Abstract
Keywords
The metabolic syndrome is a constellation of interrelated metabolic disturbances including central obesity, insulin resistance, hypertension and dyslipidaemia, that is associated with a significantly increased risk of type 2 diabetes and cardiovascular disease. The central unifying feature of this syndrome is insulin resistance, of which obesity is a significant determinant [1]. Adipose tissue secretes a number of bioactive substances (adipocytokines), which appear to mediate the development of coronary heart disease through neuroendocrine dysregulation and low-grade inflammation. Adiponectin, the most abundant and tissue specific of the adipocytokines, has been found to inhibit inflammatory reactions and protect against metabolic and cardiovascular disease. Low plasma levels of adiponectin, which are found in obese patients, are closely associated with obesity-related diseases, including the metabolic syndrome [2–4].
The prevalence of the metabolic syndrome is highly variable within and between populations and rises significantly with increasing age and obesity, particularly central obesity [5]. In 2002 the age-adjusted prevalence in the US adult population was estimated to be approximately 24% [6]. A similar prevalence of 23% has been described recently in adult New Zealand Caucasian subjects. [7]. The reported prevalence of the metabolic syndrome is higher in bipolar patients than in the general population [8], and rates of diabetes and cardiovascular disease are correspondingly increased, contributing to significantly poorer treatment outcomes, increased burden of disease and higher mortality rates in these patients [9, 10]. Estimates of the relative frequency of the metabolic syndrome, however, may be confounded by the use of selected samples of hospitalized bipolar patients who are unrepresentative of those living in the community [11], and the use of normative population data unadjusted for age and body mass index (BMI).
The high risk of the metabolic syndrome in bipolar patients may simply be a function of lifestyle factors, particularly low dietary fibre intake, low physical activity level and smoking [12, 13]. Psychotropic medication may also exacerbate this risk [14, 15], with the common end-point being obesity. There may be a pre-existing vulnerability, however, to disturbances of glucose homeostasis in patients suffering from bipolar disorder [16].
Fasting hyperinsulinaemia has been found in patients taking sodium valproate [17] but whether this is an independent effect of sodium valproate or simply a consequence of the associated weight gain has not been satisfactorily resolved [18]. Mean insulin and glucose levels during an oral glucose tolerance test were reduced after 1 month on sodium valproate, indicating an improvement in insulin sensitivity [19], but a more recent study described insulin concentrations increasing in epileptic patients taking valproate, irrespective of weight gain [20]. A similar debate exists regarding treatment with atypical antipsychotic medications [21].
We hypothesized that because of a combination of pre-existing vulnerability in bipolar disorder and treatment with sodium valproate, insulin resistance measured using HOMA-IR and the metabolic syndrome assessed according to Adult Treatment Panel III (ATP III) criteria would be more common in bipolar disorder than in subjects matched for age, gender, BMI and ethnicity.
Methods
The participants were overweight bipolar patients who had volunteered to take part in a weight reduction study [22] and overweight volunteers without psychiatric illness who were recruited from the general population by newspaper advertisement [23]. Bipolar patients who met the DSM-IV criteria for bipolar disorder and who had been taking sodium valproate for ≥6 months, with clinically significant weight gain that appeared to be related to valproate treatment, were eligible for the study. All participants had a BMI > 25 kg m–2. Sixty bipolar patients and 60 controls matched for age, gender, BMI and ethnicity were included in the study. Patients and control subjects taking insulin, oral hypoglycaemic agents, anti-obesity medication or with serious pre-existing cardiovascular disease were excluded. The study was approved by the Canterbury Ethics Committee.
All the bipolar patients were assessed using the Structured Clinical Interview for DSM-IV [24], the Young Mania Rating Scale [25]and the Montgomery–Asberg Depression Rating Scale [26].
Anthropometric and biochemical measurements were carried out after an overnight fast. Height was measured to the nearest millimetre with the subjects in bare feet. The subjects were weighed in light indoor clothing with bare feet on an electronic scale (Model TBF-501; Tanita, Arlington Heights, IL, USA). Waist measurements were made at the minimum circumference between the rib cage and the iliac crest. Blood pressure was measured with a digital blood pressure monitor (IBS Model SD-700A; IBS, Waltham, MA, USA) in patients, and in controls (Omron 70JCP; Omron Matsusaka, Mie-Ken, Japan).
Plasma glucose, total cholesterol, high-density lipoprotein cholesterol (HDL-C) and triglyceride levels were measured using an Aeroset analyser (Abbott Laboratories, Abbott Park, IL, USA) and plasma insulin using electrochemiluminescent immunoassay (Roche Diagnostics, Indianapolis, IN, USA). Plasma low-density lipoprotein cholesterol (LDL-C) levels were calculated using the Friedewald equation [27], and insulin resistance determined by the homeostatic model assessment using the formula: IR=(insulin×glucose)/22.5 (HOMA-IR) [28]. Plasma adiponectin was measured in duplicate on radioimmunoassay (Linco Research, St Charles, MO, USA). The metabolic syndrome was defined using ATP III criteria [29].
The data were analysed using SPSS for Windows version 13 (SPSS, Chicago, IL, USA). Unpaired t-tests were used to compare continuous variables between groups. Non-parametric data were analysed with the Mann–Whitney U-test or the Kruskal–Wallis test. The χ2 test or, for data for which the expected values were <5, Fisher's exact test, were used to compare categorical variables between groups. Results were considered significant at two-tailed p < 0.05.
Results
Details of the patient group have been published previously [22]. The majority of patients had bipolar 1 disorder and all were taking sodium valproate. The mean age at onset of illness (first manic episode) was 26±10 years. The majority of patients (n = 37) had been taking sodium valproate for >1 year (range = 6 months– > 5 years) and the mean plasma valproate level was 470±158 µmol L−1. The mean weight gain that patients attributed to taking sodium valproate was 15±10kg. Adiponectin results were available for 53 patients and 60 controls.
The demographic and clinical characteristics of the patients and controls are shown in Table 1. There were no significant differences between the groups for age, gender, BMI, and ethnicity. Differences in the numbers in each group taking antihypertensive or lipid lowering medication were not statistically significant, but more patients than controls were smokers (p = 0.04).
Subject characteristics
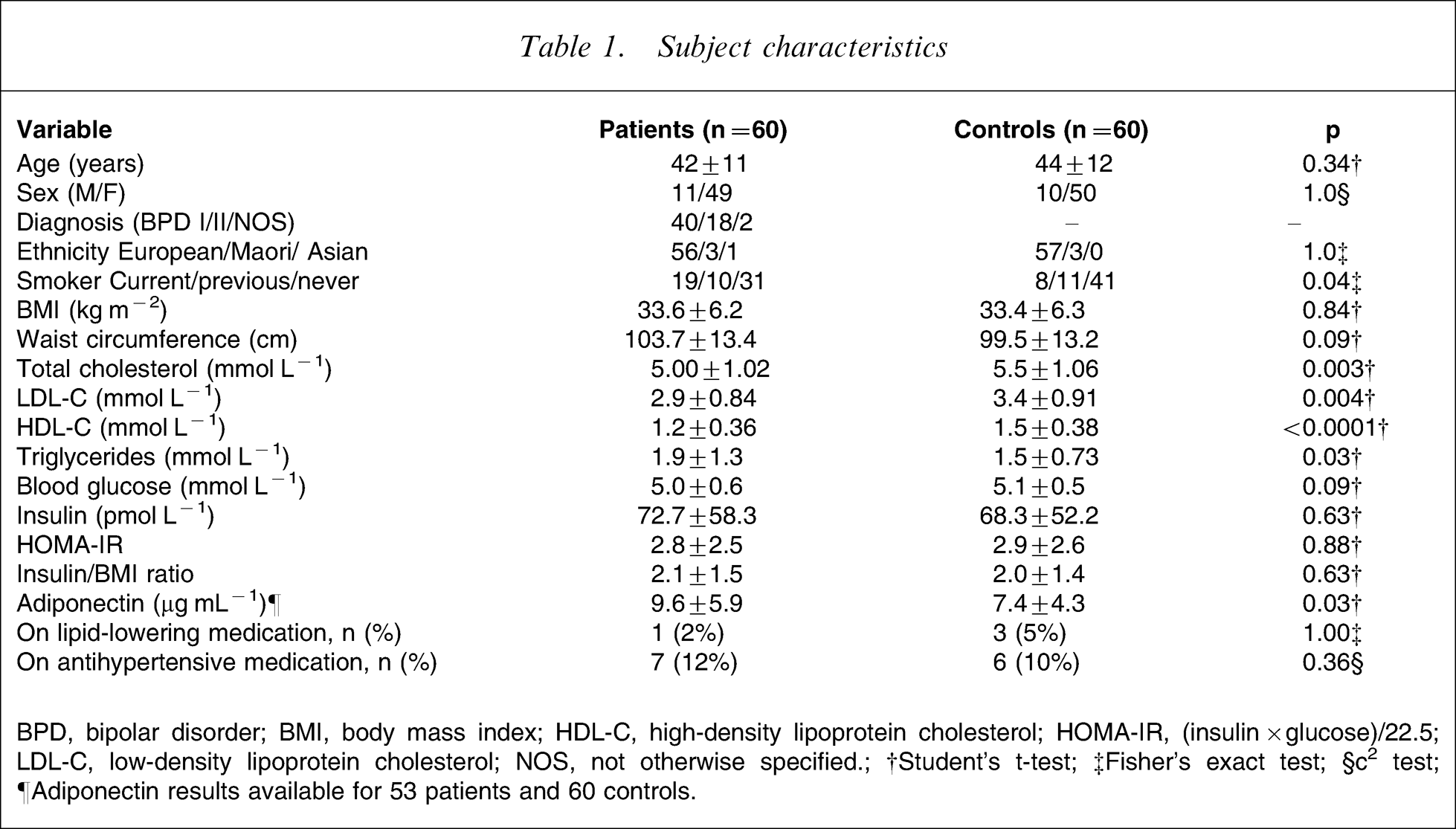
BPD, bipolar disorder; BMI, body mass index; HDL-C, high-density lipoprotein cholesterol; HOMA-IR, (insulin×glucose)/22.5; LDL-C, low-density lipoprotein cholesterol; NOS, not otherwise specified.; †Student's t-test; ‡Fisher's exact test; §c2 test.; ¶Adiponectin results available for 53 patients and 60 controls
The frequency of the metabolic syndrome was slightly higher in patients compared to controls (50% vs 32%, respectively), but this did not reach statistical significance (p = 0.06, Table 2). The total number of ATP III criteria, however, was higher in patients. Similar proportions of patients and controls met ATP III criteria for waist circumference, hypertriglyceridaemia and hypertension, whereas significantly more patients met the criterion for low HDL-C (p = 0.006, Table 2). The proportions of patients with BMI ≥30, insulin resistance (HOMA-IR ≥2.5) and fulfilling ≥3 ATP III criteria for the metabolic syndrome were not significantly different in patients taking atypical antipsychotic medication (Table 3) compared to patients not taking this medication.
Frequency of insulin resistance and metabolic syndrome
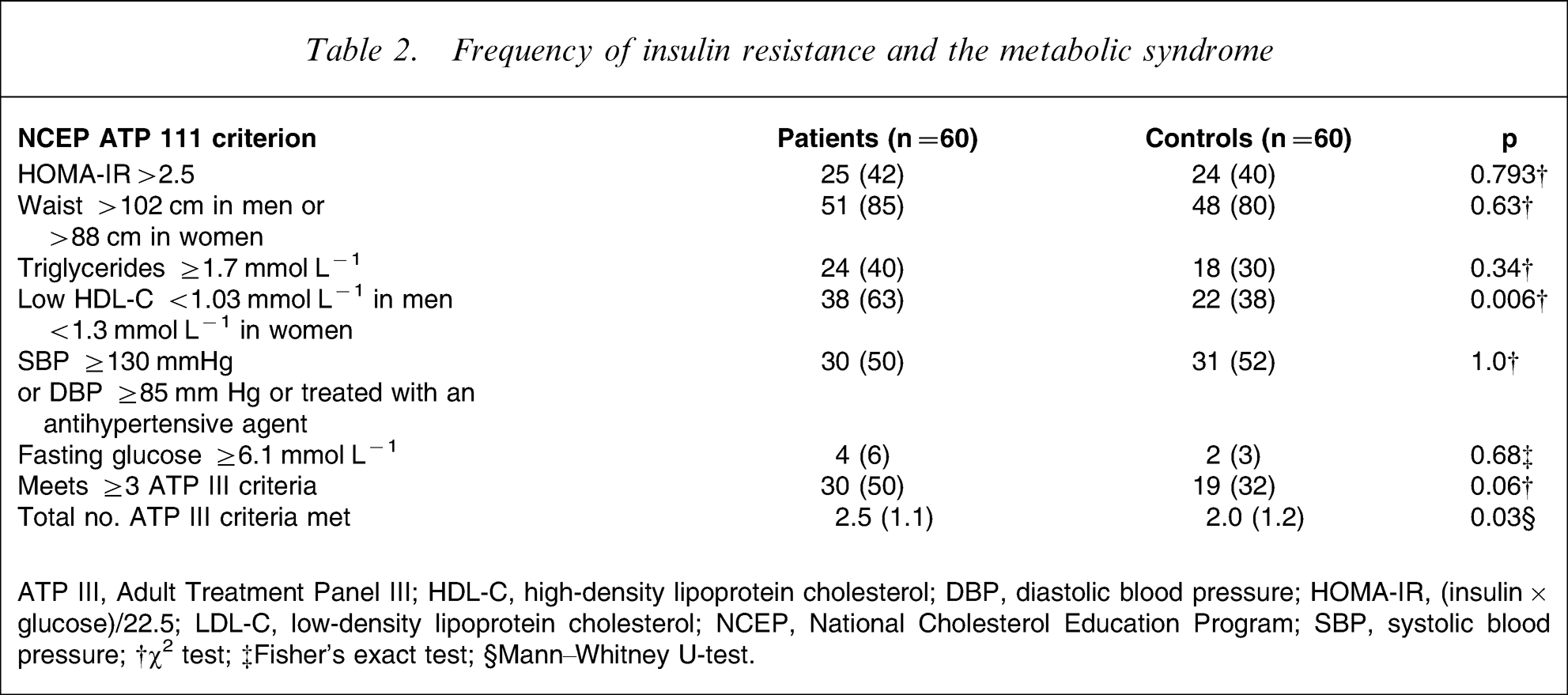
ATP III, Adult Treatment Panel III; HDL-C, high-density lipoprotein cholesterol; DBP, diastolic blood pressure; HOMA-IR, (insulin×glucose)/22.5; LDL-C, low-density lipoprotein cholesterol; NCEP, National Cholesterol Education Program; SBP, systolic blood pressure; †χ2 test; ‡Fisher's exact test; §Mann–Whitney U-test.
BMI, insulin resistance and the metabolic syndrome characteristics in patients vs presence of atypical antipsychotic
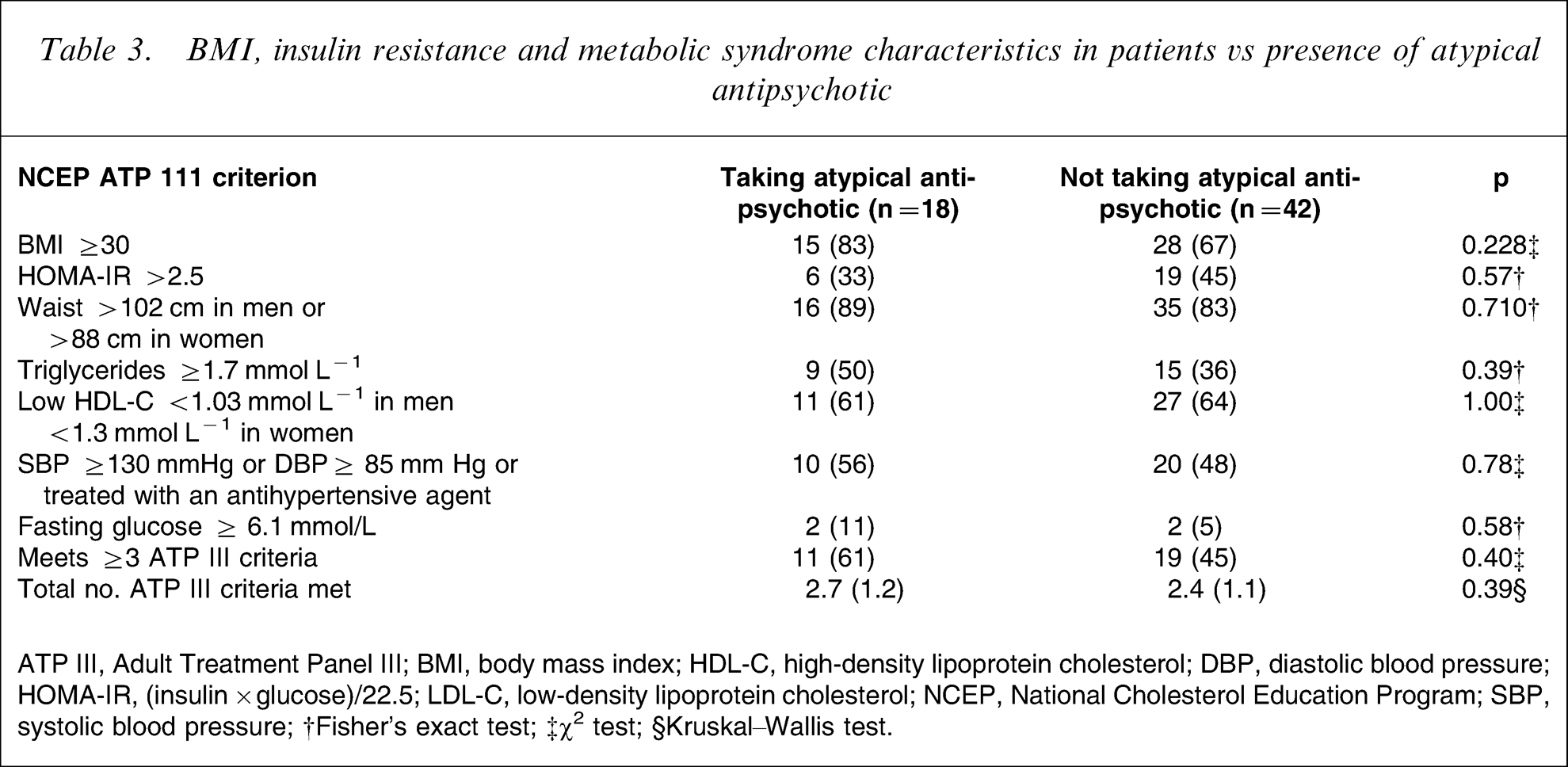
ATP III, Adult Treatment Panel III; BMI, body mass index; HDL-C, high-density lipoprotein cholesterol; DBP, diastolic blood pressure; HOMA-IR, (insulin×glucose)/22.5; LDL-C, low-density lipoprotein cholesterol; NCEP, National Cholesterol Education Program; SBP, systolic blood pressure; †Fisher's exact test; ‡χ2 test; §Kruskal–Wallis test.
Biochemical results for the two groups are shown in Table 1. Total cholesterol, LDL-C and HDL-C were all significantly lower in the bipolar patients, while triglycerides and adiponectin were higher. There were no between-group differences in waist circumference, fasting plasma glucose, insulin, insulin/BMI ratio and HOMA-IR.
Discussion
To our knowledge this is the first study to compare the components of the metabolic syndrome, insulin resistance and adiponectin levels in a group of overweight bipolar patients taking sodium valproate and members of the general population matched for age, gender, BMI and ethnicity. The aim was to examine whether metabolic abnormalities in this group are primarily related to bodyweight or whether other independent factors are involved. The frequency of the metabolic syndrome was not significantly different between bipolar patients and a well-matched control group. The null results were not explained by group differences in gender, age, BMI or ethnicity, and show that once the confounding effects of BMI, age and gender are taken into account, the frequency of the metabolic syndrome is not significantly different between bipolar patients taking sodium valproate and the general population. Differences, however, were seen for some metabolic parameters, in that total cholesterol, LDL-C and HDL-C were significantly lower in patients, and triglyceride and adiponectin levels significantly higher.
We expected a higher prevalence of the metabolic syndrome in the present patients based on a number of crucial differences between the patients and controls. First, there is a documented increased risk of the metabolic syndrome in patients with bipolar disorder. Second, all the patients in the present study were taking sodium valproate, which is known to affect plasma insulin concentrations. Hyperinsulinaemia has previously been described with valproate treatment [14] but decreased insulin sensitivity has not been demonstrated consistently [20, 30]. Increases in both basal and post-prandial insulin concentrations have been reported, together with increased appetite and weight gain [17, 31]. In the present study, mean fasting insulin concentrations in both groups were near the upper end of the normal range established by Canterbury Health Laboratories (10–80 pmol L−1), as would be expected given the BMI, waist circumference and age of the study participants. This finding is consistent with that of Verrotti et al., who reported that serum insulin increased after 1 year on valproate only in patients who gained weight [32, 33]. In contrast, Pylvanen et al. found that the circulating insulin/BMI ratio was greater in patients on valproate monotherapy than in controls and was highest in those who had been on the medication the longest [17]. These findings were interpreted by the authors as evidence of valproate-induced hyperinsulinaemia leading to weight gain. In the present study the mean insulin/BMI ratio was not significantly different between patients and controls and there was no association between the duration of treatment with sodium valproate and the insulin/BMI ratio in patients. The present two study groups were well matched for key variables known to contribute to the development of insulin resistance (age, BMI, and ethnicity) and there were no significant differences between the groups in fasting insulin or HOMA-IR. Taken together, these results suggest that obesity is a cause but not a consequence of increased serum insulin in valproate-treated patients. A prospective study design, however, would be better able to answer this question definitively. Third, as in previous studies in psychiatric population samples, smoking was more prevalent in the patients [34]. In the general population, smoking is associated with a number of features of the metabolic syndrome including truncal obesity (which is more pronounced in heavier smokers), insulin resistance, raised triglycerides, low HDL-C and raised fasting plasma glucose [35–37]. Also, adiponectin levels are usually lower in smokers than in non-smokers [38]. Despite, however, the significantly greater number of smokers among patients in the study, the frequency of the metabolic syndrome was similar in both groups and adiponectin levels were higher in patients. Fourth, some of the patients in the present study were taking atypical antipsychotic agents. Clozapine, olanzapine, risperidone and quetiapine have been found to significantly increase the risk of treatment-emergent type 2 diabetes and the metabolic syndrome in psychiatric patients [21, 39, 40]. In the present study 18 patients were taking an atypical antipsychotic: five were on olanzapine and 13 were on risperidone or quetiapine. The number of patients fulfilling ≥3 ATP III criteria for the metabolic syndrome was not significantly higher in those taking atypical antipsychotic medication compared to those not on these medications, but because the group of patients taking atypical antipsychotics was small, further conclusions cannot be drawn. Once again it may simply be that the increased frequency of features of the metabolic syndrome in those taking atypical antipsychotics is purely a function of weight gain.
There were significant differences between patients and controls in cholesterol, triglycerides and adiponectin. These may reflect underlying biological differences related to bipolar disorder itself and/or its treatment, or differences in lifestyle secondary to ongoing symptoms of depression. Sedentary lifestyles and high sucrose and alcohol intake increase the risk of dyslipidaemia [41, 42], and bipolar patients’ lifestyles may make them particularly vulnerable to this metabolic disorder [12, 43]. The contribution, however, of these variables to the present results cannot be quantified because of the lack of dietary and physical activity data for the control group.
Furthermore, the differences in metabolic parameters may directly relate to taking sodium valproate. In several previous studies valproate treatment has been associated with low total and HDL cholesterol, and elevated triglyceride levels [44–46], supporting the present results. In contrast high adiponectin levels are usually associated with a less atherogenic lipid profile. The significantly higher adiponectin levels, however, in the present patients did not protect them from this adverse effect. This finding may be especially important because a recent study has suggested that dyslipidaemia is more strongly predictive of future cardiovascular disease than other features of the metabolic syndrome [47]. Those authors found that overweight people with dyslipidaemia (plasma triglyceride > 1.7 mmol L−1, HDL < 1.03 mmol L−1 in men and 1.3 mmol L−1 in women) were more likely to have a generalized metabolic disorder than people of comparable BMI with normal plasma lipid levels. In the present study dyslipidaemia was more common in patients, suggesting that the risk of developing cardiovascular disease may be greater in overweight bipolar patients taking sodium valproate than in a general population control group with similar BMI.
Adiponectin is produced in large quantities by adipocytes, with circulating levels being inversely correlated with obesity, insulin resistance and dyslipidaemia [47, 48]. The literature concerning psychiatric illness, psychotropic medication and adiponectin levels is conflicting. This may relate to methodological differences between studies as well as genetic differences in study populations [49]. In cross-sectional studies, increased plasma adiponectin levels have been reported in patients with major depression controlled on maintenance anti-depressant therapy compared to normal subjects [50], while lower levels have been reported in drug-free schizophrenia patients [51] and patients taking olanzapine and risperidone [52, 53]. In a prospective study, however, in rats given olanzapine for 3 weeks, adiponectin levels rose during the observation period [54]. Adiponectin levels in bipolar patients have not been reported previously. In the present study, adiponectin levels were higher in patients than in a well-matched control group despite comparable BMI and waist circumference. The present findings are in contrast to a study in epileptic patients in which BMI, waist–hip ratio, plasma glucose, leptin, insulin, adiponectin and grehlin were measured before starting valproate monotherapy and after 2 years on the medication [55]. That study included a control group matched for age and BMI. Fifteen patients became obese during the follow-up period and BMI increased slightly but not significantly in controls. Adiponectin and grehlin levels fell significantly in those patients who became obese. The authors concluded that the decrease in adiponectin was the result of negative feedback inhibition as a consequence of increased fat mass; in other words, the weight gain caused the decrease in adiponectin. Cell culture studies have shown that valproate reduces the expression of the adiponectin gene [56]. The reasons for the higher adiponectin concentrations observed in the present patients are unclear. Although recent studies suggest that this difference is unlikely to be pharmacologically mediated [55, 56] further research in this area is clearly warranted.
Limitations
This study had several limitations, including the cross-sectional study design, limited sample size and lack of a comparison group of bipolar patients not on sodium valproate. The power of the study is also reduced by the heterogeneity among the patients with regard to antipsychotic drugs. Another important limitation is the lack of information on dietary intake, physical activity and substance abuse in control subjects. Despite these limitations the study provides useful information about the relative rates of insulin resistance and the metabolic syndrome in valproate-treated bipolar patients and the general population.
Conclusion
The metabolic abnormalities seen in the present overweight valproate-treated patients with bipolar disorder differ from the abnormalities seen in obesity in the general population. The frequencies of insulin resistance (HOMA-IR) and the metabolic syndrome in bipolar patients taking sodium valproate and general population controls matched for age, gender and BMI were not significantly different, whereas dyslipidaemia was significantly more common in patients. Plasma adiponectin levels were higher in bipolar patients taking sodium valproate despite similar anthropometry and degree of insulin resistance in controls. This finding suggests that adiponectin does not protect against the development of the metabolic abnormalities observed in the bipolar patients, and further studies are required concerning the association between adiponectin and cardiometabolic risk in these patients. To more clearly elucidate predictors of the metabolic syndrome and potential treatment targets in bipolar patients, future research will need to consider the role of modifiable environmental factors such as habitual dietary intake, physical activity levels and substance use in addition to illness-related factors such as age of onset, severity and medication history.
Footnotes
Acknowledgements
This project was funded by grants from the Stanley Medical Research Institute and the New Zealand National Heart Foundation. The authors would like to acknowledge research assistance from Andrea Bartram. Professor Peter Joyce has received payment for conference presentations and participation on a Sanofi-Synthelabo Advisory Board. Dr Elmslie and Professor Joyce and Associate Professor Richard Porter have received funding assistance from Sanofi-Synthelabo for conference attendance and honoraria from Janssen-Cilag and Eli Lilly. Dr Hunt has received funding assistance for conference participation from Novartis and Pfizer.
