Abstract
Background
Gadolinium-based contrast agents (GBCAs) have been used in magnetic resonance imaging for the past 30 years, where they have significantly improved the effectiveness of imaging studies. However, the increased usage of gadolinium in the medical community has also resulted in unexpected interferences in other laboratory assays. This has been particularly the case in clinical elemental analysis using inductively coupled plasma mass spectrometry (ICP-MS).
Methods
By conducting ICP-MS interference experiments, we describe how gadolinium interferes with elemental analysis by space charge effect, double charge effect and the creation of polyatomic interferences. Additionally, by reviewing more than a year of reference laboratory data from our laboratory information system, we determined the number of elemental tests cancelled due to gadolinium interference.
Results
Interference experiments show that gadolinium normally found in GBCAs can interfere with heavy metals, platinum and selenium analysis of biological fluids using ICP-MS. Within one year, our institution’s metals laboratory had to cancel 42 selenium serum tests and 19 heavy metal urine panels due to potential interference caused by gadolinium.
Conclusions
GBCAs will continue to be utilized in hospitals around the world. However, clinical laboratories should be wary of potential interferences caused by GBCAs. Relevant interferences include space charge effect, double charge interference, and the formation of polyatomic interferences caused by gadolinium. These interferences can negatively affect patient care by resulting in cancelled laboratory tests and causing patients to have blood redrawn and analysed at a later date leading to delays in their diagnosis/treatment.
Introduction
Gadolinium-based contrast agents (GBCAs) have been used in magnetic resonance imaging (MRI) for the past 30 years. During this time, GBCAs have significantly improved the effectiveness of imaging studies. 1 These GBCAs have improved the imaging of tumours, blood vessels and inflammation. 1 Due to these improved capabilities, an estimated 30-million gadolinium-enhanced MRIs are conducted around the world each year. 2 The increased utilization of GBCAs in medical practice has, however, increased the incidence of high concentrations of GBCAs found in laboratory specimens including blood, serum and urine. GBCAs present in biological samples at these high concentrations have caused significant challenges in the clinical laboratory. GBCAs have been shown to interfere with numerous clinical laboratory tests, thus resulting in delayed result turnaround times, method redevelopment and the potential release of inaccurate laboratory test results.
Known analytical interferences
It has been demonstrated that GBCAs potentially can interfere with numerous clinical tests throughout the field of laboratory medicine including inductively coupled plasma mass spectrometry (ICP-MS) methods for metal concentration determination in biological matrices. There has been significant research into how GBCAs interfere with serum calcium analysis by photometric testing using ortho-cresolphthalein.3–5 Furthermore, GBCAs can interfere with creatinine analysis by the Jaffe reaction and total iron binding capacity by the ferrozine method.3,6 The presence of significant concentrations of GBCAs has also been shown to affect ICP-MS methods for the evaluation of selenium concentrations. 7 However, the analysis of selenium by ICP-MS is certainly not the only ICP-MS method affected by GBCAs. Through multiple mechanisms, gadolinium from GBCAs can potentially interfere with other ICP-MS-based elemental assays when significant concentrations of gadolinium are present. These gadolinium-based interferences have resulted in the cancellation of clinical tests, which ultimately impacts patient care (e.g. re-collection of blood samples and delay in test results and clinical intervention).
This paper describes how gadolinium, primarily present as GBCAs, interferes with the analysis of multiple elements by ICP-MS in the clinical laboratory. Additionally, the clinical impact of these GBCA-based interferences will be discussed using data from our institution’s metals laboratory. Finally, a means of how gadolinium interferences can be attenuated during the development of ICP-MS methods will be described.
Gadolinium as an analytical interference in ICP-MS analysis
ICP-MS is an effective way to analyse metals in human samples including blood, urine, serum and tissue. The large dynamic range and multielement capabilities make ICP-MS an essential piece of equipment in any clinical metals laboratory. ICP-MS quantifies elements by determining the mass to charge ratio of the target analyte. ICP-MS accomplishes this by first ionizing the sample using an inductively coupled plasma. The metal ions are then separated by a mass filter and ultimately quantified by an ion detector. 8 This allows for the accurate analysis of most elements in the parts per billion (ppb) range. However, as with most analytical platforms, ICP-MS suffers from a number of potential interferences. Three types of interferences seen in biological samples collected from patients recently administered GBCAs and analysed by ICP-MS include: space charge effect, double charge interference and polyatomic ion formation interference.
Space charge effect
‘Space charge is a net charge imbalance caused by an excess of charged particles, which are positive ions in the beam extracted from the inductively coupled plasma’. 9 For example, a patient urine sample is collected after recently receiving a GBCA MRI. Due to the patient’s history, physical signs and symptoms, the physician orders heavy metal testing (arsenic, mercury, cadmium and lead) in a urine sample. In the clinical laboratory, internal standards (e.g. other elements) are added to the sample for proper quantitation. Appropriate internal standard elements usually consist of elements with a similar atomic mass and ionization potential as the element of interest. These internal standard elements are used to monitor the ionization and detection capabilities of the test method. After ionization in the plasma, an excess of gadolinium ions from the urine sample can push the internal standard analyte ions to the periphery of the ion beam. This can result in less internal standard analyte ions travelling through the instrument quadrupole and ultimately result in an under-recovery of target analyte ions at the ion detector.
Double charge interference
Another mechanism by which gadolinium can interfere with elemental analysis by ICP-MS is by double charge interference.7,10,11 For example, 156Gd can interfere with selenium analysis if a clinical laboratory chooses 78Se as the ideal isotope to analyse. Theoretically, most inductively coupled plasma assays remove one electron from the outer shell of the atom resulting in a + 1 charge. However, it is also possible to remove two electrons from the atom resulting in a + 2 charge. The ability to form this double charge state is dependent on the ionization energy of an analyte. As a result of the doubly charged ion formation and the fact that ICP-MS identifies ions based on the mass to charge ratio, the mass spectrometer identifies the 156Gd2+ as 78 amu. Therefore, patients who received a GBCA immediately prior to having a sample drawn for selenium may have a falsely elevated result for selenium due to the presence of double charged ions in the instrument plasma.
Polyatomic interference
A third way in which gadolinium from GBCAs can interfere with elemental analysis by ICP-MS is by formation of polyatomic ions in the plasma. For example, platinum in human matrices can be analysed using isotope 195Pt. However, within the inductively coupled plasma, residual gadolinium found in the sample can combine with argon or oxygen from the plasma, or chloride from the sample to form various polyatomic interferences. All of these polyatomic ions can have an atomic mass of 195, which happens to be the same mass as the analyte of interest. If not properly attenuated, these polyatomic ions will reach the ion detector and be quantified as 195Pt. The formation of gadolinium polyatomic ions associated with the determination of platinum has been previously described. 12 , 13
Methods
Space charge experiment
A PerkinElmer NexION 300 ICP-MS (Waltham, MA) was utilized to quantify potential internal standard elements including gallium, germanium, yttrium and rhodium with the use of helium in kinetic energy discrimination (KED) mode. Single-element gallium, germanium, yttrium and rhodium standards from Inorganic Ventures (Christiansburg, VA) were used to create an internal standard in-line solution. A slightly basic diluent was also utilized for this assay. Five human urine samples were analysed using this method to acquire a baseline recovery of internal standard elements in a human urine matrix. This was attained by first diluting the urine samples with the basic diluent. Then, the urine samples were mixed with the in-line internal standard solution immediately prior to being introduced into the ICP-MS. Baseline internal standard counts per second were then acquired from the ICP-MS. A single-element standard from Inorganic Ventures containing 1000 parts per million (ppm) gadolinium was then spiked into each of the five urine samples. Using the same in-line approach, the internal standard solution was mixed with the spiked urine sample and then introduced into the ICP-MS. Recovery of potential internal standard elements was then determined in the urine samples spiked with gadolinium at 1000 ppm. The average percent recovery among the urine samples for each internal standard element was then determined.
Double charge experiment
A PerkinElmer NexION 350D ICP-MS with PC3 Peltier cooler (Waltham, MA) was utilized to quantify selenium with the use of ammonia in dynamic reaction cell (DRC) mode. Salt-matrix standards and a slightly basic diluent were utilized in this assay. A single-element gadolinium standard from Inorganic Ventures was used to spike concentrations of 0, 100, 1000, 10,000, 500,000 and 1,000,000 ppb into an acidic matrix. For the double charge experiment, one part of the aqueous spiked standard was added to the appropriate matrix of patient samples to determine the percent of recovery.
Polyatomic interference experiment
Using a PerkinElmer Elan ICP-MS (Waltham MA) in standard mode, a slightly acidic solution containing 1 ppb of platinum was aspirated into the instrument, and platinum counts per second were recorded at isotopes 194Pt, 195Pt, 196Pt and 198Pt. A single-element platinum standard from Inorganic Ventures was used to create the 1 ppb platinum solution. Next a 2% hydrochloric acid solution containing 10,000 ppb of gadolinium was aspirated into the instrument. In theory, the gadolinium would combine with the chloride ions from the hydrochloric acid solution within the sample introduction system and the instrument creating a gadolinium chloride polyatomic interference at mass 195. The instrument recorded counts per second at mass 195 to quantitate this interference created by the gadolinium chloride complex.
Laboratory results review
Using our laboratory information system, we reviewed data from clinical samples analysed and reported in 2017. Specifically, we identified samples that were cancelled and/or commented on due to potential gadolinium interference. For clinical ICP-MS methods that have the potential for gadolinium interference, our laboratory uses a gadolinium standard that is included in the assay calibration. The assay then monitors patient samples for gadolinium throughout the analysis. If a sample has a gadolinium concentration above the threshold where it can affect or interfere with the ability to accurately quantify the specific element being measured according to the laboratory standard operating procedure, the sample is either cancelled or commented on in the laboratory information system. For this study, our review of the laboratory information system included samples analysed in 2017. It also only included patient samples analysed on clinical assays where gadolinium was actively being monitored, because during method validation, it was determined that gadolinium could significantly affect those assays. The assays included a heavy metal urine panel (arsenic, cadmium, lead and mercury) and a selenium, copper and zinc serum assay.
Results
Space charge experiment
As is shown in Figure 1, gadolinium at a concentration of 1000 ppm can suppress the signal of commonly used internal standard elements used in elemental urine analysis. Gadolinium at a concentration of 1000 ppm decreased the average per cent recovery of gallium, germanium, yttrium and rhodium by roughly 15%.
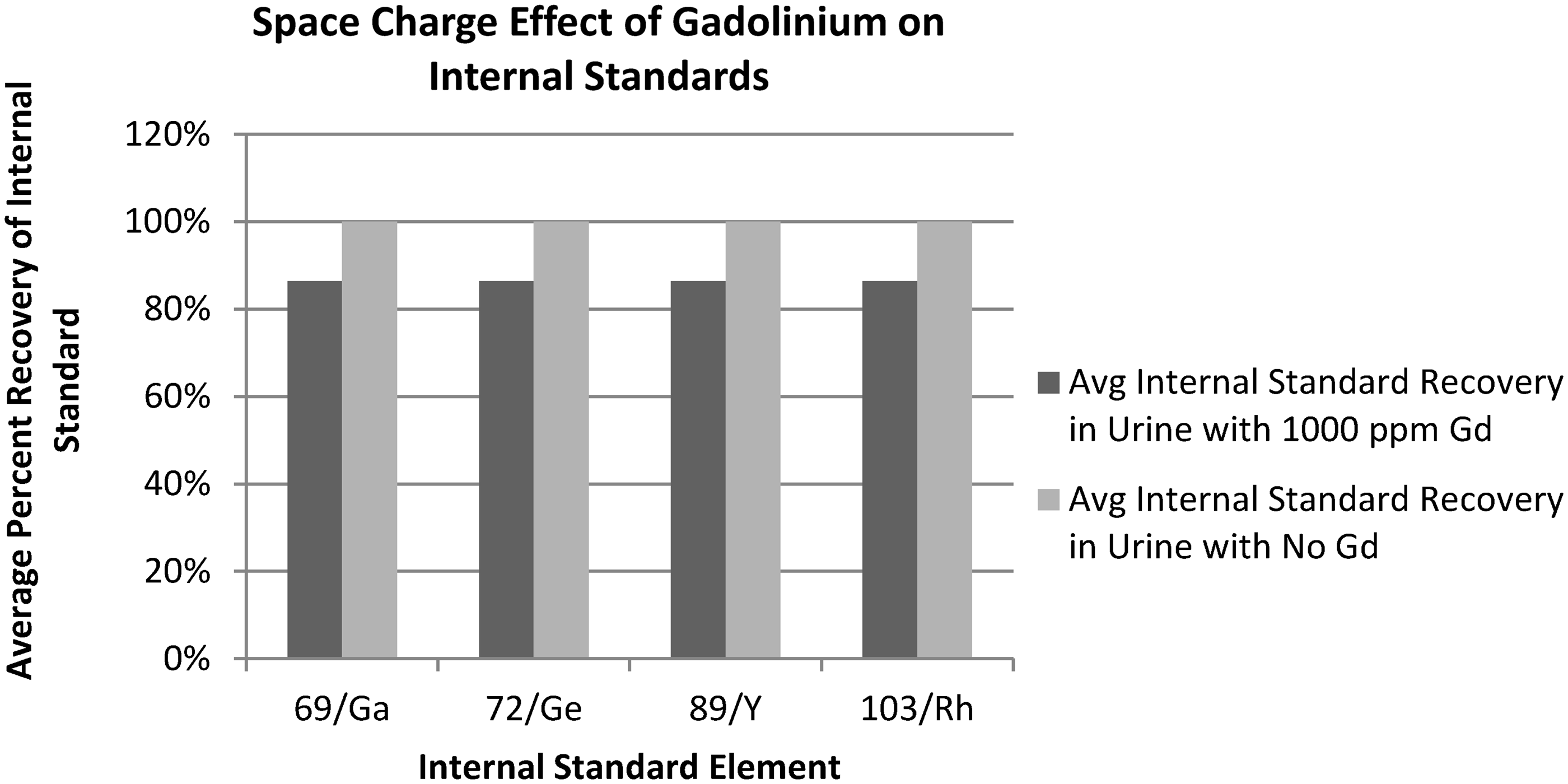
Space charge effect of gadolinium on possible internal standards.
Double charge experiment
A depiction of the effect of gadolinium double-charge can be seen in Figure 2. At 10,000 ppb, selenium in whole blood recovery is 113% and in serum is 111%. In samples with up to 500,000 ppb of gadolinium, the percent recovery is 720% in blood and 632% in serum due to the double charge interference on selenium.
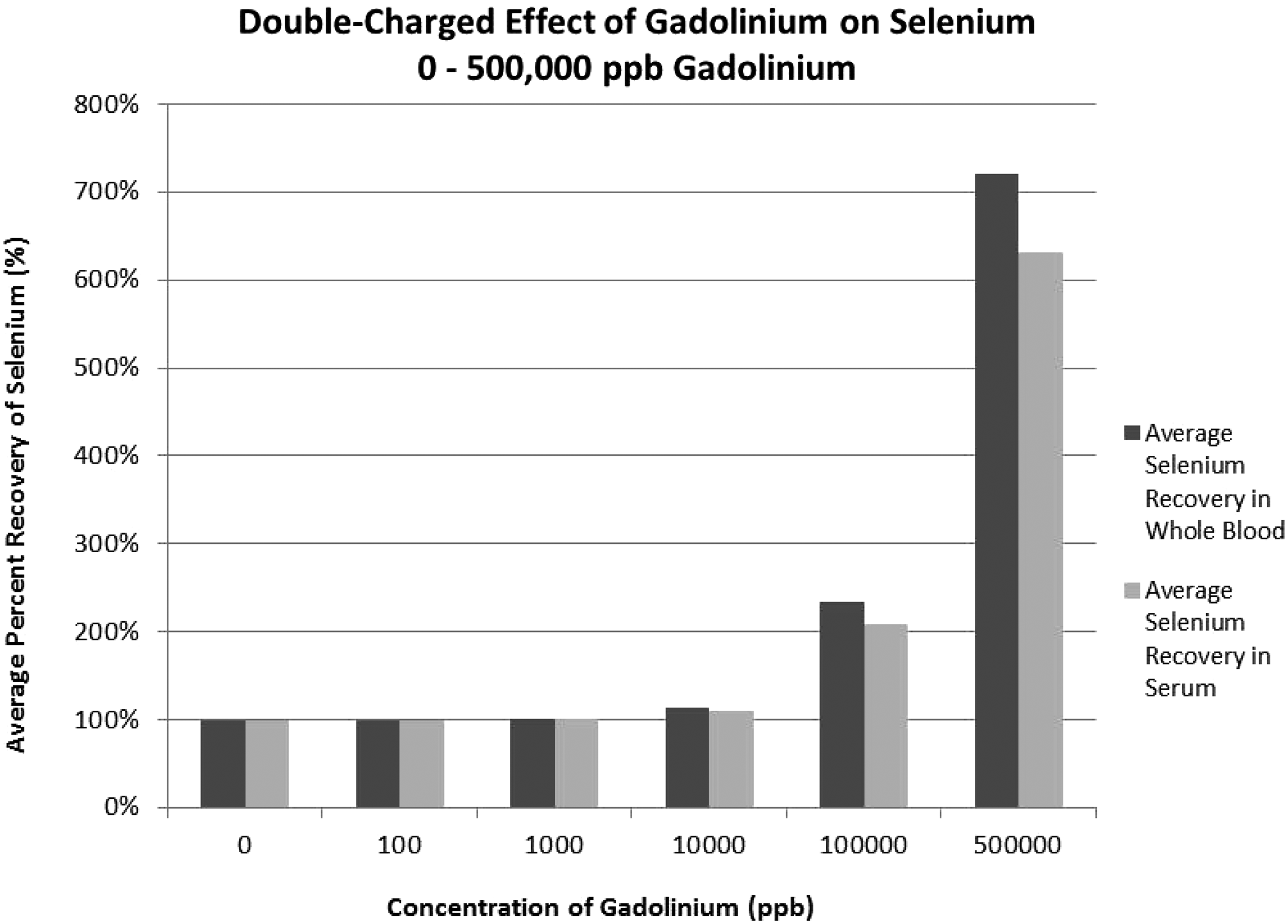
Double charge effect of gadolinium on selenium concentration determination.
Polyatomic interference experiment
Figure 3 depicts how gadolinium chloride polyatomic ion formation results in a falsely elevated platinum result due to the formation of polyatomic ions. The addition of 10,000 ppb of gadolinium resulted in a gadolinium chloride polyatomic interference at mass 195. This results in a falsely elevated signal when measuring platinum at mass 195.
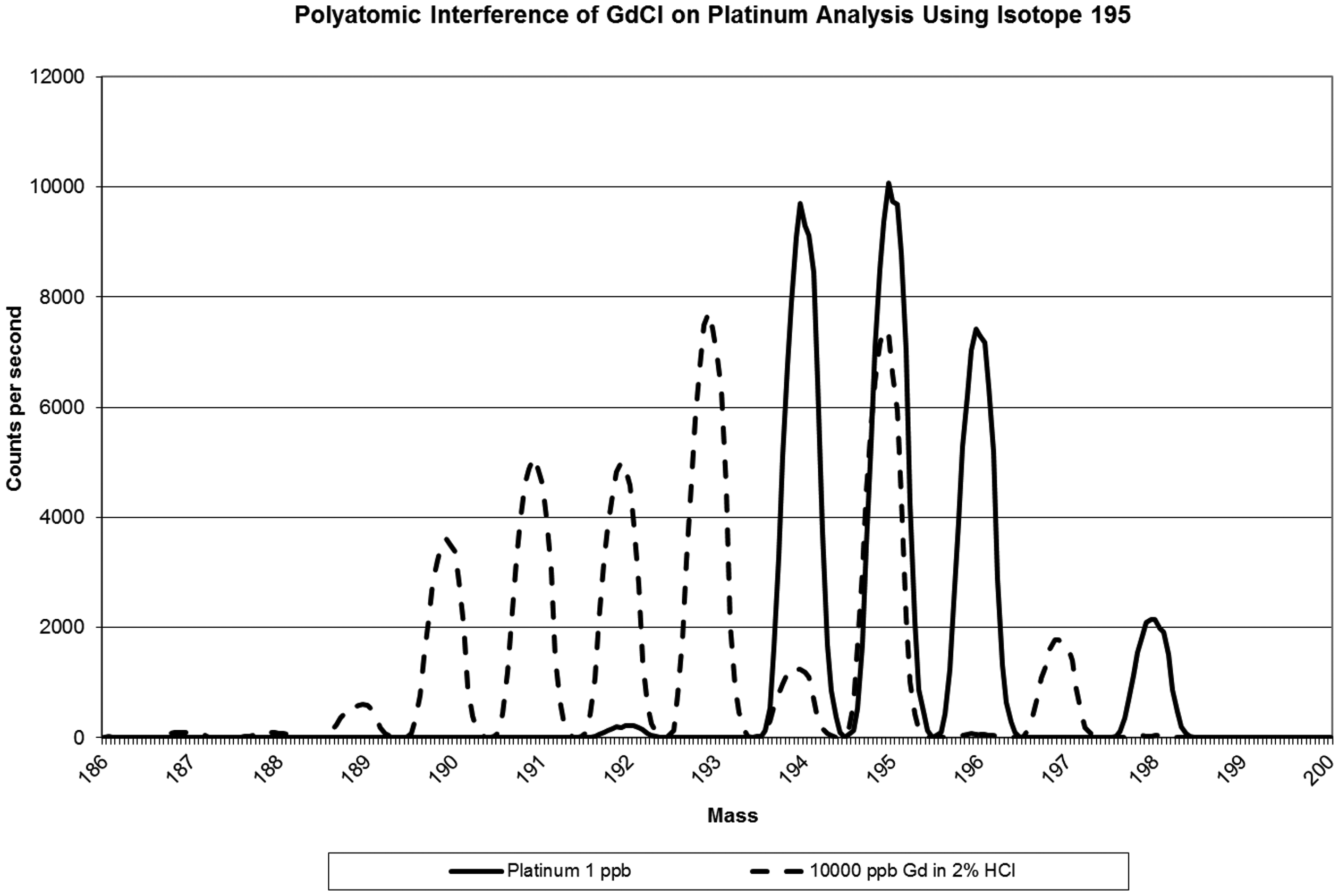
Polyatomic interference of gadolinium chloride on platinum analysis.
Specimen cancellations
Selenium
At our institution’s clinical metals laboratory, gadolinium-based interferences have resulted in tests being cancelled and specimens having to be redrawn. These situations can potentially affect and delay patient care. Using our laboratory information system, in 2017, the metals laboratory had to cancel 42 selenium serum tests due to the presence of an elevated concentration of gadolinium, and thus the potential of gadolinium double charge interference falsely biasing the selenium result. The laboratory incorporated a gadolinium single-point calibrator in the selenium serum assay calibration to monitor gadolinium concentration of samples because method validation demonstrated that gadolinium present at concentrations around 1000 ppb could falsely bias the selenium result by roughly 9%. At a concentration of 5000 ppb, gadolinium could falsely bias a selenium serum result by roughly 40%. Due to this significant bias, the laboratory decided to cancel any sample with a gadolinium concentration greater than 1000 ppb in order to prevent reporting a falsely elevated sample due to the gadolinium double charge interference. In addition, 998 comments were added to the electronic medical record for copper and/or zinc serum tests, indicating that selenium could not be ordered as an add-on test due to an elevated concentration of gadolinium that would interfere with the analysis of selenium. This is clinically troublesome because of the importance of selenium within the body. Selenium is an essential trace element, which is a constituent of greater than 30 biologically active enzymes including: glutathione peroxidase, iodothyronine deiodinase and thioredoxin reductase. 14 Symptomatic selenium deficiency is present in Keshan Disease and nutritional depletion in hospitalized patients. 14 Marginal deficiencies are also thought to be involved in immune function, mood disorders, thyroid function, reproductive disorders, inflammatory conditions, cardiovascular disease, viral virulence and cancer chemoprevention. 14 In addition, toxicity can also occur in cases with excessive dietary exposure. The inability to provide a selenium serum result can potentially impact the ability to clinically manage, diagnose or treat patients.
Heavy metal analysis
Using a gadolinium calibrator in the heavy metal urine panel, gadolinium concentrations are also monitored in patient urine samples. In the first six months of 2017, the metals laboratory had to cancel 19 heavy metal urine panel results, which included the quantitation of arsenic (As), mercury (Hg), cadmium (Cd) and lead (Pb). As described by the above space charge experiment and depicted in Figure 1, it was determined that gadolinium present at a concentration of roughly 1000 ppm in the collected urine samples could falsely decrease the assay internal standard by roughly 15%, making accurate quantification of these elements impossible. Therefore, these patient urine samples had to be recollected, causing a delay in the detection and potential diagnosis of heavy metal exposure. Urine is the preferred screening matrix for occupational, environmental, dietary or intentional exposure to these elements (As, Hg, Cd) with the exception of lead where blood is the preferred matrix. However, to diagnose heavy metal toxicity, the source of the exposure, signs and symptoms of toxicity and measurement of an abnormal concentration of the element in the appropriate matrix must be demonstrated. 14 As a result, the metals laboratory had to subsequently redevelop this heavy metal urine panel, utilizing different internal standards unaffected by gadolinium interference, to avoid further cancellations. These clinical situations resulted in additional troubleshooting, assay redevelopment and most importantly a delay in getting results to the ordering provider which ultimately could impact a patient’s care and management.
Discussion
There are several ways that clinical laboratories can attenuate the various ways that gadolinium can interfere with elemental analysis by ICP-MS. The easiest way to minimize potential gadolinium interference is by identifying and removing samples with potentially elevated concentrations of gadolinium from being analysed. In order to effectively remove elevated gadolinium samples from analysis, it is important to understand the excretion rate of GBCAs. These contrast agents are distributed rapidly within the body and are eliminated by the kidneys. A fast initial elimination (half-life approximately 2 h) is followed by a slow elimination phase (half-life approximately six days), reflecting slow release from tissues. 15 Based on these findings, recommendations can be made to the physician about delaying collection of samples within a certain period of time after the administration of GBCA. Our institution’s metals laboratory has incorporated test instructions stating that samples should not be collected less than 96 h after a GBCA MRI. These ordering instructions have been effective at lowering our test cancellation rate due to gadolinium interference. These ordering instructions in tandem with continued physician education are front-line barriers used to ensure that samples with potentially elevated concentrations of gadolinium are not analysed for various elements by ICP-MS.
Previous work has shown that urine samples from patients with normal renal function and collected three days after administration of GBCAs contained on average 1944 μg of gadolinium per 24 h collection. 16 Furthermore, our laboratory has received patient samples with gadolinium concentrations >160,000 ppb. As a result, the laboratory needs additional safeguards other than ordering instructions to prevent samples with elevated concentrations of gadolinium from being analysed and inaccurate results being reported. Therefore, it is imperative that clinical laboratories first conduct interference experiments during validation of clinical assays like those described above. Clinical laboratories should also calibrate and cancel elemental tests for gadolinium in certain circumstances that indicate interference by gadolinium.
Specifically related to the reduction of space charge effect, validation experiments can be conducted to determine the concentration of gadolinium which causes the space charge effect to interfere with the analyte of interest or internal standard analytes. Once this concentration is determined, workflow rules can be established to cancel samples when the gadolinium concentration exceeds this threshold. As stated above, our laboratory has received patient samples with gadolinium concentrations >160,000 ng/mL in our gadolinium assay. For this reason, a gadolinium concentration range of 0 to 1,000,000 ppb was deemed clinically appropriate when conducting space charge interference experiments.
Double charge interference can be resolved by choosing a different isotope of the target analyte or the internal standard used in the clinical assay. However, prior to switching to a different isotope for analysis, the isotopic abundance of the new isotope and possible polyatomic and isobaric interferences must be considered. For some elements, there is only one or two viable isotopes for analysis, so double charge interference can be troubling. Additionally, choosing a different isotope to quantitate will involve the development of a new analytical method which will result in time and resources being spent to develop a new assay.
Polyatomic interferences can be resolved through various techniques including the use of KED and DRC technology. Polyatomic interferences can be attenuated with either the use of DRC and KED instrument modes at specified cell gas flows. Many DRC-based ICP-MS methods use ammonia as a reaction gas to undergo a gas-phase chemical reaction with the interfering species and ultimately remove these interferences from the ion pathway via charge-transfer reactions. Various DRC-based methods have been previously described. 17 , 18 Another way to attenuate polyatomic interferences when using ICP-MS involves utilizing KED technology. Numerous KED-based methods have used helium as a non-reactive collision gas which, in tandem with a bias voltage at the cell exit aperture, effectively limits entry of interfering ions into the analyser quadrupole. 8 , 19 , 20 While there are ways to address each of these potential interferences, each involves additional test development time and resources.
While there are certainly different ways in which gadolinium from GBCAs can interfere with elemental analysis using ICP-MS, there are other ways that GBCAs can also interfere with ICP-MS laboratory assays. These interferences must be considered when developing ICP-MS elemental laboratory assays. First, preanalytic sample preparations must be carefully considered when developing assays where gadolinium from GBCAs can potentially interfere with assay results. It is imperative that sample cross-contamination during sample preparation and analysis is eliminated. If, for example, a urine sample is collected from a patient right after a GBCA MRI and it accidently contaminates a sample from a different patient needing elemental analysis; the gadolinium from the MRI patient could interfere with the other patients results. This could result in patient results being delayed because of sample cross-contamination.
Secondly, GBCAs not only contain gadolinium but also powerful chelators. 21 Gadolinium on its own is a toxic element. 22 In order to utilize its paramagnetic properties safety in the MRI application, linear or macrocyclic chelators are used to bind or encapsulate the gadolinium element while in vivo. This helps prevent the toxic elemental gadolinium from absorbing into tissue during the imaging process and potentially causing health problems. However, there are two types of GBCAs (e.g. linear and macrocyclic) based on their structure. Linear GBCA have been shown to result in more retention and retention for a longer time than macrocyclic GBCAs. 23 While these chelated gadolinium complexes are excreted from the body usually through urine after the imaging process, it is not understood if these chelating agents are affecting the elemental analysis by ICP-MS. The concentration of these powerful chelators can be quite high if a sample is drawn soon after a MRI, which could result in a matrix effect interference. To further complicate the situation, depending on the manufacturer of the GBCA, different chelators are used and ultimately excreted through the urine. The presence of these chelators in the urine sample could drastically alter the sample matrix, which in turn could interfere with the accurate analysis of the target analyte. A usual way to attenuate this type of interference is to matrix match the calibrators to the type of sample being analysed; however, it would not be possible to include every type of chelator used in GBCAs within the calibrating standards. Further studies must be conducted to study the matrix effects of these different chelators on ICP-MS elemental analysis.
Environmental gadolinium contamination could also potentially interfere with the accurate analysis of gadolinium itself. As gadolinium is found in the environment, gadolinium contamination of samples remains a possibility in the collection or analytical phase of ICP-MS analysis. Gadolinium is persistently found in water sources, which might be a potential source of analytical contamination.24,25 Although the putative gadolinium concentrations associated with general environmental contamination are thought to be only potentially disruptive in the analysis of gadolinium itself and have not been confirmed in other assays to date, avoidance of this type potential environmental contamination should be considered.
Conclusion
GBCAs will continue to be effectively utilized in hospitals and clinics around the world. However, clinical laboratories must be aware of potential interferences caused by GBCAs. In clinical metals laboratories, these potential interferences include space charge effect, double charge interference and the formation of polyatomic interference caused by gadolinium. These interferences do indeed affect patient care, resulting in cancelled laboratory tests, and delayed patient results. Furthermore, these analytical interferences have caused numerous laboratory methods to be re-developed, resulting in increased time and expenses to the laboratory. It is imperative that clinical laboratories attenuate interferences and continue to further research possible matrix effect interferences caused by these GBCAs.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethical approval
Not applicable
Guarantor
PD.
Contributorship
All authors contributed equally to this work.
