Abstract
Background
Multiple myeloma patients may present with spuriously elevated serum phosphate levels resulting from the presence of paraproteins interfering in the phosphomolybdate UV assay. If this phenomenon is not recognized, patients possibly receive unnecessary treatments. This short report highlights the existence of paraprotein-related pseudohyperphosphatemia, and aims to provide accessible solutions to eliminate this interference.
Material and Methods
In a patient known with IgG multiple myeloma and unexplained hyperphosphatemia, the correlation between serum phosphate levels (phosphomolybdate UV assay) and IgG concentrations (immunoturbidimetry) was evaluated. To investigate the effect of the paraprotein on phosphate levels, phosphate was measured in one serum sample before and after protein removal by either dilution, protein precipitation with sulfosalicylic acid or zinc sulphate, or ultrafiltration.
Results
A patient with multiple myeloma presented with an unexplained hyperphosphatemia which correlated positively with serum IgG concentrations. As serum dilution normalized the phosphate level, it was hypothesized that precipitation of the paraprotein during the assay reaction interfered with the measurement and resulted in pseudohyperphosphatemia. Protein removal by precipitation with sulfosalicylic acid or zinc sulphate efficiently reduced the IgG level below the detection limit but did not result in a reliable phosphate measurement. Successful removal of proteins and a serum phosphate level that matched the patient’s other biochemistry parameters and clinical condition were obtained by ultrafiltration.
Conclusion
Paraproteins can interfere with the reaction components in the phosphomolybdate UV assay and result in pseudohyperphosphatemia. If the presence of this phenomenon is established, a reliable phosphate concentration can be obtained after ultrafiltration of the sample.
Keywords
Background
Hyperphosphatemia presents when serum phosphate level exceeds 1.45 mmol/L and is most frequently caused by renal failure. 1 As renal impairment is a common issue in multiple myeloma, hyperphosphatemia may occur in these patients. 2
The phosphomolybdate UV assay is commonly used for the routine measurement of serum phosphate. 1 Paraproteins can precipitate during the assay reaction, resulting in an increased turbidity that ultimately leads to spuriously elevated phosphate levels. 3 It is impossible to predict paraprotein interference, as it is influenced by numerous factors such as assay composition and the physicochemical properties and concentration of the paraprotein. 4
This report highlights the existence of interfering paraproteins resulting in falsely elevated phosphate levels, and aims to provide and evaluate accessible solutions to eliminate this interference.
Materials and methods
Phosphate levels (phosphomolybdate UV assay) and IgG or IgA concentrations (immunoturbidimetry) were measured on an Atellica Solution (Siemens-Healthineers) in serum samples from three patients with multiple myeloma and unexplained hyperphosphatemia. One patient was diagnosed with IgG kappa multiple myeloma (patient 1), and two patients were diagnosed with IgA multiple myeloma, one IgA kappa (patient 2) and one IgA lambda (patient 3). For each patient, correlation between serum phosphate levels and IgG or IgA concentrations in multiple serum samples collected during a period of 1 year was assessed using Spearman’s rank correlation (MedCalc® v 18.11). A single serum sample from patient 1 was diluted in physiological saline (1/3 to 1/12) to verify whether the unexplained hyperphosphatemia resulted from the presence of interfering paraproteins. The phosphate and IgG concentrations were determined in each dilution and the dilution factor was taken into account.
The potential of protein removal techniques to eliminate interfering IgG paraproteins was investigated by measuring phosphate levels in one serum sample (patient 1) before and after protein removal. First, the serum sample was incubated for 15 min with either sulfosalicylic acid or zinc sulphate in methanol for protein precipitation. The second method to remove paraproteins was ultrafiltration of the serum by placing it in a tube with a Microcon 30 kDa membrane (Sigma-Aldrich) that was centrifuged at 10 500 × g for 30 min.
Results
Three patients with multiple myeloma (one IgG and two IgA) presented with an unexplained hyperphosphatemia (normal kidney function, calcium, vitamin D, and parathyroid hormone (PTH) level) which correlated positively with serum IgG (patient 1 ρ = 0.84) or IgA (patient 2 ρ = 0.75 and patient 3 ρ = 0.87) (Figure 1). Correlation (Spearman’s rank) between phosphate levels and IgG or IgA concentrations in multiple serum samples collected over time from three patients with IgG or IgA multiple myeloma. (a) Patient 1: correlation between phosphate and IgG levels is significant (n = 31; ρ = 0.84; P < .0001). (b) Patient 2: correlation between phosphate and IgA levels is significant (n = 15; ρ = 0.75; P = .0012). (c) Patient 3: correlation between phosphate and IgA levels is significant (n = 12; ρ = 0.87; P = .0003).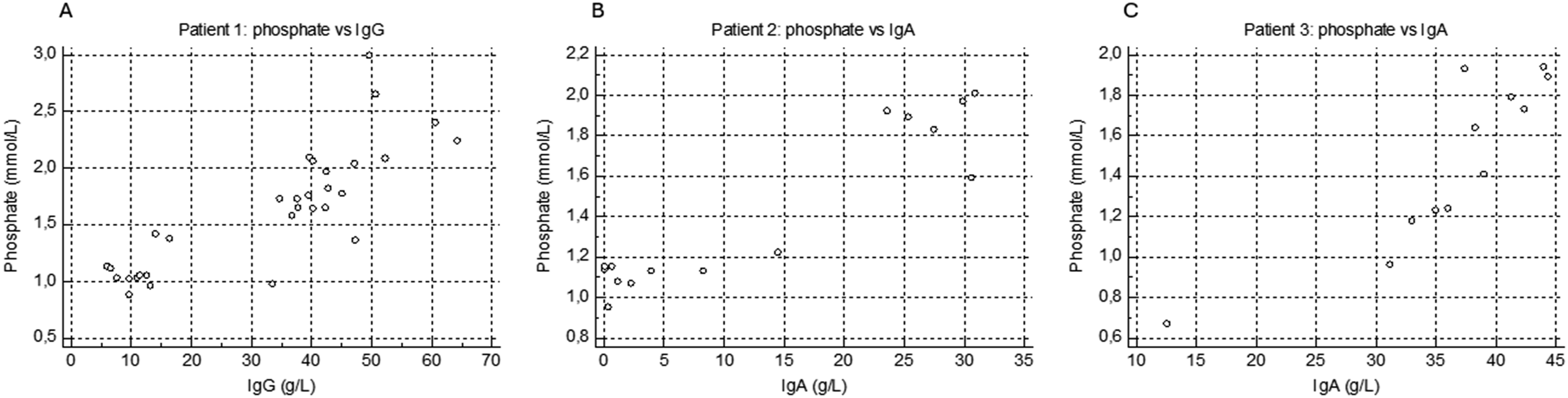
Phosphate (mmol/L) and IgG (g/L) Levels Measured in Serum From Patient 1 (IgG Kappa Multiple Myeloma and Unexplained Hyperphosphatemia) Diluted With Physiological Saline. Phosphate was Measured With the Phosphomolybdate UV Assay on Atellica Solution (Siemens-Healthineers) With Measurement Range 0.1–6.46 mmol/L. IgG was Measured Using Immunoturbidimetry on Atellica Solution (Siemens-Healthineers) With Measurement Range 1.40–34.00 g/L.

In a second serum sample from patient 1, the measured phosphate concentration was 2.45 mmol/L. Protein removal by precipitation with sulfosalicylic acid efficiently reduced the IgG level below the detection limit (<0.70 g/L) but further increased the phosphate level to 3.90 mmol/L. This indicates efficient protein removal but suggests a different interference in the phosphate assay. Similarly, protein precipitation with zinc sulphate reduced IgG below the limit of detection but interfered with the assay as it resulted in an unmeasurably low phosphate level (<0.10 mmol/L). Successful removal of the paraprotein and a phosphate level that matched the patient’s other biochemistry parameters and clinical condition were obtained by ultrafiltration: IgG levels were below the detection limit, and phosphate decreased from 2.45 mmol/L to 1.07 mmol/L.
Discussion and conclusion
Hyperphosphatemia in multiple myeloma patients occurs with advanced renal failure. However, pseudohyperphosphatemia can be present in these patients due to interference of the paraprotein in the phosphomolybdate UV assay. Clinical chemists should be aware of this phenomenon, especially when a patient presents with hyperphosphatemia in the presence of a paraprotein and a normal kidney function, calcium, PTH, and vitamin D level.
As previously observed, phosphate levels correlated with IgG and total protein concentration in a multiple myeloma patient with unexplained hyperphosphatemia.3,5 Dilution of the patient’s serum efficiently reduced the IgG paraprotein and resulted in a measurable phosphate level. However, dilution is not optimal for correct phosphate measurement as it increases the measurement error.
Incubation with precipitation agents completely removed the interfering IgG paraprotein but did not result in a correct phosphate measurement. However, several reports describe normalization of the phosphate level in serum samples from multiple myeloma patients with pseudohyperphosphatemia after incubation with sulfosalicylic acid or other agents such as polyethylene glycol or trichloroacetic acid.3,5–8 The undetectable phosphate concentration after incubation with zinc sulphate might be due to precipitation of phosphate, or interference of methanol during the assay reaction, and reagent or device-specific interference cannot be excluded. Therefore, protein precipitation with zinc sulphate is not applicable for the deproteinization of samples for phosphate measurement. Finally, efficient protein removal and a more credible phosphate level were obtained by ultrafiltration of the serum. Similar principles are observed in dry chemistry methods, in which the first layer of the reagent slide mimics ultrafiltration as it retains high-molecular weight substances such as serum proteins.8,9
In conclusion, paraproteins can interfere with reaction components in the phosphomolybdate UV assay, causing pseudohyperphosphatemia. As it is impossible to predict whether the paraprotein of a specific patient will interfere in a specific assay, unexplained hyperphosphatemia should always lead to further investigations, especially when not matching other biochemical parameters. If the presence of this phenomenon is confirmed by normalization of the phosphate level after serum dilution, a reliable phosphate concentration can be obtained after ultrafiltration of the sample. It is important to be aware of the existence of paraprotein-related pseudohyperphosphatemia to avoid unnecessary clinical investigations and therapy.
Supplemental Material
Supplemental Material - Pseudohyperphosphatemia in multiple myeloma: Removal of interfering paraproteins
Supplemental Material for Pseudohyperphosphatemia in multiple myeloma: Removal of interfering paraproteins by Michelle De bruyn, Matthias Cuykx in Annals of Clinical Biochemistry.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethical approval
Not applicable.
Guarantor
Matthias Cuykx.
Contributorship
MDB researched literature, performed experiments and data interpretation. MDB and MC were involved the content development. MDB wrote the first draft. All authors reviewed and edited the manuscript and approved the final version of the manuscript.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
