Abstract
Background
To explore the role of large-artery atherosclerosis in migraine, we investigated the association between migraine and arterial calcification in different intracranial and extracranial vessels.
Methods
1856 participants were included, mean age (standard deviation) 67.4 (5.8) years, from the population-based Rotterdam Study cohort. Migraine was assessed by validated questionnaire and vascular calcification was assessed by computed tomography (expressed in Agatston score for the coronary arteries and volume in mm3 for the aortic arch, intracranial, and extracranial carotid arteries). Per vessel, the association of migraine with calcification was investigated by linear regression, adjusted for age, sex, cardiovascular risk factors, and calcification in other vessels.
Results
Of the participants, 279 (15%) were identified as persons with lifetime migraine. In multivariable adjusted models, migraine was associated with smaller intracranial carotid artery calcification volume (difference in log-transformed calcification volume in persons with migraine compared to persons without migraine: −0.19[−0.29, −0.08]). While subjects with migraine also showed a lower calcification burden in the remaining arterial beds, those associations did not reach statistical significance.
Conclusions
Persons with migraine, compared to those without, had less arterial calcification in the intracranial carotid artery, but not in other arterial beds. Future studies are needed to confirm these findings.
Introduction
Migraine is a recurring neurovascular disorder and has been recognised as one of the top 25 diseases associated with disability worldwide (1). In addition to the individual and economic burden of migraine, increasing evidence positions migraine as a potential risk factor for cardiovascular diseases (CVDs) including stroke and cardiovascular mortality (2,3). Additionally, migraine has been associated with subclinical manifestations of cerebral small vessel disease, such as white matter lesions and microbleeds (4). However, the role of large-artery atherosclerosis has not been extensively studied in migraine.
Previous studies on migraine and carotid-intima media thickness (cIMT) show inconsistent associations including either no difference or higher cIMT in persons with migraine compared to those without migraine (5–9). While cIMT may reflect non-atherosclerotic remodeling of the arterial wall that might be associated with an early vascular aging process (10), vascular calcification may reflect advanced atherosclerosis and has shown robust associations with cardiovascular events (11). One previous study has found no association between migraine and coronary artery calcification (8). Similarly, migraine in persons who have had ischemic stroke was not associated with calcification across the vessel beds (12).
Calcification burden differs considerably across vessel beds (13,14) and carries a differential predictive value for various manifestations of CVD depending on the proximity of the vascular bed to the organ affected in the manifestation under study (15,16). Location-specific atherosclerosis, rather than vascular pathology in distant vessels, may thus play an important role in disease pathogenesis. Hence, investigation of the association between migraine in the general population with calcification in different vessels may provide information on the relationship between migraine and CVD. Therefore, we investigated whether migraine is associated with arterial calcification across the coronary arteries, aortic arch, and intra- and extracranial carotid arteries in a large group of middle-aged and elderly persons from a population-based study.
Methods
Settings and study population
This study was based in a prospective population-based cohort among the middle-aged and elderly inhabitants of the Ommoord district in Rotterdam, the Netherlands: The Rotterdam Study. The rationale and design of the study have been described previously (17). The study was initiated in 1990 with the first cohort (RS-I) of 7983 persons aged 55 years or older and was extended in 1999 with 3011 participants and the same inclusion criteria (the second cohort: RS-II). Data was collected by home interview and by physical testing and examination at the research center. Participants underwent CT assessment of calcification score/volumes during the fourth wave of the first cohort (RS-I-4, 2002–2004) and the second wave of the second cohort (RS-II-2, 2003–2006). Lifetime migraine prevalence was assessed in the fifth wave of the first cohort (RS-I-5, 2009–2011) and the third wave of the second cohort (RS-II-3, 2010–2012). We excluded participants with missing data on either migraine or calcification score/volumes. For the current study, 1856 participants who had data on migraine and arterial calcification were included. All subjects gave written informed consent, and the study protocol was approved by the medical ethics committee according to the Wet Bevolkingsonderzoek ERGO (Population Study Act Rotterdam Study), executed by the Ministry of Health, Welfare and Sport of the Netherlands.
Assessment of migraine
Migraine was assessed by a migraine questionnaire based on the migraine headache criteria from the International Classification of Headache Disorders, second edition (ICHD-II) (18) and modified from the questionnaire validated for use in the Genetic Epidemiology of Migraine (GEM) study in Leiden (19,20) during the home interview. The questionnaire assessed lifetime occurrence of headache attacks that fulfilled the following diagnostic criteria: More than five headache attacks of severe pain intensity, with a duration of 4–72 hours (when untreated), with at least two of the four headache characteristics (unilateral pain, pulsating quality, severe pain intensity, and aggravation by or causing avoidance of routine physical activity), nausea and/or photophobia and phonophobia, and not attributable to another disorder. All participants who met the criteria for lifetime history of migraine were classified as persons with migraine. The classification of migraine with aura was modified from the ICHD-II criteria as previously described (19), due to limited questions related to aura in the survey. Therefore, migraine with aura was assessed only in those participants who fulfilled all migraine criteria. We furthermore dichotomised persons with migraine as those with active migraine ( < 1 year since the last attack) or non-active migraine ( > 1 year since the last attack).
Assessment of arterial calcification
Imaging was performed with a 16- or 64-slice multidetector CT scanner (Somatom Sensation, 16- 64, Siemens, Forchheim, Germany). No contrast material was administered. We performed a cardiac and an extra-cardiac scan to visualise calcification in the coronary arteries, the aortic arch, and the extra- and intracranial internal carotid arteries. Detailed information regarding the scan protocol has previously been described (13). Quantification of calcification in the coronary arteries, aortic arch, and the extracranial internal carotid arteries was performed using commercially available software (Syngo Calcium Scoring, Siemens, Forcheim, Germany). Coronary artery calcification was quantified using the Agatston method, based on the sum of the individual coronary arteries (13,21). For calcification in the intracranial internal carotid artery, we used a dedicated semi-automated method, which has been described in detail before (22). Coronary artery calcification (CAC) is expressed as a score, while aortic arch calcification (AAC), extracranial carotid artery calcification (ECAC) and intracranial carotid artery calcification (ICAC) are expressed as volume in mm3.
Assessment of covariates
Anthropometric measures were obtained during the research centre visit. Body mass index (BMI) was calculated from the weight of the participant divided by the square of the height of the participant. Blood pressure was measured by a random-zero sphygmomanometer at the right brachial artery with the participant in the sitting position. The mean of two consecutive measurements was taken. Serum total cholesterol and high-density lipoprotein (HDL) cholesterol were measured using standard laboratory techniques. Diabetes type 2 was diagnosed in the case of a fasting plasma glucose level of 7.0 mmol/L or higher, or the use of antidiabetic medication. Information on smoking behavior, antihypertensive or lipid-lowering medication use, and history of CVD was collected by a computerised questionnaire (11,23). Smoking behavior was defined as past, current, or no history of smoking.
Statistical analysis
Population characteristics were presented as mean ( ± standard deviations) or median (25th – 75th percentile) for continuous variables as appropriate and counts (percentages) for dichotomous variables. Differences in characteristics between persons with and without migraine were tested by ANCOVA (continuous variables) and logistic regression (categorical variables), adjusted for age and sex. Due to their skewed distribution, we transformed the calcification score/volumes by performing a natural log (Ln) transformation (Ln [calcification score/volumes + 1mm3]).
Linear regression analysis was used to investigate the association between migraine and calcification score/volumes. Age was modelled as both age and age2, but age2 did not improve model fit. We developed three models. Model 1 included age, sex, and cohort. Model 2 additionally included BMI, systolic and diastolic blood pressure, total cholesterol, HDL, smoking status, use of blood pressure-lowering medication or lipid-lowering medication, prevalent diabetes, and history of CVD. For model 3, each calcification score/volume was additionally adjusted for the other three calcification score/volumes.
We further stratified the analyses for migraine subtypes (migraine without aura, migraine with aura, active migraine and nonactive migraine) as well as for sex, age ( ≥ 65 years versus < 65 years), and history of CVD. The difference between the effect estimates for the association of calcification volume/score with migraine subtypes were compared using the Wald test (24). In the stratified analyses, we tested the interaction terms between migraine and the stratified characteristic (i.e. age ≥ 65 versus < 65, sex, and history of CVD). Missing values for all covariates were imputed using multiple imputation based on all variables in the full regression model. All statistical analyses were performed using the statistical software package SPSS (IBM corp., Armonk, New York, USA), version 21.0 for Windows.
Results
Characteristics of the participants.
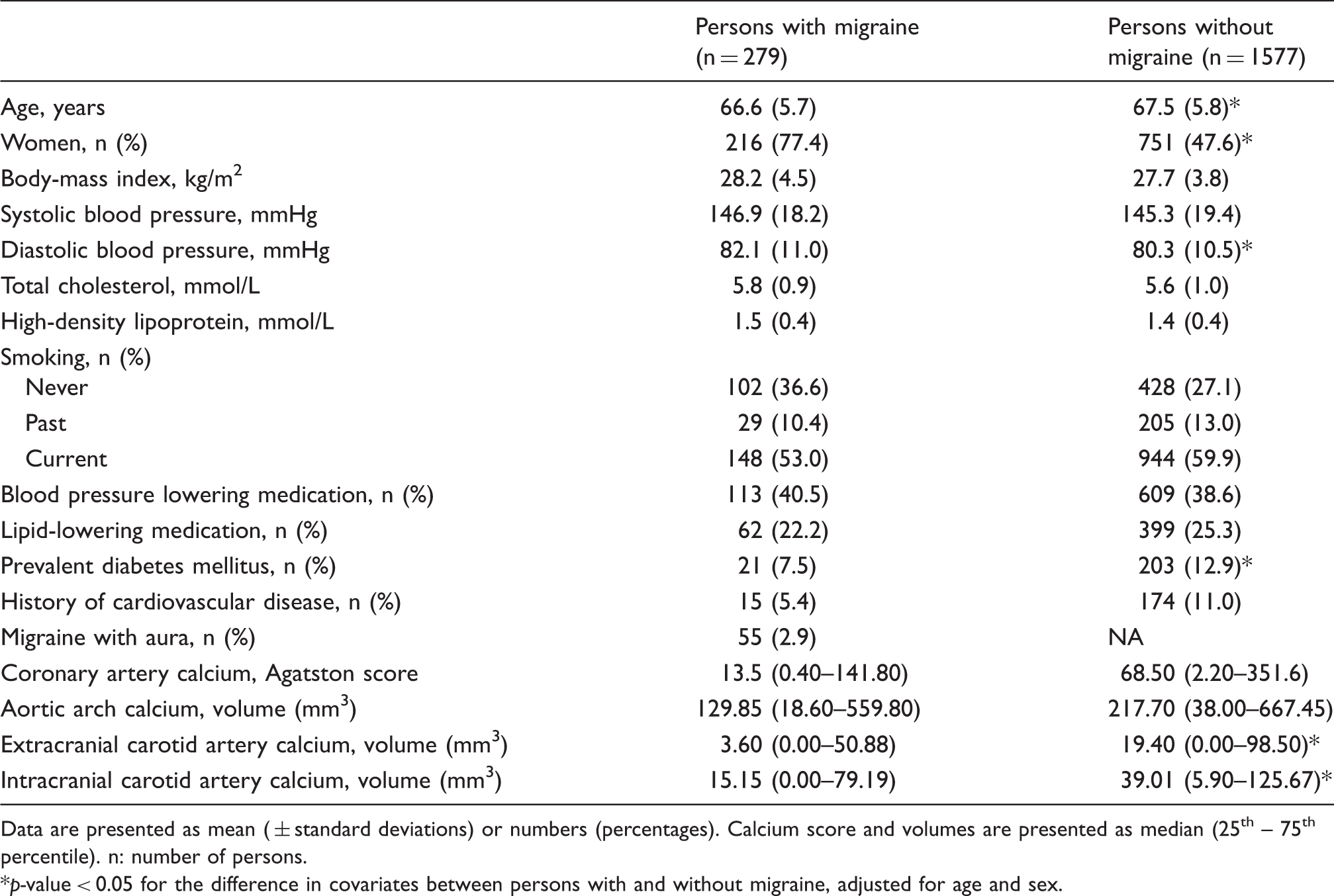
Data are presented as mean ( ± standard deviations) or numbers (percentages). Calcium score and volumes are presented as median (25th – 75th percentile). n: number of persons.
p-value < 0.05 for the difference in covariates between persons with and without migraine, adjusted for age and sex.
Difference in log-transformed calcification score/volumes between persons with and without migraine.

Values are difference in log-transformed calcification score/volumes and accompanying 95% CI for persons with migraine compared to persons without migraine. Model 1: Adjusted for age, sex and cohort. Model 2: Additionally adjusted for body-mass index, systolic blood pressure, diastolic blood pressure, total cholesterol, high-density lipoprotein, smoking, blood-pressure lowering medication use, lipid-lowering medication use, prevalent diabetes, and a history of cardiovascular disease. Model 3: Additionally adjusted for the other three calcification score/volumes.
Difference in log-transformed intracranial carotid artery calcification volume between persons with and without subtypes of migraine.

Values are difference in log-transformed intracranial carotid artery calcification volume and accompanying 95% CI for persons with different subtypes of migraine compared to persons without migraine. In all, 1839 persons had data on both intracranial carotid artery calcification and migraine. n: number of persons with a specific subtype of migraine; N: number of persons without migraine. Model 1: Adjusted for age, sex and cohort. Model 2: Additionally adjusted for body-mass index, systolic blood pressure, diastolic blood pressure, total cholesterol, high-density lipoprotein, smoking, blood-pressure lowering medication use, lipid-lowering medication use, prevalent diabetes, and a history of cardiovascular disease.
Difference in intracranial carotid artery calcification volume between persons with and without migraine in different subgroups.

Values are difference in log-transformed intracranial carotid artery calcification volume and accompanying 95% CI for persons with migraine compared to persons without migraine. In all, 1839 persons had data on both intracranial carotid artery calcification and migraine. n: number of persons with migraine; N: number of persons without migraine. Model 1: Adjusted for age, sex and cohort. Model 2: Additionally adjusted for body-mass index, systolic blood pressure, diastolic blood pressure, total cholesterol, high-density lipoprotein, smoking, antihypertensive or statin use, diabetes, and a history of cardiovascular disease. p-interaction for migraine*sex: 0.42. p-interaction for migraine*age: 0.50. p-interaction for migraine*CVD: 0.88.
Discussion
In this population-based study, we observed that persons with a lifetime history of migraine had less calcification in the intracranial carotid artery than persons without migraine, after adjusting for differences in cardiovascular risk factors. Furthermore, persons with migraine also tended to have a lower calcification burden across other vessel beds, although these differences were not statistically significant.
Previous studies assessing atherosclerosis in persons with migraine by carotid ultrasonography, a frequently used measure, found either higher cIMT (5,6) or no difference in cIMT in persons with migraine (7,8). One study also reported no difference in coronary artery calcification among persons with and without migraine (8). The findings did not appear to be related to whether persons only with current migraine (8) or with lifetime migraine (5–7) were assessed. Whereas cIMT may reflect non-atherosclerotic remodeling of the arterial wall that might be associated with an early vascular aging process (10), vascular calcification is thought to be the hallmark of more advanced atherosclerotic disease (25,26). However, there is low to moderate correlation between calcification in different arteries (13). This is thought to reflect vessel-specific atherosclerotic changes. Vascular calcification in different vessels may thus carry differential predictive value in association with various forms of CVD depending on the proximity of the vascular bed to the organ affected in the event under study (27). Indeed, coronary artery, aortic arch and extracranial carotid artery calcification have been shown to be associated with coronary heart disease, but not with cerebrovascular disease (16), while intracranial carotid artery calcification has been associated with increased risk of stroke, particularly ischemic stroke (3,15). Our finding regarding a lower calcification burden in intracranial carotid arteries among persons with migraine compared to persons without migraine additionally supports vessel-specific associations to disease. Furthermore, in line with our findings, it has recently been shown that among persons who have had ischemic stroke, subjects who had a positive history for migraine did not have more calcification of the intracranial carotid artery than persons without migraine (12). In fact, the authors suggested that atherosclerotic changes were less prevalent in stroke patients with migraine. Therefore, stroke in persons with migraine may be due to the possible impact of endothelial dysfunction related to atherosclerosis, compared to stroke in the general population.
In our study, persons with migraine had a more favorable cardiometabolic profile and therefore lower levels of large artery disease. Previous studies from the Rotterdam Study also found that persons with migraine had higher basilar artery flow than persons without migraine (19). Similarly, several studies have indicated that the association between migraine and risk of stroke is apparent only among those with a low cardiovascular risk profile (2). Our findings, in line with the others, further support the concept that traditional cardiovascular risk factors might not play a role in the association between migraine and CVD. Moreover, calcification in the intracranial carotid artery has recently been shown to be predominantly located in the internal elastic lamina and this form of calcification is nonatherosclerotic (28). Consequently, intracranial carotid artery calcification could reflect not only atherosclerosis but also other vascular pathology such as arterial stiffness (29). We also found that persons with migraine had higher diastolic blood pressure, but not systolic blood pressure, than persons without migraine, a pattern that has been suggested as an indicator of elevated peripheral vascular resistance as a consequence of increased microvascular stiffness (30). Taken together, our results suggest that other potential pathways that have been implicated in migraine, such as vascular hyperreactivity, endothelial dysfunction, microvascular pathology, or coagulation disorders, may play a role in linking migraine to CVD (31).
We further stratified our analyses for migraine subtypes including migraine with or without aura and active or non-active migraine. Previous studies have suggested that migraine with aura is an independent risk factor for CVD (32,33), all-cause mortality, and cardiovascular mortality (34). Interestingly, migraine with aura in women from a primary care population was recently shown to be associated with increased carotid thickening, whereas migraine without aura was associated with lower risk of carotid plaques and arterial stiffening (9). However, we did not find any differences in the association of arterial calcifications with migraine across different migraine subtypes, suggesting that the increased risk of CVD in persons with migraine with aura is not related to arterial calcification in the general population. As age, sex, and history of CVD may influence associations with arterial calcification (22), we additionally stratified our analyses based on these characteristics. Although the association of migraine and ICAC tended to be stronger in men, older persons, and subjects without prevalent CVD, the differences were not statistically significant. However, lack of statistical significance might have been due to smaller numbers of persons with migraine in the stratified groups.
The current study is the first study that has examined the association of migraine with arterial calcification across different vascular beds in a population-based setting. Having access to different vascular measures as well as detailed information on cardiovascular risk factors of participants allowed for in-depth head-to-head comparisons. Our study also had a number of limitations. Migraine prevalence during the lifetime was assessed by questionnaire and classified according to a modified version of the ICHD-II criteria. This modification could have resulted in persons who had migraine with moderate instead of severe pain, and persons who only had aura symptoms without headache, being classified as persons without migraine. Therefore, this might have limited our ability to find differences between the two groups. Furthermore, participants were asked about migraine history many years after the usual age of onset of migraine (in young adulthood). Together with the finding that persons with positive migraine history were younger than persons without migraine in our population, this could suggest the occurrence of recall bias. However, the lifetime prevalence of migraine (15.0%) in our study population is in line with that of the WHO-reported prevalence of 14.9% (35). Additionally, medications commonly used by persons with migraine which may affect atherosclerosis, such as aspirin or non-steroid anti-inflammatory drugs, were not corrected for. Any such unmeasured factors could be a source of residual confounding. Reverse causality could also be an issue, as persons with migraine could have been more health-conscious and consequently had lower levels of atherosclerosis. However, we adjusted our models for atherosclerosis risk factors, which could account for possible confounding by the medication use and cardiovascular risk profile of the study subjects. Finally, our cross-sectional analysis does not allow for conclusions regarding temporality of the associations.
In conclusion, in this large population-based study, we found that persons with migraine had less calcification in the intracranial carotid artery compared to persons without migraine. Future studies are needed to confirm these findings and understand potential mechanisms underlying the risk of CVD in persons with migraine.
Public health relevance
Lifetime history of migraine was associated with less intracranial carotid artery calcification when compared to no history of migraine. Lifetime history of migraine was not associated with calcification of the coronary arteries, aortic arch, or extracranial carotid artery.
Footnotes
Author contributions
The author contributions were as follows: KW, MK, OHF, DB and MAI conceived and designed the study. KW performed the data analyses and MAI, MK and OHF supervised the analyses. KW and MK drafted the final manuscript. MK, OHF, MAI, MV, AM and DB contributed to the critical revision of the manuscript and approved the final version. KW (Department of Epidemiology, Erasmus MC University Medical Center, Rotterdam, The Netherlands) had full access to all the data in the study and takes responsibility for the integrity of the data and the accuracy of the data analysis.
Acknowledgements
The invaluable contributions of inhabitants, general practitioners and pharmacists of Ommoord are gratefully acknowledged.
Declaration of conflicting interests
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Ke-xin Wen and Oscar Franco work in ErasmusAGE, a center for aging research at the Erasmus MC funded by Nestlé Nutrition (Nestec Ltd), Metagenics Inc. and AXA. Maryam Kavousi is supported by VENI grant (91616079) from the Netherlands Organization for Health Research and Development (ZonMW). Mohammed Arfan Ikram received grants from the Netherlands Heart Foundation, ZonMW, Internationaal Parkinson Fonds, and Internationale Stichting Alzheimer Onderzoek. Antoinette MaassenVanDenBrink received grants from the Netherlands Organization for Health Research and Development ZonMw (VIDI grant 917.113.34 and other grants), the Netherlands Heart Foundation, and Netherlands Brain Foundation, as well as research grants and/or consultation fees from Amgen/Novartis, Lilly/CoLucid, Teva and ATI. The Rotterdam Study is supported by the Erasmus MC University Medical Center and Erasmus University Rotterdam, the Netherlands Organisation for Scientific Research (NWO), The Netherlands Organisation for Health Research and Development (ZonMW), the Research Institute for Diseases in the Elderly (RIDE), the Netherlands Genomics Initiative (NGI), the Ministry of Education, Culture and Science, the Ministry of Health, Welfare and Sports, the European Commission (DG XII), and the Municipality of Rotterdam. None of the funding organisations or sponsors were involved in the design and conduct of the study; collection, analysis, and interpretation of the data; and preparation, review, or approval of the manuscript.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
