Abstract
Background
Severe hyperammonaemia is associated with significant morbidity and mortality. Rapid analysis and reporting of ammonia results is essential to prevent patient harm. The aim was to investigate the laboratory sample acceptance criteria, ammonia analysis and the reporting of ammonia results.
Methods
A questionnaire was distributed to clinical biochemistry laboratories in the United Kingdom. The results were collated and compared to updated best practice guidelines on hyperammonaemia issued by the Metabolic Biochemistry Network (MetBio.net).
Results
Seventy-six laboratories responded to the audit questionnaire. Although 83% laboratories are aware of the updated MetBio.net hyperammonaemia guidelines, most laboratories continue to reject samples for ammonia that are ‘too old’ for analysis (64%), haemolysed (72%) or not sent on ice (24%). Rapid ammonia analysis is available in 96% laboratories and all laboratories offer ammonia analysis on a 24/7 basis. Nearly all laboratories had implemented critical phoning limits for ammonia.
Conclusions
Laboratories are rejecting samples for ammonia analysis that have not been collected/transported in an optimal manner. Laboratories should review their sample acceptance criteria for ammonia and accept all samples in order to avoid delaying the diagnosis and management of hyperammonaemia.
Keywords
Introduction
Causes of hyperammonaemia.

Although there are many publications on the pre-analytical issues with ammonia analysis, there is no consensus on sample acceptance criteria. In the diagnostic setting, the important question is whether the patient has significant hyperammonaemia. If the blood/plasma ammonia is within the reference interval, then significant hyperammonaemia can be excluded. The Metabolic Biochemistry Network (MetBio.net) is a group of the diagnostic clinical laboratories in the United Kingdom with a special interest in inherited metabolic disorders. In December 2018, MetBio.net published their updated guidelines on the investigation of hyperammonaemia and the measurement of plasma ammonia. 23 An important new recommendation is that laboratories should accept all blood samples for ammonia analysis. If the quality of the sample is less than ideal, the result should still be reported, with an appropriate comment and an urgent repeat requested in order to confirm hyperammonaemia. Samples for ammonia analysis should not be rejected.
Five months after the new guidelines were published, a national ammonia audit of UK clinical biochemistry laboratories was conducted. The aim was to audit UK practice of ammonia measurement against standards based on the MetBio.net guidelines and raise awareness of the new guidelines.
Methods
A questionnaire was devised by the author. The questionnaire was piloted by the MetBio.net stakeholder laboratories. The questionnaire was subsequently revised following discussion and comments from MetBio.net stakeholders. The audit standards were based on the MetBio.net best practice guidelines on the investigation of hyperammonaemia and the measurement of plasma ammonia (2018). 23 A number of additional survey questions were included in the questionnaire in order to determine current practice in UK laboratories. The proposed audit was approved by the National Association for Clinical Biochemistry and Laboratory Medicine (ACB) Audit Committee and the ACB Office emailed the audit questionnaire to senior ACB members and collated the replies.
Results
Seventy-six responses were received. The responses to each of the audit/survey questions are summarised below, together with the questions, shown in italics and the audit standards based on MetBio.net recommendations, where relevant.
[Q1] What sample tubes does your laboratory accept for blood/plasma ammonia measurement?
Most laboratories accepted EDTA sample tubes (68%) or lithium heparin (24%). Both EDTA and lithium heparin sample tubes are accepted by 8% (6/76) of laboratories. The sample type accepted appears to reflect the kit manufacturers recommendation, for example, 38/40 laboratories that use Roche kits only accept EDTA sample tubes. No laboratory accepted serum.
[Q2] Do you regularly check blank sample tubes for ammonia contamination? If yes, how often have you detected contamination in the past five years?
Seven percent (5/76) regularly check blank tubes for ammonia contamination, two out of the five laboratories that check for ammonia contamination found contamination more than once a year.
[Q3] Do you advise that samples for ammonia analysis are delivered (a) on ice, (b) avoiding pneumatic chute, (c) immediately or within a specific time-interval?
The transport of samples on ice and avoiding using a chute were recommended by 78% (59/76) and 26% (20/76) respondents, respectively (Figure 1). All but two (97%) respondents advised that samples are sent immediately or within a specific time-interval (Figure 1). Percentage of laboratories recommending that samples for ammonia analysis are sent (a) immediately or within a specific time-interval, (b) on ice or (c) avoiding pneumatic chute.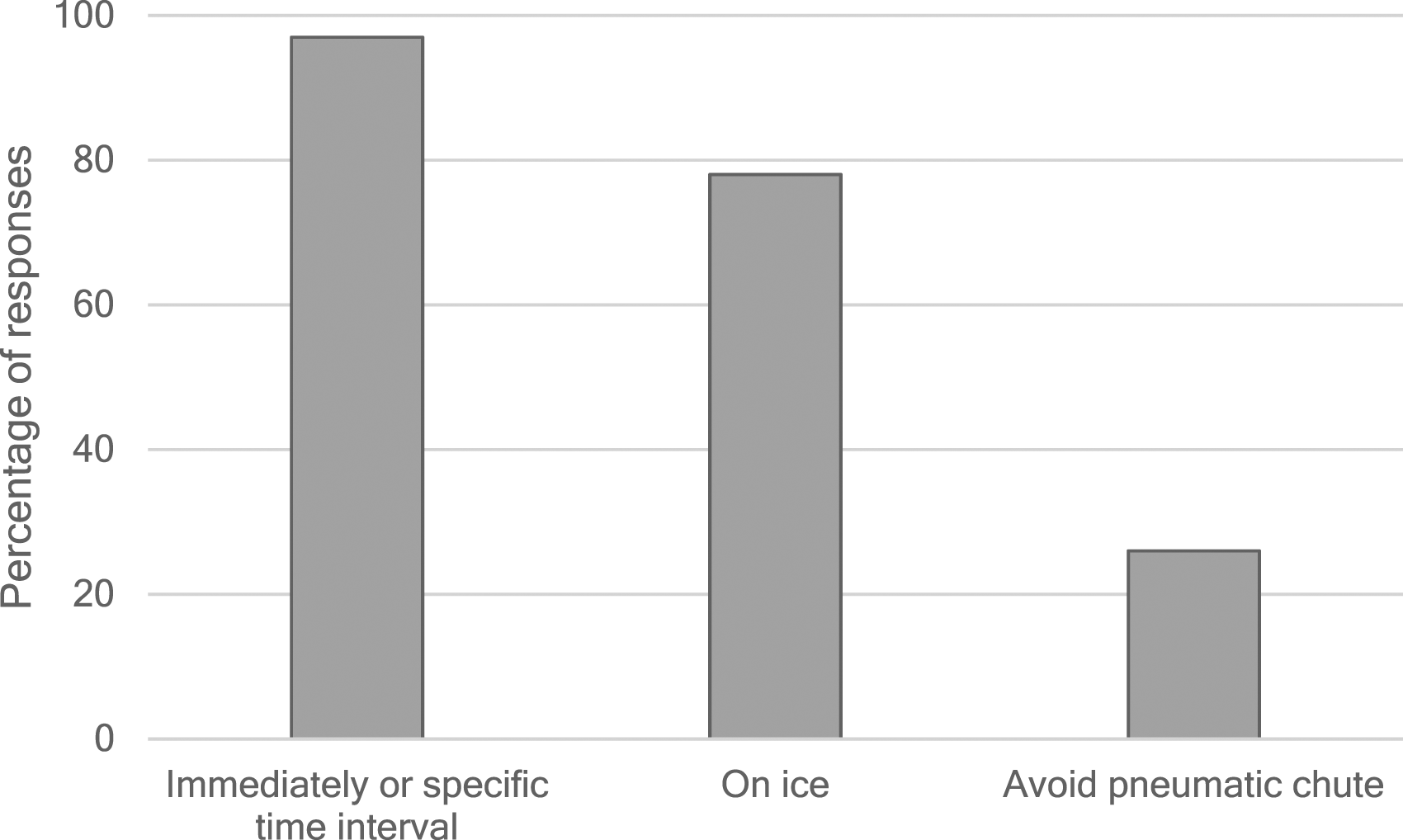
[Q4] Are samples for ammonia analysis fast tracked/prioritised once received in the laboratory?
Almost all respondents (96%, 73/76) fast track samples for ammonia analysis.
[Q5] Do you reject samples for ammonia analysis that are received ‘too old’ for analysis?
The majority of laboratories reject samples on the basis of age (64%, 49/76) (Figure 2). There was a wide range of ages of sample rejected, from 15 min and 24 h. Percentage of laboratories who 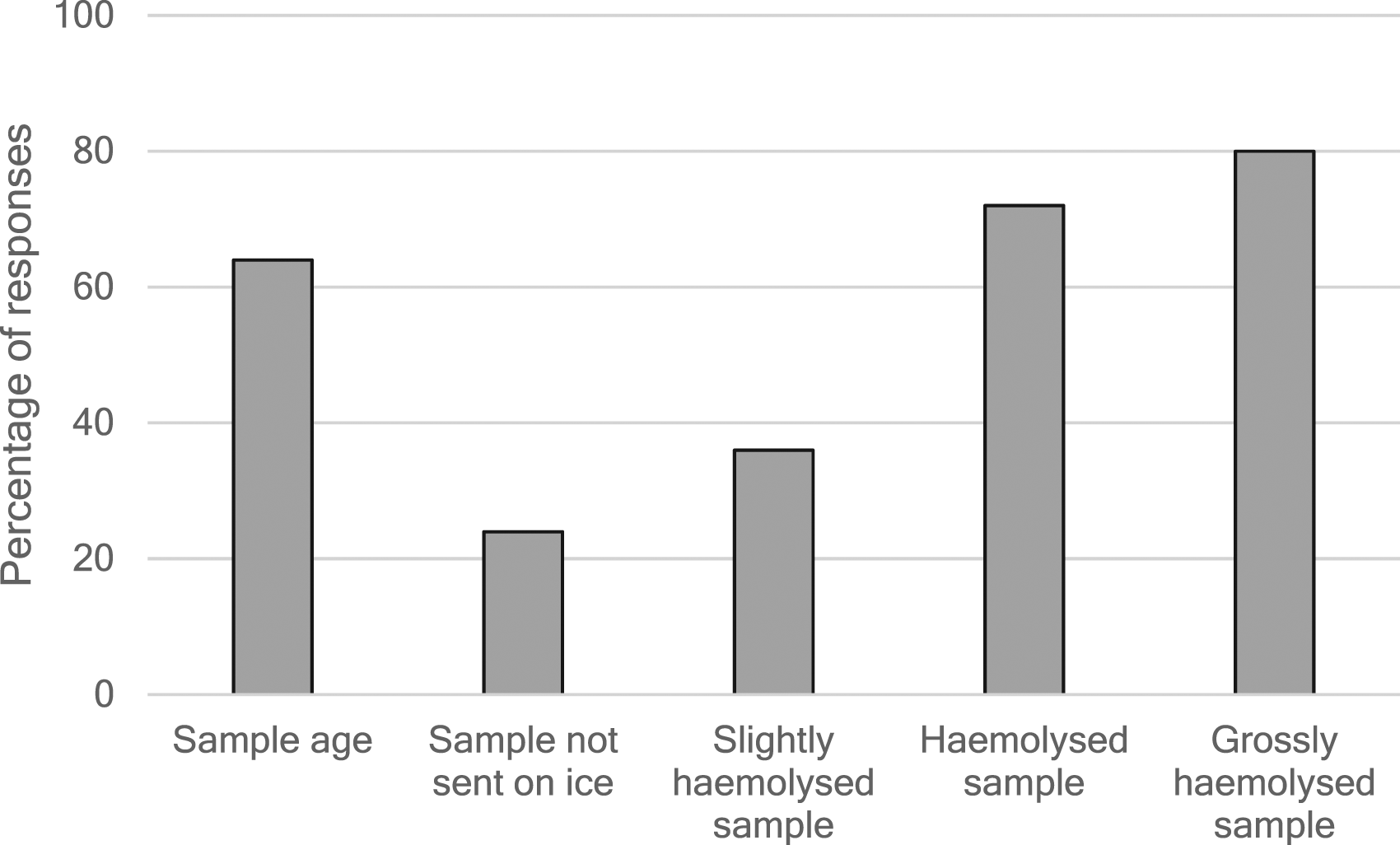
[Q6] If you accept ‘old’ samples, do you report the result with a comment on its validity?
Most respondents (85%, 23/27) who accept ‘old’ samples report the result with an appropriate comment.
[Q7] Do you reject samples for ammonia analysis that are not sent on ice? If you accept samples not sent on ice, do you report the result with a comment on its validity?
Three-quarters (58/76) of laboratories accept samples which have not been sent on ice. Of these laboratories, 55% (32/58) report the results with a comment. Nearly a quarter of laboratories (17/76) reject such samples (Figure 2).
[Q8] Do you assess plasma samples for haemolysis? [Q9] Do you indicate the degree of haemolysis on your reports?
All but one laboratory assess the haemolysis status of plasma samples for ammonia analysis and a quarter (19/76) indicate the degree of haemolysis on their reports.
[Q10] Do you reject samples for ammonia analysis that are slightly haemolysed, haemolysed, grossly haemolysed?
Whether laboratories accept or reject samples depends on the degree of haemolysis. Eighty-four percent (64/76) laboratories accept slightly haemolysed samples, whereas only 28% (21/76) and 20% (15/61) laboratories accept haemolysed and grossly haemolysed samples, respectively (Figure 2).
[Q11] If you accept haemolysed samples for ammonia analysis, do you report the result with a comment on its validity?
Out of those who accept haemolysed samples, 59% (23/39) report the result with a comment.
[Q12] If you reject a sample for ammonia analysis for any reason, do you report with a comment suggesting a repeat sample? (Survey question)
Two-thirds laboratories (50/76) suggest a repeat sample if they reject a sample for any reason.
[Q13] Do you suggest a repeat sample if the ammonia is significantly raised for the first time?
Over half of respondents (43/76) suggest a repeat if the ammonia is significantly raised for the first time.
[Q14] Which manufacturers’ method/instrument do you use for ammonia analysis? (Survey question)
Most widely used methods for ammonia analysis were enzymatic/glutamate dehydrogenase-based methods on automated clinical chemistry analysers, manufactured by Abbott Diagnostics, Beckman Coulter Diagnostics, Roche Diagnostics, Siemens Healthineers. Other methods included a dry slide bromphenol blue dye method (Ortho Clinical Diagnostics) and a bromocresol green dye whole blood method (Menarini Diagnostics).
[Q15] Does your method include a sample blank correction step?
Twenty percent (15/76) of respondents reported that their ammonia method includes sample blank correction whereas 39% (30/76) reported that their method does not. Forty-one percent (31/76) of respondents indicated that they did not know whether their method includes a sample blank correction step or not.
[Q16] Does your manufacturer recommend rejecting haemolysed samples for ammonia analysis? (Survey question)
The majority of respondents (79%, 60/76) stated that the assay manufacturer recommended rejecting haemolysed samples.
[Q17/Q18] Are you concerned that your ammonia method is affected by icterus and lipaemia? (Survey question)
Thirty-five (46%) laboratories expressed some concern about their method being affected by icterus, particularly those using Roche and Beckman methods. Thirty-eight (51%) laboratories using enzymatic glutamate dehydrogenase-based methods expressed some concern about their method being affected by lipaemia.
[Q19] What internal quality control material do you use for ammonia?
Third party quality control (QC) is used by 61% (46/76) laboratories. The majority of laboratories using Siemens, Abbott and Beckman analysers use third party QC whereas the majority of laboratories using Roche, Menarini and Ortho Clinical Diagnostics analysers use QC material provided by their assay manufacturer.
[Q20] What are the typical internal QC ranges? (Survey question)
A wide range of internal QC ranges were provided which could be grouped into low (<80 μmol/L), medium (≥80–160 μmol/L) and high (≥160 μmol/L). All but three laboratories use either two levels of QC (67%) or three levels of QC (23%).
[Q21] Which ammonia external quality assurance scheme do you participate in?
All laboratories participate in either the Welsh External Quality Assurance Scheme (WEQAS) (89%, 68/76) or Randox International Quality Assessment Scheme (RIQAS) (11%, 8/76).
[Q22] Please provide your age-matched reference ranges for ammonia
Most laboratories (75%, 53/71) quote separate reference intervals for neonates. Seventy percent (50/71) laboratories quote a reference interval of <100 μmol/L for neonates. Thirty-nine percent (28/71) quote a separate range for premature neonates with the majority of those (26/28) quoting <150 μmol/L. Forty-five percent (32/71) quote separate reference intervals for children. Just over half of laboratories (37/71) quote <40 or <50 μmol/L as their reference interval for patients >1 month old. There were 11 laboratories that quoted sex partitioned adult reference intervals, and these were all users of Roche Diagnostics analysers.
[Q23] What is the source of your reference intervals?
Various sources of the reference intervals were quoted. Twenty percent laboratories (15/76) quoted different sources for their adult and paediatric reference intervals. The source of reference intervals was unknown/historic for 39% (30/76) laboratories. The kit insert was the source of sex partitioned adult reference intervals for 11/42 of the laboratories using Roche Diagnostics analysers and a further four laboratories (non-Roche Diagnostics) used their manufacturers’ reference intervals. Guidelines were the sources for the reference intervals for 39 laboratories with 22 laboratories using Pathology Harmonisation/Scottish/North-East Harmony guidelines, 14 using MetBio.net and three using All-Wales reference intervals. Four laboratories reported that their reference intervals were taken from the literature/published data and a further three had derived their own reference intervals.
[Q24] Do you provide a 24/7 service for ammonia analysis?
All laboratories provide a 24/7 service for ammonia analysis.
[Q25] Do you have a critical/phoning limit for ammonia?
Almost all laboratories (93%, 71/76) have critical/phoning limits in place. Most (72%, 51/71) use a single critical limit for all results whereas 8% (6/71) laboratories phone all results above the upper reference limit(s). Ninety percent (46/51) of those, who quote a single critical limit, quote 100 μmol/L whereas two laboratories quote 150 μmol/L, and one quotes 200 μmol/L. Different limits are in place for neonates and adults in 15% (20/71) laboratories. Three laboratories (4%) only have critical limits for neonates/children. One laboratory phones all ammonia results.
[Q26/Q27] Are you aware of the MetBio.net guidelines for the investigation of hyperammonaemia (updated 2018) 23 and British Inherited Metabolic Diseases Group (BIMDG) emergency guidelines for treatment of hyperammonaemia? 24
Whilst 83% (63/76) respondents were aware of the MetBio.net guidelines, only 57% (43/76) were aware of the BIMDG emergency guidelines.
[Q28] What is your approximate annual workload for ammonia? (Survey question)
Annual workload varied between <100 requests per year for 10 laboratories to over 2000 requests per year for one laboratory. Most laboratories (56) receive between 100 and 1000 requests per year.
Discussion
Sample Rejection
The audit clearly shows that laboratories are aware of the need for the rapid transport and analysis of samples for ammonia. Once a blood sample has been collected, the ammonia levels start to rise. Red blood cells release ammonia into the plasma and ammonia continues to be produced by the deamination of amino acids by plasma enzymes. Increases in ammonia in whole blood are greater than those in plasma, with greater increases at higher temperatures.14–20 In a study where blood was collected from healthy volunteers (n = 36), whole blood ammonia concentrations increased by 3.9 μmol/L per hour at 0°C, 5.2 μmol/L at 20°C and 25.2 μmol/L per hour at 37°C from a mean ammonia concentration of 23.0 μmol/L. In plasma ammonia concentrations increased by 0.4 μmol/L per hour at 0°C, 1.4 μmol/L per hour at 20°C and 4.2 μmol/L per hour at 37°C from a mean ammonia concentration of 23.0 μmol/L. 14 Therefore, speedy delivery of the sample to the laboratory and rapid analysis is required to minimise such increases. 14 Chilling blood samples, by transporting them on wet ice, reduce enzyme activity and sample deterioration. To avoid artefactual increases in ammonia, good sampling technique is required to avoid haemolysis. 15 Although a poor quality sample may result in an overestimate of plasma ammonia, reporting a high result, with an appropriate comment and making a request for an urgent repeat, is not likely to do any harm. 25 Clinicians do not require a highly accurate result in the first instance to diagnose severe hyperammonaemia; rather, they need to know whether the ammonia level is normal/slightly raised or is very high so that the patient can be treated and appropriate investigations initiated. The majority of laboratories reject samples for ammonia analysis that fail to reach the laboratory within a specific time period and/or are not sent on ice and/or are haemolysed despite the release of updated guidelines.
The rejection of compromised blood samples for ammonia has contributed to significant delays in identifying and treating patients with hyperammonaemia, resulting in the issuing of two patient safety alerts by the Royal College of Pathologists.21,22 It is important that all ammonia requests are processed and reported with an appropriate comment. Repeat samples should be requested urgently to check an abnormal result if the quality of the original sample has been compromised. It is surprising that the majority of laboratories are rejecting samples for ammonia analysis as 83% laboratories indicated that they are aware of the updated MetBio.net guidelines which were issued five months prior to the audit. This may have been because laboratories had yet to implement these new guidelines and update their policies and standard operating procedures.
It is important that the tests performed in medical laboratories are accredited to ISO15189:2012. It is possible that some laboratories have become more averse to deviating from manufacturers’ instructions and are not willing to analyse haemolysed samples or samples which are delayed in reaching the laboratory as this would deviate from such instructions. El-Khoury and colleagues have questioned whether the effect of haemolysis by kit manufacturers has been overstated. They compared two methods of producing haemolysates: the more commonly used osmotic shock method and the shearing method (repeatedly passing blood through a needle and syringe). Using the shearing method, a greater than 10% change in measured ammonia was seen at a haemolysis index (HI) of 220 (at plasma ammonia of 34 μmol/L) and HI 814 (at a plasma ammonia of 59 μmol/L). Whereas using the osmotic shock method a greater than 10% change in measured ammonia was seen at much lower HIs: HI 29 (at plasma ammonia of 35 μmol/L) and HI 116 (at a plasma ammonia of 53 μmol/L). The shearing method may better reflect the haemolytic process during blood collection. 26 The ISO15189:2012 standard contains two important clauses (4.1.2.2 and 4.14.6). 27 Clause 4.1.2.2 requires the laboratory to ‘meet the needs of patients’ and clause 4.14.6 states that ‘The laboratory shall evaluate the impact of work processes and potential failures on examination results as they affect patient safety, and shall modify processes to reduce or eliminate identified risks…’
Environmental Contamination
The laboratory has an important role in optimising pre-analytical processes in order to reduce the likelihood of compromised samples. It is also important that the laboratory promotes awareness of the potential environmental causes of artefactually high ammonia results and assists in minimising their impact. There are various potential sources of ammonia contamination in the hospital environment such as cleaning products, diagnostic reagents, 28 contaminated sample collection tubes and refrigerants. The MetBio.net hyperammonaemia guidelines were amended in 2018 to reflect the important of recognising all potential environmental sources of ammonia contamination in clinical areas rather than focussing on blood collection tubes.
This survey showed that only 7% laboratories check for potential ammonia contamination of blood collection tubes. This compares to 89% laboratories when audited in 2006 (UK Ammonia Audit (WEQAS) 2006, unpublished data). Feedback comments from participants in this survey indicated that they no longer check as they never found contamination and it is difficult to assess. However, out of the five laboratories that check for ammonia contamination, two laboratories detected contamination of their blood collection tubes. Checking for sample collection tube contamination is no longer specifically recommended in the MetBio.net guidelines; however, contamination from blood collection tubes remains an important consideration when investigating the cause of spuriously high ammonia results.
Sample Blanking
Forty-one percent respondents were not aware whether their ammonia assay includes a blank correction step. One-step enzymatic assay formats do not allow for sample blanking which means that these methods are prone to interference from liver enzymes such as dehydrogenases and alanine aminotransferase. 29 This is particularly important in patients with liver disease. In such patients, the interference from liver enzymes may be significant when using one-step enzymatic ammonia assays and may give rise to falsely elevated ammonia results. 30
Critical Limits
Whilst most laboratories have critical phoning limits for ammonia, not all have adopted the critical limit in the Royal College of Pathologists guideline ‘The communication of critical and unexpected pathology results’. 31 Whilst a few laboratories have set more conservative limits, others have higher critical limits and one laboratory only phones high ammonia results in infants and children. The reasons for this were not stated. Timely communication of high ammonia results for all patients is vital in order to identify and treat all patients with hyperammonaemia as quickly as possible.
Reference Intervals
The audit showed that a range of reference intervals are quoted by laboratories. Published reference intervals for plasma ammonia are sparse and are generally not well defined. Therefore, it was not surprising that many laboratories stated that the source of their reference intervals was unknown/historic and that many laboratories have adopted reference intervals proposed by Pathology Harmonisation/Scottish Harmony and MetBio.net which are similar. MetBio.net provides the following plasma ammonia age-related reference intervals in the guidelines: premature neonates <150 μmol/L, term neonates <100 μmol/L, infants/children <40 μmol/L and adults <40 μmol/L. Some of the laboratories using Roche Diagnostics analysers quote the sex partitioned reference intervals for adults provided by the manufacturer. Whilst most manufacturers appear to provide adult reference intervals, paediatric reference intervals are lacking.
Summary
The audit has identified several areas of good practice • laboratories are aware of the pre-analytical issues with ammonia and the need to optimise sample quality • most laboratories are using at least two levels of QC and all participate in external quality assurance schemes • all laboratories offer a 24/7 analytical service • the majority of laboratories have adopted appropriate critical phoning limits for ammonia • although reference intervals for ammonia are poorly defined, the reference intervals quoted seem appropriate
The audit has shown that, although most laboratories are aware of the updated MetBio.net guidelines, many had yet to implement them fully at the time of this audit. It is recommended that • laboratories review their sample acceptance criteria in view of the findings of this audit, the updated MetBio.net guidelines and the Royal College of Pathologists patient safety alerts. • laboratories accept and analyse all samples received for ammonia analysis even if the quality is less than ideal as prompt measurement of ammonia is required for early identification of hyperammonaemia. • laboratories familiarise themselves with the operating characteristics and limitations of their ammonia methods. • if the ammonia is high (>100 μmol/L) then an urgent repeat should be requested before treatment is initiated. Once hyperammonaemia is confirmed, the patient can be managed appropriately reducing the risk of significant morbidity and mortality.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethical approval
Not applicable.
Guarantor
HA.
Contributorship
HA conducted the audit, analysed the data, drafted and revised the manuscript.
