Abstract
Objective:
The co-occurrence of type 2 diabetes and psychosis is an important form of medical comorbidity within individuals, but no large-scale study has evaluated comorbidity within families. The aim of this study was to determine whether there is evidence for familial comorbidity between type 2 diabetes and psychosis.
Method:
Data were analysed from an observational study of a nationally representative sample of 1642 people with psychosis who were in contact with psychiatric services at the time of survey (The 2010 Australian National Survey of Psychosis). Participants were aged 18–64 years and met World Health Organization’s International Classification of Diseases, 10th Revision diagnostic criteria for a psychotic disorder (857 with schizophrenia, 319 with bipolar disorder with psychotic features, 293 with schizoaffective disorder, 81 with depressive psychosis and 92 with delusional disorder or other non-organic psychoses). Logistic regression was used to estimate the association between a family history of diabetes and a family history of schizophrenia.
Results:
A positive family history of diabetes was associated with a positive family history of schizophrenia in those with a psychotic disorder (odds ratio = 1.35, p = 0.01, adjusted for age and gender). The association was different in those with an affective versus non-affective psychosis (odds ratio = 0.613, p = 0.019, adjusted for age and gender) and was significant only in those with a non-affective psychosis, specifically schizophrenia (odds ratio = 1.58, p = 0.005, adjusted for age and sex). Adjustment for demographic factors in those with schizophrenia slightly strengthened the association (odds ratio = 1.74, p = 0.001, adjusted for age, gender, diagnosis, ethnicity, education, employment, income and marital status).
Conclusion:
Elevated risk for type 2 diabetes in people with schizophrenia is not simply a consequence of antipsychotic medication; type 2 diabetes and schizophrenia share familial risk factors.
Introduction
People with psychosis are more likely to develop type 2 diabetes than people in the general community (Foley et al., 2014), and risk is increased after treatment with antipsychotic medication (Nielsen et al., 2010). The prevalence of type 2 diabetes in young adults is typically low, but at a mean age of 29 years, before any treatment with antipsychotic medication, Danes just diagnosed with schizophrenia already had a 1.27–1.63 times higher risk for diagnosed type 2 diabetes than those without schizophrenia (Cohen and De Hert, 2011). The elevated risk for type 2 diabetes in people with psychosis is therefore unlikely to be just a side effect of antipsychotic medication (Ramaswamy et al., 2006; Smith et al., 2008), and a variety of historical data and small proof of principle studies support the Danish population registry findings. The recent focus on diabetes risk associated with antipsychotic medication has sometimes obscured the fact that the co-occurrence of diabetes and schizophrenia within individuals was noted long before the advent of antipsychotic medication, before diabetes was sub-divided into types, and with different diagnostic systems for defining schizophrenia (Rouillon and Sorbara, 2005). Evidence of a familial link between diabetes and schizophrenia is more tentative.
The influential 19th-century psychiatrist Sir Henry Maudsley published in 1879 his observation that diabetes was often present in the families of people with psychosis (Maudsley, 1879). More recently, an important proof of principle study found an elevated rate of impaired glucose tolerance in physically healthy and non-obese people admitted to hospital with their first episode of psychosis but never medicated with antipsychotic drugs, and in their psychiatrically well relatives (Spelman et al., 2007). This study showed that antipsychotic medication effects and illness-related effects cannot account for all of the increased risk for diabetes-related traits within families, and is therefore consistent with a familial link between diabetes and psychosis, but it is based on a small and unrepresentative sample (of 38 cases, 38 controls and 44 relatives of cases). Two small family history studies of people with psychosis found an increased prevalence of type 2 diabetes in parents or grandparents (Fernandez-Egea et al., 2008a; Mukherjee et al., 1989), but these studies also ascertained small and unrepresentative samples of people with psychosis (34 and 70 hospitalized cases). The evidence for a familial link between diabetes and psychosis is therefore suggestive but still necessarily tentative. The aim of this study was to test if there was evidence for comorbidity between familial risk for schizophrenia and familial risk for diabetes in a large representative national sample of adults with psychosis.
Materials and methods
Data source
The Australian National Survey of Psychosis is a population-based prevalence study using a two-phase survey design with a target population defined by psychosis, place of residence, age 18–64 years and contact with public mental health services or relevant non-government organizations (Morgan et al., 2014, 2012). The survey was conducted in 2010 at seven sites across the five mainland Australian states. The research protocol for the study was approved by relevant institutional ethics committees, and all participants gave written informed consent. Of the 1825 participants in the study who screened positive for psychosis, 1624 met full criteria for a psychotic disorder, and their data are the subject of this report.
Measurements
Psychosis diagnosis
The Diagnostic Interview for Psychosis (Castle et al., 2006) was used to diagnose psychotic disorders following the International Classification of Diseases, 10th Revision (ICD-10) (World Health Organization [WHO], 2008). The psychoses can be sub-divided into non-affective (schizophrenia, schizoaffective disorder, delusional disorder or other non-organic psychoses) or affective (bipolar disorder with psychotic features, depressive psychosis) or individual psychosis diagnoses.
Diabetes diagnosis
Current type 2 diabetes was diagnosed according to the American Diabetes Association (2008) on the basis of a current fasting blood glucose reading of at least 7.0 mmol/L (126+ mg/dL) or current use of anti-diabetic medication.
Family history
Family history of schizophrenia was assessed by asking participants ‘Do you know of anyone in your family (including aunts, uncles or cousins) who has had schizophrenia?’
Family history of diabetes was assessed by asking participants ‘Do you have a family history of diabetes? (First degree relatives only; parents, siblings, children)’.
Demographic factors that may be associated with population structure
Country of birth was used to estimate ethnicity and was coded to identify those in high risk groups for diabetes in Australia – those of Aboriginal or Torres Strait Islander, Melanesian, Micronesian or Polynesian, Southern European, North African or Middle Eastern, or Asian, including those from the Indian subcontinent, descent. Secondary education was coded to reflect years completed and an additional variable coded if the participant had obtained a post-school qualification. Income was coded to reflect current fortnightly income ranging from less than AUD300 to more than AUD1000. Employment was coded in reference to the past year and was coded as employed (in any job), not employed but had a defined role (any combination of homemaker, caring for children, caring for other relatives, involved in volunteer or unpaid work, formally retired) or not employed with no formal activity (includes registered unemployment). Fortnightly income was coded in reference to the past 12 months. Marital status referred to current status at the time of survey.
Statistical analysis
Logistic regression was used to estimate the association between a family history of schizophrenia and a family history of diabetes. The outcome variable in all analyses was a family history of schizophrenia and the predictor variable was a family history of diabetes, adjusted for age in years and sex.
Associations are sometimes affected by population structure and this may lead to false positive results or failures to detect genuine associations. We therefore tested if any observed association between a family history of schizophrenia and a family history of diabetes after adjustment for variation in age and sex was altered after further adjustment for variation in ethnicity, education, employment, income and marital status. Diabetes in participants may affect the likelihood of detection of diabetes in relatives, and vice versa, and therefore reports of having a family history of diabetes. Accordingly, we tested whether the observed association between family history of schizophrenia and family history of diabetes, controlling for age, sex, ethnicity, education, employment, income and marital status was altered after further adjustment for participants’ diabetes diagnosis.
Tests for heterogeneity
All analyses were repeated for the sample stratified by sex, affective or non-affective psychosis, and for each individual psychosis diagnosis. The significance of any observed difference in the association between a family history of schizophrenia and a family history of diabetes by sex, affective versus non-affective psychosis, or by individual psychosis diagnosis was evaluated using the likelihood ratio test. All data analysis was conducted using SPSS Version 22 (IBM Corp., 2014).
Results
Characteristics of the sample
Most participants in the survey were diagnosed with a non-affective psychosis (75.6%) (Table 1). The most common psychosis diagnosis was schizophrenia (52.2%). A positive family history of schizophrenia was reported by 28.1% and a positive family history of diabetes was reported by 37.2% of participants. Most participants had not completed secondary school (68%), were currently unemployed (67.5%) and had never married (63.0%). A minority of participants were from high risk ethnic groups for type 2 diabetes, the most common of which was Aboriginal or Torres Strait Islander (5.1%). The mean age of the sample was 38.24 years (standard deviation [SD] = 11.08, median = 37, range = 18–64 years) and 38.6% were female.
Characteristics of the sample.
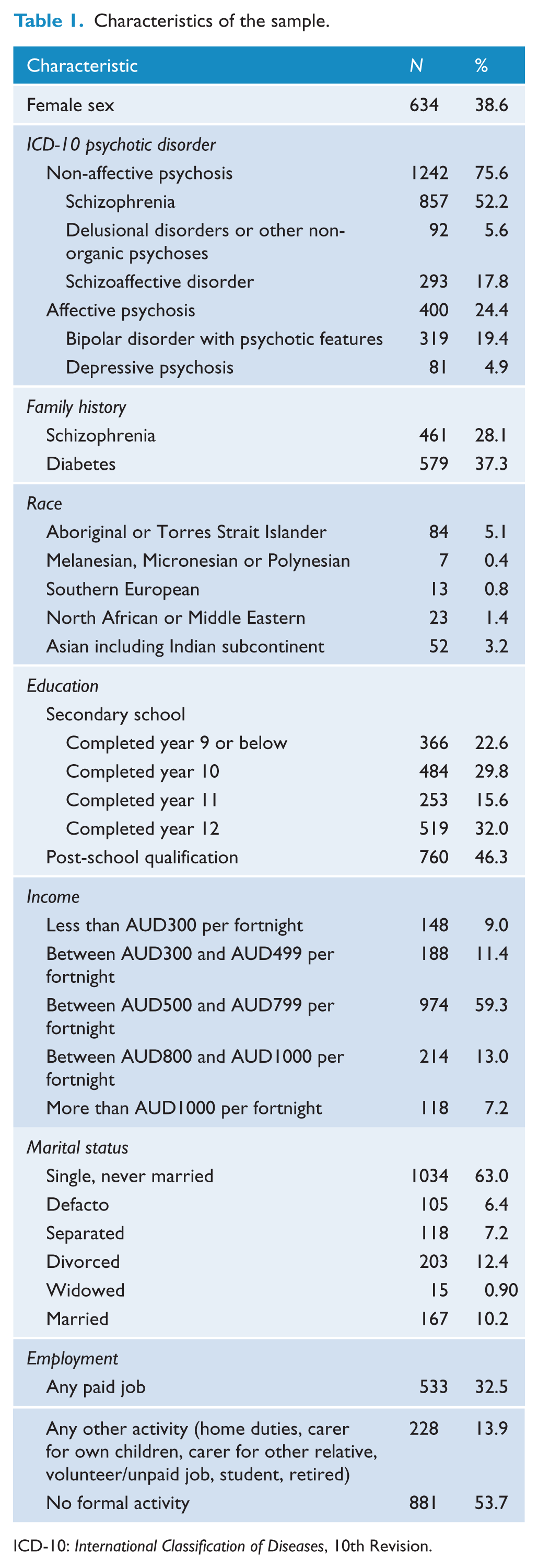
ICD-10: International Classification of Diseases, 10th Revision.
Familial comorbidity between schizophrenia and diabetes
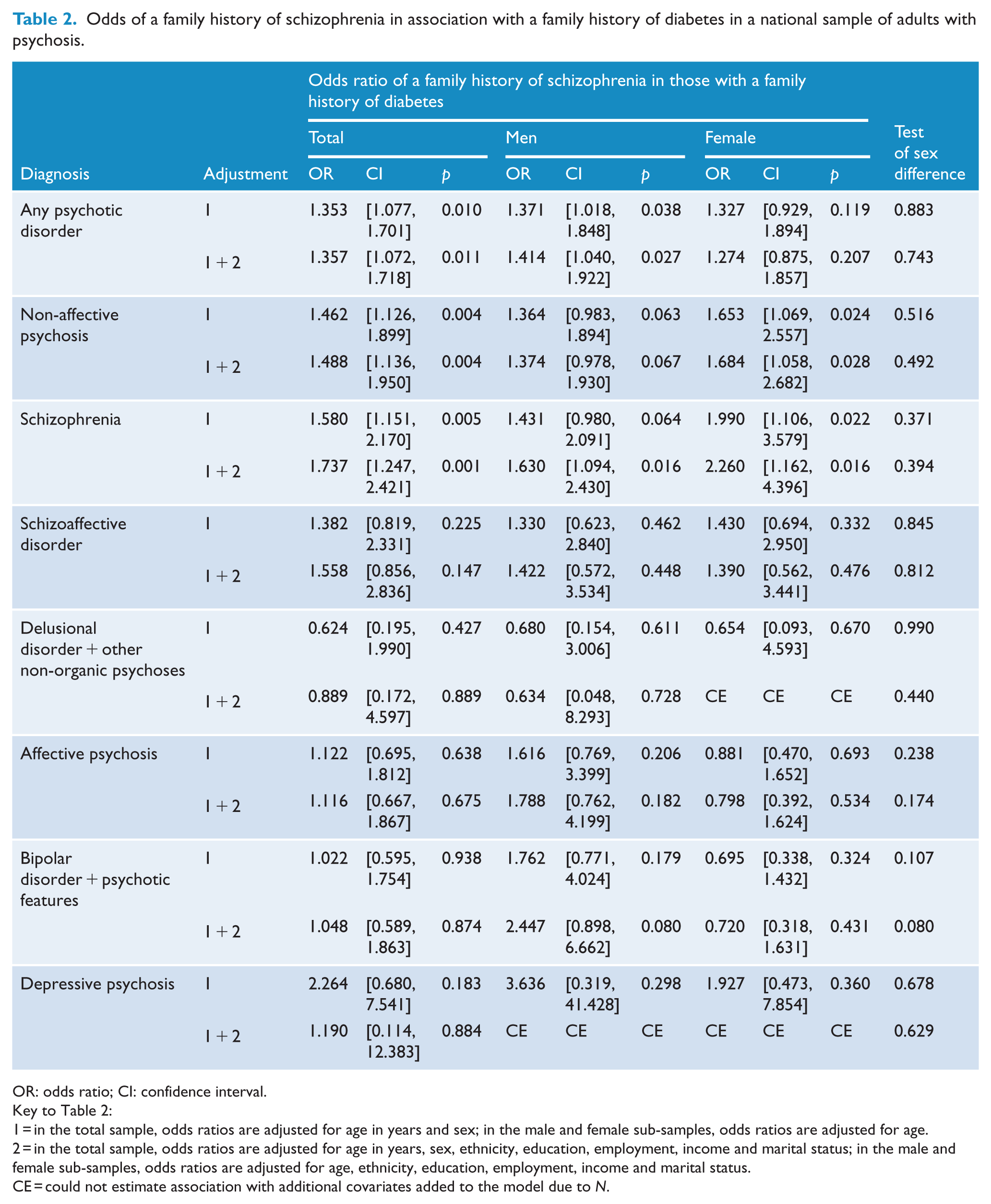
A family history of diabetes predicted a family history of schizophrenia in adults with a psychotic disorder (odds ratio [OR] = 1.35, 95% confidence interval [95% CI] = [1.08, 1.70], p = 0.01, adjusted for age and sex; OR = 1.35, 95% CI = [1.07, 1.71], p = 0.01, adjusted for age, sex, ethnicity, education, employment, income and marital status) (Table 2). This association was not altered by adjustment for affective versus non-affective psychosis (OR = 1.38, 95% CI = [1.09, 1.73], p = 0.006, adjusted for age, sex and affective versus non-affective psychosis; OR = 1.38, 95% CI = [1.09, 1.74], p = 0.008, adjusted for age, sex, affective versus non-affective psychosis, ethnicity, education, employment, income and marital status) or by adjustment for individual psychosis diagnoses (OR = 1.38, 95% CI = [1.09, 1.673], p = 0.006, adjusted for age, sex and schizoaffective disorder, bipolar disorder with psychotic features, depressive psychosis or delusional disorder or other non-organic psychosis versus schizophrenia; OR = 1.37, 95% CI = [1.08, 1.74], p = 0.008, adjusted for age, sex and schizoaffective disorder, bipolar disorder with psychotic features, depressive psychosis or delusional disorder or other non-organic psychosis versus schizophrenia, ethnicity, education, employment, income and marital status). Depressive psychosis, however, had a significantly weaker association with a family history of schizophrenia than schizophrenia (OR = 0.53, 95% CI = [1.09, 1.73], p = 0.006, adjusted for age, sex and family history of diabetes). The association between other psychosis diagnoses and a family history of schizophrenia was not significantly different from the association between schizophrenia and a family history of schizophrenia (schizoaffective disorder: OR = 1.23, 95% CI = [0.91, 1.65], p = 0.17; bipolar disorder with psychotic features: OR = 0.79, 95% CI = [0.58, 1.09], p = 0.15; delusional disorder or other non-organic psychoses: OR = 0.82, 95% CI = [0.49, 1.37], p = 0.45; all adjusted for age, sex and family history of diabetes).
Odds of a family history of schizophrenia in association with a family history of diabetes in a national sample of adults with psychosis.
OR: odds ratio; CI: confidence interval.
Key to Table 2:
1 = in the total sample, odds ratios are adjusted for age in years and sex; in the male and female sub-samples, odds ratios are adjusted for age.
2 = in the total sample, odds ratios are adjusted for age in years, sex, ethnicity, education, employment, income and marital status; in the male and female sub-samples, odds ratios are adjusted for age, ethnicity, education, employment, income and marital status.
CE = could not estimate association with additional covariates added to the model due to N.
After stratifying the sample by type of psychosis, the association between a family history of diabetes and a family history of schizophrenia was only significant in those with a non-affective psychosis, specifically schizophrenia (OR = 1.58, 95% CI = [1.15, 2.17], p = 0.005, adjusted for age and sex) (Table 2). Adjustment for demographic factors slightly strengthened the association in those with non-affective psychosis, including those with schizophrenia (OR = 1.74, 95% CI = [1.24, 2.42], p = 0.001) (Table 2). Adjustment for demographic factors slightly weakened the association in those with affective psychosis, specifically depressive psychosis, but the association between a family history of diabetes and a family history of schizophrenia in those with depressive psychosis was non-significant with or without adjustment for demographic variables (Table 2).
Further adjustment for the participant’s diabetes diagnosis did not alter the association observed between family history of diabetes and family history of schizophrenia in those with schizophrenia: after adjusting for age, sex and participant diabetes (1.47, p = 0.049) or after adjusting for age, sex, demographic variables and participant diabetes (1.74, p = 0.001).
Tests for heterogeneity
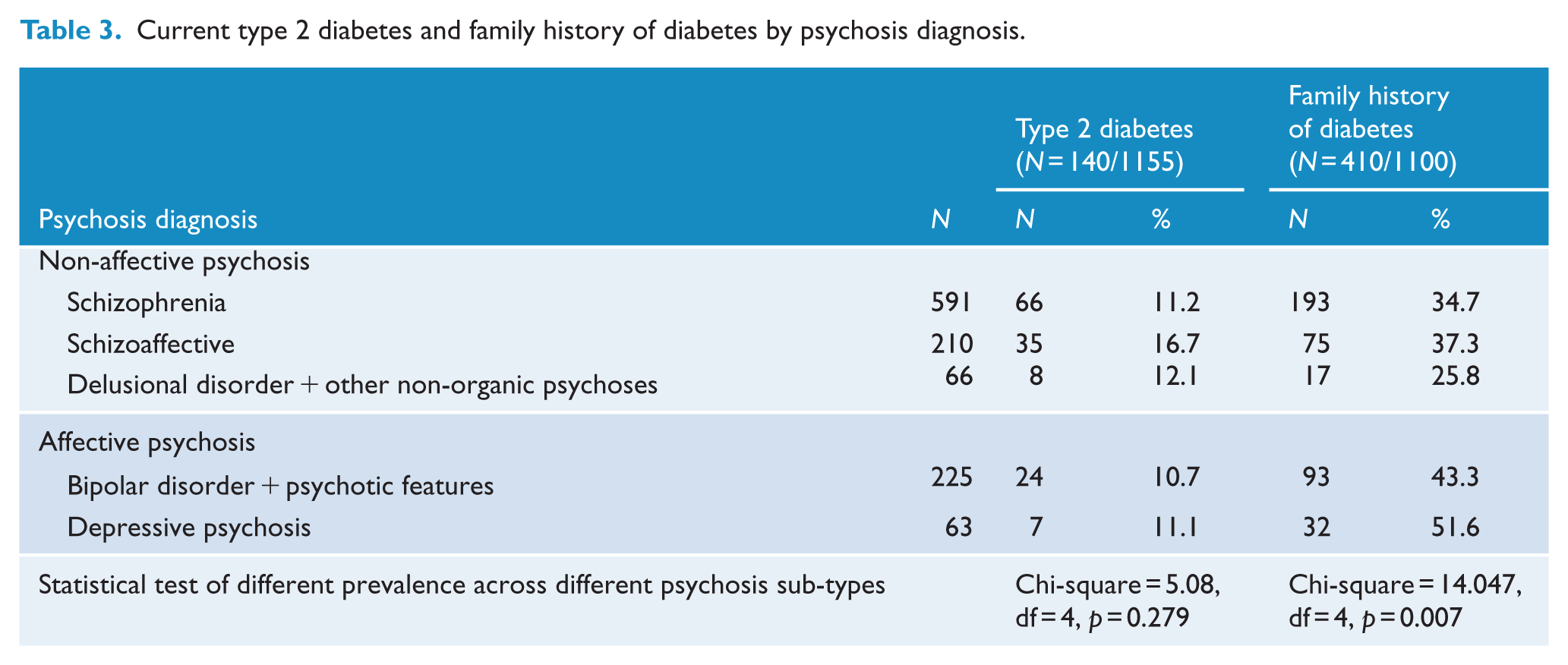
The prevalence of a family history of diabetes, but not current type 2 diabetes, varied markedly according to psychosis sub-type (Table 3).
Current type 2 diabetes and family history of diabetes by psychosis diagnosis.
There was a significant difference in the association between a family history of diabetes and a family history of schizophrenia in those with an affective versus non-affective psychosis (OR = 0.613, p = 0.019).
There was no significant difference in the association between a family history of diabetes and a family history of schizophrenia in those with delusional disorder or other non-organic psychosis (OR = 0.46, p = 0.13), depressive psychosis (OR = 0.59, p = 0.18), schizoaffective disorder (OR = 1.18, p = 0.47) or bipolar disorder with psychotic features (OR = 0.62, p = 0.053) versus schizophrenia.
There was no significant difference in the association between a family history of diabetes and a family history of schizophrenia in men and women with any psychotic disorder, men and women with an affective psychosis, men and women with a non-affective psychosis or men and women with any specific psychotic disorder (Table 2).
Discussion
A family history of diabetes is significantly associated with a family history of schizophrenia in this large national sample of adults with psychosis. The observed association could not be explained by demographic factors that may create population structure, including ethnicity, which is a common source of spurious associations (Knowler et al., 1988). The present study therefore demonstrates evidence of a familial link between diabetes and psychosis in a large, unselected sample that is consistent with previous findings from small selected samples of people with psychosis (Fernandez-Egea et al., 2008a; Mukherjee et al., 1989; Spelman et al., 2007). The association between a family history of diabetes and a family history of schizophrenia was only significant here in those with a non-affective psychosis, specifically schizophrenia.
It has previously been suggested that schizophrenia and type 2 diabetes may share genetic risk factors (Bellivier, 2005; Gough and O’Donovan, 2005; Lin and Shuldiner, 2010; Van Lieshout and Voruganti, 2008). Gene pathways that have been associated with type 2 diabetes and schizophrenia include those associated with calcium, g-secretase mediated ErbB4, adipocytokine, insulin and AKT signalling (Liu et al., 2013). Not all people with psychosis develop diabetes or have family histories of diabetes, and identification of possible genetic heterogeneity within schizophrenia (Arnedo et al., 2015) may therefore prove helpful for understanding the source of familial comorbidity with diabetes. Given abnormalities in glucose metabolism or a diagnosis of type 2 diabetes are found in some people with psychosis very early in the course of illness, before treatment with antipsychotic medication (Cohen and De Hert, 2011; Spelman et al., 2007), and at rates that exceed those in controls, we cannot exclude the possibility that glucose abnormalities may be implicated in the aetiology of psychosis in some cases.
Several environmental factors have been proposed to explain comorbidity between type 2 diabetes and psychosis within individuals, including the effect of prenatal stress, such as the effect of famine, pregnancy and birth complications, foetal iron deficiency, low birth weight, and a gene–environment interaction involving the maternal immune system and exposure to infectious agents during pregnancy (see discussions in Fernandez-Egea et al., 2008a, 2008b; Gilvarry et al., 1996; Van Lieshout and Voruganti, 2008). For any of these putative candidates to constitute a familial environmental risk factor that might explain some portion of the familial comorbidity between schizophrenia and diabetes found in the present study, these risk factors must be shared by relatives with diabetes, and therefore constitute a common environmental risk factor for diabetes, and shared by relatives with psychosis, and therefore constitute a common environmental risk factor for psychosis. The environmental risk factors proposed as possible explanatory factors for comorbidity within individuals are more likely to represent individual specific environmental risk factors than shared familial environmental risk factors common to diabetes and psychosis. Lifestyle factors such as diet and physical activity are possible familial environmental candidates (that may interact with genetic risk). Study designs that can separate genetic and familial environmental influences on these behaviours will be required to test this hypothesis (D’Onofrio et al., 2013). The sevenfold increased risk for schizophrenia associated with maternal diabetes during pregnancy (Van Lieshout and Voruganti, 2008) is consistent with a complex interplay between genetic effects and associated intrauterine effects and their sequelae on risk for schizophrenia and serves to remind us that there is more to the relationship between diabetes and psychosis than antipsychotic medication effects after psychosis has been diagnosed. Provocative statements have been made about this issue and we hope they serve to stimulate further research in this area: Perhaps the next era in psychiatry must approach schizophrenia as a systemic disease that first presents as altered behaviour rather than as a brain disease per se. We might feasibly learn more about the disease from its comorbidities than from enhanced measures of brain functioning. This step would be yet another echo from the idea that schizophrenia may be a form of familial diabetes. (Malaspina, 2013)
Limitations
Family history was assessed by asking participants in this survey about their relatives. We are not aware of any large-scale family interview and screening studies that document familial comorbidity of psychosis and type 2 diabetes. Direct assessment of diabetes in relatives would require screening and not just personal interview or access to medical records that record diabetes diagnoses because half of all type 2 diabetes cases in Australia are not diagnosed (Dunstan et al., 2002) and self-report will therefore underestimate the true level of familial risk. Based on the epidemiology of diabetes in the general Australian population we can estimate that 85–90% of diabetes cases are likely to be type 2. Assessment of family history of diabetes did not distinguish maternal and paternal histories of diabetes, and we therefore could not test for maternal effects on familial comorbidity.
Footnotes
Acknowledgements
The members of the survey study group are: V Morgan (National Project Director), A Jablensky (Chief Scientific Advisor), A Waterreus (National Project Coordinator), R Bush, V Carr, D Castle, M Cohen, C Galletly, C Harvey, B Hocking, A Mackinnon, P McGorry, J McGrath, A Neil, S Saw and H Stain. This report acknowledges, with thanks, the hundreds of mental health professionals who participated in the preparation and conduct of the survey and the many Australians with psychotic disorders who gave their time and whose responses form the basis of this publication.
Declaration of interest
Dr Foley, Professors Mackinnon, Morgan and Watts, and Ms Waterreus have no conflict of interest to disclose. Professor Castle has received grant monies for research from Eli Lilly, Janssen Cilag, Roche, Allergen, Bristol-Myers Squibb, Pfizer, Lundbeck, Astra Zeneca, Hospira; Travel Support and Honoraria for Talks and Consultancy from Eli Lilly, Bristol-Myers Squibb, Astra Zeneca, Lundbeck, Janssen Cilag, Pfizer, Organon, Sanofi-Aventis, Wyeth, Hospira, Servier; and is a current or past Advisory Board Member for Lu AA21004: Lundbeck; Pristiq: Pfizer; Varenicline: Pfizer; Asenapine: Lundbeck; Seroquel: Astra Zeneca; Bitopertin: Roche; Aripiprazole LAI: Lundbeck. He has no stocks or shares in any pharmaceutical company. Professor Galletly reports advisory board membership with Janssen Cilag, travel costs and honoraria for providing educational sessions for Lundbeck, support for conference attendance from Servier, and clinical trials for Lundbeck, Forum Pharmaceuticals, Bristol-Myers Squibb and Janssen Cilag.
Funding
The collection of data and the associated prevalence study was funded by the Australian Government Department of Health and Ageing. Debra Foley and Andrew Mackinnon thank the Colonial Foundation (Australia) and the Australian Federal Government for their support of Orygen, The National Centre of Excellence in Youth Mental Health and their support of their work on this manuscript.
