Abstract
The purpose of laboratory medicine is to facilitate better decision making in clinical practice and healthcare delivery. Decision making implies an unresolved issue, problem or unmet need. The most important criterion for any investigation to be of value in clinical practice is that it addresses an unmet need. The different ways in which laboratory investigations are utilized in patient care can be represented in the form of questions. It is important that these questions are articulated to highlight the variables that will impact on the effectiveness of the investigation in the scenario being considered. These variables include the characteristics of the patient (or population) and clinical setting, the nature of the decision and action taken on receipt of the test result and the expected outcome. Asking a question is the first step of the evidence-based laboratory medicine (EBLM) cycle, the other steps being acquiring the evidence, critically appraising the evidence, applying the evidence and auditing use of the evidence. Getting the question right determines the quality of the whole process, thus, defines the quality in practice of laboratory medicine. Whilst the main focus of the EBLM cycle is to provide a strong evidence base for use in clinical practice, it is clear that the five steps are equally applicable in commissioning, delivery and audit (performance management) of services. Asking the right question is crucial to improving the quality of evidence, and practice, in laboratory medicine, and should be used in routine laboratory medicine practice and management throughout healthcare.
Introduction
‘A prudent question is one-half of wisdom’.
Francis Bacon
The care of patients is a multi-professional team activity with continuing calls for a more integrated approach to healthcare. 1 At the core of this activity are the needs and values of those involved in delivering, and receiving, care – and the information that is shared between them; optimizing patient outcomes depends on the quality of the information that is available, the plan for translation of this information into clinical action and the effectiveness of communication. 2 Asking the right question improves the quality of communication between individuals, a point recognized in the quotation at the head of this article. Thus to provide the correct information, laboratory professionals must be aware of the questions that are posed by clinicians, which can be broadly categorized as those that (i) enhance their dialogue with patients and (ii) deliver the best knowledge on how to effectively deal with a specific clinical situation. For example, patient-caregiver communication is recognized as an important part of the care of patients with cancer. 3 Ely et al. 4 developed a taxonomy of generic questions asked by the primary care physician that could be used to help understand clinical information needs and improve our ability to meet those needs and answer such questions. The three most common generic questions reported were (i) ‘What is the drug of choice for condition x?’, (ii) ‘What is the cause of symptom x?’ and (iii) ‘What test is indicated in situation x?’. However, the importance of asking the right question is not limited to the patient-carer interaction and extends across all aspects of healthcare from patient involvement, through education, research and clinical practice to management and policymaking. 5 –10
The Oxford English Dictionary describes a question as (i) ‘a sentence worded or expressed so as to elicit information’, (ii) ‘a problem requiring resolution’, or (ii) ‘a matter or concern depending on conditions’. 11 Therefore, asking a question involves clearly articulating the nature of a problem. In medicine, questions have been described as background questions or foreground questions; 12 the former relates to the knowledge surrounding the problem (and in the context of laboratory medicine, e.g. the pathology of the disease), whilst the latter relates to the application of the knowledge. An example of the former would be ‘why does the serum C-reactive protein concentration rise in rheumatoid arthritis?’ An example of the latter would be ‘will the measurement of C-reactive protein be helpful in the diagnosis of rheumatoid arthritis?’, or ‘will the measurement of C-reactive protein be helpful in the management of rheumatoid arthritis, e.g. for the assessing the effectiveness of treatments?’ It is the latter style of question that is most important in the delivery of healthcare, including laboratory medicine. 13 –15 This resonates with the points made by Glasziou et al. 16 that Evidence-Based Medicine is about ‘…. trying to improve the quality of the information on which decisions are based ….’ and ‘…. thinking not about mechanisms but about outcomes….’.
Richardson et al. 17 concluded that the majority of questions asked in clinical practice were about (i) ‘how to gather clinical findings properly and interpret them soundly’ (clinical evidence), (ii) ‘how to select and interpret diagnostic tests’ (diagnosis), (iii) ‘how to anticipate the patients’ likely course’ (prognosis), (iv) ‘how to select treatments that do more good than harm’ (therapy), (v) ‘how to screen and reduce the risk of disease’ (prevention) and (vi) ‘how to teach yourself, the patient and the family what is needed’ (education). Crucially these authors stressed the importance of employing a ‘well-built’ clinical question, now common practice in many areas of medicine. 18 –20
The evidence-based laboratory medicine (EBLM) cycle
There are five steps in the practice of evidence-based medicine proposed by Strauss and Sackett:
21
(i) converting the need for information by asking questions that are clinically relevant and answerable, (ii) finding the best evidence to answer the question, (iii) critically appraising the evidence for its validity and usefulness, (iv) applying the evidence to clinical practice and (v) audit, evaluating your performance. These steps have been adapted for use in laboratory medicine as the EBLM cycle (Figure 1)
13,14
and modified to add an ‘Analyse’ step between the Appraise and Apply step for Laboratory Medicine Best Practice systematic reviews.
15
Christenson et al.
15
have advocated the addition of the ‘Analyse’ element for aggregating the data from several studies when ‘Appraise’ has focused on data from individual studies, whereas it can be considered as part of the ‘Appraisal’ step. The ‘Analyse’ step was also introduced by Kingsnorth et al.
22
; however, they were using an ‘Ask – Acquire/Appraise –Apply – Analyse –Adapt/Adopt’ cycle, in which the ‘Analyse’ element was applied to the output from the application of the results from the ‘Acquire/Appraise’ step which could be considered as part of the ‘Assess’ step in the cycle described in Figure 1. The objective of laboratory medicine is to assist in solving clinical problems and facilitating clinical decision making and so the use of laboratory medicine is driven by the existence of a problem, which can be articulated in a question – or a problem statement. There are several ways of preparing a problem statement, probably the simplest being (i) what is the problem? This should explain why an answer is needed; (ii) who has the problem? This should explain who needs the solution, in what clinical setting, and who will decide the problem has been solved; and (iii) how might the problem be resolved? This will describe the solution and effectively, the business case for its adoption. The exact nature of the question will, however, depend on who is asking the questions, e.g. patient, doctor, manager, policymaker, etc.
The core of the EBLM cycle.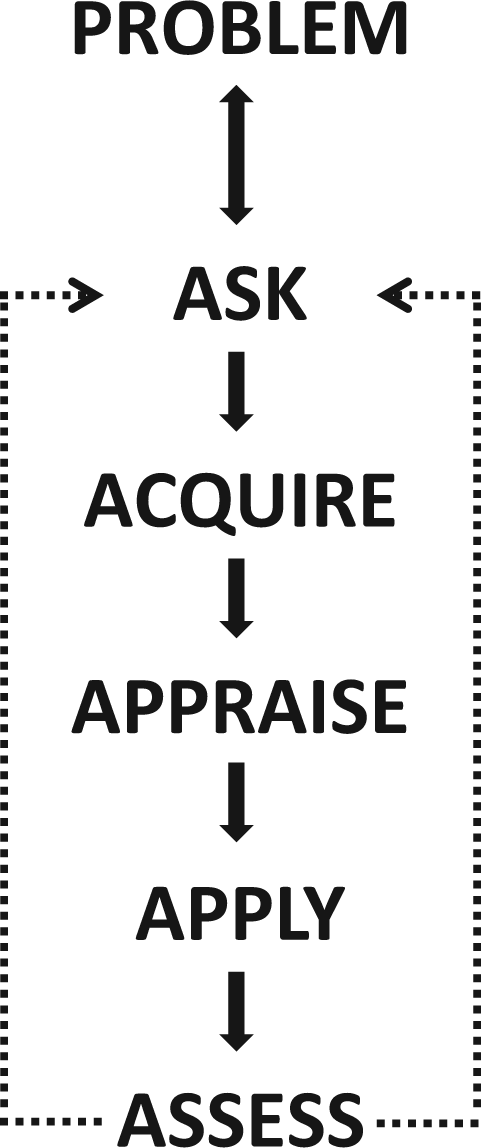
The structured question
Types of questions, the generic PICO elements and examples of structured questions relevant to laboratory medicine.
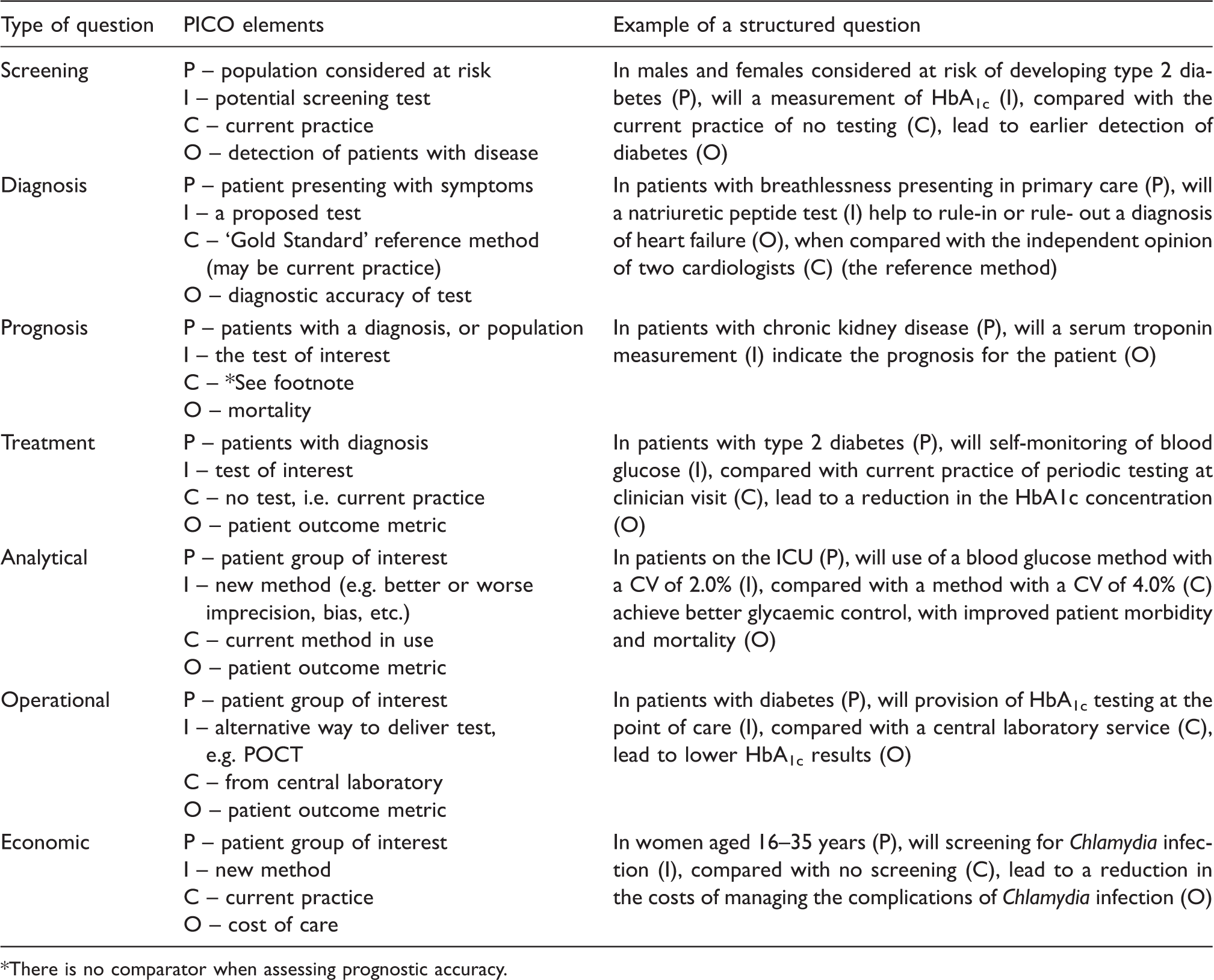
There is no comparator when assessing prognostic accuracy.
The PICO framework for the development of a structured question is also employed in the assessment of evidence for the development of clinical practice guidelines advocated by the GRADE Working Group. 26 The reader is referred to the discussion by Brozek et al. 27 regarding the GRADE approach to grading of evidence for diagnostic tests, and a specific example is described by Hsu et al. 28
Tips on formulating questions
This is not an exact science; however, there are a few points that offer guidance:
Succinctly describing the patient or population of interest is vital, as test results change with the evolution and progression of disease. Failure to do so risks leaving the study open to patient spectrum bias. For example, it is well known that patients presenting in primary care, when their condition may not yet be symptomatic or severe, are likely to have a different spectrum of results compared with those presenting to the Emergency Department when their condition is urgent (e.g. natriuretic peptide levels in breathless patients).
29
Similarly results may vary according to gender, age or lifestyle. Specifying the characteristics of the patient or population is essential; When asking a question about risk stratification, e.g. assessing prognostic performance of a test, the comparator is not always used. For example, formulating a question about the prognostic value of increased natriuretic peptide values in a population will likely not include a comparator. This is because natriuretic peptide results are a continuous variable so some of the cohort will have very low values and others comparatively higher values. Comparing the different natriuretic peptide ‘dose’ levels in the cohort and the association with outcomes will serve the comparative function. However, it is frequently helpful to indicate whether it is mortality or morbidity that is documented, and at a fixed time (see below for comment on time); Outcomes can be considered in terms of impact on the patient, the care delivery process (operational) or the cost (economic); As noted earlier when assessing the impact of a test on outcomes, it is important to remember that the test does not exist in isolation and is only effective coupled with the treatment. So, in addition to it being a ‘test-and-act’ intervention when evaluating evidence or practice, it is important to ensure that both test and treatment are delivered appropriately; In a treatment question, a validated surrogate outcome measure is often used, e.g. HbA1c in relation to glycaemic control and as a practical alternative to morbidity and mortality; The analytical question typically examines the effect of differences in method performance, e.g. imprecision or inaccuracy, although the framework can limit elegant construction of the question. In other words, in order to investigate the relationship between method imprecision (or inaccuracy) and outcome, it will be necessary to define issues such as total error (bias + [2SD]) and therefore a range of imprecision and bias values should be considered. A relevant analytical question is to define the intervention in terms of a particular assay characteristic compared with current practice. An example is to ask if there will be benefit to patients presenting with signs and symptoms of cardiac ischaemia by implementing a high-sensitivity troponin method with superior imprecision compared to use of contemporary sensitive assays that are currently the state-of-the-art methodology; The operational questions can look at impact on patient outcome or on process outcome, e.g. length of stay, hospital visits, re-hospitalization, emergency admissions – all valid surrogate process outcomes.
The issues and questions provided above are only intended as examples; further examples can be found in the included reference. 14 It is worth stressing that the objective of the structured question is to improve clarity and specificity in the understanding and communication of the problems and their resolution in the various facets of professional practice. 15
Variants of the PICO
There have been additions to the PICO framework described, all with the intention of providing greater clarity to articulation of the problem. Sometimes an
EBLM and relevant questions
There are five stages in the EBLM cycle, starting with asking the question (Figure 1). The basic structure should be viewed as a problem resolution cycle and in so doing it can be applied to a wide range of activities involving laboratory medicine (Table 2). Thus whilst the principles of evidence-based medicine (from which EBLM has evolved) were originally focussed on the interaction between patient and clinician (and associated education and research), this culture of evidence-based practice can be applied to all aspects of professional activity in laboratory medicine from strategic thinking and commissioning of services,
30
to daily practice, quality improvement
31
and organizational research
32
– as well as personal development through education, training and research. The concordance of the steps in the EBLM cycle with the steps in commissioning and performance management (audit) of diagnostic services is illustrated in Figure 2. Thus in the case of audit (the right-hand column), it is important to understand the process being audited and so the question is crucial. It is then important to understand the evidence base that underpins the utility of the test and the ‘diagnostic process’ being reviewed, e.g. if auditing the use of a test you need to understand why the test is being used, the patient characteristics, the clinical setting – as well as the decisions and actions informed by the result. This helps to inform the data collected for the audit. The data are then reviewed in the context of the evidence base and the clinical guideline (protocol) for that test’s utility(ies), and practice modified according to the outcome of the data review. This cycle can then be used to design local performance management and continuous quality improvement initiatives – linking closely to the broader annual review and strategic planning cycle. The emphasis then transfers to the commissioning (left-hand column) cycle, where the use of the laboratory medicine service to address unmet commissioning or providing needs should be considered, e.g. the use of point-of-care testing to address length of stay in the Emergency Department or in the management of patients with long-term conditions in primary care. Establishing the need is followed by development of the business case and specification of the service – informed by the evidence base, including an economic assessment. Monitoring adoption, in essence performance management of the service, has many of the similarities to audit as noted earlier. Successful completion of each step of the EBLM cycle in all of these activities is predicated in each case on the clarity with which the problem and question to be addressed are articulated.
Illustrating how asking the right question is the first step in the practice of EBLM (inner column), and in commissioning (left column) and audit (performance management) (right column) of laboratory medicine services. Illustrating the range of health professionals asking questions, the settings in which they may be asked and range of issues being addressed.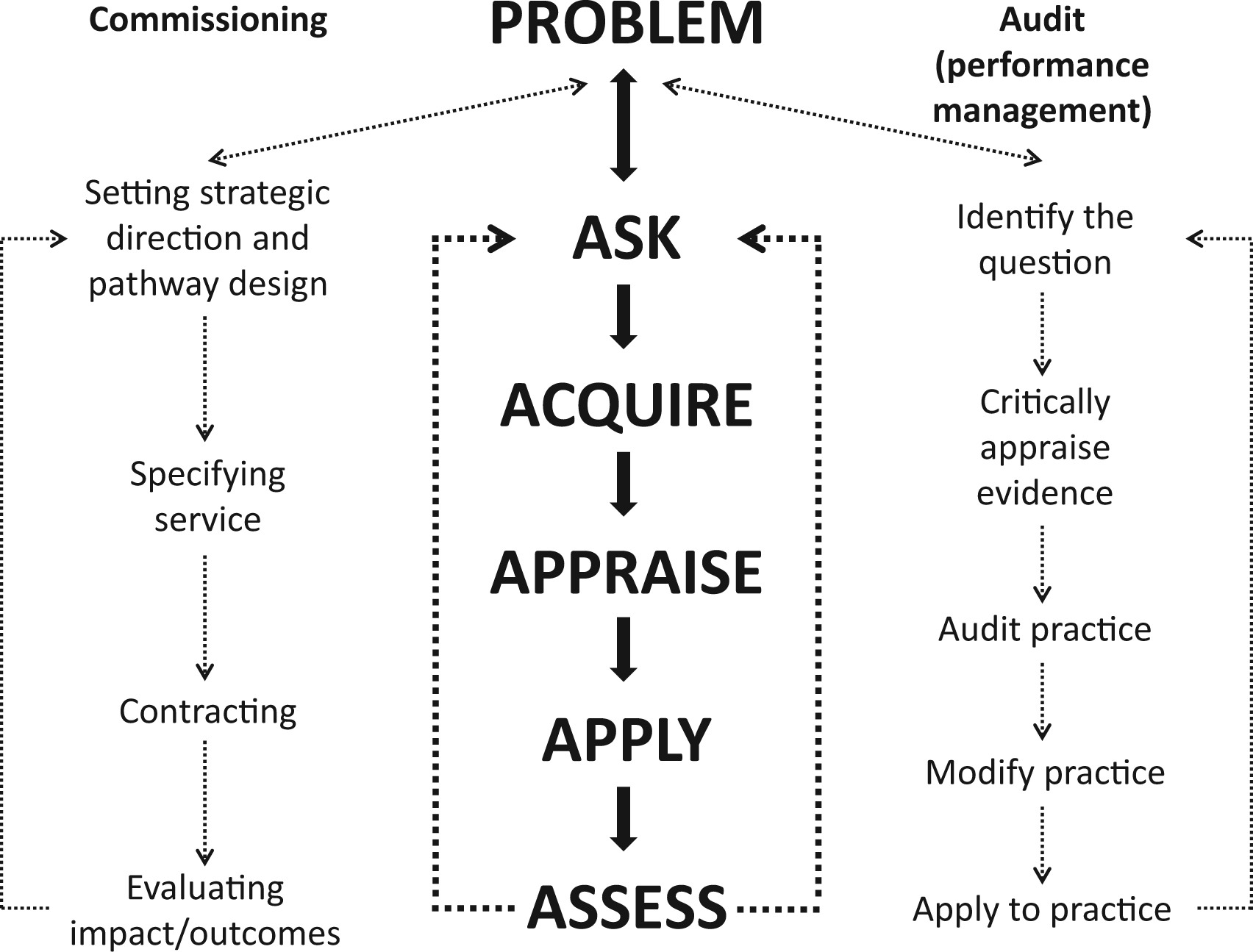

Translating EBLM into routine practice
A strategy for translating EBLM into routine practice makes a number of basic assumptions (i) laboratory medicine is part of a clinical team so test information can be converted to action; (ii) the main goal is to add value to the care pathway; (iii) a test is only used because there is a problem or unmet need; (iv) the result is acted upon at the appropriate time; and (v) resource utilization is managed
The easiest way to illustrate how EBLM can be translated into practice is with the introduction of a new test. There are a number of generic questions to ask of the requestor at this point, set out below with some explanatory notes.
Why do you want the test? What will you do with the result? What decision will you make? What action will you take? What outcome would you expect? Is the test any good, i.e. is there any evidence available? What will be the cost? What additional benefits might be expected, and where in the care pathway?
The responses to these questions help to (i) establish the nature of the problem or unmet need, (ii) review the evidence to demonstrate that the need can be met, (iii) develop the business case and (iv) set out the requirements for adoption and implementation, if the business case is successful. The questions are relevant to any number of problems that might be experienced and they reflect the use of the test and the complementary activities across the care pathway, with the potential for solutions with clinical, operational and/or economic impact. However, insofar as the questions reflect the use of the test across the care pathway they can also be used to inform audit, performance management and continuous quality improvement activities. They also provide a framework for many of the enquiries made of the laboratory, e.g. to the duty biochemist/doctor and accessing the knowledge base.
This approach is illustrated in the request for the introduction of natriuretic peptide testing – where the request was made by the local cardiac network manager. 30 The local need was based on a combination of poor access to echocardiography for patients presenting in primary care with suspected heart failure and awareness of a revised clinical guideline supported by a good evidence base for the use of a natriuretic peptide as the first line test. The PICO question developed is the second in Table 1. The discussion with stakeholders established the requirement and provided the basis of the answers to the first seven questions outlined above and enabled the development of the business case for adoption of the test, as well as the framework for the implementation and performance management of the service. This involved details of current practice including time to diagnosis from initial presentation, cost of current diagnostic pathway, level of activity, number of patients presenting with suspected heart failure, number of patients on heart failure register and cost of natriuretic peptide test. These data together with the evidence from a relevant systematic review 29 were used to model the revised pathway and establish the economic elements of the business case. The core of the business case was the reduction in time to diagnosis as well as the additional benefits of a significant reduction in the requirement for echocardiography and associated outpatient clinic visits – the savings being used to fund the natriuretic peptide service as well as reduction in overall cost to the commissioner/purchaser. The initial discussion also highlighted additional clinical questions around the potential use of the test for (i) screening women receiving chemotherapy for breast cancer and at risk of developing heart failure, (ii) screening other patient groups at risk of developing heart failure and (iii) guiding therapy in patients diagnosed with heart failure. This provided the opportunity for business cases to be developed including the extent of the relevant evidence base, to be submitted to the local annual strategic review and prioritization process.
The core of translating EBLM into routine practice is the link between articulation of the problem (the question), the quality of the evidence, the decision made on receipt of the result and the action taken. This has then to be evaluated within the context of the delivery and performance management of an integrated healthcare service, i.e. integrated care pathways. The practical questions outlined above can be adapted to address clinical, operational and economic issues as outlined in Table 2, across the full spectrum of healthcare – from policymaking to delivery.
Conclusions
In routine laboratory practice, it is recognized that (i) the evidence base is poor, 34 and (ii) there are many reports of both under- and over-utilization of tests; the second observation is an obvious corollary of the first! Over a decade ago, George Lundberg 35 alluded to this problem when identifying the need for an outcomes agenda for laboratory medicine. There is also a recognized problem around translation of evidence into routine practice with the associated transformational change, 36 a problem not limited to laboratory medicine. 37 The demand for evidence-based practice extends to laboratory medicine as one of the tools for improving the quality of healthcare. The practice of EBLM is the core for all of the facets of laboratory medicine from commissioning of a service through to audit and continuous quality improvement. The starting point in all cases remains identifying unmet needs and the articulation of the problem (the problem statement) through clinical questions.
Footnotes
Acknowledgements
None.
Declaration of conflicting interests
None.
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
Ethical approval
Not required.
Guarantor
CPP.
Contributorship
The authors contributed equally to the content of this review.
