Abstract
Immunoassays are widely used for laboratory assessment of endocrine functions including thyroid hormones. While usually adequate for patient evaluation, they are known to potentially suffer from interference from a variety of factors. We report the case of a 44 year-old male patient without clinical symptoms of thyroid disease who presented for specialist evaluation after pathological thyroid function tests prompted a transferal by his primary care practitioner. Thyroid function tests showed discrepant results across immunoassays and platforms of different manufacturers. Polyethylene glycol precipitation prompted the diagnosis of macro-thyroid-stimulating hormone, while heterophilic and non-specific antibody blocking reagents proved ineffective in eliminating the interference in thyroid-stimulating hormone, free triiodothyronine and free thyroxine measurements. Further assessment ruled out a diagnosis of familial dysalbuminemic hyperthyroxinemia, leaving an exclusion diagnosis of manufacturer-specific interference in free triiodothyronine and free thyroxine assays due to unknown factors. Both clinicians and laboratory specialists must be aware of potential interference in immunoassays which otherwise might be misleading, potentially triggering unnecessary (invasive) follow-up procedures or therapeutic interventions. Close communication is required for successful troubleshooting. To our knowledge, no other case of both macro-thyroid-stimulating hormone and manufacturer-specific interference in a single patient has been documented thus far.
Keywords
Introduction
Immunoassays are widely used for laboratory assessment of endocrine functions including thyroid hormones. While usually adequate for patient evaluation, they are known to potentially suffer from interference due to a variety of factors. These encompass endogenous antibodies (EA) – that is, heterophilic antibodies, specific human anti-animal antibodies and autoantibodies including macro-complexes.1–3 EA usually cause false positive results in sandwich immunoassays, but they can also cause false negative results in competitive immunoassays and infrequently in sandwich immunoassays. 3 The overall incidence of immunoassay interference due to EA was estimated to be less than 2.0% but the number of potentially serious errors is still relevant because of the large quantity of performed immunoassays.1,4,5 Ismail et al. showed that 28 (i.e. 47.5%) out of 59 suspect results were indeed affected by interference. 4 Undetected interference can lead to unnecessary clinical consultations, further resource intensive testing as well as potentially harmful medical interventions. 3 While proactive approaches for detection of immunoassay interference have been concluded to be unwarranted, retroactive approaches might become necessary if immunoassay results are inconsistent with patient presentation. 6 This presents a significant challenge since erroneously normal or abnormal but not implausible results can be near impossible to detect by the laboratory, and clinical information is usually required for successful troubleshooting. 1 Here, we present a case of rare coincidence of interferences in a single patient.
Case presentation
Initial patient work-up
In January 2023, a 44-year-old male attended an endocrinology practice following a referral by his general practitioner to further investigate grossly abnormal thyroid function test results. He showed no clinical signs of thyroid disease, indicated no current complaints, and had no personal or family history of thyroid dysfunction. His past medical history was unremarkable. The patient took no medication or biotin supplements. His personal history included only rabbits as pets from 2011 to 2018. He was occupied as an information technology specialist. Clinical work-up including thyroid sonography revealed no anomalies. Laboratory tests initiated by his endocrinologist indicated subclinical hypothyroidism with a remarkably high thyroid-stimulating hormone (TSH), normal free triiodothyronine (fT3) and normal free thyroxine (fT4). An assay for human anti-mouse antibodies (HAMA) was negative. The combination of these laboratory values and the low likelihood of subclinical hypothyroidism in a young male patient prompted the endocrinologist to arrange further testing, initially under the suspicion of possible interference due to heterophilic antibodies.
First comparative laboratory testing
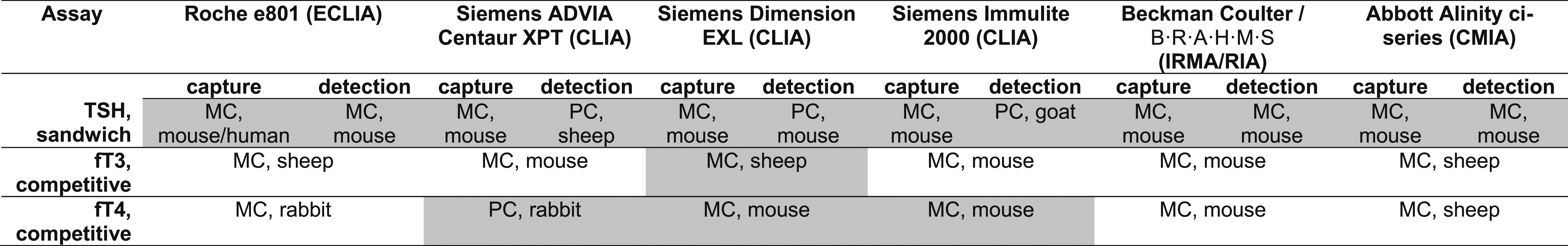
Comparison of immunoassay antibodies and platforms used in this study. Assays with suspected interference are shaded. Information about antibody origin and nature were extracted from the package inserts of the manufacturers.

CLIA: chemiluminescence immunoassay, CMIA: chemiluminescence microparticle immunoassay, ECLIA: electrochemiluminescence immunoassay, fT3: free triiodothyronine, fT4: free thyroxine, IRMA: immunoradiometric assay, MC: monoclonal, PC: polyclonal, RIA: radioimmunoassay, TSH: thyroid-stimulating hormone.
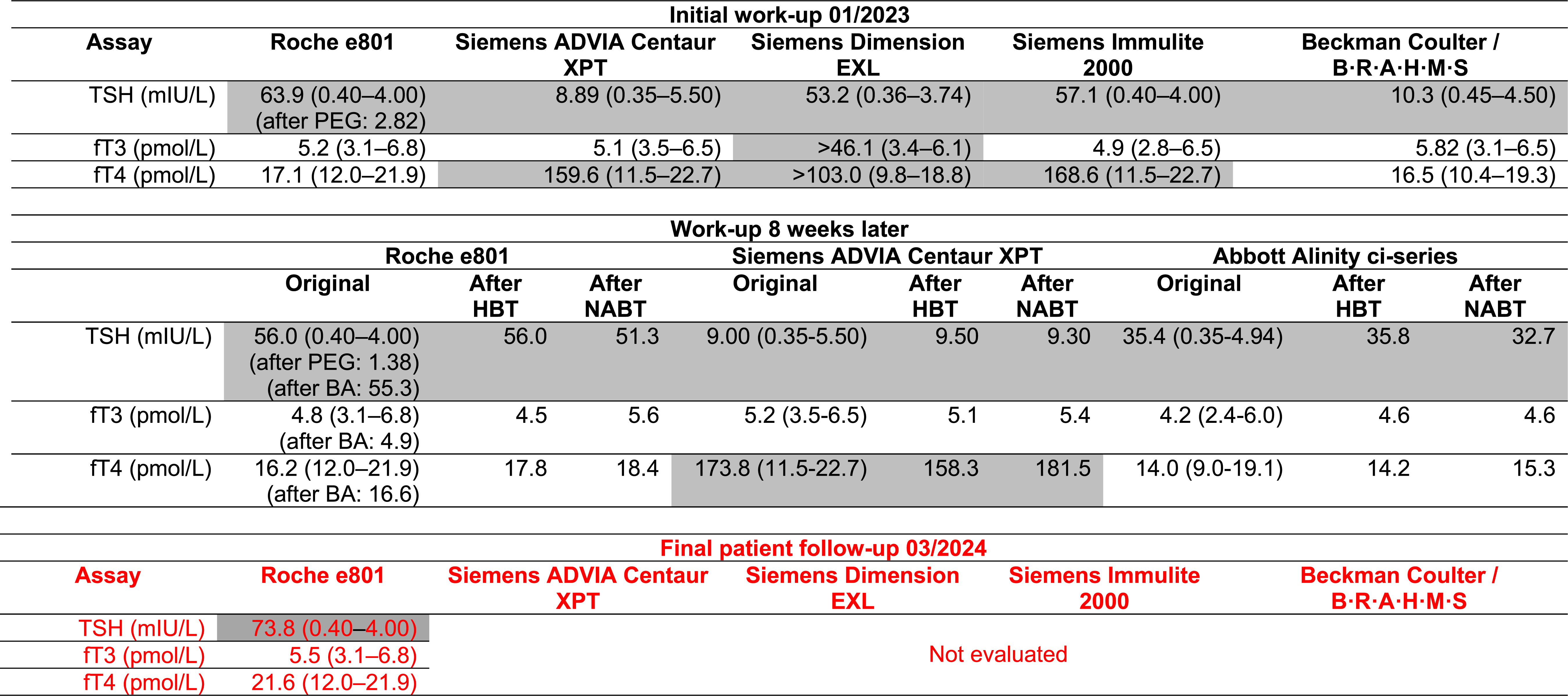
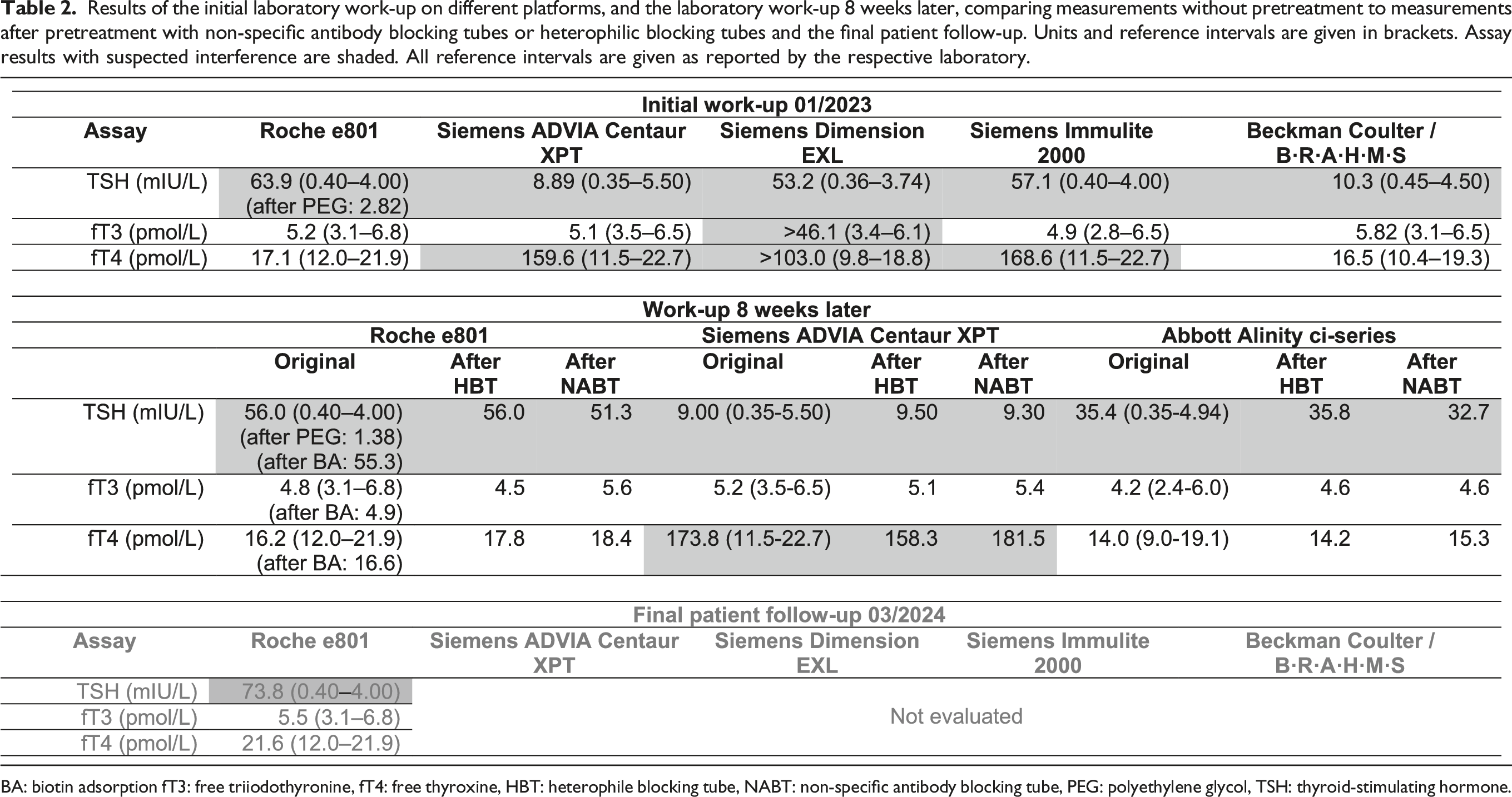
Results of the initial laboratory work-up on different platforms, and the laboratory work-up 8 weeks later, comparing measurements without pretreatment to measurements after pretreatment with non-specific antibody blocking tubes or heterophilic blocking tubes and the final patient follow-up. Units and reference intervals are given in brackets. Assay results with suspected interference are shaded. All reference intervals are given as reported by the respective laboratory.
BA: biotin adsorption fT3: free triiodothyronine, fT4: free thyroxine, HBT: heterophile blocking tube, NABT: non-specific antibody blocking tube, PEG: polyethylene glycol, TSH: thyroid-stimulating hormone.
Our laboratory is equipped with Roche platforms, and we performed additional assessments. We ruled out rheumatoid factors (7.4 kIU/L, reference interval [RI]: <14), elevated serum levels of immunoglobulin A (IgA) (1.3 g/L, RI: 0.7–4.0), immunoglobulin G (IgG) (8.7 g/L, RI: 7.0–16.0) and immunoglobulin M (IgM) (1.0 g/L, RI: 0.4–2.3) as well as haemolysis (index: 12), icterus (index: 1) and lipaemia (index: 11) as potential causes of interference. Hyperprolactinemia (0.46 nmol/L, RI: 0.20–0.92) and anti-thyroglobulin (5.5 kIU/L (RI: <100), anti-thyroid-stimulating hormone receptor (<0.80 IU/L (RI: <1.75) and anti-thyroperoxidase antibodies (9.6 kIU/L (RI: <34) were also excluded.
To investigate the possibility of macro-TSH, we decided to perform a dilution series (2-, 3-, 4-, 5- and 6-fold) with Diluent MultiAssay (Roche Deutschland Holding GmbH, Mannheim, Germany) as well as a polyethylene glycol (PEG) precipitation with PEG 6000 (concentration 250 g/L, Roche Deutschland Holding GmbH, Mannheim, Germany) according to Roche’s protocol for suspected macroprolactin. 7 The dilution series resulted in near perfect linearity. TSH measurement after PEG precipitation was 2.82 mIU/L, a recovery of 4.4% (Table 2).
Second comparative laboratory testing
Eight weeks after the initial assessment, the patient presented for his follow-up and a second survey using Roche e801, Siemens ADVIA Centaur XPT and Abbott Alinity ci-series (Abbott GmbH, Wiesbaden, Germany) was conducted.
After initial measurement, serum samples were treated with heterophilic blocking tubes (HBT) or non-specific antibody blocking tubes (NABT, both Scantibodies Laboratory, Inc., Santee, CA, USA) according to the manufacturer’s instructions. Results did not differ markedly after pretreatment with the respective reagents; however, NABT pretreatment led to some increase in fT3 and fT4 measurements (Table 2). Possible interference from circulating anti-thyroxine autoantibodies was ruled out via negative testing (qualitative assay) in an external laboratory.
Biotin adsorption according to the 1-h protocol published by us in 2020 ruled out theoretically possible (but highly unlikely) interference due to biotin as well as anti-streptavidin-antibodies (Table 2). 8
Renewed PEG precipitation in our laboratory confirmed the earlier finding with a TSH recovery of 2.5%. Follicle-stimulating hormone (FSH) (2.16 IU/L, RI: 1.00–15.0), luteinizing hormone (LH) (3.08 IU/L, RI: 1.00–12.0), beta-human chorionic gonadotropin (β-hCG) (<0.20 U/L, RI: <3.0), albumin (47 g/L, RI: 35–52) and cortisol assays (0.23 µmol/L, RI: 0.17–0.50) yielded unremarkable results.
Due to the interference in fT3 and/or fT4 measurements across Siemens platforms and immunoassays as well as the apparent elimination of more likely causes, the added diagnosis of familial dysalbuminemic hyperthyroxinemia (FDH) was suspected. However, genetic investigation via direct sequencing of the albumin gene did not reveal any pathogenic gene variant or known missense mutation associated with FDH.
After this consultation, the patient received an international emergency certificate detailing the findings of macro-TSH and manufacturer-specific interference in fT3 and fT4 assays.
Final patient follow-up
During a telephonic follow-up in November 2023, the patient did not have any complaints. In March 2024, a final consultation revealed no clinical signs of thyroid dysfunction. The patient continued to be without complaints. Measurements of fT3 and fT4 via Roche ECLIA indicated an euthyroid state, while TSH results remained consistent with macro-TSH (Table 2).
Discussion
Numerous ways to identify and investigate putative immunoassay interferences and aberrant TFTs have been proposed. These include general consideration of the clinical context (e.g. pregnancy, medications, etc.), alternative measurement methods/instruments, sample dilution, PEG precipitation, antibody blocking tubes, gel filtration chromatography, equilibrium dialysis and nonsense sandwich immunoassays.3,9,10
Commonly, measurement with an alternative method/instrument should eliminate the interference if interfering EA are causative. Additionally, a dilution series will reveal a nonlinear relationship in up to 60% of EA interferences including macro-complexes.4,11,12
In our case, the dilution series of TSH produced near perfect linearity similar to findings of Hattori et al. in some patients with macro-TSH. 13 However, PEG precipitation repeatedly led to a <5.0% recovery while different platforms produced highly variable but elevated TSH results. Both findings were consistent with the observations of other authors.14–16 The differently elevated TSH results across platforms were likely caused by varied epitopes used for assay design.14,16 Pretreatments with HBT and NABT did not relevantly alter the results and interference due to heterophilic antibodies across multiple platforms is exceedingly rare while HAMA were ruled out by the initial laboratory.4,11,14 Therefore, we diagnosed the patient with macro-TSH. An additional evaluation of TSH using gel filtration chromatography or protein A/G columns would have been desirable but was not available to us.9,10
Macro-TSH is an autoimmune complex of >150 kDa molecular mass with low bioactivity. 17 The associated anti-TSH is usually of IgG type but a case of IgA paraprotein associated macro-TSH has also been reported in 2022.12,14,15,18 Recently, a case of both macro-TSH and macro-LH possibly due to autoantibodies against the common α-subunit shared by β-hCG, FSH, LH and TSH was outlined. 17 However, β-HCG, FSH and LH were within the reference ranges in our patient, suggesting antibodies targeting the specific β-subunit of TSH. The prevalence of macro-TSH was estimated to be 0.6%–1.6% in patients with suspected subclinical hypothyroidism.13,16,18
While recommended dilution series in suspected EA interference do not consistently work and are not applicable in some assays (e.g. fT3 and fT4), PEG precipitation comes with its own shortcomings and is known to potentially produce inconclusive results (e.g. in macroprolactinemia), or interfere with immunoassays by itself sometimes.2,3,19 Similarly, blocking substances can be ineffectual in 20%–30% of samples showing interference due to EA and might fail to eliminate the interference completely as demonstrated by García-González et al. Additionally, HBT pretreatment can cause spuriously high recovery of hormones in some assays.3,19,20
As it is unlikely that all approaches fail simultaneously, a combination of available options is usually required to successfully evaluate potential EA interference.9,11
Since fT3 and/or fT4 results on Siemens platforms also appeared to suffer from interference, while none of the other platforms revealed values outside the respective reference ranges, we investigated the added possibility of FDH.
FDH has a reported prevalence of 0.01%–1.8% depending on ethnicity. 21 Inconsistent interference in fT4 and/or fT3 immunoassays across platforms is the trademark of this condition and was observed by other authors.22–24 However, contrary to the results of Khoo et al. and Zhao et al., we did not observe any interference in fT4 measurements performed on Roche, Beckman Coulter or Abbott modules, while we also detected major interference on Siemens platforms.24,25
Genetic investigation revealed no pathologic variant of the albumin gene or any of the known FDH mutations which alter the equilibrium between free and albumin-bound thyroid hormones during laboratory investigation with some immunoassays. Additionally, both serum albumin and cortisol were quantitatively unremarkable.21–23,26
Therefore, we hypothesized that the patient suffers from manufacturer-specific interference. This might be due to heterophilic antibodies to a common but unknown factor found in all three fT4 assays of the Siemens platforms and the fT3 assay of the Siemens Dimension EXL used in this case. Apparently, both HBT and NABT tubes failed to eliminate this interference as described by García-Gonzáles et al. 3 Dilution series and PEG precipitation are not applicable to free thyroid hormone assays due to the equilibrium shift between free and protein-bound hormones. Equilibrium dialysis or protein A/G columns would have been ideal complements for the evaluation of EA interference in fT3 and fT4 assays but were unavailable to us. 9
Unfortunately, package inserts supplied by Siemens did not reveal any specific substance not found within at least some of the other assays employed in this study. Therefore, the causative reagent remains elusive.
Since the combination of macro-TSH and manufacturer-specific interference in a single patient is exceedingly rare, both were recorded in the patient’s medical file as well as in his medical emergency ID to avoid unnecessary future work-ups and potential interventions unless clinical signs of thyroid disease are present.
This case illustrates some of the intricacies of immunoassay interference and the necessity of close communication and cooperation between clinical and laboratory specialists to investigate possible interference in order to avoid potentially harmful outcomes for patients.
A limitation of our case report is that the evidence for heterophilic antibody interference on the Siemens platforms remains circumstantial and must be regarded as a diagnosis by exclusion, especially since the direct sequencing of the albumin gene might only detect about 70% of FDH cases. However, since none of the other platforms showed interference in fT3 and fT4 measurements even though other authors observed such in cases of FDH, we believe this differential diagnosis to be sufficiently unlikely.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethical approval
The patient gave verbal and written informed consent to present this case.
Guarantor
VD.
Contributorship
IM did the literature review and drafted the paper. VD participated in patient care. RK, MU, SS, JRI and MB contributed to the laboratory work-ups. All authors reviewed and edited the manuscript and approved its final version.