Abstract
Objective:
Oxidative imbalance has emerged as a treatment target in bipolar disorder. As very limited data are available on the clinical use of antioxidants for mania, we report here results from a post hoc and exploratory subgroup analysis of a randomized, placebo-controlled trial of N-acetyl cysteine (NAC).
Methods:
This was a placebo-controlled, randomized, clinical trial assessing the effect of NAC over 24 weeks in mania or hypomania. Symptomatic and functional outcomes were collected over the study period.
Results:
Fifteen participants were available for this report; two participants in each group failed to complete all assessments. Within-group analyses pointed to an improvement in the NAC group on manic symptoms and worsening in the placebo group on depressive symptoms at endpoint.
Conclusions:
Although the sample size was small, these results indicated within-group efficacy for this glutathione precursor as compared to placebo. Future trials specifically designed to demonstrate the efficacy of NAC in mania are needed.
Introduction
Oxidative imbalance has been shown to be pervasive in bipolar disorder (Berk et al., 2011b; Grande et al., 2012; Magalhães et al., 2012b, 2012c). Animal and clinical studies converge to suggest that acute manic episodes are especially associated with oxidative damage (Frey et al., 2006, 2007; Kapczinski et al., 2011). Adding to this, disturbances in antioxidant enzymes have also been found during mania (Andreazza et al., 2007). With these new findings, there is growing interest in developing new interventions that directly target oxidative biology in bipolar disorder (Dean et al., 2009; Ng et al., 2008).
One promising avenue has been the study of the glutathione system (Berk et al., 2008b). Glutathione, the brain’s major antioxidant, is a generic, non-enzymatic, cellular, free-radical scavenger (Dodd et al., 2008). Recent data showing reduced brain glutathione, further points to its involvement in bipolar disorder (Andreazza et al., 2010). In this respect, N-acetyl cysteine (NAC), a glutathione precursor, may provide symptomatic relief by replenishing its levels and thus mitigating redox imbalance (Berk et al., 2008b; Choy et al., 2010; Dean et al., 2011a, 2011b).
Considering the extant evidence, adjunctive antioxidants have been little explored in mania. As previously reported, add-on NAC was effective for bipolar disorder in a naturalistic, randomized, placebo-controlled trial with patients in the different phases of the disorder (Berk et al., 2008a; Magalhães et al., 2011a, 2012b). In this report, we explore the effects of this compound in a subset of those participants who had mania at baseline.
Methods
A detailed description on the study recruitment and evaluation procedures have been published elsewhere (Berk et al., 2008a). Briefly, consenting individuals were randomized to receive 2 grams of NAC or placebo in addition to treatment as usual in a double-blind fashion. They needed to fulfill DSM-IV (Diagnostic and Statistical Manual of Mental Disorders, Fourth Edition) criteria for bipolar disorder I or II disorder, and be on stable therapy for at least 1 month prior to randomization. For this report, we only included those individuals with a manic or hypomanic episode at baseline. The trial was conducted in an outpatient setting. Exclusion criteria were kept to a minimum, and included systemic medical disorders, pregnant or lactating women, current use of NAC (500 mg/day), selenium (200 μg/day) or vitamin E (500 IU/day) and previous known intolerance or contraindication to NAC.
All participants provided written informed consent. The participating institutions’ research and ethics committees approved the trial. The study was registered with the Australian Clinical Trials Registry (registration number: 12605000362695).
The participants received two NAC (500 mg) capsules twice daily or matching placebo. They were screened at baseline with the Mini-International Neuropsychiatric Interview (Sheehan et al., 1998). Interviewers assessed mood and functional outcomes at baseline and at weeks 2, 4, 8, 12, 16, 20 and 24. The analysis included all randomized participants with at least one post-baseline assessment.
Interviewers assessed mood using the Bipolar Depression Rating Scale (BDRS; Berk et al., 2004, 2007), the Montgomery–Asberg Depression Rating Scale (MADRS; Montgomery and Asberg, 1979), and the Young Mania Rating Scale (YMRS; Young et al., 1978). The Clinical Global Impression (CGI; Spearing et al., 1997) was obtained as a measure of overall illness severity. Functioning and quality of life were assessed with the Global Assessment of Functioning (GAF; Jones et al., 1995), the Social and Occupational Functioning Assessment Scale (SOFAS; Morosini et al., 2000), the Streamlined Longitudinal Interview Clinical Evaluation for the Longitudinal Interval Follow-up Evaluation (SLICE-LIFE), the Longitudinal Interval Follow-up Evaluation – Range of Impairment Functioning Tool (LIFE-RIFT; Keller et al., 1987) and the Quality of Life Enjoyment and Satisfaction Questionnaire (Q-LES-Q; Endicott et al., 1993).
All analyses are based on the intention-to-treat population. Sample size precluded parametric analyses, so the investigation of change trajectory in this report was not possible (for results on the whole sample as well as original sample size estimation, see Berk et al., 2008a). We considered participants to have had a depressive switch when they did not have any relevant depressive symptoms at baseline, but did so at endpoint. Similarly, we considered participants to have persistent manic symptoms when a remission was not achieved. For these outcomes, we used a MADRS score of 10 and an YMRS score of 7 as cut-off points (Berk et al., 2008c). We present distributions on quantitative measures as medians and ranges to give an idea of extremes. Differences from baseline to endpoint were compared using the Wilcoxon rank sum test and differences between groups were compared with Mann–Whitney’s U.
Results
Fifteen participants were available for this report: eight in the NAC group and seven in the placebo group (Table 1). As expected from the recruitment in outpatient settings, participants were moderately ill, with a median CGI score of 4 in the NAC group and 3 in the placebo group. Table 2 shows baseline and endpoint scores for the two groups; between-group differences were not significant, as expected. Within-group analyses pointed to an improvement in the NAC group on YMRS scores (Z = 2.05, p = 0.041) and worsening in placebo group on BDRS scores (Z = 1.96, p = 0.050) at endpoint. Figure 1 illustrates the categorical outcomes in the two groups.
Demographical, clinical and treatment characteristics of the study sample at baseline.
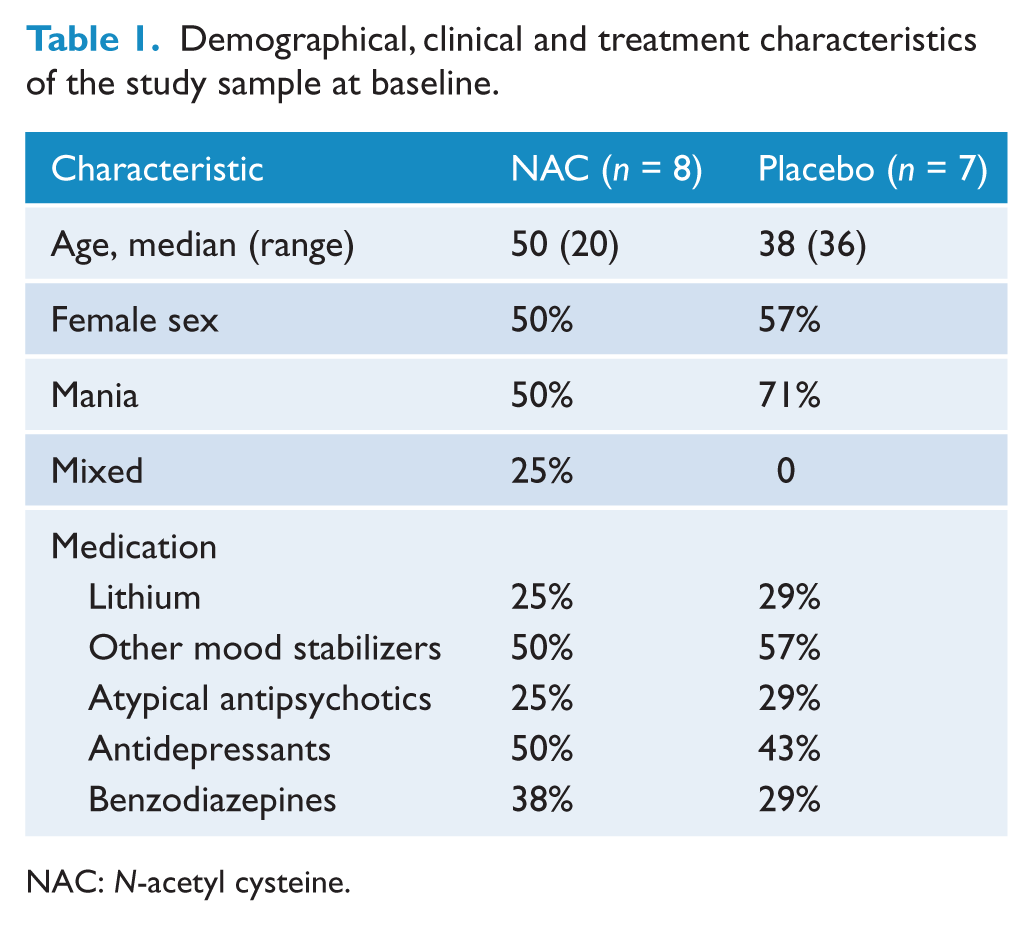
NAC: N-acetyl cysteine.
Baseline and endpoint rating scale scores according to intervention group.
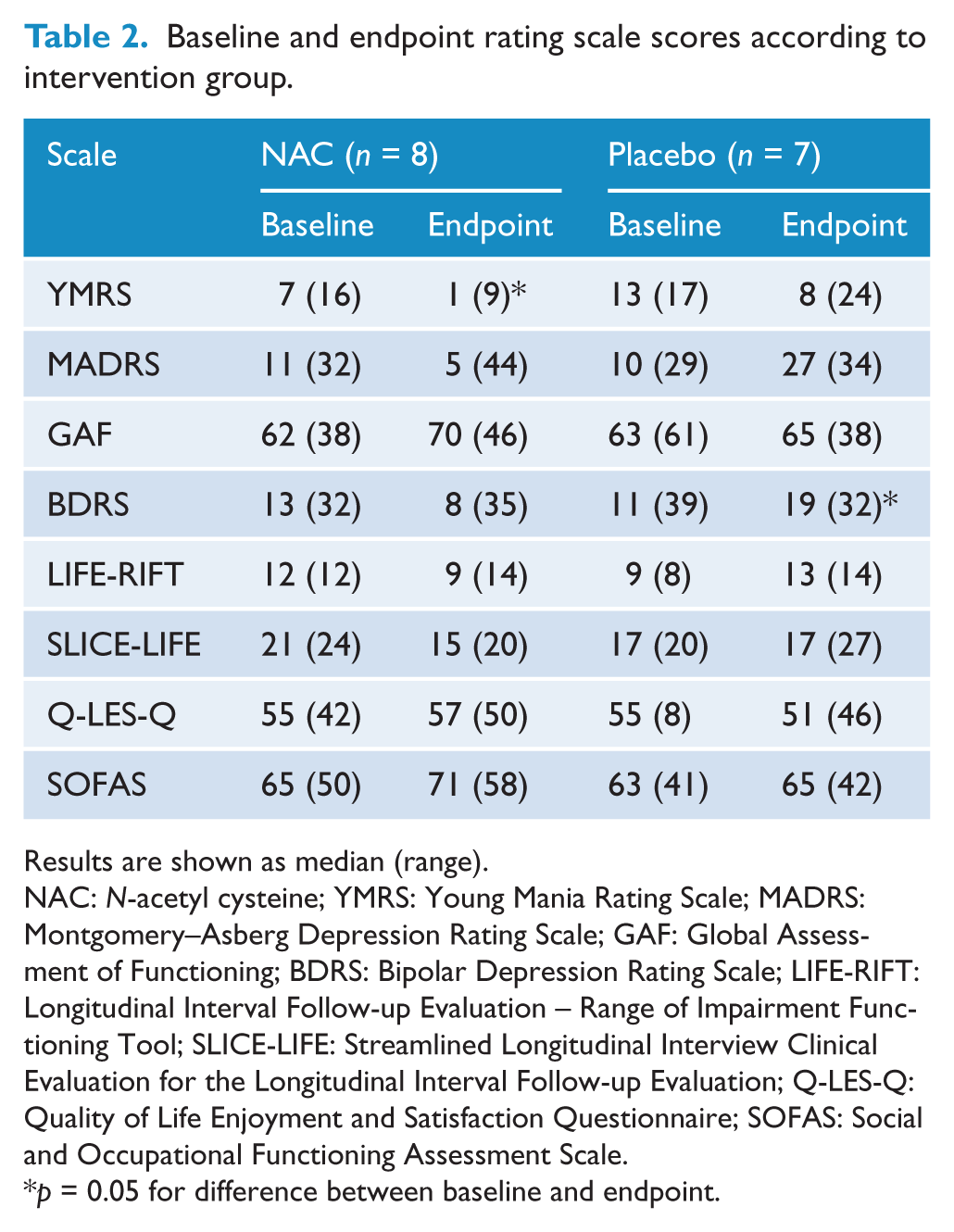
Results are shown as median (range).
NAC: N-acetyl cysteine; YMRS: Young Mania Rating Scale; MADRS: Montgomery–Asberg Depression Rating Scale; GAF: Global Assessment of Functioning; BDRS: Bipolar Depression Rating Scale; LIFE-RIFT: Longitudinal Interval Follow-up Evaluation – Range of Impairment Functioning Tool; SLICE-LIFE: Streamlined Longitudinal Interview Clinical Evaluation for the Longitudinal Interval Follow-up Evaluation; Q-LES-Q: Quality of Life Enjoyment and Satisfaction Questionnaire; SOFAS: Social and Occupational Functioning Assessment Scale.
p = 0.05 for difference between baseline and endpoint.
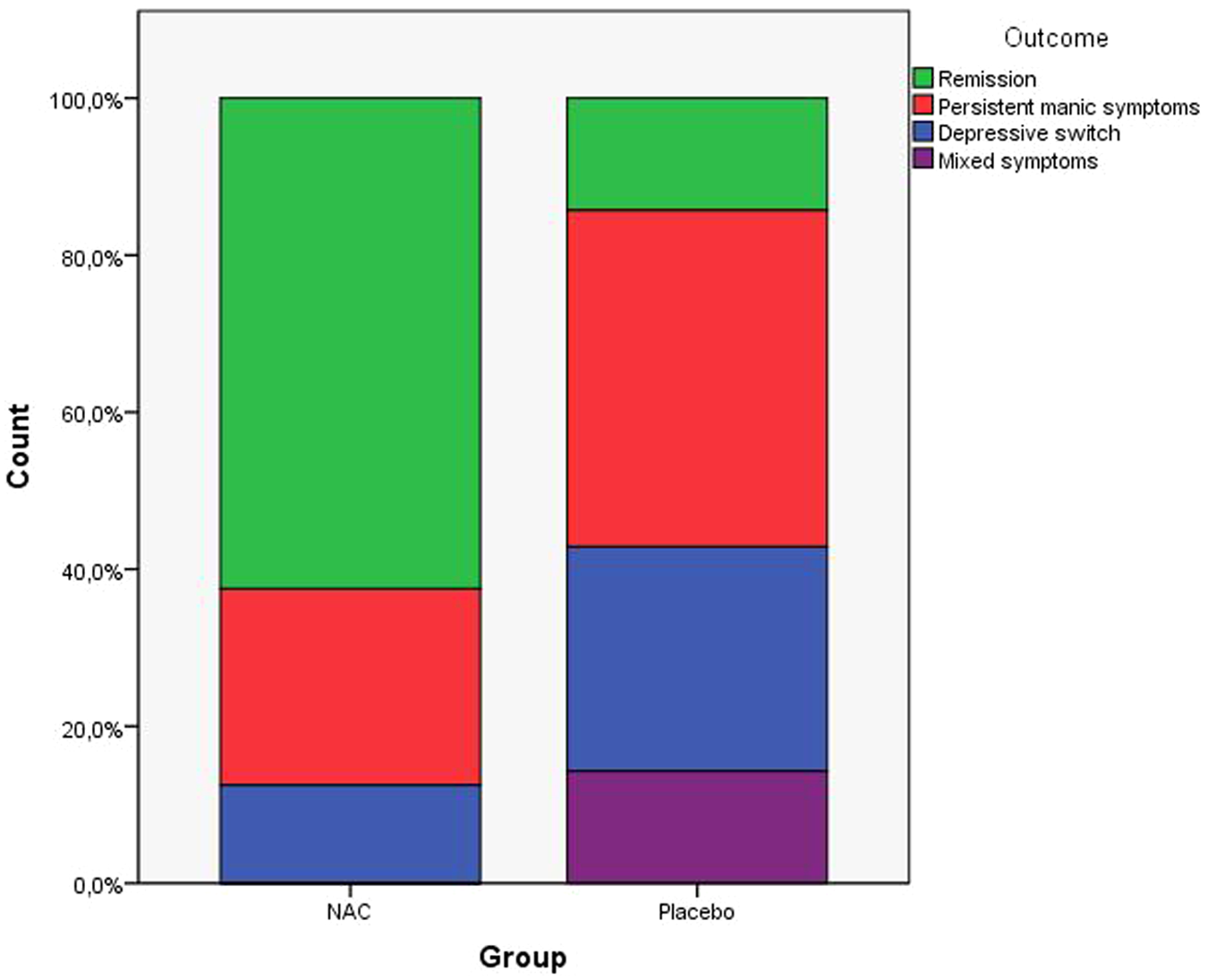
Categorical outcomes at endpoint according to treatment group. We considered participants to have had a depressive switch when they did not have any relevant depressive symptoms at baseline, but did so at endpoint. Similarly, we considered participants to have persistent manic symptoms when a remission was not achieved. For these outcomes, we used a MADRS score of 10 and an YMRS score of 7 as cut-off points.
Two participants in each group failed to complete all assessments: two participants in the placebo group and one in the NAC group withdrew their consent, and one participant in the NAC group discontinued due to an adverse event.
Discussion
In this small subset of participants with mania or hypomania, add-on NAC showed preliminary evidence of efficacy. These exploratory within-group analyses suggest that those individuals on NAC tended to have less manic symptoms at endpoint, while those on placebo tended to have more depressive symptoms at endpoint. Although not statistically significant, more participants in the NAC group also had complete symptom remissions at endpoint.
There is current interest in investigating interventions with novel mechanisms of action in bipolar disorder (Yatham et al., 2009). Oxidative stress is one of these new relevant targets (Machado-Vieira et al., 2010) and (with the possible exception of allopurinol (Machado-Vieira et al., 2008)) clinical trials of antioxidants for mania are largely lacking. Clinical and animal studies demonstrate that mood stabilizers improve oxidative stress parameters (Ng et al., 2008; Frey et al., 2006; Machado-Vieira et al., 2010). It also should be mentioned that NAC appears to have glutamate-modulating properties (Moussawi et al., 2009) and, given that increased glutamatergic neurotransmission has been found in bipolar disorder (Yildiz-Yesiloglu and Ankerst, 2006), this may represent an additional therapeutic pathway. On the whole, NAC has proven to be an interesting add-on drug in psychiatric disorders (Berk et al., 2011a; Dean et al., 2011a, 2012), both due to its benign profile of side effects (Dodd et al., 2008, 2009) and growing evidence regarding its efficacy (Berk et al., 2000, 2008a). There is also a suggestion from this same clinical trial (Magalhães et al., 2012a) that NAC is especially effective in later stages of bipolar disorder. This would concur with preclinical data showing that there needs to be increasing oxidative stress as a substrate for NAC to have an effect (Harvey et al., 2008). Although there is hope that NAC may prove to be an agent able to halt neuroprogression, at this point there is insufficient clinical evidence (Berk et al., 2012).
Despite being double-blind and placebo-controlled, the limited number of participants with mania or hypomania included is a major limitation of this report. Mania was not the a priori hypothesis of the original study. Participants were also recruited from outpatient settings and had moderate illness severity at baseline. Because of this, it is difficult to generate clinical recommendations solely based on the extant data. Future trials specifically designed to demonstrate the efficacy of NAC in mania are needed. Provided it is effective and safe, the addition of an efficacious nutraceutical agent to the current standard treatments would be welcome.
Footnotes
Funding
Declaration of interest
Professors Berk and Bush are co-inventors on two provisional patents regarding the use of NAC and related compounds for psychiatric indications, which, whilst assigned to the Mental Health Research Institute, could lead to personal remuneration upon a commercialization event.
Dr Magalhães is supported by a post-doctoral scholarship from Conselho Nacional de Desenvolvimento Científico e Tecnológico (CNPq), Brazil. Prof. Bush is a shareholder and consultant for Prana Biotechnology Ltd and a shareholder in Cogstate Ltd. Prof. Malhi has served on a number of international and national pharmaceutical advisory boards, received funding for research and has been in receipt of honoraria for talks at sponsored meetings worldwide involving the following companies: AstraZeneca, Eli Lilly, Janssen-Cilag, Lundbeck, Organon, Pfizer, Ranbaxy, Servier and Wyeth. Prof. Berk has received research support from: Beyond Blue, Bristol Myers Squibb, Eli Lilly, Geelong Medical Research Foundation, Glaxo SmithKline, Mayne Pharma, MBF, NHMRC, Novartis, Organon, Servier and Stanley Medical Research Foundation. He has been a speaker for: AstraZeneca, Bristol Myers Squibb, Eli Lilly, Glaxo SmithKline, Janssen-Cilag, Lundbeck, Merck, Pfizer, Sanofi Synthelabo, Servier, Solvay and Wyeth. He has been a consultant for: AstraZeneca, Bristol Myers Squibb, Eli Lilly, Glaxo SmithKline, Janssen-Cilag, Lundbeck and Servier.
