Abstract
Objective:
Impaired regulation of the hypothalamic-pituitary-adrenal (HPA) axis and hyper-activity of this system have been described in patients with psychosis. Conversely, some psychiatric disorders such as post-traumatic stress disorder (PTSD) are characterised by HPA hypo-activity, which could be related to prior exposure to trauma. This study examined the cortisol response to the administration of low-dose dexamethasone in first-episode psychosis (FEP) patients and its relationship to childhood trauma.
Method:
The low-dose (0.25 mg) Dexamethasone Suppression Test (DST) was performed in 21 neuroleptic-naïve or minimally treated FEP patients and 20 healthy control participants. Childhood traumatic events were assessed in all participants using the Childhood Trauma Questionnaire (CTQ) and psychiatric symptoms were assessed in patients using standard rating scales.
Results:
FEP patients reported significantly higher rates of childhood trauma compared to controls (p = 0.001) and exhibited lower basal (a.m.) cortisol (p = 0.04) and an increased rate of cortisol hyper-suppression following dexamethasone administration compared to controls (33% (7/21) vs 5% (1/20), respectively; p = 0.04). There were no significant group differences in mean cortisol decline or percent cortisol suppression following the 0.25 mg DST. This study shows for the first time that a subset of patients experiencing their first episode of psychosis display enhanced cortisol suppression.
Conclusions:
These findings suggest there may be distinct profiles of HPA axis dysfunction in psychosis which should be further explored.
Introduction
The onset of schizophrenia and other psychotic disorders has been associated with increased levels of stress and hyper-activation of the hypothalamic-pituitary-adrenal (HPA) axis (Phillips et al., 2006), which may in turn negatively impact on illness course (Garner et al., 2009; Walder et al., 2000). Patients with psychosis, particularly those in the acute phase of illness, show elevated basal adrenocorticotropin hormone (ACTH) and cortisol levels (Ryan et al., 2003, 2004; Sachar et al., 1970), elevated levels of corticotropin-releasing-hormone (CRH) in the cerebrospinal fluid (Banki et al., 1987) and attenuated HPA axis negative feedback control (Lammers et al., 1995; Tandon et al., 1991). The latter is based on findings from neuroendocrine challenge tests such as the standard (1 mg) Dexamethasone Suppression test (DST) (Tandon et al., 1991) and the Dexamethasone/CRH (DEX/CRH) test (Lammers et al., 1995), which demonstrate a higher rate of cortisol non-suppression among schizophrenia patients, indicating impaired glucocorticoid receptor (GR)-mediated negative feedback of the HPA axis.
In other psychiatric and stress-related disorders, however, the HPA axis has been found to be hypo-active (Heim et al., 2009). For example, patients with post-traumatic stress disorder (PTSD) have been found to have low basal cortisol and enhanced GR-mediated negative feedback regulation of the HPA axis, shown by enhanced cortisol suppression in the low-dose (0.25 mg or 0.5 mg) DST (Griffin et al., 2005; Rohleder et al., 2004; Yehuda et al., 1993, 2004b). Traumatised veterans with or without PTSD also show enhanced cortisol suppression following dexamethasone (de Kloet et al., 2007), suggesting these HPA axis alterations may result from compensatory mechanisms following exposure to trauma. Enhanced cortisol suppression has also been reported in other disorders that are associated with a high prevalence of childhood trauma, including borderline personality disorder (BPD) (Carrasco et al., 2003), eating disorders (Díaz-Marsá et al., 2008; Gaab et al., 2002) and chronic fatigue syndrome (Gaab et al., 2002; Heim et al., 2009). Consistent with this, reduced pituitary volume (possibly indicative of pituitary hypo-function) has been reported among BPD patients with a history of childhood trauma compared to BPD patients with no history of childhood trauma (Garner et al., 2007). In patients with an eating disorder, enhanced cortisol suppression significantly correlates with the severity of childhood trauma (Díaz-Marsá et al., 2007). Furthermore, some studies have found that children exposed to severe childhood neglect or abuse have lower basal cortisol secretion (Gunnar and Donzella, 2002; Gunnar and Vazquez, 2001; Heim et al., 2008).
Although the prevalence of childhood trauma is high among patients with psychosis (Bendall et al., 2008; Read et al., 2001), few, if any, studies have investigated potential HPA hypo-function characterised by enhanced cortisol suppression in this population. Furthermore, studies are required early in the illness course as HPA axis changes have been shown in chronic psychotic disorder (Ritsner et al., 2004). The current study therefore investigated HPA axis function and the prevalence of childhood trauma in neuroleptic-naïve or minimally treated first-episode psychosis (FEP) patients using the low-dose (0.25 mg) DST. The 0.25 mg dose of dexamethasone allows the detection of abnormally high cortisol suppression. To our knowledge, the modified low-dose DST has not been previously investigated in a psychotic population. We hypothesised that FEP patients would report greater childhood trauma than controls, that enhanced cortisol suppression would be evident in a subset of FEP patients and, finally, that enhanced cortisol suppression would correlate with the severity of childhood trauma.
Methods and materials
Participants
Twenty-one FEP patients (12 male, nine female) were recruited from the Early Psychosis Prevention and Intervention Centre (EPPIC) at Orygen Youth Health based on the following inclusion criteria: (i) aged 15–25 years; (ii) outpatients or inpatients undergoing a first episode of psychosis defined as daily psychotic symptoms experienced for longer than a week that cannot be explained by other factors (e.g. organic disorder); and (iii) living in the north and north-western metropolitan region of Melbourne. Exclusion criteria were more than 10 days of treatment with psychotropic medication, IQ < 70, organic brain disorder, any significant medical illness including impaired thyroid function, polydipsia, asthma, diabetes, chronic fatigue, steroid medications, pregnancy and shift work.
Twenty healthy control participants (12 male; eight female) were recruited from similar socio-demographic areas through advertisements and seeking friends or neighbours of the patient participants. Participants were matched as closely as possible for age and sex. Potential control participants were administered the Structured Clinical Interview for DSM-IV for Axis 1 disorders (Non-patient edition) (SCID-NP: First et al., 2002a). Exclusion criteria included a current or past history of psychiatric illness or any psychiatric illness in the immediate family, in addition to the exclusion criteria described above for the patient group.
All participants gave informed consent prior to involvement in this study and the research was approved by the North Western Mental Health Research and Ethics Committee.
Clinical assessments
Clinical assessments for the FEP group were conducted upon entry to the study. Diagnoses were determined using the Structured Clinical Interview for DSM-IV for Axis I disorders (SCID I: First et al., 2002b). The presence of PTSD was assessed using the Clinician Administered PTSD Scale (CAPS: Blake et al., 1995). Positive and negative psychotic symptoms were rated using the Brief Psychiatric Rating Scale (BPRS: Overall and Gorham, 1962) and the Scale for the Assessment of Negative Symptoms (SANS: Andreasen, 1983), respectively. Depressive and anxiety symptoms were rated using the Hamilton Depression (HAMD: Hamilton, 1960) and Hamilton Anxiety (HAMA: Hamilton, 1959) rating scales.
Childhood trauma was assessed in both patient and control groups using the Childhood Trauma Questionnaire (CTQ: Bernstein et al., 1994).
Dexamethasone Suppression Test
Participants were asked to refrain from smoking, having caffeinated drinks and taking exercise prior to collection of the morning blood samples. Baseline blood samples were obtained by venipuncture at 9 a.m. for analysis of basal cortisol levels. At 11 p.m. that evening, the participant ingested a 0.25 mg dexamethasone tablet. The following morning (9 a.m.), a blood sample was obtained for the analysis of post-dexamethasone cortisol levels. Serum cortisol levels were analysed by a commercial laboratory (Gribbles Pathology, Clayton, VIC, Australia). Blood was collected within 1 day of the psychopathology assessment.
Dexamethasone radioimmunoassay
Post-DEX plasma dexamethasone levels were measured by radioimmunoassay. Reagents for the dexamethasone radioimmunoassay, including the dexamethasone tracer, dexamethasone standards, anti-serum and quality check controls were prepared and kindly supplied by the Endocrinology Laboratory, Royal North Shore Hospital, Sydney, Australia.
The dexamethasone radioimmunoassay was performed in polystyrene tubes (10 × 75). All samples and reagents were kept on ice throughout the set up of the assay. Standards and samples were assayed in triplicate and duplicate, respectively. The assay consisted of a series of eight dexamethasone standards (0.19–25 nM), two quality check controls (QC; 2.2 nM and 6.5 nM dexamethasone), a dexamethasone-free plasma sample with (Blank; B) or without (Non-Specific Binding; NSB) the addition of rabbit anti-serum (dexamethasone C-3 carboxymethylomine conjugate) and unknowns (patient and control post-dexamethasone plasma samples). The total assay volume was 400 µL. The assay was performed using three main steps: (1) dilution of a 100 µL sample in either 100 µL dexamethasone-free plasma (standards) or 100 µL assay buffer (0.1 M NaCl, 0.05 M NaH2PO4, 0.1% bovine albumin, 0.02% sodium azide; NSB, B, QC and unknowns); (2) addition of 100 µL rabbit anti-serum (diluted 50-fold in cold assay buffer) to all samples except NSB (replaced by aliquoting an additional 100 µL buffer) 3 hours prior to the addition of (3) 100 µL [1,2,4,6,7-3H]dexamethasone tracer (specific activity 70–110 Ci/mmol, diluted 75-fold in cold assay buffer) to all samples, including a separate tube for the measurement of total counts of radioactivity (TC). A pre-incubation step with anti-serum was incorporated to maximise assay sensitivity. Samples were vortexed briefly and incubated overnight (18–24 hours) at 40°C. At the end of the incubation period, 1 mL of cold 20% polyethyleneglycol (PEG) was added to all tubes in order to precipitate protein out of the solution (except TC). Samples were vortexed briefly and were centrifuged at 2400 g for 15 minutes at 40°C. Tubes were decanted and drained carefully in order to separate unbound tracer from the protein pellet. A 1 mL measure of distilled water was added to all tubes (including TC) and allowed to stand for 30 minutes at room temperature. In the interim, the samples were vortexed (except TC) until an even protein suspension was formed. All samples were transferred into plastic scintillation vials and 10 mL scintillant fluid (Ultima Gold; PerkinElmer, Waltham, MA, USA) was added to all tubes and mixed. The tubes were counted for 10 minutes using a beta counter.
The intra- and inter-assay coefficients of variation for the dexamethasone assay were 2.59% and 1.86%, respectively. The lower limit of detection of the assay was 0.05 nM.
Post-DEX plasma dexamethasone levels ranged from 0.05 nM to 3.6 nM. In a total of nine participants (four FEP; five controls), dexamethasone could not be detected (< 0.05 nM) in the post-DEX plasma sample. However, in three of these FEP participants, dexamethasone compliance was verified by nurse signature and in one of these control participants, dexamethasone compliance was verified by cortisol hyper-suppression (63% cortisol suppression). We could not, therefore, rely on post-DEX plasma dexamethasone levels as an indicator of DST compliance.
Statistical analysis
Data are presented as individual values, mean ± SD and SEM, as indicated. Clinical and sociodemographic differences between groups were analysed using independent Student’s t-test or Pearson’s chi-squared test. For the DST, repeated measures ANOVA was used with group as the between-subject factor and time (pre-DEX cortisol vs post-DEX cortisol) as the within-subject factor. One-way ANOVA was used to analyse group differences in relative cortisol suppression (i.e. cortisol decline (pre-DEX minus post-DEX cortisol) and percent cortisol suppression ((cortisol decline/pre-DEX cortisol)*100)). Based on previous literature (Díaz-Marsá et al., 2007), cortisol hyper-suppression was defined a priori as a post-DEX cortisol value below 5 μg/dL (i.e. 138 nmol/L). Fisher’s exact test was used to determine group differences in the rates of cortisol hyper-suppression. The relationship between childhood trauma and the cortisol response to DEX was analysed using Spearman’s correlation.
Results
Characteristics of the study group
Clinical and biological characteristics of the sample are shown in Table 1. There were no group differences in distribution of sex or in BMI; however, control participants were significantly older than patients (mean difference = 1.8 years). Among the FEP patients, seven had schizophreniform disorder, four had psychotic disorder NOS, three had schizoaffective disorder, two had schizophrenia, two had a substance-induced psychotic disorder, one had major depression with psychotic features, one had bipolar disorder I with psychotic features and one had delusional disorder according to DSM-IV criteria for Axis I (SCID-I). One patient had comorbid PTSD as defined by the CAPS. Eleven (52%) patients were neuroleptic-naïve at the time of blood sampling. The remaining patients had received on average 2.4 (SD = 2.8; range = 10) days of antipsychotic medication at the time of blood sampling.
Demographic, clinical and biological characteristics of the sample.
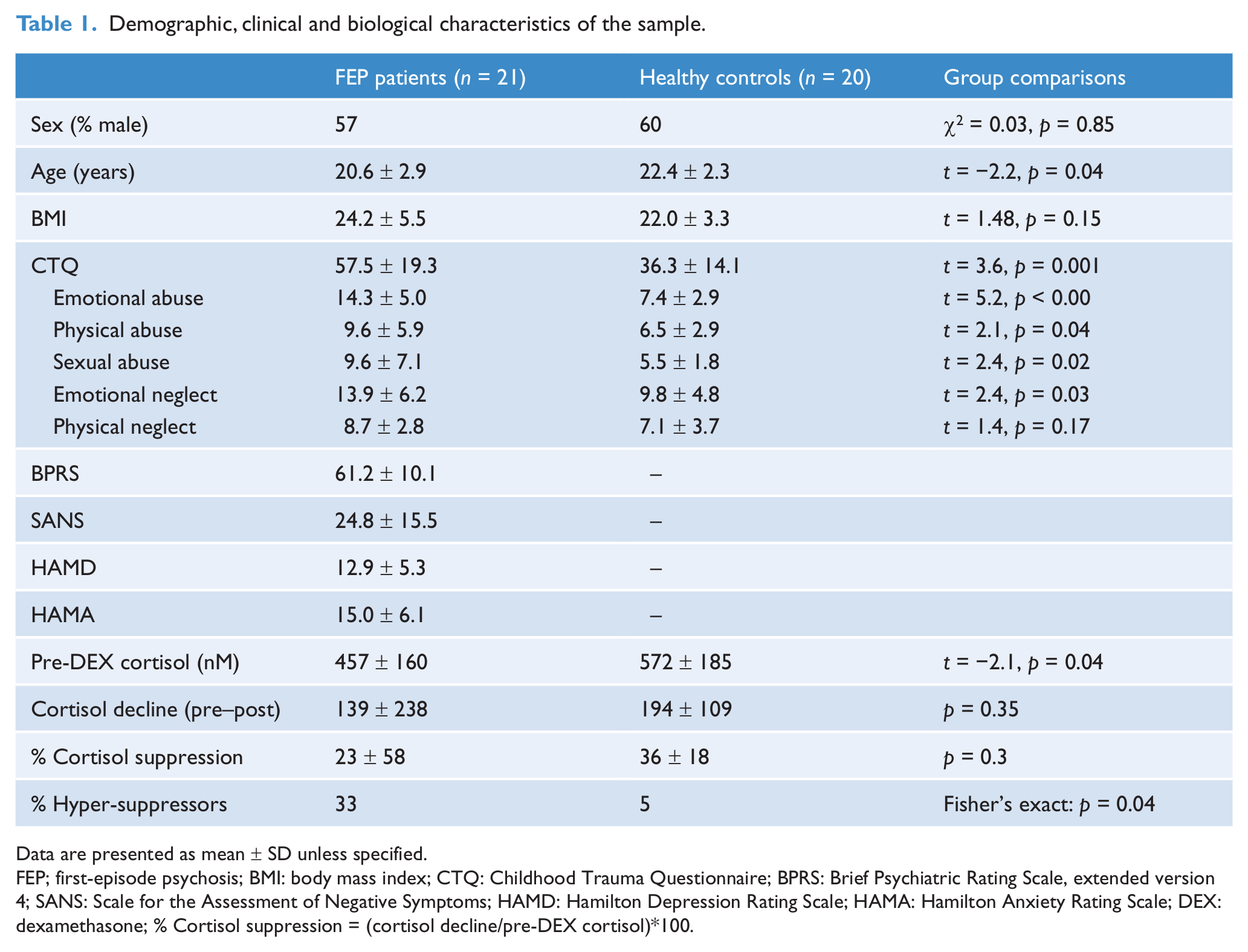
Data are presented as mean ± SD unless specified.
FEP; first-episode psychosis; BMI: body mass index; CTQ: Childhood Trauma Questionnaire; BPRS: Brief Psychiatric Rating Scale, extended version 4; SANS: Scale for the Assessment of Negative Symptoms; HAMD: Hamilton Depression Rating Scale; HAMA: Hamilton Anxiety Rating Scale; DEX: dexamethasone; % Cortisol suppression = (cortisol decline/pre-DEX cortisol)*100.
Neuroendocrine data
FEP patients had significantly lower mean pre-DEX (basal) cortisol levels compared to controls (p = 0.04; Table 1). In the DST, there was a significant main effect of time on cortisol levels (F = 32.62; df = 39; p < 0.001), with post-DEX cortisol levels being lower than pre-DEX levels (Table 1, Figure 1). However, there was no group by time effect (F = 0.86; df = 39; p = 0.36) or main effect of group on cortisol levels (F = 3.22; df = 39; p = 0.080) (Figure 1). Although there were no group differences in cortisol decline (pre-DEX minus post-DEX) (p = 0.4) or percentage of cortisol suppression ((cortisol decline/pre-DEX )*100) (p = 0.3; Table 1), the rate of cortisol hyper-suppression was significantly higher in the FEP group (p = 0.04), with seven FEP patients (three males and four females; 33%) showing cortisol hyper-suppression compared with only one male participant (5%) in the control group. Individual cortisol values for the 0.25 mg DST are shown in Figure 2.
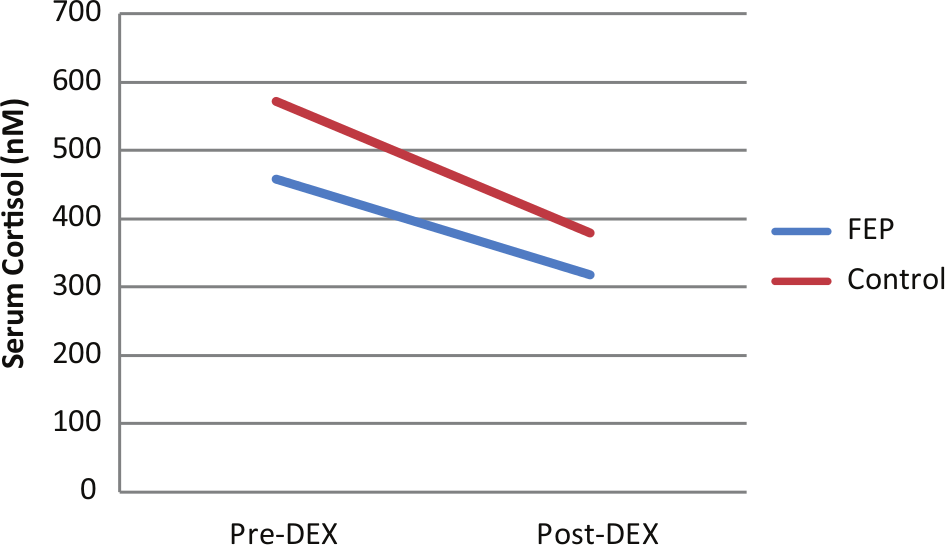
Pre and post 0.25 mg dexamethasone (DEX) cortisol levels for first-episode psychosis (FEP) patients and control participants.
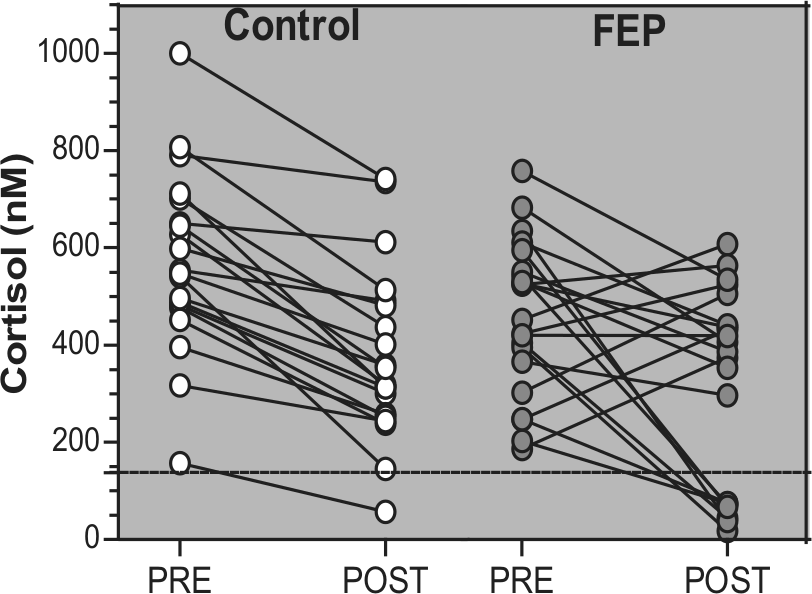
Individual serum cortisol values before (PRE) and after (POST) the administration of oral 0.25 mg dexamethasone in first-episode psychosis (FEP) patients and control participants. Cortisol suppression was defined a priori as a cut-off value of 138 nM (represented by the dashed line).
Statistical comparison of clinical variables between FEP patient non-suppressors and hyper-suppressors could not be performed owing to the small sample size. The clinical characteristics of the patient sample are shown in Table 2. Nine patients (five non-suppressors and four hyper-suppressors) had a comorbid mood or anxiety disorder, six of whom had a co-occurring mood and anxiety disorder. Comorbid major depressive disorder (MDD) was present in 21% and 29% of cortisol non-suppressors and hyper-suppressors, respectively. Only one patient (cortisol non-suppressor) had comorbid PTSD.
Clinical characteristics of first-episode psychosis (FEP) patient non-suppressors (NS) and hyper-suppressors (S).
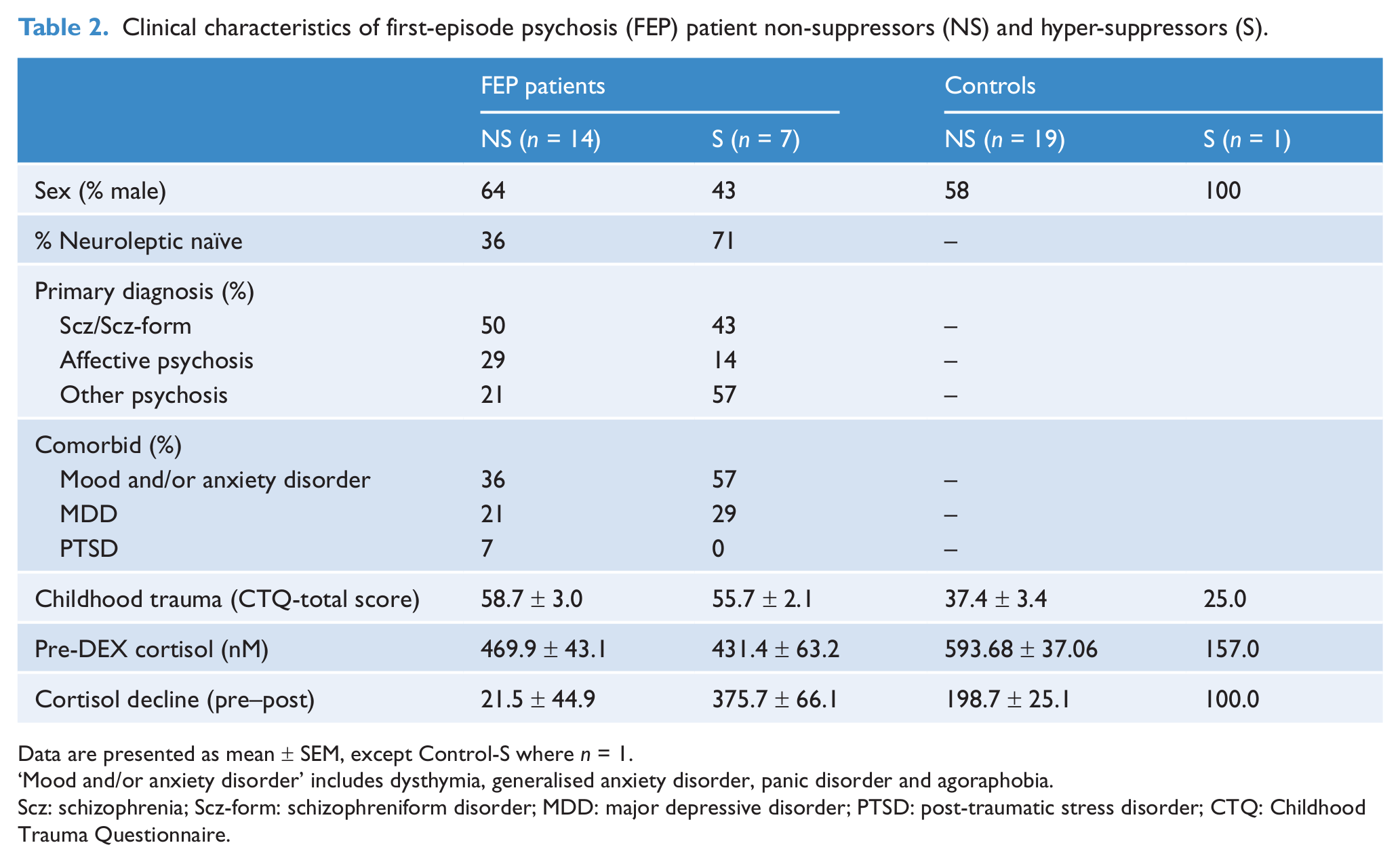
Data are presented as mean ± SEM, except Control-S where n = 1.
‘Mood and/or anxiety disorder’ includes dysthymia, generalised anxiety disorder, panic disorder and agoraphobia.
Scz: schizophrenia; Scz-form: schizophreniform disorder; MDD: major depressive disorder; PTSD: post-traumatic stress disorder; CTQ: Childhood Trauma Questionnaire.
Childhood trauma
Sixteen out of 21 FEP patients and all the control participants completed the CTQ. Total CTQ scores were significantly higher in FEP patients than in control participants (mean ± SD: 57.5 ± 19.3 vs 36.3 ± 14.1, respectively; p = 0.001) (Table 1). FEP patients also scored significantly higher on the childhood emotional abuse (p < 0.001), sexual abuse (p = 0.02), physical abuse (p = 0.04) and emotional neglect (p = 0.03) subscales of the CTQ.
Trauma and post-DEX cortisol response
The total CTQ score was not correlated with cortisol decline following dexamethasone (pre-DEX minus post-DEX) in the entire sample (r = −0.009), FEP group (r = −0.05) or control group (r = 0.06).
Discussion
The findings from this study suggest that a subset of FEP patients has a hypersensitive HPA axis response to low-dose (0.25 mg) dexamethasone administration. A significantly higher proportion of FEP patients suppressed cortisol following administration of 0.25 mg dexamethasone compared to healthy controls (33% vs 5%, respectively). This observed rate of 5% cortisol hyper-suppression in a healthy control population is consistent with previous reports (5.6%) (Díaz-Marsá et al., 2007). FEP patients in this cohort also displayed significantly lower basal cortisol levels than controls. There were, however, no group differences in mean cortisol decline (pre-DEX cortisol minus post-DEX cortisol).
This pattern of HPA hypo-function, marked by low basal cortisol and enhanced glucocorticoid sensitivity, has been frequently described in patients with PTSD (Yehuda et al., 1993, 2004a, 2004b) and more recently in patients with eating disorders (Díaz-Marsá et al., 2007, 2008), BPD (Carrasco et al., 2003; Lange et al., 2005) and other stress-related disorders including chronic fatigue syndrome (Gaab et al., 2002). To date, neuroendocrine studies in patients with psychosis have primarily been designed to detect impaired glucocorticoid-mediated negative feedback inhibition of the HPA axis by using either the standard DST or DEX/CRH challenge test. The present findings indicate that a proportion of FEP patients may in fact exhibit enhanced glucocorticoid-mediated negative feedback inhibition of the HPA axis. The observed lower basal (a.m.) cortisol levels in FEP patients in the current study is consistent with previous reports of a blunted cortisol awakening response among FEP patients (Mondelli et al., 2010; Pruessner et al., 2008), irrespective of anti-psychotic medication (Mondelli et al., 2010). Interestingly, we previously found that individuals at ultra-high risk for developing psychosis (UHR) who later developed a psychotic disorder had lower plasma cortisol levels at intake compared to UHR individuals who did not develop psychosis (Thompson et al., 2007). Based on previous and current findings, we suggest that distinct (alternate) patterns of HPA axis dysfunction may be present in subgroups of patients with early psychosis.
We were unable to compare clinical characteristics between FEP patient suppressors and non-suppressors owing to the small sample size. Cortisol hyper-suppression in response to dexamethasone in FEP patients does not appear to be influenced by comorbid PTSD as no FEP suppressors were diagnosed with comorbid PTSD. The only FEP patient who had comorbid PTSD did not show cortisol hyper-suppression. With regards to the presence of comorbid MDD, there was no apparent difference between FEP non-suppressor and hyper-suppressor subgroups (21% vs 29%, respectively). Further studies in larger samples are required to examine associations between HPA activity, symptom dimensions and comorbid disorders such as MDD and PTSD.
We hypothesised that low basal cortisol and enhanced cortisol suppression among FEP patients may be related to prior exposure to trauma (Díaz-Marsá et al., 2007; Heim et al., 2009). In the current study, FEP patients reported more childhood trauma than controls. FEP patients scored significantly higher on total CTQ scores, as well as sexual abuse, emotional abuse, physical abuse and emotional neglect subscale scores. However, childhood trauma was not correlated with the degree of cortisol suppression following 0.25 mg dexamethasone in either patients or controls. Therefore, our findings do not suggest that enhanced cortisol suppression observed in the FEP group is related to childhood traumatic experience. Previous studies have demonstrated a link between childhood trauma and hypocortisolism in both healthy and psychiatric individuals (Gunnar and Donzella, 2002; Gunnar and Vazquez, 2001; Heim et al., 2009). Some suggest that childhood trauma may be involved in the disruption of neurodevelopmental pathways, which may predispose vulnerable individuals to the development of HPA dysfunction. HPA dysregulation, in turn, may be associated with an increased risk of developing a neuropsychiatric disorder, including schizophrenia and other psychotic disorders (Read et al., 2001). Larger studies are required to investigate the link between childhood trauma and HPA function in FEP.
The experience of psychotic symptoms themselves (e.g. hallucinations, delusions) and acute psychiatric hospitalization, which often involves involuntary admission, restraint, seclusion and forced medication, can be highly traumatic for the individual (Meyer et al., 1999). Previous work by our group and others has revealed a relatively high prevalence of PTSD (up to 46%) in patients recovering from an acute psychotic episode (McGorry et al., 1991; Meyer et al., 1999). Another study by our group reported that the experience of childhood trauma increases the risk of developing post-psychosis PTSD following the initial psychotic episode (Bendall et al., 2012). Given that we did not find a link between childhood trauma and enhanced glucocorticoid sensitivity in our FEP cohort, it is still equally possible that the acute trauma following the onset of psychosis may play a role in mediating HPA dysfunction. Nevertheless, it remains unclear whether low basal cortisol and enhanced glucocorticoid sensitivity, frequently observed in patients with PTSD and our current FEP cohort, represent a consequence of traumatic events (i.e. childhood or adult post-psychosis) or a pre-existing vulnerability factor for the development of psychiatric illness. In support of the latter, low cortisol levels have been reported in the offspring of trauma survivors with PTSD, a population known to be at greater risk for PTSD (Yehuda et al., 2007). Findings in rodents suggest a blunted HPA response to stress can increase susceptibility to exhibit PTSD-like behavioural changes following a stressor, which can be prevented by administration of corticosterone (the rodent equivalent of cortisol) prior to stress exposure (Cohen et al., 2006). Whether the observed HPA axis hypo-functionin a proportion of FEP patients is a vulnerability factor, endophenotype or sequelae of acute illness or traumatic experiences, remains to be determined. Factors such as the timing, type and severity of the trauma as well as genetic and other environmental factors are likely to be important in determining the impact on HPA axis development (Ellenbroek and Cools, 2000; van Oers et al., 1997). Further research investigating the link between early childhood adversity, recent life trauma (including trauma associated with the onset of psychosis), HPA axis and GR function in psychotic disorders is warranted.
Strengths of this study include the fact that the FEP patients were neuroleptic-naïve or minimally treated, thereby minimising potential confounding effects of psychotropic medication on HPA axis function. The relatively small and heterogeneous sample size places significant limitations to these findings and restricts individual analysis of sex and diagnostic subgroups.
In conclusion, the results from this study reflect a greater prevalence of childhood trauma in FEP patients and a hypersensitive negative feedback response of the HPA axis in a subset of FEP patients. It will be important to further clarify directionality of HPA axis abnormalities in psychosis and their relation to clinical subtypes, as this could have important implications for treatment. Specifically, novel therapeutic strategies that target the HPA axis, such as the GR antagonist mifepristone, are increasingly being investigated. Enhanced cortisol suppression in response to dexamethasone may be associated with enhanced GR function and/or an increased number of GRs. Future work should extend the observations reported in this study and investigate potential GR-dependent mechanism(s) underlying HPA axis dysfunction at the onset of psychosis.
Footnotes
Acknowledgements
The authors would like to thank the participants in this study for their time and patience.
Funding
This work was supported by a NARSAD Young Investigator Award to Dr Garner and NHMRC program grant #350241. Dr Garner is supported by a NHMRC Postdoctoral Training Fellowship. Dr Phassouliotis was supported by May Stewart and Ronald John Gleghorn Bursaries (Faculty of Medicine, Dentistry and Health Sciences, University of Melbourne) and an Orygen Youth Health Studentship.
Declaration of interest
The authors report no conflicts of interest. The authors alone are responsible for the content and writing of the paper.
