Abstract
Background
Migraine is one of the most common and disabling of all medical conditions, affecting 16% of the general population, causing huge socioeconomic costs globally. Current available treatment options are inadequate. Serotonin is a key molecule in the neurobiology of migraine, but the exact role of brain serotonergic mechanisms remains a matter of controversy.
Methods
We systematically searched PubMed for studies investigating the serotonergic system in the migraine brain by either molecular neuroimaging or electrophysiological methods.
Results
The literature search resulted in 59 papers, of which 13 were eligible for review. The reviewed papers collectively support the notion that migraine patients have alterations in serotonergic neurotransmission. Most likely, migraine patients have a low cerebral serotonin level between attacks, which elevates during a migraine attack.
Conclusion
This review suggests that novel methods of investigating the serotonergic system in the migraine brain are warranted. Uncovering the serotonergic mechanisms in migraine pathophysiology could prove useful for the development of future migraine drugs.
Introduction
Migraine is a common, disabling and socioeconomically costly disorder characterised by recurrent attacks of throbbing, severe headache accompanied by multisensory symptoms (1,2). It affects 16% of the world’s population and ranks seventh highest among all causes of disability (3). The pathophysiology of migraine is complex and most likely involves peripheral mechanisms, such as sensitised perivascular trigeminal nociceptors (4), abnormal cortical sensory processing (5) and altered central pain modulation (6).
The serotonergic system in the brain has its origin in the raphe nuclei of the brainstem. From here, serotonergic neurons project to nearly every region of the central nervous system (CNS) (Figure 1), including the primary sensory cortex, the thalamus, the trigeminal nuclei and the dorsal horns of the spinal cord (7). Serotonin acts through several different receptor subtypes (see Table 1) (8) and is involved in many psychophysiological functions such as sleep, mood, appetite and pain modulation (9).
The serotonergic system in the brain. Schematic drawing depicting the serotonergic projections from the raphe nuclei. The raphe nuclei are divided into the rostral and the caudal groups.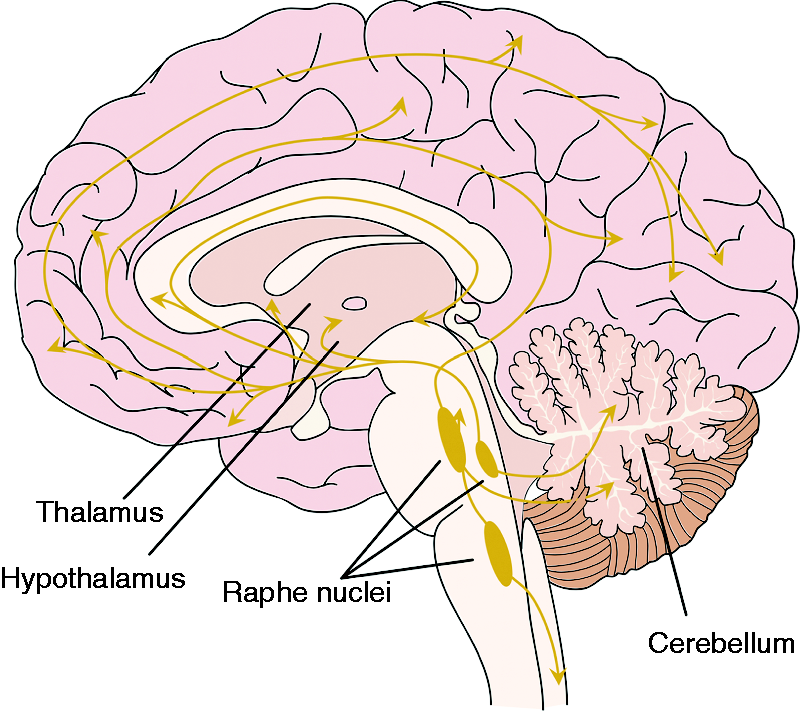
The role of serotonin (5-hydroxytryptamine (5-HT)) in migraine pathophysiology has been a matter of discussion for decades. In the early 1960s, it was observed that during migraine attacks the urinary excretion of the main metabolite of serotonin, 5-hydroxyaindoleacetic acid, was increased (10,11). Other early studies found low plasma serotonin content between attacks and increases during attacks (12,13). These observations led to the theory of migraine being a syndrome of chronically low serotonin levels with transient increases during attacks. Providing further support for serotonergic involvement in migraine pathophysiology, previously used prophylactic treatments such as methysergide and pizotifen are 5-HT2 antagonists (14) and the most efficient acute treatment to date – triptans – are selective 5-HT1B/1D receptor agonists (15–17).
Early studies of serotonin in patients employed measurements of the systemic serotonin levels and metabolism, but less is known about the serotonergic involvement in the brain of migraineurs (18). Here, we provide an overview of human studies investigating the serotonergic system in the migraine brain utilising molecular neuroimaging or electrophysiological methods as their primary tools. We discuss their methodological shortcomings and the implications of their findings for the understanding of the serotonergic involvement in migraine. Furthermore, we provide future perspectives on the continued search for serotonergic biomarkers in the migraine brain.
Investigating the serotonergic system in the brain
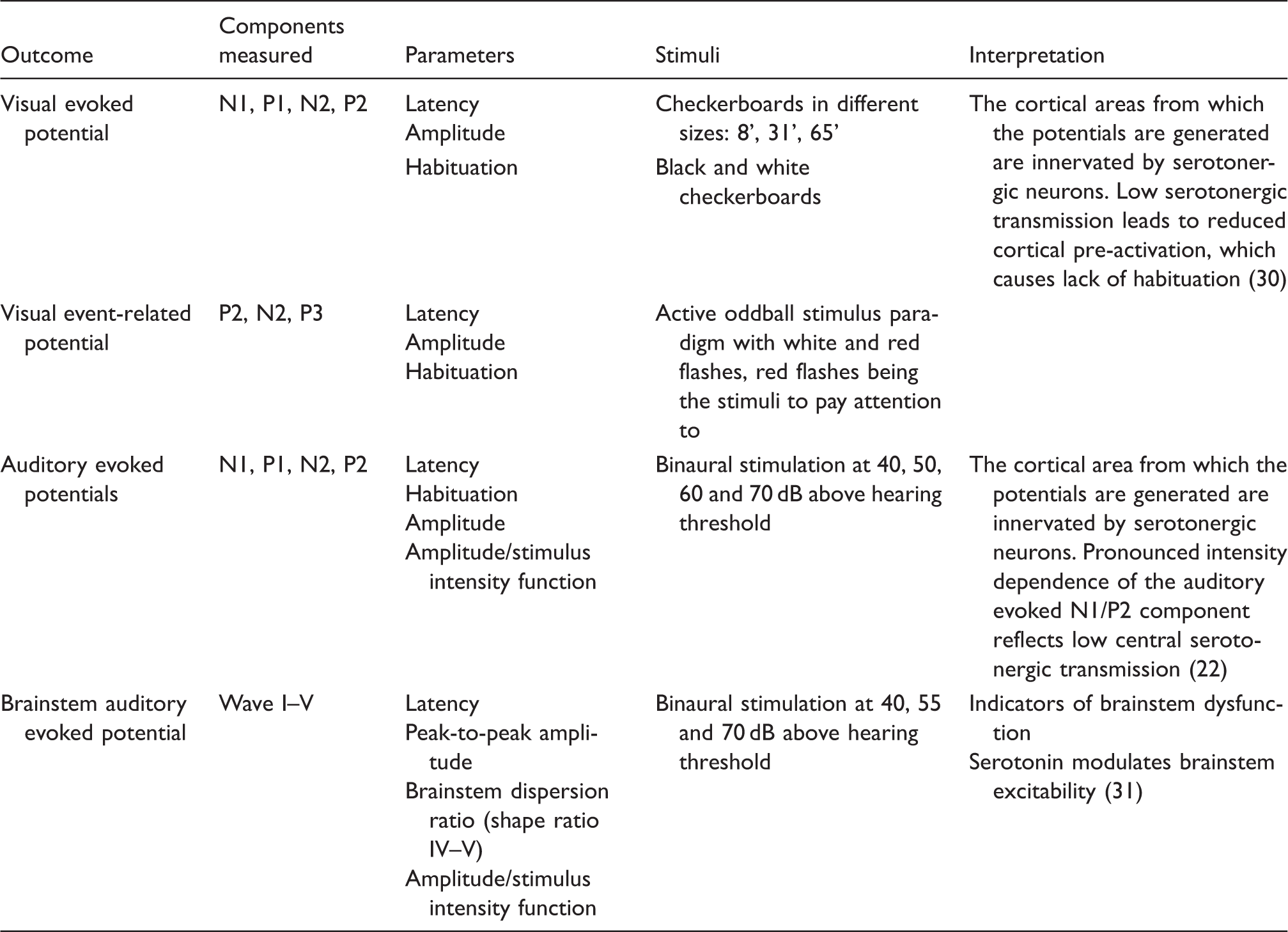
Molecular imaging such as positron emission tomography (PET) and single-photon emission computed tomography (SPECT) have been used to investigate the serotonergic system in the migraine brain. With specific radioligands, these methods enable investigations of different aspects of the serotonergic system, such as synthesis, serotonin transporter (SERT) availability and the density of serotonin receptors. These measures will be reported in this review (see Table 2). Serotonergic neurotransmission is assumed to modulate sensory processing in the visual and auditory cortices. Electrophysiological investigations with event-related potentials (ERPs) and evoked potentials (EPs) generated with visual or auditory stimuli are therefore applied in the investigation of central serotonergic neurotransmission in migraine (see Table 3) (22,23). In this review, we focus on correlations between these measures and blood, platelet or plasma serotonin.
Materials and methods
This review is based upon articles found via PubMed searching combining free text and MeSH terms for migraine and migraine-abortive treatment (“migraine” OR “migraineurs” OR “primary headache disorder” OR “triptan” OR “triptans”), serotonin and serotonin receptor (“serotonin” OR “5-HT” OR “5-HT receptor” OR “5-HT ligand” OR “serotonin receptor”) and neuroimaging and EPs (“PET” OR “neuroimaging” OR “positron emission tomography” OR “functional neuroimaging” OR “evoked potentials”). This search was carried out on September 15, 2014.
Furthermore, a review of the reference lists from the included articles was performed in order to find additional articles that were not found during the original search protocol. Human studies using molecular imaging or electrophysiological methods reflecting the serotonergic system in the migraine brain (see Tables 2 and 3) were considered to be eligible for review. Non-English studies were excluded. The initial screening was conducted based on the titles and abstracts. The remaining articles were reviewed in full text in order to determine eligibility.
Results
The PubMed search returned 59 records, of which 13 were eligible for review (Figure 2).
Results flowchart depicting the search protocol.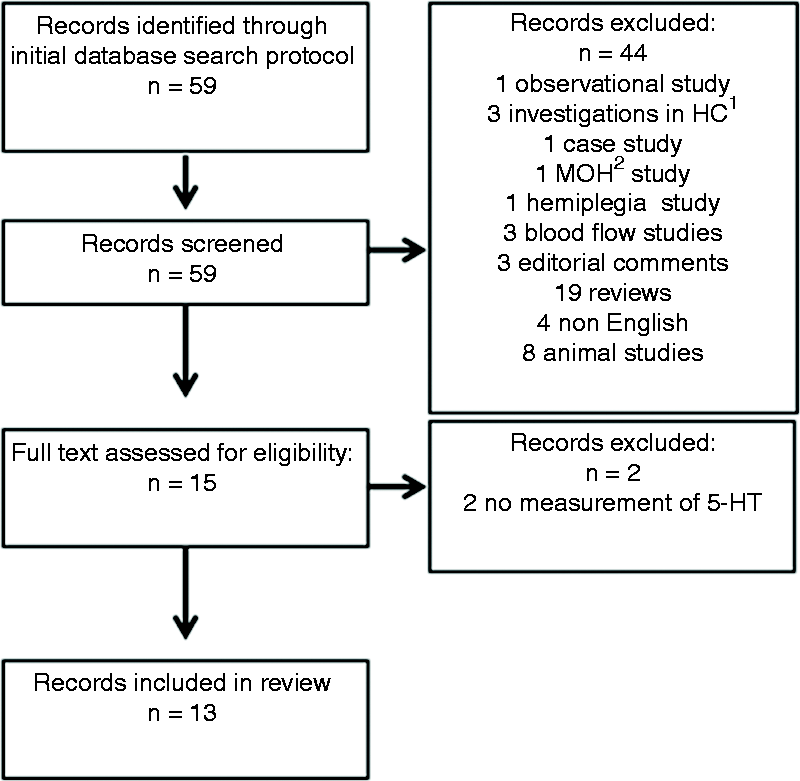
Given that migraine is a multiphasic disorder, we stratified and presented studies based on data collection timing (i.e. during interictal, preictal, ictal and postictal phases) (Figure 3). Furthermore, studies using serotonergic affecting drugs are presented.
The migraine cycle. Migraine is characterised by recurrent attacks of headache (ictal) accompanied by symptoms such as nausea, vomiting and photo- and phono phobia (2). Between attacks (interictal), patients are pain free. In the preictal phase, some patients experience premonitory symptoms such as neck stiffness, yawning, mood swings and fatigue (32).
The results and their interpretations by their authors are presented in Table 4.
Discussion
Interictal investigations
Molecular imaging
Cerebral serotonin synthesis in vivo was investigated in PET studies using the brain trapping constant, K*, for α-[11C] methyl-
Overview of serotonin receptors.

Not expressed in humans.
Molecular imaging methods used in the reviewed articles.

123I-ADAM: 2-((2-((dimethylamino)methyl)phenyl)thio)-5-iodophenylamine; SERT: serotonin transporter; SPM: statistical parametric mapping; BPND: binding potential (non displacelable); D2: dopamin type 2 receptor.
Electrophysiological methods used in the reviewed articles.
Molecular imaging and electrophysiological studies on the brain serotonin system in migraine patients.

-[11C]MTrp: α-[11C] methyl-
Electrophysiology
In migraine patients, a negative overall correlation between the platelet content of serotonin and absolute latencies of the P3 component of visual ERPs was found (43). A loss of habituation of P3 latency was also found, but this was not associated with platelet serotonin content. These results suggest that serotonergic neurotransmission is linked to the latency of P3, but not to habituation in migraine patients, contrasting with reports of low serotonin causing a lack of habituation of auditory EPs (AEPs). In a longitudinal blinded study, the correlation between brainstem auditory EPs (BAEPs) and plasma serotonin levels was investigated. A positive correlation between amplitude and plasma serotonin was reported in controls, but not in migraine patients (31). The same group reported similar results for visual evoked potentials (VEPs), in which the N1P1 amplitude for medium-sized checks correlated positively with serotonin in plasma and platelets in controls, but not in migraine patients (44). Collectively, these data suggest an interictal disturbance of the association between systemic serotonin and EPs (31,44). This could indicate an altered serotonergic neurotransmission in migraine patients, but caution should be taken when comparing systemic and central serotonin levels, since serotonin is not transported from the periphery to the CNS (17).
Preictal investigations
Electrophysiology
For stimulations with 70 dB, a trend towards a negative correlation between BAEP wave I and V latency and plasma serotonin was observed preictally (31). No correlation was seen for amplitude or the brainstem dispersion ratio (SR IV–V; i.e. a measure of brainstem function). Two VEP studies did not report any correlations between serotonin levels or any parameters measured (44,45). These data suggest that the disturbance of the normal, positive correlation (see the section entitled ‘Interictal investigations’), which is found interictally in migraine patients, persists through the preictal phase, providing further support for the role of serotonin in migraine pathophysiology.
Ictal investigations
Molecular imaging
A small PET study of the 5-HT1A receptor conducted during odor-triggered migraine attacks (46) showed higher binding in patients experiencing migraine attacks (n = 4) compared to both controls (n = 10) and to patients not developing a migraine attack (n = 6). The increased 5-HT1A binding was found in both cortical areas and in the brainstem. These results could indicate an upregulation of the receptor or changes in receptor affinity during migraine attacks (46). A decrease in endogenous serotonin, as suggested by the authors, is not likely to cause the increase in binding (see the section entitled ‘Interictal investigations’). An increase in K* for α-[11C]MTrp, which was found during migraine attacks, was suggested by the authors to reflect an increase in serotonin synthesis rate (37). The results are therefore in line with the theory of ictal increases in endogenous serotonin in the brain, but the reliability of the method and the number of participants (n = 6) should be taken into account.
Electrophysiology
Serotonin in plasma and habituation for wave IV–V amplitude and SR IV–V correlated negatively in the ictal phase (31). As in the preictal period, this was only found for one stimulus level of 55 dB, but not for 70 dB. A positive correlation between both P1N2 amplitude habituation (45) and N1P1 amplitude (44) in VEP and serotonin in platelets was found in a small group of patients (n = 8) with migraine with aura, but not in patients suffering from migraine without aura. The latter correlation was also found in controls (44,45). This indicates an ictal normalisation of the relationship between systemic serotonin and VEPs in migraine with aura.
During the migraine cycle, dynamic changes in both loss of habituation (i.e. no decrease in response after repeated stimuli) and in platelet serotonin content were observed (43). Interestingly, a decrease in platelet serotonin content coincided with an increase in habituation of the P3 latency during migraine attacks, but there was no correlation between the level of habituation and the serotonin level in platelets or in plasma. The increase in cognitive processing time, measured as P3 latency, reported during attacks was negatively correlated with the serotonin content in platelets. Based on these results, the authors suggested that the increase in cognitive processing time, but not the changes of habituation (using an oddball paradigm), during a migraine attack could be related to decreases in serotonergic neurotransmission. This is in contrast with data indicating an ictal increase in serotonergic neurotransmission (44,45). The authors did not report whether the same correlations were seen in controls, and it needs to be investigated further as to whether this is migraine specific or a common feature of the serotonergic system.
Postictal investigations
Electrophysiology
A positive correlation between wave I latency and plasma serotonin for 70 dB was found for BAEPs, in contrast to the negative correlation seen preictally. Furthermore, positive correlations between plasma serotonin and wave V amplitude, SR IV–V and amplitude/stimulus intensity function (ASF) slope were observed, which are similar to the correlations seen in controls (31). This indicates a normalisation of the correlations in migraine patients in the postictal period, which are suggested to reflect a normalisation of the serotonergic neurotransmission (31). However, these findings were derived from only one study and need to be replicated.
Investigations after treatment with serotonin-affecting drugs
Molecular imaging
In one small study (n = 5), α-[11C]MTrp brain uptake was investigated in migraine patients before and after 12 weeks of treatment with β-blockers (either propranolol or nadolol, 5-HT1A receptor antagonists and partial 5-HT1B agonists) (36). Four out of five patients showed an increase in whole-brain uptake of the radioligand, suggesting an increase in serotonin synthesis capacity after treatment. This is consistent with 5-HT1A antagonism, but contrasts with 5-HT1B receptor agonism, since activation of 5-HT1A and 5-HT1B autoreceptors leads to a decrease in serotonin synthesis and release (47). Furthermore, the same study found a higher brain uptake in migraine patients before treatment compared to controls. Based on these results, it is surprising that prophylactic treatment causes further increases in brain uptake. However, the results should be interpreted with caution because of the small sample size. Eletriptan, a 5-HT1B/1D receptor agonist that crosses the blood–brain barrier (BBB) (48), significantly decreased the α-[11C]MTrp brain uptake in migraine patients in the interictal state, but had no effect in controls (24). This indicates a difference in response to 5-HT1B receptor activation between the two groups. This difference could be due to sensitisation of 5-HT1B autoreceptors in migraine patients, caused by low interictal serotonin levels in the migraine brain. In the ictal phase, sumatriptan, another 5-HT1B/1D agonist, decreased the α-[11C]MTrp brain uptake compared to both interictal and ictal levels in migraine patients (37). This suggests a central effect of sumatriptan on serotonin synthesis during migraine attacks, which could be exerted through 5-HT1B autoreceptors (37).
Electrophysiology
An electrophysiological study with a limited number of subjects (49) demonstrated that treatment with both 5 mg (n = 8) and 10 mg (n = 6) zolmitriptan significantly increased the ASF slope of AEPs compared to both baseline and to placebo. The increase tended to be more pronounced in migraine patients compared to controls, but the difference did not reach statistical significance. Sumatriptan did not induce significant changes. The ASF slope reflects the intensity dependence of AEPs (IDAP), which is thought to be inversely related to serotonergic neurotransmission (50). The results, which are limited by the low number of subjects, thereby indicate that zolmitriptan decreases serotonergic neurotransmission, and this effect is more pronounced in migraine patients. This is in line with the effect of sumatriptan and eletriptan on α-[11C]MTrp brain uptake and the hypothesis of chronic low serotonin in the migraine brain. In another electrophysiological study, the selective serotonin reuptake inhibitor fluoxetine was administered (20 mg daily) for 1 month (51). After treatment, migraine patients without aura (but not with aura) showed increased first block amplitude of VEPs compared to baseline. The treatment also corrected the lack of habituation observed in migraine patients during baseline. There was no longer a significant difference in mean amplitude changes in the fifth block expressed as a percentage of the first block between controls and migraine patients. These data suggest that VEPs are linked to serotonergic neurotransmission and that serotonergic neurotransmission is altered in migraine patients.
Methodological shortcomings
The results from existing investigations of the serotonergic system in the migraine brain are divergent (Table 4). Methodological shortcomings are pivotal factors causing these discrepancies. Several studies measure the platelet content of serotonin as a surrogate of neuronal transmission. Even though platelets have some resemblance to neurons with regards to the receptors, release, reuptake and storage of serotonin (52), platelet content of serotonin does not reflect the synaptic mechanisms between neurons. Plasma serotonin should not be directly compared to the serotonergic tonus in the brain either, since serotonin does not cross the BBB to any appreciable extent. Another point of concern with several of the reviewed studies is that headache diaries were not collected after the investigations (24,36–39,49). Serotonin levels are thought to change in a cyclical manner in relation to the migraine cycle. Therefore, the time since the last migraine attack and the time to the next attack, relative to the time of the investigation, are highly important variables. The different subsets of subjects regarding age, gender, diagnosis and number are also highly discrepant between the studies. The stimulations used in the electrophysiological studies also differ in terms of, for example, check sizes of the alternating checkerboards for VEPs and sound frequencies and intensity levels for AEPs. Blinding of investigators is important, since three non-blinded electrophysiological studies found a lack of habituation in migraine patients (50,53,54), while blinded studies could not replicate these findings (55,56). However, a recent study comparing the habituation findings of blinded and non-blinded investigators found no differences in the results (57). Collectively, it is difficult to compare these results and draw any firm conclusions.
Brain PET studies using different serotonin receptor radioligands have limitations as well, especially due to the complex relationship between brain serotonin levels and the binding of serotonin receptor radioligands. Since the binding potentials depend on receptor affinity, neurotransmitter levels and receptor availability results must be interpreted with caution. The radioligands used in the reviewed studies are antagonists, which means that they are less sensitive to endogenous serotonin release compared to radioligands that are serotonin receptor agonists (27). Thus, the ictal increase in serotonin is less likely to be detectable with these ligands. Furthermore, the regulation of serotonin receptors is different from receptor to receptor (58,59). A low level of serotonin does not necessarily cause an upregulation of the receptor (60). Since serotonin is involved in the circadian rhythm (9), the timing of the scans should also be taken into consideration when interpreting binding potentials, as well as possible age differences between receptor distribution and density (61).
Possible serotonergic mechanisms in migraine pathophysiology
Even though the importance of serotonin in migraine pathophysiology is indisputable, the changes in the level of endogenous serotonin in the migraine brain have not yet been convincingly determined. The reviewed studies support the notion that migraine patients have alterations in the serotonergic system compared to non-migraineurs, and most likely they have a lower level between attacks, which then increases during a migraine attack (Table 4). How this difference in endogenous serotonin between migraine patients and controls and the sudden increases during attacks are linked to the sensation of head pain still remains to be fully elucidated. The headache phase in migraine depends on the activation of sensitised peripheral nociceptors and the trigeminal pathway (4), but central modulation of these signals is also involved. One prevailing theory relating serotonergic abnormalities to migraine is a central dysfunction of the pain-modulating system in migraineurs (62,63). Serotonin both facilitates and inhibits pain, depending on the site of action and the receptor subtype it activates (64). In the CNS, serotonin is predominantly analgesic, especially via mechanisms of descending inhibition (65), but a facilitatory role of serotonin has also been suggested (66). A low level of serotonin interictally could result in disinhibition of pain signals from peripheral nociceptors, thus lowering the threshold for the induction of headache. Furthermore, animal studies have shown that serotonin exacerbates pain in sensitised animals, whereas it inhibits pain in control animals (67). Low serotonergic tone could make migraine patients hypersensitive and more susceptible to stimuli, with sudden increases in serotonin during migraine attacks enhancing and maintaining the pain (e.g. through binding to proalgesic 5-HT2A receptors) (64). Collectively, the function of serotonin in the pain-modulating system is dualistic and complex. It depends on receptor subtype, availability and affinity and the physiological or pathophysiological status, making it difficult to decipher the causality between serotonin and headache.
Supporting the theory of an increase in serotonin contributing to the headache are studies showing that treatment with serotonergic agonists acting on the cerebral excitatory 5-HT2B and 5-HT2C receptors or agents increasing synaptic serotonin causes headache (17,68,69). In contrast, intravenous infusion of serotonin relieved the pain in migraine patients (70) and inhibited sensory inputs from the dural vasculature in animals (71). This effect was most likely exerted through peripheral action sites, since serotonin does not cross the BBB (21). Evidence for central pain-relieving effects of serotonin is found in animal studies showing that the release of serotonin upon stimulation of the periaqueductal grey area causes analgesia through the stimulation of inhibitory interneurons acting on dorsal horn neurons (20,72). Since serotonin increases are observed after injury to nerves or to the spinal cord in animal models (73), one might also hypothesise that the increase in brain serotonin during a migraine attack could be a protective and pain-relieving mechanism.
PET studies reported brainstem activation during spontaneous migraine attacks (74,75). The activated areas include the raphe nuclei, suggesting activation of the serotonergic neurons. The low resolution of this method should, however, be taken into account when interpreting these results. Theoretically, an increased activation in the raphe nuclei could be associated with an increase in serotonergic signaling in the projection areas and activation of the endogenous anti-nociceptive system. Interestingly, the activation persisted following treatment with sumatriptan, indicating that brainstem activation is not necessarily coupled to pain processing and 5-HT1B receptor-mediated serotonergic mechanisms (75).
The most efficient acute treatment of migraine – triptans – are 5-HT1B receptor agonists (15,48). The 5-HT1B receptor is present on the dural and cerebral vasculature, in the trigeminal ganglion and throughout the brain. It is both an autoreceptor inhibiting the release of serotonin from serotonergic neurons and also a heteroreceptor acting on non-serotonergic neurons (19,76). The central mechanisms of triptans are a subject of intense debate and have been investigated in several studies. Brain PET studies reported that zolmitriptan crosses the BBB and binds to central 5-HT1B receptors with relatively low occupancy (77,78). It is still unknown whether sumatriptan has a central effect. Extracranial but not intracranial arteries constricted after sumatriptan treatment during a migraine attack (79), arguing against its penetration of the BBB. On the other hand, sumatriptan induces more CNS adverse events compared to placebo (80). A recent study reported that sumatriptan, but not acetylsalicylic acid, caused a decrease in trigeminal–cortical coupling in healthy volunteers after activation of the trigeminal nociceptive system (81). Furthermore, there is a discrepancy between the persistent brainstem activation (75) and the decreases in serotonin synthesis (37) after sumatriptan treatment. It could be speculated that 5-HT1B receptor agonists modulate pain transmission in the CNS by primarily acting in the peripheral nervous system. On the other hand, if the pain relief is caused by central mechanisms, these could be explained by the existence of 5-HT1B receptors in the descending pain-modulating pathways in the brainstem (82), with the inhibition of serotonin release and synthesis then being secondary. This further underlines the importance – and complexity – of serotonin in the migraine pathophysiology.
Future perspectives
The present review suggests that electrophysiological investigations are not optimal in the search for serotonergic biomarkers. Firstly, EPs and ERPs only indirectly reflect serotonergic neurotransmission. Secondly, the findings from electrophysiological studies are highly inconsistent. Interictal lack of habituation, which is the most consistent finding, is not reproducible in all studies. Recently, it has been disputed whether lack of habituation, also called the ‘neurophysiological hallmark of migraine’ (83), is even specific for migraine (56,84). Low first block amplitude and increased IDAP in migraine patients are additional controversial findings that are thought to be consequences of low serotonergic disposition (30,56). Reliable methods of elucidating the central serotonergic system in the migraine brain are therefore much needed. Future studies need to focus on more direct methods of investigating the serotonergic system in migraine. A recent study suggested that central 5-HT4 receptor binding might serve as a biomarker of serotonergic tonus in the human brain (85). This method may be applied in migraine patients in order to accurately determine the differences in serotonergic levels between the migraine brain and controls. 5-HT4 receptor binding is not affected by acute changes in endogenous serotonin levels, and other methods must be applied in order to obtain knowledge of the cyclical nature of the serotonin levels in the migraine brain. Currently, few PET studies have succeeded in measuring endogenous serotonin release (27). Studies with the 5-HT1B receptor ligand [11C]AZ10419369 in non-human primates have shown some potential, with decreases in binding after fenfluramine-induced serotonin release (86). Results from human studies have been less promising, with an increase in cortical binding after a single dose of escitalopram, a selective serotonin reuptake inhibitor (87). However, there was lower binding in the raphe nuclei, suggesting that the ligand is in fact sensitive to acute changes in endogenous serotonin, thus making it possible to detect changes in the serotonergic tonus during migraine attacks. In the future, these methods should be applied in migraine research in order to gain more knowledge of the pathophysiology and serotonergic mechanisms in the migraine brain.
Clinical implications
Migraine is most likely associated with low serotonin levels between attacks and transient increases during attacks. The results of studies on the endogenous brain serotonin levels in migraine are divergent and brain serotonin levels have not yet been conclusively determined. Electrophysiological studies yielded conflicting results and might not be optimal for investigating serotonergic biomarkers in the migraine brain. Future studies should apply more direct methods of visualising the serotonergic system in the migraine brain, such as positron emission tomography with radiolabelled serotonin receptor agonists.
Footnotes
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship and/or publication of this article: this study was supported by the Lundbeck Foundation, grant number R-180-2014-3398.
