Abstract
Aims:
We aimed to explore the relationships between diet quality, dietary inflammatory potential or body mass index and outcomes of a clinical trial of nutraceutical treatment for bipolar depression.
Methods:
This is a sub-study of a randomised controlled trial of participants with bipolar depression who provided dietary intake data (n = 133). Participants received 16 weeks adjunctive treatment of either placebo or N-acetylcysteine-alone or a combination of mitochondrial-enhancing nutraceuticals including N-acetylcysteine (combination treatment). Participants were followed up 4 weeks post-treatment discontinuation (Week 20). Diet was assessed by the Cancer Council Victoria Dietary Questionnaire for Epidemiological Studies, Version 2, converted into an Australian Recommended Food Score to measure diet quality, and energy-adjusted dietary inflammatory index score to measure inflammatory potential of diet. Body mass index was also measured. Generalised estimating equation models were used to assess whether diet quality, energy-adjusted dietary inflammatory index score and/or body mass index were predictors of response to significant outcomes of the primary trial: depression symptoms, clinician-rated improvement and functioning measures.
Results:
In participants taking combination treatment compared to placebo, change in depression scores was not predicted by Australian Recommended Food Score, dietary inflammatory index or body mass index scores. However, participants with better diet quality (Australian Recommended Food Score) reported reduced general depression and bipolar depression symptoms (p = 0.01 and p = 0.03, respectively) and greater clinician-rated improvement (p = 0.02) irrespective of treatment and time. Participants who had a more anti-inflammatory dietary inflammatory index had less impairment in functioning (p = 0.01). Combination treatment may attenuate the adverse effects of pro-inflammatory diet (p = 0.03) on functioning. Participants with lower body mass index who received combination treatment (p = 0.02) or N-acetylcysteine (p = 0.02) showed greater clinician-rated improvement.
Conclusion:
These data support a possible association between diet (quality and inflammatory potential), body mass index and response to treatment for bipolar depression in the context of a nutraceutical trial. The results should be interpreted cautiously because of limitations, including numerous null findings, modest sample size and being secondary analyses.
Introduction
The depressive phase of bipolar disorder (BD) is particularly challenging to treat, with conventional treatments more effective for the management of manic symptoms (Malhi et al., 2015). Polypharmacy is common, in part due to the gap in recovery (Fountoulakis, 2010), considerable inter-individual variability and the need to treat two illness phases (Malhi et al., 2015). The common pharmacological treatments (mood stabilisers, antipsychotics and antidepressants) have side-effects, including increased risk of switching to the manic phase (due to antidepressants) and metabolic disorders (Fountoulakis, 2010; Malhi et al., 2015). The lack of recovery and the associated side-effect risk drive the need to explore new treatment avenues to reduce symptoms and improve quality of life in people with BD.
The current understanding of the pathophysiology of BD includes alterations in neurotransmitters (such as glutamate, dopamine and serotonin), increased oxidative stress, increased inflammation (Data-Franco et al., 2017; Grande et al., 2016) and mitochondrial disturbances (Stork and Renshaw, 2005). The latter may be biphasic, with increased biogenesis in mania and decreased energy generation in depression; this disturbance theoretically can be targeted pharmacologically, suggesting the possibility of treating the disorder with mitochondrial-enhancing nutraceuticals (Pereira et al., 2018). In addition, these factors might also be modified by variables such as diet quality (Sears, 2015) and body composition (Trayhurn, 2005).
Research suggests there is a bidirectional relationship between diet quality and depression. Extensive research has linked both poor diet quality and the inflammatory potential of diet to the risk for unipolar depression (Kheirouri and Alizadeh, 2019; Lassale et al., 2019; Wang et al., 2018). Diet quality is worse in people with depressive and anxiety disorders and is linked to both chronicity and severity in a dose–response manner (Gibson-Smith et al., 2018). There are also data showing poorer dietary patterns in participants with BD when compared to a sample without serious mental illness (Kilbourne et al., 2007). A study of dietary styles in an Australian sample of adult women (n = 691) found that participants with BD (n = 23) were more likely to report eating processed, nutrient-poor ‘Western’ foods compared to those who did not screen positive for a mental illness (Jacka et al., 2011). In this study, a traditional diet was deemed to be the ‘healthiest’, replete with vegetables and whole grains.
There is some evidence suggesting that modifying diet may impact mental health in clinical groups (Jacka et al., 2017; Parletta et al., 2017). A new meta-analysis supports dietary change as an efficacious treatment for depressive symptoms (Firth et al., 2019). In addition, a small proof-of-concept, open-label trial (n = 5) demonstrated the possibility that a combination of nutritional advice (encouraging a balanced diet while reducing caloric intake), exercise and general well-being might improve depression symptoms and functioning in participants with BD (Sylvia et al., 2013). While research into associations between unipolar depression and diet quality is growing, the association between diet and BD remains relatively underexplored.
Given the increasing evidence of links between diet quality and mental health, it is plausible that diet quality may influence how people with existing mental health symptoms respond to treatments. While there are various ways of describing and assessing diets, current Australian recommendations for a healthy diet are to eat a range of foods, especially fruit and vegetables, which are naturally high in fibre and low in saturated fats, and reduce consumption of sugars and alcohol (NHMRC, 2013). It is also possible to categorise dietary intake according to inflammatory potential (Shivappa et al., 2014b). For example, nutrients such as omega-3 fatty acids and polyphenols have anti-inflammatory potential (Sears, 2015). Increased inflammation has been associated with higher occurrence of treatment resistance to antidepressants in major depressive disorder (Haroon et al., 2018).
Body mass index (BMI), a crude indicator of body composition, is often higher in those with BD (Goldstein et al., 2011) and has been found to have an impact on treatment response (Kemp et al., 2010). For every increase in one unit of BMI, participants with BD showed 7.5% worse outcomes in response to lithium or valproate treatment (Kemp et al., 2010). Furthermore, higher BMI in participants with BD is associated with greater levels of hippocampal glutamate (Bond et al., 2016). One study (n = 121) also has suggested that participants with unipolar depression and lower baseline interleukin-6 inflammatory cytokine levels showed a greater reduction in depression symptoms across the study regardless of treatment received in the randomised controlled trial (RCT; Hasebe et al., 2017).
In depression intervention studies, there is some evidence to suggest that participants with higher cortisol, oxidative stress and inflammatory marker concentrations have poorer response to treatment (Carvalho et al., 2013; Lindqvist et al., 2017; Pisoni et al., 2018) including psychological therapy (Fischer et al., 2017). Contrary to this, there is some evidence to suggest that participants with both BD and a systemic illness respond better to N-acetylcysteine (NAC) treatment than participants without comorbid physical illness (Magalhães et al., 2012). In addition, participants aged >60 years with a higher cardiometabolic risk demonstrated better response to treatment for bipolar depression (Gildengers et al., 2012). Given there is inconsistent response to treatment for BD when taking into consideration the diet and body composition of an individual, and that diet can modulate similar pathophysiology as some nutraceuticals, it is important to explore how dietary and BMI factors may contribute to outcomes in a nutraceutical clinical trial.
The purpose of our study was to explore whether diet, BMI or a combination of the two may play a role in treatment response in a RCT of adjunctive nutraceuticals for bipolar depression. We focused on both diet according to the Australian Dietary Guidelines (Collins et al., 2008; NHMRC, 2013) and diet in terms of its inflammatory potential (Shivappa et al., 2014a, 2014b). The possible effect of diet and BMI indices capturing quality and inflammatory potential was analysed against the following significant outcomes of the primary trial at Week 20: depression symptom severity scores, bipolar depression rating scores, social and occupational functioning, functional impairment and clinician-rated improvement. We aimed to explore whether diet quality, energy-adjusted dietary inflammatory index (E-DII™) scores and BMI were effect modifiers for the significant relationship between treatment received and outcomes in the participants of the RCT. We hypothesised that participants’ BMI and diet, both in terms of overall quality and inflammatory potential, would be associated with their response to treatments (as assessed by change scores) received in the trial (NAC alone or a combination treatment of mitochondrial-enhancing nutraceuticals including NAC [CT]).
Methods
Trial study design
The current study utilised outcome data from a multi-site, double-blind placebo-controlled RCT. The primary study recruited a total of 181 participants with BD who were experiencing a current moderate-to-severe depressive episode as assessed by the Mini International Neuropsychiatric Interview (MINI) 5.0 (Sheehan et al., 1998) and a score ⩾20 on the Montgomery–Åsberg Depression Rating Scale (MADRS; Dean et al., 2015; Montgomery and Åsberg, 1979; ANZCTR registry: ACTRN12612000830897). Participants were randomised to receive either a CT of mitochondrial agents including NAC (n = 61), NAC alone (n = 59) or placebo (n = 61), adjunctive to treatment as usual. The study was conducted at three sites in Australia (Geelong, Melbourne and Sydney). Participants were administered the study medication in addition to treatment as usual for 16 weeks and then followed up 4 weeks post study medication cessation (Week 20). A full description of methodology, including inclusion and exclusion criteria (Dean et al., 2015) and primary results of the study (Berk et al., 2019), has been reported previously.
Ethics
The study was approved by Barwon Health Human Research and Ethics Committee (HREC), The Melbourne Clinic Research Ethics Committee, Deakin University HREC and Northern Sydney Local Health District HREC. The study was run in accordance with International Council for Harmonisation Good Clinical Practices Guidelines (Food Drug Administration [FDA], 1997). Of note, the Dietary Questionnaire for Epidemiological Studies (DQES) was included in the study as an exploratory outcome (Dean et al., 2015) for the full protocol of study, although this scale was inadvertently omitted from the ANZCTR trial registration.
Outcomes
The primary outcome of the overarching study was change in depressive symptoms (mean change in MADRS scores). At the primary endpoint (Week 16), there were no significant differences between groups on any of the outcome measures. However, the CT group was significantly superior to placebo at Week 20 on the following interviewer-rated secondary outcomes: Bipolar Depression Rating Scale (BDRS; Berk et al., 2010); Clinical Global Impressions scale bipolar version – Improvement (CGI-I; Spearing et al., 1997), Social and Occupational Functioning Assessment Scale (SOFAS; Morosini et al., 2000) and the Longitudinal Interval Follow-Up Evaluation – Range of Impaired Functioning Tool (LIFE-RIFT; Leon et al., 1999), a scale measuring impairment from psychopathology. All clinical outcome measures were assessed every 4 weeks (baseline, Weeks 4, 8, 12, 16 and 20) with the exception of CGI-I which represents a change from baseline and therefore is not measured until Week 4. A description of score ranges for each scale can be found in Supplementary File 1.
Predictors
Participants’ BMI (kg/m2) was calculated from height and weight measured at their baseline visit. Participants’ current diet was measured by the Cancer Council Victoria DQES, Version 2 (Hodge et al., 2000). The DQES was administered at the Week-4 visit to reduce participant burden at the lengthy baseline visit. Given the scale is measured as a 12-month approximation of dietary intake, it was considered sufficiently stable to be measured at Week 4. Participants (n = 133) who completed the DQES were included in the current evaluation. We examined the effect of DQES-derived diet quality, DQES-derived DII and BMI as potential independent predictors of the nutraceutical treatment response. Given outcomes were only significantly improved in the CT group compared to placebo, not NAC versus placebo; our results will focus on the CT versus placebo differences. However, results pertaining to the NAC versus placebo interactions have been included to illustrate the relationships across the whole sample.
Diet quality (Australian Recommended Food Score)
Diet quality was measured using the Australian Recommended Food Score (ARFS; Collins et al., 2008). The ARFS is a summary and evaluation of participants’ responses on the DQES administered at Week 4, in relation to Australian Dietary Guidelines (NHMRC, 2013). Scores on the ARFS range from 0 to 74, with 74 indicating an ideal diet with a variety of food group intake. Participants were awarded points for each food they consumed within the categories: vegetables, fruit, protein food, grains, dairy, fats and alcohol. For example, participants would garner 1 point for each of the following fruit categories consumed, with a total possible fruit score of 14: two or more pieces of fruit a day, orange or other citrus fruit, melons, apples, pineapple, mango/pawpaw, pears, bananas, strawberries, peach/nectarine, apricots, avocado, one or more fruit or vegetable juice per week, and canned or frozen fruit. The ARFS was calculated using STATA® Data Analysis and Statistical Software 15 (StataCorp, 2017) to convert DQES data. A median split was used to differentiate and categorise high versus low diet quality for descriptive purposes. A summary of total scores and individual food groups is shown in Table 1.
Study participants’ characteristics.
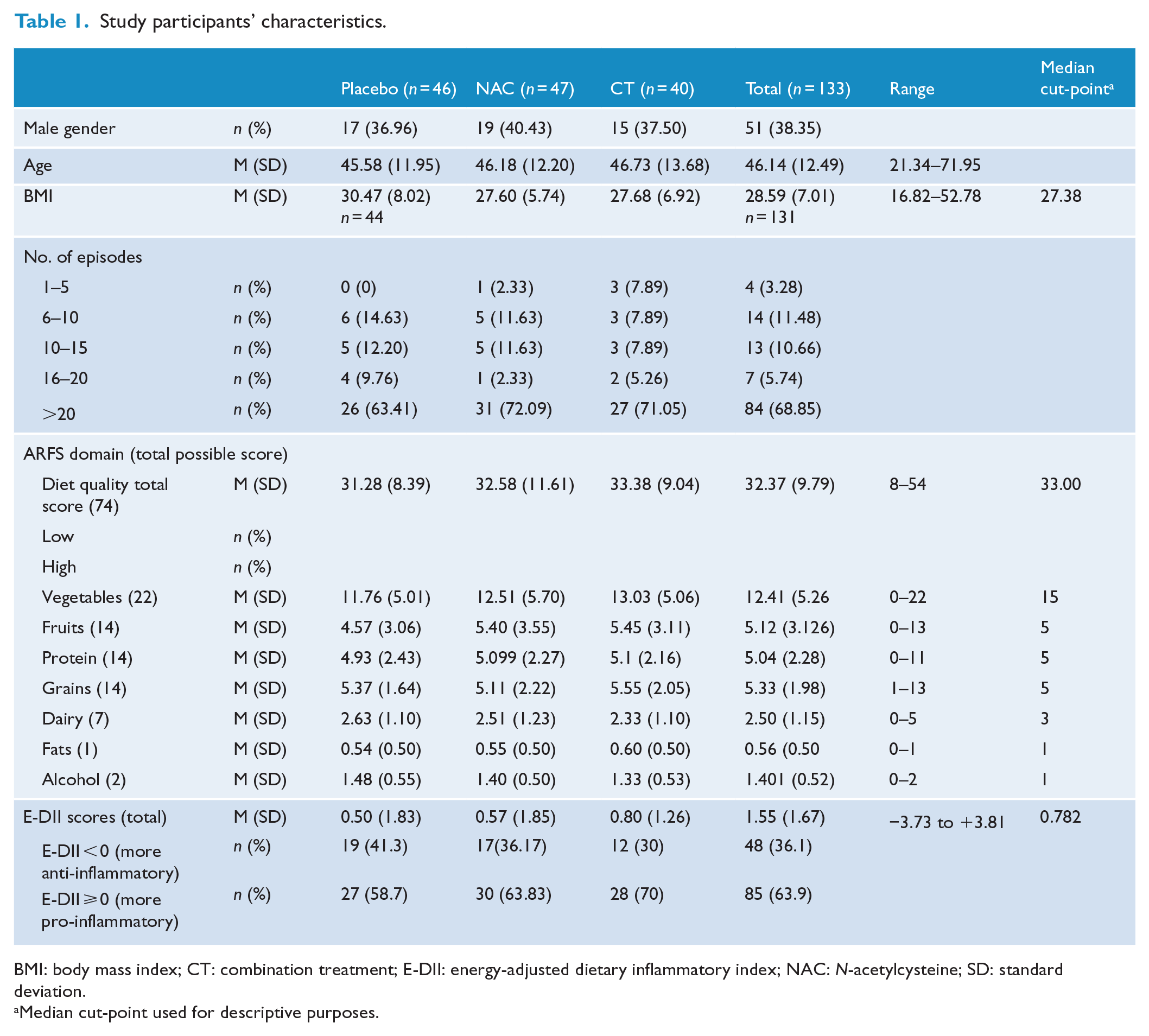
BMI: body mass index; CT: combination treatment; E-DII: energy-adjusted dietary inflammatory index; NAC: N-acetylcysteine; SD: standard deviation.
Median cut-point used for descriptive purposes.
E-DII
E-DII scores were obtained by converting DQES values, for select macro- and micronutrients with fatty acids as per the DII protocol described elsewhere (Shivappa et al., 2014a, 2014b). Lower (i.e. more negative) E-DII scores indicate a more anti-inflammatory diet, and higher (i.e. more positive) scores indicate a more pro-inflammatory diet. Out of the 45 possible food parameters for DII calculations, 24 were available from the DQES (fats, carbohydrate, protein, alcohol, cholesterol, saturated fat, mono-unsaturated fats, poly-unsaturated fats, omega-3, omega-6, fibre, retinol equivalent, beta-carotene, vitamin E, vitamin C, thiamine, riboflavin, niacin equivalent, folate, magnesium, iron, zinc, garlic and onion). The computation of the E-DII scores parallels the method used for the standard DII (Shivappa et al., 2014) that expresses food parameters per 1000 kcal and utilised an energy-adjusted global database.
BMI
BMI was calculated by dividing weight (kg) by height (m) squared using height and weight measured at baseline. BMI categories are as follows: <18.5 kg/m2, underweight; 18.5–24.9 kg/m2, healthy; 25–30 kg/m2, overweight; >30 kg/m2, obese.
Statistical analysis
To investigate the role of diet quality (ARFS), diet inflammatory potential (E-DII) and BMI as potential predictors of symptoms of BD outcomes including MADRS, SOFAS, CGI-I, BDRS and LIFE-RIFT scores, generalised estimating equation (GEE) models were used to address the longitudinal nature of data (e.g. baseline, Weeks 4, 8, 12, 16 and 20). For each study outcome, the original intention-to-treat (ITT) analysis was re-implemented using a GEE approach, and then the impact of potential predictors was evaluated using Baron and Kenny’s (1986) criteria as a guideline (e.g. a model with fixed-effect treatment group as a factor, logarithm of follow-up time and suspected predictor as covariates, and all possible two-way interactions and three-way interaction of suspected predictor, treatment group and time as first described by Kraemer et al., 2001). Each suspected predictor was examined one at a time. For the purposes of this article, we explored each individual treatment arm (CT and NAC) compared to the placebo arm. In the GEE model analysis, ARFS was analysed both as a continuous measure and as nominal factor by dichotomising according to median within a new model due to the skewness of the ARFS empirical distribution. Continuous scores were utilised in the E-DII and BMI models where they were listed as covariates.
We initially evaluated the three-way interaction of the predictor covariate, treatment group and time. The three-way interaction examined the impact of the hypothesised predictor on investigated outcome between treatment groups over the study period as a time-dependent association. If the three-way interaction was not significant, a reduced model with all main effects and two-way interactions was re-run to examine for a two-way interaction of suspected predictor and treatment group. The two-way interaction examined the effect of suspected predictor on investigated outcome between treatment groups as an average across the study period. The p-value for three-way interaction (when significant) or predictor-by-treatment group two-way interaction (from the reduced model, when three-way interaction was not significant) was reported. Beta coefficients and 95% confidence intervals (CIs) for predictor-by-treatment group two-way interaction were reported as a measure of association.
In order to fully explore the data for suspected predictors with non-significant three-way and two-way interactions, analyses of each non-specified predictor were explored. The model consisted of main effects of treatment group, time and the suspected predictor. If the main effect was significant, it was considered a non-specific predictor of the outcome because it was predictive of response in all treatment groups combined, but not associated with a differential response to treatment.
A GEE technique for continuous outcomes was used for model estimation to account for within participants’ autocorrelation. An unstructured working correlation matrix and a robust variance estimator were used in conjunction with GEE to handle any potential misspecification of correlation structure (White, 1980). Wald χ2 statistic (after fitting GEE models) was used to examine the model parameter effects. Data were split according to above and below median score in order to visualise the spread of the data. Statistical analyses were completed using IBM® SPSS® Statistics for Windows, Version 25 (IBM Corp, 2017).
Results
Participants
From the total clinical trial sample (n = 181), 33 participants did not have any post-baseline data and a further 15 participants had missing DQES data. Therefore, 133 participants who completed the DQES were included in the current study. Of these, 61.7% were females, and the average age was 46.1 (SD = 12.94) years ranging from 21 to 71 years. In total, 46 participants received placebo, 47 received NAC, and 40 received the CT.
Analysis of predictors
For descriptive purposes, a visual representation of mean change scores for each outcome measure separated by treatment arm and in terms of predictor has been included as Supplementary Files 2–4. As CGI-I is a change score in and of itself, and not measured at baseline, a summary of average Week 20 CGI-I scores is included in Table 2. Table 2 shows the mean scores and SD for high and low of each predictor in each treatment arm.
Mean Week 20 CGI-I scores.
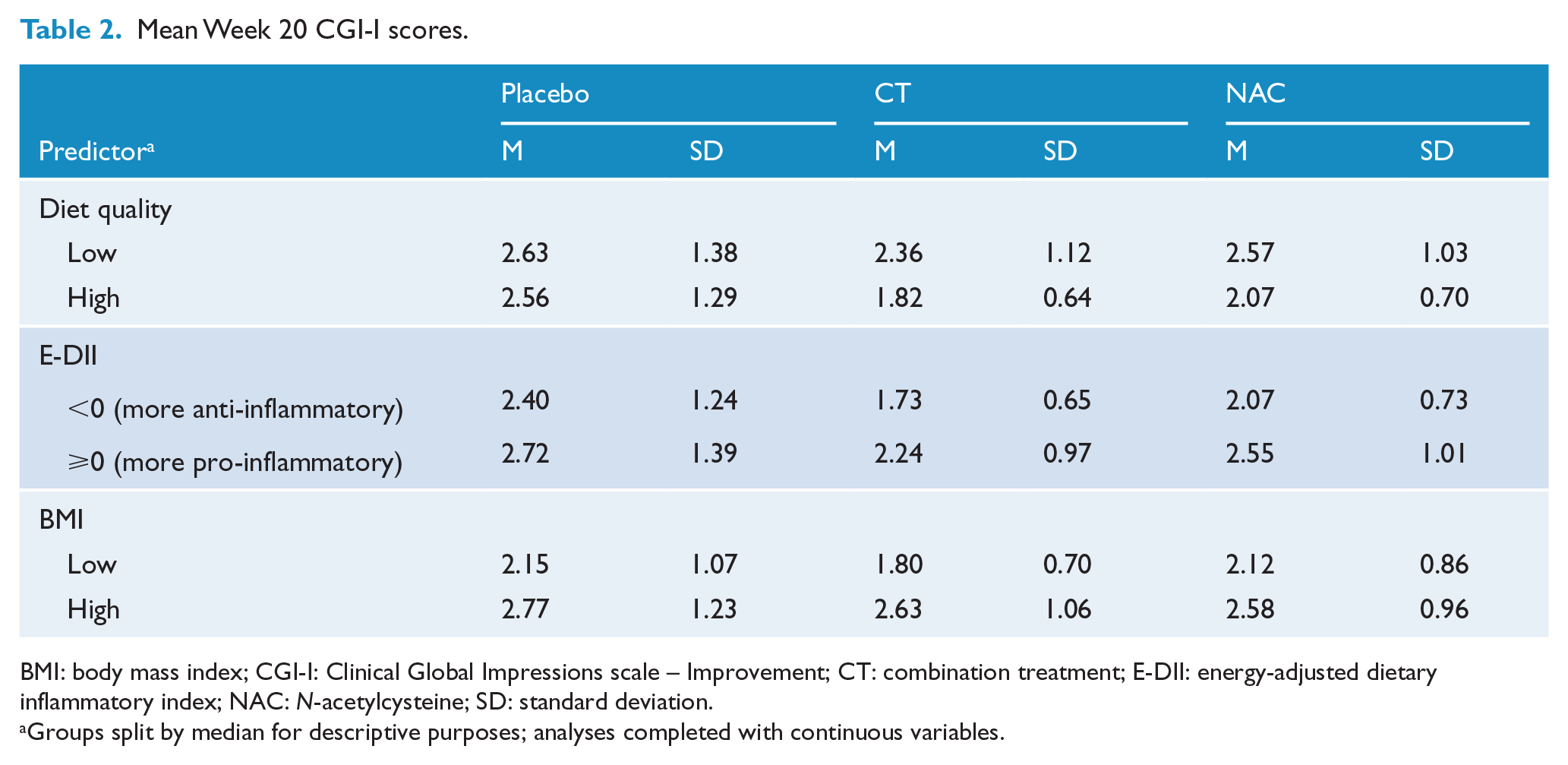
BMI: body mass index; CGI-I: Clinical Global Impressions scale – Improvement; CT: combination treatment; E-DII: energy-adjusted dietary inflammatory index; NAC: N-acetylcysteine; SD: standard deviation.
Groups split by median for descriptive purposes; analyses completed with continuous variables.
Diet quality (ARFS)
The range of scores on the ARFS for the whole sample was 8 to 54 (Mdn = 33). There was no significant difference on ARFS between placebo and NAC, or CT and placebo, F(2, 130) = 0.50, p = 0.61.
When analysing ARFS as a continuous variable in the first model, results were mainly non-significant with some spurious findings. Therefore, we focused on presenting the dichotomised ARFS model using a median split. Results of the effect modification analyses (i.e. two-way interactions between treatment group and ARFS) are shown in Table 3. All three-way and two-way interactions between ARFS, group allocation and outcome measure were not significant. Therefore, ARFS did not predict MADRS, BDRS, SOFAS, LIFE-RIFT or CGI-I scores.
Diet quality, dietary inflammatory index and BMI on mean change scores for each treatment arm.
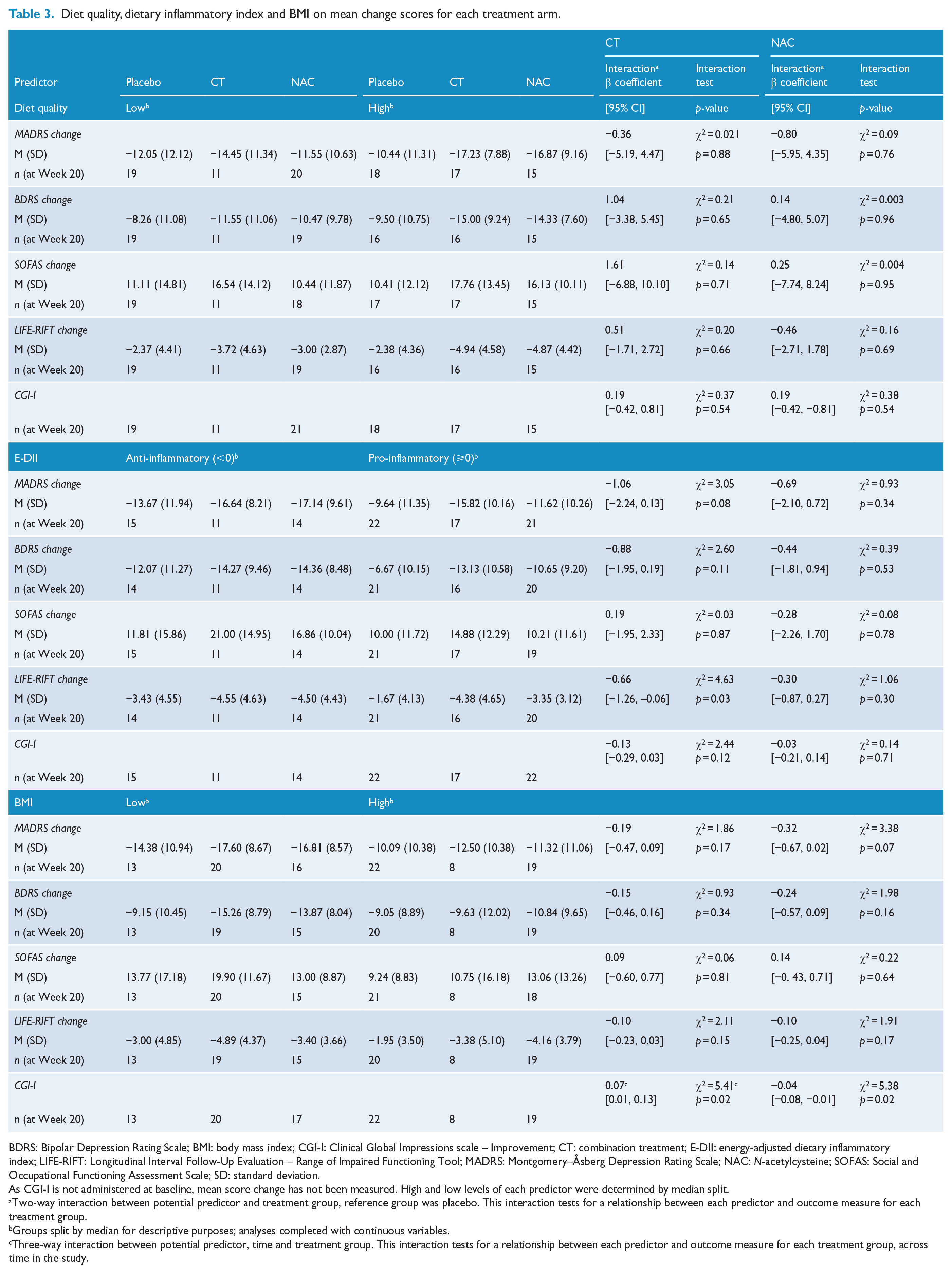
BDRS: Bipolar Depression Rating Scale; BMI: body mass index; CGI-I: Clinical Global Impressions scale – Improvement; CT: combination treatment; E-DII: energy-adjusted dietary inflammatory index; LIFE-RIFT: Longitudinal Interval Follow-Up Evaluation – Range of Impaired Functioning Tool; MADRS: Montgomery–Åsberg Depression Rating Scale; NAC: N-acetylcysteine; SOFAS: Social and Occupational Functioning Assessment Scale; SD: standard deviation.
As CGI-I is not administered at baseline, mean score change has not been measured. High and low levels of each predictor were determined by median split.
Two-way interaction between potential predictor and treatment group, reference group was placebo. This interaction tests for a relationship between each predictor and outcome measure for each treatment group.
Groups split by median for descriptive purposes; analyses completed with continuous variables.
Three-way interaction between potential predictor, time and treatment group. This interaction tests for a relationship between each predictor and outcome measure for each treatment group, across time in the study.
Diet inflammatory potential (E-DII)
E-DII scores ranged from −3.73 to +3.81, with 36.1% of participants having an E-DII score <0 (more anti-inflammatory diet). There was no significant difference between E-DII scores for CT, NAC and placebo groups, F(2, 130) = 0.08, p = 0.92.
All three-way interactions between E-DII, group allocation and outcome measure were not significant. A summary of two-way interactions between treatment group and E-DII scores can be found in Table 3. Results of the effect modification analyses showed that E-DII scores were an effect modifier for LIFE-RIFT score. For participants in the CT group compared to placebo, higher E-DII scores predicted a decrease in LIFE-RIFT score (towards improvement). For every unit increase in E-DII score (towards pro-inflammatory), LIFE-RIFT scores decreased by 0.66, representing a small reduction in impairment (β = −0.66 [−1.26, −0.06]). Scores for E-DII did not predict change in MADRS, BDRS, SOFAS or CGI-I scores when comparing placebo and both the CT and the NAC groups to placebo.
BMI
Participants had an average weight of 82.6 (SD = 20.20) kg, an average height of 170.2 (SD = 9.40) cm and an average BMI of 28.6 (SD = 7.01) kg/m2. Participants’ BMI scores ranged from 16.8 to 52.8 kg/m2, with a median score of 27.4 kg/m2 at baseline. There were no significant differences among BMI scores for CT, NAC or placebo arms, F(2, 128) = 2.44, p = 0.09. Of the 133 participants in this study, 3.0% were considered underweight, 30.8% were in the healthy range, 32.3% were overweight and 32.3% obese (including 1.5% missing data; n = 2).
A summary of results for treatment group and BMI for investigated outcomes can be found in Table 3. All three-way and two-way interactions between BMI, group allocation and outcome measure were not significant, with the exception of CGI-I. Therefore, BMI was not a significant predictor for MADRS, BDRS, SOFAS or LIFE-RIFT scores. There was a significant three-way interaction between BMI, group allocation and time for CGI-I scores when comparing the CT group to placebo. When comparing placebo and NAC, there was a significant two-way interaction between BMI and CGI-I scores. For every 10-unit decrease in BMI scores, CGI-I scores further decreased by 0.07, demonstrating greater global improvement (β = 0.07 [0.01, 0.13]).
Non-specified predictors
A summary of the main effect of potential non-specified predictors in CT compared to placebo is displayed in Table 4. The ARFS was a significant non-specified predictor of MADRS, BDRS and CGI-I outcomes. Participants with higher ARFS overall had lower post-baseline MADRS and BDRS depression symptom scores (improvement in depression), regardless of treatment group or time point. For every 10 unit increase in ARFS, MADRS scores decreased by 1.6. For every 10 unit increase in ARFS, BDRS scores would decrease by 1.4. Participants with higher ARFS overall had lower CGI-I scores, demonstrating greater improvement, regardless of treatment group or time point. For every 10-unit increase in ARFS, CGI-I scores would decrease by 0.3 (β = −0.03 [−0.05, −0.004]).
Non-specified predictors of outcomes in CT group versus placebo.
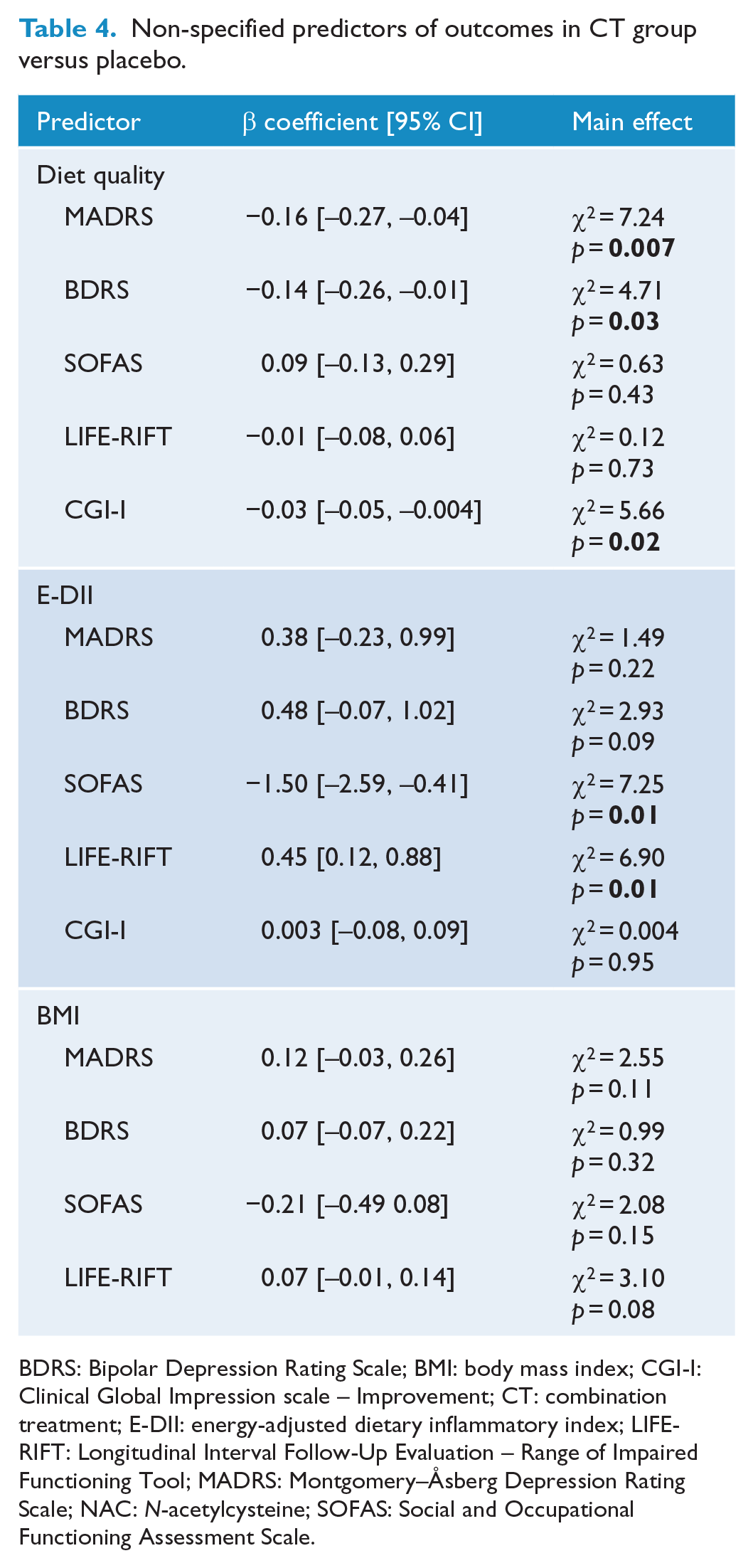
BDRS: Bipolar Depression Rating Scale; BMI: body mass index; CGI-I: Clinical Global Impression scale – Improvement; CT: combination treatment; E-DII: energy-adjusted dietary inflammatory index; LIFE-RIFT: Longitudinal Interval Follow-Up Evaluation – Range of Impaired Functioning Tool; MADRS: Montgomery–Åsberg Depression Rating Scale; NAC: N-acetylcysteine; SOFAS: Social and Occupational Functioning Assessment Scale.
Scores on the E-DII were a non-specified predictor of SOFAS score for participants in the CT group compared to placebo group. Participants with lower E-DII scores overall had higher post-baseline SOFAS scores, regardless of treatment group or time point. For every unit decrease in DII score (towards anti-inflammatory), SOFAS scores increased by 0.19 (β = −1.50 [−2.59, −0.41]). This represents a very small change in SOFAS scores towards higher social and occupational functioning. For participants in NAC group compared to placebo, E-DII was a non-specified predictor for both SOFAS and LIFE-RIFT scores.
All non-specified predictor analyses for BMI scores were not significant. BMI was not a predictor of MADRS, BDRS, SOFAS or LIFE-RIFT outcomes.
Discussion
The aim of the current study was to examine the impact of overall diet quality, dietary inflammatory potential and BMI on clinical outcomes using an exploratory analysis of a nutraceutical RCT targeting bipolar depression. Our hypothesis that diet quality, measured by the ARFS, or dietary inflammatory potential, measured by E-DII, or BMI are correlated with outcomes of CT or NAC treatment was only partially supported. Participants’ diet quality did not predict the outcomes of treatment of CT or NAC. However, participants with better diet quality were more likely to have lower MADRS, BDRS and CGI-I scores at Week 20 compared to participants with lower diet quality scores, regardless of treatment group allocation. While the 1.6-unit decrease in MADRS is considered at the lower end of clinical significance (Duru and Fantino, 2008), a 10-point increase in ARFS, thus improving diet, is a large change to achieve this difference.
With regard to the inflammatory potential of a diet, participants who adhered to a more anti-inflammatory diet (measured by the E-DII) had a greater reduction in impairment of functioning (LIFE-RIFT) regardless of treatment received. Participants consuming a more pro-inflammatory diet appeared to show the least improvement in LIFE-RIFT scores, as demonstrated by the placebo group results. However, treatment with CT appeared to attenuate the effects of the pro-inflammatory diet, thus showing a greater improvement in LIFE-RIFT scores, compared to placebo. An anti-inflammatory diet was also associated with an improvement in social and occupational functioning (SOFAS) compared to pro-inflammatory diet, regardless of treatment received.
The influence of anti-inflammatory dietary intake is particularly interesting as it aligns well with both the pathophysiology of BD and the current research into adjunctive use of anti-inflammatory medications for treating BD (see Rosenblat et al., 2016, for systematic review). These results are in keeping with previous research which has reported poorer diet quality in participants with BD (Goldstein et al., 2011; Jacka et al., 2011), and that pro-inflammatory diets are linked to the risk for depression (Lassale et al., 2019). Consumption of healthy diets may exert an effect on D2 signalling (Pritchett and Hajnal, 2011), may decrease neuroinflammation and oxidative stress, may positively influence hippocampal function and neuronal signalling pathways (for review see Knight et al., 2016) and may enhance mitochondrial activity (Lopresti and Jacka, 2015). Diet quality may also be a proxy of wider adaptive health behaviours. These results also highlight the effect of the CT through the inflammatory pathway in BD symptoms.
In addition to diet quality, results suggest that BMI may be a predictor for some treatment outcomes in BD. In particular, participants in the CT group who had lower BMI had greater clinician-rated improvement across the 20-week RCT. While BMI also predicted clinician-rated improvement in the NAC group, the relationship was not strong. Participants’ BMI did not predict any other outcomes in the study.
The relationship between BMI and response to nutraceutical treatment is in keeping with previous literature (Kemp et al., 2010). Diet and BMI may influence treatment outcomes through their shared associations of BD and operative biological pathways as well as general medical conditions (SayuriYamagata et al., 2017). Increased BMI has been correlated with higher inflammatory potential, which may predispose to higher rates of chronic disease and poor mental health (Trayhurn, 2005). BMI may thus be a contributing factor in how participants respond to treatments. Within the current study, participants with lower BMI showed better clinically rated improvement, compared to those with higher BMI. A possible interpretation is that having a lower BMI increases the potential improvement of a participant’s experience of BD symptoms. This was particularly pronounced when participants were administered the CT medication. It is unclear whether lower BMI can modulate the effectiveness of CT or whether high BMI can impede response to treatment effects, or both. It should be noted that approximately 64% of the sample were either overweight or obese, which is concordant with rates in the wider Australian population (Huse et al., 2018) and a typical psychiatric sample, who are commonly overweight (McElroy et al., 2004; Strassnig et al., 2017). This means interpretations can be generalised to the general community.
Adjunctive treatment with the CT and NAC may be optimised if diet quality and body composition are assessed and improved prior to commencing treatments. There appears to be a relationship between high-quality diet rich in antioxidant foods and lower BMI as predicting the results for participants who received CT in this study. Within the context of this study, we are unable to determine what drives this relationship. It may be that better diet quality and lower BMI are indicative of stage of illness, severity of illness, greater health literacy or self-efficacy, or medication regimen.
The results of this study need to be interpreted with caution due to several limitations. The study is likely underpowered to detect consistent associations, and the overall treatment effect for the active treatment arms versus placebo at the primary endpoint of Week 16 MADRS scores was not significant, although the CT group was superior to placebo at the Week-20 follow-up visit (reported elsewhere; Berk et al., 2019). Further limitations include the following: the sample size was calculated for the primary outcome of the main RCT; dietary data (DQES) were collected at one time point and were averaged for 12 months; and the DQES may be prone to a variety of biases (due to close-ended dietary data collection) (Cade et al., 2004; Hebert et al., 1995, 1997). BMI is also an imperfect measure of body composition as it does not take into account weight from muscle versus fat (Schneider et al., 2010). To explore these relationships further, future research should consider a combination of food frequency questionnaires and diet intake diaries and include measurement of potential confounders/effect modifiers and evaluation of change over time. Similarly, waist and hip circumferences would provide better information regarding body composition. Statistically, type I error has not been adjusted for multiple comparisons and as such, results should be interpreted cautiously. Finally, it is essential that this study is interpreted only in the context of treatment response in a clinical trial.
Nevertheless, despite some limitations, there are several strengths of this study. The data were drawn from a suitably powered double-blind placebo-controlled clinical trial conducted in multiple sites in Australia. The DQES is a well-designed and commonly utilised tool, and the converted ARFS is designed for Australian diets; therefore, this score is directly relevant to the study sample. We have utilised the E-DII, which is a widely used variant of the DII®. Both the DII and the E-DII have been validated 19 times ranging from the original validation in the Season Study (Shivappa et al., 2014b) to an Australian study of subjects with coronary heart disease (Mayr et al., 2018). Finally, there was adequate retention of participants within the primary trial with an attrition rate of 20% (Berk et al., 2019).
Results from our study provide some support for the hypothesis that having a healthy, more anti-inflammatory diet and lower BMI may enhance the efficacy of a combination of mitochondrial-enhancing nutraceuticals in the treatment of BD. In addition, the results also support the argument that dietary data should be collected in clinical trials to be controlled for in post hoc mediation/effect modification analyses. Furthermore, initial assessment of these factors in a clinical setting and individualising treatment regimens could be a possible avenue of optimised treatments for BD.
Our results provide additional information regarding the relationship between diet quality, the inflammatory potential of a diet, BMI and treatment outcomes in BD. Further research into these relationships is required, especially to explore how diet and BMI may be affecting treatment outcomes, including direction of the relationship. Recommendations for future research include the use of better tools for collecting dietary data and BMI at multiple time-points, and larger sample sizes.
Supplemental Material
Supplementary_file_1 – Supplemental material for Diet quality, dietary inflammatory index and body mass index as predictors of response to adjunctive N-acetylcysteine and mitochondrial agents in adults with bipolar disorder: A sub-study of a randomised placebo-controlled trial
Supplemental material, Supplementary_file_1 for Diet quality, dietary inflammatory index and body mass index as predictors of response to adjunctive N-acetylcysteine and mitochondrial agents in adults with bipolar disorder: A sub-study of a randomised placebo-controlled trial by Melanie M Ashton, Olivia M Dean, Wolfgang Marx, Mohammadreza Mohebbi, Michael Berk, Gin S Malhi, Chee H Ng, Sue M Cotton, Seetal Dodd, Jerome Sarris, Malcolm Hopwood, Keshav Faye-Chauhan, Yesul Kim, Sarah R Dash, Felice N Jacka, Nitin Shivappa, James R Hebert and Alyna Turner in Australian & New Zealand Journal of Psychiatry
Supplemental Material
Supplementary_file_2-4 – Supplemental material for Diet quality, dietary inflammatory index and body mass index as predictors of response to adjunctive N-acetylcysteine and mitochondrial agents in adults with bipolar disorder: A sub-study of a randomised placebo-controlled trial
Supplemental material, Supplementary_file_2-4 for Diet quality, dietary inflammatory index and body mass index as predictors of response to adjunctive N-acetylcysteine and mitochondrial agents in adults with bipolar disorder: A sub-study of a randomised placebo-controlled trial by Melanie M Ashton, Olivia M Dean, Wolfgang Marx, Mohammadreza Mohebbi, Michael Berk, Gin S Malhi, Chee H Ng, Sue M Cotton, Seetal Dodd, Jerome Sarris, Malcolm Hopwood, Keshav Faye-Chauhan, Yesul Kim, Sarah R Dash, Felice N Jacka, Nitin Shivappa, James R Hebert and Alyna Turner in Australian & New Zealand Journal of Psychiatry
Footnotes
Acknowledgements
The authors would like to thank all participants of the study. The authors would also like to acknowledge the following health services involved in this study: Barwon Health, The Geelong Clinic, The Melbourne Clinic and the University of Sydney CADE Clinic based at Royal North Shore Hospital. The authors are grateful to the Stanley Medical Research Institute and the National Health and Medical Research Council for funding the study. M.M.A. would like to thank the Australasian Society for Bipolar and Depressive Disorders, Lundbeck, Australian Rotary Health and the Ian Parker Bipolar Research Fund for providing PhD scholarship support. The authors thank Professor Graham Giles of the Cancer Epidemiology Centre of the Cancer Council Victoria for permission to use the Dietary Questionnaire for Epidemiological Studies (Version 2), Melbourne: The Cancer Council Victoria, 1996 (ANZCTR registry: ACTRN12612000830897).
Author Contributions
Initial planning of the manuscript was conducted by M.M.A., A.T., O.M.D., M.B. and W.M.; data were collected by K.F.-C. and Y.K.; M.M.A. conducted the analyses of data under the guidance of M.M.; the initial draft of the manuscript and tables was produced by M.M.A.; M.M.A., O.M.D., W.M., M.M., M.B., G.S.M., C.H.N., S.M.C., S.D., J.S., M.H., K.F.-C., Y.K., S.R.D., F.N.J., N.S., J.H. and A.T. contributed to and edited drafts of the paper and approved the final version.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship and/or publication of this article: M.M.A. has received grant/research support from Deakin University, Australasian Society for Bipolar Depressive Disorders, Lundbeck, Australian Rotary Health, Ian Parker Bipolar Research Fund and Cooperative Research Centre for Mental Health. M.B. has received grant support from NIH, Simons Autism Foundation, Cancer Council of Victoria, CRC for Mental Health, Stanley Medical Research Foundation, MBF, NHMRC, Beyond Blue, Geelong Medical Research Foundation, Bristol-Myers Squibb, Eli Lilly, GlaxoSmithKline, Organon, Novartis, Mayne Pharma and Servier; M.B. has been a speaker for AstraZeneca, Bristol-Myers Squibb, Eli Lilly, GlaxoSmithKline, Janssen-Cilag, Lundbeck, Merck, Pfizer, Sanofi-Synthelabo, Servier, Solvay and Wyeth and served as a consultant to AstraZeneca, Bristol-Myers Squibb, Eli Lilly, GlaxoSmithKline, Janssen-Cilag, Lundbeck and Servier. C.H.N. had served as a consultant for Lundbeck, Grunbiotics, Servier, Janssen-Cilag, Wyeth and Eli Lilly, received research grant support from Wyeth and Lundbeck, and speaker honoraria from Servier, Lundbeck, Bristol-Myers Squibb, Organon, Eli Lilly, GlaxoSmithKline, Janssen-Cilag, AstraZenaca, Wyeth and Pfizer. M.H. has received grant support from ISSCR, Servier, US DOD and Bionomics, has been a speaker for Janssen-Cilag, Lundbeck and Servier, and has been a consultant for AstraZeneca, Eli Lilly, Janssen-Cilag, Lundbeck and Servier. J.S. has received either presentation honoraria, travel support, clinical trial grants, book royalties or independent consultancy payments from Integria Healthcare & MediHerb, Pfizer, Scius Health, Key Pharmaceuticals, Taki Mai, Bioceuticals & Blackmores, Soho Flordis, Healthworld, HealthEd, HealthMasters, Elsevier, Chaminade University, International Society for Affective Disorders, Complementary Medicines Australia, Terry White Chemists, ANS, Society for Medicinal Plant and Natural Product Research, UBiome, Omega-3 Centre, the National Health and Medical Research Council and CR Roper Fellowship. S.D. has received grant support from Stanley Medical Research Institute, NHMRC, Beyond Blue, ARHRF, Simons Foundation, Geelong Medical Research Foundation, Fondation FondaMental, Eli Lilly, GlaxoSmithKline, Organon, Mayne Pharma and Servier; he has received speaker’s fees from Eli Lilly, advisory board fees from Eli Lilly and Novartis, and conference travel support from Servier. G.S.M. has received grant or research support from National Health and Medical Research Council, Australian Rotary Health, NSW Health, Ramsay Health, American Foundation for Suicide Prevention, Ramsay Research and Teaching Fund, Elsevier, AstraZeneca and Servier; has been a speaker for AstraZeneca, Janssen-Cilag, Lundbeck and Servier; and has been a consultant for AstraZeneca, Janssen-Cilag, Lundbeck and Servier. F.N.J. has received grant/research support from the Brain and Behaviour Research Institute, the NHMRC, Australian Rotary Health, The Geelong Medical Research Foundation, the Ian Potter Foundation, Eli Lilly, Meat and Livestock Australia, Woolworths Limited, Fernwood Gyms, The Wilson Foundation, GMHBA and The University of Melbourne and has received speakers honoraria from Sanofi-Synthelabo, Janssen-Cilag, Servier, Pfizer, Health Ed, Network Nutrition, Angelini Farmaceutica, Eli Lilly and Metagenics; furthermore, she is currently writing a book about diet and mental and brain health and has a personal belief that good diet quality is important for mental and brain health. S.M.C. has received grant support from the NHMRC, the Stanley Medical Research Institute, Beyond Blue, Movember, The University of Melbourne, Australian Catholic University, ARHRF and Mental Illness Research Fund (Victoria Department of Human Services). O.M.D. is a R.D. Wright Biomedical Research Fellow and has received grant support from the Brain and Behavior Foundation, Simons Autism Foundation, Stanley Medical Research Institute, Deakin University, Lilly, NHMRC and Australasian Society for Bipolar and Depressive Disorders (ASBDD)/Servier. N.S. has received support from the US National Institute of Diabetes, Digestive and Kidney Diseases and the US Department of Agriculture. J.R.H. has received support from the National Cancer Institute, National Institute on Minority Health and Health Disparities, National Heart Lung and Blood Institute, and the National Institute of Diabetes, Digestive and Kidney Diseases and Genentech. A.T. has received travel or grant support from the NHMRC, Deakin University, AMP Foundation, National Stroke Foundation, Hunter Medical Research Institute, Helen Macpherson Smith Trust, Schizophrenia Fellowship NSW, SMHR, ISAD and the University of Newcastle.
Disclosure Statement
Dr James R. Hébert owns controlling interest in Connecting Health Innovations LLC (CHI), a company that has licenced the right to his invention of the dietary inflammatory index (DII) from the University of South Carolina in order to develop computer and smart phone applications for patient counselling and dietary intervention in clinical settings. Dr Nitin Shivappa is an employee of CHI.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship and/or publication of this article: The study has been funded by CRC for Mental Health, the Stanley Medical Research Institute and an NHMRC Project Grant (APP1026307). M.M.A. is supported by Deakin University, Australasian Society for Bipolar and Depressive Disorders (ASBDD)/Lundbeck and Australian Rotary Health/Ian Parker Bipolar Research Fund. O.M.D. is supported by a NHMRC R.D. Wright Biomedical Research Fellowship (APP1145634). M.B. is supported by a National Health and Medical Research Council (NHMRC) Senior Principal Research Fellowship (APP1059660 and APP1156072). F.N.J. is supported by an NHMRC Career Development Fellowship (2) (APP1108125). S.C. is supported by a NHMRC Senior Research Fellowship (APP1136344). J.S. is funded by an NHMRC Clinical Research Fellowship (APP1125000). The sponsors and funding bodies have played no role in collection, analysis, interpretation of results or writing of the manuscript.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
