectivity inversely predicting symptom improvement. They hypothesise from this that relatively intact amygdala connectivity confers better fear extinction learning and, thus, treatment outcome. Examining mean connectivity parameters across the brain, Feusner et al. (2015) found that the number of connections to nearby regions (‘clustering’) increased following CBT, whereas connection length decreased, indicating an increase in overall efficiency. Because there was no correlation with symptom change, the authors interpret these changes as indicating general CBT effects on improved organisation and reflection on thoughts, feelings and behaviour. Surprisingly, when examining predictors of response, high levels on both of these measures at baseline predicted poorer outcome at follow-up, perhaps indicating that those who already utilising these self-reflective skills do not benefit as much from CBT.
Affective processing
Given that there was a large amount of variation in terms of disorder and experimental tasks, studies are discussed in separate subsections based on the regions investigated. Note that studies with multiple findings may be discussed in more than one section.
Prefronto-limbic connectivity
Three studies examined the effects of CBT on connectivity during facial affect processing tasks. Månsson et al. (2013) evaluated amygdala connectivity (constrained to prefrontal cortex) in two groups of 11 social anxiety patients who received either Internet-delivered CBT or an active control (attentional bias modification therapy). From pre- to post-therapy, there was an increase in negative coupling between amygdala and lateral prefrontal cortex (DLPFC and VLPFC), which was accompanied by reduced amygdala activation. This study was limited by a small sample with high numbers in receipt of pharmacotherapy. In addition, analyses collapsed across positive, negative and neutral affect conditions, potentially masking valence-specific effects. Addressing these limitations, Fonzo et al. (2014) recruited a larger sample of 21 medication-free patients with generalised anxiety disorder and examined longitudinal changes in amygdala connectivity for only negatively valenced faces (relative to positive faces). While amygdalo-insula connectivity was elevated in patients at baseline, there were no amygdala connectivity changes following CBT, despite reductions in amygdala activation. This may be due to differences between social and generalised anxiety disorder presentations or that their analyses contrasted positive and negative facial affect potentially obscuring change in either condition. Focusing on angry facial affect as a proxy for social threat, Mason et al. (2015) evaluated the effects of CBT for psychosis on connectivity from amygdala and DLPFC, compared to patients receiving standard care and healthy participants. They found that a baseline elevation in positive amygdala connectivity with insula and visual areas normalised following CBT. This coincided with an increase in positive coupling between amygdala and prefrontal regions, including DLPFC.
Studying a large and randomly allocated sample of social anxiety disorder patients (N = 31), Goldin et al. (2013) probed changes in DMPFC connectivity using a symptom challenge of idiosyncratic self-critical statements. When positively reappraising these statements, negative DMPFC coupling with amygdala and hippocampus increased following CBT compared to wait list. This was accompanied by greater and earlier activation of DLPFC and DMPFC in the CBT group. The authors conclude that dorsal prefrontal regions facilitate cognitive reappraisal of negative emotion through top-down regulation of the limbic regions involved in negative affect and memories associated with the self-critical content. Using a similar task, Månsson et al. (2015) examined predictors of long-term outcome following combined Internet-delivered CBT and an attentional intervention. At baseline, weaker positive coupling between dorsal ACC and amygdala and weaker activation in these two regions differentiated responders who maintained gains at 1 year follow-up from non-responders. The authors draw on evidence linking greater connectivity between this regions with stronger emotional experience (Etkin et al., 2011), concluding that this might be less desirable for long-term outcome. Interestingly, there was no change in amygdala coupling with ventral cingulate (KNT Månsson, personal communication, February 2015), which has been linked to emotion regulation (Etkin et al., 2011).
Three further studies relate to a large, randomised trial evaluating the impact of CBT for panic disorder on connectivity during an aversive conditioning task (Kircher et al., 2013; Lueken et al., 2013; Straube et al., 2014). The trial included two CBT treatment groups: in one, the CBT was delivered by a therapist (N = 22) and in the other, it was delivered in a self-help format (N = 20). Collapsing across both groups, Kircher et al. (2013) found that the panic disorder patients were characterised by elevated positive IFG coupling with amygdala, hippocampus and insula, but no change post-therapy. Lueken et al. (2013), also collapsing across groups, failed to find changes in pregenual ACC connectivity. However, pre-therapy, responders showed negative coupling with amygdala (and with DLPFC) in contrast to positive coupling in non-responders. Akin to Månsson et al. (2015), they offer pre-existing capacity for regulating the amygdala during extinction learning may be important (e.g. for exposure-based components of CBT). However, it should be noted that connectivity analyses were averaged across familiarisation and acquisition as well as extinction phases. It is also possible that sensitivity was reduced by collapsing across therapist-led and self-help groups. Addressing this issue, Straube et al. (2014) found that the therapist-led group showed a reductionin (positive) hippocampal coupling with IFG from pre-to post-therapy, potentially indicating effect on aversive learning processes in therapist-led CBT (although symptom reduction was comparable between the groups). It is worth noting that these three studies examined connectivity changes from ventral prefrontal cortical seeds and so connectivity changes from dorsal prefrontocortical regions remain a possibility.
In summary, of the seven studies reviewed, five found an effect implicating prefronto-limbic connectivity (Fonzo et al., 2014; Goldin et al., 2013; Göttlich et al., 2015; Månsson et al., 2015; Mason et al., 2015), one reported no change (Månsson et al., 2013) and one showed mixed findings across different analyses (Kircher et al., 2013; Lueken et al., 2013; Straube et al., 2014).
Insula connectivity
Two CBT studies reported insula in their connectivity analyses. Fonzo et al. (2014), described in the previous section, probed facial affect and found elevated insula connectivity with amygdala in generalised anxiety disorder compared to healthy participants. However, despite reductions in activation, this did not resolve following CBT. Simmons et al. (2013) evaluated the effects of trauma exposure therapy on insula connectivity in 24 combat veterans. Their analyses focused on symptom challenge, contrasting trauma-related combat images with positive images, pre- and post-therapy. In responders, there was a reduction in insula activation which occurred with an increase in its connectivity with dorsal anterior cingulate (extending into premotor cortex) and with cerebellum. The authors postulate that these changes could signify improved integration of salience signals (insula) with cingulate-based learning signals, facilitating unlearning of personal associations with trauma stimuli. However, this study was limited by the lack of a wait list control group and the small group of responders (N = 9).
Prefrontal connectivity with non-limbic brain regions
Lueken et al. (2013), already discussed, found that future responders showed greater inverse connectivity between pregenual ACC and DLPFC compared to future non-responders during aversive learning and extinction. These findings could indicate that improved top-down regulation of this ventral cingulate affective system from DLPFC is advantageous for therapy. In addition to the effects on inverse prefronto-limbic connectivity already reported, Goldin et al. (2013) found that CBT strengthened connectivity within prefrontal cortex during a symptom challenge. They found an increase in positive coupling between DMPFC and both DLPFC and ventromedial prefrontal cortex (VMPFC), possibly indicating an improved ability to integrate across higher order cognitive processes. Conversely, evidence from a single electroencephalography study (Miskovic et al., 2011) showed a reduction in frontal connectivity following group CBT for social anxiety disorder, as indexed by reduced synchronisation of beta and delta oscillations in this area. The authors, linking oscillations in these frequency bands to the transfer of information between subcortical and cortical targets, interpret their findings as evidence that therapy decreases the transmission of limbic threat signals to the neocortex. Further electrophysiology studies, especially in conjunction with functional MRI, will be needed to clarify whether the psychological processes captured are common or distinct across modalities.
Cognitive processing
Only one study examined cognition in relation to CBT. In a well-controlled study including both a treatment as usual and healthy participant group, Kumari et al. (2009) evaluated whether connectivity from DLPFC and IFG during a working memory task predicted response to CBT in 19 psychotic patients. Prospective reduction in psychotic symptoms was positively associated with baseline DLPFC activation and its positive coupling with cerebellum, and inversely associated with positive DLPFC-insula coupling. Neither of these connections differentiated psychosis from healthy participants, indicating that it is not the degree of pathophysiology that predicted outcome. The authors propose that greater connectivity with cerebellum may reflect relative sparing to cerebellar-thalamic-cortical circuitry and that this may confer improved executive functioning. Furthermore, the reduced connectivity of DLPFC with insula may reflect a greater ability to disengage the default mode network for task processing. Note that this study did not examine inverse connectivity.
Discussion
Despite a limited number of studies in this area, there is strong evidence that functional connectivity can predict, and is modified by, cognitive behavioural therapies. Tentative conclusions about possible predictors and mechanisms are discussed below, with greater emphasis on summarising the quality of the current literature and recommendations for future development.
Quality
There was a significant overrepresentation of anxiety disorders (80% of 15 studies) as well as methods probing affective processing (all but one of the task-based studies) and utilising functional MRI (also all but one study). Overall, the quality of studies included in this review was high, perhaps because of the recency of the studies (all but two were published in the last 2 years). For example, sample sizes were mostly optimal, with all but two (83%) exceeding minimum criteria (see Friston, 2012) in contrast to just 19% in a recent review of functional activation changes following psychological therapy spanning over 10 years (Barsaglini et al., 2014). However, of the ten studies examining mechanisms of therapy, only three included a clinical control group (only one of which was an active control treatment), making it difficult to establish how much changes were due to specific aspects of therapy rather than non-specific support. In addition, less than half (40%) of the studies included healthy participants, making it difficult to directly infer whether effects are brought about by normalisation or compensatory processes.
Emerging findings
Six of seven studies found that prefronto-limbic functional connectivity during affective processing was a significant predictor or outcome of CBT, which typically coincided with reductions in amygdala activation. These findings are consistent with predictions from cognitive neuroscience models of emotion regulation of threat and negative affect (Buhle et al., 2014; Wager et al., 2008). Considering studies reporting an effect, stronger inverse coupling between prefronto-cingulate and limbic regions predicted (Lueken et al., 2013) and mediated (Goldin et al., 2013; Månsson et al., 2013) successful therapy outcomes for anxiety disorders. Furthermore, weaker positive coupling in these circuits also predicted (Månsson et al., 2015) and mediated (Straube et al., 2014) favourable outcome, again for anxiety disorders. These findings may be integrated if we infer that positive prefronto-limbic coupling signifies dysfunctional interactions between cognitive processes in the experience of negative affect (e.g. threatening cognitions driving up the perceived threat and/or the experience of affect triggering further distressing cognitive content).
Given the marked overrepresentation of anxiety disorders, the extent to which these conclusions can be generalised to other disorders remains an open question. Interestingly, there was initial evidence of the opposite pattern in psychosis (i.e. an increase in positive coupling following CBT; Mason et al., 2015). This increase in frontal connectivity may signify disorder differences, e.g, a normalisation of the frontal disconnection that has been popularly reported in psychosis (Pettersson-Yeo et al., 2011), allowing greater reappraisal of distressing and unusual beliefs. This discrepancy reflects the need for more sophisticated tasks to better understand the functional significance of changes across disorders.
Both resting-state studies of anxiety disorders indicated that milder pathophysiology is a predictor of good outcome. There was some limited evidence that increased positive coupling between amygdala and ventral cingulate predicted better outcomes (Klumpp et al., 2014), in apparent contrast to the opposite pattern observed in one study of task-active connectivity (Kircher et al., 2013). To some extent, this is unsurprising, given that task and rest networks are highly anticorrelated (e.g. Raichle, 2011). One possibility is that at rest, there is negligible limbic activity, and so downregulation from prefrontal cortical regions is not advantageous (whereas the opposite may be true for studies probing connectivity with tasks that do engage affect). Accordingly, greater synchrony between resting prefronto-limbic fluctuations may index efficiency of this affect regulatory circuit.
Additional findings were increased in task-active connectivity within prefrontal cortex (Goldin et al., 2013; Mason et al., 2015) and with the cerebellum (Kumari et al., 2009), perhaps in keeping with a shared executive role (Bellebaum and Daum, 2007). Overall, changes in insula connectivity were rarely examined. This is in contrast with studies of mindfulness training which consistently increase insula activation, coinciding with an increase in inverse coupling (Farb et al., 2013; Kirk et al., 2014) or decoupling (Farb et al., 2007) with prefrontal cortex. This has been interpreted as a shift away from engaging cognitive and self-talk processes towards non-verbal, interoceptive awareness (Farb et al., 2007). Further work comparing different psychological interventions will be needed to clarify shared and distinct psychoneurobiological mechanisms.
A further question is to what degree the neurobiological mechanisms outlined here overlap with those of pharmacological interventions. Although connectivity mechanisms remain to be reviewed, reviews of activation studies indicate at least partially distinct mechanisms. Quidé et al. (2012) concluded that, across a range of disorders and methodologies, psychological interventions effect activation increases in higher order prefrontal cortical areas, whereas pharmacotherapy reduces activation of limbic structures. Further work directly comparing psychological and pharmacological interventions may be helpful for establishing common and distinct mechanisms.
Recommendations and future directions
Summarising the limitations above, there is a need for more studies, particular of other presentations besides anxiety disorders to enable quantitative aggregation methods (i.e. meta-analysis). Furthermore, there is a paucity of high-quality studies examining other therapy approaches besides CBT. This is surprising given the considerable literature for the neuroscience of mindfulness (mostly in healthy participants) and the ever-increasing evidence base for third-wave CBT approaches such as acceptance and commitment therapy and dialectical behavioural therapy.
For longitudinal studies, a more general methodological issue concerns whether therapy-related changes represent ‘state’ effects of reduced symptomatology, rather than mechanisms of therapy per se. It may be fruitful to examine ‘surrogate’ processes that approximate in-session therapy. For example, Cisler et al. (2014) examined the neural correlates of repeated exposure to personalised trauma narratives in people meeting criteria for post-traumatic stress disorder (PTSD). Across trials, they found increased positive coupling between amygdalo-hippocampal and prefrontal cortical regions, perhaps reflecting integration of aversive mnemonic traces with higher order cognitive processes. In other areas of social cognitive science, the simultaneous acquisition of functional connectivity data in multiple persons is being exploited for the purpose of studying reciprocal social cognitive processes (see, for example, Babiloni and Astolfi, 2014). While technical constraints currently render real-time neuroimaging of psychological therapy impractical, this may be a future area for understanding client and therapist interactions (Weingarten and Strauman, 2014).
With respect to neuroimaging methods, there is a need for more sophisticated connectivity analyses which allow the directionality of information transfer between regions to be inferred (e.g. using dynamical causal modelling; Friston et al., 2003) as none exist to date. Furthermore, studies examining changes in structural connectivity will be needed to address the current gap – ultimately, capitalising on new approaches that fuse both structural and functional connectivity, as well as analyses that enable more detailed network characteristics to be (such as the density and length of connections, e.g. Feusner et al., 2015; Göttlich et al., 2015) which can better determine the impact of therapeutic interventions on network connectivity. Finally, there was a marked lack of electrophysiology studies, greatly limiting our understanding of the temporal dynamics of therapy-related changes. More of these studies would help to ascertain, for example, to what degree therapy impacts early sensor–attentional processes as compared with later deliberative cognitive processing.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: V.K. and E.P. are part-funded by the Biomedical Research Centre for Mental Health at the Institute of Psychiatry, Psychology and Neuroscience (King’s College London) and South London and Maudsley NHS Foundation Trust, UK.
