Abstract
Background
The present study was aimed at determining the effect of the tube type used for primary sample collection and the manner of transport prior to assessment (either manual or by pneumatic tube) on the degree of pseudohyperkalaemia in leukaemic patients.
Methods
Blood from six leukaemic patients was collected into seven primary sample tubes (Monovette®, Sarstedt): sample A, heparinized blood gas syringe (potassium reference value); sample B, plasma Li-heparin without separator gel; sample C, plasma Li-heparin with separator gel; and sample D, serum with separator gel. The primary sample tubes designated B, C and D were transported to the laboratory manually. Duplicates of the same sample tubes, BPT, CPT and DPT, were sent to the laboratory by pneumatic tube.
Results
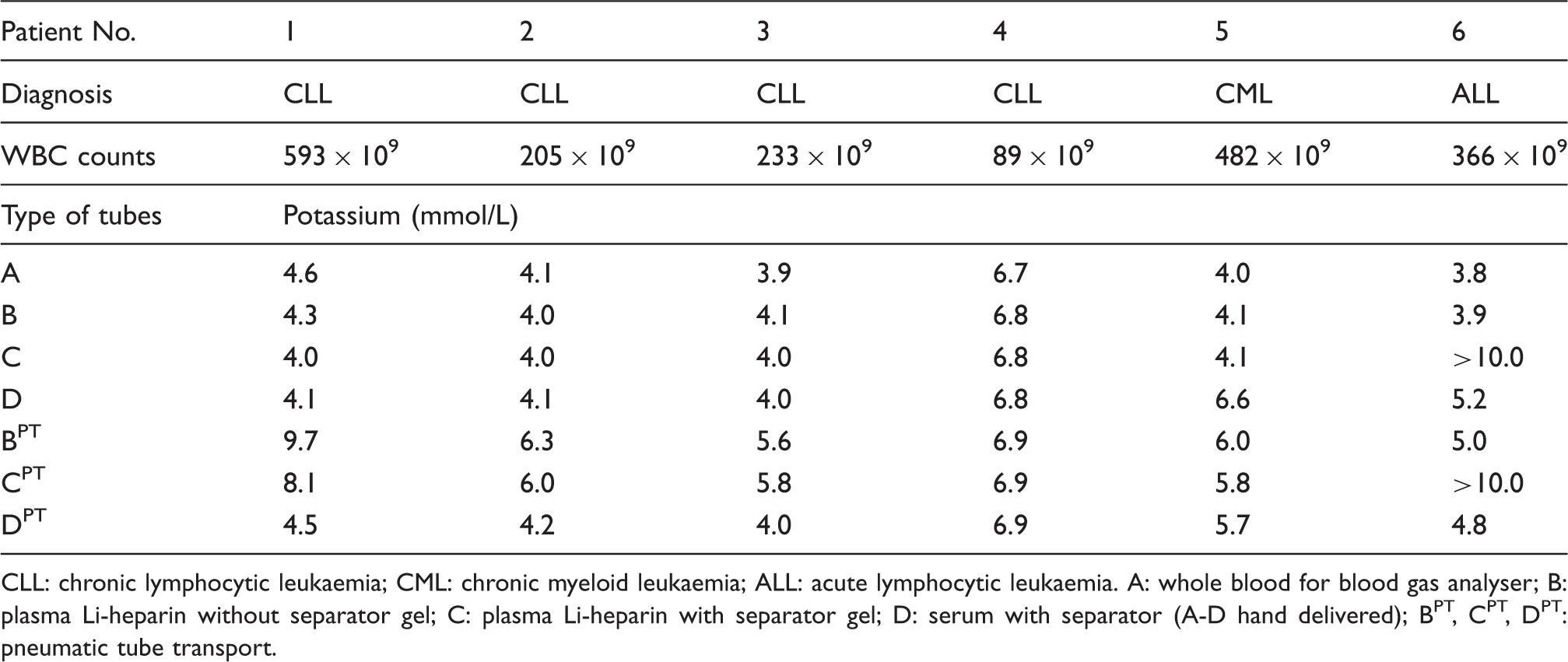
In patients with chronic lymphocytic leukaemia (CLL), there was no increase in the concentration of potassium in samples B, C and D when compared to the reference value. Transport of the samples by pneumatic tube led to a pronounced increase in potassium concentration in samples BPT and CPT, whereas there was no increase in sample DPT when compared to the reference value. In the patient with chronic myeloid leukaemia (CML), an increase in potassium concentration occurred in sample D and in samples BPT, CPT and DPT. A similar finding was observed in the patient with acute lymphocytic leukaemia (ALL), furthermore with an extremely high concentration of potassium in samples C and CPT.
Conclusions
Manual transport of non-coagulable blood (plasma Li-heparin without separator gel) to the laboratory results in the least possible artificial increase in potassium concentration in the sample.
Introduction
In both clinical and laboratory practice, artificial elevation of potassium concentration or so-called pseudohyperkalaemia has been most frequently associated with a haemolytic sample of serum or plasma and/or with late centrifugal separation of plasma from blood cells, or of serum from a blood clot.
The other causes, such as hyperleukocytosis, thrombocytosis, potassium contamination (K2 EDTA) and abnormal erythrocyte morphology, though well known, are less rooted in public awareness.1,
In leukaemic patients, the level of pseudohyperkalaemia may also be affected by the type of leukaemia, the number of leukocytes, the type of primary sample tube used (plasma, serum and separator gel) and the manner of transport (manual and pneumatic tube).3–5
In the present study, we considered the effect of the above-mentioned factors on the degree of artificial elevation of potassium concentration in patients with different types of leukaemia.
Patients, material and methods
Our mini-cohort consisted of four patients with chronic lymphocytic leukaemia (CLL); one patient with acute lymphocytic leukaemia (ALL) and one patient with chronic myeloid leukaemia (CML). The study protocol was approved by the local Ethics Committee, the University Hospital Brno. An informed consent was signed by all subjects. From each patient, venous blood was taken from the cubital vein and collected into seven 2mL primary sample tubes (Monovette®, Sarstedt, Germany).
Potassium concentration in whole blood was determined using a heparinized blood gas syringe (A). Evaluation of potassium concentration was carried out immediately after the blood was taken at the patient’s bedside using a GEM Premier 4000 analyser (Instrumentation Laboratory) and this concentration served as a reference value. Comparison of potassium quantification using a GEM Premier 4000 analyser and Cobas 8000 ISE module showed very close correlation (P/B regression Y = 1.036−0.056; r = 0.9890; n = 20).
Duplicate blood samples were taken to determine plasma potassium concentration, using plasma Li-heparin test tubes without separator gel (B) and plasma Li-heparin test tubes with separator gel (C). For the determination of serum potassium concentration, duplicate blood samples were collected in serum test tubes with separator gel (D).
The primary sample tubes designated B, C and D were transported to the laboratory by hand immediately after blood collection. The duplicates of the same sample tubes, BPT, CPT and DPT, were sent to the laboratory by pneumatic tube post(PT).
In the laboratory, the primary sample tubes were immediately centrifuged for 5 min at 2800 g in 17℃. Potassium was determined simultaneously in all samples using a Cobas 701 Roche analyser (Switzerland). The intra-assay variability for potassium determination at a concentration of 6.2 mmol/L was 1.76% (n = 20).
Results
In the patients with CLL, the concentration of potassium in samples B, C and D delivered manually to the laboratory showed no increase in comparison to the reference value (sample A). Unfortunately, we are unable to satisfactorily determine reason(s) for higher potassium concentration in sample A in comparison with samples B, C and D in patient No. 1 (CLL).
Transport of the samples by pneumatic tube led to a pronounced increase in potassium concentration in samples BPT and CPT, whereas there was no increase in sample DPT when compared to the reference value.
In patient No. 4, almost the same concentration of potassium was found in all the blood samples taken, irrespective of the type of primary sample tube or manner of transport. Thus, here it was a case of true hyperkalaemia.
In the patient with CML, an increase in potassium concentration occurred in sample D and in all samples (BPT, CPT and DPT) delivered to the laboratory by pneumatic post. A similar finding was observed in the patient with ALL, furthermore with an extremely raised concentration of potassium in samples C and CPT.
Discussion and conclusions
Generally, the cause of pseudohyperkalaemia in malignant leukocytosis has been thought to be due to a leakage of potassium from the enormously multiplied number of leukaemic cells after blood sampling. It may well be assumed that the different forms of leukaemia, with different types of leukocytes and with varying degrees of maturity, may be accompanied by pseudohyperkalaemia at various levels of severity.
We agree with Kapoor et al. that the most common cause of pseudohyperkalaemia in laboratory practice is the delay in time between blood collection and centrifugation. These cases may be easily recognized if the laboratory conforms to the ISO 15189 standard which requires times of blood collection and laboratory sample registration to be noted. We did not find any association in samples from our patients with CLL between artificially increased serum potassium concentration and precipitation and centrifugation. On the contrary, blood from the CLL patients formed precipitation and a coagulum with better sample properties following the less sensitive transport via pneumatic-tube mail.
Hypothetically, we may assume that in the patient with ALL pronounced damage to the immature leukaemic cells occurred during centrifugation in the presence of the separator gel (sample C, CPT), while no such damage occurred in the cases with a compact coagulum (sample D, DPT). Hypothetical higher coagulum ‘resistance’ for centrifugation and transportation via pneumatic-tube mail in patients with CCL did not manifest in blood samples of our patient with CML and ALL.
Our data have shown that the degree of pseudohyperkalaemia may be significantly and variably affected by different forms of leukaemia, the character of the analytical sample (serum/plasma), the type of primary sample tube used and the manner of transport.
From our results, it appears that taking non-coagulable blood (plasma Li-heparin without the separator gel) by manual delivery to the laboratory is the method which is least likely to result in an artificial increase in potassium concentration (pseudohyperkalaemia).
In cases of doubt, determination of potassium in whole blood (blood gas analyser) may be used as a decisive strategy. Because results of this analysis may be artificially affected as well, e.g. by haemolysis, general rule to interpret each laboratory result in context with other findings and with patient’s clinical status applies, not unlike in any other case.
Footnotes
Acknowledgements
The authors wish to thank nurses in Clinic of Internal Medicine – Hematology and Oncology for their assistance.
Declaration of conflicting interests
None declared.
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
Ethical approval
Ethical Committee of University Hospital Brno.
Guarantor
MD.
Contributorship
Potassium concentration in the serum and plasma of leukaemic patients from different primary sample tubes (Sarstedt) delivered to the laboratory manually and by pneumatic tube.
| Patient No. | 1 | 2 | 3 | 4 | 5 | 6 |
|---|---|---|---|---|---|---|
| Diagnosis | CLL | CLL | CLL | CLL | CML | ALL |
| WBC counts | 593 × 109 | 205 × 109 | 233 × 109 | 89 × 109 | 482 × 109 | 366 × 109 |
| Type of tubes | Potassium (mmol/L) | |||||
| A | 4.6 | 4.1 | 3.9 | 6.7 | 4.0 | 3.8 |
| B | 4.3 | 4.0 | 4.1 | 6.8 | 4.1 | 3.9 |
| C | 4.0 | 4.0 | 4.0 | 6.8 | 4.1 | >10.0 |
| D | 4.1 | 4.1 | 4.0 | 6.8 | 6.6 | 5.2 |
| BPT | 9.7 | 6.3 | 5.6 | 6.9 | 6.0 | 5.0 |
| CPT | 8.1 | 6.0 | 5.8 | 6.9 | 5.8 | >10.0 |
| DPT | 4.5 | 4.2 | 4.0 | 6.9 | 5.7 | 4.8 |
CLL: chronic lymphocytic leukaemia; CML: chronic myeloid leukaemia; ALL: acute lymphocytic leukaemia. A: whole blood for blood gas analyser; B: plasma Li-heparin without separator gel; C: plasma Li-heparin with separator gel; D: serum with separator (A-D hand delivered); BPT, CPT, DPT: pneumatic tube transport.
