Abstract
Objectives:
To provide guidance for the management of mood disorders, based on scientific evidence supplemented by expert clinical consensus and formulate recommendations to maximise clinical salience and utility.
Methods:
Articles and information sourced from search engines including PubMed and EMBASE, MEDLINE, PsycINFO and Google Scholar were supplemented by literature known to the mood disorders committee (MDC) (e.g., books, book chapters and government reports) and from published depression and bipolar disorder guidelines. Information was reviewed and discussed by members of the MDC and findings were then formulated into consensus-based recommendations and clinical guidance. The guidelines were subjected to rigorous successive consultation and external review involving: expert and clinical advisors, the public, key stakeholders, professional bodies and specialist groups with interest in mood disorders.
Results:
The Royal Australian and New Zealand College of Psychiatrists clinical practice guidelines for mood disorders (Mood Disorders CPG) provide up-to-date guidance and advice regarding the management of mood disorders that is informed by evidence and clinical experience. The Mood Disorders CPG is intended for clinical use by psychiatrists, psychologists, physicians and others with an interest in mental health care.
Conclusions:
The Mood Disorder CPG is the first Clinical Practice Guideline to address both depressive and bipolar disorders. It provides up-to-date recommendations and guidance within an evidence-based framework, supplemented by expert clinical consensus.
Mood Disorders Committee:
Professor Gin Malhi (Chair), Professor Darryl Bassett, Professor Philip Boyce, Professor Richard Bryant, Professor Paul Fitzgerald, Dr Kristina Fritz, Professor Malcolm Hopwood, Dr Bill Lyndon, Professor Roger Mulder, Professor Greg Murray, Professor Richard Porter and Associate Professor Ajeet Singh.
International expert advisors:
Professor Carlo Altamura, Dr Francesco Colom, Professor Mark George, Professor Guy Goodwin, Professor Roger McIntyre, Dr Roger Ng, Professor John O’Brien, Professor Harold Sackeim, Professor Jan Scott, Dr Nobuhiro Sugiyama, Professor Eduard Vieta, Professor Lakshmi Yatham.
Australian and New Zealand expert advisors:
Professor Marie-Paule Austin, Professor Michael Berk, Dr Yulisha Byrow, Professor Helen Christensen, Dr Nick De Felice, A/Professor Seetal Dodd, A/Professor Megan Galbally, Dr Josh Geffen, Professor Philip Hazell, A/Professor David Horgan, A/Professor Felice Jacka, Professor Gordon Johnson, Professor Anthony Jorm, Dr Jon-Paul Khoo, Professor Jayashri Kulkarni, Dr Cameron Lacey, Dr Noeline Latt, Professor Florence Levy, A/Professor Andrew Lewis, Professor Colleen Loo, Dr Thomas Mayze, Dr Linton Meagher, Professor Philip Mitchell, Professor Daniel O’Connor, Dr Nick O’Connor, Dr Tim Outhred, Dr Mark Rowe, Dr Narelle Shadbolt, Dr Martien Snellen, Professor John Tiller, Dr Bill Watkins, Dr Raymond Wu.
Keywords
Introduction
Overview
The mood disorders committee (MDC) has been mindful that effective clinical care involves the art of applying clinical knowledge and skills to the individual needs of those presenting for care. A primary motivation was to ensure that the guideline was engaging and useful to clinicians. For this reason a balance of academically focused and clinically focused experts in the field has been engaged in the writing of the guideline. Genuine involvement of clinicians of other disciplines, people with experience of mental illness, and carers has occurred to better balance the guideline.
Human beings are complex biological systems, with mind as an emergent property. For this reason there are inherent uncertainties regarding diagnostic formulation and optimal care. It is anticipated that this guideline will assist clinicians to better navigate complex and challenging clinical scenarios. Tailoring care to the individual in the context of an effective working relationship is the foundation upon which the proper application of this guideline relies.
Aim
The Mood Disorders Clinical Practice Guideline (Mood Disorders CPG) has been developed by the Royal Australian and New Zealand College of Psychiatrists (RANZCP) to guide the clinical management of real-world depressive and bipolar disorders and to advise specifically on diagnosis and treatment strategies. The guideline focuses primarily on adults and briefly addresses special populations such as children and adolescents, pregnant and post-partum women, the elderly and those with common medical illnesses.
The guideline is primarily directed at psychiatrists, psychologists and physicians with a particular interest in mental health issues. It will also be of benefit to other mental health care professionals, hospital administrators and government policy makers. It amalgamates evidence-based knowledge with clinical wisdom and makes recommendations for real-world practice.
Structure
The Mood Disorders CPG comprises four sections:
Section 1: Classification of mood disorders
Section 2: Clinical management of major depressive disorder (MDD)
Section 3: Clinical management of bipolar disorder (BD)
Section 4: Clinical management of mood disorders with complex presentations and in special populations
Scope
Ever since Kraepelin separated manic-depressive insanity from dementia praecox, the nosological status of mood disorders has been in flux (Kraepelin, 1899). In practice, two diagnostic questions that clinicians commonly face are: (1) How to diagnose depression; and (2) When to diagnose bipolar disorder on the basis of emerging (hypo)manic symptoms. Therefore, this guideline discusses the diagnosis and management of both depressive and bipolar disorders.
The reason for addressing both groups of mood disorders together is that, in recent years, the overlap between these ‘categories’ has been of particular interest and increasingly they have been conceptualized as part of a spectrum. In clinical practice mood disorders diagnoses often change reflecting movement along this spectrum, e.g. from Major Depressive Disorder (MDD) to Bipolar I Disorder (BD I), sometimes ‘via’ Bipolar II Disorder (BD II). Research has revealed ‘commonalities rather than differences’ in the two groups of disorders (depressive disorders and bipolar and related disorders, American Psychiatric Association (APA), 2013; Barlow et al., 2004) and their treatments overlap considerably; with the same medications and similar psychological interventions used in both. Thus a unified approach to the diagnosis and management of mood disorders has been adopted in this guideline (see: Figures 1 and 2 and Table 2).
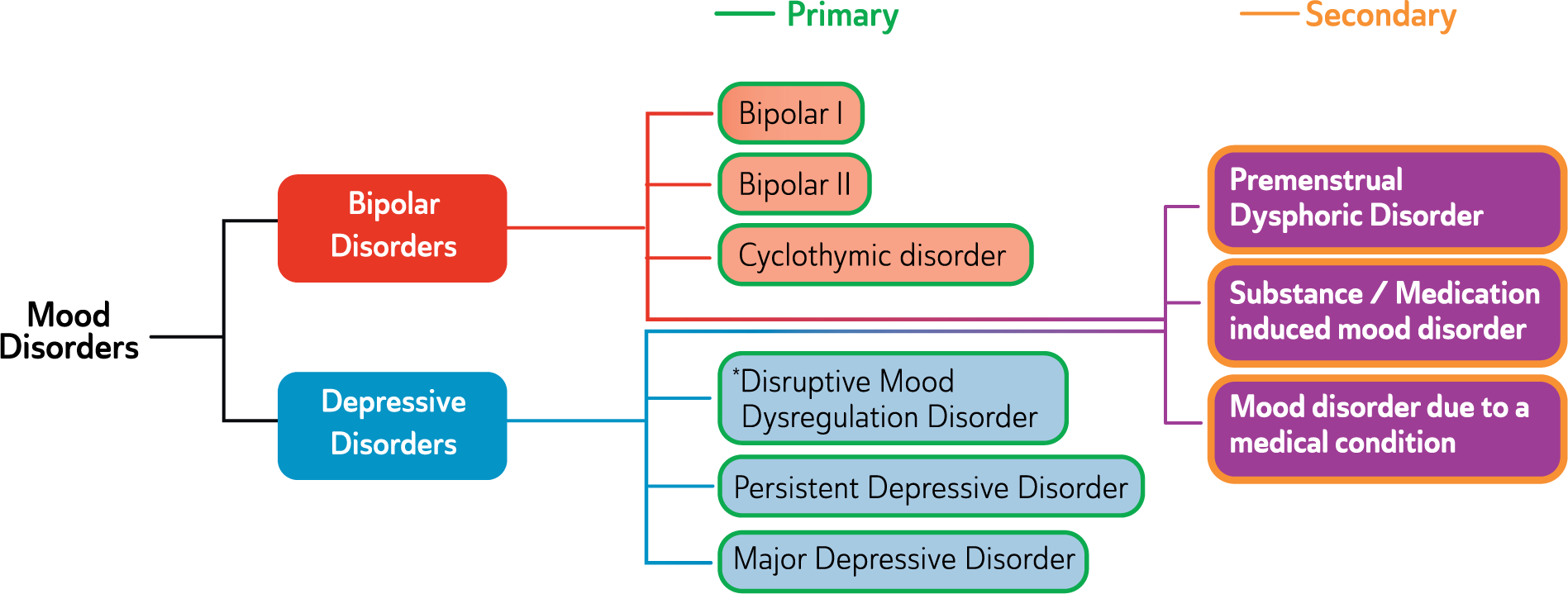
Classification of mood disorders.
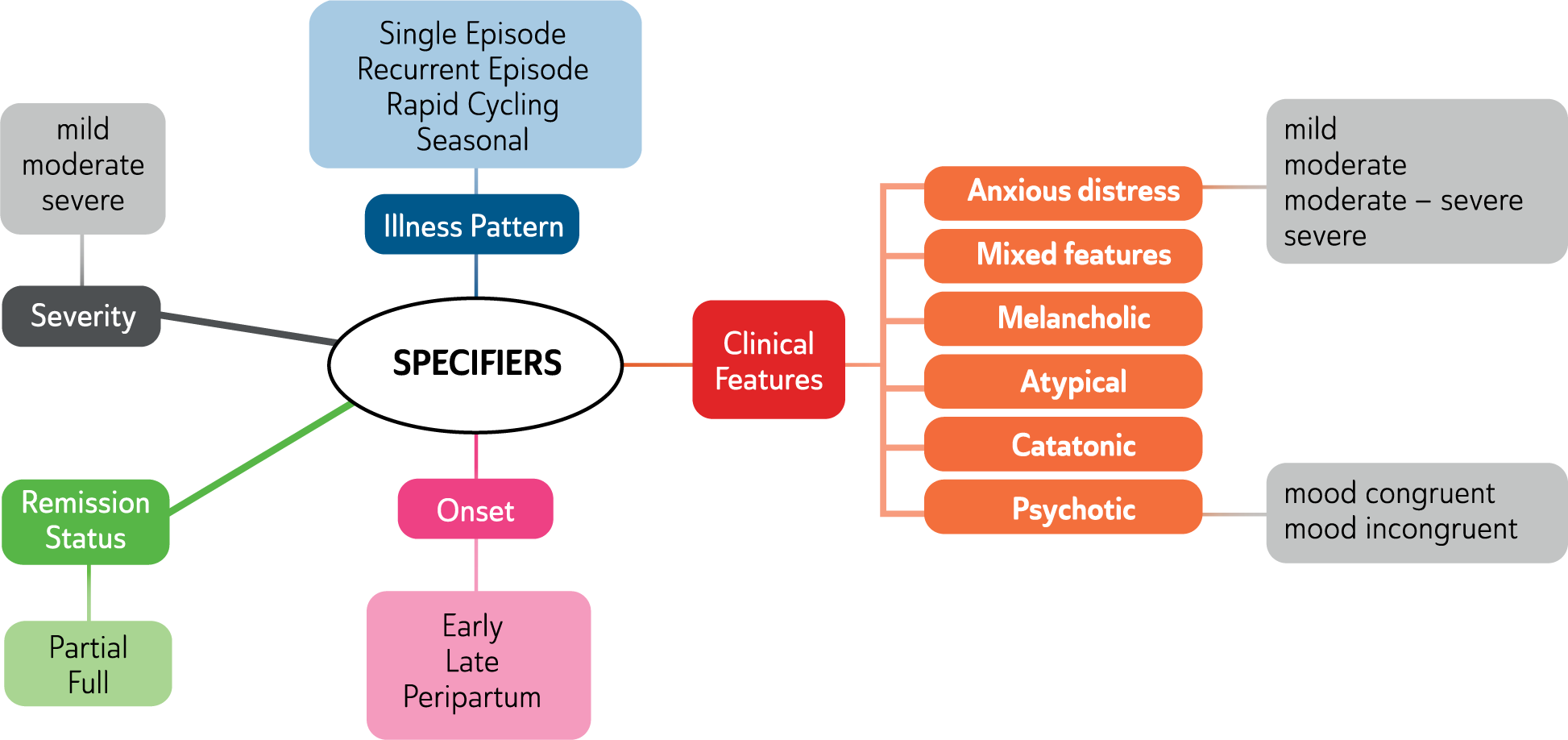
Mood disorders specifiers.
Methodology
This guideline was developed as part of the RANZCP CPG Project 2013–2015; funded solely by the College. The RANZCP called for expressions of interest from its members and appointed the Chair and the mood disorders committee (MDC) (see Appendix 5). The MDC consists of Australian and New Zealand specialists from Psychiatry and Psychology with clinical and academic expertise in the management of mood disorders.
This guideline has been developed using a number of strategies for searching and synthesizing clinical and research evidence pertaining to the diagnosis and treatment of mood disorders. Members of the MDC both individually and as part of sub-groups were charged with the task of searching the literature pertinent to particular aspects of managing mood disorders. To ensure content and form were acceptable to end-users, and to pre-empt well-recognised challenges with knowledge translation, two formal rounds of external consultation and review were undertaken with relevant experts, clinicians and consumers (see Appendix 5).
Search strategy: Articles and information sourced from search engines such as PubMed, EMBASE, MEDLINE, PsycINFO and Google Scholar was supplemented by literature known to MDC members (e.g. from books, book chapters and government reports) and from existing depression and bipolar disorder guidelines (Bauer et al., 2002a, 2002b; Cleare et al., 2015; Ellis, 2004; Goodwin, 2009; Grunze et al., 2002, 2003, 2004, 2013; Malhi et al., 2009a, 2009b; NICE, 2006, 2009; RANZCP, 2004; Yatham et al., 2013b). The search was repeated regularly between April 2013 and October 2015.
The MDC convened a teleconference in total 55 times over a 33 month period spanning March 2013–September 2015. Each meeting lasted 1 hour and involved the discussion of key aspects of the guideline and the development of clinical recommendations based on available evidence. This allowed the consideration of new evidence as it emerged and the tempering of advice if deemed necessary.
Recommendation formulation: For intervention studies, levels of evidence were assigned (see Appendix 1) and adapted from the Australian National Health and Medical Research Council (NHMRC) levels of evidence for intervention studies (NHMRC, 2009).
This guideline makes two types of recommendations that reflect the reasoning used to formulate advice. First evidence-based recommendations (EBRs) were formulated when the MDC judged there to be sufficient consistent evidence from intervention studies to support a recommendation on a given topic. For each EBR, strength of evidence was rated using the NHMRC levels of evidence for intervention studies and is graded accordingly in the recommendation box (e.g., EBR I, II, III, or IV).
It is important to remember that absence of evidence is not evidence of absence, and so, given our understanding of the nature and optimal management of many aspects of mood disorders is incomplete, a second type of recommendation was also employed. This was also derived through discussion and agreement within the MDC and termed a consensus based recommendation (CBR). CBRs were formulated when: (i) the existing intervention evidence base was absent, ambiguous, or of doubtful clinical impact in the Australian and New Zealand context; and (ii) the MDC (based on collective clinical and research knowledge and experience) reached consensus on the clinical utility of the recommendations. For example, a CBR was developed for the adjunctive psychological treatment of bipolar disorder: the recommendation that treatment should follow a published evidence-based manual has not been subject to direct test, but was inferred from the broader psychotherapy literature and the MDC’s experience in treatment and training. Similarly, the recommendation that antidepressant therapy should be avoided during mixed mood states is based on clinical experience and is a CBR. If members of the MDC held a view at variance with a CBR it was discarded and no recommendation was made. 1
The MDC produced a series of draft guidelines that were gradually refined and extensively peer reviewed.
Expert review: Australian, New Zealand and international expert advisers (clinical or academic) were invited by the Chair of the MDC to review a draft copy of the guideline and provide comment. Expert advisers are listed (see Appendix 5). Expert advisers provided comment and feedback on a voluntary basis with no remuneration and their declarations of interest are documented. The MDC assessed all comments received via expert review and produced a second draft guideline for public consultation.
Public consultation: A revised version of the guideline was released (June–July 2015) for public consultation to encourage community-wide engagement and gain broad representation across Australia and New Zealand. Specifically, the RANZCP invited review and comment from its committees and members, as well as key stakeholders, including professional bodies representing medicine, psychology, nursing, social work and occupational therapy, and special interest groups representing consumers, carers, Aboriginal and Torres Strait Islander peoples and Māori (see Appendix 5). During the consultation period, the draft guideline was publicly available for review and comment on the RANZCP website. Participants were asked to review the guideline and answer four key questions related to each section of the guideline via Survey Monkey. Question criteria were:
Are there any significant gaps (of topic, literature, other)?
Are there errors in the content?
Is the structure logical and easy to use?
Do you have any other comments?
The MDC considered all responses. For each suggestion, the MDC agreed on whether to revise the guideline. All decisions were recorded and documented. External consultation enabled the MDC to further develop, strengthen and validate recommendations to ensure all relevant issues were identified and considered.
Methodological considerations
Empirical scientific methodologies provide a rational basis to guide patient care. But they are not without their limitations. An understanding of several logistical, ethical, and methodological challenges helps to explain many of the evidence ‘blind spots’ in medical practice.
Many clinically relevant dilemmas cannot be fully elucidated empirically because of significant ethical issues. An example of this is the evidence for safety of medications in pregnancy. Prospective human comparator trials face an ethical impasse, thus the evidence base is drawn largely from animal studies and association studies in humans. Sufficient power to identify rare adverse associations unfortunately only arises after many years of post-marketing surveillance. Similarly, investigating novel therapies for severe conditions often relies on an augmentation comparator study approach, so that vulnerable patients are not unethically deprived of an established therapy. This can delay the process of demonstrating similar or superior efficacy of novel therapies as standalone treatments in severe illnesses.
Rarer clinical situations are often less well empirically studied because of their inevitably smaller public health footprint drawing less research funding, challenges of finding recruits, and the larger samples needed for rare outcome statistical power. Similar cost and recruitment challenges help to explain the relative paucity of long-term follow-up studies. Importantly, diagnostic and therapeutic areas offering potential for commercialisation are sometimes more intensively studied, with research questions shaped by intellectual property. This may result in publication bias, and relatively less study of approaches that lack commercial viability. Exclusion of common comorbidities in trials, clinical validity of rating scales and efficacy cut points, and diagnostic instability further impede evidence translation to practice. Finally, inherent clinical heterogeneity limits the utility of predictive and normative evidence-based approaches to guide practice. Hence the focus on personalized medicine has grown and clinicians are faced with having to increasingly manage clinical uncertainty.
Limitations of clinical trials
Many of the studies cited in this guideline were funded by pharmaceutical companies that manufactured the medication under investigation. Understandably, trials are designed to maximise the likelihood of identifying a favourable effect. This factor may contribute to a number of methodological weaknesses in the trial design and reported data. In addition, meta-analyses rely on all relevant trials being reported. However, this is not always the case (Ioannidis, 2009) as some studies are not reported because they are small and others because they have a negative outcome (failed trials). Data from such studies is often not made available. Furthermore the degree to which patients in clinical trials are representative of the population being treated is debatable with, for instance, many trials in depression excluding patients who are experiencing suicidal thoughts (a relatively common symptom in severe depressive disorders). Distortions in medication trial construction and interpretation may also occur when funding is being sought from third parties, such as Medicare funding in Australia. Negative outcomes from research reduce opportunities for gaining future financial support and therefore can be overlooked or misrepresented. Trials in bipolar disorder exemplify some of these problems (see Table 1). The scientific literature on psychological therapies is also influenced by publication bias (increased likelihood that positive outcome studies will be published) (Driessen et al., 2015) and researcher allegiance effects (Munder et al., 2013).
Methodological considerations in bipolar disorder trials.

Features of bipolar disorders and depressive disorders (based on DSM-5).
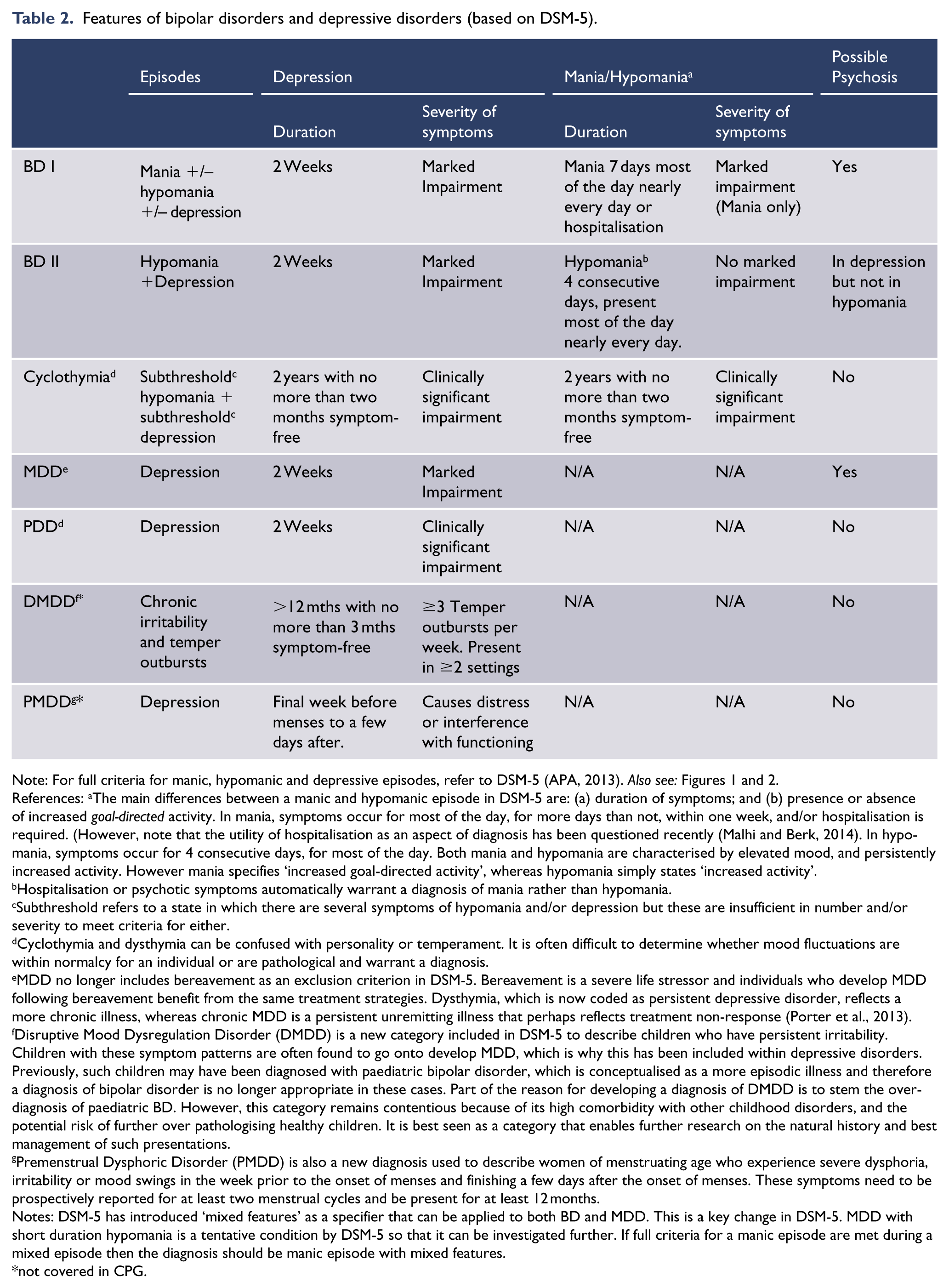
Note: For full criteria for manic, hypomanic and depressive episodes, refer to DSM-5 (APA, 2013). Also see: Figures 1 and 2.
References: aThe main differences between a manic and hypomanic episode in DSM-5 are: (a) duration of symptoms; and (b) presence or absence of increased goal-directed activity. In mania, symptoms occur for most of the day, for more days than not, within one week, and/or hospitalisation is required. (However, note that the utility of hospitalisation as an aspect of diagnosis has been questioned recently (Malhi and Berk, 2014). In hypomania, symptoms occur for 4 consecutive days, for most of the day. Both mania and hypomania are characterised by elevated mood, and persistently increased activity. However mania specifies ‘increased goal-directed activity’, whereas hypomania simply states ‘increased activity’.
Hospitalisation or psychotic symptoms automatically warrant a diagnosis of mania rather than hypomania.
Subthreshold refers to a state in which there are several symptoms of hypomania and/or depression but these are insufficient in number and/or severity to meet criteria for either.
Cyclothymia and dysthymia can be confused with personality or temperament. It is often difficult to determine whether mood fluctuations are within normalcy for an individual or are pathological and warrant a diagnosis.
MDD no longer includes bereavement as an exclusion criterion in DSM-5. Bereavement is a severe life stressor and individuals who develop MDD following bereavement benefit from the same treatment strategies. Dysthymia, which is now coded as persistent depressive disorder, reflects a more chronic illness, whereas chronic MDD is a persistent unremitting illness that perhaps reflects treatment non-response (Porter et al., 2013).
Disruptive Mood Dysregulation Disorder (DMDD) is a new category included in DSM-5 to describe children who have persistent irritability. Children with these symptom patterns are often found to go onto develop MDD, which is why this has been included within depressive disorders. Previously, such children may have been diagnosed with paediatric bipolar disorder, which is conceptualised as a more episodic illness and therefore a diagnosis of bipolar disorder is no longer appropriate in these cases. Part of the reason for developing a diagnosis of DMDD is to stem the over-diagnosis of paediatric BD. However, this category remains contentious because of its high comorbidity with other childhood disorders, and the potential risk of further over pathologising healthy children. It is best seen as a category that enables further research on the natural history and best management of such presentations.
Premenstrual Dysphoric Disorder (PMDD) is also a new diagnosis used to describe women of menstruating age who experience severe dysphoria, irritability or mood swings in the week prior to the onset of menses and finishing a few days after the onset of menses. These symptoms need to be prospectively reported for at least two menstrual cycles and be present for at least 12 months.
Notes: DSM-5 has introduced ‘mixed features’ as a specifier that can be applied to both BD and MDD. This is a key change in DSM-5. MDD with short duration hypomania is a tentative condition by DSM-5 so that it can be investigated further. If full criteria for a manic episode are met during a mixed episode then the diagnosis should be manic episode with mixed features.
not covered in CPG.
Methodological issues in clinical trials may result in ‘failed trials’ i.e. trials which do not show a difference between the effects of a drug and placebo despite one existing. This is probably a common reason for evidence not being available despite the fact that there is a general clinical impression that certain medications are effective for particular conditions.
Clinical trials of psychotherapies also have potential biases including lack of blinding and expectation effects amongst both therapists and patients. More fundamentally, psychotherapies are complex, multi-component interventions, and contrary to the logic of the randomised controlled trial (RCT), patients’ experience of and active participation in psychotherapy systematically influences the content of the intervention they receive. It has therefore been argued that RCTs are not optimal methodologies for validating psychotherapies, and instead process evaluation is required (Moore et al., 2015). Therefore while RCTs carry significant weight in this guideline, it must be remembered that they and their meta-analyses are of variable quality and validity.
Off label prescribing
In this guideline, some therapies identified as effective for the treatment of mood disorders on the basis of available evidence may have yet to receive approval for such use in Australia and/or New Zealand.
The use of such therapeutic agents outside their approved indication(s) is sometimes referred to as ‘off label’ use, and in practice this may impact eligibility for third-party payer subsidy. We recommend careful documentation supporting your clinical use of specific therapeutic agents over alternatives which are approved in your country. It is also recommended that this issue is explained to patients, including informing them that they may have to personally meet added costs due to lack of third-party payer subsidy.
Classification of mood disorders
A pragmatic approach to mood disorder classification
There is growing consensus that psychiatric diagnoses are akin to social constructs (Insel, 2014; Zachar and Kendler, 2007). It is nonetheless appropriate for the structure of this guideline to adopt an accepted mood disorder taxonomy because, (i) there is broad agreement about definitions, and (ii) diagnostic terms have accrued valuable meaning through scientific (e.g. clinical trials) and social processes (e.g. advocacy). (See: Figure 1). Using the terms as pragmatic organising constructs should not translate into their reification - the optimal classification of disorders must await a quantum leap in our understanding of the aetiology and pathophysiology of abnormal behaviour.
The origins and development of mood disorders (however described) is definitively not known, but it is certainly multi-factorial and the clinical expression is likely determined through a combination of genetic predisposition, psychological vulnerabilities and life stressors. Interplay of factors within these domains is thought to eventuate in brain/mind dysfunction. This dysfunction is sometimes observable in neural changes, and is experienced subjectively as distress that can manifest as overt behavioural change. Together these abnormalities result in clinical signs and symptoms, which are grouped into syndromes and termed mood disorders.
A variety of psychological and neurobiological models address the aetiology and pathophysiology of mood disorders (e.g. Berman et al., 2011; Caspi et al., 2003; Charney and Manji, 2004; Hasler et al., 2004; Nolen-Hoeksema, 2000). These ‘explanations’ offer important insights as to the origin of mood symptoms and provide clinicians a framework for understanding the complex pathology that underpins clinical mood disorders.
Mood disorders diagnostic criteria
Criteria for depressive disorders
Depressive disorders usually feature low mood and/or a loss of pleasure (anhedonia). These are often accompanied by somatic symptoms such as changes to weight, psychomotor disturbance, fatigue and sleep disturbances such as insomnia or hypersomnia, cognitive symptoms, such as diminished concentration and negative cognitions characterised by negative self-evaluation (low self-esteem), excessive guilt, feelings of hopelessness and helplessness, and suicidal ideation or ambivalence about living. Thus functionally, individuals with mood disorders are usually limited in their ability to perform their normal day-to-day duties (APA, 2013). (See: Table 3 Summary of DSM-5 criteria for depressive disorders.)
Summary of DSM-5 criteria for depressive disorders.
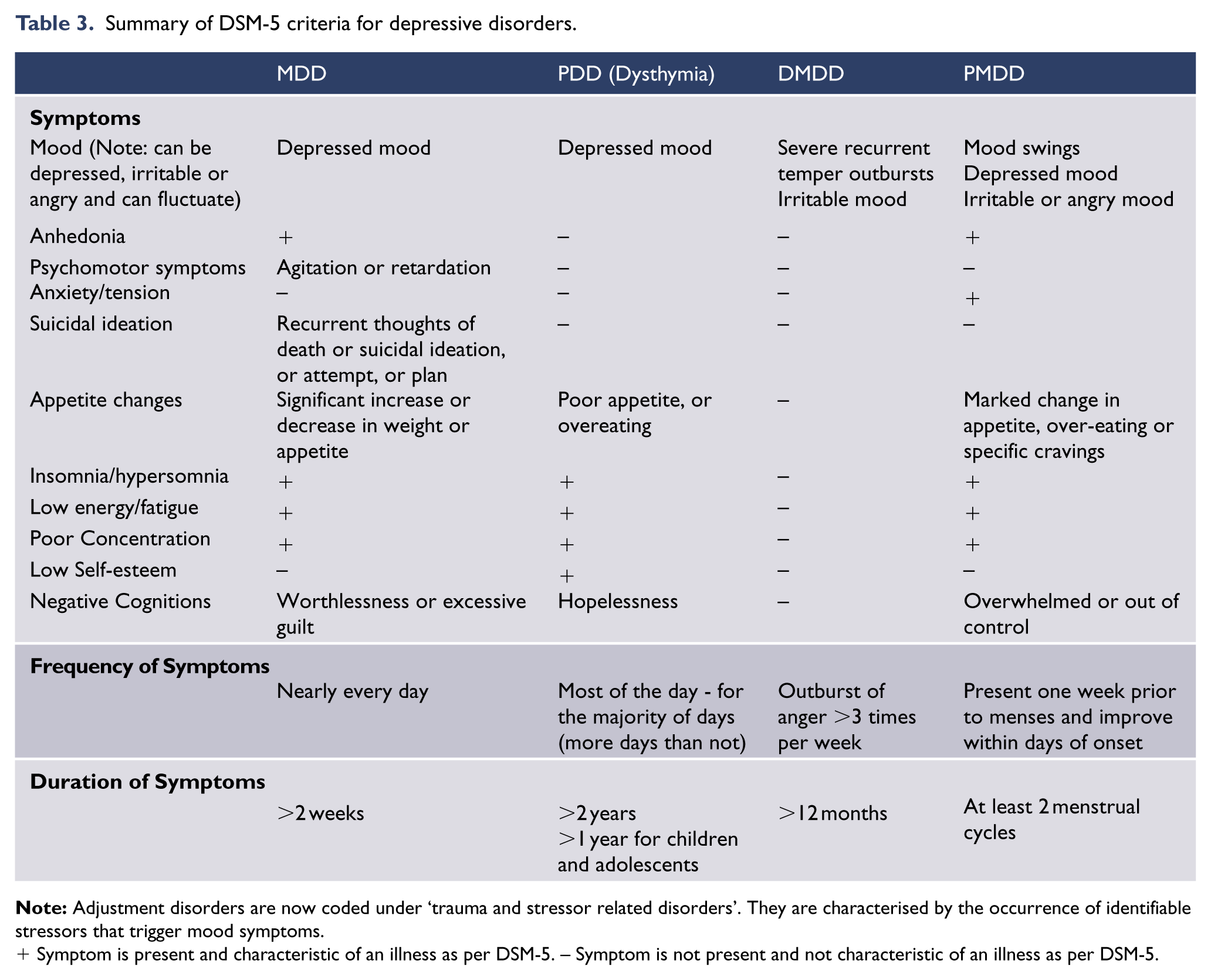
+ Symptom is present and characteristic of an illness as per DSM-5. – Symptom is not present and not characteristic of an illness as per DSM-5.
Depressive disorders comprise several diagnoses distinguished by the precipitants and/or the frequency, intensity and duration of episodes. It is important to note that in practice the term ‘major depression’ is used to refer both to an episode of illness and the illness as a whole. Technically episodes of depression are discrete single events, which also occur in the context of bipolar disorder. (See: Criteria for bipolar disorders.)
Major depressive disorder (MDD) is diagnosed in adults and is the archetypal diagnosis in this group of disorders. Persistent depressive disorder (PDD) is a more chronic presentation of depression and combines the DSM-IV diagnoses of dysthymia and chronic MDD. Disruptive mood dysregulation disorder (DMDD) is a new category in DSM-5 which is assigned to children with persistent irritability and frequent outbursts of anger. 2 Premenstrual dysphoric disorder (PMDD) is now a formal diagnosis and describes clinically problematic low mood in women that commences following ovulation and lasts until the beginning of the next menstrual cycle. Substance/medication-induced depressive disorder and Depressive disorder due to another medical condition have been added to DSM-5 (APA, 2013).
This guideline focuses on MDD and PDD because these are the depressive disorders most commonly encountered in clinical practice (see: Table 4). Consequently they also have the greatest abundance of empirical research data to help guide optimal care.
Depressive disorders (MDD and PDD) facts and figures.

These figures relate primarily to community samples.
Criteria for bipolar disorders
Presence or history of mania/hypomania is the defining element of bipolar disorders and distinguishes them from depressive disorders. An individual is diagnosed with BD I if he/she has experienced a full manic episode. One manic episode is sufficient to qualify for the diagnosis, but most individuals will also have experienced one or more major depressive episodes, which often precede the onset of mania. BD II is diagnosed if an individual has experienced both an episode of major depression and hypomania in their lifetime, but has never had a manic episode. The phases and stages of bipolar disorder are associated with varying degrees of functional impairment (see: Table 8).
In DSM-5 the core features in the diagnosis of mania have been extended to include coterminous changes in activity or energy alongside elevated, expansive or irritable mood to facilitate earlier and more accurate diagnosis in clinical settings (APA, 2013).
In individuals with bipolar disorder, the initial presentation is usually that of depression and therefore the correct diagnosis cannot be made until an episode of hypomania/mania occurs. This inevitable delay that can sometimes last up to a decade results in a period of inadvertent suboptimal management. Diagnosis is also hindered by the fact that bipolar disorder has very high rates of comorbidity with anxiety, substance misuse and personality disorders (particularly borderline personality disorder) (Malhi et al., 2012a). Once established bipolar disorder is a recurrent mood disorder that runs a lifelong course and, between episodes of illness, patients often continue to experience subthreshold symptoms, which significantly compromise their quality of life (Judd et al., 2002). Consequently, patients with bipolar disorder have high rates of self-harm and suicide (Leverich et al., 2003; Schaffer et al., 2015). The core ‘facts and figures’ of bipolar disorders are summarised in Tables 5 and 6.
Bipolar I disorder facts and figures.

Bipolar II disorder facts and figures.

Distinguishing major depression and bipolar disorder
It is important to note that current classification systems have assigned greater importance to polarity (unipolar/bipolar) than recurrence and that recurrent mood disorders feature strongly in both unipolar and bipolar mood disorders. Although not recognised in current classifications, many authorities argue that highly recurrent unipolar depression and bipolar disorder may be related conditions and that the traditional term manic-depressive illness which subsumes both is more apt.
Individuals who only ever experience depression are described as having major depressive disorder (a unipolar illness), whereas those who in addition experience (hypo)mania are diagnosed with bipolar disorder (Malhi et al., 2010). Therefore, in the absence of (hypo)mania, it is difficult to distinguish bipolar and unipolar depression. Clinically this is a common problem because of the natural history of bipolar disorder, which usually begins with depression. Indeed in the majority of cases, individuals with bipolar disorder experience several episodes of depression prior to developing (hypo)mania and for most people with bipolar disorder the predominance of depressive episodes continues throughout the course of the illness. Furthermore, even between episodes of illness, individuals with mood disorders often continue to suffer subthreshold depressive symptoms, which can compromise their quality of life, limit functioning and increase the risk of relapse (Grunze et al., 2009).
There are no definitive clinical criteria that distinguish bipolar and unipolar depression. Some depressive features and symptoms may be more likely in one presentation compared to the other, and together these may provide an ‘index of suspicion’, particularly when used in conjunction with other risk factors, such as family history (Moreno et al., 2012). It is of note that these are not diagnostic. Actuarial information should also be considered when drawing inferences about a bipolar or unipolar diagnosis from a depressive presentation (Youngstrom et al., 2005) because lifetime prevalence of unipolar depression is 10–15 times higher than bipolar disorder. If bipolar disorder is suspected on the basis of clinical features (see: Table 7) a thorough clinical assessment for (hypo)manic symptoms is essential.
Features that may distinguish bipolar and unipolar depression.
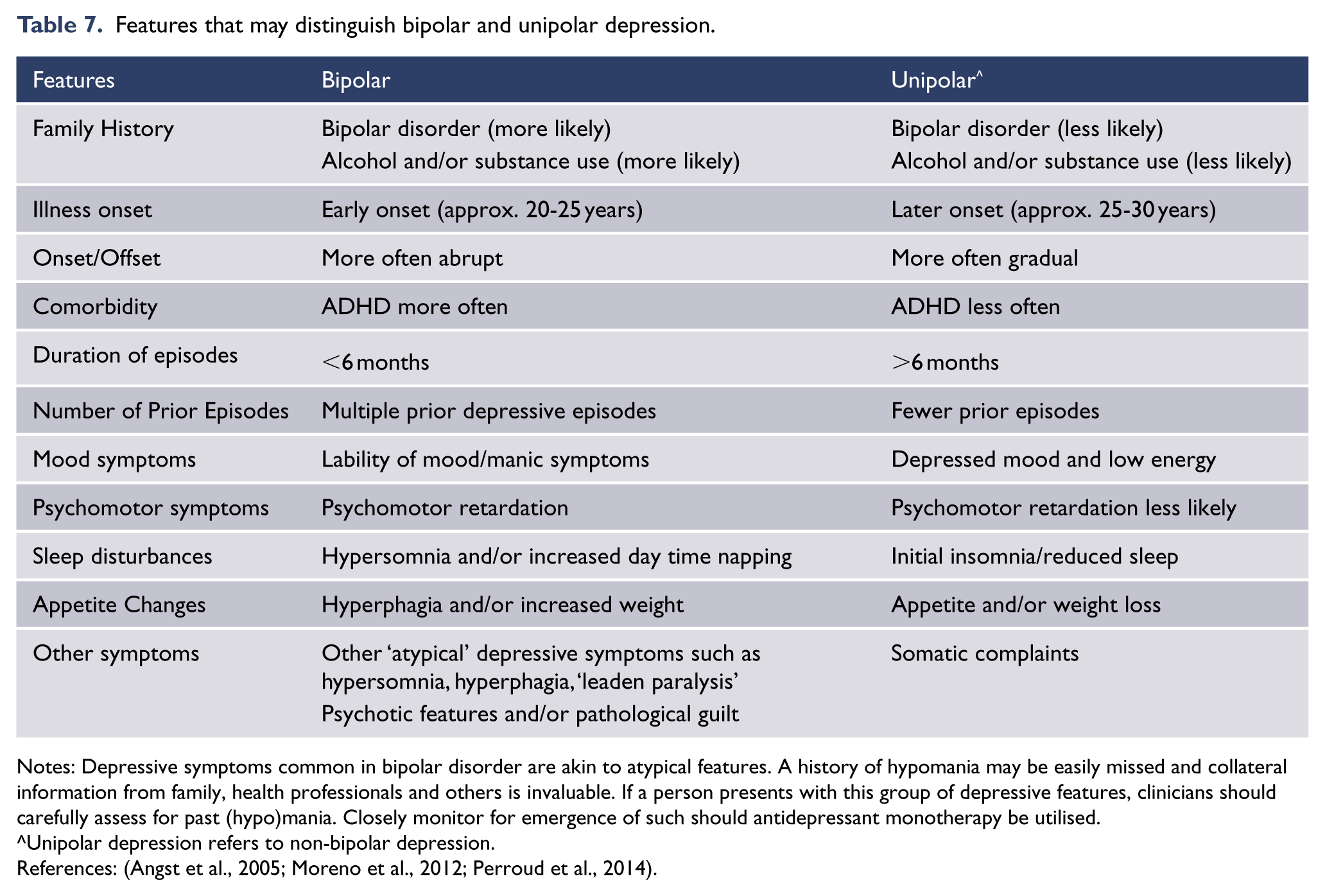
Notes: Depressive symptoms common in bipolar disorder are akin to atypical features. A history of hypomania may be easily missed and collateral information from family, health professionals and others is invaluable. If a person presents with this group of depressive features, clinicians should carefully assess for past (hypo)mania. Closely monitor for emergence of such should antidepressant monotherapy be utilised.
Unipolar depression refers to non-bipolar depression.
References: (Angst et al., 2005; Moreno et al., 2012; Perroud et al., 2014).
Summary of DSM-5 criteria for bipolar disorders.
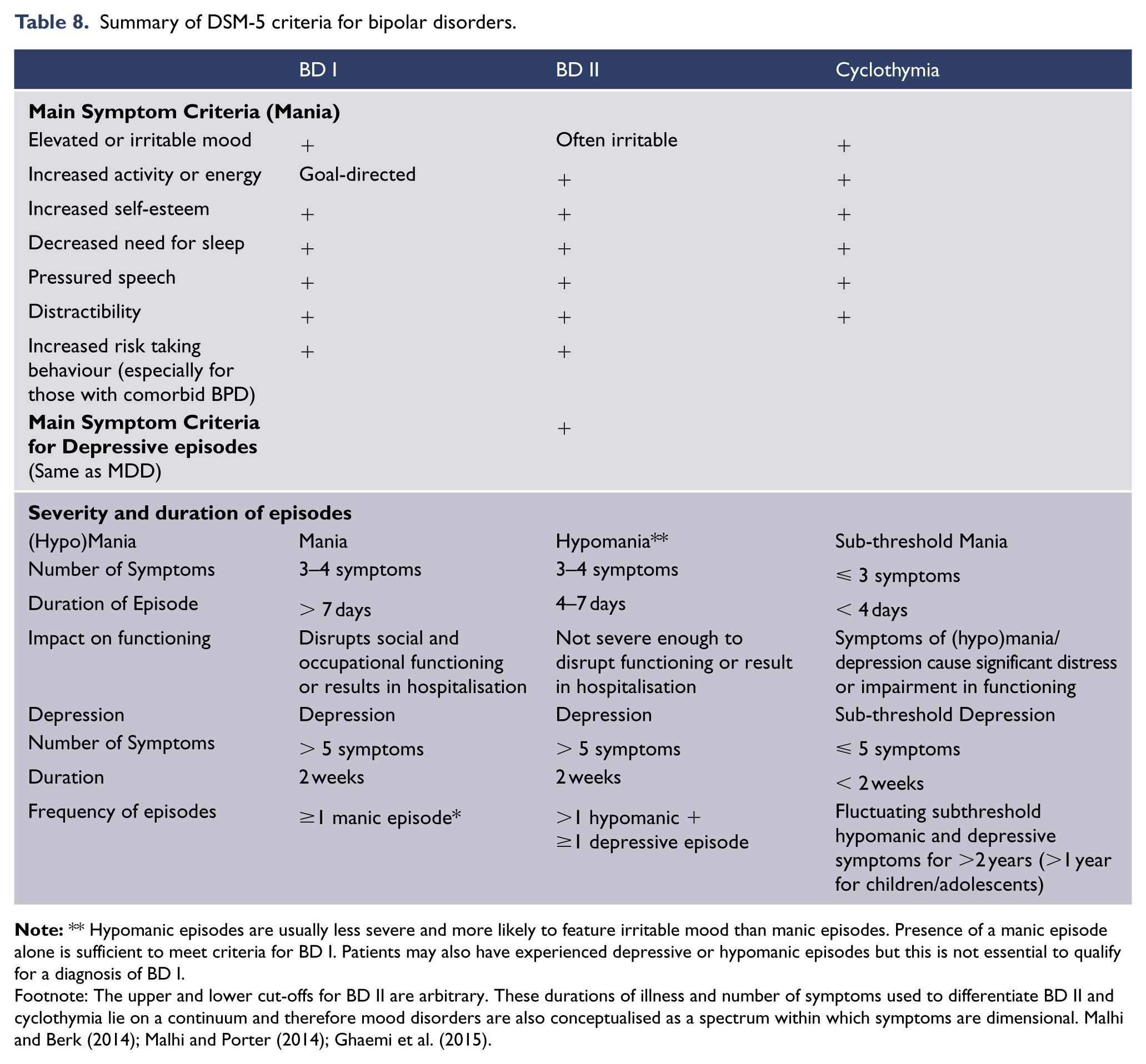
Footnote: The upper and lower cut-offs for BD II are arbitrary. These durations of illness and number of symptoms used to differentiate BD II and cyclothymia lie on a continuum and therefore mood disorders are also conceptualised as a spectrum within which symptoms are dimensional. Malhi and Berk (2014); Malhi and Porter (2014); Ghaemi et al. (2015).
Cognitive impairment in mood disorders
The issue of cognitive impairment in mood disorders has been recognised for many years, particularly in the elderly in whom the phenomenon has been described as pseudodementia. However, while it has been clear for some time now that episodes of mood disturbance are associated with significant cognitive impairment, two issues are becoming increasingly clear.
Firstly, it is clear that in recurrent mood disorders some degree of cognitive impairment remains between episodes. This is the case in both unipolar depression (Bora et al., 2013) and bipolar disorder (Baune and Malhi, 2015; Bourne et al., 2013).
Secondly, this impairment is clinically significant in terms of overall functioning, once again in both unipolar and bipolar disorders (Malhi et al., 2007; Porter et al., 2015; Withall et al., 2009).
Therefore, this is an issue which is receiving increasing attention with research into both pharmacological (Carvalho et al., 2014b) and psychological strategies (Porter et al., 2014) to improve cognition. However, currently there is no clear evidence regarding the effectiveness of specific treatments in remediating cognitive dysfunction in mood disorders but it is an important issue for clinicians to assess.
Differential diagnoses of mood disorders
Mood disorders can co-occur with all psychiatric disorders. Consequently, there are many possible differential diagnoses and so only those that are common, or particularly important because of impact on treatment pathways, have been considered.
The principal and most probable differential diagnoses for mood disorders are other depressive and bipolar disorders (see: Table 2). Delineating bipolar and unipolar mood disorders has very important implications for optimal management, prognosis, and avoiding iatrogenic worsening of affective instability. As bipolar disorders can appear to be ‘unipolar’ early in their course it is important to bear in mind a differential diagnosis of bipolar disorders in first episode major depression (see: Table 3). Therefore, as part of a diagnostic assessment of depression, the clinician should actively probe for a history of elated, excited, or irritable mood of any duration along with increased levels of energy, and ask about a family history of mania. Adjustment disorder with depressed mood should also be carefully considered as a differential diagnosis, particularly when the onset of the depressive symptoms is closely related to a major life stressor. 3 Note however, sometimes life stressors can trigger or exacerbate a major depressive episode. Similarly, it is important to consider the overlays of grief with depressive disorders noting that in some instances grief reactions may trigger adjustment disorder reactions and depressive disorders, or exacerbate or perpetuate a depressive episode. Therefore when making a diagnosis of depression the intensity, duration, functional impact, sociocultural and personality congruency of a grief reaction are important considerations.
Anxiety disorders commonly occur contemporaneously with mood disorders and often precede their onset (Malhi et al., 2002). Indeed, marked overlap between these ‘distress disorders’ is a primary driver of emerging dimensional approaches to nosology (Insel, 2014) and trans-diagnostic approaches to treatment (Buckholtz and Meyer-Lindenberg, 2012). Substance misuse, personality dysfunction and general medical disorders may coexist alongside mood disorders, and appear at any point along the course of illness (McIntyre et al., 2012; Malhi et al., 2012a). Mood disorders may also merge into psychosis, normally at the extremes of severity for both poles.
Several medical illnesses can result in specific mood disorder symptoms or indeed lead to the development of mood disorders, and these need to be borne in mind. Sleep apnoea and hypothyroidism should be considered as differential diagnoses, particularly if other features on history or mental state examination are suggestive of mood disorders. For some patients, an interplay between general medical conditions and mood disorders hampers treatment and worsens prognosis of both conditions. Among older patients presenting with first episode mood disorders, it is necessary to carefully consider organic differential diagnoses, given that major neurocognitive disorders, such as frontotemporal dementia, often present initially with affective symptoms.
Another important differential diagnosis that warrants consideration is that of personality disorders. For further details of comorbid personality disorders and other common comorbidities and how they influence management, please refer to Management of mood disorders in the context of comorbidities.
Models and formulation for assessment of mood disorders
Clinical assessment and diagnosis: setting the stage for management
Prior to formulation, it is useful to construct a template that captures the patients’ problems using the biopsychosocial and lifestyle model (BPSL Model) and considers predisposing, precipitating and perpetuating factors. Clinically, it is judicious to initially make a working diagnosis rather than prematurely foreclose on a definite diagnosis. This approach enables the commencement of initial management but at the same time limits the likelihood of assigning an incorrect diagnosis. Making a diagnosis is a sophisticated process. It involves detailed information gathering, a comprehensive clinical assessment, including a detailed mental state examination and the careful piecing together of corroborative information. Even with all of these components at hand, it is sometimes difficult to make a definitive diagnosis of a mood disorder.
As noted above, mood disorder diagnoses are critical clinical tools because of their communication value, and because much of what we know about the treatment for mood disorders is based on trials in patient samples derived using these diagnoses. However, the clinician needs to recognise their limitations and note that diagnoses form one part of an individualised, contextualised characterisation for each patient. The appraisal of context is pivotal because this provides the understanding as to why a person has a mood disorder at this particular point in their life, and is best constructed via thorough clinical assessment. To make sense of this information, and to translate it into an effective treatment pathway, it is necessary to have a meaningful framework. This process is called case formulation and there is strong consensus that this is the best strategy.
The biopsychosocial and lifestyle model (BPSL)
This model (see Figure 3) is useful for understanding potential causes of depression and bipolar disorder and for planning management. Empirically it is useful because the factors that precipitate or maintain depression and lead to its expression usually fall into one of these domains. Hence it has been widely employed in psychiatry, and recently it has been expanded to include lifestyle factors such as substance misuse, diet and smoking; all of which, along with resilience factors, have been shown to impact the development of depression and bipolar disorder.
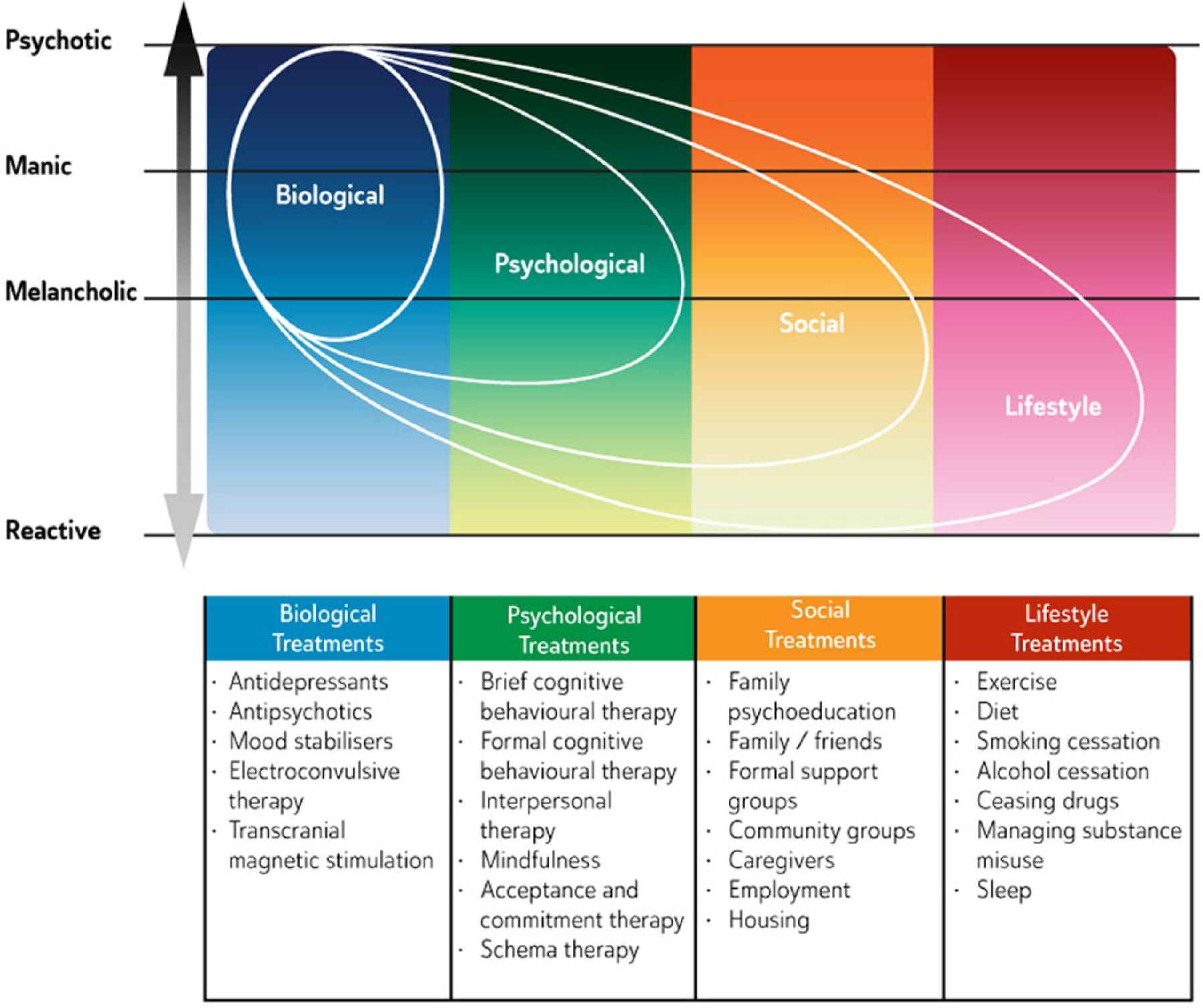
Biopsychosocial & Lifestyle Model (BPSL).
The components of the model represent seemingly distinct domains, however recent research has shown that there are important iterative links between various components and these cumulatively contribute to the onset and maintenance of mood disorders. For example, genes are typically considered to be biological factors, whereas most stressors are regarded as environmental; but recently it has been discovered that genes impact the environment and that reciprocally the environment can modify genes via epigenetic mechanisms (Niculescu, 2013). Thus, interactions between the two impact aetiology and outcome. Hence, depressive subtypes can be broadly mapped onto the BPSL model and treatments can be categorised according to its components: refer to Individualised management of mood disorders: case formulation.
Individualised management of mood disorders
The BPSL model provides the necessary framework for the development of a mood disorder formulation in an individual. Formulation builds on diagnosis, which has reliability but lacks the validity of formulation because the latter contextualises the problems of the individual and provides a richer understanding as to why he or she is unwell now. Formulation is also necessary because both treatments and diagnoses have been derived from studies of groups whereas management of mood disorders is an individual (personalised) endeavour; therefore it is important to understand the person in the context of their unique circumstances. This is the cornerstone of a successful therapeutic relationship.
Case formulation: incorporates an understanding of vulnerabilities and familial predisposition to mood disorders. It draws connections between past experiences and how these relate to the clinical presentation now. It allows mutual enrichment of the context and diagnostic framework. Traditionally the steps involved in formulation involve examining the presenting problem and predisposing, precipitating and perpetuating factors alongside protective factors. This has been termed the 5P model (see: Figure 4), but these are only a guide and may not be relevant to every person. In most instances there are one or two contributing factors from each component (Rainforth and Laurenson, 2014).
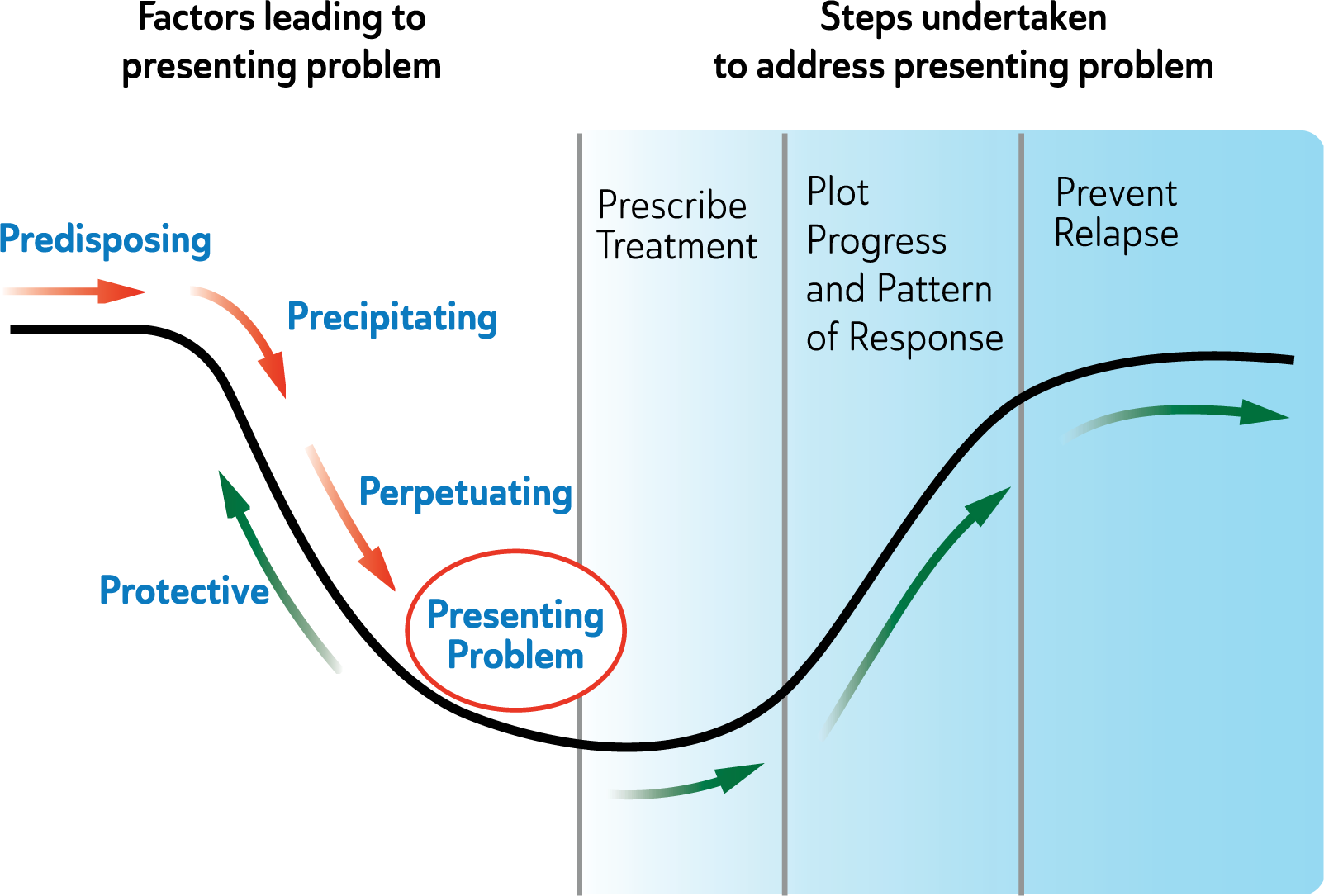
5P+ Model.
Understanding and piecing together the various factors provides a more comprehensive picture of why the individual has developed the mood disorder in the first place, and what has led to their presentation. It also acts as a forerunner to considering which interventions are most likely to be of benefit. In addition to understanding the individual, their strengths, vulnerabilities and their predicament, it is important to bear in mind that there may be alternative explanations and that different psychiatric diagnoses and general medical causes may ultimately be responsible. In this regard it is important for example in women to enquire about mood change in relation to their menstrual cycle, use of oral contraception and hormone replacement therapy (Craig, 2013; Rasgon et al., 2005; Steiner et al., 2003). Psychiatric assessment should involve a medical examination and investigations as indicated (see: Table 9). These tests need to be conducted on a case-by-case basis and tailored to individual needs. In most instances the general practitioner may be able to perform these investigations.
Physical examination and investigation of patients presenting with mood disorders.
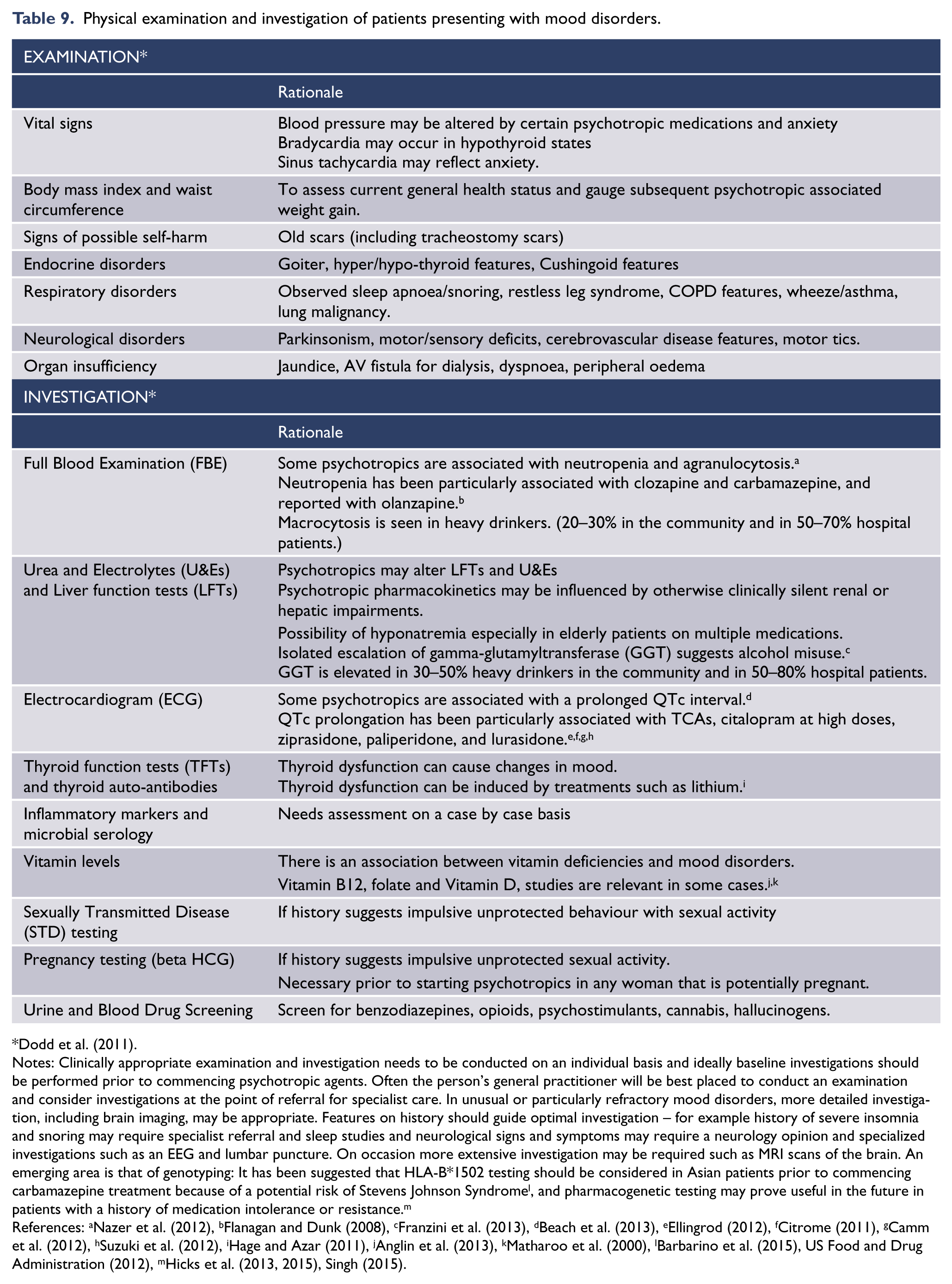
Notes: Clinically appropriate examination and investigation needs to be conducted on an individual basis and ideally baseline investigations should be performed prior to commencing psychotropic agents. Often the person’s general practitioner will be best placed to conduct an examination and consider investigations at the point of referral for specialist care. In unusual or particularly refractory mood disorders, more detailed investigation, including brain imaging, may be appropriate. Features on history should guide optimal investigation – for example history of severe insomnia and snoring may require specialist referral and sleep studies and neurological signs and symptoms may require a neurology opinion and specialized investigations such as an EEG and lumbar puncture. On occasion more extensive investigation may be required such as MRI scans of the brain. An emerging area is that of genotyping: It has been suggested that HLA-B*1502 testing should be considered in Asian patients prior to commencing carbamazepine treatment because of a potential risk of Stevens Johnson Syndrome l , and pharmacogenetic testing may prove useful in the future in patients with a history of medication intolerance or resistance. m
References: aNazer et al. (2012), bFlanagan and Dunk (2008), cFranzini et al. (2013), dBeach et al. (2013), eEllingrod (2012), fCitrome (2011), gCamm et al. (2012), hSuzuki et al. (2012), iHage and Azar (2011), jAnglin et al. (2013), kMatharoo et al. (2000), lBarbarino et al. (2015), US Food and Drug Administration (2012), mHicks et al. (2013, 2015), Singh (2015).
Resilience
An important difference between diagnostic and case formulation descriptions is that the latter recognises the resilience and strengths of a patient (Macneil et al., 2012). Resilience refers to the ability to adapt to and recover from stress (Southwick and Charney, 2012) and is not simply the absence of vulnerability: There is evidence that resilience plays an independent (mitigating) role across the three stages of mood disorder (Davydov et al., 2010; Friedman et al., 2014; Oldehinkel et al., 2014; Rutten et al., 2013).
Resilience mechanisms can also be the target of clinical work, particularly in the maintenance phase of mood disorder treatment (Malhi et al., 2013b) (see green Protective arrows in Figure 4). For instance, resilience to mood disorders can be strengthened biologically e.g., using lithium as a neuroprotective agent (Soeiro-de-Souza et al., 2012), psychologically e.g., teaching cognitive reappraisal skills (Troy et al., 2010), socially e.g., improving social support (Pietrzak et al., 2009), and through lifestyle change e.g., building exercise habits (Hare et al., 2014).
Improved resilience is a transdiagnostic treatment goal in recovery-oriented mental health services (AHMAC, Commonwealth of Australia, 2013; Australian Government, 2009; Government, 2011; Services UDoHaH, 2003; World Health Organization VHPF, University of Melbourne, 2004). Driven largely by consumers, the recovery movement highlights the importance of ‘hope’ and ‘empowerment’ in mental health service delivery, and a clinical focus on resilience supports these subjective outcomes.
General principles for managing mood disorders
Aims of treatment
The general aims of treatment of any mood disorder are to relieve symptoms, reduce the morbidity associated with the mood disorder and limit the disability and self-harm risk or potential risk of fatality. The end goal is achieving recovery to premorbid level of functioning with improved health awareness and quality of life. 4 It is important to remember the management of mood disorders is mediated entirely through the person experiencing the symptoms and as such improved subjective quality of life, personal recovery, empowerment, good therapeutic relationship – based on respect, support, warmth and empathy – are all critical to optimal outcomes.
There are broadly three
Response to treatment is the initial aim and this usually occurs within the acute phase of treatment. A clinical response is defined as a significant reduction in signs/symptoms and is normally quantified as a 50% reduction in the total score on a standardised rating scale, such as the Hamilton Depression Rating Scale (HAM-D) (Hamilton, 1960).
In the continuation phase of treatment, amelioration of symptoms continues, and the goal of treatment shifts to remission – formally defined by a cut-off score of less than 5 on the HAM-D (Hamilton, 1960), or equivalent on the Montgomery-Asberg Depression Rating Scale (MADRS) (Berk et al., 2008b; Montgomery and Asberg, 1979). In remission there are only minimal residual signs/symptoms but the individual is not yet fully functional, and without ongoing treatment remains vulnerable to relapse (Nierenberg and Wright, 1998).
Following remission, treatment continues (continuation phase) until the patient regains complete functioning and returns to their premorbid state (recovery). At this stage, ongoing treatment may still be necessary (maintenance phase) to prevent recurrence and enhance resilience but the current episode has been treated.
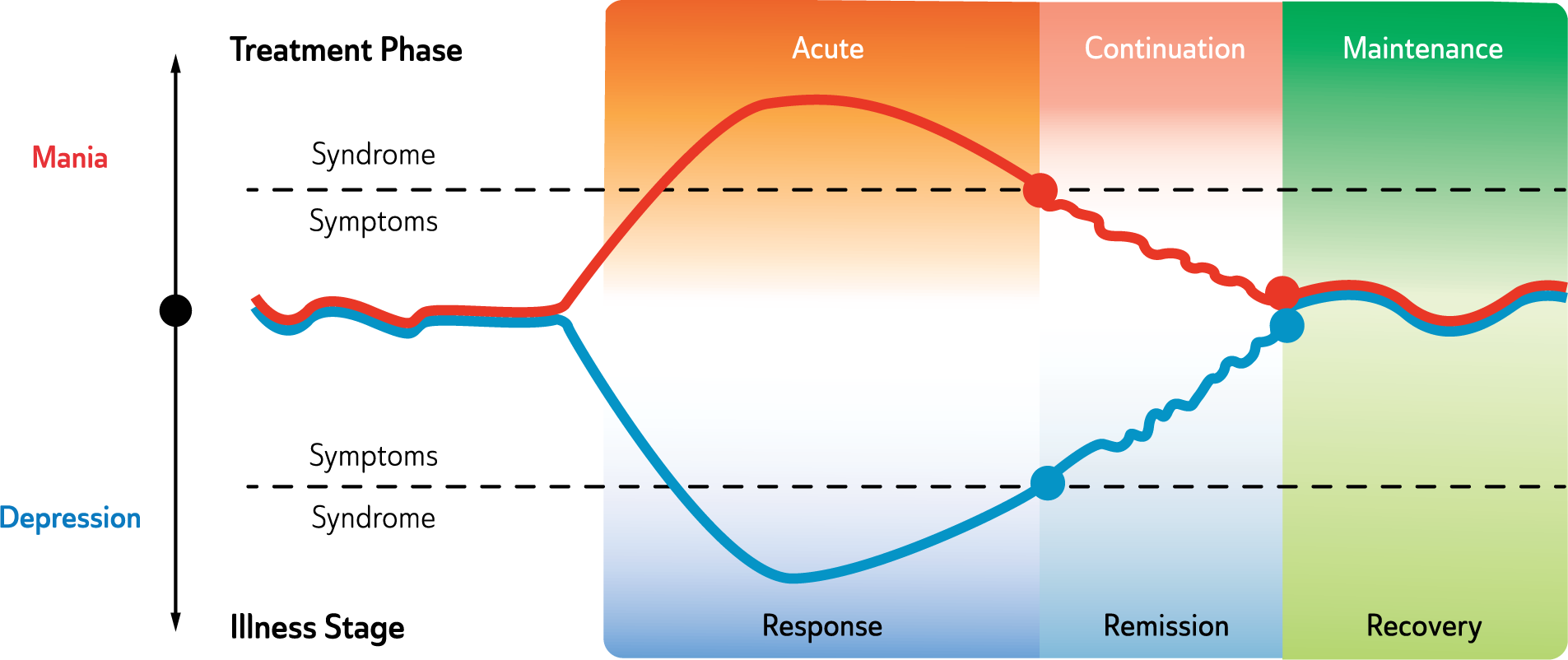
Mood disorders stages of illness and phases of treatment.
Context of treatment
The person with the disorder
There is growing recognition that mood disorders are in many cases chronic illnesses with a waxing and waning course. They are therefore best managed using a chronic illness model, which elevates the person’s active engagement in the management of their condition. Unlike acute illnesses, where the clinician’s expert tools are the primary lever for change, chronic illness management centres on the person with the disorder. Consequently, while current classification systems and the treatment science that depends on them emphasise objective features of the case, clinicians must be equally attentive to the patient’s subjective experience of the disorder and the management they are receiving (Berk et al., 2004). The patient’s perception of the quality of the therapeutic alliance is a strong predictor of adherence, leading some authorities to argue for collaborative treatment plans involving shared decision making (Berk et al., 2010).
Indeed, some guidelines argue that evidence-based practice in mood disorders should integrate patient preferences with clinical expertise and empirical information (APA, 2005). This patient-centric perspective is at the core of the recovery paradigm which dominates mental health service delivery in the Anglosphere. It also underpins efforts to provide patients with thorough psychoeducation, and aligns with calls to elevate subjective quality of life as a clinical outcome.
From multiple perspectives, therefore, a critical treatment context in the treatment of mood disorders is the person with the disorder. A key clinical challenge is to optimally integrate the clinician’s ‘technical’ expertise with the patient’s ‘lived experience’ expertise of the disorder.
Integrated care team
The optimal management of people with moderate to severe mood disorders requires not only the involvement of several health care professionals including a general practitioner (GP), mental health nurse, psychiatrist and psychological counsellors but the active partnership of family, carers and support groups as part of an integrated care team. The severity, diagnostic complexity, and treatment resistance of the presentation will influence which practitioners and how many will need to be involved for optimal care. Usually the point of entry for care is a GP or psychologist. Often patients will have an existing GP and may be more comfortable raising emotional issues with them when faced with a problem and not being able to cope. One of the key roles of the GP is to determine the severity and complexity of emotional symptoms and to make an accurate diagnosis and decide if referral to a psychiatrist or psychologist is required. For mild depressive symptoms and in the absence of suicidality or diagnostic uncertainty, management by the GP is usually sufficient with or without added psychiatric input. However, where there is diagnostic complexity, a high risk of self-harm, non-response or severe debility, referral for psychiatric assessment is indicated. For chronic severe mood disorders, a mental health case manager (usually a nurse experienced in providing mental health care) may be necessary and can be assigned by a local community mental health team or mental health team in the private sector. Health care professionals span both inpatient and outpatient services and, in conjunction with family and friends, provide crucial support throughout the course of the illness. Specialised care is of greater benefit (Kessing et al., 2013) than standard community care and assessment in specialised units is useful when managing complex and severe mood disorders (Vieta, 2013).
All providers involved in the clinical care of an individual need to be informed of the diagnosis and on-going plan for management. Therefore relevant clinical information (such as changes to treatment and hospital admission) needs to be shared regularly. Where there is lack of consensus on management, input from a psychiatrist is needed, and a second psychiatric opinion may on occasion be necessary. Clinical responsibility ultimately lies with the psychiatrist, but clinical outcomes are optimal when the various professionals involved work as a collaborative team. A successful outcome requires regular communication concerning diagnosis, formulation, treatment planning and details of current treatment, including both psychological therapy and pharmacotherapy.
Therefore, it is advisable that one of the health providers (usually the GP) acts as the primary clinical coordinator who then maintains open and regular communication between all parties to ensure that everyone is able to work together as a cohesive team. This will prevent the miscommunication of advice and provide a consistent message regarding important aspects of management such as treatment adherence. Indeed, patients often report that those involved in their care sometimes express differing views and opinions with respect to diagnosis and management. In the minds of patients and their families this can create an impression that the health care professionals are uncertain about the best course of action and this may add to their ongoing stress.
Particularly important is the relationship between providers of medication and psychological management. Patients rarely have a model of their condition that comfortably reconciles psychological and biological paradigms, and it is critical that the preferred biopsychosocial formulation is reinforced by all professionals.
Integrating online resources
There is great interest in the use of the internet as a tool to aid delivery of psychological interventions, especially CBT. Internet-based interventions have the potential to overcome many barriers to accessing evidence-based psychological interventions for mood disorders, including cost, time, and trust in professionals (Leitan et al., 2015). Internet-based interventions for depressive symptoms have demonstrated efficacy, and could be considered as an initial intervention for mild depression in the stepped management of mood disorders in primary care (RACGP, 2015). Many models of online delivery have been explored (Batterham et al., 2015), ranging from simple information, through self-help strategies, to supported time-limited structured therapies. Research is ongoing into online modalities as part of stepped care, and in hybrid formats (with psychoeducation content covered online, and more individualised therapy conducted face-to-face). Most research to date has excluded complex and/or severe presentations, and those at elevated risk of suicide or self-harm. There is clinical consensus that, in such patients, risks currently outweigh benefits for stand-alone online interventions.
For mild/moderate unipolar depression, seven RCTs provide strong effectiveness evidence for MoodGYM (
Hospitalisation and indications for treatment away from home
In most cases, mood disorders can be managed on an outpatient basis, but it is not uncommon for patients to require inpatient treatment under the care of a psychiatrist. Notably, approximately one-half of all psychiatric hospital admissions are due to a primary mood disorder (Banta et al., 2010; Brown, 2001; Hudson, 2004) and involve inpatient management in either the private or public sector. 5
Hospitalisation, particularly when involuntary, is often stressful and sometimes traumatic. The benefits of constraint, intensive monitoring and opportunity to trial new treatment regimens in the inpatient setting are often unambiguous. Nonetheless, the personal meaning of hospitalisation should be carefully addressed by the treatment team prior to and subsequent to the event to ensure that patient autonomy is maximised. In many jurisdictions, advance care directives are an important tool in this context.
Whether a patient with a mood disorder is hospitalised or not is a matter of clinical judgment (see: Table 10), and is most often considered when patients have suicidal intent or develop mania. A structured risk assessment may be helpful in identifying suicide risks, such as suicidal ideation and plans (means and opportunity to enact suicide), past history of self-harm, psychotic symptoms, impulsivity, irritability/agitation (Goossens et al., 2010; Perich et al., 2012), substance misuse and the extent of social supports.
Recommended indications for psychiatric admission.

In such instances, admission to a medical setting may be more appropriate.
Note: Decisions about admission involve weighing a number of the above clinical and psychosocial factors, together with the perspectives of carers and others involved in the community treatment setting.
Physical risk from malnourishment and dehydration, and comorbidities, such as severe medical ill health or substance misuse, may also warrant inpatient treatment. However, in cases of severe alcohol or other substance use disorders or dependence, a definitive psychiatric diagnosis should be withheld until symptoms of intoxication, detoxification, or of withdrawal, have completely abated.
Beyond risk containment, inpatient care allows detailed observation and assessment to clarify diagnosis or alter management. Hospitalisation for the treatment of acute mania is essential and particularly useful early in the episode to provide a low stimulus setting and limit risk-taking behaviours. In this context, early warning signs that indicate a manic episode is imminent can be used to facilitate an early admission to hospital so as to modify management and curtail the development of a full-blown manic episode (Perry et al., 1999), though this is not always effective (Lobban et al., 2010).
Inpatient treatment is also necessary in instances where it has not been possible to achieve effective engagement or adherence with therapy in outpatient settings, or treatments have simply failed and ongoing symptoms continue to confer significant disability. In general, hospitalisation should not be utilised when a less restrictive option is available, but for some treatments, such as ECT, hospitalisation is often necessary.
Clinical management of major depressive disorder
The successful treatment of mood disorders depends on valid diagnosis. It is important to be confident of both the mood disorder diagnosis and the stage of the illness (see: Mood disorders diagnostic criteria; see also Models and formulation for assessment of mood disorders). The evidence base for treating mood disorders is strongly tied to the currently accepted diagnoses, so identifying the diagnosis that best matches a client’s presentation is a prerequisite for evidence-informed practice.
Treatment varies according to the type of mood disorder and the stage of the illness.There are broadly three stages of illness that coincide with three phases of treatment (see: Figure 5), and three kinds of therapy (psychological interventions, pharmacotherapy and physical treatments) that are effective in treating depression.
Treatment options for mild or moderate major depressive disorder
In mild to moderate episodes of MDD, psychological management alone may be adequate, especially early in the course of illness. However, episodes of greater severity, and those that run a chronic course, are likely to require the addition of antidepressant medication, or some other combination of psychological and pharmacological treatment. (See: Figure 6)
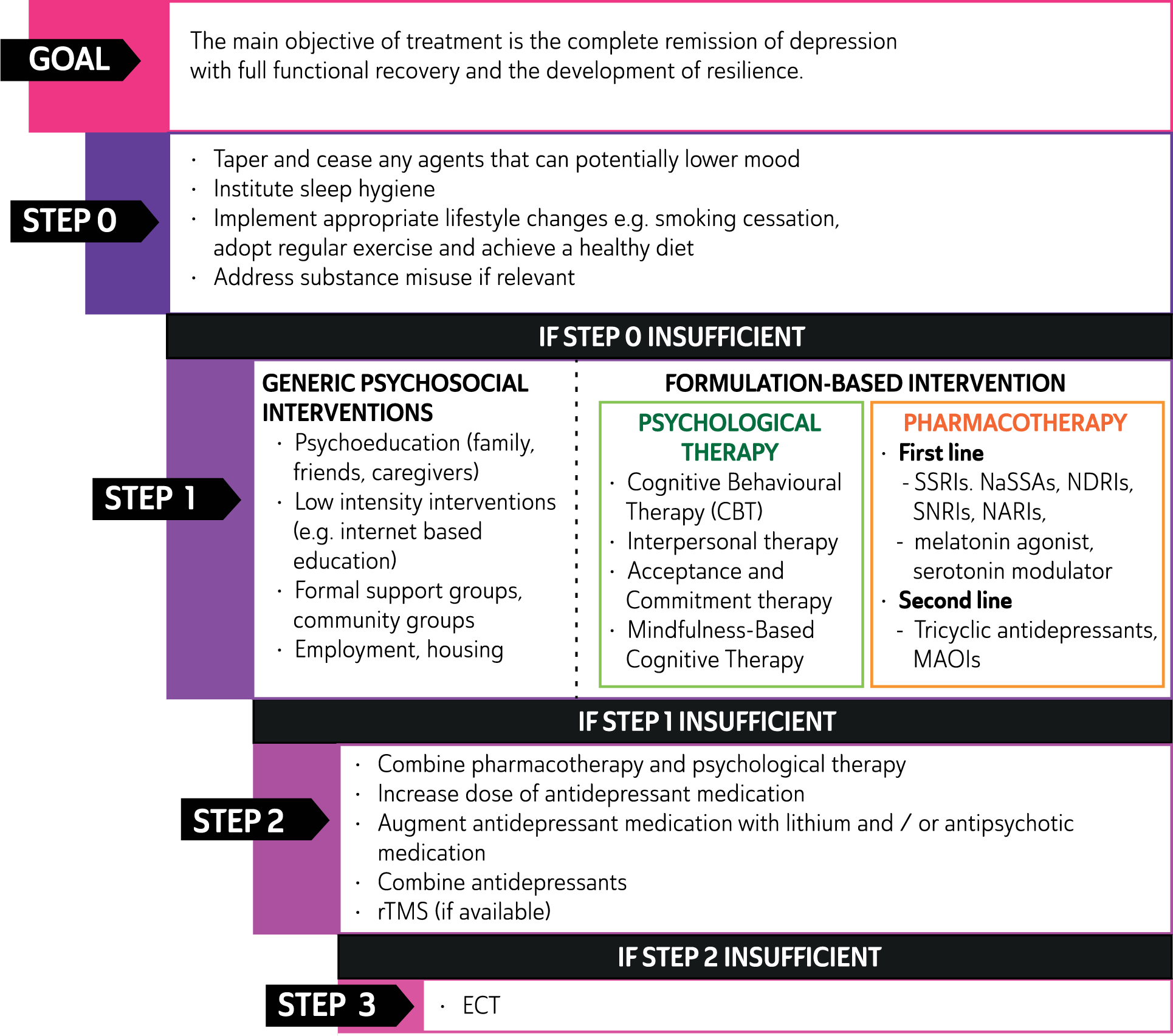
Management of major depressive disorder.
Treatment options for severe major depressive disorder
In severe epsiodes of MDD pharmacotherapy is typically needed and, where there is a high risk of suicide or when the patient’s welfare is threatened by a lack of nutrition or fluid intake, urgent intervention is sometimes necessary and may include electroconvulsive therapy (ECT). (See: Figure 6).
Treatment options available
Psychological therapy for major depressive disorder
A number of psychotherapies have been demonstrated to be efficacious for the acute treatment of depression (see: Tables 11 and 12). Effects are similar across many acute sub-populations of depressed patients (older, postpartum, inpatients, primary care, etc.), and treatment modalities (with some benefits for individual versus group delivery, and benefits for supported versus unsupported online treatments). Psychotherapies should only be delivered by competent practitioners with training in the specific approach, guided by an evidence-based treatment manual and attention to the therapeutic alliance (NICE, 2009).
Psychotherapy for depression acute phase and maintenance/relapse.
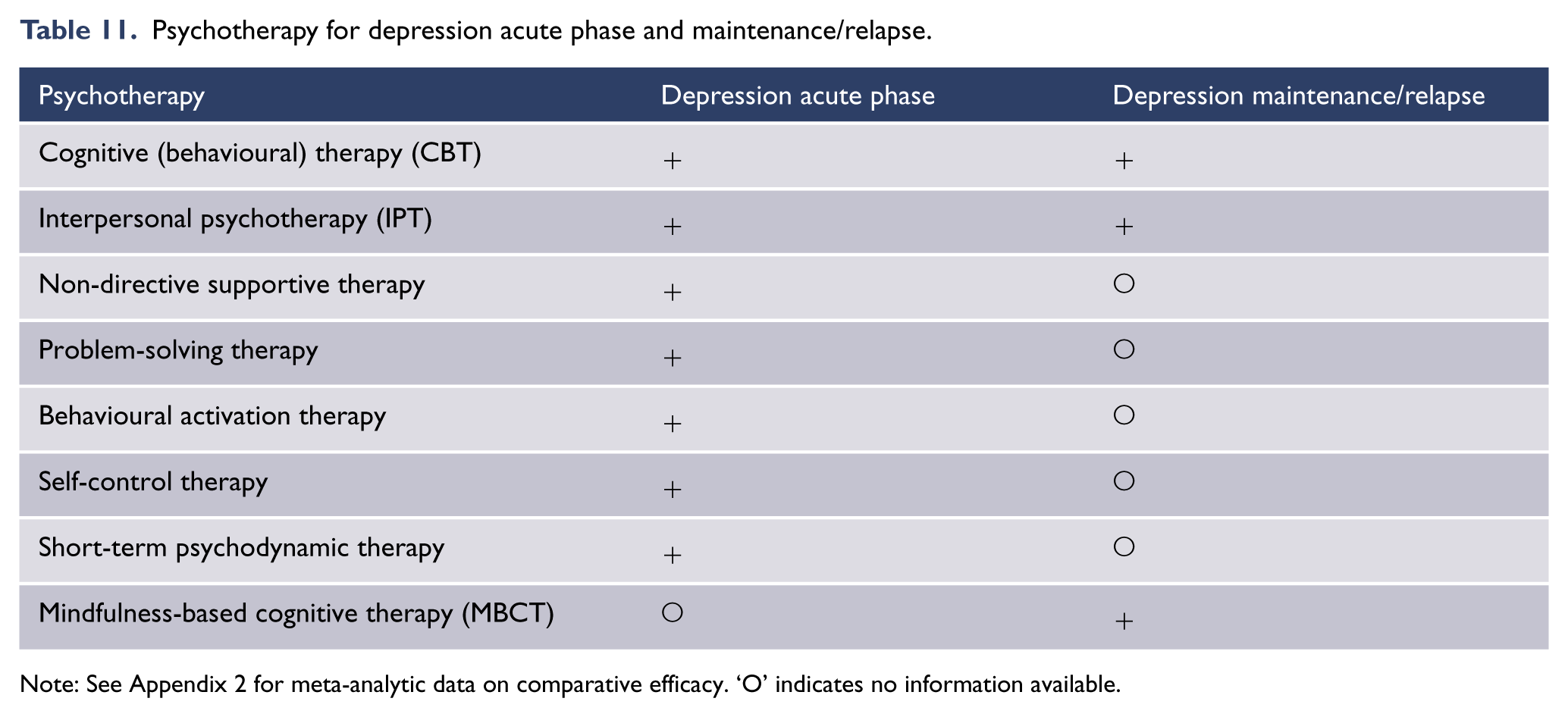
Note: See Appendix 2 for meta-analytic data on comparative efficacy. ‘O’ indicates no information available.
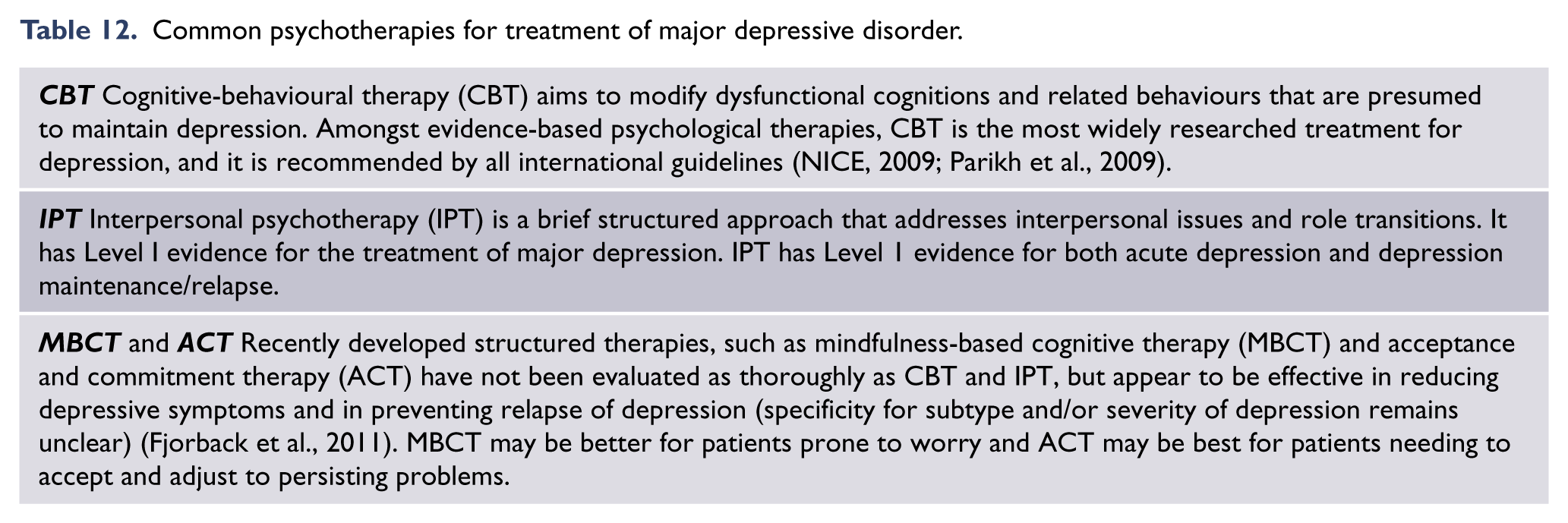
Common psychotherapies for treatment of major depressive disorder.
Effect sizes identified in the original meta-analyses are of moderate magnitude (Cuijpers et al., 2011a), but effect sizes have also been shown to be elevated in studies judged to be methodologically weaker (number needed to treat [NNT] = 2) versus stronger (NNT = 8) (Cuijpers et al., 2010a). The real effect of psychotherapies for depression is therefore likely to be modest, but not without value: Eleven high quality studies reviewed by Cuijpers et al. used active control conditions, so significant findings can be understood as specific treatment effects relative to the substantial non-specific treatment effects of control conditions (Cuijpers et al., 2014a).
Recently, two Cochrane reviews investigated the effectiveness of ‘third wave’ psychotherapies (Acceptance and Commitment Therapy (ACT), compassionate mind training, functional analytic psychotherapy, extended behavioural activation and metacognitive therapy) in the treatment of acute depression. The conclusions of both reviews were restricted by the very limited evidence base. The first found third wave psychotherapies superior to treatment as usual (risk ratio = 0.51 (Churchill et al., 2013)) and the second found no differences between third wave psychotherapies and ‘traditional’ cognitive behavioural therapy (CBT) (Hunot et al., 2013).
Relative effectiveness of psychological therapies
A series of meta-analyses and a network meta-analysis suggest that psychological treatments are equally efficacious in the acute treatment of depression (Barth et al., 2013; Cuijpers et al., 2011a). Although some other analyses have shown superiority for CBT (Tolin, 2010). There is some evidence that therapies differ in the robustness of effects: Barth et al., (2013) and Cuijpers et al., (2011a) found that, when analyses were conducted only on studies with moderate (⩾ 25) or large (⩾ 50) sample sizes, CBT, IPT and problem-solving therapy showed moderate effects versus waitlist, while effects were less robust for psychodynamic therapy, behavioural activation and supportive counselling. Underlining the similarities of effects across therapies, recent studies have found no significant differences between CBT and BA (behavioural activation, a component of CBT, but easier to deliver) (Cuijpers et al., 2007) and between CBT and brief psychodynamic psychotherapy (Driessen et al., 2013).
Similarly, although the evidence base is much wider for CBT, there is no clear evidence of superiority of one type of psychotherapy over any other for prevention of depressive relapse or recurrence: a systematic review found similar effect sizes for CBT, MBCT, and IPT (Biesheuvel-Leliefeld et al., 2015). Though there is little evidence for differential effectiveness across the various psychotherapies, this does not imply that unstructured or eclectic approaches are supported in the psychological treatment of depression. There is strong clinical consensus that treatment is best guided by an evidence-based treatment manual, tailored to the individual patient, and with proper attention to the therapeutic relationship.
Effect sizes for psychotherapies relative to antidepressant medication
There is consensus that CBT is as effective as antidepressant medication for depression of mild-moderate severity (Lampe et al., 2013). This conclusion is underlined by a recent meta-analysis of 10 relevant studies, which found the effect size of psychotherapy compared with pill placebo to be d = 0.25 (NNT = 7.14), which is comparable to the effects of antidepressant versus pill placebo (Cuijpers et al., 2014b).
Findings are more complex in relation to severe depression. Early research based on the large Treatment of Depression Collaborative Research Program found that while antidepressant medication was superior to placebo for more severely depressed patients, CBT was not (Elkin et al., 1989). However, a more recent meta-analysis found the positive effects of psychotherapy were not moderated by severity of baseline depression (Driessen et al., 2010). In fact, that analysis found some evidence for enhanced effectiveness of psychotherapy for outpatients with severe relative to more moderate forms of depression. In direct comparison with antidepressant medication, one review concluded that psychotherapies are as effective for severe depression, but only if therapists are experienced and well trained (DeRubeis et al., 2005). There is some evidence that the presence of comorbid personality disorders may predict a better response to antidepressant medication relative to psychotherapy (Fournier et al., 2008).
Suitability for psychological treatment
There are many factors that can influence whether psychological treatment is more appropriate than pharmacotherapy, including adherence to medications, timing of treatment, formulation, patient preference, psychological insight and extent of life stressors involved in onset. There is often an optimal time within an episode for an individual to engage with psychological treatments, and if a patient is psychologically minded (the capacity or preference for self-examination and introspection), psychological therapy should be initiated early in the course of the illness and this can be stand alone (monotherapy). However, if there is minimal response within a reasonable period of time, then pharmacotherapy should be considered. Depending on the severity and symptom profile of the depressed patient, psychological treatment may be best administered after initiating pharmacotherapy, and in this context clinical features such as melancholic features that may predict better response to SSRIs than CBT should be taken into consideration (Simon and Perlis, 2010). It is recommended that some form of psychological intervention (at a minimum, psychoeducation) accompany pharmacotherapy whenever possible because sometimes where medications produce response but do not achieve remission, the addition of psychological interventions may enable remission.
Overall, evidence from existing studies suggests that psychological therapies (particularly CBT and related approaches, and IPT) are as effective in reducing mild to moderate depression as pharmacological treatments, provided the therapists administering therapy are experienced and well trained (see: Recommendation Box 1).

Pharmacotherapy for major depressive disorder
Antidepressants (Level I)
The efficacy of antidepressants in the treatment of major depression has been extensively studied and substantiated relative to placebo in randomised, double-blinded, clinical trials. However, the decision to treat an individual patient with an antidepressant remains very much a matter of clinical judgment. This is because there are no reliable predictors of response to medication. Psychosocial factors such as abuse or personality traits can be informative, but the decision to pursue pharmacotherapy and the choice of antidepressant is usually based on illness severity, degree of associated impairment, patient preference and additional factors. These include previous response, the presence of atypical features, melancholic features, psychomotor retardation or psychotic features, and/or a family history of response to treatment. Significant dysfunctional premorbid personality factors (including child abuse history) (Nanni et al., 2012) and psychosocial precipitating factors suggest that antidepressants may be less likely to achieve remission as monotherapy. This step of initiating treatment and selecting a suitable antidepressant is critical and therefore any potential predictors of treatment response are invaluable. In this context, the search for pharmacogenetic predictors and other treatment biomarkers that may inform antidepressant choice continues.
With the exception of some early agents, such as the tricyclic antidepressants which are classified according to chemical structure, most antidepressants are grouped according to their primary mode of action. Table 13 lists antidepressant classes according to their principal mechanism of action and the key features they target.
Summary of antidepressant clinical use – acute.
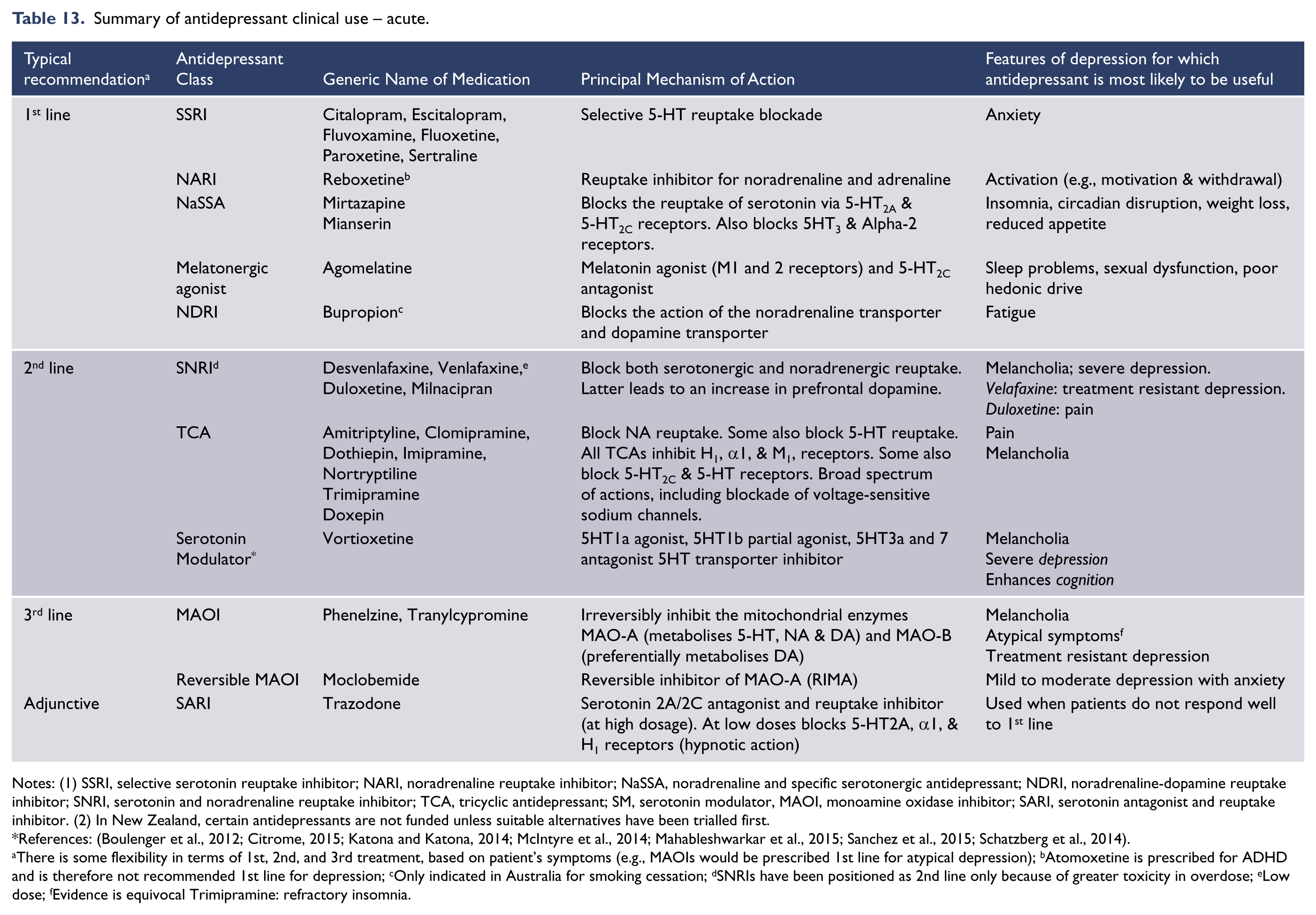
Notes: (1) SSRI, selective serotonin reuptake inhibitor; NARI, noradrenaline reuptake inhibitor; NaSSA, noradrenaline and specific serotonergic antidepressant; NDRI, noradrenaline-dopamine reuptake inhibitor; SNRI, serotonin and noradrenaline reuptake inhibitor; TCA, tricyclic antidepressant; SM, serotonin modulator, MAOI, monoamine oxidase inhibitor; SARI, serotonin antagonist and reuptake inhibitor. (2) In New Zealand, certain antidepressants are not funded unless suitable alternatives have been trialled first.
References: (Boulenger et al., 2012; Citrome, 2015; Katona and Katona, 2014; McIntyre et al., 2014; Mahableshwarkar et al., 2015; Sanchez et al., 2015; Schatzberg et al., 2014).
There is some flexibility in terms of 1st, 2nd, and 3rd treatment, based on patient’s symptoms (e.g., MAOIs would be prescribed 1st line for atypical depression); bAtomoxetine is prescribed for ADHD and is therefore not recommended 1st line for depression; cOnly indicated in Australia for smoking cessation; dSNRIs have been positioned as 2nd line only because of greater toxicity in overdose; eLow dose; fEvidence is equivocal Trimipramine: refractory insomnia.
While it is important to consider the mode of action of antidepressants, this does not predict response. Neurotransmitter systems in the brain do not act independently of each other and a medication which affects one system will also have an effect on other systems. It is still not known precisely how antidepressants exert their therapeutic effect but it is clear that increasing the concentration of synaptic neurotransmitters is only one component of a complex array of intracellular and neuroplastic mechanisms. Therefore rather than primarily relying on neurotransmitter profile the choice of antidepressant should be based on a range of factors, such as side effect profile and tolerability, cost, suicide risk and clinician’s experience.
Principles of antidepressant treatment
Once a potentially suitable antidepressant has been selected (Malhi et al., 2009b, 2013a), the first step is to ensure that it is administered appropriately. For antidepressants to be effective they must be administered at an adequate dose for a reasonable period of time. Therefore, at the outset, tolerability is key and adherence to medication can be enhanced through education, prompt management of side effects and frequent monitoring of progress (Berk et al., 2010). The support of a family member, friend or carer can be helpful in avoiding premature discontinuation. Many patients are also likely to obtain information themselves from the internet and this should be discussed with them to make best application of it.
An adequate trial of an antidepressant should be a minimum of three weeks at the recommended therapeutic dose. Note, some antidepressants require titration to the maximum recommended dose, whereas others, in particular SSRIs, generally have a flat dose-response curve and high doses do not necessarily improve efficacy (Adli et al., 2005). At the same time, it is important not to increase the dose of an antidepressant too rapidly because this increases the likelihood of side-effects and treatment discontinuation. In instances of excessive sensitivity to side effects, it is worthwhile considering metaboliser status and nocebo effects. 6
Response
An antidepressant response is usually discernible in the first two weeks of treatment (Posternak and Zimmerman, 2005; Stassen et al., 1998), and therefore, early follow-ups are important. This also ensures assessment of treatment emergent suicidality often seen in those below age 25 years (Stone et al., 2009). In some instances an antidepressant response takes longer to emerge, but if no improvement is apparent within the first three weeks of adequate treatment, a dose increase or augmentation should be considered, especially if the depressive symptoms are severe and/or disabling. If an adequate dose has been found effective, remission usually requires six weeks of treatment. Switching is an important strategy but should only be considered once an adequate trial at an adequate dose has been achieved.
Response has two important aspects that need to be considered: extent and timing. The extent of response ranges from complete (full) to no (nil) response, with partial response in between. The timing of a response can be prompt (within 7–10 days), delayed (more than 4 weeks) or never occur. Combining the two parameters provides a variety of possible outcomes, the most common of which are depicted in Figure 7.
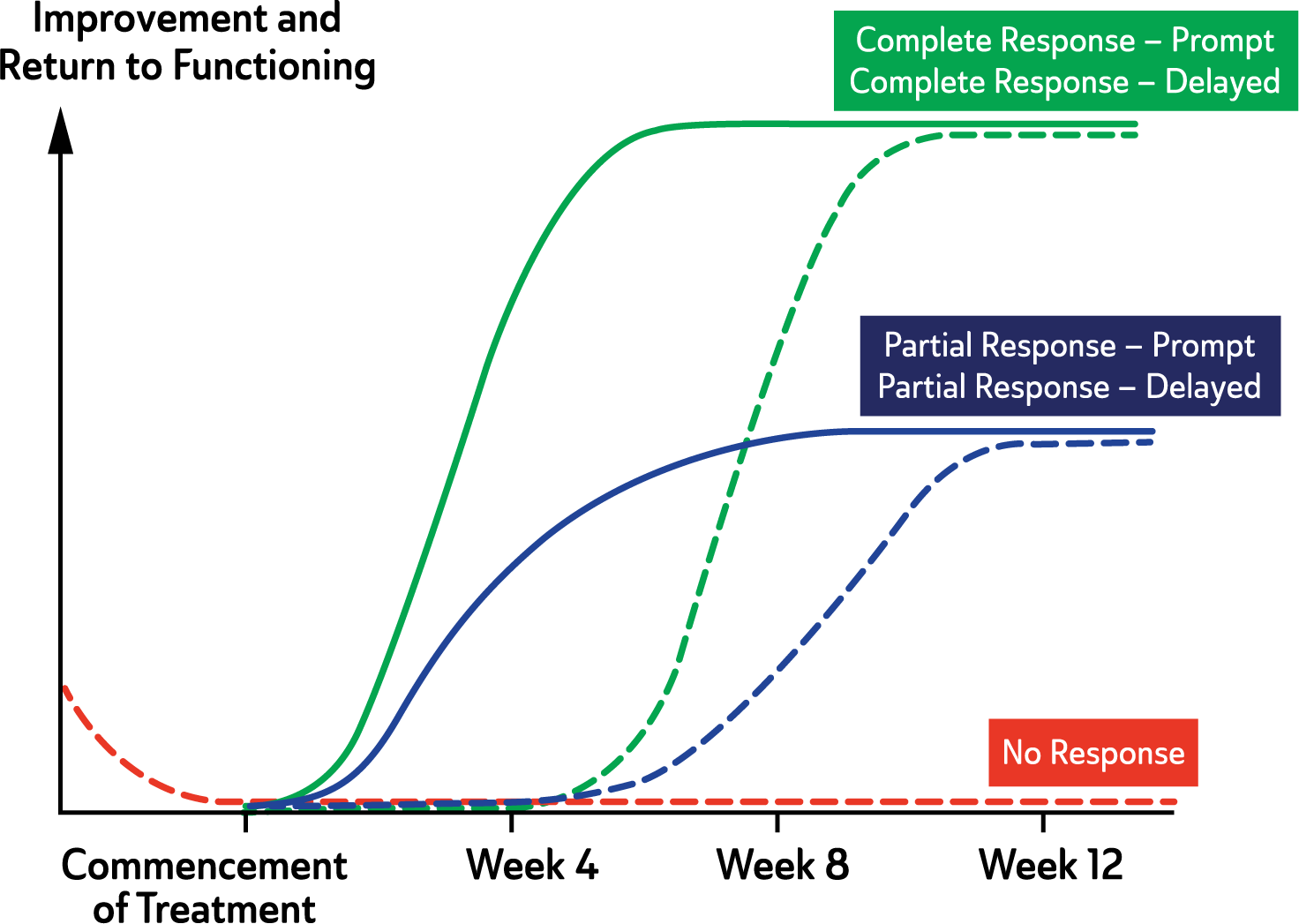
Typical Patterns of Treatment Response.
Efficacy
The efficacy 7 of antidepressants is usually determined in comparison to placebo, and though it is important to know whether an antidepressant is efficacious per se, it is particularly useful to know its differential efficacy as compared to alternative antidepressants. By using clinical trial methodology it is difficult to establish superiority of one antidepressant over another because of the large sample sizes required to detect small differences in efficacy. Therefore, the evidence for differential efficacy across the patient population, or for specific clinical profiles, is relatively modest. Meta-analyses have been used to combine the evidence from a large number of studies of individual antidepressants compared to placebo, but this can only be applied to antidepressants where a sufficient number of studies have been conducted, noting that negative studies often remain unpublished. Using this approach there is some evidence for superiority of venlafaxine over SSRIs (Bauer et al., 2009; Nemeroff et al., 2008; Weinmann et al., 2008), escitalopram over citalopram, venlafaxine, and duloxetine (Kennedy et al., 2008) and sertraline over fluoxetine and other SSRIs as a class (Cipriani et al., 2008). However, other systematic reviews have failed to identify differential efficacy between the newer generations of antidepressants (NICE, 2009; Sartorius et al., 2007). A study that used multiple treatment analysis of 12 new generation antidepressants (Cipriani et al., 2009) examined both efficacy and a measure of tolerability to rate antidepressants and found that mirtazapine, escitalopram, venlafaxine and sertraline were more efficacious than duloxetine, fluoxetine, paroxetine, fluvoxamine and reboxetine. The latter was found to be the least efficacious, although concerns regarding potential inadvertent bias have been raised because of the possibility of unpublished data not being included. The clinical utility of these differences is unclear.
The question of whether newer generations of antidepressants are as effective as tricyclic antidepressants is also unclear. A meta-analysis of the effectiveness and tolerability of TCAs versus SSRIs (Anderson, 2000), for example, failed to demonstrate a significant difference in efficacy but the SSRIs were much better tolerated. However, other studies comparing TCAs and SSRIs in patients with depression have shown that TCAs, such as amitriptyline, have superior efficacy (Barbui and Hotopf, 2001; Faravelli et al., 2003).
Vortioxetine is a new generation antidepressant, which appears to have comparable antidepressant efficacy to SRRIs, SNRIs and agomelatine, but a comparison of efficacy with TCAs and MAOIs is not yet available (Citrome, 2015; Katona and Katona, 2014; Sanchez et al., 2015; Schatzberg et al., 2014).
Overall, the differential efficacy of antidepressants in the treatment of major depressive disorders is not striking. This is partly because of the heterogeneity of the populations treated and the paucity of studies that have conducted a direct comparison. Clinically, traditional antidepressants (TCAs and MAOIs) appear to have greater efficacy, especially in the treatment of melancholic depression, but tolerability and safety concerns in overdose usually relegate these agents to second line administration.
Figure 7 shows the initial patterns of response that are common following the commencement of treatment. Remission following treatment of the first antidepressant trial, as illustrated by the green curve, only occurs in a quarter to a third of patients with major depression (Rush, 2007). In some patients there may be considerable delay before symptoms improve significantly, but there is a discernible change with treatment (see: Figure 7). Therefore, it is important to administer an adequate dose for at least three weeks and try to foster compliance.
Safety
When prescribing an antidepressant, safety issues are of considerable importance, particularly in adolescence, old age, pregnancy and breastfeeding and in the context of depression with comorbid medical illness. The effects of an antidepressant in the event it is taken in overdose is also an important consideration when selecting an appropriate medication for depressed patients who are likely to be at risk of self-harm or suicide. In this regard, the new generation antidepressants, while not without risk (particularly venlafaxine), are significantly safer than the TCAs or irreversible MAOIs.
TCAs can be lethal in overdose because of their cardiovascular side effects (tachycardia, postural hypotension, slowed cardiac conduction). They also increase cardiac morbidity and mortality in patients with ischaemic heart disease (Roose and Miyazaki, 2005). In addition, there is an increased risk of seizures with tricyclics, especially in overdose, particularly dothiepin and clomipramine, and care should be taken in prescribing these to patients with a history of epilepsy, as the seizure threshold can be reduced more so than with other agents.
While less toxic than TCAs, venlafaxine has been shown to be more hazardous in overdose than the SSRIs with an increased mortality from cardiac complications (Deshauer, 2007) and an increased risk of seizures (Montgomery, 2005).
In general, new generation antidepressants are safer options for first line treatment (Peretti, 2000). There are, however, safety issues with SSRIs that require caution (Moret et al., 2009; Roose and Miyazaki, 2005). These include hyponatraemia and osteoporosis, especially in the elderly, and a small risk of prolonged bleeding. Treatment with SSRIs during pregnancy has been associated with a small increased risk of foetal cardiac abnormalities (paroxetine) and potentially fatal pulmonary hypertension in the newborn and a neonatal withdrawal syndrome that can result in seizures has been identified (Moret et al., 2009). This needs to be balanced against the possible harmful effects of depression itself on pregnancy outcomes and its potential impact on the developing foetus (Chaudron, 2013).
Though the risk with therapeutic doses appears to be low, clinicians should be cautious about prescribing doses of SSRIs in excess of the recommended maximum, especially in patients with known cardiovascular disease, 8 due to possible QTc prolongation. Where high doses are needed, an ECG should be considered.
Concerns have been raised about the potential for antidepressants, particularly SSRIs, to cause suicidal thoughts and behaviour especially in adolescents and young adults. These concerns have prompted regulatory authorities in many countries to issue warnings to clinicians. A consensus statement by the World Psychiatric Association (WPA) (Möller et al., 2008) concluded that in the absence of randomised controlled trial evidence, the risk is difficult to assess but that the available data indicated that there was a small risk of SSRIs inducing suicidal thoughts in patients up to the age of 25. The WPA advised that this risk needed to be balanced against the known benefits of treating depression and in preventing suicide. Clinicians should therefore advise young patients and their families of the small chance of suicidal thoughts emerging during the early phase of treatment with SSRIs and monitor all patients for the emergence or worsening of suicidal thoughts during the first 2–4 weeks of treatment. The activation/agitation observed with the initial stages of taking an SSRI can be managed with a low dose of a benzodiazepine prescribed for a limited period of time.
Side-effect profile
General considerations: Tolerability to side effects is an important determinant of adherence to medication and should be weighed against the efficacy profile of the antidepressant (see: Figure 8)
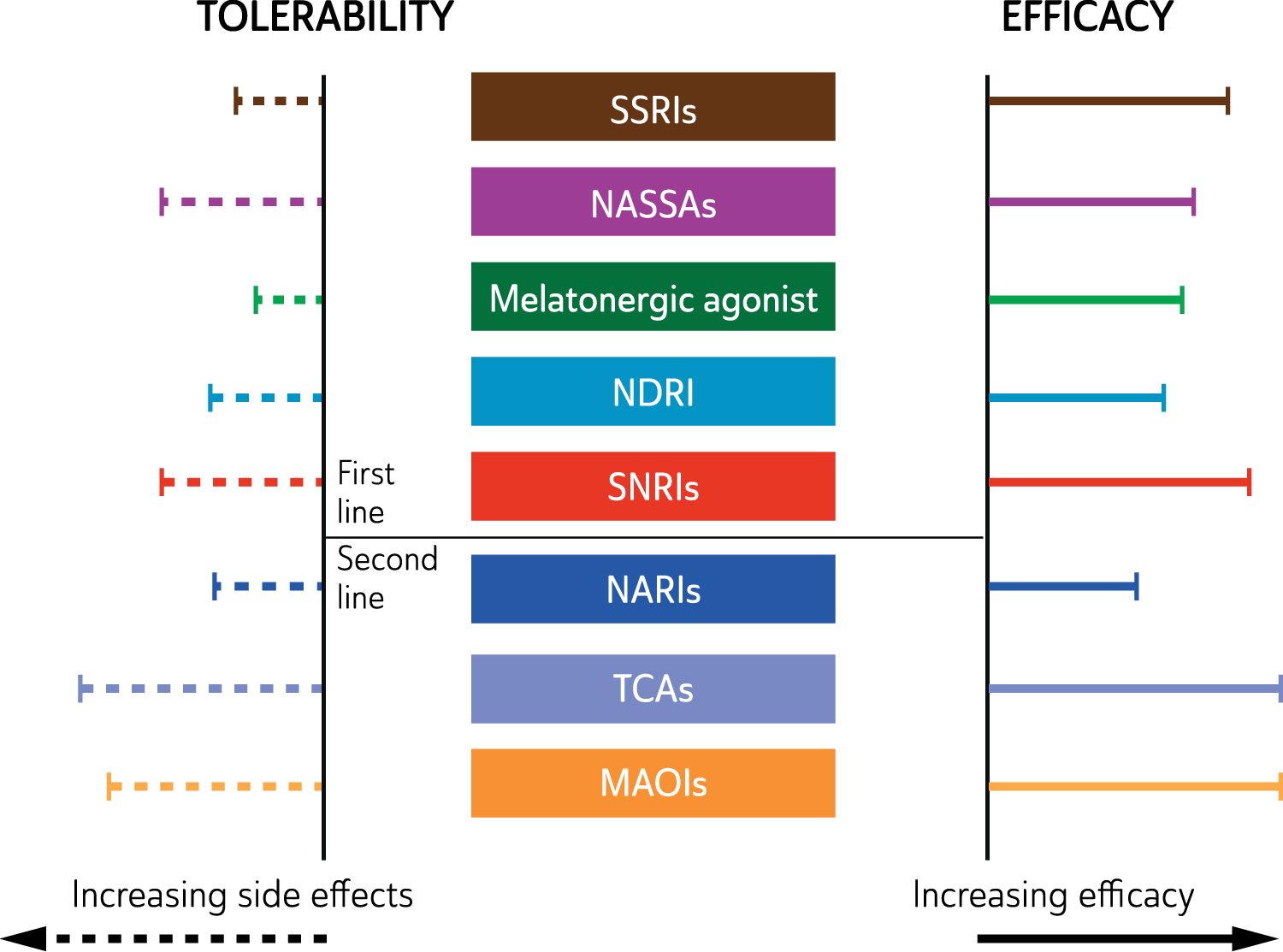
Clinical utility of antidepressants.
New generation antidepressants are in general better tolerated than tricyclics or MAOIs and cause fewer problems with anticholinergic side effects such as sedation, dry mouth and constipation (see: Tables 14 and 15).
Side effects associated with common antidepressant classes doses.

KEY: + <10%, ++ 10-30%, +++ >30%. SM, serotonin modulator. (For other abbreviations see legend of Table 13.)
Some combinations of MAOIs with other drugs can be fatal. Dietary restrictions are necessary to prevent hypertensive crisis.
Clinical considerations of antidepressant classes.
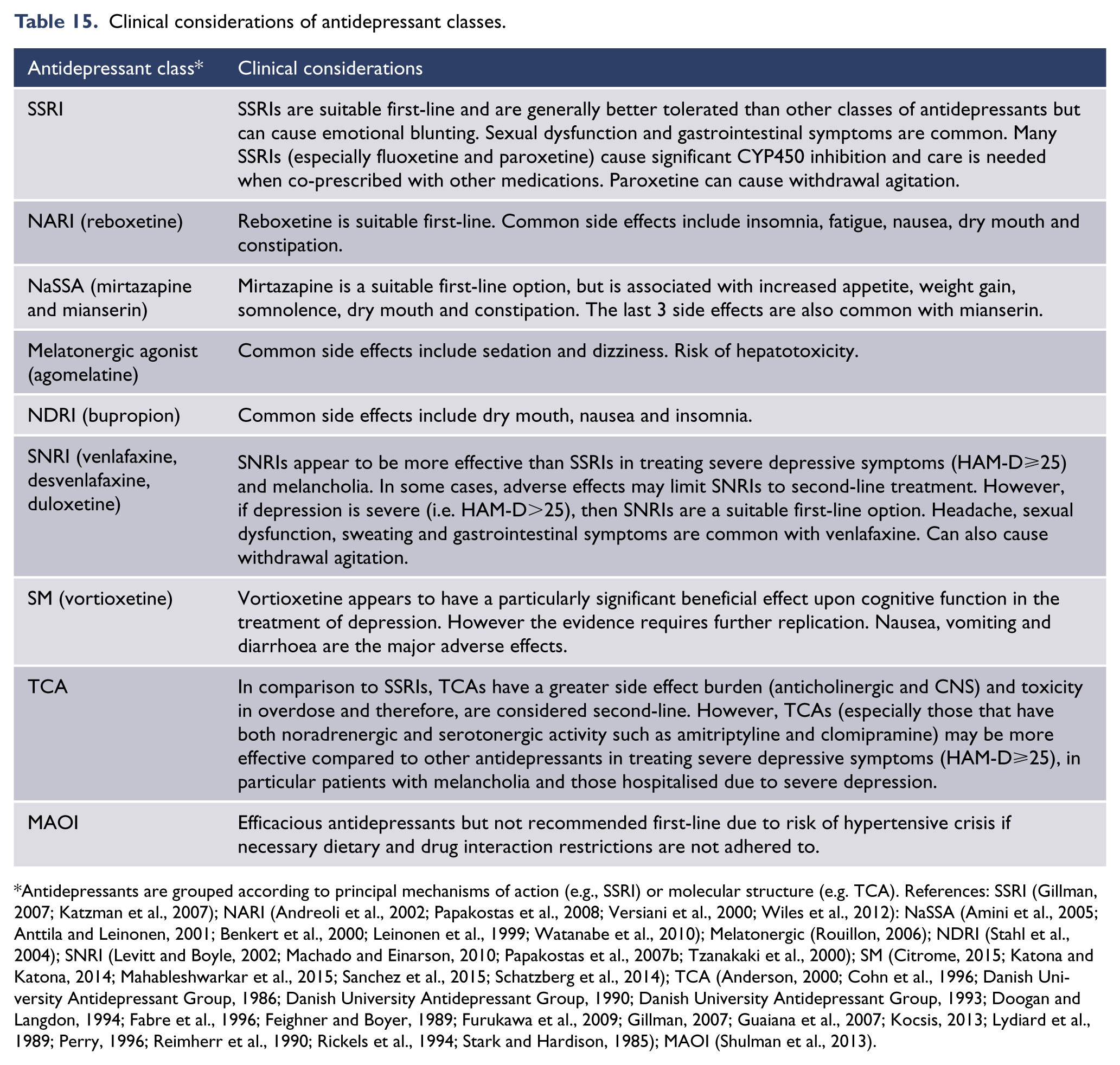
Antidepressants are grouped according to principal mechanisms of action (e.g., SSRI) or molecular structure (e.g. TCA). References: SSRI (Gillman, 2007; Katzman et al., 2007); NARI (Andreoli et al., 2002; Papakostas et al., 2008; Versiani et al., 2000; Wiles et al., 2012): NaSSA (Amini et al., 2005; Anttila and Leinonen, 2001; Benkert et al., 2000; Leinonen et al., 1999; Watanabe et al., 2010); Melatonergic (Rouillon, 2006); NDRI (Stahl et al., 2004); SNRI (Levitt and Boyle, 2002; Machado and Einarson, 2010; Papakostas et al., 2007b; Tzanakaki et al., 2000); SM (Citrome, 2015; Katona and Katona, 2014; Mahableshwarkar et al., 2015; Sanchez et al., 2015; Schatzberg et al., 2014); TCA (Anderson, 2000; Cohn et al., 1996; Danish University Antidepressant Group, 1986; Danish University Antidepressant Group, 1990; Danish University Antidepressant Group, 1993; Doogan and Langdon, 1994; Fabre et al., 1996; Feighner and Boyer, 1989; Furukawa et al., 2009; Gillman, 2007; Guaiana et al., 2007; Kocsis, 2013; Lydiard et al., 1989; Perry, 1996; Reimherr et al., 1990; Rickels et al., 1994; Stark and Hardison, 1985); MAOI (Shulman et al., 2013).
However, the newer antidepressants have a range of side effects which can be unacceptable to some patients necessitating a change of medication. It is important to note that there is considerable variability among antidepressants in the nature and severity of side-effects, and these also vary from one individual to another (Gartlehner et al., 2008, 2011).
Common side effects of SSRIs and SNRIs include nausea, headache, dry mouth, agitation, insomnia, sweating, bruxism, sexual dysfunction and weight gain. Most of these side-effects diminish to tolerable levels during the first one to two weeks of treatment but they can be troublesome and occasionally persist. Short-term use of adjunctive medications, such as benzodiazepines, anxiolytics and hypnotics, may be helpful in managing some of these initial side-effects.
Additional specific considerations: escitalopram and sertraline may have marginally better tolerability than other antidepressants (Cipriani et al., 2009). A small group of patients may experience an immediate and distressing level of agitation with serotonergic drugs, and for these patients, SSRIs may need to be avoided. In the medium to long-term, SSRI induced sexual dysfunction, though sometimes clinically indicative of adequate CNS bioavailability, may necessitate a change to another antidepressant class.
Monitoring response
Following the initial response to an antidepressant, the long-term response is equally variable. Once a patient shows short-term improvement, the symptoms either continue to improve and reach remission, or the response plateaus. When the latter scenario occurs, a change of treatment strategy may be necessary. The STAR*D study. (Rush et al., 2006) showed that after four successive trials of antidepressants a cumulative remission rate of 67% was reached, though it may require 2–3 antidepressant trials before complete remission is reached. Even with switching to an alternative medication or adding an augmenting agent, only 30% of depressed patients achieve a partial response or remain non-responsive (refractory). These patients require a more extensive re-evaluation of both the diagnosis and perpetuating factors, or are candidates for physical therapies such as ECT.
Combined psychological therapy and antidepressants (Level I)
For most patients with depression, a combined treatment approach is more effective than either psychological or antidepressant treatment alone. This applies particularly to depression of moderate or greater severity (HAM-D ⩾18) and chronic depression (Level I) (Cuijpers et al., 2009; De Maat et al., 2006; Manber et al., 2008; Pampallona et al., 2004; Schramm et al., 2007, 2008; Simon et al., 2006). In most cases of depression there are significant psychological issues, either aetiological or maintaining/perpetuating the illness. Comorbid anxiety and/or personality vulnerabilities also play a significant role. Depressive illness is inevitably a traumatic experience which impacts psychosocial, occupational, and relationship functioning, and therefore, psychological support at some point in the course of the illness is often necessary for early episodes. With regard to the structured psychotherapies, a systematic review of combined treatment with antidepressants (Oestergaard and Møldrup, 2011) concluded that pharmacotherapy combined with psychotherapy is associated with a higher probability of remission and a lower risk of relapse. Combination therapy should be considered when response to a single modality has been suboptimal or unsuccessful (Driessen and Hollon, 2010).

Footnote: Tolerability (side effect profile) is as important as efficacy when choosing an antidepressant.
Tricyclics, MAOIs and dual action agents may be more efficacious than newer agents in the treatment of moderate-severe depression.

Brain stimulation methods
Electroconvulsive therapy (Level I)
Electroconvulsive therapy (ECT) is a safe and effective treatment for the more severe forms of depression (Carney, 2003), where its antidepressant effect is found to be superior to medication strategies (Lisanby, 2007).
Therefore, in practice it is usually reserved for patients who have not responded to several trials of medication. ECT is recommended as first-line treatment in extremely severe melancholic depression, particularly when the patient refuses to eat or drink and/or is a very high suicide risk, or when the patient has very high levels of distress, has psychotic depression, catatonia or has previously responded to ECT (see: Table 16).
First- and second-line indications for ECT.

ECT is highly effective in the treatment of psychotic depression for which it is a first-line option (Lisanby, 2007). A large multicentre study of the efficacy of bitemporal ECT showed a remission rate of 95% for completers with psychotic depression (Petrides et al., 2001).
Electrode placement
The choice of electrode placement and dose of ECT needs to be determined for the individual patient, balancing efficacy against side effects. These parameters influence effectiveness, particularly for unilateral ECT (Loo et al., 2006). Doses as high as six times threshold can maximise the efficacy of unilateral ECT, while for bilateral placements (bitemporal or bifrontal) doses at 50–100% above threshold are usually sufficient.
The electrode placements currently in use are right unilateral, bitemporal and bifrontal. The relative efficacy of each is dependent on dose above seizure threshold and the data from comparative studies is difficult to interpret because of inconsistent methodologies. Generally, bitemporal ECT is more effective than unilateral, and high dose is more effective than low dose for both placements, but cognitive side-effects are greater with bitemporal placement and at higher doses (Carney, 2003). At six times threshold the efficacy of unilateral ECT approaches that of bitemporal ECT, but at the cost of greater cognitive impairment compared to lower doses (Loo et al., 2006).
Bifrontal ECT, while less studied, appears to be as effective as high dose unilateral ECT and bitemporal placement. Data from early studies (Bailine et al., 2000; Dunne and McLoughlin, 2012; Ranjkesh et al., 2005) and pooled analyses of data from studies comparing bifrontal with unilateral and bitemporal ECT show fewer cognitive side effects for bifrontal ECT compared with bitemporal ECT (Plakiotis and O’Connor, 2009). However, a much larger multicentre study failed to show a cognitive advantage for bifrontal ECT (Kellner et al., 2010) though the large amount of missing cognitive data in this study makes the findings difficult to interpret.
Overall, the evidence is insufficient to conclude a cognitive benefit for bifrontal over bitemporal ECT. Clinical experience however suggests that bifrontal ECT can be associated with fewer cognitive side effects than bitemporal ECT and may be a useful option for some patients. There is evidence that bifrontal ECT is associated with a lower rate of ECT-induced cardiac arrhythmias (Stewart et al., 2011) so it may also have safety advantages over unilateral and bitemporal ECT, particularly in patients with cardiac problems.
The introduction of ultra-brief pulse width (0.3msec) unilateral ECT has made possible effective treatment with markedly fewer cognitive side-effects (Loo et al., 2008; Sackeim et al., 2008; Spaans et al., 2013; Verwijk et al., 2012). The efficacy of unilateral ultra-brief ECT appears to be equivalent to standard brief pulse unilateral ECT but more treatments may need to be given to achieve remission (Loo et al., 2007). Ultra-brief pulse width bifrontal ECT has been shown to be effective with cognitive sparing (Sienaert et al., 2009) but bitemporal ultra-brief ECT has been shown to be ineffective (Sackeim et al., 2008) and is not recommended.
As a general recommendation and balancing efficacy against cognitive side-effects, electroconvulsive therapy for most patients should begin with unilateral ultrabrief pulse width ECT at six times threshold, or brief pulse width ECT at 3-6 times threshold. When administering ECT, both options should be available and choice should be determined by balancing the need for speed of response against concern regarding potential cognitive side effects. Consideration should be given to using bifrontal or bitemporal ECT if speed and reliability of response is paramount, for instance if patients are dehydrated or extremely suicidal. If treatment is commenced unilaterally, switching to bifrontal or bitemporal ECT should be considered after approximately six treatments if there is no response.
Following a course of ECT, relapse rates within six months have been shown to be over 50% despite maintenance pharmacotherapy (Kellner et al., 2006). Therefore in some cases maintenance ECT may be necessary.
Side effects
The efficacy of ECT needs to be balanced against potential cognitive side-effects, which increase with increasing doses of ECT. Memory impairment is the side-effect of most concern to patients. Both anterograde and retrograde memory are variably affected. Anterograde memory changes generally return to normal or may be improved compared to pre-ECT levels within 2–4 weeks (Semkovska and McLoughlin, 2010) but retrograde impairment, which is more likely with bitemporal placement, can persist for prolonged periods (Sackeim et al., 2007). It is important that patients and their immediate family/partner are made aware of the potential effects on memory before undergoing ECT.
Repetitive transcranial magnetic stimulation (rTMS)
Repetitive transcranial magnetic stimulation is a technique that, having been the subject of intensive research since the mid-1990s, is now being increasingly used in clinical practice in many countries. rTMS is a treatment method that utilises a pulsed magnetic field to stimulate and potentially alter brain activity. Repeated cortical stimulation with TMS pulses can either increase or decrease local brain activity depending on the frequency of stimulation. A typical rTMS treatment course involves daily treatment provided Monday to Friday over 4 to 6 weeks. Stimulation is applied daily for between 30 and 45 minutes. Patients receiving rTMS do not require a general anaesthetic and are typically able to return to work or drive immediately after the end of outpatient treatment. rTMS can be applied as an outpatient or to hospital inpatients. Unlike ECT there are no cognitive side effects, and unlike medications, there are no drug-drug interactions or systemic side effects.
When used to treat depression rTMS is most commonly administered as repeated short bursts of high frequency stimulation (~10 Hz) to the left dorsolateral prefrontal cortex (DLPFC). A large body of clinical sham controlled trials have demonstrated the antidepressant efficacy of this form of rTMS; supported by several meta-analyses (Level I) (Schutter, 2009; Slotema et al., 2010). There is also significant evidence (summarised in a meta-analysis) to support the antidepressant efficacy of low frequency rTMS (an alternate form of rTMS) applied to the right DLPFC (Level I) (Schutter, 2009). Bilateral rTMS does not appear to produce better results than unilateral rTMS, and therefore unilateral approaches that can be generally delivered more quickly are preferable (Chen et al., 2014).
The majority of clinical trials conducted evaluating the use of rTMS in depression have been conducted in patients who have failed to respond to one or more antidepressant medication strategies. rTMS treatment appears to be effective whether it is applied as a monotherapy or in conjunction with antidepressant medication (Slotema et al., 2010). Further research is required to evaluate whether rTMS can be successfully utilised as a maintenance treatment strategy and early studies suggest that this may be possible (Fitzgerald et al., 2013a). The findings from studies supporting the use of rTMS treatment have led to its approval and use in many countries, and it is increasingly being used in clinical practice in Australia.
Side effects
rTMS is generally well tolerated. The major treatment related side effects are headache and scalp discomfort at the site of stimulation (Rossi et al., 2009). Treatment discontinuation rates in clinical trials and in clinical practice are typically quite low. There is a potential risk of seizure induction with rTMS treatment although with suitably selected patients this risk appears extremely low (Rossi et al., 2009). In a manner similar to other antidepressant treatments, there is a risk of manic switch with rTMS treatment in patients with bipolar disorder, although the risk of this is considered low (Rossi et al., 2009), and patients with metal implants from neurosurgery or pacemakers may be unsuitable for rTMS due to risks of interference from the high power magnetic field.

Footnote: *Includes major depressive episodes occurring in context of Bipolar Disorder.
Transcranial direct current stimulation (tDCS)
tDCS involves the application of a weak electrical current through an anode and cathode to subtly modify brain activity. Initial research suggests that tDCS can modify mood in depressed patients, although it is unclear at this stage whether these effects are clinically relevant. Antidepressant effects of tDCS have been supported in recent larger studies (Brunoni et al., 2013; Loo et al., 2012) and a meta-analysis (Shiozawa et al., 2014), but another recent meta-analysis found no difference between active and sham tDCS on clinically relevant endpoints, such as response and remission rates (Berlim et al., 2013). Definitive recommendations await the outcomes of large multi-site trials of tDCS that are currently underway.
Magnetic seizure therapy (MST)
MST is a new form of convulsive therapy that is being evaluated as an antidepressant strategy. MST involves the induction of a seizure in a procedure similar to that undertaken with ECT (Fitzgerald et al., 2013a; Lisanby et al., 2001). However, instead of inducing a seizure with an electrical current, the MST seizure is produced using a high intensity transcranial magnetic stimulation device, which instigates the seizure via high frequency rTMS pulses. Initial data suggest that MST might have efficacy similar to ECT without equivalent cognitive side-effects (Kayser et al., 2011), but only limited evaluation of this technique has occurred to date.
Deep brain stimulation (DBS)
DBS is a procedure which involves the implantation of a small series of stimulating electrodes into the brain connected to a pacemaker type pulse generator which is placed below the clavicle in the chest. DBS is used extensively for patients with advanced Parkinson’s disease and a number of other neurological conditions. In recent years a number of groups have explored the use of DBS applied to several brain regions in the treatment of patients with highly refractory depression. Small studies have suggested that DBS in several brain regions including the subgenual cingulate cortex and the nucleus accumbens may have antidepressant effects (Bewernick et al., 2010; Holtzheimer et al., 2012; Lozano et al., 2012; Merkl et al., 2013). However, two recent industry sponsored trials have been terminated early due to the lack of positive interim results (Dougherty et al., 2014; Morishita et al., 2014). DBS is potentially associated with a number of serious but uncommon surgical complications including haemorrhage or infection and a wider range of reversible stimulation effects such as insomnia.
Vagus nerve stimulation (VNS)
Vagus nerve stimulation involves the surgical implantation of a pulse generator, similar to a pacemaker, in the chest wall that is connected to stimulating electrodes attached to the vagus nerve in the neck. The main existing indication for VNS is in the treatment of refractory epilepsy. A small series of studies has evaluated whether VNS has antidepressant effects (Nemeroff et al., 2006), and it appears to produce some antidepressant response but this emerges slowly and only in a minority of patients. Clinical treatment of depression using VNS is not available in most countries but a transcutaneous form of VNS that does not require surgery targeting the auricular branch has been developed and is being evaluated for clinical use.
Complementary therapies
The marked growth in complementary therapies health spending reflects the public demand for non-prescription medicines and health supplements (MacLennan et al., 2002; Xue et al., 2007). While this in part may reflect limitations of existing pharmacological therapies, it appears many people use complementary therapies not as an alternative to prescription medicine (Ennis, 2014), but to enhance a general sense of health and wellbeing (Astin, 1998; Moerman and Jonas, 2002). Clinicians should be aware of the safety and efficacy profiles of these therapies in order to help guide patients that elect to take them (Braun et al., 2010). Awareness of some potential interactions with prescription medications is also important (Gouws et al., 2012). An open discussion with patients about their use of complementary therapies can assist overall engagement with care and help build rapport. The discussion should also include advice on the importance of a healthy diet and regular exercise for optimal recovery and wellbeing (Berk et al., 2013). Complementary therapies have mainly been studied in the setting of mild to moderate depression, and as adjuncts to prescription medication in more severe depression and in bipolar disorders.
Several of the more commonly used complementary therapies to assist mood are summarised in Table 17. Given the morbidity and mortality risks of severe depression and bipolar disorders, complementary therapies should not be used instead of established prescription agents. A problematic issue with complementary therapies is the marked variability in active ingredient between brands, reflective of lower regulatory requirements (Breach et al., 2003; Petroczi et al., 2011). This makes it harder to translate the limited evidence base to many of the commercially available products. Given the sometimes substantial costs of complementary therapies, it is reasonable to discuss the merits of their ongoing use in a vulnerable patient group with often scarce resources. By the same token, if complementary therapies provide subjective benefit to patients, are affordable, are free of serious adverse effects, and do not prevent use of more proven conventional treatments, then the use of such therapies by patients should be respected. For some patients the sense of active coping and empowerment from using complementary therapies may enhance their sense of wellbeing and control, producing psychologically meaningful effects for them (Moerman and Jonas, 2002).
Complementary therapies * for depression.
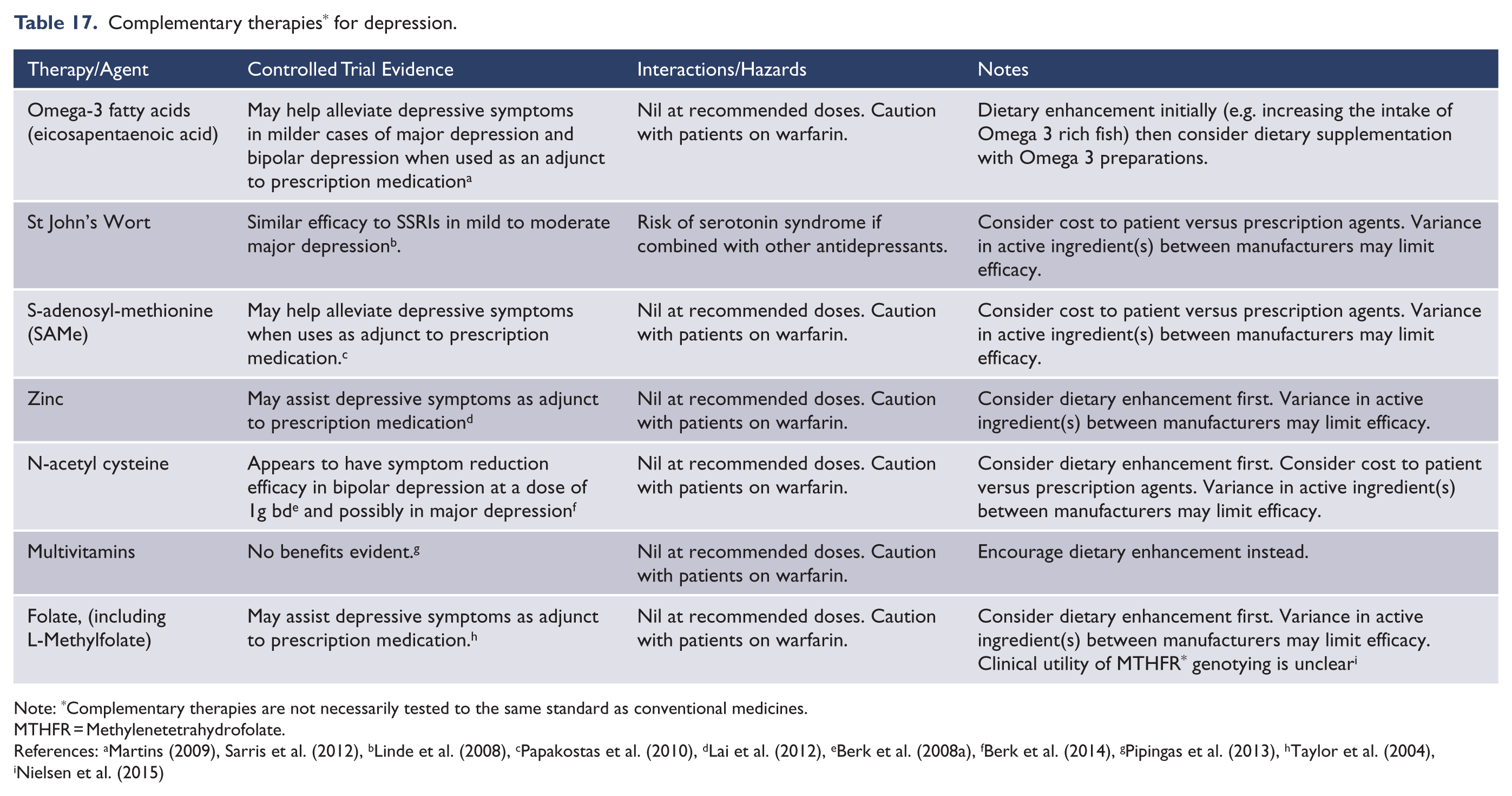
Note: *Complementary therapies are not necessarily tested to the same standard as conventional medicines.
MTHFR = Methylenetetrahydrofolate.
Health lifestyles
Diet and mental health
Since the end of 2009, there has been an exponential rise in the number of published studies documenting associations between higher diet quality and a reduced likelihood or risk for depression, as well as an increased likelihood or risk for depression with higher intakes of unhealthy food products; such studies have been published from countries across the globe in children, adolescents and adults (Jacka et al., 2010, 2011, 2013; Sanchez-Villegas et al., 2009). A recent meta-analysis has confirmed that adherence to a ‘healthy’ diet pattern is associated with a reduced likelihood of depression in adults. Although studies were too few for confirmation, there was a strong trend also observed for a positive relationship between ‘western’ style (unhealthy) dietary pattern and depression (Lai et al., 2014). Similarly, another recent meta-analysis reported that high adherence to a Mediterranean diet, also a very healthful dietary pattern, is associated with a 30% reduced risk for depression (Psaltopoulou et al., 2013). More recently still, a systematic review confirmed relationships between unhealthy dietary patterns and poorer mental health in children and adolescents (O’Neil et al., 2014).
Although the evidence base for the relationship between dietary intake and mental disorders is very new, and thus largely limited to animal studies and observational studies in humans to date, a recent large-scale dietary intervention provides important support for the findings of the meta-analyses. In the PREDIMED study, individuals at increased risk for cardiovascular events were randomised to a Mediterranean diet supplemented with either extra-virgin olive oil (EVOO) or mixed nuts, with a low-fat control diet. Although not statistically powered to assess prevention of depression, the results demonstrated a strong trend to a reduced risk for incident depression for those randomised to a Mediterranean diet with nuts, and this protective effect was particularly evident in those with type 2 diabetes (Sanchez-Villegas et al., 2013). Similarly, a recent prevention trial in older adults in the US showed that dietary counselling was as effective as psychological therapy in reducing rates of transition to case-level depression (O’Neil et al., 2013; Stahl et al., 2014). The first RCT to test causal relationships between dietary improvement and improvements in mental health is underway (O’Neil et al., 2013), and existing observational and prevention data support a causal relationship between diet quality and depression (Jacka et al., 2012) and are supported by extensive pre-clinical data.
Physical activity/exercise
The existing observational data have shown that regular exercise is protective against the development of depression, while physical inactivity is a risk factor for the development of depressive symptoms. For example, results from the Nurse’s Health Study, involving nearly 50,000 American women, showed that women who were more physically active had a reduced risk for clinical depression over a ten year follow up period (Lucas et al., 2011). In the 1958 and the 1970 British birth cohort studies, comprising nearly 30,000 people, increased leisure-time physical activity in adolescence was consistently related to increased well-being in adulthood (Sacker and Cable, 2006). Physical activity is also associated with a reduced risk of developing de-novo depression in elderly people (Pasco et al., 2011). A recent meta-analysis supports the link that physical activity prevents depression (Mammen and Faulkner, 2013). Physical activity is thus important in reducing relapse or the transition to case level disorder in those with subsyndromal symptoms as well as in preventing de novo depression.
Exercise is also established as an effective treatment strategy for depression. A meta-analysis, including results from 11 randomised controlled clinical trials, concluded that exercise is highly effective as a treatment intervention in depression with a large pooled effect size (Stathopoulou et al., 2006). Another meta-analysis including 90 articles and over 10,000 patients confirmed that exercise reduces depressive symptoms in patients with chronic illness, suggesting that patients with comorbid physical conditions also benefit from recommendations to exercise (Herring et al., 2012).
Exercise is important for general wellbeing and seems to have efficacy as an augmentation strategy in major depression (Rimer et al., 2012). A possible mechanism appears to involve brain-derived neurotrophic factor (BDNF) (Szuhany et al., 2015). However, motivating more severely depressed patients to engage is a challenge (Schuch et al., 2015). Positive studies in the field may in part be mediated by selection bias with less severely depressed cases and those improving more likely to engage in studies. Also, blinding in comparator trials is a problem. Nonetheless, it stands to common reason that for both mood and general medical wellbeing sedentary patients should be actively encouraged to engage in regular exercise.
Smoking
Evidence from observational studies supports smoking as an independent risk factor for mood and anxiety disorders (Moylan et al., 2012). Importantly, a recent systematic review and meta-analysis showed that smoking cessation is associated with reduced depression, anxiety, and stress and improved positive mood and quality of life compared with continuing to smoke. The effect sizes were as large for those with psychiatric disorders as those without and were equal or larger than those of antidepressant treatment for mood and anxiety disorders (Taylor et al., 2014). Thus, smoking cessation should receive more prominence as a clinical target in those with mood disorders.
The WHO has stated “there is no health without mental health” and therefore, recommendations and encouragement to follow national guidelines for dietary and exercise practices should be a prominent component of care for all people with depression and depressive symptoms.
Omega-3 PUFAs for mood disorders
Mood disorders are associated with pronounced reductions in Omega-3 PUFA (polyunsaturated fatty acids) status (Lin et al., 2010). Meta-analyses that have applied methodological rigour to the investigation of the role of Omega-3 PUFAs as treatments for clinical depression, rather than including studies based on self-report depression, show omega-3 PUFA, particularly EPA, to be efficacious (Lin et al., 2012).
The biological mechanisms underlying the antidepressant effects of Omega-3 PUFAs include, but are not limited to, regulation of neurotransmitters serotonin and dopamine; mood stabilization via arachidonic acid cascade; anti-inflammatory and anti-oxidant effects; and positive actions on neuroplasticity and neurogenesis. As such, the biological actions of Omega-3 PUFAs are similar to those of standard medications (e.g. antidepressants) and should be considered for use in patients for whom other treatment options are not acceptable. They should also be considered as adjunctive treatment. For this reason, clinical recommendations for the use of omega-3 PUFA, particularly those with a higher ratio of EPA to DHA, can be strengthened.
Sleep
With the exception of light treatment for seasonal affective disorder, and agomelatine (melatonergic agonist) for depression, there is not strong evidence for chronobiological treatment of acute depression (Kuiper et al., 2013).
Nonetheless, there is robust evidence for causal (often bidirectional) links between sleep and negative mood, including major depression (Harvey, 2011; Malhi and Kuiper, 2013). Consequently, a comprehensive approach to lifestyle in the maintenance phase of major depression should include assessment and where appropriate behavioural intervention for improving sleep (Asarnow et al., 2014). As with diet and exercise prescription, clinicians may not require specialised skills in behavioural sleep interventions. Rather, providing encouragement and psychoeducation to follow recognised sleep hygiene guidelines for achieving adequate sleep are likely sufficient.
Managing suboptimal response
Assessing treatment of poor response
Given the difficulties in defining treatment resistance or non-response (Malhi et al., 2005b), a practical approach is to consider partial or non-response to treatment as the persistence of depressive symptoms and a lack of functional recovery (Malhi et al., 2009b). In practice, despite a clinician’s best effort to subtype and tailor a patient’s treatment, some patients do not respond to treatment as anticipated. Therefore, this section outlines how to assess non-response and implement appropriate strategies when a patient is non-responsive to treatment. The literature on non-response to pharmacological treatment is modest, and research on non-response to psychological therapy is limited.
Poor response to psychological therapy
Time to respond to treatment is shown to be longer with psychological treatment than pharmacological treatment (Hollon et al., 2005b). But, significant early improvement has been shown even after a few weeks of psychological therapy (Keller and Hautzinger, 2007) and an early response to psychological treatment is a strong predictor of achieving a stable response after longer term treatment (Tadić et al., 2010). However, not all patients will engage with or respond to psychological treatment, and it is these patients that require further assessment.
If a patient fails to respond after multiple sessions of CBT/psychological treatment, the first step should be to re-evaluate the diagnosis, taking note that the patient may have developed new symptoms, the depression may be more complex than originally thought, personality features may be contributing, or an alternative diagnosis, for example substance abuse, better fits the patient’s symptoms. Sometimes a poor response may be the result of the therapist, such as a ‘poor fit’ between patient and therapist or the therapist not adhering to the treatment protocol, and chronic patients may indeed do worse with CBT (Scott et al., 2006). At this point it may be helpful to seek advice from colleagues. If it has been established that the original diagnosis was valid, then refer the patient to a psychiatrist for pharmacological treatment.
Poor response to pharmacological treatment
Antidepressant efficacy trials generally report response rates of 60–70% but the rate of remission is much lower at 30–40% (Nierenberg et al., 2007). Interestingly as the use of antidepressants has broadened, response rates have dropped by 3 points on the HAMD every decade antidepressants have been in use. In the STAR*D study (Trivedi et al., 2006), the remission rate following 10 weeks of citalopram therapy was 28% (HAM-D ⩽7) whereas the response rate was 47% (QIDS-SR ⩽5). As a rule of thumb, only 1/3 of patients will remit with initial antidepressant treatment. Treatment non-response is therefore a significant issue in managing depression. The first step in assessment of non-response is to review the diagnosis and treatment adherence.
After ensuring that the patient has been taking their medication as prescribed (many patients fail to collect a second script and/or or take it when they ‘feel’ depressed), several different strategies can be utilised (see: Figure 9), including increasing the dose of the current medication, augmentation, or switching to a different antidepressant (all strategies are discussed later). If at any point in the treatment process the patient is not responding to the treatment, an important step is to re-evaluate the diagnosis. Note that the patient may have developed new symptoms, the depression may be more complex than originally thought, or an alternative diagnosis better explains the patient’s symptoms. Treatment may therefore need to pursue a new treatment algorithm. During this assessment, other unique and individual factors should be taken into consideration, such as the patient’s personality and lifestyle, social factors or substance abuse, all of which can be contributing to depression. In some cases it may be necessary to seek advice from colleagues to provide a fresh perspective and framework for management. If it has been established that the original diagnosis was valid, then it may be necessary to switch the patient to a new antidepressant.
Strategies to address medication non-response.
When choosing a new antidepressant, consider one with different mechanisms of action to the original antidepressant. Before switching the patient’s medication, consider which switching strategy will be utilised (see: Switching strategies) and inform the patient about the strategy and the new medication. If there is a reduction in the severity of symptoms, continue with the treatment and assess again in a month with the goal of achieving remission. If the symptoms plateau (only a partial response has been achieved), maximise the dose of the partially effective agent and then consider adding an augmenting agent or combining antidepressants (see the section on Augmentation (Level I) and the section on Combining treatment). If, after further evaluation of the patient, it is determined that the patient is still not responsive to antidepressant medication, the patient is considered to have treatment resistant depression (TRD), defined as a lack of improvement following adequate trials of two or more antidepressants. Note, when communicating this to the patient the term ‘difficult to treat’ is preferable because treatment resistant or refractory implies no hope of a response and may also be viewed as assigning blame. In TRD, research supports persisting with pharmacological strategies, but consideration of ECT, rTMS and/or psychotherapeutic approaches is also important (Parikh et al., 2009). These strategies are addressed below.
Re-evaluate diagnosis
Diagnostic review should include re-assessment of the subtype of depression (e.g. psychotic, melancholic, atypical), personality issues that could be underpinning depressive symptoms, any history of bipolarity and investigation of any underlying neurological medical conditions, particularly in the elderly. Unrecognised comorbid medical or psychiatric conditions, such as anxiety and undisclosed substance misuse often contribute to non-response (Papakostas et al., 2012). Re-evaluation of medical comorbidity may require appropriate investigations or referral. Review of psychosocial and personality issues may reveal significant perpetuating factors that have not been adequately addressed (such as issues pertaining to compensation following injury at work). Psychological treatment may therefore have been inadequate and should be reviewed. A review of pharmacological treatment should concentrate on assessing adherence, tolerability and adequacy of dosage. Assessing the ongoing morbidity and associated disability assists in monitoring suicide risk, which may worsen with failure to respond, and will also guide decisions about continuing outpatient management or admitting the patient to hospital. Referral for a second opinion can be a valuable step in reassessing management.
Comorbidity and undiagnosed bipolar disorder
It is unlikely that core features of major depression, such as depressed mood, anhedonia and feelings of hopelessness, occur in isolation from symptoms of anxiety, substance misuse, personality, adjustment or eating disorders; in fact, a large proportion (estimated 50–70%) of patients with depression report anxiety symptoms (Fava et al., 2004, 2008; Kessler et al., 1996), which is associated with reduced efficacy of antidepressant treatment (Papakostas et al., 2012).
These commonly co-occurring symptoms and comorbidities complicate diagnosis and treatment, and generally contribute to a poorer prognosis (Coryell et al., 1995; Fiedorowicz et al., 2011; Newton-Howes et al., 2006; Rief et al., 2000). Therefore, re-evaluating the diagnosis following partial or non-response to antidepressant treatment is crucial, and it is important to consider the possibility of complex mood disorders in which depression may not be the primary or sole diagnosis.
Bipolar depression is another possibility that warrants consideration when diagnosing and treating depression, given that at least 10% of patients initially diagnosed with unipolar depression will subsequently experience hypomania/mania in the course of their illness, resulting in a revision of diagnosis to bipolar disorder and the modification of therapy – usually involving the addition of a mood stabiliser (Coryell et al., 1995; Fiedorowicz et al., 2011). There is also growing evidence to suggest that mixed symptoms commonly occur in a subset of patients with depression and these contribute to poor treatment response (Angst et al., 2010; Malhi, 2013; Malhi et al., 2014).
Paradigm shift
If the patient remains inadequately responsive despite having explored several reasonable treatment options, it is important to consider further individual patient factors, and unique illness characteristics that may be contributing to treatment resistance. In addition, it may be necessary to give consideration to alternative treatment options, outside of the standard treatment algorithm and seek a second opinion. Furthermore, it is important to bear in mind that the patient may have developed new symptoms, the depression may simply be more complex than originally thought or there may be social/lifestyle factors that are significant contributing factors. For example, since the commencement of the initial antidepressant, the patient may have experienced a change in their employment or lost their role altogether resulting in additional adjustment stress and complicating their care further. This would most likely have a negative impact on their relationships and social circumstances. In this instance, a comprehensive reassessment is needed. It is possible that the original diagnosis is incorrect or the formulation has been inadequate in capturing relevant factors. Returning to the 5P+ model and reassessing the patient’s problems is in the best interests of the patient and may open up new possibilities for treatment (see: Figure 4).
If it has been established that the original diagnosis and formulation appears to be correct and the patient’s symptoms are non-responsive to the previously suggested treatment options, novel treatment options may be warranted.
Continuing treatment
This may be an option if reassessment suggests that the duration of treatment has been inadequate, and frequent changes in medication have not allowed adequate time for effective treatment. For the most part, however, waiting before changing management unnecessarily risks delaying potential recovery. Most patients who are eventually going to respond to antidepressant treatment will show improvement within three weeks at a sufficient dose of medication (Posternak and Zimmerman, 2005; Tadić et al., 2010). If there is no improvement after three weeks, then an alternative strategy should be considered, particularly when the symptoms and impairment are severe. The involvement of a specialist (where available) is recommended and a second opinion should be sought.
Increasing the dose
Increasing the dose of antidepressant beyond the recommended maximum dose may increase efficacy for some but not all antidepressants. This option should be considered if the patient has had a partial response. This strategy is unlikely to be of benefit if there has been no response whatsoever. The evidence for the benefit of high dose treatment is sparse with few randomised studies, though STAR*D did show some differential efficacy with an increase in the dose of SSRIs. However, a systematic review (Adli et al., 2005) was supportive of high dose treatment with tricyclics and tranylcypromine, but not SSRIs, suggesting they have a relatively flat dose-response curve in the treatment of depression.There was insufficient evidence for high dose treatment with SNRIs to draw any clear conclusions, but serum levels of antidepressants, if available, may help guide dosage, and this is particularly relevant for tricyclics. In this context, pharmacogenetic approaches to guide dosing may be available in the near future (Singh et al., 2014).
Before altering any treatment it is important to allow a trial of appropriate duration, usually three weeks, at adequate dosage (Licht and Qvitzau, 2002; Ruhé et al., 2006a). It is noteworthy that in practice high dose medication can often serve as a proxy marker of non-response. Many antidepressants have a relatively narrow therapeutic range in which the agent is considered effective and safe, and research shows that increasing the dose of these medications does not always increase effectiveness but may assist some individuals (Ruhé et al., 2006a). However, antidepressants, such as venlafaxine and TCAs (other than nortriptyline), have very broad dose ranges with up to a ten-fold increase in oral dosage, e.g., venlafaxine can be safely administered at effective doses from 37.5 mg to 375 mg (Debonnel et al., 2007). Clinical monitoring at high doses is especially important as side effects and therapy discontinuations usually increase with dosage, and it is prudent to seek a second opinion and institute QTc monitoring if an increase in dose above the recommended dose range is planned.
It is difficult to be prescriptive regarding duration of medication during any particular phase of treatment because both clinical improvement and lack of response are susceptible to many factors that can alter outcome. Therefore, duration recommendations are only a rough guide. If an adequate 9 dose has been administered for three weeks and with no response then switch medications, but if a partial response occurs, continue the current medication for a further week (at the same dose; but a future dose increase can be considered if remission is not achieved). If after four weeks of treatment there is no significant improvement, alternative strategies should be implemented.
Switching antidepressants
Changing antidepressant medication and using an antidepressant from a different class is a common strategy in managing partial or non-response. This raises two considerations: 1) whether the decision to change medication is an effective strategy, and 2) when changing antidepressants, does it matter which antidepressant is prescribed?
With respect to the first question, a systematic review of three randomised controlled studies that examined switching antidepressant compared to continuation of the same antidepressant failed to show any overall advantage for switching (Bschor and Baethge, 2010). The three switching strategies studied were from fluoxetine to mianserin, from nortriptyline to fluoxetine and from venlafaxine to fluoxetine. However, clinical experience would suggest that switching is a helpful strategy in many cases and indeed there are few alternatives once an antidepressant trial has failed other than employing a case by case personalised approach.
The evidence to guide the choice of a second antidepressant agent is also inconclusive (Nierenberg et al., 2007). The STAR*D study, an open naturalistic study, showed equivalent remission rates regardless of whether the patient switched from citalopram to bupropion, sertraline or venlafaxine (Rush et al., 2006). There is randomised controlled evidence for improved outcomes with switching from an SSRI to venlafaxine and mirtazapine, and from a tricyclic to an irreversible MAOI, but the evidence for switching from an SSRI to a TCA is inadequate even though clinical experience and expert opinion supports this strategy particularly in more severe depression (Nierenberg et al., 2007).
Once a decision to switch antidepressant treatment has been made, there are two main considerations: First, which antidepressant to trial next, and second, which strategy to employ when switching. RCTs have found that switching to a different antidepressant improves response and remission rates when switching for reasons of either non-response (Nakajima et al., 2011) or intolerability. Switching within a class (e.g., from one SSRI to another) is no less effective when compared to switching out of class (e.g., from an SSRI to a non-SSRI) (Ruhé et al., 2006b; Rush, 2007; Souery et al., 2011). If treatment has been curtailed (e.g., medication has only been taken for a few days) because of side effects, then switching within a class to another agent may be a worthwhile option.
In primary care, the majority of patients are prescribed an SSRI antidepressant first-line. Patients who are intolerant to the initial SSRI often benefit from a second SSRI. It is recommended that patients use a lower dose of the second SSRI initially, and in some instances, it may be necessary to taper the first SSRI for a longer period (e.g., switching from citalopram to escitalopram can be immediate, but switching from fluoxetine to another antidepressant requires a wash-out period of at least a week before commencing the second agent at a lower dose). However, if the patient was non-responsive to the initial SSRI, switching out of class to a SNRI (i.e., venlafaxine), NDRI or TCA, is recommended (Ruhé et al., 2006b; Rush et al., 2006). Switching to a TCA carries the benefit of potentially higher response rates but is disadvantaged by the greater likelihood of side effects. If a patient begins antidepressant treatment with a TCA, for example as an inpatient, switching to another TCA has the advantage that a switch can occur without a wash-out period.
In most instances SSRIs are the starting point for antidepressant treatment, and therefore the majority of research has examined switching from SSRIs to another antidepressant. Thus, there is a lack of evidence for other switching combinations but most sequences have been trialled and there is ample empirical evidence for the majority. Of note, if side effects have contributed to poor response then switching to agents that are better tolerated is a useful option.
Switching strategies
There are three main strategies that should be considered when deciding the best way to switch medications: (i) overlap, (ii) taper or stop/start; and (iii) washout. The most common clinical strategy utilised is to introduce and withdraw medication seamlessly in order to minimise any un-medicated interim period and limit the possibility of serotonin syndrome. In practice, the choice of switching strategy depends on the degree of response achieved from the initial antidepressant trial (see: Figure 10).
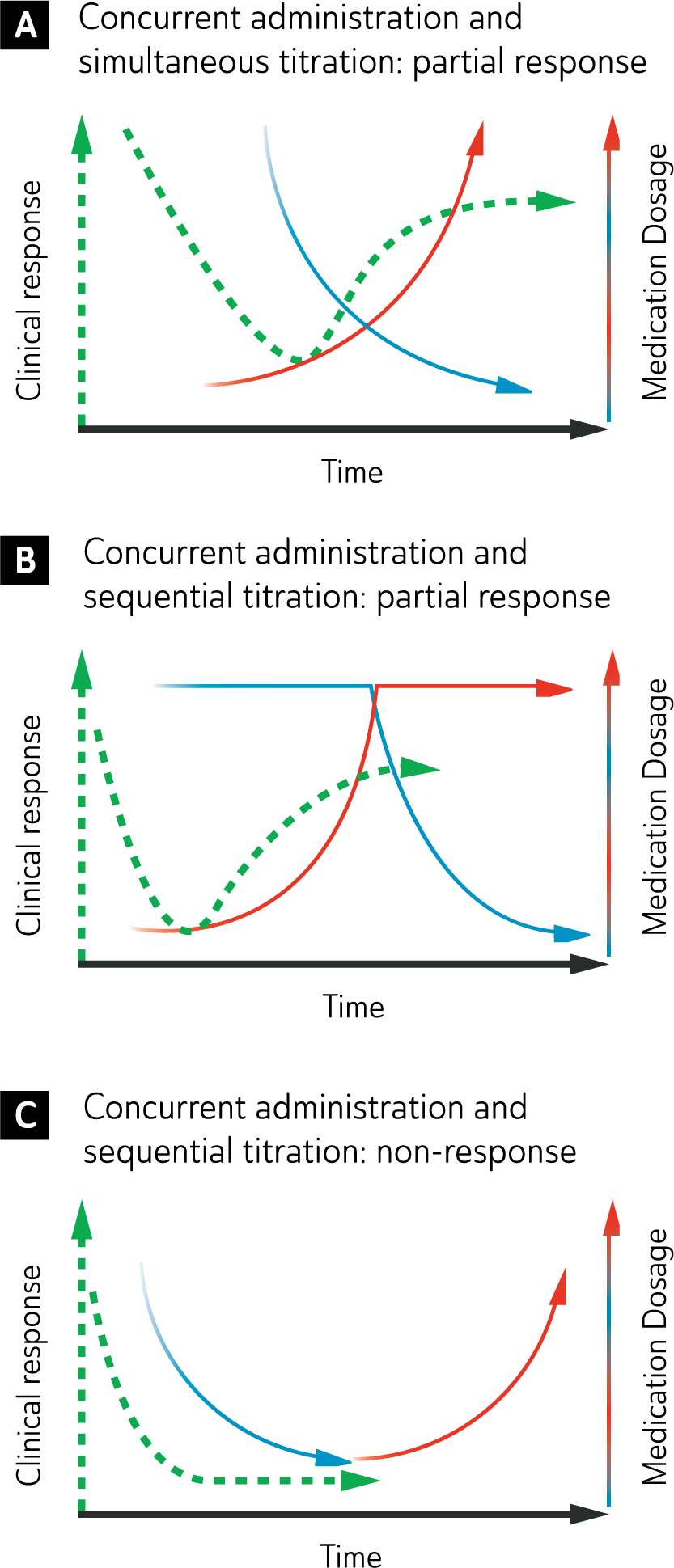
Strategies for switching antidepressant medication.
The three strategies for switching antidepressant medication are:
(A). Concurrent administration and simultaneous titration: Changes in dose of both medications are implemented simultaneously, thus, overlapping medications. Although this option offers the advantage that the individual is always medicated and switching is achieved quickly, there is an increased likelihood of interactions and side effects because the medications are administered jointly.
(B). Concurrent administration and sequential titration: Dose changes are only implemented in one medication at a time. The initial medication is continued at full dose while gradually commencing the new medication. Begin tapering the medication that is being substituted once optimal dose has been met on the new medication.
This strategy is preferred for partial response because the cross-over is important to help retain any benefit achieved from the initial medication thus far. Similar to concurrent switch, there is an increased risk of interactions and side effects because the medications overlap. However, the cause of iatrogenic side effects is more easily identified because only one medication is modified at a time.
(C). Separate administration and sequential titration: Taper the initial medication and once this has been fully withdrawn, gradually introduce and titrate the new medication to optimal dosage. This option is the cleanest way of substituting one medication for another but takes much longer, especially if it also includes a washout period. This option also increases the chance of worsening because there is a considerable period when medication is at a sub-therapeutic dose. Therefore, this strategy is preferable for those who did not respond to the initial medication, because this switch can be done quickly without a washout period and has the advantage of not overlapping medications which increases the likelihood of side effects. However, switching to, or from, an MAOI requires a clear washout period of at least a week (and up to 5 weeks in the case of fluoxetine) to limit the risk of interactions.
Precision medicine
Familial patterns of response to psychotropics have long been observed (Propping and Kopun, 1973) and posited to be mediated by inter-individual genetic variability (O’Reilly et al., 1994). Genetic and dietary variability between ethnic groups can also result in wide pharmacokinetic variance (Silva, 2013). Yet most psychotropic agents have only been extensively studied in Caucasian populations (Ninnemann, 2012). Some have described this latter area as Ethno-psychopharmacology – ethnic variations influencing the effectiveness of psychotropics (Wong and Pi, 2012). This term has now been subsumed under the broader concept of pharmacogenetics – the study of how genetic factors influence efficacy, tolerability and safety of medications. It is hoped that genetics may enable more tailored psychotropic prescribing termed personalized medicine, and this concept is gaining momentum as the cost of genotyping has reduced (Insel, 2014). ‘Precision Medicine’ is the dominant umbrella term used to describe genetically guided prescribing (Collins and Varmus, 2015).
Genetic variations (polymorphisms) of metabolizing enzymes, drug transporters (such as those at the blood-brain-barrier), and target receptors coupled with nutrition factors (potentially inducing/inhibiting metabolism) are relevant in understanding both inter-ethnic (ethno-psychopharmacology) and inter-individual (precision/personalized medicine) variability of medication response (Ninnemann, 2012; Singh et al., 2014). Despite the rapidly diminishing cost of genetic testing, randomized controlled trials to firmly elucidate the clinical utility of routine genetically guided prescribing are needed (Singh et al., 2014). Nonetheless, this emerging genetic technology may be particularly relevant in prescribing optimally to ethnic groups other than Caucasians – such as Māori and Aboriginal and Torres Strait Islander populations. It may also reduce trial and error prescribing – an issue particularly relevant in mood disorders due to the relatively long time period between commencing a medication and discerning clinical efficacy.
HLA genotyping in patients of Asian background who may be treated with the mood stabilizer and anticonvulsant carbamazepine offer a good example of precision medicine in practice. Carbamazepine has an up to 75-fold risk of causing the potentially fatal skin reaction Stevens-Johnson syndrome and its related disease toxic epidermal necrolysis in patients of Asian ethnicity compared to Caucasians (Kulkantrakorn et al., 2012). It is now recommended such testing be carried out prior to prescribing carbamazepine (Chen et al., 2011). In years to come similar guidelines for lamotrigine are likely to emerge (Li et al., 2015). The relevance of this to Māori and Aboriginal and Torres Strait Islander populations is unclear as there is a lack of empirical data and pharmacogenetic studies are needed in these groups.
Some ethnic groups have higher rates of rapid metabolizer status at the hepatic cytochrome P450 enzyme system – potentially translating to higher doses of medication being needed (Cai et al., 2006; De Leon et al., 2006). Conversely, some ethnic groups may have higher rates of poor metabolizers and require lower doses to avoid toxicity (De Leon et al., 2006). Hepatic cytochrome P450 2D6 and 2C19 metabolizer status seem the most relevant for antidepressants, with dose adjustment guidelines stemming from earlier association studies (PharmGKB, 2015), But genetically guided versus unguided randomized controlled trials are needed to robustly elucidate the clinical utility of a priori genetic testing (Singh et al., 2014). Two such trials have been conducted (Singh, 2015; Winner, 2013) and should positive findings be replicated and have a clinically meaningful effect size, precision medicine in psychiatry will likely gain wider traction. At this stage – other than HLA testing of carbamazepine among patient of Asian ethnicity – precision medicine in psychiatry remains at the ‘early adopter’ stage (Thompson et al., 2015).
Augmentation (Level I)
Augmentation refers to the addition of another agent that exerts a synergistic antidepressant effect. There is evidence from randomised controlled trials for the effectiveness of lithium and some second generation antipsychotics as augmenting agents.
Lithium, a widely supported and used augmentation agent, is found to be more effective than placebo in augmentation of TCAs, SSRIs and other antidepressants (Bauer et al., 2000; Crossley and Bauer, 2007). It is recommended that lithium be administered once daily at an oral dose that achieves trough plasma levels (blood sample taken 12 hours after last dose of lithium) within the therapeutic range (0.5 mmol/L to 0.8 mmol/l) (Berghofer et al., 2006; Malhi et al., 2011). If there is no response to lithium within 7–10 days, alternative strategies should be considered. Care should be taken when discontinuing the use of lithium as research shows that abrupt withdrawal of lithium following antidepressant augmentation with lithium results in significantly higher relapse rates (Bschor et al., 1999). Predictors of likely response to lithium augmentation include recurrent major depression with more than 3 recurrences and a family history in first degree relatives of bipolar or unipolar depression (Sugawara et al., 2010).
Second generation antipsychotics are widely used as add-on agents for TRD in clinical practice. In addition to empirical evidence, research studies support this strategy (Papakostas et al., 2007a; Philip et al., 2008). Placebo-controlled studies have found that aripiprazole, olanzapine, quetiapine and risperidone can be effective as augmentation agents. These are generally administered at much lower doses than those recommended for schizophrenia and bipolar disorder (Berman et al., 2007; Marcus et al., 2008; Philip et al., 2008) but this practice is off-label and these agents are not formally indicated for augmentation. Furthermore, adverse effects of these agents must be closely monitored, as weight gain, potential metabolic syndrome and extrapyramidal side effects are of great concern, especially in the context of long-term therapy. Once a stable response with augmentation has been achieved the gradual withdrawal of the augmenting second generation antipsychotic (SGA) agent should be considered. In some instances this may precipitate relapse and therefore ongoing use may be necessary.
Thyroid hormone has also been used to augment the effects of antidepressants in the treatment of major depression, however, the results from clinical studies are inconsistent. Most studies have been of T3 (liothyronine) augmentation, while T4 (thyroxine) has been evaluated to a lesser extent. It is reasonable to extrapolate the findings of T3 studies to T4 augmentation because T4 is a precursor of T3, which is similar in action but more potent. Earlier studies that examined T3 augmentation of tricyclic antidepressants had mixed results (Lam et al., 2009), and systematic review of T3 augmentation of SSRIs (Cooper-Kazaz and Lerer, 2008) provides equivocal evidence for its efficacy. T3 is beneficial for depressed patients with subclinical hypothyroidism, but T3 is also used as an augmentation agent for depressed patients without hypothyroidism. Patients augmented with T3 should be monitored in the same way as patients with hypothyroidism, with TSH, free T4 and T3 levels measured regularly (Rosenthal et al., 2011). In practice, despite conflicting evidence, thyroid augmentation is well tolerated (Cooper-Kazaz and Lerer, 2008) and may be a low-risk augmentation option for some patients.
A number of novel augmentation strategies have been trialled with variable success. Placebo controlled studies of augmentation with buspirone or pindolol have been unconvincing and hence these are not recommended (Lam et al., 2009; Martiny et al., 2012; Portella et al., 2011). Two randomised placebo controlled trials of augmentation with the stimulant methylphenidate have also failed to demonstrate efficacy (Patkar et al., 2006; Ravindran et al., 2008). A Cochrane systematic review of a range of stimulants that have been used for augmentation (Candy et al., 2008) found that the quality of randomised trials was overall poor and that although there was evidence that stimulants (including modafinil) reduced symptoms of depression, this was usually short-lived, clinical benefit was unclear, and effects non-specific. At present there is insufficient evidence to recommend the routine use of psychostimulants as an augmentation strategy. Similarly, the evidence base for the routine use of the dopamine agonist pramipexol is currently too limited to advise routine use (Cusin et al., 2013). As the literature is still evolving (Corp et al., 2014) clinicians are advised to exhaustively trial more evidence established therapies first and look for replicated RCT studies before considering such augmenting agents.
Combining treatment
Combining antidepressants has been a commonly used augmentation strategy among clinicians (Horgan et al., 2007).
A systematic review of clinical trials which assessed the efficacy of antidepressant combinations for the management of major depressive disorders concluded that there was evidence of greater efficiency than a single antidepressant in obtaining improvement using several combinations (mirtazapine plus SSRI and tricyclic plus SSRI), however the small number of clinical trials (5 trials) and methodological restrictions, made firm conclusions difficult (Rocha et al., 2012). A study that randomised 665 depressed patients to treatment with escitalopram plus placebo, or the combinations of bupropion with escitalopram or venlafaxine with mirtazapine (Friedman et al., 2012) showed no benefit for either combination treatment over escitalopram monotherapy.
There is some research, however, to suggest that combinations of antidepressants can be more effective than monotherapy without compromising tolerance (Bech and Csillag, 2012; Blier et al., 2010). A double-blind randomised trial where patients were treated with combinations of mirtazapine with fluoxetine, venlafaxine or buproprion showed that these were more effective for depression than fluoxetine monotherapy and that they were well tolerated (Blier et al., 2010). A recent meta-analysis found that mirtazapine and tricyclic antidepressants in combination with SSRIs are better than SSRI monotherapy in achieving remission (Rocha et al., 2012). Additionally, of the studies that reported dropout rates and side effects, there was no difference between the combined and monotherapy groups. Similarly, a double-blind study that compared mirtazapine monotherapy and paroxetine monotherapy against a combination of both antidepressants reported that remission rates after 6 weeks were highest for the latter (Blier et al., 2009). However, not all research has supported the use of combinations and not all combinations are pharmacologically sound (Licht and Qvitzau, 2002). Long-term placebo-controlled comparator studies are needed to substantiate a recommendation of polypharmacy (Rush, 2010). When determining which antidepressants to combine, a rationale based on pharmacokinetic advantage or synergistic action, should inform the decision, in addition to past experience and knowledge. For example, venlafaxine and mirtazapine are pharmacologically synergistic or complementary because of their distinct receptor profiles (Malhi et al., 2008).
Controversy remains regarding this practice but many psychiatrists use antidepressant combinations in some of their depressed patients with positive reports (Dodd et al., 2005; Keks et al., 2007; Malhi et al., 2013a; Ng et al., 2006; Rush et al., 2011; Thase, 2013). Given this uncertainty and the potential for adverse interactions with medication combinations, patients should be well advised and their consent obtained before antidepressant combinations are employed.
Brain stimulation strategies to address poor response in acute major depressive disorder
Electroconvulsive therapy
Although ECT has been seen as a last resort in treatment resistant depression it can be regarded as a reasonably efficacious option to consider before esoteric medication combinations. The response rate to unilateral ECT in patients with non-psychotic depression who have failed one or more medication trials has been shown to be as high as 48% (Prudic et al., 1996). A more recent meta-analysis (Heijnen et al., 2010) found remission rates with ECT of 48% for medication refractory patients compared to 64.9% for patients who had not received pharmacological treatment prior to ECT. Heijnen et al. (2010) made the point that most studies examined response rates to unilateral ECT at a time when doses for unilateral were low (2–2.5 x seizure threshold) relative to current practice (5–6 x threshold) and response rates may therefore be under-estimated. These remission rates can be compared with those demonstrated by the STAR*D study which showed that the remission rates for the first, second, third and fourth treatment steps were 36.8%, 30.6%, 13.7% and 13% respectively (Rush et al., 2006). For antidepressant monotherapy strategies, switching to a third antidepressant after two consecutive failures produced a low remission rate of less than 20% (Fava et al., 2006).

Footnote: If inexperienced in using medication doses above the recommended maximum, then consider seeking a second opinion. If symptoms have not significantly improved after a few weeks of treatment, re-evaluate the diagnosis.
Also refer to Recommendation Box 6.
These data suggest that ECT may be a more effective treatment than further pharmacological strategies in many patients who have failed to respond to one or more medication trials. It should therefore be considered as an option for treatment non-response early in the treatment algorithm and not left until there have been multiple treatment failures. The decision to recommend ECT after the first or subsequent medication failures needs to be made after considering the issues of ECT side-effects, safety and patient acceptance or preference. These need to be balanced against the potential adverse consequences of delaying recovery by persisting with less effective pharmacological strategies.
Repetitive transcranial magnetic stimulation
All substantive evaluations of the efficacy of rTMS have been conducted in patients with some degree of treatment resistance with many of the trials including patients who have failed to respond to 2 or more antidepressant medication trials where the likelihood of further medication response is quite low. In this context, rTMS has established efficacy, as confirmed by multiple meta-analyses (for example Schutter, 2009; Slotema et al., 2010). Therefore, it is a sensible treatment option in patients who have failed to respond to initial antidepressant therapy, or who have significant problems with tolerating medication treatment.
Treatment options in major depressive disorder maintenance phase
The clinical aim during maintenance phase is to prevent relapse/recurrence, and to decrease future risk of relapse/recurrence by increasing resilience at biological, psychological, social and lifestyle levels. A number of psychosocial variables and lifestyle factors (e.g., accommodation, social support, employment) are likely to influence the risk of relapse or illness recurrence. These include substance use, the experience of significant life events or ongoing psychosocial stressors, the presence of comorbid medical or psychiatric illnesses and the characteristics of the depressive illness itself (McGrath et al., 2006; Rucci et al., 2011). For example, more severe depression and a history of more frequent or longer duration episodes are associated with higher relapse rates. Particular attention should be paid to minimising or ameliorating residual symptoms, which are strongly linked to increased relapse rates (Kessing and Andersen, 2005; Ramana et al., 1995).
Monitoring and adherence
The therapeutic relationship established during initial treatment and further developed during the continuation phase is critical to optimal maintenance phase treatment. During this time, patients should be monitored for early signs of relapse. Sessions should include discussion of any side effects, and problem solving of stressful life events. Maintenance of a strong therapeutic relationship remains critical for continued engagement.

Footnote: The option of using ECT before or after rTMS is a matter for clinical judgement; the evidence supporting the effectiveness of ECT is stronger but the side effect profile is better for TMS.
Also refer to Recommendation Box 5.
Many patients are likely to be reluctant to continue medication treatment after the initial resolution of a depressive episode. The clinician should encourage open discussion about the benefits and burdens of maintenance pharmacotherapy versus the probability and risks of relapse.
Monitoring for early signs of recurrence is critical and a plan to do this should be formulated early in the maintenance phase. The patient should be helped to identify symptoms that have emerged early in prior depressive episodes and thought given to whether these are likely to be identified by the patient or whether other individuals, such as family members, could be engaged in a process of monitoring for these. A clear plan should be outlined as to how the patient should respond if they notice the emergence of these early signs of relapse.
There is evidence that an active depression relapse prevention plan can improve antidepressant medication adherence, and reduce depressive symptoms but not necessarily reduce overall recurrence rates (Katon et al., 2001) (Level II). Relapse rates are greatest immediately following remission and diminish with time (Belsher and Costello, 1988). The risk of relapse during the continuation phase of treatment is relatively high and is increased by a variety of factors (see: Table 18).
Factors increasing the risk of acute relapse in depression.

Psychological treatments
Depression is predominantly a recurrent condition, and existing literature has paid insufficient attention to maintenance and relapse prevention relative to acute symptom relief (Mulder, 2015). Evidence-based psychotherapies share a skill-development focus, and it is important to develop an understanding of how psychotherapies might interact with lifetime vulnerability to depressive episodes.
A 2015 meta-analysis and meta-regression (Biesheuvel-Leliefeld et al., 2015) found preventative psychological interventions across 25 identified studies were superior to treatment as usual (NNT = 5), and to a lesser extent antidepressant medication (NNT = 13).
Two psychotherapies have Level I evidence for effectiveness in preventing depressive relapse and/or recurrence. Cognitive behavioural therapy (CBT) has been found to reduce relapse and recurrence rates relative to treatment as usual in a meta-analysis limited to (n = 9) well controlled trials (Lynch, et al. 2010). In this meta-analysis, the pooled odds ratio for studies in which patients were on antidepressant treatment was 0.52, and for patients not on ADM was 0.45. A highly cited study compared relapse rates amongst three groups – responders to acute cognitive therapy who had therapy withdrawn, responders to acute medication with continuation medication, and responders to acute medication with placebo withdrawal (Hollon, et al., 2005). Those who had received cognitive therapy were less likely to relapse during continuation than those who had received medications (30.8% vs 76.2%), and were no more likely to relapse than patients who were maintained on continuation medication (30.8% vs 47.2%). There is some evidence that CBT’s maintenance effects are more pronounced amongst people who have had several episodes of depression (e.g., Bockting et al., 2005).
Mindfulness-based cognitive therapy (MBCT), designed to reduce relapse or recurrence of depression, has been found effective over control in a meta-analysis (six included studies) with a mean risk ratio of 0.66 (Piet & Hougaard, 2011). The relapse rate for MBCT participants (n = 200) was 38%, compared to 58% for controls (n = 208). A second meta-analysis (four included studies, patients with three or more prior depressive episodes), also demonstrated the effectiveness of MBCT, with an aggregate odds ratio of 0.36 versus treatment as usual (Chiesa & Serretti, 2011). As noted above in the context of CBT, there is some evidence that MBCT is more effective amongst those with a greater number of prior episodes: planned subgroup analyses by Piet and Hougaard found relative risk reduction was 43% for participants with three or more previous episodes, and not significant for those with only two.
There is limited research into interpersonal therapy (IPT) as prophylactic against depressive recurrences, but initial studies have been positive (Beshai, et al., 2011). Some, but not all studies have found IPT to match the prophylactic effect of antidepressant medication (Lampe et al., 2013).
Pharmacological treatments
Antidepressant medication in maintenance phase
Ongoing antidepressant treatment following an initial treatment response has been shown to be of significant value in the prevention of relapse and/or recurrence of depression, with better medication adherence associated with lower recurrence rates (Akerblad et al., 2006) (Level I). A significant reduction in relapse or recurrence rates has been demonstrated for several classes of antidepressant medications including TCAs and SSRIs (Hirschfeld, 2001). Nearly all studies were limited to 12 months duration or less. There is some evidence of efficacy for up to three years in patients with recurrent depression (Kupfer et al., 1992) but the numbers studied are very small. The relapse rate between antidepressants and placebo lessen significantly after six months post randomisation so the evidence for continuing them beyond 12 months remains weak. Even with ongoing maintenance treatment recurrences can occur (Byrne and Rothschild, 1998).
Lithium in maintenance phase
In the majority of circumstances, patients should continue receiving the antidepressant medication that they responded to during the acute phase. If this was administered as an adjunct to lithium, then this too should be continued throughout the continuation and maintenance phases. However, in major depression, lithium monotherapy is an effective alternative maintenance treatment option (Cipriani et al., 2006) and may be considered if antidepressants cannot be tolerated (e.g., due to sexual dysfunction). Multiple trials over time have established the effectiveness of lithium monotherapy in the prevention of depressive relapse as confirmed by meta-analyses (Souza and Goodwin, 1991), with recommended levels typically at the lower end of the therapeutic range (Malhi et al., 2011; Schou, 1989). However, long-term administration of lithium can be associated with serious side effects, such as renal and thyroid dysfunction (Van Gerven and Boer, 2006) which may complicate long-term treatment (Malhi et al., 2012d). Therefore, lithium levels must be monitored regularly.
Combined psychological and pharmacological treatments
Combining psychological and pharmacological treatments has been shown to improve clinical outcome and significantly decrease relapse and recurrence rates (Beshai et al., 2011), which may be partially due to the effect of psychological treatments on adherence (Pampallona et al., 2004). More specifically, research trials have found that maintenance CBT in combination with antidepressant medication was superior in preventing relapse to maintenance antidepressant medication alone (Beshai et al., 2011; Paykel et al., 1999), and the observed advantage persisted several years later (Paykel et al., 2005). IPT in combination with pharmacotherapy is more effective than either treatment alone (Cuijpers et al., 2011b), but more research is needed on IPT in combination, as well as other psychological interventions.
Electroconvulsive therapy treatment in maintenance phase
Continuation and maintenance phase ECT is sometimes utilised in patients who have achieved acute clinical response to this treatment. However, the evidence base for this approach is relatively limited and comprises predominately uncontrolled case series. A RCT of maintenance ECT compared with continuation pharmacotherapy (lithium plus nortriptyline) showed no difference between the groups, both having approximately 46% of patients with continued remission after 6 months. This is a relatively high relapse rate for either therapy given that approximately 40% of the patients who remitted with ECT were then excluded within one week and before randomisation, 20% because of relapse (Kellner et al., 2006). Further, in a retrospective chart review based case-control analysis, relapse rates with maintenance ECT over a number of years were shown to be substantially lower than that with antidepressant medication alone in a similar group of patients who had responded to acute phase ECT (Gagne et al., 2000) (Level III). One small randomised trial in elderly patients with psychotic depression compared maintenance treatment with nortriptyline to nortriptyline plus ECT. They found that relapse occurred sooner in the medication alone treatment group compared with those who were treated with combination pharmacotherapy and ECT (Navarro et al., 2008) (Level II).

Notes: Once remission has been achieved, antidepressant medication confers preventive benefit.
The choice of antidepressant dose is also determined by additional factors such as prior illness severity and response to treatment, comorbid disorders and medication tolerance.
Footnote: a. This is particularly important if a recurrent pattern of illness has been established.
Duration and discontinuation of maintenance treatment
Limited research has explored optimal duration of medication treatment in the maintenance phase. One clinical trial has shown that following maintenance therapy the relapse rate when transferring from active treatment to placebo is lower if maintenance therapy has continued for a longer period of time (Reimherr et al., 1998). Recurrence rates when patients remain on medication past one year are likely to be lower over subsequent years (Kornstein, 2008). A second trial demonstrated that antidepressant medications continue to have relapse prevention benefits for up to 5 years following commencement of treatment (Kupfer et al., 1992) (Level II). Once remission has been achieved, expert consensus recommends continuation of antidepressant treatment at the therapeutic dose that was effective during acute treatment for at least 1 year following an initial episode and at least 3 years for recurrent episodes (Bauer, 2007) and episodes which included psychotic features (Viguera et al., 1998) (CBR). Maintaining the therapeutic dose that was effective during acute treatment is important because this dosage is generally more efficacious in preventing relapse or recurrence than lower ‘maintenance doses’ (Dawson et al., 1998; Reynolds et al., 1999; Thase, 2006).
When patients have received maintenance antidepressant medication, cessation of medication should occur slowly with a taper over an extended period of time because withdrawal symptoms may be experienced. Discontinuation symptoms may emerge following the cessation of all classes of antidepressants. The risk of discontinuation symptoms is greatest with higher doses of antidepressants and longer duration of treatment, however symptoms are usually transient and mild, and resolve with antidepressant reinstatement (Perahia et al., 2005). Amongst the antidepressants, discontinuation symptoms are more likely with venlafaxine (Haddad and Anderson, 2007; Tint et al., 2008) and short-acting SSRIs (especially paroxetine), but less likely with fluoxetine (Michelson et al., 2000; Schatzberg et al., 2006). Abrupt cessation of TCAs may cause cholinergic-rebound phenomena (flu-like illness, myalgia and abdominal cramps). Therefore, a slow taper of medication over a longer period of time is preferred in order to reduce the likelihood of discontinuation symptoms but may not impact the likelihood of early relapse (Viguera et al., 1998) (Level II).
Further, when patients are withdrawn from medication a clear plan for monitoring for potential symptom re-emergence should be put in place. During medication withdrawal, more frequent visits should be scheduled to ensure adequate monitoring and detection of early signs of depression recurrence. Patients should be informed as to the potential symptoms that might emerge. Family members or carers may also need to be engaged in this process and, it would generally be prudent to avoid tapering off medications when patients are concurrently negotiating life events.
Clinical management of bipolar disorder
Compared with major depressive disorder, bipolar disorder is more complex and difficult to treat and polypharmacy is more common. There is no evidence for psychological monotherapy and adjunctive psychological therapies are primarily used during the maintenance phase of treatment.
Management of mania
Manic symptoms can occur within depression as mixed features, and with increasing severity from hypomania and mania through to mania with psychosis.
Severe mania
Patients suffering from acute mania often have moderated insight – especially when experiencing psychotic symptoms. In these instances involuntary treatment may be required and this should be given with as much empathy and information as possible, while recognising that this may not be fully accepted or assimilated at the time. The purpose of treatment is to protect the patient and others from the adverse effects of the illness while actively relieving distress and treating the core symptoms of the disorder, which comprise acute symptoms of mania, behavioural disturbance and cognitive disturbance (See Tables 2 and 8 and Figure 11).

Management of Mania.
Acute mania is a medical emergency, and often necessitates use of mental health legislation. Care should be provided in a low stimulus environment with support from health professionals. Treatment usually requires short-term use of a combination of benzodiazepines and antipsychotics, but delirious mania and mania with catatonic features (not attributable to an organic cause) also respond particularly well (and promptly) to ECT. Therefore ECT should not be unduly delayed, but at the same time adequate consideration needs to be given to the restraint needed for anaesthesia (England, et al. 2011; Grenier et al. 2011; Jacobowski et al. 2013; Malhi et al. 2012c).
Hypomania
At the other end of the spectrum manic symptoms and signs often present in less severe form and for shorter periods of time. Hypomania can be difficult to distinguish from normal experiences and behaviour and patients are often unaware that their thoughts, feelings and behaviour are in any way unusual. Consequently they may be reluctant to accept that anything is wrong or that they need treatment. Indeed, the hypomanic state is one of the primary reasons for patient ambivalence about the diagnosis of bipolar disorder and prophylactic treatment (Lobban et al., 2012). By definition hypomania is self-limiting and often resolves with only modest use of medications and psychosocial interventions, but usually some adjustments to treatment are necessary and these should follow the general principles of managing mania. (APA, 2013; Walsh et al. 2015; Woo et al., 2015).
Mania
It is crucial to taper and cease any agents with mood-elevating properties (e.g. antidepressants, stimulants) and institute general measures such as reducing stimulation, lowering activity level, delaying the individual from making important decisions and maintaining a structured routine. To achieve this safely, it may be sufficient to develop a therapeutic alliance and offer a diagnosis, but often admission to a suitable containment facility is required (Berk et al., 2004). Individuals suffering a manic episode can be persuasive, push limits and rationalise their impulsive or reckless behaviour to make a case for not being admitted. Medications are essential to address the biological symptoms of mania, and counter heightened arousal (Geddes and Miklowitz, 2013; Malhi et al., 2009d, 2012a; Nivoli et al., 2012; Vieta et al., 2008b; Yildiz et al., 2011). But equally, inclusion and informative counselling of family members/carers is an important consideration from the outset and their involvement will be particularly important during the patient’s subsequent care (Berk et al., 2011).
Pharmacotherapy
The pharmacotherapy of mania involves treatment with anti-manic agents. The fundamental goals of such medications are to reduce arousal, agitation and aggression, and begin the process of treating core manic symptoms including behavioural disturbances and psychosis, if it is present. Therefore, a combination of medications is often necessary. See: Figure 11, Tables 19 , and 20 for a step-by-step approach to medication choices for the treatment of acute mania. The choice of medications and their route of administration is determined by the availability of medications, the required speed of onset of action, the degree of cooperation by the patient with prescribed treatment, and the proposed future choice of medications (Cipriani et al., 2011) (see: Table 19 ).
Medications used in the management of mania.
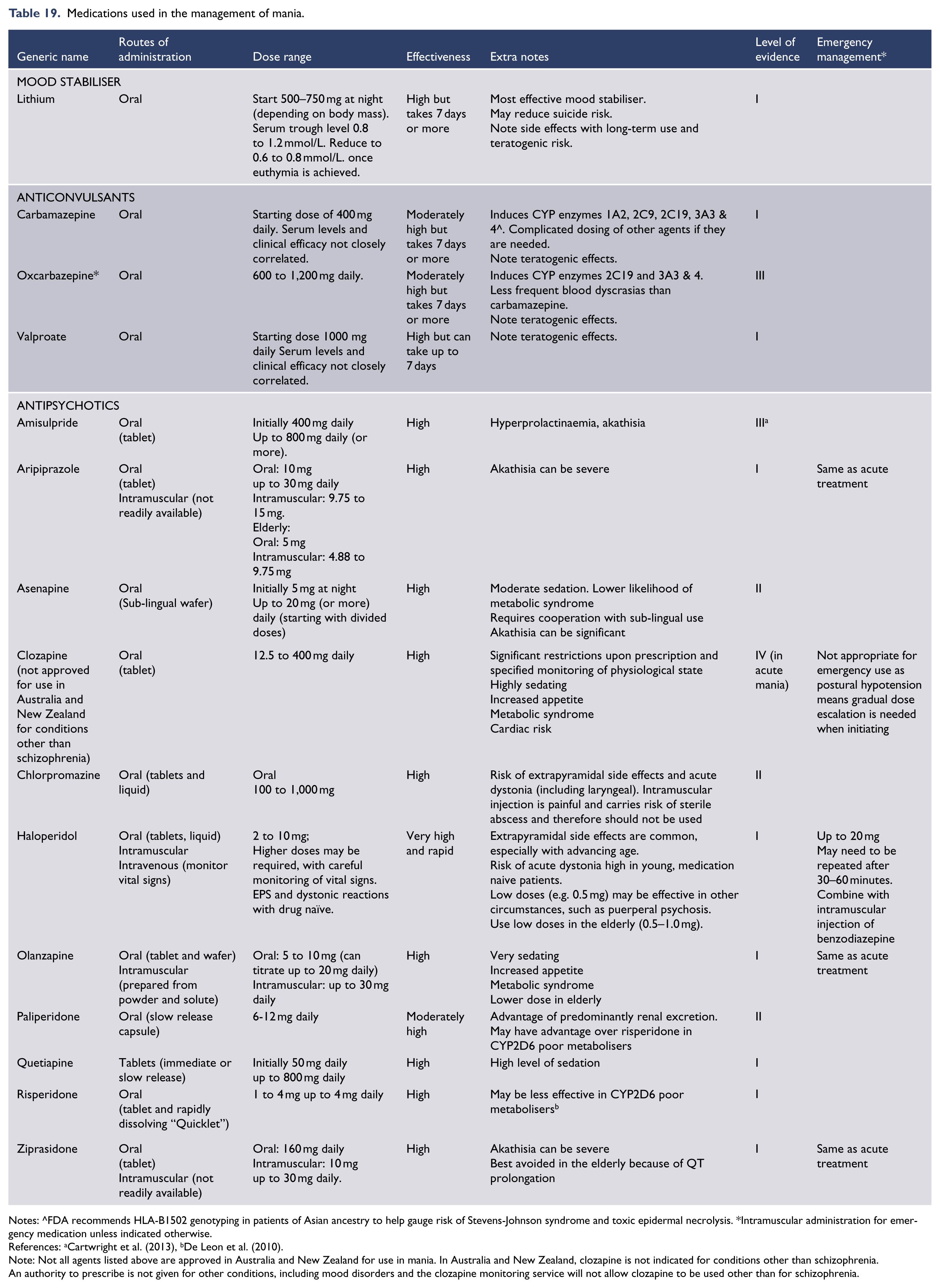
Notes: ^FDA recommends HLA-B1502 genotyping in patients of Asian ancestry to help gauge risk of Stevens-Johnson syndrome and toxic epidermal necrolysis. *Intramuscular administration for emergency medication unless indicated otherwise.
References: aCartwright et al. (2013), bDe Leon et al. (2010).
Note: Not all agents listed above are approved in Australia and New Zealand for use in mania. In Australia and New Zealand, clozapine is not indicated for conditions other than schizophrenia. An authority to prescribe is not given for other conditions, including mood disorders and the clozapine monitoring service will not allow clozapine to be used other than for schizophrenia.
Adjunctive treatments.
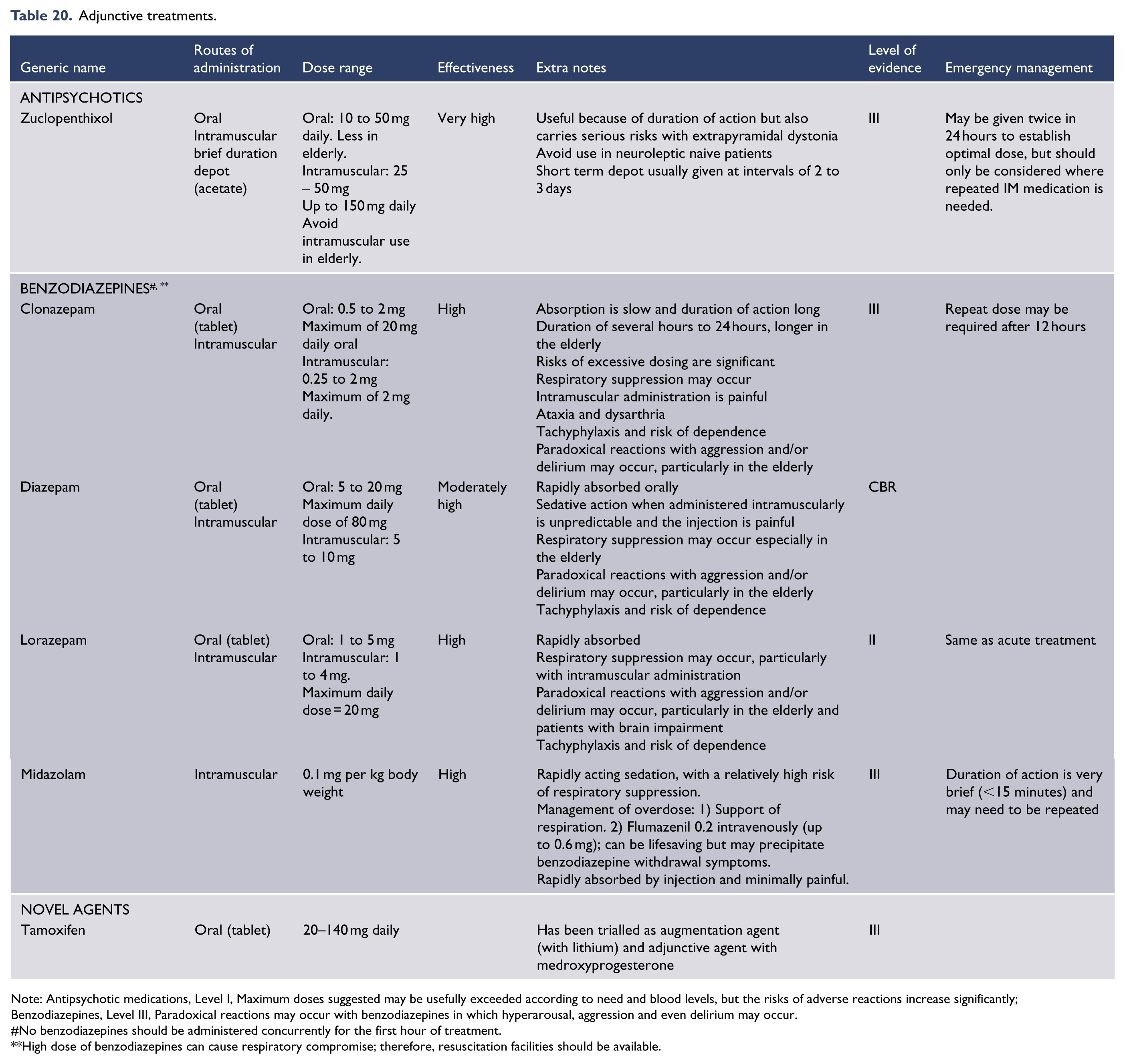
Note: Antipsychotic medications, Level I, Maximum doses suggested may be usefully exceeded according to need and blood levels, but the risks of adverse reactions increase significantly;
Benzodiazepines, Level III, Paradoxical reactions may occur with benzodiazepines in which hyperarousal, aggression and even delirium may occur.
No benzodiazepines should be administered concurrently for the first hour of treatment.
High dose of benzodiazepines can cause respiratory compromise; therefore, resuscitation facilities should be available.
Brain stimulation methods
There is a limited but consistent evidence base supporting the use of ECT for the treatment of acute mania. A recent systematic review (Versiani et al., 2011) identified 28 studies investigating ECT as a treatment for acute mania. Although identifying methodological shortcomings in the studies reviewed, the systematic review concluded that ECT was an effective treatment for mania, especially in more severe or refractory cases. A recent literature review (Loo et al., 2011) concluded that in acute mania, ECT has clinically meaningful efficacy, including in patients who have failed to respond to medication. However, there is insufficient evidence to support one electrode placement over another in treating mania and methodological problems such as inadequate dosing for unilateral ECT hamper meaningful comparisons between unilateral and bilateral placements.
Although preliminary studies have explored the use of rTMS in mania, especially the use of high frequency stimulation applied to the right prefrontal cortex, there is insufficient evidence to support its clinical use at this stage.
Psychological interventions
The cognitive, emotional and behavioural dysregulation of acute mania is a barrier to the skill-development targets of the structured psychological interventions (Geddes and Miklowitz, 2013). A handful of preliminary studies have investigated the efficacy of psychological interventions for inpatient treatment of acute mania, providing some suggestion that chronobiological strategies (especially those that can be imposed, like dark therapy) warrant further investigation (Crowe and Porter, 2014).
There is strong clinical consensus about the benefits of engaging with people even in acute mania, making all reasonable efforts to work in a genuinely collaborative and normalising way (Cooke et al., 2010). Unstructured supportive psychotherapy (which may include psychoeducation about bipolar disorder) is likely to be useful for the patient and their family during acute mania. Acute settings also provide an opportunity to engage individuals for future psychological therapy, especially after the most severe elements of mania begin to subside and the patient is starting to reflect on the consequences of the episode. Supportive psychotherapy may be particularly indicated if involuntary treatment has been required.
Patients with mania test boundaries and cause interpersonal conflicts within a treating team. Clear communication and limits on behaviour are essential psychological strategies underpinning all management (Janowsky et al., 1970). While a lack of insight and involuntary treatment early in the illness makes establishing a therapeutic relationship difficult, the capacity of the therapist to engage the patient (and their family) in a therapeutic alliance and introduce psychoeducation about the disorder and its management is of fundamental importance to long-term success. Mania is often provoked and exacerbated by emotional stressors. Relief from these stressors and the adoption of more effective coping strategies, will assist in recovery.
Supportive psychotherapy provides authentic concern for the patient’s welfare, basic information about the illness and its treatment, and conveys hope for a productive recovery: Importantly, this mode of counselling is less likely to overstimulate the patient.
Simple verbal interventions to reduce conflict and enhance meaningful relationships with significant others (such as family members) can be productive.
Treatment non-response
Assessing treatment non-response
Review the diagnosis
Often, the features of mania are clearly persistent, but may also change in form and/or relative intensity. The most obvious features may resolve, but a manic process may continue with less obvious features, such as ‘racing thoughts’, increased energy without obvious increased activity (including speech), expansive or negative thought content or rage that is not expressed, increased libido without sexual behaviour, and reduced need for sleep, which is not obvious to an observer. Psychotic symptoms may be easily missed, particularly if the patient has sufficient insight to know that their thoughts are considered an indication of illness. Therefore it is important to assess the patient’s symptoms through self-report, observation and corroboration from family in order to understand the patient’s entire manic process. Observation by health professionals will be an essential source of information, and an overall pattern of subtle core manic signs and symptoms may only be apparent when assessed continuously over hours or days, and even weeks. Further, the nature of non-response might extend beyond the persistence of core manic symptoms to comorbidity, such as substance abuse and lack of compliance with treatment. Clarification of the source of such further difficulties in management requires careful clinical assessment (Fountoulakis et al., 2012a; Grunze et al., 2013; Sienaert et al., 2013) (Level III).
It is also important to consider ‘rapid cycling’ bipolar disorder (see: Rapid cycling bipolar disorder) or ‘mixed’ features, with implications of potentially less satisfactory response to treatment (Cirillo et al., 2012; Czepielewski et al., 2013; Farren et al., 2012; Iacovides and Andreoulakis, 2011; Karaahmet et al., 2013; Krishnan, 2005; McElroy et al., 2006; Skirrow et al., 2012) (Level IV).
Review investigations
Routine investigations should be repeated such as full blood count, electrolyte levels, renal, liver, and thyroid function tests, along with (if indicated) neuro-imaging of the brain and urine screens for substances to determine whether there are rare organic reasons for non-response.
Strategies to address non-response
Address adherence to treatment
Firstly, it is important to identify if the patient is non-responsive as a result of poor adherence to treatment. Review both the patient’s medication administration and blood levels, when available. Psychoeducation regarding the nature of the illness, rationale for treatment and its benefits is essential, within the limits of the patient’s capacity for comprehension (Velligan et al., 2009) and insight (Berk et al., 2010; Rouget and Aubry, 2007) (Level I). Clearly in acute mania engagement is difficult and unlikely. If the patient has poor adherence, careful supervision of medication administration, use of dosette devices and improved routines for medication use (for example linking consumption to another regular life event) can be very helpful, as are the use of olanzapine wafers, risperidone quicklets, haloperidol and chlorpromazine syrups and asenapine wafers, as they offer a more reliable means of oral administration. Asenapine wafers are more difficult to use than the other options, because they need to be applied sublingually and the patient has to refrain from imbibing any foods or fluids. Further, depot antipsychotic medications, such as risperidone injection, olanzapine pamoate monohydrate, haloperidol decanoate, zuclopenthixol decanoate, flupenthixol decanoate, fluphenazine decanoate and paliperidone palmitate, may offer greatly improved adherence to treatment, so it may be worth adding these to the patient’s treatment regime (CBR). However, it is important to recognise that some agents, such as olanzapine, can cause acute sedation and others, such as risperidone, may take weeks to achieve a therapeutic level. Risperidone Consta is slow to achieve adequate bioavailability, and therefore, is not useful for acute mania. Paliperidone palmitate is slow to become efficacious and therefore is more difficult to use in acute mania. If it has been verified that the patient has adhered to their treatment plan, review medication dosages and increase to maximal tolerated levels, using blood levels as a guide (CBR). Ultimately, managing side effects and ongoing dialogue with patients about preferred side effect profile of different medication options will assist compliance.
Change medication
Changing medication to a different anti-manic or second-generation anti-psychotic medication can be effective. However, prior to switching maximise doses of existing agents in case of ‘pharmacokinetic’ treatment resistance.
Combination therapy
Combining an atypical antipsychotic with lithium or valproate is a common strategy employed by clinicians. It is an option that is supported by recent research, for example aripiprazole is superior compared to placebo as an adjunct to lithium or valproate. However, although efficacy is greater with combination therapy, discontinuation rates are also higher because of adverse effects (De Bartolomeis and Perugi, 2012; Geoffroy et al., 2012; Yatham et al., 2013a).
Conventional treatments such as lithium have also been combined with hormonal therapies such as tamoxifen, hormone replacement therapies and oral contraceptives in the treatment of mania. Tamoxifen is a selective oestrogen receptor modulator with both agonist and antagonist actions peripherally and within the brain. Specifically, it is a protein kinase C (PKC) inhibitor, which has shown some promise as an effective treatment of mania when augmenting lithium and could be useful in the management of treatment resistance. It has been shown to be superior as an adjunct to lithium compared with lithium monotherapy (Amrollahi et al., 2011; Zarate et al., 2007) (Level III), but may be less effective than alternative combinations such as medroxyprogesterone augmentation (Kulkarni et al., 2014; Nguyen and Low, 2012).
ECT
ECT is highly effective in the treatment of refractory mania (Level II) and can be particularly useful in the management of treatment resistant mania (Sienaert and Peuskens, 2006).
Management of bipolar depression
Bipolar depression is the predominant and most disabling component of both BD I and BD II (Judd et al., 2003a) and patients with bipolar disorder spend a considerable proportion of their lives with syndromal or subsyndromal depressive symptoms (Judd et al., 2003a, 2003b). This is also important because much of the increased risk of suicide in bipolar disorder is a consequence of depression. The treatment of bipolar depression is difficult and outcomes are often poor, partly because no medications have been specifically developed to treat this phase of bipolar disorder but also because management requires careful consideration of complex issues, such as treatment emergent affective switching (TEAS) into mania or hypomania, possible cycle acceleration, and the precipitation of mixed symptoms.
Treatment options for bipolar depression
Patients with bipolar depression either present de novo – on no medication – or have a breakthrough episode whilst on maintenance treatment.
De novo bipolar depression: patients on no medication are either medication naive or are yet to commence treatment for the first time, or have simply stopped taking treatment that has been previously prescribed.
Breakthrough bipolar depression: patients have either recently commenced treatment, which is still being titrated and they are yet to respond, or more commonly they have become unwell whilst taking treatment.
In terms of prescribing medications there are two options:
monotherapy or combinations.
Both options can be administered in either context: de novo or breakthrough bipolar depression.
This section, therefore, considers monotherapy and the combination of agents including the option of adding medication to ongoing treatment. Where possible, monotherapy is preferable but in most instances combinations of medications are more effective making this the preferred option even though it may be less well tolerated (see Figures 12 and 13).
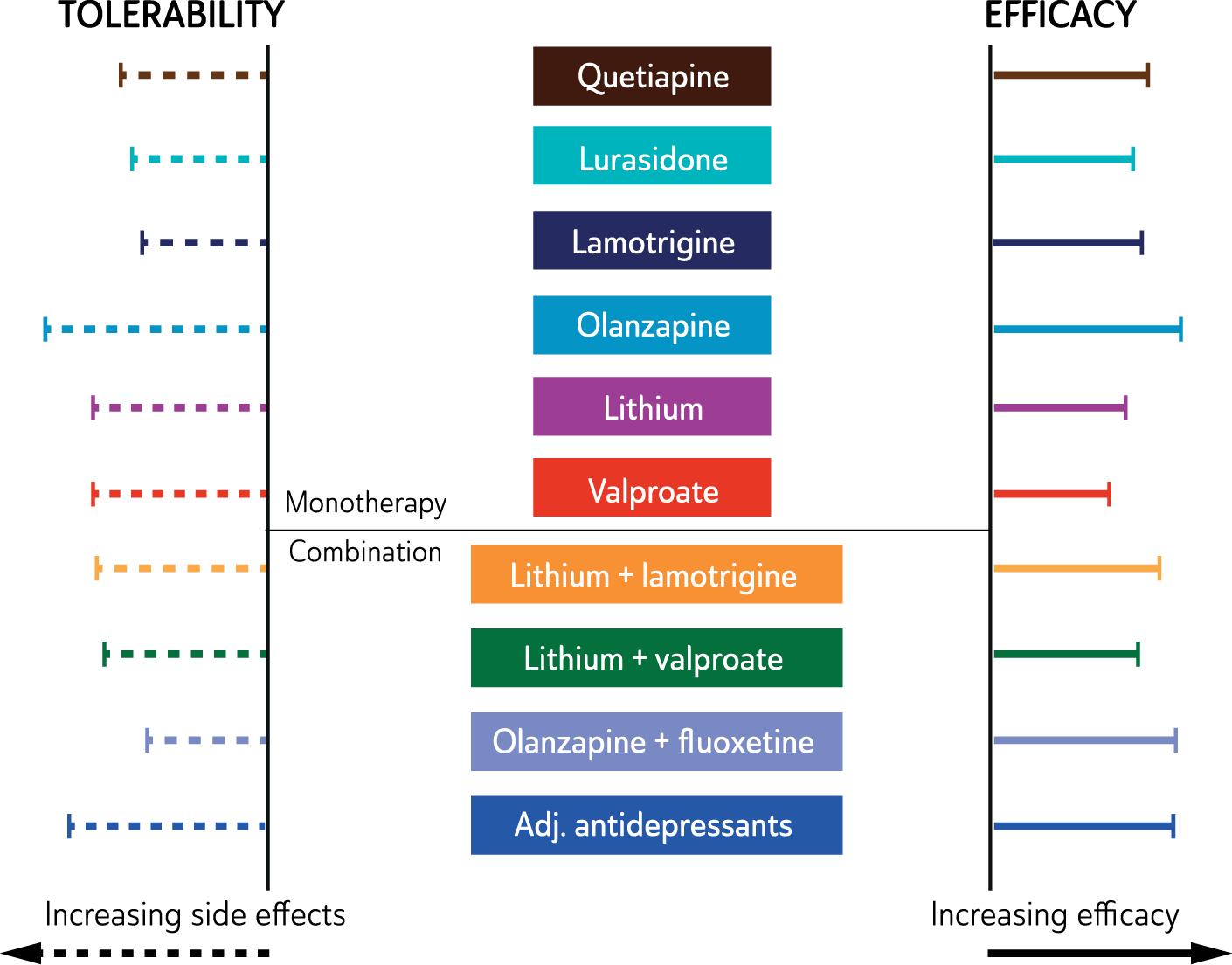
Effectiveness of medications used to treat bipolar depression.
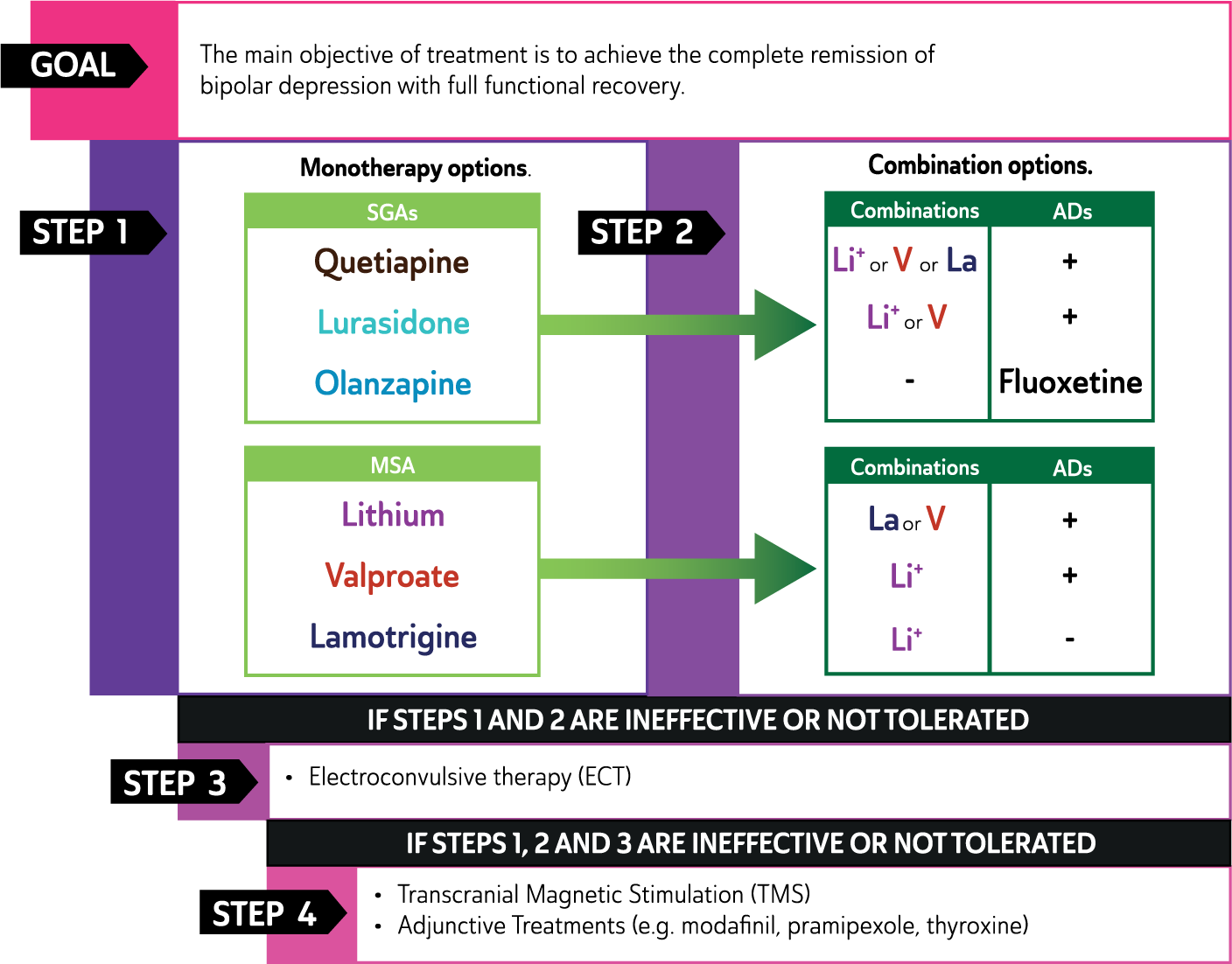
Management of Bipolar Depression.
The evidence for treating bipolar II depression is still emerging and the extent to which the evidence from BD I can be extrapolated to BD II is not clear. The key points of difference are noted below.
The treatment options for the management of bipolar depression include psychological, pharmacological, and physical interventions. These can be administered separately or in conjunction with one another. Psychological interventions are potentially useful early in the course of the illness and should be considered soon after diagnosis but only as adjunctive to pharmacotherapy. Pharmacological treatment is the mainstay of long term bipolar disorder management, though ECT may be necessary in providing relief when patients are acutely depressed or less commonly manic, especially in urgent situations such as when patients are experiencing substantive suicidal ideation.
Pharmacological strategies for bipolar depression
Monotherapy
As can be seen from Figures 12 and 13 monotherapy has demonstrated efficacy in the treatment of bipolar depression and comprises two groups of medications: second generation antipsychotics (SGAs) and mood stabilising agents (MSAs). Notably the use of adjunctive antidepressants remains contentious and the efficacy of this strategy longterm is unclear. In contrast, there is growing consensus that, if possible, antidepressant monotherapy should be avoided.
The second generation antipsychotics include quetiapine, lurasidone and olanzapine (rank order–noting that the latter is least favoured because it has the greatest likelihood of causing metabolic syndrome). The MSAs; lithium, lamotrigine and valproate can also be prescribed as individual agents. Therefore the SGAs and MSAs constitute a total of six possible monotherapy options. Note, the specific combination of olanzapine and fluoxetine (OFC) which is available in some countries as a single formulation is not classified as monotherapy because it consists of two established pharmacological agents.
In practice, a trial of monotherapy is often necessary to identify those patients who can be managed with a single agent. But while monotherapy is ideal, frequently patients require combinations of medications.
Combination
Whereas monotherapy is the preferred strategy for treating major depression, the effective treatment of bipolar depression often requires combinations of medications. The second generation antipsychotics and mood stabilising agents with efficacy as monotherapy agents can be combined or used in conjunction with antidepressants.
Second Generation (Atypical) Antipsychotics: Effective monotherapy options can be added to either mood stabilising agents or antidepressants.
Mood Stabilising Agents: Lithium can be combined with all other monotherapy options; valproate can similarly be combined with all monotherapy options but should be used with caution in women of childbearing age and when combined with lamotrigine. 10
Antidepressants: antidepressants can be added to all effective monotherapy options with the exception of lamotrigine, however the benefits of this strategy remain unclear. There is specific clinical trial evidence that fluoxetine is more effective than placebo when added to olanzapine.
In practice there may also be instances in which a SGA is added to a MSA and an antidepressant (triple therapy). However, evidence for efficacy and tolerability of such combinations is lacking.
Regarding the prescription of antidepressants in the treatment of bipolar I depression, these should be avoided if there have been one to two manic symptoms as part of the depressive episode, or psychomotor agitation, or rapid cycling is a feature of the illness (Pacchiarotti et al., 2013a) or if there is a history of antidepressant-induced mood elevation, or ongoing active substance use (see Recommendation Box 8).

Pharmacological strategies for treatment of depressive episodes in bipolar II depression
The evidence for bipolar II depression is still emerging. In practice evidence from the treatment of bipolar I depression is extrapolated to the treatment of bipolar II depression. However key differences regarding the evidence as compared with bipolar I depression are that antidepressants may be used as monotherapy noting that the evidence is modest at best, and that benefits are most likely early in the course of illness. Amongst the two groups of monotherapy agents (SGAs and MSAs), quetiapine and lamotrigine are the most favoured respectively in clinical practice.
Monotherapy evidence
Antidepressants
The issue of the use of conventional antidepressant agents in bipolar depression remains controversial. As the recent ISBD task force report noted, “There is a striking incongruity between the wide use of and the weak evidence base for the efficacy and safety of antidepressant drugs in bipolar disorder” (Pacchiarotti et al., 2013a). In practice, antidepressants are often prescribed for bipolar depression as monotherapy or in combination with other medications. The most recent meta-analyses provide conflicting evidence regarding the efficacy of antidepressants in bipolar depression compared with placebo (Sidor and MacQueen, 2011; Vázquez et al., 2011). Inconsistent research findings do not allow for a final evaluation and recommendation of antidepressants in the treatment of bipolar depression, therefore the ISBD task force concludes that “clinical trials do not provide adequate support for the efficacy of antidepressant monotherapy in treating bipolar depression”.
Bipolar depression refractory to mood stabilisers is a common clinical problem (Sachs, 1996). It appears that some mood stabilisers may have greater efficacy during the depressive phase, therefore consideration of adding or switching to such agents is advisable (Chiesa et al., 2012; De Fruyt et al., 2012; Van Lieshout and MacQueen, 2010). While some patients appear to benefit from carefully monitored use of antidepressants, others suffer iatrogenic mood destabilization due to induced elevated states (Amsterdam and Shults, 2010b; Sidor and MacQueen, 2011; Tondo et al., 2010b; Vázquez et al., 2011).
Two recent studies provide contrary findings (Malhi, 2015a), with one suggesting that long-term continuation of antidepressants in patients with rapid-cycling bipolar disorder leads to a threefold increase in mood episodes during the first year of follow up (El-Mallakh et al., 2015) whereas continuation of antidepressant monotherapy in BD II provides prophylaxis with only minimal risk of switching (Amsterdam et al., 2015). Given these complex issues, use of antidepressants in bipolar patients should be generally overseen by a psychiatrist where possible. Prior to considering an antidepressant, mixed states, substance induced states, akathisia and adjustment disorders should be excluded. Patients and carers must be clearly informed of the risk of elevated states being induced with antidepressant therapy (Miklowitz et al., 2003).
Therefore, clinically, when treating bipolar depression the patient’s mental state needs to be closely monitored for the emergence of elevated states. Where possible it is best to use an antidepressant that has been previously effective and tolerable for the particular patient.
SNRIs and TCAs may have greater propensity to induce elevated states, making other classes of antidepressants initially preferable (Gijsman et al., 2004; Giorlando et al., 2013; Post et al., 2006). Furthermore, antidepressant agents that can assist sleep cycle restoration may also have a clinical advantage over more activating agents except in patients with significant hypersomnia and anergia where activation may be desired. However, a recent study that investigated adjunctive therapy for bipolar depression found that agomelatine was not superior to placebo (Yatham et al., 2015). In the treatment of bipolar depression, adjunctive antidepressants should be prescribed at usual dose ranges as there is no biological basis for employing doses not able to lead to adequate CNS bioavailability. The clinical risk benefit ratio of antidepressants in bipolar depression needs to be determined on a case-by-case basis given considerable clinical heterogeneity in response patterns.
In addition, treatment choice should take into account the likelihood that an agent may be used to provide prophylaxis against future episodes of depression (see: Treatment options in major depressive disorder maintenance phase).
For these reasons antidepressants should be used with caution in bipolar depression (limiting duration as far as possible), always co-prescribed with a mood stabilising agent, and with very close mental state review given the risk of mood destabilisation in some patients. However, this potential hazard needs to be balanced against the reality that in clinical practice many bipolar patients appear to benefit from antidepressants, and the risks associated with refractory depression. Mood stabiliser monotherapy is therefore preferable because, if it provides adequate control of the depressive phase, the short and long term risks of iatrogenic elevated states are averted. Such deliberations are best made by an experienced clinician.
Second generation antipsychotics
Randomised controlled trials report that olanzapine (Tohen et al., 2003c, 2012), quetiapine (Calabrese et al., 2005b; McElroy et al., 2010; Thase, 2006; Young et al., 2010) and lurasidone (Loebel et al., 2014a, 2014b) are effective treatments for bipolar depression, but these studies have significant methodological issues that moderate the strength of the evidence. In considering treatment choice, the high drop-out rate and long term likelihood that olanzapine and quetiapine will lead to metabolic disturbance need to be taken into consideration. In this regard, based on available evidence, lurasidone is preferable (Loebel et al., 2014b; Selle et al., 2014) also because the combination study demonstrating its efficacy has fewer methodological issues.
Mood stabilising agents (MSA)
Lithium monotherapy has not been demonstrated to be convincingly superior to placebo as monotherapy in the treatment of bipolar depression (Young et al., 2010). However, empirical evidence, along with its proven value in mania prophylaxis and efficacy in unipolar depression as an augmentation strategy, make it a suitable first choice, particularly in classic bipolar I disorder depression (Malhi et al., 2015b). In addition, lithium has the added value of reducing the risk of suicide in mood disorders (Cipriani et al., 2013) (see: Lithium in maintenance phase).
Evidence regarding the beneficial effects of valproate monotherapy in the acute treatment of bipolar depression is limited to three very small (n = 18, n = 25, n = 54) trials, a long-term trial and a small meta-analysis, all of which showed significant benefit compared with placebo (Bond et al., 2010; Bowden, 2003; Davis et al., 2005; Ghaemi et al., 2007; Muzina et al., 2011; Smith et al., 2010).
Lamotrigine monotherapy has modest efficacy in the treatment of bipolar I depression. Its suitability as a bipolar antidepressant agent in patients with BD I who are drug naïve or who have ceased medication should be considered, in the context of the overall history of the illness and in particular, the severity and frequency of manic episodes. A trial of a recognised mania prophylactic should also be considered. Clinically, lamotrigine seems to have better utility in bipolar II depression. See Figure 13 for treatment recommendations in the management of bipolar depression.
Antidepressant-induced mania, switching and cycle acceleration
Given the spontaneous occurrence of switching in bipolar disorder, it has been difficult to determine the extent to which switching can be attributed to antidepressants (Malhi et al., 2015). The natural history of BD I and BD II differs with respect to switching, which is more a feature of BD I as compared to BD II (Judd et al., 2003a, 2003b). Furthermore, the rate of switching appears to vary depending on the class of antidepressant. For example, TCAs (7–11%) and venlafaxine (13–15%) are associated with a higher risk of inducing mania compared with SSRIs and bupropion (Gijsman et al., 2004; Leverich et al., 2006; Nemeroff et al., 2001; Peet, 1994; Post et al., 2006; Sachs et al., 1994; Vieta et al., 2002). Indeed, a recent analysis reported that SSRIs or bupropion had a switch rate only 1.16% higher than placebo (Vázquez et al., 2013). Amongst the SSRIs fluoxetine seems to be less likely to cause switching (Amsterdam and Shults, 2010a) but a paucity of studies warrants cautious use.
Despite longstanding concerns and suggestions that antidepressants may induce cycle acceleration (Altshuler et al., 1995), which is not only more disruptive clinically but also more difficult to treat, there is little evidence regarding whether antidepressants do, in fact, cause this. No increased rate of switching compared with placebo has been found in other trials of monotherapy with lamotrigine (Calabrese et al., 2008; Geddes et al., 2009), second generation antipsychotics (quetiapine, olanzapine, aripiprazole, ziprasidone) (De Fruyt et al., 2012), or lithium (Young et al., 2010).
However, a recent consensus statement from experts in the field recommends that, (a) patients should not use antidepressants if they have a history of past mania, hypomania or mixed episodes emerging during antidepressant treatment, and (b) antidepressants should be avoided in patients with high mood instability or with a history of rapid cycling (Pacchiarotti et al., 2013a, 2013b).
Psychological interventions for bipolar depression
(See Table 21.) The majority of research into psychological interventions for bipolar depression has focused on the maintenance phase, and there is some suggestion that psychological interventions are best commenced outside acute episodes (see below). However, the large STEP-BD trial provides Level II evidence for the efficacy of adjunctive psychological intervention when commenced during acute bipolar depression. Up to 30 sessions of Cognitive-Behavioural Therapy (CBT), Interpersonal and Social Rhythm Therapy (IPSRT) or Family-Focused Therapy (FFT – see Table 21) were compared against a minimal psychological intervention of 3 sessions of ‘collaborative care’ for patients recruited in an acute depressive episode and continuing on pharmacotherapy (Miklowitz et al., 2007). The structured, intensive psychotherapies were associated with more frequent (and rapid) recovery from depression and better psychosocial functioning than collaborative care: year end recovery rates were 64.4% compared to 51.5%, and in any given month of the 12-month follow-up, patients in the intensive interventions were 1.6 times more likely to be clinically well. No differences were found between the three specific psychological therapies, so the evidence supports psychological intervention generally without specifying which type (see below).
Psychotherapy for depressive phase and for maintenance phase of Bipolar Disorder.

Insufficient studies directly address the treatment of acute bipolar depression, so this column refers to studies reporting outcomes for depression syndromes and symptoms.
Indirect evidence for the efficacy of adjunctive psychological interventions in acute bipolar depression includes: (a) Level I evidence for the efficacy of psychological interventions for the residual depressive symptoms of bipolar disorder (see Table 21), and (b) Level I evidence for psychological interventions in preventing depressive relapse in bipolar disorder. One meta-analysis (8 included studies) found CBT to be as efficacious for symptoms of bipolar depression as unipolar depression (Zaretsky et al., 2007), and another (10 studies) found significant benefits for bipolar depression symptoms at 6 and 12 months follow-up (Szentagotai and David, 2010).
Consequently, recent NICE guidelines recommend psychological interventions (particularly CBT and FFT) for acute bipolar depression (National Collaborating Centre for Mental Health (NCCMH), 2014). These guidelines also recommend that evidence-based psychological interventions for unipolar depression are potentially useful for the depressions of bipolar disorder.
In sum, although research is limited, there is strong clinical consensus that the highly prevalent and highly impairing depressive phase of BD should be managed in a comprehensive biopsychosocial manner (Berk et al., 2013; Malhi et al., 2012b), and adjunctive structured psychological interventions should be offered to help stabilise depressive episodes (Goodwin, 2009; Kendall et al., 2014; Yatham et al., 2013b).
Electroconvulsive therapy and other brain stimulation methods
Compared to ECT in unipolar depression, much less attention has been paid to the investigation of ECT in treating bipolar depression, and most studies are of treatment resistant cases. A systematic review (Versiani et al., 2011) identified only 9 studies, none of which were prospective randomised controlled trials. However, this review identified 10 controlled studies that compared the efficacy of ECT in treating bipolar versus non-bipolar depression, with half of these being prospective. All but one of these studies demonstrated that ECT was equally effective for both bipolar and non-bipolar depression with some evidence that the response was more rapid in bipolar depression.
Another review of ECT in bipolar disorder (Loo et al., 2011) identified four studies that suggested a more rapid response in bipolar depression. Studies comparing ECT and medication in the treatment of bipolar depression have been inconclusive and no difference has been found though this may be a consequence of methodological shortcomings (Loo et al., 2011; Versiani et al., 2011). A large multicentre study (n = 220), comparing unilateral, bitemporal and bifrontal ECT in the treatment of depression (Kellner et al., 2010) included 50 bipolar subjects. The study compared the response rate in the bipolar subgroup with the non-bipolar group (Bailine et al., 2010) and found no difference.
ECT-induced mania
While ECT-induced switching into mania is recognised as a potential adverse effect, few studies have examined the rate of switching (Loo et al., 2011) and estimates of the rate vary. Reports of switching in controlled studies of ECT in bipolar depression are relatively uncommon. For example, of the 50 bipolar depressed patients treated with ECT in the multicentre study quoted above, none switched into mania (Bailine et al., 2010).
There are insufficient data to guide the treatment of ECT-induced mania and its management may depend on the availability of services to treat a sudden switch. Many clinicians advocate continuing with ECT, as it is an effective treatment for mania as well as depression. There may be some risk that this approach further escalates mania and, if appropriate facilities for managing mania are not immediately available, then ceasing ECT and treating pharmacologically is a suitable alternative.
Treatment non-response
Assessing treatment non-response
Ongoing subsyndromal depressive symptoms are common in bipolar disorder, as is a high rate of treatment non-response. Treatment resistant bipolar I depression can be defined as failure to reach remission with adequately dosed lithium (blood level 0.6–0.8 mMol/L) or to other adequate ongoing mood-stabilizing treatment, plus lamotrigine (50–200 mg/day) or with full dose quetiapine (⩾ 600 mg/day) as monotherapy. Similarly, treatment resistant bipolar II depression can be defined as failure to reach remission with adequately dosed lithium (blood level 0.6–0.8 mMol/L) or to other adequate ongoing mood-stabilizing treatment, plus lamotrigine (50–200 mg/day) or with full dose quetiapine (300–600 mg/day) as monotherapy. If treatment resistance is diagnosed, then a total reassessment of the nature of the bipolar depression is indicated. This is similar to the re-assessment recommended for the treatment of unipolar major depression (see: Managing suboptimal response), but there are a number of additional factors in bipolar depression that need to be considered. These include: thyroid dysfunction (Cole et al., 2002), non-adherence with pharmacological treatment (Perlis et al., 2010), rapid cycling (Coryell et al., 2003) (in which the episode is likely to resolve spontaneously and more quickly), and mixed features (Goldberg and McElroy, 2007).
Pharmacological strategies to address non-response
Research and clinical practice have provided options for the management of treatment resistant bipolar depression using medications, although the results vary widely and clinicians must use their knowledge of each patient to guide their choices. The following are suggested (Pacchiarotti et al., 2009) steps for such management and should involve supervision by a specialist:
Bipolar I depression
Quetiapine (or other atypical antipsychotic or mood stabilizer if preferred) alone → Quetiapine or Lithium + Lamotrigine (latter with care) or Olanzapine + Fluoxetine → Lithium or Quetiapine + SSRI or Bupropion → Pramipexole or Modafanil → Electroconvulsive therapy.
Bipolar II depression
Similar to management of bipolar I depression but include irreversible monoamine inhibitors as options when combining Lithium or Quetiapine (or other atypical antipsychotic or mood stabilizer if preferred) with an antidepressant (with care if combining lithium with MAOI).
Brain stimulation approaches
Very few studies have evaluated the use of rTMS treatment in homogeneous groups of patients with bipolar depression. However, many depressed patients with bipolar disorder have been included in trials of rTMS with no evidence that bipolar patients are less likely to respond to rTMS than patients with unipolar depression (Fitzgerald et al., 2013b), and overall, the rate of manic switch with rTMS treatment appears to be low.
Physical treatments such as ECT, appropriately timed bright light therapy (particularly if a seasonal component is involved) (Poon et al., 2012), vagus nerve stimulation (Rizvi et al., 2011) and repeated transcranial magnetic stimulation (George, 2010) should all be reserved for specialist centres and patients need to be monitored medically and for the emergence of manic switches.
Adjunctive strategies
Pharmacotherapy
Several medications may be added to existing treatment regimens including thyroid hormones (Bauer et al., 2005), glutamate antagonists (memantine/riluzole) (Owen, 2012), intravenous ketamine (Zarate et al., 2012), and modafinil/armodafinil (latter is the R-enantiomer of modafinil) (Calabrese et al., 2010). All these medications require further study and are best regarded as experimental.
Amongst these agents, ketamine has emerged as a potential antidepressant. Studies investigating its therapeutic effects have found a reduction in depressive symptoms in those suffering from treatment resistant depression along with mitigation of suicidality (Murrough et al., 2013; Price et al., 2009). Improvement is also seen in bipolar depression, however, the effects are short-term and often transient.
Specifically, following an initial improvement, mood can rapidly decline after a single treatment of ketamine, thus potentially enhancing the risk of suicide. Nevertheless, ketamine does appear to be more effective than placebo at reducing the symptoms of depression, albeit for a week or so (McCloud et al., 2015). The evidence favouring bipolar depression is even more tentative (McCloud et al., 2015). Based on available evidence, it is not possible to recommend ketamine as an option for treatment non-response in either unipolar or bipolar depression.
Complementary therapies
Omega-3-fatty acids (Sarris et al., 2012), N-acetyl cysteine (NAC) (Berk et al., 2008a) and other nutraceuticals (Sarris et al., 2011) may also be considered. Meta-analytic evidence supports the adjuvant use of Omega-3 for bipolar depression, although not for mania (Sarris et al., 2012). According to a recent review (Sylvia et al., 2013), preliminary data yields conflicting but mainly positive evidence for the use of chromium in the treatment of bipolar depression. Moreover, preliminary randomized, controlled trials suggest that adjunctive choline, magnesium, folate and tryptophan may be beneficial for reducing symptoms of mania (Sylvia et al., 2013).
Another recent systematic review (Sarris et al., 2011) found positive evidence with large effect sizes for NAC (d = 1.04) and a chelated mineral and vitamin formula (d = 1.70) in the adjunctive treatment of bipolar depression. Regarding the treatment of bipolar mania, several adjunctive nutraceuticals reduced the symptoms of mania with strong clinical effects: a chelated mineral formula (d = 0.83), L-tryptophan (d = 1.47), magnesium (d = 1.44), folic acid (d = 0.40), and branched-chain amino acids (d = 1.60) (Sarris et al., 2011).
Long-term management of bipolar disorder
Bipolar disorder is a recurrent illness with episodes occurring on average every 17 to 30 months (Angst and Sellaro, 2000). Once an acute episode has resolved, the clinical focus of management shifts to continuation and then to maintenance treatment. The primary aims of long-term treatment are to prevent relapse or recurrence of manic and depressive symptoms and to optimise functional recovery.
Successful long-term management of bipolar disorder requires a collaborative therapeutic relationship in which there is a clear understanding of these goals and management is tailored to the individual. Treatment needs to take into consideration comorbid medical and psychiatric illnesses, personality variables (Lozano and Johnson, 2001) and lifestyle factors (e.g., accommodation, social support, employment), all of which can contribute to life events or psychosocial stressors, impeding functional recovery. The long-term treatment of bipolar disorder requires both psychotherapeutic and pharmacological approaches.
Phases of long-term treatment
The transition from acute symptoms to remission and subsequent achievement of recovery divides long-term treatment into continuation and maintenance phases (see: Figures 5 and 14) and separates any recrudescence of symptoms into relapse and recurrence, respectively.
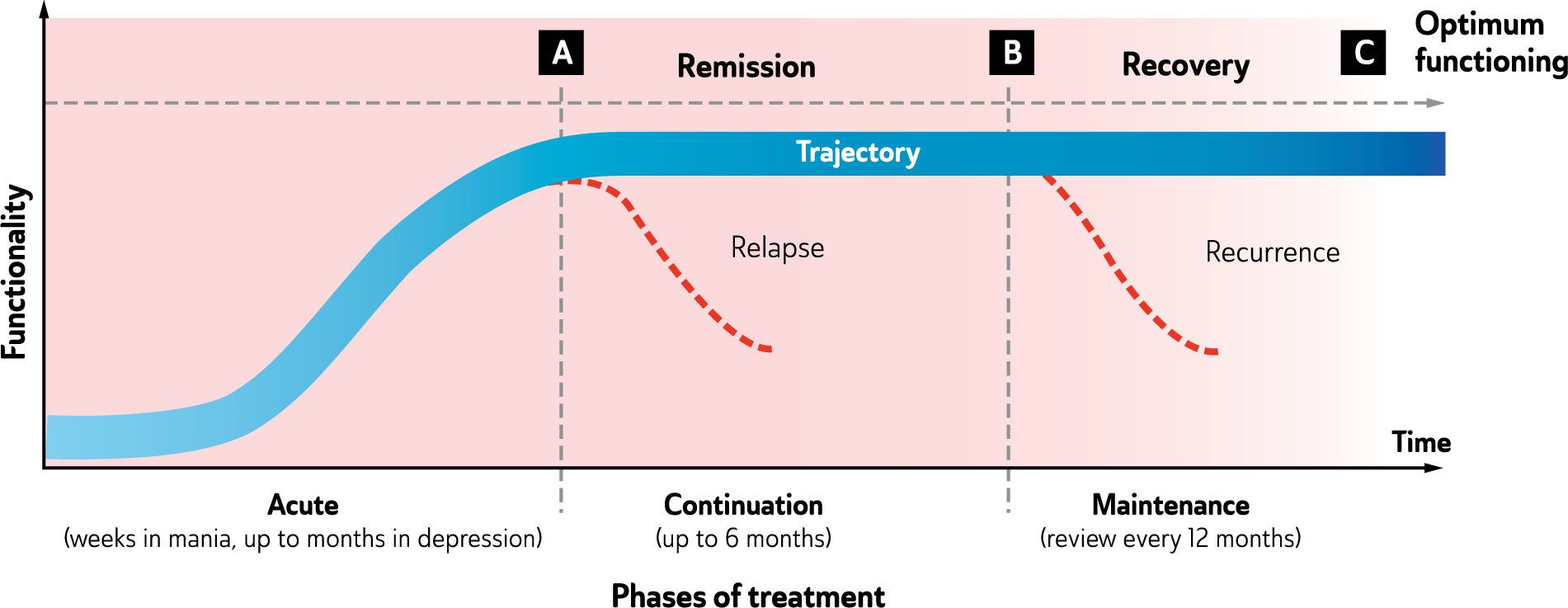
Trajectory of functional recovery.
The majority of patients with bipolar disorder will suffer from multiple episodes of depression and mania during their lifetime and, therefore, in almost all cases long-term treatment is necessary. The course of illness and trajectory are difficult to predict and even with adequate treatment, almost half of all bipolar patients will have another episode within two years, and the majority will experience further illness within five years.
In long-term treatment, the first decision point occurs at the transition from acute treatment to continuation treatment (Point A in Figure 14). At this point it is important to begin withdrawal of adjunctive agents such as benzodiazepines that have been used to manage acute behavioural and cognitive disturbance associated with a mood episode. However, it is important to maintain euthymia and only if this can be ensured should a gradual reduction of medication dosage be considered along with withdrawing medication to achieve monotherapy. Maintaining compliance and ensuring adequate adherence to treatment instructions is extremely important and therefore active engagement is necessary and should include regular review for therapeutic monitoring and reappraisal of the benefits and risks of ongoing therapy.
The second decision point occurs at the transition from continuation treatment to maintenance treatment (Point B in Figure 14). At this point it is important to reassess the efficacy of treatment thus far, the mood stability that has been achieved with acute and continuation treatment, and the number, polarity and severity of relapses/recurrences, which have occurred over the individual’s lifetime. It is also important to assess the extent to which the onset of episodes is predictable; for example, whether they are insidious, abrupt, related to life stresses or contain a seasonal component. Often, symptoms gradually increase in number and severity. These are described as ‘early warning signs’ and individuals can learn to detect these and anticipate the onset of episodes of illness. Adjunctive psychological interventions aimed at relapse prevention are typically introduced in the inter-episode period. Inter-episode periods should also be appraised to determine whether they feature subsyndromal symptoms and assess the maximum functional capacity that an individual has achieved. Specifically, it is important to gauge their quality of life whilst on maintenance treatment. This requires evaluating the tolerability of past treatments and the severity of illness overall, in particular its impact on the individual’s capabilities. This is an important exercise and is critical in informing maintenance treatment. In most cases of BD maintenance therapy is necessary, but it is particularly mandated if the individual has had many past episodes (which increase the risk of relapse and impede response to treatment) and is therefore at considerable risk of recurrence.
Continuation treatment
The continuation phase of treatment begins immediately after the remission of acute symptoms. The primary aim of treatment in this phase is to continue to achieve mood stability and prevent relapse. Ongoing treatment during the continuation phase is determined by the treatment which has been effective in the acute phase of illness and the predominant mood polarity, the current polarity, the illness stage and the history of the patient’s previous response to intervention. Time to achieve mood stability varies, but once three months of continuous mood stability has been achieved, treatment considerations shift to prophylaxis and maintenance therapy.
Key tasks during continuation treatment include maintaining clinical improvement and monitoring for side effects, which can prove troublesome and decrease adherence to treatment. During this stage of treatment, medications used to treat acute symptoms are usually tapered gradually and eventually stopped altogether as the illness stabilises. This ensures that only necessary medications are continued long-term and side effects are avoided, or kept within tolerable levels. Medication withdrawal necessarily risks relapse, and therefore, treatments should be withdrawn gradually with regular clinical monitoring.
Continuation therapy (Marked ‘A’ in Figure 15) aims to achieve optimal mood stability and prevent the occurrence of a relapse into the most recent mood episode. Monotherapy is desirable but combination strategies are often necessary. Suitable combination strategies include lithium or valproate in combination with olanzapine, quetiapine, lamotrigine or aripiprazole. These combinations are useful for treating bipolar disorder in which depression predominates or both depression and mania occur equally. With respect to monotherapy, lithium and olanzapine are more useful where mania has been a significant feature of the illness, whereas lamotrigine is better suited to a pattern in which depression predominates. Quetiapine monotherapy is useful where both phases of illness feature strongly.
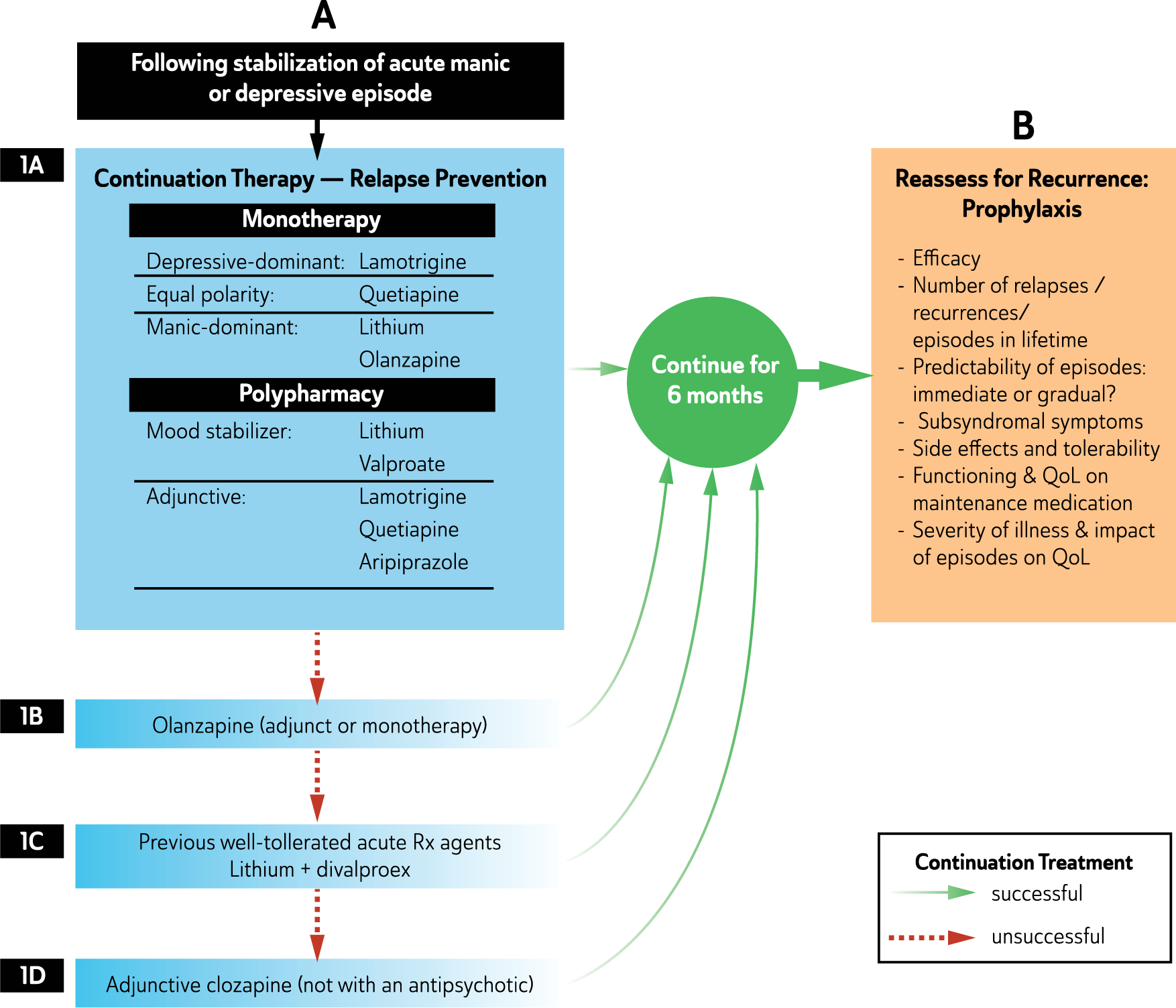
Continuation Therapy of Bipolar Disorder.
Continuation therapy may last for up to six months during which time a period of at least 6 months mood stability should be achieved, after which it transitions to maintenance therapy. During this time, if any of the initial strategies for relapse prevention are unsuccessful in achieving optimal mood stability, then ongoing treatment can be substituted or augmented adjunctively. Other previously well tolerated treatments utilised successfully in acute treatment can also be prescribed. In some cases, a combination of lithium and valproate may be necessary, and in exceptional cases clozapine may be considered in conjunction with an anticonvulsant or lithium.
Longitudinal mental state review and continuity of care enable identification of illness profile and treatment efficacy on a case-by-case basis. This facilitates optimal clinical outcome and may from time to time involve soliciting a second opinion from a psychiatric colleague or specialist clinic.
Maintenance treatment
The main goal of maintenance treatment is to prevent future episodes of illness (recurrence) and enhance resilience. In practice, even with optimal treatment, complete prophylaxis is seldom achieved; therefore, subsidiary goals warrant consideration. These include reducing the number, intensity and length of episodes and achieving functional mood stability with fewer inter-episode subsyndromal symptoms.
Maintenance treatment should be considered after the first episode of mania and subsequently it is useful to gauge the predominant polarity of the illness (Kessing et al., 2014). Because the occurrence of mania confers the diagnosis of bipolar disorder, it raises clinical issues that can take considerable time to resolve (Malhi and Porter, 2014). For example, acceptance of both the diagnosis and the need for long-term treatment are difficult issues to come to terms with, especially when patients dealing with these concerns are usually adolescents or young adults. Failure to support patients through this acceptance process can hinder long term engagement with care and moderate outcomes (Berk et al., 2010).
In terms of treatment, monotherapy is the gold standard of maintenance medication, but in reality it is often an elusive goal. Most individuals with bipolar disorder require more than one medication long-term in order to maintain mood stability. Generally, the initiation of medications is easier than their cessation and each additional medication adds potential side effects and increases risk of poor compliance. Therefore, the benefits and risks of each medication need to be carefully balanced, and long-term medication necessitates regular monitoring for clinical symptoms and the emergence of side effects to ensure ongoing efficacy and maximise compliance and adherence.
When planning maintenance treatment, the first step is to review the treatments that have been used during the continuation phase of management. These will have been determined by the nature of the acute episode and it is useful to gauge the predominant polarity of the illness. Medications necessary for treating acute symptoms may have been tapered or completely withdrawn during continuation treatment and long-term maintenance may require the initiation of new medications. The choice of medication in maintenance is governed by different factors to those relevant to continuation treatment and the cost/benefit ratio of side effects to efficacy may require further consideration.
The principal aims of maintenance treatment are:
Maintaining mood stability; 11
Achieving complete functional recovery;
Providing long-term prophylaxis; or
Reducing attraction to the euphoria and enhanced abilities of past hypomanic states
Building resilience and improving QoL
A clinical review of previous inter-episode treatment is essential, so as to determine which therapies and combinations of medications have been effective in the past and to consider potential problems with treatment adherence. This information is critical in formulating maintenance treatment.
Mood stability is the core objective of maintenance treatment and therefore a ‘mood stabilizing’ medication(s) is an essential ingredient and should be the first to be considered. At this stage it is useful to gauge the predominant polarity of the illness (Carvalho et al., 2014a). A past history of predominance of one pole or other provides an indication of future recurrences, and this informs prophylaxis as medications differ with respect to efficacy in preventing relapse of depression or mania (Yatham et al., 2013a) (see: Figure 17). However, bipolar disorder is capricious by nature and in many cases a clear pattern of polarity may be difficult to determine.
Medications prescribed during continuation treatment can be continued in maintenance treatment if side effects are tolerable and safe and the patient has managed to demonstrate compliance. If, however, on reviewing past history, recurrence is less likely, then medications can be tapered especially if they are likely to be difficult to tolerate at the present dosages, or cause serious side effects when prescribed long-term. On occasion, switching may be necessary, especially if the individual is experiencing breakthrough symptoms and current medications lack efficacy. Ongoing maintenance treatment requires regular re-evaluation to assess the effectiveness of treatment (efficacy and tolerability) and overall functioning and quality of life. (See Figure 16)
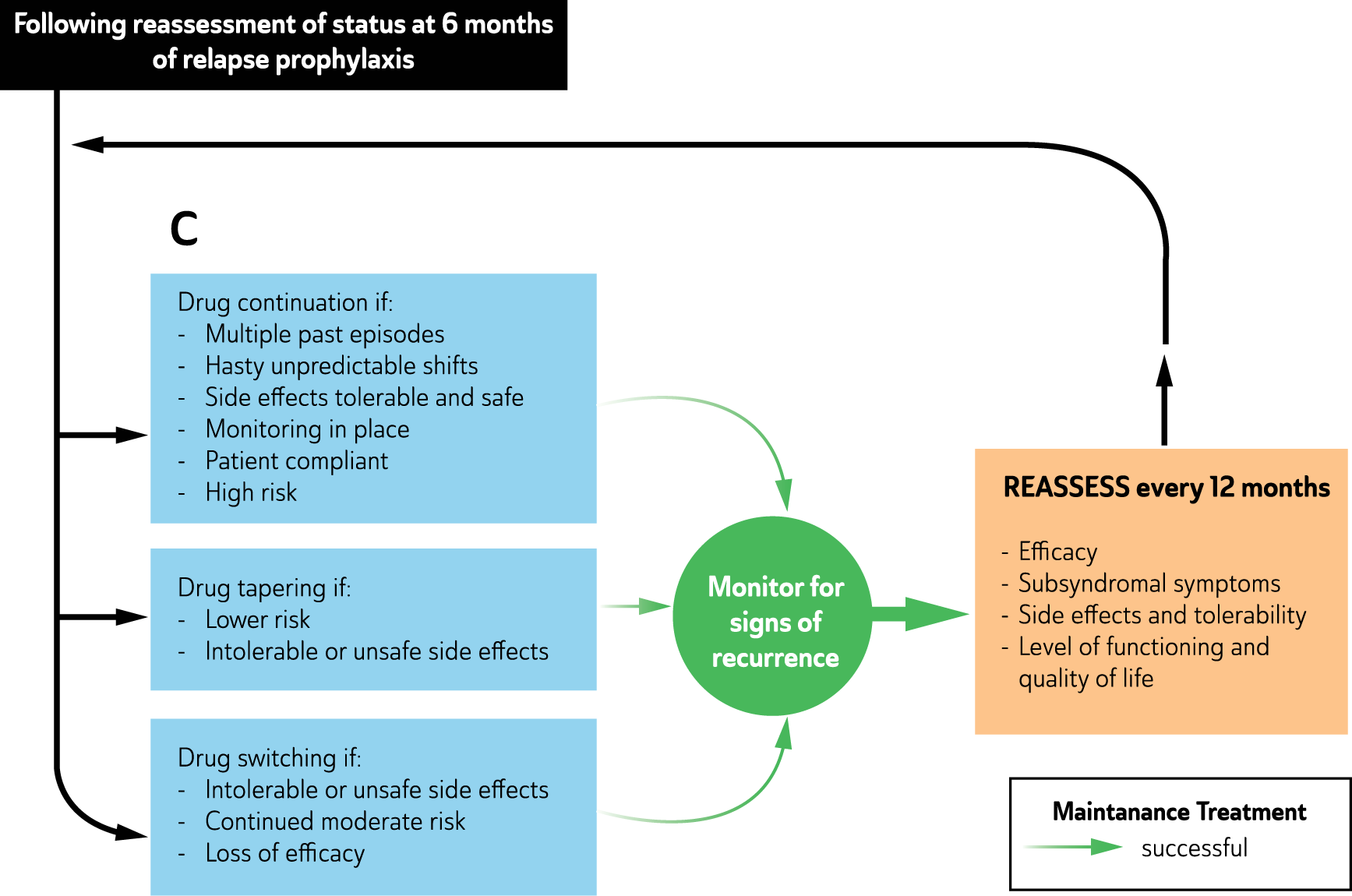
Maintenance Therapy of Bipolar Disorder.
Duration of prophylaxis
In instances of long-term mood stability, individuals often question the need for ongoing prophylaxis and want to stop maintenance treatment. This is more likely:
Early in the course of illness
If the individual: - suffers intolerable/unsafe side effects - has only experienced a few episodes in their lifetime - is yearning for the perceived pleasure of the elevated subjective state
Stopping treatment or interrupting it for a brief period to gain respite or test its feasibility (‘a medication holiday’) requires a careful discussion and evaluation of the impact of the medication on quality of life and whether its benefit outweighs any associated risks. Patients often need to be reminded that recurrence carries significant risks to their health and may provoke considerable losses (e.g. occupation, relationships, financial status). Furthermore, there is no guarantee that medications that have maintained mood stability in the past will once again be successful if reinitiated after stopping. However, there is a small possibility that some individuals remain well in the absence of long-term ongoing maintenance treatment, but it should be emphasised that this is rare (Angst et al., 2003; Simhandl et al., 2014).
Recommended treatments
Pharmacological treatments
Medications used in bipolar disorder prophylaxis have differing levels of efficacy, and some agents show selectivity in their ability to prevent mania or depression (Miura et al., 2014; Yatham et al., 2013a). Much of the evidence for prophylaxis pertains to bipolar I disorder, with relatively little data to inform the management of bipolar II disorder.
Selection of maintenance medication
Maintenance medication should be selected on the basis of both efficacy and tolerability profiles (see: Figure 17). The latter is critical for long-term treatment and these factors need to be balanced alongside individual patient considerations (including preference), past response and safety considerations (risk of suicide). The efficacy of conventional antidepressants (e.g., TCAs, SSRIs and SNRIs) in the maintenance phase of bipolar disorder is not established (Ghaemi et al., 2008), and in some instances treatment may worsen mania with no improvement of depression however, maintenance treatment with adjunctive antidepressants may be considered if a patient relapses into a depressive episode after stopping antidepressant therapy (Pacchiarotti et al., 2013a). Antidepressant treatment in combination with a mood stabilising agent is less likely to precipitate a manic mood switch, but antidepressants should be avoided if there is a past history of (hypo)mania, mixed states or rapid cycling during antidepressant treatment. In some instances, attempts to cease antidepressants during maintenance treatment can precipitate the recurrence of depression, and therefore, adjunctive antidepressant maintenance therapy alongside a mood stabilising medication may be necessary.
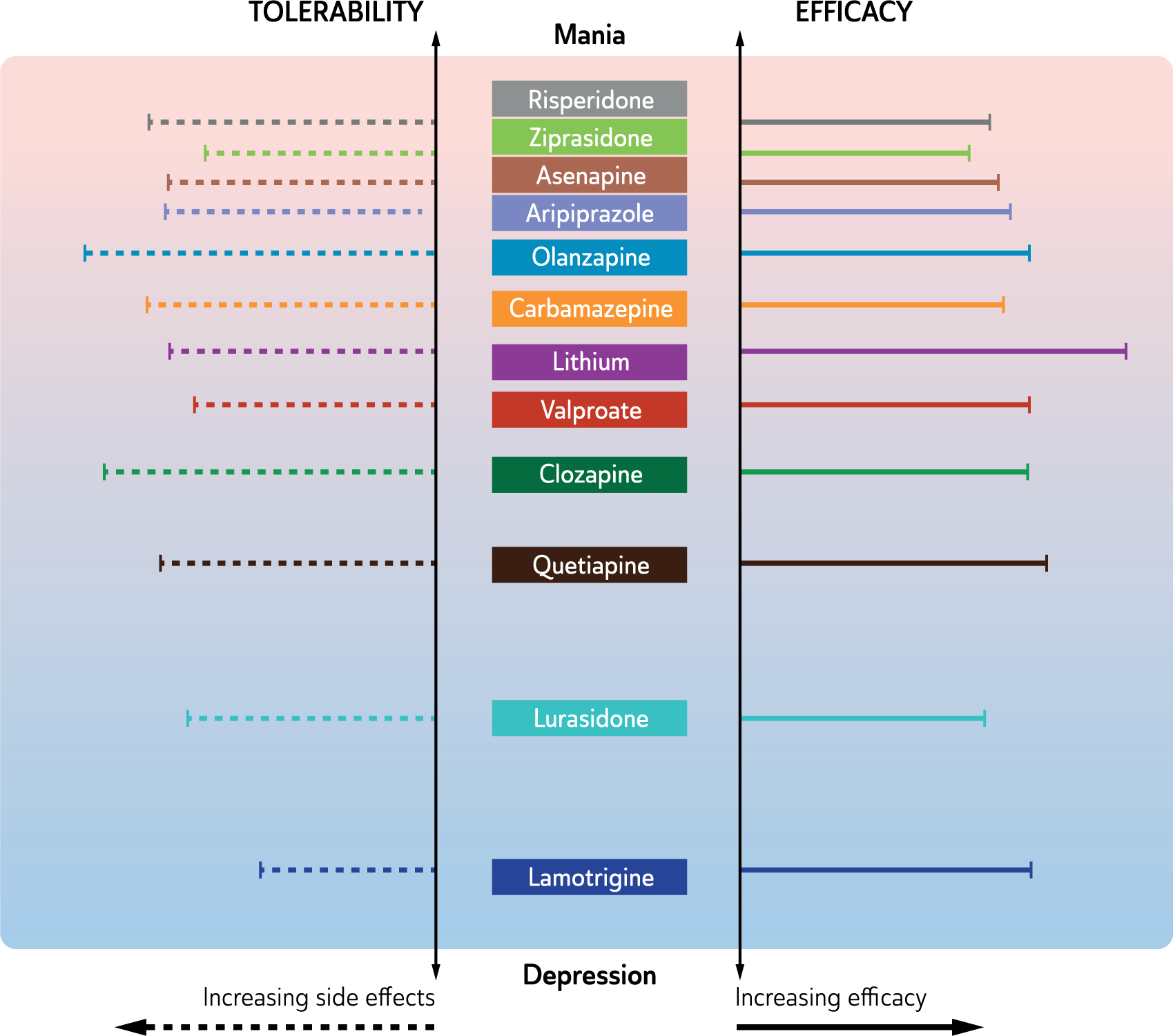
Clinical utility of medications used for maintenance therapy in bipolar disorder.
Lithium
Lithium is the oldest and only agent developed specifically for the treatment of bipolar disorder (Malhi, et al., 2009c). Therefore, it has the largest body of evidence in relation to its efficacy, though much of this is anecdotal and borne out of clinical experience. A meta-analysis of clinical trials up to a decade ago demonstrated that lithium may greatly reduce the overall risk of relapse of a manic episode by 40% to 61%, with modestly lower efficacy in the prevention of depressive relapse (Geddes et al., 2004). Amongst the many studies that have demonstrated its superiority, the BALANCE study (Geddes et al., 2010) is noteworthy because it demonstrated that lithium alone and in combination with valproate is effective in prophylaxis. Lithium is more effective alone than valproate alone, and carries the extra significant benefit of reducing suicidal behaviour and death by suicide. Clinically, lithium has a specific therapeutic serum range that necessitates regular monitoring (Malhi et al., 2011) and long-term administration runs the risk of potential, serious adverse effects, including renal insufficiency and thyroid dysfunction (see: Table 20 Adjuctive treatments) (McKnight et al., 2012; Malhi et al., 2012d; Malhi, 2015b; Shine et al, 2015; Van Gerven and Boer, 2006).
Anticonvulsant agents
Valproate is not formally approved for use as a maintenance agent and remarkably there are no RCTs that have demonstrated its efficacy in long-term prophylaxis. In comparison to lithium, it is less effective (Geddes et al., 2010) but does have modest efficacy in acute mania (Calabrese et al., 2005a; Macritchie et al., 2001; Tohen et al., 2003a). It is therefore often advocated in those patients that have a predominance of manic episodes.
Lamotrigine has greater efficacy in the prevention of depressive relapse but relatively modest impact on risk of manic relapse (Level I) (Bowden et al., 2003; Calabrese et al., 2003; Goodwin et al., 2004; Licht et al., 2010; Van der Loos et al., 2011). Its use is further complicated by the need for slow titration of its dose to limit the risk of severe skin reactions, such as Stevens-Johnson syndrome. Its advantages include its better overall tolerability and relatively low risk of weight gain or sedative side effects (Miura et al., 2014).
Carbamazepine is less effective than lithium in preventing mood episodes in bipolar disorder. It is probably better suited to patients with mixed features (Weisler et al., 2004) and may be useful in combination with lithium, especially where there is marked mood instability. In practice, carbamazepine should be regarded as third/fourth line treatment. Serum levels should be monitored with long-term use, mostly for monitoring adherence or to avoid toxicity and side effects such as skin reactions. A derivative of carbamazepine, oxcarbazepine, has been trialled in a controlled maintenance study as an adjunctive treatment with lithium, and was found to reduce relapse into depression and decrease impulsivity. The evidence for efficacy of oxcarbazepine remains weaker than for carbamazepine (Pratoomsri et al., 2006; Vieta et al., 2008a).
It is noteworthy that there is no evidence for the use of other anticonvulsants such as gabapentin and topiramate in the long-term maintenance treatment of bipolar disorder.
Second generation antipsychotic agents
Research on the efficacy of second generation antipsychotics for bipolar maintenance is rapidly evolving, with current evidence supporting quetiapine and olanzapine in the prevention of manic and depressive relapse as monotherapy or adjunctive therapy (both Level I), but care should be taken especially with olanzapine because of its propensity for metabolic syndrome and because its effect on depressive relapse may not be substantive (Miura et al., 2014). Long-acting injectable risperidone and ziprasidone have some support in the prevention of both manic and depressive episodes (Level II) (Yatham et al., 2009), and paliperidone has been trialled in the prevention of mania (Berwaerts et al., 2012) and aripiprazole monotherapy has evidence for the prevention of manic relapse (Level II).
Despite a lack of RCT evidence, clozapine is widely regarded as an option for treating severe refractory bipolar disorder. However, its significant side effects and ongoing need for monitoring limit its use long term.
Evidence on the use of other atypical antipsychotic agents is emerging, but most studies are of insufficient duration to properly study the maintenance stage of bipolar disorder (McIntyre et al., 2010a). Many patients do not achieve remission with medication monotherapy. The combination of medications increases the risks of adverse interactions and the balance of risks and benefits must be considered. However bipolar disorders are highly disruptive to patients’ lives and cause considerable distress to patients and others, even when substantially recovered but not in remission.
Research on antipsychotic long-term use for all second generation antipsychotics recommends doses within the established recommended dose range for other indications, but it is desirable to keep the maintenance dose to the minimum effective level so as to prevent side effects. In practice, this requires clinical consideration of the risk of relapse and an estimate of the minimum effective dose needed once it is established that antipsychotic maintenance therapy is required. Usually, this is achieved by a gradual taper to the estimated minimum dose.
Prospective advances in long-term pharmacological treatment
A number of medications that have existing psychiatric indications are being trialled for use in the long-term treatment of bipolar disorder. These include lurasidone, a second generation antipsychotic that has been used in the treatment of schizophrenia. In a recent study it has shown efficacy in the treatment of acute bipolar depression, both as monotherapy and as an adjunct to lithium or valproate with better tolerability (Loebel et al., 2014a).
Paliperidone in extended release form is also being investigated for maintenance treatment of bipolar disorder. Early indications are that it is effective in delaying recurrence as compared to placebo, especially with respect to manic symptoms, with possibly little effect on depressive symptoms.
In addition to the prevention of relapse/recurrence, the reduction of residual symptoms is also an important outcome/endpoint. In this context, novel agents, agomelatine and ramelteon, that have an effect of stabilising circadian rhythms, could theoretically be used. Ramelteon has been used with some positive findings as an adjunctive agent to treat insomnia (McElroy et al., 2011; Norris et al., 2013) and agomelatine has had mixed findings for the treatment of bipolar depression (Calabrese et al., 2007; Fornaro et al., 2013; Yatham, 2015).
Psychological treatment in long-term management of bipolar disorder
Compared with the evidence base for psychological interventions for major depressive disorder, evidence for efficacy of psychological interventions for bipolar disorder is of low quality (small number of studies, inconsistency of methods and outcome measures, weak control conditions, elevated risk of bias, limited blinding, etc.). All studies to date have investigated psychological interventions as adjunctive to pharmacotherapy. To understand the literature, it is important to appreciate that (a) there has been almost no research comparing the evidence-based ‘brands’, and (b) all have significant shared content, aims and therapeutic process; consequently, existing guidelines refer interchangeably to ‘psychotherapies’ and specific psychotherapies (CBT, etc.).
There is Level I evidence for the effectiveness of structured psychological interventions as a set (group, individual and family-based) in preventing relapse of any kind, with one meta-analysis suggesting a 40% reduction in relapse compared to standard treatment alone (Scott et al., 2007). There is some evidence that relapse prevention is most effective for the depressive pole (Lauder et al., 2010).
Other reviews question the magnitude of effects across different outcome measures (Lynch et al., 2010; Szentagotai and David, 2010) but on balance the literature supports structured psychological interventions for improvement of depressive symptoms, delaying or minimising recurrences and hospitalisations, and enhancing psychosocial functioning (Goodwin, 2009; Kendall et al., 2014; Yatham et al., 2013b). The literature also indicates that psychological interventions are attractive to many people with bipolar disorder, do not generate negative effects, and have important secondary benefits for quality of life, and personal and functional recovery. For example, CBT has been found in a meta-analysis (10 studies) to improve medication adherence, decrease depressive symptoms and enhance quality of life and social adjustment (Szentagotai and David, 2010). Psychoeducation and CBT have shown strong effects in reducing the subjective burden of disease (Vieta and Colom, 2004; Zaretsky et al., 2007).
Existing adjunctive psychological interventions for maintenance treatment of bipolar disorder are based on a biopsychosocial diathesis-stress model and focus on empowerment, knowledge and skills (Mansell et al., 2005). They arise out of growing evidence that initial onset and course of bipolar disorder are moderated by social and psychological factors, particularly stressful events, family conflict, social and circadian rhythm disruption and medication non-adherence (Gilman et al., 2014; Miklowitz and Johnson, 2009). Research into moderators and mediators of outcome is limited, but there is some evidence that psychological interventions may be more effective when provided earlier in the course of the disorder (Geddes and Miklowitz, 2013) and outside acute episodes (Scott et al., 2007).
Across various outcomes, the best quality evidence is for the efficacy of structured psychological interventions in general, while evidence for specific psychological therapies is weaker. Importantly, the specific psychological therapies investigated to date have substantial content overlap (see: Table 22), and there is little evidence to support efficacy ranking of the ‘brands’. Consequently, guidelines often recommend offering any of the evidence-based treatments (e.g., NCCMH, 2014). As with guidelines for unipolar depression, absence of evidence differentiating the psychosocial treatments should not be taken as support for unstructured eclecticism. There is strong clinical consensus that psychosocial treatment is best guided by an evidence-based treatment manual, tailored to the individual client, and with proper attention to the critical therapeutic relationship.
Shared content elements of evidence-based psychological interventions.

In sum, evidence suggests that a structured psychological intervention should be part of long-term management of bipolar disorder, but does not determine which particular therapy to offer. To ensure that all key content elements are covered (Table 22), therapy should be informed by a published treatment manual for one of the four evidence-based therapies (Table 23). Choice of approach will then be influenced by the local availability of therapeutic expertise (training in, e.g., CBT, family systems, social rhythms, etc.).
Specific psychological interventions for bipolar disorder.
Four specific psychological interventions can be considered evidence-based (i.e., have at least one positive RCT), and have associated published manuals to guide treatment.

Translation and models of care
In the absence of evidence distinguishing the effectiveness of existing psychological interventions, ease of dissemination is a critical consideration. Psychoeducation is a relatively simple, cost-effective psychological intervention (Parikh et al., 2012). Brief psychoeducation (3–6 sessions, preferably in a group format) has been proposed as the first offering to all patients in a stepped care approach to management of bipolar disorder, with more intensive interventions reserved for more complex cases (Parikh, 2014). This suggestion aligns with preliminary evidence for the effectiveness of web-delivered, group-based, and other self-management interventions (Castle et al., 2010; Lauder et al., 2013; Murray et al., 2011b, 2015; Todd et al., 2014), and with cautions that the more complex formulation-based interventions should only be delivered by psychologically trained clinicians with expertise in bipolar disorder (Goodwin, 2009).
People with bipolar disorder can ‘fall between the gaps’ in mental health services, because of their fluctuating levels of functioning and the dearth of specialist psychological expertise around bipolar disorder. Two large RCTs have used managed-care cooperatives in the US to investigate collaborative care programmes, integrating pharmacotherapy, group psychoeducation and intensive patient monitoring by a nurse care manager (Bauer et al., 2006). Neither found significant reductions in relapse rates, but collaborative care was associated with fewer weeks in manic episodes, improved social functioning and quality of life.
Kessing et al. showed that, compared with standard decentralised care, patients receiving evidence-based psychoeducation and pharmacotherapy had improved readmission rates (Kessing et al., 2013). Specialised care was also associated with increased consumer satisfaction and greater mean costs per patient in the specialist clinic were more than offset by reduced readmission costs.
Some of the content elements shared across evidence-based therapies have been subjected to systematic review, with Level I evidence for both relapse prevention training and psychoeducation (particularly group-delivered) as effective in preventing any kind of relapse (Bond and Anderson, 2015; Morriss et al., 2007).
Electroconvulsive therapy
Electroconvulsive therapy can be invaluable as maintenance treatment for a small number of patients with otherwise treatment resistant bipolar disorder, including recurrent mania. Before such treatment is indicated, patients have usually had inadequate response from multiple appropriate pharmacological and psychosocial interventions. Maintenance ECT is usually administered weekly and the frequency of treatment is usually progressively reduced to treatments every four weeks. Maintenance ECT is usually well tolerated and cognitive impairment does not appear to be progressive during maintenance ECT (Level III).
Duration and discontinuation of maintenance treatment
Patients with bipolar disorder are best advised to use mood stabilising and prophylactic medication indefinitely to prevent future episodes of illness. The discontinuation of long-term treatment risks relapse/recurrence, but equally the long-term use of medications can lead to significant adverse effects. Therefore, a balanced discussion of both the benefits and negative consequences in conjunction with the individual and their carers is necessary at the outset of treatment and whenever a significant change is being considered.
Requests to discontinue medication from patients need to be dealt with promptly. One special circumstance is when a woman is considering becoming pregnant, in which case a detailed risk/benefit analysis should be conducted (see section Pregnant and breastfeeding women). Such requests usually reflect one or more of three concerns.
First, the current regimen is ineffective and the person has ongoing symptoms. This should prompt re-evaluation of medication, and its optimisation so as to alleviate ongoing symptoms.
Second, the side effects of medication are intolerable. In this instance medication dosage should be reviewed and tapered where possible, otherwise substitutions may be necessary.
Third, the individual feels they have recovered but because of continuing to take medication are reminded of their illness or fail to appreciate its preventative benefit. This requires detailed discussion and perhaps further psychoeducation.
Patient choice is paramount unless the patient is deemed not to have the capacity to make an informed decision regarding their illness and treatment. Therefore, ongoing psychoeducation about the risks and benefits of treatment discontinuation is essential. Note rapid discontinuation, which is more likely to occur when patients are disengaged and decide to ‘manage themselves’, incurs significant risks, especially of relapse/recurrence and possible suicide. This underscores the need for a highly effective therapeutic alliance.

In situations where a pattern of relapse and recurrence has not been shown, a carefully monitored gradual withdrawal of medication may be appropriate. This may be indicated for example, if planning a pregnancy. The empirical evidence to guide best practice in this instance is lacking and clinical experience is varied. In cases where a single catastrophic mood episode necessitated treatment, there may be reason for ongoing medication, even in the absence of an established pattern of relapse/recurrence. In instances where substance abuse or possible antidepressant-induced elevation obfuscated diagnosis, 12 a lower threshold may apply when considering medication withdrawal after a sustained period of euthymia.
Principles of monitoring
Regular contact with health professionals is essential for appropriate management, but the frequency of visits can vary considerably, and rapid access for additional consultations when early warning signs are emerging, or significant life changes are about to happen, should be allowed for. The frequency of contact is usually no less than biannual visits once long-term stability is achieved and/or only specialist management is required regarding the management of mood stability. Follow-up is more frequent depending upon the history of the illness and time since the last mood episode.
Monitoring sessions are important because they strengthen rapport and ensure the maintenance of an ongoing alliance, which provides an opportunity for additional psychoeducation, psychological interventions, life-style management, monitoring of blood levels and side effects, and continuing tailoring of treatment. In addition, patients will usually have more frequent follow-up appointments with other health professionals in their treating team, such as their general practitioner, psychologist, or case worker. Regular visits may gradually become less frequent if the illness remains in remission, but access to help in a crisis situation must be available to both the patient and significant others, and the plan of action should be known to the patient and all members of the treating team.
Patient self-monitoring is also essential and should be combined with psychoeducation and identification of early warning signs with an action plan to deal with them, particularly a plan to get rapid access to a psychiatrist (or other health professional). The use of mood charts to monitor mood is recommended, which can be a focus of discussion at monitoring visits. Recently wearable devices and ‘apps’ in mobile phones have become popular means of monitoring activity levels. These may have clinical applications in the near future for monitoring mood stability.
Many medications utilised in the long-term management of bipolar disorder are associated with significant metabolic effects that potentially contribute to the excess medical morbidity and mortality associated with bipolar disorders. In any patient with bipolar disorder, and particularly those on medications with known metabolic risks there is thus a requirement for regular monitoring of weight, abdominal girth, blood pressure and serum lipids and glucose. These measures should be performed at least annually and more frequently dependent on need. A range of guidelines exist for metabolic monitoring (see: Table 24 ) and these can readily be utilised to provide a planned process that is easily instituted. For some patients it may be possible for their GP to undertake monitoring and conduct the necessary tests but the treating psychiatrist should ensure these are conducted and take overarching responsibility.
Tests to be conducted and parameters to be monitored during long-term treatment of bipolar disorder.
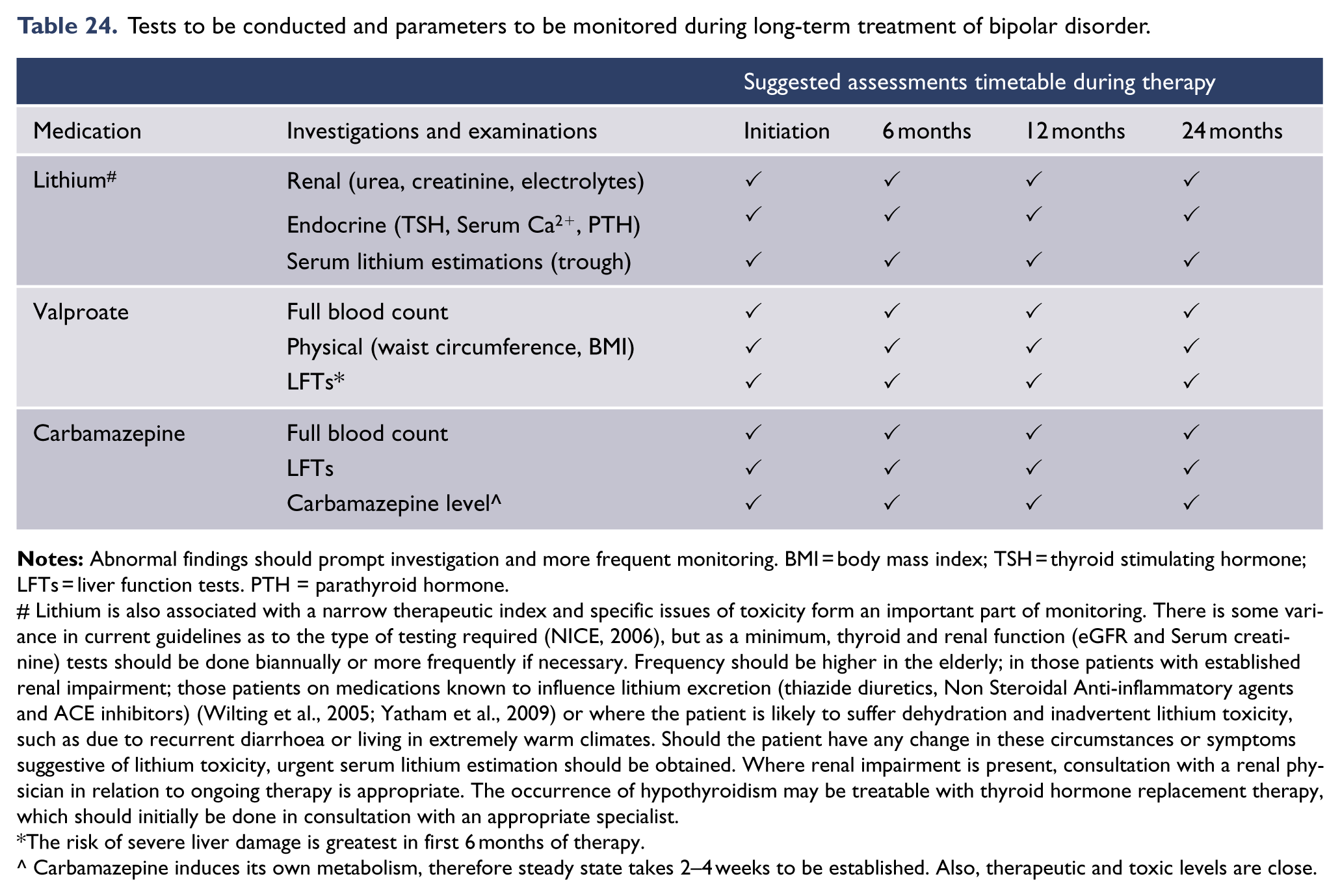
Lithium is also associated with a narrow therapeutic index and specific issues of toxicity form an important part of monitoring. There is some variance in current guidelines as to the type of testing required (NICE, 2006), but as a minimum, thyroid and renal function (eGFR and Serum creatinine) tests should be done biannually or more frequently if necessary. Frequency should be higher in the elderly; in those patients with established renal impairment; those patients on medications known to influence lithium excretion (thiazide diuretics, Non Steroidal Anti-inflammatory agents and ACE inhibitors) (Wilting et al., 2005; Yatham et al., 2009) or where the patient is likely to suffer dehydration and inadvertent lithium toxicity, such as due to recurrent diarrhoea or living in extremely warm climates. Should the patient have any change in these circumstances or symptoms suggestive of lithium toxicity, urgent serum lithium estimation should be obtained. Where renal impairment is present, consultation with a renal physician in relation to ongoing therapy is appropriate. The occurrence of hypothyroidism may be treatable with thyroid hormone replacement therapy, which should initially be done in consultation with an appropriate specialist.
The risk of severe liver damage is greatest in first 6 months of therapy.
Carbamazepine induces its own metabolism, therefore steady state takes 2–4 weeks to be established. Also, therapeutic and toxic levels are close.
Carers and the care of individuals with bipolar disorder
In the care of an individual with bipolar disorder, it is central to acknowledge the impact of the illness upon the affected individual, their family, and other carers. Equally, the carers form an integral part of the management team with their capacity to provide often-crucial additional information, and to assist in the implementation of interventions. It is further essential to acknowledge the impact of serious mental illness like bipolar disorder on carers, the level of stress that carers may experience and their heightened risk for the development of their own mental health problems such as anxiety or depression.
Involving carers from assessment and throughout management should now be considered standard care. At times this may lead to concerns in relation to the privacy of the patient, including occasional active discouragement of carer involvement by the patient. In practice, this is considerably less common than a failure by treating clinicians to consider involvement of carers and their needs and roles. Pragmatically most patients will welcome carer involvement and involvement of their carers will only enhance their recovery. It is thus appropriate to raise carer involvement early in therapeutic engagement. This discussion also needs to involve a consideration of any specific aspects that may be highly sensitive, such as issues related to substance abuse or other disinhibited behaviour during manic episodes.
The issues surrounding carer involvement and confidentiality are often particularly sensitive where capacity to consent to treatment is impaired and involuntary treatment is required. It is important to be aware that several jurisdictions have recently reviewed their mental health legislations to specifically deal with such situations. It is a requirement for practitioners to be familiar with the relevant legislation in their region.
Clinical management of mood disorders with complex presentations and in special populations
Thus far, these guidelines have addressed the management of relatively straightforward mood disorder presentations in adults. In practice, the identification of mood disorders is far more complex with subtypes of mood disorders manifesting throughout the life cycle, often with varied patterns, and usually accompanied by comorbidities, all of which warrant more sophisticated management.
Managing particular types of mood disorders
This section considers some of the recognised subtypes of mood disorders and the intricacies of management.
Mood disorder subtypes
It is important to note at the outset that there are no definitive kinds of mood disorders; even major depression can eventually manifest as bipolar disorder over time. However, some features of mood disorders do appear to co-occur and form characteristic syndromes – and these are termed subtypes.
Melancholia
Features
There is reasonable consensus that there is one subtype of depression that has a neurobiological basis that requires pharmacological treatment. However, the clinical features of this subtype of depression remain elusive; a consequence of the commonality of clinical features associated with all forms of depression. This has resulted in considerable debate about whether melancholia (endogenous or autonomous depression) is a distinct subtype of depression or whether it is a marker of severity (Parker and Hadzi-Pavlovic, 1996; Taylor and Fink, 2006).
While the clinical descriptors of this subtype of depression are elusive, there are some clinical features, treatment outcomes and biomarkers associated with it. Clinical features include: psychomotor change, anhedonia, affective non-reactivity, vegetative symptoms and cognitive changes. Treatment outcomes are superior with biological treatments (dual action antidepressants, ECT and a low placebo response). Biomarkers include: abnormal Dexamethasone Suppression Test or Corticotrophin Releasing Hormone test, high night time cortisol levels, reduced Rapid Eye Movement latency or another sleep related biomarker (such as reduced duration of Slow Wave Sleep).
Melancholia is a historically and conceptually important subtype of depression, with a putatively stronger biological basis than other unipolar depressive presentations (Hadzi-Pavlovic and Boyce, 2012; Parker and Hadzi-Pavlovic, 1996). It is characterised by psychomotor change, anhedonia, affective non-reactivity, vegetative symptoms and cognitive changes. In DSM-5, with melancholic features is one of the specifiers for depressive episode, characterised by anhedonia, non reactivity and 3 or more of: distinct quality of depressed mood; depression worse in the morning; early morning awakening; marked psychomotor change; significant anorexia or weight loss and excessive or inappropriate guilt. Major depressive disorder with melancholic features has been associated with elevated suicide risk in some studies (Grunebaum et al., 2004; Jeon et al., 2013), and because of these features, melancholic depression is an important subtype clinically.
Treatment
Despite much investigation, consistent empirical associations between melancholic features and superior response to antidepressant medication have not been established (Dodd and Berk, 2004; Georgotas et al., 1986; Mallinckrodt et al., 2005; Nierenberg, 2003; Peselow et al., 1992; Souery et al., 2007; Trivedi et al., 2006; Uher et al., 2011; Uher et al., 2012). However, most studies have not consistently stratified differential efficacy by depression severity, possibly limiting interpretation of these findings (Cameron et al., 2014; Fournier et al., 2010). Clinical opinion would suggest that serotonin noradrenalin reuptake inhibitors (SNRIs) and TCAs may have an advantage over SSRIs for this patient population (Clerc et al., 1994; Malhi et al., 2005; Perry, 1996; Singh et al., 2013), but SSRIs have also been shown to have efficacy (Yang et al., 2012). Psychotherapy may be less effective than medications in this group (Parker et al., 2013; Thase and Friedman, 1999), but combined medication and subsequently added CBT may provide optimal care (Gilfillan et al., 2014).
ECT appears to have preferential efficacy in severe refractory cases with melancholic features (Hickie et al., 1996; Pagnin et al., 2004; Parker et al., 1999), preliminary studies suggest that rTMS may also have benefit in this subtype of depression (Baeken et al., 2010, 2014).
From a clinical perspective, should melancholic features be pronounced it is reasonable to tend towards biological approaches, but remain mindful that integrated biopsychosocial care is needed for optimal functional recovery. Equally when the severity of depression is significant and melancholic features are pronounced, aggressive biological therapies are indicated and the recommended cornerstone of care.
Atypical depression
Features
Along with common symptoms of depression, atypical depression is characterised by reactive mood, marked fatigue and weakness, hypersomnia, hyperphagia with weight gain, and feeling extremely sensitive to rejection (Davidson et al., 1982). Atypical depression may be more common in bipolar depression (Akiskal and Benazzi, 2005; Baldessarini et al., 2010; Blanco et al., 2012; Mitchell et al., 2008).
Treatment
Cognitive therapy, antidepressants and ECT have been shown to be effective in treating atypical depression. A double-blind, randomised control trial studying the differential efficacy of the MAOI phenelzine versus cognitive therapy found both were superior to placebo, and each mode of treatment had a similar response rate of 58% (Jarrett et al., 1999). A lack of difference between medications and counselling was also noted in a retrospective study of 715 subjects (Parker and Crawford, 2009).
It has been proposed that atypical depression responds best to MAOIs. Four randomized controlled trials demonstrated superior efficacy for phenelzine compared to imipramine (Liebowitz et al., 1988; McGrath et al., 1993; Quitkin et al., 1990, 1991). A meta-analysis of 11 studies concluded that MAOIs may be more effective for atypical major depressive disorder than tricyclic antidepressants but there were insufficient data to demonstrate a differential response between MAOIs and SSRIs (Henkel et al., 2006). Conversely, patients with atypical depression appear less likely to remit with the SSRI citalopram than those without atypical features (Stewart et al., 2010; Trivedi et al., 2006). A recent study of 811 subjects with MDD treated with SSRI or tricyclic antidepressant (TCA) found atypical depression did not predict differential antidepressant response (Uher et al., 2011), indicating that TCAs and SSRIs appear to have similar efficacy.
ECT has been shown to be an effective treatment. A prospective study of 489 severely depressed patients treated with ECT demonstrated a superior response rate among patients with atypical features (80%) compared to typical depression (67%) (Husain et al., 2008).
Psychotic depression
Features
Psychotic depression typically refers to an episode of severe depression (melancholia) characterised by the presence of psychotic symptoms, most commonly delusions and/or hallucinations. It can also produce catatonia in which the individual loses motor skill and holds rigid poses for prolonged periods of time or becomes hyperactive and excitable (Fink, 2013).
Treatment
Psychotic depression has potentially dangerous consequences, frequently warranting intensive care (Vega et al., 2000). Combined use of an antidepressant and antipsychotic medication or ECT are the preferred treatments (Davidson, 2010; Lam et al., 2009; Nutt et al., 2010; Wijkstra et al., 2009). This is because antidepressant monotherapy in psychotic depression has lower efficacy than ECT or combined antidepressant and antipsychotic medication (Farahani and Correll, 2012), but the literature is mixed with regard to the optimal pharmacotherapy (Birkenhager et al., 2008; Künzel et al., 2009; Meyers et al., 2009; Parker et al., 1992; Rothschild et al., 2004; Simpson et al., 2003; Spiker et al., 1985; Wijkstra et al., 2010).
Most of the studies demonstrating the efficacy of the combination of antidepressants and antipsychotics have utilised tricyclic antidepressants and typical antipsychotics. Psychotic major depression appears to have a preferential response to ECT, making ECT a treatment of choice (Andrade et al., 1988; Parker et al., 1992). Similarly, ECT is an effective treatment in the context of psychotic bipolar depression. Catatonia necessitates careful organic screening, and is typically responsive to ECT, but there has been little study of major depression with catatonic symptoms probably because of the rarity of this subtype (Parker and Hadzi-Pavlovic, 1996). Following recovery, adequate maintenance treatment should be continued for 6–12 months or longer if the illness is recurrent. This will require ongoing combined antidepressant/antipsychotic medication or maintenance ECT depending on individual circumstances.
The role of TMS in psychotic depression has not been systematically examined, and a single case report noted a negative finding (Krisanaprakornkit et al., 2010; Ren et al., 2014). A recent meta-analysis has suggested superiority of ECT over TMS in psychotic depression specifically, with much less differentiation between the treatments in non psychotic depression.
CBT and other forms of psychotherapy should not be utilised as monotherapy in this group, but be employed in the recovery phase once gross mental state disturbances have been corrected with biological therapies such as medication and ECT.
Persistent depressive disorder (dysthymia)
Features
Persistent depressive disorder (dysthymia) is a consolidation of chronic major depressive disorder and dysthymic disorder. It describes a state in which the person experiences more days than not in a depressed mood for at least 2 years (mood can be irritable and duration can be 1 year in children and adolescents). During this time there has never been a symptom free period of 2 months or more. Typical symptoms include, 2 or more of, poor appetite or overeating, insomnia or hypersomnia, low energy or fatigue, low self-esteem, poor concentration or difficulty making decisions, and/or feelings of hopelessness. It is important to note that personality disorders and traits are common comorbidities with prolonged depression.
Treatment
Compared to major depression there is far less empirical data on optimal management of dysthymia. Nonetheless, these conditions do present clinically and may impair quality of life. A recent meta-analysis suggests that antidepressants are superior to placebo for the treatment of PDD, with SSRIs having better tolerability than TCAs (Levkovitz et al., 2011; Von Wolff et al., 2013). But the utility of antidepressants for subthreshold depressive symptoms is not established (Cameron et al., 2014), despite earlier data indicating antidepressants appear to have efficacy (De Lima et al., 1999). CBT appears less effective than SSRIs, but the combination of therapies appears more effective than either alone (Cuijpers et al., 2010b). Internet based CBT may be effective for minor depressive symptoms among older adults (Spek et al., 2008). Given the lack of clear efficacy of antidepressants in this group, psychosocial interventions should be encouraged prior to considering medications.
Cyclothymia
Features
Cyclothymic disorders (cyclothymia) describes prolonged states (1 year in children and adolescents, 2 years in adults) during which periods of subsyndromal hypomania and depressive symptoms have been present for the majority of the time (>50%) and there has been no period of more than 2 months when the individual has been symptom free.
Treatment
Despite the higher prevalence of bipolar spectrum disorders (Merikangas et al., 2007, 2011), cyclothymic disorder has the least empirical base to guide optimal management, because of greater focus on the more debilitating bipolar I and II disorders. As with bipolar II disorder, much of the clinical management of cyclothymic disorder is extrapolated from the management guidelines for bipolar I disorder. The validity of this is unclear, and the key clinical issue is the risk-benefit analysis between hazards of the untreated condition versus hazards from treatment. Where distress and dysfunction arising from cyclothymic disorder are mild, conservative non-pharmacological approaches alone should be trialled initially. Indeed, even with more severe symptoms it is unclear if pharmacotherapy is useful (Findling et al., 2007).
Research on psychosocial treatments for cyclothymia is limited. One randomised controlled trial found augmented CBT was effective for depressive and hypomanic symptoms, compared with clinical management (Fava et al., 2011). Over half those in the intervention group no longer met criteria for the disorder post-treatment, and gains were maintained at 1- and 2-year follow-up (Fava et al., 2011).
However, cyclothymia may be a precursor to bipolar I or II disorder and, therefore, longitudinal mental state review and psychoeducation of patients as to when to seek further review is advisable (Alloy et al., 2012). Family involvement may be particularly beneficial in the care of such patients (Pfennig et al., 2014). Borderline personality disorder needs to be considered as a potential differential diagnosis in some cases (Bayes et al., 2014).
Bipolar II Disorder (BD II)
Features
BD II differs from BD I along a dimension in which the severity of manic features become progressively more severe with respect to elevation of mood (euphoric or irritable), intensity of energy, disruption of function (both in mental state and daily activities) and the recognition by others that a mental state problem is present. The boundaries of hypomania are ill defined (Malhi et al., 2010) but the absence of psychosis, and the absence of severe life disruption because of manic symptoms (Malhi and Berk, 2014), are useful parameters. BD II is also characterised by the predominance of depressive features, both as discrete depressed episodes and as mixed affective states. The impact of BD II upon quality of life is at least as substantial as with BD I, if not greater because of the tenacity of chronic depressive features (Judd et al., 2003b). The risk of suicide is also high and comparable with BD I (Holma et al., 2014; Novick et al., 2010). No neurobiological differences with BD I have been established, further blurring the distinction between these two forms of bipolar disorder.
Treatment
Optimal management of BD II has been much less studied than BD I disorder, with many BD II treatment recommendations ‘extrapolated’ from the BD I literature (Tamayo et al., 2013). For this reason, the management of BD II disorder clinically (currently) mirrors that of BD I except that the focus is more upon depressive episodes and residual depressive and mixed symptoms, than mania. Lithium and sodium valproate are less suited to relieving mood instability in BD II disorder as compared to their role in BD I disorder. In selecting therapies, attention should be paid to the balance of hazards of the untreated condition versus hazards from treatment. For this reason, biological therapies with lower toxicity profiles are generally preferred in managing BD II disorder, but by the same token risk of suicide in the depressive phase should not be underestimated. Mood stabilisation is critical for prophylaxis and alongside pharmacotherapy, psychotherapies appear to be of value (Berk et al., 2010; Lauder et al., 2010; Schöttle et al., 2011).
Indeed, because of the predominance of depressive symptoms (for which psychosocial treatments are effective in MDD), we might expect psychological treatments to have particular application in BD II. Research on this issue is limited.
Post-hoc analyses of two large RCTs suggest that adjunctive psychosocial interventions effective in BD I are equally effective in BD II. Decreased relapses and symptom load have been found amongst BD II patients receiving adjunctive group psychoeducation (Colom et al., 2009). In the large STEP-BD trial, cognitive behaviour therapy (CBT), interpersonal and social rhythm therapy (IPSRT) and family-focused therapy (FFT) were (equally) superior to collaborative care in accelerating recovery from a depressive episode and maximizing the probability of remaining well across 12 months: No main effect of diagnosis (BD I vs. BD II) was found (Miklowitz et al., 2007). One small randomised trial of acute Bipolar II depression found IPSRT monotherapy to be equivalent to quetiapine monotherapy (Miklowitz et al., 2007). In sum, as with pharmacological intervention, clinicians are asked to cautiously generalise evidence-based psychological treatments from BD I to BD II.
Lamotrigine has received considerable support from many clinicians and some controlled trials (Bowden and Singh, 2012; Reinares et al., 2013; Selle et al., 2014; Sienaert et al., 2013; Trankner et al., 2013), in the management of bipolar depression. Whilst lamotrigine has been found to be effective for some patients with BD II, for other patients it may not provide any benefit. Atypical antipsychotic medications such as quetiapine, olanzapine and lurasidone, have shown significant benefits in bipolar depression (Level II) (Azorin et al., 2013; Bawa and Scarff, 2015; Janicak and Rado, 2011; Loebel et al., 2014a; Sanford and Dhillon, 2015; Sanford and Keating, 2012; Tohen et al., 2013; Vieta and Valenti, 2013). Asenapine may also be helpful (Azorin et al., 2013). The use of antidepressant medications is a controversial issue and perhaps more so in BD II because depressive features are so much more prominent. However, expert opinion recommends caution in their use (Mauer et al., 2014; Pacchiarotti et al., 2013a). The risks of rapid cycling, mixed states and switching into mania, are potential consequences, and efficacy is sometimes disappointing. Agomelatine, pramipexole and modafinil have found some supportive evidence but remain of uncertain value beyond intervention for treatment resistant patients (Fornaro et al., 2013), and there is some preliminary evidence that adjunctive NAC may be of benefit in the treatment of BD II (Magalhaes et al., 2011).
Mood disorders patterns
The description of mood disorders is largely cross-sectional but the course of illness is equally important. Some patterns such as mixed states/features defy classification and therefore require separate consideration (see Figure 18).
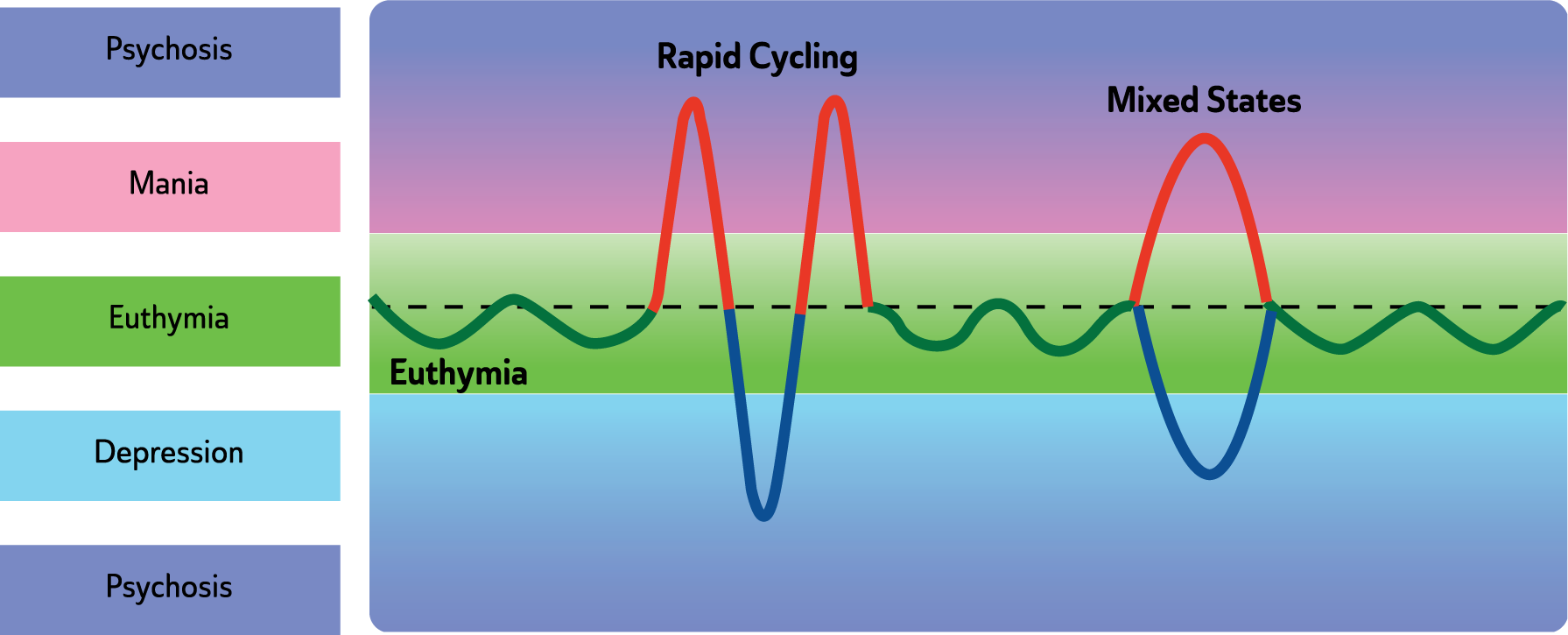
Rapid cycling and mixed states in bipolar disorder.
Mixed features
Features
In DSM-5, a ‘mixed features’ ‘specifier’ is used to indicate the occurrence of three or more symptoms from the opposite pole. Therefore depression, mania and hypomania can each be further specified with ‘mixed features’. This change is recent and therefore little is known about the diagnosis and treatment of mixed features presentations as defined by DSM-5 (Malhi, 2013, 2014). Earlier studies that examined DSM-IV mixed episodes rarely studied these separately. Usually, mixed episodes have been grouped alongside acute mania. Therefore, treatment guidelines for mixed features draw heavily on clinical experience and consensus recommendations.
Treatment
For depressed patients with mixed features, taper and cease substances with a mood-elevating effect or those that may induce inter-episode switching (e.g., antidepressants, stimulants). Consider treating the patient with olanzapine (Level II) (Tohen et al., 2014), quetiapine, or valproate as monotherapy or in combination with an antidepressant, or valproate in combination with olanzapine (Level II) (Benazzi et al., 2009). Lamotrigine adjunctive to an antimanic agent may be a useful option for mixed states to treat depressive symptoms (Krüger et al., 2005; Swann et al., 1997), though the therapeutic effect is likely to be delayed as this requires slow titration, especially when administered in conjunction with valproate. If symptoms and/or behavioural disturbance are severe or protracted, then consider ECT (Level III) (Valenti et al., 2008).
The mixed features specifier in DSM-5 for mania or hypomania emphasises dysphoria or depressed mood, loss of interest or pleasure, psychomotor retardation, fatigue, feeling of worthlessness or guilt and recurrent thoughts of death being present during an episode. There are limited trials of specific medications to use for acute dysphoric manic states with most of the data coming from retrospective analyses of trials of mania that have included those with mixed states.
For manic patients with depressive features (dysphoric mania), antidepressants should be prescribed with care, as these may aggravate the mixed state or promote rapid cycling (Fountoulakis et al., 2012b; Pacchiarotti et al., 2013b; Swann et al., 2013; Valentí et al., 2011).
Second generation antipsychotic medications, in conjunction with a mood stabiliser (Muralidharan et al., 2013) are more effective than mood stabilisers alone and are recommended for use in dysphoric mania, with Level II evidence for asenapine (McIntyre et al., 2013; Muralidharan et al., 2013; Ouanes et al., 2014), olanzapine (Fountoulakis et al., 2012b; Muralidharan et al., 2013; Ouanes et al., 2014), aripiprazole, ziprasidone and risperidone (Muralidharan et al., 2013). There is an absence of evidence regarding the use of quetiapine for the acute treatment of mixed states.
With regard to maintenance therapy, valproate appears to be more effective than lithium (Krüger et al., 2005), but the evidence regarding carbamazepine is equivocal.
Rapid cycling bipolar disorder
Features
Rapid cycling bipolar disorder refers to the presence of at least four mood episodes of mania, hypomania or depression in the previous 12 months. Described in the early 1970s as a course specifier for bipolar illnesses (Dunner and Fieve, 1974) a rapid cycling pattern is relatively common – and affects almost one third of bipolar disorder patients. In a proportion of these patients rapid cycling is associated with female gender, hypothyroidism, depression, greater illness severity and longer illness duration (Carvalho A et al., 2014; Fountoulakis et al., 2013). Clinically, rapid cycling is a transitional stage of bipolar disorder that can be easily confused with mixed states. The presence of rapid cycling complicates management and increases the risk of suicide attempts and greater depressive morbidity (Coryell et al., 2003) partly because of poorer treatment adherence (Perlis et al., 2010; Schneck, 2006; Tondo et al., 2003; Vieta et al., 2004). It is important to screen for rapid cycling, and where possible, address factors that may precipitate or exacerbate cycling, such as antidepressants, substance misuse, and medical illness, such as hypothyroidism (Schneck, 2006).
Treatment
Rapid cycling has significant implications for the treatment of depressive symptoms, with the focus being more on the long term stabilisation of mood than the treatment of the particular episode.
The evidence-base for rapid-cycling is limited and overall treatments are less effective in countering depressive symptoms than manic symptoms (Calabrese et al., 2003; Schneck, 2006). Choice of treatment is governed by the current mood state and the need to prevent cycling (Carvalho et al., 2014; Fountoulakis et al., 2013). Pharmacological monotherapy options include valproate (Level II), lithium (Level II), olanzapine (Level II) (Sanger et al., 2003), lamotrigine (Level II) (Calabrese and Delucchi, 1990; Calabrese et al., 1993, 2000, 2005a; McElroy et al., 1998; Tondo et al., 2003; Vieta et al., 2004), quetiapine (Level III) (Goldberg et al., 2008), aripiprazole (Muzina et al., 2008). There is limited evidence for medication combinations but where necessary combinations, such as lithium and valproate (Calabrese et al., 2005c) (Level III), lithium and carbamazepine (Level III) (Denicoff et al., 1997), or adjunctive lamotrigine (CBR) (Calabrese et al., 2005a) can be trialled. Adjunctive psychological interventions (CBR), as discussed may also be of benefit. Similarly, ECT has been found to be effective in some cases and can be trialled when necessary (Level III) (Calabrese et al., 2001; Papadimitriou et al., 2007).
Seasonal patterns
Seasonal effects on mood have been discussed since antiquity, and there is some scientific evidence for seasonal variation in mood: a prospective study in the general population found some 2% of variance in positive affect to be explained by decreases in winter and increases in spring/summer (Murray et al., 2001). It has also been hypothesised that objective seasonal factors (particularly decreased daylight in winter) are causal in episodes of mood disorder. A strong version of this hypothesis is that some forms of mood disorder are predominantly explained by objective seasonal factors: Seasonal Affective Disorder (SAD), first defined in the 1970s, is such a hypothesis.
Winter pattern SAD (winter depression) is characterised by episode onset in autumn/winter and remission in spring/summer (Rosenthal et al., 1984). Symptoms are generally mild to moderate in severity, and include hypersomnia with difficulty waking and daytime fatigue, appetite increase, carbohydrate craving and weight gain. Bright light is the first line treatment for winter pattern SAD, with effect sizes comparable to antidepressant medication in nonseasonal depression (Golden et al., 2005) and parallel benefits for quality of life (Michalak et al., 2007). There is some evidence that light treatment can be effective at lower intensity if the source emits blue-enriched light (Meesters et al., 2011). Guidelines for bright light treatment of winter SAD with an MDD diagnosis have been published, and there is growing research interest in bright light for depressions in BD (Benedetti, 2012).
Evidence that SAD also responds to antidepressants (Lam et al., 2006) and CBT (Rohan et al., 2009), while nonseasonal depressions also respond to bright light (Golden et al., 2005) raises doubts about SAD as a distinct clinical entity. Indeed, DSM has never accepted SAD as a diagnosis, recognising instead that some people with recurrent mood disorders have seasonal patterns to their recurrence. Although autumn/winter pattern depressions are most typical of the seasonal pattern course specifier, DSM recognises any seasonal pattern not explained by psychosocial factors. In DSM-5, the seasonal pattern specifier can be applied independently to any of depressive, manic or hypomanic episodes.
Seasonal pattern of mood episodes has a complex relationship to mood disorder diagnosis (Roecklein et al., 2010). It is slightly more common amongst people with BD (15–22%) than MDD (10–20%); amongst people with BD, seasonality may be associated with BD II and depressive polarity; amongst people with winter SAD, 80–90% warrant a diagnosis of MDD, while 10–20% warrant a BD diagnosis. Seasonal patterns of symptoms vary between people and within people across time: In BD populations, rigorous prospective studies have failed to find coordinated seasonal patterns in depression or mania (Murray et al., 2011a).
Management of mood disorders in the context of comorbidities
It is important to remember that unusual or atypical presentations may reflect concomitant psychopathology because of comorbid psychiatric or medical illness. Furthermore, new symptoms in patients already engaged in treatment may be a consequence of medication or psychological interventions.
Anxiety
Depression with comorbid anxiety
Depression with comorbid anxiety is a common clinical presentation (Malhi et al., 2002). While antidepressant medications have evidence of efficacy in this group, outcomes are often poorer than for subjects without comorbid anxiety (Papakostas et al., 2012; Souery et al., 2007; Tollefson et al., 1994). This is also true for psychotherapeutic approaches (Deckersbach et al., 2014). Nonetheless, second generation antidepressants appear to have some efficacy in this patient group (Fawcett and Barkin, 1998; Rudolph et al., 1998; Tourian et al., 2010) with a recent study suggesting agomelatine may have superior efficacy (Stein et al., 2013). Research also supports the use of psychological therapies (Cape et al., 2010), but head-to-head and combined psychological and pharmacological treatment studies are lacking. In less severe and/or less refractory cases, non-pharmacological approaches are preferred due to better risk/benefit profiles. Long-term use of clonazepam in otherwise refractory cases of depression with marked anxiety (especially panic) have been reported, but constitute the exception rather than the rule due to risks of tachyphylaxis and impaired alertness (Nardi et al., 2012; Tesar et al., 1987; Winkler et al., 2003). Nonetheless, clinically it seems there is a small proportion of patients with otherwise disabling depression and panic symptoms who do well with long-term clonazepam without tachyphylaxis or side effect burden (Nardi et al., 2012). Before such an approach is considered, psychiatric opinion is advisable.
Bipolar disorder with comorbid anxiety
Anxiety symptoms and disorders are commonly present in bipolar disorder either as comorbid conditions or a component of bipolar disorder itself (McIntyre et al., 2006; Schaffer et al., 2012) and are linked to a poorer outcome (Freeman et al., 2002; Kim et al., 2014). When an anxiety disorder is present along with bipolar disorder, the first aim of management is to achieve mood stabilisation (Freeman et al., 2002). There is limited evidence for specificity of treatments for treatment of comorbid anxiety disorders in bipolar disorder, especially for psychological treatments and serotonergic antidepressants. Psychological interventions for anxiety comorbidity in bipolar disorder have great potential, given the challenges of adding anxiolytics to mood stabilising pharmacotherapy regimens. Studies to date have investigated psychological interventions targeting both anxiety and bipolar disorders (N = 7), specific anxiety disorders in bipolar disorder populations (N = 7) and bipolar disorder only but reporting on anxiety outcomes (N = 8). Research has overwhelmingly focused on the inter-episode period, and has found significant if modest benefits with no evidence of harm (Stratford et al., 2015). The set of studies are extremely heterogeneous, but CBT for bipolar disorder augmented with anxiety strategies may have the strongest evidence.
Pregabalin has been shown to have modest efficacy in the treatment of comorbid anxiety and depression (Stein et al., 2008), and there is evidence for the efficacy of the second generation antipsychotic medications such as quetiapine (Sheehan et al., 2013) and olanzapine (Level II) (Suppes et al., 2014) but not risperidone (Sheehan et al., 2009) and the anticonvulsant mood stabilisers (sodium valproate and lamotrigine (Level II)) for the treatment of comorbid anxiety disorders (Stein et al., 2008). Part of risk/ benefit analysis in using atypicals to manage anxiety comorbidities is to consider risk of metabolic syndrome and balance this against risk of other treatment options to control anxiety.
Based on clinical experience, short-term use of benzodiazepines (less than 4 weeks) may be of benefit for severe anxiety symptoms prior to mood stabilisation being achieved, but supportive research evidence is lacking (Bobo et al., 2014).
Substance use
Comorbid substance use and mood disorder is often seen in clinical practice. Substance use may cause, mimic, underlie or complicate mental health disorders. It is important to ensure that the symptoms of a mood disorder are not attributable to either the physiological or pharmacological effects of a substance or to signs and symptoms of substance withdrawal.
Comorbid substance use may be a consequence of:
The underlying primary mood disorder, reflecting attempts to ‘self-medicate’ mood symptoms. However, eventually depressive symptoms may emerge because of the depressogenic effects of the substance or because the mood disorder has taken hold. In essence, there is a complex and interactive relationship between substance misuse (especially alcohol) and depressive symptoms.
The mood disorder being a psychiatric complication of substance use (e.g. euphoria during the initial phase of psychostimulant use followed by depression during the ‘crash’ phase). It is noteworthy that co-morbid alcohol and substance use disorders are associated with a worse course of bipolar disorder including more mood symptoms, recurrent mood episodes and decreased likelihood of recovery, more suicides, more hospitalisations and higher levels of aggressiveness (Suppes and Cosgrove, 2014).
If appropriate assessment, diagnosis and management of substance use are not undertaken, comorbid substance use may result in incorrect diagnosis and ineffective treatment of mental health disorders such as depression or bipolar disorder (Gao et al., 2010; Worthington et al., 1996). The risk of suicide increases, as does the probability of hospitalization and of aggressive behaviour. Importantly, many patients with comorbid bipolar disorder and substance abuse need to recover from elements of their mood disorder before their substance abuse can be usefully addressed.
For this reason, a full assessment and diagnosis of substance use and its management including detoxification is necessary, ideally prior to diagnosing and treating underlying mood disorders both to enable accurate diagnosis and effective therapy (Magidson et al., 2013). However, it is important to note that in many instances mood disorder patients with comorbid substance dependency will not be able to shift their usage until aspects of their mood disorders are contained. Often in outpatient practice settings, treatment of the mood disorder and substance use disorder needs to occur concurrently.
Treatment of depression with comorbid substance use disorder
A meta-analysis of comorbid substance use disorder with major depression found depressive symptoms did not significantly improve with SSRI treatment, but other classes of antidepressants (notably TCAs) may have some efficacy (Torrens et al., 2005). No antidepressant effect was found in comorbid cocaine dependence or opioid dependence (Torrens et al., 2005). Interestingly, venlafaxine has been shown to be ineffective in treating depressive symptoms comorbid with cannabis use, and may in fact be associated with greater cannabis use (Levin et al., 2013).
Patients with nicotine dependence and depression may potentially benefit from bupropion or nortriptyline, possibly because these agents are associated with greater smoking cessation rates (Hughes et al., 2007). Special caution is needed when prescribing antidepressants to patients who misuse stimulant drugs, as there may be heightened risk of serotonin syndrome (Silins et al., 2007). Among patients with hepatic impairments (e.g. alcoholic cirrhosis, hepatitis C from IV drug use) desvenlafaxine may offer some benefit over antidepressants which are metabolised by the liver (Baird-Bellaire et al., 2013).
Treatment of bipolar disorder with comorbid substance use disorder
Substance use disorders are commonly comorbid with bipolar disorders and rates as high as 67% have been described. Bipolar disorder with comorbid substance use is associated with greater illness morbidity (Mazza et al., 2009) and mortality (Level IV evidence: (Yoon et al., 2011)).
Empirical evidence to guide optimal management of bipolar disorder and comorbid substance abuse is scarce, partly because such patients are difficult to engage in trials (Malhi et al., 2012a). One study suggests that integrating CBT with group drug counselling has better outcomes than group drug counselling alone (Weiss et al., 2009). Alcohol has been studied more than other comorbid substances, but again the evidence base to guide clinicians remains poor (Azorin et al., 2010).
Use of valproate may help reduce alcohol consumption in some patients (Salloum et al., 2005; Sattar, 2007) with possible additional benefit if naltrexone is added (Salloum et al., 2006). Adjunctive quetiapine may help reduce alcohol use comorbid with bipolar disorder (Martinotti et al., 2008), but the limited literature is mixed (Brown et al., 2008), preventing empirical guidance. Importantly, patients with bipolar disorder have a five-fold greater chance of smoking compared to the general public (Diaz et al., 2009). Identifying nicotine dependence among bipolar patients and attempts to tackle it should form a routine element of patient care especially as treatments targeting smoking cessation are now emerging (Chengappa et al., 2014; Evins et al., 2014).
Appropriate assessment, diagnosis and management of alcohol or substance use disorder prior to mood therapy are advisable to best inform underlying diagnosis and best enhance odds of mood therapy efficacy. This may require a period of inpatient care in some instances, and engagement with formal drug and alcohol relapse prevention programs to encourage sustained abstinence. Ongoing comorbid substance misuse has a poor prognosis with mood disorder. A variety of strategies may be employed including supportive programmes (telephone, group), nicotine supplements and varenicline (Chengappa et al., 2014; Evins et al., 2014).
Personality disorder
Personality disorders are common in patients with major depression, with studies reporting prevalence rates ranging from 43–58% (Mulder, 2004). Cluster C personality disorders (avoidant, dependant and obsessive compulsive) are most commonly associated with major depressive disorders (Friborg et al., 2014). Depressed patients with a comorbid personality disorder have twice the chance of poor outcome with an OR 2.16 (CI 1.83 to 2.56) (Level I) (Newton-Howes et al., 2014). There is some evidence SSRIs may be superior to TCAs in depressed patients with comorbid Cluster B personality disorders (Level II) (Mulder et al., 2003), and preliminary evidence suggests CBT may be more effective than IPT in patients with comorbid major depression (Level II) (Joyce et al., 2007).There is consistent evidence that active treatment of major depression is associated with improvement in personality disorder symptoms (Fava et al., 2002; Mulder et al., 2010) Overall, however, there is insufficient evidence to formulate evidence-based recommendations in the management of co-occurring mood disorders in patients with personality disorders.
Borderline personality disorder and mood disorders
Clinical features
It is common for people with Borderline Personality Disorder to also suffer from a Major Depressive Disorder, and Bipolar Disorders (more commonly BD II) are present in about 9–16% of people with a Borderline Personality Disorder (Paris et al., 2007). This comorbidity amplifies the symptoms of each disorder and increases the time before remission (Gunderson et al., 2014). The presence of Borderline Personality Disorder with Major Depressive Disorder increases interpersonal difficulties (Yoshimatsu and Palmer, 2014), while the co-occurrence of Borderline Personality Disorder and Bipolar Disorder is associated with more impulsive behaviour and transient dissociative symptoms (Zimmerman et al., 2013, 2014). It is important to note that the concurrent treatment of both Borderline Personality Disorder and mood disorders offers a better probability of improvement in both sets of illness symptoms and in function (Beatson and Rao, 2013; Gunderson et al., 2014; Paris and Black, 2015; Yoshimatsu and Palmer, 2014).
The relationship between Borderline Personality Disorder and Bipolar Disorder remains subject to controversy, but the consensus is that they are distinct disorders (Antoniadis et al., 2012; Bassett, 2012; Belli et al., 2012; Coulston et al., 2012; Yoshimatsu and Palmer, 2014). Possible over-diagnosis of BD II can arise as a result of the overlap of symptoms between Borderline Personality Disorder and BD II. Table 25 summarizes the similarities and differences between Mood Disorders and Borderline Personality Disorder. Accurate diagnosis is based upon a detailed longitudinal history, with emphasis upon the accurate assessment of hypomanic episodes, and interpersonal and family history (Bayes et al., 2014; Ghaemi and Dalley, 2014; Paris and Black, 2015). The management of co-morbid occurrence must therefore address the specific needs of each patient, with attention to the contributions of each of these disorders, while accepting a degree of commonality.
Summary of the relationships of mood disorders and borderline personality disorder.
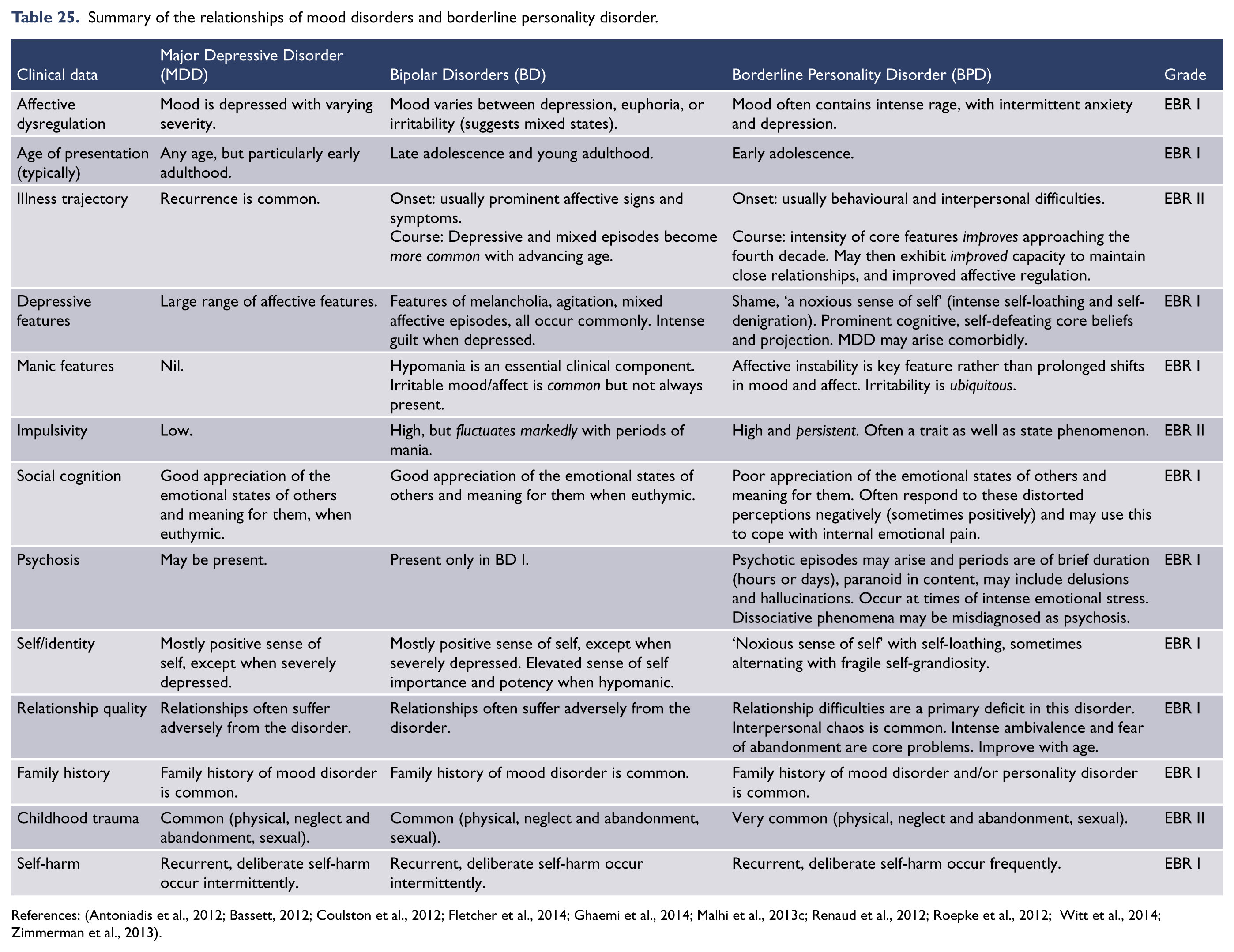
Psychotherapy
Psychotherapy is fundamental to the management of Borderline Personality Disorder and of considerable importance in the management of mood disorders. The approach in psychotherapy is different with Borderline Personality Disorder from mood disorders, requiring close attention to the disrupted sense of self and the development of a capacity to establish and maintain close, meaningful relationships. However, the management of patients suffering both disorders at the same time has not been the subject of study. While no specific form of psychotherapy has been shown to be optimal in efficacy in Borderline Personality Disorder, Dialectical Behaviour Therapy (DBT) is particularly useful (Bateman et al., 2015), and Psychodynamic Psychotherapy can be helpful. Cognitive and Behavioural Therapies (CBT) are of particular value in Major Depressive Disorder and Bipolar Disorders. The cognitively orientated therapies (such as DBT and CBT), tend to provide benefit within months, while the transference orientated therapies (such as Psychodynamic Psychotherapy) tend to require periods of years to provide significant benefits. Patients in both groups of disorders respond to supportive psychotherapies and both can benefit from DBT and CBT (Bassett, 2012; Bayes et al., 2014; Belli et al., 2012; Friborg et al., 2014; Ghaemi et al., 2014; Martín-Blanco et al., 2014; Zimmerman and Morgan, 2013). There is evidence that comorbid Borderline Personality Disorder and Bipolar Disorder increases the risk of suicide attempts (Zimmerman et al., 2014). Therefore, it is important to diagnose both conditions when they are present, because they identify the comorbid patients who are at particularly high risk for suicide attempts.

Pharmacotherapy: Borderline Personality Disorder
Medications have limited benefits in the management of Borderline Personality Disorder, with atypical antipsychotic agents having the best established efficacy (Ripoll, 2013; Stoffers et al., 2010). Atypical antipsychotics and mood stabilzers (including lithium, sodium valproate and lamotrigine) can be valuable to assist with concurrent Bipolar Disorders, affective instability, impulsivity and aggression. Antidepressants and atypical antipsychotics have value when Borderline Personality Disorder is comorbid with Major Depressive Disorder.
Schizophrenia
Co-occurring depression is common in patients with schizophrenia. In the Australian Survey of High Impact Psychosis, 55% of patients with a psychotic disorder, such as schizophrenia, had depressed mood in the year prior to the survey (Morgan et al., 2012). When a patient with schizophrenia presents with significant depressive symptoms, a number of differential diagnoses need to be considered (Siris, 2000): first, rule out the presence of medical factors contributing to depressed mood (e.g., cardiovascular disorders or cancer); second, the role of prescribed drugs, such as beta-blockers or the use of illicit substances, including alcohol; and third, the role of dopaminergic blockers in the development of dysphoric symptoms, given that antipsychotics block dopamine receptors with high potency potentially disrupting activity in reward related brain pathways, resulting in anhedonia or dysphoric symptoms.
Another important diagnostic consideration is the overlap between depression and the negative symptoms of schizophrenia (Fitzgerald et al., 2002). The presence of cognitive features of depression, such as guilt, hopelessness and suicidal thoughts, along with distinct sadness rather than blunting of affect are more suggestive of depression.
Post-psychotic depression has been used to describe the onset of depressive symptoms following the resolution of an acute psychotic episode (Birchwood et al., 2005). Given the particularly traumatic nature of psychosis and the disruption this has on patients’ lives, developing a stress-related reaction including symptoms of post-traumatic stress disorder or depression is likely to be a relatively common occurrence in patients, especially those experiencing traumatic events around the time of hospitalisation. At any point in time, the impact of life stressors, the impact of chronic illness and stigma and discrimination associated with the diagnosis of schizophrenia should be considered as possibly related to emerging depressive symptoms.
Finally, in patients who are relatively clinically stable, it is possible that the onset of depressive symptoms is a prodrome of psychotic relapse (Siris, 2000). This might be accompanied by other non-specific symptoms, such as anxiety. Evaluation requires a careful exploration of the possible presence of subtle subthreshold psychotic symptoms which would confirm that the mood change is related to a relapse of psychosis.
The management of emerging depression in a patient with schizophrenia will take all of these factors into account whilst ensuring any emerging risks are appropriately managed. Once organic factors are excluded, any suggestion of emerging psychosis should be treated appropriately with antipsychotic medication (Siris, 2000). The patient should be provided appropriate support and monitoring including supportive psychotherapy to address illness and lifestyle related stressors. The dose and type of ongoing antipsychotic medication should be reviewed with possible dose reduction or switch to a less potent dopamine blocker considered if this is thought to be an aetiological factor.
The role of antidepressant medication in the treatment of depression and schizophrenia has been evaluated by a series of clinical trials, although most of the clinical trials have been conducted with tricyclic antidepressants being co-prescribed with older first generation antipsychotic medications. A meta-analysis of this data was equivocal reflecting the lack of substantial studies (Whitehead et al., 2002). Studies, however, have suggested that antidepressant medications may reduce suicide in patients with schizophrenia (Tiihonen et al., 2012; Zisook et al., 2010). Lithium has also been suggested as a potentially useful agent in the treatment of depression and schizophrenia although this is not supported by substantial clinical trials (Siris, 2000).
Attention-deficit/hyperactivity disorder
Attention-Deficit/Hyperactivity Disorder (ADHD) is a disease characterised by attention-deficit, impulsivity and hyperactivity (see also: Children and adolescents). It is known that 3% to 7% of school-age children have ADHD globally (Kessler et al., 2006). The relationship between ADHD and Major Depression has been well examined with clear evidence that the presence of childhood and adult ADHD is associated with an increased incidence of depression throughout the lifespan. For example, a major North American national survey of healthy children aged 6–17 years reported depression in 14% of ADHD children versus 1% of those without ADHD (Larson et al., 2011). The incidence of depressive disorders in adolescents with ADHD ranges from 12% to 50% (Angold et al., 1999), and the likelihood of developing depression is five times greater in adults with ADHD compared to adults without ADHD. The pathway from ADHD to Major Depression often progresses through behaviour disorders (more often in males) and anxiety (more often in females) (Burke et al., 2005; Roy et al., 2014). Adults with ADHD also experience an earlier onset of depression, increased morbidity, more severe symptoms and a higher suicide rate compared with adults without ADHD (Nierenberg et al., 2005).
A prospective population study found higher rates of emergent bipolar disorder amongst individuals with a childhood diagnosis of ADHD than those without ADHD (Chen et al., 2013). Data suggest that 9 to 21% of adults with bipolar disorder also have comorbid ADHD (Kessler et al., 2006; Marin et al., 2013; Nierenberg et al., 2005). Diagnostic differentiation between the symptoms of ADHD and bipolar disorder can be difficult. What is thought to be ADHD could be an early expression of the emergent bipolar disorder. This tension was emphasised in a study of inpatients with recent bipolar disorder relapse, of whom 82% scored positively on a standardised diagnostic instrument for a diagnosis of lifetime ADHD (Marin et al., 2013). When a childhood history was taken carefully, this figure reduced to 26%, still a significant proportion. In the absence of a clear diagnostic marker, careful exploration of history of symptomatology, inter-episode functioning and the presence or absence of contemporaneous evidence of ADHD in childhood remain the most useful clinical tools.
Data on the effective treatment of mood disorders in the presence of comorbid ADHD is generally lacking. The treatment of depression in the setting of comorbid ADHD is generally more difficult and treatment resistance is more common (Biederman et al., 2008; Nierenberg et al., 2005). This underlines the importance of identifying comorbid ADHD as early as possible. The Texas Algorithm Project provides a guide to the treatment of ADHD comorbid with depression. In the first instance treatment is directed at the more severe of the two conditions, but the algorithm allows for combination therapy when needed (Pliszka et al., 2006). Sometimes treatment for ADHD must be withdrawn if it is aggravating the depression. Some mood disorder guidelines suggest that combinations of antidepressants with stimulant medication are relatively safe and that antidepressants with dopaminergic activity may be helpful for symptoms of both ADHD and depression. Perhaps more significantly, they have also warned of the possibility of stimulant medication utilised for comorbid ADHD destabilising the treatment of bipolar disorder, particularly in those with rapid cycling or mixed affective states (Malhi et al., 2009d; Pacchiarotti et al., 2013a).
Medical conditions, including ‘vascular depression’
Mood disorders and physical health have a strong and complex bidirectional relationship that is important to recognise and manage. Depression can impede engagement in therapies for comorbid medical conditions thus worsening prognosis. Medical comorbidities – particularly chronic pain – appear to reduce antidepressant efficacy (Bair et al., 2003; Howland et al., 2008; Pohle et al., 2009), but antidepressants still appear to have efficacy over placebo (Gill and Hatcher, 2000). SSRIs are effective in ischemic heart disease and comorbid major depression (Pizzi et al., 2011), and appear more effective than psychological counselling alone (Dickens et al., 2013; Lespérance et al., 2007). Psychological therapies, including on-line formats, are effective, especially where chronic pain exists (Buhrman et al., 2013; Hoffman et al., 2007). Vascular depression is not part of the current DSM nosology, but has been proposed to be a subtype of depression (Alexopoulos et al., 1997), possibly related to microvascular events in fronto-limbic tracts and with poorer antidepressant efficacy and tolerability noted (Baldwin et al., 2004). Citalopram has been associated with prolonged QTc interval at high doses but clinical relevance is unclear (Vieweg et al., 2012). SSRIs have been associated with greater perioperative bleeding but it is unclear if they are causative or mediated by covariates (Auerbach et al., 2013).
Recent population-based studies confirm that bipolar disorder is commonly comorbid with a range of medical disorders, with consequent increased morbidity and mortality. Measured prevalence rates of comorbid medical conditions range from 41% to 58% (Carney and Jones, 2006; Kemp et al., 2014; Magalhaes et al., 2012). Conditions most commonly reported are obesity, migraine, hypertension, hyperlipidaemia, asthma, diabetes, endocrine and metabolic syndromes. Less commonly reported conditions include thyroid and parathyroid disorders, liver disease, hepatitis C, HIV, peptic ulcer disease, pancreatitis, autoimmune disorders and renal impairment. Medical comorbidity is so common that it has been suggested that it should be regarded as a core feature of bipolar disorder (Magalhaes et al., 2012), or that bipolar disorder should be regarded as a multi-system disorder (Leboyer et al., 2012).
The clinical characteristics associated with medical comorbidity were explored in a study of over 3000 bipolar patients from the STEP-BD study (Magalhaes et al., 2012). Comorbid medical conditions were more likely to occur in those patients who had an early onset and highly recurrent disorder, with comorbid anxiety and substance abuse and a history of smoking. Bipolar disorder is linked to an increased risk of developing dementia, and with a greater number of episodes of increased illness (Kessing and Andersen, 2004).
It is useful to consider medical illness in bipolar disorder patients as being: truly comorbid; conditions related to treatment; and conditions related to behaviours associated with bipolar disorder (Carney and Jones, 2006).
Comorbid disorders that may be related to lifestyle or treatment include migraine (Oedegaard et al., 2011), diabetes, hypertension and metabolic syndrome. An association has been found between bipolar disorder and thyroid dysfunction, particularly in women, independent of the effect of lithium. Dysfunction identified includes hyperthyroidism in hospitalised patients (Thomsen and Kessing, 2005); increased levels of TSH especially in women (Ozerdem and Rasgon, 2014); and increased levels of T3 (Krishna et al., 2013). High rates of subclinical hypothyroidism and thyroid antibodies have been found mainly in female bipolar patients and are associated with poorer response to mood stabilisers (Bauer et al., 2014). This study also showed that female patients have less ability to compensate for the effects of lithium on thyroid function and respond better than males to thyroid supplements.
Cardiovascular disease is a leading cause of mortality in bipolar disorder, even above the mortality from suicide (Leboyer et al., 2012). It can be comorbid, secondary to lifestyle behaviours or may relate to treatment. Obesity, metabolic syndrome, hypertension and diabetes can be complications of treatment with lithium, second generation antipsychotics and anticonvulsants. Hyperphagia during depressive episodes, a sedentary lifestyle, poor diet and smoking contribute to cardiovascular disease. However, a recent review (Leboyer et al., 2012) found that when the effects of lifestyle and medication were controlled for, the association between bipolar disorder and cardiovascular disease remains significant. Likewise the metabolic syndrome has been shown to be associated with bipolar disorder independent of medication effects and is associated with a more complex illness presentation, less favourable response to treatment and adverse outcomes including increased disease burden and suicide (McIntyre et al., 2010b).
Other treatments which can induce medical conditions include lithium (obesity, hypothyroidism, hyperparathyroidism, renal impairment and lithium toxicity); valproate (obesity, ataxia, menstrual disturbance, polycystic ovarian syndrome, foetal abnormalities, abnormal liver function, pancreatitis, leucopaenia); carbamazepine (weight gain, neurological symptoms, abnormal liver function, leucopaenia, foetal abnormalities, Stevens-Johnson syndrome); and lamotrigine (Stevens-Johnson Syndrome).
Illness-related behaviours such as substance misuse, smoking, promiscuity and impulsive recklessness can lead to infections with hepatitis B and C and HIV; alcohol-related liver disease and pancreatitis; sexually transmitted disease; asthma and obstructive pulmonary disease; and injury through accidents.
Management of mood disorders in the context of special populations
Pregnant and breastfeeding women
The prevalence of major depression and bipolar disorder peaks during the childbearing years, and so there are high rates of depression across the perinatal period (the time from conception to the end of the first postnatal year). The postpartum period is also a time of high risk for bipolar relapse and for the development of postpartum or puerperal psychosis, which is currently thought to be a variant of bipolar disorder (Bergink et al., 2015; Munk-Olsen et al., 2012; Pfuhlmann et al., 2002). A key consideration in the management of perinatal mood disorders is the neurodevelopmental impact of the disorder, and its treatment, on the foetus, the nursing infant and quality of the mother-infant relationship and attachment (Lewis et al., 2014; Stein et al., 2014).
Whenever psychotropic medication is prescribed to pregnant and breastfeeding women, informed consent should be obtained. In order to obtain informed consent for the provision of pharmacological treatment during pregnancy and breastfeeding, a risk/benefit analysis needs to be performed giving consideration to the risks for the mother as well as the risks to the foetus, and the risks to either of non-treatment. All risks need to be identified and disclosed, both proven and speculative, giving particular consideration to what is of significance to any particular patient. Details of any risk/benefit discussion and the woman’s decision should be carefully documented (Snellen et al., 2014).

Footnote: *(Nicholas et al., 2009).
The organisation of services for women with mood disorders over the perinatal period and the use of screening instruments have been described in specialised guidelines from the United Kingdom’s National Institute for Clinical Excellence (NICE, 2007), Australia’s Beyond Blue (BeyondBlue, 2011), and the Scottish Intercollegiate Guidelines Network (SIGN, 2012).
DSM-5 includes a peripartum onset specifier that can apply to the current episode or, if the full criteria are not currently met for a mood episode, most recent episode of mania, hypomania or depression in BD I or BD II, if onset of mood symptoms occurs during pregnancy or in the 4 weeks following delivery. This specifier does not take into account onsets of depression prior to conception or episodes of depression that arise any time in the first year postpartum that are commonly considered as perinatal depression or perinatal mood disorder. The specifier starts off differently for major depressive disorder. This specifier can be applied to the current or, if full criteria are not currently met for a major depressive episode, most recent episode of major depression if onset of mood symptoms occurs during pregnancy or in the 4 weeks following delivery.
The prevalence of perinatal depression lies between 6.5% to 12.9% (Gavin et al., 2005). While it used to be considered most episodes of depression arose postpartum, it is now recognised that major depression commonly occurs during pregnancy, emanating either from a pre-existing depressive episode or a new onset episode. A clinical assessment of depression in pregnancy needs to take into account the fact that some ‘depressive’ symptoms can arise from physical symptoms associated with pregnancy, such as insomnia and weight change (Matthey and Ross-Hamid, 2011; Yonkers et al., 2009), the same caveat applies postpartum when ‘depressive-like symptoms’ may be the consequence of being a nursing mother. The clinical picture of perinatal depression (whether arising in pregnancy or postnatally) is, in essence, no different from major depression arising at other times in life. Treating perinatal depression is important because the illness can adversely affect pregnancy, the developing foetus and the mother-infant relationship.
Treatment of depression in pregnant and breastfeeding women
Maternal depression (depression during pregnancy) has been associated with increased risk of premature delivery, low birth weight, gestational hypertension (Grigoriadis et al., 2013c; Grote et al., 2010) and perinatal death (Howard et al., 2007). Depression in pregnancy may have an adverse impact on infant emotional and cognitive development (Chaudron, 2013; Deave et al., 2008), and postnatal depression may have an adverse effect on the mother-infant relationship, that in turn leads to poor infant development and outcomes (Goodman et al., 2011; Tronick and Reck, 2009).
The potential adverse effects on the foetus and on infant development make it an imperative to treat depression during pregnancy.
The treatment of perinatal depression follows the same approaches used for the treatment of depression at other times; women with mild-to-moderate depression should be offered formulation based psychological treatments such as interpersonal therapy or cognitive behaviour therapy (Dennis et al., 2010; Spinelli, 1997). Women with more severe depression should be offered antidepressant medication even though there is an absence of evidence regarding the efficacy of antidepressants in the perinatal period.
The use of antidepressant medication for women in the perinatal period, especially during pregnancy, requires a careful risk/benefit analysis, as the developing foetus and the breastfeeding infant will invariably be exposed to antidepressant medication. The essence of this is shown in Table 26: Risks vs benefits of medication in pregnancy.
Risks vs benefits of medication in pregnancy.
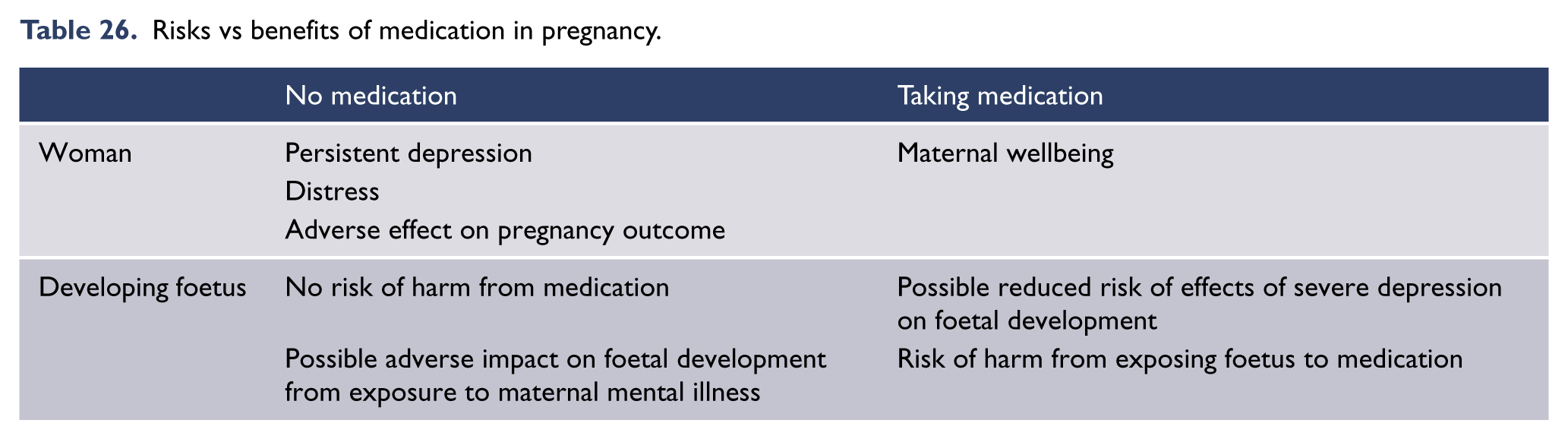
Risks associated with antidepressant use in pregnancy are small, but meta-analyses have identified statistically significantly increased risk of congenital abnormalities with first trimester exposure to antidepressant medications (Grigoriadis et al., 2013b; Myles et al., 2013). They have also identified a small, but significant, risk of the foetus developing persistent pulmonary hypertension of the newborn (Byatt et al., 2013; De Vera and Bérard, 2012). There is also a significantly increased risk of the infants developing a poor neonatal adaptation syndrome (PNAS) in the first week postpartum (Galbally et al., 2009; Grigoriadis et al., 2013a) with up to 30% of infants having symptoms. Tapering down the dose of an SSRI or SNRI in the late third trimester has been proposed as a strategy to reduce the severity of the PNAS (Byatt et al., 2013; Grigoriadis et al., 2013a). However, there is insufficient evidence to demonstrate that it does reduce PNAS, and therefore, this strategy needs to be balanced against the risk of the woman having a depressive relapse (Koren and Boucher, 2009; Koren et al., 2005; Warburton et al., 2010).
There is also an increased risk of women developing gestational hypertension and potentially pre-eclampsia following exposures to SSRIs (Toh et al., 2009) and suggestions of an increased risk of postpartum hemorrhage (Palmsten et al., 2013).
ECT has been safely used for the treatment of severe depression during pregnancy, and it may be the treatment of choice for patients with severe depression or bipolar episodes carrying a significant risk to the mother and/or the foetus (BeyondBlue, 2011). Anaesthesia in pregnancy carries increased risk of rapid desaturation and reflux with aspiration (Lakshmana et al., 2014). For these reasons ECT should be undertaken in a unit where there is ready access to foetal and maternal monitoring and access to appropriate post-anaesthetic recovery services, especially in the third trimester and late second trimester.
TMS may become an alternative to medications in this group in the future, but to date there is limited data available in pregnancy (Garcia et al., 2010; Nahas et al., 1999; Tan et al., 2008).
Bright light therapy has been evaluated for the treatment of depression during pregnancy (Wirz-Justice et al., 2011) in two trials for postnatal depression (Crowley and Youngstedt, 2012). While these small trials provide some evidence for its use, there is insufficient evidence to recommend its use as a first line treatment.
Postpartum depression
The treatment of postpartum depression depends on the type and severity of the depression. Episodes of depression arising within the first 4 weeks postpartum tend to be melancholic in nature and require assertive management with antidepressants. ECT should be used for women with psychotic (delusional) depression to ensure quick recovery from the depressive episode to minimise disruption to the mother-infant relationship.
In addition to providing treatment to the depressed mother, attention also needs to be paid to the mother-infant dyad. First, the safety of the infant and the woman’s capacity to provide a safe and nurturing environment needs to be assessed. This will also require an assessment of the woman’s intimate relationship as this may impact on safety issues, especially when domestic violence is present (and may be contributing to the woman’s depression). In such cases, liaison with child protection services is recommended. Second, an assessment of the quality of the mother-infant relationship and if problems are identified, specific interventions to address this may need to be implemented.
Psychosocial treatments, such as CBT, non-directive counselling and IPT (Dennis and Hodnett, 2007) are the first line of treatment for mild-moderate depression arising postpartum. Women with mild to moderate postnatal depression require additional support to assist them in compensating for fatigue induced by sleep deprivation as a result of nursing the baby. The provision of both practical and emotional support is of critical importance in helping women with postnatal depression.
For more severe depression antidepressant medication is indicated, though here consideration needs to be taken whether the mother is breastfeeding. If this is the case then the risk of the nursing infant being exposed to the medication needs to be balanced against the importance of the mother continuing breastfeeding and the necessity for medication. In the main, only small amounts of SSRIs and TCAs pass through to the breast milk and are generally considered safe (Eberhard-Gran et al., 2006) except for fluoxetine, which has a long half-life increasing the risk of reaching higher levels than other SSRIs and SNRIs (Weissman et al., 2004). Information about individual antidepressants and their passage through breast milk can be found on websites such as LactMed (NLM NLoM, 2014). Consideration needs to be paid to the side-effects of the medication and the impact upon the nursing mother, such as sedation that might interfere with night time feeds and breastfeeding.
Postpartum with pre-existing bipolar disorder
Women with pre-existing bipolar disorder are at high risk of having a relapse postpartum, with estimates suggesting that up to 50% of women will relapse in the first 3 weeks postpartum. Postpartum psychosis affects 1–2 women per 1000 deliveries and has a rapid onset of illness within the first three weeks following childbirth. Indeed, bipolar disorder is the most likely cause of admission for psychotic illness in the first three weeks postpartum (Munk-Olsen et al., 2012). Follow-up studies of puerperal psychosis would suggest that it is a variant of bipolar disorder (Munk-Olsen et al., 2012).
The management of bipolar disorder over the perinatal period is intrinsically complicated because it is a time of high risk for relapse, and mood stabilisers can have a deleterious effect on the developing foetus.
Pregnancy planning for women with bipolar disorder
For women with pre-existing bipolar disorder who wish to conceive, a careful risk-benefit analysis for remaining on mood stabilising medication needs to be done. It is essential that the women should be provided with good antenatal care and referral to a high risk clinic is recommended. There is good evidence that the anticonvulsant mood stabiliser sodium valproate can cause significant harm to the foetus, which has a foetal congenital abnormality rate ranging from 5 to 17% (Galbally et al., 2010a). Sodium valproate has also been shown to contribute to intellectual impairment in children who have been exposed to it in utero (Meador et al., 2009, 2012), whereas the risks of congenital abnormalities are lower for carbamazepine (3%) and lamotrigine (2%) (Hernandez-Diaz et al., 2012). Lithium has also been associated with a small increased risk of foetal cardiac defects (Cohen et al., 1994) but a recent meta-analysis (McKnight et al., 2012) did not find an increased risk for Epstein’s anomaly and concluded that the evidence that exposure to lithium is teratogenic is quite weak, and findings accord with the notion that the risk has been overestimated. A recent study found a significantly increased risk (OR 4.75) of cardiovascular anomalies compared to a non-exposed group (Diav-Citrin et al., 2014). For women with severe bipolar disorder lithium appears to be the safest option amongst available mood stabilisers. Its use has to be balanced against the potential risks for the developing foetus and the risk of relapse during pregnancy if lithium is discontinued. Women should have folate supplementation and an ultrasound to determine whether an abnormality is present (along with a plan as to what to do should an abnormality be found).
If mood stabilisers are to be withdrawn, it is recommended that the dose be tapered down slowly as this reduces the risk of relapse (Baldessarini et al., 1999).
Lithium is effective in preventing relapse (Bergink et al., 2012), and lithium prophylaxis should be offered to women with bipolar disorder with the proviso that breastfeeding is contraindicated with lithium. Lithium can be safely reintroduced in the latter stages of the pregnancy in order to provide prophylaxis against relapse. Lithium management in the latter stages of pregnancy is more complex and requires careful monitoring and dose reduction prior to delivery to avoid exposing the foetus to toxic levels of lithium (Galbally et al., 2010b). The use of the anticonvulsant mood stabilisers to prevent relapse is controversial, as evidence to support their use in preventing postpartum relapse is minimal.
SGA medications can be used as an alternative to mood stabilisers; there is insufficient evidence to suggest harm to the foetus and they are generally regarded as relatively safe. They can be used to treat psychotic symptoms during pregnancy, and as an alternative to mood stabilisers, but caution needs to be taken because of the possible risk of excess weight gain and/or gestational diabetes.
Postpartum relapse of bipolar disorder
There is a high risk (~50%) of a bipolar relapse in the first month following delivery. Women need to be monitored carefully over this period for any emergent symptoms and appropriate treatment applied when they emerge. There is evidence to suggest that sleep preservation reduces the risk of manic relapse and consideration should be given to the use of hypnotics, and the short term use of benzodiazepines can be of benefit in reducing anxiety symptoms or emergent psychotic symptoms (Bergink et al., 2012) (Plante and Winkelman, 2008). If a mother and baby unit is available, it is advisable to have a planned admission to monitor progress of the first month postpartum.
Mood stabilisers can be reintroduced following delivery. The reintroduction of lithium should be at the pre-pregnancy therapeutic dose (with serum lithium assessed shortly after the re-introduction). Anticonvulsant mood stabilisers should be at the previous therapeutic dose. Breastfeeding is contraindicated with lithium but recent data suggest that anticonvulsant mood stabilisers and second-generation antipsychotics can be taken if the woman is breastfeeding. Benzodiazepines, or a sedating antipsychotic can be used in the short term to assist in restoring a stable sleep–wake cycle if the woman is having trouble with her sleep. This needs to be done cautiously, as this could impact on her capacity to care for the newborn overnight, and to safely manage feeds. It is particularly important that women who receive sedating medications be advised to never co-sleep with their babies due to the inherent risks.
It is important to ensure adequate contraception and contraceptive advice is provided to the woman, especially if the woman has had a manic relapse with its attendant impulsivity.
There is no evidence to guide us about when to reintroduce mood stabilisers. General considerations would include, the risk of the medication passing through breast milk (if the woman is breastfeeding), side effects that could interfere with the woman’s capacity to care for her infant (sedation) and the severity of her illness (frequency of episodes). Ideally the woman should return to the effective mood stabilizer she had previously taken. Notwithstanding this, contraception should be discussed as soon as possible following delivery.
If prophylaxis is not used it is recommended that women are reviewed regularly over the first few months postpartum to ensure that mood stability has been achieved. A postpartum relapse could be to either pole of illness. Should a relapse occur, the safety of the woman and her infant becomes paramount. It is recommended that the woman be treated in a specialised mother and baby unit to allow for high quality psychiatric treatment while ensuring the safety of her infant and allowing for the mother-infant relationship to develop.
The treatment for the relapse is no different to the acute treatment of either (hypo)mania or major depression at any other time, using either antipsychotics, mood stabilizers or antidepressants or ECT as indicated.
While breastfeeding is recognised as desirable, in some instances, it may be necessary to cease breastfeeding if the woman is too unwell to breastfeed, requires sedation to ensure sleep, and disruption to this (to feed the infant) would have an adverse effect on her mental state or if lithium is indicated.
Children and adolescents
Major depression
In Australia, approximately 1.1% of children (4–11 years) and 5.0% of adolescents (12–17 years) meet diagnostic criteria for major depressive disorder based on parent/carer report. Based on self-report, the prevalence in adolescents increases to 7.7%. As measured on the K10, some 20% of adolescents report high or very high levels of psychological distress (Lawrence et al., 2015). However, closer to 10% are estimated to have subsyndromal symptoms. Depression is the major risk factor for suicide: in the management of depression in young people, therefore, there is a tension between the goal of conservative intervention for the developing brain/person and the need for prompt symptom relief.
Symptoms of mood disorders in children and adolescents often appear similar to those in adult patients, but with some differences that are attributed to their ongoing emotional, cognitive and social development (Fergusson et al., 2005; Kaufman et al., 2001; Klein et al., 2005). Children may display mood lability, temper tantrums, somatic complaints, and/or social withdrawal rather than verbally expressing their feelings of depression. Indeed, recognition and treatment of depressive and bipolar disorders in children and adolescence is often complex and has been hampered by a limited existing evidence base (Goldstein et al., 2012; Hazell, 2011; Hetrick et al., 2012). Partly, this relates to how to include appropriate allowances for the developmental variability and immaturity of children and adolescents (Carlson, 2000). To address this complexity, DSM-5 introduced Disruptive Mood Dysregulation Disorder (DMDD), a new diagnosis intended to decrease the use of the bipolar disorder diagnosis in young people. DMDD has poor test-retest reliability (kappa = 0.25), and is highly contentious because of its elevated comorbidity with other childhood disorders, and the potential risk of pathologising healthy children (Fristad and Algorta, 2013).
Psychological interventions, particularly CBT and IPT, are indicated as first-line treatments for child and adolescent MDD, especially when it is of mild to moderate severity. A recent network meta-analysis (a relatively new technique that integrates direct and indirect evidence from randomised controlled trials) compared the efficacy and acceptability of psychotherapies for depression in young people. Comparing 52 studies (N = 3805), post-treatment effects over control were significant for interpersonal therapy and cognitive behavioural therapy (standardised mean differences range from -0.47 to -0.96) (Zhou et al., 2015).
Evidence and treatment guidelines support parent/family involvement, particularly with children (David-Ferdon and Kaslow, 2008). Two recent Cochrane reviews address the treatment of major depression in children and adolescents, both highlighting significant methodological limitations of the available literature. The first focused on antidepressant medication, and found Level I evidence for a small beneficial effect of SSRIs amongst young adults (6–18 years): remission rates increased from 380 per 1000 to 448 per 1000 with antidepressant relative to placebo. The same review also highlighted the increased risk (58%) of suicide-related outcome amongst those receiving antidepressants relative to placebo (Hetrick et al., 2012). Of the SSRIs, fluoxetine has the most consistent evidence of efficacy over placebo, and it is recommended as the first line antidepressant for young people in treatment guidelines (NICE, 2005). A highly cited systematic review concluded that the efficacy of antidepressants correlates with depression severity, and that the benefits over placebo are substantial for severe symptoms, but minimal or nonexistant with mild-moderate depression (Fournier et al., 2010).

Footnote: aSnellen and Malhi (2014), bFor mild to moderate cases of MDD psychological interventions, particularly CBT and IPT, should be the preferred treatment modality. cIn instances of treatment resistance it may be necessary to trial sodium valproate.
A second Cochrane review compared the efficacy of psychotherapy, antidepressants and the combination of psychotherapy and medication. The authors found limited evidence for an advantage of antidepressants in young people (Cox et al., 2014). Specifically, two studies (a total of 220 participants) found a significant advantage for antidepressants (67.8% remission rates) over psychotherapy (53.7%), but only on clinician-rated measures immediately post-intervention. Hetrick et al. (2012) noted that there was evidence of increased suicidal ideation with medication: one study (N = 188) found suicidal ideation to be 3–4 times more common amongst participants receiving medication relative to psychological therapy, a ratio that was maintained at 6 to 9 months. The review also found evidence that adding psychotherapy to medication was beneficial for remission rates immediately post intervention, but no evidence that adding medication to psychotherapy was beneficial.
In sum, much remains unknown about the optimal treatment of depression in children and adolescents. As with adult major depression, there is Level I evidence for psychological treatments (CBT and IPT), and Level I evidence for the efficacy of antidepressant medication (fluoxetine) for young people. Little is known about the moderation of psychotherapy’s effectiveness by depression severity, but antidepressant medication appears to be more effective for severe relative to mild/moderate depression. Given the absence of evidence for superiority of medication monotherapy over psychotherapy monotherapy, the risks associated with antidepressant medication have influenced treatment guidelines in this area. The evidence-supported psychotherapies (individual CBT and IPT) are considered first line for management of MDD in young people of all levels of severity, with potential addition of fluoxetine being considered when depression is moderate–severe, or when psychotherapy has been trialled and found ineffective. Clinicians with child/adolescent expertise can consider non-fluoxetine medications (sertraline, citalopram, and if necessary antipsychotic augmentation) for recurrent, unresponsive or psychotic depression. Young people prescribed antidepressants of all kinds must be closely monitored for emergent suicidality, hostility, agitation, mania, and unusual changes in behaviour (NICE, 2005).
Bipolar disorder
Prevalence estimates are unclear as epidemiological surveys have seldom addressed BD in children and adolescents and there are significant international differences in the rate of diagnosing early onset BD (Stringaris and Youngstrom, 2014). Estimates for BD I range between 0.4–1.6%, with up to an additional 1% meeting criteria for BD II disorder (McClellan et al., 2007).
Most controversially there has been a suggested broadening of the definition of childhood bipolar disorder, whereby irritability has been adopted as a defining symptom, together with permitting a persistent or chronic pattern of symptoms to define the course rather than a relapsing or remitting pattern. As noted by Goodwin et al. (2008) this recent broadening of the diagnosis has tended to confound the bipolar diagnosis with alternative childhood psychopathology resulting, for example, in extremely high co-morbidity rates with ADHD, with figures between 70% and 98% being reported.
Such broadening of diagnostic criteria has also been associated with a lack of predictive validity and stability in juvenile bipolar disorder (Hazell et al., 2003), compared to the high diagnostic stability of the narrowly defined phenotype. The resulting risk has been the extrapolating of adult bipolar treatment options to significant numbers of young children, including preschoolers, identified with this broadly defined phenotype, before adequate efficacy and safety studies have been performed (Goodwin et al., 2008; McClellan et al., 2007). In contrast, there has also been concern over under-recognition of the onset of narrowly defined bipolar disorder (Goodwin et al., 2008). DSM has adopted an intermediate position on the phenotype: children and adolescents whose mood is predominantly irritable, if episodic, are considered part of the bipolar spectrum (Fristad and MacPherson, 2014).
The DSM-5 has explicitly reserved BD I for episodic presentation of bipolar symptoms (with mania taken to include states defined by severe irritability). Non-episodic presentations of severe irritability are thought to be common, and are now captured by the contentious DMDD diagnosis (see above) (APA, 2013). Importantly, outcome studies have generally not supported children and adolescents with ADHD (see also: Attention-deficit/hyperactivity disorder) or dysregulated mood symptomology being at increased risk of developing BD in adulthood (McClellan et al., 2007).
A diagnosis of BD in a young person is prognostically significant. The development of BD in early adolescence has been linked to a range of adverse outcomes as compared with a later onset, including cormorbidity with other psychiatric disorders, primarily ADHD (Moreno et al., 2007; Soutullo et al., 2009) and substance abuse (Soutullo et al., 2009), severe manic irritability (Soutullo et al., 2009), and treatment resistance (Jolin et al., 2005). Risk of suicide is also increased amongst people whose BD appears in childhood/adolescence. Although the aetiology of BD in adolescence is not known, some factors which have been linked with the development of early-onset BD, include having an immediate family member with a mood disorder, a history of childhood abuse, comorbid substance misuse, stimulant and antidepressant use, and omega-3 fatty acid deficiency (McNamara et al., 2010).
Reviews and practice guidelines recommend a combination of pharmacotherapy and psychosocial intervention for adolescents with BD I (Fristad and MacPherson, 2014). The existing empirical literature disproportionately focuses on the treatment of manic and mixed episodes, at the expense of bipolar depression and prophylactic treatment. Open label studies suggest lamotrigine may be effective, either as monotherapy or adjunctive therapy, for treating depressed bipolar adolescents (Level IV) (Chang et al., 2006).
Acute mania
Second generation antipsychotics, such as olanzapine (Tohen et al., 2007), quetiapine (DelBello et al., 2006), ziprasidone (DelBello et al., 2008), risperidone (Haas et al., 2009) and aripiprazole monotherapy (Findling et al., 2009) and quetiapine and sodium valproate combination therapy (DelBello et al., 2002), have Level II support (McClellan et al., 2007). However, the potential for metabolic side effects with this class of drugs with long term treatment warrants serious consideration (Thomas et al., 2011). Despite common usage, there is a lack of published RCT studies regarding lithium and valproate in acute mania and such use is reliant upon open studies (Level III) (Goldstein et al., 2012).
Maintenance
Currently, there are few well-controlled studies examining the efficacy of pharmacological treatments for prophylaxis in adolescents with BD. Many of these studies did not include a placebo group and have involved antipsychotic comparisons to either lithium or valproate. Overall, these studies suggest that antipsychotics may be more effective than mood stabilizers in acute presentations. Partly, this is the result of lithium receiving FDA approval being grandparented in on the basis of adult data (Goldstein et al., 2012). Further controlled studies are needed to determine the safety and efficacy of these pharmacological treatments in this population.
There is support for continuation and/or maintenance use of lithium and valproate from observational and naturalistic studies over 6 to 18 month periods. A direct double blind comparison between lithium and valproate showed no significant difference in median survival time to relapse, which was relatively short (both fewer than 115 days). Notably, when compared with lithium, valproate divalproex was associated with significantly fewer side-effects. Aripiprazole (Findling et al., 2011) and risperidone augmentation of lithium (Pavuluri et al., 2006) have been shown to be efficacious for the treatment of adolescents with BD. However, as with BD in general, the long-term use of atypicals in the treatment strategy for BD should be monitored regularly as medication-emergent side effects are not uncommon. Refer to Malhi and colleagues, 2012b and Ng and colleagues, 2009 for safety monitoring procedures.
The use of ECT in adolescents is rare, but numerous case reports support its efficacy and safety in the treatment of severe affective, psychotic and catatonic conditions. A review of 42 patients aged 14–18 years being treated with ECT in NSW (Walter and Rey, 1997) concluded that ECT was effective in 50% of cases, with mood disorders being the most responsive.
Psychosocial treatments
There has been only limited research into the efficacy of psychosocial treatments for bipolar disorder in adolescents. As with investigations of pharmacotherapy, conclusions must be qualified by ongoing debate about the optimal description of the disorder (ranging from narrow to broad definitions). In terms of specific interventions, a recent systematic review found evidence that family psychoeducation was probably efficacious (Fristad and MacPherson, 2014).

Expert consensus recommends the use of adjunctive psychosocial intervention, and as in the adult case, highlights the overlapping content of the various branded therapies (McClellan et al., 2007). Patients’ families should be provided with psychoeducation concerning aetiology, symptoms, course, medications, risk and protective factors, and effective treatments. Adolescents should receive skills training around communication, problem solving, CBT, and emotion regulation skills. Both families and adolescents should be trained in relapse prevention strategies. Important unanswered clinical questions include the matching of treatments to level of severity, particularly the relative cost/benefits of psychosocial and pharmacological intervention across the spectrum of presentations.
Older people
There are multiple issues that will need to be considered when treating depression in older people. The following is designed as a list that may be helpful in clinical practice, but is not necessarily comprehensive.
Evaluation
Late onset depression (onset in adults 60 years or older) should be distinguished from older adults reporting an initial depressive episode at a younger age. Late onset depression is associated with greater neuropsychological deficit, more brain imaging abnormalities and increased risk for dementia (Diniz et al., 2013; Hickie et al., 2005; Salloway et al., 1996). This should prompt more stringent monitoring of cognitive function in older people.
It should be remembered that older people may not declare alcohol use unless specifically asked and that this may be significant (Merrick et al., 2008).
Age is a well-recognised risk factor for suicide. Suicide risk is significant in older people and clinicians should be aware of the possible risk of suicide (Conwell et al., 2011).
Medication issues
Older people may be particularly sensitive to medication for a number of reasons. These include increased sensitivity to the constipating effects of medication, increased risks of falls (Van den Brand et al., 2009), reduced renal function, other medical problems, increased risk of delirium and, for this reason, possibly increased sensitivity to anticholinergic agents (Campanelli, 2012). Drugs that cause falls may give rise to further loss of confidence, which may be a particular issue in older people (Mulsant and Pollock, 2013).
As well as increased sensitivity to medication, older people may be on multiple other drugs giving rise to an increased risk of drug interactions (Mulsant and Pollock, 2013).
For various reasons it may take a longer time for a response to be seen in older people (Calati et al., 2013).
There has been some debate regarding the relative effectiveness of serotonergic compared with noradrenergic drugs in older people with depression (Bose et al., 2008; Navarro et al., 2001). Generally speaking, evidence suggests that there is equivalence between these types of drugs (Calati et al., 2013) and that treatment should be based on symptom and side effect profile and the need to avoid interactions (Coupland et al., 2011).
The combination of antidepressants with antipsychotics for psychotic depression or augmentation of antidepressant response (Steffens et al., 2011) should be approached with more caution in older people given their sensitivity to medication and the evidence that antipsychotics may increase the risk of cardiovascular accidents in patients with dementia (Ray et al., 2009).

Sleep may be a particular issue in older people. The prescription of benzodiazepines (which should only be prescribed short term) should be balanced against the risk of falls (Ray et al., 2000).
There remains a lack of robust high quality evidence for the use of stimulants (Hardy, 2009).
Maintenance treatment
Maintenance treatment with antidepressant medication may reduce recurrence compared to maintenance psychotherapy (Reynolds et al., 2006).
Psychotherapy
There are good studies regarding the use of psychological treatments in older people. These should be considered. There is particularly good evidence for the use of cognitive-behavioural therapy (Gould et al., 2012), interpersonal psychotherapy (Van Schaik et al., 2006) and problem-solving therapy (Alexopoulos et al., 2011).
The effect of CBT and IPT may be less in people with physical comorbidity or impaired cognition (Pinquart et al., 2007; Reynolds et al., 2006).
Problem solving therapy is effective for people with impaired cognition (Alexopoulos et al., 2011; Areán et al., 2010).
Lifestyle changes
Structured exercise is helpful for reducing depression severity (Bridle et al., 2012). Improving nutrition, increasing social interactions can also be considered (Solomon and Taylor, 2014).
Māori
Epidemiology
In the 1950s Māori adults were recorded as having lower prevalence of mental disorders than non-Māori (Durie, 1999). However, recent mental health data shows that Māori mental health is now an area of high priority in New Zealand with significant recorded disparities between the mental health of Māori and non-Māori. The rate of diagnosed mood or anxiety disorders in Māori adults at some time in their life has increased to 21.7% in 2013, compared to 17.3% in 2006/07 (Ministry of Health, 2014a).
These changes over time demonstrate the need to develop effective practice guidelines that are responsive to Māori mental health needs and also provide hope that clinicians, health services and researchers can work with Māori to reduce current disparities. An analysis of Māori mental health needs from community and hospital data has been completed and the findings relevant to mood disorders are briefly highlighted in this section (Baxter, 2008).
There are discrepancies between rates of mood disorders in Māori in community samples, primary care and specialist mental health services. Te Rau Hinengaro (The New Zealand Mental Health Survey) measured the prevalence rates of mental disorders using a large nationally representative sample of Māori (n = 2595) (Baxter et al., 2006a, 2006b). This identified higher rate 12-month prevalence of mood disorder in Māori (11.6%) compared to non-Māori/non-Pacific ethnicities (7.5%). One in four Māori (24.3%) had a lifetime mood disorder, the most common lifetime mood disorders being major depressive disorder (15.7%) followed by bipolar disorder (8.3%). Adjusting for contributory factors such as age, gender and socioeconomic status reduced much of the inequity in prevalence. Therefore treatment initiatives for Māori that reduce relative poverty, socioeconomic disadvantage, improve education and physical health outcomes will benefit Māori mental health outcomes. However the unadjusted rates highlight the high health burden associated with mood disorders for Māori. Māori with mood disorders was the most likely group to access a health service compared to other mental disorders. However 3 in 5 (56.9%) Māori with a mood disorder had no contact with any service.
There is consistent evidence of biomedical, social, political and cultural factors that contribute to health inequities of indigenous communities internationally and a broad framework which includes these factors can assist health practitioners working with Māori patients and whānau (families) to contribute to improved Māori health outcomes (King et al., 2009; Pitama et al., 2014).
Major depressive disorder
Community
Te Rau Hinengaro also provides prevalence rates for major depressive disorder (Baxter, 2008). Baxter (2008) This identified marginally higher 12-month prevalence of major depressive disorder in Māori (6.9%) compared to other ethnic groups (5.6%) with the same prevalence after adjustment. Māori females were more likely to have depression than non-Māori in the Youth 2000 survey (Clark et al., 2008). The New Zealand Health Survey also demonstrates Māori experience higher rates of psychological distress than other adults: Māori are 1.6 times as likely to have experienced high levels of psychological distress (indicating a high probability of a depressive or anxiety disorder) as non-Māori (Ministry of Health, 2014a).
Primary care
There is variability in the reported prevalence of depression in Māori in primary care. For example there was no difference in 12-month prevalence of depression by ethnicity in a primary care sample in Auckland, which included 721 Māori participants (Arroll et al., 2009). A smaller study of 81 Māori attending Wellington primary care identified significantly higher rates of depression compared to non-Māori (The MaGPIe Research Group and John Bushnell, 2005). Differences in study methods particularly sampling strategies, sample size and case ascertainment methods may account for much of variation between the studies. Despite the increase in rate of diagnosed mood disorders in recent years, differences between the prevalence of psychological distress and diagnosed mood disorders remain (Ministry of Health, 2014a).This may indicate differences in people’s understanding of their own symptoms, health service provision and barriers to health services. Māori continue to be more likely to experience barriers to primary health care (Ministry of Health, 2014a).
Specialist mental health services
Despite the high prevalence of depression identified in the community Māori are less likely to be admitted to hospital for depression (Baxter, 2008; Te Puni Kokiri, 1996). Different pathways to and through care for Māori may influence this discrepancy.
Bipolar disorder
Māori have a higher prevalence of bipolar disorder than other ethnicities with a 12-month prevalence of 4.6% (Baxter et al., 2006b). This discrepancy persisted after adjustment for age, gender and socio-economic status. Māori have a significantly higher hospitalisation rate for bipolar disorder than non-Māori (Robson and Harris, 2007). Manic episodes and bipolar disorder are one of the leading causes of hospitalization among Māori, and 1 in 6 (16.1%) of all hospitalisations in Māori are for these diagnoses (Robson and Harris, 2007).
Suicide and self-harm hospitalisations
Suicide and hospitalisation for intentional self-harm are often associated with mood disorders. In 2011, the total Māori suicide rate was 16.8 per 100,000 Māori population; 1.8 times higher than the non-Māori rate (9.1 per 100,000 non-Māori population) (Ministry of Health, 2014b). The biggest discrepancies were seen in the youth suicide rates with Māori youth 2.4 times higher than the equivalent rate for non-Māori youth. Māori also had higher hospitalisation rates for deliberate self-harm than non-Māori. Unfortunately reductions in non-Māori self-harm between 1996 and 2011 have not been observed in Māori self-harm rates.
Experience of discrimination
There is increasing recognition internationally regarding the adverse effect of discrimination on health and mental health in particular (Paradies, 2006). Māori are more likely to report experiences of self-reported racial discrimination, and are almost ten times more likely to experience discrimination in three or more settings than were Europeans (4.5% [95% CI 3.2–5.8] vs 0.5% [0.3–0.7]) (Harris et al., 2006). After adjustment for discrimination and deprivation, odds ratios comparing Māori and European ethnic groups reduce from 1.30 (1.11–1.54) to 1.02 (0.85–1.22) for low mental health. Māori may experience discriminatory attitudes from clinicians within mental health services, which may contribute to worse outcomes for Māori (Johnstone and Read, 2000).
Specific considerations
Clinical engagement and assessment
There is increasing recognition of need for effective engagement relationship building to work effectively with Māori patients (Mäuri Ora Associates, 2006). There is a range of resources available which aim to provide recommendations for developing clinicians’ skills in engaging with Māori (See Appendix 3).
Diagnostic considerations
There are a variety of social and cultural factors that may influence the way some Māori may present. Discussion of this is beyond the scope of these guidelines, but readers are referred to the following resource:
Recognising and managing mental health disorders in Māori (Best Practice Advocacy Centre New Zealand, 2010).
Developing evidence for best hauora Māori practice for mood disorders
The majority of studies identified in these guidelines have been guided by selecting the highest level and quality of evidence. However this process may come at the cost of reducing the relevance of the evidence for different patient groups including Māori. For example few of the studies referenced in these guidelines include sufficient numbers of Māori participants to allow recommendations specific to Māori to be made. In the following section recent publications involving treatment of Māori with mood disorders are briefly reviewed.
Psychotherapy
In a study investigating ethnic differences in care for all patients admitted to an adult psychiatric inpatient unit Māori were less likely to be referred for psychotherapy in the community (Kumar et al., 2008). However in a primary care study treatment for psychological problems was offered by the GP at similar rates to both Māori and non-Māori (The MaGPIe Research Group and John Bushnell, 2005).
Several Māori adaptions of common psychotherapy approaches have been reported. These include a brief cognitive behavioural therapy based intervention for sub-threshold mental health syndromes in primary care (Mathieson et al., 2012) and cognitive-behavioural therapy (Bennett, 2009b). Common features of the adaption for Māori include the use of Māori language and imagery and emphasis on relationships. Whilst initial results demonstrate the acceptability of these approaches for Māori larger studies of effectiveness are awaited. Additional resources for psychotherapy for Māori include:
He rongoa kei te korero. Talking therapies for Māori (Te Pou, 2010).
Medications
Māori may be less likely to receive anti-depressant medication. In a primary care study Māori with depression were significantly less likely to be treated with antidepressant medication although it was uncertain if patient or prescriber factors accounted for this discrepancy (Arroll et al., 2009). In an analysis of national prescription medicine dispensing claims data, Māori have lower age-standardised prescription rates for anti-depressants than non-Māori and non-Pasifika with an estimated shortfall of 46,300 prescriptions of ‘newer anti-depressants’ after adjusting for population size, age, and disease burden (Metcalfe et al., 2013).
Potential barriers to accessing anti-depressant medication include primary care access, diagnosis, prescribing and collection of prescriptions. Some general practitioners reported beliefs that Māori were less likely to acknowledge depression and had greater stigma towards mental health despite little evidence supporting ethnic differences in stigma towards mental health in New Zealand (Thomas et al., 2010). Prescription costs are another potential barrier with Māori adults 2.4 times as likely not to have collected a prescription due to cost than non-Māori and non-Pacific adults (Ministry of Health, 2014a).
Access to alternative treatments
Clinicians should also be aware Māori may access alternative treatments for mood disorders including traditional healing practices (rongoā rākau-native flora, karakia-prayer, mirimiri/komiri/romiromi-massage). In 2006 10.6% of Māori with a mood disorder saw a non-health service provider with high levels of satisfaction with care reported (Baxter, 2008).
Service delivery models
Both mainstream services and Māori services have a role to play in meeting Māori mental health needs. Mainstream clinical services have been challenged to be more culturally appropriate for Māori and to increase community access to services. Clinical Kaupapa Māori services aim to improve mental health outcomes and recovery through having access to cultural and clinical resources and prioritise a service run by Māori for Māori. Common principles of Kaupapa Māori mental health services include whānaungatanga (emphasis on relationships), whakapapa (genealogy), empowerment of Māori patients and whānau, cultural assessment, Te Reo Māori (Māori language), Tikanga Māori (customs and culture), Kaumātua guidance, access to traditional healing, access to mainstream health services, and quality performance measures relevant to Māori (Ministry of Health, 2002) (te pu). Translating this set of values to service delivery for Māori and their whānau who present with mental illness needs to occur not only with Kaupapa Māori services but also within general community teams, crisis and impatient services and forensic and other specialty areas and requires further development of a Māori mental health workforce.
Aboriginal and Torres Strait Islander peoples
The National Strategic Framework for Aboriginal and Torres Strait islander Peoples’ Mental Health and Social and Emotional Wellbeing 2014–2019 will replace the Social and Emotional Wellbeing Framework 2004–2009 as the guiding document informing policy and priorities for Aboriginal and Torres Strait Islander peoples. It prioritises an holistic approach concordant with the cultural and spiritual beliefs of Australia’s first peoples. It is a paradigm that recognises the complex interplay of cultural, historical, social, personal and spiritual factors determining health and mental health outcomes while acknowledging and foregrounding resilience and strength in the face of adversity.
There are a number of important documents including The National Strategic Framework for Aboriginal and Torres Strait Islander People’s Mental Health, (Australian Government Department of Health and Ageing, 2005), and The Mental Health and Social and Emotional Wellbeing of Aboriginal and Torres Strait Islander Peoples, Families and Communities (NMHC, 2012b) (which is a supplementary paper to A Contributing Life Closing the Gap) (AHMAC, 2009) that acknowledge the historical context of colonisation, dispossession and displacement and call for a more nuanced understanding of the diversity of the Aboriginal and Torres Strait Islander population with multiple language groups, cultures and traditions. While there are now many learning and practice resources (Commonwealth Australia (collaboration with the University of Western Australia), 2014) there is a lack of comprehensive epidemiological data for the Aboriginal and Torres Strait Islander people for mental health. A number of issues have contributed to this lack of research including methodological issues relating to the need for culturally appropriate tools and research approaches, the diversity of Aboriginal and Torres Strait Islander populations which makes accurate generalisation difficult, and the complicating influence of comorbid conditions and high levels of social adversity, particularly in remote communities. Suffice it to say that depression is common, may be identified through screening but may also present in culturally informed ways (Brown et al., 2012; Esler et al., 2008; Lee et al., 2008; Schlesinger et al., 2007). Practitioners should not presume exotic phenomenology but should be alert to the importance of culture, context and communication in assessment and interventions (Hunter, 2014).


These recommendations are therefore based on current knowledge base and, for the most part, are not specific to mood disorders but reflect a culturally concordant approach (See Appendix 4).
Footnotes
Appendix 1
Levels of evidence for intervention studies*.

| Level | Design |
|---|---|
| I | A systematic review of level II studies |
| II | A randomised controlled trial (RCT) |
| III | A pseudo-randomised controlled trial (i.e. alternate allocation or some other method) |
| A comparative study with concurrent controls: | |
| Non-randomised, experimental trial | |
| Cohort study | |
| Case-control study | |
| Interrupted time series with a control group | |
| A comparative study without concurrent controls: | |
| Historical control study | |
| Two or more single arm studies | |
| Interrupted time series without a parallel control group | |
| IV | A case series with either post-test or pre-test/post-test outcomes |
Adapted from: NHMRC levels of evidence for intervention studies (NHMRC, 2009).
Appendix 2
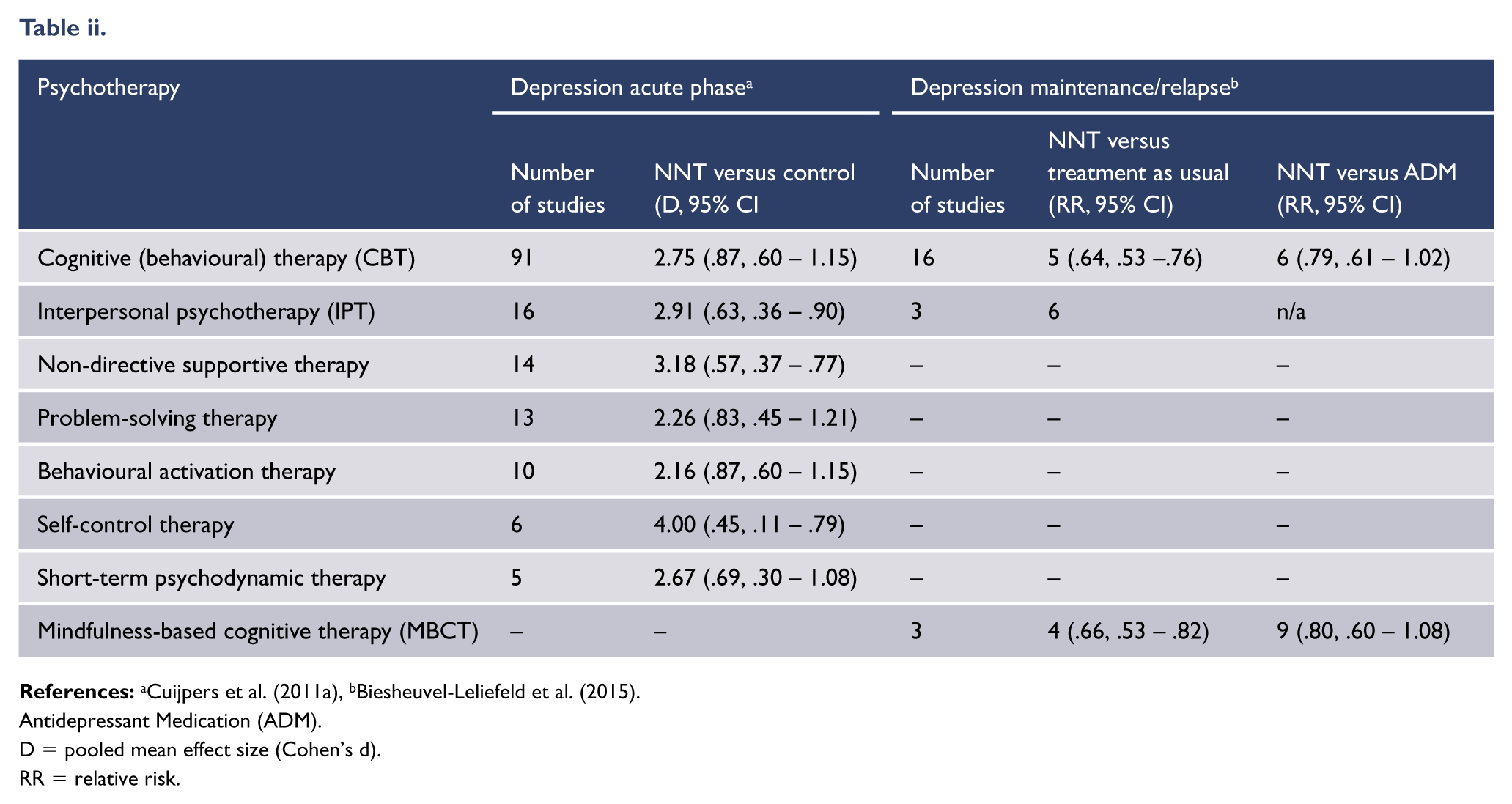
| Psychotherapy | Depression acute phase
a
|
Depression maintenance/relapse
b
|
|||
|---|---|---|---|---|---|
| Number of studies | NNT versus control (D, 95% CI | Number of studies | NNT versus treatment as usual (RR, 95% CI) | NNT versus ADM (RR, 95% CI) | |
| Cognitive (behavioural) therapy (CBT) | 91 | 2.75 (.87, .60 – 1.15) | 16 | 5 (.64, .53 –.76) | 6 (.79, .61 – 1.02) |
| Interpersonal psychotherapy (IPT) | 16 | 2.91 (.63, .36 – .90) | 3 | 6 | n/a |
| Non-directive supportive therapy | 14 | 3.18 (.57, .37 – .77) | – | – | – |
| Problem-solving therapy | 13 | 2.26 (.83, .45 – 1.21) | – | – | – |
| Behavioural activation therapy | 10 | 2.16 (.87, .60 – 1.15) | – | – | – |
| Self-control therapy | 6 | 4.00 (.45, .11 – .79) | – | – | – |
| Short-term psychodynamic therapy | 5 | 2.67 (.69, .30 – 1.08) | – | – | – |
| Mindfulness-based cognitive therapy (MBCT) | – | – | 3 | 4 (.66, .53 – .82) | 9 (.80, .60 – 1.08) |
Antidepressant Medication (ADM).
D = pooled mean effect size (Cohen’s d).
RR = relative risk.
Appendix 3
Further reading for developing clinicians skills in engaging Māori.
Appendix 4
All mental health staff working in Australia should have mandatory training in Aboriginal and Torres Strait Islander people cultural awareness which should include understanding of the following:
Appendix 5
External consultation specialist groups approached.
| Key stakeholders, professional bodies, special groups and interested parties |
|---|
| Internal College committees, fellows and trainees: |
| Community Collaboration Committee |
| Aboriginal and Torres Straight Islanders Committee |
| Te Kaunihera Committee |
| Fellows and trainees |
| Australia and New Zealand Mental Health and Disability Commissions |
| Mental Health Council of Australia |
| Committee of Presidents of Medical Colleges |
| Council of Medical Colleges New Zealand |
| Australian and New Zealand Colleges of Nurses and Mental Health Nurses |
| Australian Psychological Society |
| New Zealand Psychological Society |
| New Zealand College of Clinical Psychologists |
| Australian Clinical Psychology Association |
| Australian Association of Social Workers |
| Aotearoa New Zealand Association of Social Workers |
| Occupational Therapy Australia |
| New Zealand Association of Occupational Therapists |
| Mental Health Foundation NZ |
| SANE |
| Mental Illness Fellowship of Australia |
| Mental Health Professionals’ Network |
During the consultation period, the draft guideline was publicly available for review and comment on the RANZCP website. Participants were asked to review the guideline and answer four key questions related to each section of the guideline via Survey Monkey. Question criteria were:
Are there any significant gaps (of topic, literature, other)?
Are there errors in the content?
Is the structure logical and easy to use?
Do you have any other comments?
Acknowledgements
The following people and organisations contributed information that was used in the RANZCP Clinical Practice Guideline for Mood Disorders.
Feedback received during Public and Stakeholder consultations
RANZCP Project Team.
Ms Rosie Forster, Senior Department Manager, Practice, Policy and Partnerships.
Dr Huseyin Mustafa, Project Manager, Policy Operations and Committees.
Ms Edwina Ward, Project Officer (from October 2014), Practice, Policy and Partnerships.
Ms Helen Walters Project Officer (until December 2014), Practice, Policy and Partnerships.
Ms Claire Leslie, Project Officer (from June – September 2014), Practice, Policy and Partnerships
Disclaimer
Compiled for the Royal Australian and New Zealand College of Psychiatrists (RANZCP), this information and advice is based on current medical knowledge and practice as at the date of publication. This CPG is intended as a general guide only, not as a substitute for individual medical advice. The RANZCP and its employees accept no responsibility for any consequences arising from relying upon the information contained in this publication.
Funding
The development of this guideline was supported and funded by the RANZCP. The RANZCP acknowledges the significant pro-bono input of RANZCP Fellows and other expert contributors in the development of this guideline. The RANZCP thanks those who have given of their time, experience and expertise.
Declaration of interest
MDC disclosure of potential conflicts of interest management process.
Members of the Mood Disorders Committee (MDC) signed a deed of undertaking at the time of appointment in which they agreed to declare any conflict, whether actual, potential, perceived or likely to arise. To manage conflicts of interest of the MDC during the CPG development process: As a part of the standing items of all meetings, all MDC members were asked to declare their conflicts of interest during each teleconference meeting and these were recorded. If a conflict of interest was declared, the individual(s) concerned was excluded from the discussion and right to vote/contribute. The MDC members signed an up-dated COI form at the time of submitting the mood disorders guideline for publication.
Committee member’s declarations of interest are listed in Table iv of Appendix 5. MDC member disclosure statements will be available at ![]() .
.
