Abstract
Background
Urine oxalate measurement is an important investigation in the evaluation of renal stone disease. Primary hyperoxaluria (PH) is a rare inherited metabolic disease characterised by persistently elevated urine oxalate, but the diagnosis may be missed in adults until renal failure has developed. Urine oxalate results were reviewed to compare oxalate:creatinine ratio and oxalate excretion, and to estimate the potential numbers of undiagnosed PH.
Methods
Urine oxalate results from August 2011 to April 2013 were reviewed. Oxalate excretion and oxalate:creatinine ratio were evaluated for 24 h collections and ratio alone for spot urine samples.
Results
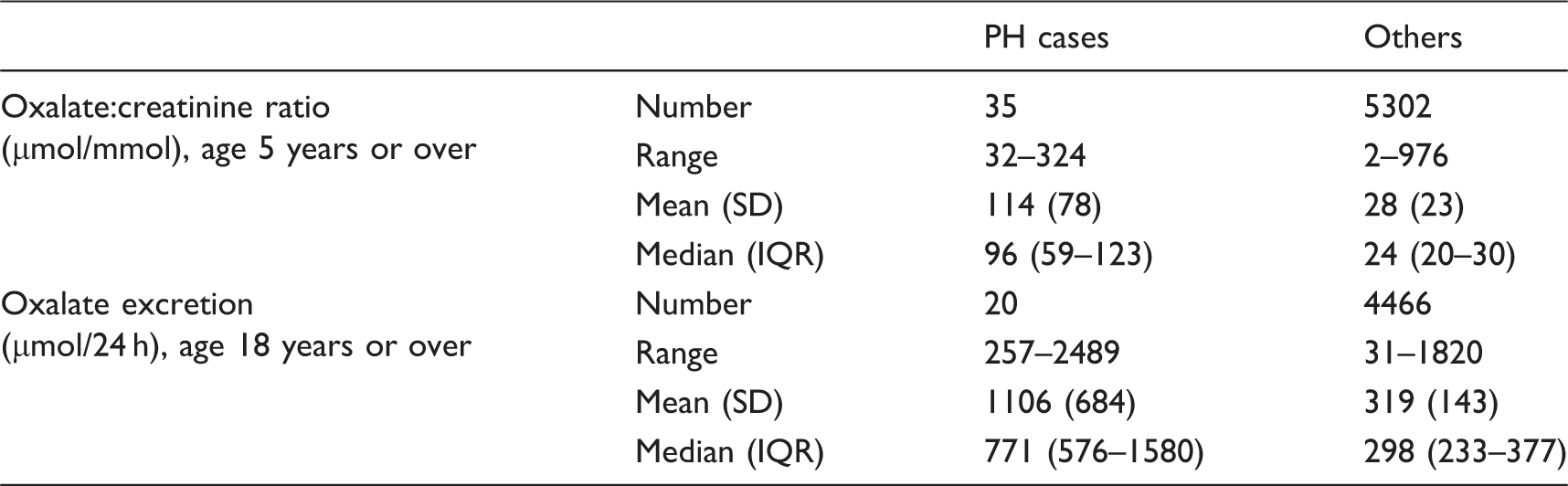
Oxalate:creatinine ratio and oxalate excretion were moderately correlated (R = 0.63) in 24-h urine collections from patients aged 18 years and above. Sex-related differences were found requiring implementation of male and female reference ranges for oxalate:creatinine ratio. Of samples with both ratio and excretion above the reference range, 7% came from patients with confirmed PH. There were 24 patients with grossly elevated urine oxalate who had not been evaluated for PH.
Conclusions
Oxalate:creatinine ratio and oxalate excretion were discordant in many patients, which is likely to be a result of intra-individual variation in creatinine output and imprecision in the collection itself. Some PH patients had urine oxalate within the reference range on occasion, and therefore it is not possible to exclude PH on the finding of a single normal result. A significant number of individuals had urine oxalate results well above the reference range who potentially have undiagnosed PH and are consequently at risk of renal failure.
Introduction
Measurement of urine oxalate is a common investigation in the work up of patients with renal stone disease, and is the first line test when considering primary hyperoxaluria (PH). Hyperoxaluria is a well-recognised risk factor for calculus formation 1 because of the very low solubility of calcium oxalate, which can consequently lead to urine supersaturation.
Hyperoxaluria is usually a secondary phenomenon and due to exogenous sources of oxalate. Dietary oxalate is relatively widespread; examples of food with high oxalate content include rhubarb, chocolate, spinach and beetroot. Dietary calcium normally complexes oxalate in the gut lumen, which reduces the amount of oxalate that is absorbed. 2 If calcium availability is limited, then there will be more free oxalate and enteric hyperoxaluria can occur. Fat malabsorption is one cause of this, when calcium fatty acid salts are formed in the gut lumen. Enteric hyperoxaluria is found in malabsorptive conditions, such as cystic fibrosis, 3 and post bariatric surgery. Roux-en-Y gastric bypass surgery has been shown to increase oxalate absorption from the gut, 4 and has also been associated with increased urine oxalate excretion and urolithiasis.5,6
The primary hyperoxalurias are rare inherited metabolic diseases of glyoxylate metabolism that cause a build up of endogenous oxalate (for a recent review see Cochat and Rumsby, 2013). 7 Hyperoxaluria causes renal damage indirectly by recurrent stone formation, as well as via direct oxalate toxicity, leading to end stage renal failure (ESRF) after which systemic deposition of calcium oxalate occurs. Three forms of disease, PH1, PH2 and PH3, are caused by mutations in AGXT, GRPHR and HOGA1, respectively. Of these, PH1 accounts for the majority (∼90%) of cases with a prevalence of ∼1 per million in Europe. 8 The clinical spectrum is very broad in PH; PH1 patients can have a severe phenotype presenting with ESRF and systemic oxalosis in infancy, but can also have a later presentation with recurrent urolithiasis. All three forms have a similar range of age of onset 9 and degree of oxaluria, 10 but PH2 11 and PH3 12 are thought to have milder clinical phenotypes; for example, ESRF and systemic oxalosis have not yet been reported in PH3, though this is a relatively newly identified disease. Despite the heterogeneity in clinical symptoms, an elevated urine oxalate is the common hallmark in PH, and is the essential first line investigation. 13 Definitive diagnosis of these conditions requires the finding of a known pathological mutation on gene sequencing, or demonstration of impaired enzyme activity in a liver biopsy.
Although PH is an infrequent cause of renal stone disease, the consequences of missed diagnosis are severe. Without treatment, patients can progress to ESRF and if transplanted they can lose the graft; 14 a double tragedy if a living related donor organ is used. 15 There is increasing recognition of the impact of rare diseases on individuals and families as illustrated by the move to implement a national strategy on Rare Disease (http://www.raredisease.org.uk/uk-rare-disease-strategy.htm). PH should be considered in unexplained nephrocalcinosis, recurrent calcium oxalate stone formers, bilateral stone formers or unexplained renal failure, particularly when oxalate crystals are seen in the kidney.
The department of Clinical Biochemistry at University College London Hospitals (UCLH) is a tertiary referral centre for the investigation of PH. It is the only UK centre that can provide definitive diagnosis of all forms of PH, and a database of all confirmed cases of PH in the UK is maintained. As we have recently seen an increase in adult presentation of PH, all of whom had a history of recurrent stone disease, we performed a review of our data with the following aims: to investigate the comparability of oxalate:creatinine ratio and oxalate excretion for the diagnosis of hyperoxaluria, to survey urine oxalate results stratified by the PH status of the patients, and to estimate the potential numbers of undiagnosed PH.
Materials and methods
Oxalate was measured on an Aries autoanalyser (Instrumentation Laboratory SpA, Milan, Italy) using oxalate oxidase (Trinity Biotech plc, Wicklow, Ireland) with a pre-treatment step to remove interference from ascorbate. 16 Creatinine was measured on a Cobas 8000 autoanalyser (Roche Diagnostics GmbH, Mannheim, Germany) using a kinetic Jaffé reaction according to the manufacturer’s instructions.
The laboratory information management system was searched from 1 August 2011 to 30 April 2013 for samples on which urine oxalate measurement had been requested. The following data were extracted for each sample: patient age and sex, urine oxalate concentration, oxalate:creatinine ratio, oxalate excretion, urine creatinine concentration, creatinine excretion, urine volume and urine time. External quality assessment, research and dummy samples were removed. Samples without recorded patient age or sex, or without a urine oxalate concentration result were also removed. For the purposes of this analysis, where oxalate had been reported as <50 µmol/L, a value of 25 µmol/L was substituted and the oxalate:creatinine ratio was recalculated for these samples, provided that the urine creatinine was >1 mmol/L (thus very dilute urine samples were excluded).
The database of PH cases contains all UK patients in whom a definitive diagnosis had been made. Urine oxalate results were cross-checked against this database to identify known PH cases. The dataset was split into two groups: those in whom a definitive diagnosis has been made (PH group) and all other patients without confirmed PH (‘Others’ group).
Excel (Microsoft Corporation, Redmond, USA) was used to manipulate the dataset and parametric and non-parametric descriptive statistics were used as appropriate. Means of populations were compared using the t test and statistical significance was defined as a p ≤ 0.05.
Results
Demographic details of the dataset.
Age dependence of both oxalate excretion and oxalate:creatinine ratio was observed in the ‘Others’ group. Oxalate excretion (not corrected for body surface area) levelled out from approximately age 18 years (data not shown); for this reason, the dataset was limited to those aged 18 years or over when considering this variable. Oxalate:creatinine ratio fell rapidly over the first years of life and achieved stability from approximately age five years (Figure 1); for this reason, the dataset was limited to those aged 5 years or over when considering this variable. Where patients had multiple samples in the dataset, the mean oxalate values were calculated so that there was just one data point for each patient.
Age dependence of oxalate:creatinine ratio in patients without a diagnosis of PH.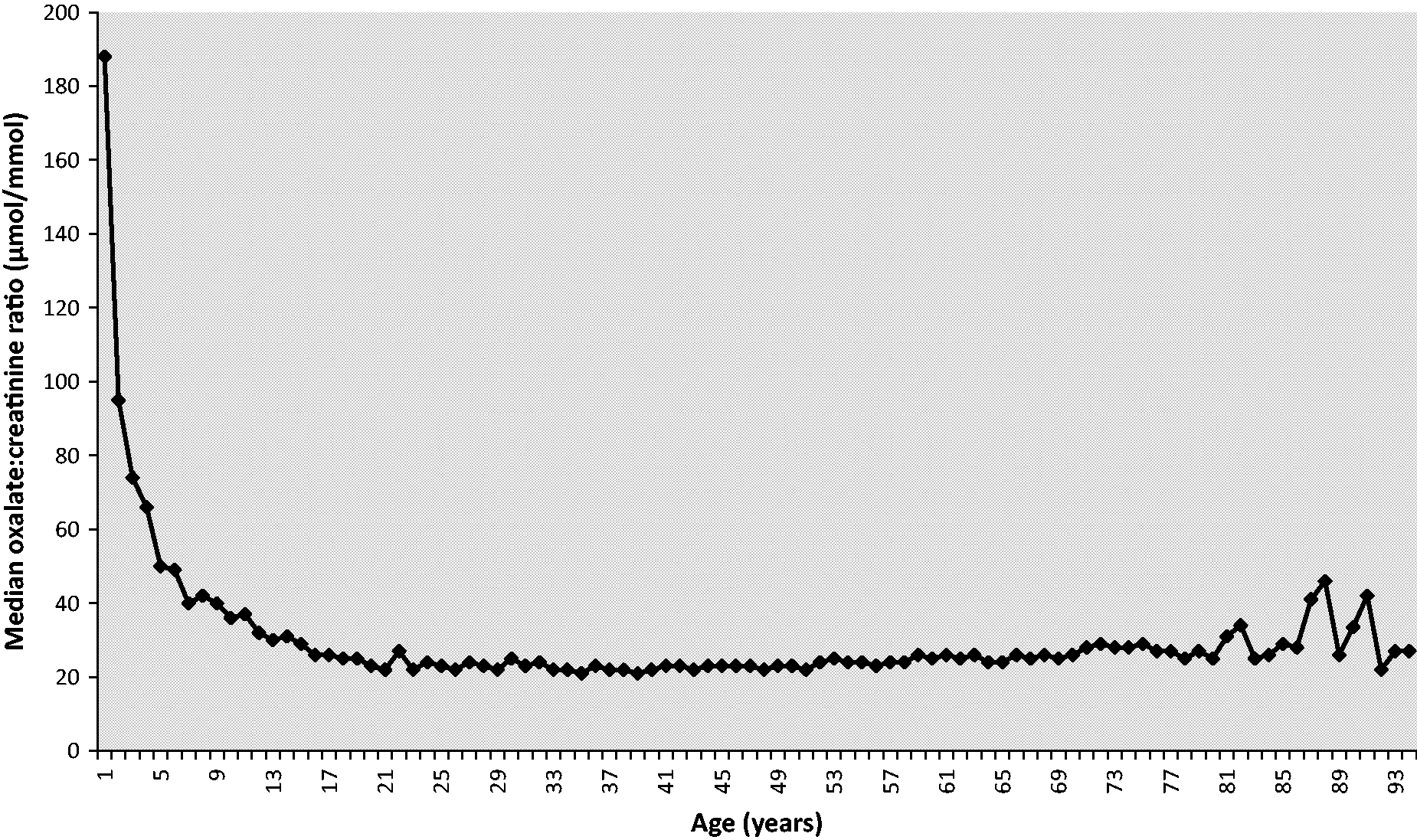
Using data from 24 h collections from patients aged 18 years or over, oxalate excretion and oxalate:creatinine ratio were plotted against each other after first removing samples with likely under and over-collection (under-collection defined as 24 h urine volume <500 mL and creatinine excretion <4 mmol/24 h; over-collection defined as 24 h urine volume >4000 mL and creatinine excretion >21 mmol/24 h) (Figure 2). Pearson’s correlation coefficient was 0.63. To address this imperfect relationship, we reviewed the distribution of results for males and females in the ‘Others’ group (Table 2). Both mean oxalate excretion and mean creatinine excretion were significantly higher in men (340 vs. 281 µmol/24 h, p < 0.001 and 14.7 vs. 9.8 µmol/24 h, p < 0.001), whereas mean oxalate:creatinine ratio was significantly higher in women (32 vs. 26 µmol/mmol, p < 0.001). After removing 626 24 h collections with the oxalate excretion result above the laboratory’s upper reference limit (URL) of 460 µmol/24 h, the distribution of oxalate:creatinine ratio results was re-calculated. The 95th centile was 39 µmol/mmol for all subjects, 33 µmol/mmol for men and 45 µmol/mmol for women.
Scatter plot of oxalate:creatinine ratio and oxalate excretion. PH cases are represented by the hollow triangles, and others are represented by shaded diamonds. The lines represent the laboratory’s current URLs: oxalate:creatinine ratio 38 µmol/mmol, and oxalate excretion 460 µmol/24 h. Distribution of oxalate results for men and women in the ‘Others’ group.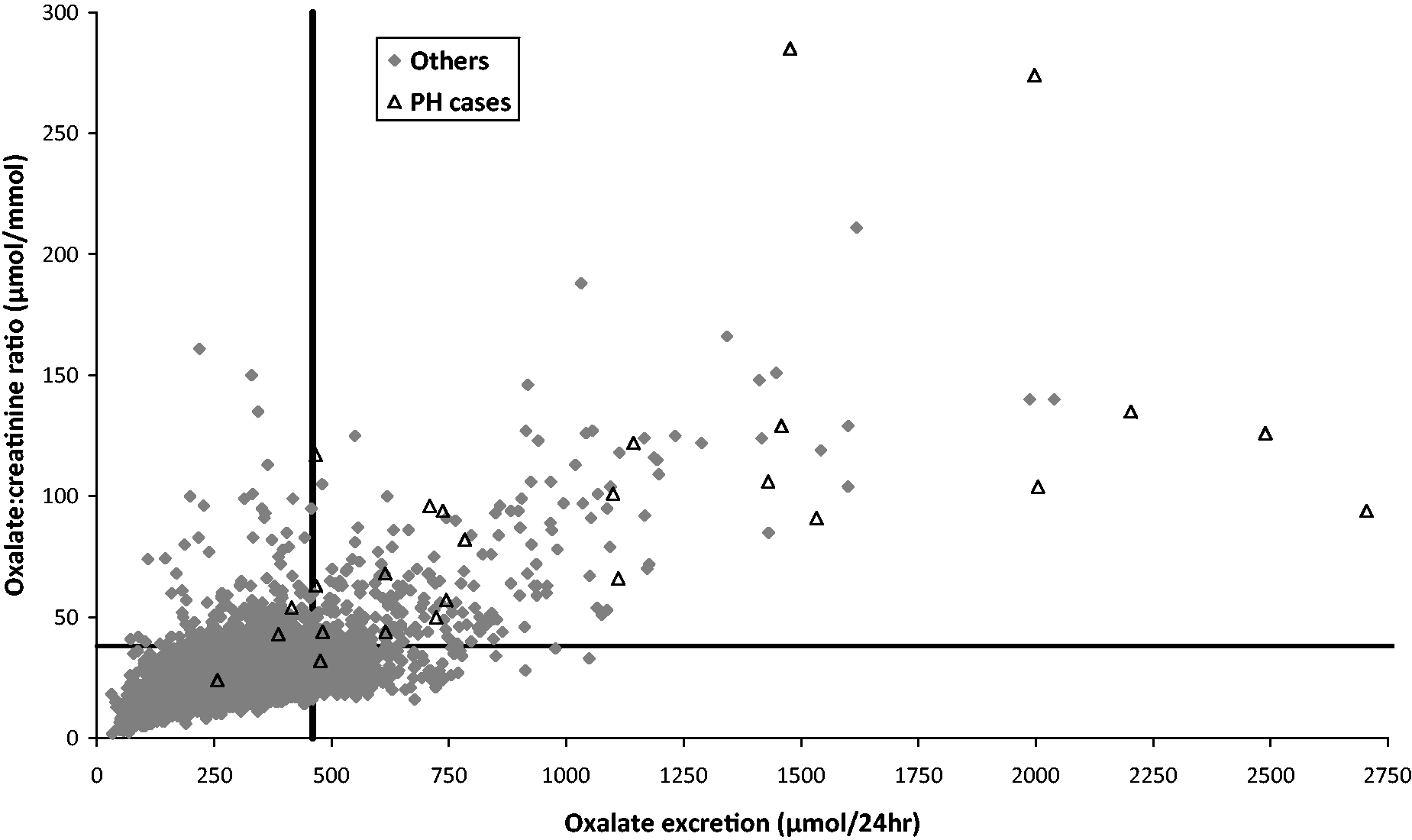

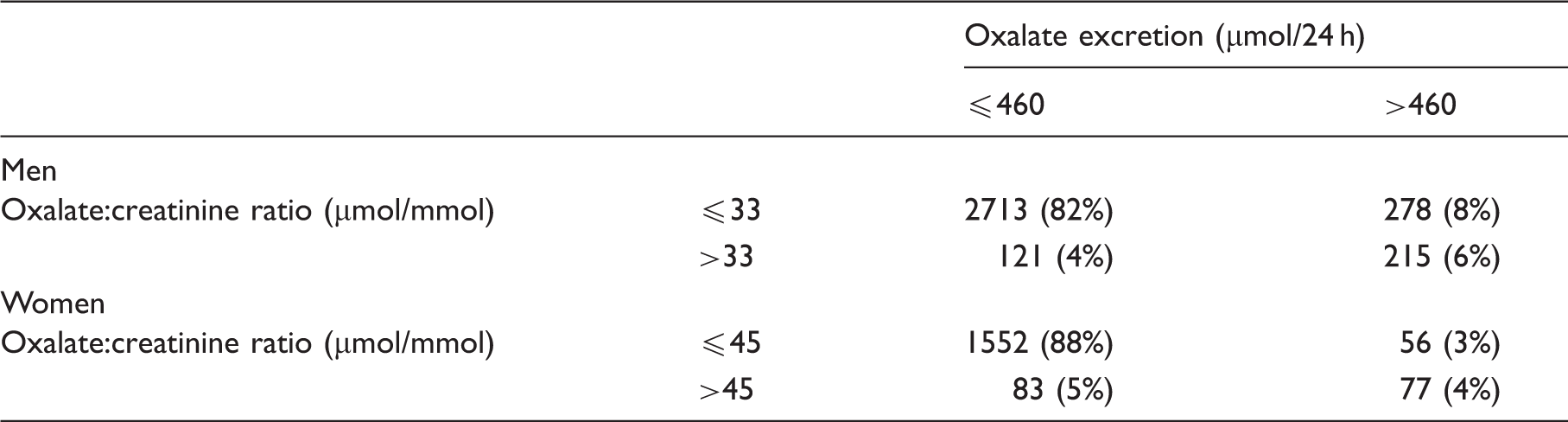
Categorisation of oxalate results for men and women in the ‘Others’ group, according to the laboratory’s URL for oxalate excretion and sex related cut-offs for oxalate:creatinine ratio.
The distribution of results for the PH and ‘Others’ groups is summarised in Table 4. Both mean oxalate:creatinine ratio and mean oxalate excretion were significantly higher in the PH group (114 vs. 28 µmol/mmol, p < 0.001 and 1106 vs. 319 µmol/24 h, p < 0.001), and there was no difference in mean creatinine excretion (11.8 vs. 13.0 mmol/24 h, p = 0.272). There was overlap in the distribution of results for the PH and ‘Others’ groups for oxalate:creatinine ratio (Figure 3 and Table 4) and oxalate excretion (data not shown). There was no statistically significant difference between patients with each type of PH by one way ANOVA for oxalate:creatinine ratio (p = 0.46, Figure 3) and oxalate excretion (p = 0.999).
Box and whisker plot of oxalate:creatinine ratios from patients aged five years or over, stratified according to diagnosis (PH1, PH2, PH3, all PH and ‘Others’). The circle represents the mean, the horizontal line the median, the box the interquartile range, the whiskers the 2.5th and 97.5th percentiles and the diamonds the 24 patients from the ‘others’ group with grossly elevated results. Distribution of oxalate results for PH cases and ‘Others’.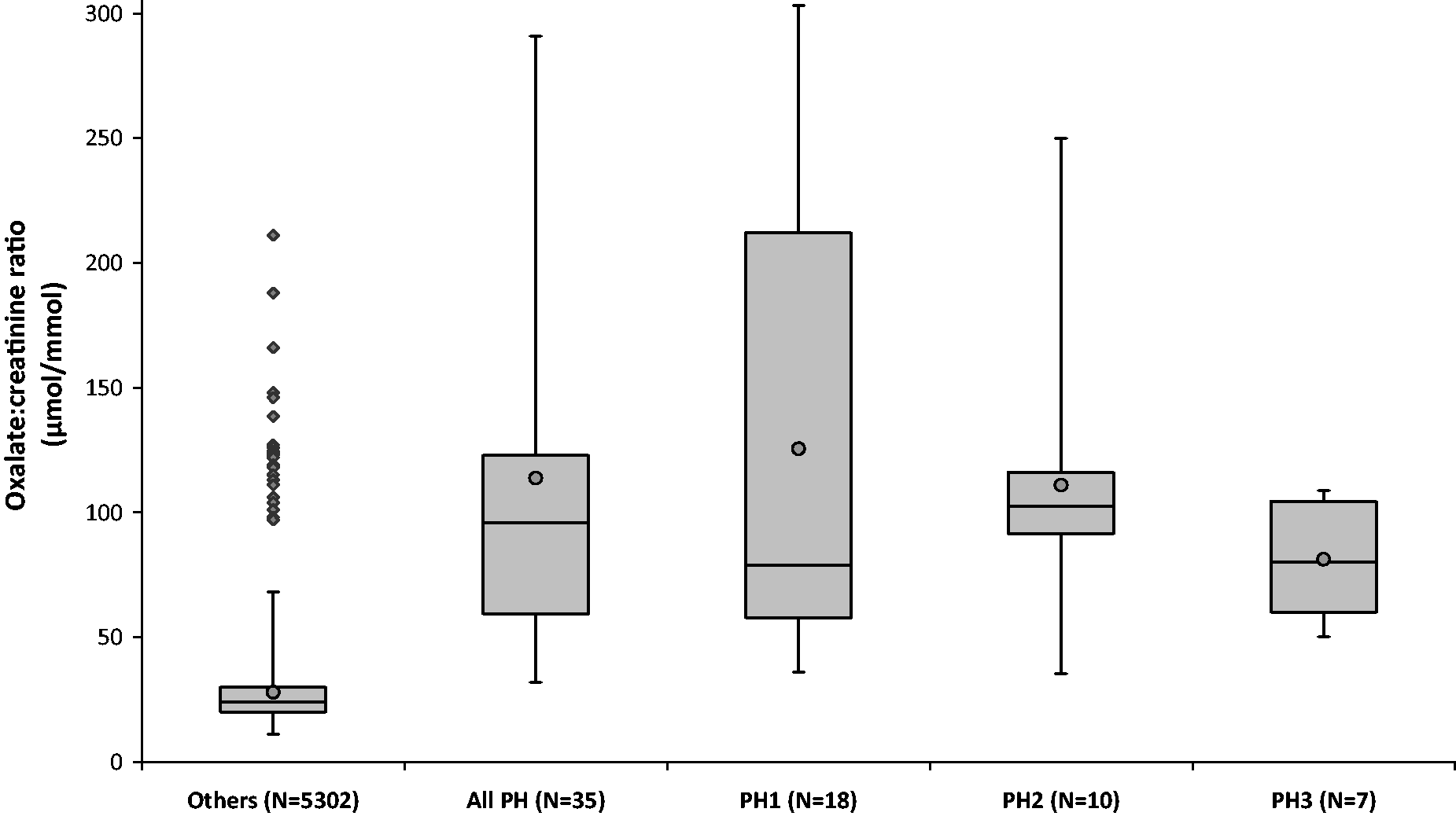
The majority of samples from PH cases (21) had both elevated oxalate:creatinine ratio and oxalate excretion; PH cases accounted for 7% of all samples with both parameters elevated. Five PH cases had an oxalate result within the reference range: One of these, who had both parameters in the reference range (excretion 257 µmol/24 h, ratio 24 µmol/mmol), was in renal failure. The other four were on pyridoxine treatment at the time of testing; oxalate excretions were 476, 415, 481 and 387 µmol/24 h, respectively, and oxalate:creatinine ratios were 32, 54, 44 and 43 µmol/mmol, respectively.
The wide distribution of oxalate results in PH limits the value of a single cut-off for diagnosis. However, we took the median result from the PH group (oxalate:creatinine ratio 96 µmol/mmol and oxalate excretion 771 µmol/24 h) to define a cut-off for grossly elevated oxalate in the ‘Others’ group, i.e. those in whom a diagnosis of PH should be considered. There were 24 patients aged 18 years or over in this category who had not been followed up with a definitive diagnosis, and these results are highlighted on Figure 3. This number does not count any potential missed cases of infantile onset hyperoxaluria, because paediatric results were not included in this part of the analysis.
Discussion
When patients are investigated for recurrent renal calculi or unexplained renal failure, PH must be considered as an important, albeit rare, diagnosis, and measurement of urine oxalate is central to the initial evaluation of these patients. We surveyed more than 6500 urine oxalate results from a 20-month period in conjunction with the database of known PH cases in the UK, allowing separate analysis of results from patients with PH and results from ‘Others’. The ‘Others’ group are from patients who were likely to be under investigation for renal calculi and so will almost certainly include many with secondary hyperoxaluria, and possibly some with undiagnosed PH. The demographic profile showed a predominance of middle-aged men: 65% of samples came from men with a median age of 46 years (Table 1). In contrast, the patients with confirmed PH had a more equal male:female distribution (57% male) as is to be expected for an autosomal recessive inherited disease. They also had a lower age profile, reflecting the fact that in many cases PH presents in infancy. Unsurprisingly both mean oxalate excretion and mean oxalate:creatinine ratio were higher in the PH group (Table 4) and there was no significant difference in creatinine excretion (data not shown).
The age dependence of urine oxalate is well known17–19 and our data confirmed a rapid fall in oxalate:creatinine ratio over the first few years of life (Figure 1). Rapid growth and increase in muscle mass leads to larger creatinine production and hence excretion, therefore oxalate:creatinine ratio in urine decreases. Inspection of Figure 1 reveals that the fall in median oxalate:creatinine ratio starts to plateau after about 5 years of age, which was the justification for removing data from patients aged under 5 years from the analysis. There is also apparent increased variability in oxalate:creatinine ratio above age 80 years; however, this probably reflects the lower numbers of samples from elderly patients. Oxalate excretion results should be corrected to a body surface area of 1.73 m2 in children to allow interpretation, 13 because unadjusted results may appear misleadingly normal (this is where the oxalate:creatinine ratio can be particularly helpful). Data on body surface area was unavailable and the median unadjusted oxalate excretion varied widely until about age 18 years; therefore, oxalate excretion data on patients aged under 18 years were removed from the analysis.
We investigated the comparability of two parameters: oxalate output and oxalate:creatinine ratio, both determined from the same 24 h urine collection. The use of a 24 h collection is often preferred over a spot urine sample; it indicates the overall level of oxaluria and should be less subject to variation in oxalate and creatinine excretion, for example from dietary sources in the post prandial phase. However, it also has the effect of smoothing out peaks and troughs and may hide episodic hyperoxaluria: it is the absolute oxalate concentration in the urine that affects lithogenesis, so it is possible to have a 24 h output within the reference range but still be at high risk of stone formation. 20 Another drawback to 24 h collections is that accurate timing is known to be problematic and their duration can often be either under or over 24 h in length. In addition, there are other factors affecting the reliability of a 24 h collection, such as the patient missing a urine pass, the quantity of acid added to the container or the possibility of other liquid being added. Oxalate:creatinine ratio is not subject to error from an inaccurately timed collection and can be calculated from a spot urine sample as well as from a 24 h collection, which is an advantage especially for children. However, as a ratio it is a composite parameter that includes two variables with associated imprecision. Although the added analytical variation from urine creatinine measurement is not great, there is a wider biological variation, including sex differences, which is evident from the large creatinine excretion reference interval (7–21 mmol/24 h in our laboratory). Oxalate excretion from a 24 h collection and oxalate:creatinine ratio calculated from a separate sample have previously been shown to be moderately correlated: using an early morning urine in stone formers 21 and using a random sample in a patient with PH. 22 In our data, both parameters were calculated from the same 24 h collection and better agreement might be expected because of the smoothing out of variability described above. We obtained a Pearson’s correlation coefficient of R = 0.63. Inspection of Figure 2 shows that there is a large scatter of results, even after exclusion of obvious under and over-collections. In particular, there are a large number (391) of samples with normal oxalate:creatinine ratio but elevated oxalate excretion (bottom right quadrant of the graph). Of these, just 38 (10%) are from women, which is much lower than the proportion in the entire dataset (Table 1).
Men had statistically significant higher excretion of both oxalate and creatinine than women in this dataset (Table 2). This finding has previously been reported in healthy individuals 23 and patients with renal stone disease, 24 and is a reflection of the larger average body size of men leading to a greater production of both analytes. Women had statistically significant higher oxalate:creatinine ratios than men (Table 2). This finding may be explained by the relative magnitude of the differences in oxalate and creatinine excretion already noted; whilst women had an on average 17% lower oxalate excretion, this figure was 33% for creatinine excretion. The reduced oxalate is more than offset by the reduced creatinine, so that the ratio of the two analytes is increased. It is possible therefore that many of the discordant pairs of oxalate:creatinine ratio and oxalate excretion results actually reflect variation in oxalate and creatinine excretion according to body size, which correlates on average with sex. Because body size data are not often available, the use of sex-related URLs as a surrogate could be considered. To estimate these, we calculated the 95th centile of the oxalate:creatinine ratio distribution, with elevated excretions excluded. The value for all samples (39 µmol/mmol) is very close to our current, non-sex related URL (38 µmol/mmol). As expected, amongst men the value was lower (33 µmol/mmol) and amongst women it was higher (45 µmol/mmol). These cut-offs were used along with the URL for oxalate excretion to categorise pairs of results (Table 3).
There were only a limited number (26) of 24 h collections from known PH cases aged 18 years or over in the dataset. When these results were categorised according to the cut-offs for oxalate excretion and oxalate:creatinine ratio, unsurprisingly the great majority (21) had both elevated. In fact, 7% of all results with both oxalate:creatinine ratio and oxalate excretion elevated were from known PH cases, compared to just 0.6% of results with only one parameter elevated. This finding suggests that an elevated oxalate:creatinine ratio and oxalate excretion in combination greatly increases the likelihood of PH, and that in the absence of secondary causes, further investigation is warranted. Our data also demonstrate that patients with PH may on occasion have urine oxalate concentrations within the reference range, a finding that has previously been reported. 25 These apparently normal results may reflect patients in renal failure, which may give misleading results as oxalate excretion is impaired and systemic oxalate deposition occurs. Alternatively, there are some patients who are particularly responsive to pyridoxine,26,27 a cofactor for the enzyme alanine:glyoxylate aminotransferase that is deficient in PH1. Two of the pyridoxine responsive cases had elevated oxalate results on another occasion, with no difference in treatment. This shows that repeat measurement of oxalate may be indicated as a single urine oxalate measurement within the reference range cannot exclude PH. Patients in whom there is a strong clinical suspicion of PH (for example recurrent or bilateral calcium oxalate urolithiasis or unexplained renal failure, especially in children) should have a repeat measurement of urine oxalate if a single value is within the reference range. However, our data show that if renal function is intact and in the absence of pyridoxine therapy, the combination of normal oxalate excretion and normal oxalate:creatinine ratio is not consistent with a diagnosis of PH.
It is highly likely that there are patients in the ‘Others’ group who have undiagnosed PH. It is our practice to highlight such cases on results review and advise on further testing. While the prevalence of PH is thought to be around 1 per million, 8 our data come from a population of stone formers and thus the prevalence in this cohort may be higher. We used the median urine oxalate from the PH group to define a cut-off for grossly elevated results. Using this cut-off, there were 24 patients aged 18 years or over in the ‘Others’ group with grossly elevated oxalate (both ratio and excretion), who are marked in Figure 3. This suggests that there may be a substantial number of individuals in whom the diagnosis has been overlooked. It is important to state that there is not an oxalate cut-off that can be used in clinical practice to diagnose PH, so our definition of ‘grossly elevated’ is only arbitrary. The rationale for picking this level is that it is sufficiently high to exclude many cases of secondary hyperoxaluria which is not usually so extreme, although overlap between primary and secondary disease has been shown 6 and enteric hyperoxaluria should therefore be excluded prior to further investigation for PH. A recent study showed that the median oxalate excretion of patients with enteric hyperoxaluria pre-treatment was 715 µmol/24 h, 28 which is similar to the median of PH cases in this study (771 µmol/24 h). However, in the absence of any clear secondary cause, a genetic basis should be sought, and it is likely that there are cases of PH within this group that have been missed or not fully investigated. The clinical consequence of failing to make a diagnosis is the risk of ESRF, which may be delayed by appropriate management of the patient. There is also the risk to other family members and possibility of incorrect transplant choices being made.15,29 PH is not excluded in any patient simply by urine oxalate results below this grossly elevated cut-off, or even within the reference range.
The above analysis of grossly elevated results does not include patients under 18 years of age, so we cannot comment upon the potential for missed diagnoses amongst children. As previously discussed, there is age-related variability in oxalate:creatinine ratio which affects interpretation and makes the use of a blunt cut-off inappropriate. Urine oxalate results on children should always be interpreted with reference to age-related reference ranges (and body surface area if available). The incidence of PH is higher for children, 7 and there should be a higher index of suspicion of PH in any child with calcium oxalate stones and/or unexplained renal impairment. However, adult presentation of PH does not necessarily equate to a less severe phenotype, 30 so that the use of urine oxalate testing to distinguish these patients from other stone formers is also important.
PH2 11 and PH39,12 are considered to have a potentially milder phenotype than PH1, although recent data suggest that the outcome may be just as poor for PH2 (Rumsby and Hulton, unpublished data). It might be anticipated that this would correlate with lower urine oxalate concentrations and this has been shown previously in some patient cohorts10,11 but not others. 12 Our dataset showed no statistical difference in oxaluria between the three types of PH (Figure 3). In patients with either PH1 or PH2, the oxalate:creatinine ratios ranged from within the reference range to grossly elevated, and although results from patients with PH3 were not as high, there was still considerable overlap with PH1 and PH2. Our results illustrate that urine oxalate itself is not a useful indicator as to which type of PH the patient may have. Currently, a definitive diagnosis can only be made by finding a known pathological mutation on gene sequencing, or by measurement of decreased enzyme activity on liver biopsy. A genetic diagnosis should be pursued in patients with suspected PH and family studies undertaken as appropriate.
Conclusions
This study highlights the variability in results of 24 h urine collections for urine oxalate, which may be a reflection of both pre-analytical errors (such as under and over collection) and physiological variation in excretion. Calculation of both oxalate excretion and oxalate:creatinine ratio helps to identify cases of genuine hyperoxaluria, and patients with both elevated are much more likely to have PH. Because of differences in production of oxalate and creatinine by body size, we now recommend the use of sex-related URLs for oxalate:creatinine ratio.
Patients with PH can have hugely variable urine oxalate concentrations by both measures, and there is no clear cut-off that can make the diagnosis with certainty. Our results suggest that differentiation between the three forms of PH is not possible based on urine oxalate measurement alone. It is advisable to obtain repeat measures from patients in whom the clinical suspicion is high, and then progress to genetic testing for a definitive diagnosis.
The high number of individuals with grossly elevated urine oxalate above the median of PH patients suggests that there may be cases of PH amongst adult stone formers that remain undiagnosed. If our results are extrapolated to other laboratories offering this service, there is likely to be a sizeable number nationwide. The finding of persistent hyperoxaluria in a patient should always be followed up because of the potential severity of a diagnosis of PH.
Footnotes
Declarations of conflicting interests
Not applicable.
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
Ethical approval
Approved by UCLH R&D department as an audit.
Guarantor
GR.
Contributorship
GR conceived the study, OCM analysed the data and prepared the first draft of the manuscript, CT contributed to assay performance and data interpretation, and all authors reviewed and edited the manuscript and approved the final version.
