Abstract
Background
Reference intervals are a vital part of reporting numerical pathology results. It is known, however, that variation in reference intervals between laboratories is common, even when analytical methods support common reference intervals. In response to this, in Australia, the Australasian Association of Clinical Biochemists together with the Royal College of Pathologists of Australasia published in 2014 a set of recommended common reference intervals for 11 common serum analytes (sodium, potassium, chloride, bicarbonate, creatinine male, creatinine female, calcium, calcium adjusted for albumin, phosphate, magnesium, lactate dehydrogenase, alkaline phosphatase and total protein).
Methods
Uptake of recommended common reference intervals in Australian laboratories was assessed using data from four annual cycles of the RCPAQAP reference intervals external quality assurance programme.
Results
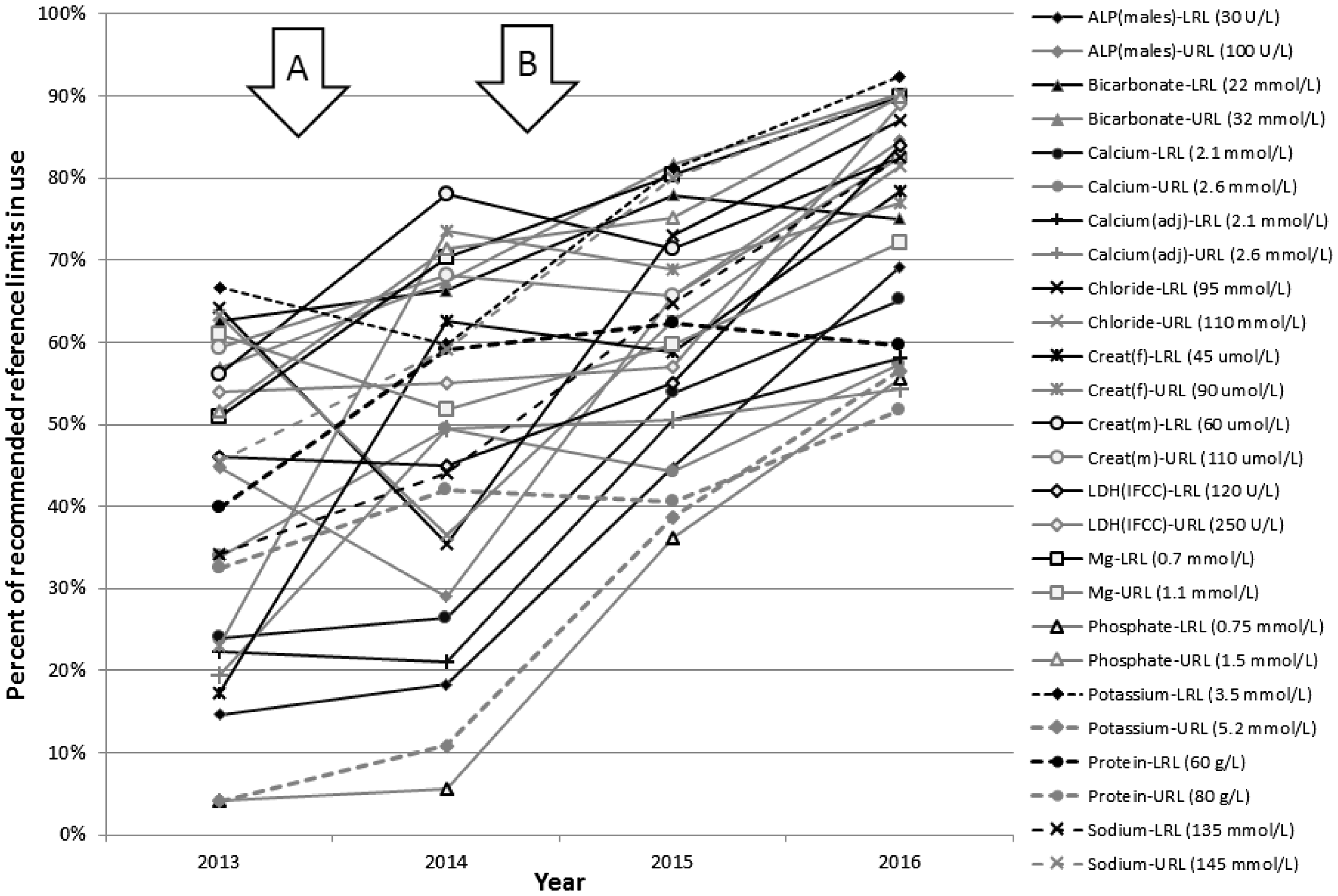
Over three years, from 2013 to 2016, the use of the recommended upper and lower reference limits has increased from 40% to 83%. Nearly half of the intervals in use by enrolled laboratories in 2016 have been changed in this time period, indicating an active response to the guidelines.
Conclusions
These data support the activities of the Australasian Association of Clinical Biochemists and Royal College of Pathologists of Australasia in demonstrating a change in laboratory behaviour to reduce unnecessary variation in reference intervals and thus provide a consistent message to doctor and patients irrespective of the laboratory used.
Introduction
Reference intervals have been described as the most widely used decision support tool for numerical pathology results. 1 Variation in reference intervals without method or population differences leads to different information being provided by different laboratories. 2 Previous work has demonstrated this to be a significant problem in the reporting of chemical pathology results in Australia. 2 Being aware of this issue, in 2013, the Australasian Association of Clinical Biochemists (AACB) commenced a project to harmonize reference intervals for common chemical pathology tests. The process included a series of annual workshops involving interested laboratory professionals. The meetings considered all relevant issues and available data to develop recommended common reference intervals (CRI). This lead to a formal proposal for CRI for 11 frequently requested routine serum chemical pathology tests which was supported by the Royal College of Pathologists of Australasia (RCPA) and published in 2014. 3
In 2013, the RCPAQAP introduced an EQA programme for reference intervals for common serum analytes. Participants submit the lower and upper reference limits (LRL, URL) for analytes in the programme which are measured in their laboratory. The requested reference intervals are those for a 40-year-old male, except for ALP, AST, ALT and creatinine, where sex-specific intervals are requested. This paper shows the uptake of the AACB recommended CRI using data from the RCPAQAP reference intervals programme from 2013 to 2016.
Methods
Data were extracted from participant returns from the reference intervals programme from 2013 to 2016. Data included the number of enrolled laboratories, the grouping of laboratories in networks, the participation of laboratories over the four years of the programme to date as well as the LRL and URL for all of the analytes for which CRI were recommended.
3
The included analytes and the recommended reference limits are shown in Figure 1. All methods were included for each analyte except for LDH, where only laboratories using results traceable to the IFCC reference method were included.
4
Use of AACB recommended common reference intervals. Data from the RCPAQAP reference intervals programme from 2013 to 2016 have been used to show the frequency of use of the AACB recommended CRI. Letter A shows the time of the intervals being initially proposed and circulated for likely response and letter B shows the time of formal publication.
3
Results
The numbers of enrolled laboratories in the RI programme in each of the years were as follows: 2013: 123, 2014: 93; 2015: 105; 2016: 92. This compares with 163 enrolments in the liquid serum chemistry programme and approximately 400 laboratories in the General Serum Chemistry programme in Australia. Of the 92 labs enrolled in the RI programme 2016, 72 (78%) were also enrolled in 2013, 69 (75%) also in 2014 and 82 (89%) in 2015 with 56 (61%) enrolled in all four years. Not all laboratories submitted reference intervals for all analytes. The enrolled laboratories in 2016 represented 29 pathology organizations, with between one and 26 laboratories enrolled from each organization.
The percentage of laboratories reporting with the recommended URL and LRL is expressed as a percentage of the number of laboratories reporting their reference limits for that test. The median uptake for the different intervals in 2016 was 82% compared with 40% in 2013. The results for the individual reference limits are shown in Figure 1. For all analytes, the recommended CRI are the most common ones in use for the 2016 results. This is in contrast with 2013, where eight of the most common reference limits were different to those subsequently recommended. The current level of use of the CRI has been driven by changes made by laboratories rather than inclusion of different laboratories in the programme with a 46% of intervals reported in 2016 having changed since previous years.
Discussion
The AACB and RCPA are addressing the need to reduce unnecessary variation in reference intervals in Australia, in line with similar activities in the UK. 5 Data from the RCPAQAP Liquid Serum Chemistry programme, which is based on fresh unprocessed frozen serum, have been used to determine whether common reference limits are appropriate and the RCPQAQAP reference interval programme has now provided important information about uptake of the recommended CRI.
The data presented show a high level of current usage of the recommended CRI. The median uptake for the intervals is 82%. With the lowest uptake for the total protein URL (56%) and the highest for the sodium URL (93%). The data show that laboratories have acted in response to the guidelines or to the discussions which took place prior to the formal adoption of the guidelines (Figure 1). Further evidence to show that laboratories have acted in response to the guidelines is that many intervals for currently enrolled laboratories have changed since 2013. It is clear that the current agreement between laboratories is the product of the collaborative process in the AACB meetings rather than formalizing a pre-existing status.
The ability to establish common reference intervals for these analytes has been dependent on there being minimal between-laboratory or between-method differences in results. All the measurands included, with the exception of serum bicarbonate, have listings on the JCTLM database providing higher order reference materials and methods for these tests. 6
There are a number of limitations to the current data-set. In 2016, only 92 laboratories were enrolled in the RI programme. However, the coverage represented by the enrolled laboratories is significantly higher as many individual laboratories are part of networks which use single computer systems and apply the same reference intervals throughout the network. Thus, an enrolment of one laboratory can be representative of many individual laboratories. While the exact effect of this is unknown, enrolments include laboratories from most large public and private pathology networks. It is also possible that the enrolled laboratories may reflect a selection bias, with laboratories interested in CRI more likely to participate in the RI programme.
In conclusion, there is good evidence that there has been a marked reduction in unnecessary variation of reference intervals for a number of common biochemistry tests in Australia. This reflects the activities of the professional organizations such as the AACB and RCPA as well as the involvement of the individual laboratories. Recently, additional CRI have been proposed for further biochemistry tests, and it is hoped that harmonization in this field will continue.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethical approval
Not applicable.
Guarantor
GJ.
Contributorship
SK extracted the data, GJ and SK analysed the data and wrote the manuscript.
