Abstract
Introduction
The climate crisis presents a complex and growing challenge for healthcare systems around the world. Healthcare systems can contribute to substantial global emissions, with the UK’s National Health Service (NHS) alone responsible for 4%–5% of the country’s total carbon footprint. A wide range of clinical disciplines have already begun to assess and design interventions to tackle this issue. However, clinical and diagnostic laboratories remain underexplored.
Aims
What studies have been undertaken to assess and improve the environmental impact of clinical laboratories?
Methods
This scoping review undertook a multi-database search from date of inception to 5th February 2024. All primary studies that assessed the environmental outcomes of clinical laboratories were included. Studies were screened and data extracted by one reviewer with a 10% verification process at each stage. Studies were assessed based upon year of publication, geographical region, interconnectivity and area and type of clinical laboratory or test.
Findings
There has been some longstanding interest in understanding the environmental impact of clinical laboratories, and this field of investigation has gained popularity within the scholarly community in the last decade. Despite this recent increase in popularity there is a relatively limited number of intervention studies aimed at improving sustainability within clinical laboratories. Most research in this area originates from the United States, United Kingdom, and Australia, although the topic appears to be of global scholarly interest. There is limited interconnectivity of studies included in this review. Studies in this field have primarily been conducted at the clinical laboratory level, with a focus on quantifying waste in kilograms, measuring carbon dioxide equivalent (CO2e) emissions, and categorizing laboratory waste by type. To a lesser degree these outcomes have been assessed for specific clinical tests. Across both clinical laboratory and specific test assessments there is notable heterogeneity in both methods used, and areas explored.
Discussion
While this scoping review highlights a growing interest and awareness in this important field, the diversity of reported outcomes and the limited interconnectivity of studies indicate that it remains a developing area. The lack of consensus in methodologies and outcome measures suggests that establishing a baseline analysis remains a distant goal. Ideally, future efforts should prioritize improving the assessment of individual laboratory tests, fostering greater standardization, and enhancing repeatability to strengthen the reliability of environmental impact evaluations.
Introduction
The ongoing climate crisis presents escalating challenges, 1 influencing various sectors, including medical practice2,3 and scientific research. 4 The crisis will mandate adaptation both at the clinical level as environmental changes affect human health, 5 as well as the operational level as climate variation impacts the infrastructure that supports health services. 6 In the UK, the National Health Service (NHS) was the world’s first national health provider to declare a net-zero policy, with a variety of pathways mapped depending on level of action taken. 7 This is crucial to the UK’s net-zero ambitions, as the NHS is responsible for an estimated 4%–5% of the UK’s carbon emissions. 8
Work has begun to look at health systems infrastructure, 9 as well as many of the clinical specialties that make-up a complete health system.10–12 While challenging, notable academic and policy progress has been made in a variety of such specialties, such as surgery, 13 general practice, 14 emergency medicine, 15 and research laboratories. 16 Such research has led not only to estimated impacts of clinical specialties, but burgeoning programs aimed at mitigating the emissions associated, such as the Royal College of Emergency Medicine’s GreenED programme. 17 Clinical and diagnostic laboratories have yet to undergo such a revolution.
The requirement of in vitro diagnostics (IVDs) has increased significantly globally, 18 in part due to the COVID-19 pandemic. 19 Each year, approximately 14 billion tests are conducted in the US, 20 1.2 billion in the UK, 21 and 500 million in Australia. 22 Market research estimates that the global diagnostic laboratory market will increase from $297 billion in 2021 to $514 billion by 2028, representing an immense rate of growth. 23 IVDs take place within laboratory facilities, which in themselves have the potential to be energy intensive due to their significant heating, cooling, and particularly ventilation requirements.24,25 Beyond energy, IVDs require analytical equipment which comes with the potential of ecological impact, 26 as do the immense volumes of single-use plastics. 27 IVDs also can require transport or delivery, which can increase their ecological impact. 28
The predicted growth of IVDs has the potential to significantly increase the environmental impact of clinical laboratories. This is driven by the resource-intensive nature of laboratory operations, including environmental requirements, specialist equipment, waste products, transport, and energy costs. As a result, it is imperative to better understand what research has been conducted to date.
Recent systematic reviews in this field have examined the environmental impact of broader healthcare systems 29 and hospitals 30 or assessed interventions aimed at reducing their environmental footprint.31,32 Previous review papers have highlighted the need for environmental assessment and interventions at a specific clinical laboratory level.33–35 Despite this recognition, no comprehensive synthesis currently exists on the environmental impact of clinical laboratories, or the interventions implemented to mitigate their effects.
Aims/questions
This scoping review aims to answer two questions. These are. (1) Q1: What studies have been undertaken assessing the environmental impact of clinical laboratories? (2) Q2: What interventions have been studied to mitigate the environmental impact of clinical laboratories?
Methods
This scoping review was conducted following methodological guidance provided by Peters et al 36 and Levac et al. 37 This scoping review is reported in accordance with the PRISMA Extension for Scoping Reviews (PRISMA-ScR) guidance. 38
Search
The following databases were searched on 5th February 2024: MEDLINE (Ovid), Embase (Ovid), Environment Complete (EBSCOhost), and Web of Science (indexes: SCI-EXPANDED; SSCI; AHCI; CPCI-S; CPCI-SSH; ESCI). Search terms were identified by the review team and included a combination of relevant subject headings and keywords. The search strategy was developed by an information specialist (CH) and was adapted for each database. The search strategies used for each database can be found in Appendix 1. No date limits were applied, but searches were limited to studies in English. References were downloaded into EndNote and duplicates were removed before being uploaded into Rayyan for screening. 39 Backwards citation searching using Web of Science was conducted for included papers to identify additional studies.
Study selection
Any type of primary study which assessed any kind of environmental outcomes for clinical laboratories was included. For the purposes of this review clinical laboratories were defined as healthcare facilities providing a wide range of laboratory procedures which aid the physicians in carrying out the diagnosis, treatment, and management of patients. 40 This definition was extended to include the screening for disease, as well as the use of Point of Care testing (POCT), which use similar technologies and often come under the operational management of clinical laboratories.
Title/abstract screening was conducted by a single reviewer using Rayyan. 39 Study selection was piloted with 10% of the titles and abstracts being screened by five reviewers. A Kappa Score was calculated to assess reviewer agreement during this pilot process, and substantial agreement (0.61–0.80 agreement) between all reviewers was needed before single screening of the remaining references continued. 41 Full paper screening followed the same 10% verification process and reasons for exclusion at the full paper screening stage were documented and recorded.
Data extraction (selection and coding)
After the 10% verification process data extraction was performed by one reviewer using a pre-piloted form. Reviewers used Microsoft Copilot AI (generative artificial intelligence software) (Azure OpenAI-powered AI: 01/04/24 - 01/09/2024) to verify the data items extracted. 42 The data items extracted included: year of publication, country of study, city/town, study type, study aims, clinical setting, single or multi-centre, population (description of laboratory diagnostics or clinical laboratory activities under investigation), sample size, diagnostic focus, specimen and test types, outcomes, economic evaluation, and description of interventions implemented to reduce environmental impact, type of intervention, intervention description, and control group where applicable.
Strategy for data synthesis
Given the wide variation in study design, interventions, and outcomes, a narrative synthesis approach was used to organize the review findings. Bar charts were employed to analyse the growth in the number of studies per year over time. A geographical map was utilized to pinpoint the regions of the world that have explored this area and investigated interventions to reduce the environmental impacts of clinical laboratories. 43 Additionally, a citation map was employed to examine the interrelationship of citations within this field using Litmaps’ citation mapping software. 44 Individual study characteristics was described based upon two global categories of clinical laboratory assessment/interventions and specific tests.
Results
Database searches yielded 2417 unique citations after duplicate removal, with 322 full-text papers retrieved following title and abstract screening. After full paper screening 40 studies (40 primary papers and one link to paper) met the inclusion criteria for the scoping review. Screening the references of the included studies led to the review of an additional 442 titles and abstracts, with one paper identified through hand searching. This process resulted in the screening of 13 full-text papers, from which three additional studies were included. This resulted in 43 studies (43 primary papers and one link to paper) being included in this review (Figure 1). Screening process for paper inclusion.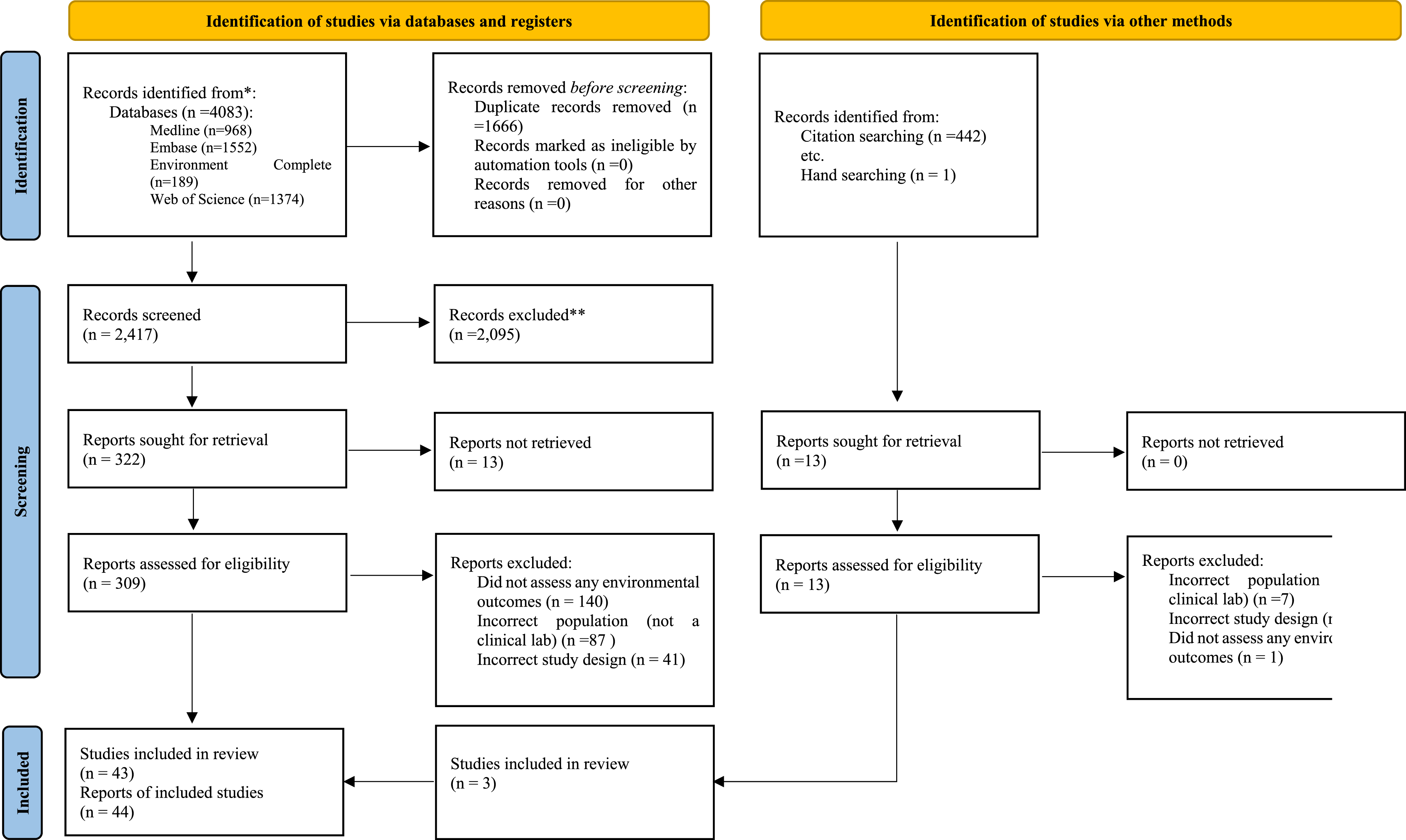
During abstract and title screening the review team were able to achieve a substantial agreement within one round of consensus development (% of agreement: 98.8–99.6, Cohen’s k: 0.65–0.90). Two rounds of consensus building were required during full paper screening to reach substantial agreement (% of agreement: 90.3–96.8, Cohen’s k: 0.61–0.89). The primary reason for exclusion during full-text screening was the lack of reporting on environmental outcomes (n = 141). This was followed by studies that did not assess a clinical lab. (n = 94) and those with inappropriate study designs (n = 43).
Temporal analysis of studies on clinical laboratories’ environmental impact
The number of studies per year evaluating the environmental impact of clinical laboratories is presented in Figure 2. The topic area of environmental impact of clinical laboratories was first assessed in 1975 (Pragay, 1975). For the following three decades (1975–2004) a relatively low number of studies were published (11.6%, n = 5). Subsequently, there has been a notable growth in the number of publications during the last 20 years, with 10 studies (23.2% of studies identified) published between 2005 and 2014 and 27 studies (65.1% of studies identified) between 2015 and 2024. The first intervention study was published in 2012, and the remaining five studies were published in 2022 and 2023. Number of studies per year evaluating the clinical impact of clinical laboratories. *inclusion dates for the literature search ran until 5th February 2024, Geographical Distribution of Research on Clinical Laboratory Environmental Impact.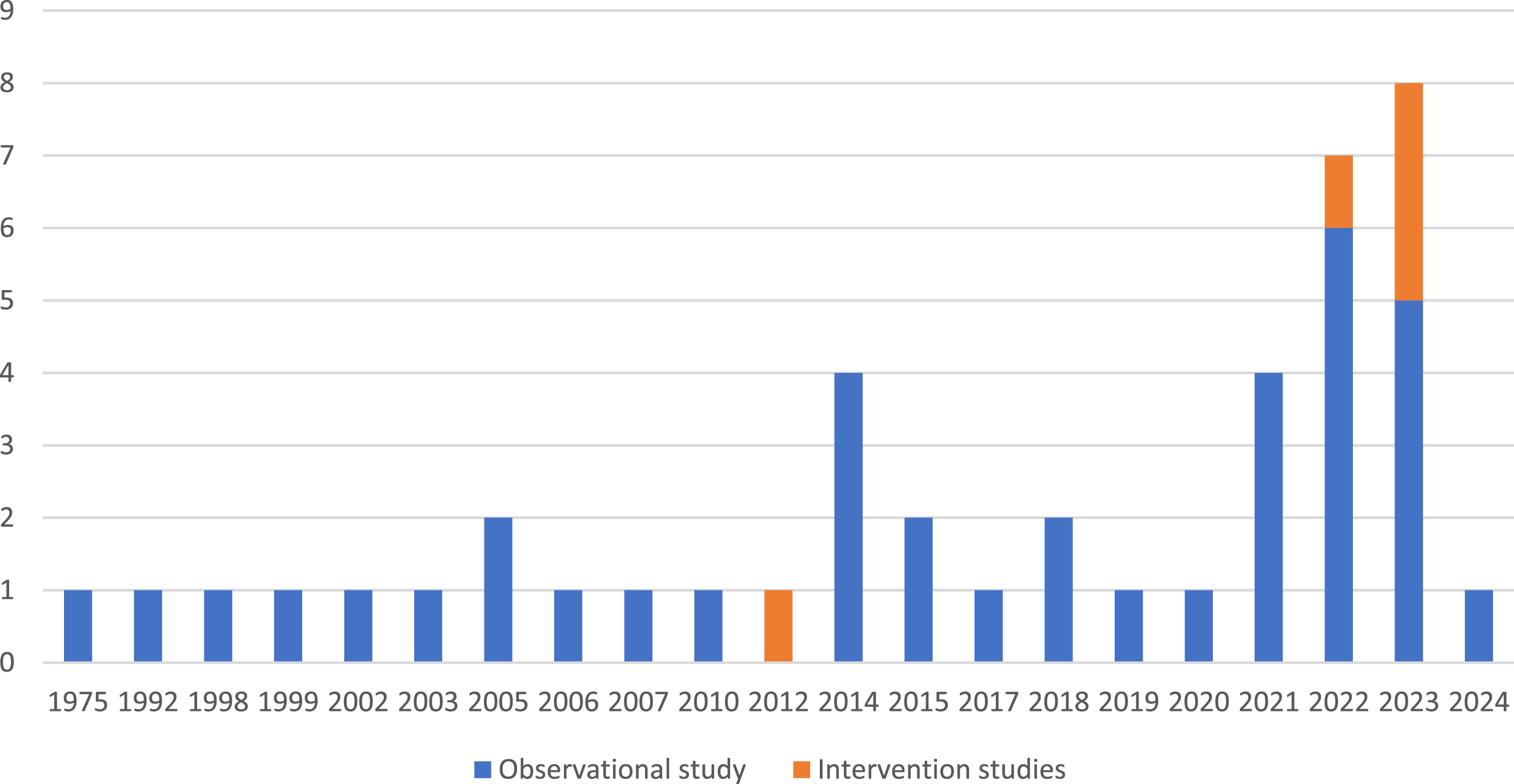
Exploring the geographical locations of the studies, nearly half were completed in three countries the United States (n = 9), United Kingdom (n = 6), and Australia (n = 6) (Figure 3; Tables 1 and 5). It is important to note that of these 21 studies, 15 were published in the last 10 years. Geographical location of included studies. Characteristics of Clinical Laboratory Level – Observational Studies.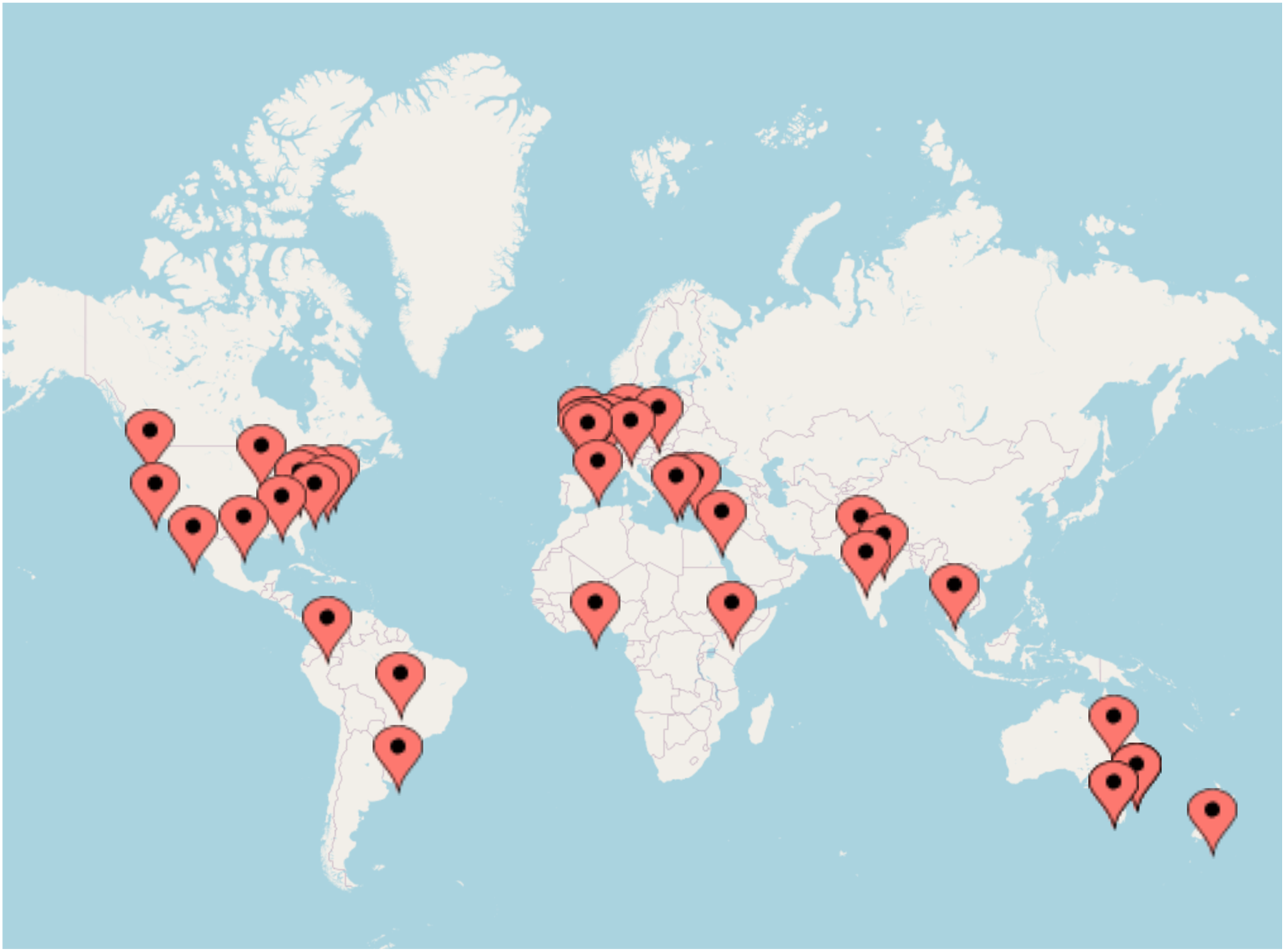
The second most common regional areas were Greece (n = 2), Germany (n = 2), India (n = 2), and Brazil (n = 2). Despite the relatively geographically clustered nature of the studies, there has been a wide range of single studies carried out in multiple countries, including Mexico, Jordan, Ethiopia, Poland, France, Iran, Pakistan, Sao Tome and Principe, Colombia, Canada, Thailand, Togo, Turkey, and the Netherlands.
Citation patterns in research on the environmental impact of clinical laboratories
The Litmaps citation map illustrates the current body of evidence evaluating the environmental impact of clinical laboratories and their citation relationships (see Figure 4 for Litmaps citation map of all included studies). Out of the 43 included studies only two studies were unable to be identified using the Litmap software. Of these 41 studies, only 14 studies (34.1%) cited previous research within this domain. Litmaps citation map of all included studies. This citation map visually represents each study as a circle (node), with lines (edges) indicating citation relationships between them. The horizontal position reflects the year of publication, while the vertical position corresponds to the total number of citations each paper has received.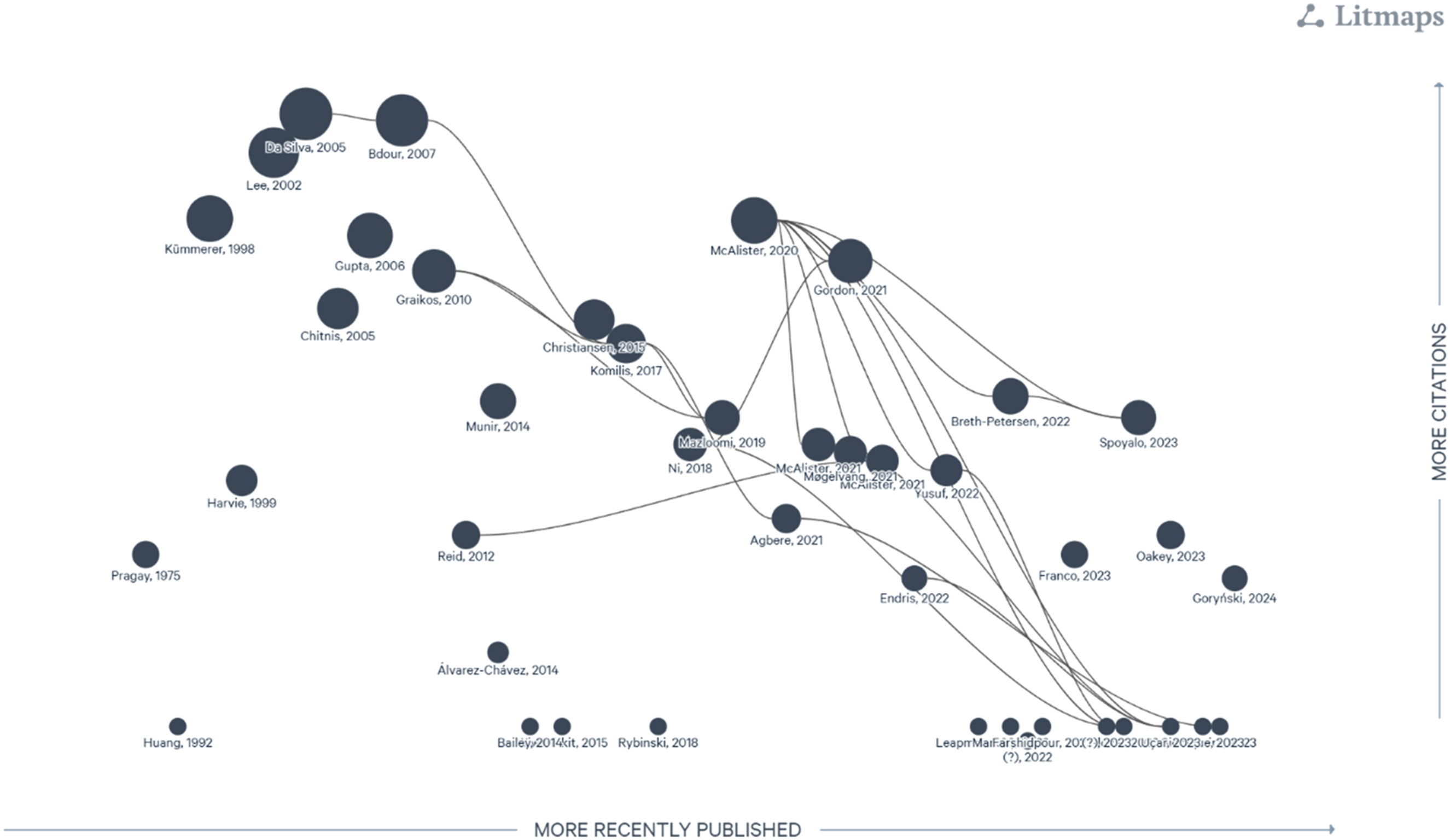
Externally (number of all citations of included studies), the included studies vary in the number of citations, with an interquartile range of 14 citations. The lower quartile (Q1) shows that 25% of the articles have 3 or fewer citations, while the upper quartile (Q3) indicates that 75% have 17 or fewer citations (Range = 0 to 308 citations). Out of the 41 included studies, the median number of citations was 10.
Stuthe vertical position corresponds to the total number of citations each paper has received dy characteristics
Out of the 43 included studies, the assessment of environmental impact of laboratory testing was undertaken either at a clinical laboratory level (Clinical Laboratory-Wide Environmental Assessment) (n = 28) or for a specific clinical test (n = 15).
Clinical laboratory level – observational studies
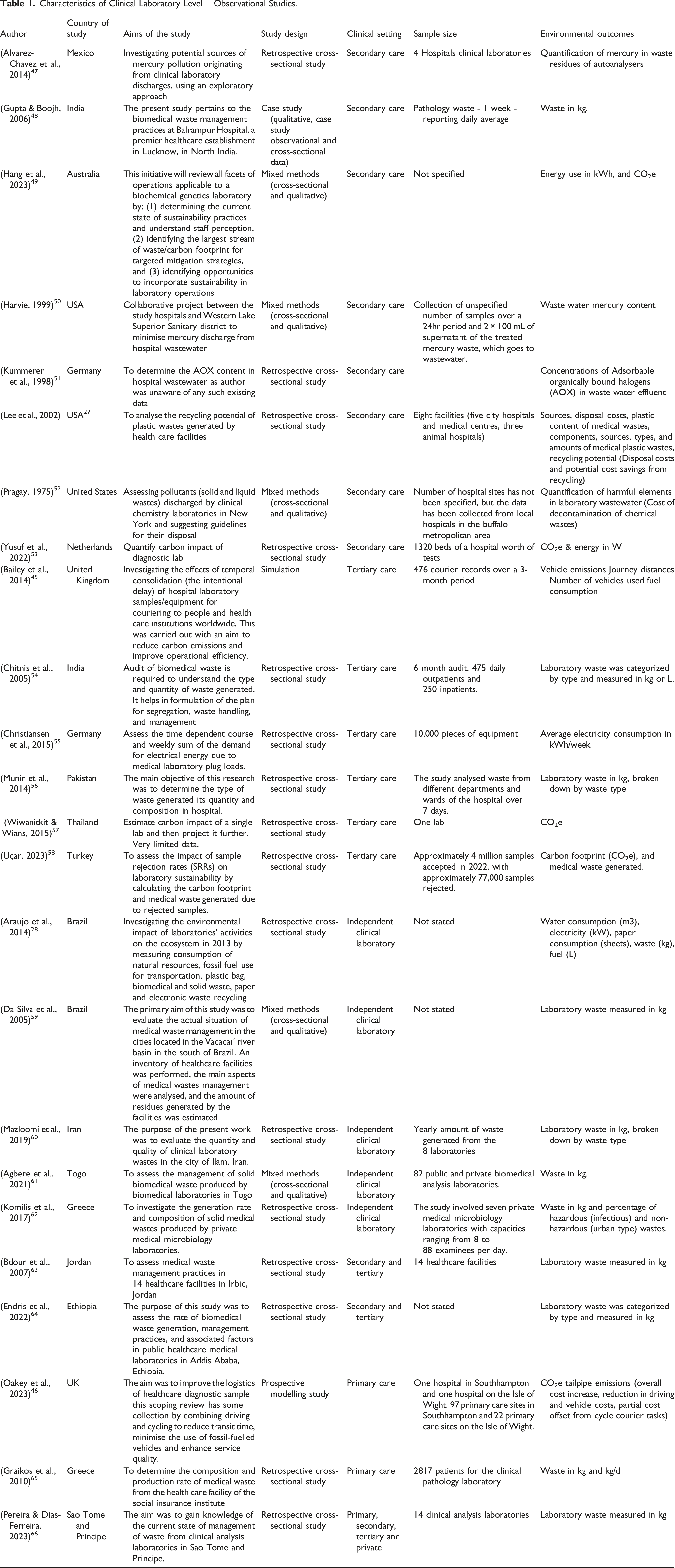
Of the 28 studies at the clinical laboratory level, 24 were observational studies, and four were intervention studies (Table 1). Among the 24 observational studies assessing the environmental impact of clinical laboratories, the majority evaluated the environmental impact of laboratory testing at a specific point in time, utilizing either a retrospective cross-sectional design (n = 16) or a mixed-methods approach combining qualitative and cross-sectional data (n = 6). The remaining two studies that assessed the environmental impact of clinical laboratories at a clinical laboratory level presented environmental impact data collected using a cross-sectional design, which was then utilized as part of a simulation scenario aimed at reducing environmental impact.44,46
The assessments of environmental impact at the clinical laboratory level were conducted in various care settings: secondary care (n = 8), tertiary care (n = 6), private independent laboratory (n = 5), secondary and tertiary care (n = 2), primary care (n = 2), and across primary, secondary, tertiary, and private independent laboratory (n = 1).
At a specific clinical laboratory level setting (primary, secondary, tertiary, private independent laboratory) there was limited commonality of reporting of environmental outcomes. Among the 24 studies that assessed the environmental impact at the clinical laboratory level, there was limited consistency in the metrics used. The most common measurement was laboratory waste quantified in kilograms (n = 12), followed by carbon dioxide equivalent (CO2e) emissions (n = 5), laboratory waste categorized by type and measured in kilograms or litres (n = 4), and energy usage reported in kilowatt-hours (kWh) (n = 3). In addition to these outcomes, there were 14 additional varying outcomes reported in two or fewer studies. Additionally, three studies evaluated the financial costs at the clinical laboratory level. These costs included disposal expenses, potential cost savings from recycling, overall cost increases, driving and vehicle costs, partial cost offsets from cycle courier tasks, and the cost of decontaminating chemical waste.
Clinical laboratory level – experimental studies
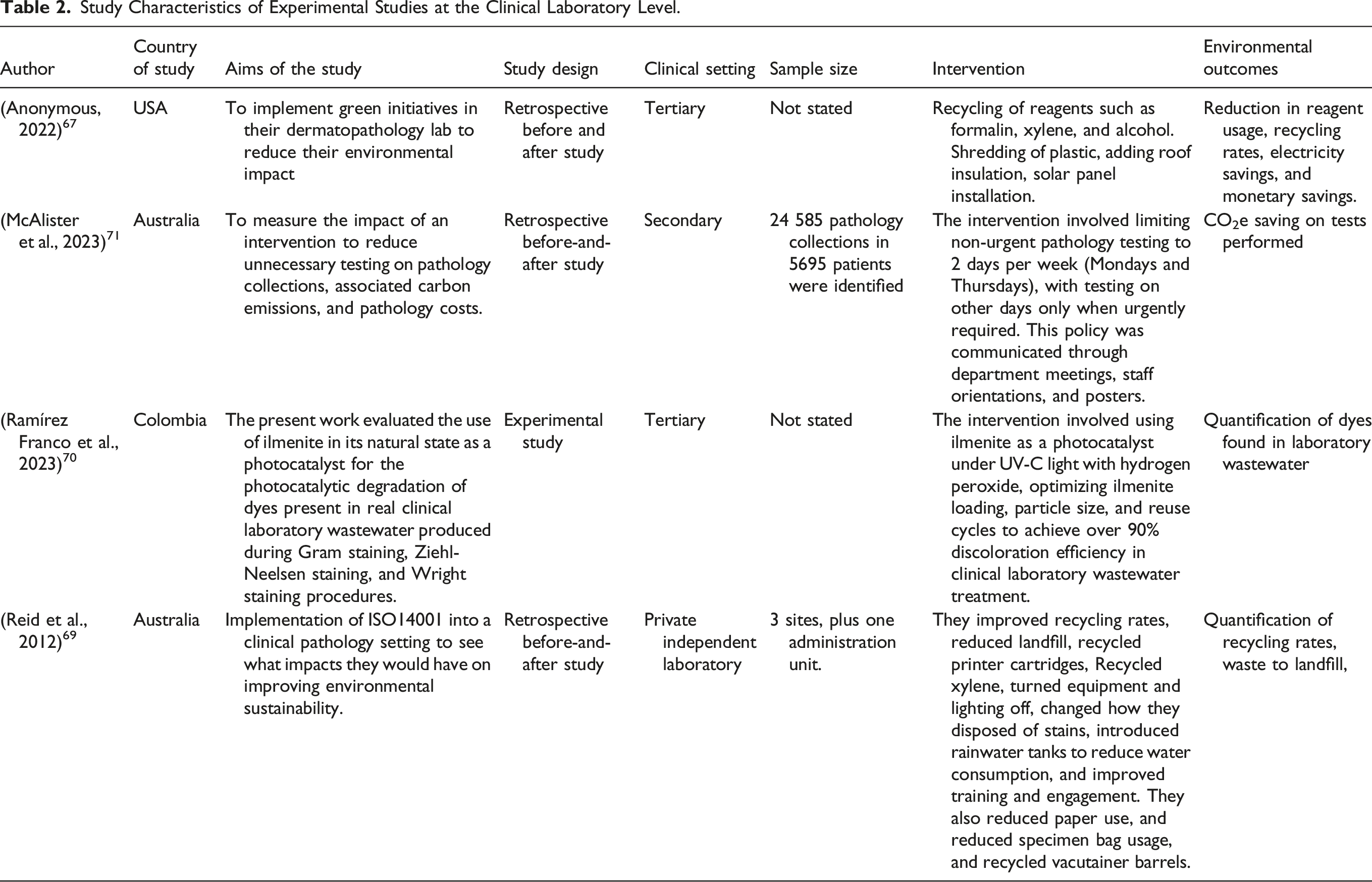
Four of the studies were intervention studies at a clinical laboratory level, based in tertiary care (n = 2), secondary care (n = 1), and in a private independent laboratory (n = 1) setting. These employed either a retrospective before-and-after study design67–69 or experimental design. 70 Across the four studies, the interventions focused on four main approaches: resource recycling and waste reduction (e.g. reagents, plastic, and water),67,69,70 energy efficiency and renewable energy integration (solar panels and insulation 67 ), and operational changes (limiting non-urgent testing and enhancing staff engagement68,69). These efforts collectively aimed to promote environmental sustainability and operational efficiency.
Study Characteristics of Experimental Studies at the Clinical Laboratory Level.
Specific clinical test
Of the 15 studies which assessed the environmental impact of a specific clinical test, 14 were observational studies, and 1 was an intervention study. Of the 14 observational studies, seven studies established environmental impact in a single time period using a cross-sectional (n = 9), life-cycle assessment (n = 2), cohort study (n = 1), observational study (n = 1), and case series design (n = 1).
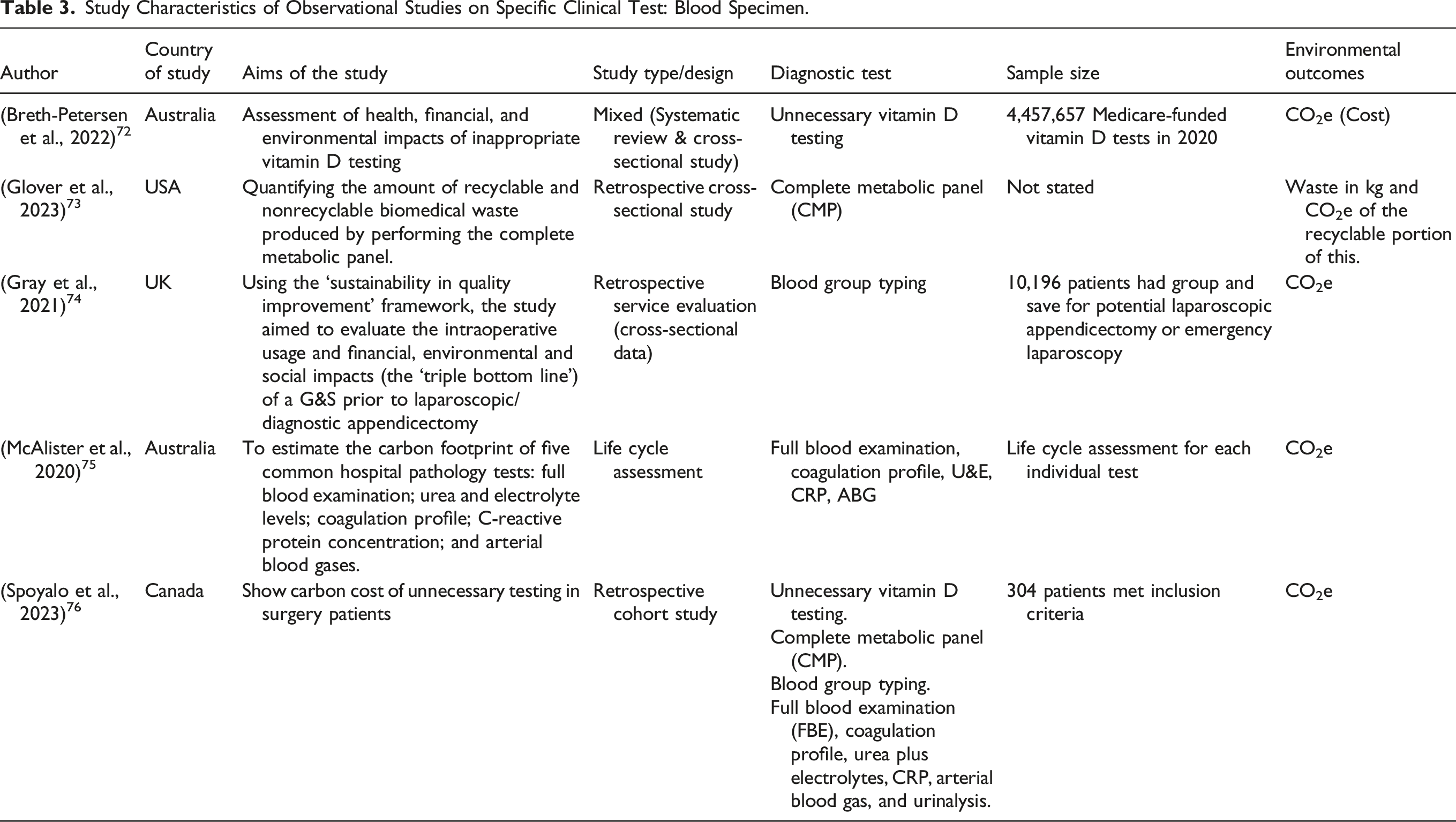
Specific clinical test – blood specimen tests
Study Characteristics of Observational Studies on Specific Clinical Test: Blood Specimen.
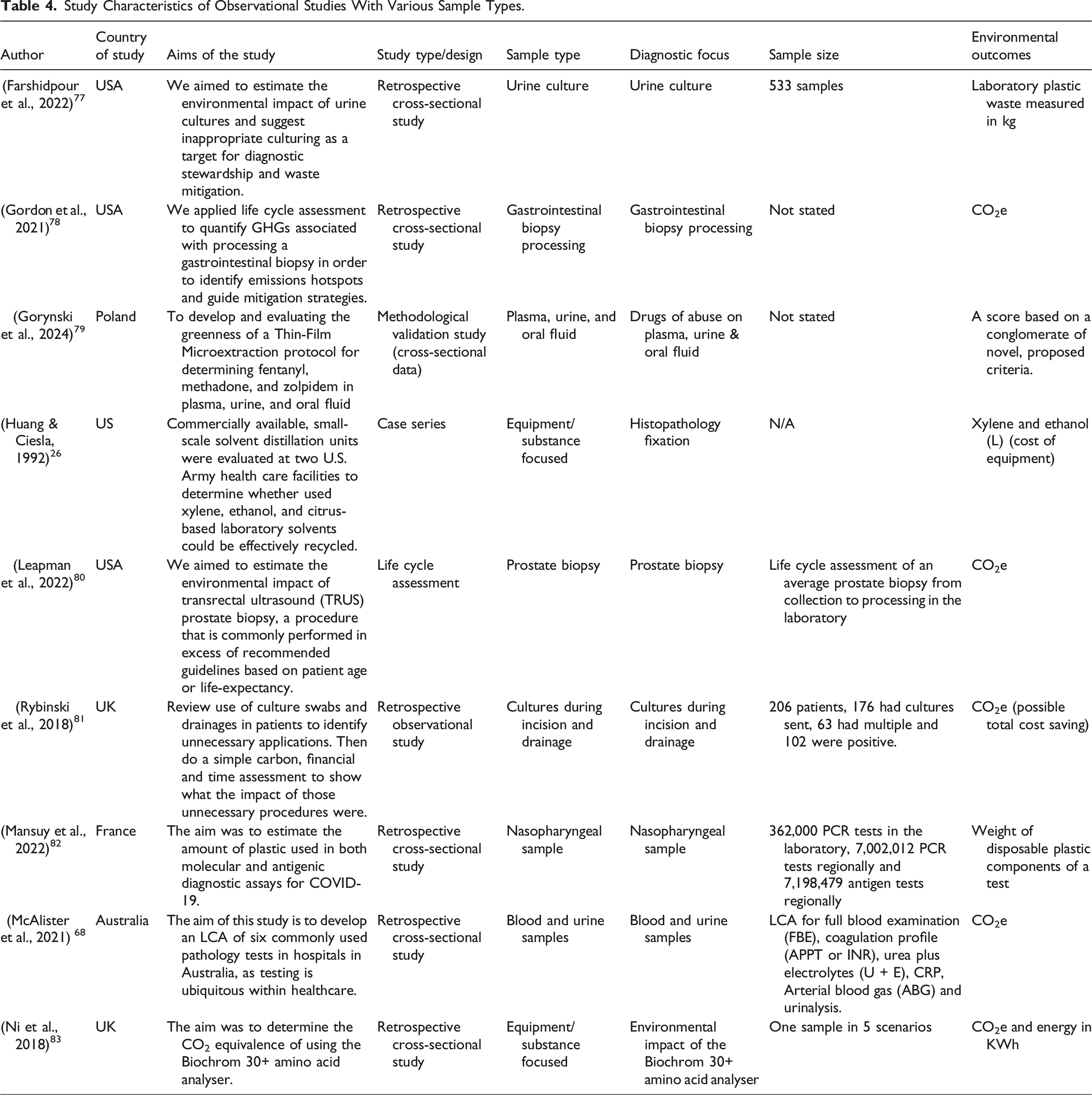
Specific clinical test – Various Sample Types
Study Characteristics of Observational Studies With Various Sample Types.
Specific clinical test – experimental studies
Study Characteristics of Intervention Study for Pre-Surgery Blood Typing.

Discussion
It is evident that there has been some historical scholarly interest in the environmental impact of laboratory testing with the first paper being published 50 years ago. However, more recently this interest has increased with the majority of papers being published within the last 10 years. Despite this growing interest there have been relatively few studies assessing interventions within clinical laboratories through the lens of the environmental impact. All but one of the intervention studies were conducted after 2021. The search dates for this scoping review concluded in early February 2024; hence, the small numbers of studies included from that year. It is reasonable to predict that at least a similar number of studies were published in 2024 as in the preceding few years, if not more considering the recent trend is showing increasing publications.
Analysis of geographical location of the studies suggests a preponderance of high-income settings. This highlights the need for further research within low to middle income countries, as there is substantial potential to reduce the environmental impact of clinical laboratories within these regions. 85 However, with individual studies being performed in geographically diverse settings, the environmental impact of clinical laboratories appears to be of scholarly interest in multiple regions around the world. This growing global interest in the environmental impact of laboratory testing may be influenced by international groups and organizations advocating for and promoting sustainable practices.86–88
There was limited connectivity of papers and internal citations which may suggest a lack of coordination within this field. However, this lack of limited connectivity may relate to the heterogeneity within the areas under investigation but could also be suggestive of a paucity of globally diverse papers. Despite this possible moderating factor, it does highlight the need for greater coordination, as coordinated interventions, both individually and at an organizational level, can be more effective in reducing environmental impacts. 89
Studies were grouped into two levels: laboratory and clinical test levels, encompassing both observational and interventional studies. At both levels there was notable heterogeneity regarding clinical environment and test type factors for both types of studies. There was some consistency in the observational studies regarding the outcomes assessed, with waste quantified in kilograms, and CO2e calculated at both the laboratory and clinical test levels.
At the laboratory level, it is vital that future observational studies establish a common outcome set and adopt a systematic categorization by test type or volume to enable cross-comparison of laboratories of different sizes and types. This research direction would enable a method of establishing the degree of contribution that clinical laboratories are having on the environment by presenting a range of effect. This will also facilitate in establishing benchmarks and goals for future development at this level. A similar lack of consistency in effect may also be observed within the intervention studies at this level due to the wide variation in approaches and interventions implemented. Current work on greater standardization in assessing the environmental impact of clinical laboratories is being undertaken in England through the development of laboratory networks. 90 These pathology networks are being assessed for their maturity against a nationally set list of key performance indicators. Environmental sustainability has now been added to this process, and this may eventually mandate laboratories to consider their impact.
To improve the reliability of the outcomes of such studies, reducing the granularity of assessments may be beneficial. Focusing on single laboratory tests with standardised, repeatable procedures could enhance comparability between laboratories and allow for more reliable estimates of environmental impact. From an interventional perspective, this principle of repeatability would also be evident. The focus of the studies is crucial if the aim is to establish a consistent and repeatable estimate of an effect. The substantial variation within laboratories makes achieving a standardised estimate of effect challenging, particularly from a repeatability perspective. Attempts have been made to estimate the environmental impact of various healthcare systems and processes, including renal care, 91 cataract surgery, 92 surgery overall, 93 and healthcare in its entirety. These analyses detail the scale of the environmental impact of these processes in CO2e via detailed life cycle assessment. This level of detail is required to identify resource-intensive hotspots that could benefit from targeted interventions, as well as establishing a baseline from which improvements can be made. 94
Whilst this review demonstrates an increasing academic interest in this topic, it is clear that significant further work is required. We propose that the life cycle assessment of commonly performed pathology tests be undertaken with robust, reproducible methodology. This would allow pragmatic comparison of the contribution of the different elements (e.g. patient travel, phlebotomy, specimen transport, laboratory analysis, and waste) of the testing pathway to the overall carbon footprint. Research focussed on optimising these stages, with an emphasis on reducing unnecessary work could lead to significant gains.
Limitations of the scoping review
This scoping review has some methodological limitations. Screening and data extraction were conducted by a single reviewer; however, a 10% verification process was implemented for both. Additionally, Copilot was used to double-check all extracted data items. While we consider our search strategy to be robust, we identified three additional studies through hand searching and citation screening, which may indicate potential recall issues. Nonetheless, we do not believe this would substantially alter the overall findings of this scoping review. Furthermore, we included only English-language papers in both the search strategy and screening, which may have introduced geographical bias in the included studies.
Conclusion
Whilst this scoping review demonstrates an increasing interest and awareness in this important field, the diversity of reported outcomes and limited interconnectivity of the studies suggest that this is a still a developing area. The first stage of improving a process is to establish a baseline or starting point from where advancements may be measured. With a lack of consensus in methodologies and outcomes, this baseline analysis of the environmental impact of clinical laboratories seems distant. Future efforts should focus on enhancing the assessment of individual laboratory tests, promoting greater standardization of methodologies and outcomes, and repeatability to improve the reliability of environmental impact evaluations. This challenge will also be evident in intervention studies, as the underlying inconsistency would lead to a similar variability in intervention effects. This lack of consistency would make repeatability and standardization difficult, ultimately preventing long-term improvements and refinements of the intervention based on key moderating factors.
Supplemental Material
Supplemental Material - Environmental sustainability of clinical laboratories: A scoping review
Supplemental Material for Environmental sustainability of clinical laboratories: A scoping review by RJ Shorten, A Sanders, M Farley, S Josse, S Shafiq, C Harris, A Clegg, and J Hill in Annals of Clinical Biochemistry.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: JH, CH, and AC are part-funded by the National Institute for Health and Care Research Applied Research Collaboration North-West Coast (NIHR ARC NWC). The views expressed are those of the authors and not necessarily those of the NHS, the NIHR, or the Department of Health and Social Care.
Ethical approval
Not applicable.
Guarantor
RJ Shorten.
Contributorship
Data Curation: JH; Formal Analysis: JH, CH; Investigation: RS AS MF SJ SS CH AC JH; Methodology: RS AS MF SJ SS CH AC JH; Project Administration: RS JH; Software: CH JH; Supervision: RS JH AC; Validation JH CH; Visualization JH; Writing – Original Draft Preparation: RS AS MF SJ SS CH JH; Writing – Review & Editing: RS AS MF SJ SS CH AC JH.
Supplemental material
Supplemental Material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
