Abstract
Objective:
Individuals with bipolar disorder experience a disproportionately high incidence of medical co-morbidity and obesity. These health-related problems are a barrier to recovery from mood episodes and have been linked with unfavorable responses to pharmacological treatment. However, little is known about whether and how these characteristics affect responses to adjunctive psychotherapy.
Method:
Embedded in the Systematic Treatment Enhancement Program for Bipolar Disorder was a randomized controlled trial of psychotherapy for bipolar depression comparing the efficacy of intensive psychotherapy plus pharmacotherapy with collaborative care (a three-session psycho-educational intervention) plus pharmacotherapy. We conducted a post-hoc analysis to evaluate whether medical burden and body mass index predicted and/or moderated the likelihood of recovery and time until recovery from a depressive episode among patients in the two treatments.
Results:
Participants who had medical co-morbidity and body mass index data constituted 199 of the 293 patients in the original Systematic Treatment Enhancement Program for Bipolar Disorder trial. Higher medical burden predicted a lower likelihood of recovery from depression in both treatment conditions (odds ratio = 0.89), but did not moderate responses to intensive psychotherapy vs collaborative care. Intensive psychotherapy yielded superior recovery rates for individuals of normal body mass index (odds ratio= 2.39) compared with collaborative care, but not among individuals who were overweight or obese.
Conclusion:
Medical co-morbidity and body weight impacts symptom improvement and attention to this co-morbidity may inform the development of more personalized treatments for bipolar disorder.
Introduction
Bipolar disorder is a chronic and debilitating psychiatric illness characterized by recurrent episodes of mania and depression (American Psychiatric Association [APA], 2000). The standard first line of treatment for bipolar disorder is pharmacotherapy, but this treatment alone often fails to bring patients to sustained remission (Gelenberg et al., 1989; Judd et al., 2008). Adjunctive psychosocial interventions increase the odds of remission, sustained euthymia and prevent future mood episodes. A number of randomized controlled trials have demonstrated that psychosocial treatments such as family-focused therapy (FFT) (Miklowitz et al., 2003; Rea et al., 2003), cognitive behavioral therapy (CBT) (Cochran, 1984; Lam et al., 2005), interpersonal and social rhythm therapy (IPSRT) (Frank et al., 2005, 2008) and group psycho-education (Colom et al., 2003) are effective in reducing residual symptoms, improving medication adherence, preventing future mood episodes and improving overall psychosocial functioning in conjunction with pharmacotherapy. Despite the significant gains that can be made by pairing psychotherapy with medication, there remains variability in response rates, underlining the need to determine which patients are most likely to respond to adjunctive psychotherapy.
Medical co-morbidity is one illness characteristic that may contribute to variability in response to psychotherapy or pharmacotherapy. Medical burden, defined as the number of lifetime medical conditions, is associated with a number of adverse longitudinal outcomes, including a more chronic course, longer duration of illness, poorer global functioning and higher rates of disability (Magalhaes et al., 2012). Preliminary evidence suggests that bipolar patients with more medical illnesses respond more poorly to pharmacologic treatment. Kemp et al. (2010) found that patients with a higher body mass index (BMI) and medical illnesses of the endocrine and metabolic systems are less likely to achieve full remission (Calkin et al., 2009; Kemp et al., 2010). Similarly, Thompson et al. (2006), found that higher severity of baseline medical co-morbidities predicted slower decreases in depression among bipolar patients receiving adjunctive IPSRT or pharmacotherapy (Thompson et al., 2006).
Prior work has demonstrated that medical burden adversely affects response to pharmacological treatment; however, no prior studies have investigated how BMI or medical problems affect the outcome of psychosocial interventions of different intensity in bipolar depression. Accordingly, the objective of this study was to conduct a post-hoc analysis evaluating whether medical burden and BMI predict or moderate response to adjunctive intensive psychotherapy (up to 30 sessions over 9 months of FFT, IPSRT or CBT) or collaborative care for depression (three individual sessions of education and instruction in self-care) in the Systematic Treatment Enhancement Program for Bipolar Disorder (STEP-BD) psychotherapy trial. Because of studies linking medical problems and obesity with symptom severity and impairment in functioning (Magalhaes et al., 2012), we hypothesized that patients with a greater number of medical conditions and higher BMI would have poorer treatment response overall. We predicted this result because individuals with high medical burden and BMI are less likely to be physically active than those with low medical burden and normal body weight, which could interfere with their engagement in between session homework assignments. Secondarily, we hypothesized that patients high on medical burden and BMI would benefit more from intensive psychotherapy because they may be less likely to maintain consistency with medications or sleep/wake cycles, or to follow-up with assignments such as exercise, family skills training tasks or pleasurable events scheduling, each of which may be central to mood improvement.
Method
Study design
STEP-BD was a longitudinal nationwide study, funded by the National Institute of Mental Health. It was designed to study course, outcome and treatment effectiveness in bipolar disorder. Embedded within the prospective, longitudinal study were a number of clinical trials, evaluating the efficacy of pharmacotherapies such as mood stabilizing medications, antidepressants and atypical antipsychotics, as well as psychosocial interventions such as brief psycho-education, FFT, CBT and IPSRT. (For a full description of the nature and scope of the STEP-BD study, refer to Sachs et al. (2003)).
One of the clinical trials in STEP was a randomized controlled trial of intensive psychotherapy vs collaborative care for acute bipolar depression (Miklowitz et al., 2007). Participants were randomly assigned to receive 9 months (up to 30 sessions) of manualized treatment with intensive psychotherapy, or 6 weeks (up to three sessions) with collaborative care. Stratification variables included study site, bipolar I or bipolar II status, availability of a family for treatment and, if simultaneously in the randomized acute depression study, pharmacological treatment (mood stabilizer with or without an antidepressant). Patients were assigned to intensive psychotherapy or collaborative care within each stratum using a 60:40 randomization formula. Collaborative care was a brief psycho-educational intervention. Participants in the intensive psychotherapy condition received FFT, CBT or IPSRT. These three intensive psychotherapies were also grounded in psycho-education, but were designed as enhanced treatments, each with a unique theoretical foundation. FFT was centered on the impact of the family system on the course of bipolar disorder, and accordingly involved educating the family about bipolar disorder and enhancing family communication problem solving (Miklowitz et al., 2003). CBT focused on the role of maladaptive cognitions, and accordingly involved restructuring cognitive distortions, challenging negative thought patterns, problem solving and activity planning (Lam et al., 2005). IPSRT focused on stabilizing social rhythms that are common precipitants of mood episodes, and accordingly involved solving interpersonal problems, regulating sleep, eating and behavior patterns and addressing relationship difficulties and role disputes (Frank et al., 2005, 2008).
Participants
A full description of participant inclusion/exclusion criteria has been previously reported (Miklowitz and Otto, 2007; Miklowitz et al., 2007). Eligible participants (n = 293) met the Diagnostic and Statistical Manual of Mental Disorders-IV (DSM-IV) criteria for bipolar I or II disorder. Diagnoses were established by the Mini-International Neuropsychiatric Interview (MINI) (Sheehan et al., 1998), with information corroborated by the Affective Disorders Evaluations (ADE) (Sachs et al., 2003). All eligible participants met MINI criteria for a current major depressive episode. Participants were also treated or willing to initiate treatment with a mood stabilizing medication. Of the 293 participants enrolled in the STEP-BD psychosocial pathway, this study includes a subsample of participants (n = 199), with information available from their baseline visit regarding medical conditions and BMI.
Measures
Treatment outcome
Symptoms of depression and mania were assessed at each treatment visit using the Clinic Monitoring Form (Sachs et al., 2003). Recovery was defined as experiencing ⩽ two moderate mood symptoms for ⩾ eight consecutive weeks. Participants were designated non-recovered status if they failed to meet these criteria. Intraclass inter-rater reliability coefficients for Clinic Monitoring Form depression and mania items ranged from 0.83 to 0.99 (Sachs et al., 2003).
Medical burden
Participants reported the lifetime presence or absence of a variety of medical conditions, including cancer, diabetes, sleep apnea, cardiovascular problems, thyroid disease, seizures, hepatitis, multiple sclerosis, migraines, Stevens-Johnson syndrome, peptic ulcers, diastolic murmur, headaches, allergies, asthma, eczema, Raynaud’s phenomenon, head trauma with loss of consciousness and other loss of consciousness. This history of medical co-morbidities was obtained through patient interview by a physician using the ADE (Sachs et al., 2003). All medical conditions were coded for lifetime presence or absence. An index of medical illness severity was not available. An index of cumulative medical burden was created by summing across categories, resulting in a continuous variable—total number of medical conditions endorsed.
Body mass index (BMI)
BMI is a ratio of height and weight, defined as an individual’s body weight divided by the square of their height. BMI was used as the best proxy of obesity. Waist measurement was not recorded as part of the routine physical screening. Participants’ height and weight were measured at baseline during their physical screening, and BMI was calculated based on these measures. BMI below 25 is considered normal, 25–29.9 is considered overweight and above 30 is considered obese. In this sample, 28% (n = 55) of participants were of normal weight, 38% (n = 76) were overweight and 34% (n = 68) were obese. There were no significantly under-weight participants.
Psychosocial treatment outcome
Miklowitz et al. (2007) documented the superiority of adjunctive psychotherapy relative to adjunctive collaborative care for achieving recovery and time to recovery. Due to reduced power in the smaller subsample of participants included in this analysis (n = 199), the effect of treatment condition on recovery, χ2(1, n = 199) = 0.59, p = 0.44, and time to recovery, b = 0.18, standard Eerror [SE] = 0.19, p = 0.33, was no longer significant. To dissociate whether this change in treatment effects was due to sample reduction or bias on behalf of the selected subsample, we tested whether a dummy coded variable (included selected subsample = 1, excluded = 0) interacted with treatment condition. Subgroup membership did not interact with recovery rates (p = 0.08) or time until recovery (p = 0.20). Furthermore, established parameters for testing moderators in randomized clinical trials do not necessitate a main effect of treatment condition (Kraemer et al., 2008). Our approach for testing medical burden and BMI as predictors and moderators in this subsample are detailed below.
Data analytic approach
Preliminary analyses compared the selected sample with the full original STEP-BD sample on clinical and demographic variables. The hypothesized predictors and moderators of treatment outcome were medical burden (number of lifetime medical conditions) and BMI. Treatment outcome was defined using two variables: (1) recovery (a binary, or success/failure outcome variable) and (2) time until recovery. When examining recovery, we used a series of conditional logistic regression analyses, and when examining time to recovery, conditional Cox proportional hazard (survival) models. All analyses were by intention to treat. Patients were included until their final assessment point, with a maximum of 365 days in the study (mean [M] = 176.94 days, standard deviation [SD] = 104.53 days) (Sachs et al., 2003). The proportionality of risk assumption was met for all survival analyses.
Medical burden, defined as the number of lifetime medical conditions, is associated with a number of adverse longitudinal outcomes including, a more chronic course, longer duration of illness, poorer global functioning and higher rates of disability (Magalhaes et al., 2012). Medical burden and BMI were evaluated independently in separate regression models as continuous predictors. To evaluate the significance of these variables for predicting recovery status in patients receiving each randomized treatment (intensive psychotherapy or collaborative care), we examined treatment type by predictor interactions in the model. To probe the form of a significant interaction with BMI, simple slopes of BMI on recovery were tested at the conventional values for denoting normal weight, overweight and obese (Aiken and West, 1991).
Medical burden and BMI have demonstrated associations with age and sex (Goldstein et al., 2011; Post et al., 2015). Also, in our previous studies, we investigated co-morbid anxiety disorders, number of mood episodes, illness duration, affective instability and sleep duration as predictors or moderators of psychotherapy treatment outcome in STEP-BD (Deckersbach et al., 2014; Peters et al., 2014; Stange et al., in press). To understand the unique contribution of medical burden and BMI to the prediction of outcome, we first examined associations between age, sex, anxiety, illness course and sleep and our medical predictors. Subsequently, we included these variables as covariates.
Lifetime anxiety was coded as present or absent (present = 1, absent = 0), number of mood episodes was dummy coded with 1–9 episodes as the reference category (1–9 episodes = 0, 10–20 episodes = 1, 20+ episodes = 2) and illness duration was a continuous variable reflecting number of years with a bipolar disorder diagnoses (e.g. years since mania or hypomania onset). Affective instability was determined by calculating a mean square successive difference (MSSD) score, which reflects the size and temporal order of which consecutively measured moods differ from one another (Ebner-Priemer et al., 2009; Jahng et al., 2008). Thus, the MSSD for mania and depression reflects the degree to which manic and depressive fluctuate over time, taking into account initial symptom severity. The MSSD was computed for each symptom of depression and mania from consecutively assessed mania and depression ratings in the ADE.
Sleep duration was operationally defined as the average number of hours of sleep in the past week by averaging the minimum and maximum sleep duration values reported on the clinical monitoring form (Gruber et al., 2009). Participants were divided into three groups based on their average weekly sleep duration: short sleepers (<6.5 hours/night), normal sleepers (6.5–8.5 hours/night) and long sleepers (>8.5 hours/night). These cutoffs have been validated in other studies based on their distinct clinical correlates (Edinger et al., 2000; Gruber et al., 2009; Kaneita et al., 2007).
The effect size estimate (odds ratios) for the effect of medical burden or BMI on treatment outcome is presented before and after adjusting for each of these covariates. Covariates were added separately to examine the effect of each covariate. Because of missing data, the sample was reduced with the addition of each covariate. A minimum of n = 118 participants was needed to detect a medium-sized effect in a regression model, including randomization, medical burden (or BMI) and one additional covariate with 95% confidence.
Results
Study sample
Demographic and clinical characteristics of the total sample (n = 199) are presented in Table 1. The selected sample was comparable to the original STEP-BD sample on demographic and clinical variables with two exceptions: the current sample was approximately 2.5 years younger (F = 4.89, p = 0.028) and reported slightly higher ratings of depression at baseline (F = 4.73, p = 0.030).
Demographic and illness characteristics of 199 bipolar-depressed participants.
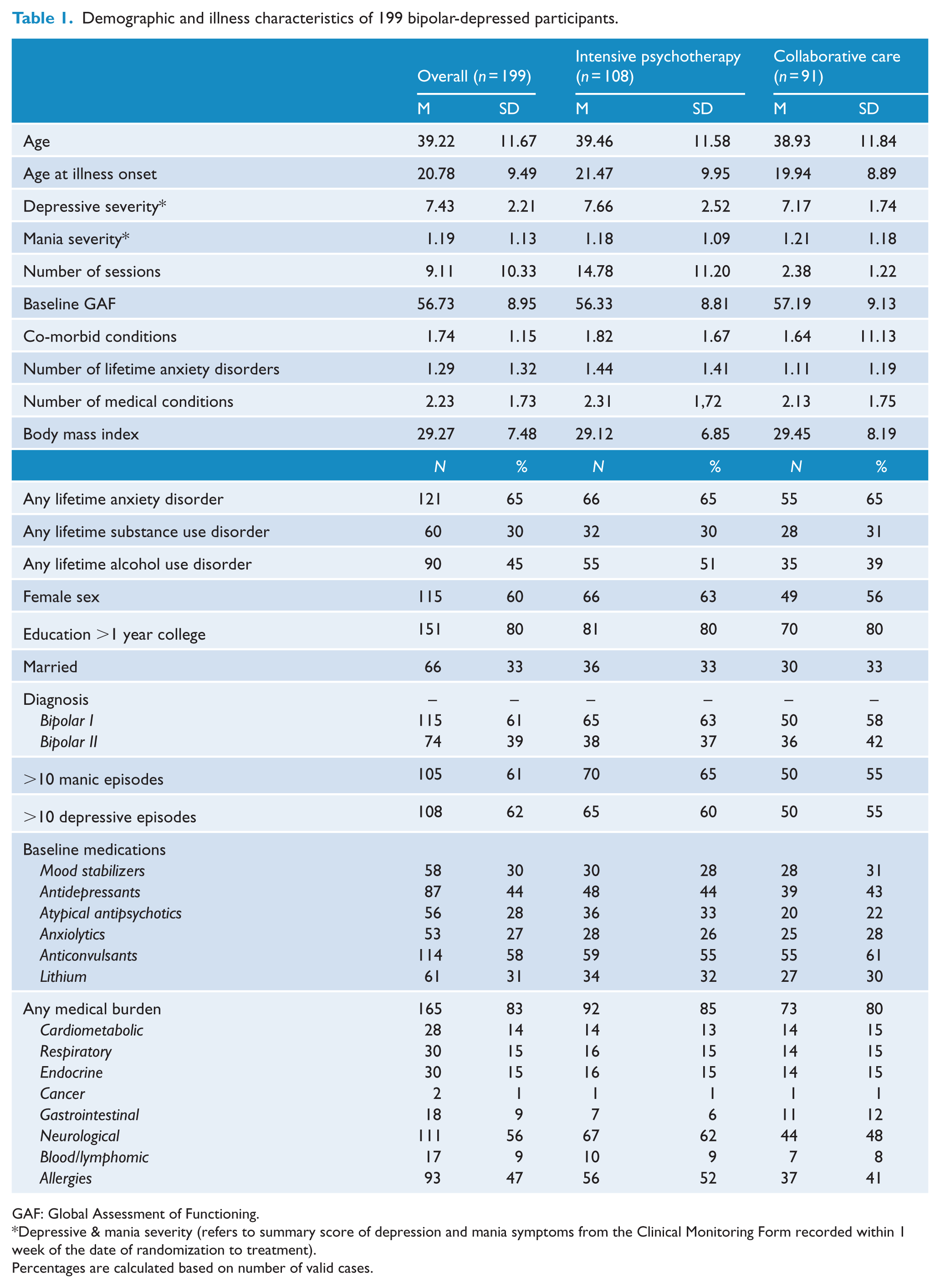
GAF: Global Assessment of Functioning.
Depressive & mania severity (refers to summary score of depression and mania symptoms from the Clinical Monitoring Form recorded within 1 week of the date of randomization to treatment).
Percentages are calculated based on number of valid cases.
Associations between medical burden, BMI and course markers
Table 2 displays the bivariate correlations between medical burden, BMI and markers of illness course that we have previously found to predict or moderate psychosocial treatment outcome in STEP-BD (Deckersbach et al., 2014; Peters et al., 2014; Stange et al., in press). At baseline, medical burden was significantly (p < 0.05) associated having a lifetime history of anxiety disorders (r = 0.34; moderate strength). Medical burden was also significantly, but weakly associated with higher BMI (r = 0.22), number of lifetime anxiety disorders (r = 0.38), number of manic episodes (r = 0.20), illness duration (r = 0.23), older age (r = 0.16) and female sex (r = 0.22). At baseline, BMI was associated with number of manic episodes (r = 0.19).
Associations between medical burden, BMI and course markers.
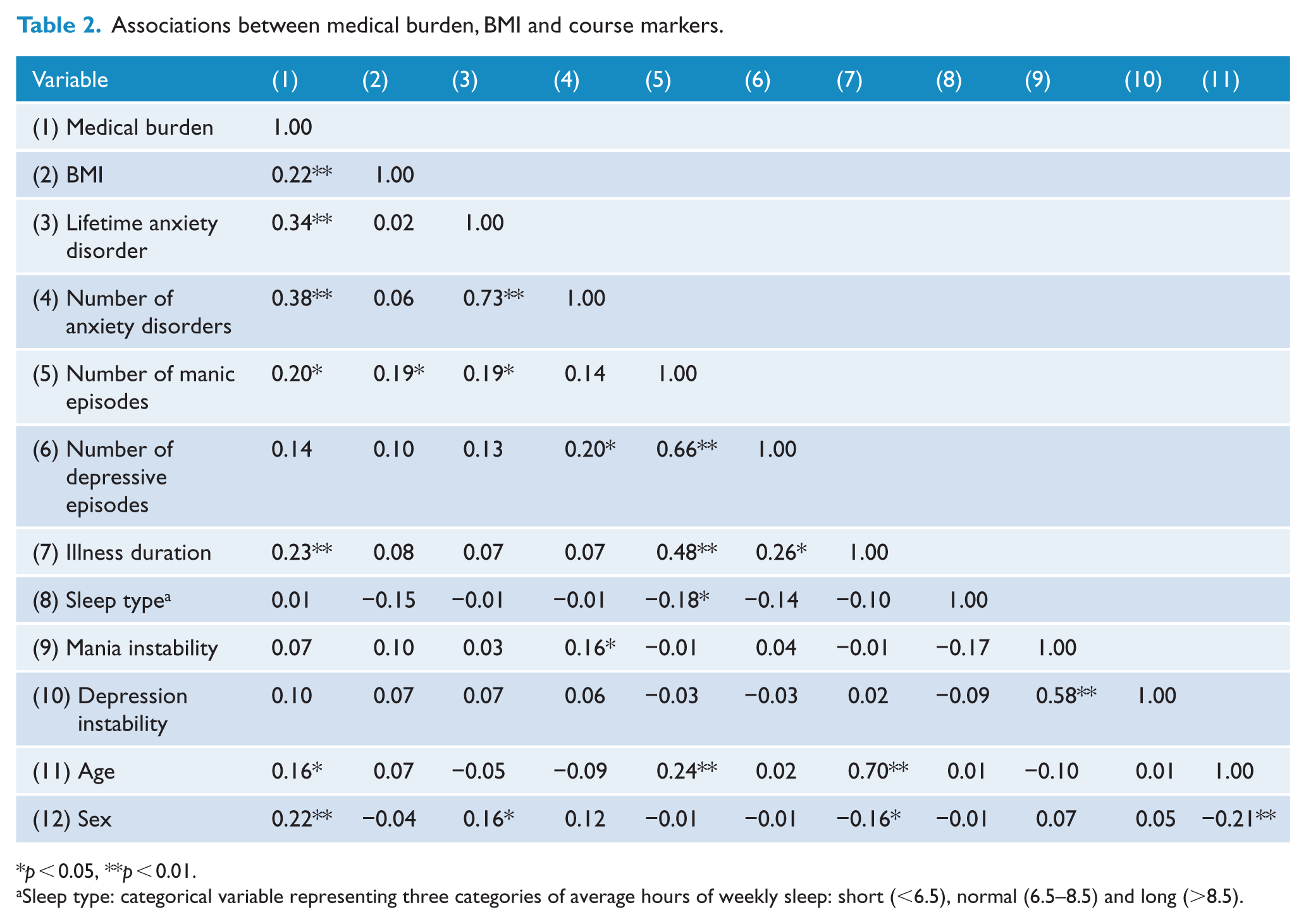
p < 0.05, **p < 0.01.
Sleep type: categorical variable representing three categories of average hours of weekly sleep: short (<6.5), normal (6.5–8.5) and long (>8.5).
Does medical burden or BMI predict treatment outcome?
Cumulative medical burden and BMI were first examined as predictors of recovery independent of treatment assignment (Step 1 of the modeling sequence; see Table 3).
Logistic regression and Cox regression analyses evaluating effects of medical burden and BMI on likelihood of recovery and time until recovery (n = 199).
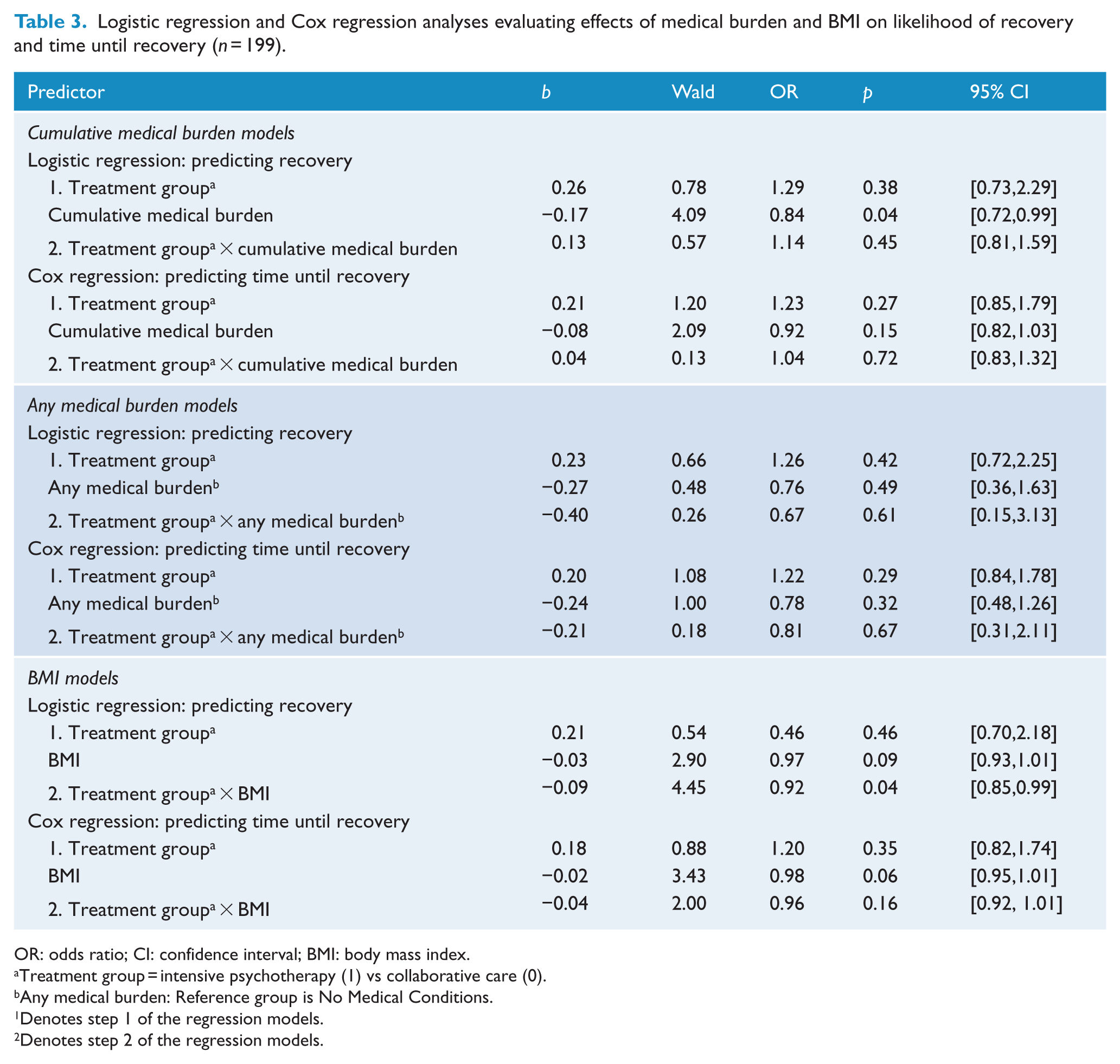
OR: odds ratio; CI: confidence interval; BMI: body mass index.
Treatment group = intensive psychotherapy (1) vs collaborative care (0).
Any medical burden: Reference group is No Medical Conditions.
Denotes step 1 of the regression models.
Denotes step 2 of the regression models.
Cumulative medical burden and treatment outcome
As shown in Table 3, greater cumulative medical burden were associated with a decreased likelihood of recovery from depression (p = 0.04). Specifically, for every additional medical condition, individuals were 0.84 times less likely to recover. The effect of cumulative medical burden on recovery was reduced to non-significance after adjusting for age (p = 0.07), sex (p = 0.06), lifetime anxiety disorder (p = 0.34), number of anxiety disorders (p = 0.46), number of manic episodes (p = 0.18), number of depressive episodes (p = 0.14), illness duration (p = 0.08), sleep type (p = 0.30) or affective instability (p = 0.41). However, the effect size estimates (odds ratios; Table 4) for cumulative medical burden remained almost identical to its uncorrected value (odds ratio, OR = 0.84) despite reductions in sample size due to missing data in the covariate of interest. Cumulative medical burden did not predict time until recovery (OR = 0.92, p = 0.15).
Effect of medical burden and BMI on likelihood of recovery and time until recovery after adjusting for clinical covariates.
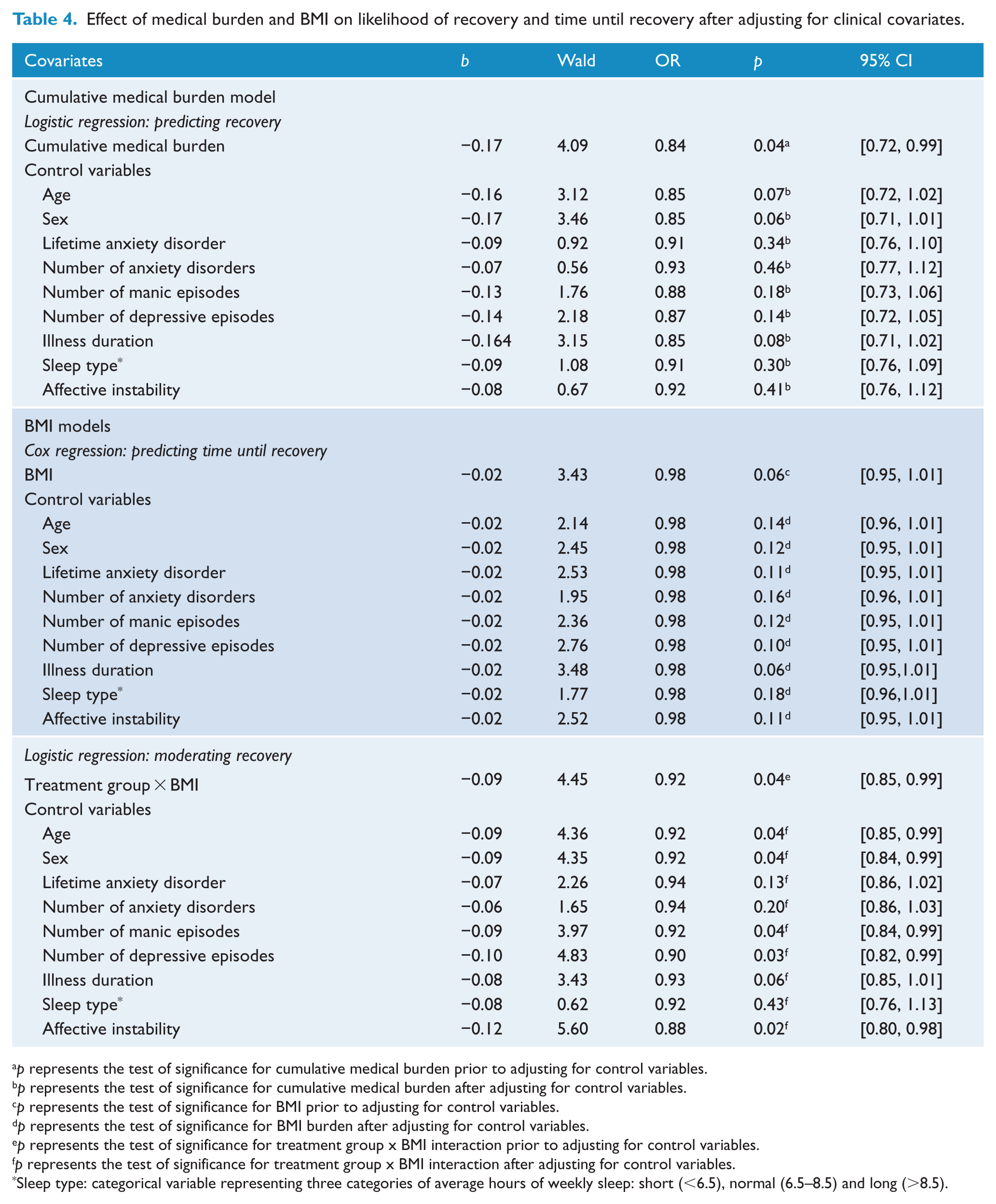
p represents the test of significance for cumulative medical burden prior to adjusting for control variables.
p represents the test of significance for cumulative medical burden after adjusting for control variables.
p represents the test of significance for BMI prior to adjusting for control variables.
p represents the test of significance for BMI burden after adjusting for control variables.
p represents the test of significance for treatment group x BMI interaction prior to adjusting for control variables.
p represents the test of significance for treatment group x BMI interaction after adjusting for control variables.
Sleep type: categorical variable representing three categories of average hours of weekly sleep: short (<6.5), normal (6.5–8.5) and long (>8.5).
BMI
BMI did not predict likelihood of recovery (see Table 3). There was a trend for BMI to predict time to recovery, such that higher BMI was associated with longer time until recovery (OR = 0.98, p = 0.06). This trend for BMI on time to recovery was unaffected by adjusting for illness duration, but was reduced after adjusting for age (p = 0.14), sex (p = 0.12), lifetime anxiety disorder (p = 0.11), number of anxiety disorders (p = 0.16), number of manic episodes (p = 0.12), number of depressive episodes (p = 0.10), sleep type (p = 0.18) and affective instability (p = 0.11). However, the effect size estimates (OR) remained almost identical to the unadjusted OR (0.98), despite reductions in sample size due to missing data in the covariate of interest (see Table 4).
Do medical burden or BMI moderate treatment outcome?
Cumulative medical burden, any medical burden or BMI were next examined as moderators of treatment outcome by including a treatment condition interaction term in the second step of the model. Results of this step of the modeling sequence are also displayed in Table 3.
Neither cumulative medical burden, nor any medical condition interacted with treatment condition to predict likelihood of or time until recovery (see Table 3). However, BMI interacted with treatment condition to predict recovery status (p = 0.04, OR = 0.92; see Table 3). To probe the form of this significant interaction, simple slopes of BMI on recovery were tested at values that are routinely used to characterize how much an individual’s body weight departs from what is normal or desirable for a person of his or her height (Aiken and West, 1991). Participants with a normal range BMI were significantly more likely to recover (OR = 2.39) with intensive psychotherapy than collaborative care, b = 0.87, SE = 0.43, Z(2.04), p = 0.04. In contrast, there was no difference in recovery rates between psychotherapy or collaborative for overweight participants, b = 0.22, SE = 0.29, Z(0.76), p = 0.45 or obese participants, b = −0.43, SE = 0.42, Z(−1.01), p = 0.31.
BMI remained a significant moderator of recovery status—i.e. intensive treatment was associated with more recovery than collaborative care in normal weight individuals—after controlling for age, sex, number of manic episodes number of depressive episodes and affective instability (Table 4). The treatment moderating effect of BMI held up after controlling for number of manic episodes (p = 0.04), number of depressive episodes (p = 0.03) and affective instability (p = 0.02). The moderating effects of BMI were reduced to a statistical trend (p = 0.06) after adjusting for illness duration, and reduced to non-significance after controlling for lifetime anxiety (p = 0.13), number of anxiety disorders (p = 0.20) and sleep type (p = 0.43; Table 4). Although all p were reduced following the inclusion of each covariate, the effect size remained almost identical to the unadjusted omnibus value (OR = 0.92; Table 4). BMI did not moderate the effects of treatment condition on time to recovery (Table 4).
Discussion
Medical burden and obesity have been linked with a more chronic course, longer duration of illness, poorer global functioning and higher rates of disability in bipolar disorder. Our objective was to evaluate whether medical burden or BMI predicted or moderated responses to adjunctive psychotherapy among patients with bipolar I or II depression. Overall, we found that increased cumulative medical burden predicted a lower likelihood of recovery. In addition, there was a trend that increased BMI was associated with longer time until recovery. The predictive value of medical burden was reduced to non-significance after controlling for other predictors and moderators of treatment, but this was due to reduction in sample size or inter-correlation with additional predictors/moderators of treatment outcome. Notably, effect size estimates remained the same after adjusting for these variables. These results are consistent with previous findings that medical co-morbidities pose a barrier to recovery in bipolar disorder and are associated with poor outcomes with pharmacotherapy (Fagiolini et al., 2003; Keck and McElroy, 2003; Kemp et al., 2014; Thompson et al., 2006) and IPSRT (Thompson et al., 2006). Our findings supplement this literature, consistent with Thompson et al.’s (2006) study in IPSRT indicating that recovery from treatment is more difficult when patients have significant medical problems (Thompson et al., 2006).
With regard to the two treatment conditions (intensive psychotherapy vs collaborative care), high medical burden was associated with similar rates of and time to recovery in both treatment conditions. When BMI was examined in relation to treatment condition, we found that individuals with a BMI of less than 25 (normal range) were more likely to recover with intensive psychotherapy compared with collaborative care. In contrast, for individuals with BMI in the overweight or obese categories, there was no difference in between response rates to psychotherapy and collaborative care. This is contrary to our hypothesis that patients with high BMI who were assigned to intensive psychotherapy would have an advantage in recovery. In fact, overweight or obese participants demonstrated comparably low rates of response to both intensive psychotherapy and collaborative care.
There are a wide variety of potential explanations of the association between medical burden/BMI and treatment resistance. Medical burden reduces quality of life and increases stress, and this stress may correspondingly contribute to depression maintenance (Bender and Alloy, 2011). Alternatively, medical burden and BMI may reduce engagement in adaptive responses that promote mood stability. For example, regular exercise and a Mediterranean diet are both associated with lower rates of mood disruption (Sanchez-Villegas et al., 2013; Stathopoulou et al., 2006), a finding also shown in bipolar disorder (Jacka et al., 2011). Furthermore, both obesity and some of the medical co-morbidities have potential inflammatory diatheses, which are linked with increased risk for mood disorders (Berk et al., 2013; Moylan et al., 2014; Sanna et al., 2014), and our cross-sectional data cannot rule out that obesity is not due to the medications used to try to control the bipolar disorder. Obesity is linked to lower levels of physical activity, yet physical activity is demonstrated to have beneficial effects in depression and possibly in bipolar disorder through mechanisms that include reductions in inflammation and increased brain-derived neurotrophic factor (BDNF) (Ng et al., 2007; Thomson et al., 2015). Finally, obesity and poorer health behaviors may be linked to other issues that impact treatment engagement or adherence, such as greater discounting of future rewards in favor of immediate rewards (Bickel et al., 2012; Fields et al., 2013; Jarmolowicz et al., 2014). Indeed, the environmental factors that increase risk for mood disorders such as smoking, poor diet and decreased physical activity are shared risk factors for other common non-communicable disorders such as cardiovascular disease, osteoporosis and diabetes (O’Neil et al., 2015).
Increasing treatment resistance associated with high medical burden and BMI suggests a need for targeted interventions for individuals with medical co-morbidity and weight problems. Interventions that target health risk, obesity and mood might provide the best fit and overall benefit for these patients. For instance, Frank et al. (2014) demonstrated that the combination of psychiatric treatment, medical monitoring and lifestyle coaching resulted in faster reductions in BMI relative to psychiatric care, particularly among those with higher c-reactive protein, total cholesterol and less sleep instability (Frank et al., 2015). In addition, exercise interventions are both feasible and effective in reducing depression (Stathopoulou et al., 2006), and there is increasing evidence that exercise interventions can benefit co-morbid anxiety as well (Asmundson et al., 2013).
In addition to lifestyle management, because weight gain and other adverse metabolic issues in bipolar disorder are often attributed to second-generation antipsychotic medications which are a frequent first-choice treatment for bipolar (Soreca et al., 2009; Sylvia et al., 2015), a key component of more targeted interventions may include patient advocacy for managing complex pharmacological and medical prescriptions and choosing when a more weight-neutral atypical antipsychotic when available. In particular, because complex medication regimens are often managed by multiple providers, one component may be teaching patients tools to help navigate the medical system and facilitate more streamlined care. Further, coping skills for tolerating chronic pain and side effects of medications may help improve quality of life.
This study used a well-characterized sample enrolled in a large clinical trial. It was the first study to investigate the effects of medical burden and obesity on psychotherapy outcomes in bipolar depression. However, analyses were performed post-hoc on a clinical trial that was not originally designed or powered to detect differences in treatment according to medical burden and BMI, or control for other variables that may be linked to these predictors. Data on medical illness and BMI were missing for a portion of the original STEP sample, so it is not possible to determine whether the sample was biased due to data not missing at random. Thus, these findings await replication in future prospective studies that stratify randomization according to medical burden/BMI. In addition, measures of medical burden were completed retrospectively by self-report and did not include information regarding additional medications participants may have been taking for their medical problems. Because this report could not be verified against their medical records, this report may have been subject to recall bias. Further, although BMI is a figure that can be calculated easily, the number only reflects generalized obesity. Because abdominal obesity is one of the most commonly observed parameters of metabolic syndrome in bipolar disorder and is a much stronger predictor of insulin resistance than overall obesity (Kemp et al., 2010), future studies should also examine the role of adiposity distribution in predicting treatment response.
In summary, our results suggest that medical burden and high BMI complicate treatment, not only with pharmacotherapy but also psychotherapy. Contrary to our hypothesis, intensive psychotherapy did not yield better response rates among individuals with high BMI. These results may call for the development of specific psychotherapies that target health management, nutrition and lifestyle in addition to depressive symptoms. Indeed, a recent proof of concept pilot study of a nutrition, exercise and wellness treatment for bipolar disorder yielded improvements in depressive symptoms and functioning (Sylvia et al., 2013). A randomized controlled trial is currently underway.
Footnotes
Declaration of interest
Amy Peters reports no relevant conflicts of interest. Leah W. Shesler reports no relevant conflicts of interest. Louisa Sylvia was employed by the Massachusetts General Hospital, served as a Consultant for Bracket Global and Clintara, received research support from the National Institute of Mental Health (NIMH), is a former stockholder in Concordant Rater Systems, and has received support from New Harbinger Publishers. Pedro Vieira da Silva Magalhães reports no relevant conflicts of interest. David J. Miklowitz has received research support or honoraria from NIMH, Brain and Behavior Research Foundation, Danny Alberts Foundation and Attias Family Foundation. He has received other financial or material support from Guilford Press and John Wiley and Sons. Michael Otto has served as a consultant for MicroTransponder, Inc., receives research support from NIMH and royalties from Oxford University Press and Routledge. Ellen Frank has served as a consultant for Servier International, and has received other financial or material support from Guilford Press and the American Psychological Association Press. Michael Berk is an employee of Barwon Health and Deakin University; he has received research support from the National Institutes of Health (NIH), the National Health and Medical Research Council (NHMRC), the Cooperative Research Centres (CRC), Rotary, Beyond Blue, Stanley Medical Research Institute and the Simons Foundation; he has received honoraria from Lundbeck, Astrazeneca, Servier, Lilly, Janssen, Pfizer, and Merck; he has served as a speaker or on the advisory board for Astrazeneca, Lundbeck, Lilly, and Janssen; and he has received financial or material support from Allen &Unwin and Cambridge University Press. Darin D. Dougherty has served as a consult to Medtronic; has received grant/research support from Medtronic, Eli Lilly and Cyberonics, travel/research support from Roche and has received honoraria from Reed Elsevier. Andrew A. Nierenberg has served as a consultant to Appliance Computing Inc. (Mindsite), BrainCells, Inc., Brandeis University, Bristol-Myers Squibb, Clintara, Dianippon Sumitomo (Now Sunovion), Eli Lilly and Company, EpiQ, Forest, Novartis, PamLabs, PGx Health, Shire, Schering-Plough, Sunovion, Takeda Pharmaceuticals, Teva and Targacept. He has consulted through the Massachusetts General Hospital (MGH) Clinical Trials Network and Institute (CTNI) to Astra Zeneca, Brain Cells, Inc., Dianippon Sumitomo/Sepracor, Johnson and Johnson, Labopharm, Merck, Methylation Science, Novartis, PGx Health, Shire, Schering-Plough, Targacept and Takeda/Lundbeck Pharmaceuticals. Andrew Nierenberg received honoraria or travel expenses including continuing medical examination (CME) activities from the American Professional Society of ADHD and related disorders (APSARD), Belvoir Publishing, Boston Center for the Arts, University of Texas Southwestern Dallas, Hillside Hospital, American Drug Utilization Review, American Society for Clinical Psychopharmacology, Bayamon Region Psychiatric Society, San Juan, PR, Baystate Medical Center, Canadian Psychiatric Association, Columbia University, Douglas Hospital/McGill University, IMEDEX, International Society for Bipolar Disorders, Israel Society for Biological Psychiatry, John Hopkins University, MJ Consulting, New York State, Massachusetts Association of College Counselors, Medscape, MBL Publishing, Physicians Postgraduate Press, Ryan Licht Sang Foundation, Slack Publishing, SUNY Buffalo, University of Florida, University of Miami, University of Wisconsin, University of Pisa and SciMed. Andrew Nierenberg is a presenter for the Massachusetts General Hospital Psychiatry Academy (MGHPA). The education programs conducted by the MGHPA were supported through Independent Medical Education (IME) grants from the following pharmaceutical companies in 2008: Astra Zeneca, Eli Lilly, and Janssen Pharmaceuticals; in 2009 Astra Zeneca, Eli Lilly and Bristol-Myers Squibb. No speaker bureaus or boards since 2003. Andrew Nierenberg owns stock options in Appliance Computing, Inc. and Brain Cells, Inc. Additional income is possible from ![]() depending on overall revenues of the company but no revenue has been received to date. Through MGH, Andrew Nierenberg is named for copyrights to the Clinical Positive Affect Scale and the MGH Structured Clinical Interview for the Montgomery Asberg Depression Scale exclusively licensed to the MGH Clinical Trials Network and Institute (CTNI).He has received grant/research support from the Agency for Healthcare Research and Quality (AHRQ), Cephalon, Forest, Mylin, NIMH, PamLabs, Pfizer Pharmaceuticals, Takeda and Shire. In the next 2 years, it is possible that he will receive grants from Dey Pharmaceuticals, Sunovion and Targacept. ThiloDeckersbach has been funded by the NIMH, the National Alliance for Research on Schizophrenia and Depression (NARSAD), the Tourettes Syndrome Association (TSA), the International Obsessive Compulsive Disorder Foundation (OCF) and Tufts University. He has received honoraria, consultation fees and/or royalties from the MGH Psychiatry Academy, BrainCells Inc., Systems Research and Applications Corporation, Boston University, the Catalan.
depending on overall revenues of the company but no revenue has been received to date. Through MGH, Andrew Nierenberg is named for copyrights to the Clinical Positive Affect Scale and the MGH Structured Clinical Interview for the Montgomery Asberg Depression Scale exclusively licensed to the MGH Clinical Trials Network and Institute (CTNI).He has received grant/research support from the Agency for Healthcare Research and Quality (AHRQ), Cephalon, Forest, Mylin, NIMH, PamLabs, Pfizer Pharmaceuticals, Takeda and Shire. In the next 2 years, it is possible that he will receive grants from Dey Pharmaceuticals, Sunovion and Targacept. ThiloDeckersbach has been funded by the NIMH, the National Alliance for Research on Schizophrenia and Depression (NARSAD), the Tourettes Syndrome Association (TSA), the International Obsessive Compulsive Disorder Foundation (OCF) and Tufts University. He has received honoraria, consultation fees and/or royalties from the MGH Psychiatry Academy, BrainCells Inc., Systems Research and Applications Corporation, Boston University, the Catalan.
Funding
STEP-BD was funded in part by contract N01MH80001 from the National Institute of Mental Health (Gary Sachs). Support for the development of the psychosocial treatments was provided by grants MH29618 (Ellen Frank), MH43931 (David Miklowitz) and MH55101 (David Miklowitz) from the National Institute of Mental Health and by the National Alliance for Research on Schizophrenia and Depression (David Miklowitz).
