Abstract
This is a case of a 61-year-old lady who presented to the lipid clinic with possible familial hypercholesterolaemia (Simon Broome Criteria). She was commenced on atorvastatin; however, 4 weeks later, she developed hepatitis, and therefore her atorvastatin was discontinued. Following that, her liver function tests normalized, and she was diagnosed with statin-induced hepatitis. Three years later, she was seen again in the lipid clinic with an uncontrolled lipid profile, and she was commenced on alirocumab, a Proprotein Convertase Subtilisin/Kexin type 9 (PCSK9) inhibitor. A few days later, she developed hepatitis, and subsequently, the alirocumab was discontinued. She underwent a liver biopsy, which confirmed that she had Autoimmune Hepatitis (AIH) with presumed superimposed drug injury. This is the first reported case of autoimmune hepatitis associated with alirocumab.
Introduction
Familial hypercholesterolaemia is an inherited metabolic disorder characterized by high serum total cholesterol and LDL-cholesterol (LDL-C) concentrations. A large number of mutations have been described, with the commonest affecting LDL-C receptors or the structure of apolipoprotein B, leading to a defect in the uptake and catabolism of LDL-C. 1
Its management usually involves statin therapy as first-line treatment with other oral agents, such as ezetimibe, if a high dose of a potent statin does not control the lipids adequately. 2 In patients who are intolerant of statins or who are not responding to combination oral treatment, Proprotein Convertase Subtilisin/Kexin type 9 (PCSK9) inhibitors can be considered.3,4
PCSK9 inhibitors are monoclonal antibody proteins that bind free PCSK9 molecules. This prevents PCSK9 from attaching to the LDL receptor, thus increasing the number of LDL receptors, leading to an increase in LDL-C clearance. 5 Currently, there are two approved PCSK9 inhibitors for use: alirocumab (manufactured by Sanofi as Praluent) and evolocumab (manufactured by Amgen as Repatha).
Although it has not been reported in the literature or the drug licence that alirocumab can cause AIH, there are other monoclonal antibodies that have also been reported to cause this condition, such as Natalizumab, Pembrolizumab and Infliximab.6–8
Case presentation
Hepatic profile, coagulation profile, urea, and electrolytes.
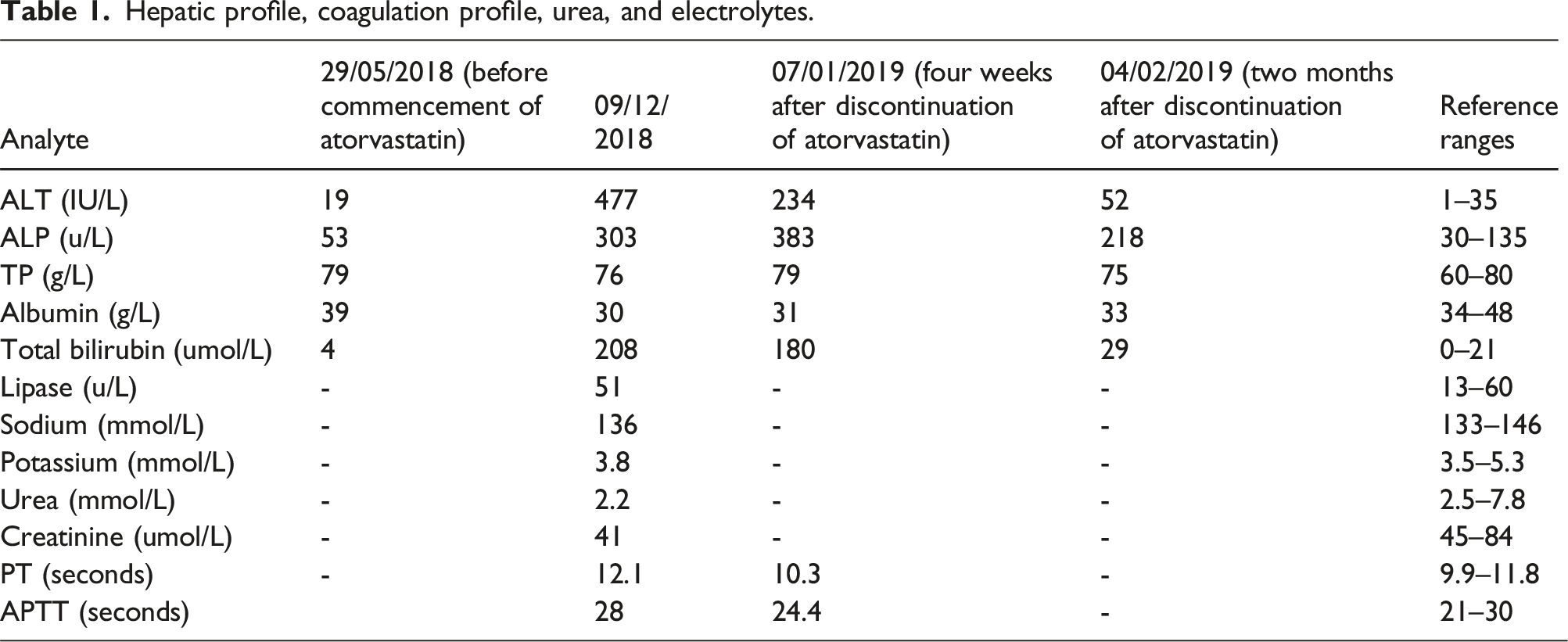
Due to the persistently deranged LFTs, significantly raised bilirubin and features suggestive of possible autoimmune liver disease which, included positive anti-smooth muscle antibody (SMA), a liver biopsy was organized, and it showed a mixed lymphoplasmacytic and neutrophilic infiltrate, together with accumulation of Kupffer cells, in addition to minor swelling of hepatocytes. The presence of very occasional lymphoid aggregates suggests the possibility of autoimmune hepatitis, so the possibility of pre-existing liver disease, or even progression from drug toxicity to an autoimmune process, cannot be absolutely excluded.
Her liver function tests improved 2 months after discontinuation of atorvastatin by conservative management, as shown in (Table 1). She continued to be seen in the lipid clinic and after 3.5 years following discontinuation of atorvastatin, her hepatic profile was completely normal; however, her cholesterol was elevated at 7.6 mmol/L with an LDL-C of 5.07 mmol/L, HDL of 1.35 mmol/L and triglycerides of 2.7 mmol/L.
Hepatic profile, coagulation profile.
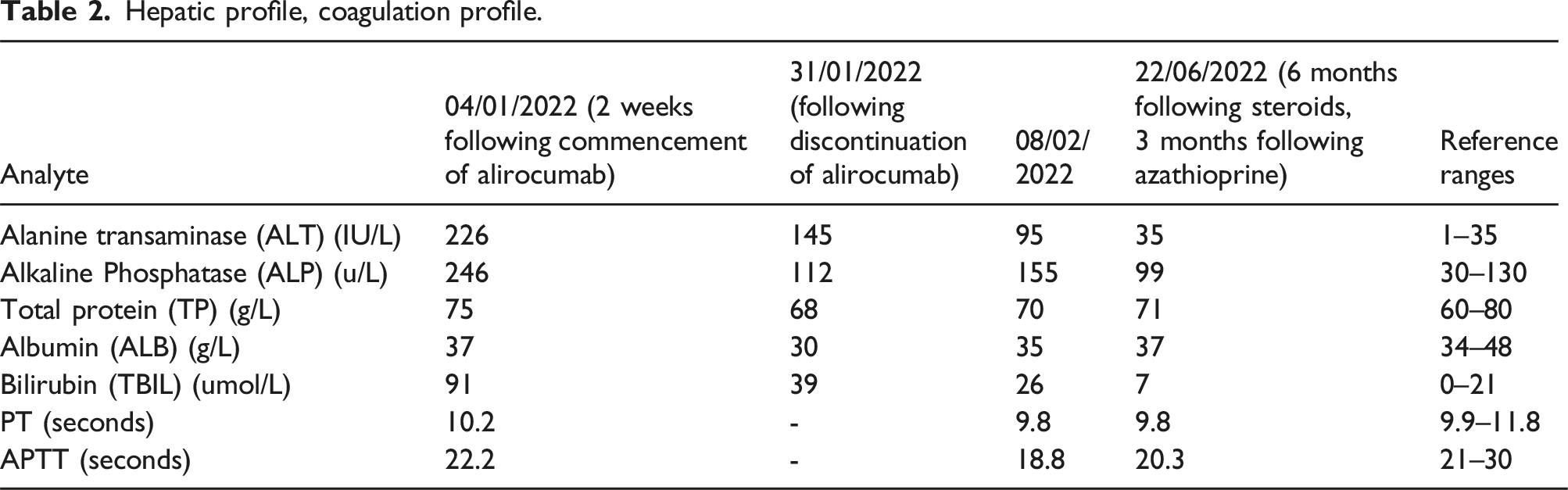
She was admitted to the hospital and had a CT liver triple-phase and liver biopsy done. Her CT liver showed no biliary obstruction, no significant focal liver parenchymal abnormality and normal perfusion to the liver. The liver biopsy revealed mild steatosis, a mixture of lymphocytes, plasma cells and mild interface hepatitis; features similar to the liver biopsy done previously.
She was commenced on prednisolone, and later, she was also started on azathioprine to treat likely autoimmune hepatitis, and her LFTs slowly improved with this treatment (Table 2). The hallmark histological features from her liver biopsies to suggest AIH are interface hepatitis with plasma cell-rich lymphoplasmocytic infiltrates and hepatocytes swelling.
Following the normalisation in her liver function, colesevelam treatment was recommended as her lipid profile was still not adequately controlled. The AIH has made the management of her hypercholesterolaemia very challenging.
Discussion
This is the first report of alirocumab being implicated in the development of autoimmune hepatitis. The mechanism by which alirocumab causes liver injury remains speculative but could be explained by several mechanisms. 8 Firstly, a hypersensitivity reaction is a possibility, although this is usually associated with fever, eosinophilia and urticaria, and this current patient did not exhibit any of these. Secondly, hepatotoxicity, as alirocumab has been reported to cause drug-induced liver injury in 0.16% of patients in the Evaluation of Cardiovascular Outcomes After an Acute Coronary Syndrome During Treatment with Alirocumab trial (ODYSSEY trial) 9 . Lastly, alirocumab is a human monoclonal antibody, and monoclonal antibody therapy sometimes causes immune mediated liver injury, but such events have not been described in alirocumab or evolocumab. 10 It is very difficult to differentiate between drug-induced liver injury (DILI) and drug-induced AIH. 7 With a drug-induced liver injury (DILI) we would expect the LFTs to normalize and not increase again once the offending agent has been discontinued. In our case, this doesn’t occur and despite stopping the offending drug LFTs continue to peak and trough. Moreover, her histological findings and optimal response to corticosteroids and immunosuppressive therapy argue for autoimmune hepatitis; however, during her second episode, the SMA was negative.
It is challenging to distinguish if this lady had hepatotoxicity due to an autoimmune immunological effect or if alirocumab unmasked a genetic predisposition to autoimmune hepatitis. These conditions share several similarities: both are driven by immunological processes and may need management with corticosteroids. In addition, the histological features overlap. 6 This lady is more likely to have an underlying AIH, given that she had a further flare with a different drug, and we suspect the alirocumab drove this.
In respect of AIH being a possible side-effect amongst PCSK9 inhibitors as a whole, a recent conference proceeding poster suggested there may also have been a case of AIH in a patient prescribed with evolocumab. 11 If so, then this condition may be a class side-effect, albeit rare, which requires consideration when prescribing.
Conclusion
Many monoclonal antibodies have been reported to cause AIH, and we now report, for the first time, a case of alirocumab associated with AIH. This case also indicates the importance of early monitoring of the liver profile in patients taking alirocumab and likely for the PCSK9 inhibitors class, in general, and may need to take place much sooner than the traditional 3-month monitoring with statin treatment.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethical approval
Not applicable.
Guarantor
Amira Ibrahim.
Contributorship
Amira Ibrahim conducted literature search, data collection, and wrote the manuscript. Geeta Prasad, E S. Kilpatrick and Timothy J. Morris contributed to the writing, reviewing and approval of the final article. All authors critically reviewed and approved the final version of the manuscript.
Correction (November 2024):
The article has been updated to correct the date in the heading ‘07/01/2019 (four weeks after discontinuation of atorvastatin)’ from ‘07/12/2019’ to ‘07/01/2019’ in Table 1.
