Abstract
Background
Progranulin (GP88) is an 88-kDa glycoprotein growth factor with important biological effects in tumorigenesis and tumour survival. We investigated the usefulness of measuring serum GP88 concentrations as a predictive biomarker for hepatocellular carcinoma in patients with viral hepatitis C after treatment with direct-acting antiviral agents.
Methods
We measured the serum GP88 concentrations by using a sandwich enzyme-linked immunoassay from 67 healthy control subjects and 29 patients (20 patients who did not develop hepatocellular carcinoma and 9 patients who developed hepatocellular carcinoma after treatment) with viral hepatitis C after treatment with asunaprevir and daclatasvir.
Results
The serum GP88 concentrations of patients with chronic hepatitis C prior to antiviral treatment were significantly higher than those of healthy control subjects. After antiviral treatment, the serum GP88 concentrations of patients who eventually developed hepatocellular carcinoma were significantly higher than those who did not develop hepatocellular carcinoma. The changes in the serum GP88 concentrations before and after treatment in patients who developed hepatocellular carcinoma were significantly lower than those in patients who did not develop hepatocellular carcinoma. The cumulative incidence of hepatocellular carcinoma was significantly higher in either patients with high serum GP88 concentrations after treatment or those with small changes of serum GP88 concentrations pre- and post-treatment.
Conclusions
Sustained high concentrations of serum GP88 in patients treated with direct-acting antiviral agents are correlated with the risk of developing hepatocellular carcinoma.
Keywords
Introduction
Liver cancer is the fourth most common cause of cancer-related death worldwide, with an estimated 841,080 new cases diagnosed in 2018. 1 Hepatocellular carcinoma (HCC) comprised 75%–85% of primary liver cancer, with one of the main causes being chronic hepatitis C1. While direct-acting antivirals (DAAs) have been successful in eliminating the hepatitis C virus in almost all of these patients, the risk of HCC remains even after sustained virologic response (SVR).2–5 Therefore, it is necessary to continue surveillance for liver cancer in patients treated for chronic hepatitis C virus.
Progranulin (GP88) is an 88-kDa cysteine-rich glycoprotein also known as granulin–epithelin precursor, 6 acrogranin, 7 and PC cell-derived growth factor. 8 GP88 is a granulin precursor protein; however, in contrast to proinflammatory granulin, it is generally considered to be anti-inflammatory. 9 In vivo and in vitro studies have shown many biological effects of GP88, such as cellular proliferation,10,11 invasiveness,11–14 angiogenesis,13,15 malignant cell transformation,16,17 resistance to anticancer drugs,18–21 and immune evasion. 22 GP88 overexpression in tissues, or the increase of GP88 concentrations in biological fluids, has been observed in many different diseases. In fact, GP88 is an expected biomarker with diagnostic, predictive and prognostic qualities.23–28 In HCC, several studies have demonstrated GP88 overexpression in liver tissue,29,30 but the relationship between GP88 in serum and the development of HCC after chronic hepatitis C virus infection has not been reported.
The aim of the present study was to investigate the usefulness of measuring serum GP88 (sGP88) as a predictive biomarker for HCC in patients with chronic viral hepatitis C after DAA treatment.
Materials and methods
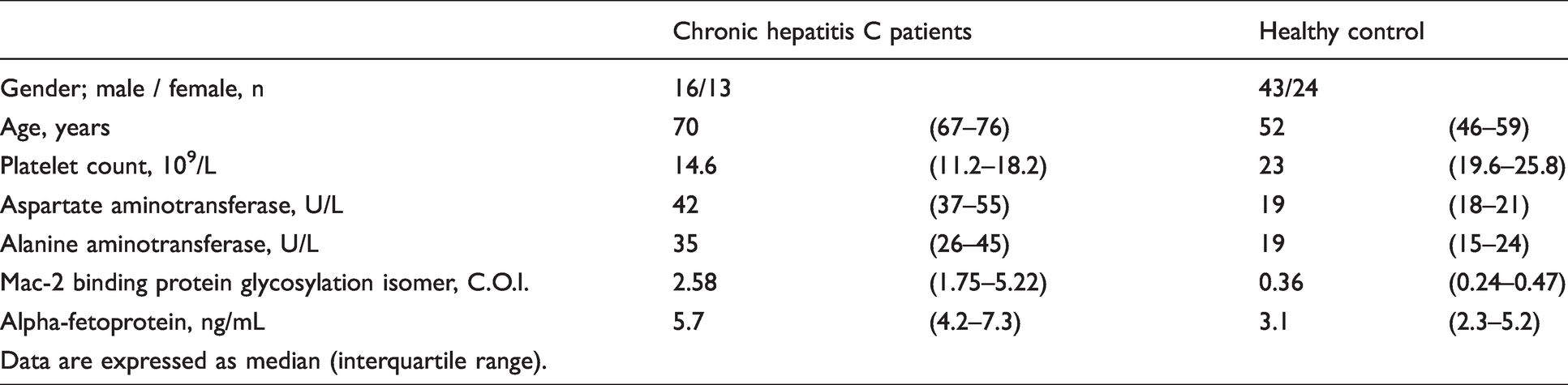
Characteristics of healthy control subjects and patients with chronic hepatitis C
A retrospective observational study was performed at Gifu University Hospital and Gifu Municipal Hospital. Study approval was obtained from the Gifu University Hospital Clinical Research Ethics Board (2018–031) and Gifu Municipal Hospital Ethics Board (27–255). For the present study, we enrolled 67 healthy control subjects and 29 patients diagnosed as having chronic hepatitis C in 2014 and 2015 at Gifu University Hospital or Gifu Municipal Hospital. The characteristics of the study subjects are shown in Table 1. Patients who were not administered any anti-inflammatory agents were treated with asunaprevir 200 mg/day and daclatasvir 60 mg/day DAAs for 24 weeks. Patients were declared as sustained virological response if they were hepatitis C virus-RNA negative for 24 weeks after the completion of antiviral therapy. Among these patients, 9 developed HCC during the median observational period of 1.2 years after SVR (HCC group), whereas 20 did not develop HCC after SVR during the median observational period of 3.2 years (non-HCC group). No patient had events such as resistance-associated mutations in NS3 and NS5A before treatment. Participants who had concentrations of aspartate aminotransferase (AST) <30 U/L, alanine aminotransferase (ALT) <40 U/L, and estimated glomerular filtration rate >90 mL/min/1.73 m2 were enrolled as the healthy control subjects. We used serum from the healthy control subjects that had been collected by the Resource Center for Health Science in Japan. These were sampled before treatment (pre-treatment) and at the end of the 24-week treatment period (post-treatment). The serum samples were stored at −80°C until analysis. To examine GP88 expression in liver tissues via an immunohistochemical assay, we used liver tissue samples from patients with HCC. Ethics approval was obtained from the Gifu Municipal Hospital.
Baseline characteristics of patients with chronic hepatitis C and healthy control subjects.
Measurement of sGP88 concentration
We used a sandwich enzyme-linked immunosorbent assay system established by a previous report to measure the GP88 concentrations in the serum. 26 This assay does not recognize processed granulin and is specific for full-length GP88 by using a pair of anti-human GP88 mouse monoclonal antibody and anti-human GP88 rabbit antibody (A&G Pharmaceutical, Inc., Precision Antibody, Columbia, MD, USA). A total of 100 μL of 1:20 diluted serum in 1% skim milk were applied to each well, which had been coated overnight with 10 μg/mL anti-human GP88 mouse monoclonal antibody (6B3 antibody) and was incubated for 2 h at room temperature. After washing each well, 100 μL of anti-human GP88 rabbit antibody (A&G Pharmaceutical, Inc.) was added and then incubated for 1 h. After washing again, 100 μL of horseradish peroxidase (HRP)-goat anti-rabbit IgG antibody (KPL Inc., MD, USA) was added to each well and incubated for 1 h. After further washing, TMB substrate reagent (BD Biosciences, Inc., San Diego, CA, USA) was added, and absorbance was measured at OD 620 nm. This assay has been verified that within-run repeatability below 4.4%, between-day repeatability below 7.7%, no effects of exiting substances, and limit of detect 1 ng/mL.
Immunohistochemical analysis
Formalin-fixed paraffin-embedded sections of liver tissue obtained from partial HCC resections were used to evaluate the histopathological characteristics and GP88 expression by immunohistochemical staining using 6B3 anti-GP88 monoclonal antibody (A&G Pharmaceutical). Five-micron tissue sections on positively charged glass slides were deparaffinized using xylene, rehydrated in graded ethanol solutions and then blocked in 10% normal goat serum. The sections were incubated with an anti-human GP88 primary rabbit monoclonal antibody (clone from 6B3; A&G Pharmaceutical Inc.). Histofine® Simple Stain™ MAX PO peroxidase-labelled anti-rabbit IgG polyclonal antibody (Nichirei Biosciences, Tokyo, Japan) was used as the secondary antibody. After washing, bound HRP was detected by 3,3′-diaminobenzidine as a chromogen, turning positive signals into a dark brown colour. The washed slides were contrast-stained with Mayer’s hematoxylin.
Statistical analysis
All analyses were performed using the statistical software JMP® 13 (SAS Institute Inc., Cary, NC, USA). The concentrations of sGP88 and other markers and the changes of the pre- and post-treatment concentrations were compared among each group by the Wilcoxon rank-sum test. A comparison of the concentrations of sGP88 and other markers between pre- and post-treatment was performed by the one-tailed Wilcoxon signed-rank test. The cumulative incidence curve was assessed using the Kaplan–Meier method, and the differences between the groups were assessed using the log-rank test. The hazard ratios were determined by the Cox proportional hazards regression models. A P-value <0.05 was considered as statistically significant.
Results
sGP88 concentrations in patients with chronic hepatitis C before treatment and in healthy control subjects
We compared the sGP88 concentrations in healthy subjects and in patients with pre-antiviral treatment for chronic hepatitis C prior to the start of antiviral treatment. The median sGP88 concentration in the healthy control group was 49.0 (interquartile range, 43.1–59.6) ng/mL, which was significantly lower than that in patients with chronic hepatitis C (106.0 (90.8–131.2) ng/mL) (P < 0.001) (Figure 1).
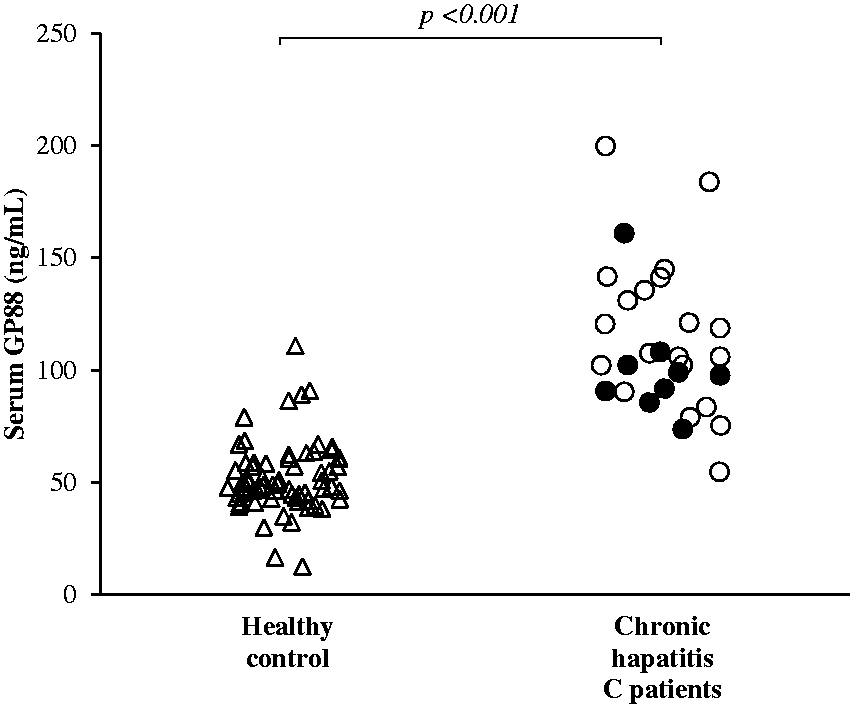
Serum GP88 concentrations in healthy control subjects and patients with chronic hepatitis C pre-antiviral treatment. △, healthy control subjects (n = 67). ○, patients without hepatocellular carcinoma (HCC) after treatment with direct-acting antivirals for hepatitis C (n = 20). ●, patients who developed HCC after treatment with direct-acting antivirals for chronic hepatitis C (n = 9). The P-values were analysed by the Wilcoxon rank-sum test.
sGP88 concentrations and other liver-related markers in patients before and after treatment for chronic hepatitis C
We then compared the sGP88 concentrations in patients with chronic hepatitis C pre- and post-treatment (Figure 2). In addition, these patients were further stratified into two groups: those who developed HCC (n = 9) and those who did not develop HCC post-treatment (n = 20). The pre-treatment medians for the sGP88 concentrations in patients who developed and did not develop HCC were 97.7 (90.8–102.3) ng/mL and 113.2 (99.3–137.1) ng/mL, respectively, and no significant difference was observed (P = 0.14). The post-treatment medians of the sGP88 concentration in the HCC and non-HCC groups were 85.0 (77.6–95.0) ng/mL and 67.0 (63.0–78.0) ng/mL, respectively, and a significant difference was observed (P = 0.005). A significant difference was also seen in both the HCC and non-HCC groups between the pre- and post-treatment sGP88 concentrations (P = 0.074 and P < 0.001, respectively). The medians of the changes between the pre- and post-treatment in the HCC and non-HCC groups were significant (P = 0.002) at −8.0 (−23.0 to −3.7) ng/mL and −42.0 (−60.0 to −32.1), respectively. In the HCC group, the pre-treatment concentrations of AST, ALT and Mac-2 binding protein glycosylation isomer (M2BPGi) were significantly higher than those of post-treatment. In the non-HCC group, the pre-treatment concentrations of AST and ALT were significantly higher than those of post-treatment. Additionally, significant differences in the pre-treatment concentrations of platelet counts, albumin, AST, ALT, M2BPGi and alpha-fetoprotein (AFP) between the HCC and non-HCC groups were observed. However, there was a significant difference only with the albumin and sGP88 concentrations post-treatment (Supplementary Table S1).
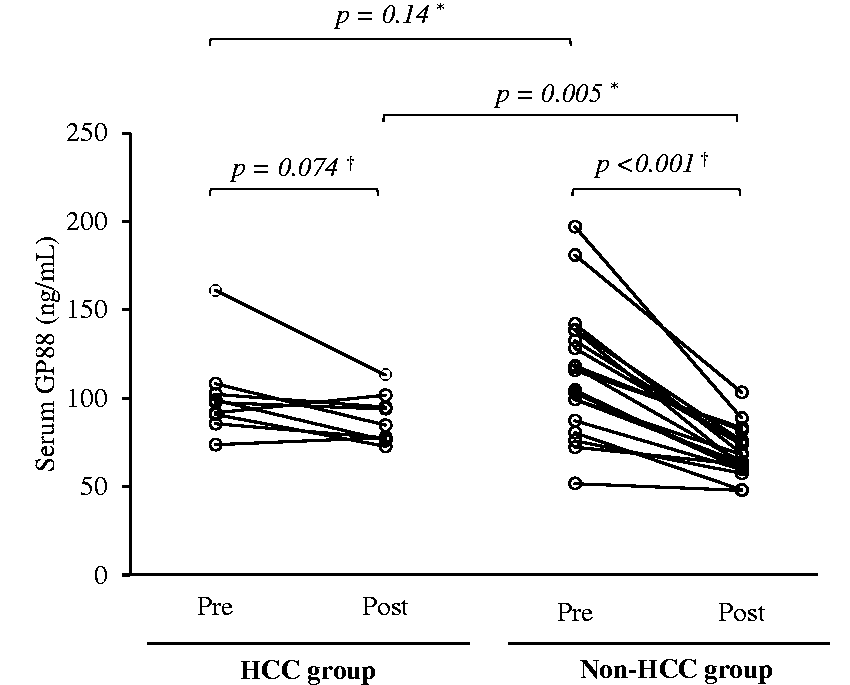
Serum GP88 concentrations pre and post antiviral treatment. Pre-treatment: the serum GP88 concentrations in patients with pre-antiviral treatment for chronic hepatitis C prior to the start of antiviral treatment. Post-treatment: the serum GP88 concentrations in patients after 24 weeks of direct-antiviral treatment. Hepatocellular carcinoma (HCC) group: patients who developed HCC after treatment with direct-acting antivirals for chronic hepatitis C. Non-HCC group: patients without HCC after treatment with direct-acting antivirals for hepatitis C. *The P-values were analysed by Wilcoxon rank-sum test. †The P-values were analysed by Wilcoxon signed-rank test.
Risk factor for the development of HCC
The univariate analysis for the risk of HCC development after DAA treatment was performed by the Cox proportional hazards regression models. The following factors were found to significantly promote HCC development: (1) at pre-treatment, albumin (every 1 g/dL increase), AST (every log 10 U/L increase) and Mac-2 binding protein glycosylation isomer (M2BPGi; every log 1 cut-off-index increase); (2) at post-treatment, GP88 (every log 10 ng/mL increase); and (3) for the change ratios of post/pre-treatment, M2BPGi (every log 0.1 decrease) and GP88 concentrations (every log 0.1 increase). The multivariate analysis revealed the pre-treatment albumin concentrations and the change ratios of post-/pre-treatment in the sGP88 concentrations as independent risk factors for increasing the development of HCC post-DAA treatment (Table 2).
Univariate and multivariate analysis for the development of hepatocellular carcinoma.
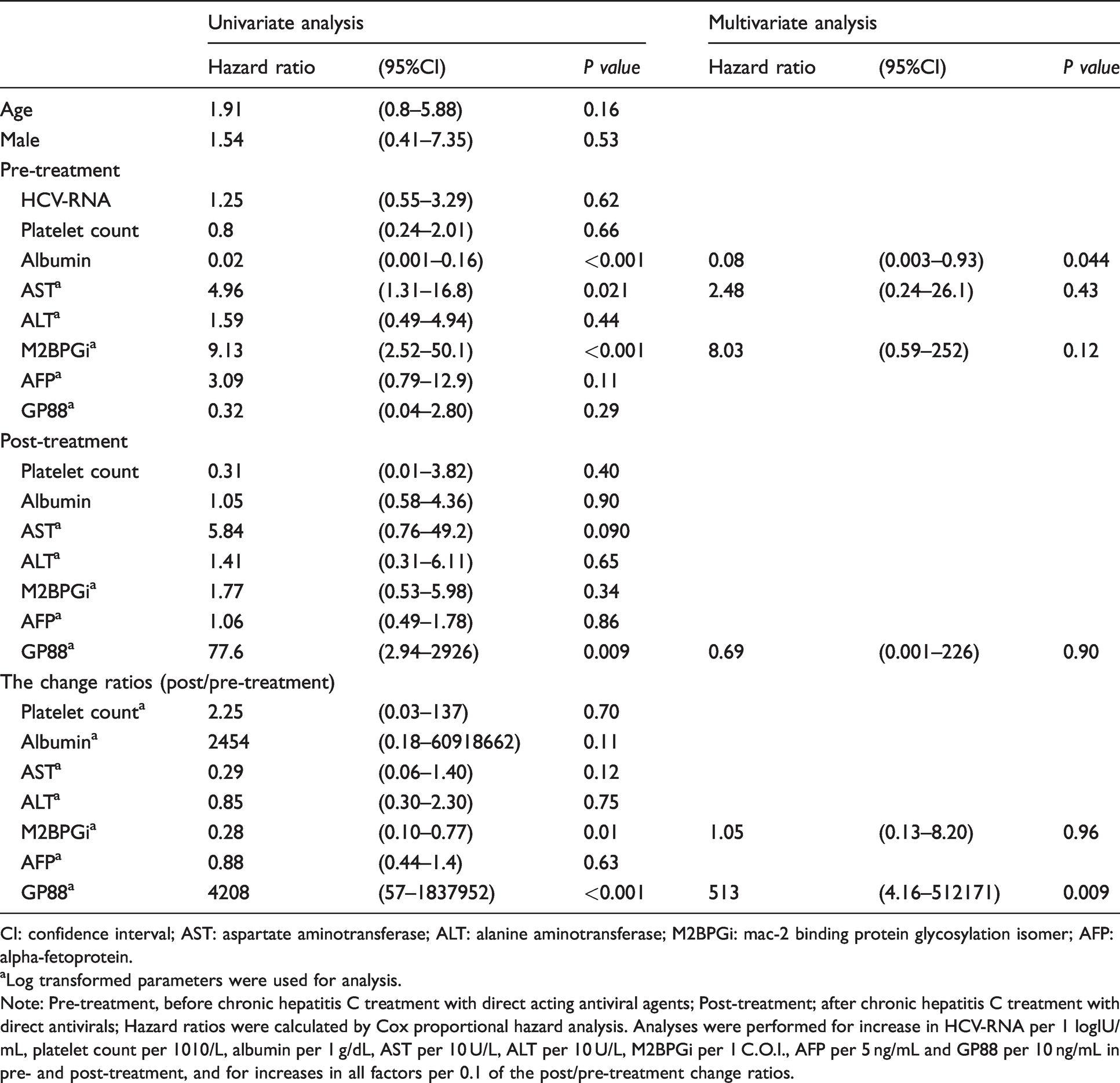
CI: confidence interval; AST: aspartate aminotransferase; ALT: alanine aminotransferase; M2BPGi: mac-2 binding protein glycosylation isomer; AFP: alpha-fetoprotein.
aLog transformed parameters were used for analysis.
Note: Pre-treatment, before chronic hepatitis C treatment with direct acting antiviral agents; Post-treatment; after chronic hepatitis C treatment with direct antivirals; Hazard ratios were calculated by Cox proportional hazard analysis. Analyses were performed for increase in HCV-RNA per 1 logIU/mL, platelet count per 1010/L, albumin per 1 g/dL, AST per 10 U/L, ALT per 10 U/L, M2BPGi per 1 C.O.I., AFP per 5 ng/mL and GP88 per 10 ng/mL in pre- and post-treatment, and for increases in all factors per 0.1 of the post/pre-treatment change ratios.
Cumulative incidence of HCC development among patients treated with DAAs
We calculated the cumulative incidence of HCC by employing the Kaplan–Meier method stratified using the median of the pre-treatment, post-treatment and change ratios (post-/pre-treatment) of the sGP88 concentrations in all patients. The cumulative incidence of HCC was analysed for the median pre-treatment sGP88 concentrations above (n = 13) or below (n = 16) 106 ng/mL (Figure 3(a)), median post-treatment concentrations above (n = 14) or below (n = 15) 76.2 ng/mL (Figure 3(b)), and median post-/pre-treatment change ratios in the sGP88 concentrations above (n = 14) or below (n = 15) 0.70 (Figure 3(c)). In the log-rank test, although the cumulative incidence of HCC for the pre-treatment sGP88 concentrations over 106 ng/mL was not significant compared with that for 106 ng/mL or less (P = 0.17), the cumulative incidence of HCC for the post-treatment sGP88 concentrations over 76.2 ng/mL was significantly higher than that of 76.2 ng/mL or less (P = 0.026). In addition, the cumulative incidence of HCC for the change ratios of the post-/pre-treatment GP88 concentrations over 0.70 was significantly higher than that of 0.70 or less (P = 0.002).
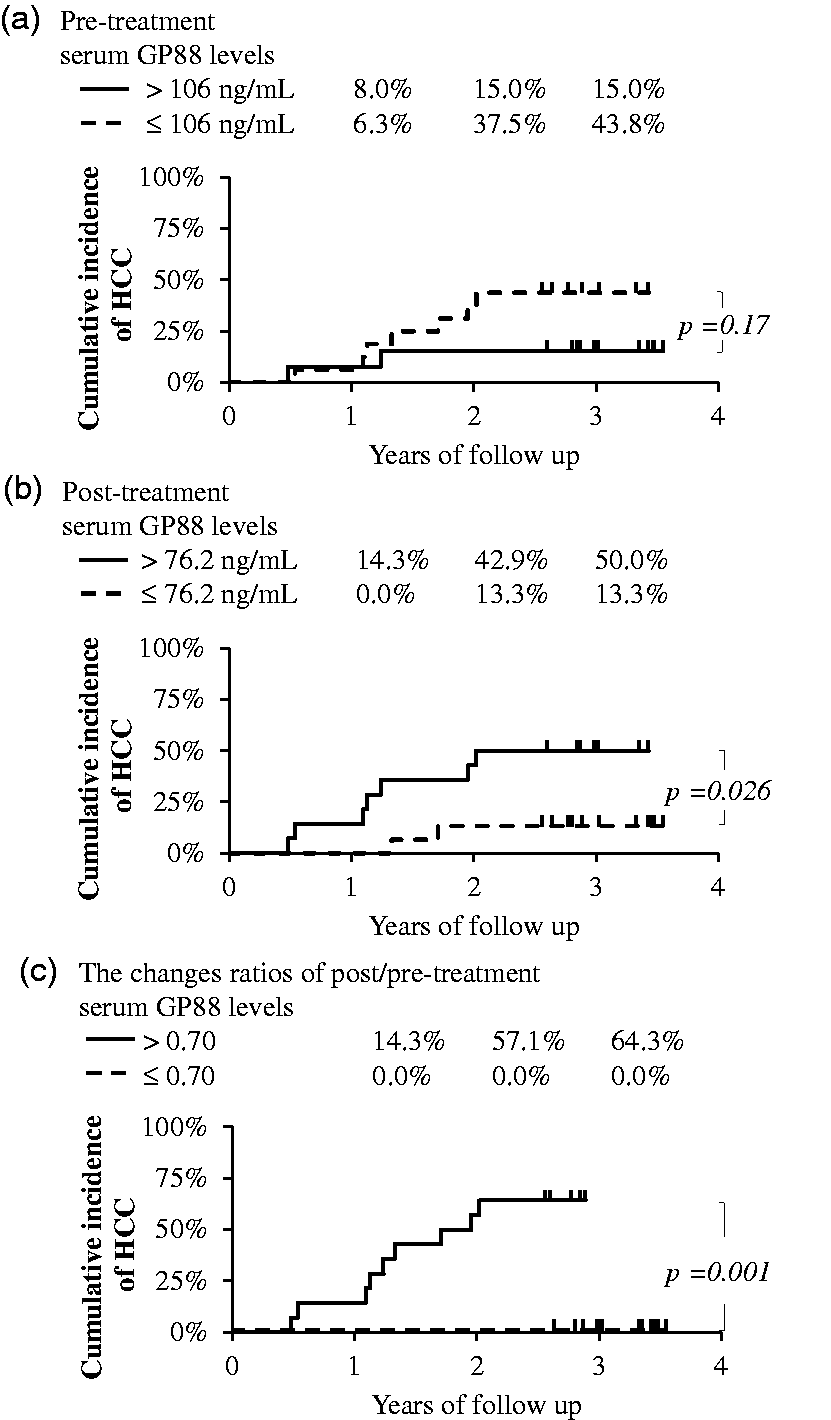
Cumulative incidence of hepatocellular carcinoma stratified by the serum GP88 concentrations. (a) Stratified by the median serum GP88 concentrations of before antiviral treatment (106 ng/mL), (b) stratified by the median serum GP88 concentrations after 24 weeks of antiviral treatment starting (76.2 ng/mL), (c) stratified by the median change ratios of the post-/pre-treatment serum GP88 concentrations (0.70). Data are arranged into strata based on the medians of the serum GP88 concentrations or the change ratios. The P-values were analysed by log-rank test.
Immunohistochemical analysis of GP88 in the liver tissues of patients with HCC
We performed histological analysis by immunohistochemical staining of GP88 in the partially resected liver tissues of patients with HCC to evaluate GP88 protein expression in HCC and non-HCC tissues. GP88 immunohistochemical staining was observed in the tumour cell cytoplasm, and HCC tissue section was strongly positive for GP88. In the non-tumour cells, GP88 stained negative or weakly positive (Figure 4). Thus, GP88 was expressed in both HCC and non-HCC tissues, but the amount of expression visually was greater in the HCC than in non-HCC tissues.
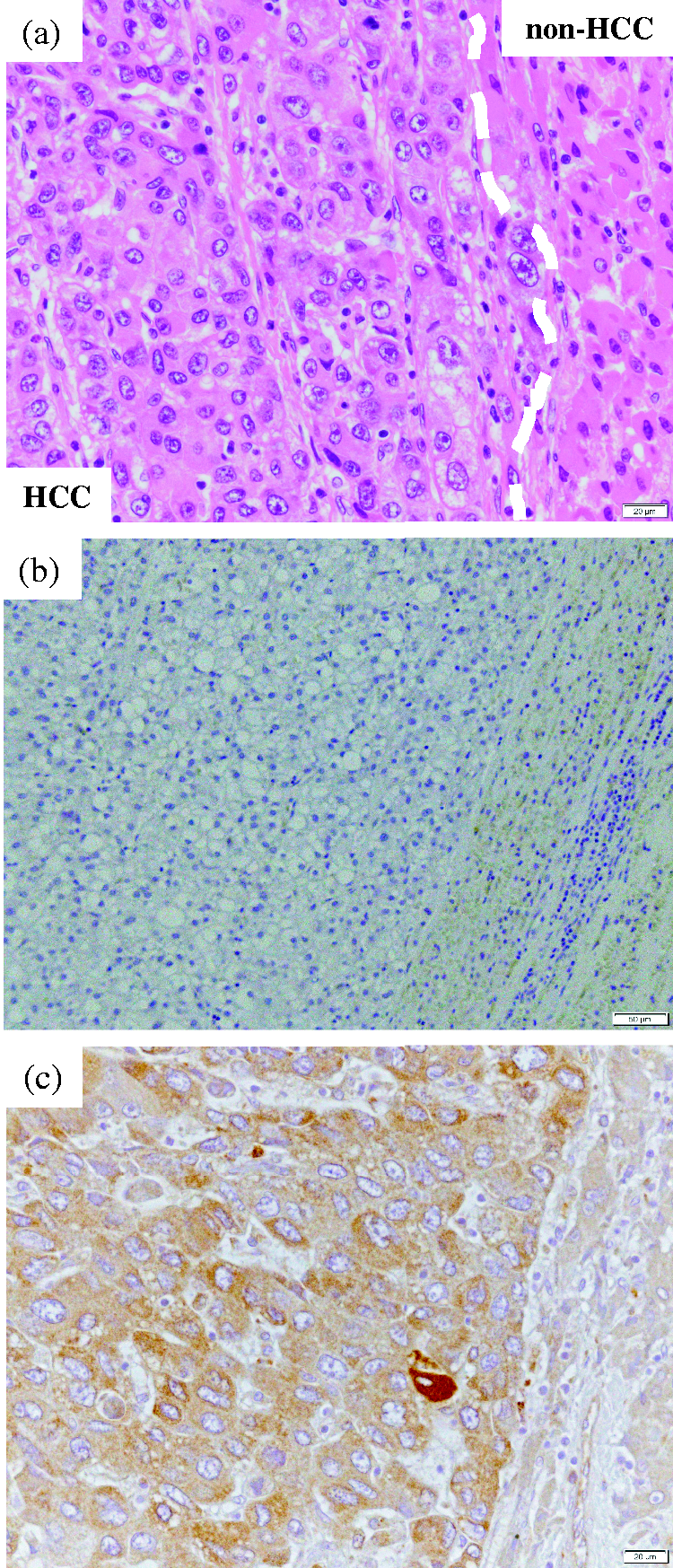
Hematoxylin and eosin staining and immunohistochemical staining of GP88 in partially resected liver tissues of patients with hepatocellular carcinoma. (a) Hematoxylin and eosin staining. (b) Negative control. (c) Immunohistochemical staining of GP88. Immunohistochemical staining conditions are described in the Materials and Methods section. The results are strongly positive in the tumour tissue area and weakly positive in the non-tumour tissue area.
Discussion
Previous reports have demonstrated that GP88 concentrations are overexpressed in human or animal cancer tissue or cell lines, such as breast cancer,31,32 epithelial ovarian cancer, 33 glioblastoma, 34 prostate cancer, 35 and biliary tract cancer. 36 In addition, the elevation of sGP88 concentrations has been revealed for patients with cancer, such as breast cancer, 37 epithelial ovarian cancer,38,39 non-small cell lung carcinoma, 23 and malignant lymphoma. 25 In HCC, previous histological studies have shown that the GP88-RNA concentrations and GP88 protein expressions in HCC tissues were significantly higher than those in non-tumour tissues and normal liver tissues.14,29 The present results also showed that high concentrations of GP88 expression were detected in the serum and in the histological analysis of patients with HCC.
The present study demonstrated a significant difference in the sGP88 concentrations between healthy subjects and patients with chronic hepatitis C treated with DAAs. Although the sGP88 concentrations in patients with chronic hepatitis C were significantly higher than those in healthy controls, there was no significant difference in the pre-treatment GP88 concentrations among the groups that did and did not develop HCC after DAA treatment. In contrast, the post-treatment sGP88 concentrations in the HCC group were significantly higher than those in the non-HCC group; hence, the change ratios of the post-/pre-treatment sGP88 concentrations in the HCC group were significantly higher than those in the non-HCC group. Moreover, we showed that the pre- to post-treatment changes of the sGP88 concentrations were associated with an increased probability of developing HCC. Although the limitations of the current study include the limited number of patients who developed HCC and the use of only asunaprevir and daclatasvir as DAAs, to the best of our knowledge, this is the first report suggesting that the sGP88 concentrations may be a predictive biomarker for the development of HCC in patients who have undergone DAA therapy.
AST, ALT and M2BPGi are markers associated with the development of hepatitis and liver cancer. We observed decreases in the AST, ALT, M2BPGi and sGP88 concentrations in the patients in both the HCC and non-HCC groups after antiviral treatment, which was in agreement with a previous retrospective study showing that patients treated with DAAs have decreased concentrations of serum AST, ALT and liver fibrosis markers, such as M2BPGi, autotaxin and transient elastography score. 40 These results indicate that hepatitis is immediately improved by DAA therapy. In addition, decreases in the M2BPGi and sGP88 concentrations are reasonable because they are positively correlated with the progress of hepatitis. In patients with hepatitis C, the platelet counts decrease with the progression of hepatitis and liver fibrosis, but the results of the present study did not show a significant correlation between the sGP88 concentrations and the platelet counts (data not shown). The production of GP88 in platelets has not been fully defined, and its production in platelets has not been clarified, but there are reports that there is no correlation between the two even in patients with acute lymphoblastic leukemia. 41 There appears to be no direct relationship between the sGP88 concentrations and thrombocytopenia associated with hepatitis progression.
A meta-analysis study has demonstrated that interferon treatment, a conventional treatment for hepatitis C virus infection, slows the development of HCC, 42 and that interferon-free DAA treatments have been as effective as interferon treatment. However, the risk of HCC development remains after the elimination of the hepatitis C virus, with reported incidences at 5- and 10-year follow-up examinations of 2.3%–8.8% and 3.1%–11.1%, respectively. 43 The meta-analysis study shows that a defined risk factor for the development of HCC is liver fibrosis, and that the HCC incidence rate in patients who progressed to liver fibrosis (4.2%) is significantly higher than that in overall patients (1.5%). 42 Hepatitis C virus-induced HCC is triggered by a long-term and multistep process via molecular mechanisms such as direct effects derived by the virus and indirect host immune response. 44 In particular, the hepatitis C virus core protein and sustained inflammation with cytokine production induce reactive oxygen species production, and this upregulates transforming growth factor beta, which contributes to the progression of liver fibrosis and the development of HCC. 44 Moreover, Asahina et al. 45 reported that pre-treatment is not the only factor that progressed liver fibrosis, but also aging, male sex, fatty liver, low albumin concentration at pre-treatment and high concentration of ALT and AFP at post-treatment were risk factors for the development of HCC in patients who underwent interferon antiviral therapy. In the present study, aging and male sex were not detected as risk factors by Cox proportional hazard analysis. This may be due to the differences in the patient backgrounds, such as different ages between interferon and DAA therapy studies. 46 Moreover, a retrospective study of 1922 patients who received DAA therapy showed that in addition to the pre-treatment M2BPGi concentrations, the post-treatment AFP and M2BPGi concentrations were significant risk factors. 46 Our results showed that only the pre-treatment M2BPGi concentrations after antiviral treatment in the HCC group were significantly higher than those of post-treatment, and that it was identified as a significant risk factor for the development of HCC by the univariate analysis. These results support a previous report that liver fibrosis is one of the major risk factors for the development of HCC. 46 However, the M2BPGi concentrations may be not suitable as a follow-up marker for the development of HCC after DAA treatment because M2BPGi concentrations immediately decrease following antiviral therapy, and there is no significant difference between the HCC and non-HCC groups after antiviral treatment.
Although the sGP88 concentrations in patients with chronic hepatitis C were significantly higher than those in healthy controls, there was no significant difference in the pre-treatment sGP88 concentrations among the groups who did and did not develop HCC after DAA treatment. In contrast to AST, ALT and M2BPGi, the post-treatment sGP88 concentrations in the HCC group were significantly higher than those in the non-HCC group; the high sGP88 concentrations caused by chronic hepatitis C did not decrease and were maintained.
It has been reported that the hepatitis C virus core protein and non-structural region NS5A activate β-catenin, 44 which is increased in HCC tissues and significantly correlates with GP88 expression. 47 Previous studies have demonstrated that GP88 promotes the invasion of HCC 30 and confers resistance to natural killer cell cytotoxicity. 22 Although further studies are necessary before recommending other DAA treatments, such as sofosbuvir, high concentrations of sGP88 may be one of the possible causes for the development of HCC, and the changes of the sGP88 concentrations pre- and post-treatment may be an independent risk factor that can make a difference in the cumulative incidence of HCC. Therefore, the concentrations of sGP88 post-DAA treatment may be an excellent predictor of HCC development and may be highly useful for follow-up.
In summary, sustained high concentrations of sGP88 after chronic hepatitis C treatment with DAAs may promote the development of HCC, which may be used as a predictive follow-up biomarker for HCC. Furthermore, additional studies for other treatments for chronic hepatitis C and other disorders that cause HCC, such as chronic hepatitis B or non-alcoholic steatohepatitis, should be conducted.
Conclusion
We performed a retrospective study on the sGP88 concentrations in patients treated with the DAAs asunaprevir and daclatasvir. Since we observed a correlation between the consistently high concentrations of sGP88 and the development of HCC, the sGP88 concentrations may be used as a predictive and follow-up biomarker for the development of HCC after treatment for chronic hepatitis C.
Supplemental Material
sj-pdf-1-acb-10.1177_00045632211036723 - Supplemental material for Serum GP88 as a predictive biomarker for hepatocellular carcinoma in patients with viral hepatitis C after direct-acting antiviral agents
Supplemental material, sj-pdf-1-acb-10.1177_00045632211036723 for Serum GP88 as a predictive biomarker for hepatocellular carcinoma in patients with viral hepatitis C after direct-acting antiviral agents by Hidekazu Ishida, Masao Takemura, Atsushi Suetsugu, Takafumi Naiki, Takuji Tanaka, Tomita Eiichi, Ginette Serrero, Hidetoshi Matsunami, Yasuko Yamamoto and Kuniaki Saito in Annals of Clinical Biochemistry
Footnotes
Acknowledgements
I would like to express my appreciation to Professor Hiroyasu Ito and Professor Masahito Shimizu who gave me permission to commit the Ph.D. program.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
This research was supported by Fujita Health University Graduate School.
Ethical approval
Gifu University Hospital Clinical Research Ethics Board approved this study (2018–031) and Gifu Municipal Hospital Ethics Board (27–255).
Guarantor
KS.
Contributorship
HI and TM researched literature and conceived the study. HI gained ethical approval, data analysis and wrote the first draft of the manuscript. HI, TM and YY involved in the investigation. AS, NT, ET, GS and HM involved in the supply of samples or reagents. TT involved in the pathological evaluation. KS involved as supervision and project administration. All authors reviewed and edited the manuscript and approved the final version of the manuscript.
Supplemental Material
Supplementary material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
