Abstract
Background:
In light of emerging evidence questioning the safety of antidepressants, it is timely to investigate the appropriateness of antidepressant prescribing. This study estimated the prevalence of possible over- and under-treatment with antidepressants among primary care attendees and investigated the factors associated with potentially inappropriate antidepressant use.
Methods:
In all, 789 adult primary care patients with depressive symptoms were recruited from 30 general practices in Victoria, Australia, in 2005 and followed up every 3 months in 2006 and annually from 2007 to 2011. For this study, we first assessed appropriateness of antidepressant use in 2007 at the 2-year follow-up to enable history of depression to be taken into account, providing 574 (73%) patients with five yearly assessments, resulting in a total of 2870 assessments. We estimated the prevalence of use of antidepressants according to the adapted National Institute for Health and Care Excellence guidelines and used regression analysis to identify factors associated with possible over- and under-treatment.
Results:
In 41% (243/586) of assessments where antidepressants were indicated according to adapted National Institute for Health and Care Excellence guidelines, patients reported not taking them. Conversely in a third (557/1711) of assessments where guideline criteria were unlikely to be met, participants reported antidepressant use. Being female and chronic physical illness were associated with antidepressant use where guideline criteria were not met, but no factors were associated with not taking antidepressants where guideline criteria were met.
Conclusions:
Much antidepressant treatment in general practice is for people with minimal or mild symptoms, while people with moderate or severe depressive symptoms may miss out. There is considerable scope for improving depression care through better allocation of antidepressant treatment.
Keywords
Introduction
Worldwide there has been a substantial increase in antidepressant use which is not explained by changes in either incident or prevalent depression (Exeter et al., 2009; Hollingworth et al., 2010; Ilyas and Moncrieff, 2012; Olfson and Marcus, 2009; Spence et al., 2014). Some of the rise in antidepressant use may be explained by antidepressants being prescribed to people not meeting severity criteria set by clinical guidelines and clinicians not deprescribing antidepressants among patients who do not meet criteria for continued use (Ambresin et al., 2015; Hepner et al., 2007; Kendrick et al., 2015). Mounting evidence suggests a potential link between antidepressant use and a range of adverse events including mortality, fractures, cardiovascular events and gastrointestinal and intracranial bleeding (Coupland et al., 2018; Frellick, 2018; Maslej et al., 2017; Rabenda et al., 2013; Yuet et al., 2019). Therefore, there is concern that the growing number of individuals taking these medications in the absence of clinical need is unnecessarily exposed to increased risk of harm.
Current guidelines support the use of antidepressants as part of first-line treatment for adults with moderate to severe major depression (Malhi et al., 2015; National Institute for Health Clinical Excellence (NICE), 2010). Once symptom remission is achieved, a period of treatment maintenance is recommended prior to gradual cessation of antidepressants. As yet there is no strong evidence to support the appropriate duration of treatment maintenance, and guidelines suggest anywhere from 6 months to at least 2 years, and typically longer in those with a history of recurrent depression. The guidelines recommend that antidepressants are not routinely used to treat subthreshold or mild depression because the benefits do not outweigh the potential risks (including but not limited to those listed above; see Bet et al., 2013; Coupland et al., 2018; Frellick, 2018; Hill et al., 2015; Maslej et al., 2017; Rabenda et al., 2013; Salvi et al., 2017). However, they can be considered in the presence of chronicity, poor response to non-pharmacological interventions, comorbidities or other complicating factors as determined by the prescribing clinician (Malhi et al., 2015; NICE, 2010).
Clearly, published guidelines allow for considerable scope in their clinical application and it is difficult to apply clear criteria as to what constitutes appropriate or inappropriate antidepressant treatment (Piek et al., 2011). As a result, researchers have used varying definitions of inappropriate antidepressant treatment for depression, making it difficult to compare findings. For example, two studies have used the Composite International Diagnostic Interview (CIDI) to determine the appropriateness of antidepressant treatment. A US study of over 5000 patients with clinician identified depression found that 56% of antidepressant prescriptions were for patients who did not meet criteria for 12-month major depressive episodes (MDEs; Mojtabai, 2013). While another study applied a broader definition (absence of 12-month mood or anxiety disorder or alcohol dependence) and identified that antidepressants were not indicated for 28% of the 526 patients taking them (Sihvo et al., 2008). In Scotland, 38% of 120 primary care patients prescribed antidepressants had ‘no depression’ on the Depression subscale of the Hospital Anxiety and Depression Scale (HADS-D; Cameron et al., 2009; Zigmond and Snaith, 1983), while an Italian survey found that 35% of 80 patients who were prescribed antidepressants for depression did not meet criteria for current depression on the World Health Organization (WHO) 10th revision of the International Statistical Classification of Diseases and Related Health Problems (ICD-10) Symptom Checklist for Depression (Berardi et al., 2005; Janca et al., 1993).
The influence of definitions on the prevalence of inappropriate antidepressant treatment was further demonstrated by a study of 1531 primary care patients in the Netherlands. The researchers first considered that antidepressants were definitely, or possibly, justified for 12-month mild, moderate or severe major depressive disorder (MDD); any depression in the previous 5 years; lifetime history of anxiety or lifetime history of dysthymia (Piek et al., 2011). Under these criteria, only 5% of patients were receiving antidepressant treatment not justified by guidelines (Piek et al., 2011). However, when they excluded dysthymia, lifetime history of recurrent or chronic major depression and lifetime history of anxiety from their definition, patients using antidepressants without guideline justification increased to 52%.
Regardless of how depression and appropriate antidepressant treatment are defined, it is clear that at least some people who are taking antidepressant medication are doing so without clinical indication. Concurrently, however, many people who might benefit from antidepressant treatment miss out (Hämäläinen et al., 2009; Harris et al., 2015; Wang et al., 2005). In Australia, Harris et al. (2015) found that among people who had consulted a health professional for mental health reasons, at least 40% of those who met criteria for moderate major depression and 29% of those who met criteria for severe major depression in the past year were not taking antidepressants.
To understand antidepressant treatment across the primary care population, it is helpful to examine possible under-treatment and over-treatment concurrently and to identify the drivers of both aspects of treatment misallocation. Therefore, the aims of this study were to (1) estimate the prevalence of possible over-treatment, when antidepressants are not indicated according to guidelines, and possible under-treatment, when guidelines suggest they are indicated, among primary care patients; (2) assess the prevalence of use of non-pharmacological mental health care by indication and use of antidepressants and (3) to identify the factors associated with guideline discordant antidepressant treatment.
Materials and methods
Study design
This study comprised an analysis of longitudinal data collected between 2005 and 2011 in a prospective, observational cohort study: Diagnosis, Management and Outcomes of Depression (diamond) (Gunn et al., 2008). Details of diamond study methods, including the setting, sample size, participant recruitment and eligibility, baseline and follow-up procedures and instruments used have been published previously (Gunn et al., 2008). diamond was approved by the Human Research Ethics Committee at the University of Melbourne (ID: 030613X).
Setting
diamond was conducted in 30 general practices across Victoria, Australia. Practices were recruited from a list of 200 randomly selected general practitioners (GPs) provided by the Australian Health Insurance Commission. The list was stratified by population distribution to ensure a representative rural and metropolitan sample. All GPs had provided at least 1500 consultations in the previous year. Thirty GPs, from different practices, participated in the study.
Participants
A random list of patients seen by the GP in the previous year was generated from computerised records. GPs identified patients meeting the following inclusion criteria: aged 18 to 75 years; literate in English; no terminal illness and resided in the community. A total of 17,780 people met these criteria and were sent a screening survey which included the Centre for Epidemiologic Studies Depression Scale (CES-D: Radloff, 1977). Surveys were returned by 7509 (42.2%) people, and 1793 (23.9%) of whom scored
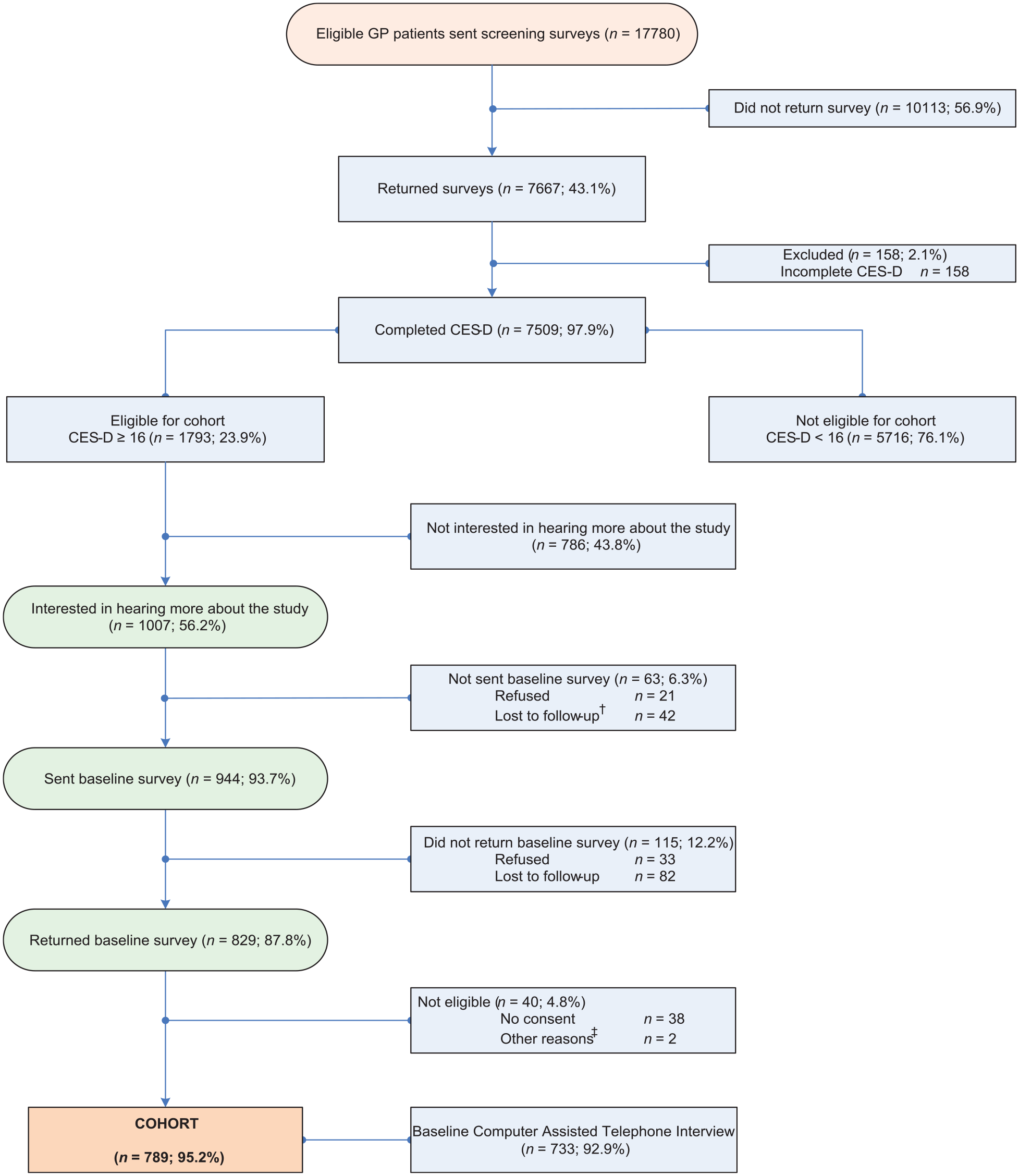
Flow chart of the diamond study cohort from screening to baseline.
Procedure
From 2005 to 2011, diamond participants completed self-reported surveys and computer-assisted telephone interviews that documented their experiences, service use, treatment and health outcomes (Gunn et al., 2008). Assessments were completed at baseline, at three monthly intervals during the first 12 months, and annually thereafter. This study reports on the data from five annual assessments, starting in the second year of the cohort (2007–2011; see the following for the rationale).
Measures
Antidepressant medications
At each assessment, participants were asked by a trained research assistant via a computer-assisted telephone interview ‘Have you been taking any medication for your emotional or physical well-being in the past 12 months?’. Those who answered yes were asked a follow-up question about what medications they had taken (including prescribed, complementary and alternative medicines or over-the-counter medication taken for a health/illness reason). Participants were asked to get their medicines so that the exact name and dose could be accurately recorded. The medications were entered into the database using a pull-down list of medications based on the MIMS (MIMS Australia, 2004) and subsequent questions asked about the frequency, strength, dose, times per day taken, condition medication was taken for, who prescribed the medication and length of time the medication was being taken. Data collected for the medications were checked and cleaned by a research pharmacist. Participants who listed medications falling into the monamine oxidase inhibitor, reversible monamine oxidase inhibitor, melatonergic, selective serotonin or serotonin norepinephrine reuptake inhibitor and tricyclic or tetracyclic classes (MIMS Australia, 2004) were classified as having taken antidepressants.
MDE
Twelve-month MDE was assessed using the ‘Depressive and Dysthymic Disorder module’ of the CIDI Auto (12-month version 2.1; World Health Organisation, 1997) at baseline and at each annual assessment, except at 12-month follow-up when there was a funding gap. For logistical reasons related to the cost of administering the CIDI and to allow recruitment from a wide geographical area, the CIDI was administered by telephone with trained and experienced interviewers who had at least an undergraduate degree (Gunn et al., 2008).
Depressive symptoms at 3, 6, 9 and 12 months were assessed using the nine-item Patient Health Questionnaire (PHQ-9: Spitzer et al., 1999). Total scores on the PHQ-9 range from 0 to 27; people who scored between 15 and 19 or 20 and 27 at 3, 6, 9 or 12 months were classified having moderate or severe depression during the first 12 months of the study. Although not a diagnostic measure, validation studies indicate that the PHQ-9 is a reasonable proxy measure of depression in the absence of a gold standard diagnosis (Gilbody et al., 2007).
Anxiety
Anxiety was measured at each annual follow-up using the PHQ anxiety module which assesses the presence of panic disorder and other anxiety disorder in the past 4 weeks (Spitzer and Kroenke, 1999). The 15-item panic module provides a probable diagnosis of panic disorder in respondents who answer ‘yes’ to four questions establishing the presence of anxiety attacks and subsequently endorse more than four of 11 somatic symptoms that occurred during their last attack. The other anxiety disorder module comprises seven items asking about symptoms of generalised anxiety in the last 4 weeks. Participants were asked whether, in the last 4 weeks, they had felt nervous, anxious, on edge or were worrying a lot about different things. If they endorsed ‘Several days’ or ‘More than half the days’ on this first question, they were asked six further questions about their anxiety symptoms. Participants met the criteria for anxiety disorder if their answer to the first question and to at least three additional questions in this module was ‘More than half the days’.
Clinical indication for antidepressants
Clinical indication for antidepressants was first assessed in 2007, at 2 years post-baseline to allow for history of depression to be considered. For each year between 2007 and 2011, MDE measured using the CIDI was used to identify moderate to severe MDE in the past 12 months, MDE at previous annual assessment was used to identify a history of moderate to severe MDE in the preceding year and anxiety measured using the PHQ anxiety module was used to identify anxiety in the last 4 weeks. Box 1 summarises how clinical indication for antidepressants according to adapted NICE guidelines was defined at each annual assessment. We defined antidepressants as being likely to be indicated if moderate or severe MDE was present in the past 12 months and possibly indicated if (1) no depression or only mild MDE was present in the past 12 months, but moderate or severe MDE was present in the 12-month period before that or (2) no depression or only mild MDE was present in the previous 12 months, but anxiety disorder was present in the previous 4 weeks. Antidepressants were unlikely to be indicated if moderate or severe MDE was not present in the past 24 months (i.e. at current or previous assessment) and anxiety disorder was not present in the previous 4 weeks.
Criteria for determining clinical indication for antidepressants according to guidelines.

MDE: major depressive episode.
Measured at current assessment.
Measured at previous assessment.
Anxiety disorder was present in the previous 4 weeks as measured at the current assessment (Spitzer et al., 1999).
Predictor variables
Variables collected at baseline were participant sex, age at recruitment, geographic location of GP surgery (i.e. urban or rural) and history of childhood abuse. Childhood physical and sexual abuse was measured using the Child Maltreatment History Self-Report (CMHSR), which asks about physical or sexual abuse before 16 years of age (MacMillan et al., 1997).
At each assessment, the following were collected: having a partner, financial stress, negative life events, chronic physical illness and hazardous drinking. Financial stress was assessed by asking participants how well they managed on their available income on a 5-point Likert-type scale (Easy, Not too bad, Difficult some of the time, Difficult all of the time; Impossible). Difficulty managing on income was coded as yes if participants responded difficult all of the time or impossible and as no otherwise (Gunn et al., 2008). Stressful life events in the past 3 months were assessed by asking participants if any of the 16 events had happened to them (e.g. major personal illness) and what impact it had (e.g. extremely negative, extremely positive; Norbeck, 1984; Sarason et al., 1978). Chronic physical illness was assessed by asking participants if, in the past 12 months, they had any one of the 11 most commonly managed conditions in Australian primary care (i.e. asthma, emphysema, diabetes, arthritis, back problem, chronic sinusitis, high cholesterol, heart disease, cancer, stroke or dermatitis; Britt et al., 2007; Gunn et al., 2012). Probable hazardous drinking was defined using the two-step scoring method 2 of the four-item FAST Alcohol Screening Test (FAST) (Hodgson et al., 2002).
We also asked participants if they had received non-pharmacological mental health care in the previous 12 months, including psychoeducation, psychological support from a GP, referral to a mental health specialist and consultation with a mental health specialist.
Statistical analysis
Patient characteristics were summarised at baseline, and psychiatric symptoms and psychological therapies were summarised by year of assessment. Over the five annual assessments, we estimated the prevalence of antidepressant use by when guidelines suggested they were clinically indicated, and the frequency of psychological therapies stratified by antidepressant treatment and by whether antidepressants were clinically indicated according to guidelines. We assessed factors associated with possible under-treatment using only assessments where antidepressants were likely to be clinically indicated and antidepressant use was not reported. Factors associated with possible over-treatment were assessed using only assessments where antidepressants were unlikely to be clinically indicated but antidepressant use was reported. We used separate logistic regressions using generalised estimating equations with robust standard errors to evaluate factors associated with possible under- and over-treatment with antidepressants and to accommodate the clustering of patients in GP surgeries and the repeated outcome measures for each patient. Initially, all factors were fitted individually in the logistic regression models and then they were fitted jointly. Multiple imputations using multivariate normal model were used to handle incomplete data, under the assumption that data are missing at random. The model incorporated all analysis and auxiliary variables potentially associated with incomplete participation. Full details of the multiple imputation approach used are provided in Supplemental Appendix 2.
All estimates were obtained by averaging results across 50 imputed datasets with inferences under multiple imputation obtained using Rubin’s (1987) rules. Frequency estimates were calculated using imputed percentage estimates and total number of participants or assessments. All main effects were tested for statistical significance using Wald tests. Data were analysed using Stata 13.1 (StataCorp, 2013).
Results
Participants
Figure 2 shows retention rates of the cohort participants from baseline to year 6. Reasons for withdrawal included no longer wishing to take part in the study, feeling that the study was not relevant (e.g. not worried about mental health), being too busy, other personal reasons (unwell, moved overseas) or death. Of 789 adult primary care patients recruited in 2005, 574 (73%) participated in at least one annual assessment between 2007 (year 2) and 2011 (year 6) and contributed data to at least one assessment. Of the 574 participants, 409 (71%) patients participated in all five assessments, 47 (8%) in four assessments, 36 (6%) in three assessments, 48 (8%) in two assessments and 34 (6%) in one assessment.
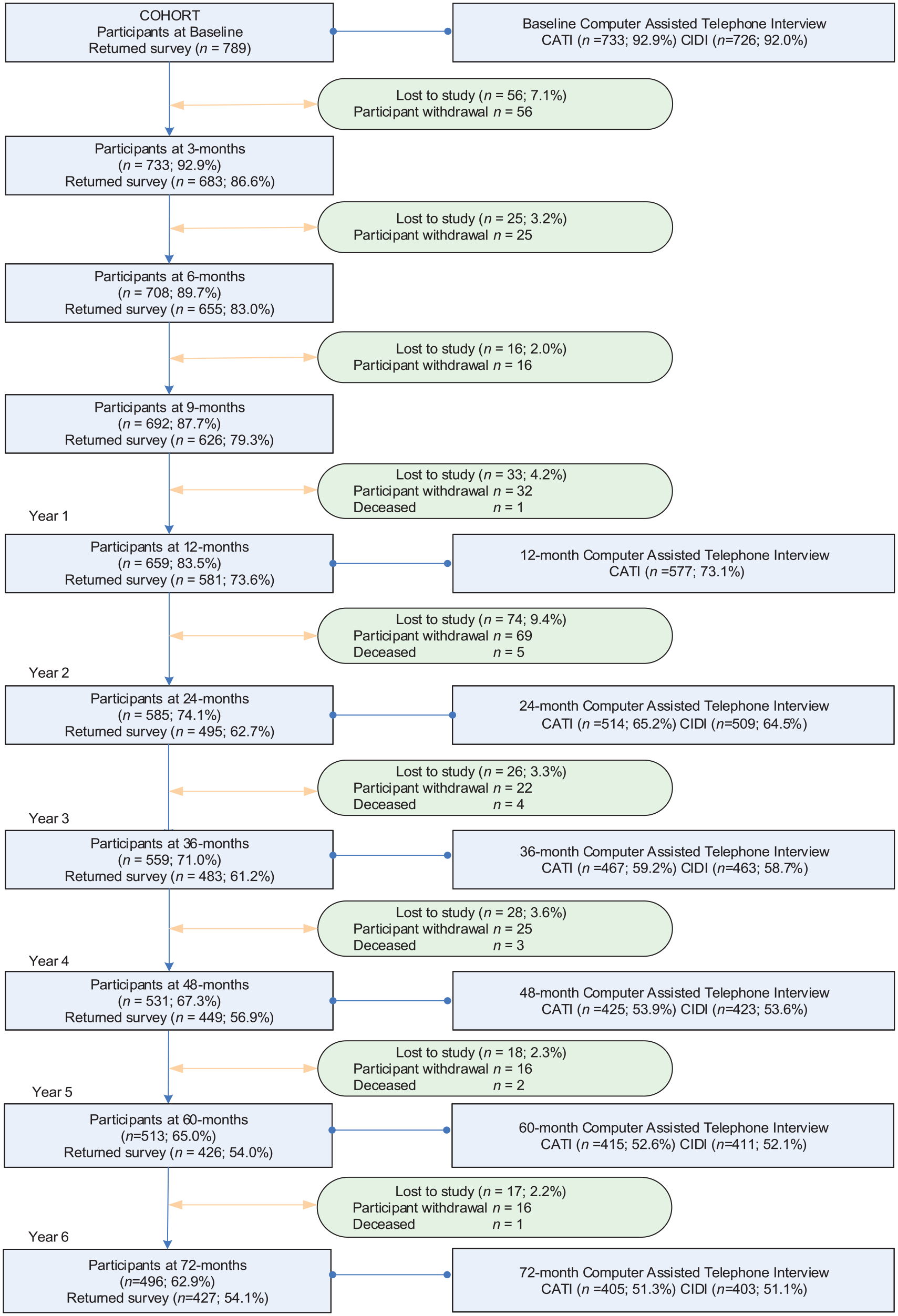
Flow chart of the diamond study cohort from baseline to year 6.
Table 1 summarises participant characteristics, psychiatric symptoms and psychological therapies. One-third (33%) of participants were recruited from clinics in rural and regional areas, 27% were men and the mean age at baseline was 48 years (SD = 12.9 years). Around a third of patients were not in a relationship at each annual assessment, a fifth had financial stress, 20–24% experienced negative life events, only 7–11% of patients were not experiencing chronic pain, and around 15% reported hazardous drinking. At year 1, 46% had depressive symptoms, and around 20% had depressive symptoms and anxiety at years 2–6.
There were no differences in baseline characteristics between the 574 patients that were used for this study (responders) and the 215 that were not included as they dropped out of the study before year 2 (non-responders; Supplemental Appendix 1).
Prevalence of antidepressant use according to guideline criteria
Taking antidepressants for emotional well-being in the past 12 months was reported in 41% (1182/2870) of all assessments (Table 2). Based on criteria in Box 1, of the 2870 assessments, antidepressants were likely to be indicated according to guidelines for 20% of assessments, possibly indicated for 20% of assessments and unlikely to be indicated for 60% of assessments. Of the 1182 assessments reporting antidepressant use, 29% (343/1182) were likely to be clinically indicated in the past 12 months, 24% (282/1182) were possibly clinically indicated and 47% (557/1182) were unlikely to be clinically indicated as they did not meet criteria for moderate or severe depression in the past 2 years and did not have current anxiety.
Patient characteristics, psychiatric symptoms and psychological therapies in 574 patients.
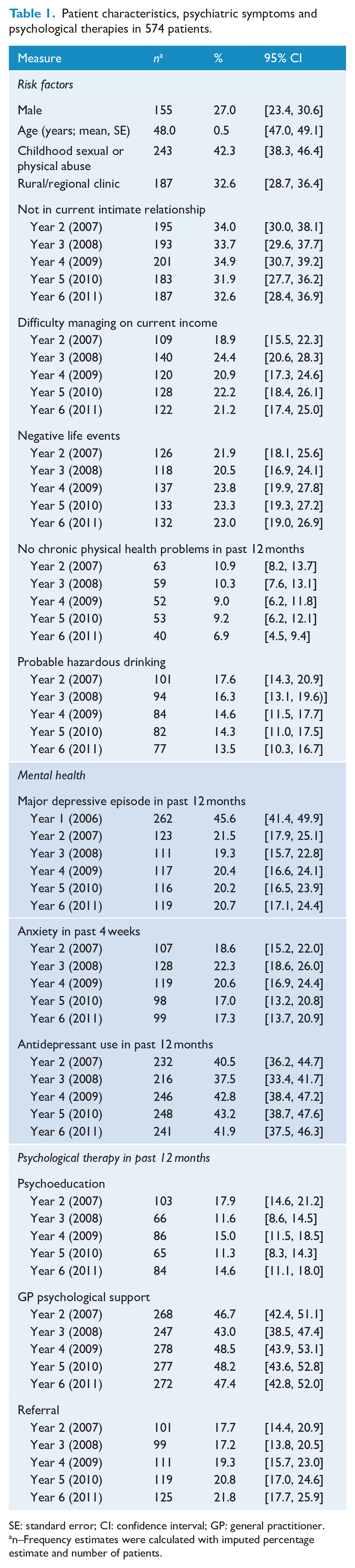
SE: standard error; CI: confidence interval; GP: general practitioner.
n–Frequency estimates were calculated with imputed percentage estimate and number of patients.
In 59% (343/586) of assessments where antidepressant treatment was likely to be indicated according to guidelines, antidepressant use was reported, suggesting appropriate treatment, and for 41% of assessments, patients were identified as being possibly under-treated. In a third (557/1711) of assessments where antidepressants were unlikely to be indicated, participants reported taking antidepressants, which we classified as possible over-treatment.
In an ancillary analysis, we expanded the criterion in which antidepressants were considered justified to include the presence of dysthymia, which requires 2 years of subthreshold symptoms. This expanded criterion did not greatly impact the prevalence of possible over-treatment among people without clinical indication according to guidelines because nearly all instances of dysthymia were already accounted for by the presence of anxiety, with fewer than 1% of assessments with complete data re-classified from unlikely to be indicated to possibly indicated.
Other mental health interventions
Frequency of mental health interventions received by antidepressant use and clinical indication are shown in Table 3. At assessments where antidepressants were being used, lower levels of psychological therapy were being used where antidepressant use was unlikely to be clinically indicated according to guidelines compared to where it was for psychoeducation (14% for unlikely to be indicated compared to 23% when likely to be indicated), GP psychological support (64% vs 87%) and referral (18% vs 42%). At assessments where antidepressants were not being used, lower levels of psychological therapy were being used where antidepressant use was unlikely to be indicated compared to where it was for psychoeducation (9% vs 19%), GP psychological support (24% vs 59%) and referral (8% vs 27%).
Antidepressant treatment by clinical indication according to guidelines in 574 patients over five annual assessments.
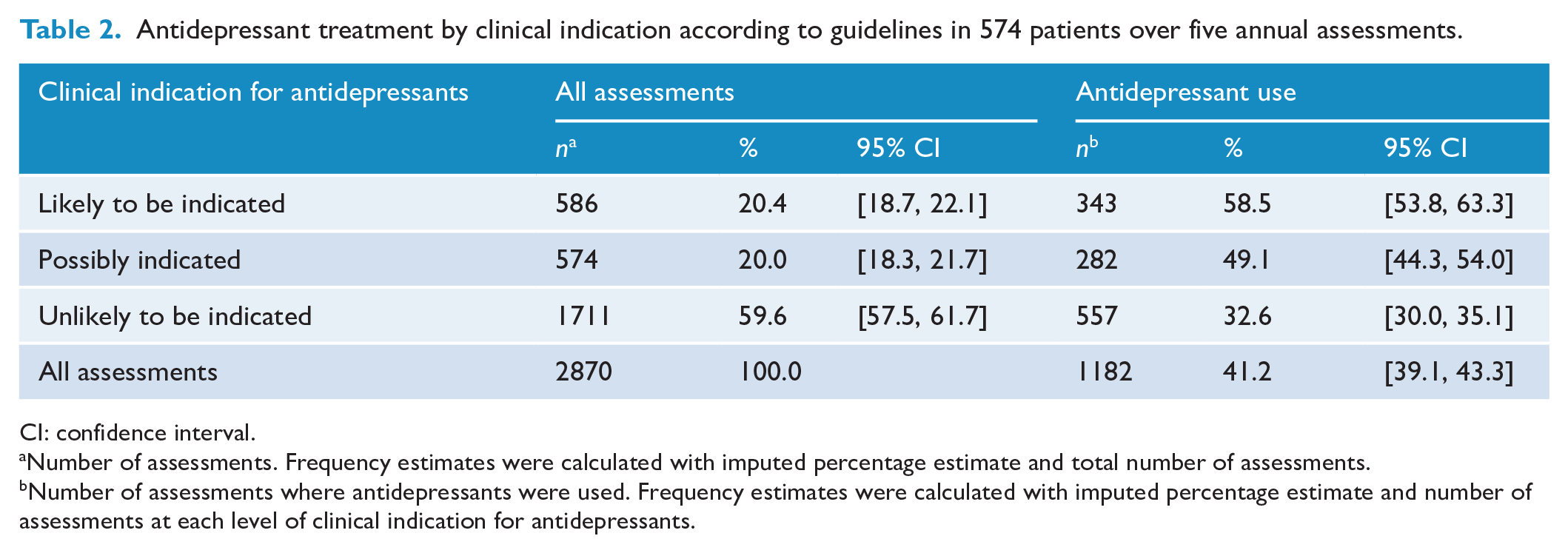
CI: confidence interval.
Number of assessments. Frequency estimates were calculated with imputed percentage estimate and total number of assessments.
Number of assessments where antidepressants were used. Frequency estimates were calculated with imputed percentage estimate and number of assessments at each level of clinical indication for antidepressants.
Predictors of possible over- and under-treatment of antidepressants
We found no statistical evidence of associations between risk factors and not taking antidepressants when they were likely to be clinically indicated according to guidelines (Table 4). Being female and having a chronic illness were both found to be associated with taking antidepressants when they were unlikely to be clinically indicated (Table 5).
Psychological therapies received in 12 months by antidepressant use and clinical indication in 574 patients over five annual assessments.
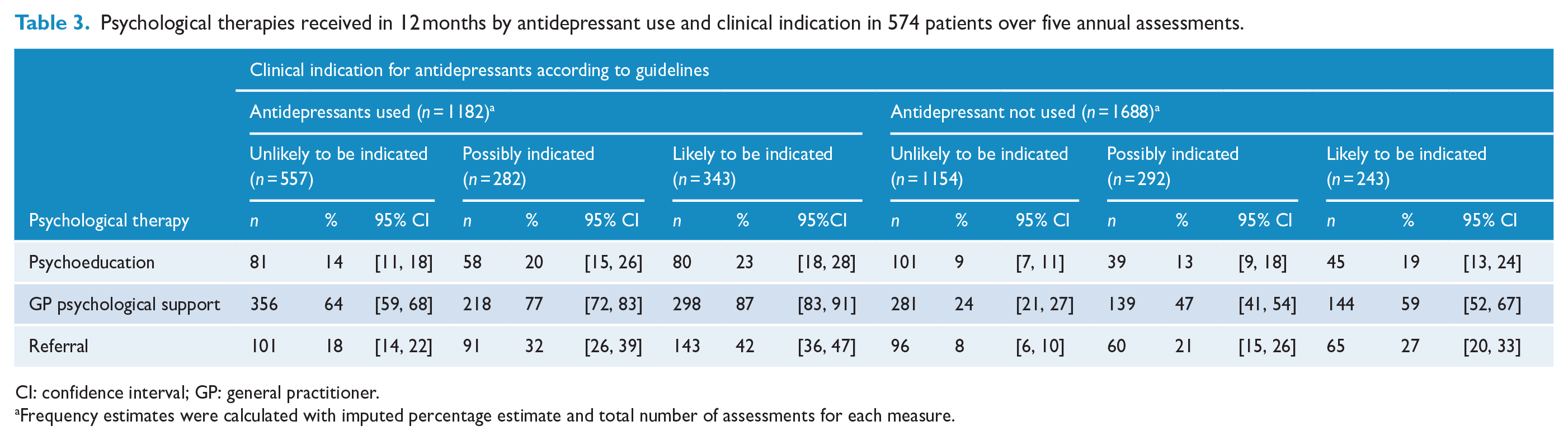
CI: confidence interval; GP: general practitioner.
Frequency estimates were calculated with imputed percentage estimate and total number of assessments for each measure.
Factors associated with possible under-treatment with antidepressants when they were clinically indicated.
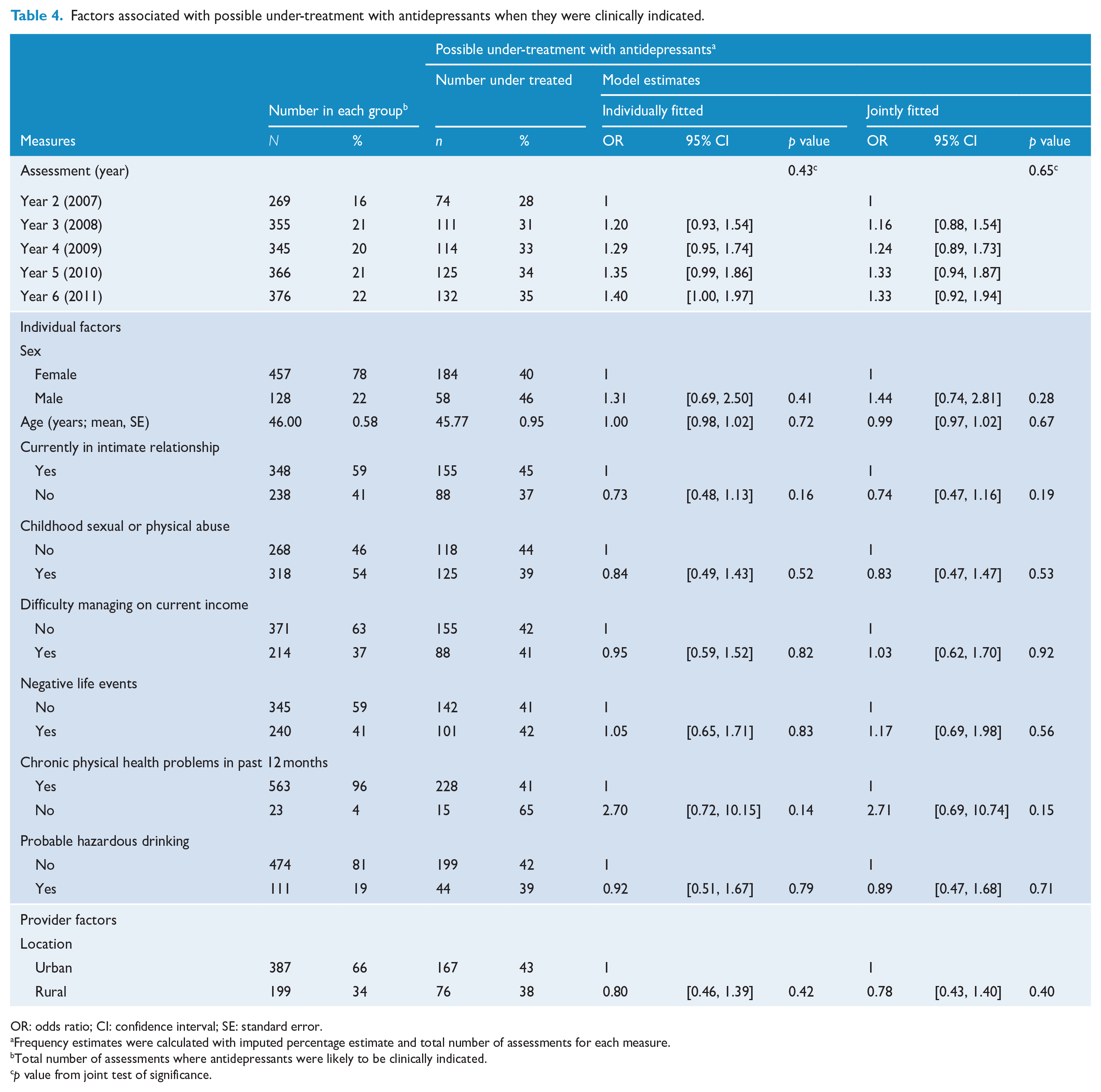
OR: odds ratio; CI: confidence interval; SE: standard error.
Frequency estimates were calculated with imputed percentage estimate and total number of assessments for each measure.
Total number of assessments where antidepressants were likely to be clinically indicated.
p value from joint test of significance.
Factors associated with possible over-treatment with antidepressants when they were not indicated. a
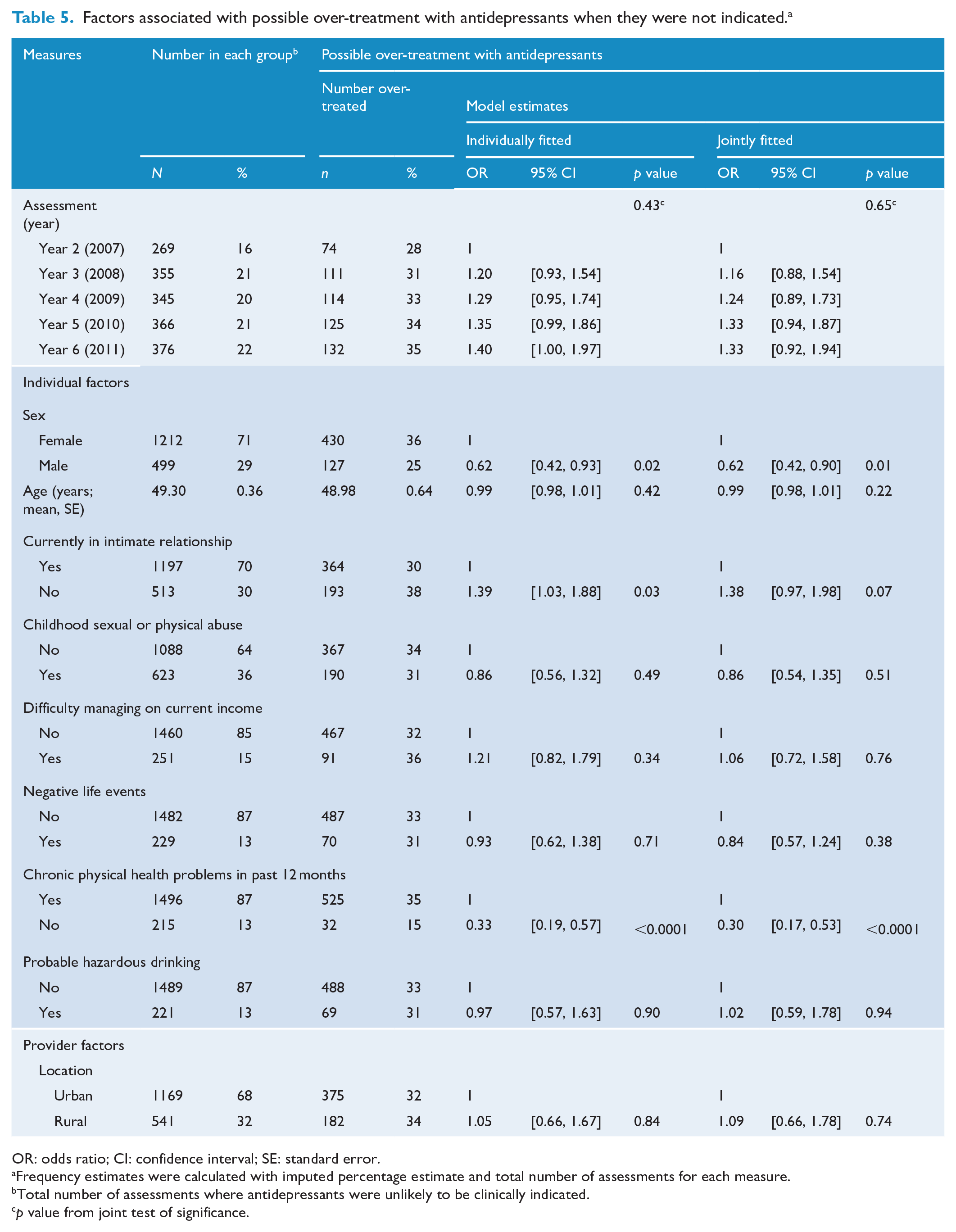
OR: odds ratio; CI: confidence interval; SE: standard error.
Frequency estimates were calculated with imputed percentage estimate and total number of assessments for each measure.
Total number of assessments where antidepressants were unlikely to be clinically indicated.
p value from joint test of significance.
Discussion
In this Australian primary care cohort, we found that reported antidepressant use was not always consistent with current guidelines (NICE, 2010). According to our study definitions, possible over-treatment was identified in around one in three assessments, while possible under-treatment was identified in around two in five assessments. Possible over-treatment of antidepressants was more likely to occur in the context of one or more chronic illnesses such as back pain, arthritis, emphysema and cancer.
Limitations and strengths
The prevalence of possible over-treatment may be inflated as there are indications other than MDE or anxiety where antidepressants may be used. We could not exclude whether possible over-treatment with antidepressants could be explained by prescribing antidepressants in line with guideline recommendations for the treatment of other common conditions (e.g. social anxiety disorder Andrews et al., 2018; NICE, 2013), posttraumatic stress disorder (Phoenix Australia, Centre for Posttraumatic Mental Health, 2013) or common off-label uses (e.g. in the treatment of chronic pain, Urits et al., 2019), as this information was not collected in the diamond study. Furthermore, some participants may have had an earlier severe or recurrent episode of major depression which had responded to treatment, and a reasonable clinical decision may be to continue the antidepressant for longer than 2 years.
Antidepressants may also be prescribed for people with persistent subthreshold symptoms which have not responded to psychological treatment. In our sample, almost two-thirds of antidepressant use that did not meet guideline criteria was reported by participants who also received psychological treatment in the previous 12 months from a GP. It is possible that antidepressants were not the first treatment option for these participants; however, we were unable to confirm this with the available data. This finding may also indicate that GPs use a mix of counselling and antidepressants as the main approach to managing mild depressive symptoms.
Likewise, the prevalence of possible under-treatment may also be inflated. For instance, participants who reported not using antidepressants although clinically indicated according to guidelines may have been prescribed antidepressants by their clinician but the patient decided not to take the medication.
History of MDE in the first year was measured using depressive symptoms using the PHQ-9 at three monthly intervals. As the PHQ-9 measures depressive symptoms in the previous 2 weeks, this could potentially underestimate MDE in first year which could also bias the prevalence of possible over-treatment upwards when assessed in year 2. Similarly, the presence of anxiety over the past 12 months could be underestimated as it was only measured in the past 4 weeks at each yearly assessment, thus could lead to inflated estimates of possible over-treatment with antidepressants when unlikely to be clinically indicated. Finally, the classification of possible over- and under-treatment relied on patient self-report of depression severity, anxiety and antidepressant use and should be interpreted in light of the potential for response bias.
A strength of this study was the measurement of outcomes prospectively over time, providing almost 3000 assessments which lead to a high degree of precision around the prevalence estimates. In addition, the longitudinal analysis enabled us to account for changes in depressive symptoms over time providing a better representation of the overall patterns of antidepressant use, including long-term use of the same individual than previous cross-sectional studies (Berardi et al., 2005; Cameron et al., 2009; Piek et al., 2011).
Generalisability and transferability
Generalisability may be limited to patients who attended GPs who were particularly interested in mental health and that they practice mental health care in a way that is not representative of all GPs in Victoria. For example, it is possible that GPs in this study were more likely to provide psychological support to their patients than other GPs. The findings are generalisable to patients seen in primary care across the spectrum from mild to severe depression; however, may be limited to individuals who are more likely to experience multiple comorbidities because of the diamond recruitment approach and inclusion criteria (Gunn et al., 2008). Although participants were surveyed between 2005 and 2011, the findings still remain applicable as treatment with antidepressants in the primary care setting is unlikely to have changed significantly in recent years.
Comparisons with other studies
Our findings contribute to the growing body of literature on this topic, with our estimates of possible inappropriate treatment falling in the mid-range of those reported previously. In our study, of assessments where patients reported taking antidepressants for mental health in the past 12 months, 47% were unlikely to be indicated. This was consistent with previous literature reporting that 28–56% of primary care patients taking antidepressants did not meet criteria for major depression (Berardi et al., 2005; Cameron et al., 2009; Mojtabai, 2013; Sihvo et al., 2008). Although the percentages are not directly comparable because of the different criteria used for defining depression and antidepressant use, they demonstrate that in about 30% to almost 60% of patients taking antidepressants, such treatment may not be indicated.
For possible under-treatment, we found that antidepressants were taken in just 41% of assessments where the patient had moderate to severe 12-month major depression, compared to Berardi et al (2005) who reported that 33% of people who met ICD-10 criteria for depression in the past month were not receiving antidepressant treatment. Similarly, Harris et al. (2015) reported that at least 32% of people with moderate and severe major depression were not taking an antidepressant.
The estimated prevalence for possible over-treatment of 33% was higher than the 14% reported by Berardi et al (2005). However, it is lower than Mojtabai’s (2013) estimation of 74% who did not exclude patients with anxiety and lower than Piek et al’.s (2011) revised estimation, using stricter criteria, that 52% of antidepressant prescriptions were not justified.
Conclusion
There is considerable scope to both improve depression care and minimise potential unnecessary harm through more targeted allocation of antidepressant treatment. Strategies are needed to identify people who could benefit from treatment with antidepressants when likely to be clinically indicated according to guidelines but are not receiving it and to provide people with mild depressive symptoms alternative pathways to provide them with support for their mental health when treatment with antidepressants may not be appropriate.
Supplemental Material
AD_trmt_in_primary_care_Appendix_2_MI – Supplemental material for Antidepressant treatment for primary care patients with depressive symptoms: Data from the diamond longitudinal cohort study
Supplemental material, AD_trmt_in_primary_care_Appendix_2_MI for Antidepressant treatment for primary care patients with depressive symptoms: Data from the diamond longitudinal cohort study by Sandra K Davidson, Helena Romaniuk, Patty Chondros, Christopher Dowrick, Jane Pirkis, Helen Herrman, Susan Fletcher and Jane Gunn in Australian & New Zealand Journal of Psychiatry
Supplemental Material
AD_trmt_in_primary_care_Appendix_Revised – Supplemental material for Antidepressant treatment for primary care patients with depressive symptoms: Data from the diamond longitudinal cohort study
Supplemental material, AD_trmt_in_primary_care_Appendix_Revised for Antidepressant treatment for primary care patients with depressive symptoms: Data from the diamond longitudinal cohort study by Sandra K Davidson, Helena Romaniuk, Patty Chondros, Christopher Dowrick, Jane Pirkis, Helen Herrman, Susan Fletcher and Jane Gunn in Australian & New Zealand Journal of Psychiatry
Footnotes
Acknowledgements
The named authors submit this publication on behalf of the diamond study investigators which include the following: Professor Jane Gunn, Professor Helen Herrman, Professor Michael Kyrios, Professor Kelsey Hegarty, Professor Christopher Dowrick, Associate Professor Victoria Palmer, Dr Gail Gilchrist, Associate Professor Grant Blashki, Professor Dimity Pond and Dr Patty Chondros. We acknowledge the 30 dedicated GPs, their patients and practice staff for making this research possible. We thank the cohort participants for their involvement in the study. We also thank the diamond project team and associate investigators involved in the study: Associate Professor Cathy Mihalopoulos, Professor Lena Sanci, Professor Frances Griffiths, Dr Sandra Davidson, Ms Maria Potiriadis, Ms Konstancja Densley, Dr Aves Middleton and the casual research staff.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship and/or publication of this article: The diamond study was initiated with pilot funding from the Beyondblue Victorian Centre of Excellence and the main cohort study has received Project Grant funding from the National Health and Medical Research Council (ID 299869, 454463, 566511 & 1002908). The one-year Computer Assisted Telephone Interview was funded by a Stream 3 grant from the Australian Primary Health Care Research Institute (APHCRI). No funding body had a role in study design; the collection, analysis and interpretation of data; the writing of the manuscript or the decision to submit this manuscript for publication.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
