Abstract
The recent
Keywords
Introduction
Biotin, also referred to as vitamin B7, is a water-soluble vitamin that is essential for the metabolism of proteins, fats, carbohydrates and glucose. Biotin functions as a coenzyme involved in the transfer of carboxyl groups by five carboxylases that are important in key metabolic pathways.
An adequate intake of biotin is thought to be 30–70 μg/day for an adult, with some increase in the requirement during pregnancy. Biotin deficiency in healthy individuals is believed to be rare as a typical diet contains sufficient biotin for normal requirements. 1 However, biotin deficiency can occur in some individuals; risk factors for deficiency include some inherited metabolic diseases (listed below), long-term parental nutrition, anticonvulsant or protein pump inhibitor medication, Crohn’s disease, alcoholism and the consumption of large quantities of raw eggs (as avidin in the eggs binds biotin; cooking eggs denaturates the avidin removing this effect). The signs and symptoms of deficiency can include disturbances in metabolism, muscle pains, fatigue and defects in the hair, nails and skin.1,2
When prescribed, the biotin doses given range between 5 and 300 mg per day. Biotin may be used to treat several rare inherited metabolic diseases, with typical biotin doses of: Biotinidase deficiency 5–10 mg/day, holocarboxylase synthetase deficiency 30–40 mg/day, biotin-thiamine-responsive basal ganglia disease 100–300 mg/day and as a novel therapy in multiple sclerosis (MS) 300 mg/day.3–5
Even at high doses, the toxicity of biotin appears to be very low, with no or few reports of adverse effects of biotin supplementation, and hence no upper safe limit of dosage has been established, except perhaps for caution in taking very high doses of biotin in pregnancy. 1
Outside of clinical use, biotin is available in multivitamin preparations, typically at doses of 30–300
In recent years, laboratories have become aware of the potential for biotin interference to cause unexpected assay results, one example being in thyroid function test results, where the typical pattern of interference seen is low TSH results, high FT4, FT3 and TRAb results (see the Case study section).4–9 However, individuals taking biotin and many health-care providers are likely to be unware of the potential for biotin interference in some immunoassays.
Case study
A 55-year-old man with MS was referred to a hospital thyroid unit because of markedly elevated FT4 and FT3 results, and low (apparently suppressed) levels of TSH as these results suggested that he had hyperthyroidism. The ‘TSH receptor binding inhibiting antibody’ concentration was also elevated. Clinically, the patient did not appear to have hyperthyroidism and an Iodine-123 thyroid scan appeared normal. As the biochemical results were discordant with the patient’s clinical assessment, investigations for assay interference from drugs or other compounds were conducted. The patient had been receiving 300 mg/day of biotin as a treatment for his MS. The patient stopped taking biotin temporarily, and two weeks later, thyroid function tests showed completely normal results. Treatment with biotin was then restarted. 6
Recently, there have been increasing numbers of case reports and studies published linking biotin use and interference with many analytes, not just with thyroid function tests, for example troponin, NT-proBNP, cortisol and 25OH-vitamin D. These studies have helped further describe the risk to patient care and the actions the laboratory can take to mitigate this risk.4,5,10–13
The concentration of biotin at which biotin interference is seen varies between immunoassays. For some immunoassays, interference can occur at biotin concentrations as low as 10 ng/mL (40.9 nmol/L – to convert ng/mL to nmol/L, multiply by 4.09). 2 For other immunoassays, interference may only occur at concentrations of 200 ng/mL of biotin (818 nmol/L) or more.12,13
The use of biotin and streptavidin in some immunoassay methodologies
The binding of biotin to streptavidin is a useful reaction, used in some immunoassay formats, as it allows the rapid separation of the analyte or assay component from the sample. It is relatively simple to conjugate a small biotin molecule (C10H16N2O3S molecular weight 244.31) to antibodies or analyte (for competitive immunoassays) with minimum effect on the reaction dynamics of the biotinylated molecules. The biotinylated molecule can then be captured on a streptavidin microparticle or other solid phase, as streptavidin has a high affinity for biotin and a tightly bound complex of streptavidin–biotin can be rapidly formed. This technique is used to separate the analyte from the sample in many immunoassays.
Some immunoassays that use biotin may use biotin only as a structural component of either the microparticle or conjugate to join assay components together. These immunoassays do not use streptavidin–biotin binding to capture the analyte, and biotin in the patient sample will not interfere in the assay.12,13
It should be noted that excess biotin in the sample does not interfere with all immunoassay formats because some immunoassays do not use streptavidin–biotin binding at all (no biotin or streptavidin is in the reagents). These immunoassays use antibodies (sometimes analyte) conjugated to the microparticle to separate the analyte from the sample.
However, many immunoassays do use streptavidin–biotin binding in their assay formats. A recent review of 374 immunoassays in the USA revealed that the majority (221/374, 59%) of the surveyed immunoassays used biotin in the assay format and that nearly a quarter (82/374, 22%) of the immunoassays were at ‘high risk’ of interference from biotin (the authors deemed this to be where biotin interfered at a concentration of <51 ng/mL). 14
Mechanism of interference
All immunoassays that use streptavidin–biotin binding as part of the assay reaction are thought to be susceptible to interference from excess biotin in patient samples.5,12,13,15
When biotin–streptavidin binding is used as part of a ‘sandwich’ immunoassay format excess biotin in the sample can block the binding of biotinylated antibodies to the biotin binding sites on the streptavidin-coated solid phase, resulting in falsely low results. In contrast, for ‘competitive’ immunoassays, excess biotin in the specimen can block the binding of biotinylated analyte to the biotin binding sites on the streptavidin-coated solid phase, resulting in falsely high results.12,13 Hence, the mechanism of interference is quite simple and relates to loss of signal in the immunoassay because biotin in the patient sample bindings to, and blocks, biotinylated reagents being captured by the biotin binding sites on the streptavidin-coated solid phase, see Figure 1.
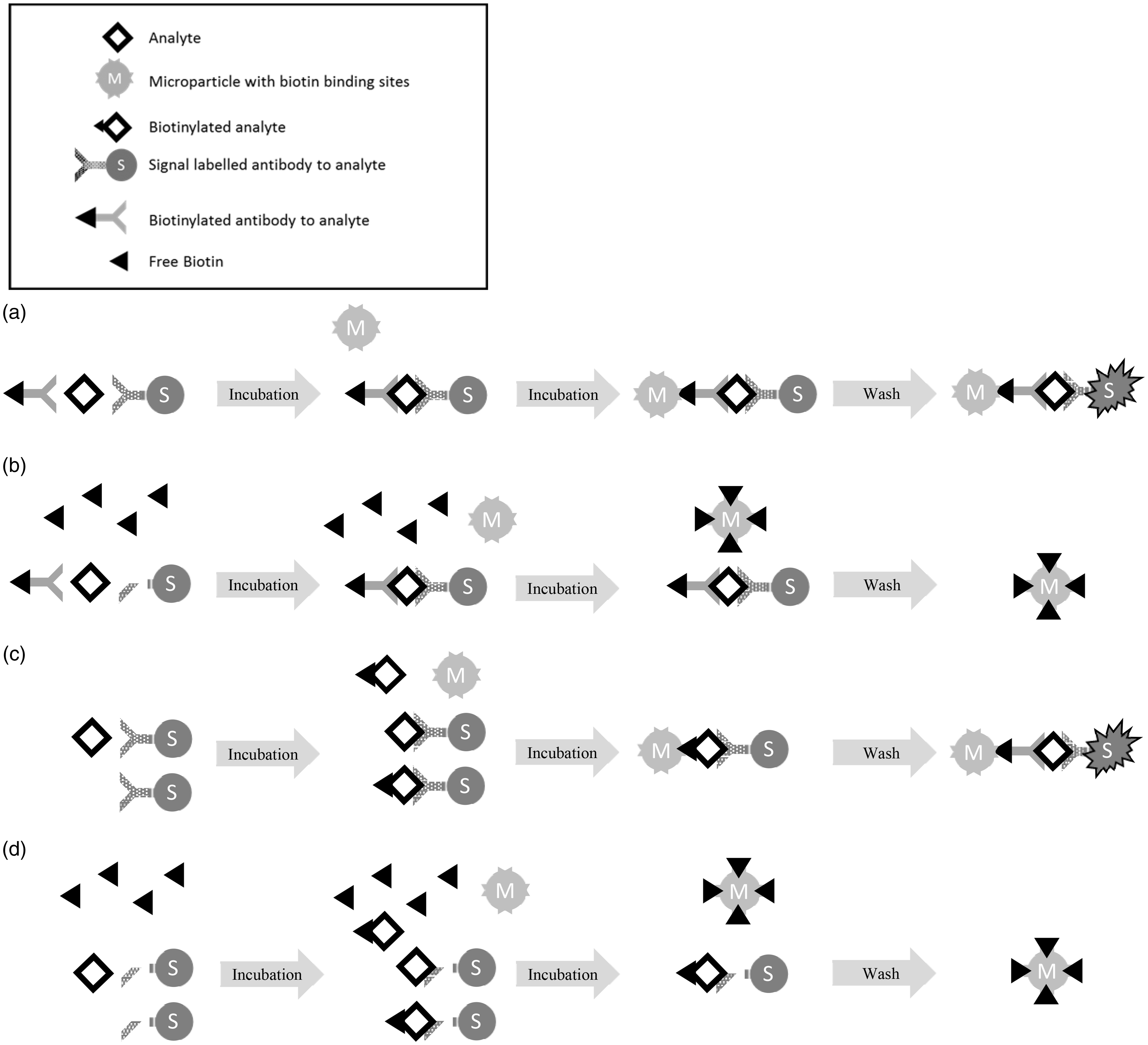
The mechanism of biotin interference in immunoassay. (a) One-step sandwich assay using free capture biotin-streptavidin binding in the absence of biotin in the sample, e.g. TSH: In a typical sandwich immunoassay, in the absence of excess biotin, the analyte in the sample is bound by two antibodies (one biotinylated). Then streptavidin-coated microparticles are added, which captures the antibody-analyte sandwich. Next, any unbound material is washed away, and then signal is generated which is proportional to the analyte concentration. (b) One-step sandwich assay using free capture biotin-streptavidin binding in the presence of biotin in the sample, e.g. TSH: In the presence of biotin in the sample, some of the biotin-binding sites on the streptavidin-coated microparticles are blocked leading to loss of the biotinylated antibody-analyte sandwich in the wash step and a loss of signal resulting in falsely low results. (c) Competitive immunoassay using free capture biotin-streptavidin binding in the absence of biotin in the sample, e.g. FT4, FT3: In a typical competitive immunoassay, in the absence of excess biotin, a single antibody is added and binds to the analyte. Then biotinylated-analyte and streptavidin-coated microparticles are added, the biotinylated-analyte binds to vacant binding sites on the antibody and the complex is captured by the streptavidin-coated microparticle. Next any unbound material is washed away, and then signal is generated which is inversely proportional to the analyte concentration. (d) Competitive immunoassay using free capture biotin- streptavidin binding in the presence of biotin in the sample, e.g. FT4, FT3: In the presence of biotin in the sample, some of the biotin binding sites on the streptavidin-coated microparticle are blocked leading to loss of the biotinylated antibody-analyte sandwich and a loss of signal resulting in falsely high results.
Which immunoassays are affected, in what circumstances and by how much?
Several recent papers have investigated biotin interference with streptavidin–biotin binding immunoassays by spiking samples with biotin to the concentrations expected to be seen in patients receiving moderate (5–10 mg/day) and very high doses (up to 300 mg/day) of biotin. Interference correlated with biotin concentration and could be reduced by absorbing biotin with a pretreatment step using streptavidin microparticles.11,15–17
A recent study looked at the immunoassays available on one analyser and found that at biotin concentrations >500 ng/mL (2045 nmol/L) all of the immunoassays tested showed significant interference from biotin, but the magnitude of interference was variable. Some immunoassays, such as hsTroponin T, TSH and anti-thyroid antibodies showed interference at relatively low biotin concentrations (15.6 and 31.3 ng/mL, 63.8 and 128.0 nmol/L, respectively), and that these concentrations of biotin could be reached and surpassed by, respectively 5 mg, 10 mg or higher biotin doses.3,15,18
UKNEQAS in August 2018 performed spiking experiments in the Steroid, Thyroid, Peptide and Cardiac Schemes using high concentration spikes of 750 and 1500 ng/mL of biotin. These biotin spikes resulted in falsely low or falsely high results (including < LOD or above the upper limit of the measuring interval) for many of the assays using streptavidin–biotin binding as part of the assay format; no interference was seen with assays that did not use streptavidin–biotin binding.
The clinical significance of biotin interference depends on three factors: (1) the susceptibility of an immunoassay to biotin interference (e.g. does a low concentration of biotin cause interference), (2) the prevalence of biotin at that concentration and (3) the consequences of a change in the result, which depends on the analyte. Analytes where incorrect results might be considered as having a high risk of leading to errors in patient management include hsTroponin, NT-proBNP and procalcitonin where ‘false negative’ results might be easy to miss, and could lead to patients not receiving appropriate management. For troponin, an interesting overview of how to investigate troponin values that are questioned, including when there is a possibility of biotin interference, has recently been published. 19
While little information has been published to date on the possibility of interference with immunoassays for infectious diseases, it is possible that immunoassays for HIV and hepatitis, for example, could be affected by biotin interference as streptavidin–biotin binding is used in some of these immunoassays and interference might lead to a false-negative result and a missed or incorrect diagnosis. 20
A short listing of immunoassays in which biotin interference may be critical from a clinical standpoint is detailed in Table 1.
Short list of immunoassays where results may be misleading if results are falsely decreased or increased by biotin interference. Effect direction based on typical assay format, this may differ for specific immunoassays. The magnitude of change and the susceptibility of assays to interference are highly variable.
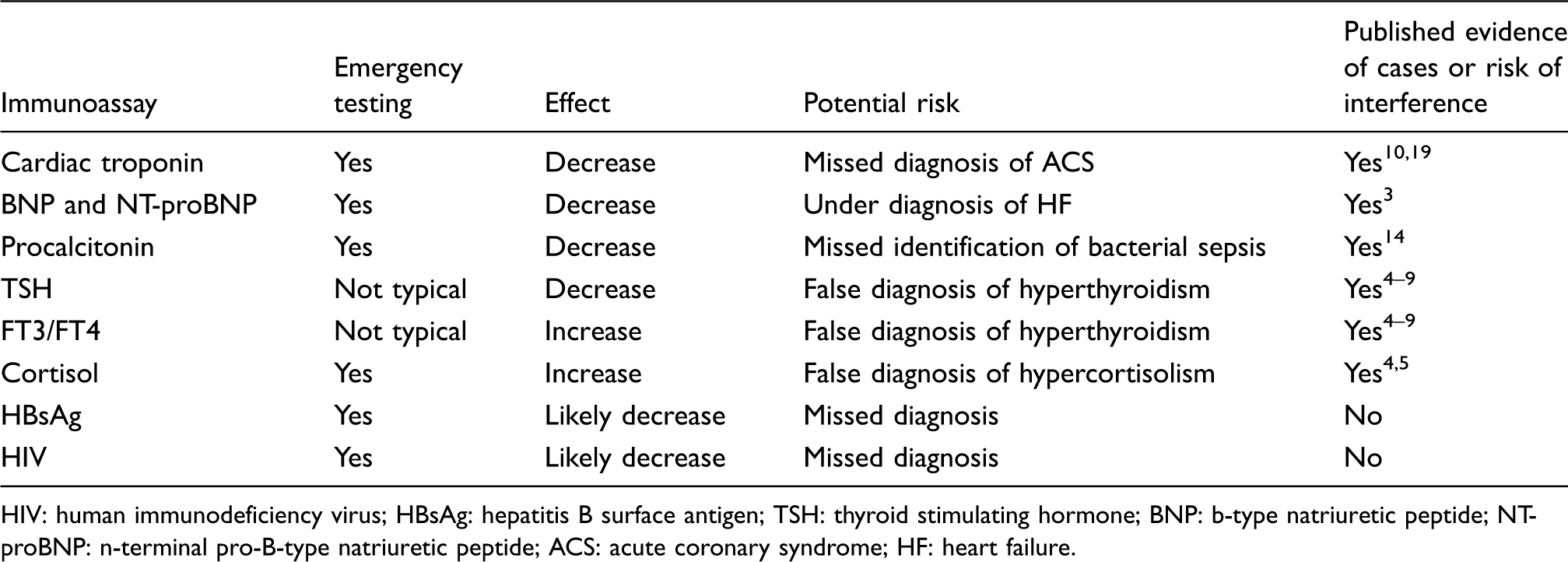
HIV: human immunodeficiency virus; HBsAg: hepatitis B surface antigen; TSH: thyroid stimulating hormone; BNP: b-type natriuretic peptide; NT-proBNP: n-terminal pro-B-type natriuretic peptide; ACS: acute coronary syndrome; HF: heart failure.
The kinetics of biotin in the blood
Biotin is rapidly absorbed from the gastrointestinal tract, with peak serum levels of biotin typically occurring 1 to 3 h after ingestion. Only a limited amount of catabolism of biotin occurs, with excess biotin rapidly appearing in urine. The metabolites of biotin are thought also to bind to streptavidin, albeit with lower affinity than biotin. There is some possibility that biotin metabolites contribute to assay interference, but may not be measured in LC-MS/MS or immunoassays for biotin; hence, biotin-spiking experiments may not fully reflect the interference caused by
With a normal dietary intake of biotin, the biotin concentration in serum or plasma may typically be between 0.1 and 0.8 ng/mL (0.409–3.27 nmol/L).18,21
Biotin doses of 5–10 mg may result in peak levels of biotin in the blood of 30 ng/mL (120 nmo/L), a biotin dose of 20 mg may result in peak levels of 200 ng/mL (818 nmol/L) or more and a biotin mega-dose of 300 mg may give a peak level of 1200 ng/mL (4908 nmol/L) or more, approximate peak serum biotin levels after a single biotin dose are shown in Figure 2.10,13 Peak levels will vary between patients, depending on renal function, frequency of dosing and normal inter-individual variation. Serum biotin concentrations, in theory, will reach a plateau after a few days of constant doses. In some studies, and not in others, higher peak concentrations of biotin after repeated doses over several days have been found.18,22
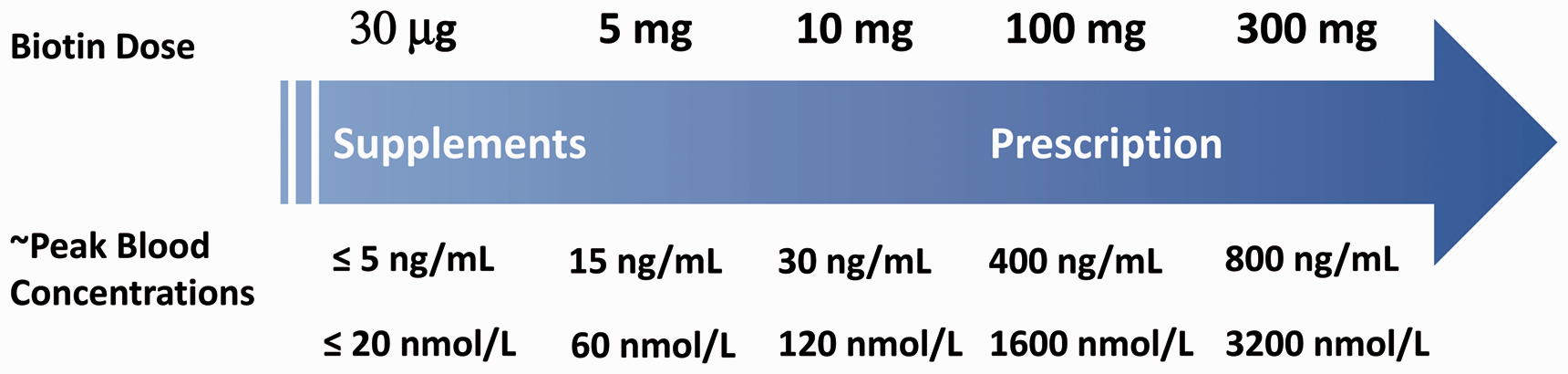
Schematic of approximate peak serum concentrations after a single dose of biotin.
Studies of the pharmacokinetics of biotin suggest in healthy individuals a median half-life of approximately 15 h. When a patient has renal impairment, it is thought that the half-life and peak biotin concentrations increase in comparison to the half-life and peak biotin concentrations seen in healthy individuals taking the same dosing of biotin.5,21
The ACB Scientific committee has suggested that assays with an interference threshold <30 ng/mL (120 nmol/L) of biotin could be classified as more susceptible to interference, as this corresponds to the expected peak serum biotin concentration from OTC supplements of 5–10 mg. 23 Other authors have suggested that biotin concentrations as low as 10 ng/mL may cause interference in the most susceptible immunoassays. 21
A study using healthy individuals found that the peak levels of biotin reached, and the time for biotin concentrations to fall below 30 ng/mL (122.7 nmol/L) was highly dependent on the dose taken and the frequency of dosage (the study investigated dosing from once a day to four times a day). The longest time for the serum biotin concentration to fall below 30 ng/mL (128.7 nmol/L) was 31 h for the highest tested dose, 20 mg taken four times a day, and for the serum biotin concentration to fall to 10 ng/mL (40.9 nmol/L) in these individuals took 73 h. For biotin regimens of ≤10 mg taken four times a day, the serum biotin levels were below 30 ng/mL (122.7 nmol/L) 8 h after the last dose. 18
In contrast where studies have looked at the duration of interference seen
A recent study surveyed an outpatient population for biotin use using a questionnaire and quantified biotin, by an LC-MS/MS methodology, in plasma samples collected from patients presenting to an emergency department (ED). The study reported for the returned surveys (
If a patient is known to have taken a significant (typically 5 mg or more) dose of biotin, then a delay before taking blood for testing with assays susceptible to biotin interference may be necessary to avoid inaccurate results. Some manufacturers recommend a delay of 8 h before testing after a significant (5 mg) biotin dose, but literature sources and some guidelines suggest that a delay before testing of longer than 8 h is needed; the delay may need to be one day, two days or even longer.5,8,18,21 Indeed, the FDA stated, in their 2017 Biotin Safety Communication, that there is insufficient data to recommend a safe interval to delay testing after a biotin dose to obviate biotin interference. 10
Pretreatment techniques to identify biotin interference
Several publications have now reported techniques which use streptavidin microparticles, obtained as excess reagent from assay kits, to absorb and remove excess biotin from the sample as a pretreatment step before immunoassay testing. This pretreatment can involve up to a 1 h incubation of the sample with the streptavidin microparticles. This technique can be used to confirm the presence of biotin interference, and some studies report that these techniques can be used to report results that are free of biotin interference.11,16,17 However, it would be up to each laboratory to validate the effectiveness of these techniques and to decide if patient results could be safely reported.
Recently, the Clinical Laboratory Standards Institute (CLSI) has revised its testing requirements for biotin interference. The CLSI publishes guidelines for Interference Testing (CLSI EP07-A3 and CLSI EP37-A). CLSI EP07 provides study design, data analysis and labelling recommendations. CLSI EP37 provides tables of recommended concentrations to test for potentially interfering substances. CLSI EP-37 has been changed recently to recommend that testing for biotin interference is now performed by manufacturers at biotin concentrations up to 3510 ng/mL (14,356 nmol/L); this is a change from the previously recommended testing at 7.5 ng/mL (30.7 nmol/L) of biotin.24,25
Summary
In November 2017, the FDA released the ‘
The advice given by the ACB Scientific Committee and the FDA which should be considered by laboratories, includes10,23:
Educating health-care professionals (HCPs) of the potential for biotin interference and encouraging them to ask patients if they are taking any prescribed or non-prescribed supplements containing biotin and then to alert the laboratory. Manufacturers should make information describing the interference of biotin with their immunoassays available, including the concentration of biotin at which interference starts to occur and whether interference is likely to be negative or positive (falsely low or falsely high results). Ideally, manufacturers should try and build more robustness to biotin interference into their assay design. The laboratory should have a list available for internal use (and perhaps for external review by HCPs or national laboratory regulatory bodies such as UKAS or INAB, or used in comments on patient results) of immunoassays that might be susceptible to biotin interference, and ideally, the concentrations of biotin that are likely to have a significant impact on results. That the currently available data are insufficient to support recommendations for safe testing using affected tests in patients taking high levels of biotin, including about the length of time for biotin to clear from the blood.
10
To consider the possibility of biotin interference as one of the possible causes of immunoassays results that are discrepant with each other or with clinical findings. The laboratory having contingency plans for obtaining biotin interference free results, either from immunoassays not affected by biotin interference, or by having a validated procedure to remove biotin interference.
Footnotes
Acknowledgements
This article was prepared at the invitation of the Clinical Sciences Reviews Committee of the Association for Clinical Biochemistry and Laboratory Medicine. The author would like to thank Dr Claudio Galli and Dr Randal Schneider, Abbott Laboratories, for their advice and comments.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: GA is a current employee of Abbott Laboratories and holds Abbott stock.
Ethical approval
Not applicable.
Guarantor
GA.
Contributorship
GA.
