Abstract
Background
The detection of deficiencies in B12 and folate children is important. However, despite the availability of various markers to assess B12 and folate metabolism, there are limited studies describing the reference intervals (RIs) and changes during growth and development for these markers in healthy children.
Methods
Using samples collected from 378 children aged 30 days–< 18 years, we derived continuous RIs for holotranscobalamin, homocysteine and red cell folate.
Results
The lower RI for holotranscobalamin was lowest at birth, rising during early childhood and then declining following ages 4–6 years whereas red cell folate was highest early in life and then declined steadily towards adulthood. Total homocysteine, reflective of both B12 and folate status was elevated early in life, reaching a nadir at age 2 and then increasing towards adulthood.
Conclusions
Continuous central 95th percentile RI for holotranscobalamin, homocysteine and red cell folate for children ages 30 days to <18 years were established. Each marker shows dynamic changes throughout childhood and adolescence which will assist clinicians in more appropriately assessing B12 and folate status in this population.
Keywords
Introduction
Vitamin B12 (cobalamin) and folate are water soluble vitamins acting as cofactors in the re-methylation cycle and in the conversion of homocysteine to methionine by methionine synthase. 1 They are, therefore, essential in the de novo synthesis of thymidine for DNA synthesis and repair. 1 Both vitamin B12 and folate deficiency can present similarly, with haematologic manifestations such as megaloblastic anaemia and, neurological manifestations, such as peripheral neuropathy due to impaired DNA synthesis. 1 During childhood, it is particularly important that vitamin B12 and folate deficiency can be confidently detected. 2
There are several biochemical markers of vitamin B12 and folate status. As a substrate for methionine synthase, increased total homocysteine in the blood can be an indicator of vitamin B12 and/or folate deficiency. Additionally, vitamin B12 as adenosyl-cobalamin, is a cofactor for the conversion of methyl-malonyl-Coenzyme A to succinyl-Coenzyme A and increases in serum or urine methylmalonic acid can indicate vitamin B12 deficiency. 1 Vitamin B12 status can also be assessed through the measurement of circulating vitamin B12 bound to its transport proteins haptocorrin and transcobalamin (Total B12) or to transcobalamin alone (holotranscobalamin (HoloTC)). HoloTC is thought to be a better marker of vitamin B12 status, as it represents the fraction available for cellular uptake. Folate itself can be measured in the serum or within the red cell, with serum responding more rapidly to dietary intake and red cells obtaining their folate during erythropoiesis and therefore representing folate stores over a longer period. 3 Despite the existence of these various markers to assess vitamin B12 and folate metabolism, there are limited studies describing their reference intervals (RIs) in healthy children and how they change during growth and development.
The paediatric population is known to undergo progressive, age-related developments that result in physiological changes of many biomarkers. Thus, paediatric RIs are often organized through the partitioning of results into varying age groups and often different categories for sex.4–6 For some analytes, however, there may be a large difference between the RI of one partitioned age group and the next, and misclassification can occur when a child has reached an age ‘cut-off’. 7 Continuous RIs have been suggested as a solution to this issue. Continuous RIs establish smooth curves for reference limits when compared against partitioned RIs and may more accurately reflect the dynamic relationship between target biomarkers and age. Recently, continuous RIs have been successfully utilized for several age-dynamic biomarkers, such as alkaline phosphatase, alanine aminotransferase and creatinine,8,9 illustrating higher accuracy in results. Additionally, Hoq et al. compared 30 biochemistry analytes in the same reference population, demonstrating the feasibility and superiority of continuous RIs. 10 More recently, Wilson et al established continuous reference intervals using an LMS based approach in a Canadian cohort of healthy children and adolescents for a number of laboratory parameters. 11
The Harmonising Age Pathology Parameters in Kids (HAPPI-Kids) study is a prospective cross-sectional study which aims to address current gaps in paediatric RIs by collecting paediatric blood samples from healthy children. 12 Using samples from the HAPPI-Kids study we derived continuous reference limits for a number of markers of B12 and folate status, namely, holotranscobalamin, homocysteine and red cell folate.
Methods
Study design
The study protocol was approved by The Royal Children’s Hospital (RCH), Melbourne, Australia, Ethics in Human Research Committee (HREC) (HREC 34183) and conducted in accordance with the declaration of Helsinki. The study protocol has been previously published. 12
Study subjects and samples
The majority of participants (91%) were born in Australia with the birth country of the parents of participants including Australia (56%), Southeast Asia (8%), South Asia (6%), Europe (4%), Middle East (2%) and Africa (2%), similar to the population described in Hoq et al. and matching the distribution of the general Australian population. 10 Australia has mandatory folate supplementation and millers are required to add folic acid to wheat flour for bread-making purposes.
Details of pre-analytic procedures have been previously published. 12 Briefly, venous blood samples from healthy children ages 30 days to < 18 years were obtained from children prior to undergoing minor elective day surgery. Samples for holotranscobalamin were collected in S-Monovette serum gel tubes (Sarstedt). Samples for red cell folate were collected in S-Monovette EDTA tubes (Sarstedt). Samples for homocysteine were collected in S-Monovette lithium heparin gel tubes (Sarstedt) on an ice pillow. Serum and plasma samples were separated by centrifugation for 5 min at 5000 r/min (4472 G) at 6°C and 400 μL aliquots were stored at -80°C until analysis.
Sample analysis
Holotranscobalamin and total homocysteine were measured in serum samples and red cell folate was measured in lysed whole blood samples. Total homocysteine and holotranscobalamin were analysed on an Abbott Architect i1000SR analyser (Abbott) and red cell folate on a Roche Cobas e601 analyser (Roche Diagnostics) in accordance with the manufacturer’s instructions.
All analyses were performed at RCH Laboratory Services which was accredited to ISO15189:2012 at the time of analysis. Regular quality control was performed for each assay and assays were in control when samples were tested. Long term coefficients of variation (CV) for each assay were as follows: holotranscobalamin 4.6% at 20 pmol/L, red cell folate, 5.9% and 4.5% at 433 nmol/L and 1038 nmol/L, respectively, and homocysteine, 6.5% and 6% at 6.4 μmol/L and 11.5 μmol/L, respectively.
Statistical analysis
All statistical analysis was performed using Stata 15.1 (StataCorp LLC). Continuous RIs were established as described previously.10,12 In summary, visual inspection of scatter plots of analyte concentration against age were used to identify extreme outliers. Best-fitting fractional polynomial (FP) regression models of age against each analyte concentration were derived with a maximum FP degree of 2 (i.e. x2, x1/2, x, ln(x), x−1/2 and x−1 x−2). Further outlier exclusion was undertaken if the absolute residual >3*IQR and the FP age functions re-run. The power functions of best-fitting model were then used in quantile regression models to predict continuous 2.5% and 97.5% reference limits using bootstrapping methods (20 replications). The process was also repeated for the same data partitioned for gender. Where the 95% confidence intervals (CI) for each gender specific reference limit overlapped, the decision was made to opt for non-gender specific reference intervals. For holotranscobalamin, samples with results above the analytical measurement range (>128 pmol/L) were included as 128 pmol/L for the statistical analysis. Due to this the upper 97.5% reference limit was not calculated.
To facilitate use of the reference limits in most laboratory information systems, age partitioned reference limits for each year were calculated by substitution of the midpoint of each year (0.5, 1.5, etc.) into each reference limit equation.
Results
Sample numbers and patient characteristics.
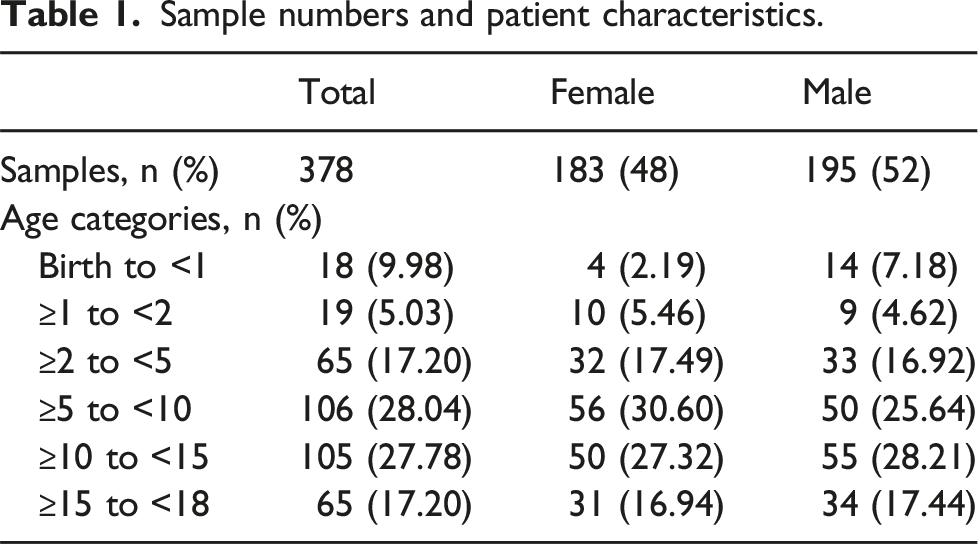
Continuous reference intervals
Holotranscobalamin
Parametric equations for upper and lower reference limits for each analyte.
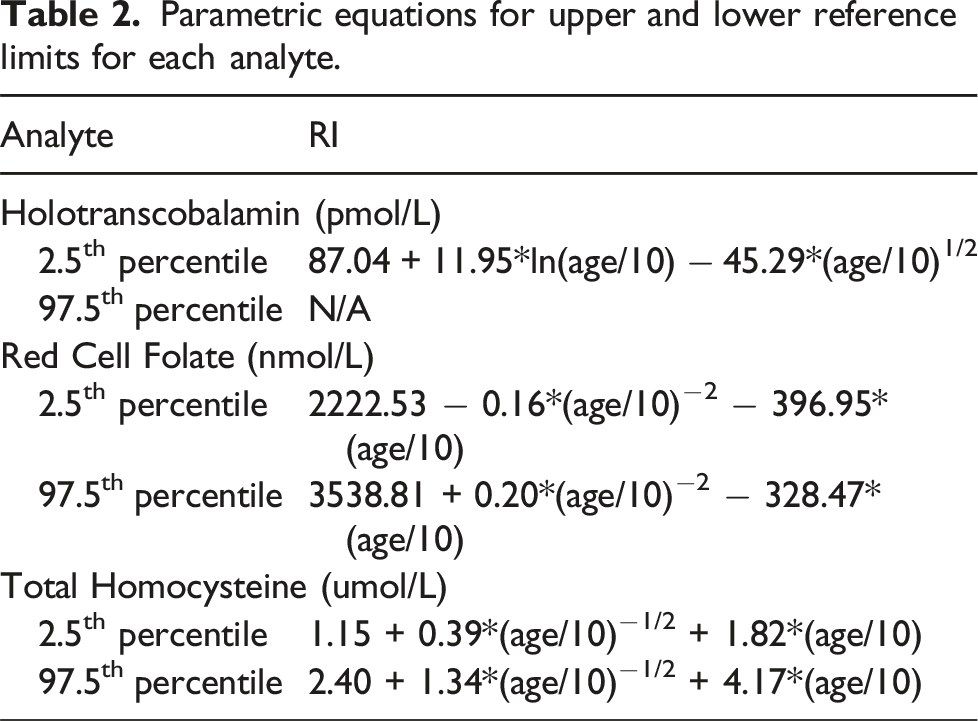
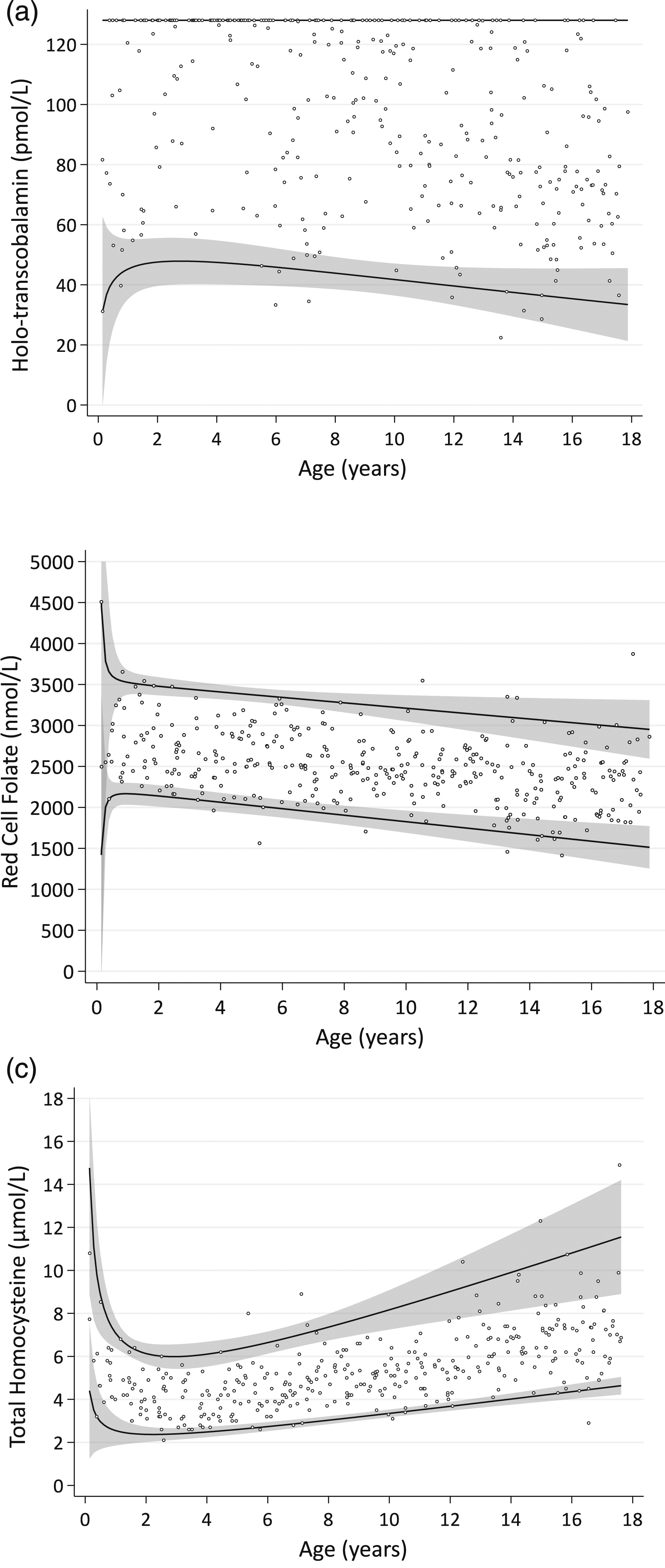
Continuous RIs obtained from best-fitting fractional polynomial models for holo-transcobalamin (n = 370) (Abbott Architect i1000SR) (1A), red cell folate (n = 372) (Roche Cobas e601) (1B) and total homocysteine (n = 365) (Abbott Architect i1000SR) (1C). Solid lines represent the 2.5th percentile and 97.5th percentile limits, respectively, and the grey areas represent the 95% confidence intervals around those limits.
Red cell folate
For red cell folate, reference intervals were derived on a sample of 372 individuals. No outliers were identified or excluded, and gender specific reference limits were not identified. The continuous 2.5th and 97.5th percentile reference limits are plotted in Figure 1(b).
Homocysteine
For total homocysteine, reference intervals were derived on a sample of 365 individuals following exclusion of 2 mathematical outliers. Again, gender specific reference limits were not identified. The continuous 2.5th and 97.5th percentile reference limits are plotted in Figure 1(c).
Age partitioned reference limits
Age partitioned reference limits for each year of age appear in supplemental table 1.
Discussion
Using paediatric blood samples from healthy infants and children, we derived continuous 2.5th and 97.5th percentile reference limits for holotranscobalamin and total homocysteine for the Abbott Architect platform and red cell folate on the Roche Cobas platform. Figure 1(a) shows that the lower reference limit for holotranscobalamin was lowest at birth, rises during early childhood and then declines following ages 4–6 years. This pattern was also observed by Heiner-Fokkema et al., in a smaller cohort of 170 Dutch children and was correlated with total B12 concentrations. 13 This pattern was thought to be related to lower cobalamin intake with exclusive breastfeeding earlier in life with subsequent increases with the introduction of solid food, including meat. 13 Data on diet was not collected for the infants in this study, so we cannot confirm if this pattern in our cohort may be related to lower cobalamin intake with exclusive breastfeeding.
Red cell folate was highest early in life and then declined steadily towards adulthood. Few studies have assessed red cell folate in healthy children. Studies of serum folate by Bailey et al., 6 and Bohn et al., 14 showed lower serum folate concentrations in younger children, a peak at ages 6–7, followed by a decline into adulthood. The contrast in our red cell folate results with these serum folate findings may be due to serum folate reflecting more recent dietary intake and red cell folate reflecting intake over the previous 2–3 months. The CALIPER cohort used by Bailey et al. and Bohn et al., was from a Canadian population with mandatory folate supplementation, similar to the Australian population tested here, so it is unlikely that the lower levels in the younger children were due to subclinical folate deficiency.6,14
Total homocysteine, being a substrate for methionine synthase, is a marker of both folate and cobalamin sufficiency within tissues. Figure 1(c) shows that total homocysteine is elevated early in life, reaches a nadir at age 2 and then increases towards adulthood. These changes were also observed by Monsen et al., in a population of 700 children aged 4 days to 19 years. 15 The authors found that in early life the plasma total homocysteine was inversely correlated with total B12 and may have been due to total B12 deficiency in this age group. 15 Due to the very low number of subjects in the younger age group (n = 7 for < 6 months) in our cohort, we could not confirm this finding. It should be noted that vitamin B6 also influences plasma homocysteine. Vitamin B6 was not measured in our cohort although our healthy cohort would not be considered at high risk for deficiency. 16
The importance of paediatric specific reference limits is illustrated by the example of homocysteine (Figure 1(c)). The manufacturer’s specific adult upper reference limit for the total homocysteine assay 15 μmol/L. This is not reached until ∼ age 18 with our continuous upper reference limit. If used for a paediatric population, there is a risk of missing elevated total homocysteine and underlying B12 or folate deficiency. Continuous limits, also provide a lower risk of misclassification at age ‘cut-offs’ and appear to better capture the rapid changes in childhood, for example, total homocysteine from birth to age 2 (Figure 1(c)) or the smooth decline in red cell folate from birth to age 18 (Figure 1(b)). Continuous reference limits, however, are difficult to incorporate into current laboratory information systems and adoption is hampered by the current capabilities of laboratory information systems and electronic medical records. One approach could be to have the electronic medical record extract the result data and plot it on a chart similar to those in Figure 1. This approach is used in our institution to plot height and weight for age on a growth chart within the medical record. To facilitate incorporation of these reference limits into commonly used laboratory information systems, yearly age-partitioned limits were also calculated by substitution of the midpoint of each year (0.5, 1.5, etc.) into each reference limit equation and are presented in supplemental table 1.
The strengths of this study lie in the carefully selected reference population of healthy neonates, infants, and older children representative of our local population. Sample numbers were evenly distributed across each year of life. However, data was not obtained for neonates and there were limited numbers of infants less than 6 months old which is reflected in the wider confidence limits for ages < 6 months. Another limitation of the study is the total sample size of 378 samples. There is significant variation in total sample sizes for estimating continuous reference intervals, 17 with recent studies using over 15,000 samples. 18 For the quantile regression-based approach used here, Hoq et al showed a median sample size of 1,029 (IQR 549, 1802). 17 The sample size in our study (378) lies at the lower end of this range. Royston et al suggested a sample size of 292 is adequate for time specific reference intervals with an even spread of samples within each age group. 19 A recent simulation study showed prediction accuracy of lower and upper reference limits by a fractional polynomial-quantile regression approach was 80% and 90%, respectively, for a sample size of 500, increasing to 91% and 95%, respectively, for a sample size of 1000. 20 While it is accurate that larger samples sizes improve confidence in continuous reference limits, using smaller sample sizes can still produce valid reference limits while balancing the logistical and ethical difficulties in collecting samples from large numbers of healthy children.
In summary, we have established continuous central 95th percentile reference intervals for holotranscobalamin, homocysteine and red cell folate for children ages 30 days to <18 years. Each marker shows dynamic changes throughout childhood and adolescence which will assist clinicians in more appropriately assessing B12 and folate status in this population.
Supplemental Material
Supplemental Material - Continuous reference intervals for holotranscobalamin, homocysteine and folate in a healthy paediatric cohort
Supplemental Material for Continuous reference intervals for holotranscobalamin, homocysteine and folate in a healthy paediatric cohort in Joel D Smith, Vasiliki Karlaftis, Stephen Hearps, Chantal Attard, Helen Savoia, Janine Campbell and Paul Monagle, on behalf of the HAPPI-Kids study team in Annals of Clinical Biochemistry
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was funded by The Royal Children’s Hospital Foundation. CA has received funding unrelated to this manuscript from the National Health and Medical research Council of Australia, Medical Research Future Fund (Australia), National Blood Authority (Australia), Bayer, Janssen and Takeda and is the Executive Director Australian Society for Medical Research.
Ethical approval
Not applicable.
Guarantor
JS.
Contributorship
All authors contributed equally to the manuscript.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
