Abstract
Background
Biotin interference in biotin-streptavidin-based immunoassays is increasingly reported due to individuals taking biotin-containing supplements and patients prescribed biotin. The reported prevalence of serum biotin above the lowest threshold (≥10
Methods
Biotin was measured in 524 anonymized surplus serum samples in which at least one immunoassay test had been requested.
Results
The median (95% confidence intervals) for serum biotin was 0.27
Conclusions
These data indicate that the probability of biotin immunoassay interference in our patient population is extremely low, with the exception of assays reporting the lowest interference thresholds (e.g. Ortho Troponin I assay [threshold ≥2.5
Keywords
Introduction
Immunoassays utilizing the streptavidin–biotin interaction in the capture of antibodies to the solid phase may be subject to biotin interference. In brief, the biotinylated antibody–analyte complex is captured on a streptavidin microparticle or other solid phase, as streptavidin has a high affinity for biotin. The rapidly formed tightly bound complex of streptavidin–biotin is then used to separate the analyte from the sample. Excess biotin in the sample, however, may occupy binding sites on the microparticle and the resultant loss of signal in the immunoassay gives falsely low or falsely high results in sandwich and competitive immunoassays, respectively.
Biotin immunoassay interferences are a potential problem in patients with high serum biotin concentrations, resulting from an increased use of high dose (5–20 mg) biotin-containing supplements marketed as beauty products and recent trials reporting benefits of very high-dose biotin (300 mg) in patients with multiple sclerosis.
1
The magnitude of biotin interference is dependent on the serum biotin concentration as well as the specific immunoassay type and design. The lowest interference thresholds for some of the common manufacturers include 10
The prevalence of high serum biotin concentrations (≥10
Materials and methods
In a six-month period between January and June 2019, surplus patient serum samples in which at least one immunoassay test had been requested were identified in nine batches to assess the local prevalence of potential biotin immunoassay interference.
Samples were anonymized, stored at 4°C and analysed in duplicate within three days of sample collection for biotin using the IDK® Biotin ELISA (Immunodiagnostik (IDK), AG, Bensheim, Germany). ELISA plates were read using a TECAN Sunrise Absorbance Reader and data analysed using Magellan software (Tecan, Austria, GmbH). The measurement range of the biotin assay is 0.048–1.1
Normality of data distribution was assessed using Q-Q and P-P plots using IBM SPSS Statistics for Windows, version 25 (IBM Corp., Armonk, N.Y., USA). Biotin data were non-parametric and therefore expressed as medians with 95% confidence intervals in parentheses.
Results
Biotin concentrations were measured in 524 patient samples. The median (95% confidence limits) serum biotin concentration was 0.27
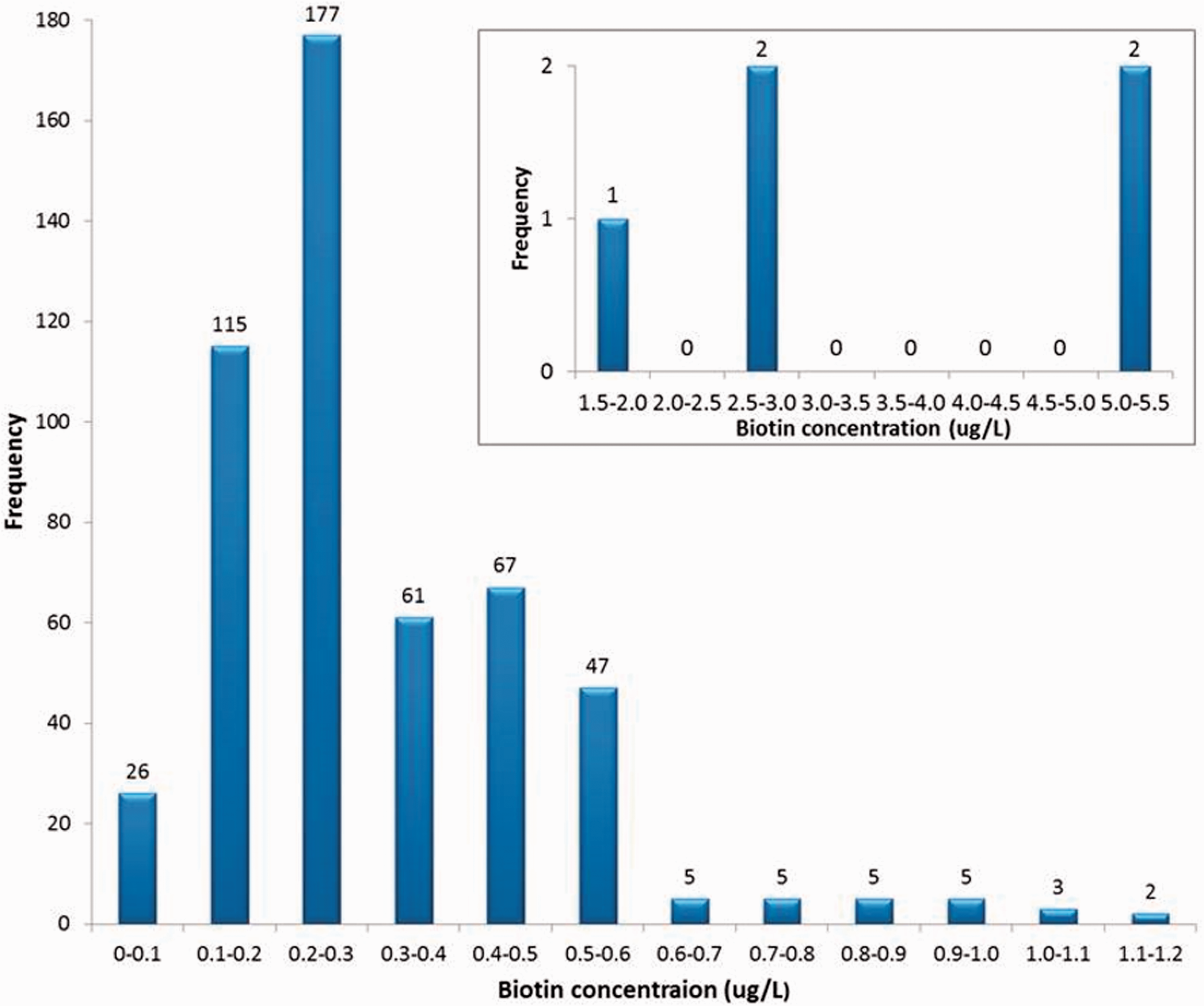
Distribution of biotin concentrations in the 524 serum samples tested.
Discussion
The extremely low prevalence (<0.2%) of high serum biotin (≥10
In healthy subjects, mean (range) plasma biotin concentrations are 0.19 (0.12–0.32)
It is reassuring that none of the samples tested contained biotin at concentrations known to interfere in the Roche immunoassays which are currently used in our laboratory. It is, however, important to note that 4/524 (0.8%) samples contained biotin at concentrations above 2.5
The FDA Biotin (Vitamin B7): Safety Communication – May Interfere with Lab Tests 7 and a statement from the ACB Scientific Committee regarding biotin/vitamin B7 interference in immunoassays 8 have raised laboratory awareness regarding potential biotin interference in specific immunoassays and provided patient education and advice to health-care providers and manufacturers. Manufacturers, such as Roche, have already begun re-issuing kits with higher thresholds for biotin interference. In suspected biotin interference, a simple protocol using left-over streptavidin beads from assay kits to absorb and remove excess biotin from the sample as a pre-treatment step before immunoassay testing, is a relatively quick and cheap means of neutralizing any potential interference and may be used to identify interference, and if present report results that are free of biotin interference. 1 , 9 In our population, the rare cases of possible biotin immunoassay interference will require good clinician–laboratory communication to recognize discordance between clinical features and immunoassay results which may then be identified by measurement on a non-streptavidin–biotin assay, and if present confirmed to be due to biotin by preanalytical treatment with streptavidin beads as described above.
In summary, our data indicate that the probability of biotin immunoassay interference in our population is extremely low. Clinicians and laboratorians, however, must remain vigilant to detect erroneous laboratory results resulting from interference in immunoassays, including that due to biotin.
Footnotes
Acknowledgements
We thank Birmingham NEQAS for providing the biotin ELISA kits for this study, and Dr T Kalaria for statistical advice.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethical approval
Not applicable.
Guarantor
PM.
Contributorship
AS and PM conceived and designed the study. AS researched the literature, collected and analysed samples, analysed the data and wrote the first draft. RG, HA and PM contributed to data interpretation. All authors critically reviewed, revised and approved the final version of the article.
