Abstract
This case report describes the positive interference of the commonly used skin protective barrier cream used together with urine collection bags on the benzethonium chloride method for urine protein measurements in a 6-month-old female baby, leading to falsely elevated results. The interference was identified by both artificially mixing urine samples with this cream and comparing the results obtained using the benzethonium chloride method with those obtained using the pyrogallol red method.
Introduction
Urine protein measurement is an essential diagnostic tool for monitoring renal function and disease. Among the numerous methods available for urine protein quantification, the benzethonium chloride (BTC) method has gained considerable attention due to its simplicity, rapidity, and cost-effectiveness. 1 This colorimetric method is based on the formation of an insoluble complex between benzethonium chloride and urinary proteins, which can be measured spectrophotometrically at 630 nm. 2 However, the accuracy of the BTC method can be affected by the presence of interfering substances (levodopa, methyldopa, Na2-cefoxitin, organically bound iodine from radiopaque media, high levels of homogentisic acid, and administration of gelatin-based plasma replacements) as is mentioned in the kit insert.3–5
Urine collection bags are commonly used to get urine samples from babies. Barrier cream (BC) is a topical formulation which is frequently used to protect the sensitive skin of infants’ nappy area. This case report describes the positive interference of BC in the quantitative determination of urine protein by the BTC method in a 6-month-old girl suspected of having a congenital nephrotic syndrome (CNS). This case report for the first time highlights the impact of BC interference on the accuracy of urine protein measurements using the BTC method and emphasizes the importance of considering such interference when interpreting results.
Case presentation
A 6-month-old girl diagnosed with PMM2-CDG, a congenital disorder of glycosylation caused by phosphomannomutase 2 deficiency, was evaluated for renal abnormalities known to occur in this disease, especially nephrotic syndrome.
6
At 5 months old, her urine protein level was 0.7 g/L with a protein-to-creatinine ratio of 7730 mg/mmol creatinine (reference interval; <80 mg/mmol creatinine for 0.2–0.5 year ages), and her plasma albumin level was 30 g/L (reference interval; 25–46 g/L for 15 days–1 year age). At a specialized CDG centre a week later, her plasma albumin level decreased to 26 g/L and urine protein was not measured. The decreasing albumin level suggested she could be developing a nephrotic syndrome. At 6 months old, during a follow-up appointment with the nephrology department, a urine dipstick test was performed for the first time. Initial measurements using urine strips (Clinitek Status + Analyser, Siemens Healthcare Diagnostics) showed negative results for urine protein, while a quantitative determination using a commercial assay (Roche Diagnostics) based on the BTC method in a Roche Cobas 8000 c502 analyser (Roche Diagnostics, Hoffmann) revealed high protein levels (3.33 g/L, reference interval; <0.14). To evaluate this elevated protein concentration, immunometric urine albumin (Roche Diagnostics) and beta-2 microglobulin measurements (Roche Diagnostics) were performed. Although those levels were increased (119.41 mg/mmol creatinine (reference interval <3.5) and 0.25 mg/mmol creatinine (reference interval <0.030), respectively), these concentrations could not explain the high total protein concentration in urine. Furthermore, urine protein electrophoresis (Hydrasys 2 system, Sebia) showed no visible band for any protein type in the urine sample (Figure 1). Urine protein electrophoresis of patient sample (lines 34 and 35) and positive control (line 36).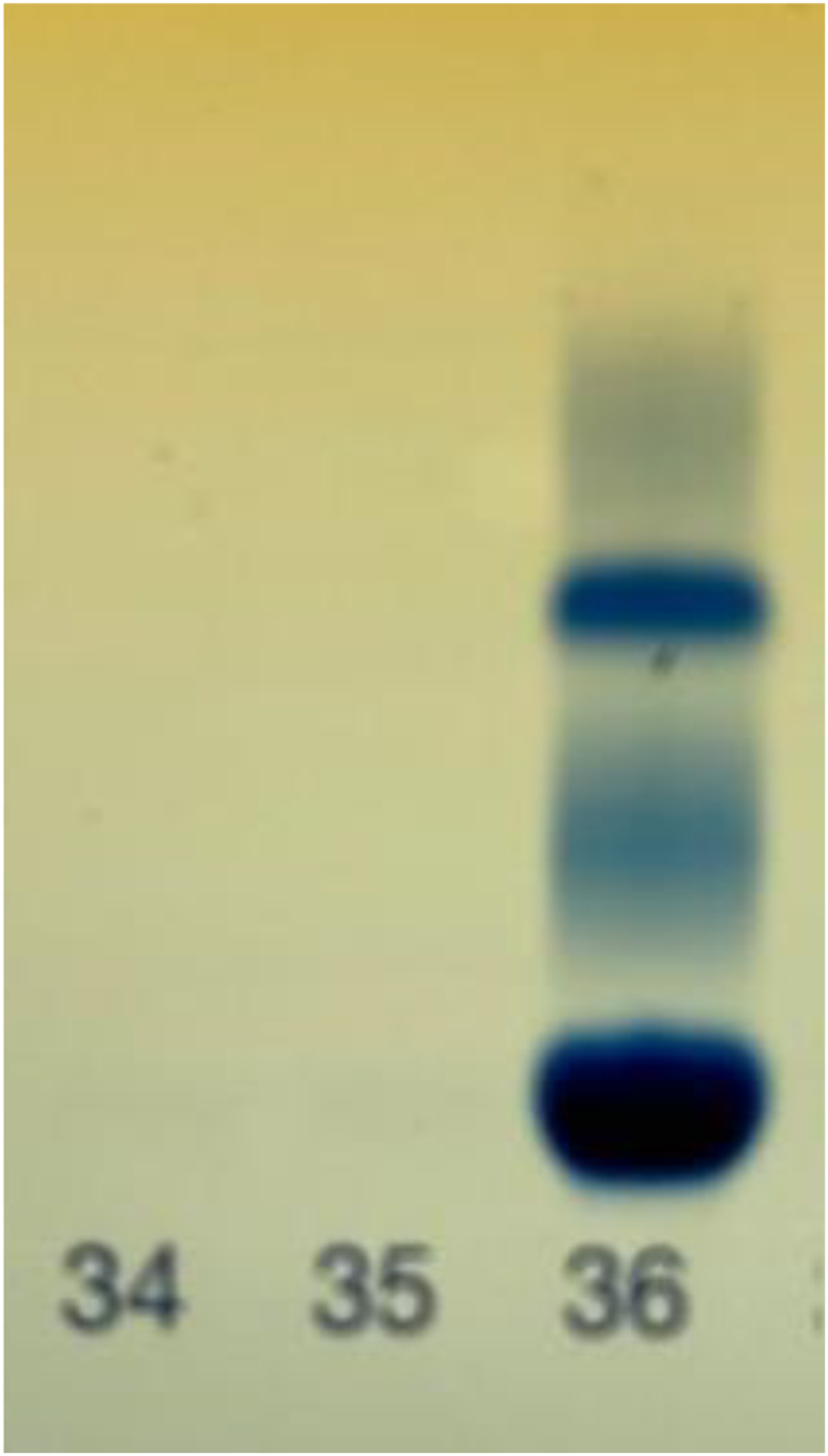
Protein levels in control and patient urine samples before and after the ultrafiltration with a 10,000 Da cut-off.

Urine protein levels measured by the BTC method before and after mixing and incubation of urine sample with BC.

Discussion
High molecular weight substances that are present in barrier cream used together with urine collection bags may lead to positive interference in the benzethonium chloride method for urine protein measurements.
In this case, the initial extremely high protein levels obtained using the BTC method raised suspicion of interference, as they were inconsistent with the negative urine dipstick result. Due to the possibility of laboratory error, no further actions were taken regarding this high level of proteinuria. The medical team’s decision to repeat the urine protein measurement using the pyrogallol red method, which is less susceptible to interference, and the catheter urine showing no protein in the urine, helped to confirm the presence of interference and establish the correct protein levels in the patient’s urine.
This report presents the first case of positive interference by a barrier cream in urine protein measurements. The interference likely occurred due to the presence of high molecular weight molecules that may interfere with the BTC protein quantification method, leading to falsely elevated protein concentrations.
Conclusion
In patients with renal disease, it is crucial to ensure accurate measurements of urine protein levels. This case report highlights the importance of considering potential interferences, including those from barrier creams, when interpreting urine protein measurements. Clinicians and laboratory professionals should be aware of this potential interference and consider alternative collection methods, such as catheterization, or measurement with dye binding methods to obtain accurate results in patients using such products. Further research is warranted to investigate the extent of interference caused by high molecular weight substance-containing products and develop strategies to mitigate their impact on clinical measurements.
Footnotes
Acknowledgements
The authors thank the clinical laboratory of the Rode Kruis Ziekenhuis at Beverwijk (The Netherlands) for measurement of total protein in urine with the pyrogallol red method.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethical approval
Our institution does not require ethical approval for reporting individual anonymous cases. A written informed consent to publication from the patient's guardians was obtained.
Guarantor
WPJE.
Contributorship
JAE and AMB were involved in patient recruitment and gaining written informed consent. J.C.F. was involved in laboratory and data analysis. A.C.H, W.P.J.E., and Ö.Ö. researched literature and conceived the study. Ö.Ö. wrote the first draft of the manuscript. All authors reviewed and edited the manuscript and approved the final version of the manuscript.
Informed consent
A written informed consent to publication from the patient’s guardians was obtained.
