Abstract
Background
Thyroid function tests (TFTs) are routinely requested by general practitioners (GPs) in the clinical biochemistry laboratory. Hypothyroxinaemia (low fT4) accompanied by TSH within the reference interval (RI) is a discordant pattern which is seen commonly in non-thyroidal illness and also as result of medications. Hypopituitarism is a lot rarer, but a serious condition the laboratory does not want to miss.
Methods
All thyroid hormone samples from primary care meeting the discordant case definition under investigation [fT4<10 pmol/L and TSH within RI (0.3–4.2 mU/L)] had partial anterior pituitary profiles [PAPP (cortisol, oestradiol/testosterone, prolactin, gonadotrophins)] added as reflex tests and results interpreted by a chemical pathologist. From January to June 2023, we conducted structured interviews with the requesting GPs, and, where indicated, requested repeat samples for full anterior pituitary profile [FAPP (PAPP, growth hormone (GH) and insulin-like growth factor 1 (IGF-1)]. We also reviewed the laboratory records of patients with previously known hypopituitarism to determine their fT4 and TSH values at diagnosis.
Results
Over the 6 months 41,487 GP TFTs were requested; 54 (0.13%) fitted the discordant case definition and had PAPP reflexed. 13 FAPPs were requested. We identified 3 cases of hypopituitarism. The number of additional tests required to diagnose 1 case of hypopituitarism was 129. In 74% of reflex-tested cases, there was a plausible explanation for the TFT pattern (medications, known thyroid dysfunction, non-thyroidal illness, pregnancy).
Conclusion
This study highlights the importance of medical liaison and early intervention in a biochemistry laboratory in identifying cases of unsuspected hypopituitarism.
Introduction
Thyroid function tests (TFTs) are one of the tests most requested by general practitioners (GPs) from the clinical biochemistry laboratory. In Ireland, the standard TFT profile includes both TSH and fT4. Irish national guidelines 1 recommend ordering TFTs in primary care in patients with signs and symptoms of thyroid dysfunction. Screening in asymptomatic patients is not recommended, particularly in patients under 50 years. Irish guidelines advise primary care physicians to discuss cases of secondary hypothyroidism with their laboratory consultant or endocrinologist in order for further investigations to be arranged prior to referral to Endocrinology. UK National Institute for Health and Care Excellence 2 recommends measuring thyroid stimulating hormone (TSH) alone when investigating thyroid dysfunction, unless secondary hypothyroidism is being considered. While NICE highlight that cases of secondary hypothyroidism require further investigation with a pituitary profile, UK guidelines suggest this testing is not carried out until under the care of an endocrinologist. 3 Similarly, the American Association of Clinical Endocrinologists and the American Thyroid Association recommend consulting an endocrinologist if there is a discordant pattern of TFTs. 4
Hypothyroxinaemia accompanied by TSH within the reference interval (RI), 5 is a discordant pattern which is seen commonly in non-thyroidal illness and also as result of medications. Hypopituitarism is a further differential diagnosis and, while rare, with an annual incidence rate of 4.21 cases /100,000, 6 it is a serious condition the clinical diagnostic laboratory is uniquely positioned to help identify. Early detection and treatment is essential, given the associated mortality with secondary adrenal insufficiency. 7 Common causes of hypopituitarism include pituitary tumours, pituitary surgery, head injury, cranial radiotherapy, hypophysitis, infiltrative conditions and medications (immune checkpoint inhibitors).7,8
Reflective and reflex laboratory testing protocols enable the laboratory to play an important role in the early identification of patients with hypopituitarism. Reflex testing refers to rules built into the laboratory information system that leads to tests being added automatically when predefined criteria are met. Conversely, reflective testing refers to further testing performed following review of the clinical findings and other laboratory results. While reflective testing to evaluate for hypopituitarism in those with hypothyroxinaemia exists widely in the United Kingdom, as evident from the recent national survey by McKeeman et al, 9 to our knowledge, Tallaght University Hospital and Naas General Hospital, where the authors have set this reflex system up in 2019, are the only hospitals in Ireland systematically using reflex testing for identification of hypopituitarism. We currently reflex test a partial anterior pituitary profile (PAPP) on all hypothyroxinaemic samples (fT4 <10 pmol/L) received on adult patients from primary care, where TSH is within the RI. However, the thyroid axis is traditionally described as being affected late in hypopituitarism, with the GH axis described as the first axis affected in cases of hypopituitarism. 7 PAPP comprises of tests which can be performed on the primary lithium-heparin sample (cortisol, prolactin, oestradiol/testosterone and gonadotrophins [follicle-stimulating hormone (FSH) and luteinising hormone (LH)] on our testing platform. We are unable to test growth hormone (GH) and insulin-like growth factor 1 (IGF-1) on the primary sample due to strict preanalytical requirements (sample must be separated within 4 hours and therefore, not achievable with primary care samples).
The most appropriate fT4 cut-off for investigating hypopituitarism has not been established. Data published on fT4 concentrations in patients at diagnosis of hypopituitarism have reported mean ± SD fT4 values of 7.7pmol/L ± 2.57 pmol/L, using the two-step Diasorin method (Diasorin, Stillwater, MN, USA) 10 . Of note, 18% of adults with newly diagnosed hypopituitarism in this study had normal TFTs, that is, fT4 and TSH within the reference interval. In our laboratory we use the one-step Roche Cobas Elecsys fT4 method (Roche Diagnostics, Mannheim, Germany). As fT4 assays are not yet standardized, fT4 values between different methods are not interchangeable and cannot be directly compared. 11 By reviewing the outcomes from our current practice and incorporating a review of discordant TFT patterns in patients with hypopituitarism, we aim to locally evaluate the clinical utility of reflex testing for hypopituitarism.
Methods
From January to June 2023, we prospectively evaluated all primary care patients’ samples with fT4<10 pmol/L (RI 12–22 pmol/L) and TSH within RI (0.3–4.2 mU/L) in whom partial PAPP tests were added (cortisol, oestradiol/testosterone, prolactin, gonadotrophins), as per standard practice in Tallaght University Hospital laboratory. Our Clinical Chemistry laboratory is an ISO 15189 accredited testing laboratory. This study aimed to evaluate the efficiency of our clinical pathway. Pituitary hormones were analysed by immunoassay on the Roche Cobas 8000 e801 platform (Roche Diagnostics, Mannheim, Germany), while GH and IGF1 were analysed by immunoassay on the IDS-iSYS instrument (IDS-iSYS, Immunodiagnostic Systems, Boldon, UK). Our fT4 method is the one-step Roche Cobas Elecsys, fourth generation (Roche Diagnostics, Mannheim, Germany). All analytes are measured in vacuette lithium heparin blood collection tubes. Internal quality control and external quality assessment (EQA) performance was satisfactory throughout the study period for all hormones. The FAPP hormone panel is enrolled in RIQAS and UK NEQAS EQA schemes. Chemical Pathology Specialist Registrar (SpR) reviewed request forms, patient letters and results. The SpR then phoned all the requesting GPs and conducted a structured interview to determine the reason the test was ordered, symptomatology, past medical history, existence of illness in the weeks preceding thyroid hormone level testing, sampling time and medications.
In cases where hypopituitarism was biochemically and clinically highly likely, in discussion with the GP, repeat testing of a full anterior pituitary profile (FAPP), under optimal conditions, was performed by testing an early morning fasting sample for fT4, TSH, FSH, LH, oestradiol/testosterone, cortisol, prolactin, GH and IGF-1. Where cortisol was very low [morning cortisol <115 nmol/L (local decision)], hydrocortisone replacement in physiological doses was advised (10 mg upon awakening, 5 mg at 14:00).
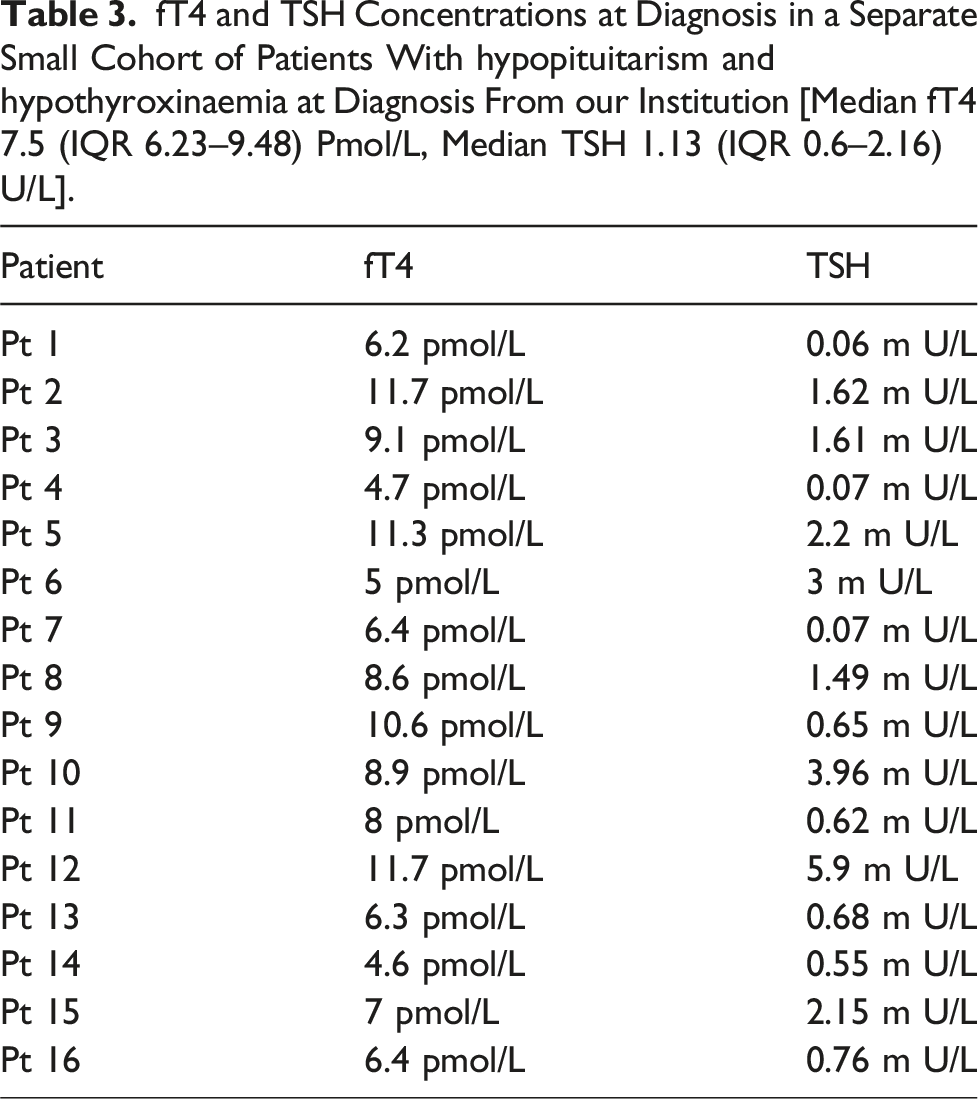
In addition to the above, we reviewed the laboratory records of a previously diagnosed cohort of 16 patients from our unit with known hypopituitarism and hypothyroxinaemia (based on imaging and dynamic function tests), to determine their fT4 level at diagnosis.
Results
Most Likely Cause of hypothyroxinaemia in 54 Primary Care Patients Evaluated by reflex PAPP and FAPP

In three patients, we advised hydrocortisone replacement therapy (10 mg upon awakening, 5 mg at 14:00) until review by an endocrinologist. While IGF-1 was within the reference interval in all cases in whom hypopituitarism was excluded, it was low in only 1 of the 3 hypopituitarism cases.
The aetiology for discordant TFTs was undetermined in 11 patients. In 5 of these cases, repeat TFTs were suggested (if minor perturbation noted in another hormone axis or low cortisol due to afternoon sampling time). TFTs were found to have in 4 out of these 5 cases. In the one case where the TFTs remained discordant, the patient was referred to endocrinology. Furthermore, low cortisol results (morning cortisol <300 nmol/L) due to afternoon sampling times all normalized on repeat 09:00 sample. This cortisol cut-off is based on local consensus opinion.
Repeat TFTs were not suggested in the remaining 6 undetermined cases as all other hormone axes were robust. These cases were reported as ‘No evidence to support hypopituitarism’.
Results of Full Anterior pituitary Screen in Patients With Confirmed hypopituitarism.
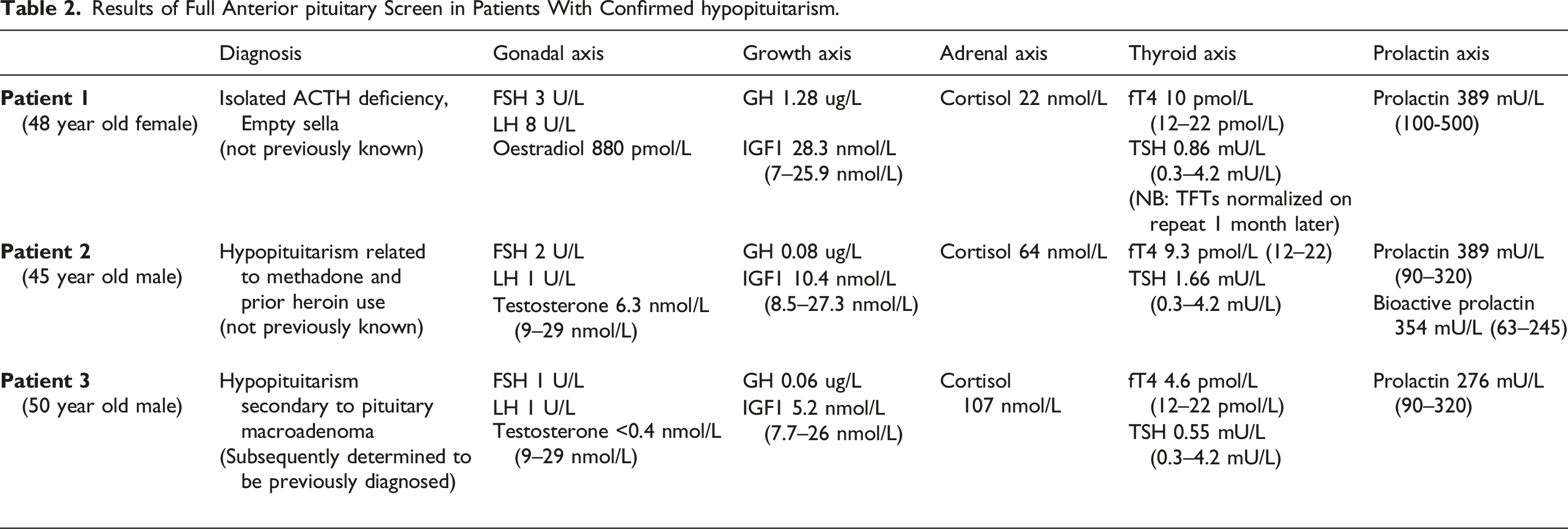
Furthermore, we identified 4 patients with isolated suboptimal cortisol concentrations on 09:00 samples (morning cortisol <300 nmol/L) requiring endocrinology referral for short Synacthen test. Following review by endocrinology, 2 of these 3 patients were deemed not to require a short Synacthen test but are due to have a repeat pituitary profile in 4–6 months. One of these patients had a short Synacthen test performed and passed it, that is, 30 min cortisol >450 nmol/L. In this patient, the 09:00 cortisol was 214 nmol/L. The Synacthen test result was 165 nmol/L for cortisol at time 0 and 708 nmol/L at 30 min. The fourth patient has also been reviewed by endocrinology and is awaiting MRI pituitary and a short Synacthen test at time of paper submission.
After excluding hypopituitarism by PAPP and FAPP, the aetiology of hypothyroxinaemia was attributed to medications in cases where the patient was taking medications known to cause this biochemical pattern – antidepressants, antipsychotics and antiepileptics.3,12 Following the structured GP interview, we classified patients as having a known thyroid dysfunction if they had a history of established thyroid disease and were on either thyroid replacement or antithyroid treatment. In those on thyroid hormone replacement (levothyroxine), previous TFTs were also reviewed to confirm the accurate diagnosis of hypothyroidism, that is, at least one high TSH level in the past (≥10 mU/L). 13
fT4 and TSH Concentrations at Diagnosis in a Separate Small Cohort of Patients With hypopituitarism and hypothyroxinaemia at Diagnosis From our Institution [Median fT4 7.5 (IQR 6.23–9.48) Pmol/L, Median TSH 1.13 (IQR 0.6–2.16) U/L].
Discussion
The merits of a laboratory testing protocol to further evaluate cases of hypothyroxinaemia are evident: we identified 3 cases of hypopituitarism over a 6-month period in a catchment population of approximately 450,000. While one of these cases was subsequently determined to be a known patient with hypopituitarism who had stopped taking their medication, it further demonstrates the protocol in place is robust in detecting patients with hypopituitarism. Two new cases of clinically confirmed hypopituitarism were clinically unsuspected: both patients had TFTs ordered routinely by their GP, were clinically euthyroid and there was no clinical suspicion of either a thyroid or a pituitary dysfunction.
Reflex testing refers to rules built into the laboratory information system that leads to tests being added automatically when predefined criteria are met. Conversely, reflective testing refers to further testing performed following review of the clinical findings and other laboratory results. The benefits of reflective testing by medical laboratories are well documented, including the recent randomized controlled trial on reflective testing by Oosterhuis et al.14–16
The fT4 cut off <10 pmol/L used in this reflex laboratory testing protocol is based on our calculated uncertainty of measurement, marking the level 2 standard deviations below the lower reference limit of 12 pmol/L. We also calculated the reference change value (RCV) with the 99% probability of unidirectional change. RCV was 16.9%, which also landed at the <10 pmol/L value 17 . The clinical utility of the fT4 RCV in assessing real change (critical difference) has been confirmed previously by D’Aurizio et al. 18
We evaluated our hospitals cohort of patients with hypopituitarism and found only 16 with secondary hypothyroidism at diagnosis, approximately 16 %. While these patients’, median fT4 at 7.5 (IQR 6.23–9.48) pmol/L was lower than our cut-off level, several patients with higher fT4 at diagnosis would have been missed even with the fT4 cut-off level for triggering reflex testing set at <10 pmol/L. Interestingly, our findings differ to that reported by Alexopoulou et al, 10 regarding the presence of abnormal TFTs in adults with newly diagnosed hypopituitarism (16% vs 82%).
The number needed to diagnose refers to the number of additional tests required to diagnose one new case and is used in illustrating the clinical effectiveness of testing.9,19 The number of additional tests required to diagnose 1 case of hypopituitarism in this survey was 129.
This study has demonstrated automatic reflex testing in cases of hypothyroxinaemia is not entirely efficient. In the majority of cases (74 %) automatically reflex tested by the laboratory, a plausible clinical cause was noted (known thyroid dysfunction, non-thyroidal illness, pregnancy, medications). However, in the case of medications (No. = 15), we would argue anterior pituitary testing is still indicated initially prior to attributing the low fT4 to medication. Based on this, automatic reflex testing of an anterior pituitary profile by the laboratory in the remaining 25 patients resulted in 125 unnecessary tests. Reflective testing, including a conversation with the primary care physician as undertaken here, collecting data on past medical history, symptoms, pregnancy status and medication use would result in a more efficient laboratory service. Had reflective testing been the model used over the 6-month period, the number of additional tests required to detect 1 case of hypopituitarism would have been 87.
Many commonly prescribed medications have been shown to be implicated in the biochemical pattern of hypothyroxinaemia. These include antipsychotics, antidepressants, antiepileptics, dopaminergic medication and glucocorticoids.3,12,20 The various classes of drugs exert their effects by different mechanisms. Dopaminergic medication and glucocorticoids can suppress TSH while antiepileptics can increase hepatic metabolism of thyroid hormone. 3 In the 28% of cases ultimately attributed to medication, it is the opinion of the authors that full anterior pituitary testing should be performed at least once to exclude an underlying pituitary condition potentially masked by a hypothyroxinaemia-inducing medication.
Biochemical assessment of the GH axis can aid in the identification of cases of hypopituitarism, given it is traditionally described as the first axis affected in cases of hypopituitarism. 7 Due to strict preanalytical requirements (sample must be separated within 4 h and therefore, not achievable with primary care samples) GH and IGF-1 tests were not included in our reflex partial anterior pituitary profile.
The clinical utility of adding GH and IGF-1 as part of repeated full anterior pituitary profile in select cases, where the pre-test probability for hypopituitarism is high, was also explored in this survey. Based on the survey’s findings, assessment of the GH axis was not particularly sensitive in detecting hypopituitarism in patients with hypothyroxinaemia, with only one patient with hypopituitarism having a low IGF-1. However, the very small number of hypopituitarism cases is a noteworthy limitation. The pulsatile nature of GH secretion is a further limitation. In the 10 patients in whom hypopituitarism could be excluded based on full anterior pituitary testing, IGF-1 levels were within the reference intervals in all cases.
The survey demonstrated a large number of patients (20%, No. = 11) with no clear cause to their discordant TFT pattern identified. In 5 of these cases, repeat TFTs were suggested (if minor perturbation noted in another hormone axis or low cortisol due to afternoon sampling time). TFTs were found to have normalized in 4 out of these 5 cases. In the 1 case where the TFTs remained discordant, the patient was referred to endocrinology. Furthermore, low cortisol results due to afternoon sampling times all normalized on repeat 09:00 sample.
Repeat TFTs were not suggested in the remaining six undetermined cases as all other hormone axes were robust. The included reflective TFT algorithm (Figure 1) is intended as an aid to interpretation in discordant TFT cases (low Ft4, TSH within RI). This algorithm has been developed based on our learning from this study and will be in use in our laboratory going forward. Preiss et al
21
report their protocol for investigating patients with discordant thyroid function tests is to first repeat the fT4 after calibrating the assay to reduce the risk of an analytical issue. Interestingly this brought 54% of their low fT4 samples into the reference interval. While the fT4 assay in our laboratory is calibrated as required, it is not specifically performed when a hypothyroxinaemia pattern is observed. However, on review of all cases who had FAPP and therefore repeat TFTs within a very short time interval (approximately 2 weeks), the discordant TFT pattern remained in all but 1 case. Sample analysis on an alternative two-step fT4 assay is not included in our protocol. Based on our institution’s experience, analysis of fT4 samples with concentration values in the range 10–12pmol/L were comparable to that obtained on the Abbott Architect fT4 two-step assay (Abbott, Chicago, IL, USA) (data not shown). Strengths of this study include its prospective nature as well as its focus on the patient’s clinical presentation, by telephoning all GPs. The small sample size and short duration are recognized limitations. Proposed reflective algorithm for investigating hypothyroxinaemia.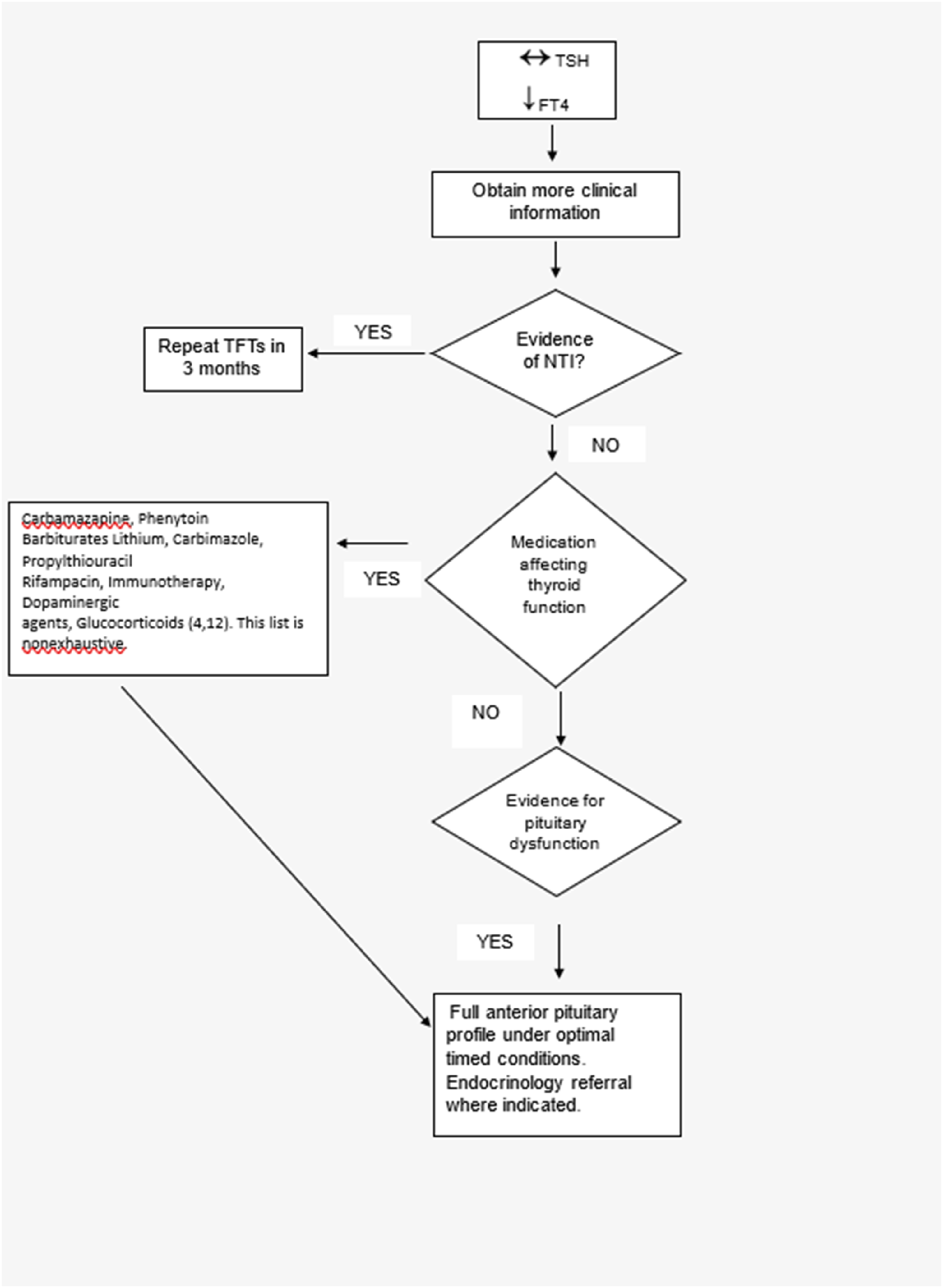
Conclusion
The biochemistry laboratory has an important role to play in identifying cases of hypopituitarism based on the discordant TFT pattern of low fT4 with TSH in the reference interval. This study highlights the importance of liaison and early intervention by laboratory medical staff. Reflective pituitary testing is a more efficient model of clinical liaison than reflex testing. We encourage other medical laboratories to adopt a similar reflective model to aid in identifying cases of hypopituitarism in primary care and improve patient outcomes.
Footnotes
Acknowledgements
The authors would like to acknowledge the help of the staff in the Department of Chemical Pathology, Tallaght University Hospital.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethical approval
Not required for the study.
Guarantor
GB.
Contributorship
CF conducted structured interviews with General Practitioners and wrote the first draft of the paper. All authors reviewed and edited the manuscript and approved the final version of the manuscript.
