Abstract
Background
Lipaemic interference on automated analysers has been widely studied using soy-based emulsion such as Intralipid. Due to the greater adoption of fish oil-based lipid emulsion for total parenteral nutrition in view of improved clinical outcomes, we seek to characterize the optical properties of SMOFlipid 20% (Fresenius Kabi, Bad Homburg, Germany), a fish oil-based emulsion, on the Roche Cobas 6000 chemistry analyser (Roche Diagnostic, Basel, Switzerland).
Method
Various amounts of SMOFlipid were spiked into pooled serums. We plotted Roche Cobas Serum Index Gen.2 Lipaemia Index (L-index) against the amount of SMOFlipid added. We then studied the interference thresholds for aspartate aminotransferase, alanine aminotransferase, albumin and renal panel analytes using SMOFlipid. We subjected five levels of spiked lipaemia to high-speed centrifugation and analysed the specimens pre- and post-centrifugation. To postulate whether fish oil-based lipid emulsion interferes with laboratory results in the clinical setting, we calculated concentrations of SMOFlipid post-lipid rescue therapy and steady-state concentration of a typical total parenteral nutrition regime using pharmacokinetic principles.
Results
SMOFlipid optical behaviour is similar to Intralipid using the Serum Index Gen.2 L-index, with 1 mg/dL of SMOFlipid representing 1 unit of L-index. Manufacturer-stated interference thresholds are accurate for alanine aminotransferase, aspartate aminotransferase, albumin, urea and creatinine. High-speed centrifugation at 60 min 21,100g facilitates the removal of fish oil-based SMOFlipid.
Conclusion
Based on the interference thresholds we verified and pharmacokinetics parameters provided by SMOFlipid manufacturer, total parenteral nutrition may not interfere with chemistry analytes given sufficient clearance, but lipid rescue therapy will interfere. Further studies assessing lipaemic interference on immunoassays are needed.
Background
Lipaemic interference on automated analysers using soybean oil-based lipid emulsion such as Intralipid has been widely studied for past two decades.1–3 Recently, there was a shift from soybean towards fish oil-based lipid emulsion for total parenteral nutrition (TPN) due to improved clinical outcomes. 4 , 5 There is currently a paucity of lipaemic interference studies on biochemical parameters using newer fish oil-based lipid emulsion. We therefore seek to characterize the optical properties of SMOFlipid 20% (Fresenius Kabi, Bad Homburg, Germany), a fish oil-based lipid emulsion with medium chain triglycerides (MCTs), on the Roche Cobas 6000 automated clinical chemistry analyser (Roche Diagnostic, Basel, Switzerland). We also conducted interference studies using SMOFlipid 20% and assessed the effectiveness of high-speed centrifugation in removing fish oil-based exogenous lipaemia. We then use pharmacokinetic principles to postulate whether TPN and lipid rescue therapy using fish oil-based lipid emulsion will interfere with laboratory results.
Methods
To determine optical properties of SMOFlipid, we spiked various amounts of SMOFlipid into pooled serums and ran the samples in duplicates. We plotted Roche Cobas Serum Index Gen.2 Lipaemia Index (L-index) against the amount of SMOFlipid added in mg/dL. L-index, a measure of lipid turbidity, was obtained via photospectrometry on the Roche Cobas 6000 by measuring absorbances at 660 nm (primary wavelength) and 700 nm (secondary wavelength). 6 A Passing Bablok regression was then performed using mcr package on R version 3.5.2 (R Core Team 2018, Vienna, Austria).
To study lipaemic interference from exogenous fish oil-based lipid emulsion therapy, pooled serums were tested for three common liver analytes (aspartate aminotransferase [AST] without pyridoxal-phosphate, alanine aminotransferase [ALT] without pyridoxal-phosphate, albumin using bromocresol green) and four common renal panel analytes (sodium [Na], potassium [K], enzymatic creatinine and urea) on Roche Cobas 6000. For each analyte, two pools of serums with different analyte concentrations were prepared. Varying amounts of SMOFlipid were added to pooled serums to obtain different levels of lipaemia. For each level of lipaemia, an equivalent amount of deionized water was added to base serum to act as control to account for dilutional effect of the added SMOFlipid for non-electrolytes. Electrolytes were compared to baseline. Baseline pooled serums, deionized water controls and lipaemic samples were analysed in triplicates. The differences were compared against The Royal College of Pathologists of Australasia (RCPA) allowable limits.
To assess the effectiveness of high-speed centrifugation for the removal of exogenous fish oil emulsion lipaemia, a further five different concentrations of spiked lipaemia were obtained and analysed for AST, ALT, albumin, Na, K, creatinine and urea in duplicates. They were transferred to microtubes and centrifuged at relative centrifugal force of 21,100g for 60 min using Sorvall Legend Micro 21 Microcentrifuge (Thermo Scientific, Waltham, United States). The lipid faction was then removed, and aqueous phase was analysed for AST, ALT, albumin, Na, K, creatinine and urea in duplicates. Results of the spun specimens were compared to controls and baseline.
To postulate whether fish oil-based lipid emulsion will interfere with laboratory results in the clinical setting, we calculated concentrations of SMOFlipid post-lipid rescue therapy and steady-state concentration of a typical TPN using pharmacokinetic principles.
Results
Optical properties of SMOFlipid on Roche Cobas L-index
We observe a linear relationship of SMOFlipid with the Roche L-index up to 2000 mg/dL (Figure 1). Slope is 0.98 (95% confidence interval 0.97–1.00). Intercept is 19.17 (95% confidence interval 13.28–29.78). The Passing Bablok regression line is close to the line of identity. This is consistent with the manufacturer’s claim that 1 L-index is equivalent to the turbidity of 1 mg/dL soy-based Intralipid. We have thus shown fish oil-based SMOFlipid behaves the same way optically as soy-based Intralipid using the Serum Index Gen.2 L-index on Roche Cobas 6000.
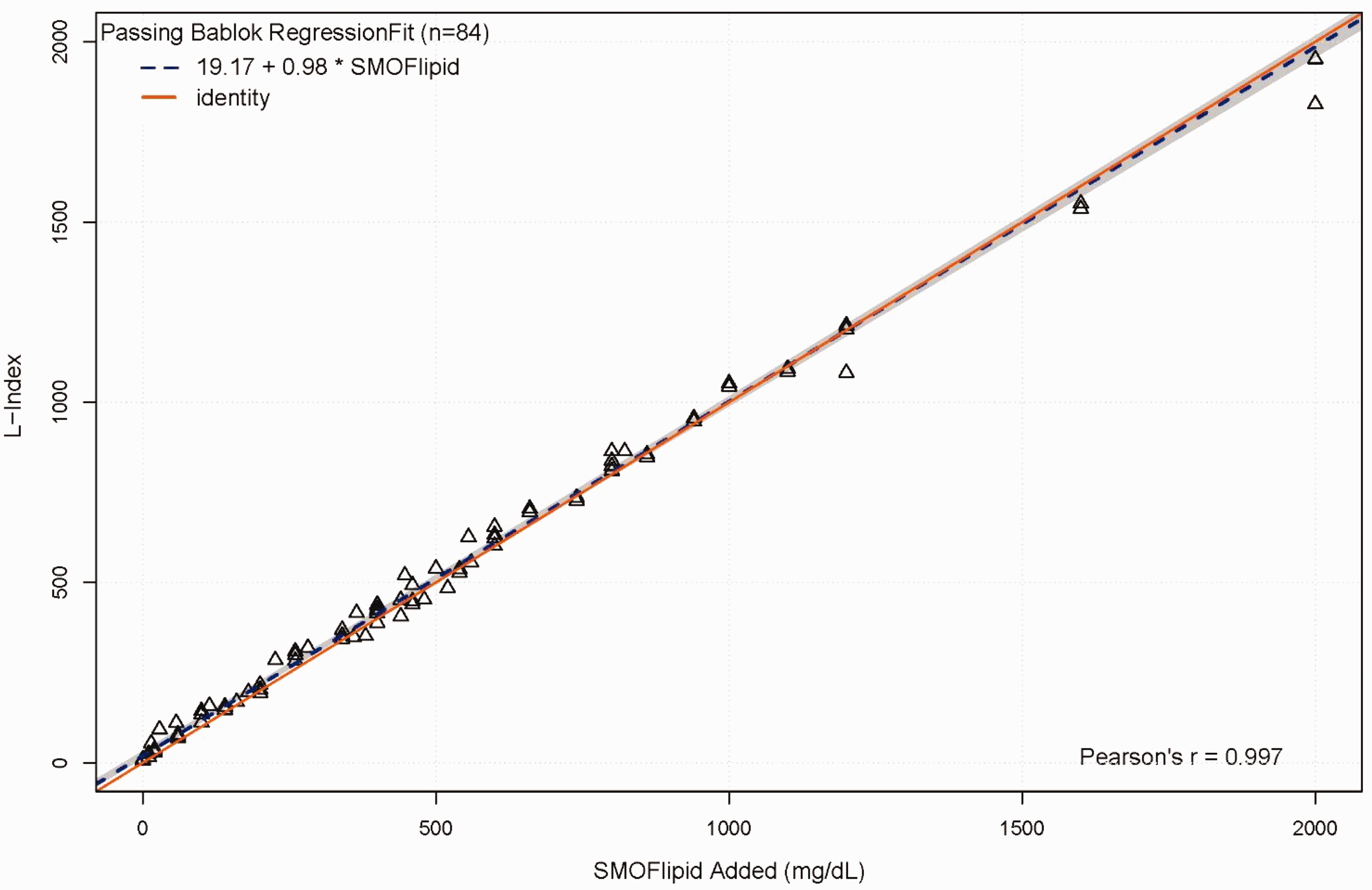
Passing Bablok regression line of L-index on SMOFlipid. Dashed blue line is the Passing Bablok regression line. Red solid line is the line of identity. 95% confidence interval is shown as the shaded region.
Lipaemic interference study
AST, ALT and albumin
Lipaemic interference on AST was tested using two serum pools with concentrations of 18.4 U/L and 142.9 U/L. For both pools, AST concentrations were not affected at low levels of lipaemia (L-index of up to 158 and 153) when compared to water spiked controls, meeting the manufacturer’s claims of 150 L-index. The 18.4 U/L pool was able to meet the RCPA criteria of ± 5 U/L at an L-index of 158. The 142.9 U/L pool was able to meet the RCPA criteria of 12% at an L-index of 153. However, the difference from water spiked controls increased as L-index increases. Negative AST values were seen for the 18.4 U/L pool at L-index of 520, 626 and 864 (Table 1).
Degree of interference for various levels of lipaemia on AST, ALT and albumin.
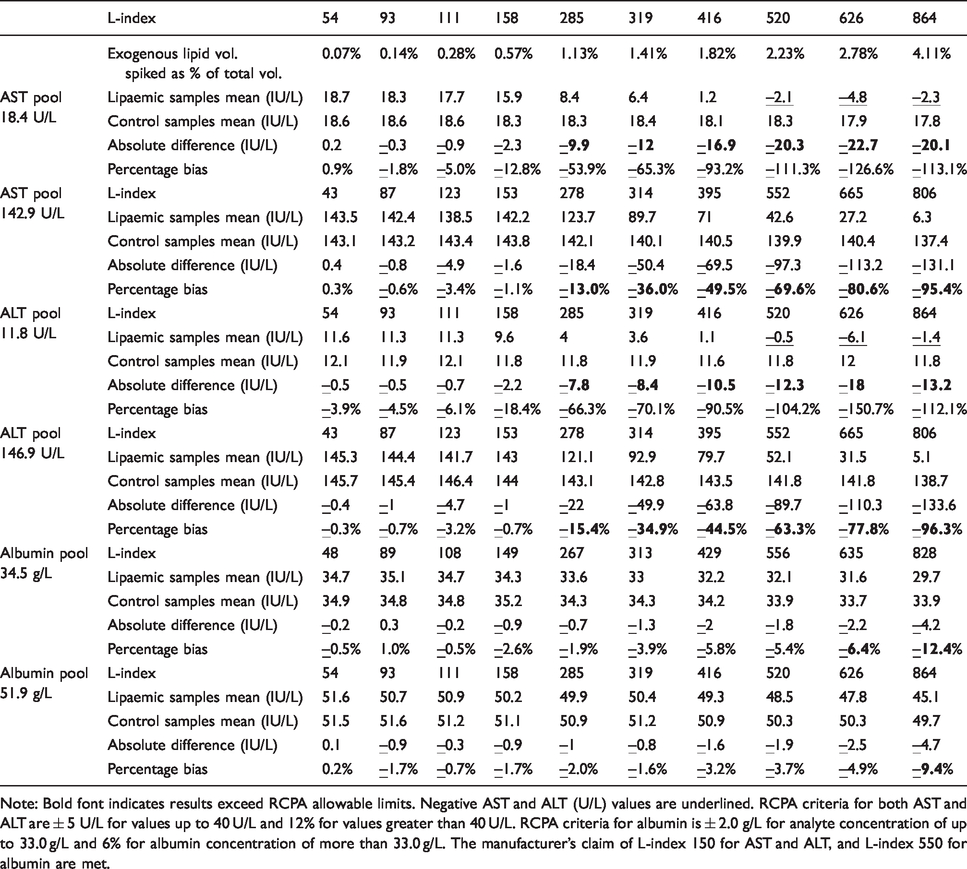
Note: Bold font indicates results exceed RCPA allowable limits. Negative AST and ALT (U/L) values are underlined. RCPA criteria for both AST and ALT are ± 5 U/L for values up to 40 U/L and 12% for values greater than 40 U/L. RCPA criteria for albumin is ± 2.0 g/L for analyte concentration of up to 33.0 g/L and 6% for albumin concentration of more than 33.0 g/L. The manufacturer’s claim of L-index 150 for AST and ALT, and L-index 550 for albumin are met.
ALT was tested using two serum pools with concentrations of 11.8 U/L and 146.9 U/L. Similar to AST, both ALT pools were not affected at low levels of lipaemia (L-index of up to 158 and 153) when compared to water spiked controls, meeting the manufacturer’s claims of 150 L-index. The 11.8 U/L pool was able to meet the RCPA criteria of ±5 U/L at an L-index of 158. The 146.9 U/L pool was able to meet the RCPA criteria of 12% at an L-index of 153. ALT becomes increasingly negative as L-index increases (Table 1).
Two albumin pools with concentrations of 34.5 g/L and 51.9 g/L were prepared. For the 34.5 g/L pool, the difference from water spiked controls varies from –5.8% to +1.0% when tested up to an L-index of 556. For the 51.9 g/L pool, the difference from water spiked controls varies from −4.9% to 0.2% up to an L-index of 626. Both pools satisfy the RCPA allowable limits of performance up of 6%, at the manufacturer’s claim of L-index of 550 for albumin. However, albumin is susceptible to interference at higher lipaemia levels. At an L-index of 828, the 34.5 g/L pool shows a negative bias of 12.4%, while the 51.9 g/L pool shows a negative bias of 9.4% at an L-index of 864 (Table 1).
Creatinine and urea
Lipaemic interference on enzymatic creatinine was tested using two serum pools with creatinine concentrations of 84 μmol/L and 158 μmol/L. For the pool with a baseline concentration of 84 μmol/L, creatinine is free from lipaemic interferences up to the tested L-index of 1214. For the pool with a baseline concentration of 158 μmol/L, creatinine is free from lipaemic interferences up to the tested L-index of 1193 (Table 2).
Interference for various levels of lipaemia for creatinine and urea.
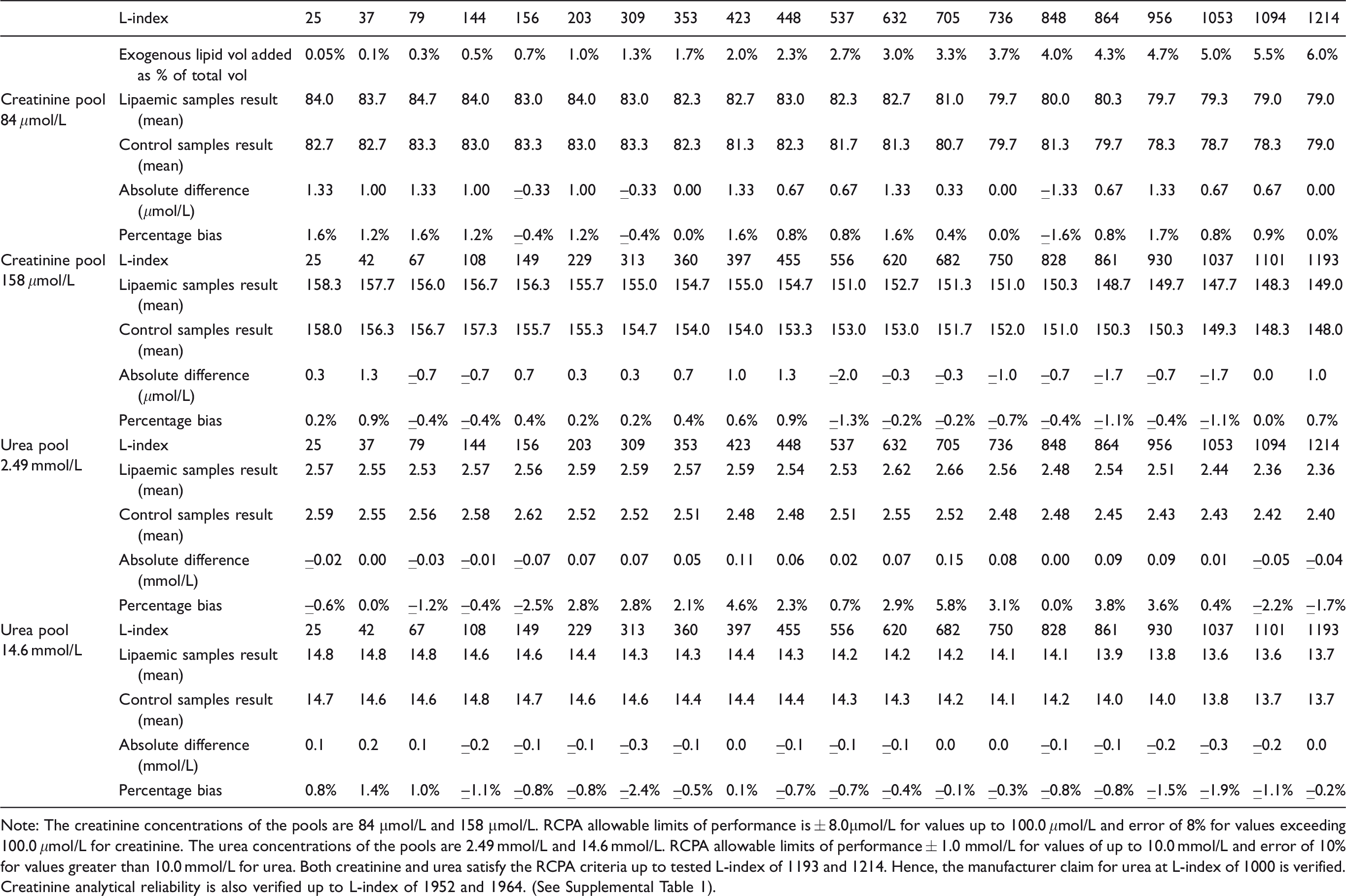
Note: The creatinine concentrations of the pools are 84 μmol/L and 158 μmol/L. RCPA allowable limits of performance is ± 8.0μmol/L for values up to 100.0 μmol/L and error of 8% for values exceeding 100.0 μmol/L for creatinine. The urea concentrations of the pools are 2.49 mmol/L and 14.6 mmol/L. RCPA allowable limits of performance ± 1.0 mmol/L for values of up to 10.0 mmol/L and error of 10% for values greater than 10.0 mmol/L for urea. Both creatinine and urea satisfy the RCPA criteria up to tested L-index of 1193 and 1214. Hence, the manufacturer claim for urea at L-index of 1000 is verified. Creatinine analytical reliability is also verified up to L-index of 1952 and 1964. (See Supplemental Table 1).
For creatinine, the manufacturer claims that the analytical reliability is 2000. We then tested up to L-index 1952 and 1964 for creatinine with another two sets of pooled serum and found no interference (Supplemental Table 1).
The baseline pooled serum values for urea were 2.49 mmol/L and 14.6 mmol/L. Both urea pools meet the RCPA criteria up to the tested L-index of 1214 and 1193, respectively. Thus, the manufacturer’s claims that the analytical reliability for urea is up to L-index of 1000 is satisfied (Table 2).
Sodium and potassium
The pooled serum Na concentrations were 126 mmol/L and 143 mmol/L. For the 126 mmol/L pool, Na absolute difference from baseline meets RCPA criteria of absolute difference of ±3 mmol/L up to L-index of 676. However, beyond L-index of 676, the deviation for the 126 mmol/L pool from baseline value exceeds 3 mmol/L, reaching –6.0 mmol/L at an L-index of 1198. For the 143 mmol/L pool, Na absolute difference from baseline meets RCPA absolute difference of up to L-index 848 (Table 3). Therefore, Na did not meet the manufacturers’ claim of 2000 at both concentrations.
Interference for various levels of lipaemia for sodium and potassium.
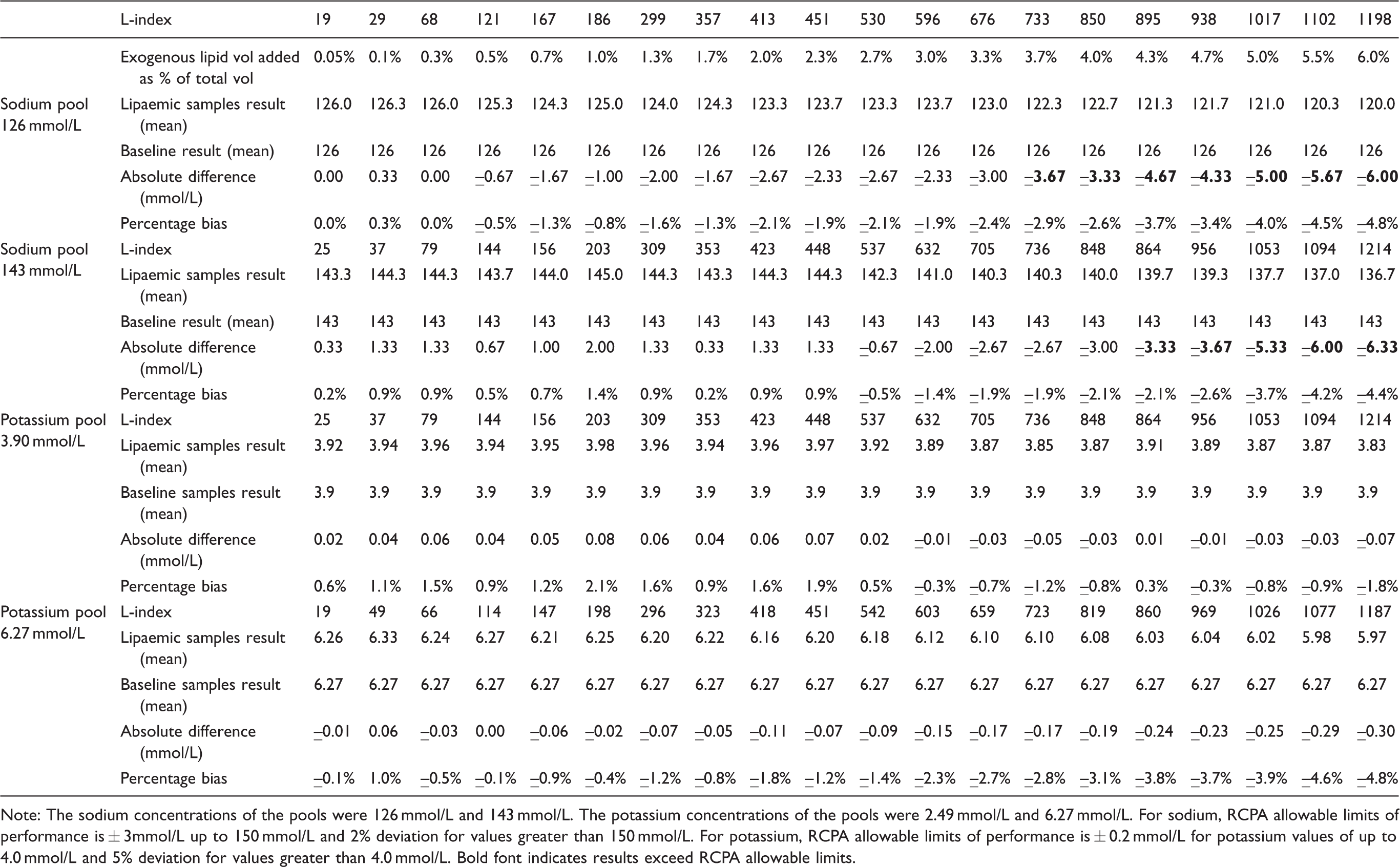
Note: The sodium concentrations of the pools were 126 mmol/L and 143 mmol/L. The potassium concentrations of the pools were 2.49 mmol/L and 6.27 mmol/L. For sodium, RCPA allowable limits of performance is ± 3mmol/L up to 150 mmol/L and 2% deviation for values greater than 150 mmol/L. For potassium, RCPA allowable limits of performance is ± 0.2 mmol/L for potassium values of up to 4.0 mmol/L and 5% deviation for values greater than 4.0 mmol/L. Bold font indicates results exceed RCPA allowable limits.
The pooled serum K concentrations were 3.90 mmol/L and 6.27 mmol/L. For the 3.90 mmol/L pool, K absolute difference from baseline meets RCPA criteria of absolute difference of ± 0.2 mmol/L up to L-index of 1214. For the 6.27 mmol/L pool, K percentage difference from baseline meets RCPA criteria of 5% deviation up to an L-index of 1187 (Table 3). Roche package insert indicated that the lipaemic interference threshold is L-index of 2000 for K. We then tested up to L-index 1952 and 1964 for K using another two sets of pooled serum at concentrations of 4.03 mmol/l and 6.23 mmol/L, respectively (Supplemental Table 1). We noted the difference from baseline exceeded the RCPA criteria of 5% deviation for both concentrations. Hence, K did not meet manufacturer’s claim of L-index of 2000.
Effects of high-speed centrifugation
We subjected five levels of lipaemia to high-speed centrifugation, The L-index mean values decreased from 426.5, 823.5, 1212.5, 1551.5 and 1952.0 to 19.5, 24.0, 29.5, 35.5 and 41.5, respectively (Figure 2). High-speed centrifugation improved analyte recovery of the five levels of lipaemia (Supplemental Table 2).
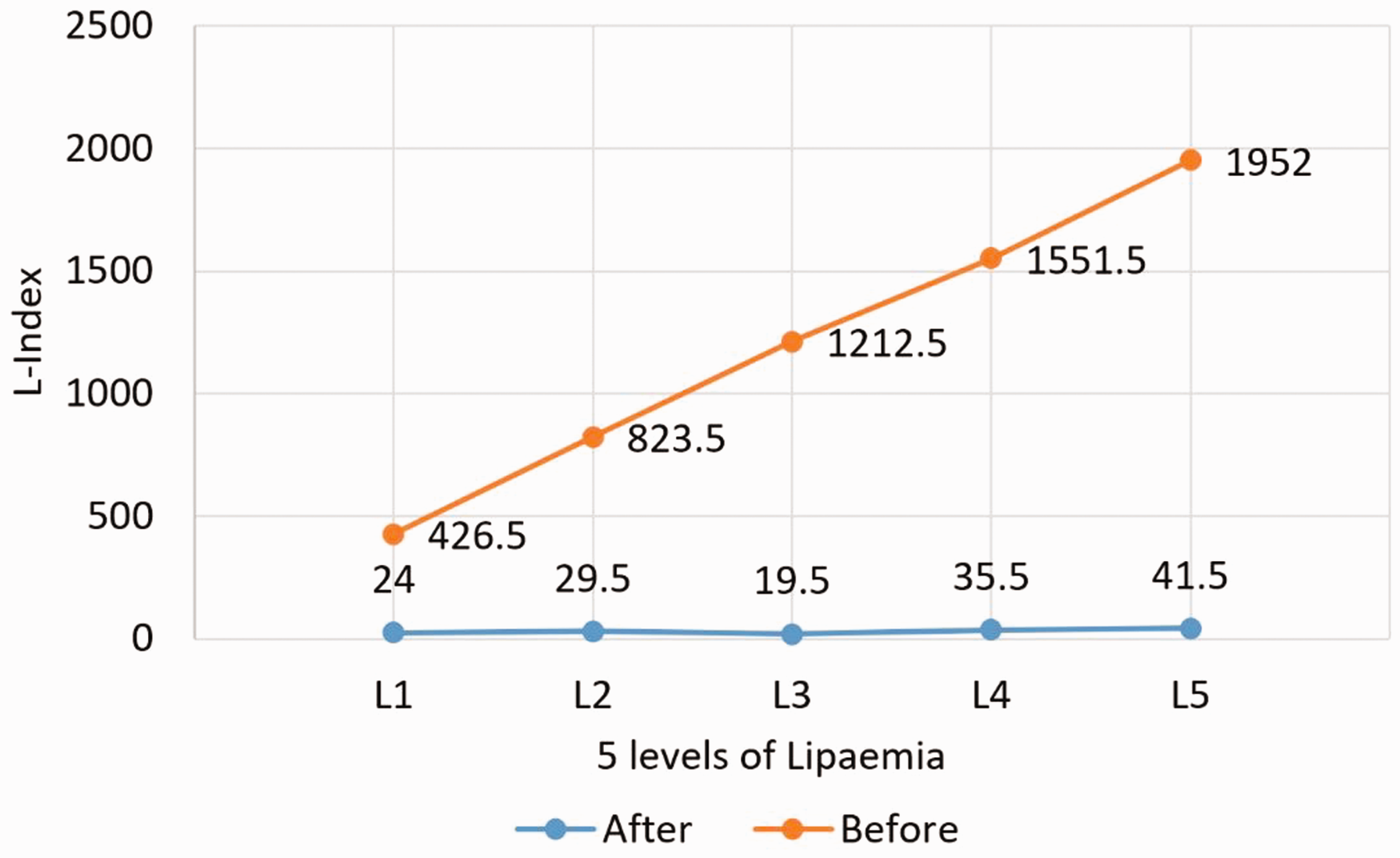
Removal of lipaemia after post-centrifugation for five different levels of spiked lipaemia.
Prior to centrifugation, ALT and AST in lipaemia samples showed huge variation between duplicate runs (Figure 3) and there were even negative values. Repeatability (in terms of percentage coefficient of variation, CV) was poor. Post-centrifugation, CVs improved for both ALT and AST (Supplemental Table 3).
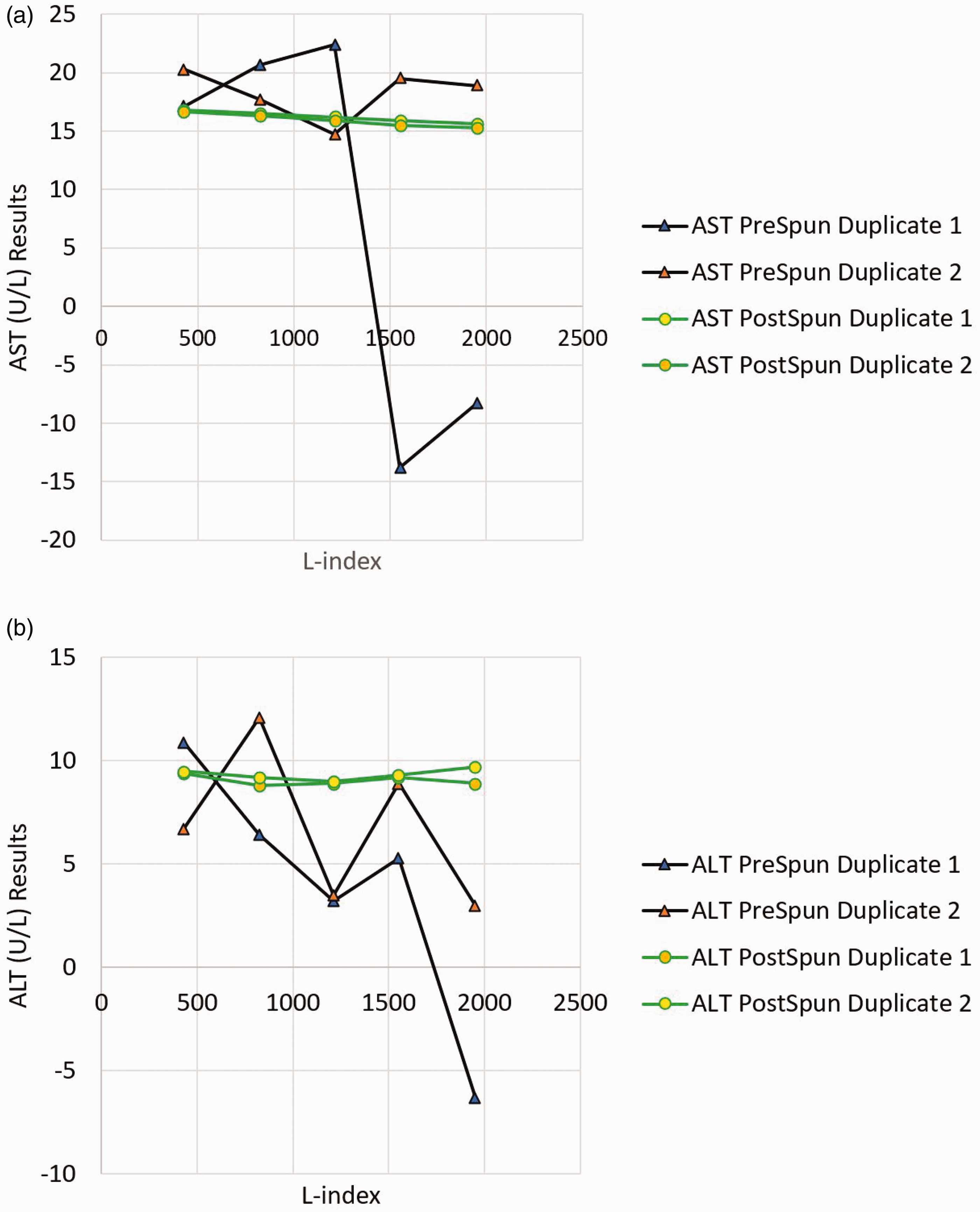
Effect of centrifugation on ALT and AST results in samples spiked with SMOFlipid. Note that the post-centrifugation, the repeatability improved (green lines are closer to each other). Baseline ALT and AST are 9.4 U/L and 17.3 U/L, respectively.
Predicting lipaemic interference of SMOFlipid in clinical setting
We used pharmacokinetic principles and lipaemia thresholds to determine if TPN and lipid rescue therapy will result in interference.
Lipid rescue therapy
For acute local anesthetic overdose with systemic toxicity, the Association of Anaesthetists of Great Britain and Ireland recommends administration of 1.5 mL/kg 20% lipid emulsion as a bolus over 1 min as initial therapy, 7 , 8 followed by infusion of 20% lipid emulsion at the rate of 15 mL/kg/h for 5 min before reassessment. We assume the average human plasma volume to be about 45 mL per kg body weight. 9 Hence, the initial bolus translates to a 3.3% volume of SMOFlipid over the total plasma volume, corresponding to 660 mg/dL of SMOFlipid or an L-index of 660. This exceeds the threshold for ALT, AST and close to the limit of albumin.
The next five-min infusion at 0.25 mL/kg/min will increase the volume of lipid emulsion administered to 6.1% of the plasma volume or 1220 mg/dL or an L-index of 1220. Samples taken from patients that underwent lipid rescue therapy are therefore likely to suffer from interference, and we thus recommend high-speed centrifugation before analysis on mainframe chemistry analysers.
Lipid emulsion in TPN
Lipid emulsion is also a significant component in TPN to provide adequate calories. The recommended daily dose for parenteral lipids in adults is 0.7–1.3 g triglycerides/kg body weight. 10 For a typical 25–30 kcal/kg/day TPN caloric requirement, 11 1 g of fat/kg/day (5 mL/kg/day of SMOFlipid 20%) will supply 9 kcal/kg/day and accounts for 30–36% of total calories. The hourly rate will be 0.208 mL/kg or 0.0416 g fat/kg if administered as a constant infusion over 24 h.
Based on the pharmacokinetic parameters provided in the SMOFlipid insert, 12 the median area under the curve (AUC) is 2598 mg×h/dL (range 1288–6164). A 70-kg subject, at the infusion rate stated in the insert of 125 mg/kg/h for 6 h, will receive a total dose of 70 × 125 × 6 = 52,500 mg of SMOFlipid. Median clearance will approximately be Dose/AUC = 52,500/2598 = 20.2 dL/h.
Given the infusion rate of 0.0416 g SMOFlipid fat/kg bodyweight/h in the TPN regime, the steady-state concentration would be infusion rate/clearance = 41.6 mg/kg/h × 70 kg
If the blood sample is taken from contralateral arm of the TPN infusion, interference is unlikely assuming median clearance. However, when there is significant decreased clearance in critically ill patients, the steady-state lipid concentration may increase beyond the AST and ALT threshold.
Discussion
The new generation of lipid emulsions containing fish oil are gaining wider prominence. In fact, some authors propose the use of SMOFlipid as the gold standard of parenteral nutrition for intensive care unit (ICU) patients. 13 Position papers by the American Society of Parenteral and Enteral Nutrition and European Society for Parenteral and Enteral Nutrition noted the role of fish oil-based emulsion and MCTs with reduced omega-6 for critically ill patients. 14 , 15 Mertes et al. showed SMOFlipid may be associated with better liver tolerance and a shorter length of hospitalization. 16 Due to the benefits of fish oil-based emulsion in ICU patients,17–19 our hospital compounding pharmacy has switched to SMOFlipid emulsion in 2014. While traditionally soybean lipid emulsion is used as a rescue therapy for calcium channel blockers, beta-blockers, tricyclic antidepressants overdose and local anaesthetic systemic toxicity, 20 , 21 fish oil-based lipid emulsion has been used as well. 22 , 23
Most recent lipid emulsion interference studies are still performed with the traditional soy-based Intralipid. 24 , 25 There is a paucity of data on the interference of fish oil-based exogenous lipaemia on in vitro diagnostic instruments, and hence it was timely for us to evaluate optical properties and verify interference thresholds using SMOFlipid.
For recovery of non-electrolytes, we compared results of samples supplemented with SMOFlipid to corresponding samples supplemented with an equal volume of deionized water control. This is because of dilutional effect. Given that the lipid emulsion does not contain the measured analyte, addition of lipid emulsion on the lipid supplemented samples will decrease the analyte concentration. Comparing lipaemic supplemented specimens with deionized water controls which experience a same magnitude of volume dilution will negate the dilutional effect and more accurately identify any interference resulting from turbidity. For electrolytes (Na and K), we have chosen to compare with the baseline as the lipid emulsion may contain Na and K in small concentrations. Comparison with samples spiked with deionsied water as control is incorrect, as this will falsely result in superior recovery, because the spiked deionized water does not contain Na and K (falsely lowering the control value), while lipid emulsion may contain both Na and K (falsely increasing Na and K value).
Our results show that the manufacturer’s claims for ALT, AST, albumin, creatinine, urea are accurate using SMOPFlipid. However, K and Na are susceptible to interference at levels of lipaemia below the manufacturer’s claims of L-index of 2000. Ji and Meng evaluated the interference of lipaemia on Roche Cobas 6000 assays using Intralipid and generally found good agreement with Roche package inserts. 26 They found that ALT is venerable to lipaemic interference at an L-index of 150, similar to our study. However, they found that Na did not suffer interference up to manufacturer claimed L-index. This is probably due to the fact Ji and Meng defined significant difference as ±10% while we have chosen to use the more stringent RCPA allowable limits. Allowable limits of performance is the acceptance criteria used for the RCPA external quality assurance programs. The criteria takes into account of imprecision or total error derived from biological variation. 27
For the triglycerides constituents, SMOFlipid comprises of 30% soybean oil, 30% MCTs, 25% olive oil, 15% fish oil. Intralipid comprises of 100% soybean oil for its triglycerides component. High-speed centrifugation has been previously reported to be effective in removing lipaemia. 28 , 29 However, the inclusion of MCTs and fish oil may change the overall density of the emulsion and centrifugation characteristics, with an altered effectiveness in removing exogenous lipaemia by high-speed centrifugation for SMOFlipid. We have shown that high-speed centrifugation is as effective in removing exogenous lipaemia resulting from fish oil-based emulsion.
We have shown that lipid rescue therapy will interfere with analytes based on pharmacokinetic parameters. For TPN, interference is unlikely to happen based on the median clearance of healthy individuals. However, if there is a significant decrease in clearance, accumulation of SMOFlipid and lipaemic interference will occur. It is important to note that TPN may be infused via central venous line or peripheral inserted central line. If the specimens are drawn from the same line, there is a risk of lipid contamination.
Our study was aimed at addressing interference from exogenous SMOFlipid lipaemia and not endogenous lipaemia. Endogenous lipaemia may arise from primary lipid disorders, diabetes mellitus, alcoholism, hypothyroidism and recent ingestion of triglyceride rich food. 30 Turbidity of the endogenous lipaemia is caused by accumulation of various lipoprotein particles, mainly chylomicrons, very low density lipoprotein and remnants with negligible light scattering effect from low density lipoprotein and high density lipoprotein factions. 31 No preparation of synthetic lipid emulsion can accurately represent the heterogenous proportions of lipoproteins found in human. Exogenously supplemented lipaemia has been shown not to interfere in the same way as endogenous lipameia by Nanji et al.,32 Bornhorst et al.33 and Cobbaert et al. 34 We thus acknowledge that while our study is fully applicable to exogenous SMOFlipid emulsion, but caveats must be applied when interpreting L-index interference thresholds in the context of native lipaemia.
Other limitations of our study include the fact that the results are only applicable to Roche Cobas 6000. Further studies using SMOFlipid or other synthetic fish oil containing emulsions to assess interference of other analytes, especially immunoassays, at multiple concentrations will be needed.
Given that lipid emulsion therapy may have an increasing role in emergency departments and ICUs, clinical laboratories may receive greater number of lipaemic samples due to exogenous interference including fish oil-based emulsion. It may be inappropriate to refuse to analyse these exogenously lipaemic samples as clinicians will need a result to guide patient management. Laboratories should therefore verify lipaemic thresholds with same type of lipid emulsion used by the hospital formulary and assess the performance of their chosen method of lipid removal.
Conclusion
Our study shows that the SMOFlipid optical behaviour is similar to the Intralipid using the Serum indices L-index, on the Roche Cobas 6000. For laboratories that do not have an ultracentrifuge in view of space and budget constraint, high-speed centrifugation at 60 min 21,100g facilitates the removal of fish oil-based SMOFlipid, and accurate results can be obtained. Based on the interference thresholds and pharmacokinetic parameters provided by SMOFlipid manufacturer, TPN will not interfere with chemistry analytes given sufficient clearance, but lipid rescue therapy will interfere.
Supplemental Material
sj-pdf-1-acb-10.1177_0004563220986593 - Supplemental material for Does the use of fish oil-based lipid emulsion in the clinical setting of total parenteral nutrition and lipid rescue therapy interfere with common laboratory analytes on Roche Cobas 6000?
Supplemental material, sj-pdf-1-acb-10.1177_0004563220986593 for Does the use of fish oil-based lipid emulsion in the clinical setting of total parenteral nutrition and lipid rescue therapy interfere with common laboratory analytes on Roche Cobas 6000? by Jun Guan Tan and Moh Sim Wong in Annals of Clinical Biochemistry
Footnotes
Acknowledgements
We would like to thank our pharmacy colleagues, Ms Siu Ling Khoo and Mr Herby Wi Lee Soon (Khoo Teck Puat Hospital, Department of Pharmacy), for their assistance and the supply of the SMOFlipid 20% used in the interference study.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethical approval
Not applicable. The study was done as a part of the laboratory validation procedures, and Institutional Review Board approval was exempted.
Guarantor
MSW.
Contributorship
JGT and MSW conceived the study. Both JGT and MSW were involved in protocol development. JGT performed the interference experiments and wrote the first draft of the manuscript. All authors reviewed and edited the manuscript and approved the final version of the manuscript.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
