Abstract
Objectives:
To provide advice and guidance regarding the management of mood disorders, derived from scientific evidence and supplemented by expert clinical consensus to formulate s that maximise clinical utility.
Methods:
Articles and information sourced from search engines including PubMed, EMBASE, MEDLINE, PsycINFO and Google Scholar were supplemented by literature known to the mood disorders committee (e.g. books, book chapters and government reports) and from published depression and bipolar disorder guidelines. Relevant information was appraised and discussed in detail by members of the mood disorders committee, with a view to formulating and developing consensus-based recommendations and clinical guidance. The guidelines were subjected to rigorous consultation and external review involving: expert and clinical advisors, key stakeholders, professional bodies and specialist groups with interest in mood disorders.
Results:
The Royal Australian and New Zealand College of Psychiatrists mood disorders clinical practice guidelines 2020 (MDcpg2020) provide up-to-date guidance regarding the management of mood disorders that is informed by evidence and clinical experience. The guideline is intended for clinical use by psychiatrists, psychologists, primary care physicians and others with an interest in mental health care.
Conclusion:
The MDcpg2020 builds on the previous 2015 guidelines and maintains its joint focus on both depressive and bipolar disorders. It provides up-to-date recommendations and guidance within an evidence-based framework, supplemented by expert clinical consensus.
Mood disorders committee:
Gin S Malhi (Chair), Erica Bell, Darryl Bassett, Philip Boyce, Richard Bryant, Philip Hazell, Malcolm Hopwood, Bill Lyndon, Roger Mulder, Richard Porter, Ajeet B Singh and Greg Murray.
Keywords
1. Introduction
1.1. Overview
This guideline updates the Royal Australian and New Zealand College of Psychiatrists Clinical Practice Guidelines for Mood Disorders (MDcpg2015) that were published in 2015 (Malhi et al., 2015). The core composition of the Mood Disorders Committee (MDC) driving the development of the guideline (MDcpg2020) has remained largely the same, as has the process of evaluating the evidence and synthesising the findings.
The 2020 guidelines are published at a time of change in mental health service delivery. The COVID-19 illness and governments’ attempts to manage its spread bring a range of new social, physical and psychological risks for clinicians to consider (see https://oxfordhealthbrc.nihr.ac.uk/our-work/oxppl/covid-19-and-mental-health-guidance). Acceptance and uptake of telepsychiatry has accelerated during the pandemic, with consequences for all aspects of professional practice (Smith et al., 2020). As we note below, a key challenge for clinicians is keeping abreast of technological developments, which emerge not through traditional scientific processes (across time frames of years), but through the marketplace (across time frames of months).
1.2. Methodology
Initially, the MDC identified areas within the MDcpg2015 where significant changes had occurred, including the development of new therapies, or where practice and conceptualisation have evolved. Recommendations of the MDcpg2015 were also carefully reviewed in light of any new evidence. The MDC then developed a draft which was informed by regular teleconferences, and a full-day face-to-face meeting. 1 The draft then underwent further revisions in light of feedback from key College committees (see Appendix A).
Once the foci of the MDcpg2020 agreed, the MDC resolved to develop evidence-based recommendations (EBR; see Appendix B), and where this was not possible, for example because of insufficient evidence, to develop consensus-based recommendations (CBR). To this end, literature in PubMed, EMBASE, MEDLINE, PsychINFO and Google Scholar was searched to identify current research and clinical guidance pertinent to proposed new or updated sections. Searches prioritised recent evidence, systematic reviews and meta-analyses. These literature searches were supplemented by resources known to the MDC.
The MDC comprised members of the 2015 MDC from the disciplines of psychiatry and psychology with specific expertise in the management of mood disorders in adults, children and adolescents. An additional member with expertise in child and adolescent mental health was appointed to the MDC in 2019. RANZCP committees, faculties and sections were notified of the MDcpg2020 and the terms of reference were approved by the RANZCP Committee for Practice, Policy and Partnerships and tabled at the RANZCP Board.
Preparation of the MDcpg2020 has been funded solely by the RANZCP, with acknowledgement of the significant pro-bono contributions of MDC members.
Finally, in addition to extensive iteration with College committees, the MDcpg2020 underwent comprehensive peer review conducted formally by the ANZJP.2
1.3. Scope
The MDcpg2020 retains its focus on mood disorders and continues to uniquely combine both depressive disorders and bipolar disorders, reflecting the reality of clinical practice; where mania (bipolar disorder) usually emerges in the context of pre-existing depression that may, or may not, have been diagnosed as yet. However, more emphasis has been placed on diagnosis, classification, assessment and formulation. A new framework for treatment is presented along with the introduction of a number of novel paradigms. The relationship between psychological and pharmacological approaches has also been clarified, and new sections have been added on child and adolescent mood disorders, physical treatments and response to treatment.
As per the original guidelines, the MDcpg2020 is intended for use by specialists (psychiatrists and psychologists) and all those involved in the clinical management of mood disorders in different settings, in particular general practitioners. Catering for such a diverse audience means that any recommendations made by the MDcpg2020 should be actively appraised in the reader’s own professional context. Professional context includes the clinician’s training (e.g. psychiatrist, GP, psychologist), expertise and interest (e.g. mood disorder specialisation, child/adolescent disorders, psychological experience) and the networks in which they practice (e.g. sole practitioner, secondary or tertiary centre, managed care). To provide one example of how context should influence application of these recommendations, while psychological treatment may be a safe initial monotherapy for severe depression when delivered within a specialist mood disorders clinic, the recommendation may not be appropriate if adopted by a sole practitioner.
Four further underpinning assumptions should be noted:
First, the guidelines assume a model of collaborative care across professionals and disciplines. We acknowledge that this is not always possible but advocate that it is the ethical responsibility of professionals to attempt to create this collaboration even in sometimes suboptimal contexts.
Second, the focus of these guidelines is clinical management (i.e. treatment managed by clinicians such as psychiatrists, GPs and clinical psychologists), but such management occurs in the context of other determinants of mental health and well-being, including the important role played by carers, family and social support networks and services.
Third, as the title suggests, the guidelines adopt a largely nomothetic, group-level approach to mental health, focusing on mood disorders/diagnoses and evidence-based practice (Courtois and Brown, 2019; Van Os et al., 2019), while also pointing to idiographic considerations such as individual differences, including ethnicity, class, gender, minority group status (e.g. sexual identity), personality and common physical/mental health comorbidities. Clearly, the number and variety of these factors precludes detailed analysis and at many points the guidelines simply emphasise the clinical importance of individualised case formulation and the use of practice-based evidence to inform personalised care (see also MDcpg2015).
Finally, the guidelines assume an active engaged patient, with whom decision-making is shared in a strong, supportive clinical relationship (Rush, 2017). This assumption is consistent with the chronic illness self-management model widely accepted in the management of mood disorders (Yatham et al., 2018), and the recovery-oriented, resilience-development emphasis of the MDcpg2015. Patient preference has been shown to improve engagement, retention and outcomes (Mergl et al., 2011) and it is therefore a factor in treatment choice in some of the recommendations.
1.4. Navigating the MDcpg2020
The MDcpg2020 uses the MDcpg2015 as a foundation. Many of the principles and general recommendations of the MDcpg2015 still apply; however, the specifics of management have been revised and updated considerably. Therefore, to navigate the MDcpg2020 it is useful to briefly review its structure and to refer to the roadmap provided in Figure 1, which outlines its key components and the order in which they have been presented.
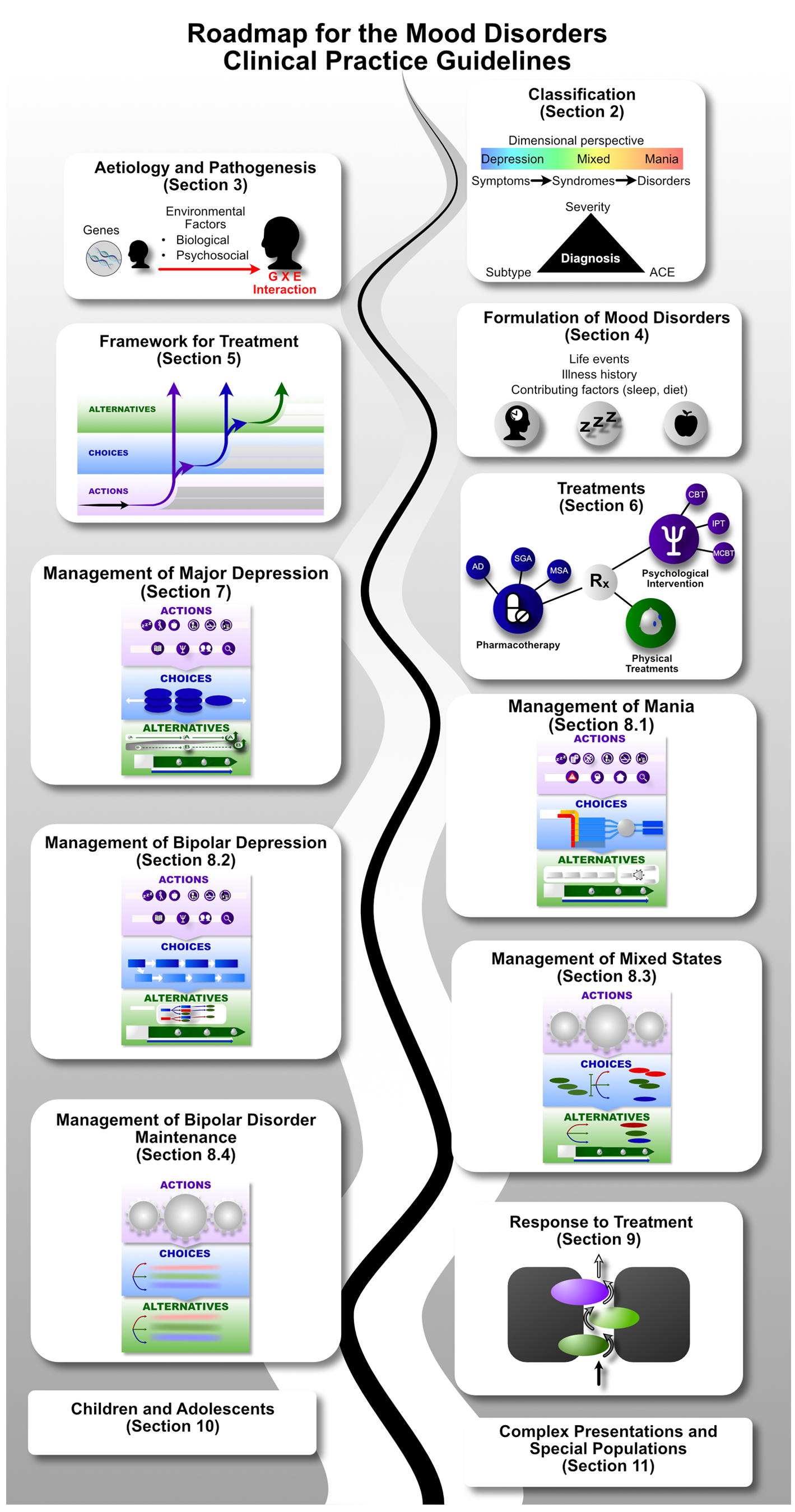
Roadmap to the 2020 Mood Disorders Clinical Practice Guidelines.
Structure
The MDcpg2020 begins with a new section on the aetiology and pathogenesis of mood disorders reflecting advances in basic and clinical neuroscience yielding new knowledge that is relevant to understanding the basis of mood disorders. This is followed by an update on classification and refinement of assessment and formulation, and a new section introducing novel models for the clinical management of mood disorders.
The treatment of mood disorders is broadly divided into the management of depressive and bipolar disorders with greater emphasis on suboptimal response and the introduction of a response perspective instead of treatment resistance. More detailed management figures are presented that summarise treatment approaches for Major Depression, Bipolar Depression, Mania and Maintenance therapy for bipolar disorder (BD). In addition, totally new management figures are presented for mixed states, the appraisal of maintenance therapy in BD and the channelling of response. The clinical management of complex presentations and special populations has also been updated with particular emphasis on children and adolescents.
2. Classification
The MDcpg2020 adopts a pragmatic approach to mood disorders taxonomy so that diagnostic and treatment recommendations can be easily applied to clinical practice.
2.1. Phenomenology
The signs and symptoms that define mood disorders are essentially those shown in Figure 2. Traditionally, these have been divided into those that characterise depression and those that signify mania (Malhi and Bell, 2019a; Malhi and Mann, 2018). Many symptoms belong to one syndrome or the other depending on the direction of change. For example, a lack of energy (fatigue, lassitude) reflecting depression versus increased energy that typifies mania. However, some symptoms are more specific for one pole as compared to the other, such as guilt in depression and prolixity in mania.
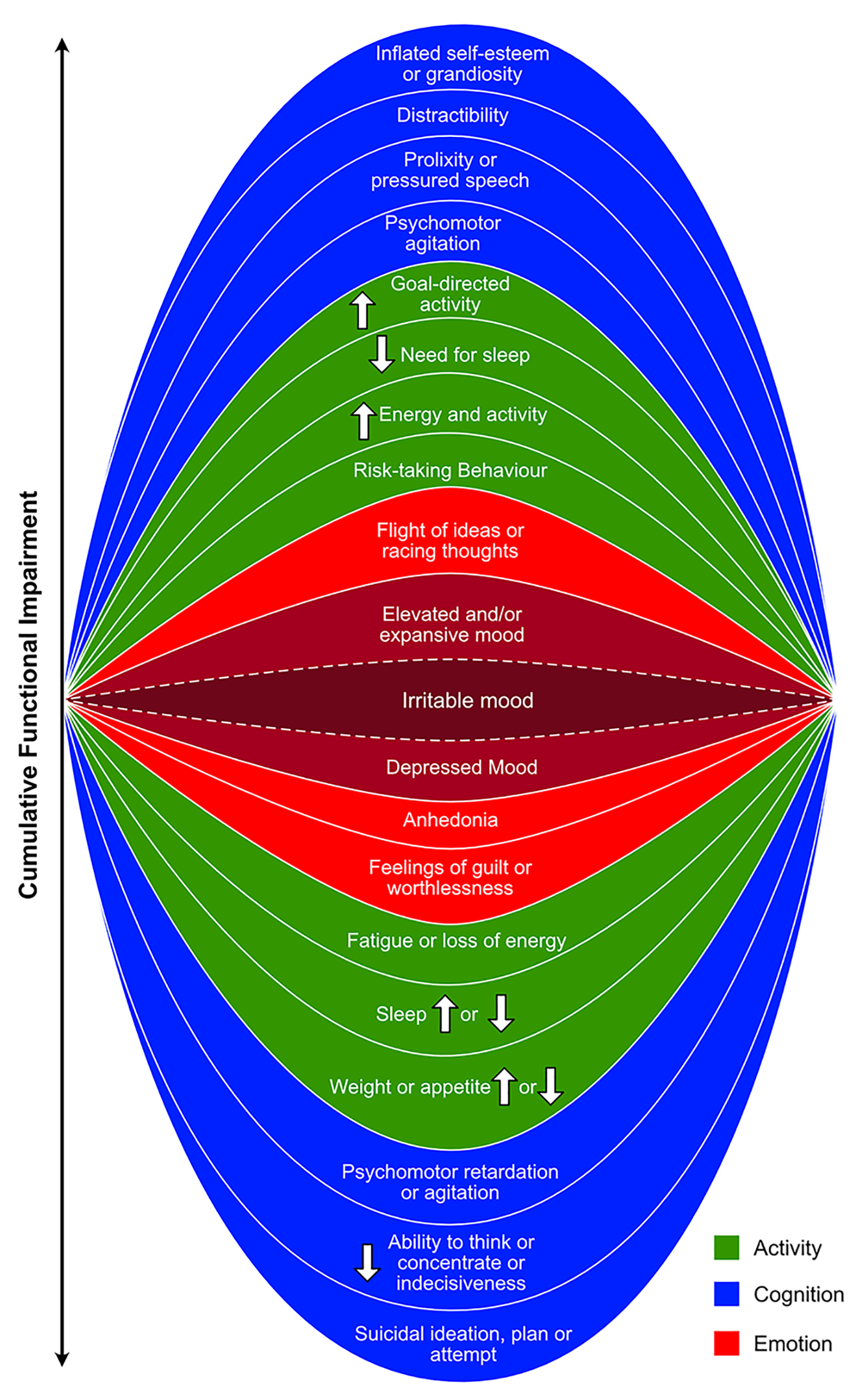
Symptoms of depression and mania according to DSM-5.
Notably, few symptoms are unique to either syndrome and some, such as irritability, seem to occur equally in both (Bell et al., 2020). Furthermore, many of the symptoms are not too dissimilar to normal changes that individuals experience, and it may be simply the duration and severity that is deemed unusual or cause for concern. For example, changes in sleep and appetite, vicissitudes of mood, being able to attend and concentrate, and having motivation and drive. Normally, all of these vary considerably in normal health and so determining cut offs can be difficult both subjectively and objectively. Therefore, clinically, it is important to elicit these symptoms if they are not spontaneously reported. In other words, it is necessary to ask systematically about each of the symptoms and also inquire as to whether they are causing any functional impairment, that is to say, imposing any limitations on what the person can do in their day-to-day activities, both work and leisure.
Some symptoms may require more active probing by the clinician. For example, patients may be reluctant to raise issues of self-harm and suicide (Malhi et al., 2019a). Most patients will not volunteer these ideas unless specifically asked, and even then, may be somewhat guarded. The onset of some symptoms can be insidious and indeed so gradual that the individual themselves is unaware of any significant change. For example, the loss of the ability to experience pleasure (anhedonia) may be gradual and subtle and may only become evident through targeted inquiry. In addition to eliciting the full gamut of symptoms, it is important also to screen for these over a reasonable period of time. While diagnoses require symptoms to be present for at least 2 weeks for depression and a week for mania, these duration criteria are somewhat arbitrary. Clinically, it is more important to review the history for several months prior to presentation to identify the onset of an episode of illness. As with all psychiatric history (anamnesis), it is important to obtain a corroborative account, and this is particularly useful when assessing for change.
Finally, it is important to note that typically, emotional symptoms fluctuate and may do so even within the period of 1 day (diurnal variation), and so it may be necessary to assess patients at different times of the day to obtain an accurate picture of their mental state (Kaufmann et al., 2020). It is also important to remember that symptoms can be masked by medication.
2.2. Spectrum
The term mood disorders in the MDcpg2020 encompasses both depressive and bipolar disorders which exist on a continuum (see Figure 3). The concept of a spectrum is important to bear in mind when considering the diagnosis and the management of mood disorders. This dimensional perspective applies to both syndromes individually, such as depression and mania, and to the juxtaposition of disorders within the broader category of mood disorders. It also applies more granularly to individual symptoms such as low mood, lack of energy and cognitive slowing. The concept of dimensionality is important and is reflected in a number of substantive changes to classification in this guideline.
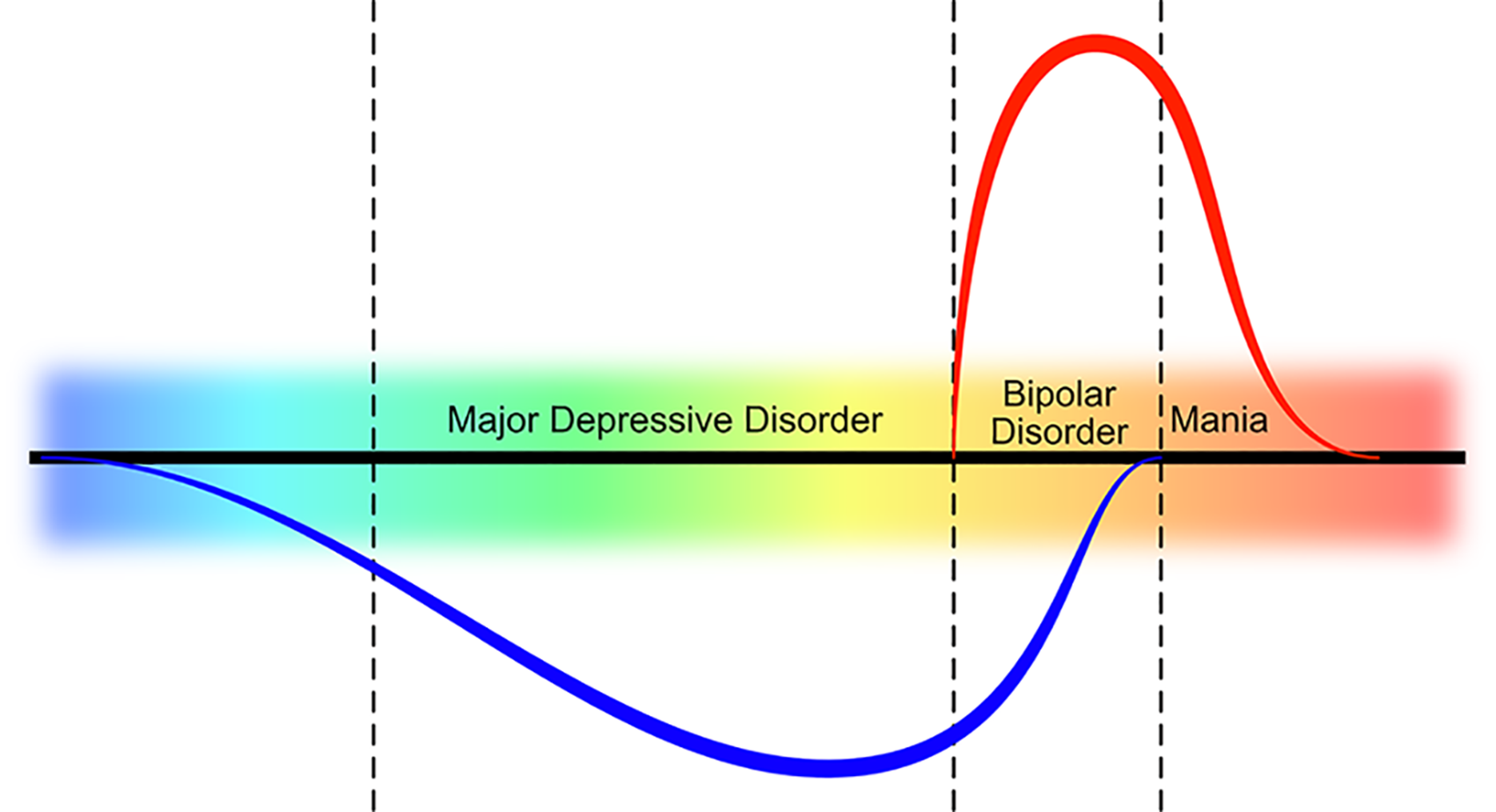
Spectrum of mood disorders.
This spectrum differs from what has been described as the ‘bipolar spectrum’ in which the affective instability of borderline personality disorder, cyclothymia and bipolar disorder subtypes are considered to be on a continuum (Akiskal and Pinto, 1999).
2.3. Classification systems: DSM-5 and ICD-11
In clinical and research practice, the definitions of mood disorders are drawn from the major classificatory systems DSM-5 and ICD-11. The MDcpg2020 recognises practical strengths of these taxonomies such as their reliability, familiarity and frequency of uptake and therefore they remain an option for describing the clinical presentations of mood disorders. However, there are key aspects (see 1–3 below) that we consider to be important for achieving more accurate and meaningful diagnoses (Malhi and Bell, 2019c). In addition, the MDcpg2020 provides alternative schemas that better approximate clinical description with reality and enrich diagnostic formulation:
First, the MDcpg2020 no longer distinguishes bipolar I and bipolar II disorder. It is our basic premise that all syndromes characterised by symptoms of mania are best described simply as bipolar disorder (see Bipolar disorder subtypes, in section 2.3).
Second, a new model for grouping the phenomenology of mood disorders is presented (see ACE model, in section 2.3). It offers an alternative to the standard classification models and the subtyping approach. Used in conjunction with the existing schemas, it enables the diagnosis of mood disorders to be more specific and their management to be more sophisticated.
Third, mixed mood states are conceptualised differently to how they are in DSM-5 and ICD-11, and this allows for their coexistence as an independent mood state alongside depression and mania.
Bipolar disorder subtypes
Both DSM-5 and ICD-11 divide bipolar disorders into bipolar I and bipolar II (Nierenberg, 2019). However, the MDcpg2020 no longer makes this distinction because partitioning bipolar disorder in this manner is arbitrary and does not meaningfully inform management (Malhi et al., 2019f). Categorising bipolar presentations as bipolar II also fails to capture those individuals that have mania for very short periods of time (e.g. 2–3 days) and the alternatives offered by DSM-5, for example, short-duration hypomania, are rarely used (Gitlin and Malhi, 2020; Malhi et al., 2019f). Thus, in the MDcpg2020 all syndromes characterised by symptoms of mania are simply referred to as bipolar disorder.
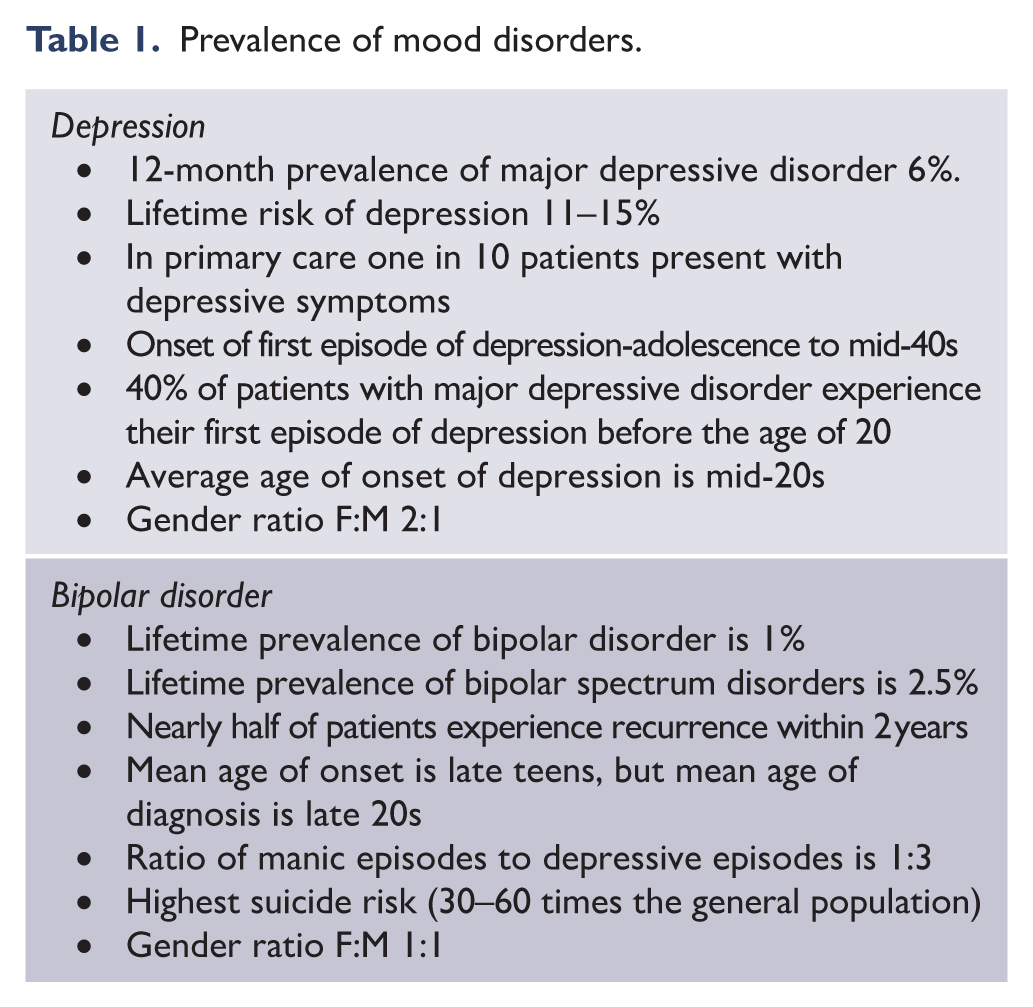
To quantify the duration of mania, the number of days can be specified (e.g. symptoms of mania lasting for 5 days) (Malhi et al., 2019g) and to measure impact the degree of impairment can be described as mild, moderate or severe. The relevant prevalence statistics for both depression and bipolar disorder are briefly summarised in Table 1.
Prevalence of mood disorders.
ACE model
DSM has long conceptualised mood disorders as discrete syndromes. A key problem is that conceptualising mood disorders in this categorical and largely dichotomous manner fails to capture clinical reality, in which admixtures of the two mood states are common. The ACE model, which emphasises the domains of activity and cognition alongside emotion, provides one means of addressing these problems (Grunebaum, 2019; Malhi et al., 2018a).
Using the ACE model, the primacy given to mood states can be supplanted by the three main components: activity, cognition and emotion. For the most part, the severity of symptoms within a domain, and the domains themselves, varies in unison, reflecting the intrinsic coupling of the various pathophysiological components. Naturally, some separation may occur because of inherent latencies and qualities of certain symptoms and domains. The model is useful, however, because, unlike the dichotomous, polarity-driven model used to define depression and bipolar disorder, the ACE model allows for the conceptualisation of mixed states as an asynchrony of the domains. The ‘pure’ states of depression and mania therefore simply reflect the occasions when the direction of change and the severity of symptoms are similar. Mixed states, therefore, arise when there is an uncoupling between the various domains and the symptoms they contain.
In the ACE model, symptoms can be mapped along each of the domains (axes) in unison or separately (see Figures 4–8). Asynchrony between the various domains allows for the expression of mixed states – for example, permitting individuals to be simultaneously extremely low in mood and yet agitated in activity and accelerated in thinking (cognition). The model also explains how treatments, which differentially impact the various domains, may result in a complex mixed picture in which the domains are uncoupled – for example, in response to an antidepressant with cognitive and activating properties these domains may separate from mood (see Figure 7). Furthermore, the ACE model facilitates the detection of mood disorders by drawing attention to those symptoms that patients more commonly experience. For instance, the inability to think, concentrate or remember things, or be motivated and active in terms of functioning. By assigning equal importance to a much broader range of phenomenology, the ACE model ensures that, in the context of treatment, goals such as remission and recovery are gauged across the full spectrum of symptoms.
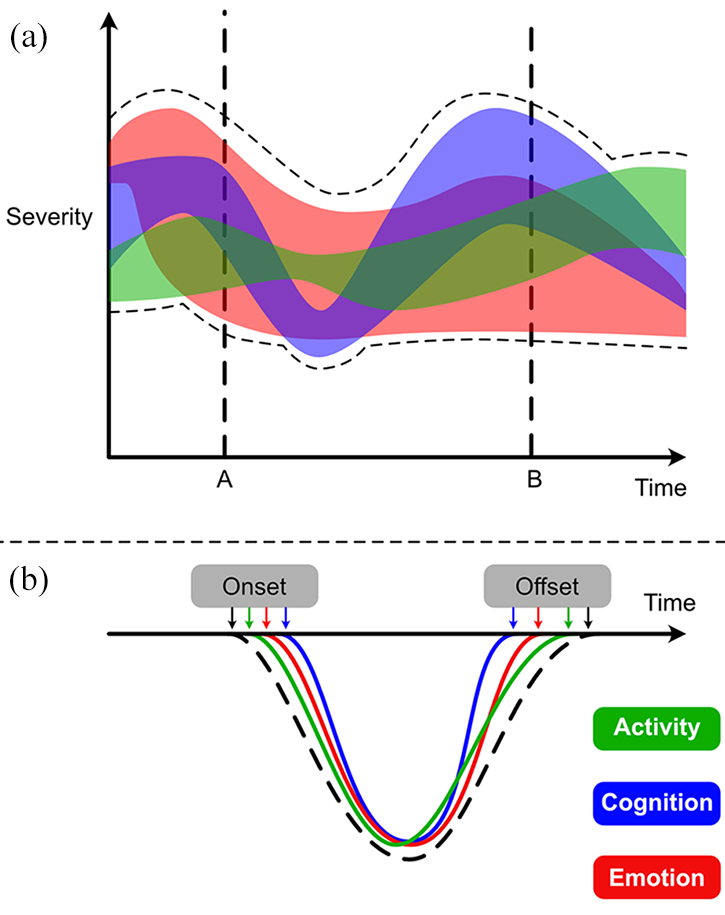
Differential components and timing of ACE components within mood episodes (adapted from Malhi et al., 2018a). (A) The schematic shows the three activity (green), cognition (blue) and emotion (red) that vary in severity over time. The overall range of severity (peak and trough) is shown with a black dashed line. At points A and B, the overall severity seems to be the same. However, it is evident that the various domains vary significantly such that the most prominent at point A is emotion, followed by cognition, however, at point B this order is reversed, and cognition is the major contributor to severity. Thus, by mapping domains and individual symptoms, rather than the syndrome as a whole, the ACE model provides a more granular understanding of changes over time. (B) The differential onset and offset of symptoms over time. The black dashed line shows the overall change in symptoms with depression emerging gradually and resolving slowly. However, within this, it can be seen that changes in activity precede those of emotion and cognition and that in response to treatment or spontaneous remission, it is the cognitive symptoms that recover first followed by emotion, with activity taking the longest. This is important when monitoring the effects of treatments and also when communicating to patients what to anticipate as regards recovery. Often as initial symptoms remit, they may lead a patient to believe that they have recovered and may even prompt them to consider stopping treatment. However, as is evident, some symptoms take considerably longer to remit and therefore it is important to emphasise to patients that they continue with treatment until they have regained full function.
Ways in which uncoupling of symptoms from ACE domains leads to mixed presentations.
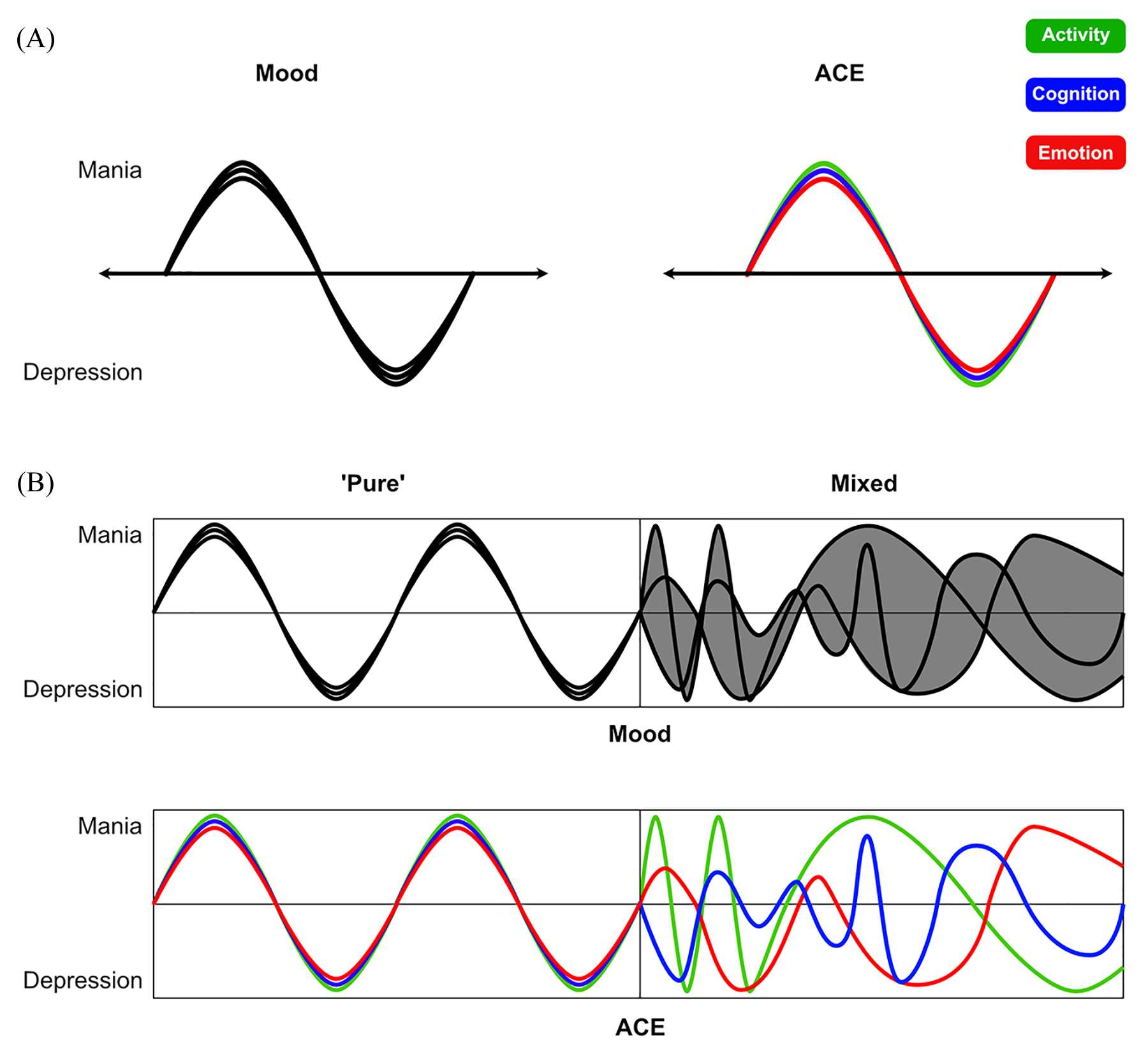
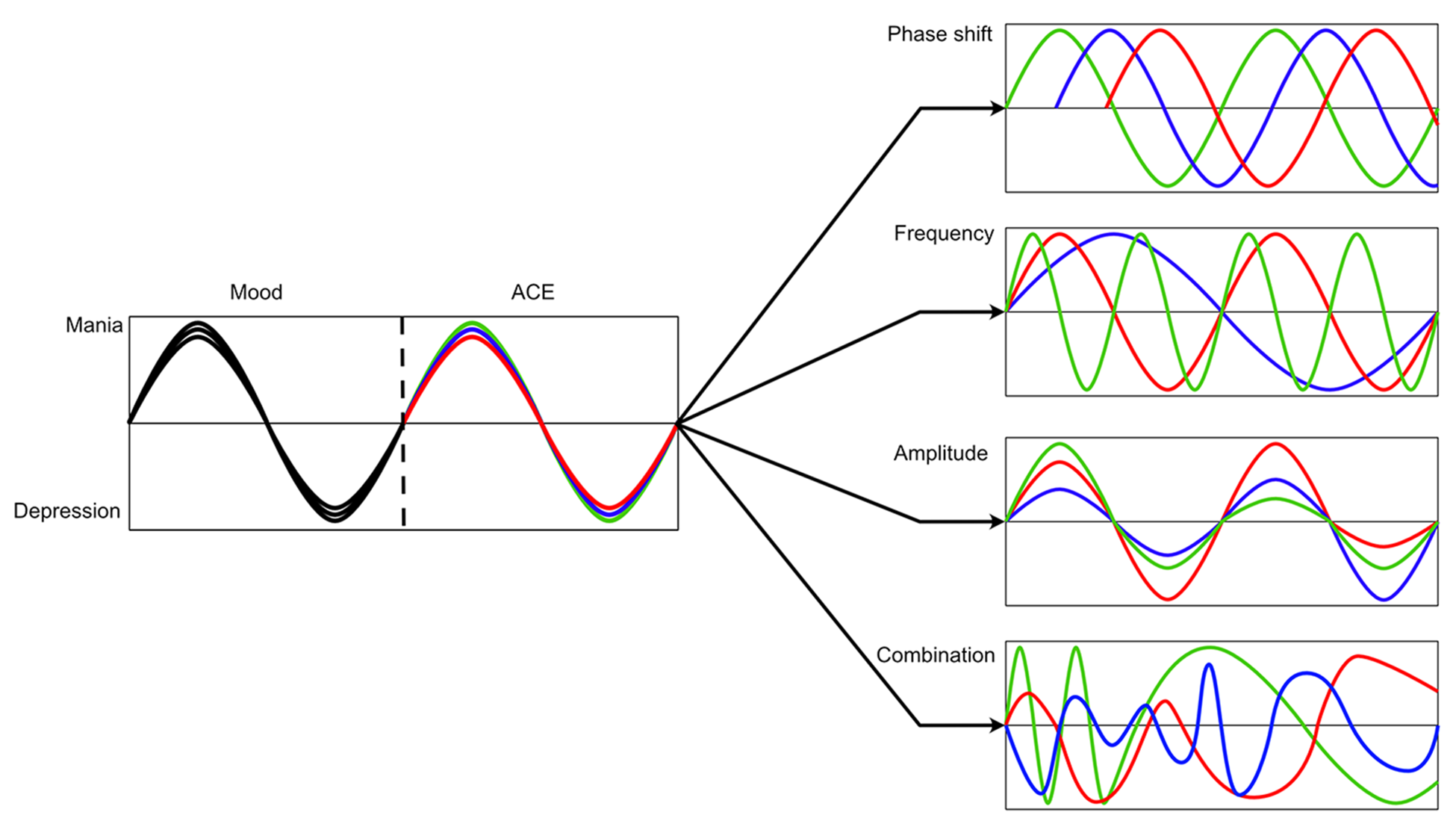
Conceptualisation of mixed states according to the ACE model. (A) The conventional model of mood disorders with changes in mood (along with other symptoms) contributing to expressions of mania and depression. These can be partitioned into activity, cognition and emotion shown respectively in green, blue, and red as part of the ACE model. (B) The pure states of depression and mania are contrasted with the common mixed presentations seen in clinical practice. If only mood is mapped, as in the upper part of the diagram, mixed states do not follow from the bipolar dichotomy of mania and depression (denoted by grey shading). They are, therefore, challenging to diagnose clinically and have proven difficult to investigate. However, if mood states are mapped using the ACE model, then mixed states can be readily understood as uncoupling of the various symptoms from different domains leading to a variety of patterns and manifestations as seen in clinical practice.
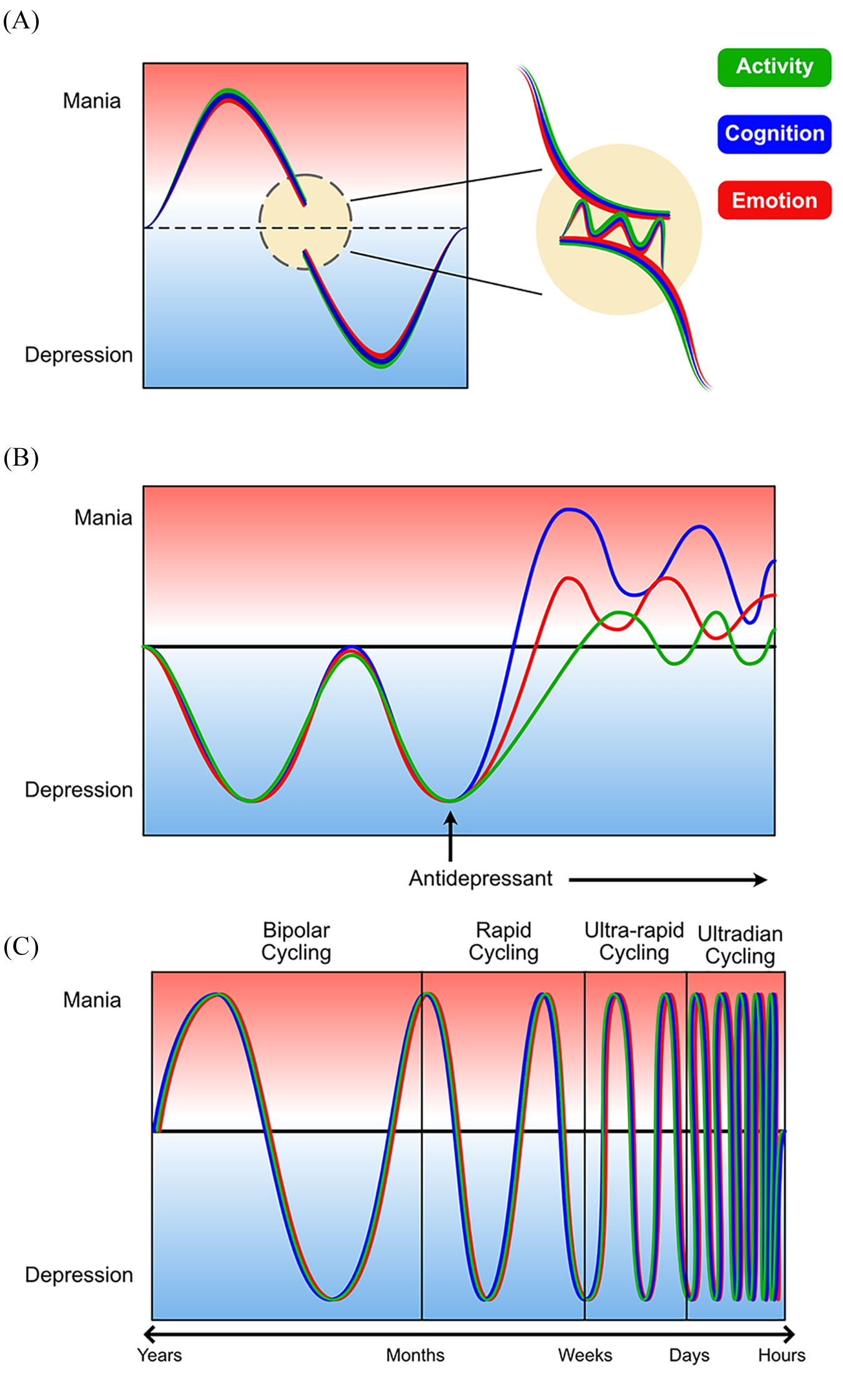
Aetiological subtypes of mixed mood states (adapted from Malhi et al., 2019e). This schematic shows the heterogeneity of mixed states illustrating three alternative phenocopies of idiopathic mixed states. (A) Transitional mixed states in which the period of transition between mania and depression inevitably involves a brief time during which symptoms from both depression and mania may be present (yellow circle). This can give the impression of a mixed mood state when in fact it is the straightforward shifting of mood from one pole to the other. Note, although the transition is shown from mania to depression, the converse is also possible. The mixed state can be further extended if the latency of individual domains is different. (B) The effect of an antidepressant which can lead to the uncoupling of the domains and thus produce a mixed state. The differential impact of an antidepressant – for example, affecting cognition more so than emotion and activity – may lead to a difference emerging in the rate of change of symptoms such that the various domains are uncoupled for a period of time. This is a treatment induced mixed state, which is different to an intrinsic (idiopathic) mixed state. (C) Shows how rapid cycling can occur at extremely high frequency such as ultra-rapid and ultradian cycling with mood swings occurring in days and hours. The rapid swings can give the impression of a mixed state with an individual appearing depressed in the morning and manic in the afternoon, for example. Such rapid changes would also be subject to transitional mixed states; however, again, this is not a true intrinsic mixed state. The aetiology of these three types of mixed states is important as treatment approaches to manage these mixed states are different to those potentially useful for intrinsic mixed states (see section 8.3. ‘Mixed states’).
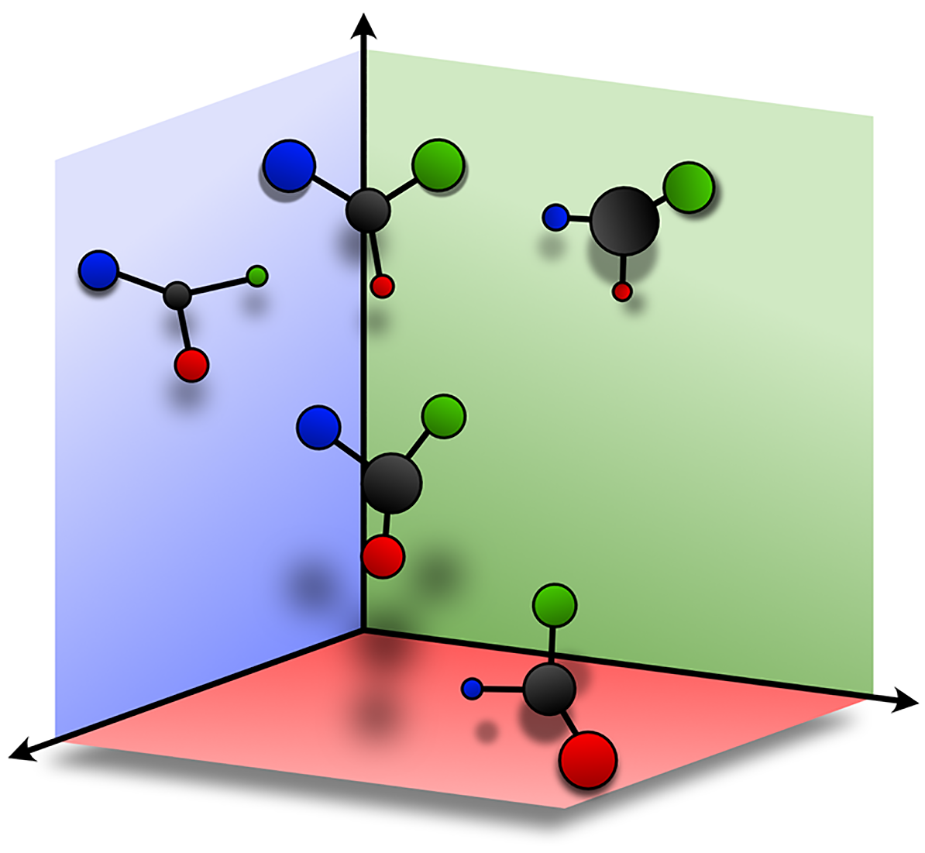
Schematic showing how symptoms relate to each of the ACE domains.
Thus, in addition to providing greater cross-sectional richness, the ACE model also affords a more granular consideration of the natural chronology of symptoms, allowing for differential response times across domains (see Figure 4).
The ACE model is yet to be widely used in clinical outcomes research, so treatment recommendations in MDcpg2020 remain largely informed by trials using the categorical DSM/ICD diagnoses. The ACE model is offered here as a useful dimensional addition to case formulation and treatment planning (Henry, 2019) (see section 4 ‘The formulation of mood disorders’). The categorical diagnoses remain pragmatically useful, and next we introduce some clinical nuances concerning these diagnoses.
Mixed states
Mood states featuring symptoms that are regarded typically as those of depression or mania, when occurring concurrently, are described as mixed states. Research studies have shown that these mixed states are relatively common in clinical practice, but remarkably they are seldom diagnosed. Previously (in DSM-IV) mixed states could be captured and coded as mixed episodes; however, this is no longer the case and so data regarding the prevalence of mixed states are scarce. In place of mixed episodes, DSM-5 introduced a specifier (‘with mixed features’, referring to the presence of symptoms from ‘the opposite pole’) that can be ‘attached’ to a mood episode such as depression or mania (Kontis and N Fountoulakis, 2019).
However, the DSM-5 mixed states specifier fails to accurately capture mixed states because, somewhat ironically, its definition of ‘at least three of the following symptoms’ (from the opposite pole) does not specify which symptoms are most important and key symptoms such as distractibility, irritability and psychomotor agitation do not feature. This means all manner of mixed states are lumped together with no consideration of the number or nature of the symptoms (Barbuti et al., 2019). Recently developed rating scales may be able to capture the clinical profile of mixed states in granular detail but as yet these are not being widely used (Malhi et al., 2017a; Zimmerman et al., 2014). In addition to using rating scales, the ACE model may assist in defining mixed states.
In the ACE model, there is no tension between having mixed states and ‘pure’ forms of depression and mania to conceptualise the nature of the component symptoms and domains of mood disorders. This is in contrast to DSM, where an impression of mixed states can only be captured by specifying symptoms occurring in a predominant depressive or manic mood state, which does not reflect reality.
This conceptualisation of mixed states is not new and it draws upon that originally proposed by Weygandt and Kraeplin. Recently, it has once again been described in detail (Malhi et al., 2018a, 2019e).
It is important to note that mixed states themselves are likely to be very heterogeneous. First, because of different causes bringing about mixed states, and second, simply because of the very many symptoms that can be variously combined to produce a myriad of presentations. Dissecting the latter clearly requires further research; however, it is important to separate the various aetiological subtypes as this greatly informs treatment strategies.
Given the potentially recurrent nature of mood disorders, the typical patterns of distribution of mood episodes in which depression and mania alternate can appear such that they are juxtaposed or that they are interspersed, shifting from one episode to the next, or dispersed with periods of euthymia separating them. It is therefore natural at times for patients to experience transition directly from mania into depression and vice versa. As one episode subsides, the other takes hold, presumably because of the underlying mechanisms gearing such changes. In these instances, there will inevitably be brief periods of time where there is intermingling of depressive and manic mood symptoms. These periods are best described as transitional mixed states, as they occur ‘naturally’ and reflect the changing pattern of mood disorders.
Another potential kind of mixed state is in fact a phenocopy wherein extremely rapid cycling of mood symptoms from depression to mania and vice versa can appear to create a state in which both depressive and manic symptoms co-occur. Rapid cycling is broadly and non-specifically defined as four or more episodes of either mania or depression occurring over a period of 12 months. 3 However, in practice, it is not uncommon for many more episodes to occur within a period of 12 months and there are many instances in which rapid cycling appears to occur with a periodicity of weeks and sometimes days. These kinds of presentations have been described as ultra-rapid cycling and ultradian. Given that an episode of mania requires symptoms to be present for 7 days or more, and depression requires symptoms to be present for at least 2 weeks, it can be seen that if the polarity of symptoms is changing within these time frames, then technically it is not possible to define either episode and so essentially a mixed state exists. However, this is not a ‘true’ mixed state per se as the symptoms are not necessarily comingled or coterminous, but instead occur with a rapidity and frequency that cannot be captured descriptively. This then is a cycling mixed state.
The third kind of mixed state that is separate from idiopathic mixed states is that which is caused/triggered by a treatment. Usually, it is when a treatment is first commenced that this type of mixed state occurs. However, sometimes it can be because of a change in dose. This type of mixed state occurs because treatments have differential effects on the various symptoms and domains and if this difference in efficacy is marked, then an effect may occur almost exclusively within symptoms within one or two of the three domains. This then leads to uncoupling between activity, cognition and emotion, which clinically manifests as a mixed mood state.
Therefore, in sum, there are four types of mixed states: transitional, treatment-induced, cycling and idiopathic.
Mixed (mood) states versus affective dysregulation in borderline personality disorder
Patients with borderline personality disorders (BPD) are known to suffer significant periods of affective dysregulation which can resemble Mixed States. While the clinical presentations of emotional disturbance in each of these disorders are characterised by common features of multiple emotional, cognitive and behavioural disturbances, there are important distinguishing features of each (Bassett, 2012; Bassett et al., 2017; Bayes et al., 2016a, 2019; Chapman, 2019).
The affective dysregulation evident in BPD is typically characterised by rage, irritability, affective instability or lability (changes occurring over minutes or hours), and often prominent efforts to prevent abandonment despite abrasive interpersonal behaviours. Depressed and anxious mood may be prominent, but usually less intensely than anger. Indeed, interpersonal difficulties frequently precipitate affective instability. The pursuit of important life goals is often severely disrupted, particularly significant relationships.
In contrast, mixed mood states in bipolar disorders typically comprise combinations of a variety of affective symptoms (e.g. depression and mania), with concomitant disruptions of thought processes and behaviours. Changes in these states extend over hours or days, and only occasionally include rage as a prominent emotional state. Depressed mood is more common, although elevated mood also occurs. The development of mixed affective states tends to occur with a prominent level of autonomy, only being linked to external events and interpersonal interactions to a limited degree.
2.4. Severity
The categorisation of mood disorders according to severity is widely used in clinical practice. Severity serves as a shorthand for acuity of illness and the need for different kinds of interventions. It is also used to indicate different types of depression. For example, ‘severe depression’ is often used to indicate melancholic and psychotic presentations of mood disorders and those featuring suicidal thinking. Clinically, depression is also often described as mild or moderate – suggesting that it can perhaps be managed within the community or that it is likely to be responsive to psychological interventions alone.
In DSM-5, severity is implied by the number of symptoms the individual has and the extent to which they are functionally impaired. Similarly, the effect on functioning is captured in ICD-10/11 with depressive episodes described as mild, moderate or severe on the basis of the number of symptoms they are experiencing and the degree of distress this causes along with their impact on activities.
However, these broad bands of severity (mild, moderate, severe), while pragmatic and sometimes useful, are difficult to apply with consistency and often do not provide sufficient detail to meaningfully inform treatment. Therefore, where possible, other schemas (e.g. ACE model and subtypes) should be used in conjunction with an assessment of severity to inform management (see section 4.1. ‘Formulation’ and Figure 12).
2.5. Subtypes
Depression is heterogeneous and is thought to comprise a number of subtypes (see MDcpg2015, page 102). Each subtype is characterised phenotypically by the prominence of particular symptoms (see Figures 9–12). Clinically, specifying depression as a subtype can be helpful in determining management. Anxious distress, for example, denotes high levels of anxiety and possible risk of suicide. Mixed features reflect a bipolar diathesis and require caution when prescribing conventional antidepressants. Some subtypes respond preferentially to particular medications. For example, melancholia, which is often characterised by anhedonia, psychomotor changes and guilt, is more likely to benefit from broad-spectrum antidepressant treatment and agents that engage dopaminergic and noradrenergic neurotransmission (Malhi et al., 2005). This is even more the case with psychotic depression in which the features of psychosis can be mood congruent or incongruent and typically involve delusions and sometimes even hallucinations. In this subtype, antipsychotic medication along with antidepressants is more effective than either alone and ECT is sometimes needed (see section 4.1. ‘Formulation’, for integration of schemas when determining treatment).
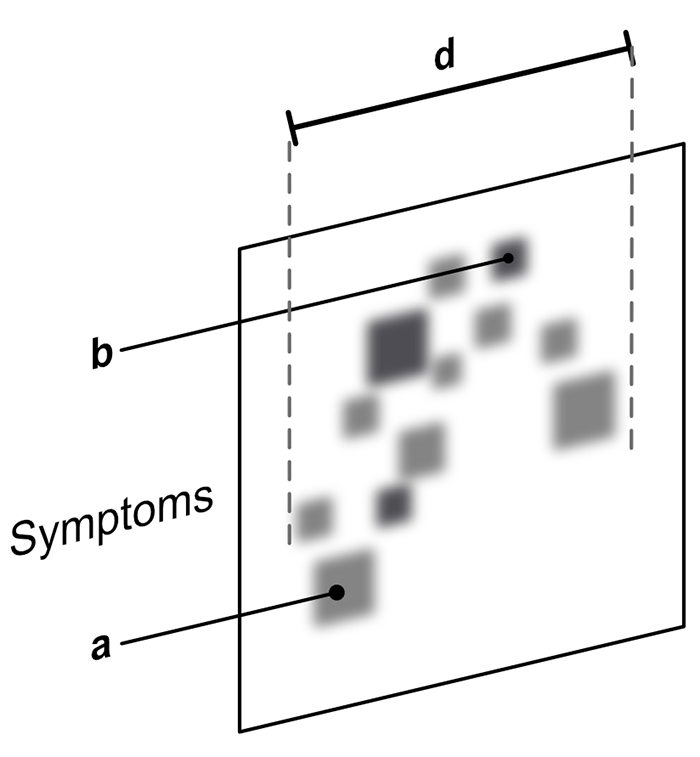
Diagnostic approaches to mood disorders – symptoms.
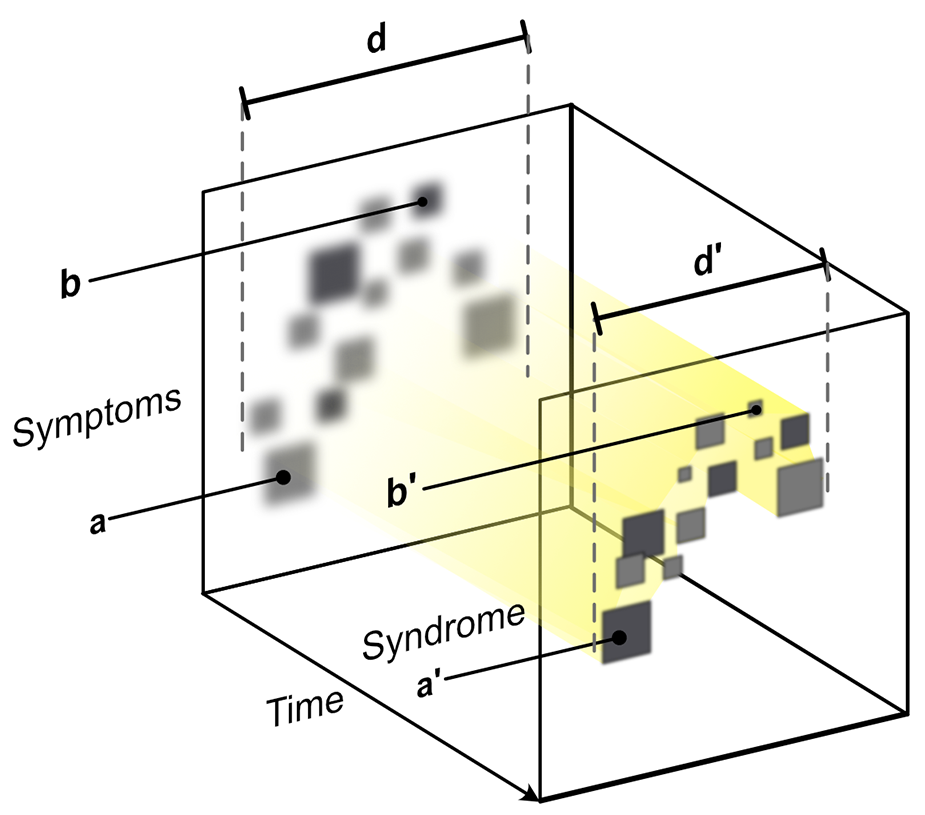
Diagnostic approaches to mood disorders – syndromes.
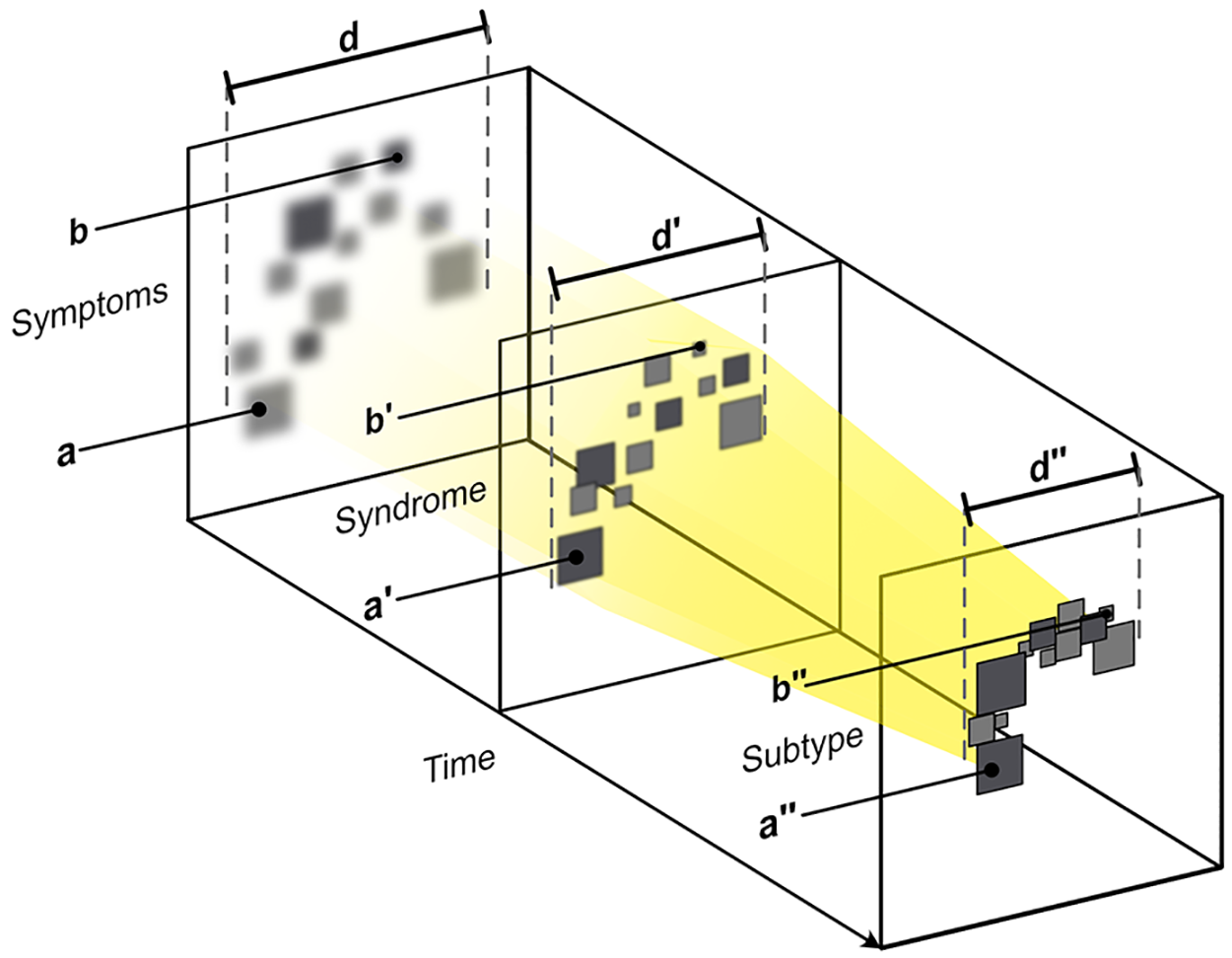
Diagnostic approaches to mood disorders – subtypes.
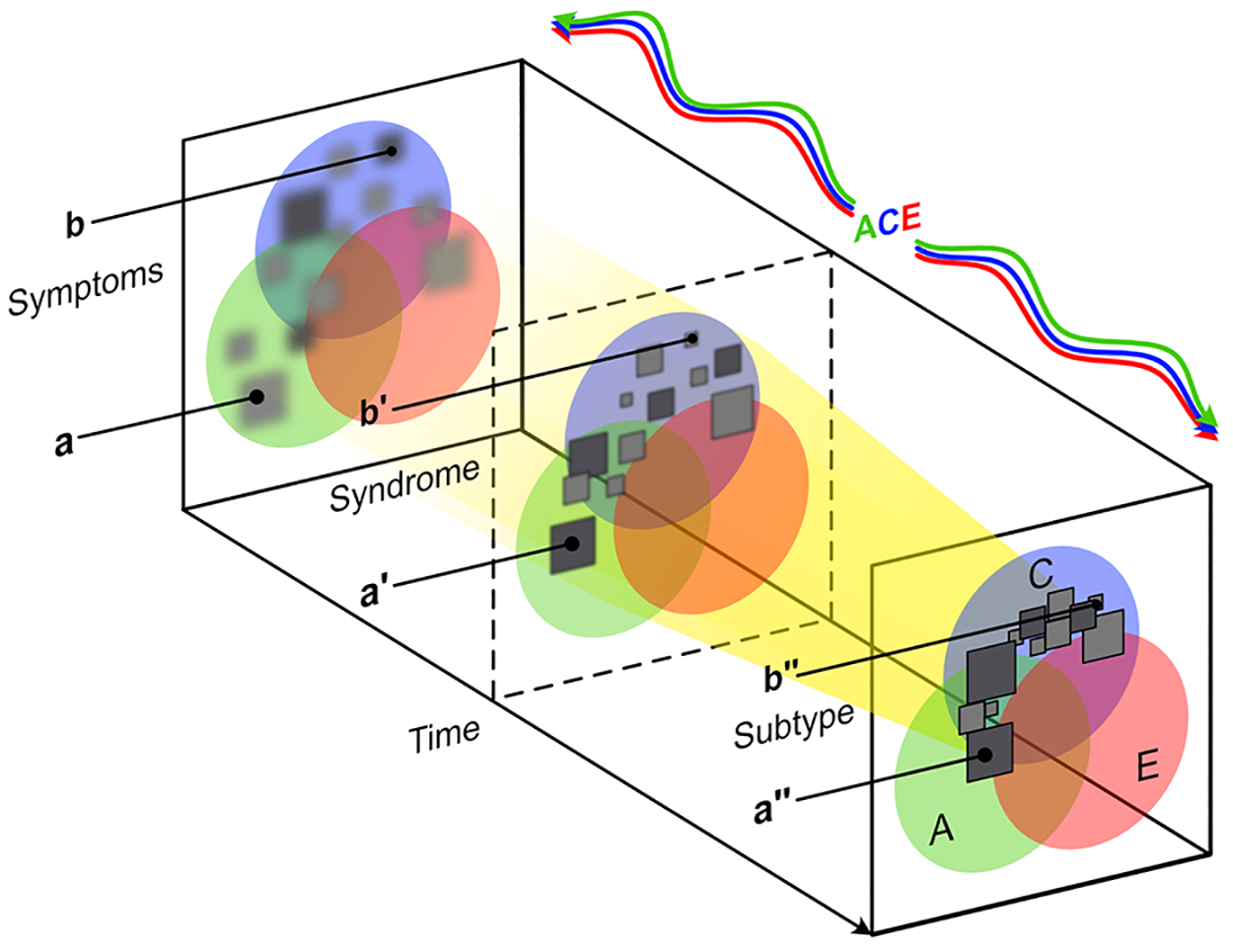
The unification of diagnostic approaches for mood disorders.
The unification of diagnostic approaches for mood disorders
Greater precision can be achieved by employing a number of diagnostic approaches in unison. Figures 9–12 show the emergence of a mood disorder and illustrate the integration of several approaches (Figure 12). These are explained separately in Figures 9–12 as symptoms progress to form a syndrome and then attract a mood disorder diagnosis (Figures adapted from Malhi et al., 2020b).
2.6. Bereavement
Depression after bereavement is common, with longitudinal assessments indicating that 20–25% of bereaved people develop depression at some point (Galatzer-Levy and Bonanno, 2012; Maccallum et al., 2015; Pham et al., 2018). Prior to DSM-5, clinicians were advised to use caution in diagnosing depression after bereavement because it may result in over-diagnosis in the context of expected levels of grief. In DSM-5, this ‘bereavement exclusion’ was removed because of evidence that depression in the wake of bereavement is not qualitatively different from depression following other life stressors (Kendler et al., 2008) and subsequent evidence has supported this decision (Jozwiak et al., 2013).
DSM-5’s decision to remove the bereavement exclusion from major depressive disorder (MDD) was widely criticised as ‘medicalising grief’, but DSM-5 is clear that (a) understandable and culturally appropriate responses to life events (like grief as a response to bereavement) are not mental disorders and (b) MDD is a mental disorder which can be triggered by stressors including, but not limited to, bereavement (see section 3.1 ‘Stress’).
Considerable evidence has now accumulated that a third possible mood outcome of bereavement can be delineated. ICD-11 has introduced the new diagnosis of prolonged grief disorder, a symptom constellation that can be differentiated from both normal grief and major depression. The disorder is characterised by persistent longing for the deceased that is associated with emotional pain and can also involve a sense of meaninglessness, inability to accept the death, difficulty in engaging with future activities or other relationships, and loss of identity. In practice, it is very common to have comorbid major depressive disorder and prolonged grief disorder, and therefore clinically, the main point to note in making a differential diagnosis is to elicit the presence of persistent yearning or longing for the deceased that impedes one’s ability to function (Shear, 2015).
The clinical significance of this new diagnosis is that prolonged grief disorder may warrant a specific treatment approach. A series of controlled trials have shown that cognitive behaviour therapy involving emotional processing of the loss via a form of exposure therapy, cognitive reframing of beliefs about the loss, and structured activities to bolster future goal setting and activity scheduling leads to a marked reduction of prolonged grief symptoms (Bryant et al., 2014; Shear et al., 2014). Importantly, this approach has been shown to be superior to interpersonal psychological therapy (Shear et al., 2005) and antidepressant treatment (Shear et al., 2016).
2.7. Mood disorders and personality disorders
The forthcoming version of the International Classification of Diseases (ICD-11) allows clinicians to describe the core personality function as mild, moderate or severe when characterising personality disorder (PD; Tyrer et al., 2019). Clinicians can then choose to describe features of personality disorder by specifying one or more trait domains: negative affectivity, detachment, disinhibition, dissociality and anankastia. Additionally, a borderline pattern qualifier may be used. Assessing core personality functioning may be more useful than assessing specific PD types since it is more predictive of psychosocial functioning (Crawford et al., 2011; Morey et al., 2013). Patients struggling with impaired capacities for emotional regulation, self-worth and intimacy may be more likely to experience depression and less likely to remit (Bach, 2018). Accessing core personality functioning should help inform clinical management of patients with mild to moderate depression according to the level of their personality dysfunction. Those with more personality dysfunction may need more intensive and supportive treatment (Bach, 2018).
The distinction between BPD and mood disorders has become clearer. Developmentally, patients with BPD have been reported to have higher prevalence and greater severity of adverse childhood events, particularly sexual abuse, and depersonalisation during childhood (Bayes et al., 2016b). Clinically, the traits of emotional lability, anxiousness, separation insecurity, hostility, impulsivity, risk taking and chaotic interpersonal relationships are significantly more prominent in BPD than bipolar disorder (Fowler et al., 2019).
2.8. Adolescents and children
Major depression
MDD is relatively uncommon in children and unlike its distribution in adults, it affects males and females equally. However, the incidence of MDD rises from mid-adolescence, when the M:F ratio begins to approach that seen in adult populations. The predominant symptom of childhood depression may be irritability rather than depressed mood, and other typical features include an insidious onset and periods of normal reactivity, such as when playing with friends. Melancholic features are extremely rare and invite a search for an organic cause such as prolactinoma. Hallucinations may be reported but are often a marker of traumatic experiences rather than psychosis (Hielscher et al., 2018; Nam et al., 2016). Finally, death by suicide is very uncommon in prepubertal children, but many depressed children harbour suicidal thoughts.
Bipolar disorder
Early-onset BD can occur in young adolescents but is rare in children. The unofficial term ‘paediatric bipolar disorder’ has been used to describe young people with classic episodic and severe mood fluctuation, but confusingly has also been applied to children with persistent and severe hyperactivity, impulsivity, affective dysregulation and explosive temper (Duffy et al., 2020; Vaudreuil et al., 2019). The case for the latter being a variant or subtype of BD is unproven and the reliable diagnosis of bipolar disorder in young children remains questionable (Malhi and Bell, 2020).
Disruptive mood dysregulation disorder (DMDD)
A new category Disruptive Mood Dysregulation Disorder (DMDD) was created in DSM-5 to describe children who have persistent irritability. Children with persistent irritability may go onto develop MDD, which is why DMDD was included for classificatory purposes within depressive disorders. Part of the motivation for developing a diagnosis of DMDD was to stem the over-diagnosis of BD in children, and DMDD remains contentious because of its high overlap with Oppositional Defiant Disorder (Mayes et al., 2016) and because of difficulties in accurately applying its criteria in practice (Malhi and Bell, 2019b). As a risk marker for the subsequent onset of MDD, the diagnosis of DMDD is redundant, as its features are already captured by the irritability dimension (as opposed to the vindictive/defiant dimension) of Oppositional Defiant Disorder (Déry et al., 2017). Indeed, the World Health Organization’s International Classification of Diseases, 11th Revision (ICD-11) panel of experts recommend that DMDD symptoms are classified as an ODD specifier rather than a separate diagnosis.
3. Aetiology and pathogenesis of mood disorders
The aetiology and pathogenesis of mood disorders includes a multitude of biopsychosocial factors operating in an interacting and dynamic fashion (see MDcpg2015, Figure 3). Beyond this general statement about complexity, the specific aetiological cascades ending in diagnosed mood disorders remain elusive. Indeed, disappointing progress towards understanding specific aetiologies of the existing diagnoses – neither neuroscience nor genetics has produced a laboratory test for any mood disorders – is a primary motivation for the sea-change towards dimensional approaches like the ACE model (Insel, 2014; Malhi et al., 2018a).
Four prominent and complementary approaches to mood disorder aetiology are outlined here: the role of stress and coping; genetics and gene-environment interactions; emerging evidence for the role of circadian function; and the importance of cognition in both the aetiology and ongoing functional compromise associated with mood disorders (Figure 13).
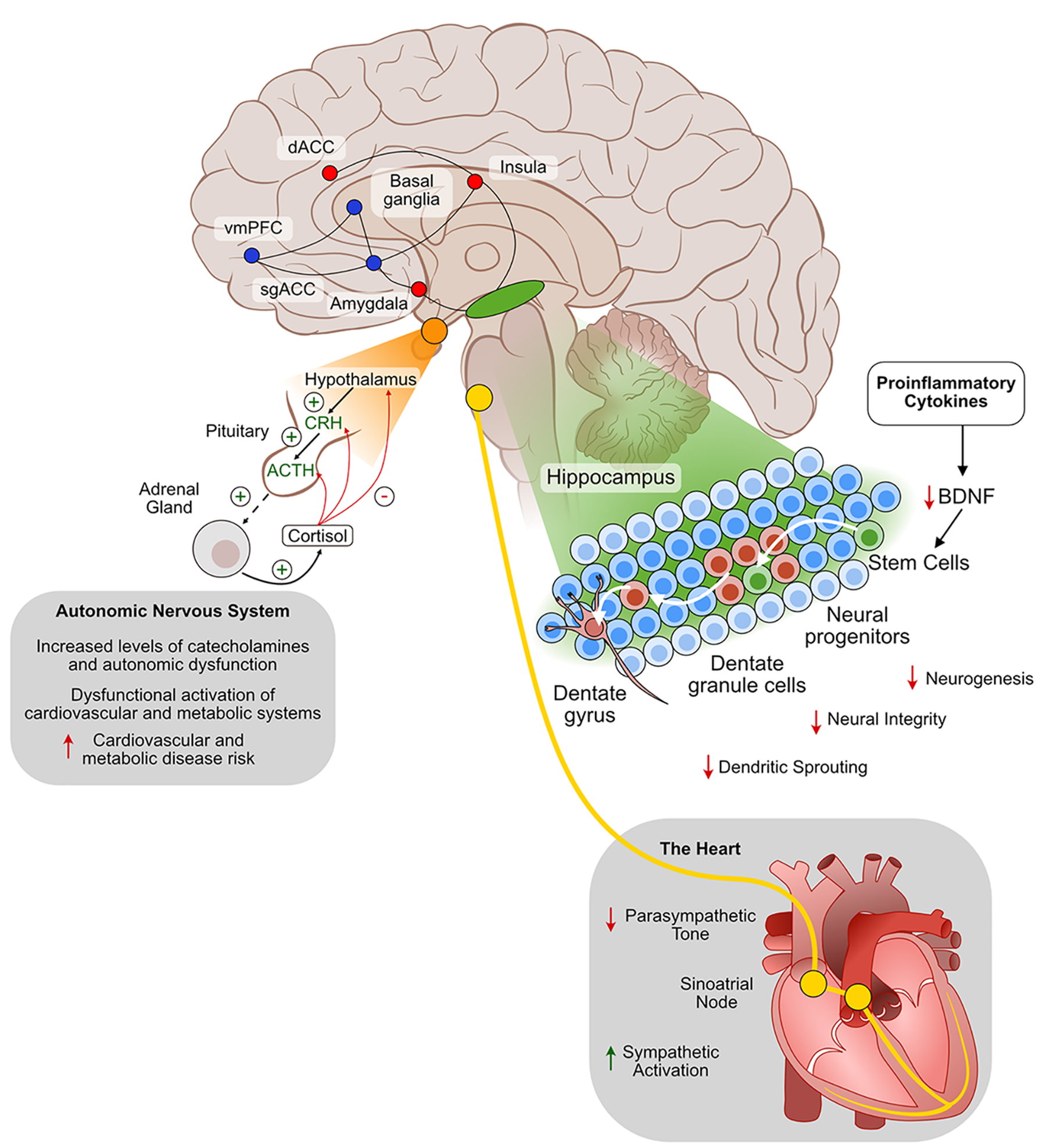
The neurobiology of mood disorders.
3.1. Stress
Research into stress as a risk factor for mood disorders has focused primarily on two classes of psychosocial stressor, distal stress related to childhood adversity and proximal stress of life events in adulthood (see Uher, 2014, for a comprehensive list of environmental risk factors across development).
Childhood maltreatment (trauma/abuse/neglect) is an important environmental factor in the aetiology of mood disorders (Brühl et al., 2019; Buckman et al., 2018). A large cohort study of over 11 million adults in the United Kingdom, for example, found that those who disclosed childhood maltreatment were 2.14 times more likely to have a psychiatric diagnosis – with mood disorders featuring particularly prominently (Chandan et al., 2019). Earlier literature identified an even higher relative risk, with a fourfold increase in risk of depression in women who have survived childhood abuse (Mullen et al., 1996). Girls are 2–3 times more likely to be victims of sexual abuse, the sequelae of which are recognised as a significant driver of the 2 to 1 predominance of depression in females in adolescents and adults (Piccinelli and Wilkinson, 2000).
While extant research has shown that early life stressors are particularly influential in lifetime risk for depression, adult stressors also increase risk (Gibb et al., 2003; Harkness et al., 2006). An association between stressors and risk for depression has been demonstrated for both acute (Kendler et al., 1998) and chronic (Hammen et al., 2009) stressors. Stressful life events include life-threatening or chronic illness, financial difficulties, loss of employment, separation, bereavement and being subjected to violence. Interpersonal trauma appears to be more prognostic of relapse than daily stressors (Beshai et al., 2011). There is some evidence that stressful life events may be more strongly associated with first and second episodes of depression than later ones (Stroud et al., 2008), and a similar pattern of decreasing (though still significant) impact with episodes has been demonstrated in bipolar disorder (Kemner et al., 2015).
The role of stress has been less researched in bipolar disorder than unipolar depression, but similar patterns are emerging. Childhood adversity (including early emotional trauma, parental psychopathology and family conflict) increases risk of developing bipolar spectrum disorders (Palmier-Claus et al., 2016) and is linked to a poorer clinical picture (Farias et al., 2019) and prognosis (Agnew-Blais and Danese, 2016) among people with a bipolar diagnosis. Among people diagnosed with bipolar disorder, recent life stress and other negative events increase depressive relapse risk (Rowland and Marwaha, 2018), while manic relapse may be more strongly linked to goal-attainment events (e.g. getting married, graduating) (Johnson et al., 2017).
There is evidence that physical abuse and sexual maltreatment in childhood predict first onset and recurrent mania (Gilman et al., 2015). Among those with childhood adversity, Gilman et al. found that past-year life events in adulthood (interpersonal instability and financial hardship) contributed additional risk for first onset mania, but only among those with a history of childhood physical abuse or neglect. For those with a childhood history of sexual maltreatment, on the other hand, Gilman et al. (2015) found that the extreme effects of adverse childhood environment on first onset mania obscured any signal of additional stress arising in adulthood.
The impact of stress on multiple bodily systems (with potentially sensitising consequences for future stress) has been well demonstrated. These effects include neurochemical changes implicated in depressive states (Anisman, 2009). Stressors can activate cytokines, stimulate growth factors (e.g. brain-derived neurotrophic factor) and trigger the release of HPA axis-related hormones, which can in turn impact depression (Cassiers et al., 2019). Experiences of chronic and uncontrollable stress (as emphasised in learned helplessness models of depression) exacerbate negative attributional style (Alloy et al., 1984) and trigger a self-reinforcing cascade of neuroendocrine and inflammatory processes that result in further sensitisation to depressive states among susceptible individuals (Cassiers et al., 2019; Richter-Levin and Xu, 2018). Differential methylation in human spermatozoa has been found in victims of childhood maltreatment, providing a possible mechanistic link between such environmental adversity and epigenetic modification of gene expression (Roberts et al., 2018).
Animal and clinical studies have linked early childhood trauma to depression via changes in the HPA axis, particularly glucocorticoid receptor hypofunction (Stetler and Miller, 2011), and even more specifically childhood adversity results in DNA methylation of key sites in the glucocorticoid receptor gene reducing its expression (Entringer et al., 2015). Not surprisingly, there is also evidence of effects of early childhood adversity on psychological processes (including dysfunctional cognitive schemas, learned helplessness, etc.), which, analogous to the sensitisation of the HPA axis and DNA methylation, predispose the individual to strong emotional responses and an inability to cope in the face of later stressors (Tafet and Nemeroff, 2015). Therefore, through neurobiological, psychological and behavioural mechanisms, exposure to emotional neglect, or sexual and physical abuse, has a significant and profound effect on the likelihood, severity and chronicity of major depression (Malhi and Mann, 2018). Diathesis-stress models of mood disorder posit that these underlying genetic, neurobiological, or cognitive vulnerabilities interact with environmental stressors to produce symptoms and syndromes (Border et al., 2019; Levinson, 2006; Southwick et al., 2004).
In the context of stress, it is particularly worth noting that prolonged and repeated stressors can impact the biological, psychological and social systems that modulate mood. Prolonged and repeated exposure to natural disasters, such as bushfires, floods and earthquakes can increase mood disorders (Bryant et al., 2017). For example, there is emerging evidence in relation to the COVID-19 pandemic that suggests social isolation, fears of transmission and financial stress arising from economic downturn can lead to increased mood disorders (Li et al., 2020). Clinicians should be sensitive to contextual factors that may affect the mood states of people who ordinarily would not have developed mood disorders.
Thus far, we have presented data on stress as an objective phenomenon, but of course the same life event may have very different meanings and consequences for different people (Luhmann et al., 2012; Park, 2010). Relevant scientific models of this dynamic include Lazarus and Folkman’s seminal transactional theory of stress and coping (Lazarus and Folkman, 1984), which emphasises that personal outcomes of stress depend on the attributions (interpretations of the event) and coping behaviours (attempts to deal with stress) that the person brings to the challenge. This critical mediating role of the person in determining the consequences of stress has dominated psychological approaches to stress for the past 50 years and still underpins CBT and related psychological treatments of mood disorders.
Coping explains moderate amounts of variance in cross-sectional and prospective depression risk, playing multiple roles in developmental cascades towards mood disorder (Zimmer-Gembeck and Skinner, 2016). Successful coping with adversity can also decrease risk for mood disorder and resilience is a protective factor with adaptive components that are developed primarily in the context of adversity (Malhi et al., 2019b). As highlighted in MDcpg2015, the development of resilience is a key target of all treatments for mood disorders.
Consistent with Lazarus and Folkman’s transactional theory of stress, there is evidence for bidirectional relationships between stress and mood problems. Specifically, the psychological sequelae of depression may contribute to the occurrence of future stressful events, which in turn can compound the likelihood of further depression thus creating a vicious cycle (Hammen, 1991). Contributing factors include cognitive attributional style, personality traits (especially trait neuroticism), attachment styles, interpersonal relationships and coping behaviours (Liu and Alloy, 2010) (contemporary research in this domain is often framed in terms of emotion regulation (Aldao et al., 2010). There is some evidence that the hypothesis can be extended to bipolar spectrum disorders (Bender et al., 2010).
Gender differences in the experience of, and response to, stress are one component of the substantial gender differences in prevalence of major depressive disorder. There is robust evidence that biological (e.g. hormonal), social (e.g. exposure to sexual abuse, see above) and psychological factors (e.g. attributional and coping styles) play interacting roles in the much greater risk for females (Hyde et al., 2008). An influential approach (Response Styles Theory; Nolen-Hoeksema et al., 2008) proposes that gender differences in depression are partly attributable to gender differences in coping. Specifically, that rumination can be a coping strategy that is conducive to depression, and females tend to respond to dysphoria with internalised coping (particularly rumination), while males more commonly employ externalised distraction (Leadbeater et al., 1999). Interestingly, early functional brain changes have been found in adolescent girls with emotional symptoms – thought to be the neural antecedents of later mood disorders (Das et al., 2013; Malhi et al., 2019d).
For some patients with mood disorders, there will be a less clear link to past and current stressors. While the concept of endogenous and exogenous depression has faded from prominence, there is still merit in the concept that some mood states are more closely tied to environmental stressors than others (Mendels and Cochrane, 1968). Nonetheless, robust evidence for stress as a ubiquitous (albeit non-specific) risk factor for mood disorders encourages clinicians to ask how life has been treating their patients and (from an appraisal/coping perspective) how their patients have been responding (Nemeroff, 2016; Tunnard et al., 2014).
3.2. Genetics
Family studies and monozygotic twin concordance studies demonstrate an important heritable risk for mood disorders (particularly bipolar disorders) suggesting that they are to a large degree attributable to genetic factors (McGuffin et al., 2003; Malhi et al., 2000). While this broad conclusion is beyond dispute and is a core plank in the explanation of mood disorders, more recent molecular genetic research has complicated the notion of ‘genetic factors’ in two major ways.
First, early assumptions that the mechanism of inheritance would involve a small set of genes, linked specifically to binary diagnoses, acting independently of the environment have been shown to be false (Uher and Zwicker, 2017). Genetic mechanisms likely involve many thousands of genetic variants, in reciprocal interaction with each other, environmental exposures and random factors (Craddock and Sklar, 2013; Mullins et al., 2016). The pathways from genes to diagnostic phenotypes are complex and extensive, passing through poorly understood intermediate traits at many biobehavioural levels (see the influential watershed metaphor; Keller and Miller, 2006). Moreover, there is no one-to-one association between any identified genetic characteristics and any recognised mental disorder: some two thirds of genetic associations are shared across schizophrenia and the mood disorders, for example (Cross-Disorder Group of the Psychiatric Genomics Consortium, 2013). To date, no clinically useful links between candidate gene polymorphisms and genome-wide association study genetic loci have been identified (Stahl et al., 2019; Wray et al., 2018).
Second, the heritability estimates derived from twin studies have become contentious. The ‘heritability gap’ refers to the difference between the large phenotypic variance attributed to genes by twin and family studies (approximately 60% and 45% for bipolar and unipolar disorders, respectively), and the much smaller variance attributable to genes by molecular estimates (approximately 25% and 5%, respectively; Uher and Zwicker, 2017). Explanation for the gap is not settled, but it has thrown the ‘equal environments’ assumption of twin studies in doubt (Young, 2019) and encouraged rigorous work into gene-environment (G × E) interactions (Uher and Zwicker, 2017).
Early G × E studies pointed to an association between variants of the serotonin transporter promoter region and risk of depression, contingent on exposure to trauma (Caspi et al., 2003). This finding was of great interest in providing evidence of the theoretically important notion of gene–environment interactions and also encouraging research into epigenetic mechanisms in pathogenesis. However, findings for specific gene interactions with environmental stress (including Caspi et al., 2003) have proven difficult to replicate (Border et al., 2019).
Polygenic risk scores (PRS) have greater promise as the genetic variable in putative G × E interactions. Effect sizes remain small for the PRS main effect (e.g. <2% of variance in diagnostic outcomes in a recent meta-analysis; Mistry et al., 2018). Explanatory power is smaller again for hypothesised multiplicative interactions between PRS and adverse life events (e.g. 0.12%; Arnau-Soler et al., 2019; Colodro-Conde et al., 2018). There is no evidence that any extant G × E score is clinically useful as a risk tool: early studies are showing some promise with the polygenetic risk approach (Fang et al., 2020), but effect sizes lack clinical utility. Future risk scores will combine multiple risk gene loci (polygenetic risk) with environmental risks factors (stress, trauma, microbiome) in order to help stratify vulnerability at a population level and target preventative resources to those most at risk of developing mood disorders (Malhi, 2020). The extent to which human volition (e.g. coping and attribution, see above) sets a ceiling on such predictive models is unknown.
Current trends in G × E research include a pivot from the genetics of vulnerability towards the genetics of plasticity (sensitivity to both positive and negative features of the environment, for example, Belsky and Pluess, 2009); and attention to the challenge of heterotypic continuity (the presence of disorder tends to be stable across development, while its manifestations vary; Uher and Zwicker, 2017). Mechanisms under investigation include epigenesis (stressors altering gene expression; McEwen, 2020) and changes to the microbiome (Marin et al., 2017; Painold et al., 2019). There is currently little evidence that therapeutic approaches can modify pathogenic epigenetic effects, and further research is needed before clinical applications can be considered (Cai et al., 2015; Smith et al., 2016).
In sum, we strongly encourage mood disorder case formulations to include genetic (through the proxy of family history) (Mistry et al., 2018), environmental (especially childhood trauma, but also adult stressors) and psychological risk/resilience factors (including attributional style and coping habits; Figure 14).
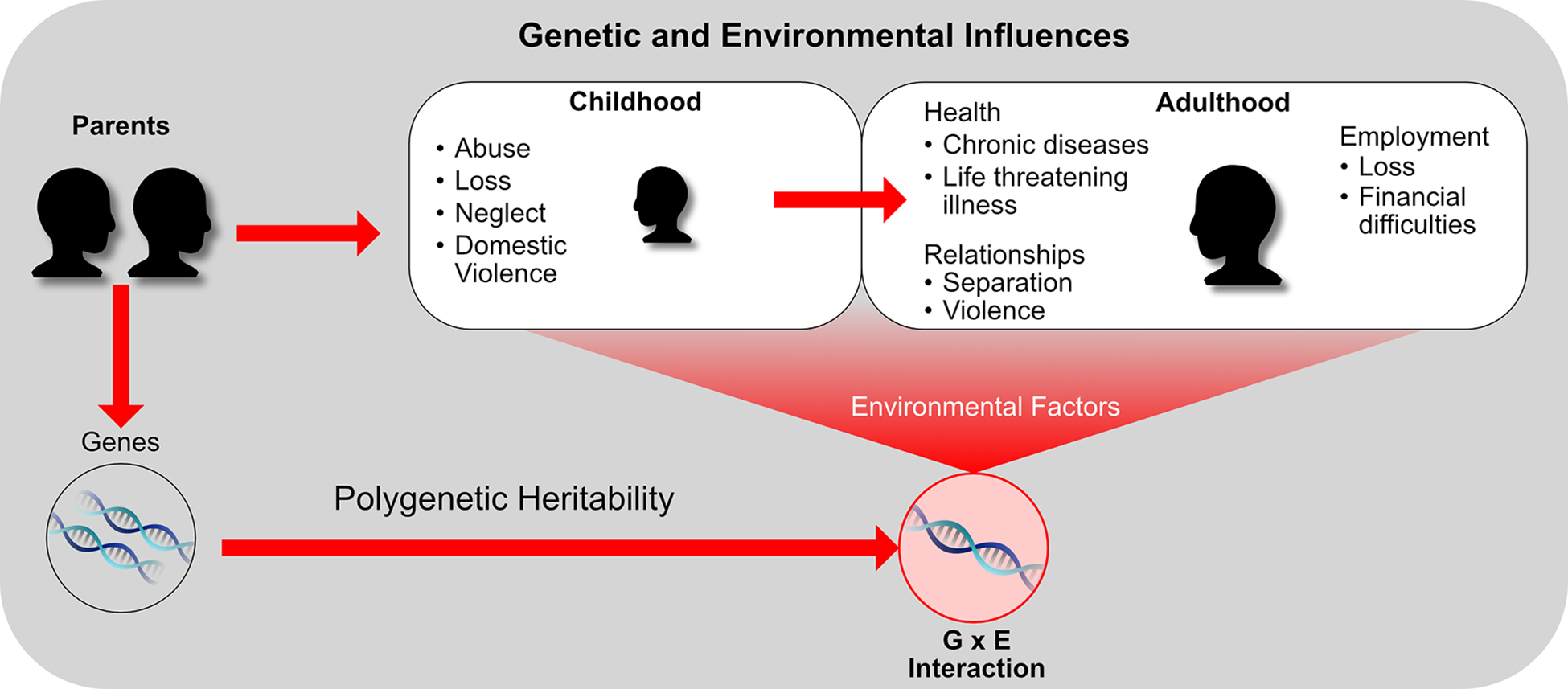
Genetic and environmental influences in the development of mood disorders.
3.3. Circadian function
Motivated by the lack of recent breakthroughs in pharmacotherapy or psychotherapy for mood disorders, increasing research attention has been directed to circadian function as a risk factor and a potential intervention target.
The circadian system is adapted to optimise coordination of internal biological, neurocognitive and psychological processes, and synchronisation of these with the planet’s 24-hour light/dark cycle (see Figure 15). In humans, the circadian system is best understood as an open, multi-level motivational system that provides the temporal framework and impetus to support more complex engagements with the environment (Murray, 2019a). Not surprisingly, then, disorders of activity, cognition and emotion are associated with circadian abnormalities.
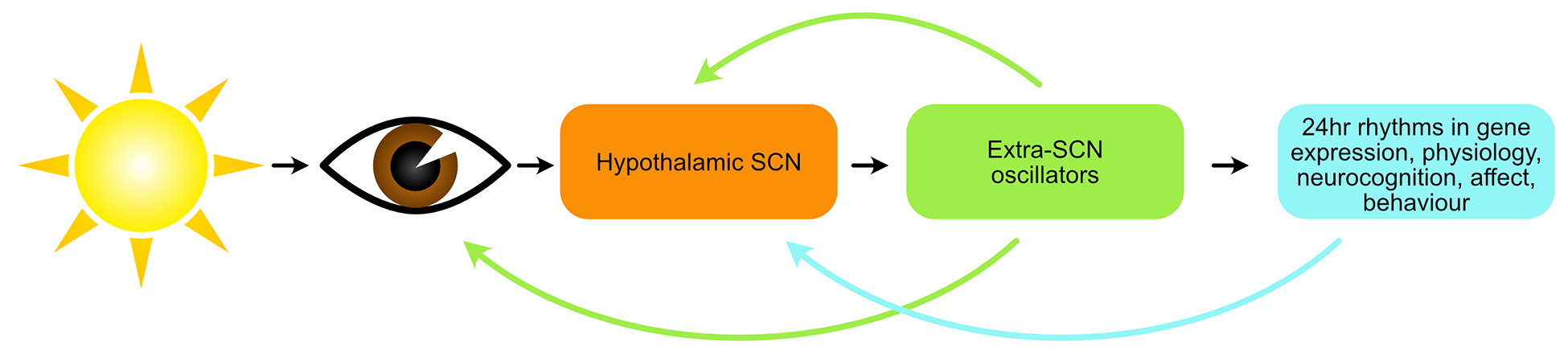
The human circadian system. Schematic representation of the human circadian system, emphasising its open, multi-level and hierarchical nature. The light-eye pathway is the primary zeitgeber mechanism in humans, but activity, meals and socialising are also known zeitgebers. (modified from Murray, 2019a). Circadian biology is well characterised at the molecular genetic level. Self-sustained rhythmicity is generated by a primary intra-cell autoregulatory transcription-translation feedback loop involving CLOCK and BMAL1, and their target genes Per1, Per2, Cry1 and Cry2, whose products form a negative-feedback loop taking approximately 24 hours (Takahashi, 2017).
This is perhaps most evident in BD, where marked disturbances of circadian rhythms (measurable physiological, cognitive or behavioural processes exhibiting the approximately 24-hour imprint of the circadian system) are evident during episodes of illness. However, abnormalities persist during periods of remission and manifest as disturbances of sleep (Harvey, 2008), social (Jones et al., 2005) and physiological rhythms (Boland et al., 2012). Abnormalities have been reliably demonstrated in daily rhythms of body temperature, melatonin secretion, activity and sleep timing, and sleep/wake cycles (Soreca, 2014). Such abnormalities could simply be epiphenomena or consequences of the disorder, but a wide range of evidence suggests that circadian abnormalities may be causal in BD and as such are of interest as a potential treatment target (Logan and McClung, 2019; Murray and Harvey, 2010).
The open nature of the circadian system (adapted to ensure daily synchronisation with seasonally-varying daylength via light information and other zeitgebers [‘time-givers’]) is important to the social zeitgeber theory of mood disorders, which proposes that weakened zeitgeber scaffolding due to stressful life events perturbs circadian function (Grandin et al., 2006).
It is useful to briefly explicate the relationship between circadian function and sleep. The circadian system is phylogenetically older and more pervasive than sleep and provides an oscillatory foundation upon which the sleep system functions. From a circadian viewpoint, then, the sleep–wake cycle can be considered the most apparent circadian rhythm in humans: sleep and wake are simply different phases of a 24-hour rhythm in behavioural engagement (Murray, 2019a). From the viewpoint of sleep per se, the circadian system is one of two primary drivers of sleep propensity. Borbély’s influential two-process model of sleep regulation (Borbély, 1982) proposes that the circadian system regulates sleep timing and architecture in a bidirectional interaction with sleep homeostasis (the build-up of sleep pressure during the day, and its dissipation at night during sleep).
Circadian and sleep disturbances may also be important in the pathophysiology of depressive episodes of MDD (Hühne et al., 2018). The core symptoms of depression imply circadian disruption such as: changes in the sleep/wake cycle; diurnal variation in the severity of mood symptoms; and variations in the daily cyclic levels of hormones and neurotransmitters (Edgar and McClung, 2013). There is also evidence that the disruption of circadian rhythms is a potential trigger for depressive episodes in vulnerable individuals (Ehlers et al., 1988).
The circadian level of explanation encapsulates many of the themes raised above in the broad overview of aetiology and pathogenesis. The involvement of genes is suggested by animal research using knock-out models; the pathways from molecular genetic clockwork to functional phenotypes are complex and varied; circadian processes are not pathologies per se but are involved in phenotypes that may underpin mood and other diagnostic syndromes (eveningness, decreased 24-hour amplitude of activity, poor sleep, etc.); a diathesis in circadian function may predispose to disorder or relapse; this vulnerability can be activated by stressors, but also moderated by coping behaviours as taught in behavioural therapies.
3.4. Cognition
Many patients with mood disorders have significant cognitive impairment upon objective assessment (Douglas et al., 2018; Miskowiak et al., 2019) that is closely linked to general functioning (Baune and Malhi, 2015; McIntyre et al., 2013) and perhaps also to the risk of relapse (Schmid and Hammar, 2013). In addition, some patients perceive that they have significant cognitive impairment. This group do not necessarily overlap with those who have objective impairment, and in practice there is often little correlation between objective and subjective impairment of cognition (Miskowiak et al., 2016; Petersen et al., 2019).
Cognitive impairment occurs during episodes of both mania and depression, but may also be present between episodes of illness in patients with recurrent mood disorders (Porter et al., 2015). The long-term impact of this impairment is not clear given the difficulty of longitudinal studies.
Given the effect on functioning and relapse, it is likely that cognitive impairment significantly worsens the course of the illness; but data is lacking. There is some evidence of progression of cognitive impairment in unipolar patients which may be related to the number of episodes individuals experience (Gorwood et al., 2008; Semkovska et al., 2019). However, it may be that episodes and cognitive impairment are markers of a more functionally debilitating illness (Porter and Douglas, 2019). Longer follow-up studies of bipolar disorder are few and the long-term outcome of cognitive impairment in bipolar disorders not well established but the current data does not suggest inexorable progression (Bora and Ozerdem, 2017). Once again, the issue of whether episodes of illness progressively worsen cognitive impairment is not yet clear.
Interestingly, not all patients are impaired and there is the suggestion that patients with both unipolar and bipolar mood disorders cluster into three groups – those with global impairment, specific impairment or not impaired in comparison with healthy subjects (Burdick et al., 2014). There are unlikely to be clear boundaries between these groups and a spectrum of impairment seems more likely. While cognitive impairment is of critical importance to patients and likely holds significant clues as to the pathophysiology of mood disorders, as yet surprisingly little is known about its determinants.
The role of cognition in mood disorders is even more complicated in the elderly, where vascular changes and Alzheimer’s disease are more common than in the general population. The onset of these diseases will compromise their cognitive functioning compounding their pre-existing dysfunction.
Some cognitive compromise arises as a consequence of treatment, although it is not clear which particular treatments increase the risk of cognitive impairment in mood disorders (Bourne et al., 2013). However, there are commonly used treatments that have the potential to cause cognitive impairment. These include, for example, tricyclic antidepressants (particularly in the elderly) and lithium (Malhi et al., 2016c). The effects of ECT on cognition are complicated and are discussed in detail below (see section ‘ECT’).
Assessment of cognition
Instruments and tools
Clinicians need to be mindful of possible cognitive impairment and should routinely enquire about this with their patients. Standardised instruments are of benefit to quantify, and monitor, the degree of impairment and can aid clinicians understanding their patients’ perception of cognitive impairment. One useful instrument, specifically designed for use in patients with bipolar disorder that can also be used in major depressive disorder, is the COBRA 4 (Ott et al., 2016; Rosa et al., 2013). When possible, patients should have brief cognitive testing. Various short and easily administered tools are available including the THINC-it computerised battery (McIntyre et al., 2017) and the screen for cognitive impairment in psychiatry (SCIP), a brief pen and paper tool which can be administered at the bedside (Ott et al., 2016).
Timing
In general, it is best to assess cognitive function when patients are euthymic. In practice, this may be difficult to achieve, in which case a pragmatic approach can be taken especially given a relative lack of evidence of correlation between severity of depression and severity of cognitive difficulties. Suggested questions to use clinically to screen for cognitive difficulties are given in Box 1.
Suggested questions for clinicians to screen for cognitive impairment in mood disorders.
Do you find that you are slower in your thinking than you used to be?
Do you find that you are more distractible than you used to be?
Do you find that you cannot hold things in your mind, such as shopping lists and telephone numbers?
Do you find that it is more difficult to learn new things?
Do you find that you forget people’s names or other things that you used to remember?
Do you find that it is difficult to plan and carry out activities that have a number of steps?
Do you find that you are able to concentrate when reading a book or newspaper, but have to keep re-reading a paragraph?
4. The formulation of mood disorders
The assessment and formulation of a mood disorder is arguably the most important aspect of clinical management as this determines all future steps. Adherence to the biopsychosocial and lifestyle model, entailing both cross-sectional and longitudinal appraisal of the clinical picture, remains the cornerstone of diagnosis and treatment. Therefore, much of the advice in the MDcpg2015 remains relevant. However, in some areas where there has been considerable development, for example, because of technological advances, more detailed monitoring of physiological and behavioural aspects of mood disorders is now possible.
To address the need for a more sophisticated approach to the assessment and formulation of a mood disorder diagnosis, the MDcpg2020 provides a multifaceted approach for the diagnosis of mood disorders.
4.1. Formulation
The formulation of a mood disorder is the end result of a process that comprises a number of components. From the clinician’s perspective, the first of these is the detection and diagnosis of an illness. This component can take place in a variety of settings (see section 4.2. ‘Setting of care’) and initially requires the individual (or those around them) to recognise that there is a problem and for them to seek help. This is usually driven by the patient experiencing distress. Therefore, at the outset of an assessment it is important to acknowledge the patient’s feelings and demonstrate that the troubling symptoms they are experiencing are recognised and that their concern is appropriate. Eliciting the symptoms of depression is relatively straightforward using open-ended questions, active listening and focused questions to ensure all mood symptoms are assessed. This can be aided by the use of questionnaires which can be self-report or clinician-administered. However, in order to arrive at a diagnosis, a threshold needs to be determined. Focusing on symptoms alone, a diagnosis can be made based on the specifications set out in classificatory systems such as DSM-5 and ICD (see section 2, ‘Classification’). However, these are summated rather crudely, and the diagnosis does not convey sufficient meaningful information regarding the clinical profile of the individual. To enhance the diagnostic usefulness of the clinical symptoms, they may be grouped according to the ACE model (see ‘Ace model’ in section 2), which provides an indication of which domains are most affected. This model also ensures a broader perspective is maintained and allows for the possibility that the main problem areas concern activity and/or cognition and not just emotion. Adding a further layer of sophistication, symptoms can be rated in terms of severity and the extent to which they impact functioning, that is, the degree to which they impair activities the individual would normally be engaged in. Severity can be rated and communicated broadly as mild, moderate or severe, indicating to some degree the acuity of the illness and the urgency with which treatment is needed. The diagnosis can be further nuanced by considering whether the symptoms at the level of syndromes form specific subtypes such as anxious distress or melancholia. These are potentially indicative of the ‘kinds’ of treatments that are likely to be of benefit. This multi-level approach to diagnosis provides considerable depth and allows for a three-dimensional perspective on any particular syndrome (see Figures 9–12). This provides the basis for planning more sophisticated treatment which allows targeting of specific domains, as well as levels of severity and subtypes of depression. This granular approach to phenomenology can be enriched further by adding a longitudinal perspective, as discussed in greater detail in the MDcpg2015, in which each episode is characterised in terms of its course, context and pattern over time. Still further texture can be added by considering the broader psychosocial factors associated with the current mood episode.
The MDcpg2015 considers in detail some important aspects of clinical assessment, such as setting the stage for management. Assessment makes use of the 5P+ model and is set against the Biopsychosocial lifestyle model (see Figure 3, MDcpg2015). Mood disorders can be further characterised with respect to the illness history and comorbidities, both past and present, and these are summarised in Figure 16. Key among these, and worthy of particular consideration, are the comorbidities of anxiety and substance misuse, concurrent medical illnesses such as heart disease, diabetes and inflammatory disorders.
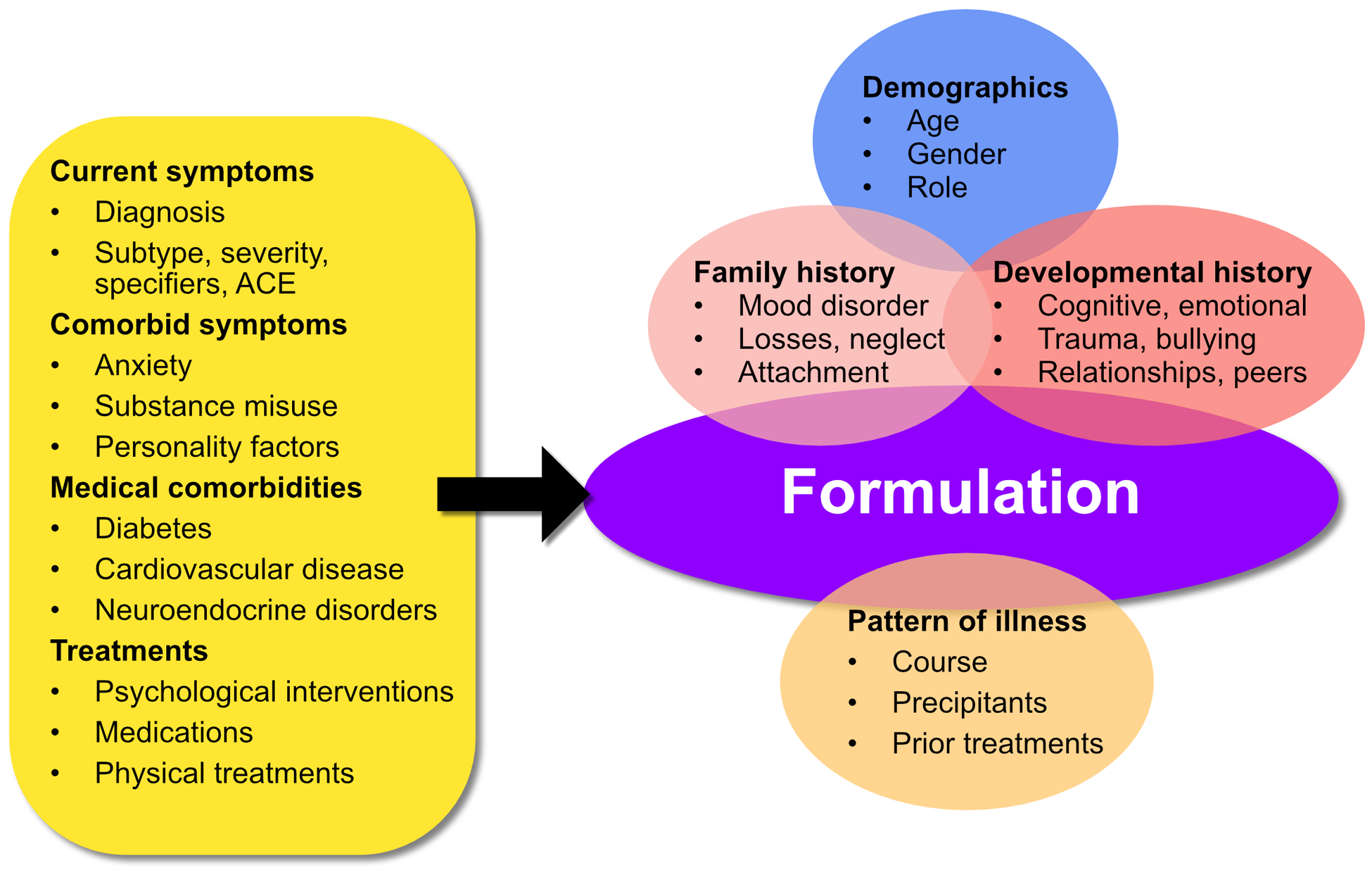
Clinical assessment and formulation of mood disorders.
4.2 Setting of care
Adults
Adult mental health services assisting Australians and New Zealanders with mood disorders are diverse and include services within private, public, primary and tertiary sectors. This diversity can result in what may seem a complex system to navigate, a point noted in many recent reviews of mental health care in both countries (NMHC, 2014). For the individual with a mood disorder this can result in inconsistent access to care and inefficient targeting of resources. The specific risk mostly relates to adequate capacity for escalation across levels of care when required. A systematic approach to mapping population need can then be utilised to enable sufficient resourcing.
In defining the desirable characteristics of a service system response to mood disorders, it is valuable to refer to the seminal work from the United Kingdom, by Goldberg and Huxley (1980), who described a tiered approach to mental health care. Specifically, they identified differing levels (and prevalence) of morbidity and levels of care. At the base is morbidity in the community, where public health measures, such as improving health literacy and self-help, are paramount. The resources available at this level of care have expanded considerably in the last decade (see www.headtohealth.gov.au/) but questions remain about appropriateness of targeting and linkage to traditional clinical services.
At the next level is morbidity in primary care, where identification of mood disorders is essential and is a filter to receiving treatment. The filter to either public or private secondary care is based to a large degree on access (geographical and economic) and on the severity and complexity of the disorder. In an Australian and New Zealand context, it is important to acknowledge the need for services that are culturally appropriate for Aboriginal and Torres Strait Islander and Māori people. Further complicating access is divergence in resource allocation between private and public psychiatry, between urban and rural or remote regions and across primary and secondary care. For example, general practitioners in isolated areas are often forced to assume responsibility for higher levels of morbidity than their metropolitan counterparts and yet have fewer specialist services.
The impact of the COVID-19 pandemic is forcing mental health professionals to utilise technology for clinical activity on a larger scale than ever before, which may ultimately assist with this issue by leading to enduring practice change.
The final level of care is that provided by inpatient care (public or private) for those with the most severe disorders. Goldberg and Huxley noted the frequent failure to recognise other levels of care by practitioners, in particular that specialist services often fail to recognise the role of primary level care. This is important because the role of primary care is critical in meeting the challenges of common mental health disorders such as depression.
An additional issue is the question of access to novel treatments through research, particularly for those patients with treatment-resistant disorders. Clinical trials relevant to patients with mood disorders are almost exclusively available in a small number of tertiary clinical or academic centres. Access is thus limited and selection criteria for trials often exclude the kind of complex comorbid pathology that is the clinical norm in specialist care. This is in contrast to other areas of medicine such as oncology where clinical trials and other research exist in a clearer continuum with clinical care.
This simple tiering of services helps direct both planning of resources and guiding individuals to the appropriate level of care. In recognition of changing treatment resources, we have suggested an additional, more formalised layer for interventions at the community level that includes the use of online education and therapy. These may occur prior to any face-to-face consultation (see Table 2).
Levels of care for mood disorders.
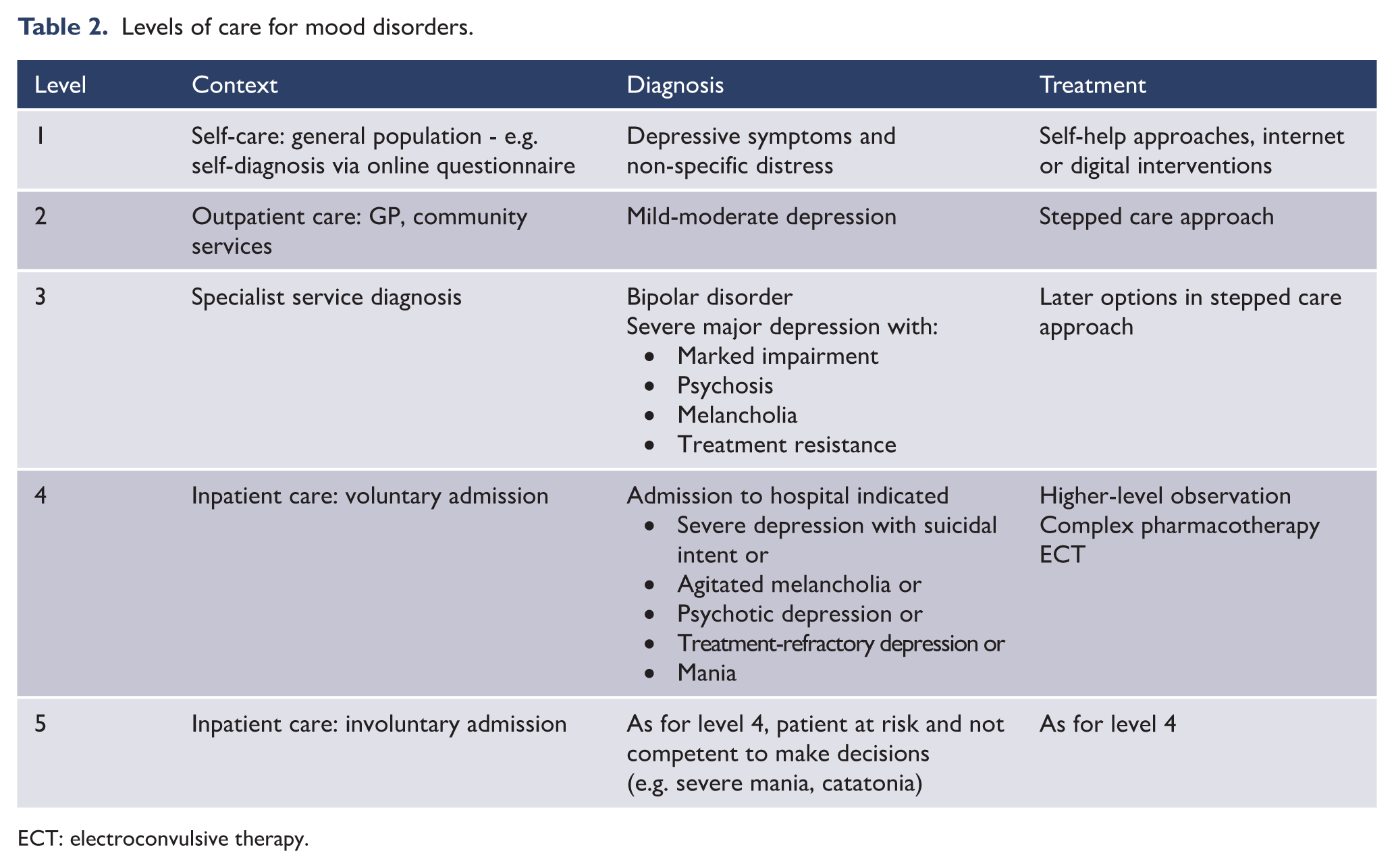
ECT: electroconvulsive therapy.
It is appropriate to consider current symptom level and issues of risk in making clinical decisions about level of care. For example, an individual may need to access the service system directly at level 5 if they have imminent risk of suicidal behaviour. Table 2 shows the five levels of care in which, depending on context and diagnosis, different levels of care are provided for the management of mood disorders.
This tiered model may also be helpful for service system development. It is vital that all patients with mood disorders have adequate access to the required resources independent of their financial situation. Currently one of the challenging access points is the transition from level 2 to 3 with either a lack of publicly accessible specialist outpatient services and/or private specialist services. This requires addressing as a public health priority.
It is important to consider the needs of Indigenous people in Mental Health Services. Various data currently suggest inequitable access. For example in New Zealand, Māori experience barriers to accessing primary health care (Ministry of Health, 2014), are less likely to be admitted to hospital for depression (Baxter, 2008) and may experience discrimination within mental health services (Johnstone and Read, 2000). Detailed discussion on alternative ‘service delivery models’ for Indigenous people was presented in the MDcpg2015, and further detailed discussion is presented in Te Rau Matatini (2015).
Children and adolescents
Child and adolescent mental health services in Australia and New Zealand are typically organised around the tier system developed for the National Health Service (NHS) in the United Kingdom. This allows some flexibility, with each level of severity bridging two tiers, and as severity of depression increases, management moves to successively higher tiers and more specified services. The system is under review, but presently is configured as follows (see Table 3 and Figure 17).
Tier system for setting of care for children and adolescents.

CAMHS: child and adolescent mental health services.

Settings of care for children and adolescents.
5. A framework for treatment: actions, choices, alternatives
A key translational challenge for any clinical practice guideline is to encourage fidelity to best practice in the general case while supporting clinicians’ expert flexibility in working with their particular patients. MDcpg2020 addresses this challenge by presenting recommendations in a hierarchy from most to least prescriptive: the guideline is organised around treatments that are essential in all cases (Actions), treatments that are available if these actions aren’t sufficient (Choices) and remaining treatments that may be considered (Alternatives). As shown in Figure 18, this approach also acts as a treatment framework to assist clinicians organise their decisions around treatments. It offers clinicians and patients alike clarity regarding treatment decisions and can be applied to determining broad options such as whether to prescribe medication, or consider physical treatments, or specific treatment decisions such as stopping or starting treatment, increasing dosage or the selection of a specific medication or intervention.

Foundations of treatment.
5.1. Aims of treatment
Starting with the World Health Organization’s holistic definition of health as not merely the absence of disease, the importance of flourishing as a focus of mental health efforts is seen in the recovery-oriented practice prescribed by national mental health policies (e.g. Commonwealth of Australia, 2013; Paterson et al., 2018; UK Department of Health, 2011; US Department of Health and Human Services, 2003), the elevation of quality of life (Morton et al., 2017) and the personal growth targets of positive psychology (Celano et al., 2020). The MDcpg2015 recommended that the aims of mood disorder treatment should go beyond symptom relief to include resilience and improved well-being, and this position continues in the MDcpg2020.
Consistent with these more holistic views of mental health, resilience refers to the ability to adapt to, and recover from, stress; not simply the absence of vulnerability (Malhi et al., 2019b). There is evidence that resilience plays a mitigating role across all stages in the management of mood disorders (see MDcpg2015, and consideration of coping responses above). Personal recovery (as contrasted with symptomatic or functional recovery) refers to the process of adaptation to serious mental illness and encourages clinicians to have a more active engagement with patients. Quality of life is a patient-valued treatment outcome which can provide a constructive compass for collaborative treatment planning (Murray et al., 2017). The consequent emphasis on empowerment and hope is particularly important in the management of mood disorders, which typically require a chronic illness self-management framework (Yatham et al., 2018).
These humanistic approaches are sometimes caricatured as overlooking the real disruptions and risks of psychopathology. In contrast, MDcpg2020 proposes that the clinician’s task is to pay proper attention across the spectrum of treatment aims (see Figure 19). The different types of treatment targets will be weighted differently for different patients at different times, but the full suite of aims should always be held in mind. As considered below, for example, the maintenance phase in mood disorder management provides a window of opportunity for clinicians and patients to shift focus from managing acute symptoms to building resilience and clarifying positive, personally meaningful goals for treatment.
The spectrum of treatment targets in mood disorder management.
5.2. Social context of clinical practice
The assessment and management of mood disorders occurs in a social context that influences access to, and cost of, care. It is our position that all individuals with mood disorders should have equitable access to affordable, evidence-based high-quality mental health care. As highlighted in recent government-supported reviews (Productivity Commission, 2019), this ideal is often not met, with lower socio-economic status and rurality associated with more limited treatment options. Multiple reports have acknowledged that access is not equitable for Aboriginal and Torres Strait Islander or Māori peoples, highlighting that this is an area that clearly still requires resolution. While digital platforms are starting to impact these inequities (see section 6.1 ‘Digital therapies’), it is incumbent upon each practitioner to maintain a sophisticated awareness of the local clinical services and social supports (e.g. financial, disability, housing, employment and educational).
Access may also vary in relation to specific treatment options. For example, there are significant variations in the availability of antidepressant medications and evidence-based psychotherapy between Australia and New Zealand and indeed other countries in our region. It is again incumbent on practitioners to have up-to-date expertise on rules and regulations in their jurisdiction, including specific indications/reimbursements in the treatment of mood disorders. We believe that it is also incumbent on organisations and individuals to advocate for full access to medications and high-quality evidence-based psychological treatments. The emphasis on digital delivery of psychological treatments in MDcpg2020 (below) is driven in part by the importance of the issue of equity of access.
6. Treatments
To do justice to the expansive literature and significant clinical challenges associated with treating mood disorders, the MDcpg2020 organises recommendations about treatment in two distinct ways. First, the existing kinds of evidence-based treatment are briefly reviewed, including discussion of their effectiveness and some recommendations for administration. For easy reference, some of these topics are presented in boxes. Section 6 adopts the Actions, Choices, Alternatives framework, beginning with those interventions that form the foundation of treatment (Actions, according to our management framework): establishment of a therapeutic relationship and an outcome monitoring regime (see Box 2 and Box 3), lifestyle interventions (exercise, diet, sleep hygiene; see Boxes 4 - 6), psychoeducation, social support and finally, provision of evidence-based psychological interventions (see Boxes 9 - 12). Section 6 also introduces newer therapies such as digital therapies and chronobiological treatments and briefly reviews the different classes of pharmacotherapy (antidepressants, mood-stabilising agents and second-generation antipsychotics) before concluding with a detailed discussion of stimulation therapies.
Therapeutic relationship.
The therapeutic relationship is a purposeful, goal-focused interpersonal relationship seeking to advance the best interests and outcomes of the client. Numerous models exist, but the therapeutic relationship is commonly associated with concepts of therapeutic friendliness (warmth and openness), respect, commitment, affirmation, recognition, responsiveness, positive regard, empathy, trust, hopefulness, receptivity, honesty and reflexivity.
It is useful to think about the many ways that a therapeutic interaction differs from a social interaction, including: confidentiality, non-judgemental stance, one-way focus of attention and care, presence of a shared problem and goal, professional’s contained emotions and limited disclosure, delivered in a care setting.
The requisite professional skills for maintaining a therapeutic relationship include self-awareness, self-knowledge, empathy, and awareness of boundaries and limits of the professional role.
In the context of psychological therapy specifically, the ‘therapeutic relationship’ is considered a sine qua non of positive outcomes. The ‘common factors’ position in psychological research argues that features of the therapeutic relationship (particularly facets of the therapeutic alliance) are not just necessary but also sufficient for positive outcomes.
For more information on maintaining effective therapeutic relationships, see 1, below.
Websites
Strategies for implementing assessments.
Many models of assessment are recognised in mental health, and we assume readers are fluent in at least one. Throughout these guidelines, for example, reference is made to assessment from a biopsychosocial and lifestyle perspective, individualised case formulation, diagnostic and dimensional approaches, acuity and the consideration of risk. However, as risk requires tailored assessment, depending on context, it is not considered here as part of the assessment of mood disorders*.
Here, we highlight one distinction that is relevant to implementing assessments with mood disorder patients, namely, the distinction between three contrasting aims of assessment – diagnosis, case formulation and monitoring.
Strategies to aid diagnostic assessment
A challenge for the clinician is to balance the need for structured information collection with the need for patients to share their experience in their own language. A useful strategy is to conduct a funnel interview, starting with the patient’s story, then signpost, for example, ‘Thank you. I think I have a good starting idea of what’s going on for you. Is it OK if I ask some more specific questions?’ Structured diagnostic information and quantitative syndrome severity information may be collected by self-report questionnaires completed and scored prior to interview.
Considerations for individualised case formulation
Develop a characterisation of the presenting problem in the context of the person’s strengths, vulnerabilities and treatment goals. The assessment template should include fields for the broader biopsychosocial and lifestyle context (e.g. genogram, current living situation). The patient should be explicitly asked for their goals for treatment (preferably framed in positive terms), and these should be among the agreed treatment goals (the clinician may have additional goals, such as reacting promptly to newly emerging symptoms).
Strategies to support treatment outcome monitoring
Given the challenge of extrapolating from clinical trial data to individual patients, clinicians must collect data about the impact of treatment (practice-based evidence). This data will be first collected at baseline as a measure of severity, and then throughout treatment. Self-monitoring can be tedious, and potentially demoralising
Reinforce monitoring by ensuring that any data provided by the patient is discussed. Address barriers to monitoring, by minimising the number of items, and providing digital data collection. Direct patients to webpages comparing mood monitoring apps for their consideration and trial (see 1, below). Choose items that validly capture treatment goals. In some cases, this will be a single bespoke item (e.g. Get through the week without becoming distressed at work), in others we may want to monitor for emergent psychiatric symptoms (e.g. using the Internal State Scale) (Bauer et al., 1991).
To support the patient’s understanding of the assessment process, see 2 below.
* For detailed discussion of risk assessment, see the RANZCP Clinical Practice Guideline for the management of deliberate self-harm see 3, below.
Websites
Strategies for instituting sleep hygiene.
Sleep hygiene refers to behaviours and environmental features that impact sleep quality and quantity (the term originally meant, literally, the cleanliness of the sleeping environment). Sleep hygiene is just one component of CBT for insomnia, and is a necessary but not sufficient intervention if significant sleep problems are present.
Steps in instituting sleep hygiene:
Explain that addressing sleep hygiene is a first, minimally intrusive step in ameliorating the sleep problems that often co-exist with mood disorders. Direct patients to good quality information (see 1, below). Sleep problems are also particularly complex and prevalent in BD, and BD-specific information can be found online (see 2, below). Follow-up by asking (at the next appointment) what information the patient found useful or surprising, and ‘Are there any barriers to applying this information in your own life?’ Patients can personalise the concept of sleep hygiene by completing the Sleep Hygiene Index (SHI) (Mastin et al., 2006). The SHI is a freely available 13-item measure. It should be augmented with one additional item addressing technology use: ‘I check email, texts, or social media during my sleep time (between going to bed and getting up)’. Patient responses to the Sleep Hygiene Index are useful to stimulate discussion and problem-solving. Targets to address may be practical (e.g. noise, light and temperature in the bedroom), behavioural (exercising too close to bedtime) or psychological (taking worries to bed).
This Way Up offers a free online CBT for insomnia program, which includes comprehensive coverage of sleep hygiene (see 3, below).
CBT: cognitive behavioural therapy; BD: bipolar disorders.
Websites
Second, we approach treatment decisions from the starting point of particular mood disorder syndromes and presentations (Sections 7 through 11). These sections build on the information presented in Section 6 by considering particular clinical challenges as treatments are delivered, paying attention to issues around sequencing and combining treatments.
6.1. Actions
Lifestyles
While the acute episodes of illness in mood disorders are important, it has become evident of late that most prophylaxis occurs when patients are relatively well. For the majority of patients, this is also the period (between episodes) in which they spend most of their lives and so it is vital that the appropriate strategies are implemented that aid psychological treatments and pharmacotherapy. These are grouped as lifestyle interventions and include both the institution of positive changes such as exercise and diet, and addressing negative habits such as smoking and substance misuse (see Boxes 7 and 8).
Nutrition and diet
The benefits of dietary modifications in the treatment of depressive disorders have been the subject of intense research and the evidence supporting the ‘Mediterranean diet’ characterised by high vegetable, fruit, fish and grain components, with low animal fat components, has increased (see Box 5 and Table 4). However, to warrant definitive recommendation, this dietary profile still requires further confirmation in well-designed clinical trials in clinical populations (Firth et al., 2019a; Li et al., 2017; Marx et al., 2017; Molendijk et al., 2018).
Strategies for instituting a healthy diet.
The content of diet has an impact upon mood disorder symptoms, psychological well-being and overall health. An optimal diet is not difficult to devise and can be very helpful, but clinicians must remain mindful of well-documented socioeconomic challenges in making healthy food decisions.
A helpful diet is a flexible, appetising and sustainable one, which is also characterised by:
Ample amounts of fruits, vegetables (including potatoes), breads (particularly whole grain), nuts and seeds. Fresh rather than processed food. Fresh fruits as preferred desserts. Dairy, poultry and fish in moderate quantities. Red meats in small amounts. Reduce saturated fats. Olive oil (or other vegetable oil if preferred) as prime source of dietary fat. Alcohol in modest quantities: maximum 1–2 standard drinks per day for males and 1 for females. Methylfolates, probiotics and Omega-3 fatty acids may be helpful.
Strategies for instituting regular exercise.
Exercise improves sleep and mood in people with mood disorders and improves general health. Motivational challenges of developing and maintaining an exercise plan are well known, and are particularly important to address for people with mood disorders.
Helping a patient build an exercise plan:
Inform the patient that exercise is a helpful adjunctive treatment for depressive disorders because it improves brain function, offers pleasurable experiences, improves self-esteem, enhances sleep quality and increases the opportunities for interactions with other people. Daily exercise is preferred over intermittent activity. Aerobic (e.g. walking, running, cycling) and resistance exercise (e.g. using weights) are both effective for depressive disorders, and the combination may be better than either alone. Some patients may find exercising with other people more motivating, but this is not universally the case. It is important to choose the form of exercise according to personal preference, remembering that previous preferences for exercise activities may help with this choice. Ideally the exercise should be pleasurable and require some effort, but not be exhausting or painful. Documenting an exercise plan is helpful, along with regular reviews of progress and difficulties. Some patients with depressive disorders struggle to find the ‘right exercise’ for them. Suggestions based upon their past activities and a review of options (e.g. start with a walk every day, perhaps with another person or pet) may be helpful. Patients will benefit from building on their exercise plans to make these activities part of their daily lives. This may start as ‘treatment’ but could progress to becoming a ‘lifestyle’.
Harvard University Medical School has a useful website to assist patients start an exercise program (see 1, below).
VicHealth has a useful survey of health-related apps, including ones to encourage regular exercise (see 2, below).
Websites
Strategies for addressing smoking.
Cigarette smoking is much more common among patients with mood disorders compared with the general population, and people with mood disorders tend to smoke more heavily than general population smokers. Smoking may also interfere with the metabolism of antidepressant and antipsychotic medications, and smoking plays a part in the decreased life expectancy that comes with a mental illness diagnosis. Nicotine is highly addictive, associated with significant withdrawal symptoms, and quitting smoking is a complex and enduring challenge for most people.
Patients benefit from reflecting on their motivations to smoke (e.g. habit, emotional benefits, social pressures), why they want to stop (e.g. health concerns, cost, pressure from others) and what would make cessation difficult (e.g. withdrawal symptoms).
Most smokers struggle to stop, and support through ‘lapses’ is essential.
Behavioural strategies for coping with withdrawal symptoms include: Exercise. Relaxation exercises/activities. Distraction at times of craving with pleasurable activities. Attention to sleep hygiene. Rest when dizzy. Being prepared is essential as unexpected discomfort is most unsettling.
Medications that can assist with withdrawal:
Nicotine replacement may help with the process of withdrawal as part of a cessation plan. Bupropion can be helpful for some patients as long as behavioural measures are also employed. Bupropion has the added benefit of being an antidepressant. Varenicline can be very effective as adjunctive treatment, but secondary depression is a significant side effect and this medication is best avoided in depressed patients.
High-quality resources to assist people with the challenge of decreasing cigarette use are freely available online (see 1 and 2, below).
Assistance for clinicians in helping people with a mental illness to quit smoking can be found online (see 3, below).
Websites
Strategies for addressing alcohol and substance misuse.
Alcohol and substance misuse problems are common in the community and are often a hidden contributing factor to mood disorders and poor treatment response. Management of drug and alcohol conditions in mood disorder populations is a multidisciplinary challenge, and clinicians should be expert on local referral networks.
Offer psychoeducation about alcohol/substance use, balancing the need to engage/support with risk of enabling/avoiding. Explain potential harmful effects of alcohol or illicit substance abuse, including impact on prognosis of mood disorder, and recommended safe levels of intake.
Good quality patient psychoeducation can be found online (see 1 and 2, below)
Assessment should include measurement of current usage (e.g. AUDIT; Saunders et al., 1993), role of substances in risk profile (particularly risk for impulsivity, suicidality, foetal alcohol syndrome). Consider Gamma-Glutamyl Transferase (GGT) screening, and collateral history.
Gauge readiness for change. Good quality information on preparedness to change behaviour is available online for patients and for clinicians (see 3 and 4 below, respectively).
Hello Sunday Morning (see 5, below) provides useful tips and an app to assist people modify alcohol use.
Turning Point website provides an overview of various approaches to addiction and substance use problems for patients and clinicians (see 6 and 7, below).
AUDIT: Alcohol Use Disorders Identification Test.
Websites
Healthy lifestyles in mood disorders.
Exercise
Regular exercise is associated with improved quality of life, and antidepressant effects in depressive and bipolar disorders (Carter et al., 2019; Melo et al., 2016; Sarris et al., 2020). Exercise – particularly aerobic exercise – should be encouraged in all patients, not least because of its general health benefits. However, a practical issue that can limit engagement with exercise is the reduction of drive (lack of motivation) that is typically associated with severe depression.
Although the optimal amount and type of exercise is yet to be determined, we do know that health benefits require aerobic activity to be vigorous (enough to require concentrated effort) and regular (at least two to three times weekly) (Sarris et al., 2020). Resistance-based exercise is also beneficial in the management of depressive symptoms, and a combination of aerobic and resistance exercises are probably optimal. The benefits of exercise for bipolar disorder management remain less clearly defined, but the benefits for general health are beyond dispute (Ashdown-Franks et al., 2019; Gordon et al., 2018; Harvey et al., 2018; Kvam et al., 2016; Schuch et al., 2016). Personal supervision of an exercise regime can assist with motivational challenges.
Chronobiological treatments
Mood and sleep processes are deeply connected with circadian function (Figure 20), and there has been growing interest in mood disorder interventions targeting circadian pathways and/or changes in sleep (Murray, 2019a).
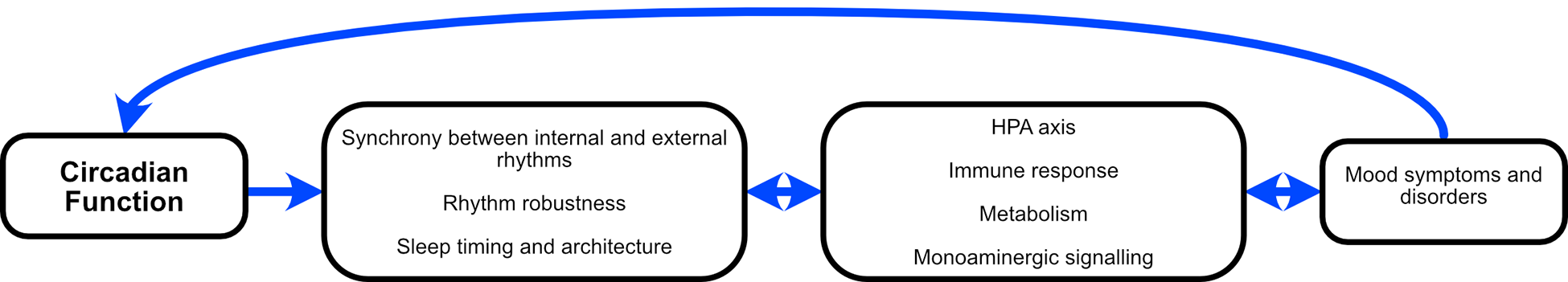
Pathways between circadian function and mood.
Five types of chronobiological interventions are recognised: bright light therapy (BLT), sleep deprivation (SD), dark therapy (DT), melatonergic agonists (MA) and behavioural interventions designed to address social rhythm instability (see ‘Psychological Treatment for Bipolar Disorder’ in section 6.1, and Figure 21). Research remains limited, with few trials comparing chronotherapies with existing first-line treatments (Cunningham et al., 2019; Gottlieb et al., 2019). However, the relatively benign side-effect profiles of most chronobiological interventions have encouraged their clinical use in circumstances where more evidence-supported modalities have failed or are inappropriate. Effective delivery requires specialist knowledge and expertise in these interventions (a good clinical introduction is provided in Wirz-Justice et al., 2009).
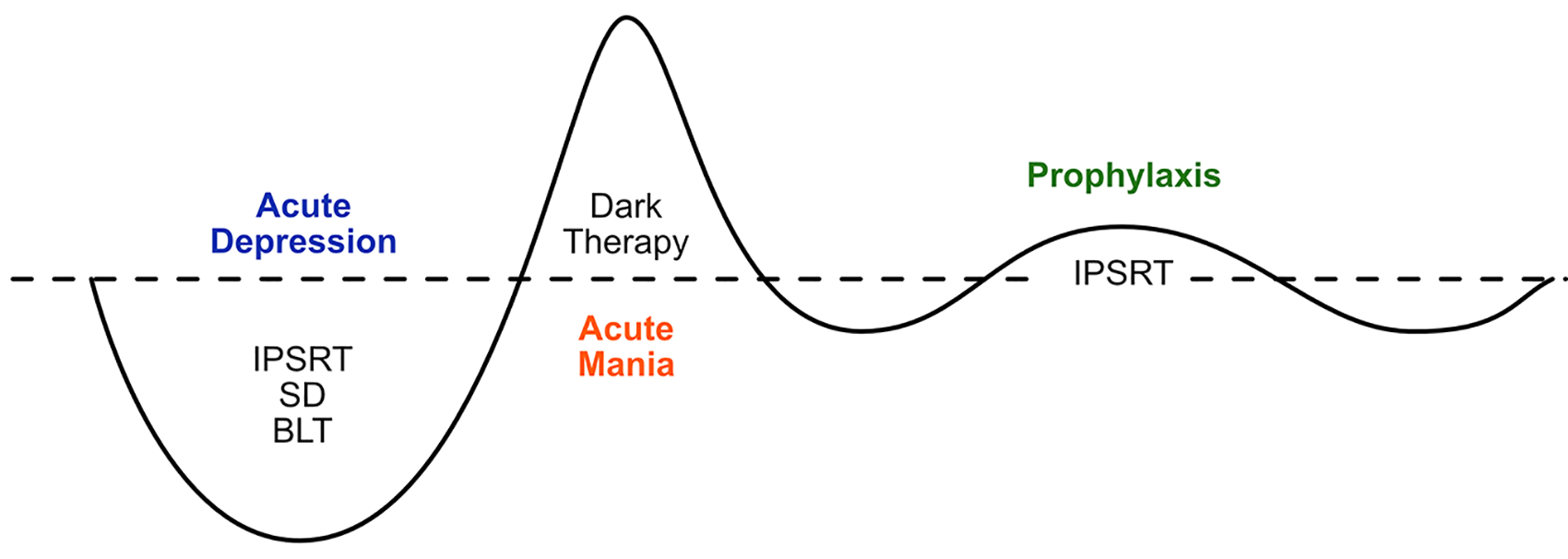
Figurative representation of the application of chronobiological interventions across the phases of mood disorder.
Acute MDD
Bright light therapy (BLT) was originally studied as a treatment for winter depression (Seasonal Affective Disorder, or MDD with seasonal pattern). Three early placebo controlled trials showed positive effects of BLT for winter depression (Eastman et al., 1998; Lam et al., 2006; Terman et al., 1998). Large placebo controlled studies and meta-analyses have subsequently confirmed its benefits as monotherapy or as adjunctive to antidepressants (Al-Karawi and Jubair, 2016; Chojnacka et al., 2016; Geoffroy et al., 2019; Martiny, 2004), and BLT can be recommended as the first-line treatment for winter depression.
Subsequently, BLT has been investigated in nonseasonal depressions. A 2016 meta-analysis concluded that moderate duration (2–5 weeks) BLT was effective in the treatment of acute nonseasonal MDD (Al-Karawi and Jubair, 2016). Interestingly, Al-Karawi and Jubair (2016) found (consistent with an earlier meta-analysis with largely non-overlapping studies, Golden et al., 2005) that benefits of BLT were only significant when light treatment was provided as monotherapy. By contrast, a more recent meta-analysis of 6000 patients (Cunningham et al., 2019) found BLT effective as an adjunct to antidepressant medication, but concluded that evidence for its antidepressant effects as monotherapy was mixed. A recognised strength of BLT is its relatively benign side-effect profile in nonseasonal MDD, and so BLT can be safely recommended as an addition to address non-response to antidepressant medication, or as monotherapy in people for whom antidepressant medication is contraindicated.
One of the challenges of BLT has been the lack of evidence-based treatment guidelines to support translation into everyday practice. Maruani and Geoffroy (2019) recently presented a version of such guidelines, distinguishing between delivery parameters and risk considerations for the different diagnostic groups. It is recommended that clinicians consult this material before applying BLT for any form of depression (including the special case of bipolar depression as discussed below).
Acute mania
Two small inpatient studies of dark therapy for acute mania have been published. Barbini et al. (2005) found 14 hours of dark (6 p.m. to 8 a.m., n = 16) adjunctive to treatment as usual (TAU) produced rapid antimanic response compared with TAU alone (n = 16) over 3 days. Henriksen et al. (2020) found amber (blue-blocking) glasses (6 p.m. to 8 a.m., n = 12) were superior to clear glasses (6 p.m. to 8 a.m., n = 11) in reducing manic symptoms across 7 days of intervention (effect size = 1.86). Barbini et al. (2005) note no adverse events, while 2 of 12 patients in the treatment group developed depressive symptoms (successfully addressed by delaying or stopping use of the amber glasses). A recent review by a chronobiology taskforce of the International Society for Bipolar Disorders (ISBD) recognised the limitations of the literature, but concluded there was evidence for blue-blocking glasses as an adjunct in the treatment of acute inpatient mania. The use of blue-blocking glasses (between 6 p.m. and 8 a.m.) should be considered a potentially effective, non-intrusive adjunctive option for mania in an inpatient setting.
Acute bipolar depression
The recent ISBD task force review concluded that there was evidence for three chronobiological interventions for bipolar depression: BLT, sleep deprivation and the behavioural Interpersonal and Social Rhythm Therapy (Gottlieb et al., 2019).
BLT (administered in the morning or at midday) has been found effective in one meta-analysis (Tseng et al., 2016) and four subsequent randomised controlled trials (RCTs) (Sit et al., 2017; Yorguner Kupeli et al., 2018; Zhou et al., 2018). However, a more recent systematic review that was limited only to RCTs, unlike Tseng et al., 2016; or Gottlieb et al., 2019, and excluded studies based on samples with less than 80% of participants diagnosed with bipolar disorder, did not find evidence of a beneficial effect compared to controls (Takeshima et al., 2020). Takeshima note that a sensitivity analysis of their data (restricted to those studies with low overall indirectness) did nonetheless replicate the positive findings for BLT reported by Gottlieb et al. (2019) and argue that more research is required.
There was originally some concern about elevated switch rate from BLT in the treatment of acute bipolar depression (Leibenluft et al., 1995; Wirz-Justice et al., 1999). Data from RCTs do not support this concern (Sit and Haigh, 2019), as long as BLT is combined with mood-stabilising medication (Zhou et al., 2018). In sum, BLT can be recommended as a safe and potentially effective treatment (adjunctive to mood-stabilising medication) for acute bipolar depressions. In terms of delivery, it is recommended that BLT for bipolar depression should be given at around midday (Sit et al., 2007), and light exposure titrated (15 minutes initially, increasing by 15 minutes per week up to 60 minutes at 1 month). Clinicians are directed to the recent diagnosis-specific guidelines developed by Maruani and Geoffroy (2019) for further clinical guidance about administration in this vulnerable population.
Total sleep deprivation (TSD) for one or more nights has long been known to bring about rapid resolution of depressive symptoms in a high proportion of patients. However, the stand-alone therapy is ultimately ineffective because rapid relapse is the norm, and the challenge has been to identify augmenting strategies to hold the antidepressant effect. A small meta-analysis (including both MDD and bipolar depression) has suggested that with adjunctive treatment (usually bright light) there is a positive effect of TSD after 7–9 weeks (Humpston et al., 2020). Most studies used 1 or 3 nights of TSD followed by morning BLT with some also using sleep phase advance. In the meta-analysis, authors note many limitations and caveats to any conclusion regarding efficacy. The TSD literature specific to BD was reviewed recently by the ISBD task force (Gottlieb et al., 2019). Once again, the evidence was considered to consist of small and variable quality studies but the treatment was recommended by this expert group on the basis that side effects and switching appeared low. However, TSD will involve a complex treatment regimen, the parameters of which remain unclear, so it cannot be recommended for general use at this time.
Redressing circadian vulnerability for BD relapse prevention
The social zeitgeber hypothesis and associated behavioural therapies for BD (IPSRT and its variants) (Crowe et al., 2020; Grandin et al., 2006) propose that circadian function can be strengthened by increasing the regularity of daily behaviours and regularising the timing of light exposure. Similarly, pre-emptive attention to forthcoming circadian rhythm disruptors (time zone travel, unusual working hours, etc.) may minimise impacts on circadian pathways to mood symptoms (Inder et al., 2015). Consequently, treatment guidelines for BD encourage daily routine for people with BD. Some specific recommendations are contained in Box 9. Sleep and mood have a bidirectional relationship, and from a circadian health perspective, stabilisation of daily routines (as prescribed within IPSRT, for example) requires that any problems with sleep itself be addressed first. Consequently, clinicians working with mood disorders need to be attentive to sleep components in their patient’s presentation and where necessary elevate sleep problems as targets of clinical attention in their own right (Fang et al., 2019; Morton and Murray, 2020b; Wang et al., 2019).
Self-management strategies for increasing daily routine.
A range of evidence suggests that more regular daily routines (‘social rhythms’ in the literature) may benefit the mood and sleep of people with mood disorders. Clinicians can assist people to appreciate the importance of routine and proactively address routine-disrupting life events.
The patient should be encouraged to
Set a regular wake time and sleep time (within a 1-hour window across 7 days of the week): there is evidence that regular wake time may be the most important strategy for increasing mood-relevant daily routine Seek exposure to morning bright light Avoid naps during daylight hours (especially later in the day) Ensure stability of key social events, for example, daily activities and meals Plan sleep, activity and medication timing when travelling across time zones, and other foreseeable lifestyle disruptions (e.g. perinatal period) In the evening, follow sleep hygiene strategies Aim to sleep at the correct circadian time for the individual
Hypnotic medications should be used judiciously to address short-term sleep problems only
Information for clinicians about strengthening social rhythms is contained in the free online training for Interpersonal and Social Rhythm Therapy (IPSRT) www.ipsrt.org/training
Circadian assessment in clinical practice
Circadian parameters cannot be directly assessed in everyday life, but three downstream behavioural variables reciprocally related to endogenous circadian function (chronotype, social rhythm stability, and the 24-hour activity/rest cycle) can be readily assessed as part of characterising and potentially strengthening circadian function (see Table 5; Morton and Murray, 2020a).
Clinical assessment of circadian function.
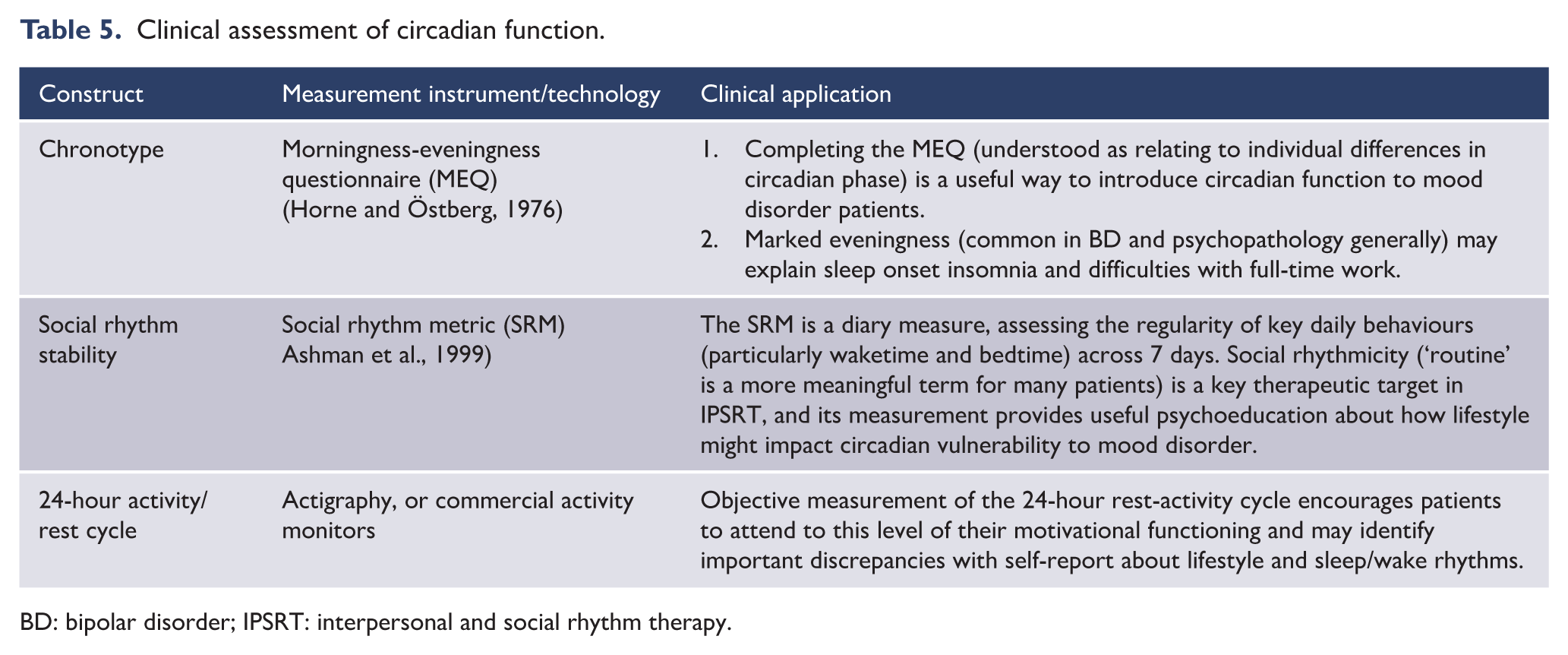
BD: bipolar disorder; IPSRT: interpersonal and social rhythm therapy.
The general clinical principles underpinning these assessments are twofold. First, their measurement provides relevant psychoeducation about the circadian motivational aspect of the individual. Second, assessment of chronotype, social rhythmicity and 24-hour activity/rest cycle provides data to inform behavioural recommendations about optimal lifestyle from a circadian viewpoint (namely, maximised activity in the daylight hours, maximised disengagement at night, qualified by individual differences in preferred daily timing). Anecdotally, patients find this level of analysis compelling, agreeing that stability of mood and emotion is strongly associated with the strength of their 24-hour behaviour/lifestyle rhythm.
Psychoeducation.
Psychoeducation aims to improve outcomes by ensuring the patient understands the nature of, and treatments for mood disorders, encouraging self-management and decreasing stigma. Critically, psychoeducation should adopt an adult learning approach rather than ‘teaching’: psychoeducation prioritises active learning, open discussion and valuing of the experience the learner brings to the topic (see Box 10).
Strategies for implementing psychoeducation.
Psychoeducation aims to improve outcomes by ensuring the patient understands the nature of, and treatments for mood disorders, encouraging self-management and decreasing stigma.
For good quality psychoeducation about mood disorders, patients should be directed to websites 1–4, below.
Psychoeducation can be initiated by asking the patient about their diagnosis, for example, ‘What do you understand by the term depression/bipolar disorder?’ This will uncover common misunderstandings (e.g. genetic determinism), or unproductive stereotypes (e.g. personal associations with the term through observation of other family members).
The clinician should follow up by asking (at the next appointment) what information the patient found useful or surprising, and ‘Are there any barriers to applying this information in your own life?’
Lived experience perspectives are an important complement to diagnostic information emphasised in most psychoeducation resources. Lived experience stories capture a range of challenges people face in managing mood disorders, while also focusing on hope and recovery goals. Useful curated lived experience perspectives can be found online (see 5, below), in books (e.g. An Unquiet Mind: A Memoir of Moods and Madness by Kay Jamison), and via support groups.
Support groups provide powerful opportunities for learning and sharing, but come with the caution that hearing others’ stories in an unstructured environment can be challenging (see 6 and 7, below).
Websites
Social support
Strategies for implementing social support.
Social support is the provision of emotional assistance (empathy, concern, acceptance, encouragement, caring), instrumental support (financial assistance, goods and services), informational assistance (advice, suggestions, factual information) and companionship (acceptance into a social/cultural group and environment, with identity and recognition). Social support should not be addressed only as part of an initial assessment: the patient’s progress in accessing social support should be reviewed over time and evaluated for value and comprehensiveness.
Clinicians can assist patients with information about services, advice regarding strategies to cope with social needs, referral to appropriate services and enquiry into available resources.
The nature of the social support services and opportunities will vary throughout regions and treatment contexts. They will include government organisations and services, social groups (self-help organisations, community groups), religious organisations and private business services (private hospitals, private health practitioners).
Allied health professionals (social workers, occupational therapists, physiotherapists) are well-placed to provide local information, as are government departments responsible for social security and health care.
Patients can be encouraged to improve their social networks with some simple tips (see 1, below).
Organisations focussing on support and education for people with mood disorders can be particularly helpful (see 2–4, below).
Websites
Psychological treatments
Differentiating psychological treatments and understanding mechanisms
Research into psychological treatments has primarily investigated treatment outcomes; psychotherapy process research is much less advanced, and the mechanisms by which psychological treatments work is poorly understood (Harmer et al., 2017). Thus, a central question remains: do therapies work through specific techniques associated with the individual foci of treatments (e.g. changing cognitions in CBT), or by ‘common factors’ shared across the various treatment approaches (Cuijpers et al., 2019b)? There is consensus that these common factors (collaborative therapeutic alliance, empathy, collecting patient feedback, etc.) (Parikh et al., 2016) are necessary to therapeutic outcomes (Norcross, 2011). The unanswered question is whether such relationship-based factors are also sufficient for change (see Mulder et al., 2017).
Psychological treatments for acute MDD
Evidence
Evidence for the effectiveness of psychological treatments as outpatient monotherapy has expanded (Cuijpers, 2017). Six therapies (CBT, IPT, Problem-solving therapy, Behavioural activation therapy, Nondirective supportive therapy and Short-term psychodynamic therapy) have now been tested in at least 10 RCTs and shown to be more effective than wait-list control. Importantly, beneficial effects have been shown to be maintained for 6 and 12 months. A number of meta-analyses and network analyses have concluded that in the outpatient treatment of major depression, there are no significant differences in the benefits derived from antidepressant therapy in comparison to psychological therapy (Cuijpers, 2017). Recent findings demonstrating effectiveness of psychological treatment in real-world practice have strengthened confidence in the value of such therapies for major depression (see, for example, Connolly Gibbons et al., 2016; Lorenzo-Luaces et al., 2018b; Saloheimo et al., 2016).
Evidence and methodological considerations for psychological treatment as monotherapy for acute MDD.
Evidence for the efficacy of psychological treatment is derived from studies in which the intervention is delivered by trained therapists, under supervision, in a manualised form with high fidelity to a particular treatment brand. The evidence base therefore does not generalise to eclectic selection of elements from existing evidence-based treatments.
CBT is by far the most commonly employed intervention and the most widely tested.
Therapies with very different assumptions have indistinguishable effects, and the perennial ‘common versus specific factors’ debate remains open (Mulder et al., 2017).
Clinical trials of psychological interventions have significant limitations, including the impossibility of patient blinding and the lack of long-term follow-up (see MDcpg2015)
Demonstrated effect sizes depend partly on the chosen control group (waiting list is probably a ‘nocebo’, generating the largest effect sizes).
Publication bias has overestimated treatment effects, just as it has for antidepressant medication (Driessen et al., 2015b).
MDD: major depressive disorder; CBT: cognitive behavioural therapy.
Delivery of psychological treatments
Three quarters of patients seeking treatment for depression prefer psychological treatment over medication (McHugh et al., 2013; Raue et al., 2009). Gender is a factor in the preference for psychological over medication treatment – the tendency is significantly more marked in female patients (McHugh et al., 2013), and males’ (relative) reticence to seek psychological assistance for depression is a growing focus of research into the problem of male help seeking (e.g. Seidler et al., 2016).
There is strong clinical consensus that manual-informed treatment (based on one of the evidence-based psychological interventions in which the therapist is trained, and supported by appropriate ongoing peer supervision) is superior to eclectic practice (see Petrik et al., 2013). However, manual-informed practice should not be understood as slavish mechanical application, or a ‘one size fits all approach’. Rather, treatment manuals for the evidence-based psychological interventions support ‘flexibility within fidelity’ by providing guidelines, suggestions and strategies to be tailored to the individual client (see Lilienfeld et al., 2013).
In most jurisdictions, quality control of the delivery of evidence-based psychotherapy is indirect, being via the professional ethics of the clinician (including the responsibility to only work in areas in which the clinician has appropriate training and expertise), in the context of centrally regulated training programs and ongoing professional development. In Australia, an additional layer of responsibility (namely, to only deliver evidence-based treatments) applies to psychologists providing services under Medicare. In some instances, divergence from manualised depression treatment may reflect best-practice case-formulation-based treatment (The British Psychological Society, 2011); however, alternatively it may reflect a suboptimal occasion of care (e.g. a purposeless shift to unstructured or eclectic psychotherapy).
Evidence for psychodynamic treatments is summarised in Box 13 (Leichsenrin et al., 2015). Short-term psychodynamic therapy appears to be effective in the treatment of depression, both as monotherapy (Driessen et al., 2015a, 2017) and as an adjunct to antidepressant medication (Driessen et al., 2020) and recent findings await robust replication. However, it is important to note that not all depressive presentations benefit from this therapeutic approach (Town et al., 2017) (see Box 14).
Psychodynamic treatments and evidence-based clinical practice.
It is important to note some polarisation between proponents of psychodynamic (particularly long-term and psychoanalytic variants) versus structured intervention approaches like CBT (see, for example, Hofmann, 2016; Leichsenring et al., 2015). Proponents of psychodynamic therapies highlight their particular potential for treating depression in patients with marked attachment, trauma or personality features (see ‘Personality disorders’ within section 11.1 ‘Complex presentations’). Detractors warn about the lack of standardisation (particularly in long-term variants). Both camps would agree that psychodynamic approaches elevate therapist insights and the therapeutic relationship over the more concrete therapeutic strategies that characterise the behaviourally based therapies like CBT: for proponents, this is an appropriate and powerful prioritisation of psychotherapy process underpinning long-term change; for detractors, this constitutes a missed opportunity for skill-development, and a risk factor for unaccountable practice.
Psychodynamic theory has been influential in illuminating the relationship and alliance-based elements of psychological interventions, and of course is visible in the logic of CBT (particularly its schema-focused variants). Psychodynamic therapies promote regression, which can be distressing for some patients, and even generate transitory deterioration in mental state. Similarly, the development of insight is very important in this form of therapy, and some patients struggle to apply this new knowledge.
Debate continues, in part because of our inadequate understanding of mediators (see above), and moderators of psychological treatments generally (Beutler et al., 2016; Driessen et al., 2016). Reviews in the area are subject to allegiance effects (Fonagy, 2015), and some disagreements probably rest on paradigmatic differences about personality and behaviour change (Mulder et al., 2017). We encourage clinicians to remain skeptical about the evidence base generally and open to emerging data about the processes and outcomes of psychological interventions (e.g. Blease et al., 2016).
CBT: cognitive behavioural therapy.
Psychological treatments for acute depression.
MDcpg2015 described CBT and IPT as the primary recommended approaches for the treatment of acute depression. In MDcpg2020, we note that in addition to CBT and IPT, four further approaches now have replicated evidence for efficacy and (to some extent) real-world effectiveness for acute depression. These four are presented here for comprehensiveness, but CBT and IPT remain the primary recommended approaches because they have been subjected to more investigation across sites, are more commonly taught in training programs and are familiar to current practitioner networks.
Problem-solving therapy comes in various forms, but is characterised by articulating personal problems, generating multiple solutions for each, selecting strategies and a systematic plan of action, and evaluating the solution. Problem-solving therapy encourages skill development and empowerment, so is antithetical to the clinician attempting to solve patient problems or advising them on how to act (Cuijpers et al., 2018; Pierce, 2012).
Behavioural activation therapy (developed originally by Lewinsohn et al. (1979) involves activity scheduling aimed at increasing pleasant activities and positive interactions (social skills training is present in some variants). Behavioural activation (encouraging physical and social activity, for its physiological and motivational benefits) is a component of CBT (typically rolled out prior to addressing cognitions).
Nondirective supportive therapy is typically understood as an unstructured therapy with no specific psychological techniques. It is not aimed at solutions or acquiring new skills, but instead focuses on opportunities to ventilate and receive empathy. In the literature, this approach is sometimes referred to as counselling.
Short-term psychodynamic therapy aims to improve the person’s understanding of repetitive conflicts by exploring them from the viewpoint of childhood experiences, and past conflicts. In contrast to long-term psychodynamic therapy, short-term psychodynamic therapy is structured and time limited (maximum of 25-30 sessions delivered weekly) and prioritises improved adjustment to present day challenges. There is no evidence to support open-ended or long-term psychodynamic therapy.
CBT: cognitive behavioural therapy; IPT: interpersonal therapy.
Combined psychological and pharmacological treatments for acute major depression
Replicated analyses now support the combination of psychological treatment and pharmacotherapy as the most effective therapeutic strategy for acute major depression, all else being equal (see Box 15). In brief, meta-analytic studies have shown that combination treatment is more effective than pharmacology alone, psychological treatment alone and the combination of psychological treatment and placebo (Cuijpers et al., 2014b; De Maat et al., 2007; Pampallona et al., 2004). Moreover, a recent trial has shown benefits of both adding psychological treatment to antidepressant medication and adding antidepressant medication to psychological treatment (CBT) in patients who did not achieve remission with either alone (Dunlop et al., 2019). This finding is important clinically because it suggests that the sequence in which the treatments are combined (CBT or antidepressant monotherapy commenced first) does not affect the extent to which the patient can benefit from combining treatments.
A Cochrane review concluded that adding psychological treatment to AD is beneficial for acute treatment, and also for medium- and long-term outcomes (as recommended in MDcpg2015), but that there is insufficient evidence to support the theoretically compelling suggestion of switching from antidepressant medication to psychological treatment (Ijaz et al., 2018). Persistent or chronic depression and non-responsive depression have been a frontier for research into psychological treatments for major depression (McPherson, 2020). In MDcpg2015, Managing Suboptimal Response was divided into poor response to psychological therapy, and poor response to pharmacological treatment. While these considerations remain useful within the ‘silo’ of either psychological or pharmacological treatment, the evidence for superiority of combined treatment suggests that a primary option for poor response is the addition of the other modality (poor response to psychological treatment, consider adding antidepressant medication; poor antidepressant medication response, consider adding psychological treatment).
Evidence for the combination of psychological and pharmacological therapy for the management of acute depression.
Adding psychological treatment to medication has been shown to improve outcomes over medication alone with a g = 0.43 (NNT = 4) (Cuijpers et al., 2014a). Conversely, adding medication to psychological treatment has been shown to improve outcomes over psychological treatment alone with a d = 0.35 (NNT = 5) (Cuijpers et al., 2009). In addition, Cuijpers et al. (2015) conducted a meta-analysis in which combined treatment was compared with placebo-only, with psychological treatment, with pharmacotherapy, and with the combination of psychological treatment and placebo. Moderate benefits for combined treatment were found when compared with pill placebo (q = 0.46), small to moderate against pharmacotherapy (q = 0.38), psychological treatment (q = 0.34) and psychological treatment plus placebo (g = 0.23). Karyotaki et al. (2016) reported that combined treatment outperformed antidepressants alone at 6 months or longer post-randomization and during maintenance treatment. Bashir et al. (2018) found that combined therapy was more effective than pharmacotherapy or psychological monotherapies in terms of response, remission and reduction of symptoms. Most recently, a network meta-analysis (101 studies, N = 11,910 patients with moderate-severe depression) found benefits of combined treatment relative to psychotherapy alone, and antidepressant medication alone in terms of both response and remission rates (Cuijpers et al., 2020). Findings were the same in treatment-resistant and chronic subgroups. Thus, the combination of the two types of intervention is superior to either alone (Cuijpers, 2017; Cuijpers et al., 2020)
NNT: number needed to treat.
Psychological treatments for bipolar disorder
Adjunctive psychological treatment for bipolar disorder
Outcomes in BD are improved by augmenting medication with an evidence-based psychological treatment, and adjunctive psychological intervention is a central component of a chronic disease management approach to BD (Yatham et al., 2018). Concerns still remain about the strength of the evidence base for psychological interventions (Goodwin et al., 2016; Murray, 2018; Oud et al., 2016), but best practice treatment of BD clearly involves the combination of medication and psychological treatment. As discussed in MDcpg2015, the strongest evidence is for delivery in the maintenance and depressive phases of BD, with benefits for time to remission, time to recurrence and functional outcomes (Chen et al., 2019a). Here, we introduce the four evidence-based interventions (Psychoeducation, Cognitive-behavioural therapy, Family-focused therapy, Interpersonal and social rhythm therapy) and emerging treatments.
Psychoeducation
Psychoeducation delivered in a group format appears to be one of the most effective psychotherapies for BD maintenance phase and is more economical and less demanding on therapists than more complex individual therapies (Novick and Swartz, 2019). Small ‘p’ psychoeducation (i.e. information to support self-management and engagement with professionals, as contrasted with the manualised group-delivered Psychoeducation intervention [Big ‘P’] tested in clinical trials) is considered by the British Association for Psychopharmacology to be integral to patient care (Goodwin et al., 2016). There is insufficient evidence to recommend Psychoeducation for the treatment of acute bipolar depression, but there is no reason to think that it would be detrimental.
The core content elements of psychoeducation (to be delivered to all patients) are the content elements shared across the psychological treatments, including improved ability to recognise early warning signs, knowledge about and acceptance of BD, and daily monitoring of mood and sleep (Duffy et al., 2019; Faurholt-Jepsen et al., 2019) (see Table 22 in MDcpg2015). These elements are often conceptualised as part of self-management approaches to BD (Gliddon et al., 2017; Michalak et al., 2019). Recovery-oriented, evidence-based versions of this self-management material can be found online (e.g. www.bdwellness.com) (Morton et al., 2019; Murray et al., 2011).
Cognitive-behavioural therapy (CBT)
CBT has strong support as an acute treatment for bipolar depression and as a maintenance treatment for BD (Goodwin et al., 2016; Yatham et al., 2018). A meta-analysis of 19 RCTs suggests that effect sizes are small to moderate (Chiang et al., 2017), with benefits on relapse rate, depressive symptoms, severity of mania and psychosocial functioning.
Family-focused therapy (FFT)
FFT is recognised as efficacious for both acute depressive symptoms and maintenance treatment in a number of guidelines (Murray, 2018), as it has illness burden benefits for the family in addition to the individual with BD. However, in practice its use is limited somewhat because naturally FFT requires at least one family member to participate in the treatment. Since MDcpg2015, a narrative review by the program’s developers (Miklowitz and Chung, 2016) highlights emerging research into preventative family-focused treatments for at-risk youth (Miklowitz et al., 2019).
Interpersonal and social rhythm therapy (IPSRT)
The small number of existing trials of IPSRT have generated complex findings (hence its lower recommendation level in international guidelines including the MDcpg2015), with some evidence that it may be more effective for acute depressive symptoms than for maintenance treatment.
Peer support
Peer support is premised on the notion that the lived experience of others constitutes valuable information for people with BD and is a strengths-based approach consistent with recovery principles (Proudfoot et al., 2012). Peer support specifically for BD (as contrasted with the more common transdiagnostic peer support for serious mental illnesses) has now been the focus of at least four trials. There are various manifestations, with the trend in scientific literature towards more structured variants with formalised training and delivery. Not surprisingly, its benefits may be most apparent in self-efficacy and empowerment outcomes. Recent North American guidelines (Yatham et al., 2018) consider that there is insufficient evidence to consider peer support as an evidence-based intervention for BD. A challenge in this area is the absence of consensus around the definition of ‘peer support’ or ‘intentional peer support’, and a consequent lack of standardisation of interventions and training approaches across trials. Anecdotally, there may be a risk in peer-support interventions if the information does not support adherence to pharmacotherapy or encourages substance use (Yatham et al., 2018). Proper training and support of peer workers, and combining lived experience with clinical expertise in the development of programs would appear to be the solution to this concern. In Australia, peer support is receiving growing attention through face-to-face support groups informed by clinical guidelines (e.g. Bipolar Life) and aspects of contemporary online treatment packages (e.g. peer moderated discussion boards).
Mindfulness-based interventions
Mindfulness-based interventions have been of growing interest because of their popularity in the community generally, and their appeal to people with BD as consistent with more person-focused interventions for this chronic condition (Murray et al., 2017). Three reviews have recently been published on the topic. One meta-analysis in patients with BD found benefits of mindfulness-based therapies for depressive and anxiety symptoms over baseline (in nine uncontrolled studies, N = 142), but no relative benefits over various control conditions in three studies (Chu et al., 2018). Lovas and Schuman-Olivier (2018) identified 13 reports of Mindfulness-Based Cognitive Therapy (MBCT), but heterogeneity across studies and small samples precluded a meta-analysis. Their narrative review concluded that there was preliminary evidence of benefits for anxiety, residual depression, mood regulation, and broad attentional and frontal-executive control. Finally, Bojic and Becerra (2017) concluded that MBCT is a promising treatment, based on 13 studies (N = 429) that found evidence of benefits for cognitive functioning, emotional regulation, and symptoms of anxiety, depression and mania. However, the lack of adequately powered randomised trials means that such interventions cannot as yet be recommended. Further research is warranted especially given growing evidence that earlier concerns about meditation precipitating mania can be addressed (Fletcher et al., 2018a; Lovas and Schuman-Olivier, 2018).
Cognitive remediation
Cognitive remediation refers to a set of behavioural interventions targeting cognitive difficulties with the aim of improving functional outcomes. Cognitive remediation has typically been provided in a structured group format, teaching skills and problem-solving strategies to compensate for cognition-related challenges, but other modalities including digitally delivered interventions are under development (Sole et al., 2017). Functional remediation refers to a subset of this approach that addresses neurocognitive deficits (particularly attention, memory and executive functioning deficits associated with BD) (Vieta and Torrent, 2016) while providing psychoeducation about minimising the impact of cognitive deficits in daily life. Functional remediation has shown evidence of preliminary benefit in two trials (Demant et al., 2015; Torrent et al., 2013). An earlier review (Anaya et al., 2012) found preliminary evidence of moderate effect size improvements in cognitive performance in an aggregate sample of bipolar disorder and unipolar disorder subjects. And most recently, a systematic review of 11 studies (3 RCTs, n = 354) concluded that there was some support for cognitive and functional improvements.
Digital therapies
A significant development in the treatment of mood disorders over the past 5 years has been the worldwide expansion of research into digital interventions. The accessibility and cost advantages of digital interventions for mental health have led to large-scale digital investments by Australian and UK governments among others (Clark et al., 2018). Public recognition and apparent acceptance of evidence-based online treatments, particularly since the COVID-19 pandemic, has dramatically improved access to evidence-based psychological treatments, enabling the MDcpg2020 prioritisation of psychological treatments as foundational in mood disorder management (Figure 18).
This expansion has been so rapid and pervasive that terminology is struggling to keep up, and the literature suffers from a variety of characterisations and aggregations of hardware (e.g. smartphones, computers, tablets), software (apps, websites, video conferencing) types of intervention (monitoring, education, information, social networking, psychological treatment, self-help) and connectivity (telephone, local delivery, internet delivery) (Andersson et al., 2019).
A further challenge of the rapid expansion of digital interventions is that many practicing clinicians were trained at a time when these interventions did not exist. Consequently, applied knowledge of how to direct, support and monitor patients progressing through online offerings is not widespread. As noted in the RANZCP position statement on e-mental health, it is critical that clinicians upskill on this fast-moving space. The Australian Government’s eMHPrac (e-mental health in practice, see www.emhprac.org.au) is designed to address this need, and the Australian Commission for Safety and Quality in Health Care is currently developing national standards for digital mental health services (see www.safetyandquality.gov.au/standards/national-safety-and-quality-digital-mental-health-standard).
It is timely to acknowledge increased use of (and government funding for) treatment delivered by telephone or videoconferencing (telehealth) during the period of social isolation associated with COVID-19. It is likely that this trend will continue because telehealth can partly ameliorate regional disparities in access. Telehealth also has convenience benefits for patients and may equalise the power imbalance inherent in patients’ travelling to attend physical clinics (see for discussion, Topol, 2015). On the other hand, the telehealth modality is still emerging: Digitally mediated and in-person consultations clearly have contrasting strengths, and new hybrid models of care will soon be subjected to rigorous tests (Smith et al., 2020). In-person consultations would appear to be particularly beneficial, for example, during the initial assessment phase and turning points in illness trajectory.
Digital therapies for major depressive disorder
In the treatment of major depression, online therapies (when delivered with some human support, such as emails with a coach) can be as effective as face-to-face management and are highly acceptable to patients (Apolinário-Hagen et al., 2018) (see Table 6). As with face-to-face psychological interventions, the majority of work in digital treatment has focused on Internet-delivered CBT (iCBT) and its variants.
Online clinics and supporting information for clinicians.
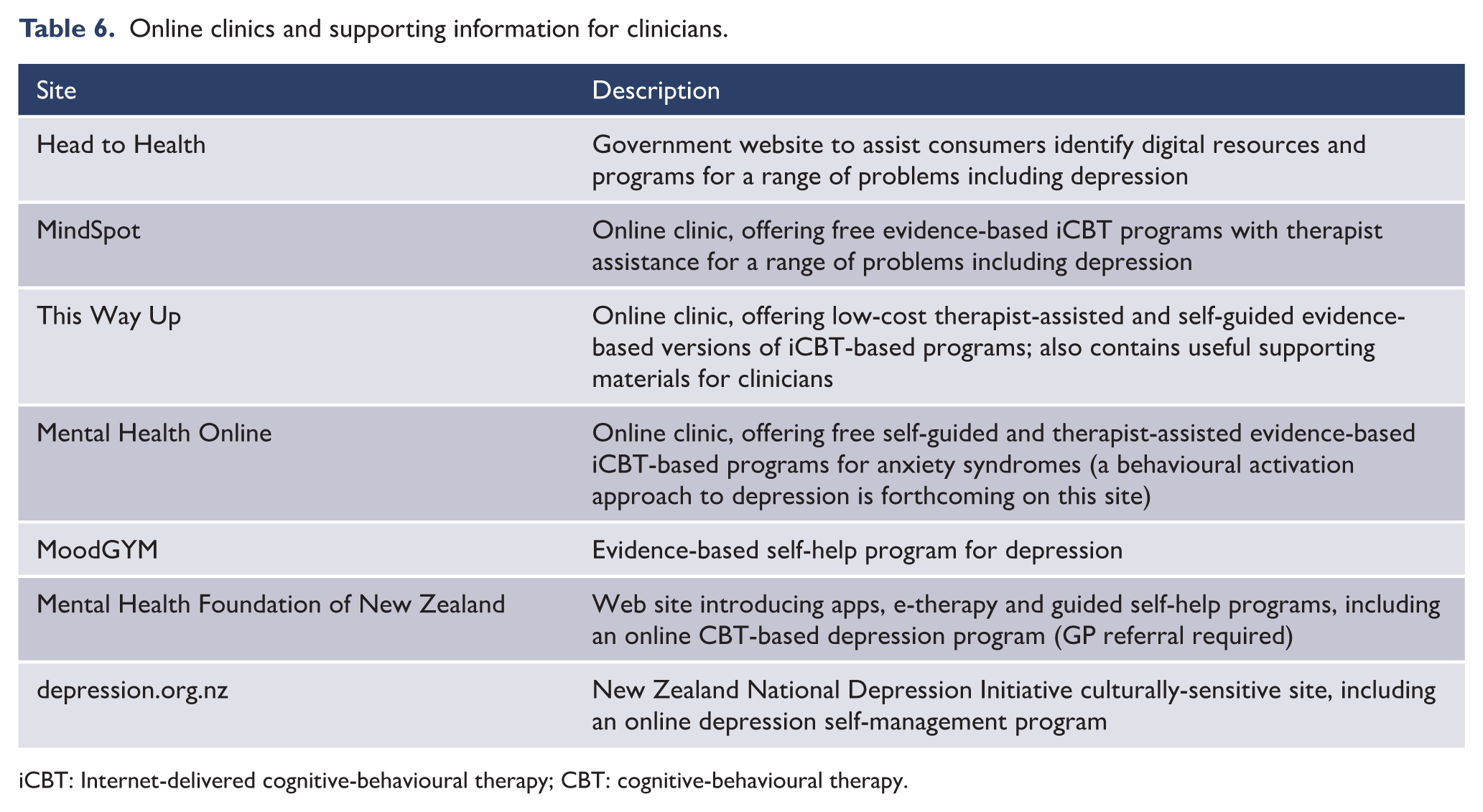
iCBT: Internet-delivered cognitive-behavioural therapy; CBT: cognitive-behavioural therapy.
A range of evidence suggests that iCBT is as effective as the original face-to-face modality in the treatment of acute MDD. A direct meta-analytic test of the therapeutic equivalence question (Carlbring et al., 2018) found no efficacy difference between Internet-delivered and face-to-face CBT across 20 trials (across all disorders, but specific to depression symptoms in four reviewed studies). Similarly, a network meta-analysis (155 trials, N > 15,000 patients) concluded that delivery format does not moderate the outcomes of CBT for acute depression (Cuijpers et al., 2019a). Cuijpers et al. also highlighted that self-help therapy (whether delivered through the Internet or other modalities) was only superior to care as usual when provided in a guided format (i.e. with some human coaching or support).
The same conclusion has been drawn by separate systematic reviews (Ahern et al., 2018; Andrews et al., 2018; Josephine et al., 2017). The strength of support for iCBT is bolstered further by evidence that trials included in iCBT meta-analyses include severe and complex presentations of depression (see also Bower et al., 2013; Lorenzo-Luaces et al., 2018a) and that effect sizes from controlled trials generalise to everyday practice (Flygare et al., 2020; Staples et al., 2019).
The role of digital interventions in the management of mood disorders
As an evidence-based therapy for major depression that may be as effective as face-to-face psychological treatment for a range of severities, Table 7 outlines some factors for clinicians considering commencing digital interventions for a particular case. Table 8 highlights that digital and face-to-face modalities have different strengths, but on balance, face-to-face consultations are recommended for the initial assessment phase to ensure thorough diagnosis and tailored case formulation.
Summary of digital interventions for major depression.
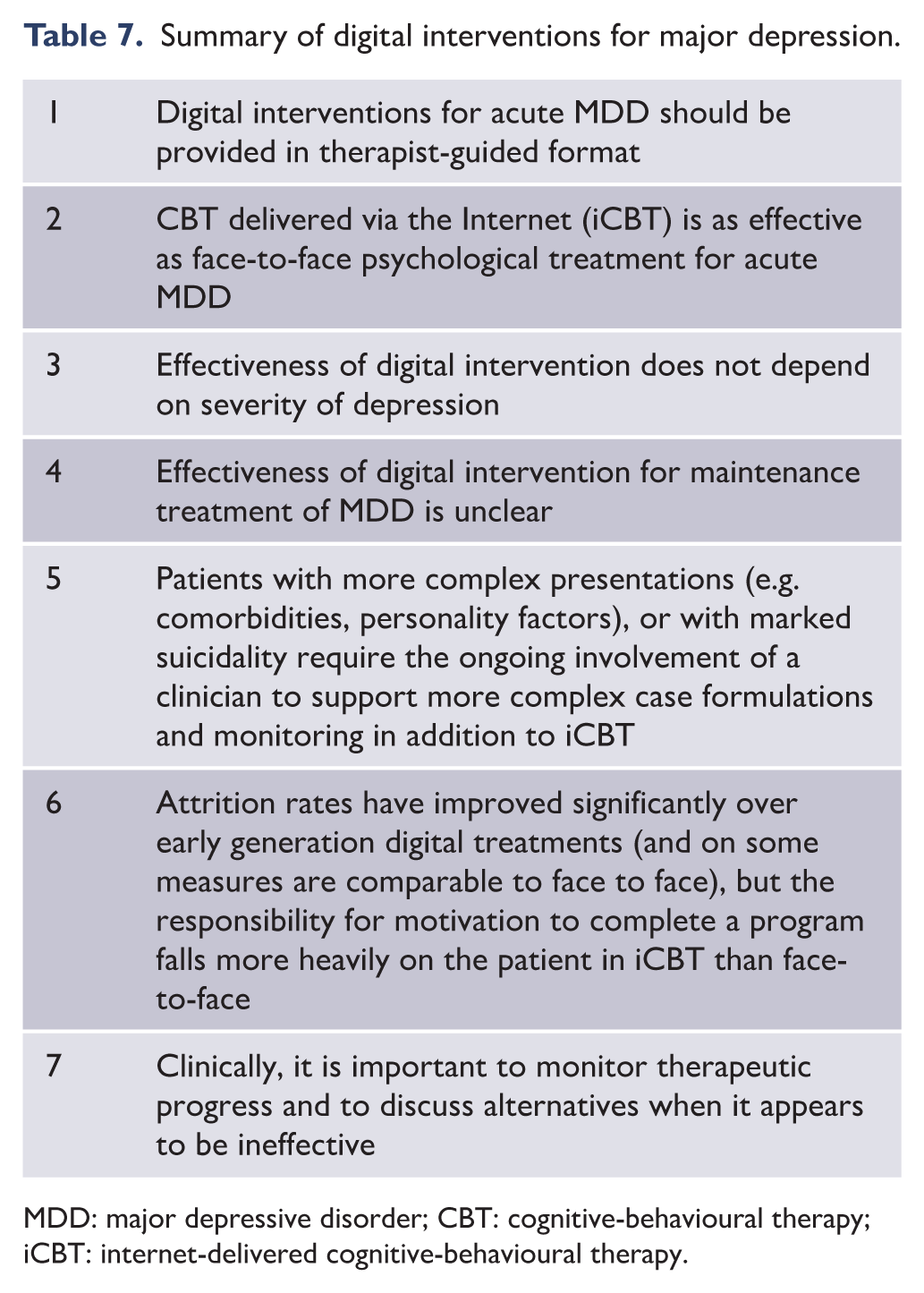
MDD: major depressive disorder; CBT: cognitive-behavioural therapy; iCBT: internet-delivered cognitive-behavioural therapy.
Strengths and weaknesses of digital, relative to face-to-face intervention.
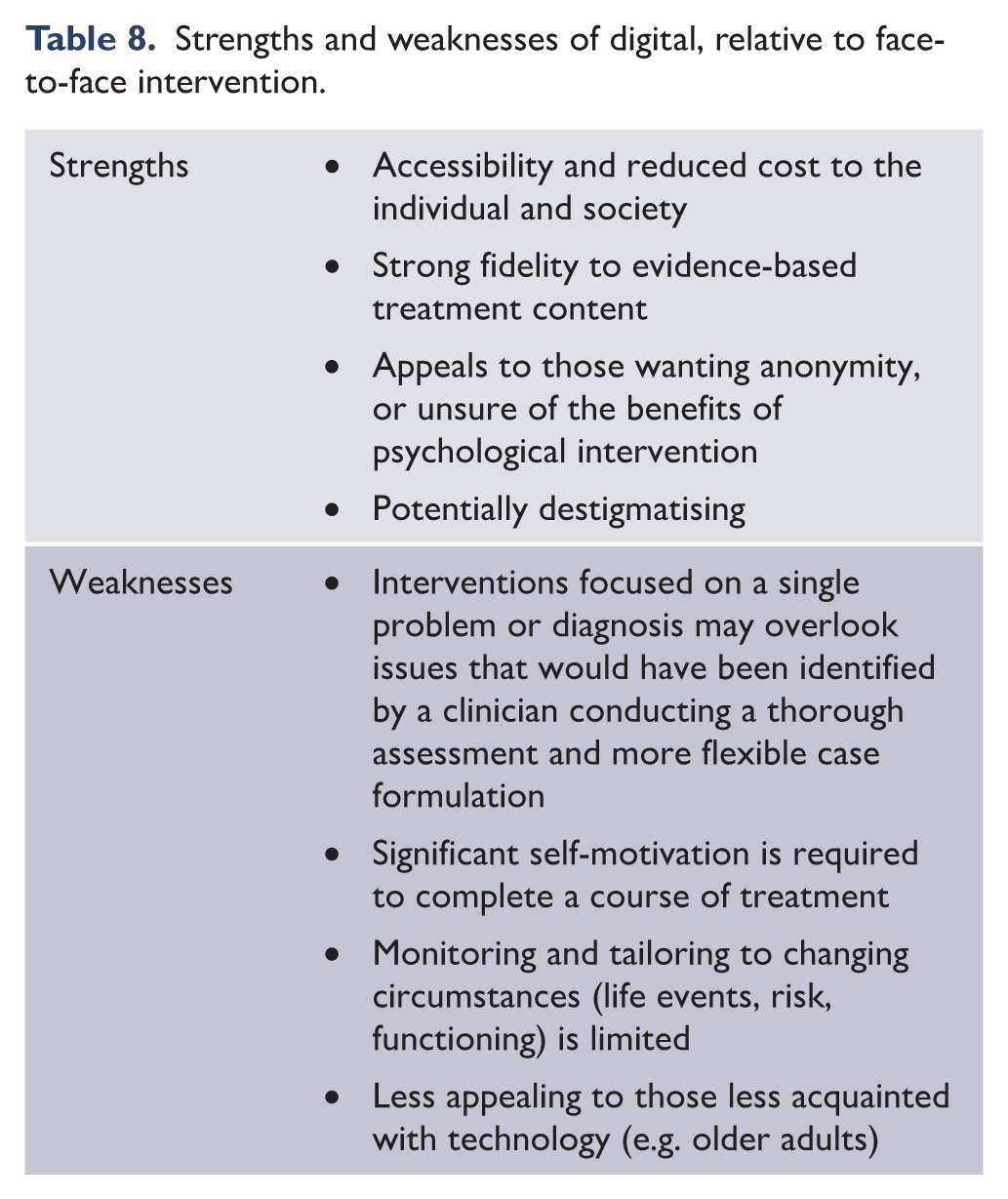
Recent bipolar guidelines (Yatham et al., 2018) highlight the paucity of data on digital therapies for BD, a point reiterated by a recent review (Dean et al., 2018). One large RCT has found evidence for small-moderate benefits on depression (but not mania) in one of two active arms (that included Psychoeducation and CBT) over control (a peer support forum) (Gliddon et al., 2019). Further research is forthcoming (e.g. Fletcher et al., 2018b), and enthusiasm for digital intervention in BD remains high, because of its acceptability, feasibility and apparent safety even in high risk mood disorder populations (Fletcher et al., 2018a; Leitan et al., 2015), and the fact that world-wide less than half of people on the BD spectrum receive any treatment (Merikangas et al., 2011). The chronobiological interventions described above, in particular, would benefit from digital dissemination because they rely on biobehavioural models that are not widely taught to clinicians. While digital versions of the psychological treatments are yet to be tested, good quality psychoeducation for BD is available online (e.g. www.bdwellness.com; www.cci.health.wa.gov.au; www.dbsalliance.org).
A key next step in digital mental health will be development of sophisticated online assessment portals (utilising artificial intelligence to streamline user experience and refine behavioural phenotypes) which would support treatment seeking among those unlikely to seek face-to-face consultation. Research is also beginning to emerge into hybrid psychological treatments that combine the didactic strengths of online delivery (structured, engaging delivery of specific content) with the process strengths of face-to-face consultations (flexible, supportive, embodied engagement) (Erbe et al., 2017).
6.2. Choices
Pharmacotherapy
The pharmacotherapy of mood disorders involves the administration of medications that have diverse molecular structures but overlapping mechanisms of action. Attempts have been made to introduce a more standardised nomenclature, either on the basis of specific pharmacological properties such as receptor binding profiles (Zohar et al., 2015), or on the basis of principal clinical usage; these are yet to be widely adopted. Hence, the MDcpg2020 adopts a pragmatic approach and considers the evidence concerning the actions of new medications, or new evidence of established medications under three broad headings: antidepressants, mood-stabilising agents (MSAs) and second-generation antipsychotics (SGAs, with a range of actions on mood disorders). These groups are not mutually exclusive but instead reflect classes that are clinically recognised (Table 10).
Pharmacological treatment based on clinical profile.
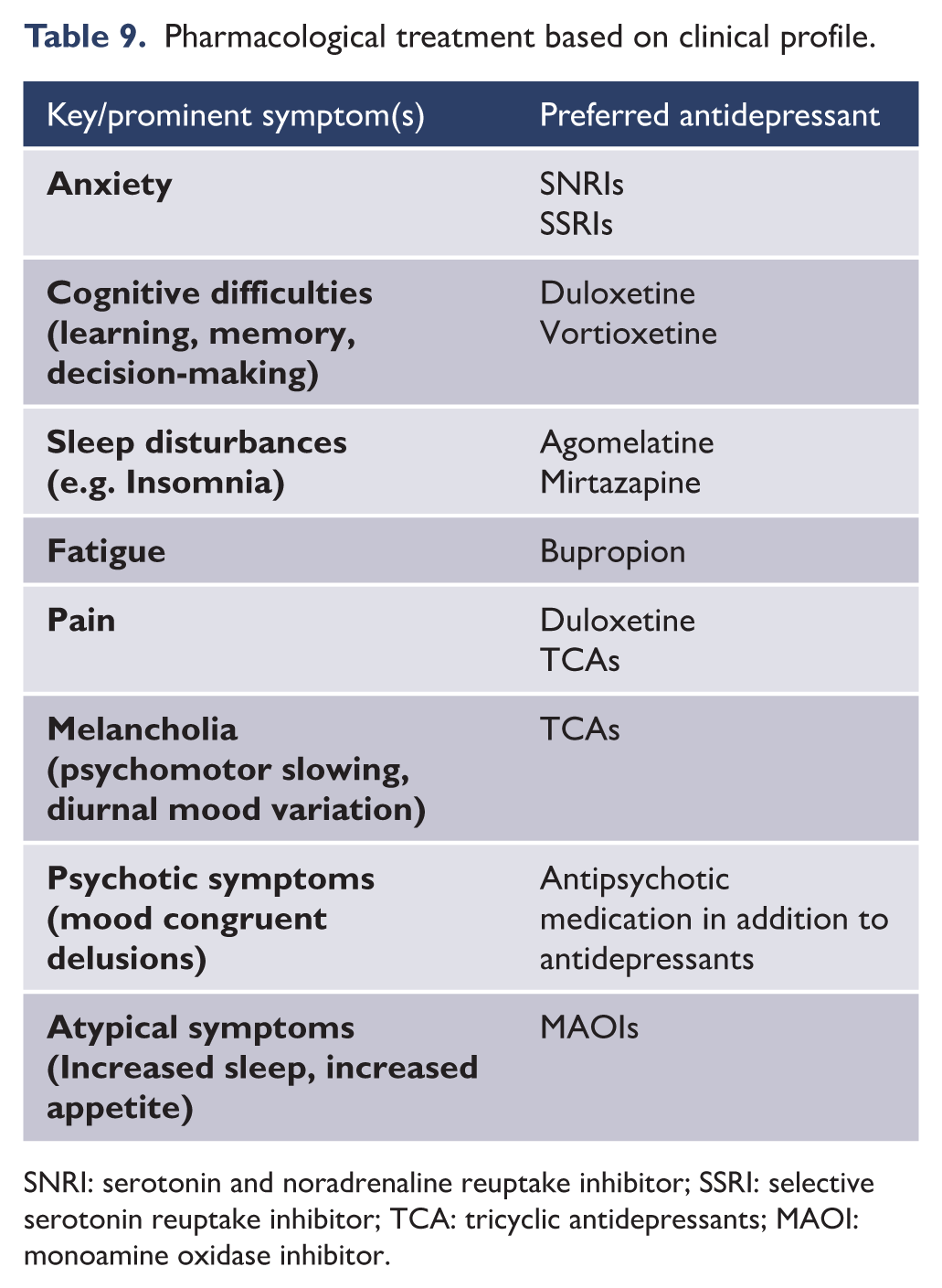
SNRI: serotonin and noradrenaline reuptake inhibitor; SSRI: selective serotonin reuptake inhibitor; TCA: tricyclic antidepressants; MAOI: monoamine oxidase inhibitor.
Antidepressants
Based on their mechanism of action, antidepressants can be divided into more than a dozen different classes (see Table 10). Most antidepressants interact with membrane-bound receptors, and/or reuptake transporters serving monoaminergic neurotransmission, that is, they modulate serotonin, noradrenaline and dopamine. However, a few classes of antidepressants also involve different mechanisms of action, for example, the inhibition of monoamine oxidase, interaction with melatonergic transmission and NMDA glutamatergic receptor blockade. In recent years, relatively few new antidepressants have become available clinically, though a number are in development (see ‘Rapidly acting antidepressants’ within section 6.2 ‘Choices’).
Classes of antidepressants.
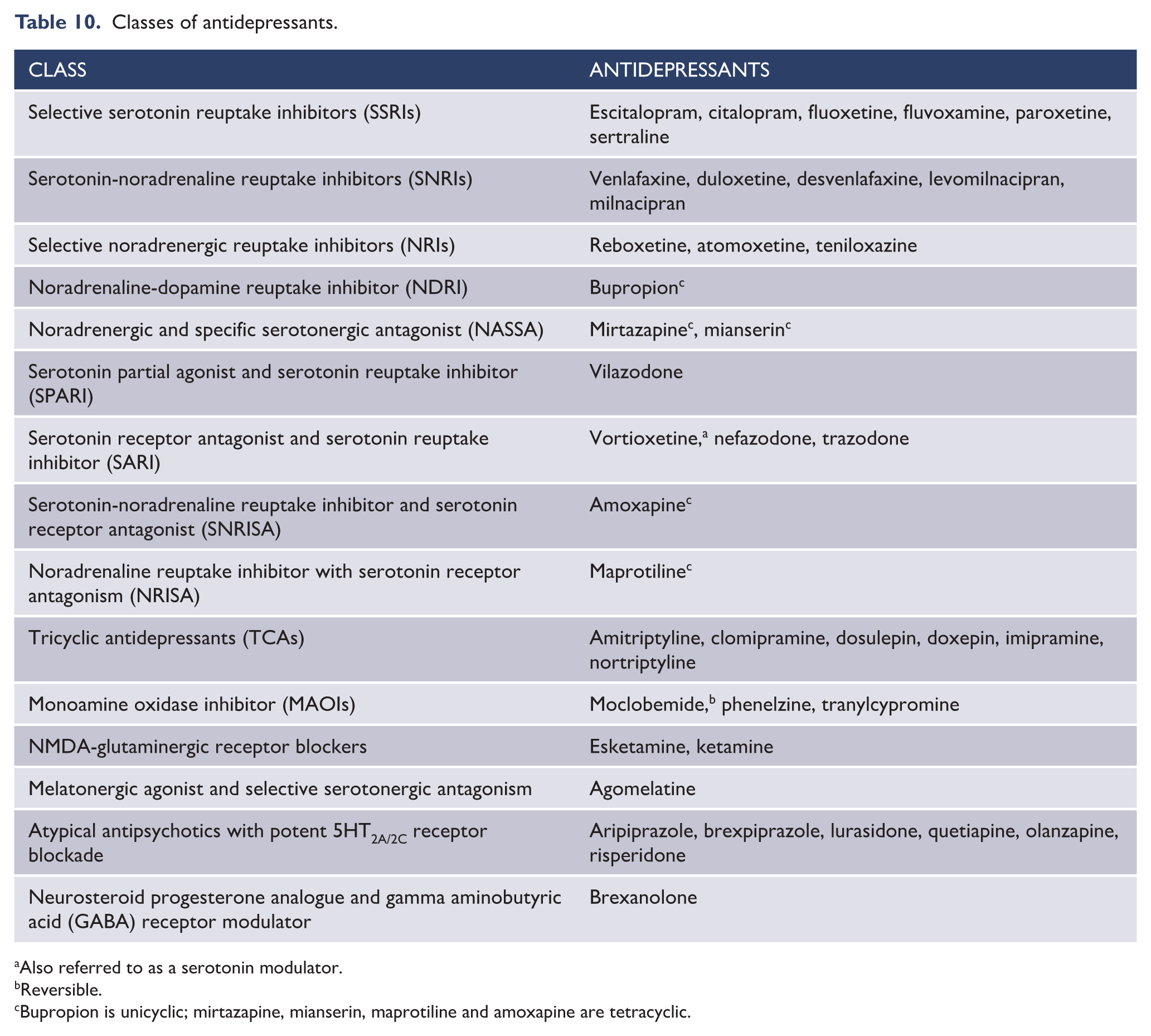
Also referred to as a serotonin modulator.
Reversible.
Bupropion is unicyclic; mirtazapine, mianserin, maprotiline and amoxapine are tetracyclic.
The side effects of antidepressants determine treatment choice, as much as their efficacy, and like their therapeutic actions are determined by their receptor interactions (see Figure 22). However, clinically, it is useful to consider the principal concerns expressed by patients, namely, sexual dysfunction, weight gain and sedation; these are shown schematically (Figure 23) alongside the antidepressants that are least likely to cause these side effects. It is important to note that these are only indications of likely tolerability and that in individual cases, antidepressants may cause these particular side effects, and many others. Therefore, asking the patients about side effects, and maintaining vigilance and close monitoring is essential throughout management but especially when initiating medication or modifying dosage (see Figure 24).
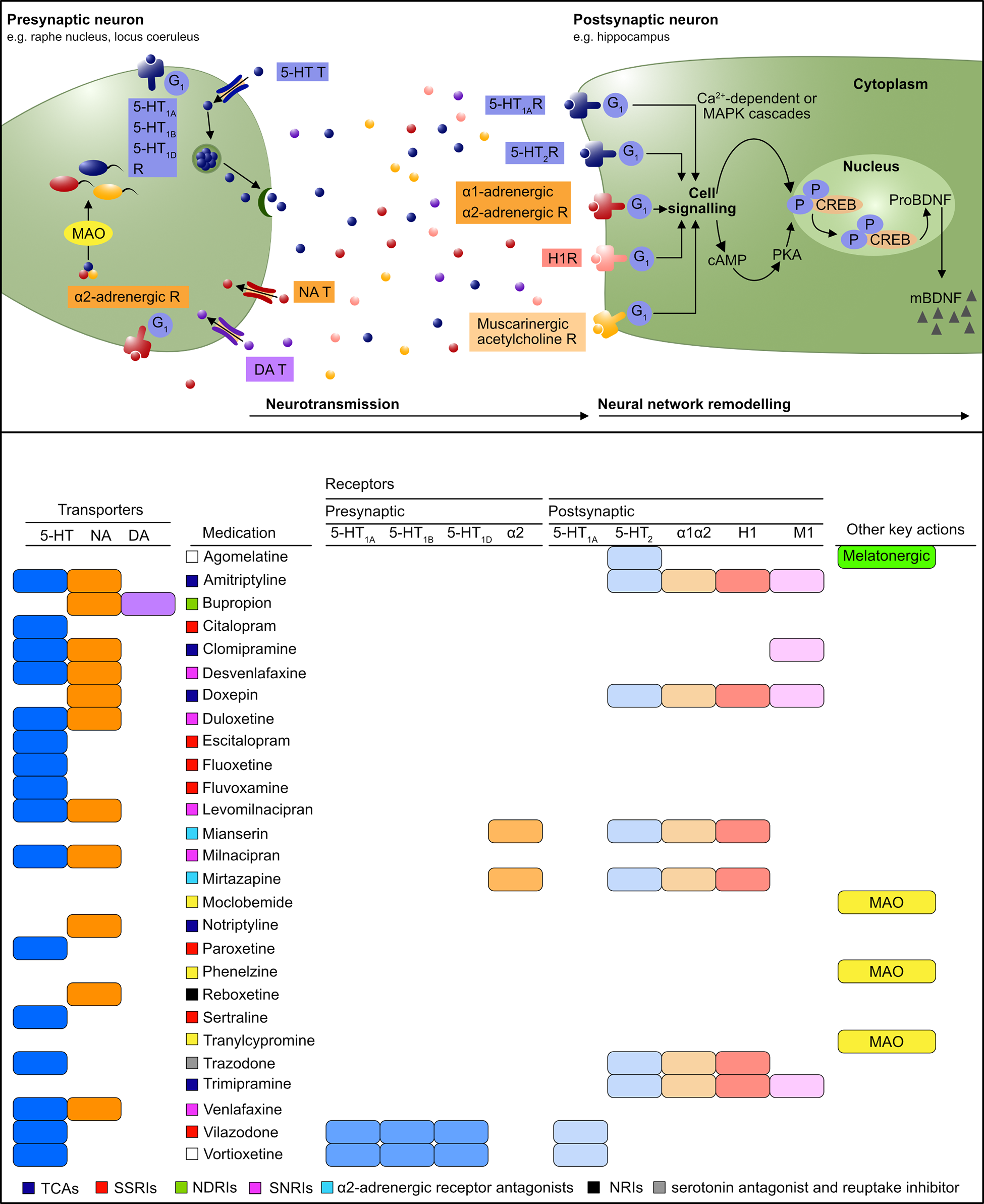
Antidepressant mechanisms (adapted from Malhi and Mann, 2018).
Antidepressant side effects.
Properties of antidepressants.
Antidepressants are not recommended as monotherapy for acute bipolar depression and are not a first-choice medication; they should only be prescribed in conjunction with MSAs or SGAs. Furthermore, the efficacy of antidepressants in this context is supported by only limited evidence. However, empirically, there is sufficient evidence of benefit to warrant their inclusion as an Alternative – hence their listing as part of double or triple therapy (see section 8.2 ‘Bipolar depression’) (Gijsman et al., 2004; Tohen et al., 2003; Vazquez et al., 2011).
Treatment of cognitive difficulties
When cognitive difficulties are identified, the clinical situation needs to be carefully assessed, recognising that the relationship between mood symptoms and cognitive function may be bi-directional. First, residual mood symptoms should be assessed and addressed if possible. Second, determine whether the antidepressant medications are contributing to cognitive impairment and, if necessary, they should be adjusted. For example, reducing the dose of, or switching from, tricyclic antidepressants, reviewing the dose and levels of lithium or reviewing the dose of antipsychotic medication. The issue of co-morbid substance use or medical comorbidity should also be reviewed.
The elderly are more likely to suffer from cognitive side effects of treatment. SSRIs are generally well tolerated and do not appear to have cognitive side effects. Paroxetine has a strong evidence base in old age depression (Reynolds et al., 2006) but is more anticholinergic than other SSRIs and theoretically more likely to cause cognitive difficulties.
Should patients continue to have cognitive difficulties despite optimising pharmacotherapy, then referral for a more detailed neuropsychological assessment is indicated. This may then provide information regarding the domains of cognition that are affected, and help to differentiate between dementia and the effects of a mood disorder, and guide future treatment.
There are currently no well-established cognitive enhancers in mood disorders. There is some positive data regarding erythropoietin in bipolar disorder (Miskowiak et al., 2014) but this is not generally available. There are data suggesting that vortioxetine may be helpful for some aspects of cognitive function in major depression, but the effects are relatively narrow and are related to the treatment of acute depression (McIntyre et al., 2016). Duloxetine, an SNRI, has been shown to have beneficial effects on working memory in elderly depressed patients (Katona et al., 2012), and one trial of modafinil in remitted major depression has been shown to have weak benefit (Kaser et al., 2017).
Given the overlap between cognitive impairment in old age depression and dementia, anti-dementia drugs have been trialled. However, while earlier reports were encouraging, the largest clinical trial suggests an increased risk of relapse when donepezil is added to antidepressants in depressed elderly patients with cognitive impairment (Reynolds et al., 2011). If, however, it becomes apparent that the patient is suffering from an Alzheimer’s or Lewy body type dementia, then the use of cholinesterase inhibitors may be indicated.
A new area of interest is that of cognitive remediation in mood disorders. Functional remediation which includes a cognitive focus has been shown to improve function in euthymic bipolar disorders (Torrent et al., 2013). Interventions with a focus on cognitive training (consisting of computerised tasks and strategy coaching) have shown promise in small randomised controlled trials (Porter et al., 2013) but definitive trials are awaited. More specific treatments include problem-solving therapy for depressed elderly patients with executive dysfunction (Alexopoulos et al., 2011); compared with a comparison treatment, this significantly improved overall functioning over 12 months, but did not improve cognitive function. Preliminary studies of cognitive rehabilitation in elderly patients who have recovered from depression have produced encouraging results (Morimoto et al., 2012).
Treatment response paradigm
The emergence of newer agents has reignited discussion concerning antidepressant response and, in particular, the delay in response that occurs with conventional antidepressant medications. A new paradigm that has been recently postulated to address this gap is briefly outlined (Malhi et al., 2020e).
In practice, there is a substantial delay, usually a matter of weeks, following the commencement of an antidepressant before there is any significant improvement in depressive symptoms. This delay is a problem for several reasons. First, there is the risk that treatment may be stopped as the individual feels it is not of benefit. Second, if the treatment is not effective, then the underlying illness continues to cause impairment and is associated with ongoing risks such as the possibility of self-harm or suicide.
A pragmatic approach to speed up the action of antidepressants, or bridge the delay in antidepressant response, is to prescribe additional agents thought to accelerate the process of recovery. It is important to note, however, that this is theoretical and that there is little evidence both to support and indeed inform such strategies, which may in some instances cause severe adverse effects.
Drawing on collective clinical experience, we have outlined an approach that may be useful for framing considerations in relation to this delay in antidepressant effect. This Windows of Antidepressant Response Paradigm (WARP) has been illustrated in Figure 25 and is explained in more detail in the accompanying legend.
Windows of antidepressant response paradigm (WARP) (adapted from Malhi et al., 2020c).
An additional reason for including this paradigm is the development of agents that may have potential to fulfil such functions. It is important to note however, that as yet, there is no specific evidence to support specific medications and that this guidance has been provided primarily to inform and raise awareness. Specifically, it does not extend to making any particular recommendations regarding particular agents. Nevertheless, in real-world practice, doctors often utilise medications such as those with anxiolytic or sedating properties (e.g. second-generation antipsychotics, benzodiazepines) to achieve prompt relief from some of the more troubling symptoms (such as agitation) that patients with depression may be experiencing while waiting for the underlying antidepressant to take effect. And while this may be facilitatory in the short term, it is an approach that is risk-laden because of the likelihood of persisting with these medications, which in the case of benzodiazepines can lead to dependence. Finally, it should also be noted that agents such as esketamine and brexanolone are not indicated for rapid response per se, and nor is there sufficient evidence as yet for their use in this manner; hence, they are not included in the main recommendations in these guidelines, regarding the management of mood disorders.
Rapidly acting antidepressants
Most currently available antidepressants take time to produce a clinically significant response. Faster onset of action is clearly desirable, particularly in severe or high-risk cases. However, no currently established antidepressant agent has consistently shown a faster onset of action. But recently, two new agents appear to show that a more rapid antidepressant action may be possible.
The first agent is esketamine – the s-enantiomer of ketamine. It is a novel antidepressant that has recently been granted an indication in the United States for adjunctive use in the treatment of ‘resistant’ depression by the FDA, and in Europe by the EMA. However, upon initial assessment by NICE, in the United Kingdom, funding for the administration of esketamine under the NHS was not approved immediately because of concerns regarding benefit and cost.
The second agent is brexanolone, a potent positive allosteric modulator of synaptic and extrasynaptic GABA receptors (Kanes et al., 2017), that has to be administered by intravenous infusion, 5 which has been approved for the treatment of postpartum depression in the United States.
Ketamine
Ketamine has been available as an anesthetic agent for more than half a century and in addition to its use in humans, is used widely in veterinary practice. It has also been used for the management of chronic pain in humans for over 30 years but has gained notoriety because of recreational use.
In anesthetic doses ketamine has been associated with neuropsychiatric complications such as hallucinations and dissociation, and it lends itself to abuse because of its euphoric and hallucinogenic actions. Pharmacologically, ketamine is referred to as a ‘pharmacologist’s nightmare’ because of its numerous receptor interactions and complex mechanism of action. However, it primarily acts as a competitive antagonist at the NMDA glutamatergic receptor, and its effects at opioid receptors may contribute to its mood elevating effects (Williams et al., 2018).
The antidepressant effects of subanaesthetic intravenous doses of ketamine were first demonstrated in open and then placebo-controlled trials over a decade ago (Berman et al., 2000). Ketamine showed a reduction in mood symptoms in a proportion of subjects with treatment-resistant depression (see section 9. ‘Response to treatment’) observable within 6–12 hours. This effect generally lasted less than a week following a single dose. Subsequent evidence has emerged showing a similar pattern of response in smaller studies in subjects with refractory bipolar depression (Zarate et al., 2012). This use of low-dose ketamine is associated with transient side effects (1–2 hours) including dizziness, blurred vision, headache, nausea or vomiting, dry mouth, poor coordination, difficulties in concentration and restlessness and occasionally transient elevation in blood pressure and heart rate (Zarate et al., 2006). The practical limitations of intravenous use and concerns over the risks of ongoing abuse have limited its widespread utilisation, although considerable off label use of various formulations of ketamine has subsequently occurred (Malhi et al., 2016a). Currently no form of ketamine has regulatory approval for the treatment of Major depression in Australia or New Zealand.
Esketamine
More recent research has focused on the clinical development of an intranasal formulation of esketamine, a racemic isomer of ketamine which may have more potent affinity for the NMDA receptor. In some studies, esketamine has shown a sustained (beyond 1 month) pattern of response in subjects with treatment-resistant depression, and maintenance of the acute response has been achieved with repeated administration (Daly et al., 2018; Popova et al., 2019; Zheng et al., 2020a). A placebo-controlled relapse prevention trial has also demonstrated prevention of depressive relapse after acute treatment (Daly et al., 2019). However, the clinical validity and significance of these short, and longer term data together with ongoing uncertainty around the issues of abuse potential is still a matter of pointed debate (Horowitz and Moncrieff, 2020; Kryst et al., 2020; Mahase, 2020; Schatzberg, 2014; Sial et al., 2020). The trials of esketamine showed a similar pattern of side effects to those of ketamine including transient post dose hypertension and dissociation in a proportion of subjects. Esketamine has been licensed for use in treatment-resistant depression (see section 9.3 ‘Difficult-to-treat depression (DTD) and treatment-resistant depression (TRD)’) by the FDA with a requirement that patients require a minimum of 2 hours post dose monitoring in a clinically supervised setting to assist in the management of side effects. This is appropriate given the above safety concerns.
Currently there is no specific data directly comparing the relative efficacy and tolerability of intravenous or other formulations of ketamine to each other or to esketamine. It is also noteworthy that the majority of the available esketamine data stems largely from industry sponsored trials.
One frequently reported anecdotal observation from early trials of intravenous ketamine was a specific benefit for suicidality. This possible benefit was examined in 2 recent randomised controlled trials comparing the benefits of esketamine or placebo added to new antidepressant therapy in inpatients with severe major depression with suicidal intent (Fu et al., 2019a; Ionescu et al., 2019). There were no differences between esketamine and placebo groups in terms of a reduction in suicide risk, which was observed in all groups; and a more recent small sample size (n = 68) RCT suggests some benefit to suicidal ideation measures (Canuso et al., 2019). Hence, further studies are needed.
Neurosteroids
Molecules termed neurosteroids are also under investigation for potential rapid antidepressant effect; these include a group of synthetic progesterone analogues with potential central actions as modulators of the GABA receptor in addition to potentially relevant actions at central gonadotrophin receptors. These actions have led to the early investigation of their use in major depression and more specifically major depression occurring in either the postpartum or perimenopause periods. The first of these agents to achieve FDA approval is brexanolone which has randomised controlled trial evidence in the treatment of severe postpartum depression (Meltzer-Brody et al., 2018). However, currently brexanolone is only available as a prolonged (60 hours) intravenous infusion and this, along with its prohibitive cost, limits its clinical utility. A number of other compounds in this class are currently under investigation but are not yet sufficiently developed for clinical use.
Mood-stabilising agents
The term mood stabiliser, although intuitively appealing and clinically meaningful, is somewhat confusing as no medications have an indication as a mood-stabilising agent (MSA) (Goodwin and Malhi, 2007; Malhi et al., 2018b). In recent years, attempts have been made to operationalise the definition of MSAs and various criteria have been proposed. However, none are as yet widely adopted. Clinically, the key actions of an agent that qualify it as a useful mood stabiliser, are its effects at both ends of the mood spectrum, namely depression and mania, and its ability to maintain euthymia by preventing future mood instability. Prophylaxis is essential to the definition and clinical utility of a MSA. With these factors in mind, the most effective MSAs are lithium and the anticonvulsant, valproate, though the anticonvulsant lamotrigine is also included alongside these two because it is not an SGA (the other ‘class’ that putatively has mood stabilising properties). Lithium remains the most effective MSA, with further evidence supporting its use in mood disorders as monotherapy and as an adjunct (Malhi et al., 2017c; Nolen et al., 2019). It is also preferable because of its additional anti-suicidal and neuroprotective properties, although it can be a little more complicated to manage and can on occasion be problematic with respect to tolerability (Malhi et al., 2012; Nederlof et al., 2019).
In the management of bipolar depression, the mood stabilisers lithium and valproate have been positioned as monotherapy Choices even though evidence for their efficacy in the treatment of acute bipolar depression is not as robust as other agents such as quetiapine (Young et al., 2010).
Regarding lithium, there is abundant empirical evidence for its efficacy in bipolar depression, added to which it is a potent anti-manic agent and arguably the best prophylactic agent (Malhi et al., 2017b). Given that long-term tolerability is a key consideration, even when managing an acute episode of illness, lithium is granted primacy (Malhi et al., 2020a). In addition, it is important to note that in many instances the lack of efficacy of lithium is partly because a ‘lithium-responsive’ patient has not been adequately identified. The key feature of a lithium-responder is the pattern of the illness in which the mood episodes are discrete and recurrent, and in the periods between episodes, patients achieve full remission. This classic episodic pattern of illness involving
Lithium.
In the management of mood disorders, lithium has a special role (Álvarez et al., 2019; Malhi et al., 2020d). It is the only agent that is specific to the treatment of an illness, namely, bipolar disorder, formerly known as manic depressive illness (Malhi et al., 2020a). However, it is important to note that bipolar disorder, as described in the modern day, is not the same as manic depressive illness, and lithium specificity is more attuned to recurrent depressive disorders and those that have clear episodes with periods of remission. In recent years, the use of lithium has declined, despite new evidence supporting its efficacy and overall effectiveness in the management of mood disorders and the discovery of significant additional benefits such as the prevention of suicide and direct neuroprotective effects. This is largely attributed to a lack of marketing, as compared to newer agents, and misconceptions regarding its tolerability and difficulties in prescription. However, as has been described elsewhere, lithium is relatively easy to prescribe and manage long term and its side effects have been exaggerated (Malhi et al., 2017c). It remains an important molecule for the treatment of mood disorders and hence why it features strongly throughout these guidelines taking pole position in many of the recommendations. Its therapeutic window is readily achieved because its plasma levels can be monitored, and this should be considered a benefit as opposed to a hindrance, and provided its levels are monitored carefully and unnecessary peaks are avoided, its toxicity long term can be limited (Malhi et al., 2011, 2016b). However, lithium does have side effects, particularly involving thyroid and renal function, and can cause acute side effects, such as tremor, change in taste and a flare up of psoriasis. In most cases these are transient. To avoid side effects, the lowest dose necessary should be prescribed and plasma levels can be tailored according to whether it is being used to treat depression, mania or to provide long-term mood stability and prophylaxis. Lithium is mainly used in the treatment of bipolar disorder but is also effective as an augmentation strategy in the treatment of depression. Underscoring once again its versatility, it can be combined with all antidepressants and in addition to having an independent antidepressant effect, it acts synergistically, augmenting the effects of other medications (Malhi et al., 2017d).
Unlike lithium, there is little evidence for the use of valproate in the treatment of major depression. However, while the evidence regarding its use in the treatment of acute bipolar depression is modest it features as a Choice indication alongside lithium. This is because, like lithium, valproate has efficacy in mania in addition to which, empirically it has been shown to be effective for maintenance and prophylaxis with a possible reduction in depressive symptoms long term (Bowden et al., 2000). The trials supporting the use of valproate for acute bipolar depression are few, but all show benefit in favour of valproate as compared to placebo (Bond et al., 2010; Davis et al., 2005; Ghaemi et al., 2007; Muzina et al., 2011). It is important to note that valproate is not recommended for use among women of childbearing age because of its high rate of teratogenicity, polycystic ovarian syndrome risk, and its potential to lead to developmental problems in offspring.
Lamotrigine has the converse problem to that of lithium and valproate. A meta-analysis of a number of double-blind placebo-controlled trials of acute bipolar depression indicate that it has efficacy (Geddes et al., 2009), even though the individual trials fail to separate consistently from placebo. This is possibly because of methodological issues such as the need for slow titration of lamotrigine and the overall short duration of some of its efficacy trials. Because of these factors, effective doses in these RCTs were possibly only achieved towards the conclusion of studies, in other words for a relatively short period of time. Additional support for lamotrigine as a Choice indication for acute bipolar depression comes from its use as an adjunct to lithium (Van Der Loos et al., 2009) and quetiapine (Geddes et al., 2016), respectively, compared to placebo adjunctive therapy, in which lamotrigine produced a significant benefit. However, unlike lithium and valproate, lamotrigine has only demonstrated antimanic actions in a pooled analysis of two RCTs (Goodwin et al., 2004) and is probably less effective than these agents long-term for maintenance and prophylaxis of mania. Nevertheless, given its effectiveness in treating bipolar depression and in particular because of its tolerability, it is a justified monotherapy Choice.
Second-generation antipsychotics (SGAs)
The second-generation antipsychotics (SGAs), also known as atypical antipsychotics, are weak D2 receptor antagonists but potent blockers of 5HT2A/2c receptors. They are also 5HT1A receptor partial agonists. Clinically, SGAs such as lurasidone, quetiapine and olanzapine may be used either as monotherapy or adjunctively in the treatment of major depression and comparatively new agents such as aripiprazole and brexpiprazole have formal indications for adjunctive treatment of major depression.
In the treatment of acute bipolar depression, the Choice SGAs include quetiapine, lurasidone and cariprazine, as there is randomised controlled trial evidence for each of these (Calabrese et al., 2005; Durgam et al., 2016; Loebel et al., 2014). However, it is important to note that a number of the trials have significant methodological issues that moderate the strength of the evidence. On the basis of empirical evidence, lurasidone may be best suited as the Choice SGA, especially as it also has evidence when used in combination with other agents. In comparison to MSAs, the SGAs are generally ‘second Choice’ because of their poorer tolerability longer term.
Nutritional supplements
Probiotics
The use of probiotic supplements to alter the gut microbiome has gained attention in recent years. There is emerging support for the benefits of these supplements in the management of depressive disorders (Huang et al., 2016), but further research is necessary to confirm and clarify their clinical utility, effect sizes and optimal probiotic formulations (Goh et al., 2019).
Methylfolate
The use of methylfolate in the management of mood disorders may be beneficial, especially when patients have polymorphisms of the methylene tetrahydrofolate reductase gene (MTHFR), which result in less efficient conversion of folate to methylfolate (its active metabolite) in the single carbon cycle (Zheng et al., 2020b). The evidence for the benefits of such supplements has received mixed empirical support (Young et al., 2019; Zheng et al., 2020b). MTHFR polymorphisms can now be determined by genetic analysis and may help target patients where methylfolate containing supplements may have clinical utility (Dartois et al., 2019).
Omega-3 fatty acids
The evidence for beneficial effects from Omega-3 polyunsaturated fatty acid supplements containing eicosapentaenoic acid (EPA) in depressive disorders has empirical backing (Deacon et al., 2017; Liao et al., 2019). However, the optimal proportion of EPA is yet to be determined, and quality assurance of available products remains problematic (Heller et al., 2019). It is notable that EPA composing over 60% of the supplement is the only formulation that appears to be effective (Dartois et al., 2019).
Hypericum perforatum
Hypericum perforatum (St John’s Wort) continues to appear helpful in the management of both unipolar and bipolar depressive disorders. However, the benefits have only been confirmed for depression of mild to moderate severity and interactions with other medications can be significant (e.g. serotonin syndrome with serotonergic compounds) as can the risk of precipitating mania in patients with bipolar disorder. Given the availability of low-cost pharmaceutical grade SSRIs, there is little case for use of Hypericum perforatum in the contemporary era.
S-adenosylmethionine
The benefits of S-adenosylmethionine in the management of depressive disorders have not been confirmed by further investigations (Sarris et al., 2020; Targum et al., 2018).
Cannabidiol
There has been increased interest in cannabinoids, in particular cannabidiol (medical marijuana), in the treatment of specific forms of epilepsy, chronic pain, nausea and vomiting (Whiting et al., 2015) and there is interest in its potential use for psychiatric disorders (Premoli et al., 2019). The RANZCP has produced a clinical memorandum to provide information for psychiatrists about the therapeutic use of medical cannabis (www.ranzcp.org/files/resources/college_statements/clinical_memoranda/cm-medical-use-of-cannabinoids.aspx). Since its use has become more widespread, it is being used to treat depression (Corroon and Phillips, 2018), however, as shown in recent systematic reviews (Bonaccorso et al., 2019; Pinto et al., 2019), there is a lack of evidence for its efficacy in the treatment of depression. The effectiveness of cannabidiol for treatment of mania has been examined in case reports that concluded it was not effective in reducing manic symptoms but that it did not cause harm (Zuardi et al., 2008).
It should be emphasised that there are risks of harm from the effects of street marijuana, especially with high levels of Δ-9-tetrahydocannabidiol (THC), with the risk of inducing relapse in patients with bipolar disorder or worsening depression (see Table 11).
Supplements in depression.

MTHFR: methylene tetrahydrofolate reductase gene; EPA: eicosapentaenoic acid; SSRI: selective serotonin reuptake inhibitor.
6.3. Alternatives
Physical treatments
ECT
Recently published studies and reviews have continued to support the efficacy and safety of ECT which remains the most effective form of neurostimulation for the treatment of severe and psychotic depression. Detailed guidelines for ECT practice have recently been published by the RANZCP (Weiss et al., 2019). In addition, clinical practice recommendations for maintenance ECT have been published by an Australian consensus workshop (Gill and Kellner, 2019). Both of these documents are based on a combination of evidence and consensus opinion and are valuable references for those administering ECT.
In general, the current recommendations for the use of ECT in the management of mood disorders have not altered substantially from the previous guidelines (Malhi et al., 2015). However, important information relevant to clinical practice has been published in the past 5 years and this is briefly summarised below, together with updated recommendations (see Recommendation Box 1).
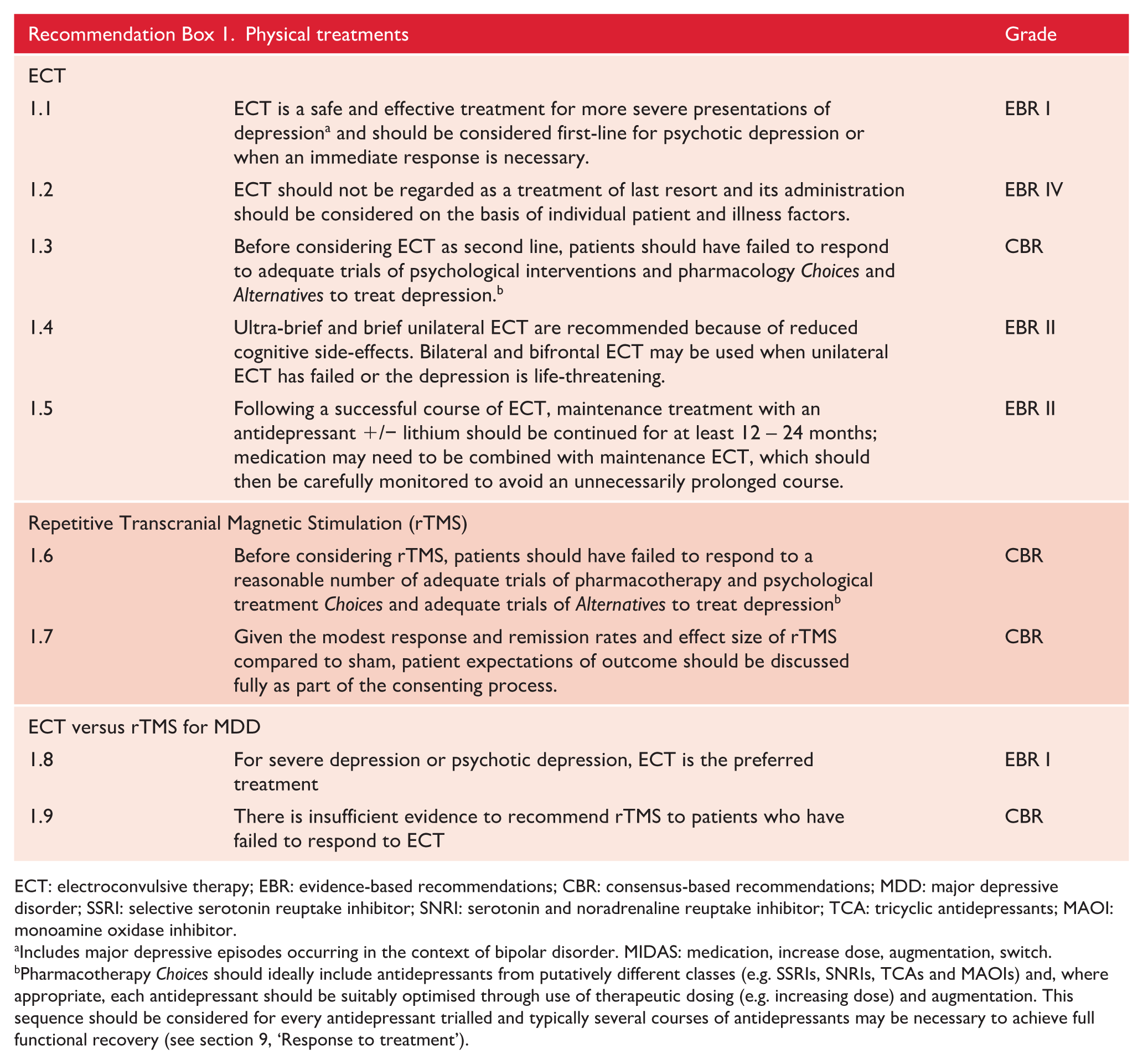
ECT: electroconvulsive therapy; EBR: evidence-based recommendations; CBR: consensus-based recommendations; MDD: major depressive disorder; SSRI: selective serotonin reuptake inhibitor; SNRI: serotonin and noradrenaline reuptake inhibitor; TCA: tricyclic antidepressants; MAOI: monoamine oxidase inhibitor.
Includes major depressive episodes occurring in the context of bipolar disorder. MIDAS: medication, increase dose, augmentation, switch.
Pharmacotherapy Choices should ideally include antidepressants from putatively different classes (e.g. SSRIs, SNRIs, TCAs and MAOIs) and, where appropriate, each antidepressant should be suitably optimised through use of therapeutic dosing (e.g. increasing dose) and augmentation. This sequence should be considered for every antidepressant trialled and typically several courses of antidepressants may be necessary to achieve full functional recovery (see section 9, ‘Response to treatment’).
Depression
ECT continues to be recommended as first line treatment for severe depression when a rapid response is necessary, such as when a patient is at risk from suicide, malnutrition or dehydration and for psychotic depression and catatonia (Dessens et al., 2016; Luchini et al., 2015), or where there are high levels of distress or there is a past history of good response. More commonly, it is recommended for depression that has failed to respond to combined pharmacological and psychological management. The use of ECT in this situation should be based on the assessment of the individual patient’s circumstances, including the degree of impairment, severity of symptoms, the adequacy of prior treatment and the duration of illness. Where indicated (see Table 12) it can be recommended after one treatment failure, but for most cases of treatment resistance (non-response) it should be considered after two failures (Rasmussen, 2019). A recent cost-effectiveness evaluation of ECT (Ross, 2018) concluded that third line ECT, after two failures of treatment, is the optimal strategy (for a more detailed discussion of responsivity, TRD and DTD; see section 9, ‘Response to treatment’).
Indications for ECT in mood disorders.
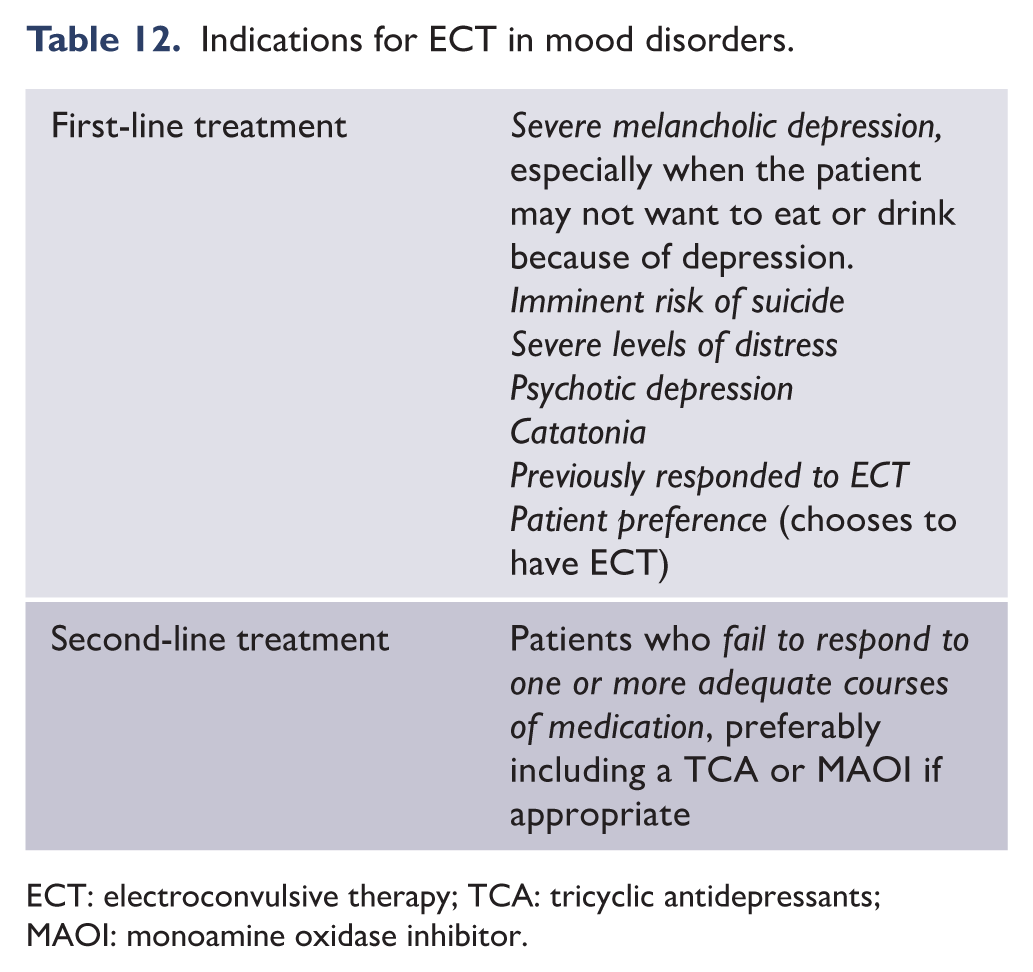
ECT: electroconvulsive therapy; TCA: tricyclic antidepressants; MAOI: monoamine oxidase inhibitor.
Bipolar disorder
ECT is effective for bipolar depression. It may produce a more rapid response than in unipolar depression (Agarkar et al., 2018; Bahji et al., 2019) and may be more effective than pharmacological treatment (Schoeyen et al., 2014). Care should be taken to monitor and manage the risk of ECT-induced mania. Mania also responds to ECT and is recommended when pharmacological management is unsuccessful (Perugi et al., 2017; Weiner and Reti, 2017; Wong et al., 2019). ECT is also indicated for bipolar mixed states unresponsive to pharmacological management (Medda et al., 2015; Perugi et al., 2017), but maintenance ECT as a ‘mood stabiliser’ has not been studied sufficiently, although there is evidence that it can be effective in reducing episodes and the frequency of hospitalisation (Santos Pina et al., 2016).
Despite some recent research recalibrating the risks associated with ECT in terms of cognitive side effects, the risk of ECT-related cognitive impairment remains a potential limiting factor for ECT practice. The decision to offer ECT to patients must only be made after careful consideration of the potential adverse effects on cognition and its consequences for the individual patient, including any possible effects on psychosocial and occupational functioning. The aim should be to achieve a balance between efficacy and side effects by selecting the treatment parameters that are most appropriate to the individual patient and the illness characteristics. Patients and family, where appropriate, should be fully informed about the potential benefits to be expected, as well as the potential adverse effects, including the possible effects on memory and the low risk of long-term retrograde memory impairment. The cognitive status of patients undergoing ECT should be monitored during and after a course of treatment, using an appropriate measuring instrument.
Safety of ECT
A recent systematic review of the mortality associated with ECT (Tørring et al., 2017) found that in studies published from 2001 there was one ECT-related death reported in a total of 414,747 treatments. It concluded that death caused by ECT is an extremely rare event.
ECT and cognitive effects
Despite improvements in ECT administration, including the introduction of ultra-brief pulse width unilateral ECT, bifrontal placement and refinements in anaesthetic technique, the risk of some degree of ECT-related cognitive impairment remains a concern.
Memory
The available data indicate that the majority of negative cognitive effects, including anterograde memory impairment, are short-lived and reversible and that shortly following ECT many cognitive functions are improved compared with baseline function because of the negative impact on cognition of the untreated illness (Andrade et al., 2016; McClintock et al., 2014; Semkovska and McLoughlin, 2010). However, although still debated, there are concerns about the long-term effects of ECT on retrograde memory, particularly autobiographical memory (Andrade et al., 2016; Fraser et al., 2008; Sackeim et al., 2007). Adverse cognitive changes are more common with bitemporal placement compared to unilateral, higher doses, greater numbers of treatments and with three times weekly treatment compared to twice weekly (Semkovska et al., 2016; UK ECT Review Group, 2003).
Confusion
While the effects on memory are of most concern to patients, impairment in other cognitive domains can occur. A brief period of post-ECT disorientation is common and rarely this can persist as a confusional state (delirium) which may last hours or exceptionally, days. While self-limiting, such prolonged confusion can be distressing to patients and their families.
Other cognitive effects
Other cognitive domains that can be affected include executive functioning, processing speed, verbal fluency, attention and spatial orientation, all of which have been shown to rapidly resolve after the end of a course of ECT (Andrade et al., 2016; Semkovska and McLoughlin, 2010).
ECT in special populations
The elderly
A systematic review and recent studies of ECT-related cognitive impairment in the elderly (Kumar et al., 2016; Lisanby et al., 2020; Obbels et al., 2019) suggest that ECT does not cause significant impairment as measured by a commonly used cognitive screening test (MMSE), and that with ultra-brief pulse right unilateral ECT there are few adverse effects. However, there may be an adverse effect on autobiographical memory in some patients (Lisanby et al., 2020).
Although clinical experience supports the efficacy of ECT in the elderly, controlled data in this age group remain sparse. More recent literature, however, provides strong support for its use in acute depression and as maintenance treatment (Geduldig and Kellner, 2016; Kellner et al., 2016a, 2016b). A recent review of ECT in the elderly (Meyer et al., 2018) concluded that it was a safe and effective treatment in this age group, that the cognitive side effects are transient, and that it does not worsen dementia.
Children and adolescents
The use of ECT in depressed adolescents has been, and remains, controversial, with some jurisdictions banning ECT in this age group. However, available data support the efficacy and safety of ECT in adolescents and young adults (Weiner and Reti, 2017). The RANZCP guidelines for ECT (Weiss et al., 2019) recommend that ECT should be available to treat adolescents. Three recent retrospective studies examined the outcomes of a total of 304 adolescents who were treated with ECT for a range of psychiatric disorders (Benson et al., 2019; Karayağmurlu et al., 2020; Maoz et al., 2018) and found ECT to be safe and effective for mood disorders, schizophrenia and catatonia. However, adolescents and young adults may have a slower response to ECT and may require more treatments (>12 applications of ECT) than older patients and are also at more risk of prolonged seizures (Maoz et al., 2018).
ECT and depression with comorbid personality disorder
It has been recognised that patients with borderline personality syndrome and comorbid depression are less likely to respond to ECT and have a higher relapse rate than those without personality disorder (Rasmussen, 2015a). The use of ECT in these patients has therefore been questioned. However, a recent prospective study comparing outcomes of 29 depressed borderline patients with 108 non-borderline patients treated with ECT found equal efficacy between the two (Lee et al., 2019). Although a challenging group to assess and manage, a diagnosis of borderline personality disorder should not preclude ECT as a treatment for comorbid depression.
ECT and pregnancy
There have been five systematic reviews of the safety of ECT in pregnancy since 2015 (Calaway et al., 2016; Coshal et al., 2019; Leiknes et al., 2015; Sinha et al., 2017; Spodniakova et al., 2015), with Calaway et al, focusing on the safety of ECT in the first trimester. These overlapping reviews examined several hundred case reports and concluded that ECT in pregnancy was relatively safe. The review by Leiknes et al. (2015), was more cautious, concluding that the risks were significant and that ECT should only be used as a last resort in pregnant women. However, in this review, they included all adverse outcomes, some of which could not be considered to be a consequence of the ECT. The most common adverse effects that have been identified include transient foetal bradycardia, uterine contractions (due to oxytocin release) and transient maternal hypertension. Less common or rare events include mild vaginal bleeding, premature labour and one report of maternal status epilepticus with foetal death (Balki et al., 2006).
Although the use of electroconvulsive therapy to treat severe psychiatric illness in pregnant women remains controversial, the available evidence indicates that it is a safe and effective treatment (Ward et al., 2018; Weiss et al., 2019), and should be considered for women with severe psychiatric disorders where Alternative treatments have not been effective or when there are high risks associated with the disorder. The reported low risk of adverse foetal effects need to be balanced against the adverse effects of untreated maternal illness on both mother and foetus, such as prematurity, low birth weight, pre-eclampsia, drug and alcohol abuse, psychosis, suicide and impairment of maternal-infant bonding.
During the second and third trimester ECT should only be given in an appropriate setting where obstetric care and foetal monitoring are available. Close liaison between psychiatrist, obstetrician and anaesthetist is essential to assessment and management. The patient and partner should be fully informed of the potential risks and benefits of both ECT and alternative treatments. Obstetric unit staff need education and counselling about ECT in pregnancy to ensure that they are supportive of the patient.
Maintenance post-ECT
In order to prevent the recognised high rates of relapse and recurrence following a successful course of ECT, some form of maintenance treatment is required. The strategies that have been shown to be effective are maintenance ECT, pharmacological maintenance with an antidepressant and/or lithium, and the combination of both ECT and medication (Weiner and Reti, 2017). Of these strategies the combination of maintenance ECT and medication appears to be more effective than medication alone (Kellner et al., 2016a). Maintenance ECT has not been shown to worsen cognitive function over time (Kirov et al., 2016; Weiner and Reti, 2017).
Consensus based recommendations for relapse/recurrence prevention post-ECT have recently been made by an Australian expert group (Gill and Kellner, 2019). The group recommends post-ECT maintenance to consist of antidepressant medication +/− lithium for at least 12–24 months. This will often be accompanied by a maintenance ECT program which should be carefully monitored to prevent it being unnecessarily prolonged.
Lithium
There is increasing interest in the role of lithium to prevent suicide as well as maintain wellness post-ECT (Rasmussen, 2015b). A large Swedish cohort register-based study of over 7000 patients treated with ECT for unipolar depression (Brus et al., 2019) demonstrated that at 12 months post-ECT there were no suicides among those patients who had been treated with lithium compared to 56 suicides in the non-lithium group. The readmission rate for the lithium group was significantly lower than the non-lithium group. These findings suggest an important role for lithium as part of maintenance therapy following ECT.
Repetitive transcranial magnetic stimulation (rTMS)
Recently, the RANZCP has developed guidelines for the administration of rTMS, (see RANZCP Professional Practice Guideline 16 November 20186), however, there are considerable financial and time costs associated with the administration of rTMS, and so comparisons of rTMS with Alternative strategies such as antidepressant switch or augmentation are important in determining whether rTMS is appropriate at an early stage of treatment (see Recommendation Box 1). Data comparing rTMS to other interventions (e.g. medication, psychological intervention and ECT) is limited (Benadhira et al., 2017; Needs et al., 2019; Sehatzadeh et al., 2019; Trevizol et al., 2020). For example, after a mean of between 2.2 and 2.7 failed antidepressant trials, there is no difference between a switch to venlafaxine compared with rTMS (Brunelin et al., 2014). This means that Level I data is absent regarding the stage at which rTMS should be used and given the comparative convenience and accessibility of pharmacotherapy our recommendation is that pharmacotherapy Choices should ideally include antidepressants from putatively different classes (e.g. SSRIs, SNRIs, TCAs and MAOIs) and, where appropriate, each antidepressant should be suitably optimised through use of therapeutic dosing (e.g. increasing dose) and augmentation (provided there has been a response to the antidepressant). This sequence should be considered for every antidepressant trialled and typically several courses of antidepressants may be necessary to achieve full functional recovery and that such pharmacotherapeutic optimisation should occur prior to considering rTMS.However, several recent systematic reviews and meta-analyses of randomised controlled trials, including sham-controlled, provide modest evidence for the efficacy of rTMS in the treatment of unipolar and bipolar depression that has failed to respond adequate pharmacological management (Berlim et al., 2014; Brunoni et al., 2017; Garnaat et al., 2018; Hovington et al., 2013; Kedzior et al., 2015; McClintock et al., 2018; Milev et al., 2016; Mutz et al., 2018, 2019; Perera et al., 2016). And on the basis of this, in many countries, rTMS is regarded as an appropriate treatment for depression that has failed to respond to adequate antidepressant treatment (McClintock et al., 2018; Milev et al., 2016).
Efficacy of rTMS in depression
The interpretation of the results of meta-analyses and systemic reviews of rTMS for the treatment of depression is made complex by the considerable variation in methodology between studies and the various ways of analysis and presentation of results. Many randomised controlled studies have been underpowered and studies have varied in the target population studied (unipolar, bipolar); medication status and degree of treatment resistance; quality of the sham condition; and the total number of treatments given (Garnaat et al., 2018). Additionally, there is considerable variability in treatment parameters, such as site and frequency of stimulation, numbers of stimuli delivered and the type of stimulation employed.
Most studies have examined the efficacy of high-frequency stimulation of the left dorsolateral prefrontal cortex (LDLPFC) with a smaller number studying low-frequency stimulation of the right DLPFC, with systematic reviews showing equal efficacy between these two forms of treatment (Brunoni et al., 2017; Milev et al., 2016).
Virtually all RCTs reviewed systematically or by meta-analysis have been conducted on treatment resistant subjects who have failed one or more adequate antidepressant courses. To date, there have been no controlled trials of rTMS monotherapy in a treatment-naïve, non-resistant depressed population (Kiebs et al., 2019). Early studies employed a relatively short course of treatment (5–10 sessions) compared with 20–30 sessions in later studies. There is sham-controlled evidence that longer courses of treatment have greater efficacy (Teng et al., 2017).
Different meta-analyses of sham-controlled studies report results differently. Efficacy has been reported as response (a 50% reduction of depression rating scale score) and remission (MADRS < 10 or HAMD-17 < 7) rates (with many reporting only response rates); as odds ratios; effect sizes; number needed to treat (NNT) or as weighted mean difference in depression rating scale scores. Reported response rates from RCTs have ranged up to 50%, and remission rates up to 35% (Perera et al., 2016; Rostami et al., 2017; Sehatzadeh et al., 2019; Taylor et al., 2017). Odds ratios reported for response range from 3.3 to 3.75 and for remission from 2.51 to 3.3 (Berlim et al., 2014; Brunoni et al., 2017; Mutz et al., 2018, 2019). Effect sizes for rTMS over sham range from small (0.33) to moderate (0.6) (Health Quality Ontario, 2016; Kedzior et al., 2015; Slotema et al., 2010; Taylor et al., 2017). Numbers needed to treat (NNT) for a response range from 6 to 10 and for remission from 8 to 10 (Berlim et al., 2014; Health Quality Ontario, 2016).
A recent systematic review and meta-analysis of randomised sham-controlled trials of high-frequency left DLPFC rTMS for depression that had failed at least one adequate course of antidepressant (Health Quality Ontario, 2016) reported response and remission rates of 25.1% and 17.4%, respectively, compared to response and remission rates of sham treatment of 12.3% and 6.7%. The effect size for response or remission was small at 0.33, with NNT of 10 for both. The weighted mean difference in end-point depression scores between rTMS and sham was reported as statistically significant, but small, and did not meet pre-specified criteria for clinical significance.
Naturalistic (‘real-world’) effectiveness studies report similar or higher rates of response and remission. A recent naturalistic multicentre study (Taylor et al., 2017) used three different assessments, QIDS-SR, PHQ-9 and CGI, to measure response and remission. The QIDS-SR showed response and remission rates of 29.4% and 5.9%, respectively, while the corresponding rates for the PHQ-9 were 39.2% and 15.7% and for the CGI were 50.9% and 17.9%. An earlier multicentre naturalistic study (Carpenter et al., 2012) showed response and remission rates of 58% and 37.1%, respectively (using CGI).
To summarise, systematic reviews and meta-analyses of sham-controlled studies of rTMS for depression show statistical superiority of active treatment over sham. However, the clinical significance of this difference has been questioned (Health Quality Ontario, 2016), and overall, greater efficacy is reported for high-frequency left DLPFC and low-frequency right DLPFC stimulation, and for longer treatment courses using higher numbers of stimuli per session.
Predictors of response
Studies exploring potential predictors of response have yielded inconclusive and inconsistent results (McClintock et al., 2018). Some clinical and demographic variables can impact on response but there is insufficient evidence for reliable predictors (Fitzgerald et al., 2016). Some studies suggest rTMS is less effective in older than younger patients (Beuzon et al., 2017; Kar, 2019; Rostami et al., 2017) while others find no age effect (Fitzgerald et al., 2016) or a non-linear effect (Trevizol et al., 2020). Illness related factors that have some positive predictive value include less severe depression, shorter duration of episode, recurrent rather than single episode depression, a predominance of cognitive/affective symptoms over somatic symptoms and previous response to rTMS (Beuzon et al., 2017; Fitzgerald et al., 2016; Kar, 2019; Rostami et al., 2017; Trevizol et al., 2020). Bipolar depression is as likely to respond as unipolar depression (Rostami et al., 2017). Negative predictors of response include high levels of treatment resistance, longer duration of episode, significant comorbid anxiety, the presence of psychotic symptoms and previous lack of response to ECT (Brunoni et al., 2019; McClintock et al., 2018; Voigt et al., 2019).
rTMS compared to other treatments (positioning of rTMS)
The antidepressant efficacy of rTMS has been compared to electroconvulsive therapy in a number of studies. Several meta-analyses consistently report the superiority of ECT over rTMS for the treatment of depression (Brunoni et al., 2019; Chen et al., 2017; Health Quality Ontario, 2016; Micallef-Trigona, 2014; Mutz et al., 2019; Ren et al., 2014), particularly for psychotic depression (Ren et al., 2014).
A consensus statement (McClintock et al., 2018) contrasted the response and remission rates of rTMS for treatment-resistant depression with those of antidepressant treatment stages found in the STAR-D study (Rush et al., 2006) and suggested equivalent outcomes. However, controlled data comparing rTMS with antidepressants in treatment-resistant depression are scarce. One randomised controlled trial compared low-frequency rTMS applied to the right DLPFC against venlafaxine and a combination of the two treatments in patients with treatment-resistant depression (Brunelin et al., 2014) and found no significant difference in response and remission rates between the three arms, in patients who had failed to respond to a mean of between 2 and 3 previous antidepressant trials. Thus, currently, there is insufficient evidence for differential efficacy of rTMS and antidepressants in treating resistant depression.
Variations in rTMS technique
rTMS can be delivered in a number of different ways. The site of stimulation can be right or left DLPFC, dorso-medial PFC or bilateral. Frequency can be high or low and the number of stimuli per session can vary. Differing coils can be used to vary the depth of stimulation and different strategies – altering the nature of the stimulation such as theta-burst, synchronised, priming and accelerated can be employed. A systematic review and network meta-analysis (Brunoni et al., 2017) of various forms of rTMS for depression found that high-frequency, low-frequency, priming low-frequency, bilateral and theta-burst techniques were more effective than sham and had equivalent efficacy, but that accelerated, synchronised and deep rTMS were no more effective than sham. Several systematic reviews of sham-controlled studies of high-frequency left DLPFC and low-frequency right DLPFC demonstrate equivalent efficacy (Brunoni et al., 2017; Cao et al., 2018; Milev et al., 2016; Mutz et al., 2018).
Guidelines for an appropriate choice of rTMS parameters are outlined in RANZCP Professional Practice Guideline 16 (November 2018).
Maintaining response post-TMS
In common with pharmacotherapy and ECT, relapse without maintenance treatment following treatment with rTMS is common (Garnaat et al., 2018; Health Quality Ontario, 2016; Kedzior et al., 2015; Sackeim, 2016) with estimates of relapse at 6 months as high as 77% (Milev et al., 2016). Several studies and reviews demonstrate that maintenance antidepressant medication post-rTMS significantly reduces relapse rates within 12 months (Dunner et al., 2014; Perera et al., 2016; Philip et al., 2016). Continuation/maintenance rTMS has also been shown to prolong acute benefit of rTMS and reduce relapse rates over 12 months, as well as being effective when re-introduced to treat a relapse in an acute responder (Garnaat et al., 2018; Milev et al., 2016; Perera et al., 2016; Philip et al., 2016; Rachid, 2018; Sackeim, 2016). Therefore, maintaining response with medication and/or rTMS post-rTMS is likely to be necessary, but to date, there are insufficient data to determine the best approach (Garnaat et al., 2018).
Other brain stimulation methods
Transcranial direct current stimulation
Transcranial direct current stimulation (tDCS) is a method of neurostimulation that has been proposed as a treatment for acute depression. It involves the application of a low-intensity direct current passing between two electrodes (anode and cathode) placed on the scalp. A recent large multicentre, international randomised sham-controlled study of tDCS in unipolar and bipolar depression (Loo et al., 2018) found that there was no difference between active and sham treatments in either group, although there was a question about the possible biological activity of the sham condition. A recent systematic review of tDCS (Borrione et al., 2018) found that most studies were small, underpowered and most returned negative results.
In summary, at this stage, there is insufficient evidence from randomised sham-controlled trials to recommend tDCS for clinical use.
Magnetic seizure therapy (MST)
Magnetic seizure therapy involves using high-powered transcranial magnetic stimulation, which penetrates the superficial layers of the cortex. Therefore, unlike electroconvulsive therapy, the induced seizure is focal, limited to the cortex and does not involve deeper structures, thereby reducing the risk of cognitive impairment compared to ECT (Lisanby et al., 2003).
In summary, while showing promise as an effective treatment, possibly rivalling ECT in efficacy but with much reduced cognitive side effects, MST remains an experimental treatment and insufficient data are available as yet to recommend its clinical use for depression.
Focal electrically administered seizure therapy (FEAST)
FEAST is a method of inducing a focal seizure using high-intensity direct current (Sackeim, 2004). The current passes from a small circular anterior electrode placed above the midpoint of the right eyebrow (right anterior orbitofrontal cortex) to a large oblong posterior electrode placed anteriorly to the right motor cortex (Sahlem et al., 2016). The effect is to induce a focal, frontal seizure as demonstrated by a regional cerebral blood flow study (Chahine et al., 2014). A feasibility study (Nahas et al., 2013) demonstrated the antidepressant efficacy of FEAST, with 8 and 5 of 16 patients reaching response or remission, respectively. A more recent study of 20 patients (Sahlem et al., 2016) demonstrated that FEAST was effective for depression, with response and remission rates of 65% and 55%, respectively. Furthermore, cognitive testing revealed no anterograde or retrograde memory side effects.
In summary, FEAST is an experimental treatment that may be as effective as ECT for depression but with significantly reduced cognitive side effects.
7. Management of major depressive disorder
It is important to bear in mind that while depression can, and does, occur in isolation, depressive symptoms and syndromes are frequently encountered alongside general medical illnesses such as cardiac diseases, neurological disorders (e.g. Parkinson’s), endocrine disorders and chronic medical conditions such as cancer and autoimmune syndromes. Within the realm of psychiatry, depression is often comorbid – overlapping with anxiety disorders, personality dysfunction, substance misuse and, in particular, alcohol misuse. Furthermore, phenomenologically, it is no different from bipolar depression, and a small proportion of patients initially diagnosed as having major depression will be rediagnosed as having bipolar depression, once they manifest manic symptoms.
However, clinically, depression is also common singularly, and in many instances, it is the only medical problem a patient is facing. Therefore, this section discusses the management of major depression that is uncomplicated by any concurrent illness.
Depressive disorders, such as major depression, are a major part of psychiatry, and yet, in the majority of cases, the management of depression begins in primary care, and this is also where the initial diagnosis is usually made.
7.1. Actions
Laying down a strong foundation is key to the management of depression, not only for the successful resolution of the current episode but for reducing the likelihood of recurrence. The Actions are therefore essential, and from the outset it is important to emphasise their benefits, for the effective management of both the current episode of depression and for preventing future episodes.
Throughout management, it is important to bear in mind that depression is a chronic, often lifelong, illness, and that while acute episodes can be successfully treated, and a reasonable level of functioning restored, the underlying vulnerability is unlikely to be rectified. In other words, acute exacerbations of the disorder (major depressive episodes) can be resolved using currently available treatments but as yet, it is not possible to cure the illness altogether. Nevertheless, resilience against depression can be enhanced, and many of the recommended Actions as part of laying down a foundation for the management of depression, likely contribute to the development of resilience through adaptive processes such as tempering and fortification (Malhi et al., 2019b). Therefore, the value of the initial Actions should not be underestimated and these should be implemented as comprehensively as possible.
A lack of restorative sleep, with early morning waking following a poor night’s sleep is characteristic of major depression, although excessive sleep at night-time and during the day with accompanying fatigue and tiredness is also common (as part of atypical depression; see classification MDcpg2015). This is because the biology of sleep is closely geared to the biology of depression (see section 3.3. ‘Circadian function’) and so the restoration of a normal sleep pattern with good quality sleep is essential and this can be achieved by adopting healthy sleep habits (see Box 4). Briefly, this involves setting appropriate times for sleeping at night-time and waking up in the morning.
Alongside sleep, the institution of a healthy diet and regular exercise are essential. These interventions have been addressed in detail in the treatments section (see ‘Lifestyles’, in section 6.1), and the principal Actions are summarised in Boxes 5 and 6. The key point to note is that in practice, exercising and dieting are challenging prospects for anyone as they require strong motivation and consistent monitoring. This means that in the context of mood disorders extra effort and resources are required and both the goals, and progress towards them, need to be reviewed regularly. On occasion, it is useful to engage the assistance of a specialist dietician or exercise physiologist to identify the most appropriate diet and exercise regime. However, straightforward and effective advice and monitoring can be provided by a practice nurse or allied health staff with knowledge of these areas.
Alongside instituting these Actions, it is important to address other contributing lifestyle factors. For example, smoking, excess intake of alcohol and substance misuse should be stopped as soon as possible or, at the very least, moderated.
Again, these are not easy habits to reform and additional assistance may be necessary from specialist drug and alcohol services. In addition, medications that lower mood should also be stopped; and this is particularly important as, in practice, it is a contributory factor that is often overlooked.
The foundation of management should also include the implementation of measures ranging from psychoeducation and psychological interventions to examining social factors and ensuring that all aspects of the illness are adequately monitored and assessed.
It is important to involve family and friends as well as the individual suffering from depression in psychoeducation because often the illness is poorly understood, and the individual’s behaviour can be negatively misconstrued. For example, a lack of motivation and drive may be seen as being lazy or simply uninterested. Also, friends and family can often help in ensuring the institution of healthy habits and routines and making sure the individual complies with medication and attends appointments.
Psychological interventions (see ‘Psychological treatments’ in section 6.1) are particularly important because from the perspective of patients they are often the preferable treatments. Formal psychological help is also increasingly and more widely available and, because of government subsidy in many countries including Australia, more readily accessible.
Pragmatically, a challenge to instituting psychological intervention can be that patients lose interest in engaging psychologically when symptoms improve with antidepressant medication (the component of treatment instituted most quickly in primary care). To circumvent this suboptimal outcome, it is critical that pharmacotherapy and psychological intervention are presented to patients as complementary components of holistic treatment of depression. A key attraction of the psychological component is that it offers an opportunity for learning new cognitive and behavioural skills to prevent future relapse and recurrence.
Other factors that should be assessed include considering the extent of social support the individual has in terms of family, friends and social networks and also in terms of having a role, purpose and place to live. Often factors in these social aspects of the individual’s life are major sources of the stressors that precipitate and maintain depression. Hence, although clinicians may not be able to address these aspects directly, acknowledgement of their importance, and advice and referral to those that can help (e.g. social worker, community mental health team) is of immense value.
In order to maintain a secure foundation, it is important to put in place measures that allow the objective assessment of depression and associated risk, and ensuring that there is regular clinical review with respect to outcomes. This can be achieved using either self-report rating scales (such as the K-10, BDI, DASS or PHQ-9), observer-rated scales (such as the MADRS or Hamilton rating scale) or by asking the patient directly.
In the context of mood disorders, it is important to assess whether a person is at risk of attempting suicide; and the key factors to note in this regard are a past history of any suicide attempt and their current mental state. Specifically, it is useful to assess the degree of hopelessness and suicidal ideation and whether they have intent and have developed a plan to commit suicide. It is important that these questions are put to the patient explicitly.
7.2. Choices
While some mild presentations of MDD may be managed successfully by only implementing the Actions outlined above, in particular the use of psychological interventions, many cases are likely to require antidepressant medication. Depending on locale, more than 20 antidepressant formulations, belonging to more than a dozen pharmacological classes, may be available for the treatment of major depression (see Table 10). The vast majority of these act via monoaminergic modulation and differ mainly in terms of tolerability and only somewhat in efficacy. Nevertheless, these differences are important and are useful for tailoring choice to the clinical profile of the depressed individual. Recent network meta-analyses (Cipriani et al., 2009, 2016, 2018) have shown that all antidepressants are effective (compared with placebo) and that some are more effective than others (head to head studies). Therefore, based on both evidence and clinical experience there are seven Choice antidepressants. It is noteworthy that each antidepressant is from a different class (see Table 10). The Choice antidepressants are shown in Figure 26 and they are positioned along an axis that reflects their differential tolerability and efficacy, comprising two groups of three antidepressants and with amitriptyline on its own. The two groups of three comprise escitalopram, vortioxetine and agomelatine (EVA) in one group, and venlafaxine, mirtazapine and bupropion (VMB) in the other. There is another subtle gradient of effectiveness favouring those agents that are listed higher. However, it is important to note that these differences are marginal and not as significant as the impact of tailoring choice to the patient’s unique profile. Nevertheless, it provides some indication of putative preferences (Figure 26).
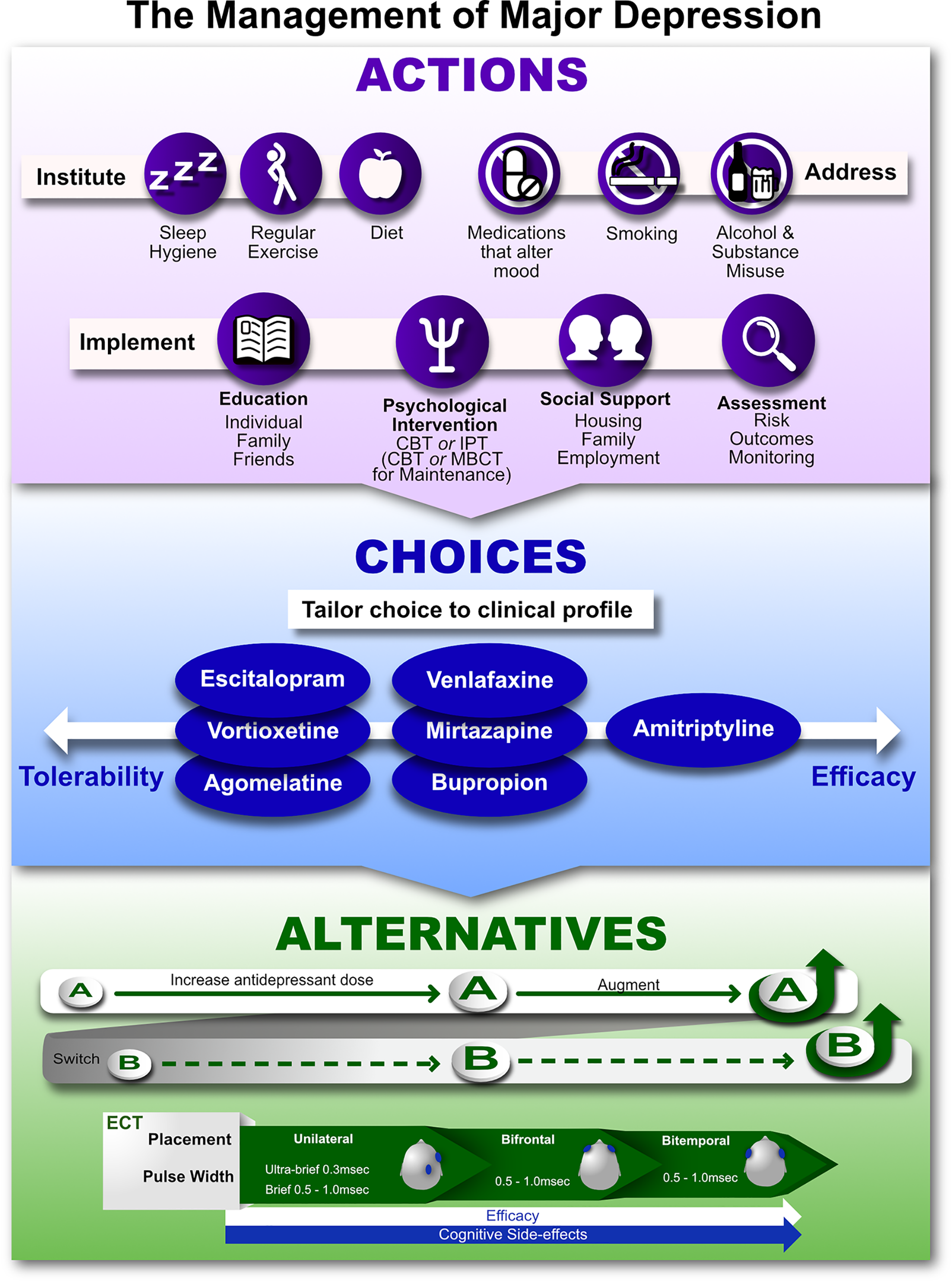
The management of major depression.
In practice, pharmacotherapy can commence with any Choice antidepressant and the selection will ultimately depend upon a number of additional practical and clinical factors – such as availability of a particular agent and whether any antidepressants have been used previously, and if so, to what extent were they effective. The clinical profile is also a key determinant and should be used to gauge treatment selection (see Table 9). For this it is useful to consider the symptom profile and perhaps draw on the ACE model to determine if any particular domain of symptoms is more prominent and in need of specific targeting. Symptom severity is also important and, in conjunction with the degree of functional impairment caused by the clinical syndrome, can be used to determine the speed with which to act. Finally, if the clinical features of a particular subtype are present this can be useful in determining antidepressant selection.
If a Choice antidepressant is ineffective then an Alternative can be trialled, and any number of the Choice medications can be considered provided there is sufficient clinical indication for their use. However, in instances where a partial response is achieved (see section 9, ‘Response to treatment’) it may be useful to consider Alternative strategies before switching medication, and here there are a number of options.
7.3. Alternatives
After suitable Choice antidepressants have been trialled any of the remaining agents can be prescribed using the same principle of tailoring treatment where possible to subtypes, symptoms and severity. The initial response to pharmacotherapy can result in the resolution of symptoms, a partial response (<50% reduction is symptom severity from baseline) or no response whatsoever. It is noteworthy that in some cases the response is delayed and so may not be evident immediately.
In cases where there is no response or only a partial response, three strategies are available. The first is to increase the dose of the antidepressant to the maximum specified in the Approved Prescribing Information. This generally enhances both pharmacokinetic and pharmacodynamic properties and ensures an adequate level is being achieved at target receptors (for further guidance, see Recommendation Box 6). The second strategy is to augment the actions of the antidepressant already being prescribed (the primary antidepressant) by adding a suitable agent. This strategy is particularly useful if a partial response has already been achieved indicating that the primary antidepressant is having some effect. The third strategy is to switch to a different antidepressant and while this can be done at any stage it is probably most useful if there is no, or minimal (<20% improvement) response to the primary antidepressant or once an increased dose and augmentation have been trialled.
Therefore, when faced with partial or no response to antidepressant medication any of the three strategies may be considered next. However, partial response would favour trialling an increase in dose (but not above the recommended effective dose) and or augmentation before switching whereas no response provides less of an indication of what is likely to be effective. Nevertheless, all of these strategies may still be effective as the response to the primary antidepressant may be delayed and an increase in dose or augmentation with an additional agent may produce improvement. If, however, these strategies are ineffective then a switch to another antidepressant is necessary, and once this is achieved the same strategies of increasing the dose and adding an augmenting agent can be employed once again.
In practice, it is often the case that more than one antidepressant trial is needed to achieve a satisfactory antidepressant response, namely, one that leads to remission and functional recovery. And in some cases, several antidepressants may need to be trialled. Suboptimal response, which includes no response, or a partial response is a common problem and therefore it is discussed in further detail separately (see section 9, ‘Response to treatment’).
Another Alternative for managing major depression is electroconvulsive therapy (ECT). While ECT is usually only considered as an option once pharmacotherapy has been trialled (as shown in Figure 26) it is a Choice treatment in a number of specific instances, such as when depression is severe, or accompanied by suicidal behaviour, or it features marked psychotic symptoms, or when there is an imminent risk of becoming severely medically unwell because of not eating or drinking, or when there are marked psychomotor symptoms. It should also be regarded as a Choice treatment when patients have previously responded well to ECT.
In most cases ECT is usually considered once a number of antidepressant trials have not been successful. In this context ECT is an effective treatment for depression and there are options regarding the mode of delivery (electrode placement) and the amount of current that is applied (pulse width). It is important to note that with increasing efficacy ECT is prone to cause greater cognitive side effects and therefore, as shown in Figure 26, it is advisable to begin with unilateral placement, and utilising as brief a pulse width as possible and then to progress, as need be, to bifrontal and then bitemporal placement with an increase in the amount of current being applied. The administration of ECT, its indications and potential complications, are discussed in greater detail in the Treatments section (see section ‘ECT’, in section 6.3).

MDD: major depressive disorder; CBR: consensus-based recommendation; EBR: evidence-based recommendation; CBT: cognitive behavioural therapy; IPT: interpersonal therapy.
Two contextual factors regarding psychological treatments are important in the management of acute MDD: patients generally prefer psychological intervention over antidepressant medication; and psychological interventions are becoming more accessible due to increased numbers of trained clinicians, online self-management programs and telehealth platforms (see section ‘Digital therapies’ within section 6.1. ‘Actions’).

MDD: major depressive disorder; CBR: consensus-based recommendation; CBT: cognitive behavioural therapy; MBCT: mindfulness-based cognitive therapy; EBR: evidence-based recommendation.
Note: Treatments aiming to prevent relapse are more effective if full remission of the initial episode is achieved. The choice of antidepressant dose is also determined by additional factors such as prior illness severity and response to treatment, comorbid disorders and medication tolerance.
This is particularly important if a recurrent pattern of illness has been established.
7.4. Maintenance
Medication
A key practical issue is how long to continue prescribing medication after a clinical response has been achieved. The MDcpg2015 advocated maintaining treatment for at least 6 months and up to 1 year (Recommendation 7.4). A minimum duration of 6 months remains a reasonable recommendation, supported by a recent meta-analysis. This showed that active treatment remained significantly superior to placebo with consistent effect sizes of between 0.27 and 0.34 after 8, 12, 16, 20 and 24 weeks (Henssler et al., 2018).
However, beyond 6 months there is less data on the efficacy of maintenance therapy. Sim et al. (2016) identified only 35 reports involving treatment with an antidepressant for more than 1 year. Overall, long-term controlled trials showed significantly reduced recurrences with antidepressants versus placebo treatment (RR = 2.03, CI = [1.80, 2.28], p < 0.0001) with a relatively low number needed-to-treat (NNT = 3.8, CI = [3.3, 4.6]).
Of note, there is no evidence regarding maintaining antidepressant treatment beyond 3 years, and there is also no consistent evidence favouring any antidepressant class in maintenance treatment. For example, Henssler et al. (2018) reported higher effectiveness for TCAs and SNRIs at 8 and 12 weeks but Sim et al. (2016) reported similar efficacy for all antidepressant classes. Evidence is also somewhat limited in relation to dosing of antidepressants during maintenance therapy. The clearest study examined the use of older tricyclic antidepressants in maintenance therapy and clearly showed that lowering dosing in maintenance therapy after acute therapy was associated with a higher risk of relapse (Frank et al., 1993).
Therefore, generally speaking, pharmacotherapy should be continued beyond 6 months and ideally for more than a year for those with recurrent depressive episodes. Furthermore, the dose utilised for maintenance should be the same as that used in acute therapy.
Similar to pharmacotherapy, for ECT there are few randomised trials of maintenance treatment in depression, however a recent meta-analysis reported that ECT plus pharmacotherapy was associated with fewer relapses and recurrence than pharmacotherapy alone at 6 months and 1 year (Elias et al., 2018). This suggests that continuing and maintaining treatment is of benefit, at least for the first 12 months.
Psychological treatments
The MDcpg2015 recommended psychotherapies (particularly CBT and MBCT; see section ‘Psychological treatments’ within section 6.1. ‘Actions’) to be delivered in the maintenance phase of MDD for prevention of relapse and/or recurrence. A recent systematic review and meta-analysis focusing specifically on the effects of interventions (compared to controls) delivered when MDD patients were in remission concluded, (1) CBT is superior in protecting against relapse, (2) MBCT is also superior in protecting against relapse, but only among patients with 3 or more episodes (Zhang et al., 2018). As mentioned in the MDcpg2015, a meta-analysis and meta-regression (Biesheuvel-Leliefeld et al., 2015) found preventive psychological interventions (Cognitive Therapy, IPT and MBCT) across 25 identified studies were superior to treatment as usual (NNT = 5), and to a lesser extent antidepressant medication (NNT = 13): there was a signal that prevention was more powerful when the treatment was preceded by acute-phase psychological treatment, but no signal of differential efficacy between the three types of treatment. However, one large trial comparing MBCT with maintenance antidepressants reported no difference in efficacy (Kuyken et al., 2015), and a meta-analysis concluded that psychosocial intervention yielded inconsistent or inconclusive results (Sim et al., 2016). However, Sim and colleagues noted that most trials found evidence of long-term benefit.
In summary, preventive treatments including CBT and MBCT applied during the maintenance phase (either as continuation of acute phase treatment or commenced de novo) are recommended (Bockting et al., 2015).
As might be expected given their skill-based techniques, evidence has strengthened that psychological interventions delivered during the acute phase also have benefits for the maintenance phase: effects of psychological interventions endure at 6 and 12 months (Cuijpers, 2017), and there is some evidence for maintenance of benefits at 2 years (Sim et al., 2016) after treatment has terminated. A recent network meta-analysis, reported that psychological treatment may be more effective than pharmacotherapy in the longer term (Cuijpers et al., 2020), consistent with a prior meta-analysis comparing CBT with continuing pharmacotherapy (Cuijpers et al., 2013; McPherson and Hengartner, 2019).
The availability of trained therapists for the provision of preventative psychological interventions is a concern leading to increased attention to online and other digital technologies. Initial positive findings for an internet-based relapse-prevention treatment for MDD (Hollandare et al., 2013) have not been replicated (Klein et al., 2018), but enthusiasm for the potential of digital therapies for maintenance treatment remains strong.
As with the acute treatment of MDD, combining psychosocial interventions and pharmacotherapy may have advantages in the maintenance phase. Based on their meta-analysis, Karyotaki et al. (2016) reported that combined maintenance psychological treatment with antidepressants resulted in better sustained treatment response compared to antidepressants alone at 6 months or longer post randomisation. A meta-analysis investigating sequential integration (as opposed to simultaneous integration) of psychological treatment with pharmacotherapy (introduction of CBT in the residual phase to augment antidepressants delivered in the acute phase) also reported a relative advantage of the combination in preventing relapse/recurrence (Guidi et al., 2016). Specifically, meta-analysis found that patients who had antidepressants tapered and discontinued were likely to benefit from the addition of CBT. This finding is reinforced by a recent study showing that adding preventive cognitive therapy to antidepressant discontinuation resulted in a 41% relative risk reduction in the risk of relapse or recurrence (Bockting et al., 2015).
7.5. Withdrawal of antidepressants
Stopping antidepressants
It is generally recommended that patients should stop their antidepressant medication after they have been in remission for around 9 months to a year (Malhi et al., 2015). Following ceasing medication, many patients will remain depression-free, but some may have a depressive relapse and up to 40% may experience discontinuation or withdrawal symptoms (diagnostic criteria have been proposed for withdrawal, Chouinard and Chouinard, 2015), however, we prefer to focus on discontinuation and withdrawal symptoms as not all patients will meet the criteria, but will nevertheless be distressed by their symptoms). Because of this, it has been recommended that antidepressants should be withdrawn gradually, with doses tapered down over an extended period of time.
Rates of discontinuation/withdrawal symptoms
The precise rate, severity and duration of antidepressant Discontinuation and Withdrawal Symptoms (DaWS) is not clear, as there have been few systematic studies of this phenomenon (especially after longer term treatment) and antidepressants differ in their propensity to cause such symptoms, with some (e.g. paroxetine and venlafaxine) having higher rates than others. Initially, DaWS were thought to occur with only a small proportion of patients with mild and self-limiting symptoms. However, we now recognise DaWS are common, can be severe, and, in some patients, they can persist for a significant period of time. This has led to NICE modifying their recommendations (Iacobucci, 2019) regarding stopping antidepressants. However, there is ongoing debate about the evidence base that forms the basis of such clinical practice guidelines.
Early studies based on short-term (up to 12 weeks) randomised controlled trials reported that up to a quarter of patients could experience DaWS. For example, Baldwin et al. (2007) reported prevalence rates between 7% and 23% of patients undergoing antidepressant withdrawal. However, a recent systematic review reported that up to 56% of patients will experience DaWS, and of these, almost half (46%; based on four surveys) will regard their symptoms to be severe (Davies and Read, 2019). The findings of this influential review (the data for one of the studies comes from a survey collected for the All-Party Parliamentary Group for Prescribed Drug Dependence) are considered controversial and have been challenged, the main criticism being that the survey relied on internet-based data and that, in at least one survey, it specifically recruited participants who had experienced antidepressant withdrawal symptoms, while more rigorous and controlled studies were not included in the review.
While the exact rates of DaWS are not known, clinical experience, coupled with the published literature, suggests that up to 40% of patients may be affected, causing significant distress. Notably, in some patients, symptoms can persist for months rather than weeks.
Notably, DaWS may emerge following the cessation of all classes of antidepressants, other than the melatoninergic agent, agomelatine. The risk of DaWS is greatest with higher doses of antidepressants and the longer the duration of treatment. However, symptoms are usually transient and mild, and resolve with antidepressant reinstatement. DaWS are diverse and variably expressed; the acronym FINISH –
Symptoms of discontinuation and withdrawal (adapted from Haddad and Anderson, 2007, and Jha et al., 2018).

Risk of discontinuation and withdrawal symptoms
While all patients are at risk of developing DaWS, the risk is greater among patients who have had higher doses than the minimum effective dose of the antidepressant (Haddad and Anderson, 2007), or have experienced withdrawal symptoms after missing a dose(s) (Harvey and Slabbert, 2014; Ruhe et al., 2019), or had a prior experience of DaWS after previously attempting to stop the antidepressant.
Strategies for dose reduction
As DaWS occur following abrupt discontinuation of the antidepressant, it is recommended that the dose of antidepressant be reduced slowly. However, there is insufficient evidence to suggest the best regime for this. A clinical trial found that tapering over 2 weeks had no benefit over tapering over a few days (Tint et al., 2008), but this, in practice, is still considered a short-term timeframe. It has recently been suggested that antidepressants should be tapered down hyperbolically (by lowering the dose by smaller amounts over time) in the same manner as benzodiazepines (Horowitz and Taylor, 2019), which is further supported by a Dutch taskforce (Ruhe et al., 2019). However, reducing the antidepressant in such a way is impractical as current preparations of antidepressant do not allow for the dose to be reduced by such small decrements.
We suggest that, for those with risk factors for severe DaWS, the first step is to reduce the dose to the minimal effective dose. Following this, the dose should be halved, and after a week, the dose should be reduced more slowly in small decrements (allowing 2 weeks for each dose reduction), according to how the tablet can be divided. Unfortunately, this is not feasible with medications that are encapsulated (e.g. venlafaxine and duloxetine).
Differentiating discontinuation and withdrawal symptoms from relapse
A critical clinical issue is working out whether, after stopping medication, the patient is experiencing DaWS or having a depressive relapse.
Discontinuation and withdrawal symptoms typically start a few days after stopping the antidepressant and affect different systems (see Table 13). They are not the typical symptoms of a depressive relapse (loss of interest, anhedonia, loss of self-worth or cognitive changes) and they respond quickly to the re-introduction of the antidepressant.
Precision medicine
Despite ongoing enthusiasm for the concept of precision medicine, to date, it is largely industry-driven research that has been the mainstay, and there is ongoing need for independent replication of findings despite positive meta-analytic data suggesting remission rates can be doubled with genetically guided antidepressant prescribing (Bousman et al., 2018; Bousman and Eyre, 2020). At this stage – other than HLA testing of carbamazepine among patients of Asian ethnicity (Shnayder et al., 2020) – precision medicine in psychiatry remains an area of much promise, but one in need of firmer independent RCT data before it can be adopted widely (Thompson et al., 2015). Inclusion of environmental factors, epigenetics through methylation status, and machine learning models may also enhance the clinical utility of the field (Musker and Wong, 2019).

CBR: consensus-based recommendation; AD: antidepressants; EBR: evidence-based recommendation; MAOI: monoamine oxidase inhibitor.
7.6. Mixed anxiety and depression
The recognition that depression and anxiety symptoms often co-occur dates back to the beginning of psychiatric nosology. In the modern era, their separation began in the 1960s with drugs being marketed as ‘specific’ antidepressants (e.g. tricyclics) or anxiolytics (e.g. benzodiazepines) and was compounded during the drafting of DSM-III when different advisory committees for anxiety disorders and mood disorders were established (Möller et al., 2016). In 1990 ICD-10 largely followed suit but retained a mixed anxiety and depressive disorder (MADD) diagnosis in the anxiety disorder section. ICD-11 has retained this diagnosis but changed its name to mixed depressive and anxiety disorder and moved it to the mood disorders section. DSM-5 has included a new ‘with anxious distress’ specifier in the mood disorders section (Mulder et al., 2019).
The prevalence of these diagnoses remains unknown. While there is little dispute that a substantial number of individuals in the community suffer from comorbid symptoms of depression and anxiety, data on this prevalence is inconsistent due to the discordant criteria between the ICD-11 and DSM-5 diagnoses. The prevalence of the DSM-5 anxious distress specifier in one study of depressed patients was 54% (Gaspersz et al., 2017).
The clinical utility of the MADD diagnosis and DSM-5 anxious distress specifier is also unknown. One study reported the anxious distress specifier outperformed comorbid anxiety in predicting chronicity, time to remission and functional disability (Gaspersz et al., 2017). However, there is currently no evidence that either diagnosis helps in planning treatment.
Thus, matters have reached an unsatisfactory compromise. The fabricated distinction between anxiety and depression ignores the fact that the most common form of mood disorder is mixed anxiety and depression. Both current diagnostic systems allow comorbid diagnoses of depression and anxiety in ICD-11 and anxious distress specifier in DSM-5. Neither have an evidence base for their validity or clinical utility. Both appear to be common and we urgently need research on their reliability, validity and actual evidence-based treatments. Whether either has validity beyond the identification of comorbid anxiety is unknown. It is not surprising that most clinicians do not appear to use these diagnoses.
In summary, anxiety commonly co-occurs in depression but the specific treatment implications of this are unknown. Both conditions respond to similar psychological and pharmacotherapeutic approaches and therefore combining these treatments may be especially relevant for this group of patients. The mixed presentation can be regarded as an overlap of two diagnoses (anxiety and depression), which can therefore be treated as separate disorders. However, there is no evidence to inform this approach. Practically, it is possible that the use of anxiolytics and atypical antipsychotics to specifically address the co-occurring anxiety may be helpful but again there is little evidence to guide practice. The approach to treatment can be broadly predicated on the assumption that treatment is addressing either two phenotypic presentations (i.e. anxiety and depression co-occurring) or alternatively that there is a single distinct entity that consists of mixed anxiety and depression. Clearly further research is needed to study whether any treatments are better suited to the treatment of depressed patients who also have significant anxiety symptoms.
8. Management of bipolar disorder
8.1. Mania
Mania defines bipolar disorder. And clinically, florid mania is arguably the most distinctive of all psychiatric disorders. Studies investigating the treatment of mania have often been confounded by the inclusion of patients experiencing mixed mood states. In the MDcpg2020 both the diagnosis and management of mixed states is dealt with separately, and so the advice in this section mainly concerns mania.
Note, the term hypomania is often used to describe a manic syndrome that is of less severity than mania. This descriptor has not been used here because it is a product of the arbitrary subtyping of bipolar disorder (see section 2. ‘Classification’) and as such lacks specificity. Instead, mania is used to describe all manic syndromes of varying severity in which there are no concurrent depressive (mixed) symptoms. Further specificity can be achieved by noting whether there are additional symptoms such as those of psychosis, or agitation and the degree to which insight is compromised. These additional features are important when considering management as they will inform which strategies to employ. For example, a loss of insight may require selecting medications that can be administered other than orally. Severe psychosis, agitation and behavioural disturbances may also require additional safety considerations both for the individual and those caring for them.
Actions
The management of mania is distinctly different to the management of the depressive phase of bipolar disorder. In patients experiencing severe mania, safety is paramount, and a suitable setting is critical (see MDcpg2015).
Once a diagnosis of mania has been established, management begins with a number of Actions (see Figure 27), most important of which is to ensure safety (e.g. evaluate risk of self-harm) and to assess for the possibility of any ongoing or future risks (impacting, for example, the individual’s reputation, their relationships and finances). It is also important to assess for the possibility of psychosis and cognitive changes including any moderation of insight. Following a detailed assessment of mental state and behaviour it is important to ensure that there is close clinical supervision including ongoing regular monitoring. For severe mania, assessment is best achieved in hospital, but even for moderate or mild mania it is important to implement structure and routine and lower activity levels. It is also necessary to review all prescribed treatment and any illicit substances the individual may be taking and stop any medications that may be contributing to an elevation in mood, such as antidepressants or stimulants. Alcohol and substance misuse must be stopped and withdrawn appropriately.
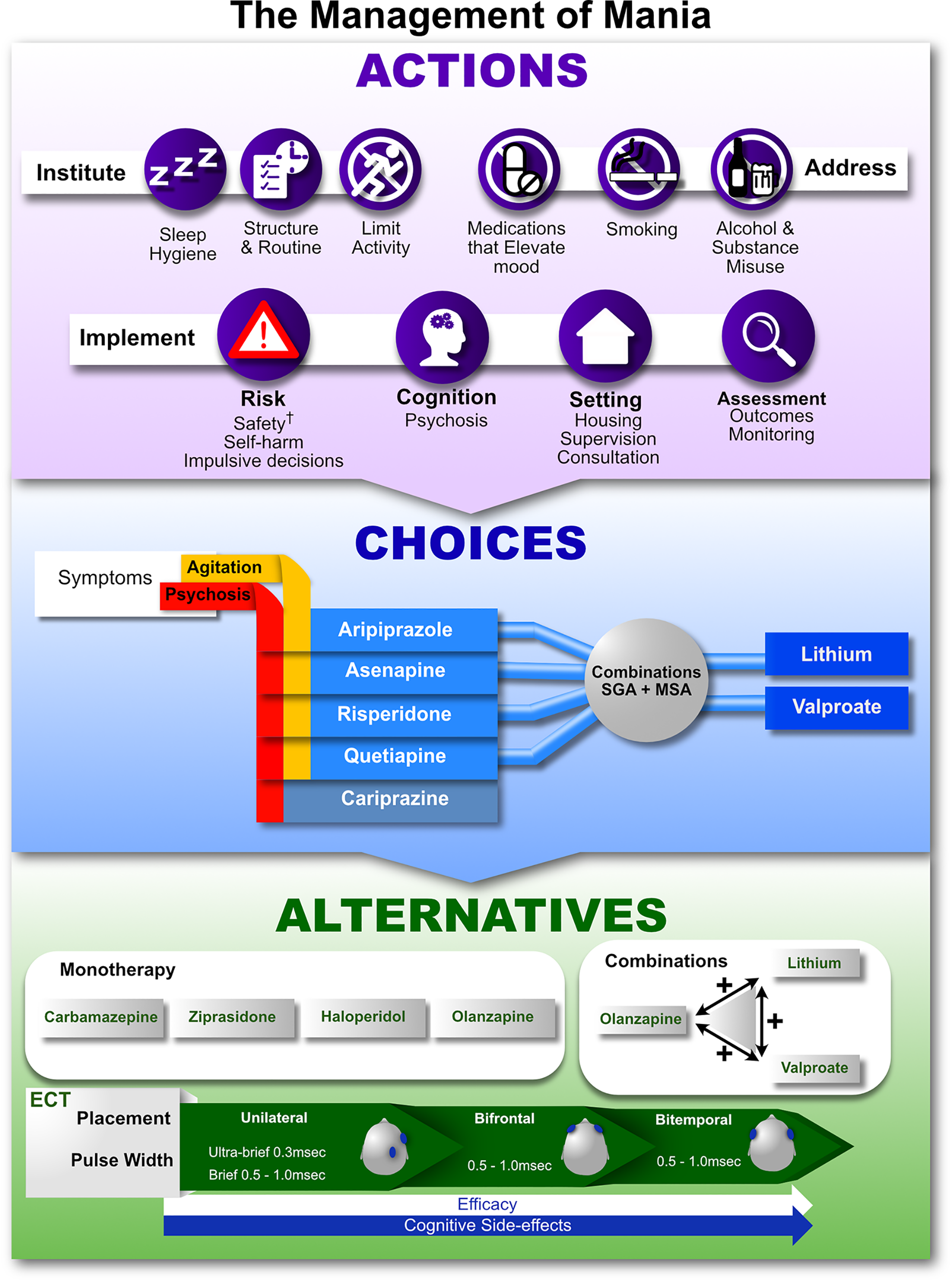
The management of mania.
Pharmacotherapy for agitation.

Sublingual.
Also available as orally disintegrating formulation.
Also available in wafer formulation.
Alternatives
Monotherapy should be given preference over combinations where possible, because of better compliance and fewer adverse effects. The monotherapeutic Alternative agents with efficacy in mania range from the anticonvulsant carbamazepine through to the atypical antipsychotic olanzapine and also include ziprasidone and haloperidol, providing a variety of means of administering these medications. If monotherapy is unsuccessful or treatment is being added to existing mood-stabilising agents, combinations can be trialled involving lithium, valproate and olanzapine, all of which can be combined as dyads (OL + Li, OL+VA, Li + VA). ECT is also a useful Alternative to bear in mind and though it is uncommonly used to treat mania, it is an efficacious strategy especially when urgent treatment is required (OL = olanzapine; VA = valproate; Li = lithium).
As with depressive disorders, sleep hygiene is essential, however in mania the main problem is sleeping less than usual and the individual does not experience a need or desire for sleep; hence initially, it may be necessary to aid sleep with sedation. In severe mania, insight is often compromised, and individuals are unable to attend or concentrate, which makes psychological interventions impractical. Therefore, from a psychological perspective, the inpatient treatment of mania should emphasise relationship building with the patient, and psychoeducation with the family (Chen et al., 2019b). Mania is typically a crisis for individuals and their families, and therefore demonstrating empathy, mitigating carer burnout, and instilling hope are key goals. Thus, once the foundation has been laid, clinical management usually moves to pharmacotherapy fairly swiftly where a number of choices have to be made.
Choices
Mania is the core feature of the syndrome and for this, second-generation antipsychotics and/or mood stabilisers can be used either as monotherapy or in combination. Clinically, the antimanic effects of mood stabilisers (lithium and valproate) take time to materialise, and so the SGAs are usually administered first, either as monotherapy or in combination with mood-stabilising agents. The SGAs also have the advantage of addressing additional symptoms that are often present in severe mania, namely agitation and psychosis (see Table 14, in legend of Figure 27). In this regard, it is important to bear in mind that all the SGAs have antipsychotic and sedating properties and will therefore assist with mania and psychosis and that the majority of these agents will also help with agitation. Thus, in many cases, an SGA is the preferred choice. However, it is important to note that immediately following amelioration of the acute phase of the syndrome, the aim will be to maintain mood stability and to prevent recurrences, and in this regard the mood stabilisers are likely to be more effective and better tolerated. Therefore, in mild or moderate mania, where there are no psychotic symptoms or concerns about agitation, mood-stabilising agents (MSAs) can be prescribed from the outset as monotherapy provided the patient is accepting of treatment.
In addition, as noted previously, chronobiological principles can be usefully applied in the inpatient setting. At a minimum, these could include instituting robust 24-hour routines (waking at the same time each day, minimising night-time disturbances), and minimising ambient light exposure at night (see Box 4).
Alternatives
If the Choice agents are unavailable, unsuitable or have already been trialled, then there are a number of Alternative options. Here again, monotherapy and combinations of medications are possible and once again, carbamazepine (a mood stabiliser) is marginally more favourable than the antipsychotics ziprasidone, haloperidol and olanzapine, primarily because it is less likely to cause long-term side-effects. However, HLA genetic testing in patients of Asian ancestry is advisable prior to initiating carbamazepine (SJS risk) (Phillips et al., 2018).
As with Choice agents, combinations of optional agents can also be used; for instance, lithium in combination with valproate is an effective option and either of these MSAs can be combined with SGAs, even those with less favourable long-term tolerability such as olanzapine. The rationale for this is that once the acute episode of mania resolves and management shifts to continuation and maintenance therapy these medications will be revised and therapies more suitable for long-term management will be instituted (see section 8.4, ‘Maintenance’). In addition to these many considerations of SGAs and MSAs, adjunctive treatments can also be prescribed, especially when symptoms are severe or there is marked agitation and/or psychosis. These are shown in Table 15.
Finally, as with major depression and bipolar depression, ECT is also a useful and effective option although it is seldom needed or used (Weiner, 2017). However, there is little data available to assist clinicians in choosing optimal electrode placement, with only one randomised controlled trial comparing bifrontal and bitemporal placement (Hiremani et al., 2008). Both placements were equally effective but the response to bifrontal was more rapid. A recent retrospective analysis (Wong, 2019) reported outcomes in mania of right unilateral brief pulse width, right unilateral ultra-brief pulse width, bifrontal and bitemporal placements. All treatments were equally effective but right unilateral ultra-brief was associated with significantly less cognitive impairment. Several case series have reported effective treatment of mania with right unilateral ultra-brief ECT (Anand, 2016; Elias et al., 2016; Sidorov and Mayur, 2017). Thus, based on current data, unilateral brief pulse and ultra-brief pulse and bifrontal ECT are as effective as bitemporal ECT, but are less likely to cause cognitive side-effects and so the initial choice should be one of these three options where possible. However, where an urgent response is needed, bifrontal ECT may be given priority as it may achieve a more rapid response.
8.2. Bipolar depression
As discussed earlier (see section 2. ‘Classification’) this guideline does not divide bipolar disorder into subtypes. This does not impact treatment because clinical trials have rarely analysed subtypes separately. Indeed, only a few pooled analyses have done so.
To determine how to treat bipolar depression, first develop a detailed profile of the clinical features of the depressive syndrome (e.g. severity, subtypes and ACE domains; see ‘ACE model’ in section 2.3.) and characterise associated illness factors (e.g. course of illness, duration of episodes, psychological and social features of the episode). This process is similar to the formulation of MDD. Treatment can then be tailored accordingly.
The management of bipolar depression is inherently difficult because of the ever-present risk of inducing manic symptoms and creating either a mixed state or triggering an episode of mania. This is most likely to occur with conventional antidepressants when prescribed as monotherapy. And this type of iatrogenic switch into mania/mixed symptoms is also referred to as a treatment-emergent affective switch (TEAS). In practice, the risk of this occurring is relatively small, but it can be further minimised if alongside antidepressant medication an antimanic medication is prescribed (e.g. mood-stabilising agent, or second-generation antipsychotic). Hence, in the MDcpg2020, antidepressant monotherapy (with conventional antidepressants) is not advocated in the treatment of bipolar depression. However, clinically, where manic symptoms have only ever been mild and intermittent, antidepressants may sometimes be needed and these can be prescribed if necessary. But if a TEAS occurs, then antidepressant medication should be stopped immediately (Figure 28).
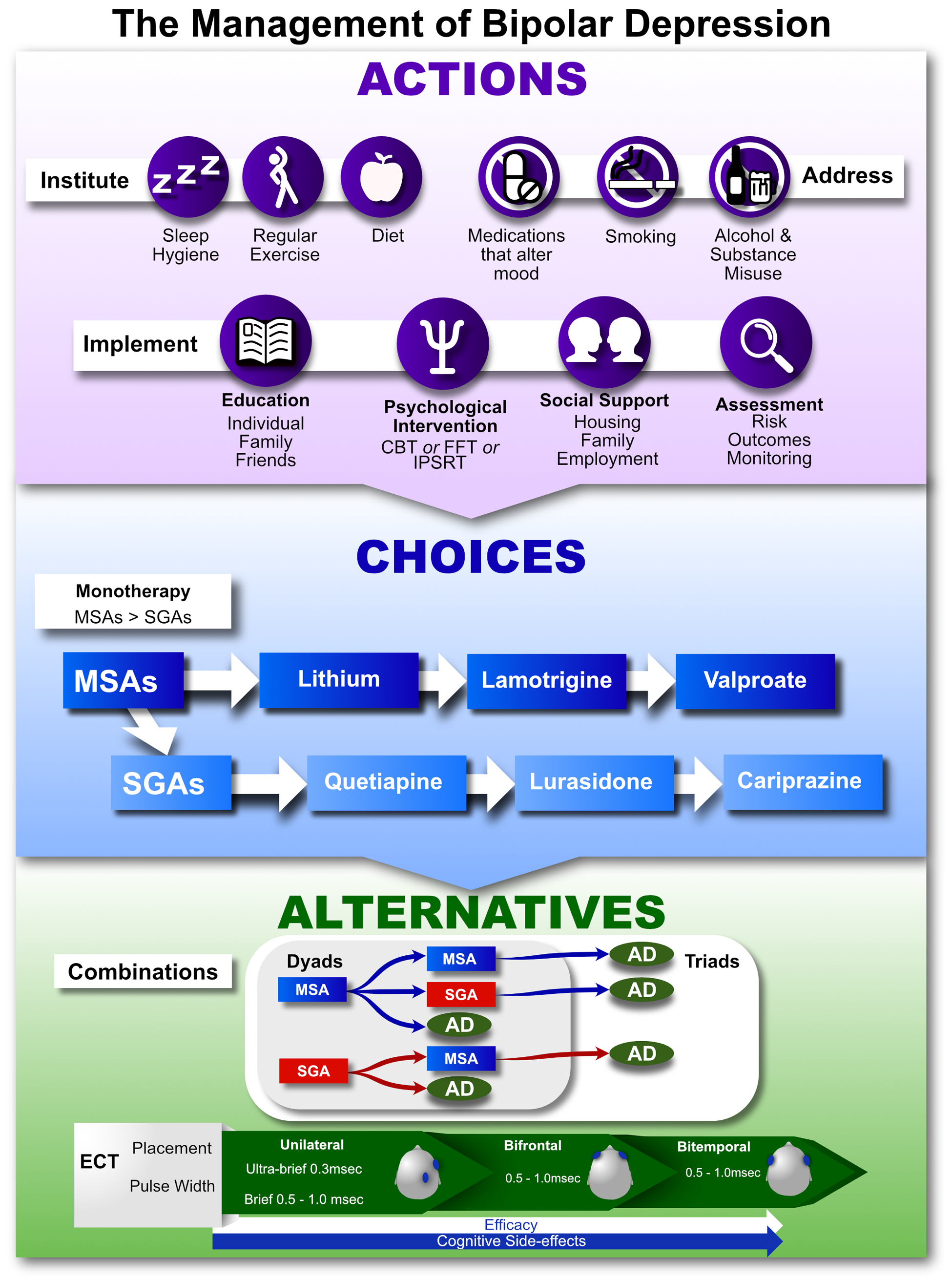
The management of bipolar depression.
Actions
Similar to the management of major depression, the management of bipolar depression begins with Actions that include the institution of regular sleep and exercise and the adoption of a healthy diet (see ‘Lifestyles’ in section 6.1. and Boxes 3–8 for details).
The restoration of sleep is important in the management of depression (see sections 3.3. ‘Circadian function’ and ‘Chronobiological treatments’ in section 6.1.) but especially so when tackling bipolar depression, because of the significant role that chronobiological mechanisms are thought to play in the pathogenesis of bipolar disorder (Gershon et al., 2019). Healthy diet and exercise also help institute routines and normalise sleep (e.g. avoiding caffeine at night-time and ensuring healthy routines by expending excess energy). In addition, it is essential that medications that can lower mood are withdrawn and that unhealthy lifestyle habits are moderated (e.g. reducing alcohol intake, avoiding substance misuse and stopping smoking – using, for example, nicotine replacement therapy).
At the same time, it is important to provide suitable psychoeducation both to the individual and where relevant, to family and friends. This is particularly important if the illness is new and the patient is treatment naïve. It is important to inform patients that the treatment of bipolar depression requires a multipronged approach in which, in addition to instituting Actions that normalise biological rhythms and metabolism, and addressing substances that alter mood, it is necessary to engage with psychological treatment and examine sources of stress, such as financial and relationship pressures. Where necessary and available, assistance with special support may need to be provided, for example, by engaging a social worker or by referring to social services (e.g. housing or employment).
Key among the Actions is the implementation of psychological treatment (CBT, FFT or IPSRT, see section 6. ‘Treatments’) and referral to a specialist should be made as soon as is practicable. Note, a suitable assessment and formulation should provide an indication of what kind of therapy is most likely to be of benefit. In instances where this is not immediately evident, an in-depth psychological assessment may be necessary. In practice, referral for psychological input regarding the management of an episode of bipolar depression can be a useful starting point for the institution of long-term psychological interventions for relapse prevention (see below).
Finally, assessment measures should also be put in place at the beginning of management to ensure the illness and outcomes of treatment are adequately monitored and any associated risk, such as that of suicide is regularly assessed.
It is important to establish that monitoring symptoms, treatment and outcome is an essential component of management and that engagement with formal assessment is a key component of laying a solid foundation for subsequent treatment.
Choices
The treatment of bipolar depression often takes weeks and months and so long-term tolerability is an important consideration, especially given that once the acute depressive episode resolves, management transitions to maintenance and prophylaxis. Hence, long-term mood stabilisation is the aim, and for this reason mood-stabilising agents are preferable to second-generation antipsychotics, which carry the risk of metabolic syndrome. For the acute resolution of symptoms agents from both classes have similar efficacy, however SGAs may be slightly faster (possibly because of easier administration) and within the two classes of agents there is a slight gradient of efficacy from lithium to lamotrigine to valproate and from quetiapine to lurasidone to cariprazine. Clearly, a number of monotherapy Choices can be trialled before selecting agents from the many other options available.
Alternatives
Before considering combinations of mood-stabilising agents and second-generation antipsychotics it is important to ensure that each medication has been prescribed optimally. Combination therapy has been found to be effective in the management of bipolar depression and both combinations of two medications (dyads) and three medications (triads) can be trialled.
Combination dyads can be achieved by adding an MSA to an MSA, an SGA to an MSA, or an AD to either an MSA or an SGA. The two combinations that do not include an AD can be converted to ‘triple’ therapy by adding an AD creating an MSA + MSA + AD combination and an MSA + SGA + AD combination (see Figure 28). Clearly such combinations need to be constructed gradually and step-by-step while monitoring for interactions and side-effects.
Further options include treatments that have some evidence and those that are experimental (see Table 15).
Further options for the treatment of acute bipolar depression.

Finally, if pharmacotherapy is insufficient or psychotic symptoms are present, then ECT is an option and this can be administered as it is for major depression; and titrating for efficacy against side effects, and graduating as necessary from unilateral (ultrabrief and brief) through to bifrontal or bitemporal.
Combinations
Where possible, monotherapy should be the goal of pharmacotherapy. However, in the management of acute bipolar depression, combinations are often necessary. In practice, given the number of monotherapy Choices, several courses of monotherapy can be trialled before combinations need to be considered. However, combinations may be considered much earlier as an Alternative if partial response has been achieved with a Choice monotherapy agent. In these instances, the addition of another MSA or SGA would be the first step towards a double-therapy combination, or alternatively an antidepressant can be added to either an MSA or an SGA to achieve the same. On occasion, triple-therapy may be needed (two MSAs + an antidepressant, or an MSA + an SGA + an antidepressant, see above). It is important to note that prior to embarking upon any combination medication regimen, it is essential to ensure that any chosen medication that is already being prescribed is optimised by checking compliance and, where possible, measuring blood levels.
Detailed assessment and implications for treatment
While the bipolar I versus II distinction that is based primarily on the severity of manic symptoms has been used to guide treatment for many years, we argue that a more nuanced approach should be adopted which draws on a range of additional clinical factors. Below we set out the factors which should be assessed and their implications for treatment.
Severity and subtype of depression
No trials have specifically examined the utility of different pharmacological agents depending on subtype of bipolar depression (e.g. atypical or melancholic). Large trials have examined differential responses to different antidepressants in unipolar depression but found no significant interaction between subtype and treatment (Uher et al., 2011). However, it is argued that DSM melancholia identifies a large group of patients and is not sufficiently specific (Parker et al., 2017). Extremely severe, suicidal or melancholic (with psychomotor features) depression may be a presentation in which venlafaxine, or other SNRIs or TCAs, are appropriate despite the clear link between these agents and an increased risk of TEAS in patients with mood disorders (Post et al., 2006). If used, there should be very close monitoring for early signs of TEAS and these antidepressants should not be used without co-prescription of an adequate dose of an agent with evidence of antimanic effect (Parker et al., 2019).
Characterisation of past episodes of mood elevation
Moving away from the bipolar I/II dichotomy allows for assessment of past episodes of mania in several dimensions. This then influences treatment decisions when treatment of the depressive episode is being considered. We suggest the following:
If episodes have been of brief duration and mild then even if switching occurs, this tends to be to episodes which are similar in nature to previous episodes (Bond et al., 2008), and it is therefore unlikely to be debilitating for the patient. In other words, the risks of switching are less of a factor in the treatment decision. In this circumstance, antidepressants should be used either in combination with a mood stabiliser or, in cases where previous elevation has been particularly mild and brief, they can be used on their own. Lamotrigine monotherapy is also more appropriate when previous episodes of mood elevation have been mild, while in the case of more severe previous episodes of elevation, co-prescription of an agent with greater proven prophylaxis against mania is recommended and antidepressants should be used with greater caution (note: particular care is needed when combining lamotrigine with valproate).
Potential consequences of treatment-induced manic symptoms should be considered. For example, in certain occupations there may be particularly severe and long-term consequences compared with others. In such cases, prophylaxis against mania is particularly important and should be the primary consideration, and antidepressants should be used with much greater caution.
If episodes have been of particularly sudden onset then this should prompt greater concern and greater emphasis on the need for effective anti-mania prophylaxis, with greater caution regarding the use of antidepressants. Conversely, if onset has in the past been gradual with, for example, onset following a period of sleep disturbance, then this can be monitored and addressed if it occurs. The treatment of the depressive episode may then include the prescription of an AD also depending of course on the severity of the depression (see above).
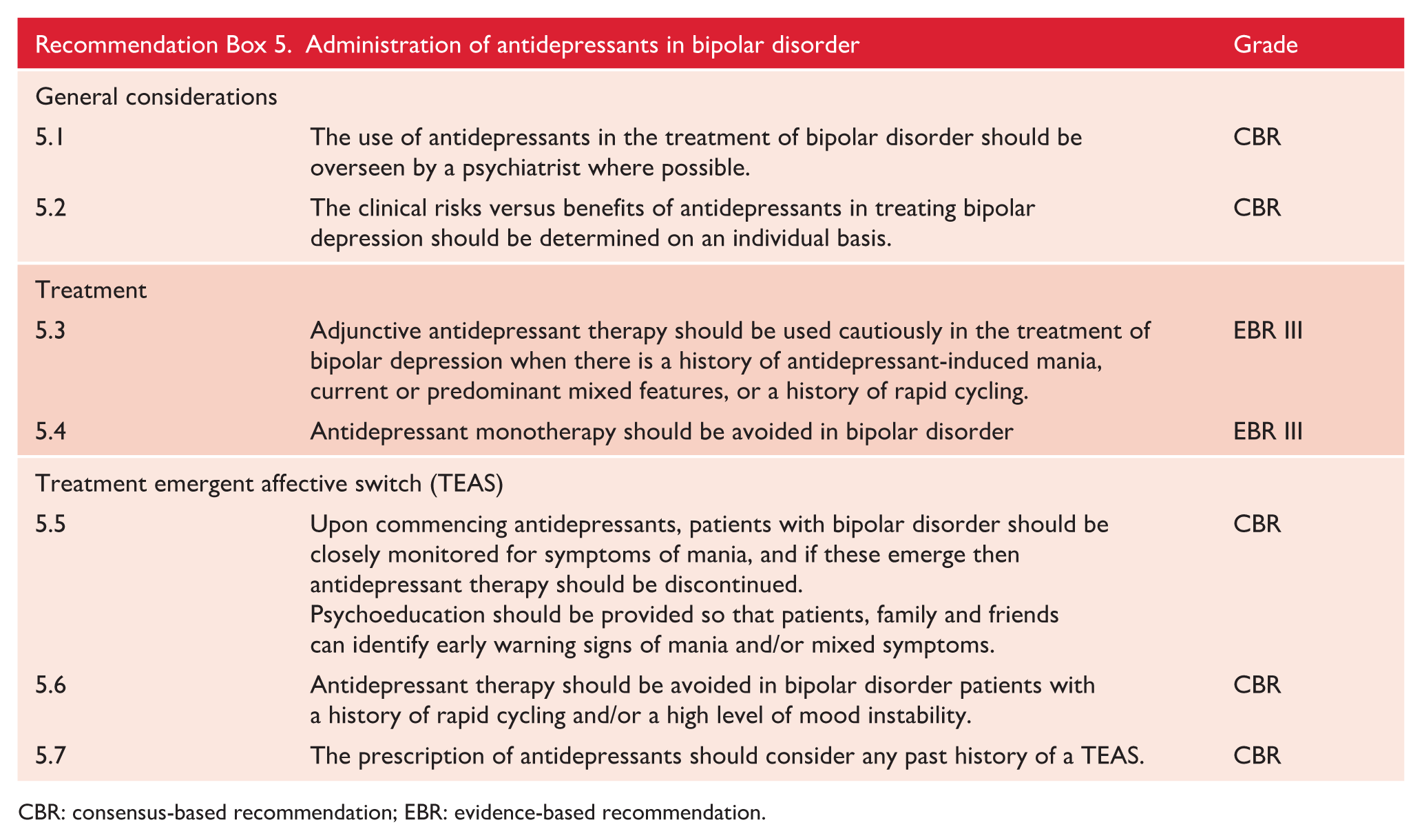
CBR: consensus-based recommendation; EBR: evidence-based recommendation.
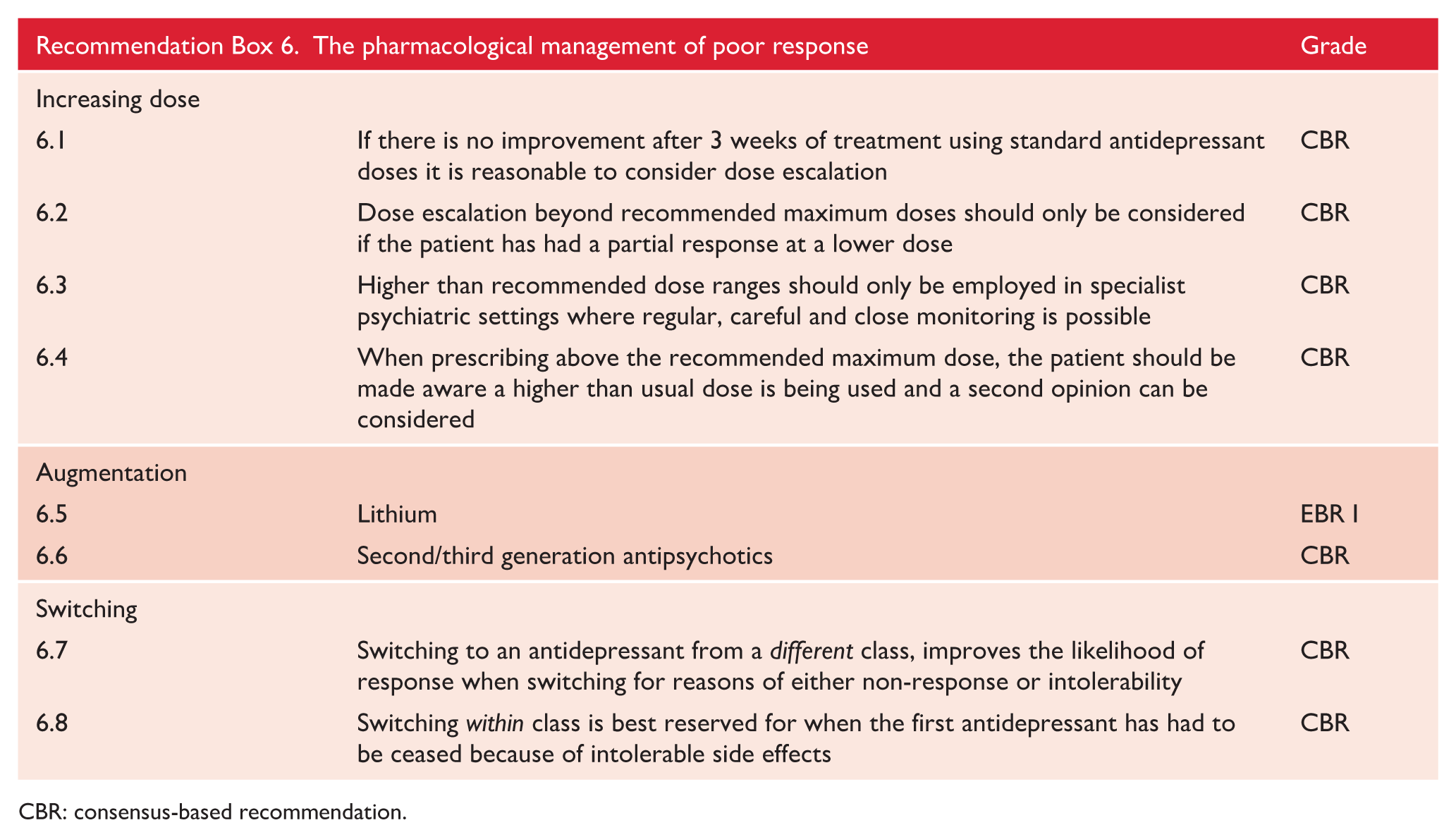
CBR: consensus-based recommendation.
8.3. Mixed states
The management of mixed states is complicated, first because of the complex nature of mixed mood states, which are intrinsically capricious and difficult to define, and second because of a paucity of evidence. Management is fundamentally dependent upon how mixed states are diagnosed, and, given that the classification of mixed states is in a state of flux because of the dramatic change from DSM-IV to DSM-5, in which mixed episodes were replaced by mixed features, evidence relating to mixed states varies depending upon which classification has been used (see ‘Mixed states’ in section 2.3.). The relatively recent change to ‘mixed features’ means that long-term evidence is not yet available and the extant clinical trial evidence has examined different kinds of mixed states – making interpretation and comparison extremely difficult.
Research studies using DSM
In DSM-IV, mixed states were captured as mixed episodes – on par with bipolar depression and mania. These episodes were codable and therefore reliable information could be gathered. However, the definition of a mixed episode was impractical as it required both a full depressive and manic episode to occur concurrently. In practice, mixed episodes were usually subsumed within mania, and most clinical trials included mixed patients as part of the manic syndrome. This is one reason why data specific to the management of mixed symptoms are sparse. Comparison of research across the two taxonomies is difficult because, while the threshold for a mixed episode, as defined by DSM-IV, was unnecessarily high, the threshold for mixed features as specified in DSM-5 is modest and, more importantly, lacks specificity – as only three symptoms from the opposite pole are required. Furthermore, the role of key overlapping symptoms (e.g. distractibility, irritability and agitation) is unclear (but currently excluded in DSM-5) and therefore, at present, mixed states can be defined in a multitude of ways. Broadly speaking, mixed symptoms (those that are predominantly from the opposite pole of illness) can occur in the context of either mania or depression, creating mania with mixed features or depression with mixed features, and instances in which symptoms from both poles are present in significant numbers can presumably be defined as either (Malhi, 2013).
A pragmatic approach to diagnosing mixed states for treatment
In the MDcpg2020 we have adopted a pragmatic approach in which a mixed state is any mood state in which symptoms traditionally described as belonging to either mania or depression are present alongside symptoms conventionally thought of as belonging to the other pole. We have not set an arbitrary threshold per se, and therefore to qualify as a mixed state even one symptom from the opposite pole may be sufficient. For example, a depressive syndrome in which the individual has guilt, anhedonia, low mood, suicidal ideation, hopelessness and poor sleep but also has marked accelerated thinking or heightened energy and agitation would qualify as having a mixed state. The key here, as with all mood episodes, is functional impairment.
We recommend that initially a mixed state is further characterised according to subtype, so that treatment can be guided. For example, ‘induced’ mixed states can be simply managed by withdrawing the treatment that has initiated a mixed state. In the same vein, once a cycling, transitional or treatment-induced mixed state has been excluded as the cause, the targeted treatment of a genuine (idiopathic) mixed state can be approached using a combination of paradigms including the ACE model, the severity of illness, predominant features and, where relevant and available, knowledge of past treatment response.
For the purposes of treatment, mixed states can be divided into two groups. The first, where there is no preponderance of symptoms from either pole (a bipolar mixed state) and a mixed state in which there is a preponderance of one pole or the other (unipolar mixed state). The latter is further divided into a depressive mixed state or manic mixed state depending on the preponderance of symptoms. This then allows three pathways of treatment as depicted in the management of mixed states (Figure 29). As per other mood states (e.g. major depression, bipolar depression, mania) the management of mixed states also commences with Actions which are roughly the same as those for the acute and maintenance phases of bipolar disorder. However, a notable and important difference is that of stopping treatments that can exacerbate a mixed state, in particular, antidepressants in the case of a manic mixed state.
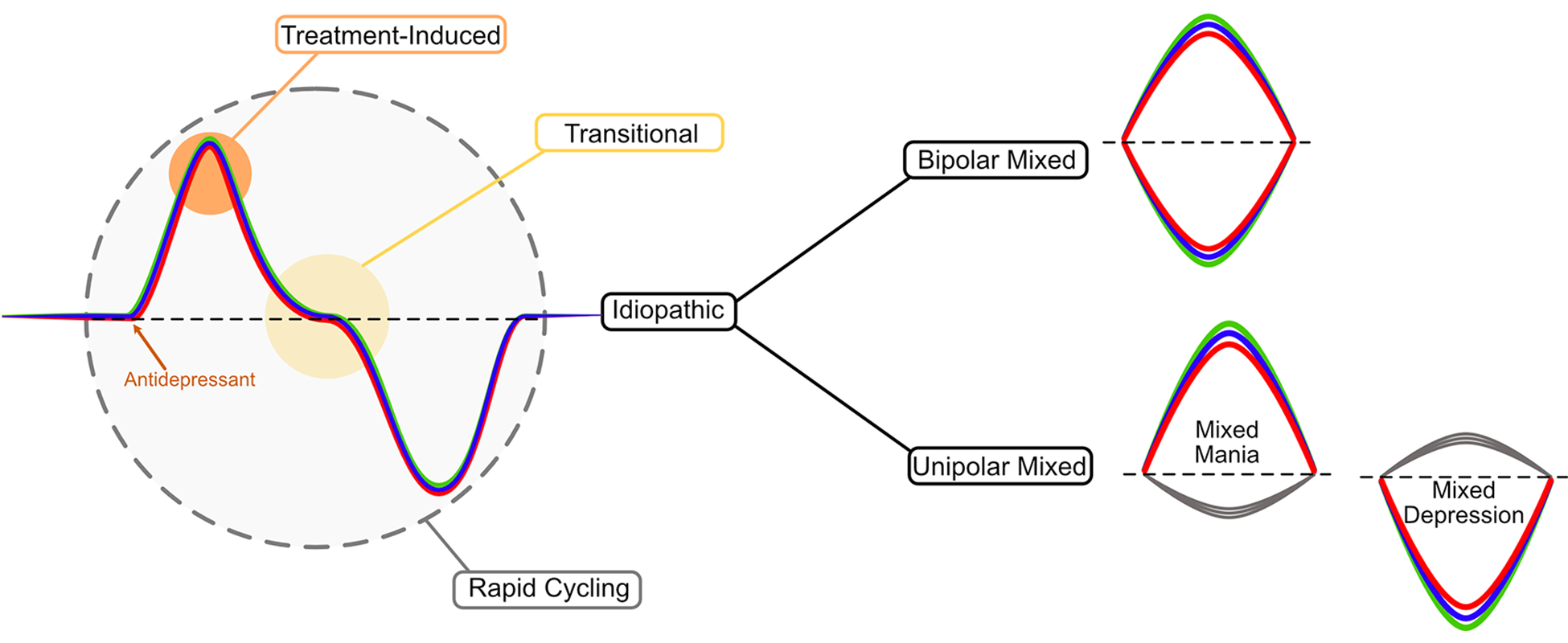
Overview of the management of mixed states.
Actions
Psychoeducation and structured psychological interventions are particularly important in the management of mixed states, where the outcomes have, thus far, been poorer as compared to treating bipolar depression or mania. Mixed states are associated with a higher likelihood of suicide, and therefore, therapeutic engagement is essential. Making patients and carers aware from the outset, that mixed states are complicated and challenging to manage is important, and it is essential that expectations are moderated accordingly. In practice, mixed states can often be confused with personality disorders and missed altogether because of comorbid substance misuse (Coles et al., 2019). Mixed states can also be particularly challenging to manage for carers and health professionals because, when patients are in a mixed state, their behaviour can be unpredictable and erratic, with often inexplicable changes in emotion and activity and they can also be overly irritable. For example, while stating they are depressed, they can become overactive, momentarily elated or demanding. Such behaviour may be confusing for carers who may not appreciate the distress the individual is experiencing. When this occurs, it is important to listen to, and appreciate, the feelings of carers and clarify that the patient’s seemingly irrational behaviour is part of the clinical picture. Hence why sophisticated psychoeducation is particularly important and should include the family.
Choices
Due to a lack of evidence, no agents can be specifically recommended with a high degree of confidence for the management of mixed states per se. Hence, the recommendations here are based on an amalgam of studies conducted in a variety of mixed mood states, along with extrapolation from the treatment of mania and bipolar depression combined with clinical experience. The principal aim of treatment in the management of mixed states is the same as that in the treatment of mania and bipolar depression, namely, to treat acute symptoms and achieve mood stabilisation. However, an important difference that sets the management of mixed states apart is the risk of creating contrapolar symptoms. In other words, overshooting and exacerbating the mixed symptoms. Therefore, it is useful to conceptualise two processes taking place in unison.
First is the treatment of acute symptoms and ameliorating their severity, and the second, involves ensuring that existing symptoms are not exacerbated, and new symptoms are not created. Clearly, this is inherently difficult and hence why the backbone of pharmacotherapy for mixed states is that of mood stabilisation, using agents that have both antimanic and antidepressant properties. It is important to note this is essential, regardless of the predominance of symptoms, and hence why agents that have both antidepressant and antimanic properties and have mood stabilising properties provide the basic axis for management. Three agents fall into this category – lithium, valproate and quetiapine, and depending on the past history of the individual and their suitability to each of these medications, one of these should be prescribed to ensure contrapolar symptoms are not generated and mixed symptoms are not exacerbated.
For bipolar mixed states, one of these agents may suffice; however, in some cases, it may be necessary to administer combinations such as lithium + quetiapine, or lithium + valproate. Medications that have some evidence to suggest effectiveness in both mania and depression can also be used in bipolar mixed states, such as cariprazine and ziprasidone. Note, these two medications can also be used in either mixed mania or mixed depression. Additional Choice agents include aripiprazole and asenapine, both of which have efficacy in mania and some early signals of efficacy in mixed mania. Similarly, lurasidone, which is indicated for bipolar depression, can be used for the treatment of mixed depression.
Where possible, monotherapy is desirable. However, given the complexity of mixed states with the concurrent need to treat symptoms, and prevent the exacerbation of others, usually combinations are necessary. Among the various recommended Choice agents, specifying a sequence is difficult but subtle preferences are indicated in The Management of Mixed States figure (Figure 30).
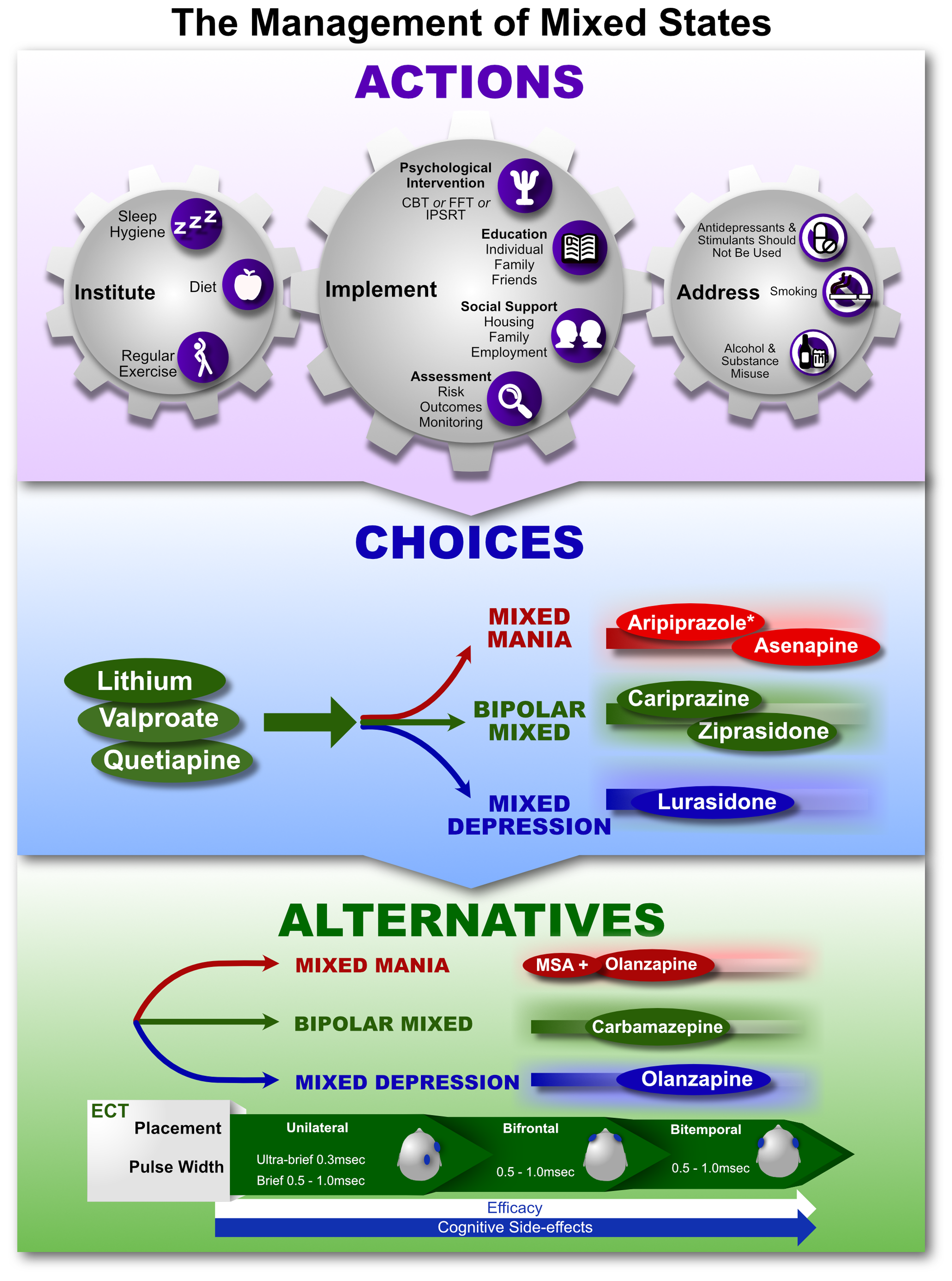
The management of mixed states.
Alternatives
There is evidence for combination strategies and also the use of agents that otherwise have significant side effects, such as olanzapine, and these are included as Alternatives if Choice agents and combinations of these are unsuccessful. However, in addition, it is important to note that ECT is effective in both mania and depression and therefore it remains a useful Alternative for consideration, once reasonable pharmacotherapeutic strategies have been trialled. Again, it is important to emphasise that although antidepressants may have some utility in the treatment of bipolar depression they should be avoided, if at all possible, in the management of mixed states as should stimulants, which should be avoided altogether.
8.4. Maintenance
The maintenance phase of bipolar disorder follows the remission of acute, depressive, manic or mixed symptoms. It is the phase in which the focus of management shifts to maintaining euthymia and mood stability and preventing the occurrence of an acute episode of illness. It is therefore the phase of illness that is also concerned with prophylaxis, and is without doubt, the most important phase in the management of bipolar disorder. This is because only during maintenance can psychological therapies be engaged to their full extent. It is the phase in which recovery can be galvanised and interventions that enhance resilience can be administered. However, it is also the phase in which there is greatest likelihood of relapse because of nonadherence and the resumption of activities and habits that can precipitate an acute episode (e.g. returning to work and taking on additional stress and resuming alcohol and illicit substance intake). When feeling well, individuals are more likely to miss appointments with their psychiatrist or psychologist and are more inclined to stop medication or moderate their medication dosages because of side effects and the belief that medication is no longer necessary or needed. These are significant risks and so it is critical that follow up is maintained and patients are educated regarding cessation of treatment. Instead, it is important that they are encouraged to focus on enhancing resilience to reduce the likelihood of future relapse.
The Actions in maintenance therapy are much the same as in other phases of the illness in terms of needing to focus on sleep, exercise and diet, stopping treatments that could cause or contribute to mood instability, and instituting psychosocial measures alongside education. However, whereas previously in the management of bipolar depression or mania there is a clear end point, namely remission of the episode, in the maintenance phase of bipolar disorder there is no end point per se, and instead a long-term view needs to be adopted. For this, it is necessary to set realistic goals and in order to achieve these, regular monitoring is required, and the process of evaluation and adjustment has to be ongoing, more so than in any other phase of bipolar disorder. Establishing a good therapeutic relationship for the maintenance phase of treatment is critical, so that the treating clinician understands the nuances of the patient’s illness and the patient is able to develop trust. As bipolar disorder is a lifelong illness, continuity of care is essential. However, sometimes care has to be transitioned to a new doctor and this requires careful discussion and the identification of a new doctor that ‘fits’ with the patient.
Another key consideration in maintenance therapy is when to transition from adopting an acute perspective on treatment to a long-term view, and when to institute medications that have predominantly mood stabilising and prophylactic actions as opposed to immediate treatment of acute symptoms. Even more specifically, it is critical to determine which strategies to employ, and what specific treatments need to be in place. This is a necessarily complex process that begins with a detailed clinical appraisal of all aspects of management as the person begins to improve and the symptoms of an acute episode of illness begin to remit.
Principal considerations
As the symptoms of an acute episode remit, and treatment attention switches to that of maintenance therapy, it is important to take note of the recent episode from which the maintenance phase of the illness emerges; in other words, whether the person is recovering from mania, depression or a mixed mood state. Simultaneously, it is important to consider the precipitants that led to this particular episode. Was it the first episode (i.e. a de nouveau episode of depression or mania)? Or does the person have a past history, such that the recent episode has occurred in the context of ongoing treatment (a breakthrough episode). Of course, it is possible that the person has simply stopped their treatment, and this too is an important consideration. Hence, it is also important to consider personal factors, namely whether the person believes they need treatment (of any kind), and whether they are likely to adhere to treatment as prescribed.
It is also necessary to consider the prior responsiveness of the illness to treatment, and in particular, which treatments have been utilised for recent episodes of depression or mania, both psychological interventions and pharmacotherapy, and to what extent they have worked. Collating and synthesising all this information is critical, because it helps guide the formulation of a prevention strategy and it is essential to be armed with this knowledge before considering specific treatments and treatment strategies (see Figures 31 and 32).

Appraisal for maintenance treatment in bipolar disorder.
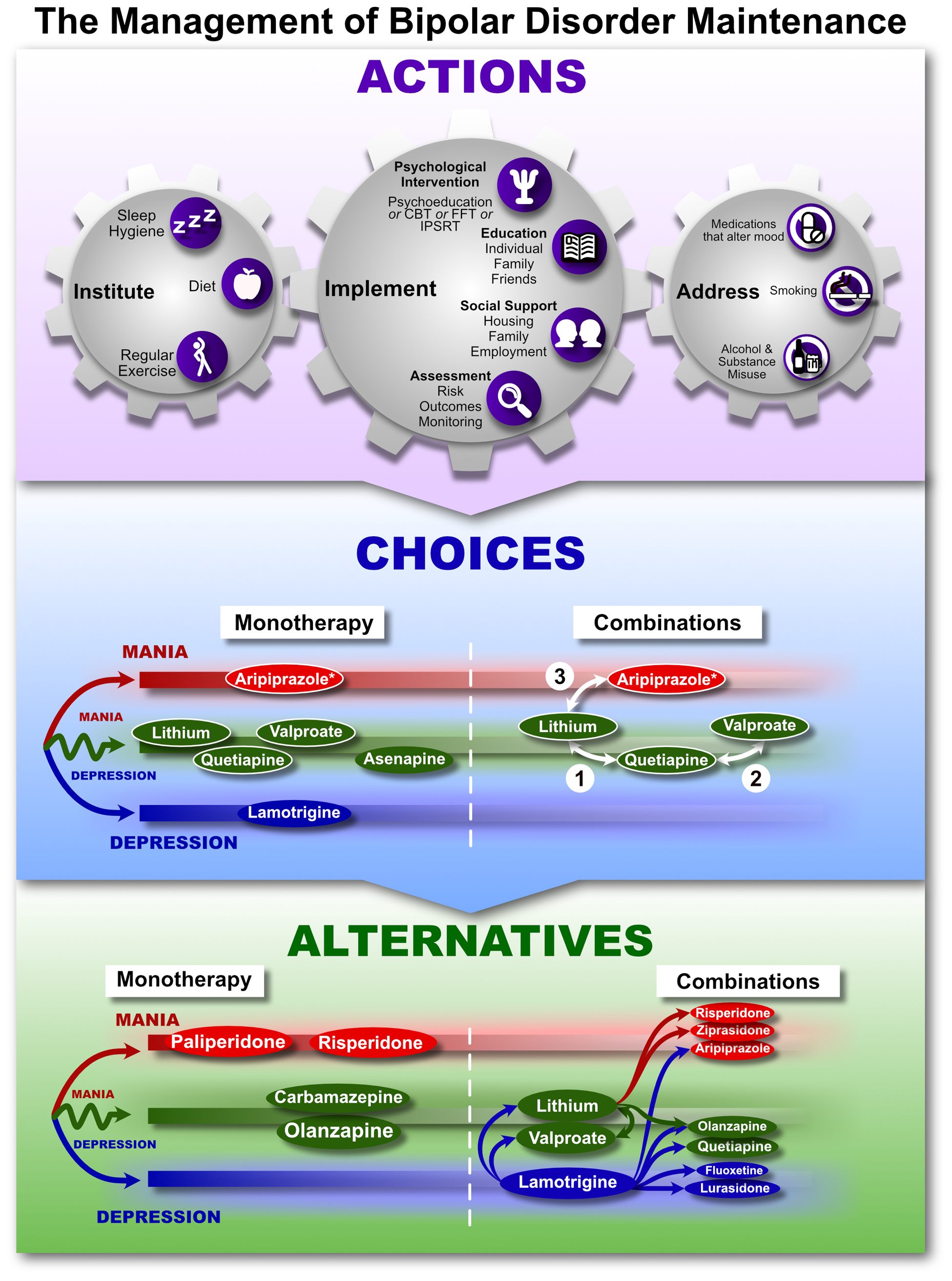
The management of bipolar disorder – maintenance.
Actions
Psychosocial and general medical health
As mentioned, the maintenance phase of bipolar disorder is an excellent opportunity for psychoeducation and psychological interventions such as CBT, family focused therapy, interpersonal and social rhythm therapy, and support from family and friends. It is therefore important that the individual is engaged with the appropriate therapist such as a psychiatrist or psychologist and has access to other health professionals as necessary, such as a social worker or a community mental health nurse. The general medical health of the individual is also important and so regular engagement with their General Practitioner is also necessary.
Apart from actively engaging in therapeutic strategies, it is also important to monitor the individual’s mental state and functioning, and therefore regular clinical follow up is essential. Such assessments should include detailed evaluation of symptoms, side effects and functioning, and ongoing clinical assessment can be aided by making use of technological means of monitoring (see ‘Digital therapies’ in section 6.1). The specific psychological therapies that are evidence based for maintenance and prophylaxis of bipolar disorder include group Psychoeducation, CBT, IPSRT and FFT (Miklowitz et al., 2020; Murray, 2018).
There is evidence that risk of recurrence increases with the number of prior episodes of bipolar disorder and a greater number of episodes is also associated with more chronic symptoms, and functional impairment (Tremain et al., 2020b). Number of episodes may also moderate the response to treatments, particularly adjunctive psychosocial treatments. For example, an early trial of CBT for bipolar disorder (Scott et al., 2006) found a bifurcation at 12 episodes: adjunctive CBT was superior to treatment as usual in participants with fewer than 12 previous episodes, but less effective in those with more episodes. Much remains unknown about treatment implications of the long-term trajectory of bipolar disorder (Tremain et al., 2019, 2020a), but clinicians should be mindful of the additional challenges likely to confront patients with a long history of relapses, growing chronicity and psychosocial challenges. In some cases, living well despite ongoing symptoms may be a reasonable goal, and the focus of psychological work may shift to values-based and self-compassion work (Murray et al., 2017).
Choices
Pharmacotherapy
The use of medications to maintain mood stability and prevent a recrudescence of an acute episode is a difficult and complex process. As discussed in the general principles of maintenance management, a detailed appraisal is needed of the most recent episode, its treatment and the nature of past episodes and their treatment in order to formulate an appropriate maintenance and preventative strategy (see Figure 31).
This information is essential as it informs which medications have been previously successful and which have not, and thus, which medications remain as available options. It is also useful to know which treatments have been ineffective and caused problems and may need to be avoided. Therefore, a thorough appraisal provides an indication of which strategies are most likely to be helpful, and may also give valuable insights into whether, for example, combinations of interventions are necessary.
Figure 32 shows the various agents available for maintenance and prophylaxis and broadly notes their efficacy relative to the phases of bipolar disorder. Reading from left to right, it also provides a rough guide to the order in which medications should be considered, reflecting their strength of evidence and relative efficacy in relation to one another. Note that the chart comprises three overlapping areas shown by different colours – namely, depression, mania and both. A key consideration at the outset is to determine whether efforts to prevent future episodes need to be focused on one of these particular phases. This has been termed polarity predominance and it is best determined on the basis of reviewing past history. For example, individuals who have had predominantly depressive episodes are described as having a predominantly depressive polarity, and therefore, maintenance medication for such individuals can be weighted towards preventing depression. However, alongside the overall predominance of polarity based on past episodes, it is important to consider the most recent episode from which the individual is recovering. Clearly, if the recent episode is mania, then the medications that have been used to treat the acute symptoms of mania will need to be gradually substituted or stopped in order to transition to medications that will provide prophylaxis against depression. This can be thought of as a treatment journey that requires determining a starting point and identifying where the various treatments are likely to lead and ultimately end. The steps that need to be taken to make this ‘journey’ can then be mapped and instituted by titrating medications accordingly. So, for example, if transitioning from acute mania to prophylaxis that is geared to providing mood stability and prevention of depression, then many of the acute antimanic agents may need to be stopped, a mood stabiliser will need to be introduced, and additional antidepressant medication may need to be instituted. When considering the steps along this treatment journey it is necessary to review which treatments are available and which have been successful. In other words, medications that have been trialled in the past may not be options, and so treatment needs to be planned with a clear understanding of both the patterns of illness, and patterns of response. On top of this, patient preferences also need to be considered. For example, some patients are especially sensitive to weight gain, while others may be intolerant of medications that cause even mild sedation. Therefore, treatment needs to be carefully planned with these considerations in mind and it is subject to constant revision based on ongoing assessment.
Figure 32 shows that lithium is the preferred Choice for long-term therapy and that this should be given initial consideration whenever planning maintenance therapy. This is because lithium provides prophylaxis against both depression and mania and can be monitored (Malhi et al., 2016b). The choices for monotherapy, where both depression and mania need to be prevented, include lithium, valproate, quetiapine and asenapine, with the latter two (valproate, quetiapine) having better evidence for the prevention of depression. In general, mood stabilisers are given preference when prescribing maintenance therapy, because of their better tolerability, but if there is a preponderance of mania, then aripiprazole (long-acting injectable formulation if adherence is a problem) is a suitable choice, whereas if depression predominates, then lamotrigine may be more suitable. However, in many instances, combinations are needed and therefore, lithium can be combined with quetiapine, or quetiapine can be combined with valproate, or where there is a greater likelihood of future episodes of mania, lithium can be combined with aripiprazole.
Alternatives
If Choice agents, either as monotherapy or combinations, are unsuccessful in maintaining mood stability, then several Alternatives can be trialled (see Figure 12). Once again, if there is equal emphasis to be placed on both prophylaxis of mania and depression, then carbamazepine and olanzapine are options as monotherapy. Where mania is the main consideration, then paliperidone and risperidone can be trialled. However, in some instances, even these will be insufficient, and combinations may be necessary. For the purposes of combining medications, it is important to think of key, foundational medications around which the combinations are constructed. These include lithium, valproate and lamotrigine. To prevent both mania and depression, lithium can be combined with valproate, or either of these can be combined with olanzapine. Where there is a predominance of mania, lithium or valproate can be combined with risperidone or ziprasidone. Where depression is the main target in terms of prophylaxis, then lamotrigine can be combined with lurasidone, lithium or valproate, or it can be combined with quetiapine, olanzapine, or aripiprazole (increasingly recruiting an antimanic effect). Note, olanzapine should be prescribed with caution in this regard, because of its long-term metabolic side-effects.
9. Response to treatment
The MDcpg2015 considered the assessment and management of a suboptimal response when treating a mood disorder in detail, and while many of the principles still hold, the MDcpg2020 provides an important change in focus.
First, we discuss poor response to various treatments in the context of the overall management of a mood disorder. With respect to the implementation of therapeutic strategies (psychological, pharmacotherapy and physical treatments), we discuss their sequencing and in relation to pharmacotherapy, we introduce the concept of MIDAS, which attempts to enhance a poor response to achieve recovery. We also examine treatment non-response in greater detail and make a distinction between difficult-to-treat depression (DTD) and treatment-resistant depression (TRD). However, the most significant development is the shift in focus from ‘poor response’ and ‘treatment resistance’ to treatment responsivity with the introduction of the concept of channelling response. In channelling response, treatment is conceptualised as comprising channels of response and the aim of management is to identify and use the most relevant channel to achieve recovery.
9.1. Response
Clinically, the desired response to treatment is rapid improvement and restoration of normal functioning. In the case of depression, the patient recovers from their depressive symptoms and returns to their normal duties. In contrast, by convention, the term response has been described in research trials as a 50% reduction in symptoms (usually gauged using a standard rating scale). In practice, this falls short of ‘clinical improvement’ and so recently there has been a shift towards emphasising the importance of remission (the absence of symptoms) and recovery (the restoration of normalcy with no need for ongoing treatment) and the resumption of normal functioning. Beyond this there are additional goals such as the prevention of future illness and the development of resilience (see section 5.1, ‘Aims of treatment’).
However, despite these meaningful developments with respect to the ultimate aims of treatment, in clinical practice and in research the most often discussed immediate goal remains that of response, and as a consequence, there is undue emphasis on inadequate response. The latter includes poor response, partial response and no response. Clinically, this focus on the lack of a suitable response translates into a preoccupation with the likelihood of a negative outcome.
9.2. Poor response
It is important to note at the outset that non-response is simply a special case of poor response. The latter is more widely used, and in most cases, poor response subsumes non-response. It is also important to acknowledge that in some cases a poor response may reflect either a complete misdiagnosis or an underlying irreversible cause for clinical depression (e.g. stroke, brain damage) that is not amenable to psychological and/or pharmacological remedies.
Technically, a poor response to treatment begins as soon as a therapy or intervention is deemed to be unsuccessful. Thus, it is an aspect of management that needs to be borne in mind at all times – and applies to all forms of intervention and treatment, including lifestyle strategies, psychological intervention, physical treatments and pharmacotherapy. However, most commonly, a poor response is considered in the context of pharmacotherapy and often equated to treatment resistance. Indeed, it is ostensibly the basis of treatment-resistant depression (TRD).
9.3. Difficult-to-treat depression (DTD) and treatment-resistant depression (TRD)
The term treatment-resistant depression (TRD) is problematic because it has been associated with a narrow focus on pharmacotherapeutic non-response, and also because it invokes a nihilistic view as regards the management of depression (Malhi et al., 2019c). The description also insinuates that TRD is a ‘kind’ of depression (a subtype); one which is somehow resistant to currently available treatments, in particular medications. To overcome these problems the term difficult-to-treat depression (DTD) has been proposed with two main advantages (McAllister-Williams et al., 2020): first, unlike TRD, the description ‘difficult to treat’ suggests that the management of the depressive illness is likely to be challenging but at the same time it is not totally unachievable. In other words, the depression is not completely resistant (refractory) per se. Second, the definition of DTD is not predicated on pharmacotherapy alone, and the management of DTD involves a much broader set of strategies – including, for example, psychosocial measures. Nevertheless, DTD does retain some of the negative connotations associated with TRD, in that it still implies the depressive illness is insufficiently responsive to therapeutic interventions. The reasons for DTD are patient defined, and, in addition to non-response, include medication intolerance and lack of acceptance of the diagnosis and need for treatment (McAllister-Williams et al., 2020). Furthermore, unlike TRD, DTD is not exclusively focused on the acute treatment of depression.
A key problem that DTD shares with TRD is that both ‘diagnoses’ require a threshold. For TRD this is often specified as failure to achieve a suitable response to two or more adequate courses of pharmacotherapy; a very modest and clinically meaningless threshold. For DTD the threshold is even lower, which makes the term nearly universal and means that it lacks specificity. For example, the designation of DTD can be applied immediately following the very first intervention has failed to achieve recovery. Thus, DTD is extremely heterogeneous, as any number and all manner of ‘difficulties’ can contribute to non-response. And therefore, although DTD confers some advantages over the more widely used term TRD, such as being potentially less stigmatising, it does not sufficiently alter the focus of management and does not resolve the problem of having to define a new ‘subset’ of depression.
Broadly speaking, TRD can be regarded as a subset of DTD, which in turn is a subset of depression. However, in practice, the boundaries of these conditions are difficult to define meaningfully, and the designations are of limited clinical value. Furthermore, they create a negative therapeutic perspective. Hence, recently, an alternative has been proposed, which dispenses with the need to define a threshold and changes the focus of treatment from non-response to responsivity (Malhi et al., 2020). The change places emphasis on the process of treatment with a view to achieving recovery. This new paradigm shifts attention to treatment response and reframes both the management of depression and the nature of the illness in terms of responsivity (see Figure 33).
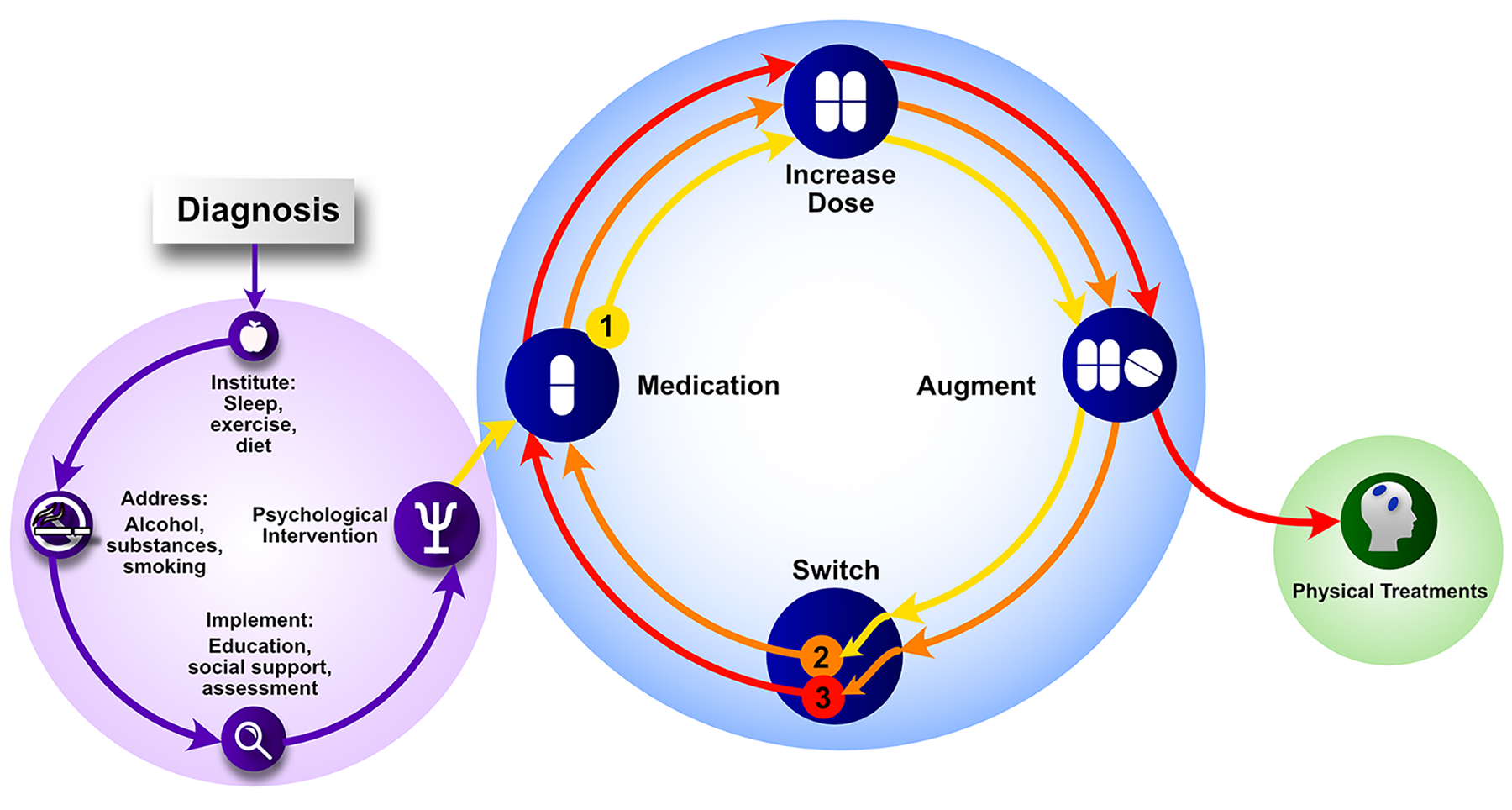
Strategies for channelling response.
9.4. Channelling response paradigm
Typically, depression is clinically heterogeneous and of variable severity. It likely comprises several subtypes – each of which is determined by both biological and environmental factors (Malhi and Mann, 2018). Thus, depression has many manifestations, each with a potentially different responsivity to treatment – and each necessitating individualised management. Note, the focus of this perspective is not on poor response or resistance to treatment, but instead on response (outcome) and responsiveness (of the depression). This is described as the response perspective and it can be adopted throughout the management of depression from beginning to end. By assuming this perspective, the need to create an artificial threshold to demarcate fluid concepts such as ‘treatment-resistant’ or ‘difficult-to-treat’ depressions is obviated. Instead, the ‘process of management’ becomes the focus, in which identifying the responsivity of the unique depressive illness is the primary aim.
The change in focus to response involves regarding depression as an amalgamation of responsivities, which can be conceptualised as units of response. Different depressions comprise different responsivities and will therefore respond to different treatments. The precise combination of responsivities in any particular case of depression is determined by a whole host of biological and psychosocial factors, and knowing these can provide clues as to which treatments are likely to be most effective. Accordingly, suitable treatments can be trialled while focussing on the responsivity of depression and treatment response.
Importantly, the response paradigm encompasses all manner of interventions (psychological, social, pharmacological and physical) and includes the combination of different kinds of treatments and strategies. In other words, it reflects the complexity of real-world management. To illustrate this paradigm, treatment response and the responsivity of depression can be conceptualised as a ‘channel’ between depression and recovery (see Figure 34). Traversing the channel reflects the journey of responding to treatment and achieving recovery with different interventions facilitating this process. It is important to note that in any particular case of depression several pathways may be available and that the journey involving response can be undertaken via any of them.
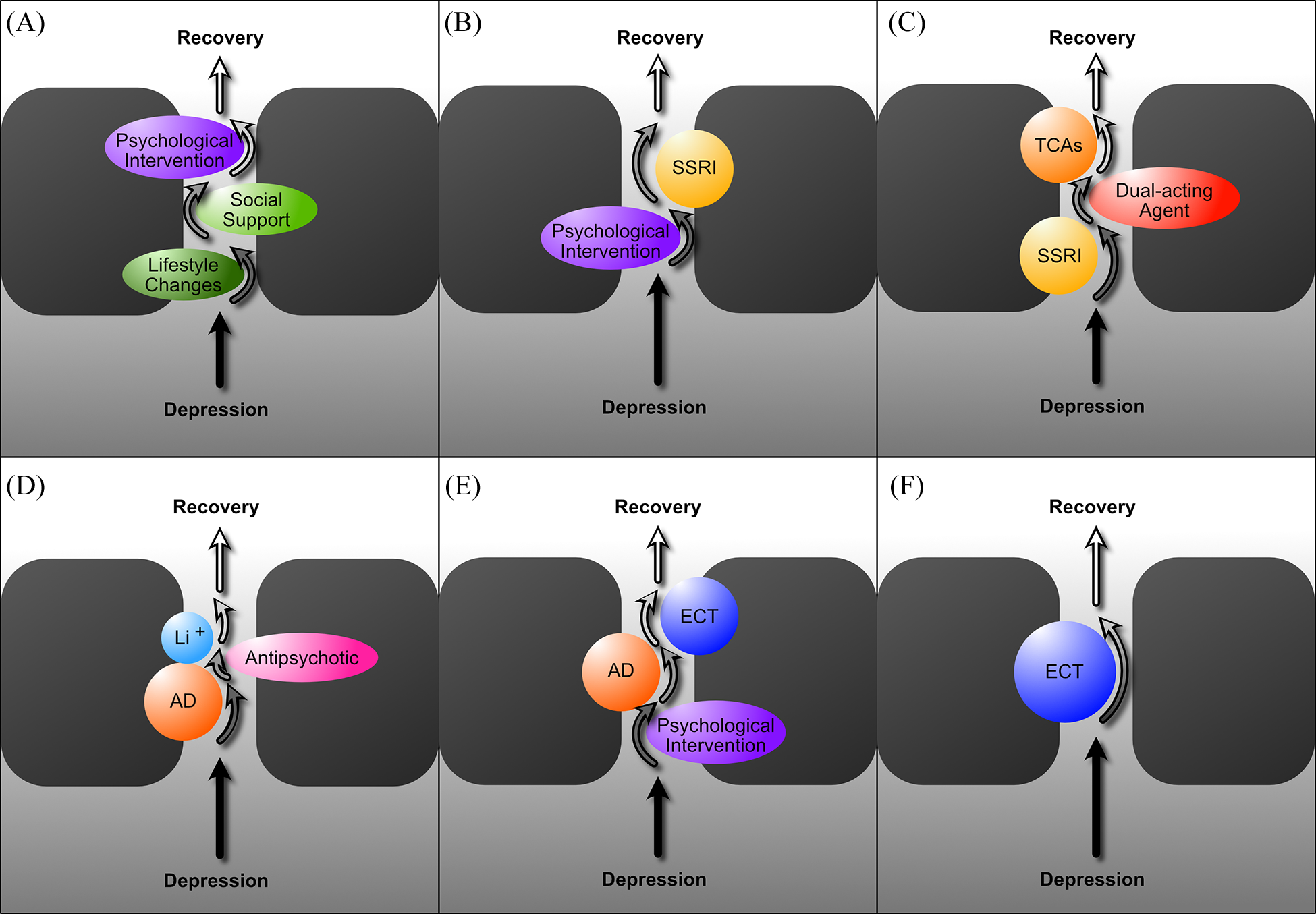
Modelling therapeutic channels of response.
In some kinds of depression both psychological and pharmacological interventions will be required, and only when employed in unison, are they sufficient and successful. In other instances, physical treatments may be needed, and sometimes this may be the case from the outset. Within different kinds of treatments, passage through some channels may require augmentation, and in some instances, the use of a number of agents may be necessary to achieve initially a partial response and then full recovery. It is important to note that the focus throughout is on response and that a partial response is not regarded as a poor response but instead, as a step along the path to recovery. In other words, some progress has been made and, in some cases, an initial step may be vital, as it then allows subsequent treatments to complete the ‘journey’.
A complete lack of response may be an indication that an altogether different channel is required, and that an alternative treatment strategy has to be engaged. However, here too, not all efforts are necessarily wasted and there may sometimes be an essential facilitatory process between channels. For example, in practice it is often the case that a course of ECT following a series of treatments that have been unsuccessful sometimes ‘opens up’ the possibility of responsivity to subsequent treatments (even those that have been trialled unsuccessfully in the past).
The advantage of the channelling response perspective is that it maintains a consistent approach throughout the management of depression and keeps the focus on treatment responsivity. In other words, it regards depression as treatable, and as an illness that will respond – provided a suitable treatment is administered appropriately. Adopting this perspective is likely to imbue confidence both in practitioners and patients and provide a more realistic expectation of outcomes, namely that the correct responsivity needs to be identified and this may take a number of treatment trials and different strategies depending upon the make up of a particular depressive illness. The channelling response paradigm also obviates the need to categorise depression using ‘difficult-to-treat’ or ‘treatment-resistant’ labels, which imply that there is something unusual about their particular depressive illness, or indeed them, as individuals. At the same time, the channelling response paradigm does allow for instances in which a specific treatment responsivity has not been found and all reasonable measures have been ineffective in achieving recovery. These are instances in which an alternative diagnosis is the likely cause of the depressive illness, for example, a stroke or neoplasm.
Poor response will also occur during the trialling of different treatments, but the attitude to this is not that the treatment has failed, leading to a sense of despair and questioning as to whether the illness is resistant to therapy, but instead understanding that the responsivity of this particular illness is yet to be identified and that a suitable and effective treatment strategy has simply not yet been administered. If this approach and perspective is adopted from the outset, then both patients and clinicians alike, are less likely to be discouraged as they progress through the many combinations and treatments that are available; many of which will lead to improvement.
Principles of the channelling response paradigm
Evaluate response
Throughout the management of mood disorders, it is important to maintain a focus on responsivity. Ideally, this perspective should be adopted from the outset of management and the treatment of mood disorders should be envisioned as an ongoing process that may require long-term engagement. While individual episodes of the illness clearly have to be treated separately, the aim of management should be to consider the likely course of the illness and institute strategies that reflect this. And although, at times, individual treatments may not be effective, longitudinal follow-up of patients suggests that eventually, most patients will respond, improve, and recover with an appropriate strategy in place. However, it is also important to acknowledge that this is partly a function of the nature of the illness, which is recurrent and self-remitting with respect to clinical episodes.
Review formulation
When managing a mood disorder, it is important to periodically review the formulation and treatment of the illness and assess its course from the beginning. This involves re-examining the diagnosis critically and within this, considering alternative formulations, in particular psychosocial factors, which are often overlooked or not assigned as much weight as biological causes. A comprehensive assessment is necessary to ensure that nothing has been missed and that the diagnosis is accurate and does not need to be changed. This is also an opportunity to gain further diagnostic clarity and specificity and attempts should be made to identify any subtypes, clinical profile or any other aspects of the patient’s phenomenology that could help inform treatment, for example, if there is a family history of depression, what kinds of treatment have worked for immediate relatives and a careful review should be undertaken of past treatments and response in family members with a similar mood disorder.
It is also important to consider concurrent psychiatric and medical illnesses and consider whether a new illness has emerged that is perhaps contributing to the mood disorder.
Review treatment
An important step is to assess whether treatment has been taken as prescribed. Note, this applies to psychological interventions as well as pharmacotherapy.
For psychological treatments it is important to gain an understanding of what aspects of the illness are being addressed by a clinician that may be providing concurrent treatment, and it is also important to review whether the patient is following advice regarding cessation of substance misuse, exercise and sleep.
With respect to pharmacotherapy, it is essential to carefully review all the medications the patient is taking – including over-the-counter medicines, alternative therapies and supplements. For each medication it is important to ensure that the correct dosage has been administered and that the patient has followed instructions carefully, for example, taking medication with food, or at a certain time of day and ensuring that they are taking an adequate dosage. These aspects of treatment may seem basic but are often the reason why a response has not been achieved. If the patient has not been adherent to treatment, it is important to find out why this has occurred, and whether there are particular reasons for non-adherence. In some cases, further psychoeducation may be necessary.
Finally, side-effects also need to be reviewed and medication may need to be withdrawn if they persist and are intolerable.
Adopt and maintain the ‘response perspective’
Throughout the management of a mood disorder, it is important to reassure patients that there are many potentially effective treatments available and that no single treatment is necessarily the final strategy in terms of options. At the same time, it is important to use every clinical interaction as an opportunity to reinforce the need for ongoing treatment and appropriate engagement with any strategy that is employed. Concerns such as dependence and addiction to medications that sometimes hinder engagement should be addressed promptly, and any misconceptions should be firmly dispelled.
Finding the channel
The management of mood disorders begins with setting a foundation for treatment and this involves a number of Actions (see section 7 ‘Management of major depressive disorder’, Figure 26). In the management of depression, following diagnosis and the institution of appropriate actions, treatment involves the implementation of psychological interventions, which can, if necessary, be combined with medication. Pharmacotherapy involves the administration of Choice medications and following the administration of a suitable medication for an appropriate duration of time, an increase in dose may be trialled in order to achieve an optimal response. Subsequent to this any response that has occurred can be augmented – for example, with the addition of lithium, but if these strategies are unsuccessful the antidepressant medication can be switched and the whole process can be repeated. Given the number of Choice antidepressants available for the management of depression, the prescription of a suitable medication (M), followed by an Increase in Dose (ID), its Augmentation (A) and finally a Switch (S) to another antidepressant should be trialled a minimum three times (repeating the cycle [MIDAS] – see Figure 35) so as to ensure that the effects of antidepressants from at least three different classes have been experienced, and many more times if considering Alternatives. However, at some point, to achieve a response it may be necessary to switch from pharmacotherapy to physical treatments and this may occur much earlier if there are specific indications that are particularly responsive to ECT such as psychotic symptoms and melancholia. The evidence supporting this process is outlined in Box 17.
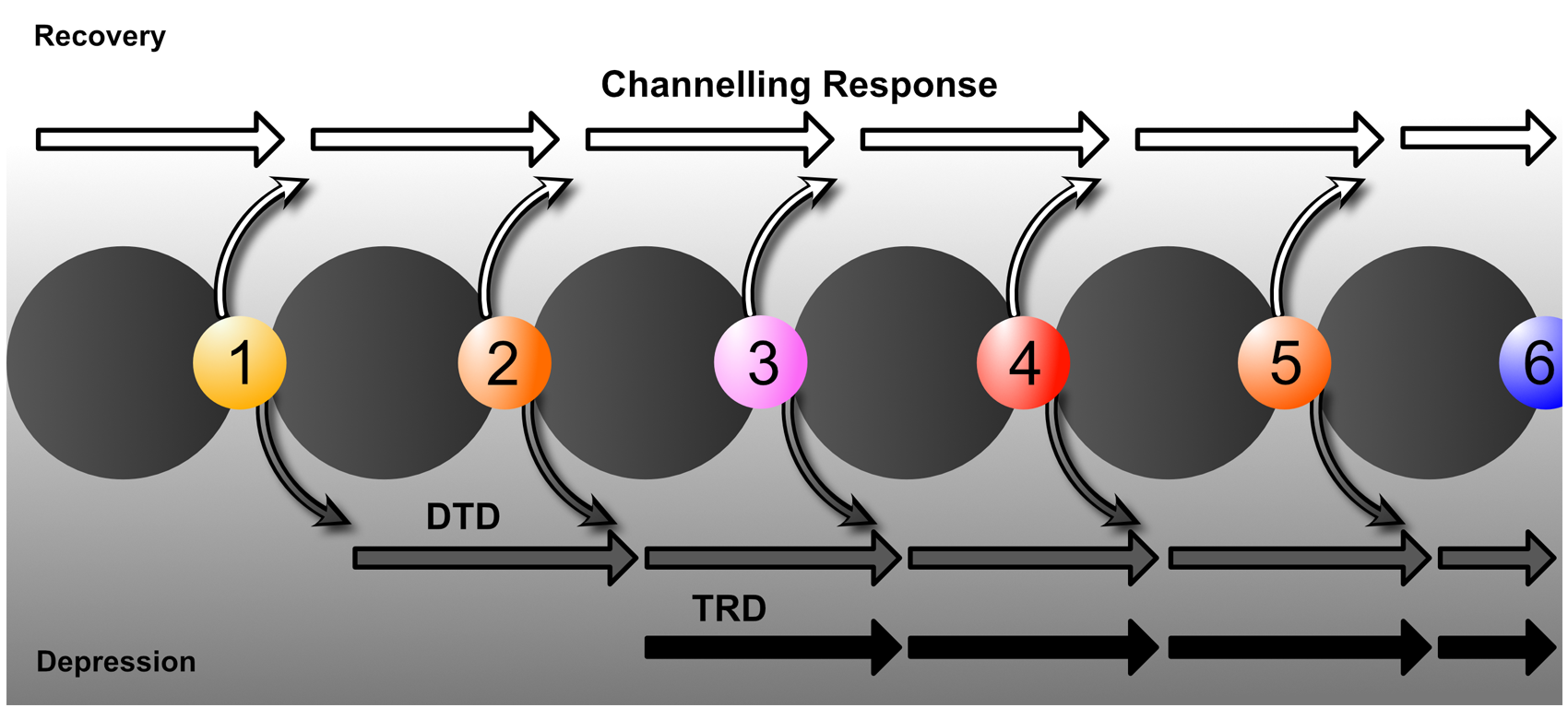
Models of treatment: DTD, TRD and Channelling Response.
Evidence for strategies and interventions used to overcome poor response to treatment
Psychological treatment
Effect sizes for psychological treatment of acute depression are only small in good quality trials (Cuijpers, 2017), and perhaps half of patients fail to achieve reliable improvement under naturalistic conditions (Pybis et al., 2017). While psychological treatment generally protects against deterioration, it is not a silver bullet – and some 5–10% of patients may still deteriorate despite psychological treatment (Rozental et al., 2019), and therefore clinicians should remain alert to this possibility.
There is a paucity of information about modifying treatment plans in the face of poor response to psychological treatments for depression. A number of principles are worth noting.
First and foremost, clinicians offering psychological intervention should monitor progress and response (in a similar way to that used to monitor response following pharmacotherapy; see Box 13).
Second, as noted above, where psychological monotherapy is insufficient, data suggests the addition of antidepressant medication should be considered.
Revisiting the case formulation (potentially in consultation with an experienced colleague) may identify barriers to progress from a psychological and behavioural perspective.
Revisiting the case formulation may lead to consideration of the ‘fit’ of the therapeutic approach to the patient. Although evidence is limited, clinical consensus suggests that varying emphases of different therapies (e.g. behavioural, cognitive, schema-focused, interpersonal, short-term psychodynamic and supportive) may resonate with patients to different degrees.
In practice, reviewing the fit between the patient and the therapy they are receiving may raise the possibility of changing the therapist; a strategy with a number of challenges if the patient is acutely depressed. Such a change should only be considered in consultation with the patient, taking care to maximise support/continuity, to minimise additional illness work on their part, and proactively address the risk of engendering feelings of rejection or abandonment.
Finally, different patients may preferentially respond to individual, supported online, or group treatment approaches, and poor response should encourage consideration of changes in therapeutic modality (keeping in mind the cautions raised above).
Pharmacological treatment
Increasing the dose of an antidepressant beyond its recommended standard dose may improve the clinical response with some antidepressants, but the evidence from clinical trials suggests that there is limited improvement in response at doses above the recommended dose for the second generation antidepressants (e.g. the SSRIs) (Furukawa et al., 2019), and that any modest benefit comes at the risk of more side effects (Jakubovski et al., 2015). Therefore, dose escalation beyond the recommended range should only be considered if the patient has had a partial response at a lower dose. However, this strategy is unlikely to be of significant benefit if there has been no response using standard doses (fluoxetine equivalents 20–40 mg) (Furukawa et al., 2019) and adherence has been assured. It is important to note that the evidence for the benefit of high-dose treatment is sparse with few large randomised studies, though STAR*D did show some differential efficacy with an increase in the dose of SSRIs. However, the literature is mixed, with other data suggesting switching to mirtazapine is more beneficial than escalating an SSRI such as sertraline (Kato et al., 2018). An early systematic review (Adli et al., 2005) was supportive of high-dose treatment with tricyclics (other than nortriptyline) and tranylcypromine, where serum levels of these antidepressants can be used to guide dosage, (Hiemke et al., 2018). But at present, there is insufficient evidence for high-dose treatment with SNRIs to draw any clear conclusions and given the very many mixed findings in the literature, a case by case risk benefit analysis is needed. Thus, higher than recommended dose ranges should only be employed in specialist psychiatric settings where regular, careful and close monitoring is possible (e.g. QTc interval for TCAs and escitalopram/citalopram) (Beach et al., 2018). In addition, when prescribing above the recommended dose, a second opinion is advisable. Also, when considering high-dose approaches it is important the patient is made aware a high dose is being used and the limited evidence, and potential risks are documented. Caution is particularly needed in older patients.
Before dose escalation is considered it is essential to allow a trial of appropriate duration, at least 3 weeks, at an adequate initial dosage (De Vries et al., 2019; Licht and Qvitzau, 2002; Ruhé et al., 2006). Having said this, there will be a subpopulation of patients who are late responders (Nierenberg et al., 2000). This creates a clinical dilemma, but taking a probabilistic approach, if there is no improvement after 3 weeks of treatment using standard doses it is reasonable to consider dose escalation (Henkel et al., 2009). It is noteworthy that in practice high-dose medication can often serve as a proxy marker of nonresponse. However, this can be misleading, especially as a small proportion of patients may rapidly or poorly metabolise certain antidepressants, explaining lack of response/side effects and need for dose escalation (in such instances monitoring serum drug levels or pharmacogenomic testing may be required).
Some antidepressants (particularly first-generation agents) have a relatively narrow therapeutic range in which the agent is considered effective and safe, and research shows that increasing the dose of these medications does not always increase effectiveness but that it may assist some individuals (Ruhé et al., 2006). However, antidepressants, such as venlafaxine and TCAs (other than nortriptyline), have very broad dose ranges with up to a ten-fold increase in oral dosage, for example, venlafaxine can be safely administered at effective doses from 37.5 to 375 mg (Debonnel et al., 2007). This is putatively related to genetic variability at P450 2D6 and/or 2C19 (Hicks et al., 2017). Clinical monitoring at high doses is especially important as side effects and therapy discontinuations usually increase with dosage.
Lithium and second/third generation antipsychotics remain preferred options for augmentation in treatment-resistant depression (Bassett et al., 2019; Mulder et al., 2018; Strawbridge et al., 2019).
Clinical observations encourage the use of liothyronine (T3) even though objective research studies remain negative (Lorentzen et al., 2020; Parmentier and Sienaert, 2018). Evidence for the use of modafanil and methylphenidate is emerging (Bassett et al., 2019) and stimulants may be useful for the management of ‘treatment-resistant depression’ (see section 9.3 for discussion of this term).
Minocycline is also showing promise as an adjunctive medication (Husain et al., 2017), as are oestrogen supplements, which have a role as augmenting agents in some perimenopausal women (Maki et al., 2018).
In addition, a combination of ‘wake’ (total sleep deprivation) and light therapies (facilitating sleep cycle with light exposure) have also shown promise in the management of poorly responding depression, especially when diurnal mood variation is a prominent feature of the illness (Kragh et al., 2018).
Despite a limited evidence base to support this strategy, switching medication, and using an antidepressant from a different class, is a common strategy for managing partial or nonresponse (Boyce et al., 2020). This raises two questions: 1) first, is switching medication an effective strategy? And second, having decided to switch, does it matter which antidepressant is prescribed?
With respect to the first question, a recent large prospective study suggests that switching to mirtazapine from sertraline is more beneficial than escalating the dose of sertraline (Kato et al., 2018). Furthermore, switching between escitalopram and nortriptyline (in either direction) may be beneficial if the initial medication is ineffective (Köhler-Forsberg et al., 2019). However, contrary to these findings, a systematic review of three randomised controlled studies that compared switching antidepressant treatment to continuation of the same antidepressant failed to show any significant overall advantage for switching (Bschor et al., 2018).
Nevertheless, on balance and on the basis of clinical experience, in many cases of non-response, switching is a strategy worthy of consideration and it is a logical choice given there are few alternatives once a trial of a particular antidepressant has failed.
As regards the second question – which antidepressant to prescribe next, RCTs have found that switching to an antidepressant from a different class, improves response and remission rates when switching for reasons of either non-response (Nakajima et al., 2011) or intolerability (Köhler-Forsberg et al., 2019). Switching within class is best reserved for when the first antidepressant has had to be ceased because of intolerable side effects. It is also indicated for patients with mild to moderate depression. Switching out of class (e.g. from an SSRI to an SNRI) especially to an antidepressant with greater efficacy is indicated when the patient has severe depression or melancholia.
SSRI: selective serotonin reuptake inhibitor; TRD: treatment-resistant depression; SNRI: serotonin and noradrenaline reuptake inhibitor.
10. Children and adolescents
10.1. Major depression
There are fewer clinical trials of first line treatments for MDD in children and adolescents than there are for adults (see ‘Pharmacotherapy’ in section 6.2). Evidence for the management of unresponsive MDD is presently limited to two controlled trials (Zhou et al., 2014).
Overall, much remains unknown about the optimal treatment of depression in children and adolescents. There is Level I evidence for psychological treatments (CBT and IPT) (Weersing et al., 2017), and Level I evidence for the efficacy of (one) antidepressant medication (fluoxetine) for young people (Zhou et al., 2020). For first line treatment, the other antidepressants do not separate from placebo (see Box 18). Little is known about the moderation of the effectiveness of psychological treatments by depression severity, but antidepressant medication appears to be more effective for severe depression as compared to mild/moderate depression. One guideline recommends that evidence-supported psychological interventions (individual CBT and IPT) are considered first line for management of MDD in young people of all levels of severity, with potential addition of fluoxetine being considered when depression is moderate to severe, or when a psychological intervention has been trialled and found ineffective (NICE, 2005). An update to the guideline (NICE, 2019b) recommends family therapy (specifically Attachment Based Family Therapy) as an alternative if the developmental needs of the young person cannot be met by CBT or IPT. Another guideline recommends active monitoring for mild depression, while moderate-severe depression is managed in the first instance with pharmacotherapy and/or psychological intervention. If symptoms are unresponsive to one treatment modality the other treatment modality should be added (Cheung et al., 2018).
Efficacy and safety of antidepressants in children and adolescents.
Rank order of efficacy (highest to lowest) relative to placebo:
Fluoxetine,a duloxetine, venlafaxine, mirtazapine, sertraline, citalopram, escitalopram, paroxetine, imipramine, amitriptyline, clomipramine, nortriptylineb
Rank order of discontinuation (least to most) relative to placebo:
amitriptyline, fluoxetine, citalopram, clomipramine, mirtazapine, paroxetine, escitalopram, duloxetine,b sertraline, venlafaxine,b imipramineb
Rank order of suicidal ideation or behaviour (least to most) relative to placebo:
imipramine, clomipramine, escitalopram, duloxetine, fluoxetine, citalopram, paroxetine, sertraline, venlafaxineb
aSignificantly better than placebo.
bSignificantly worse than placebo (Cipriani et al., 2016).
The latter recommendation is more consistent than the first with current trial evidence, which finds a faster time to response with antidepressants than psychological therapy, offset by higher rates of suicidal ideation (see Recommendation Box 7). In contrast to studies in adults (see Box 15), combined therapy is not superior to psychological or pharmacological monotherapy for first line treatment of adolescents with MDD (see Recommendation Box 7). For unresponsive MDD clinicians with child/adolescent expertise can consider switching to another SSRI or a non-SSRI antidepressant. Augmentation with a mood stabiliser or psychological therapy may be most effective if instituted at the same time as the antidepressant switch (Emslie et al., 2010). Young people prescribed antidepressants of all kinds must be closely monitored for emergent suicidality, hostility, agitation, mania and unusual changes in behaviour (NICE, 2019b) (see Table 16).
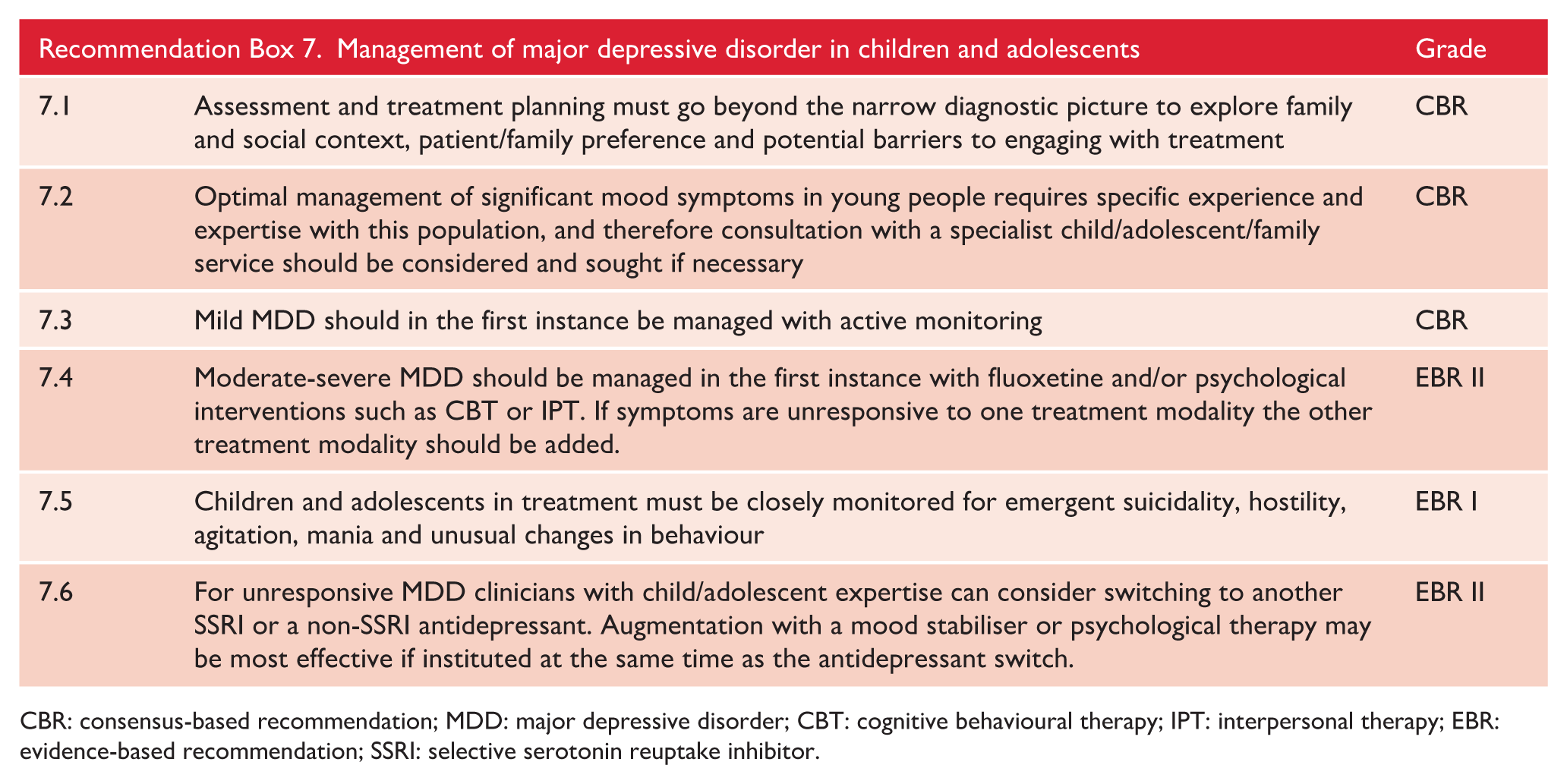
CBR: consensus-based recommendation; MDD: major depressive disorder; CBT: cognitive behavioural therapy; IPT: interpersonal therapy; EBR: evidence-based recommendation; SSRI: selective serotonin reuptake inhibitor.
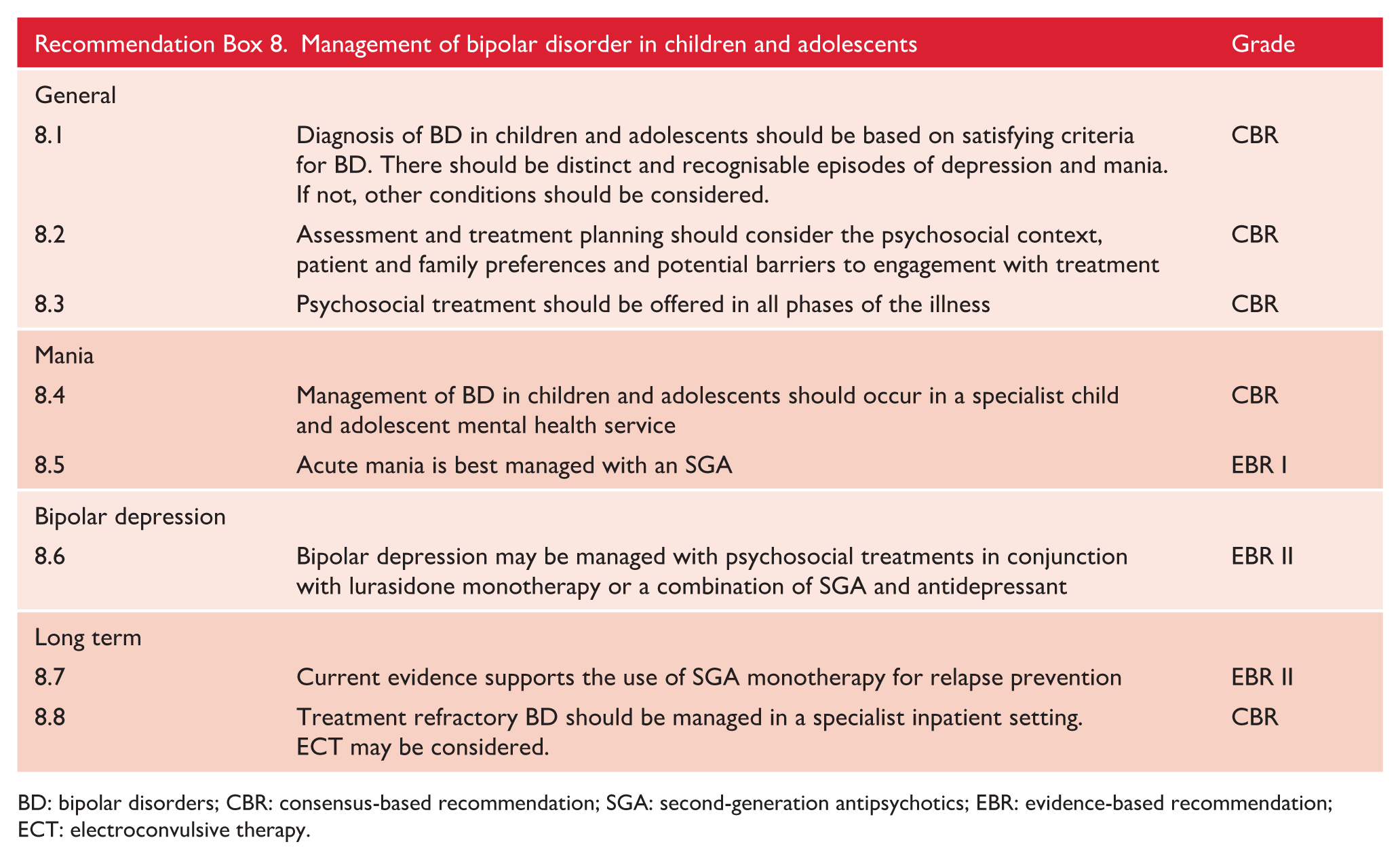
BD: bipolar disorders; CBR: consensus-based recommendation; SGA: second-generation antipsychotics; EBR: evidence-based recommendation; ECT: electroconvulsive therapy.
Psychological therapy versus antidepressant and combined treatments for depression in children and adolescents.
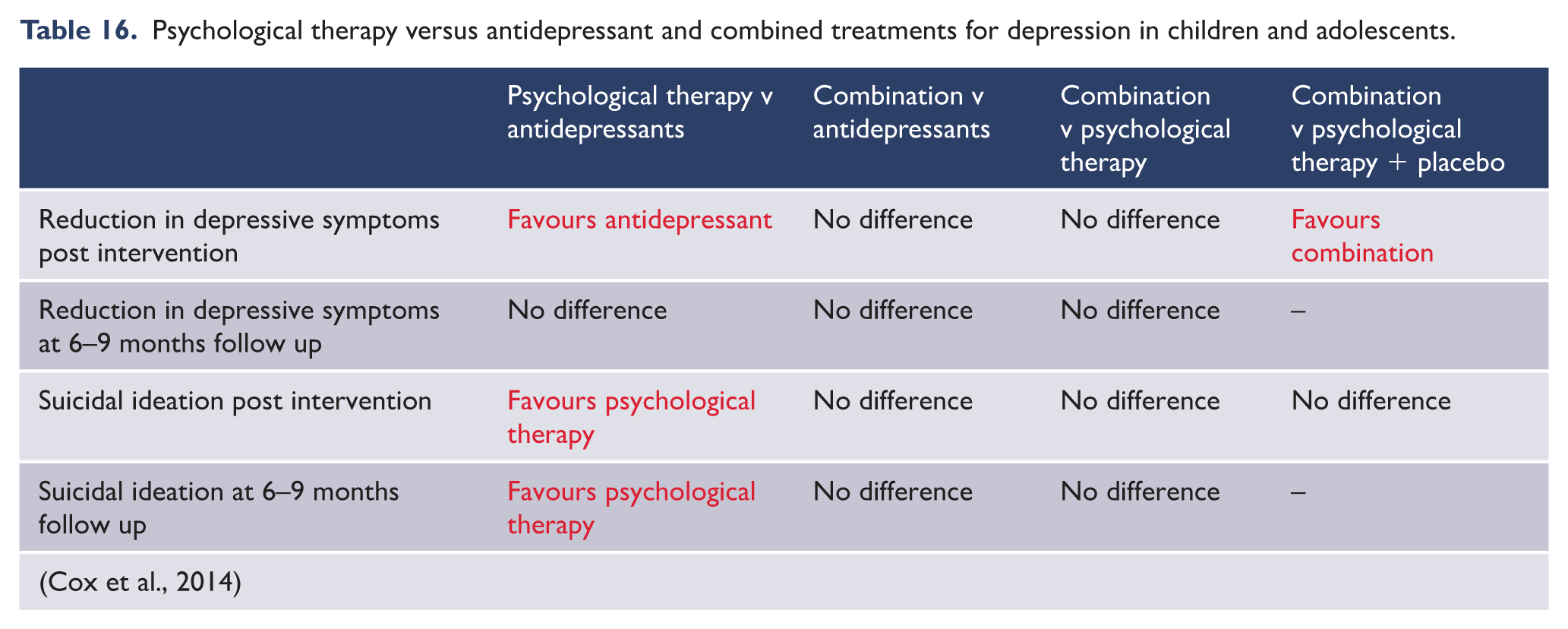
10.2. Bipolar disorder
The DSM-5 has explicitly reserved BD I for episodic presentation of bipolar symptoms (with mania taken to include states defined by severe irritability). Non-episodic presentations of severe irritability are thought to be common, and are now captured by the contentious DMDD diagnosis (see section 10.3, ‘Disruptive mood dysregulation disorder (DMDD)’) (American Psychiatric Association, 2013). Importantly, outcome studies have generally not supported children and adolescents with ADHD or dysregulated mood symptomology being at increased risk of developing BD in adulthood (McClellan et al., 2007). Reviews and practice guidelines recommend a combination of pharmacotherapy and psychosocial intervention for adolescents with BD (Fristad and MacPherson, 2014) (see ‘Psychological treatments’ in section 6.1). The existing empirical literature disproportionately focuses on the treatment of manic and mixed episodes, at the expense of bipolar depression and prophylactic treatment. Children and adolescents with BD typically have pre-existing conditions such as attention deficit hyperactivity disorder, conduct disorder or anxiety problems (Estrada-Prat et al., 2019). Those presenting for acute treatment of mania may not have new or recent onset problems, but rather a worsening of difficulties that have been present for some time. As such, samples of children and adolescents recruited for acute treatment studies are not directly comparable with samples of adults recruited for clinical trials of acute mania.
Empirical evidence supports the use of a range of SGAs in the treatment of acute mania in adolescents. Little separates these drugs in terms of efficacy or time to response (see Box 19) therefore treatment choice is informed largely by side effect profile and accessibility. In contrast, no trial evidence to date supports the use of non-SGA monotherapy for the treatment of mania. The benefit of combined pharmacotherapy is also uncertain. Adding quetiapine to valproate was superior to valproate alone, but this may simply reflect the adding of an effective treatment to an ineffective treatment.
Efficacy and safety of treatments for bipolar disorder in children and adolescents.
SGAs: aripiprazole, asenapine, olanzapine, quetiapine, risperidone, ziprasidone are superior to placebo
Non-SGA: valproate, oxcarbazepine, topiramate did not separate from placebo. No data available for other non-SGA drugs
Combination: valproatea plus quetiapine superior to valproate alone
Lurasidone, olanzapine plus fluoxetine superior to placebo
Quetiapine did not separate from placebo
CBT, DBT, family focused treatment reduce depression in young people with bipolar disorder
Aripiprazole increased time to relapse
Lamotrigine augmentation did not increase time to relapse
No difference found between lithium and valproate
SGAs: second-generation antipsychotics; CBT: cognitive behavioural therapy; DBT: dialectical behaviour therapy.
aValproate is contraindicated in females of reproductive age.
Lurasidone monotherapy, olanzapine plus fluoxetine, and several psychosocial treatments have been found effective in improving depression symptoms in bipolar disorder, while quetiapine was no better than placebo. Other treatments popularly promoted such as lamotrigine monotherapy have not been evaluated in clinical trials in young people.
Aripiprazole is superior to placebo in relapse prevention, while lamotrigine is not. Lithium and valproate are inseparable from each other, but it is not known whether either are superior to placebo in preventing relapse. Expert consensus recommends the use of adjunctive psychosocial intervention. Patients’ families should be provided with psychoeducation concerning aetiology, symptoms, course, medications, risk and protective factors and effective treatments. Adolescents should receive skills training around communication, problem solving, CBT and emotion regulation skills. Both families and adolescents should be trained in relapse prevention strategies. To maximise engagement, it is useful to encourage the young person to set positive, personally relevant goals for treatment, such as improved relationships with peers. More broadly, it is important for clinicians to be sensitive to the developmental context of adolescent BD, ensuring treatment is meaningful to the young person (Murray, 2019b). An example of a structured intervention that contains all these elements is Family-Focused Treatment for Adolescents (Miklowitz et al., 2004).
Children and adolescents with treatment refractory bipolar disorder would usually be treated in an inpatient setting, preferably a high severity or intensive care unit with staff skilled in the care of children and adolescents who are severely unwell. The National Institute for Health and Clinical Excellence (National Collaborating Centre for Mental Health, 2006) and American Academy of Child and Adolescent Psychiatry (McClellan et al., 2007) both recommend electroconvulsive therapy (ECT) for refractory mania and bipolar depression, although there is an absence of empirical evidence to support the practice. Indicators for ECT include physical compromise owing to dehydration, malnutrition and exhaustion, severe and persistent psychotic symptoms and unrelenting suicidality (see Table 12).
10.3. Disruptive mood dysregulation disorder (DMDD)
The definition and clinical meaningfulness of DMDD has been robustly questioned (see section 2 ‘Classification’). The diagnosis was introduced in DSM-5 in 2013 and so its evaluation is in its infancy. A systematic review identified only one completed RCT in which participants were specifically assessed using DMDD criteria (as opposed to the older unofficial condition severe mood dysregulation) (Benarous et al., 2017). Preadolescent children (n = 43) were randomised to receive Dialectical Behaviour Therapy or TAU. There was a higher response rate in the experimental group compared with the control group, but no significant difference in remission rate (Perepletchikova et al., 2017). Most classes of psychotropic agent have been considered as potential treatments for DMDD (Tourian et al., 2015) but no RCTs have yet been reported.
10.4. Transition to youth or adult mental health services
Owing to the recurring and sometimes chronic nature of mood disorder, many young people who have experienced a mood disorder will need treatment beyond the age at which they are eligible for child and adolescent mental health services (CAMHS) (see section 7 ‘Management of major depressive disorder’). In such cases transition to a youth or adult focused mental health service (AMHS) is necessary. Evidence suggests that this is often not executed very well. Optimal transition requires continuity of care; a period of parallel care or joint working between CAMHS and AMHS; at least one transition planning meeting, involving the adolescent, with or without a carer, and key AMHS and CAMHS professionals, prior to the handover of care; and information transfer (Paul et al., 2013). A systematic review found only 4–13% of cases examined achieved at least two of the four criteria for optimal transition (Appleton et al., 2019). NICE (2019b) recommend that if a young person aged 17 is recovering from an episode of mood disorder CAMHS should continue treatment to remission, even when the person turns 18 years of age. If the condition is non-remitting transition should commence, adhering to the above principles, prior to the 18th birthday. Young people in transition should receive comprehensive information about the management of their mood disorder in AMHS.
11. Complex presentations and special populations
A diagnosable mood disorder is only one feature of an individual for whom the guidelines provide treatment advice. Important moderators of outcome are recognised at many biological, psychological and social levels, encompassing, for example, comorbidities, individual differences, strengths, preferences and membership of minorities. This section attends to the assessment and management of a small number of these complexities, featured because of their prominence as clinical issues in everyday practice when tackling mood disorders. Note, where separate guidelines are available these have been referenced and it is advised that these also be consulted.
11.1. Complex presentations
Personality disorders
Psychological treatment
Several psychological approaches have been specifically developed for borderline personality disorder. These include Dialectical Behaviour Therapy (DBT) (Linehan, 1993), CBT (Beck and Freeman, 1990) and modified psychodynamic treatments, such as mentalisation-based therapy (MBT) (Batemann and Fonagy, 2004) and transference-focused therapy (TFT) (Clarkin et al., 1999). Each approach appears to be more effective than TAU for BPD related problems. Trials of direct comparisons between these brands of therapy have reported few differences (Giesen-Bloo et al., 2006; Kliem et al., 2010; Stoffers et al., 2012), and a recent comprehensive meta-analysis was unable to identify any differences between types of psychotherapy (Cristea et al., 2017). Importantly, the authors of this meta-analysis also noted that the overall effects of treatment are generally small, inflated by risk of bias and publication bias and are unlikely to be sustained at follow up.
Despite the high rates of comorbidity between BPD and mood disorder, no treatment studies have specifically investigated such patients. CBT has consistently been demonstrated to be effective in depression, but somewhat surprisingly it was not superior to control conditions in the five studies which have looked at this in patients with BPD (Cristea et al., 2017). Generally, psychodynamic forms of therapy target longstanding interpersonal and intrapersonal problems but there are no RCTs and only anecdotal evidence to suggest that they may be of some help. Schema focused therapy was originally developed for personality disorders but only one study to date has shown a concurrent improvement in depressive symptoms (Bamelis et al., 2014). Thus, while there is clinical consensus that diagnosing BPD in patients with mood disorders and offering concurrent treatment for both disorders is desirable it is difficult to make specific recommendations about which psychological treatment to offer. The length of psychological treatment may also be relevant, as most treatments for depression deliver 12–16 sessions over 3–6 months while treatments for personality disorders are usually administered for more than a year. Again, no studies have evaluated this aspect of therapy and so no definitive guidance can be offered.
In the context of management, it is important to note that the relationship between comorbid personality disorders and mood disorders is dynamic. Consequently, individuals with personality disorders often have mood episodes that necessitate suitable treatment. Conversely, individuals with mood disorders commonly experience co-existing personality difficulties that are often overlooked – resulting in the inadequate treatment of personality disorders. Thus, it is important that clinicians regularly review both mood and personality symptoms and carefully consider the possibility of both.
Comorbid medical illness
Patients with mood disorders and comorbid medical conditions are at greater risk of polypharmacy and various iatrogenic complications (Holvast et al., 2017). Vigilance for medication interactions, including pharmacokinetic issues where hepatic or renal impairments exist, is needed. Vigilance for pharmacodynamic interactions is also needed, especially where multiple serotonergic agents or sedating agents are prescribed. Minimising polypharmacy is advisable with regular medication review recommended (Holvast et al., 2017).
In patients with cardiovascular conditions – where clinically appropriate – it is prudent to avoid agents associated with prolonged QTc interval and agents with high risk of inducing/aggravating metabolic syndrome (Fu et al., 2019b). In chronic pain patients tricyclic antidepressants are traditionally first line when used in pain control, but literature now supports use of duloxetine (Leo and Khalid, 2019; Nicholas et al., 2009). In highly complex patients with severe medical and mood comorbidities, close communication between involved specialists is prudent. Optimising mental state outcomes must not be neglected, as optimal mental state outcomes will enhance overall engagement with care and overall health outcomes.

EBR: evidence-based recommendations; CBR: consensus-based recommendations.
Intellectual disability
In the presence of intellectual disability, it is important to note that the presentation of mood disorders can be very different and can present with behavioural features including aggression and irritability. Standard frameworks for classification may not capture the clinical picture and some modifications to diagnostic criteria may be necessary. The assessment process may also need to be modified such as the use of shorter clinical interview sessions, assessing patients in their usual environment (e.g. at home), assessing capacity to report symptoms, using simple language and visual cues. Collateral history and information is particularly important and the observations from carers and families are of critical importance. Where possible the same interventions as for other people with mood disorders should be provided but the method of delivery and duration of treatment may need to be adjusted to account for the disability. Issues relating to capacity and consent may also need to be formally considered and it may be useful to consult with an intellectual disability specialist. Several resources have been developed for the management of mental health in this population (see www.healthymind.org.au/#, www.idhealtheducation.edu.au/ and www.3dn.unsw.edu.au/sites/default/files/documents/Accessible-Mental-Health-Services-for-People-with-a-ID-A-Guide-for-Providers.pdf).
11.2. Age and life stage considerations
Pregnancy and post-partum
The management of mood disorders over the perinatal period was reviewed in the MDcpg2015 and there have been no studies to change the guiding principles. Furthermore, comprehensive guidelines on mental health care over the perinatal period (Austin et al., 2017) have been published since the MDcpg2015 that provide more detail about the management of perinatal depression.
MDD is common both during pregnancy and postpartum (Howard et al., 2014); a key point is that perinatal depression can have an adverse impact on the developing foetus (Stein et al., 2014) and also an adverse impact on the mother-infant relationship that increases the risk of the child developing mental disorders in later life. Women with a severe, or psychotic, postpartum depression are at risk of infanticide and/or suicide (Austin et al., 2007), making this a psychiatric emergency that requires prompt and assertive treatment. This highlights the importance of recognising and treating perinatal depression.
Psychological treatments are part of the management of all depressions, and, because they do not expose the developing foetus or nursling to medications, should be offered to all women with a perinatal depression. In the case of postnatal depression, a recent systematic review and meta-analysis concluded that CBT monotherapy is sufficient to improve symptoms and quality of life in postnatal depression (Huang et al., 2018), consistent with an early systematic review of intervention in primary care settings (Stephens et al., 2016). In both reviews, benefits were both immediate and maintained in the medium term.
Prescribing medications to women over the perinatal period (pregnancy and postpartum) requires a careful analysis of the benefits of the medication for the woman as well as risks of exposing the foetus to potential, but small teratogenic risks of the medication, recognising that the rate of congenital abnormalities in the population is around 3% (Abeywardana and Sullivan, 2008), and that there is a risk of the infant experiencing poor neonatal adaptation syndrome, especially with paroxetine. Small amounts of antidepressants pass through the breastmilk and so nursing infants may be exposed to them, however, most antidepressants are considered to be safe for breastfeeding.
In light of these risks, antidepressant medication should be reserved for those women with more severe depression or where psychological treatment has been ineffective or is not appropriate.
The perinatal period is a time of risk for women with bipolar disorder, first, because there is a high risk of relapse following childbirth and second, as they may be on maintenance medication that could be harmful to the developing foetus. Women should be informed about this when they are first diagnosed. Women with BD are at risk of unplanned pregnancy, especially in association with disinhibited behaviour when manic, for this reason, a discussion about contraception should take place at the time of diagnosis.
Childbirth can trigger a relapse in around 66% of women with BD, this often arising in the first month following delivery. This risk can be reduced to around 23% if prophylactic medication, in particular lithium, is used (Wesseloo et al., 2016). Relapse is increased among women with a more severe illness and recent episodes. Ideally, women with BD should have pre-pregnancy counselling prior to conceiving to discuss the risks of treatment and the need for prophylaxis.
Mood stabilisers can be teratogenic, and these risks have been reviewed in depth (Khan et al., 2016). Sodium valproate is of particular concern, as 10–11% of infants exposed in utero will have major congenital malformations (Tomson and Battino, 2012) and are at risk of significant intellectual impairment (Gentile, 2014). Hence, as per the MDcpg2015, we recommend that valproate not be used as a first line mood stabiliser in women of child bearing age (Malhi et al., 2015).
Carbamazepine is also linked to foetal abnormalities (but not with intellectual impairment) and should not be used during pregnancy (Tomson and Battino, 2012). Lamotrigine has a lower risk during pregnancy (and can be prescribed to breastfeeding mothers), with reports of 2.3% congenital malformations; however, it has limited efficacy in preventing mania.
In planning for pregnancy, it is important to aim for optimal mood stability and the use of monotherapy if necessary. Lithium is the preferred mood stabiliser (Malhi et al., 2015, 2020a, 2020d) with demonstrated efficacy in the prophylaxis of postpartum relapse (Bergink et al., 2012; Wesseloo et al., 2016) and should be considered for women with severe BD. Valproate and carbamazepine should be avoided. If already taking lithium this should be continued although the dose should be reduced in the first trimester to 500 mg per day. In the second and third trimesters lithium should be prescribed in divided doses and a trough level of 0.6 mmol/L should be achieved if possible.
Before birth, lithium dosage can be reduced 2 weeks prior and reintroduced soon after. Note, because lithium will be readily secreted into breastmilk, breastfeeding should be avoided. Lithium is also a useful mood stabiliser for peripartum disorders and should be given consideration even in those women new to lithium therapy. Again, a clinical risk benefit analysis is needed on a case by case basis.
While lithium provides effective prophylaxis, it does carry with it a risk of cardiac anomalies, however, the rates (odds ratio 4.75) are lower than previously thought (Diav-Citrin et al., 2014). If lithium is used during pregnancy foetal echocardiography and level-2 ultrasound are recommended (Bergink and Kushner, 2014). Monitoring the serum lithium levels regularly during pregnancy is essential as the serum levels can change with the changing maternal fluid volume, to maintain a therapeutic level of 0.6–0.8 mmol/L (Nolen et al., 2019; Poels et al., 2018).
Again, while lithium is effective for prophylaxis, breastfeeding while taking lithium is not recommended (Galbally et al., 2018; Poels et al., 2018) as it passes into breastmilk, and risks exposing the infant to its side-effects.
The second-generation antipsychotics (SGAs), such as quetiapine or olanzapine, are used as alternatives in the treatment of BD. They are generally considered to be safe in pregnancy; however, they may increase the risk for gestational diabetes and the likelihood of having a large baby (Chisolm and Payne, 2016).
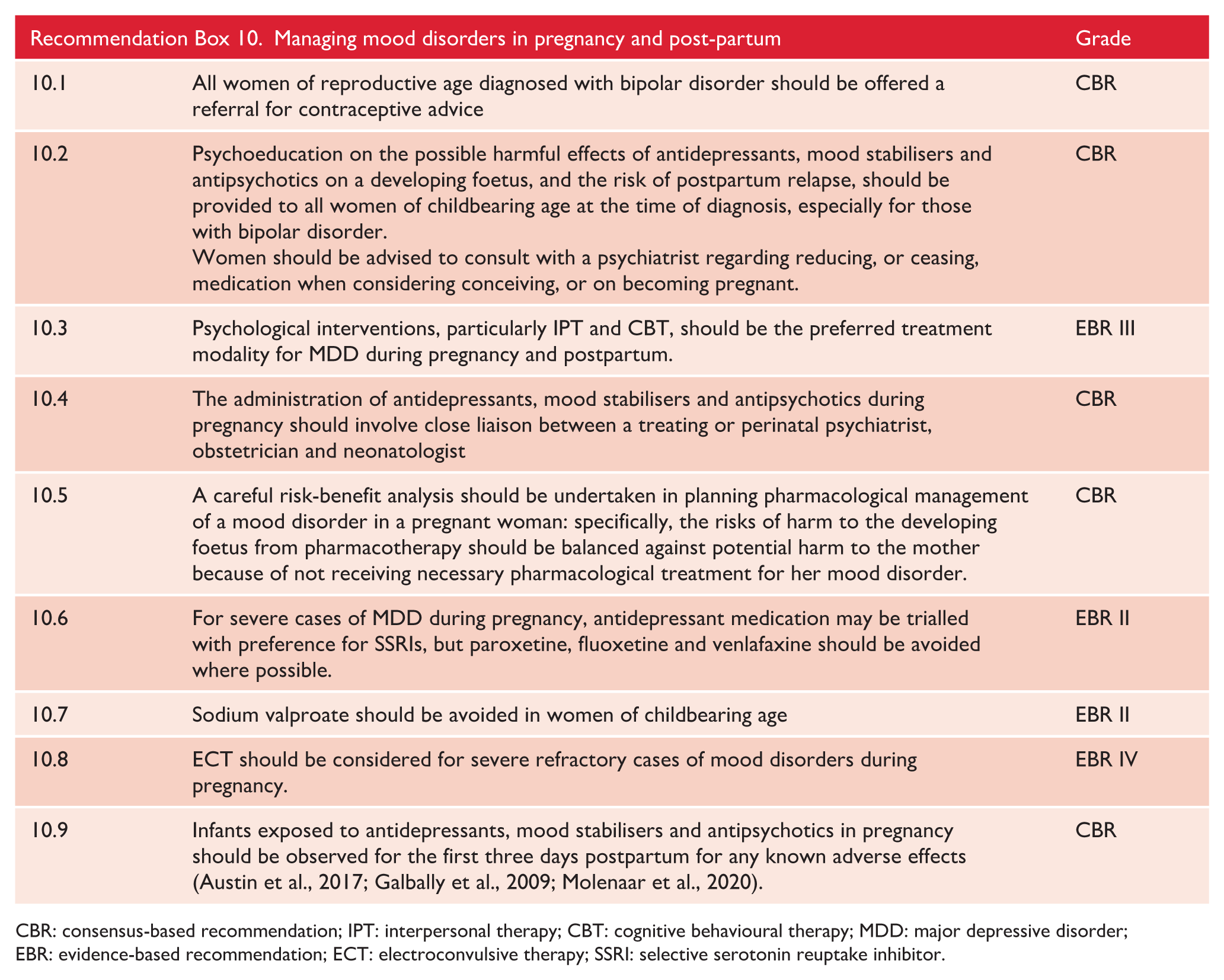
CBR: consensus-based recommendation; IPT: interpersonal therapy; CBT: cognitive behavioural therapy; MDD: major depressive disorder; EBR: evidence-based recommendation; ECT: electroconvulsive therapy; SSRI: selective serotonin reuptake inhibitor.
Menopause
Background
The menopause transition (peri-menopause) and early postmenopausal period are associated with risk for both recurrence of previous depression and new-onset depression (Bromberger et al., 2011; Cohen, 2006; Freeman, 2006). Depressive symptoms co-occur and overlap with menopausal symptoms, with hot flashes and night sweats identified as independent predictors of peri-menopausal depression (Frey et al., 2008). There are also numerous psychosocial challenges associated with the menopause transition which may contribute to a higher incidence of depression. Various factors including a history of pre-menstrual syndrome (Freeman et al., 2004), increased BMI (Maki et al., 2019), life stress (Woods et al., 2008) and a history of depression (Bromberger et al., 2011) appear to increase the risk of peri-menopausal depression. Their presence should prompt greater vigilance for depressive symptoms (Maki et al., 2019; Soares, 2019).
Assessment
When assessing women around menopausal age, clinicians should specifically elicit and assess the symptoms of menopause and psychosocial risk factors related to the perimenopause. To date, a specific rating scale for depressive symptoms in the perimenopause has not been validated. However, there are a number of validated menopause symptom and health-related quality of life scales that include mood items, that may be of use (e.g. Menopause Rating Scale, Heinemann et al., 2004; Menopause-specific Quality of Life Questionnaire, Hilditch et al., 1996). The possibility of early menopause should be considered and a menstrual history taken, especially noting any changes in menstrual function. This may be indicated by family history. Clinicians should also be aware of the additional risk of osteoporosis conferred by early menopause, which may be exacerbated by mood disorder or by medications which increase prolactin levels.
Mood swings occur more frequently during menopause transition but tend to be relatively short (Freeman et al., 2008). However, given that mood symptom severity increases in bipolar disorder late in menopause transition (Marsh et al., 2015) mood symptoms need to be evaluated carefully in patients known to have bipolar disorder.
Treatment
Antidepressants remain the first line pharmacological treatment for depression in peri- and postmenopausal women. Only desvenlafaxine has been studied sufficiently and proven efficacious for peri- and postmenopausal depression in two large RCTs (Clayton, 2013; Kornstein, 2010). However, there are also small open label studies that suggest efficacy for SSRIs, SNRIs and vortioxetine (Maki et al., 2019). Thus, based on data to date, it is recommended that medications be chosen based on prior efficacy and/or tolerability.
Evidence for oestrogen therapies
A recent systematic review that examined the efficacy of oestradiol in the treatment of depression (Rubinow, 2015), found that it may have efficacy in perimenopausal but not postmenopausal depressed women, consistent with the ‘critical window’ hypothesis, which suggests that the beneficial effects of oestradiol are observed only when it is given prior to cessation of ovarian activity (Soares, 2019).
A recent, high-quality RCT that investigated the efficacy of hormonal therapy (transdermal estradiol plus intermittent micronised progesterone) in preventing the onset of depressive symptoms in euthymic perimenopausal and early postmenopausal women (Gordon et al., 2018), found that women receiving active treatment were significantly less likely to develop depressive symptoms, compared with those receiving placebo (32.3% vs 17.3%). However, this requires replication. At present, we do not recommend oestrogen as prophylaxis of major depression in the perimenopause or menopause. In women who have previously presented with significant mood symptoms perimenstrually or had severe post-partum onset episodes of depression, the use of oestrogen prophylactically could be considered.
In women presenting with depression who are not already taking an antidepressant, it has been suggested that if there are significant menopausal and concurrent depressive symptoms, a brief trial (2–4 weeks) of monotherapy with transdermal estradiol may be helpful (Soares, 2019). Following this there should be an evaluation of the benefits and tolerability of hormone treatment, and it may then be necessary to consider augmentation with an antidepressant or switching to one altogether. However, we consider that the evidence at this point favours starting an antidepressant over first line treatment with oestrogen in this situation.
In peri and post-menopausal women already treated with antidepressants, without response, there is some evidence that estrogen-based therapy may augment the response to antidepressants. However, this should be limited to women with significant concurrent menopausal symptoms (Maki et al., 2019). If prescribing hormone replacement therapy (HRT), relevant risks and benefits must be discussed. Potential adverse effects include increased risk of venous thromboembolism and stroke with oral but not transdermal HRT (NICE guidelines 2015). A recent meta-analysis found a small but statistically significant increased risk of breast cancer associated with menopausal hormone therapy (Collaborative Group on Hormonal Factors in Breast Cancer 2019).
Evidence for psychological treatments of depression in menopause remains limited. One early review investigating cognitive-behavioral, behavioral and mindfulness-based treatments (Green et al., 2015) found only two relevant studies, both with positive results. The review was then expanded to include studies that had included depression symptoms as an outcome measure, and found some evidence that interventions targeting menopausal symptoms may be beneficial for depressive symptoms in the mild-moderate range. Psychological treatments are part of the management of all depressions (see above), and clinical tips for tailoring CBT to the physical and psychological features of menopause can be found at www.womens-health-concern.org/help-and-advice/factsheets/cognitive-behaviour-therapy-cbt-menopausal-symptoms/.

CBR: consensus-based recommendations.
Older people
Medication issues
Paroxetine has a strong evidence base (Reynolds et al., 2006) but is more anticholinergic than other SSRIs and patients are more prone to develop discontinuation effects if it is stopped suddenly (Coupland et al., 1996). The risk of QTc prolongation and associated tachyarrhythmia is likely to be greater in the elderly, in patients with cardiovascular and other co-morbidity and in those on other pharmacotherapy, especially polypharmacy (Franchi et al., 2016). It is suggested that all antidepressants may carry this risk and that therefore QTc and clinical symptoms of arrhythmia should be monitored for all antidepressants (Aronow and Shamliyan, 2020). There is debate regarding whether particular agents are more problematic in this regard. Among second generation antidepressants, it has been suggested that citalopram and escitalopram are differentially associated with QTc prolongation and arrhythmias (Qirjazi et al., 2016). However, it has been suggested that this risk exists also with other antidepressants (Aronow and Shamliyan, 2020). Where antidepressants are to be used in higher than standard doses in the elderly, baseline and steady state QTc monitoring is recommended. The maximum recommended dose of citalopram is 20 mg per day for patients older than 60 years of age (maximum dose 40 mg those under age 60) (US Food & Drug Administration, 2012). However, the risks and benefits of increasing the dose of citalopram in an elderly patient with a signal of benefit, compared with switching to another antidepressant should be considered.
Hyponatraemia is an important complication of antidepressant therapy in the elderly. The risk appears to be higher with SSRIs and SNRIs than with TCAs or NaSSAs. The risk is highest immediately after starting antidepressants and after 6 months is negligible (Lien, 2018). Urea and electrolytes should be measured at baseline and after 2 weeks of therapy.
Two antidepressants have been suggested to have cognitive enhancing properties in those with old age depression. Both duloxetine and vortioxetine (Katona et al., 2012; Raskin et al., 2007) have demonstrated improvements in verbal learning in elderly depressed patients, while vortioxetine has shown an improvement in a task measuring vigilance and processing speed. If these drugs are readily available and familiar to the practitioner they are suitable treatment options.
The benefits of lithium in severe mood disorders, and the risks of discontinuing treatment in those with bipolar disorder should be considered carefully. However, lithium should be used with greater caution in the elderly given increased susceptibility to worsening of cognitive function which can occur even at low serum levels (Malhi et al., 2016c; Shulman et al., 2019). There are concerns regarding ECG changes and cardiac safety with lithium therapy, which should prompt regular ECG monitoring (Mehta and Vannozzi, 2017).
Psychotherapy
Studies have suggested that both cognitive behavioural therapy (CBT) and interpersonal therapy (IPT) are effective in the treatment of depression in the elderly (Cuijpers et al., 2006; Reynolds et al., 1999). In the acute phase of the illness the largest trial of a specific psychological treatment designed to help with cognitive problems was of problem solving therapy for depressed elderly patients with executive dysfunction (Alexopoulos et al., 2011). Problem solving therapy, compared with a control treatment significantly improved overall functioning over 12 months. However, it did not improve cognitive function. There is no evidence regarding psychotherapy for bipolar disorder specific to the elderly.
11.3. Special populations
Refugees and immigrants
Refugees are at markedly higher risk of developing mood disorders because of prior exposure to multiple trauma, forced separation from family and social networks, detention and post-migration difficulties (Charlson et al., 2019). Mood disorders in refugees commonly occur with comorbid PTSD, prolonged grief, adjustment reactions and somatic disorders. Although there is increasing evidence of psychological treatments for refugees that are based on trauma-focused cognitive behavior therapy principles, such as Narrative Exposure Therapy, these are largely focused on reducing PTSD symptoms (Neuner et al., 2008). The evidence base for treatments that specifically target mood disorders in refugees is insufficiently robust, although there is evidence that Interpersonal Psychotherapy can reduce depression in refugees (Bolton et al., 2007).
The assessment and treatment of refugees and immigrants is complicated because of cultural and language differences and it is important that clinicians are sensitive to the cultural context within which the patient is presenting. Some cultural and religious groups are likely to under-report suicidal ideation, and may be sensitive to questions regarding libido, or history of sexual assault. Clinicians also need to be aware of issues concerning gender, and some patients may be better assessed and treated by a clinician of the same sex. Clinicians also need to be aware of the need to work with professionally trained interpreters, and the challenges that this can introduce in terms of ensuring that interpreters can reliably convey subtle meanings of questions and answers to allow the clinician to make the optimal clinical judgements.
Physical health in indigenous populations
Premature mortality is increased in bipolar disorder, partly related to an increased rate of cardiovascular disease (Correll et al., 2017; Cunningham et al., 2014; Druss et al., 2011; Firth et al., 2019b). There is also clear evidence of reduced life expectancy among those with severe mental illness in New Zealand (Cunningham et al., 2014). Higher levels of cardiovascular risk factors such as metabolic abnormalities have been seen among those from minority ethnic populations who experience severe mental illness (Carliner et al., 2014; De Caluwé et al., 2019). A recent study also suggests that in New Zealand, Māori with bipolar disorder had a higher level of physical morbidity and a higher risk of death from natural causes compared with non-Māori with bipolar disorder (Cunningham et al., 2020). These data indicate a particular need to focus on physical health in indigenous people with bipolar disorder and to be aware of the interaction between ethnicity, bipolar disorder and the metabolic risks associated with some medications for bipolar disorder.
Supplemental Material
sj-docx-1-anp-10.1177_0004867420979353 – Supplemental material for The 2020 Royal Australian and New Zealand College of Psychiatrists clinical practice guidelines for mood disorders
Supplemental material, sj-docx-1-anp-10.1177_0004867420979353 for The 2020 Royal Australian and New Zealand College of Psychiatrists clinical practice guidelines for mood disorders by Gin S Malhi, Erica Bell, Darryl Bassett, Philip Boyce, Richard Bryant, Philip Hazell, Malcolm Hopwood, Bill Lyndon, Roger Mulder, Richard Porter, Ajeet B Singh and Greg Murray in Australian & New Zealand Journal of Psychiatry
Footnotes
Declaration of Conflicting Interests
G.S.M. has received grant or research support from National Health and Medical Research Council, Australian Rotary Health, NSW Health, American Foundation for Suicide Prevention, Ramsay Research and Teaching Fund, Elsevier, AstraZeneca, Janssen-Cilag, Lundbeck, Otsuka and Servier; and has been a consultant for AstraZeneca, Janssen-Cilag, Lundbeck, Otsuka and Servier. P.B. has received research support from the National Health and Medical Research Council, speaker fees from Servier, Janssen and the Australian Medical Forum, educational support from Servier and Lundbeck, has been a consultant for Servier, served on an advisory board for Lundbeck and has served as DSMC Chair for Douglas Pharmaceuticals. R.B. has received grant support in the last 5 years from the National Health and Medical Research Council, the Australian Research Council, TAL Insurance, and support for travel for advisory meetings to the World Health Organization. M.H. has received grant or research support in the last 5 years from the National Health and Medical Research Council, Medical Research Future Fund, Ramsay Health Research Foundation, Boehringer-Ingleheim, Douglas, Janssen-Cilag, Lundbeck, Lyndra,Otsuka, Praxis and Servier; and has been a consultant for Janssen-Cilag, Lundbeck, Otsuka and Servier. R.M. has received support for travel to education meetings from Servier and Lundbeck, speaker fees from Servier and Committee fees from Janssen. R.P. has received support for travel to educational meetings from Servier and Lundbeck and uses software for research at no cost from Scientific Brain Training Pro. G.M. has received grant support in the last 5 years from the National Health and Medical Research Council, the Mental Illness Research Fund, Victorian Medical Research Acceleration Fund, Canadian Institutes of Health Research, Readiness, SiSU Wellness and Barbara Dicker Foundation. D.B. has received funding to host webinars by Lundbeck. A.B.S. has shares/options in Baycrest Biotechnology Pty Ltd (pharmacogenetics company) and Greenfield Medicinal Cannabis, has received speaking honoraria from Servier, Lundbeck and Otsuka Australia. The authors E.B., R.B., P.H. and B.L. declared no potential conflicts of interest with respect to the research, authorship and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship and/or publication of this article.
ORCID iDs
Supplemental material
Supplemental material for this article is available online.
Notes
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
