Abstract
Objective:
This study examines characteristics of individuals with bipolar disorder who sought psychotherapy versus those who did not.
Methods:
Bipolar CHOICE was an 11-site comparative effectiveness study of lithium versus quetiapine in symptomatic outpatients (N = 482) with bipolar disorder. At baseline, participants’ psychotherapy use within the past 3 months, mood, functioning, and overall health were assessed. Logistic regressions were used to test whether psychotherapy users and non-users differed on various demographic and clinical variables at baseline. Mixed-effects regression was used to determine whether psychotherapy groups differed on response to treatment over the 6-month study. Kaplan-Meier plots and log-rank tests were employed to test whether there were any differences in time to recovery (CGI-BP ≤ 2 for at least 8 weeks) between the groups.
Results:
Thirty one percent of participants reported using psychotherapy services. Psychotherapy users reported greater medication side effect burden than non-users and were more likely to have moderate to high suicide risk and at least one anxiety disorder. Participants not utilizing medications or psychotherapy had greater mania symptom severity, were younger, and less educated than medication only users. Medication only users were more likely to be married than the other participants.
Conclusions:
These data suggest that a minority of individuals with bipolar disorder attend psychotherapy services, and those that do have greater illness burden.
Introduction
Bipolar disorder is associated with significant functional impairment, lower quality of life, and higher rates of suicide (Kilbourne et al., 2004; Novick et al., 2010). The foundations of treatment are mood stabilizers and antipsychotics (Geddes and Miklowitz, 2013; American Psychiatric Association, 2002); however, pharmacotherapy alone seldom leads to full and sustained remission (Frank et al., 2000). Even with stable pharmacotherapy, close to 40% of individuals experience an episode recurrence within one year, and over 70% have a recurrence within five years (Gitlin et al., 1995; Tohen et al., 1990).
Adjunctive psychotherapy can thus be very beneficial as it targets areas that medications do not address, such as teaching individuals about the illness, providing skills to control their mood fluctuations, coping with stressful life events, improving social rhythms, sleep patterns, and interpersonal skills (Miklowitz, 2008a; Swartz et al., 2012). In a review of 18 randomized trials of psychotherapy, adjunctive psychosocial treatments were associated with a 30 to 40% reduction in relapse rates over the course of 12 to 30 months, as well as greater functional improvements compared to those taking medications alone (Miklowitz, 2008a). However, employment of psychotherapy as a treatment modality continues to be underutilized (Evans-Lacko et al., 2011; Weiss et al., 2000). In a Canadian Community Health survey of 852 bipolar respondents, over 45% reported never using any psychosocial treatment services (Schaffer et al., 2006). Furthermore, in a study that examined respondents from the 1987 National Medical Expenditure survey, psychotherapy users reported worse general health, greater functional impairment, and higher medical costs than psychotherapy non-users (Olfson and Pincus, 1994).
The aim of this study is to examine the characteristics of symptomatic individuals with bipolar disorder who utilized psychotherapy services. Given that psychotherapy services seem to be underutilized, we predicted that participants with greater symptom severity and comorbidities would be more likely to utilize psychotherapy services because traditional treatment for bipolar disorder (i.e., pharmacotherapy) may not be sufficient to reduce illness burden for individuals who have greater symptom severity. Because psychotherapy plus medications has shown to out-perform psychotherapy or medications alone for bipolar disorder (Parikh et al., 2014), we expected that psychotherapy users (while taking study medication) would have better clinical outcomes.
Methods
Procedure
The Clinical Health Outcomes Initiative in Comparative Effectiveness for Bipolar Disorder (Bipolar CHOICE) was an 11-site, 6-month randomized comparative effectiveness trial of lithium versus quetiapine (Nierenberg et al., in press). Each site’s institutional review board approved the study protocol and subjects provided written and verbal consent prior to participation. In addition to the randomized medication, all participants received adjunctive personalized treatment (APT), which was guideline-informed and evidence-based care (Suppes et al., 2005). Thus, study investigators were able to prescribe additional medications as clinically necessary, with some restrictions depending on participants’ randomization group. The rationale and methods of CHOICE are described elsewhere (Nierenberg et al., 2013).
Participants
Four hundred eighty two individuals (ages 18-68) were enrolled in the study between 2011 and 2012. To be eligible, participants had to meet DSM-IV TR criteria for bipolar I or II disorder and currently be symptomatic (Clinical Global Impression Scale for Bipolar Disorder ⩾ 3). Individuals were excluded from the study if they were currently taking therapeutic doses of lithium or quetiapine or had any contraindication to lithium or quetiapine. Inclusion and exclusion criteria were broad to maximize generalizability of the study sample (Nierenberg et al., 2013).
Measures
Psychiatric history
Current and lifetime DSM-IV TR diagnoses and suicide risk were assessed using the Extended Mini-International Neuropsychiatric Interview (Sheehan et al., 1998). Participants reported their number of bipolar episodes in the prior year and percentage of time depressed or manic/hypomanic.
Medical history
At screening, participants reported their current and past medical conditions. At weeks 0, 8, and 24, fasting serum chemistries and vital signs were conducted. Metabolic syndrome was assessed according to the National Cholesterol Education Program’s guidelines (Expert Panel on Detection, Evaluation, and Treatment of High Blood Cholesterol in Adults, 2001).
Mood and functioning
Severity of psychiatric symptoms was assessed using the Bipolar Inventory of Symptoms Scale (BISS; Bowden et al., 2007) and the Clinical Global Impression Scale for Bipolar Disorder (CGI-BP; Spearing et al., 1997). Higher BISS and CGI-BP scores indicate greater symptom severity. Life functioning was assessed at weeks 0, 12, and 24 using the Longitudinal Follow-Up Evaluation – Range of Impaired Functioning Tool (LIFE-RIFT; Leon et al., 2000). Higher LIFE-RIFT scores are indicative of greater functional impairment.
Psychotherapy utilization
At screening, the Cornell Services Index assessed the type, provider, frequency, site, and cost of health services utilized in the past 90 days (Sirey et al., 2005). Psychotherapy visits included individual, group, couples/family therapy, combined medication and psychotherapy, or other psychological service visit with the following provider types: psychiatrist (MD), psychologist (PhD), social worker, unspecified therapist or counselor, nurse practitioner, psychiatric nurse practitioner, or vocational rehabilitation counselor. Patients that reported having used at least one psychotherapy visit during the 90 days prior to baseline, inclusive of all modalities, were categorized as psychotherapy users.
Medication use and side effects
Participants reported the medications they were taking prior to starting the study. At each study visit, the medication recommendation tracking form (MRTF) assessed dose changes, missed doses, and medication changes (Reilly-Harrington et al., 2013). The Frequency and Intensity of Side Effects Ratings (FISER) assessed medication burden (Wisniewski et al., 2006), and higher scores indicate greater burden.
Statistical analyses
Statistical analyses were conducted using SAS version 9.2 (SAS Institute, Cary, NC). Logistic regressions examined whether the lithium and quetiapine groups differed in psychotherapy usage (if no difference, treatment groups were pooled together). Logistic regression models were used to compare baseline differences in demographic and clinical variables between psychotherapy users and non-users. Mixed-effects regression models were conducted to compare changes in clinical outcomes over 6 months between psychotherapy users and non-users. We included fixed effects for psychotherapy group, time, group-by-time interaction, site, site-by-time interaction, as well as random effects for intercepts and slopes. These models take into account the baseline group differences in clinical outcomes. We produced Kaplan-Meier plots and conducted a log-rank test to determine whether there was a difference in time to recovery (CGI-BP ≤ 2 for at least 8 weeks) between baseline psychotherapy users and non-users.
We categorized participants by whether they attended a psychotherapy visit only, but were not taking any psychiatric medications, attended psychotherapy and were taking medications, were taking medications only, or neither in the 90 days prior to enrollment. ANOVAs were used to compare these four groups on demographic and clinical variables. Post-hoc contrasts were conducted to determine significant pairwise differences. Given the exploratory nature of this paper, no adjustment for multiple hypothesis testing was made.
Results
Psychotherapy use prior to baseline
Treatment group
At baseline 30.7% of participants in the CHOICE sample utilized psychotherapy up to 90 days before baseline. The two treatment groups were not statistically significant in their use of psychotherapy prior to randomization (lithium group, 27.5% versus quetiapine group, 33.9%; p = .13). The average number of psychotherapy visits in the 90 days prior to enrollment for the whole sample was 3.6 (SD = 9.8). The average number of psychotherapy visits was 8.3 (SD = 13.9) among patients utilizing psychotherapy services (i.e., with at least one visit with the 90 days prior to baseline). There was no difference in the baseline frequency of prior psychotherapy visits between the lithium (M = 3.6, SD = 10.5) and quetiapine (M = 3.6, SD = 9.0) groups (p = 1.0).
Demographic and clinical characteristics
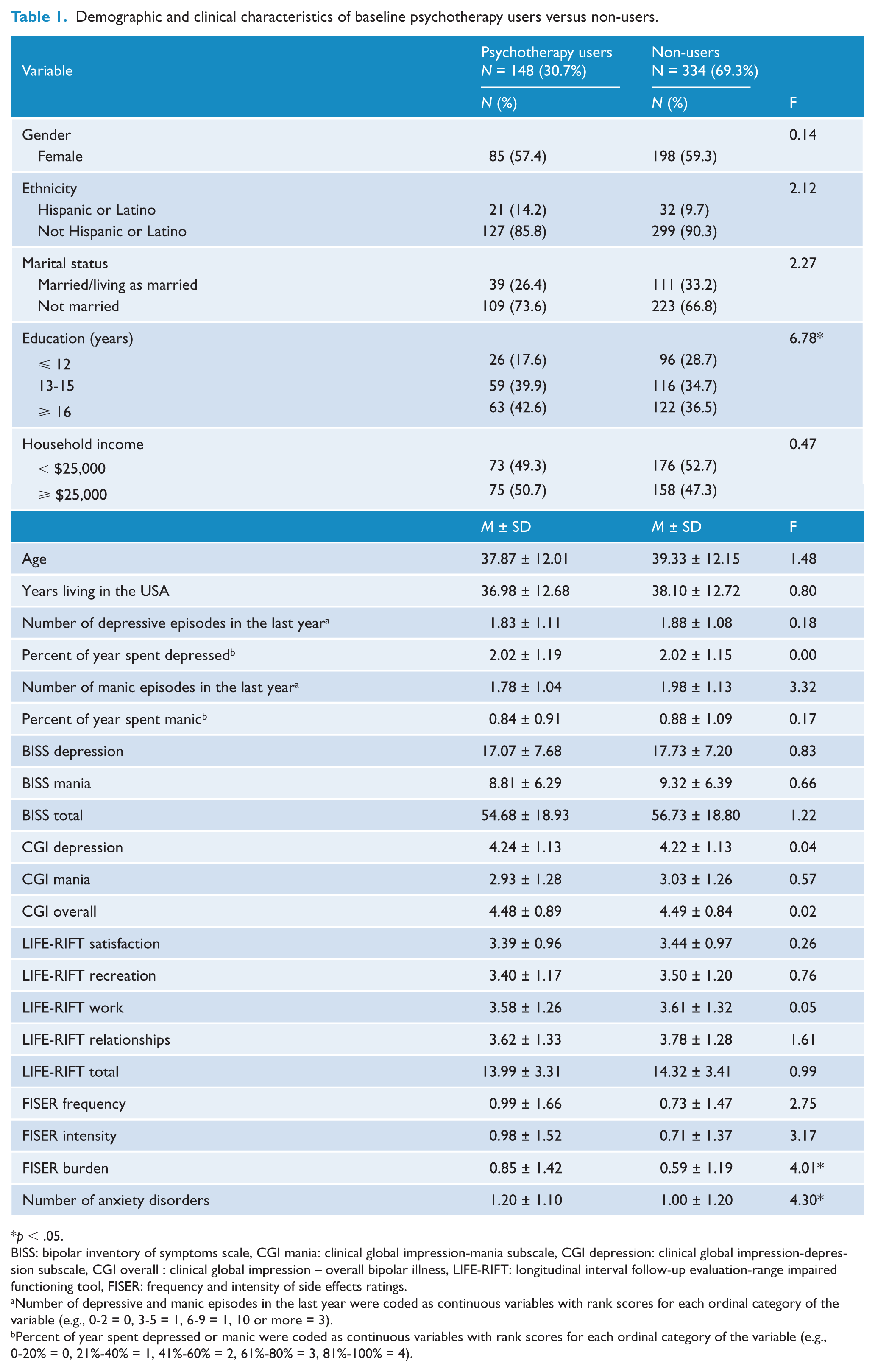
Table 1 shows the participants’ demographic and clinical characteristics. Logistic regressions revealed that psychotherapy users did not differ from non-users in most of the baseline characteristics (p’s > .05); however, psychotherapy users reported significantly greater side effect burden from their medications (p < .05). To further examine this finding, we fit a logistic regression model with therapy group as the dependent variable and both FISER side-effect burden and number of psychotropic medications as independent variables. The inclusion of the number of medications in the model caused FISER side-effects burden coefficient to become non-significant, p=.99. Therefore, the significant group difference in the self-report of side effect burden was mostly due to the group difference in the number of psychotropic medications.
Demographic and clinical characteristics of baseline psychotherapy users versus non-users.
p < .05.
BISS: bipolar inventory of symptoms scale, CGI mania: clinical global impression-mania subscale, CGI depression: clinical global impression-depression subscale, CGI overall : clinical global impression – overall bipolar illness, LIFE-RIFT: longitudinal interval follow-up evaluation-range impaired functioning tool, FISER: frequency and intensity of side effects ratings.
Number of depressive and manic episodes in the last year were coded as continuous variables with rank scores for each ordinal category of the variable (e.g., 0-2 = 0, 3-5 = 1, 6-9 = 1, 10 or more = 3).
Percent of year spent depressed or manic were coded as continuous variables with rank scores for each ordinal category of the variable (e.g., 0-20% = 0, 21%-40% = 1, 41%-60% = 2, 61%-80% = 3, 81%-100% = 4).
The two groups significantly differed in education, such that participants with at least some college compared to only a high school diploma were significantly more likely to be psychotherapy users (p’s < .02).
Psychiatric and medical comorbidities
Psychotherapy users compared to non-users were significantly more likely to have a moderate to high suicide risk (36.5% versus 25.7%; p = .02), and at least one anxiety disorder (65.5% versus 53.9%; p=.02) and a greater number of anxiety disorders (M = 1.2, SD = 1.1 versus M = 1.0, SD = 1.2; F=4.3; p = .038). Psychotherapy users were taking more psychotropic medications (M = 2.1, SD = 1.5) than non-users (M = 1.7, SD = 1.0; p < .03). There were no other group differences on comorbidity prevalence (p’s > .07, data not shown).
Clinical outcomes over the study by psychotherapy group
Mixed effects regression models showed no differences in illness severity, quality of life, bipolar symptoms, and medication side effects over time between psychotherapy users and non-users (all p’s > .49). There was also no difference in time to recovery between the two groups (log-rank χ2 (1) = 2.3, p = .13).
Baseline clinical and demographic features of individuals who used prior psychotherapy and pharmacotherapy, pharmacotherapy only, psychotherapy only, or neither
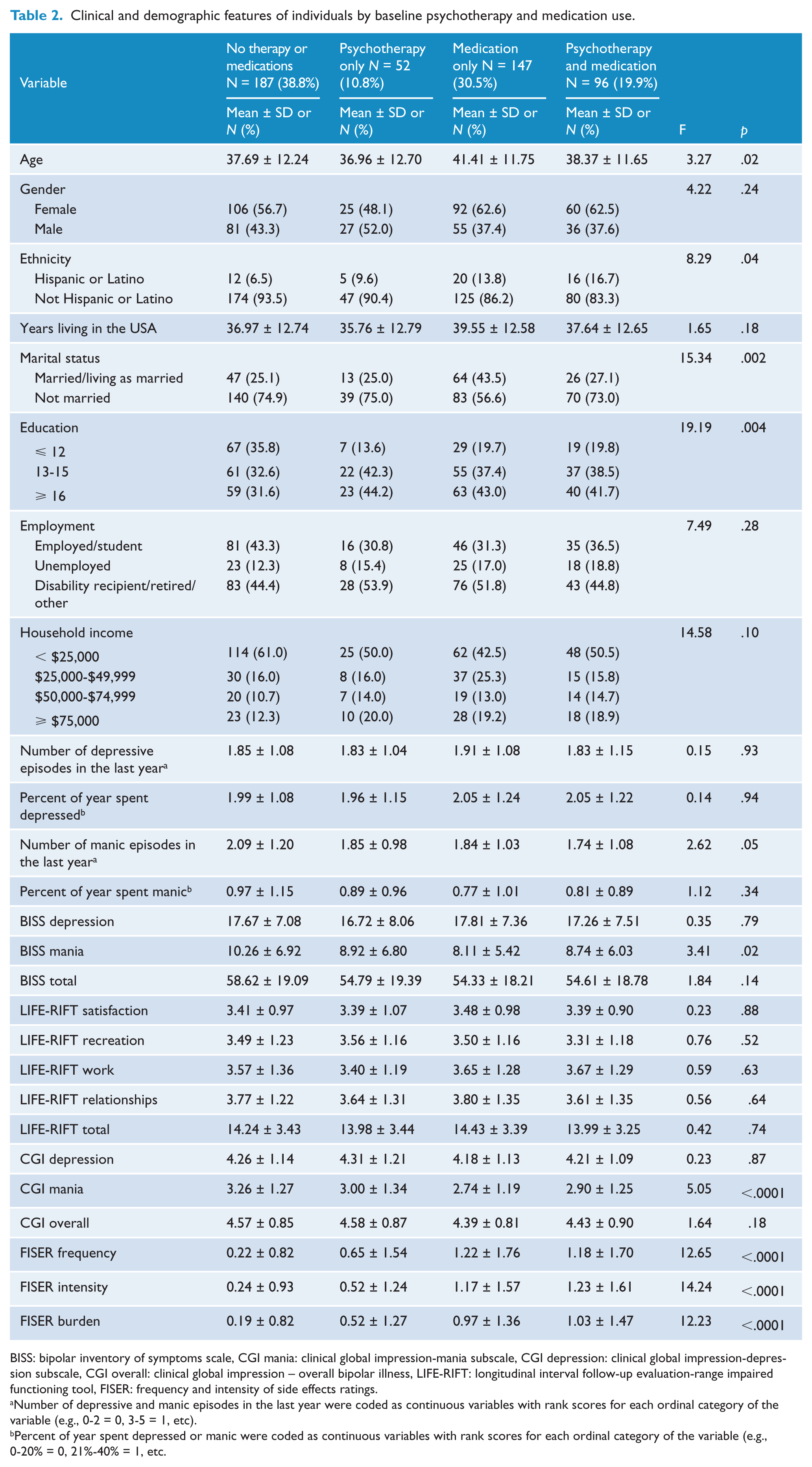
ANOVAs revealed that participants not utilizing medications or psychotherapy had greater mania symptom severity, were younger, and less educated than medication only users (both p’s=< .05). Medication only users were more likely to be married than the other participants (p’s < .01). Participants using both medications and psychotherapy were significantly more likely to be Hispanic or Latino than those not using either (p < .05). See Table 2 for a summary of these comparisons.
Clinical and demographic features of individuals by baseline psychotherapy and medication use.
BISS: bipolar inventory of symptoms scale, CGI mania: clinical global impression-mania subscale, CGI depression: clinical global impression-depression subscale, CGI overall: clinical global impression – overall bipolar illness, LIFE-RIFT: longitudinal interval follow-up evaluation-range impaired functioning tool, FISER: frequency and intensity of side effects ratings.
Number of depressive and manic episodes in the last year were coded as continuous variables with rank scores for each ordinal category of the variable (e.g., 0-2 = 0, 3-5 = 1, etc).
Percent of year spent depressed or manic were coded as continuous variables with rank scores for each ordinal category of the variable (e.g., 0-20% = 0, 21%-40% = 1, etc.
Discussion
Less than one-third of participants reported utilized psychotherapy within 90 days of baseline, and compared to non-users tended to have greater overall impairment (i.e., higher overall medication side effects, increased risk of suicide, and more anxiety). Interestingly, participants not utilizing medications or psychotherapy were younger and less educated than medication only users. Unexpectedly, psychotherapy (while taking study medications) did not improve clinical outcomes over the study duration. This study highlights the under-utilization of psychotherapy services in individuals with bipolar disorder despite evidence that adjunctive psychotherapies are effective for mood stabilization and the prevention of relapses (Sylvia et al., 2008). These data support similar findings from other studies (Evans-Lacko et al., 2011; Schaffer et al., 2006; Weiss et al., 2000), but add to our understanding of the characteristics of bipolar patients who utilize psychotherapy services (e.g., those with higher anxiety and suicide risk). We expected that more severe pathology would be associated with psychotherapy utilization as these individuals may either be more likely to be referred to treatment or to seek out additional services. Using the same rationale, psychotherapy users had a higher frequency of medication use than psychotherapy non-users (explaining their higher medication side effect burden). Alternatively, psychotherapy non-users’ lower frequency of medication use may be explained by individuals not currently seeking treatment. Less educated and younger individuals appear to be less likely to utilize psychotherapy services, perhaps due to financial constraints or not recognizing the benefit of psychotherapy. Unexpectedly, psychotherapy was associated with a lower rate of metabolic syndrome, which warrants further research. It may be that psychotherapy is increasing individuals’ awareness of their overall health, which could also explain why they are reporting more side effect burden.
We were also surprised to see that adjunct psychotherapy did not improve outcomes over the study duration. We think that this may be due to our sample being treatment-resistant given their years of illness and number of prior episodes CBT for severe and recurrent bipolar disorders (Scott et al., 2006). This was highlighted in our main outcome paper from this trial, since we did not find any group differences between the study medications, even with optimized, personalized treatment for bipolar disorder (Nierenberg et al., in press).
Our findings should be evaluated in the context of methodical limitations. First, we did not have data on different types of psychotherapy (e.g., cognitive behavioral therapy, family-focused therapy, etc) due to the limitations of the Cornell Services Index. Our definition of psychotherapy was also overly inclusive, rather than too narrow, to have the most robust effect to detect any differences in order to inform future studies. Further, we could not examine frequency of psychotherapy utilization and clinical outcomes due to a low variability in psychotherapy frequency. Generalizability may be limited by use of participants recruited for a comparative effectiveness study of lithium versus quetiapine who may have had a reductionistic view of bipolar disorder being responsive to somatic rather than psychological or combined interventions. Finally, as mentioned previously, we did not do any corrections for multiple comparisons as this paper is intended to be a hypothesis-generating paper.
This study furthers our understanding of the characteristics of patients with bipolar disorder who underutilize psychotherapy services as a treatment modality. Specifically, younger, less educated individuals as well as those with less illness and side effect burden might benefit from more encouragement to attend psychotherapy. In summary, despite the well-documented efficacy of adjunctive psychotherapy for bipolar disorder (Miklowitz, 2008b), it is an under-utilized treatment modality in this illness.
Footnotes
Declaration of interest
Dr Sylvia was a shareholder in Concordant Rater Systems and has served as a consultant for United Biosource Corporation, Clintara, Bracket, and Clinical Trials Network. She has received grant/research support from NIMH, PCORI, and AFSP. She has received Honoraria from MGH Psych Academy. She receives royalties from New Harbinger.
Dr Thase has been an advisor/consultant: to Alkermes, AstraZeneca, Bristol-Myers Squibb, Eli Lilly, Forest Laboratories, GlaxoSmithKline, Janssen Pharmaceuticals, Lundbeck, MedAvante, Merck, Mylan, Neuronetics, Otsuka, Pamlab, PharmaNeuroboost, Pfizer, Rexahn, Roche, Shire, Sunovion, Supernus, Takeda, Teva, Cerecor, Inc, Dey Pharma, LP, Gerson Lehman Group, Guidepoint Global, Novartis, Ortho-McNeil Pharmaceuticals, and Transcept Pharmaceuticals as well as the US Food and Drug Administration and the National Institute of Mental Health. During the same time frame, Dr. Thase has received honoraria for talks from AstraZeneca, Bristol-Myers Squibb, Eli Lilly, Merck, and Pfizer and he has received research grants from Alkermes, AstraZeneca, Eli Lilly, Forest, GlaxoSmithKline, Otsuka, PharmaNeuroboost, and Roche, as well as the National Institute of Mental Health and the Agency for Healthcare Research and Quality. Dr. Thase has also received royalties from the American Psychiatric Association, Guilford Publications, Herald House, and W.W. Norton & Company, Inc. He has equity holdings in MedAvante, Inc.
Dr Reilly-Harrington receives royalties from Oxford University Press, the American Psychological Association, and New Harbinger. She serves as a consultant for United Biosource Corporation, Clintara, and was a shareholder in Concordant Rater Systems.
Ms Salcedo reports no conflict of interest.
Dr Brody has received salary support over the past 3 years from grants funded by Forrest, Agency for Healthcare Quality and Research, and Pritzker neuropsychiatric disorders research consortium.
Dr Kinrys has received research support from Astra-Zeneca, Bristol-Myers Squibb Company, Cephalon, Elan Pharmaceuticals, Eli Lilly & Company, Forest Pharmaceuticals Inc., GlaxoSmithkline, Sanofi/Synthelabo, Sepracor Inc., Pfizer Inc, UCB Pharma, and Wyeth-Ayerst Laboratories. He has been an advisor or consultant for Astra-Zeneca, Cephalon, Eli Lilly & Company, Forest Pharmaceuticals Inc., GlaxoSmithkline, Janssen Pharmaceutica, Pfizer Inc, Sepracor Inc., UCB Pharma, and Wyeth-Ayerst Laboratories. Dr. Kinrys has been a speaker for Astra-Zeneca, Forest Pharmaceuticals Inc., GlaxoSmithkline, Sepracor Inc., and Wyeth-Ayerst Laboratories.
Dr Kemp has served on the speakers/advisory boards for AstraZeneca, Sunovian, Takeda, and Lundbeck. He is a consultant for Bristol-Myers Squibb, Teva, Corcept, and Janssen. He has grant/research support from NARSAD, Cleveland Foundation, and the Depressive and Bipolar Alternative Treatment Foundation. His spouse is a minor stockholder for Sanofi and Abbott.
Dr Shelton has been a consultant for Bristol-Myers Squibb Company; Cerecor, Inc.; Cyberonics, Inc.; Eli Lilly and Company; Forest Pharmaceuticals; Janssen Pharmaceutica; Medtronic, Inc.; Pamlab, Inc.; Pfizer, Inc.; Ridge Diagnostics; and Takeda Pharmaceuticals. He has grant support from Assures Health; Bristol-Myers Squibb; Eli Lilly and Company; Elan, Corp.; Euthymics Bioscience; Forest Pharmaceuticals; Janssen Pharmaceutica; Jazz Pharmaceuticals; Naurex, Inc.; Novartis Pharmaceuticals; Otsuka Pharmaceuticals; Pamlab, Inc.; Pfizer, Inc.; Repligen, Corp.; Ridge Diagnostics; St. Jude Medical, Inc.; Takeda Pharmaceuticals.
Dr McElroy is a consultant to or member of the scientific advisory boards of Alkermes, Bracket, F. Hoffman-La Roche Ltd, Corcept, MedAvante, Shire, Sunovion, and Teva. She is a principal or co-investigator on studies sponsored by the Agency for Healthcare Research & Quality (AHRQ), Alkermes, AstraZeneca, Cephalon, Eli Lilly and Company, Marriott Foundation, National Institute of Mental Health, Orexigen Therapeutics, Inc., Shire, Takeda Pharmaceutical Company Ltd., and Transcept Pharmaceutical, Inc. She is also an inventor on United States Patent No. 6,323,236 B2, Use of Sulfamate Derivatives for Treating Impulse Control Disorders, and along with the patient’s assignee, University of Cincinnati, Cincinnati, Ohio, has received payments from Johnson & Johnson, which has exclusive rights under the patent.
Dr Kocsis has received research grants and contracts from AHRQ, NIMH, NIDA, Burroughs Wellcome Trust, Pritzker Consortium, Elan, Takeda, Forest, Astra Zeneca, Roche. He is on the speaker’s bureau at Pfizer and Merck and on the advisory board at Corcept.
Dr Bobo reports no conflict of interest.
Dr Kamali has received research support from Janssen Pharmaceuticals.
Dr McInnis has received grants for research support from NIMH, the Heinz C Prechter Research Fund, and the Michigan Institute for Clinical Health Research (MICHR). MM has received consulting income from the Qatar National Research Foundation and Merck Pharmaceuticals.
Dr Friedman received grant support from NIMH, AHRQ, Novartis, St. Jude Medical, Medtronics, Repligen, Astra-Zeneca, Roche, and Takeda. He receives royalties from Springer. He has been a consultant for PamLab.
Dr Tohen was an employee of Lilly (1997 to 2008) and has received honoraria from or consulted for Abbott, AstraZeneca, Alkermes, Bristol Myers Squibb, GlaxoSmithKline, Lilly, Johnson & Johnson, Otsuka, Merck, Sunovion, Forest, Roche, Elan, Lundbeck, Teva, Pamlab, Wyeth and Wiley Publishing; his spouse was a full time employee at Lilly (1998-2013).
Dr Bowden is a research collaborator with Elan and a consultant with Teva, He has no participation with speaker bureaus, nor does he or his wife hold any equity position in any biomedical or pharmaceutical corporation.
Dr Ketter had the following financial interests/arrangements or affiliations that could be perceived as real or apparent conflicts of interest in the past three years: Grant/Research Support from the Agency for Healthcare Research and Quality, AstraZeneca Pharmaceuticals LP, Cephalon Inc., Eli Lilly and Company, Pfizer Inc., and Sunovion Pharmaceuticals; Consultant Fees from Allergan, Inc., Avanir Pharmaceuticals, Bristol-Myers Squibb Company, Cephalon Inc., Forest Pharmaceuticals, Genetech, Janssen Pharmaceutica Products, LP, Merck & Co., Inc., Sunovion Pharmaceuticals, Teva Pharmaceuticals; Lecture Honoraria from Abbott Laboratories, Inc., AstraZeneca Pharmaceuticals LP, GlaxoSmithKline, and Otsuka Pharmaceuticals; and Publication Royalties from American Psychiatric Publishing, Inc. In addition, Dr. Ketter’s spouse is an employee of and holds stock in Janssen Pharmaceuticals.
Dr Singh has been a speaker for BMS, Merck, and Sunovion. He has received research support from Novartis and Astra Zeneca.
Dr Calabrese has received federal funding from the Department of Defense, Health Resources Services Administration, and the National Institute of Mental Health. He has received research support from Abbott, AstraZeneca, Bristol-Myers Squibb, Cephalon, Cleveland Foundation, Eli Lilly, GlaxoSmithKline, Janssen, Lundbeck, NARSAD, Repligen, Stanley Medical Research Institute, Takeda, and Wyeth. He has consulted to or served on advisory boards of Abbott, AstraZeneca, Bristol-Meyers Squibb, Cephalon, Dainippon Sumitomo, EPI-Q, Inc., Forest, France Foundation, GlaxoSmithKline, Janssen, Johnson and Johnson, Lundbeck, Merck, Neurosearch, OrthoMcNeil, Otsuka, Pfizer, Repligen, Schering-Plough, Servier, Solvay, Supernus, Synosia, Takeda, and Wyeth. Dr. Calabrese has provided CME lectures supported by AstraZeneca, Bristol-Myers Squibb, France Foundation, GlaxoSmithKline, Janssen, Johnson and Johnson, Merck, Sanofi Aventis, Schering-Plough, Pfizer, Solvay, and Wyeth.
Dr Nierenberg is a consultant for Abbott Laboratories, Astra Zeneca, Basilea, BrainCells Inc., Brandeis University, Bristol-Myers Squibb, Cephalon, Corcept, Eli Lilly & Co., Forest, Genaissance, GlaxoSmithKline, Innapharma, Janssen Pharmaceutica, Jazz Pharmaceuticals, Lundbeck, Merck, Novartis, PamLabs, PGx Health, Pfizer, Ridge Diagnostics, Roche, Sepracor, Schering-Plough, Shire, Somerset, Sunovion, Takeda, Targacept, and Teva. He is a stakeholder in Appliance Computing, Inc. (MindSite); Brain Cells, Inc., InfoMed (potential share of income). He receives research support from AHRQ, Bristol-Myers Squibb, Cederroth, Cyberonics, Elan, Forest Pharmaceuticals, GlaxoSmithKline, Janssen Pharmaceutica, LichtwerPharma, Eli Lilly, Mylin (formerly Dey Pharmaceuticals), NARSAD, NIMH, Pamlabs, Pfizer, Shire, Stanley Foundation, and Wyeth-Ayerst. Honoraria include MGH Psychiatry Academy in the past 3 years (Prior to 3 years ago, honoraria from Bristol-Myers Squibb, Cyberonics, Forest Pharmaceuticals, GlaxoSmithKline, Eli Lilly, Shire, Wyeth-Ayerst). Dr. Nierenberg receives other income from legal case reviews for CRICO, MBL Publishing for past services as Editor-in-chief of CNS Spectrums, Slack Inc. for services as Associate Editor of Psychiatric Annals, and Editorial Board, Mind Mood Memory, Belvior Publications. He has copyright joint ownership with MGH for Structured Clinical Interview for MADRS and Clinical Positive Affect Scale and additional honoraria from ADURS, American Society for Clinical Psychopharmacology and Zucker Hillside Hospital and Forest and Janssen, Biomedical Development, Boston Center for the Arts, University of Pisa, University of Wisconsin at Madison, University Texas Southwest at Dallas, Health New England and Harold Grinspoon Charitable Foundation and Eli Lilly and AstraZeneca, Brandeis University, International Society for Bipolar Disorder, 2nd East Asian Bipolar Forum, Mid-Atlantic Permanente Research Institute, Up-to-Date.
Mr Rabideau and Dr Elson report no conflict of interest.
Dr Deckersbach has received research support from NIMH, NARSAD, TSA, IOCDF, Tufts University, NIH, NIA, DBDAT, Janssen Pharmaceuticals, the Forest Research Institute, Shire Development Inc., Medtronic, Cyberonics, Northstar. He has received honoraria, consultation fees and/or royalties from the following: Medacorp, MGH Psychiatry Academy, BrainCellsInc.,Clintara, LLC., Systems Research and Applications Corporation, Boston University, Tufts University, the Catalan Agency for Health Technology Assessment and Research, the National Association of Social Workers Massachusetts, Massachusetts Medical Society, and Oxford University Press.
Funding
This study was funded by the Agency for Healthcare Research and Quality (AHRQ), 1R01HS019371-01
