Abstract
Background
The CELTIC ranges project aims to deliver a comprehensive range of reference intervals for commonly ordered laboratory investigations suitable for use in an Irish population as well as enabling comparison with relevant international studies. In this paper, we describe our methodology used throughout the entire project and present paediatric reference intervals for renal profile tests in plasma (sodium, potassium, urea and creatinine).
Methods
1023 children aged up to 17 years were recruited from our hospital’s general practitioner paediatric phlebotomy clinic. Clinical chemistry analyses were performed on the Roche modular system and statistical analysis was completed in line with CLSI guideline EP28-A3c.
Results
The plasma reference interval for sodium for ages 0.45–16.99 years was 137–143 mmol/L in 1000 subjects (combined genders). For plasma potassium, the corresponding ranges between 1 and 16.99 years (combined genders) were 3.6–4.8 mmol/L. Apart from neonates and in keeping with other studies, age partitioning for electrolytes was not required. Data for plasma creatinine (enzymatic methodology) and urea is also presented and, as anticipated, required partitioning for both age and gender.
Conclusions
Our renal profile findings are broadly consistent with those of international studies, for example, CALIPER, HAPPI, NORDIC, PRINCE and KiGGs. Moreover, the CELTIC ranges study is also based on over 1000 subjects whose samples were analysed on the widely used Roche modular analytics system. We also expect the findings will improve knowledge of children’s metabolic health in Ireland.
Introduction
Laboratory investigations in sick children are now increasingly common and usually consist of panels of biochemical and haematological tests such as renal, liver and bone profiles, immunoassays and full blood counts.1,2 Progressive improvements in laboratory technology to suit the unique characteristics of laboratory testing in children have enabled accurate and reliable results using ever diminishing volumes of blood. Results are not only useful to guide better diagnosis and treatment of the presenting illness but also potentially open up the possibility of earlier detection of a range of diseases including for example acute kidney injury3–6 or chronic kidney disease.7,8
Despite improvements in laboratory technology, a substantial area of difficulty remains because, unlike in adults, knowing what is normal 9 or whether the results represent pathological findings is not always clear-cut in children. Reference intervals (RIs) are essential tools used by clinical and scientific teams to interpret patient laboratory test results. 10 While population-based RIs can represent values expected in a typical community, laboratory results that fall outside RIs may not necessarily indicate a disease but rather the requirement for medical review. 11 However, accurate and comprehensive RIs are not always readily available.11,12 Significant difficulties have been reported with the development of paediatric reference intervals.13–23
Common methodologies in paediatric practice for development of RIs include: (1) collection of samples from healthy children, (2) use of residual samples (or additional sampling) on children attending a clinic or hospital where they require laboratory testing for a medical condition,2,23–26 (3) data mining techniques from established laboratory information systems11,12,27 and (4) transference studies. 11 The latter two options are dependent on the availability of the former two for comparison. 28
Relatively few studies in healthy school children have been conducted recently due to ethical considerations and high costs. For renal profile tests, studies are additionally complicated by anthropometric changes in growing children. Over recent decades, both height and BMI in pre-teens and teenagers have been increasing and age of menarche decreasing. High protein foodstuffs and drinks currently being promoted, as well as general nutritional improvements and lifestyle may also be contributing.29,30 These trends are certainly evident in Ireland as well as recent demographic changes in the population and cast doubt on the applicability of many ranges developed some years ago and in different demographic groups.
Determination of RIs by using leftover samples from children who have attended a hospital or clinic is a widely used technique which has the advantage of being less challenging from the ethical perspective. Specific statistical tools for RI have also been to assist with analysis in these studies. 31 Of course, the choice of clinic used to collect samples has a significant impact on the applicability of the results. Arguably, children attending any clinic are unlikely to be truly representative of the unselected healthy population at large and often show greater degrees of ‘abnormality’ than studies in healthy volunteers. Nevertheless, data from these studies are useful because children with test results outside the 2.5th–97.5th percentiles are far more likely to harbour a pathological abnormality than if they just fell outside the same percentiles of a healthy population. Clinically useful decision levels can also be derived more satisfactorily from these studies.
No comprehensive studies of paediatric reference ranges for common tests have been conducted in Ireland, apart from one study in 2013 which reviewed physical activity and cardiovascular risk including fasting blood lipid levels in 50 healthy Irish school children. 32
One of the key deliverables from the CELTIC ranges project will be a comprehensive set of RIs for the most commonly ordered laboratory tests in children. This paper describes the methodology for the entire project, including how the RIs were determined. In order to illustrate the methodology, we also present RIs for renal profile tests and include a commentary on how they compare with existing ranges available.
Materials and methods
The CELTIC ranges project uses a variety of methods for determining paediatric RIs, including indirect and direct methodologies as well as data mining and transference studies. The present report describes the methods and results for a prospective cross-sectional direct methodology where we collected residual and additional blood samples if required and in line with CLSI EP28-A3c guidelines. 28
Patient recruitment
Of all, 1023 participants were recruited from children aged up to 17 years who were referred by their general practitioner (GP) to the paediatric phlebotomy clinic at our hospital between May 2019 and February 2022. Patients who had known clinical or metabolic conditions or demonstrated signs of acute infection were excluded. Prior to agreement on participation, parents were asked for details of their child’s medical history including whether their child was feeling well now. GP request forms were also examined for clinical details. Any children with unresolved illness or symptoms (e.g., vomiting or fever) were not included. Likewise, any children with known medical conditions who attended GP Phlebotomy for routine monitoring blood tests were also excluded (e.g., endocrine disorder, coeliac disease, coagulation disorder, renal or liver disease, any chronic condition including genetic conditions, immunological or rheumatological disorder or medication history).
An explanation of the project and requirements were provided by a member of the team, backed up by separate information leaflets for parents and children (as appropriate). Interested parents provided informed consent by completing written consent forms, which were verified and countersigned by the CELTIC ranges team. Participants over 7 years of age also provided written consent following a series of age appropriate questions to ensure there was complete understanding of the project and the subsequent steps. No participants received any gratuity for participating in the study.
Data collection
Once consented, parents and participants completed a structured questionnaire with a member of the team. Information obtained included time of last meal, water consumption, screen time, exercise time, ethnicity, health history, neonatal vitamin D provision, birth weight, gestation time, sleep time and quality, wellness in the previous week, medication, vitamin and supplement use and fizzy drink consumption. Fasting was not required for participation.
Anthropometric measurements included height and weight using standard stadiometer® (Holtain Limited) and step-on weighing scales (SECA® Model 799 7021129). Participants under 2 years of age were assessed using body length (SECA®) and weight measured as child was lying down (SECA® Model 727). All instrumentation was in good working order and calibrated annually.
Phlebotomy procedure
All phlebotomy procedures took place in a private room by nursing staff trained in paediatric phlebotomy. Common distraction techniques such as television, singing and stickers were used. Younger participants were permitted to sit on their parent/guardian knee during the process.
Blood sampling was carried out in accordance with standardised phlebotomy procedures and hospital policy 33 using the Becton Dickson Vacutainer® Safety lok™ (25g) blood collection system. Blood samples were drawn into Vacuette® (Greiner Bio-one) tubes (3.5mL) in line with manufacturer’s instructions and in accordance with recommended order of draw. Optimal tube requirements were determined for the tests requested. EDTA samples were included in the study when requested by the GP.
Sample handling/storage
Consent forms, request forms and samples were sent to the central laboratory specimen reception for processing. All requests were assigned a laboratory accession number and recorded on the Laboratory Information System (Clinisys® version 5.32 SP 27 Build 135). Clinical chemistry requests included an additional CELTIC ranges profile at request entry to ensure all required analysis was completed. No additional tests were added for haematology analysis (if requested by clinician). All participants were assigned a unique study number.
Plasma/serum samples were separated following centrifugation at 3000 g for 5 min (Hettich Rotixa 50 RS®) at room temperature within 2 h of blood draw. Serum samples were allowed to clot for a minimum of 10 minutes prior to centrifugation. All samples were aliquoted using plastic Pasteur pipettes into Eppendorf® and Sarstedt® cryovials, labelled with a unique study number and frozen at −80°C. A separate fresh aliquot of plasma was sent to the clinical chemistry laboratory for the requested tests and CELTIC ranges agreed profile. The full CELTIC ranges panel included renal profile (sodium, potassium, creatinine and urea), liver profile (bilirubin, albumin, alanine aminotransferase and gamma-glutamyl transferase), bone profile (total calcium, albumin, alkaline phosphatase and phosphate), full lipid profile (cholesterol, triglyceride, low-density lipoprotein and high-density lipoprotein), glucose, haemoglobin A1c, amylase, vitamin D, aspartate aminotransferase and thyroid function tests (thyrotropin and free thyroxine).
Analytical procedure
Sample analysis was performed as part of routine laboratory service which is fully accredited to BS EN ISO 15189:2012 by the Irish National Accreditation Board (INAB www.inab.ie). All equipment and analytical methods are verified according to laboratory policy in line with CLSI guidelines (www.clsi.org). Equipment had been adequately maintained according to the manufacturer’s schedule. Calibration is performed after initial assay set-up on the analyser and each time a reagent kit with a new lot number is used, as per manufacturer's instructions. Quality control procedures are performed for each assay as determined by laboratory policy in line with manufacturer’s guidance, for each day that sample testing is conducted. Quality control results are examined by trained personnel to ensure that the values are within the recommended range for each control level. All samples in the clinical chemistry laboratory undergo automated interference analysis for haemolysis, icterus and lipaemia.
Summary of analytical methods for renal profile tests in plasma on the Roche modular system.
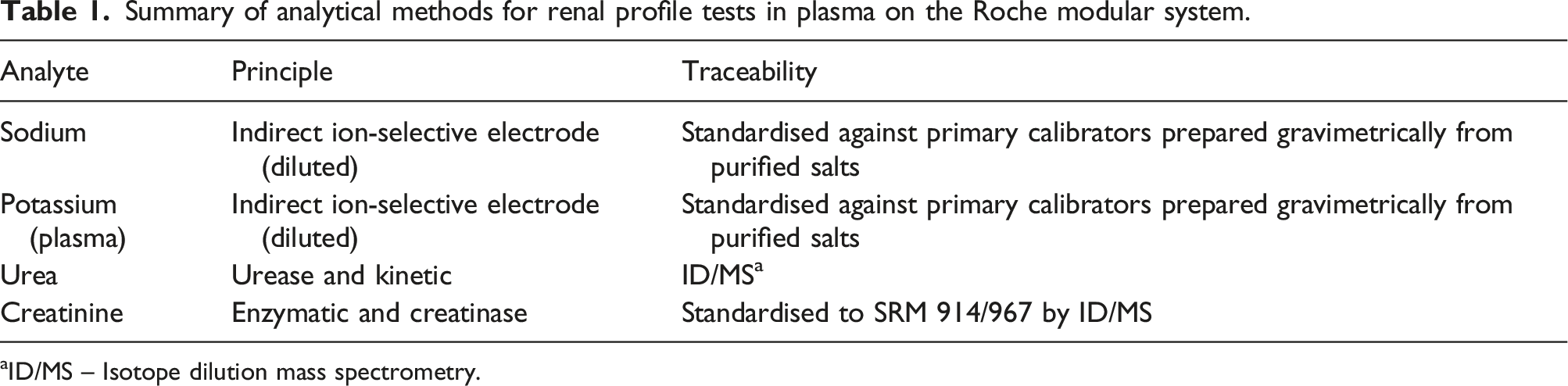
aID/MS – Isotope dilution mass spectrometry.
Clinical review
All haematology and clinical chemistry laboratory results were reported to the requesting clinician via the Laboratory Information System (LIS) in accordance with laboratory policy and procedures. Prior to release, all laboratory results were clinically reviewed and appropriate follow up was arranged where necessary.
Statistical analysis
Figure 1 illustrates the overall direct methodology process used in the project. All statistical analysis was performed in line with CLSI guideline EP28- A3c.
28
Analysis of data was performed with Microsoft Excel® 2013, IBM SPSS® version 29 and GraphPad® Prism version 8.4.3. Outline of the process employed for direct methodology in the CELTIC ranges project.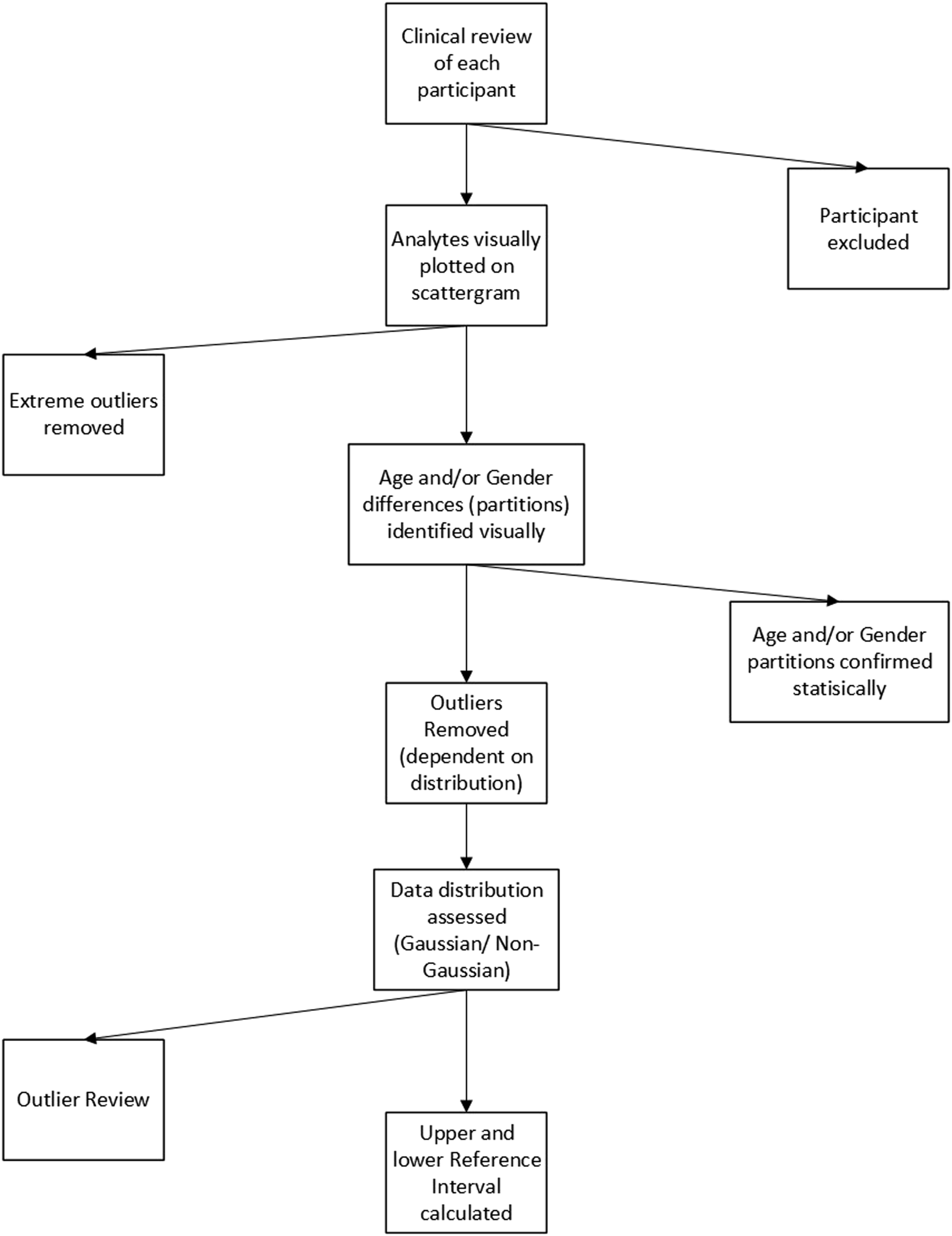
At the time of clinical review, participants that met the exclusion criteria were no longer included in the data set. Data verification clean up and review for completeness took place prior to any statistical analysis. This process included review of missing extreme and atypical laboratory results including review for transcription and contamination errors. All test results that were adversely affected by sample quality issues, for example, haemolysis and lipaemia were excluded.
Scatter plots and box-plots were initially used to determine extreme outliers by observation. Extreme outliers were confirmed through analysis by Dixon and Reed. 28 All data sets were assessed for normality and Gaussian distribution. If required, appropriate transforming techniques were applied. Age and gender partitions were initially determined by visual inspection of scatterplots of age against analyte concentration and based on physiological assumptions. Statistical significance of partitioning was determined using ANOVA 34 analysis and subsequently assessed by the Harris and Boyd method. 28 Partitioning was subsequently reviewed by clinical experts to determine if the partitioning should remain.
Once partitioning based on age or gender was complete, each partition was reviewed for distribution using normal Q-Q plots, histogram distribution and detrended normal Q-Q plots. Statistical analysis of Gaussian distribution was completed using the Kolmogorov–Smirnov statistic, skewness and kurtosis scores. If data was normally distributed, each partition was subjected to two rounds of outlier review using boxplots and the Tukey 28 test. Following outlier removal, data distribution was checked for the final time.
RIs are calculated for partitions with a sample size 120 using the nonparametric rank method. Confidence intervals (90%) were calculated using ranked observations.11,28 For partitions with sample numbers <120, the robust method of Horn and Pesce was used for reference interval calculation. The confidence intervals (90%) of the limits are calculated using percentile bootstrap estimates. 11 RIs were determined as the inter-centile range between the 2.5th and 97.5th percentile.
The methodology described here is being used for all analytes and RI analysis throughout the CELTIC ranges project.
Results
Of all, 1023 participants were recruited from the diversity of residents in the South Dublin region. Three participants with medical conditions were excluded from further analysis following clinical review. The female to male ratio was 530:485. The age range of the participants was 0.45 years to 21 years. Due to small numbers, participants over 17 years of age (Figure 2) were excluded. This resulted in a total of 1015 participants for further analysis. Participants were classified as fasting (458) or non-fasting (557) based on self-reporting. Overview of age of CELTIC ranges participants.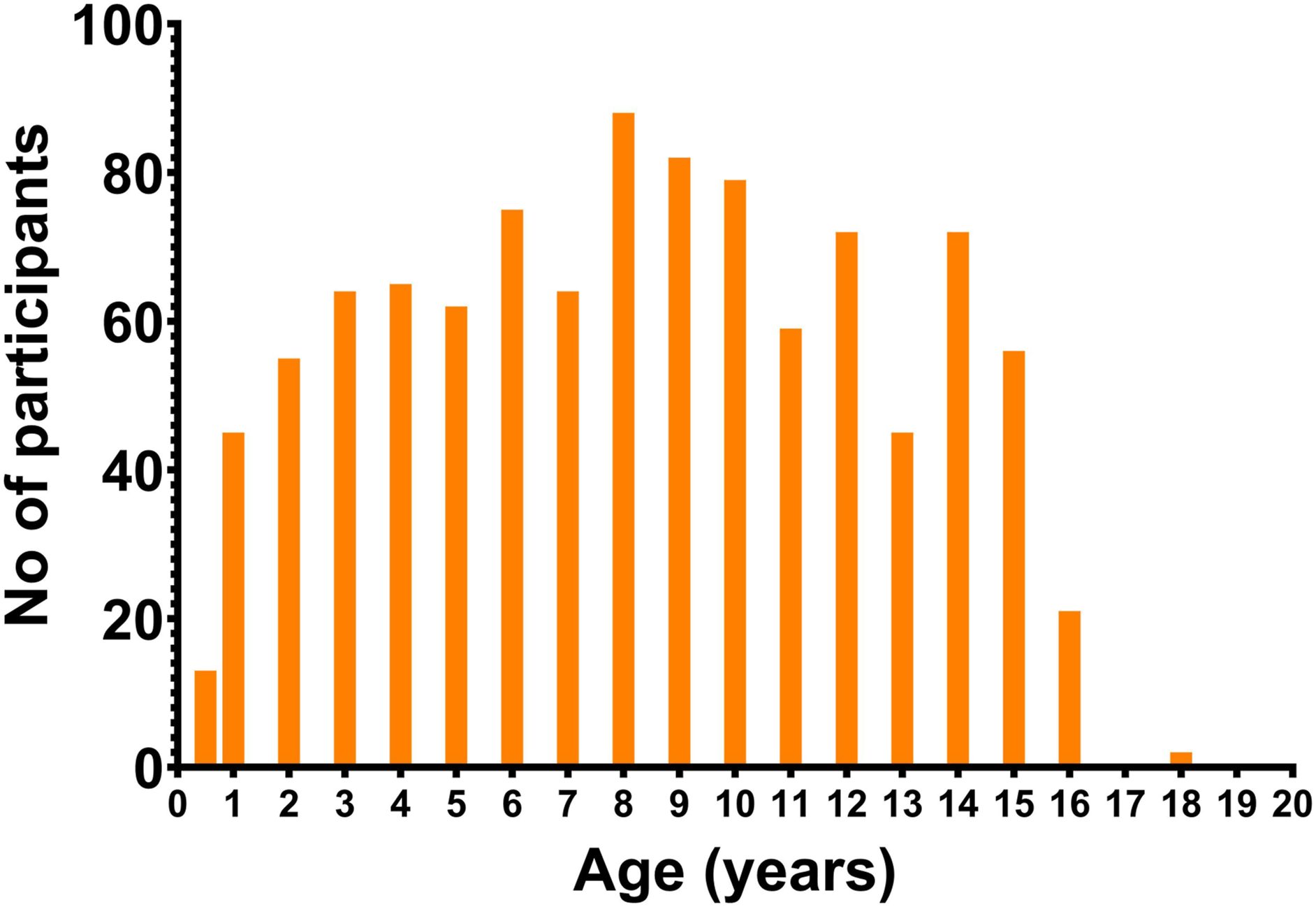
All partitions were subject to data review as outlined in Figure 1 involving visual assessment and statistical methodologies Figure 3. Final decisions on RI were based on clinical assessment. Age-related scatterplots for renal profile tests in plasma on the Roche modular system for the 1015 CELTIC ranges participants (female in pink and male in blue).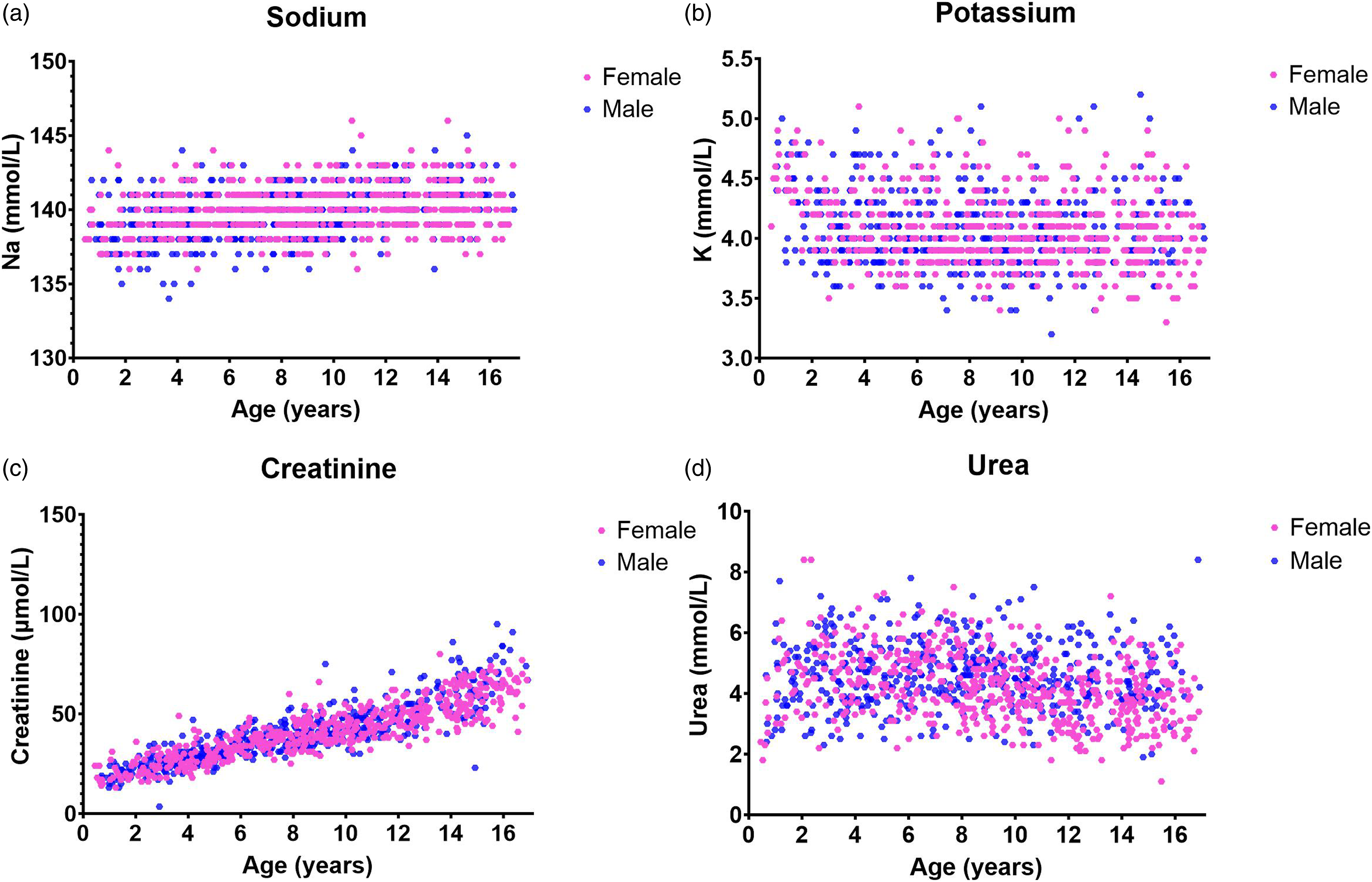
Sodium
Calculated CELTIC ranges reference intervals for renal profile tests in plasma (Roche modular systemTM).
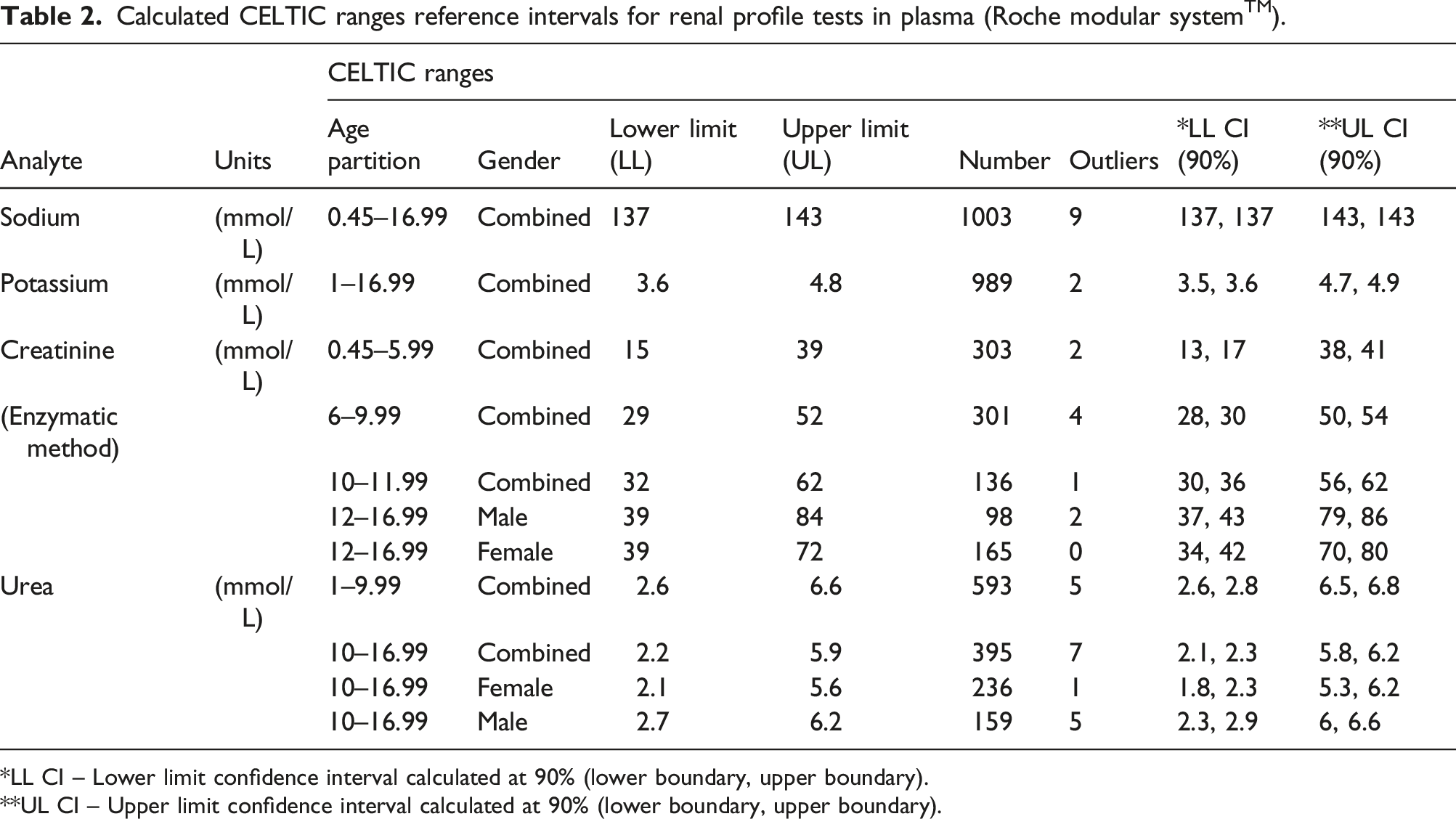
*LL CI – Lower limit confidence interval calculated at 90% (lower boundary, upper boundary).
**UL CI – Upper limit confidence interval calculated at 90% (lower boundary, upper boundary).
Potassium
Potassium also did not require any age or gender partitioning; however, participants <1 year old were removed due to low numbers, visual inspection and international evidence35–39 indicating the requirement for separate RIs for this cohort. RI calculated for age group >1 year to 16.99 years was 3.6–4.8 mmol/L with the final participant number being 989 after outlier removal. Sample size was large enough to compute RI using nonparametric methods (Figure 3).
Creatinine
Creatinine required considerable review and partitioning: 0.45–5.99 years, 6–9.99 years, 10–11.99 years and 12–16.99 years. These partitions are consistent with other studies.35–39 Gender partitioning was included in patients between 12 and 16.99 years. Sample sizes were large enough for all partitions to be calculated using nonparametric methodology (Figure 3). The confidence intervals of the limits of the nonparametric intervals for male cohort 12–16.99 years were determined using a bootstrap method.
Urea
Urea demonstrated variability in subjects less than 1 year of age on inspection due to low numbers and a decision was made to remove this cohort from further analysis (Figure 3). 1 Age partitioning resulted in two cohorts: 1–9.99 years and 10–16.99 years. Gender partitioning was included for participants greater than 10 years of age. However, a combined gender RI for 10–16.99 years is also provided. Sample sizes were large enough to compute RI using nonparametric methods.
Discussion
Establishment of reference intervals in a paediatric population is a time-consuming and cumbersome task, due to difficulty of obtaining blood samples from healthy children. Since the publication of the CALIPER study, there have been a number of additional studies published on RIs from direct/indirect methodology.21,26,27,36,37,39–42,44 The CELTIC ranges study is the first study of its size in Ireland which involved recruitment of participants attending a GP phlebotomy clinic. This allowed for use of results of requested tests, additional tests and residual samples. The unique study design, population selection, age and even distribution of study participants from 0.45 to <17 years facilitated estimation of RIs using a CLSI-based nonparametric statistical approach comparable to previous studies.11,36–39
This report outlines the RI determined for plasma sodium, potassium, creatinine and urea calculated from 1015 participants. Pre-analytical conditions for our participants were comparable to all children attending the phlebotomy clinic. Fasting for phlebotomy was not required unless indicated by requesting clinician. Physical exercise was not restricted and all phlebotomy procedures were completed by appropriately trained personnel in line with hospital procedure.
An additional challenge when establishing RIs in children is the ever-changing impact of growth and therefore many age- and gender-specific reference ranges may be required depending on the analyte. It is well known a-priori knowledge that there are no physiological events in childhood between the ages of 1 and 16 years that mandate the partitioning of plasma sodium and potassium concentrations. Sodium RIs established are similar to other studies available, even though the upmost age category differs across all studies quoted. Partitioning for age and gender was not required, in accordance with most other studies with the exception of the HAPPI study.26,35,37–39 Even so and despite advising on use of age-related calculated changes for sodium in the HAPPI study, Hoq et al. concluded these changes were not clinically significant and may not be required. 36 CELTIC ranges reported sodium RIs of 137–143 mmol/L for the age group 0.45–16.99 years. CALIPER reported 139–147 mmol/L for an extended age cohort of 0–<19 years. 35 Roche’s Instructions for Use (IFU) documentation does not provide separate paediatric RI for sodium. 43 KiGGS, 38 PRINCE 37 and NORDIC 44 reported comparable reference intervals to CELTIC ranges for sodium concentration.
As expected, potassium RIs also were not partitioned by age or gender after we excluded participants <1 years based on visual inspection.26,35,37–39 Other international studies also included a separate RI for children <1 year. 36 Ridefelt et al. reported separate RI for children <4 years for potassium when combining data from studies across Denmark and Sweden.45,37,38 The RI for CELTIC ranges in plasma on Roche modular systems was 3.6–4.8 mmol/L for age range 1–16.99 years. CALIPER reported a higher RI of 4–5.3 mmol/L for an extended age range of 1–<19 years. 35 Analogous RI of 3.3–4.6 mmol/L was reported in Germany for children >1 year. 37 Roche’s IFU for potassium provides three separate RIs: for less than 4 days, less than 2 years and less than 12 years. 43 Minimal changes are noted between those aged under 12 years (3.4–5 mmol/l) and over 12 years (3.5–5.0 mmol/L). A significant issue comparing results of potassium RI across studies is the selection of blood tubes. CELTIC ranges measurement is from lithium heparin plasma which has been shown to be 0.14–0.26 mmol/L (approximately 7%) lower than serum concentrations, 44 which causes difficulty in comparing RIs across international studies.
Inevitably creatinine and urea required significant age and gender partitioning reflective of the growth and development of children. Creatinine proved to be one of the most challenging analytes when assessing age- and gender-specific paediatric RIs. Sex-specific differences were demonstrated to be emerging during puberty and were statistically significant in a younger age cohort for urea as opposed to creatinine. This was comparable to data reported by the CALIPER study. 35 CELTIC ranges demonstrated a more complex pattern of change particularly in creatinine than other published studies.26,35,37–39 The current method in use involves enzymatic methodology as opposed to the often problematic Jaffe reaction. Colantonio et al. have previously demonstrated the lower results obtained in a paediatric population via an enzymatic method for creatinine, mainly due to the interference in the Jaffe reaction. 25 Roche’s current creatinine ranges, which were originally determined in 2002 by Schlebusch et al., 46 provide for separate RI for each 2 year age band between 1 and 15 years old. 47 No gender separation was provided unlike the CELTIC ranges cohort where clear segregation was noted both statistically and clinically from the age of 12 years. Our ranges also confirm a higher creatinine upper reference limit applies in the rapidly growing pre-teens and teenagers from 12 to 16.99 years (especially boys) where we propose up to 72 µmol/L for girls (N = 165) and up to 84 µmol/L for boys (N = 98) compared with (for the genders combined) up to 60 µmol/L (N = 42) for ages 11–13 years and up to 68 µmol/L (N = 38) for 13–15 year olds. 46 Also corroborated by CALIPER, we believe these higher values in CELTIC ranges more adequately reflect creatinine levels in today’s pre-teens and teenagers.
There has some scepticism with regard to the clinical significance of gender-specific RIs for urea. 36 Despite the fact, statistically significant partitioning was identified in the growing 10–17 age group, and the RIs are very similar and may be combined in clinical practice. Hence, we recommend combined urea ranges for the age group 10–16.99 years and also offer the gender-specific ranges for comparison. This tactic is also supported by the PRINCE 37 study which also did not provide gender-specific reference intervals. In the case of CALIPER, 35 separate gender-specific intervals were provided with an extended age range to 19 years. Roche’s IFU for urea just indicates a partition by age below 1 year without any gender partitions. 48
Conclusion
Our renal profile findings are broadly consistent with those of international studies such as CALIPER, HAPPI, NORDIC, PRINCE and KiGGs. Moreover, the CELTIC ranges study is also based on over 1000 subjects whose samples were analysed on the widely used Roche modular analytics system. We anticipate that CELTIC ranges will allow better understanding of physiological changes in renal profiles tests in growing children (particularly creatinine and urea) and will ultimately improve clinical interpretation of results in paediatric laboratory medicine.
Footnotes
Acknowledgements
The team would like to acknowledge CHI Ireland for their support in this study and in particular the phlebotomy team at paediatric General Practitioner clinic CHI Tallaght and the laboratory medicine department at Tallaght University Hospital. Finally, CELTIC ranges would like to thank all participants and their family without whom this valuable source of information would not be available. The team gratefully acknowledges a grant (reference 1713) from the National Children’s Hospital Foundation, a registered charity in Ireland.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by a grant (reference 1713) from the National Children’s Hospital Foundation, a registered charity in Ireland.
Ethical approval
Ethics approval was obtained for the study by the St James’s Hospital/ Tallaght University Hospital Ethics and Research committee. Approval included the use of residual blood (2017-07 Chairman’s Action (15)) and procurement of an additional 3.5ml serum sample from consented participants (2019-04List 15(11)).
Guarantor
GB.
Contributorship
AL and GB conceived the study, applied for ethics, recruited participants and reviewed results. TB participated in further patient MDT and referral. GB, TB and EM provided clinical governorship of project. AL and GB wrote first draft of manuscript. All authors reviewed and edited the manuscript and approved the final version for submission.
