Abstract
Background
The indocyanine green retention rate is important for assessing the severity of liver disorders. In the conventional method, blood needs to be collected twice. In the present study, we developed an automated indocyanine green method that does not require blood sampling before intravenous indocyanine green injections and is applicable to an automated biochemical analyser.
Methods
The serum samples of 471 patients collected before and after intravenous indocyanine green injections and submitted to the clinical laboratory of our hospital were used as samples. The standard procedure established by the Japan Society of Hepatology was used as the standard method. In the automated indocyanine green method, serum collected after an intravenous indocyanine green injection was mixed with the saline reagent containing a surfactant, and the indocyanine green concentration was measured at a dominant wavelength of 805 nm and a complementary wavelength of 884 nm.
Results
The coefficient of variations of the within- and between-run reproducibilities of this method were 2% or lower, and dilution linearity passing the origin was noted up to 10 mg/L indocyanine green. The reagent was stable for four weeks or longer. Haemoglobin, bilirubin and chyle had no impact on the results obtained. The correlation coefficient between the standard method (x) and this method (y) was r=0.995; however, slight divergence was noted in turbid samples.
Conclusion
Divergence in turbid samples may have corresponded to false negativity with the standard procedure. Our method may be highly practical because blood sampling before indocyanine green loading is unnecessary and measurements are simple.
Introduction
The indocyanine green (ICG) retention rate is useful for evaluating liver function and the liver functional reserve1,2 and is also used to assess the resectable range for liver surgery.3,4
In the manual method (standard procedure; employed as a conventional method in many hospitals) used to measure blood ICG, the absorbance of serum collected after an intravenous ICG injection is measured relative to that of serum collected before the injection as a control, and the blood retention rate of ICG is calculated. This method requires blood sampling at least twice, before and after an intravenous ICG injection. Moreover, when serum turbidity (chyle) changes after ICG loading, the blood ICG retention rate may be significantly influenced. In the present study, which was conducted with the aim of developing an ICG measurement method requiring blood sampling only once without the influence of turbidity, such as chyle, we developed a photometric method in which the ICG concentration in serum collected after an intravenous ICG injection is measured at two wavelengths: 805 and 884 nm.
Measurement method
Samples
Serum samples were collected from 471 patients before and after intravenous ICG injections. Written informed consent was obtained from all participants, and the study protocol was approved by the Ethics Committee of Chiba University Graduate School of Medicine (approval number: 685). The influence of co-existing substances was evaluated using Interference Check A Plus (Sysmex Corp.).
Procedure
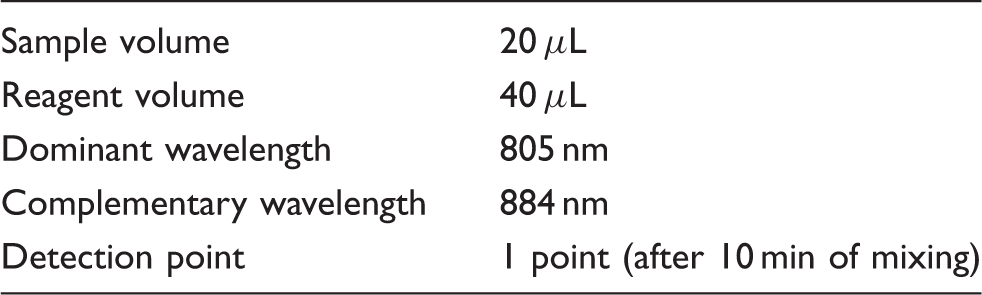
1. Manual method (standard procedure, conventional method): After the addition of 2 mL of saline to 1 mL of each serum sample and mixing, absorbance was measured at 805 nm using a 7010 clinical spectrophotometer (Hitachi High-Technologies Corp.). ICG concentrations were assessed by subtracting the absorbance of serum collected before an intravenous ICG injection from that after the injection. 2. Automated ICG method: Saline (0.9% NaCl) containing 0.1% Brij35 (HLB: 16.9) and 0.1% sodium benzoate was installed as a procedure in a BM2250 automated biochemical analyser (JEOL Ltd). Sample and reagent volumes were 20 and 40 μL, respectively.
Calibration method
Serum-added 10 mg/L ICG solution was used as the standard solution to prepare a calibration curve. An ICG value of 10 mg/L was equivalent to a plasma disappearance rate of 100%.
Results
Preliminary experiments on the automated ICG method
Absorption spectrum of ICG-added serum
ICG solution was added to pooled serum in order to adjust ICG concentrations to 1.0, 3.0, 5.0 and 7.0 mg/L, and an absorption spectrum was prepared by measuring the absorbance of each ICG-containing serum at wavelengths between 700 and 900 nm. An absorption spectrum is shown in Figure 1. Maximum absorption was observed near 805 nm at all concentrations, whereas no absorption was detected at a wavelength of 880 nm or longer. We added turbid cases (without ICG loading) of the absorption spectrum in Supplementary Figure 1.
Absorption spectrum of ICG-added serum. Each sample was spiked with ICG to pool serum. ICG values were 0, 1, 3, 5 and 10 mg/L, respectively.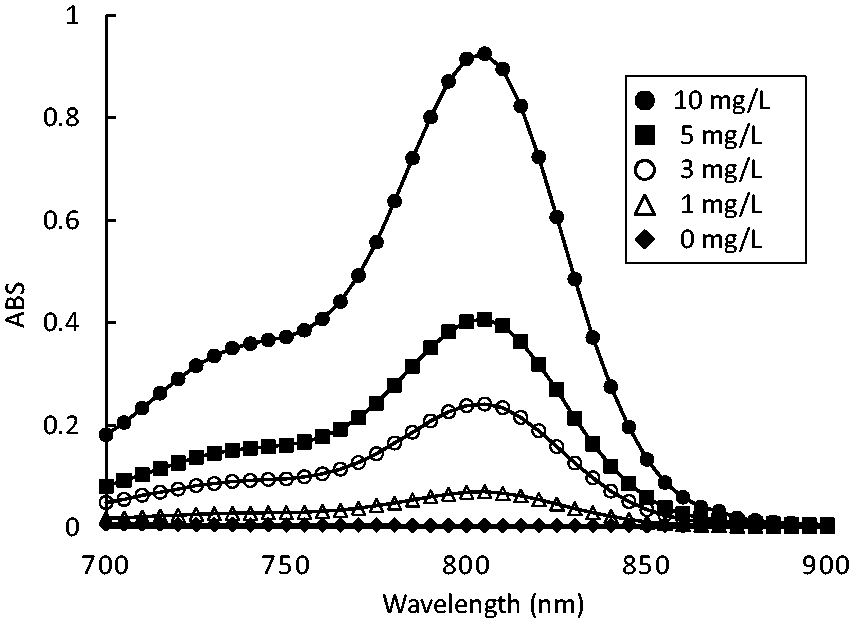
Relationship between the absorbance of patient samples measured at 805 and 884 nm
In order to confirm the absence of substances with maximum absorption at 884 nm in serum, the absorbance of serum collected before the intravenous ICG injection was measured at 805 and 884 nm, and the correlation of absorbance was assessed. The correlation of absorbance of patient samples between 805 nm (x) and 884 nm (y) was r=0.9991, y=0.90x−0.003 (Figure 2).
Correlation of absorbance of patient samples between 805 nm and 884 nm.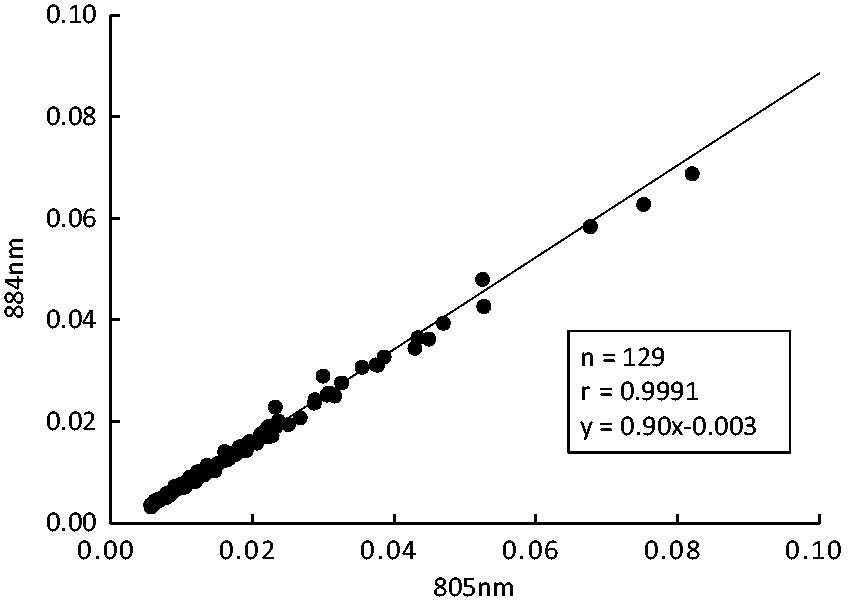
Analytical performance
The analytical conditions (BM-2250).
Reproducibility
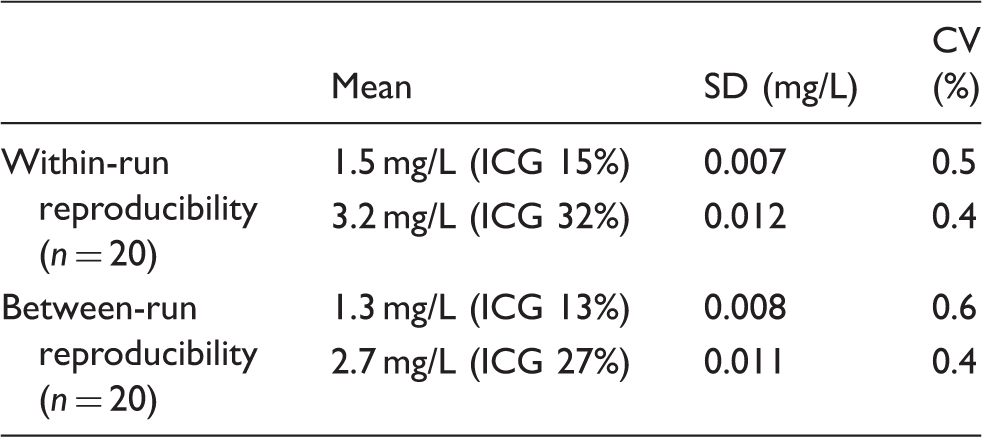
1. Within-run reproducibility: When within-run reproducibility was investigated using serum samples (pooled serum samples after the intravenous ICG injection) containing ICG at mean concentrations of 1.4 and 3.0 mg/L (n = 20), coefficient of variation (CV) was 0.5% or lower at both concentrations (Table 2). 2. Between-run reproducibility: Since ICG concentrations decrease with exposure to light, serum samples were stored at −80℃ with a shield until analysed. Between-run reproducibility was investigated, while the reagent remained installed in the analyser (continued for 30 days) using serum samples containing ICG at mean concentrations of 1.4 and 3.0 mg/L (n = 20, 30 days). CV was 1.5% or lower at both concentrations (Table 2). Reproducibility. ICG: indocyanine green; CV: coefficient of variation; SD: standard deviation.
Linearity
Linearity passing the origin (zero absorbance) was confirmed up to 10 mg/L.
Co-existing substances
Interference Check A Plus was added to serum containing 3.0 mg/L ICG, and the influence of co-existing substances was measured. The final concentrations of the added co-existing substances were as follows: haemoglobin, 4.0 g/L; conjugated bilirubin, 160 mg/L; free bilirubin, 170 mg/L and chyle, 560 formazin turbidity units (FTU). The rates of interference with the conventional method were +9.4, + 1.9, + 2.3 and +80.4%, respectively, while those with this method were +1.8, + 2.6, + 2.2 and +3.4%, respectively. In the interference test of chyle with the conventional method, 100 FTU was equivalent to 0.37 mg/L ICG.
Correlation
The correlation between the conventional method (x) and this method (y) was r=0.992, regression equation y = 1.04x + 0.06, and some divergent cases were noted (Figure 3). Serum was turbid in all divergent cases. We added divergent case (turbid cases before ICG loading) of the absorption spectrum in Supplementary Figure 2.
Correlation between the conventional method and this method. Arrows indicate divergent cases (serum was turbid in all divergent cases).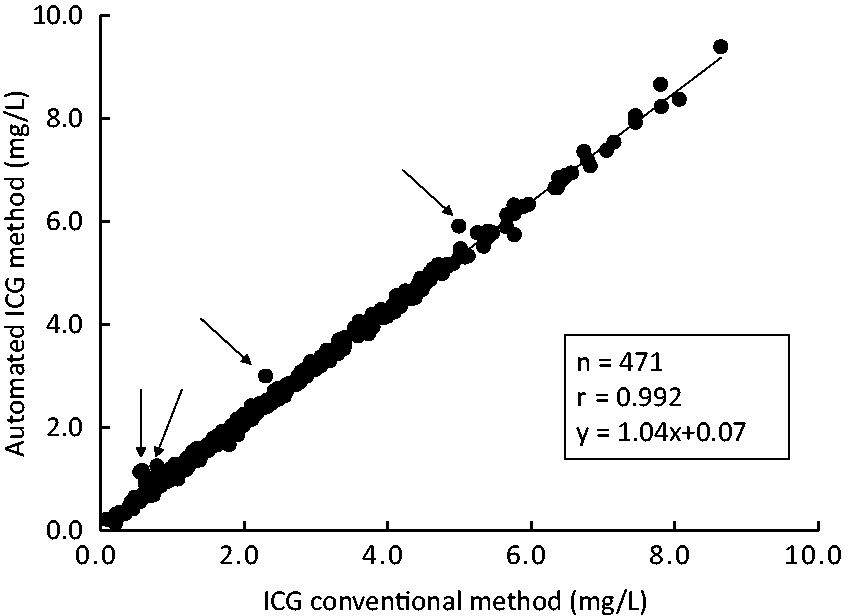
Discussion
We herein developed an automatic analysis method for ICG measurements and compared its precision with that of the conventional method. This method showed favourable quantitativity and did not require blood sampling before an intravenous injection of ICG solution. Although more than 1 mL of serum was required in the conventional method, it was possible to use less than 0.1 mL of serum in our method. And cross-contamination between samples is considered difficult to occur because the carryover of the automatic analyser is less than 1/10,000.
The Child-Pugh score is the most widely used method in this article for the evaluation of the liver functional reserve in liver function tests and is also the most frequently employed method to judge the severity and prognosis of hepatic cirrhosis. 5 However, patients with mild hepatic cirrhosis and those with metastatic liver cancer indicated for surgical liver resection are mostly classified as Child-Pugh score A. 3 Therefore, difficulties are associated with accurately judging the liver functional reserve based on the Child-Pugh score. In order to assess the surgically resectable range, Makuuchi et al. 6 designed an evaluation method (Makuuchi-Criteria) based on the presence or absence of ascites, blood bilirubin concentrations and the 15-min retention rate in the ICG test. In Japan, many institutions judge the resectable range before liver resection using the Makuuchi-Criteria, which may contribute to reducing the mortality on hepatectomy. Therefore, evaluating the 15-min ICG retention rate is important for assessing the liver functional reserve for surgical liver resection.
In the standard measurement method of the 15-min ICG retention rate, blood is collected before and then after an intravenous ICG injection, the rate is calculated by subtracting the absorbance at 805 nm of serum collected before from that collected after the intravenous ICG injection. This method requires blood sampling at least twice. Moreover, measurement errors occur due to haemolysis during blood sampling and changes in the intensity of chyle, which are disadvantageous. Furthermore, it is measured manually using a spectrophotometer, which requires a relatively large volume of blood. In contrast, in a method in which ICG in ICG-loaded serum is decolourized using hypochlorous acid and used as a control, 7 blood sampling before ICG loading is unnecessary. An automated analysis method by decolourizing ICG in ICG-loaded serum using periodic acid has also recently been investigated. However, these methods require the use of strong acid as a reagent, and periodic acid in an automated analysis method 8 may negatively influence other measurement reagents. An ICG clearance metre has also recently been developed, in which a sensor is attached to the fingertip and the ICG concentration is measured using an optical sensor.9–11 This method does not require blood sampling; however, a slightly weak correlation with the conventional method9,11 and the high price of the device are disadvantageous.
In order to overcome these disadvantages of the conventional method, we attempted to develop an automated ICG method in which the ICG concentration is measured at a long wavelength considering the complementary wavelength as a principle. In order to investigate the presence or absence of biological component-derived pigments in serum, we analysed the correlation of the absorbance of serum samples from 120 patients including those containing haemolysis and chyle between 805 nm (x) and 884 nm (y). A very strong correlation (r=0.998, y=0.91x−0.002) was observed, and the mean absorbance at 805 and 884 nm were 0.020 and 0.016, respectively. Since an absorbance of 0.01 corresponded to an ICG retention rate of approximately 1%, the error of the ICG concentration measurement employing 884 nm as a complementary wavelength was within an ICG retention rate of approximately 0.4%, which is considered to be within an acceptable error range.
The correlation between this method and the conventional method was favourable; however, some divergent cases were noted in the low concentration range, and the value measured by this method was higher in all these cases. When the properties of these samples were investigated, turbidity derived from chyle was observed in all samples, suggesting that the condition of chyle in patient blood may change (decrease) when an intravenous ICG injection is delayed after blood sampling. In this case, the conventional method is strongly influenced by turbidity, such as chyle, and causes a significant error. In contrast, our method is not influenced by chyle. Accordingly, it was assumed that absorbance was high in divergent cases due to the influence of chyle in serum collected before the intravenous ICG injection; however, chyle decreased in serum after the ICG injection, causing a negative error in the conventional method. Moreover, the conventional method is influenced by haemolysis; therefore, a significant error may be caused when haemolysis occurs in either blood collected before or after an intravenous ICG injection, whereas no such error occurs in our method.
Conclusion
The analytical performance of the automated ICG method using a long wavelength as a complementary wavelength was favourable. Since this method does not require blood sampling before an intravenous ICG injection or cause an error due to chyle or haemolysis, it may be useful to measure ICG concentrations for ICG retention rate measurements.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethical approval
Ethics Committee of Chiba University Graduate School of Medicine (approval number, 685).
Guarantor
MS.
Contributorship
MS, EH, SO designed the research; YukS, MS, TY and YujS performed the research; YukS, MS, TY, YujS, and KM were involved in analysis and interpretation of data; MS, YujS and KM were involved in gaining ethical approval; and YS and MS wrote the first draft of the manuscript. All authors reviewed and edited the manuscript and approved the final version of the manuscript.
Supplementary material
Additional supplementary information may be found with the online version of this article.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
