Abstract
The aldosterone renin ratio (ARR) is used as a screening test for primary aldosteronism (PA), and this requires measurement of both aldosterone and renin. Aldosterone is usually measured using immunoassay or liquid chromatography-mass spectrometry techniques. Antihypertensive medications should be discontinued prior to screening as they can interfere with interpretation of results. Indapamide is a thiazide-like diuretic commonly used in the treatment of hypertension. NICE guidance (NG136 2019) recommends the use of indapamide over more conventional thiazide diuretics. Indapamide has been noted to cause interference in a liquid chromatography-mass spectrometry method by interfering in the measurement of aldosterone-d4 internal standard. This leads to falsely increased concentrations of aldosterone which can lead to unnecessary further investigations.
Background
The aldosterone renin ratio (ARR) is used as a screening test for primary aldosteronism (PA), and this requires measurement of both aldosterone and renin. Current endocrine society guidelines suggest screening all patients with hypertension using the ARR. 1 Ideally, antihypertensive medications should be discontinued prior to screening but this is not always possible. Antihypertensive drugs can alter interpretation with false-positive and false-negative ARRs possible. 1
There are two common techniques used for aldosterone measurement: immunoassay and mass spectrometry. 2 Positive bias has been widely reported for immunoassay-based methods compared to liquid chromatography-tandem mass spectrometry (LC-MS/MS) methods. 3 This bias is likely due to cross-reactivity of structurally similar compounds in immunoassay methods. 2
LC-MS/MS measurement of aldosterone is technically challenging due to low endogenous concentrations (pmol/L) and poor ionization properties requiring a mass spectrometer with high analytical sensitivity. 3 Several methods have been published for sample preparation using supported liquid extraction (SLE) and solid phase extraction (SPE) techniques. 3 Most published methods use calibrators prepared in house, and both d4 and d7 internal standards have been reported.4,5
Indapamide is a thiazide-like diuretic commonly used in the treatment of hypertension. NICE guidance (NG136 2019) recommends the use of indapamide over more conventional thiazide diuretics. 6 As well as a mild diuretic effect, it is believed to have direct vascular effects causing vasodilation. 7
Indapamide is available in both immediate and sustained release formulations. It is widely distributed throughout the body, highly protein bound and undergoes extensive hepatic metabolism, with <7% excreted unchanged in the urine. 7 Studies report peak plasma levels 1–2 h post-dose, with plasma concentrations of 154 ± 35 ng/mL reported for once daily 2.5 mg dosing and an elimination half-life of 14 h. 8
Method
Calibrators (Chromsystems 6PLUS1® Multilevel Serum Calibrator Steroid Profile 1), internal quality control material and patient samples were mixed with deuterated internal standard (Aldosterone-9,11,12,12-d4, Sigma Aldrich) and loaded onto a Biotage ISOLUTE®-supported liquid extraction (SLE+) plate. The plate was eluted with MTBE (HPLC grade, Honeywell), evaporated to dryness and reconstituted with 40% methanol.
Chromatography was performed on CORTECS C18 2.1 ×100 mm column, mobile phase A was de-ionized water (18.2 mΩ) and mobile phase B was methanol (LC-MS grade, Merck Millipore) without additives. Gradient elution was used starting from 40% B to 65% B over 3 min, followed by 95% B wash step and re-equilibration to starting conditions. Aldosterone eluted at 2.0 min with a total run time of 6 min.
Mass spectrometry was performed on a Waters Xevo® TQ-XS and a Waters Xevo® TQ Absolute in negative ion mode. Analyte and deuterated internal standard (IS) were detected in multiple reaction monitoring (MRM) mode. On Waters Xevo® TQ-XS MRM transitions were m/z 359.4 > 189.3 (quantifier), 359.4 > 297.2 (qualifier) and 363.4 > 190.3 (IS). On Waters Xevo® TQ Absolute MRM transitions were m/z 359.3 > 189.1 (quantifier), 359.3 > 297.2 (qualifier) and 363.3 > 190.1 (IS).
Investigation
The aldosterone LC-MS/MS method was implemented in our laboratory in August 2024. During the routine review of results, several patient samples had unusual chromatograms not seen during the method development and validation performed in 2020–2021. Three over-lapping peaks were observed for the aldosterone-d4 IS MRM at 1.9, 2 and 2.1 min rather than the expected single peak. Aldosterone quantifier and qualifier chromatography was as expected, and no ion ratio flags were present.
For all samples with multiple peaks, clinical details and drug history were obtained from the electronic patient record. As expected, the patients were prescribed multiple antihypertensive agents as well as drugs for other comorbidities. Drugs common to all patients were reviewed and information obtained on their mass and chemical structure. Indapamide was identified as a potential interferent.
Indapamide analytical standard (Supelco, ≥99% purity) was purchased from Merck (Mw = 365.83). A solution of 100 ng/mL was prepared in delipidated human serum (0.2 µM sterile filtered, TCS Biosciences) and analysed using the LC-MS/MS aldosterone method with and without the addition of IS. No peaks were observed for the aldosterone quantifier or qualifier transitions. On the Waters Xevo® TQ-XS, a peak was observed in the IS MRM with RT 2.2 min for indapamide and 2.1 min for aldosterone-d4. On the Waters Xevo® TQ Absolute, two peaks were observed in the IS MRM at 1.9 and 2.1 min, the latter having a much greater peak area. Aldosterone-d4 peak was observed at 2.0 min. The peaks observed for indapamide overlapped with that of the internal standard (Figure 1). Chromatograms for IS MRM m/z 363.4 > 190.3 on Waters Xevo® TQ-XS for (a) aldosterone-d4 IS, (b) indapamide, (c) aldosterone-d4 IS and indapamide and MRM m/z 363.3 > 190.1 on Waters Xevo® TQ Absolute for (d) aldosterone-d4 IS, (e) indapamide, (f) aldosterone-d4 IS and indapamide.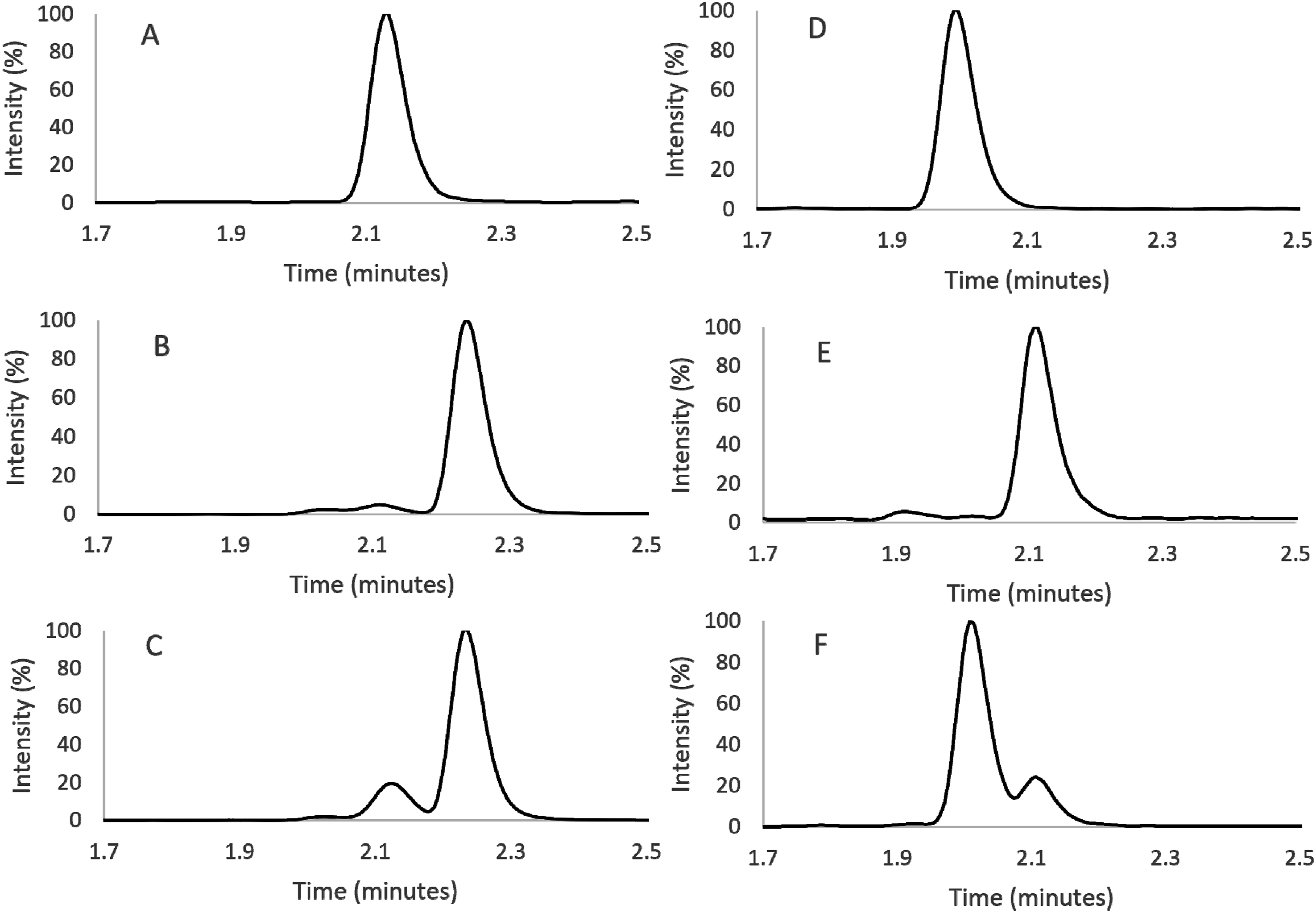
If indapamide is present, the internal standard area is underestimated and therefore aldosterone concentration is falsely increased. On the Waters Xevo® TQ-XS the aldosterone-d4 peak area is smaller than the indapamide peak area, the opposite is observed on the Waters Xevo® TQ Absolute.
Alternative chromatography conditions using the same column and alternative IS MRMs were investigated but were unsuccessful. Interference from indapamide was not observed for m/z 363.4 > 301.2; however, commercial quality control material gave variable results compared with patient samples.
Aldosterone 13C3 internal standard (10 µg/mL solution, purity >99.9%) was obtained from Cerilliant for investigation. This was added to the existing method with m/z 362.3 > 192.2. Injection of a 100 ng/mL indapamide solution prepared in delipidated human serum showed no peak for this MRM. Several routine aldosterone assays were extracted using Aldo-d4 and 13C3 labelled internal standard to allow comparison of patient results. Indapamide was tuned and MRMs added to the method, m/z 364 > 232.9 and 364 > 188.9. A batch of samples were analysed, and all samples with additional peak at 2.1 min in Aldo-d4 on this batch were confirmed to contain indapamide.
Results
Patient samples (n = 723) were analysed over different assays on the Waters Xevo® TQ Absolute and data calculated using both internal standards. One hundred and twelve samples were excluded as they were below the lower limit of quantitation (75 pmol/L). Indapamide was detected based on chromatography in 4% of samples tested (Figure 2). Comparison of aldosterone results using d4 IS versus 13C3 IS. Samples with indapamide detected (triangles) and those without (circles).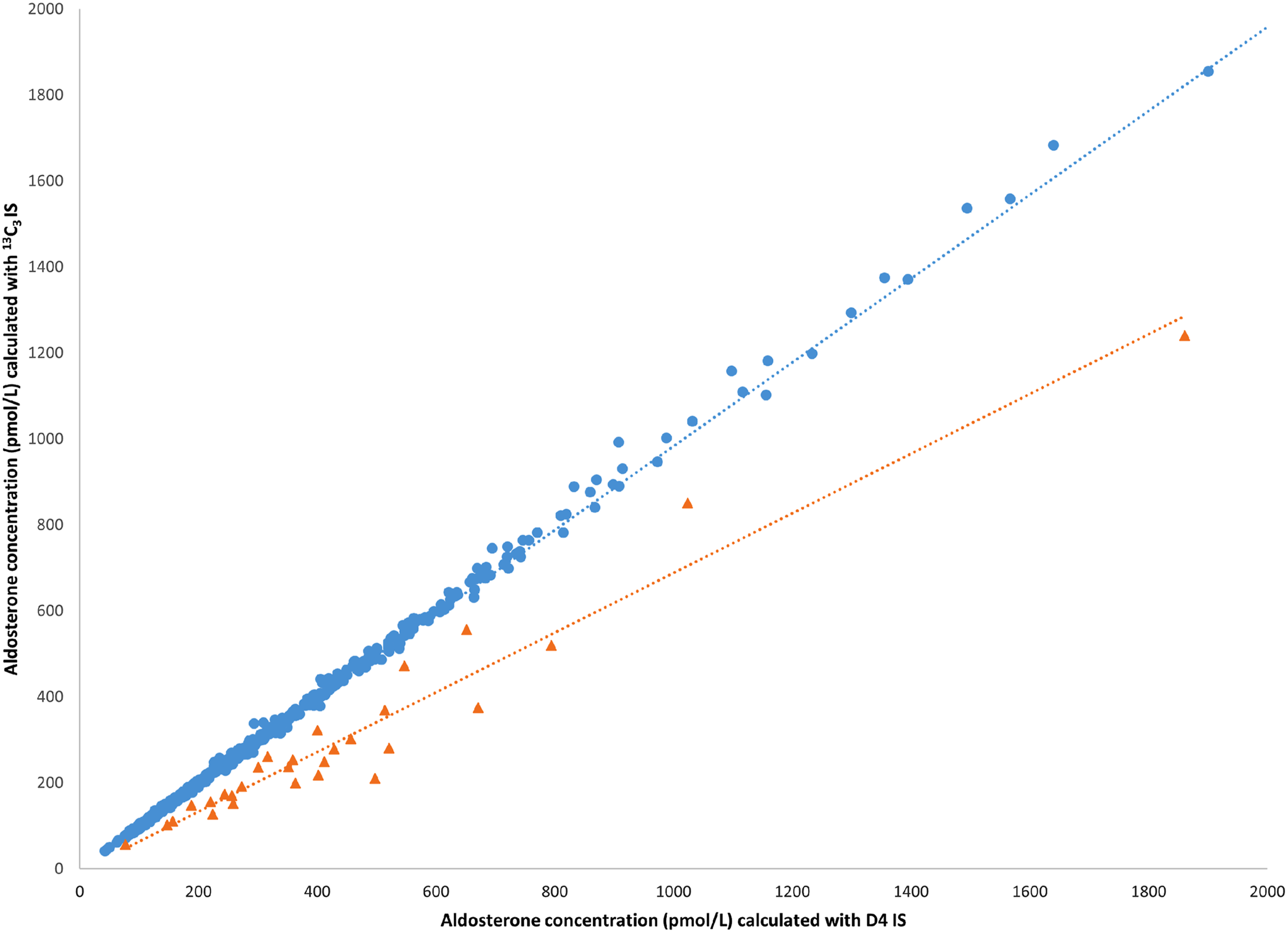
Discussion
This example highlights the requirement for thorough interference testing during validation with a special emphasis on the context in which the assay is typically used, for example antihypertensive drugs in patients investigated for PA, as well as ongoing review of established methods in clinical use due to changes in treatment guidelines. Although many guidelines recommend withdrawal of antihypertensive medications prior to screening, it is important to remember many patients are tested whilst on medications.
During the initial validation interference testing was performed, and this included other steroids, for example, prednisolone, dexamethasone, hydrocortisone, and fludrocortisone, and antihypertensives, for example, spironolactone. Indapamide interference was not specifically tested; however, a retrospective review of the chromatograms did not identify any patient samples containing indapamide during the initial validation. This suggests that a slow transition process occurred after the introduction of the new NICE guidance in August 2019 before the implementation of our method in August 2024. The guideline recommended continuing a conventional thiazide diuretic if blood pressure was well controlled. However, if starting or changing a diuretic then indapamide was recommended over conventional therapy.
It is likely that there has been an increase in indapamide prescribing since 2019; however, there is no published data available for direct comparison between 2019 and 2025. Indapamide is now the preferred thiazide-like diuretic in several Scottish formularies. The only other thiazide-like diuretic on Scottish formularies is metolazone (Mw 365.8), limited to secondary care only. Further investigation is required to determine if this causes interference.
Interference testing for drugs can be challenging as many drugs like indapamide undergo extensive metabolism in the body and therefore it is difficult to determine the extent of parent drug that remains in plasma to cause interference. As seen in our cohort of samples, 4% of patient sample analysed contained sufficient indapamide concentrations to interfere with quantification.
During the investigation, analysis was performed on two different LC-MS/MS systems; the interference was present on both analysers, but relative intensities of indapamide to IS varied between analysers. This suggests that the interference may have a less impact on the Waters Xevo® TQ Absolute method, but we have insufficient patient data to determine this at this time.
If this indapamide interference was not identified, then falsely elevated results would have been reported. The extent of the impact of indapamide interference is difficult to determine. In our cohort of patients, many of those on indapamide at the time of sampling had a raised renin. Indapamide interference may have led to unnecessary further investigations such as saline suppression tests and potential imaging investigations. This would have caused significant extra cost to the NHS and potential worry to patients.
Footnotes
Acknowledgements
The authors would like to thank the staff working in the LC-MS/MS laboratory at Glasgow Royal Infirmary for running the routine patient samples.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethical approval
Not required.
Guarantor
AF.
Contributorship
All authors reviewed and edited the manuscript and approved the final version of the manuscript.
