Abstract
Background
National Health Service England issued a Patient Safety Alert in 2014 mandating all acute Trusts in England to implement Acute Kidney Injury (AKI) warning stage results and to do so using a standardised algorithm. In 2021, the Renal and Pathology Getting It Right First Time (GIRFT) teams found significant variation in AKI reporting across the UK. A survey was designed to capture information on the entire AKI detection and alerting process to investigate the potential sources of this unwarranted variation.
Methods
In August 2021, an online survey consisting of 54 questions was made available to all UK laboratories. The questions covered creatinine assays, laboratory information management systems (LIMS), the AKI algorithm and AKI reporting.
Results
We received 101 responses from laboratories. Data were reviewed for England only – 91 laboratories. Findings included that 72% used enzymatic creatinine. In addition, 7 manufacturer-analytical platforms, 15 different LIMS and a wide range of creatinine reference ranges were in use. In 68% of laboratories, the AKI algorithm was installed by the LIMS provider. Marked variation was found in the minimum age of AKI reporting with only 18% starting at the recommended 1 month/28-days. Some 89% phoned all new AKI2s and AKI3s, as per AKI guidance while 76% provided comments/hyperlinks in reports.
Conclusions
The national survey has identified laboratory practices that potentially contribute to unwarranted variation in the reporting of AKI in the England. This has formed the basis for improvement work to remedy the situation, including national recommendations, included within this article.
Background
Acute Kidney Injury (AKI) is a common clinical syndrome 1 with high mortality and morbidity, affecting as many as 20% of hospital admissions in developed healthcare systems.2–5 Survivors are at a higher risk of later developing Chronic Kidney Disease (CKD). 6 The estimated annual cost of AKI in England is approximately £1 billion or 1% of the NHS budget. 3 The publication of the 2009 NCEPOD (National Confidential Enquiry into Patient Outcome and Death) report into AKI identified significant deficiencies in the assessment of AKI risk, its detection, assessment of severity, and management. 2 ‘Think Kidneys’, a 3-year national initiative supported by NHS England (NHSE) and the UK Renal Registry (UKRR), was established in 2013 to improve AKI care and outcomes. 7 This programme helped drive the 2014 NHSE Patient Safety Alert (PSA) to standardise AKI detection, 8 by mandating all acute trusts in England to implement AKI detection via a standardised algorithm 9 by March 2015. 8 This was initially only for patient samples collected in the hospital setting but was later rolled-out to Primary Care.8,10,11 In brief, the NHSE AKI algorithm looks at previous creatinine results over the last 365 days in comparison to current creatinine result, to determine if AKI is likely present. AKI severity is also calculated and three possible levels of AKI warning stage results are added – AKI Stage 1 (AKI1), AKI Stage 2 (AKI2) and AKI Stage 3 (AKI3). 9 The algorithm was intended to be used to detect AKI in both children and adults, with only two patient groups to be excluded – neonates and patients receiving dialysis for end-stage kidney disease.8–11
All laboratories in England were also mandated by the Patient Safety team at NHSE to submit AKI data to a central AKI master patient index hosted by UKRR for national quality improvement and assurance purposes. 12 The UK Kidney Association (UKKA) 2018 national report stated that 87% of the 190 England-based laboratories submitted data to a standard to allow data analysis. The data that were analysed demonstrated a wide geographical variation in AKI incidence. 13 This was also reflected in both the Renal and Pathology Getting it Right First Time (GIRFT) (a national NHSE programme designed to improve treatment and care of patients through in-depth review of services, benchmarking, and presenting a data-driven evidence base to support change) reports.14,15 Pathology GIRFT demonstrated a 3 to 5 fold variation in the incidence of all AKI stages both within Emergency departments and Primary Care. 14 The variation found by all three groups could only be partially explained by care quality factors as outlined in the NCEPOD and GIRFT reports.13–15
To investigate further this unwarranted laboratory variation, 14 including laboratory methodological differences, a collaboration was formed in 2021 between GIRFT, Association for Clinical Biochemistry and Laboratory Medicine (ACB), UK National External Quality Assessment Services (UK NEQAS) and UKKA, to form an AKI Taskforce. A national survey was designed to capture data on the whole AKI detection and alerting process in order to get a good understanding of laboratory processes that may lead to variation.
Method
The national AKI survey was hosted on SurveyMonkey and was open from August to October 2021, with all UK laboratories invited to participate by both ACB and UK NEQAS. We cannot confirm how many laboratories received the survey, though postulate the majority did, as we sent out survey invitations via several routes. Respondents were asked to complete 54 questions (Supplemental File 1). These covered the specific creatinine assays used, LIMS, the AKI algorithm, how and which patient results are fed into the algorithm, how the AKI results were reported to the clinicians, and reported to the UKRR. Data were reviewed such that only a single response was counted per laboratory/UKKA code, but some questions allowed more than one response. Data transfer to Microsoft Excel was accomplished using the validated functionality within the SurveyMonkey software. There was no manual data transfer to Microsoft Excel. Data analysis was performed using Microsoft Excel.
Results
Ninety-one complete responses were received from laboratories in England. Only data from England are included in this manuscript and Supplemental files as the original PSA applied to England only. A copy of all questions and responses not detailed in this manuscript is provided within Supplemental Material (Supplemental File 1 and Supplemental File 2).
Q1–3 related to the respondents details. Supplemental Figure 1 shows a breakdown of who AKI is reported to. Respondents were able to select more than one answer.
Section 1: Creatinine assays
This section includes data from Questions 4 to 13. Some laboratories use more than one manufacturer-assay combination, for example, both Abbott Alinity and Abbott Architect, in the same laboratory, or Siemens ADVIA and Siemens Atellica. These have been counted individually, but within a single laboratory.
Q4: What creatinine assays are used to feed into your AKI algorithm (across your complete hospital/network)? Enzymatic assays were used by 78% (n = 71) of laboratories. The remaining 24% (n = 22) used compensated kinetic jaffe. Two laboratories reported using both enzymatic and compensated kinetic jaffe. No laboratories reported using creatinine results generated from blood gas analysers or other POCT devices. Q9 asked the same question, but within the individual respondent’s laboratory and no differences were observed.
Q5–Q7 Laboratories were asked where different creatinine requests are processed and whether this is the same analytical platform. These data are shown in Supplemental Figures 2 to 4.
Q8: Which manufacturer does your laboratory use for creatinine analysis? Seven manufacturer-analytical platform combinations were reported, with the most popular analytical platform being the Roche Cobas (51% n = 50). The Roche Cobas enzymatic assay (43% n = 42) was also the most popular assay-platform combination (Figure 1). Manufacturer-analytical platforms and creatinine assay used by laboratories, which is subsequently fed into the AKI algorithm (more than one response per laboratory accepted).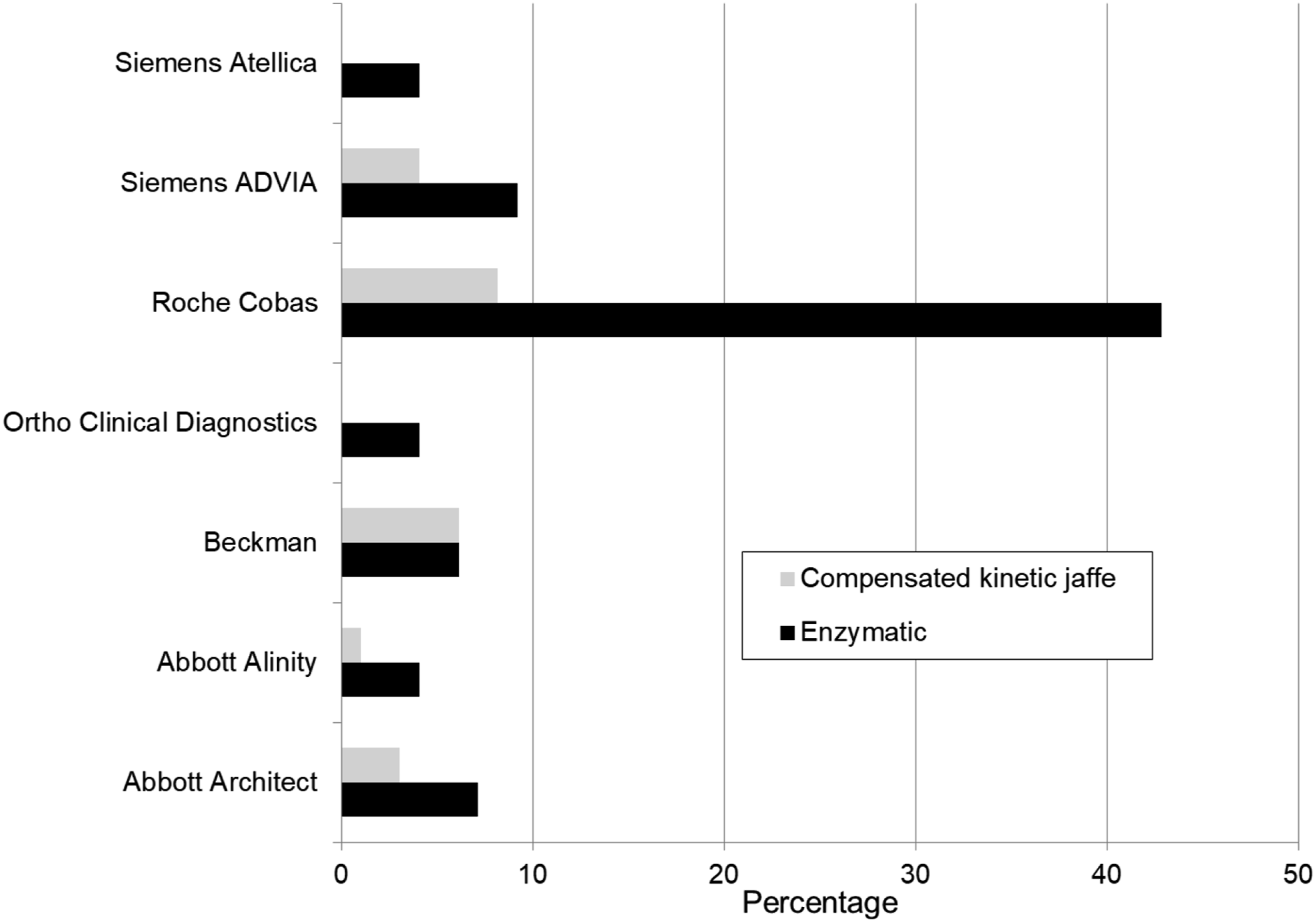
Q10 and Q11: What reference range(s) do you use for creatinine in males and females over 16 years old? The range of results are displayed in Figure 2(a) males and Figure 2(b) females. When multiple age-related reference ranges were provided the lowest and the highest creatinine concentration over the age range has been taken. There is variation both within and between manufacturers, which is not solely attributed to the two different method principles. Reference range information for creatinine for (a) Males >16 years and (b) Females >16 years (When multiple age-related reference ranges were provided the lowest and the highest creatinine concentration over the age range has been taken).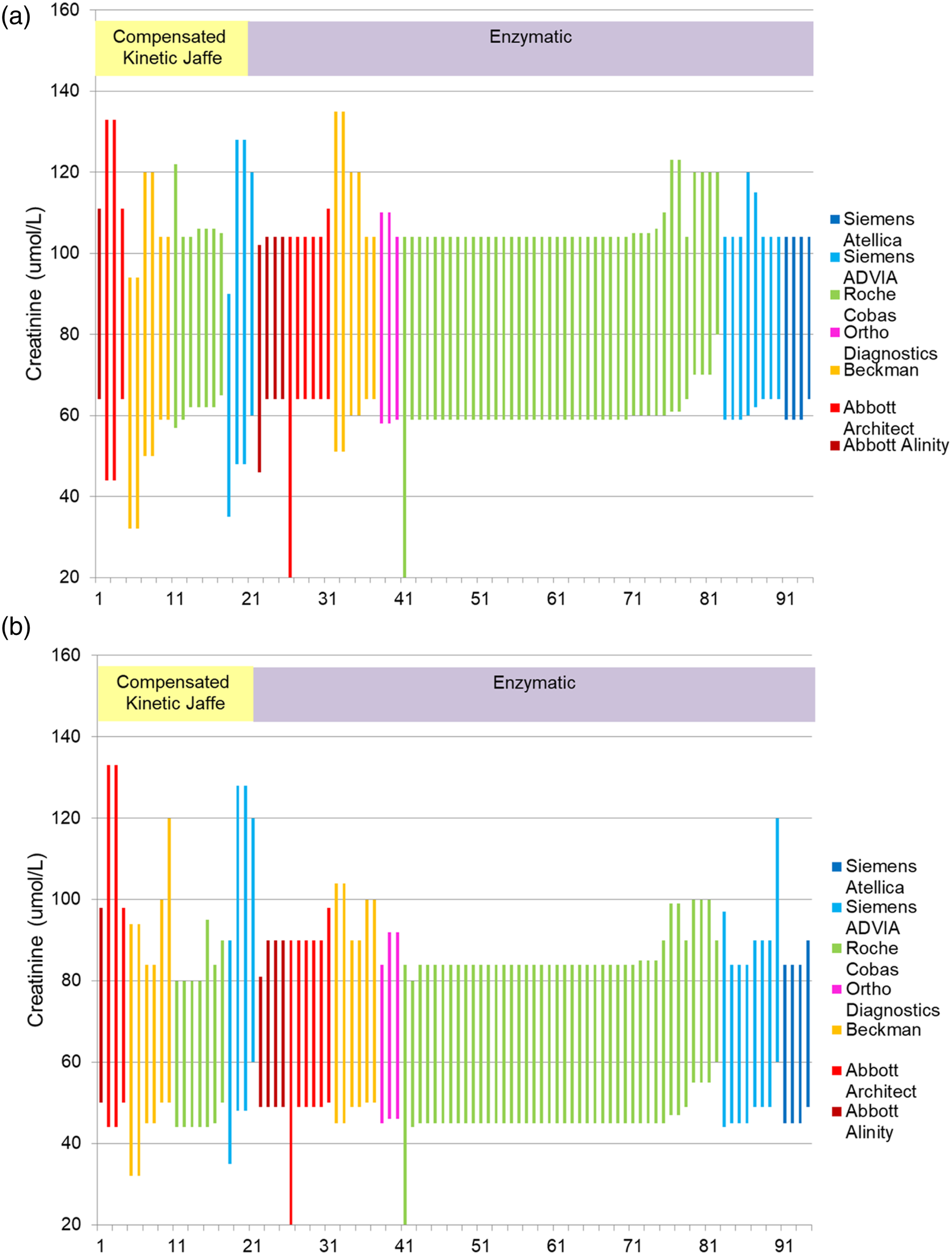
Q12 and Q13: Do you monitor the variation of creatinine across all analysers within a laboratory or network? If yes, at what frequency do you monitor this variation? From the 91 laboratories that responded, 96% (n = 87) monitored the variation across the laboratory/network. Of the four laboratories that responded ‘No’, two are in network that uses a different platform and there is insufficient information regarding the other two. The most popular frequency for the monitoring of creatinine variation across all analysers was monthly, 75% (n = 65), with an approximate even split for the other frequencies: 9% (n = 8) daily, 6% (n = 5) weekly and 10% (n = 9) less than monthly.
Section 2: LIMS and the AKI algorithm
This section includes data from Question 14 and Question 17 to 25.
Q14: Which LIMS do you use in your laboratory? Fifteen LIMS from multiple providers are in use. The most common were iLab Apex-DXC 22% (n = 21), iLab Telepath-DXC 20% (n = 19), Winpath5-Clinisys 15% (n = 14) and Winpath Enterprise-Clinisys 13% (n = 12). Two laboratories use two different LIMS within their network and one laboratory was in the middle of changing LIMS during the survey, therefore the total number of replies was 94.
Q17 and Q18: Where is the AKI calculated? If external to the laboratory, does the software include creatinine from more than one laboratory? The LIMS in the laboratory was reported by 96% (n = 87) of respondents, with one laboratory reporting separate software within the laboratory, two laboratories use external software to the laboratory and one laboratory reported using LIMS for Primary Care and Out-Patient Department (OPD) work and the Hospital PAS for In-patient work. It was only in this latter example that creatinine from more than one laboratory could be included in the algorithm.
Q19: What is the provenance of the AKI algorithm that your laboratory uses? Multiple responses were allowed. Seventy-five percent (n = 68) said the algorithm was supplied by their LIMS provider and 30% (n = 27) knew the NHSE algorithm was installed. Other responses were 11% (n = 10) ‘in-house’, 6% (n = 5) did not know, and 6% (n = 5) selected ‘other’ and completed the free text box (Figure 3). These laboratories were provided an algorithm by their LIMS provider and then modified it in-house. Provenance of the AKI Algorithm used within laboratories (more than one response per laboratory accepted).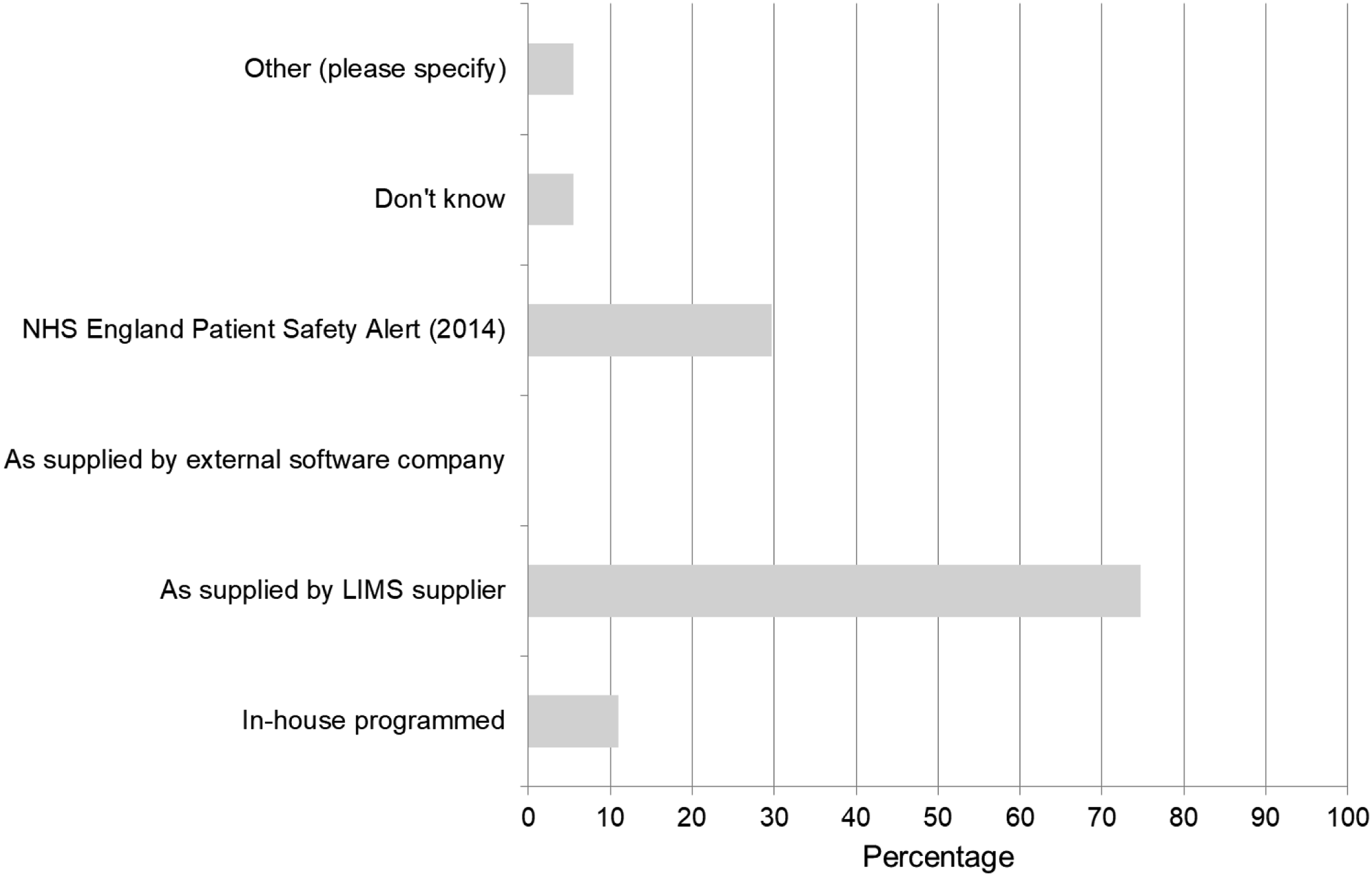
Q20: Who is responsible for ensuring the validity of the AKI algorithm? Laboratory IT or laboratory personnel accounted for 89% (n = 81) of responses. Of the remaining 10 responses, 9% (n = 8) did not know who was responsible and 2% (n = 2) replied that hospital IT were responsible.
Q21: Can your laboratory edit the AKI algorithm? Only 90 laboratories responded to this question; with yes 50% (n = 45), no 40% (n = 36) and did not know 10% (n = 9).
Questions 22 to 25 looked at the validation of the AKI algorithm and is summarised in Supplemental Figures 5 to 8.
Section 3: Reporting AKI
This section includes data from Questions 15 and 16, and Question 26 to 46.
Q15 and Q16: Are all your Primary Care and/or Secondary Care creatinine results put through the AKI algorithm? For Primary Care results, 92% (n = 81) did, 7% (n = 6) did not, and 1% (n = 1) did not know. For Secondary Care results, 87% (n = 78) did, 11% (n = 10) did not and 2% (n = 2) did not know.
Q26: When a Reference Value (RV) value is calculated but no AKI detected, what action is taken by LIMS? No action taken by 49% (n = 45), and 26% (n = 24) added an ‘AKI 0’ test code. The remaining 22 responses indicated that they would either delete the AKI request, or suppress test name/results, or add comments (e.g. ‘AKI has not been detected’ or ‘an AKI calculation has not been made’).
Q27: If you are able to generate an AKI 0, are you able to suppress this result so that it doesn’t appear in the clinical results system? This question was not applicable to 50% (n = 45) of respondents. Thirty percent replied yes (n = 27), 12% no (n = 11) and 9% (n = 8) did not know.
Q28: Are there other scenarios that may generate non-numerical variables? Comments included no sample received, no baseline creatinine available, problem with sample integrity, or AKI not detected, or an AKI calculation has not been made.
Q29 and Q30: When no previous creatinine results reported within the last 365 days and the current creatinine is above the reference range, what action is taken? Sixty-six percent (n = 57) add the recommended ‘?AKI ?CKD. Suggest repeat’ comment. No action is taken by 16% (n = 14) and the remaining 18% (n = 16) contained comments such as results reviewed by duty biochemist, results telephoned and do not know. In 56% (n = 49) of cases, a list of patients for whom comments have been appended could be retrieved.
Q31 and Q32: Do you exclude dialysis patients from the AKI alert? If yes, how do you do this? Forty-seven percent (n = 43) exclude these patients, with 54% (n = 23) using location codes and 12% (n = 5) using patient flags in their LIMS and 2% (n = 1) using consultant code in their LIMS. The free text option was completed by 33% (n = 14) with a variety of methods employed; ‘patient type’, type of patient identification number, location codes along with the consultant code, patient flags or clinical details.
Q33: Do you exclude obstetric patients? Only 89 laboratories responded to this question. Eighty-seven percent (n = 77) said yes, the remainder said no or did not know.
Q34: Do you exclude other locations/disciplines/patient groups? Twenty-four percent (n = 22) stated yes; with Paediatrics, Primary Care, OPDs, Renal Wards, Renal OPDs and Intensive Care Units (ITU) listed.
Q35: At what patient age does your laboratory start reporting AKI results? A wide range of ages was provided (Figure 4), with the majority (46%, n = 37) ‘from birth’. The minimum age laboratories use for starting to report AKI alerts.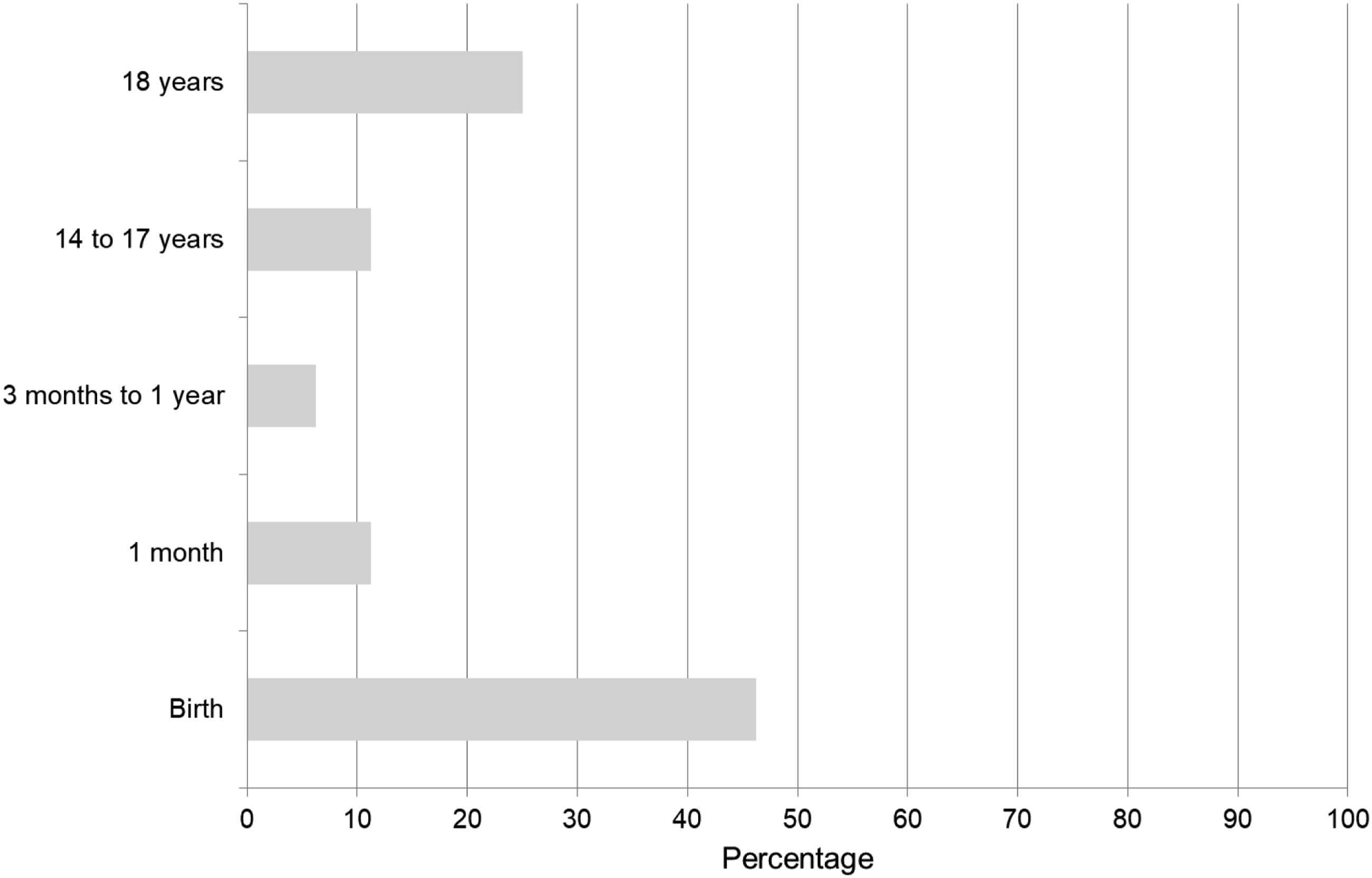
Q36: Does your laboratory phone AKI alerts? There were 89 responses to this question. Eighty-nine percent (n = 79) phone Primary Care and 81% (n = 72) Secondary Care. Four do not phone AKI alerts.
Q37: Which AKI warning stage results do you phone? Ninety-three percent (n = 79) phoned AKI2s and AKI3s, while another 66% (n = 56) phoned only AKI3. Additionally, 2% (n = 2) phone Paediatric AKI1s, 6% (n = 5) AKI1 via a local protocol/staff discretion and 24% (n = 20) AKI1 when the potassium ≥6 mmol/L (Figure 5). Multiple responses were allowed for this question and data was taken from the 85 ‘yes’ respondents to Q36. Phoning protocols for AKI results (more than one response per laboratory accepted).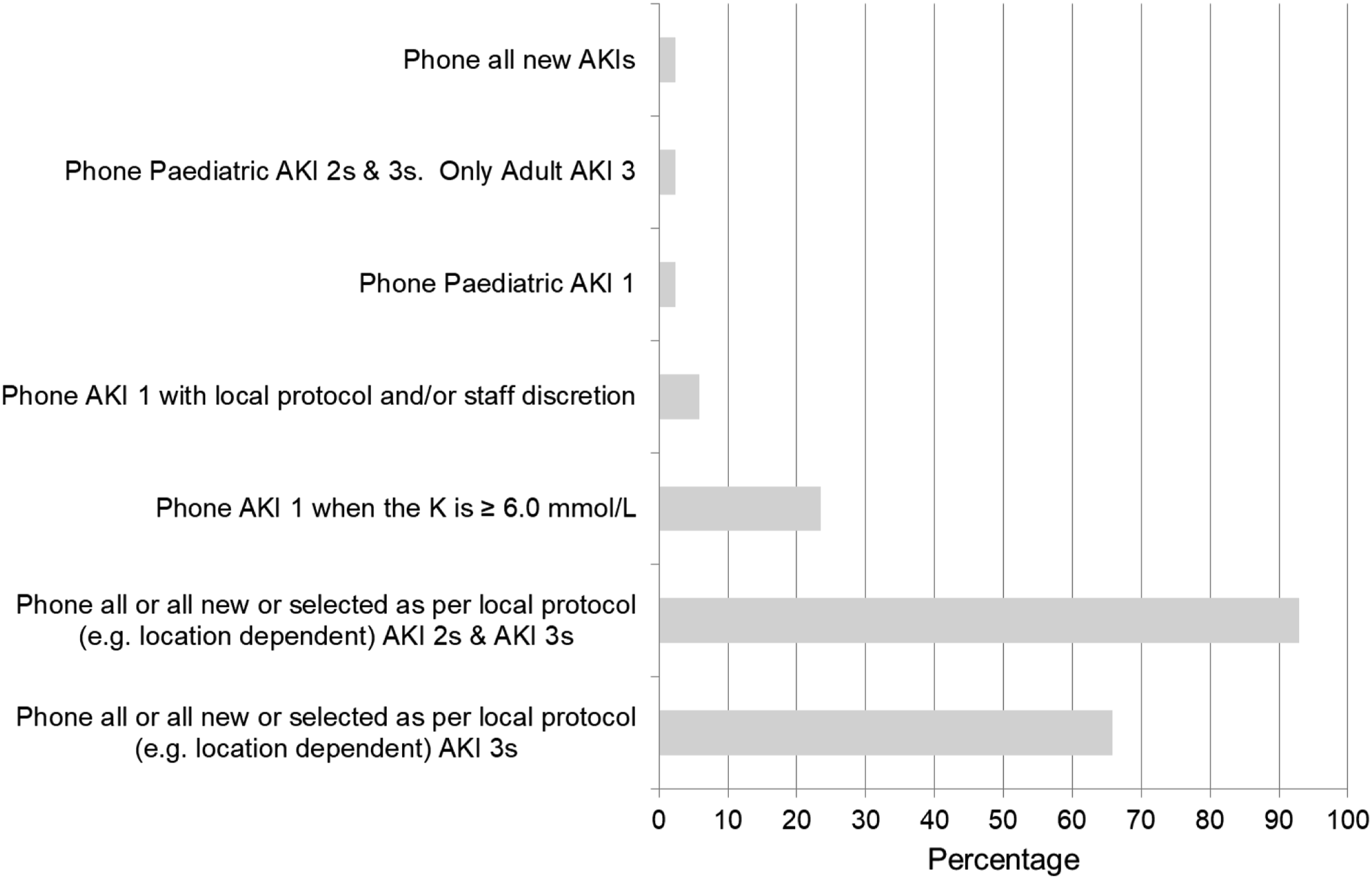
Q38 and Q39: What turn-around-time do you have for phoning Primary and Secondary Care results? A wide variation in practice, for both AKI2s and AKI3s, for Primary and Secondary Care ranging from ‘as soon as possible’ to 24 h (Figure 6). Turn-around-time for phoning AKI Stage 2 and AKI Stage 3 results to Primary and Secondary care.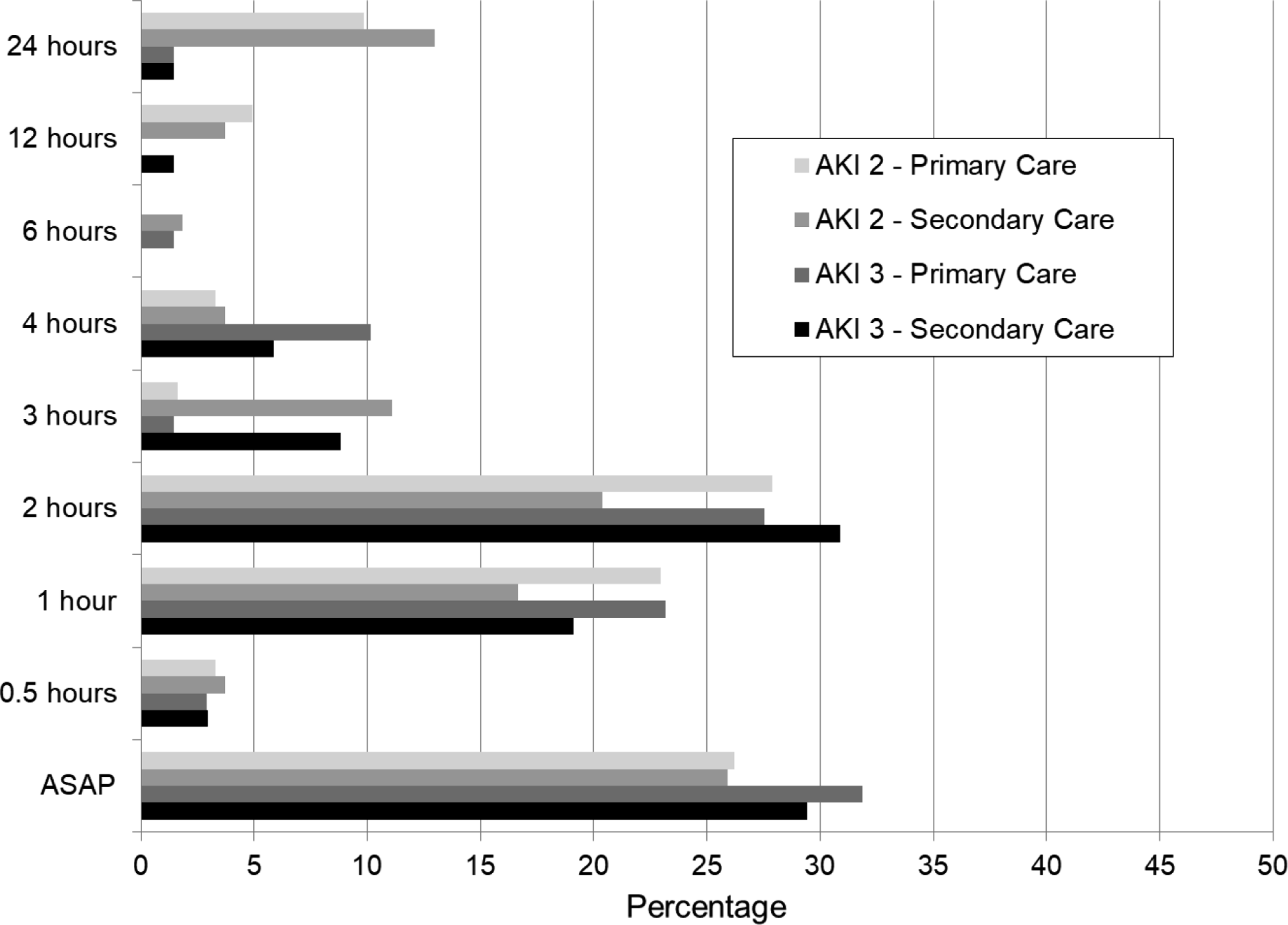
Q40: Do you have other mechanisms for alerting clinicians? There were only 76 responses to this question. Sixty-three percent (n = 48) use flagged patient reports or AKI worklists (electronic or paper or mobile apps), 21% (n = 16) contacting clinicians/teams by email and 3% (n = 2) by text alerts. Another 36% (n = 27) stated in free text patient flags in Sunquest, ICE and electronic patient records, for example, Nervecentre; or daily AKI lists provided to dedicated AKI teams, Critical Care Outreach Teams or hospital patient safety teams.
Q41–Q44: Do you add interpretative comments or hyperlinks to AKI warning stage results? Fifty-five percent (n = 50) provide comments, 24% (n = 22) comments with hyperlinks and 19% (n = 17) did not provide anything. Two laboratories did not know. Many different examples were given but are not discussed further here.
Q45: Were your Renal, Acute Medicine or Critical Care Teams involved with writing the comments? Of those that did provide comments, 73% (n = 60) did so with these teams.
Q46: For Primary Care requests, have you checked that any flagging/comments are transferred with the AKI result in ALL Primary Care reporting routes? Sixty percent (n = 47) of responses said yes, 14% (n = 11) no and the remaining 27% (n = 21) did not know.
Section 4: Reporting to the UK renal registry
This section includes data from Questions 47 to 54.
Q47 and Q48: Do you routinely send AKI warning stage results and creatinine results to the UKRR? Eighty-six percent (n = 78) send AKI and 65% (n = 58) send creatinine data. Eight percent (n = 7) did not send AKI and 14% (n = 12) did not send creatinine data. The remaining respondents did not know.
Q49: Do you have systems in place to ensure that data extraction from your LIMS before transfer to UKRR is complete? Systems were in place for 56% (n = 46).
Q50 and Q51: Do you have systems in place to ensure that data are returned every month without exception? Who is responsible for ensuring that data are returned to the UKKR? These same respondents as in Q49 also had systems in place to ensure this was done too. In 79% (n = 63) of cases the laboratory was responsible for ensuring that the data is transferred to the UKKR. In 11% (n = 9) of cases the Hospital IT were responsible, other responses included 1% (n = 1) Renal Team and the remaining did not know.
Q52: Is your laboratory aware that data returns to the UKKR are mandated by an NHS England Patient Safety Alert? Eighty-eight percent (n = 79) indicated yes.
Q53: At what frequency do you have meetings with your renal, acute medicine or critical care colleagues? The majority of responses, 57% (n = 51), were ‘As and when required’. Thirty percent (n = 27) do not meet regularly with any of these teams. Other responses ranged from 0–1 month to 6–12 months.
Discussion
Survey responses came from all parts of England. Although the UKKA reports there are 190 England-based laboratories, this includes 39 ‘satellites’ which submit data via their ‘parent’ laboratory. 13 We postulate several of the 91 survey responders therefore returned data for multiple laboratories, meaning together they likely provided AKI detection and reporting data that is broadly representative of laboratories across England. Although the majority (78%) use enzymatic assays recommended within previous and current NICE Guidance NG203-Chronic Kidney Disease: assessment and management guidance (2021), a significant minority (22%) use alternative methods. 16 The survey highlighted variation in reference ranges reported by laboratories, both between and within manufacturers (Figure 2). There is no rationale for different creatinine reference ranges, if the different methods are reliably measuring creatinine. Enzymatic creatinine assays appear to agree better for adult reference ranges and are markedly different to the compensated kinetic Jaffe assay. This is of concern as the AKI algorithm has two arms which each rely upon reference ranges. 9 Discrepant reference ranges may therefore account for some the observed variation in AKI detection.
Fifteen different LIMS are in use, provided by multiple LIMS companies, inherently challenging uniform roll out of laboratory programs such as AKI detection. All English-based laboratories are required to use the AKI algorithm as per the NHSE PSA.8–11 Though 75% of respondents stated that their LIMS provider supplied the AKI algorithm, only a minority of respondents (30%) knew the NHSE algorithm was installed, and 6% did not know the provenance at all. This may reflect a lack of knowledge by the respondent, or that this information was not recorded or not found. Of further note, 11% stated use of ‘in-house’ AKI algorithms rather than the NHSE algorithm, and 50% indicated options to modify AKI algorithms supplied by their LIMS provider and 6% indicated they had done this.
English-based laboratories are required to process all creatinine results from Primary and Secondary Care using the algorithm.8–11 We found compliance was 87% for Secondary and 92% for Primary Care. Of note, a small but significant percentage of responders stated not reporting to Primary and/or Secondary Care; the reasons are unknown but could be due to IT functionally.
The NHSE AKI algorithm has two end points where a calculated RV ratio is found to be < 1.5 and no AKI detected. Consistent with the algorithm, 49% of responders reported no action is taken, however, 26% stated their LIMS was configured to add an ‘AKI 0’ test code even though ‘AKI 0’ is not included in the NHSE algorithm. 9 The second endpoint with an RV <1.5 and no AKI detected is when no creatinine results have been reported within the previous 365 days. From this, there are three possible endpoints of ‘No flag’ (current creatinine result within the reference range), ‘Flag low’ (low creatinine) and ‘Flag high’ (high creatinine). The latter requires the additional comment ‘?AKI ?CKD. Suggest repeat’ to be reported with the current creatinine.8,9 Only sixty-six percent of responders stated they added this comment, which is required within the NHSE algorithm. 9 We postulate that LIMS providers installing the algorithm may not have included provision for this comment and/or laboratory/IT staff validating the algorithm may not recognise the requirement for this pathway-arm and comment.
It is recommended that patients receiving long-term dialysis for end-stage kidney disease are excluded from AKI algorithm reporting8,11 as comparison of sequential post- and pre-dialysis creatinine assays can generate false positive results. This is recognised as a challenge for laboratories as they often do not have access to this information, or/and it not recorded in LIMS to categorise these patients appropriately. It is thus not surprising that only 47% reported excluding these patients, with the majority (54%) using exclusion by location codes rules. Exclusion of renal patients by inpatient or outpatient location codes is a potential problem because AKI can occur in patients not receiving dialysis (e.g. kidney transplant recipients) and will be missed. The clinical risk may be relatively low, however, as renal teams will be focussed on creatinine results. Conversely, false positive AKI alerts may arise if haemodialysis patients are managed on non-renal wards, although the clinical risk is again low. A flag in the LIMS to identify dialysis patients may be preferable, though would be complex to administer and maintain. A similar question was asked about obstetric patients, with 87% also excluding these patients, even though this was not stated in the NHSE PSA8–10 or the Think Kidneys 2014 guidance. 11 Neither clinical nor laboratory criteria for AKI diagnosis in non-pregnant patients are fully validated in pregnant patients, but one early study has provided some useful data on the validity of them. 17 We asked if other patient groups were excluded and a notable 26% reported yes; with Paediatrics, Primary Care, OPDs, Renal Wards, Renal OPDs and ITUs listed. These additional exclusions were also contrary to the NHSE PSA. Special care baby/neonatal units should be excluded as neonates are excluded from AKI reporting, 11 however, children are included9–11 and there is a specific arm in the algorithm to accommodate them. 9 Patients in Primary Care and ICU settings should receive AKI alerts.8,10,11 The AKI taskforce was aware of variation regarding age laboratories start reporting AKI alerts and therefore a question was included. A range of ages was reported, with the majority (46%) answering with ‘from birth’. Only 11% were correctly reporting from age 1 month (Figure 4). Responders also indicated a number of laboratories are not reporting AKI in Paediatric populations, again contrary to the NHSE algorithm 8 and national guidance,9,10 again risking AKI being missed in these patients.
The survey indicates 55% of laboratories provide interpretative comments and a further 24% provide comments in addition to hyperlinks for guidance. It is noteworthy that the remainder did not, given the full complexity of AKI and the need for appropriate patient management. Alerting clinicians to a new AKI2 or AKI3 is important to allow prompt clinical review. We found 89% phoned Primary Care and 81% Secondary Care. The turn-around-time target for phoning was explored and highlighted a wide variation in practice, for both AKI2s and AKI3s, and for Primary and Secondary Care ranging from ‘as soon as possible’ to 24 h (Figure 6). Some laboratories interpreted this question as time from result available and others from sample receipt. The variation in turn-around-time in general may be due to laboratories following different national guidance or local protocols. The Think Kidneys 2020 guidance 11 advised all new AKI3s to be phoned immediately and all new AKI2s within 24 h. The Royal College of Pathologists 18 advises all new AKI3s and AKI 2s from both Primary and Secondary Care to be phoned within 2 h of result being available. Laboratories reported they had other mechanisms to alert clinicians. GIRFT data indicate a few laboratories have developed their AKI response such that it is fully integrated into the Electronic Health Record (EHR) with a live dashboard and decision support for clinicians. 15 This variation is consistent with the NHSE PSA, in which it was acknowledged that the most appropriate local alerting mechanisms could be utilised for communicating AKI alerts to clinicians.
Despite the NHSE PSA mandating both AKI and creatinine data are reported monthly to the UKRR,10–12 the majority (86%) did so for AKI data, but only 65% for creatinine data.
This national survey has identified marked variation in practice in all areas of AKI detection and reporting pathways. These system-wide variations in AKI detection and reporting impede the standardisation of AKI patient management. Such unwarranted variation has potentially serious implications for clinical care, patient outcomes, efficient use of healthcare resources and regional health inequality. Accordingly, this multidisciplinary AKI taskforce has published this report to raise awareness of the variation and the causes behind them, and furthermore has compiled 10 recommendations fully endorsed by the ACB, GIRFT, UKKA and UK NEQAS.
Recommendations

These recommendations have been fully endorsed by:
Association for Clinical Biochemistry and Laboratory Medicine
GIRFT Pathology and GIRFT Renal
UK Kidney Association
UK NEQAS
Supplemental Material
Supplemental Material - National recommendations to standardise acute kidney injury detection and alerting
Supplemental Material for National recommendations to standardise acute kidney injury detection and alerting by Rachel Marrington, Anna L Barton, Alexandra Yates, William McKane, Nicholas M Selby, Jonathan S Murray, James F Medcalf, Finlay MacKenzie and Martin Myers in Annals of Clinical Biochemistry
Supplemental Material
Supplemental Material - National recommendations to standardise acute kidney injury detection and alerting
Supplemental Material for National recommendations to standardise acute kidney injury detection and alerting by Rachel Marrington, Anna L Barton, Alexandra Yates, William McKane, Nicholas M Selby, Jonathan S Murray, James F Medcalf, Finlay MacKenzie and Martin Myers in Annals of Clinical Biochemistry
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethical approval
Ethical approval is not applicable.
Guarantor
RM.
Contributorship
All authors contributed to the writing of the survey questions. RM and AB reviewed and analysed the survey data. RM and AB produced the first draft of the manuscript. All authors critically reviewed, revised and approved the final version of the manuscript.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
