Abstract
Objective:
Antidepressant use is widespread. While weight gain is a commonly reported side-effect of antidepressant use and has the potential to affect population health, there is little large-scale population-based evidence on the issue, particularly for long-term use (⩾12 months). The aim of this study is to investigate the association between antidepressant use and weight change, including whether this relationship varies according to antidepressant class, recency of use, duration of use and dose.
Methods:
Annual percentage weight change was calculated from self-reported weight at two time-points from 20,751 participants aged ⩾45 years from the 45 and Up Study – a population-based cohort study from New South Wales, Australia. Antidepressant use, ascertained from linked pharmaceutical data, from 19 months before baseline until end of follow-up (mean = 3.3 years of follow-up), was categorised as current, past-only, non-persistent or non-use. The association between antidepressant use and weight change was modelled using linear and multinomial logistic regressions and according to antidepressant class, recency, duration and dose.
Results:
Antidepressants were dispensed to 23% of participants (n = 4748) during the study period. Current antidepressant users were significantly more likely to gain >3% of their body weight annually than non-users (adjusted relative risk ratio = 1.19; 95% confidence interval: [1.03, 1.38]); the risk increased with increasing dose among current users (p[trend] = 0.003). Risk of weight gain did not vary significantly according to antidepressant class, recency or duration of use; however, statistical power was limited. No significant associations were found between antidepressant use and weight loss.
Conclusion:
Current antidepressant use was associated with modest but statistically significant annual gains in weight, with similar effects observed across the different classes of antidepressants used.
Introduction
Use of antidepressant medications is widespread and has been increasing over time in Organisation for Economic Co-operation and Development (OECD) countries (OECD, 2013). Between 2000 and 2011, dispensings of antidepressants in Australia increased from an estimated 45 defined daily doses (DDD) per 1000 people per day (1000/day) to 89 DDD/1000/day (OECD, 2013). Weight gain is a common complaint among antidepressant users (Papakostas, 2008) and can contribute to medication discontinuation (Fava, 2000). At a population level, weight gain associated with antidepressant use is important in the context of the high global prevalences of both obesity (World Health Organization, 2013) and depression (World Health Organization, 2012). High body mass index (BMI) accounts for a large burden of disease globally (Lim et al., 2012). Small increases in BMI are associated with increased morbidity, mortality and hospitalisation costs (Korda et al., 2015; Whitlock et al., 2009) and, over time, can contribute to larger increases in BMI.
Results from published clinical trials have shown that short-term antidepressant use (4–12 weeks) is associated with weight gain (Serretti and Mandelli, 2010). These effects may vary depending on the class of antidepressant medication. While there is evidence that tricyclic antidepressants (TCAs) and monoamine oxidase inhibitors are associated with weight gain (Schwartz et al., 2004; Serretti and Mandelli, 2010), it was initially thought that use of selective-serotonin reuptake inhibitors (SSRIs) was associated with no weight change or with weight loss (Fava, 2000). However, there is emerging but sparse evidence that this effect is transient and that long-term use (⩾12 months) of SSRIs may be associated with weight gain (Schwartz et al., 2004). Population-based studies are well placed to assess the weight effects of long-term use of antidepressants, which are often beyond the scope of clinical trials (Serretti and Mandelli, 2010).
We were able to locate only three prospective population studies that have examined the relationship between antidepressant use and weight change, finding greater mean weight gain in users compared to non-users (Kivimaki et al., 2010; Patten et al., 2011), although the absolute average weight gain was small (~0.1–0.3 kg/year) (Blumenthal et al., 2014; Kivimaki et al., 2010; Patten et al., 2011). Moreover, there are concerns that use of ‘average’ weight gain as a measure of change in body weight may obscure important directional evidence (Paige et al., 2014).
It would be expected that if antidepressant use is causally related to weight gain, then more recent use, longer durations of use and higher doses would be associated with a greater likelihood of weight gain. However, it is unclear from population-based studies whether weight change varies by these factors.
Using detailed longitudinal survey data, linked to administrative pharmaceutical claims, this population-based study aimed to expand on current evidence by investigating the association between antidepressant use and weight change, including whether this relationship differs by antidepressant class, recency and duration of use, and dose.
Methods
Data sources and study population
We undertook analyses using data from the Sax Institute’s 45 and Up Study baseline questionnaire linked to follow-up questionnaire data and to medication data from Pharmaceutical Benefits Scheme (PBS) records.
Briefly, the 45 and Up Study is an Australian cohort of 267,153 men and women aged 45 years or over from New South Wales (NSW), Australia. The study is described in detail elsewhere (Banks et al., 2008) and questionnaires can be viewed at www.45andup.org.au. Baseline questionnaires were distributed between January 2006 and December 2008. A follow-up questionnaire was distributed to the first 100,000 participants enrolled in the 45 and Up Study, and of those invited to take part, 60,404 participants (60.4%) did so, with questionnaires completed from 2010 to 2011 (1.7–5.1 years after baseline).
The PBS dataset is an administrative dataset containing information about subsidised dispensed prescription medications for the Australian population (Duckett, 2004). Individual PBS records linked to the 45 and Up Study were available from 2004. During the study period, complete records on antidepressant dispensings were only available for people who held a valid healthcare concession-card, so we restricted our analyses to 45 and Up Study participants who were concession-card holders (those with at least one concessional and no non-concessional PBS claims) during the study period. The study period was defined for each participant from 19 months before completion of the baseline questionnaire (the minimum period of time for which PBS data were available for all participants) until completion of the follow-up questionnaire.
Only participants who completed both the 45 and Up Study baseline questionnaire and the follow-up questionnaire and had linked PBS records during the study period were included in the present analyses. Consistent with previous studies of weight change (Ball et al., 2002; Brown et al., 2005), we excluded people with a history of cancer (except for non-melanoma skin cancer), self-reported on the baseline questionnaire. Participants who self-reported holding a Department of Veteran’s Affairs (DVA) card (n = 649, 1%) were also excluded, as these people have access to a broader range of subsided medications under a separate Australian Government programme.
The Sax Institute linked the baseline 45 and Up questionnaire data, the follow-up questionnaire data and the PBS data, using a confidential unique identifier. The PBS data were supplied by the Australian Government Department of Human Services.
Measurements
Weight change
Annual change in body weight was the primary outcome, calculated as self-reported weight at follow-up minus self-reported weight at baseline divided by follow-up time in years. Annual percentage weight change was calculated as annual weight change divided by weight at baseline, multiplied by 100.
Participants were categorised into one of three groups – weight loss (defined as >3% weight loss on average per year), weight maintenance (weight change ⩽3% on average per year) and weight gain (>3% weight gain on average per year). A 3% cut-point is considered large enough to account for measurement error and normal weight variation (Stevens et al., 2006). Weight change was modelled as both a continuous and categorical variable, as previous work suggests that modelling weight change as average continuous change only may not allow the detection of important effects on weight in specific categories (Paige et al., 2014).
Antidepressant use
Antidepressant use was based on dispensings of antidepressant medications identified from the PBS dataset with Anatomical Therapeutic Chemical (ATC) classification codes beginning with N06A (World Health Organization Collaborating Centre for Drug Statistics Methodology, 2013). Study participants were categorised as current users, past-only users, non-persistent users and non-users. As PBS data only provide information about dispensing, not actual use of the medications, current and past-only users only included those with two or more consecutive dispensings of an antidepressant (Andrade et al., 2006; Eaddy et al., 2005; Paige et al., 2015). Specifically, current users were defined as those who had ⩾2 consecutive antidepressant dispensings at any time between the baseline and follow-up surveys. Past-only users were defined as participants who had ⩾2 consecutive antidepressant dispensings within the 19-month period before the baseline questionnaire but not between the two surveys. Consecutive dispensings were defined as where the time between the dispensings was less than or equal to the maximum standard supply period (based on the pack size obtained from the PBS website; e.g. if pack size is 30 tablets, the maximum standard supply period is 30 days) and an additional refill period of 8 days. Non-persistent users were defined as participants who had one or more non-consecutive dispensings of any antidepressant within the study period and no periods of consecutive antidepressant dispensings. Non-users were defined as those who were not dispensed any antidepressant in the study period.
Current users were further classified by class of antidepressant used, recency of use, duration of use and dose. Class of antidepressant was categorised as follows: SSRI (ATC codes N06AB02–N06AB10), TCA (N06AA01–N06AA23), ‘other’ antidepressant (N06AF01–N06AF06, N06AG02–N06AG03 and N06AX01–N06AX26) or more than one class.
Because participants were followed up for a mean of 3.3 years, we also examined how recently current users had been dispensed an antidepressant, based on the time between the last dispensing of an antidepressant and completion of the follow-up questionnaire, categorised as <12 months and ⩾12 months.
Duration of antidepressant use during follow-up was calculated as total number of days of dispensed antidepressants (assuming one tablet is used per day), categorised as <12 months and ⩾12 months based on definitions of acute and long-term treatment used in previous studies (Schwartz et al., 2004).
Dose was calculated as the average number of dispensed DDD per day. DDD is a World Health Organization classification system which has been validated (Lau et al., 1997) and is defined as ‘the assumed average maintenance dose per day for a drug used for its main indication in adults’ (World Health Organization Collaborating Centre for Drug Statistics Methodology, 2009). Dispensed DDD for each antidepressant dispensing was calculated as the strength (mg) of the dispensed antidepressant multiplied by the pack size divided by the DDD. This dispensed DDD was then summed for each individual and divided by follow-up time to obtain the average dispensed DDD per day.
Potential confounding factors
Factors with the potential to confound the antidepressant–weight change relationship were identified a priori from the literature (Ball et al., 2002; Blazer et al., 2005; Brown et al., 2005; Harris et al., 2011; Hollingworth et al., 2010; Paige et al., 2015). Potential confounding factors included in the final models were sex; age; education (no school certificate, school certificate, trade/certificate/diploma, or university degree); region of residence (major cities, inner regional or more remote); physical activity tertiles (based on the weighted number of reported weekly sessions of walking, moderate activity and vigorous activity (Australian Institute of Health and Welfare [AIHW], 2003)); smoking status (never, past or current); alcohol consumption (drinks/week: none, light [1–10 for men and 1–5 for women], moderate [11–35 for men and 6–20 for women] and heavy [>35 for men and >20 for women] (Power et al., 1998)); physical functioning impairment (derived from responses to Medical Outcome Scale Short Form-36 [SF-36] 10-item physical functioning scale [PF-10] (Ware, 2000), categorised as none/minor, moderate or severe); doctor-diagnosed heart disease (yes, no) or stroke (yes, no); and antipsychotic medication use (any dispensing during the study period of an item with ATC code beginning with N05A). Diet was not considered a potential confounder as it is likely to lie, at least in part, on the causal pathway between medication use and weight change.
Depression was considered to have the potential to confound the antidepressant–weight change relationship. However, limited information was available on depression, and as the relationship with antidepressant medication was assumed to be complex, difficult to quantify and collinear with exposure to antidepressants, this was explored through sensitivity analyses. Using baseline psychological distress as a proxy for depression, we conducted a sensitivity analysis adjusting for baseline psychological distress (based on responses to the Kessler 10 scale (Kessler et al., 2002) and categorised as low [score of <16], moderate [16 to <22] or high/very high [⩾22]). All potential confounders – except for region of residence (derived from the postcode obtained from Medicare data) and dispensing of antipsychotics (from PBS data) – were derived from self-reported baseline questionnaire responses.
Statistical methods
Mean weight, average weight change and the percentage of participants in each weight change category were calculated for each antidepressant user category, and the proportions of participants in each antidepressant user category were described in relation to each of the potential confounding factors. The relationship between baseline BMI and antidepressant use was examined using linear regression.
For the main analyses, examining the association between antidepressant user categories and weight change, linear regression was used for mean annual weight change and multinomial logistic regression for weight change categories. Regression models were initially adjusted for sex and age only, and then additionally adjusted for the confounding factors detailed above.
The additionally adjusted multinomial logistic models were then re-run, examining antidepressant use in current users separately according to antidepressant class, recency of use (i.e. how close to the second weight change measure that antidepressant use was estimated to occur), duration and dose. To examine whether the risk of weight gain among current users differed by these factors, differences in effect sizes between the sub-groups were tested using a weighted least squares approach (Tierney et al., 2007; Viwatwongkasem et al., 2012) and tests for trend were undertaken where appropriate. Post-diagnostic tests were used to test the assumptions of the linear regression models. Robust standard errors were used where residuals demonstrated non-normality. Influential outlying observations were excluded from the analyses (n = 7).
In contrast to odds ratios which are usually generated from logistic regression, estimates of the strength of associations from multinomial logistic regression are reported as relative risk ratios (RRRs). These RRRs express the relative risk of having the outcome compared to not having the outcome in relation to different levels of the exposure variables (StataCorp, 2011). For example, an RRR of 1.5 for current antidepressant users with weight loss can be interpreted as follows: for current antidepressant users compared to non-users, the relative risk of losing weight compared to maintaining weight is 1.5.
In all analyses, 95% confidence intervals (CIs) were generated. Analyses were performed using Stata version 12.0 and were undertaken using the Secure Unified Research Environment (Sax Institute, 2014).
Missing data
In all, 24% (4969) of participants had missing values for one or more of the confounding factors or weight at baseline or follow-up. We used multiple imputation to impute values for the missing data (see Supplementary Appendix 1 for more detail). We further re-ran the main regression models, excluding participants with missing values for weight or any of the confounding factors (complete case analysis).
Ethics approval for the 45 and Up Study as a whole was obtained from the University of New South Wales Human Research Ethics Committee. Approval for this specific project was obtained from the University of Sydney Human Research Ethics Committee and the Australian National University Human Research Ethics Committee.
Results
The final study population consisted of 20,751 participants with concessional-only PBS claims during the study period. Participants were followed up from baseline for a mean of 3.3 years, with the overall study period (from 19 months before baseline until the follow-up questionnaire) ranging from 3.3 to 6.7 years.
Summary characteristics of the study population are presented in Tables 1 and 2. Overall, 23% (n = 4748) of the sample had been dispensed at least one antidepressant during the study period, with 15% (n = 3078) classified as current users. Most current antidepressant users were long-term users who had used antidepressants for ⩾12 months (n = 2346, 76%). More females than males used antidepressants, and use decreased with increasing age, education and physical activity (Table 1).
Percentage in each antidepressant user category according to participant characteristics.
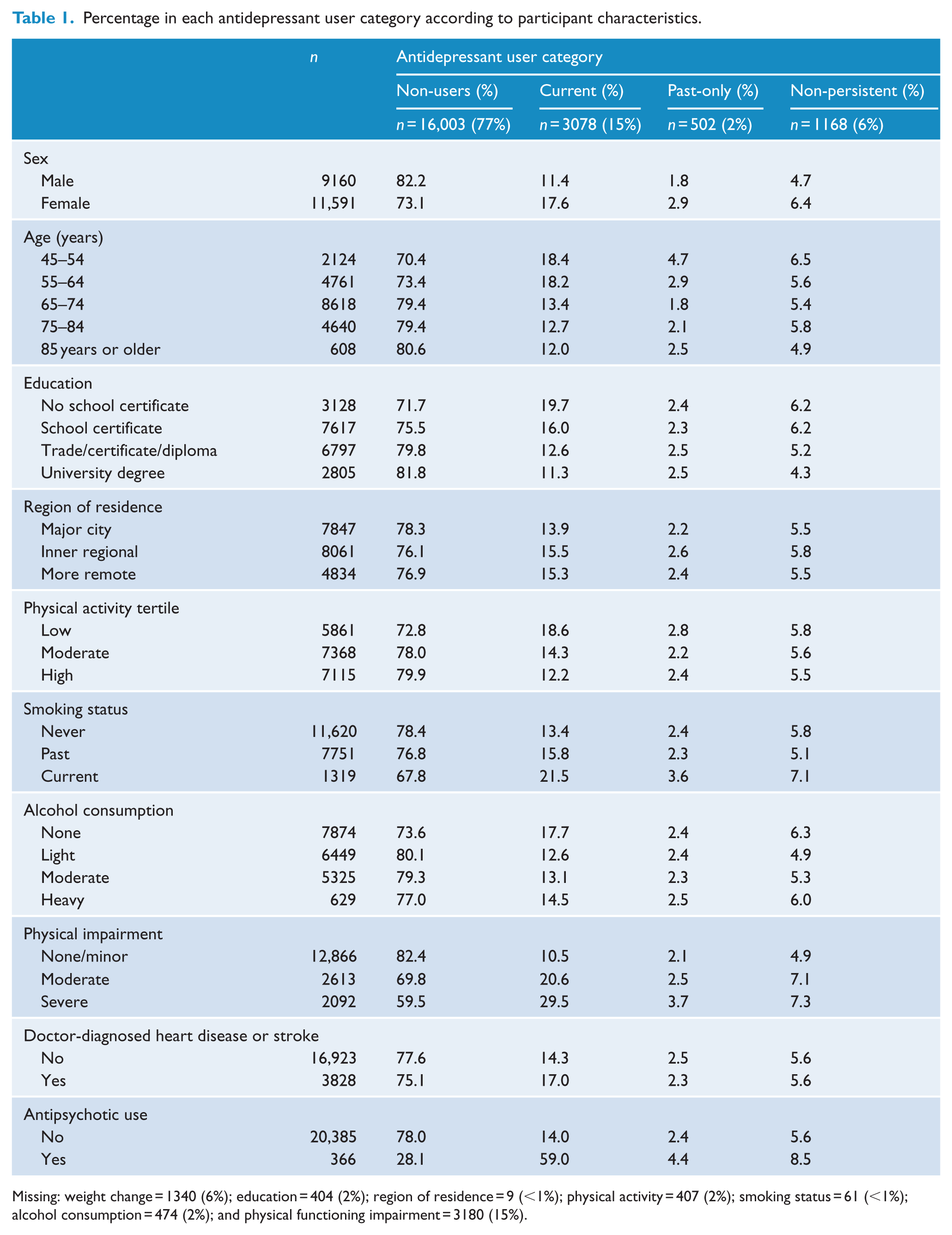
Missing: weight change = 1340 (6%); education = 404 (2%); region of residence = 9 (<1%); physical activity = 407 (2%); smoking status = 61 (<1%); alcohol consumption = 474 (2%); and physical functioning impairment = 3180 (15%).
Mean weight and weight change and proportion in each weight change category according to antidepressant user categories.
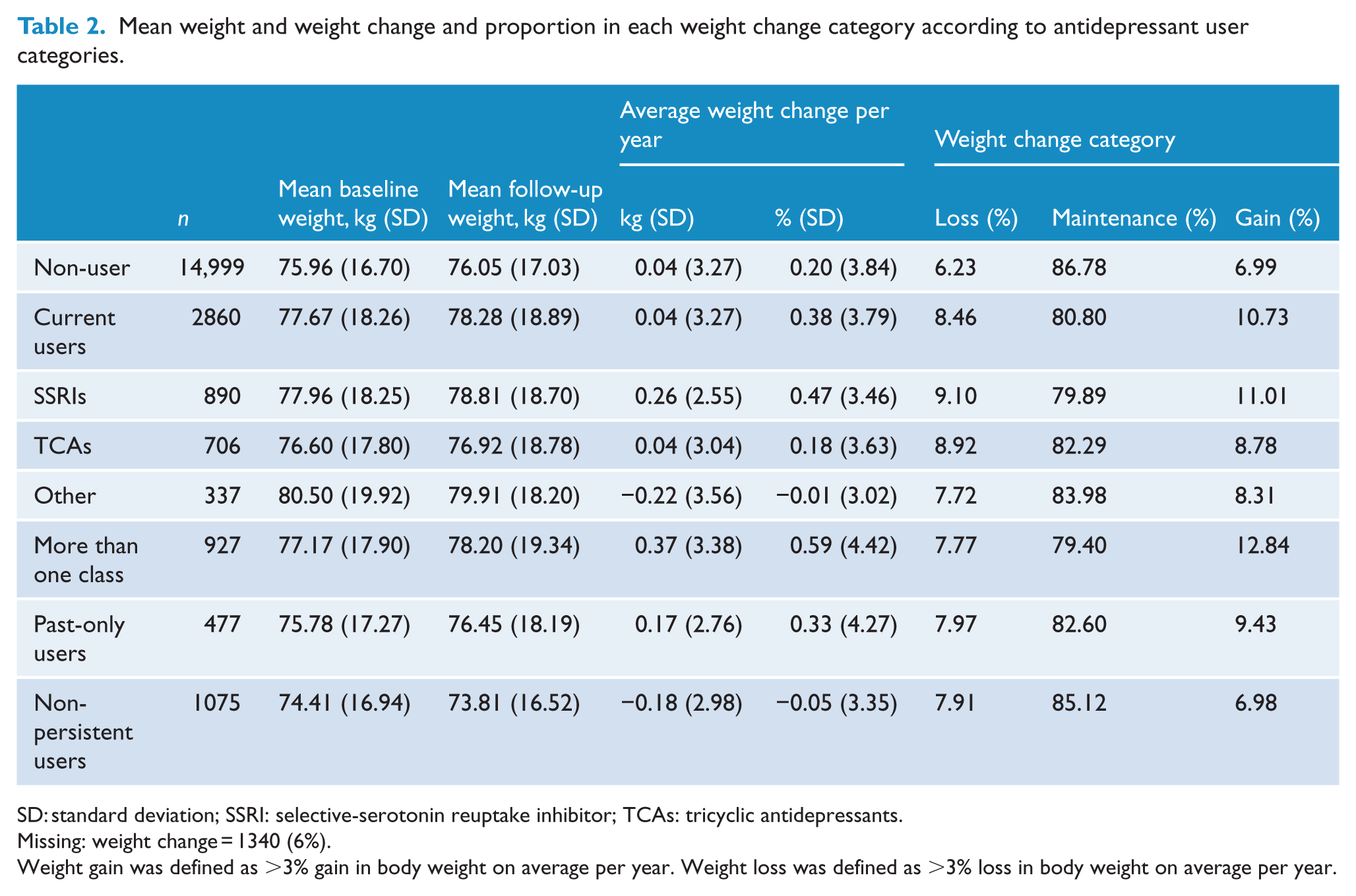
SD: standard deviation; SSRI: selective-serotonin reuptake inhibitor; TCAs: tricyclic antidepressants.
Missing: weight change = 1340 (6%).
Weight gain was defined as >3% gain in body weight on average per year. Weight loss was defined as >3% loss in body weight on average per year.
Overall, the majority of participants maintained their weight (86%, n = 16,636); 7% (n = 1300) lost >3% of their body weight on average per year and 7% (1475) gained >3% of their body weight. Average weight change per year was −0.02 kg for men and 0.12 kg for women.
Antidepressant use and baseline BMI and weight
Mean baseline BMI in non-users of antidepressants was 26.93 kg/m2. Compared to non-users, current antidepressant users had a higher baseline BMI after sex- and age-adjustment (Table 3). After further adjustment, current and non-persistent users had statistically different mean baseline BMIs compared to non-users (adjusted difference in baseline BMI of current vs non-users = 0.40 [0.18, 0.61], non-persistent vs non-users = −0.35 [−0.66, −0.03]).
Differences in baseline BMI by antidepressant use.
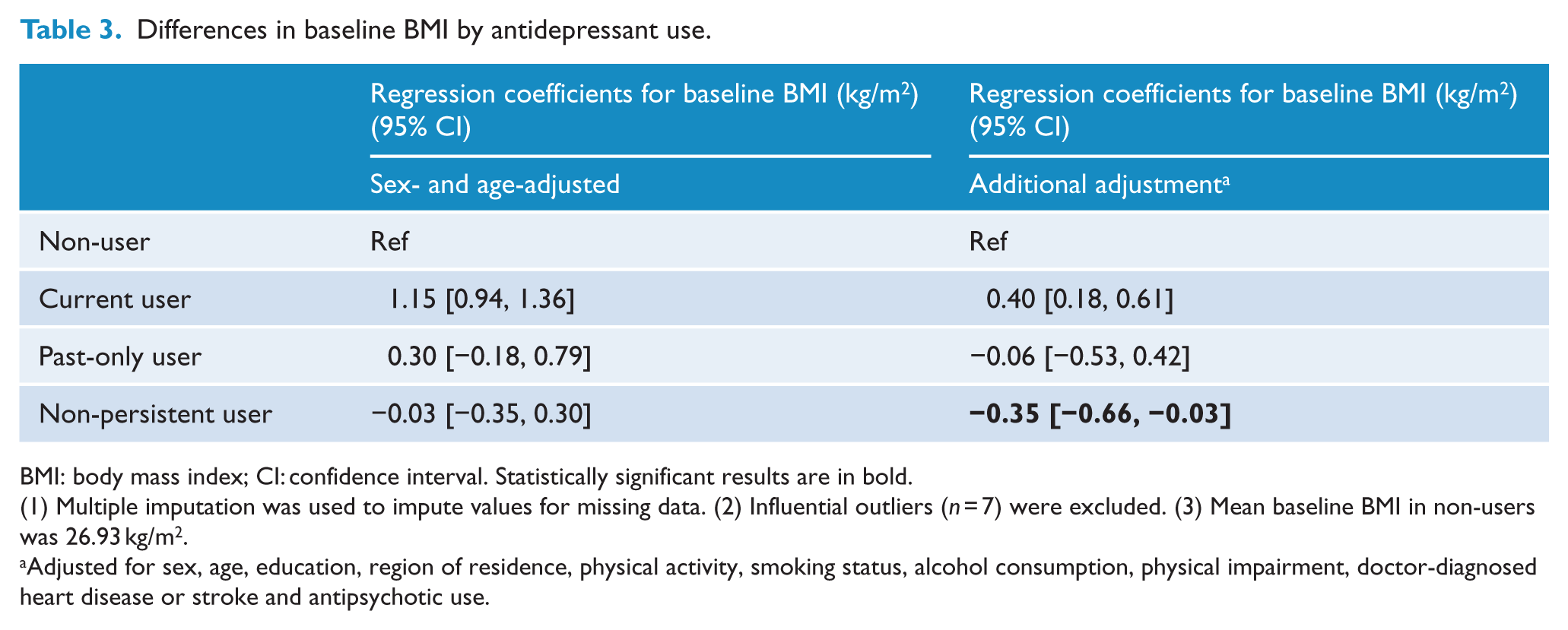
BMI: body mass index; CI: confidence interval. Statistically significant results are in bold.
(1) Multiple imputation was used to impute values for missing data. (2) Influential outliers (n = 7) were excluded. (3) Mean baseline BMI in non-users was 26.93 kg/m2.
Adjusted for sex, age, education, region of residence, physical activity, smoking status, alcohol consumption, physical impairment, doctor-diagnosed heart disease or stroke and antipsychotic use.
Antidepressant use and mean weight change
On average, non-users, current users and past-only users of antidepressants gained weight while non-persistent users lost weight (Table 2). After adjustment for potential confounding factors, there were no significant differences in average weight change per year between non-users and current users or past-only users. Compared to non-users, non-persistent antidepressant users had significantly less weight gain on average (additionally adjusted difference in relative [%] weight change = −0.26; 95% CI: [−0.50, −0.02]) (Table 4).
Differences in mean annual relative weight change by antidepressant use.
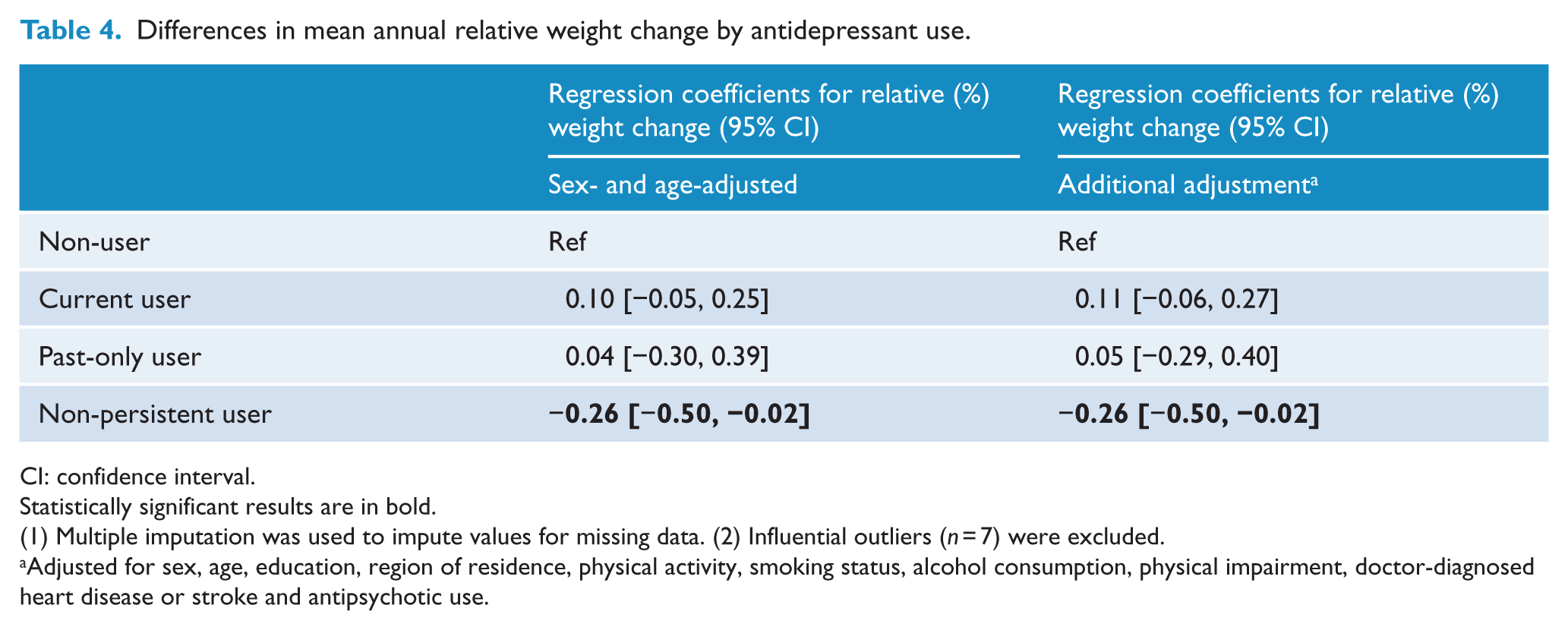
CI: confidence interval.
Statistically significant results are in bold.
(1) Multiple imputation was used to impute values for missing data. (2) Influential outliers (n = 7) were excluded.
Adjusted for sex, age, education, region of residence, physical activity, smoking status, alcohol consumption, physical impairment, doctor-diagnosed heart disease or stroke and antipsychotic use.
Antidepressant use and weight gain
Overall, 11% of current users, 10% of past-only users and 7% of non-persistent users gained >3% of their weight on average per year compared to 7% of non-users (Table 2). After adjustment for age and sex, current antidepressant users were more likely to gain weight than non-users; this relationship remained statistically significant after adjusting for additional confounders (adjusted RRR [aRRR] = 1.19 [1.03, 1.38]). Past-only users and non-persistent users were no more likely to gain weight than non-users (Table 5). Adjustment for baseline psychological distress made little material difference (<2%) to the effect sizes observed in the main study results Supplementary Tables 1 and 2).
Relative risk ratios of weight gain and weight loss compared to weight maintenance according to antidepressant use.
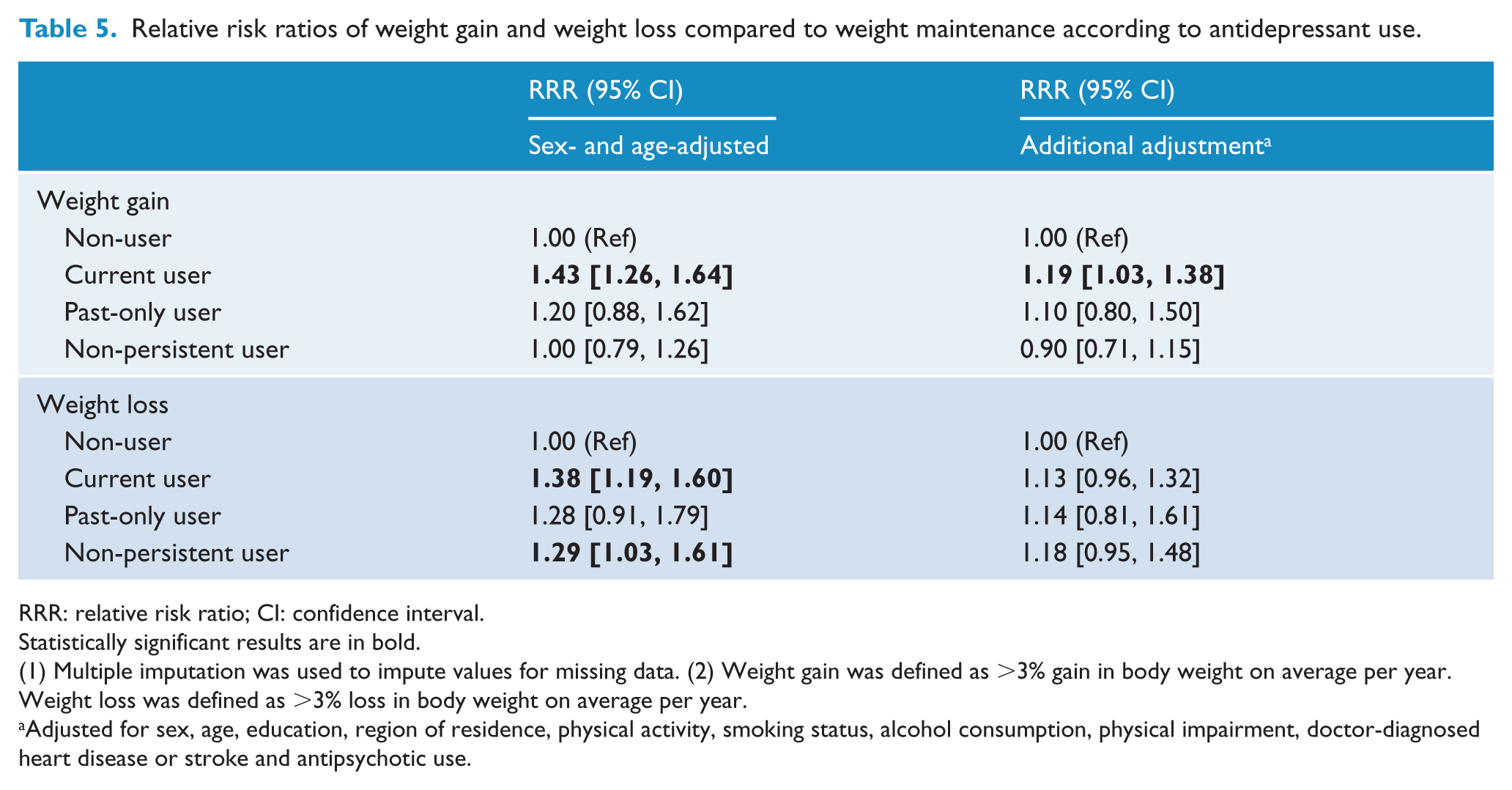
RRR: relative risk ratio; CI: confidence interval.
Statistically significant results are in bold.
(1) Multiple imputation was used to impute values for missing data. (2) Weight gain was defined as >3% gain in body weight on average per year. Weight loss was defined as >3% loss in body weight on average per year.
Adjusted for sex, age, education, region of residence, physical activity, smoking status, alcohol consumption, physical impairment, doctor-diagnosed heart disease or stroke and antipsychotic use.
The risk of weight gain among current users did not differ significantly by antidepressant class, recency or duration of use (p > 0.05, Figure 1). However, the overall pattern indicated a significantly higher risk of weight gain in users of SSRIs, more recent users and with longer durations of use, compared to non-users, for these individual categories. A dose–response relationship was observed between the average number of DDD dispensed per day and weight gain (p[trend] = 0.003), such that those dispensed >1 DDD were 54% more likely to gain weight than non-users (aRRR = 1.54 [1.18, 2.00]).
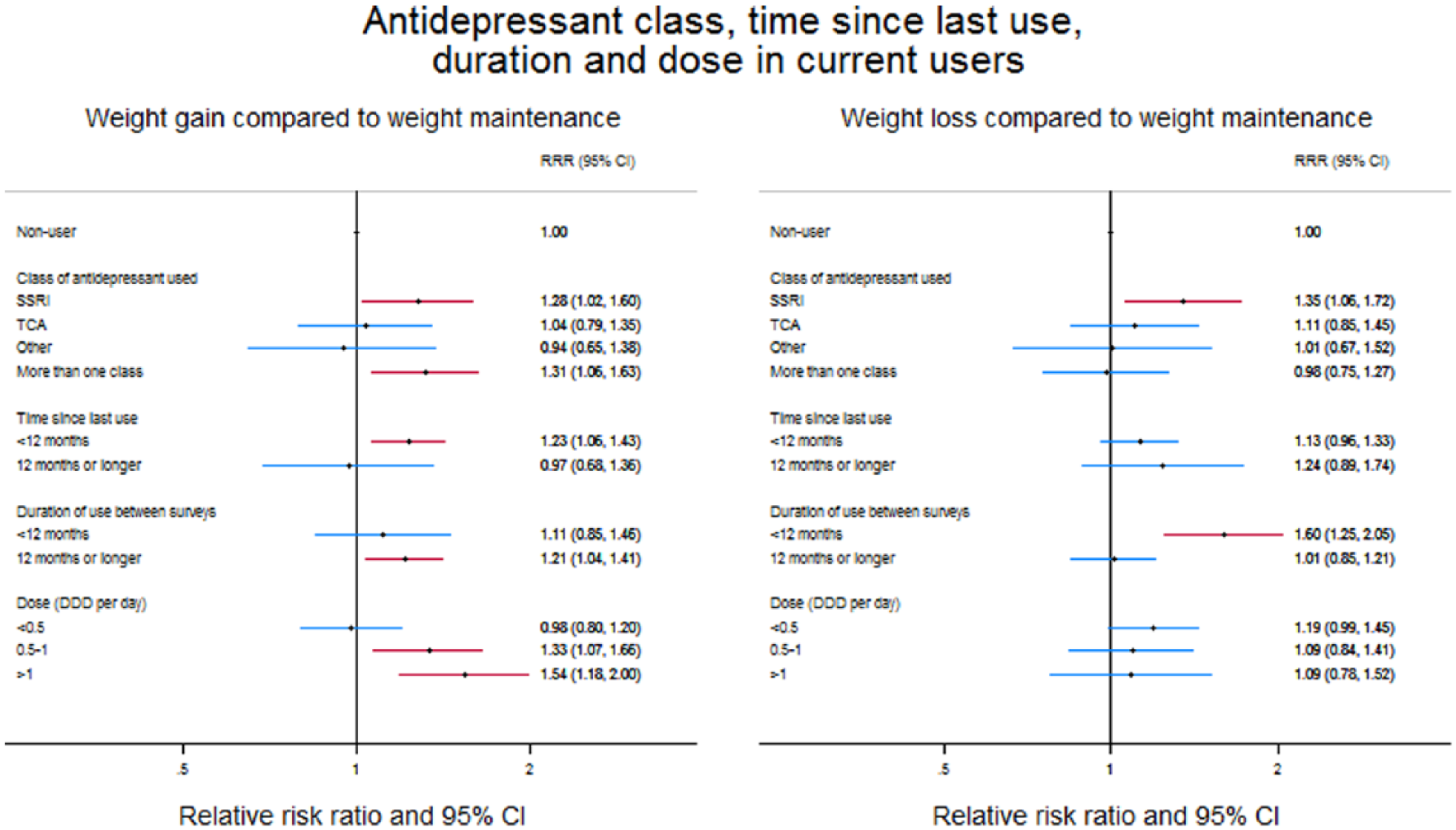
Relationship between antidepressant use and weight change according to the class of antidepressant used, recency of use, duration of use and dose, among current between survey users.
Antidepressant use and weight loss
Overall, 8% of current, past-only and non-persistent antidepressant users lost >3% of their body weight on average per year compared to 6% of non-users (Table 2). After adjusting for sex and age, current users and non-persistent users were more likely to lose weight than non-users (Table 5). However, after adjustment for additional potential confounding factors, there was no significant association between any of the antidepressant user categories and weight loss. The risk of weight loss among current users did not differ by class of antidepressant, recency of use or dose (p > 0.05, Figure 1). However, current users who had used antidepressants for <12 months during follow-up were more likely than non-users to lose weight (aRRR = 1.60 [1.25, 2.05]) and were significantly more likely to lose weight than those using for 12 months or longer (p = 0.003).
There were no material differences between the results of the main analyses, which imputed missing values, and the complete case analysis Supplementary Tables 3 and 4).
Discussion
In this large population-based study, current antidepressant use (i.e. use between the two reported weight measurements) was associated with a small but significant increased risk of weight gain, including after adjustment for a number of potential confounding factors. Compared to non-users, current users had a 19% increased risk of gaining >3% body weight on average per year, and the strength of this relationship increased with increasing dose. The risk of weight gain among current users did not differ significantly by antidepressant class, time since last use or duration of use; however, the general patterns observed for these factors were consistent with a causal relationship.
Our results are broadly consistent with previous population-based studies, which found mean weight changes of 0.11 and 0.29 kg/year among users compared to non-users (Kivimaki et al., 2010; Patten et al., 2011). Although we examined relative (%) change in weight, our analysis of average weight change showed very similar results, with current antidepressant users having a 0.11 [−0.06 to 0.27] higher average percentage weight gain compared to non-users. To address concerns that average weight change measures can obscure directional information about a risk factor–weight change relationship (Paige et al., 2014), we examined categories of weight change as well as average weight change and found a significant increased risk of weight gain in current users compared to non-users.
It is possible that the relationship we observed between antidepressant use and the risk of weight gain may be partly explained by recovery from depression or severity of depression. Nevertheless, there is evidence that antidepressants affect weight independent of their effect on mood through mechanisms such as changes to appetite regulation and metabolism (Fernstrom, 1989; Zimmermann et al., 2003). Furthermore, adjustment for baseline psychological distress in our study made little difference to the strength of the association between antidepressant use and weight change. Our findings are also consistent with those from clinical trials. We found that short-term use (<12 months compared to ⩾12 months) of antidepressants was associated with a 60% increased risk of weight loss compared to non-users. While our definition of short-term use is much longer than that considered short-term use in clinical trials (4–12 weeks), our results are broadly supportive of clinical trial findings. Results from a meta-analysis of clinical trials showed that 8 out of 17 commonly used antidepressants were significantly associated with mean weight loss during short-term use (4–12 weeks) compared to placebo (Serretti and Mandelli, 2010). Fewer antidepressant types (3/17) were significantly associated with mean weight gain compared to placebo during this time. Four of the eight antidepressants associated with short-term weight loss were SSRIs, while none of those associated with short-term weight gain were SSRIs. Although differences in definitions of short-term use make the comparison between our study and clinical trials difficult, these results suggest that the association between short-term antidepressant use and the increased risk of weight loss observed in our study may be related to use of SSRIs. Indeed, a third of current users in our study population used SSRIs only (n = 890); however, due to small sample numbers, we were unable to specifically examine current SSRI use by duration of use, so we cannot confirm this hypothesis.
Our study is larger than previous studies examining weight change in antidepressant users compared to non-users and provides some of the most reliable evidence to date on long-term use of antidepressants. We found that current long-term (⩾12 months) users were 21% more likely to gain weight than non-users. Although we found no significant difference in the risk of weight gain between those using for <12 months and those using for ⩾12 months, statistical power to detect such a difference was limited. To date, clinical trial data on long-term use of antidepressants have been scarce; of the 116 clinical trials included in the meta-analysis described above, only 12 studies examined antidepressant use of >12 months (Serretti and Mandelli, 2010). Information on moderate to long-term use (⩾4 months) was available for 11 types of antidepressants and, generally, mean weight change was greater during long-term use compared to short-term use. Only bupropion (‘other’ type of antidepressant) was significantly associated with weight loss, while two antidepressants were associated with weight gain (amitriptyline, a TCA, and paroxetine, an SSRI) compared to placebo.
There is limited evidence about whether weight change effects vary between types of antidepressants. We found no evidence that the risk of weight gain among current users varies by class – findings similar to those from the Finnish study which found that weight gain was higher in users of all antidepressant classes (SSRIs, TCAs and ‘others’) than in non-users (Kivimaki et al., 2010). However, we did not have large enough sample numbers to undertake analyses of individual types of antidepressants, and analyses of weight change by class could obscure differences in the risk of weight gain between individual types of antidepressants. Results from the aforementioned meta-analysis suggest that during medium- to long-term use, some antidepressants, including bupropion, amitriptyline and paroxetine, varied in their likelihood to cause weight change compared to some other antidepressants (Serretti and Mandelli, 2010). However, in many cases, the CIs were wide and overlapping, reflecting small sample sizes. A large population-based US study (n = 22,610) of electronic health records involving head-to-head comparisons of different types of antidepressants found small but significant differences in weight gain between users of some of the different antidepressants (Blumenthal et al., 2014). For example, while almost all antidepressants were associated with weight gain over 12 months, users of amitriptyline, nortriptyline and bupropion gained slightly less weight than users of citalopram (ranging from −0.06 kg for bupropion to −0.15 kg for nortriptyline) (Blumenthal et al., 2014). In apparent contrast, the meta-analysis results suggest that, compared to non-use, short-term use of amitriptyline and nortriptyline is associated with significantly more weight gain than citalopram use, but these variations disappeared in medium- to long-term treatment (Serretti and Mandelli, 2010). These studies suggest that the propensity of antidepressants to cause weight change may vary between some antidepressants; however, a direct comparison cannot be made between the studies due to the use of different reference groups, and differences in weight gain effects may be a function of duration. Further large-scale studies with adequate numbers of users of common antidepressants are needed.
There are a number of limitations that should be borne in mind when interpreting the results of our study. (1) Most data were self-reported and this may be an issue particularly in relation to weight change, as it is well established that people tend to underestimate their weight when self-reporting (Gorber et al., 2007). However, a validation study using participants from the 45 and Up Study found a high correlation (r = 0.99) between self-reported and measured weight (Ng et al., 2011), suggesting the results of this study based on self-reported weight change are likely to be valid. (2) Weight was reported at two time-points, so it was not possible to precisely identify when weight change occurred in relation to antidepressant use. While we reported results as average weight change per year, the weight change observed did not necessarily occur uniformly over time or concurrently with antidepressant use during the follow-up period. (3) While we adjusted for several potential confounding factors, residual confounding cannot be excluded and, as with most observational studies, confounding by indication could have affected the results, although consistency with clinical trial findings suggests this is unlikely to be a major issue. Moreover, there is evidence that physicians prescribing antidepressants take into account the possibility of side-effects such as weight gain when prescribing antidepressants (Zimmerman et al., 2004). As such, people who are considered to be at a higher risk of weight gain may be more likely to be prescribed antidepressants that are thought to be associated with weight loss or less weight gain, and the converse may also occur. Furthermore, this study included past and continuous users, not just new users. People often discontinue antidepressant treatment due to side-effects (Bulloch and Patten, 2010) such as weight gain, so it is possible that some of the users in this study are those who had already shown to be tolerant to the antidepressant, and thus, the observed effect sizes are likely to be conservative. (4) While the study population was large overall, numbers were limited within sub-groups, meaning type two errors cannot be ruled out and negative results should be interpreted with caution. (5) Our study was restricted to concession-card holders, so participants are likely to be older, sicker and poorer than the general population and not representative of antidepressant users in the Australian population. While the absolute results, particularly the prevalence of antidepressant use, should be regarded with caution, the relative measures of association, such as the RRRs generated in this study, can be generalised to the broader population (Mealing et al., 2010).
The main findings of our study indicate that current users of antidepressants have a 19% increased risk of gaining >3% of their body weight on average per year compared to people not using antidepressants. This translates into an increased risk of annual gains of more than 2.6 and 2.1 kg for the average Australian man (weighing 85.9 kg) and woman (weighing 71.1 kg), respectively (Australian Bureau of Statistics, 2012). While this weight gain may appear modest in absolute terms, previous studies have shown that ongoing weight gains of this magnitude (>10 kg during adult years) are related to an increased risk in all-cause age-specific mortality and chronic diseases including type 2 diabetes, heart disease and stroke (Colditz et al., 1995; Holbrook et al., 1989; Kawachi, 1999; Manson et al., 1995; Willett et al., 1995). Moreover, once weight has been gained it is often difficult to lose. We did not find any difference between the risk of weight gain between current, past-only and non-persistent users of antidepressants, so we cannot rule out long-term impacts of antidepressants on weight once use ceases. This is planned to be the subject of future research. In our study population, consistent with findings from the general population, use of antidepressants is widespread and long-term. Hence, although it is difficult to quantify the potential impacts of antidepressants on overweight and obesity in the population, along with previous evidence that antidepressant use is associated with incident obesity (Patten et al., 2009), it is likely to be a contributor.
Quantitative evidence on the magnitude and risk of weight gain with antidepressant use is likely to be of use for health professionals and consumers. There is clear evidence that weight gain is one of the main concerns of patients being prescribed antidepressants and can result in people not initiating use, not adhering to their treatment and/or discontinuing use (Bull et al., 2002; Bulloch and Patten, 2010; Hu et al., 2004). The results of this study suggest that most antidepressant users will maintain their weight within 3% of their baseline body weight on average per year. However, some antidepressant users will be at an increased risk of weight gain, primarily during long-term use; there is little evidence to suggest that short-term antidepressant use is associated with weight gain, but instead appears to be associated with an increased likelihood of weight loss.
Conclusion
There is evidence that current antidepressant use is associated with modest population-level weight gain in mid-aged and older Australian adults. While some features of this relationship provide supportive evidence of a causal relationship, further evidence is needed from large-scale longitudinal studies to confirm these findings. Given the widespread use of antidepressants and the increased risk of age-specific mortality and chronic disease due to excess weight, this finding of increased risk of weight gain among antidepressant users could translate into increased health risks within the population.
Footnotes
Acknowledgements
We would like to thank Professor Scott Henderson for providing clinical advice and Professor Catherine D’Este and Dr Grace Joshy for providing statistical advice. This research was completed using data collected through the 45 and Up Study (![]() ). The 45 and Up Study is managed by the Sax Institute in collaboration with major partner Cancer Council NSW and the following partners: the National Heart Foundation of Australia (NSW Division); NSW Ministry of Health; beyondblue; NSW Government Family & Community Services – Carers, Ageing and Disability Inclusion; and the Australian Red Cross Blood Service. We thank the many thousands of people participating in the 45 and Up Study.
). The 45 and Up Study is managed by the Sax Institute in collaboration with major partner Cancer Council NSW and the following partners: the National Heart Foundation of Australia (NSW Division); NSW Ministry of Health; beyondblue; NSW Government Family & Community Services – Carers, Ageing and Disability Inclusion; and the Australian Red Cross Blood Service. We thank the many thousands of people participating in the 45 and Up Study.
Declaration of interest
The author(s) declared no potential conflicts of interest with respect to the research, authorship and/or publication of this article.
Funding
Emily Banks and Bryan Rodgers are supported by the National Health and Medical Research Council of Australia (NHMRC) (Fellowship numbers 1042717 and 471429, respectively). This project was supported by the Study of Economic and Environmental Factors in health project, funded by the NHMRC (grant reference: 402810), NHMRC project grant 1024450 and the NHMRC Centre of Research Excellence in Medicines and Ageing (grant reference: 1060407).
