Abstract
Background
The early prediction of acute kidney injury (AKI) by current clinical and laboratory methods remains inadequate. Neutrophil gelatinase-associated lipocalin (NGAL) has emerged as a promising non-invasive biomarker of kidney injury. We systematically reviewed the utility of plasma and urine NGAL measurements for the prediction of AKI in humans.
Methods
We searched MEDLINE, PubMed and EMBASE for human biomarker studies that included NGAL (January 2005 to October 2013). Studies reporting on the use of NGAL for the early prediction and prognosis of AKI were analysed in three common clinical settings: cardiac surgery, critical illness and kidney transplantation.
Results
We identified 58 manuscripts that met our inclusion and exclusion criteria, reporting on more than 16,500 patients. Following cardiac surgery, NGAL measurement in over 7000 patients was predictive of AKI and its severity, with an overall area under the receiver operator characteristic curve (AUC) of 0.82–0.83. Similar results were obtained in over 8500 critically ill patients. In over 1000 patients undergoing kidney transplantation, NGAL measurements predicted delayed graft function with an overall AUC of 0.87. In all three settings, NGAL significantly improved the prediction of AKI risk over the clinical model alone.
Conclusions
We identified several studies that collectively strongly support the use of NGAL as a biomarker for the prediction of AKI. However, we noted some limitations, including lack of published studies that adhere to diagnostic study guidelines, heterogeneity in AKI definition, the lack of uniformly applicable cut-off values and variability in the performance of commercially available NGAL assays.
Introduction
Biology of NGAL
Human neutrophil gelatinase-associated lipocalin (NGAL, also known as lipocalin 2, siderocalin or 24p3) was originally isolated from the supernatant of activated neutrophils and identified as a polypeptide covalently bound to gelatinase.1,2 NGAL is expressed in a variety of human tissues, including lung, liver and kidney, in various pathologic states.3–5 Human NGAL consists of a single disulphide-bridged polypeptide with a molecular weight of 25 kDa. 1 While the majority of NGAL is in a monomeric form, NGAL also occurs as dimers and trimers, as well as in a complex with neutrophil gelatinase.1,6 The 25 kDa monomeric NGAL form is secreted by injured kidney tubule epithelial cells, whereas the dimeric form is the predominant form secreted by neutrophils.7,8 The major ligands for NGAL are siderophores,9,10 which are ferric ion-specific chelating compounds.11–14 The iron status of NGAL is a critical determinant of biological activity. Iron-containing NGAL binds to cell surface receptors such as megalin, gets internalized and releases its bound iron. The increased intracellular iron concentration drives the regulation of iron-dependent genes.15,16 On the contrary, iron-lacking NGAL binds to the same receptors, but chelates intracellular iron and shuttles it to the extracellular space. NGAL has been implicated in multiple biological processes, including attenuation of apoptosis 17 and differentiation of renal tubule epithelial cells and nephrons. 16 During nephrogenesis, the NGAL:siderophore:iron complex converts renal progenitors into epithelial tubules. 18
Preclinical studies identified NGAL to be one of the most upregulated genes and proteins in the kidney very early after acute kidney injury (AKI) in animal models.19–21 NGAL protein expression was detected predominantly in tubule epithelial cells that were undergoing proliferation and regeneration, suggesting a role in the repair process. 20 Serendipitously, NGAL protein was also detected in the urine and plasma in animal models of AKI, where it preceded the increase in plasma creatinine concentrations. The urine NGAL is derived predominantly from epithelial cells of the distal nephron, although a fraction may come from the systemic pool escaping reabsorption due to proximal tubular injury. 18 Plasma NGAL originates not only from the damaged kidneys (via tubular back-leak) but also from extrarenal organs.4,22
The clinical need for better biomarkers of AKI
An estimated 5–10% of all hospitalized patients and up to 40% of critically ill patients experience an episode of AKI during the course of their illness.23,24 The complications of AKI include increased susceptibility to infections, extrarenal organ damage, the development of chronic kidney disease (CKD) and an increased rate of hospital re-admission.25,26 To estimate the individual patient’s risk of developing AKI, clinical judgement integrates many variables, including the magnitude and course of the underlying renal insult, the increase in functional markers of glomerular filtration rate such as plasma creatinine and urea, and the decline in urine output. 27 However, little progress has been made in methods for the early laboratory diagnosis of AKI, in part due to the reliance on functional markers such as plasma creatinine in daily practice. Many factors can modulate plasma concentrations of creatinine, including age, race, gender, muscle mass, protein intake, hydration status, and certain medications. 28 A single measurement of plasma creatinine cannot differentiate between normal renal function, AKI, chronic kidney disease (CKD), and transient azotaemia with dehydration. 29 Following an acute injury, the rise in plasma creatinine concentration is delayed by several hours to days, precluding the timely institution of potential therapies. 28
NGAL fulfils many of the characteristics important for a useful AKI biomarker. NGAL represents a significant component in the pathophysiology of the disease. The concentration of NGAL in urine or plasma rises rapidly in a dose-dependent manner that is proportional to the degree of damage. NGAL is expressed early after kidney damage, when such injury is still potentially limitable or reversible. NGAL further allows differentiation between the causes of AKI (intrinsic versus transient ‘pre-renal’ AKI), risk stratification, therapy monitoring and prognostication with respect to the need for acute dialysis, duration of hospital stay and mortality. The marker is noninvasive, clinically actionable and reliably measurable on available standardized clinical platforms. Furthermore, NGAL incrementally adds value to the baseline clinical risk assessment, potentially enabling physicians to intervene early to limit the extent of renal injury. 30 Clinical studies that substantiate these statements are reviewed below.
Methods
We searched MEDLINE, PubMed and EMBASE databases for human biomarker studies that included NGAL (January 2005 to October 2013). Studies were eligible to be included in this review if they investigated plasma or urine NGAL for the prediction of AKI in at least 25 human subjects. Studies were analysed in three common clinical settings: cardiac surgery, critical illness and kidney transplantation. Within each group, studies were divided based on whether plasma or urine NGAL was measured. The primary endpoints abstracted from each study included the definition of AKI used, area under the receiver operator characteristic curve (AUC), sensitivity, specificity, cut-off values and the specific assay used. Total AUC, sensitivity and specificity values were calculated as raw (un-weighted) averages of the individual study data in each clinical setting.
Results
Urine NGAL for the early prediction of acute kidney injury after cardiac surgery.

Note: AKI was defined using various criteria. The RIFLE classification of AKI consists of three graded levels of injury (
RRT: renal replacement therapy; NGAL: neutrophil gelatinase-associated lipocalin; KDIGO, kidney disease improving global outcomes; ELISA, enzyme-linked immunosorbent assay.
Blood NGAL for the early prediction of acute kidney injury after cardiac surgery.
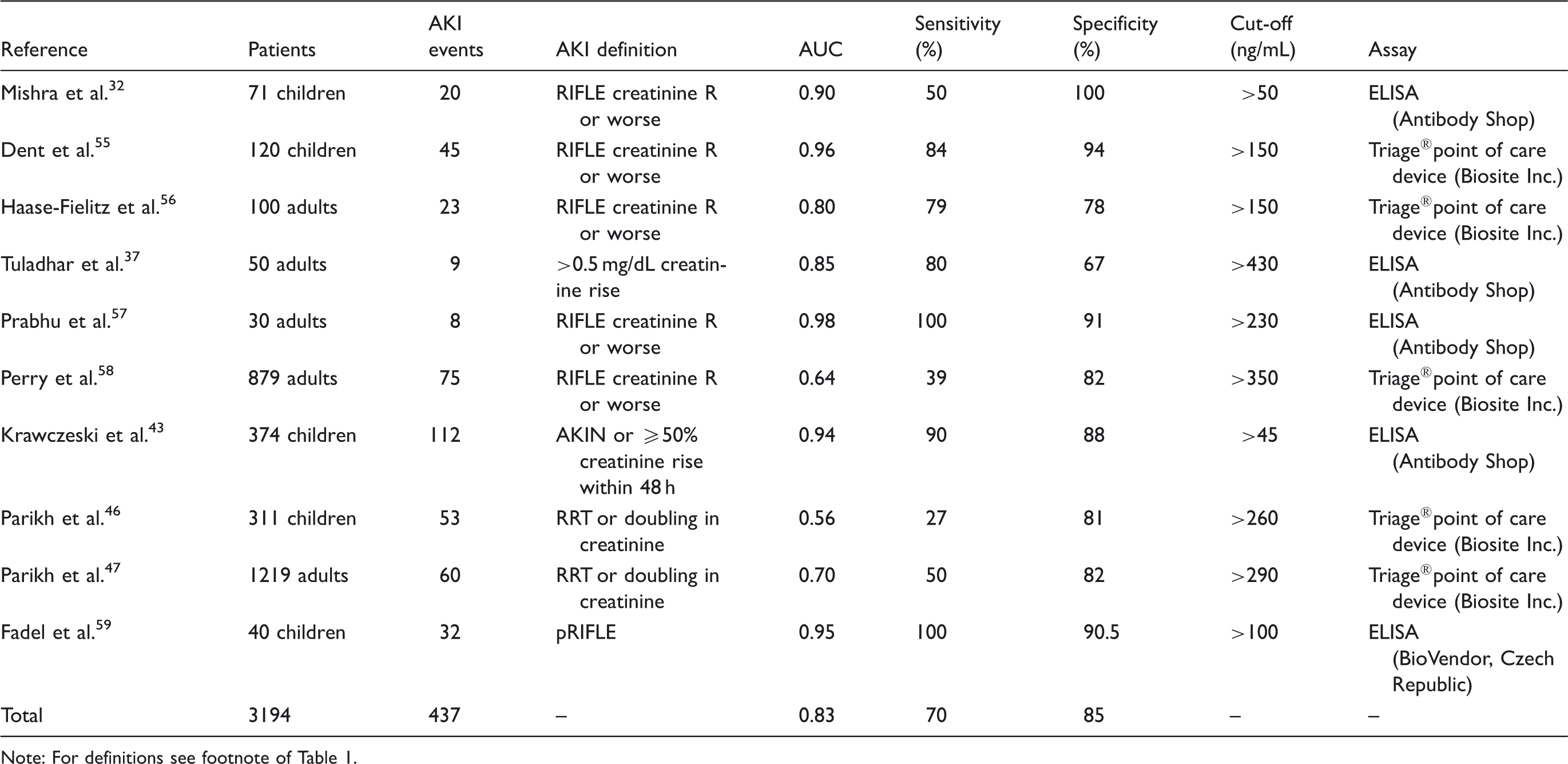
Note: For definitions see footnote of Table 1.
Urine NGAL for the early prediction of acute kidney injury in the critical care or emergency department settings.
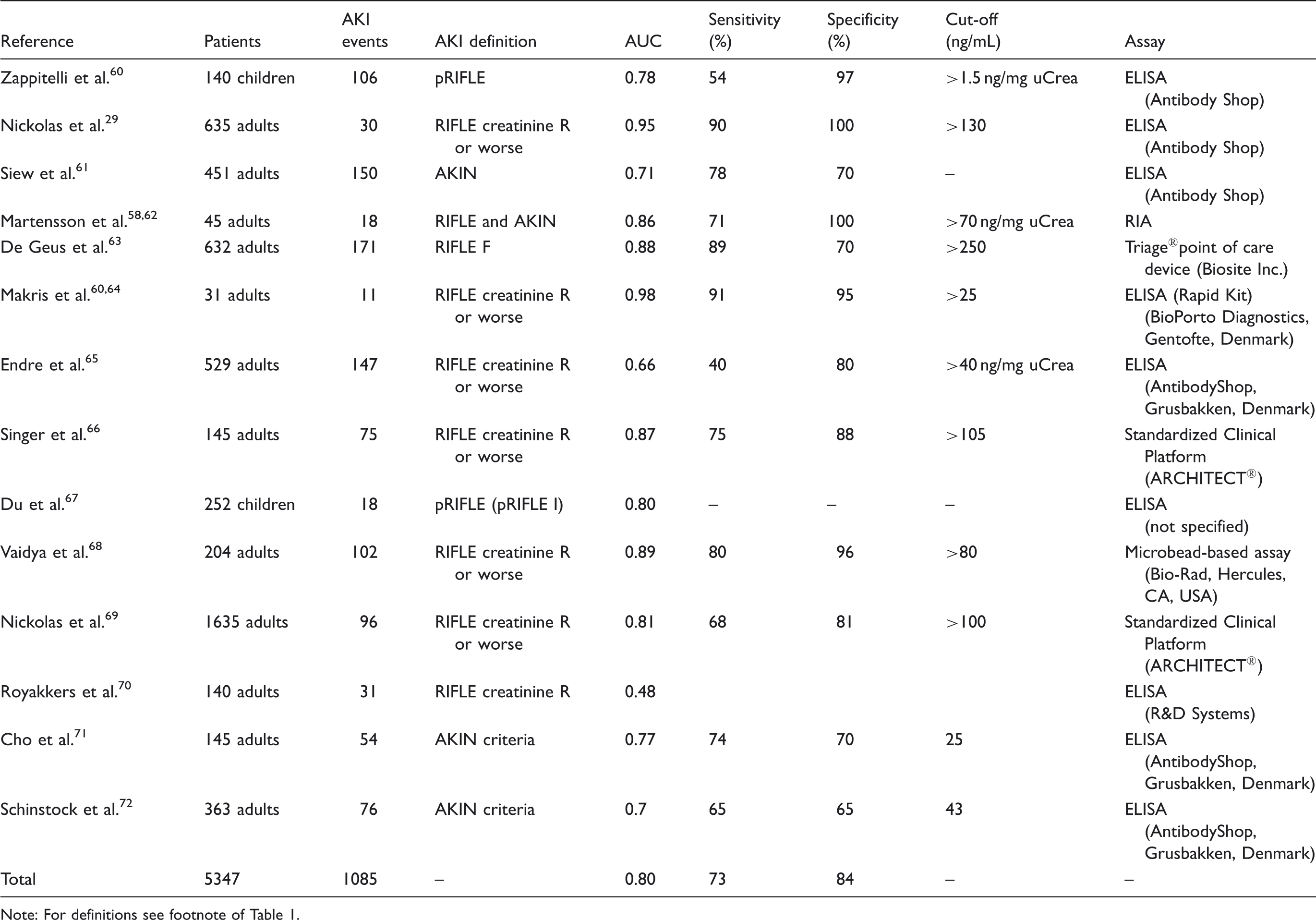
Note: For definitions see footnote of Table 1.
Blood NGAL for the early prediction of acute kidney injury in the critical care or emergency department settings.
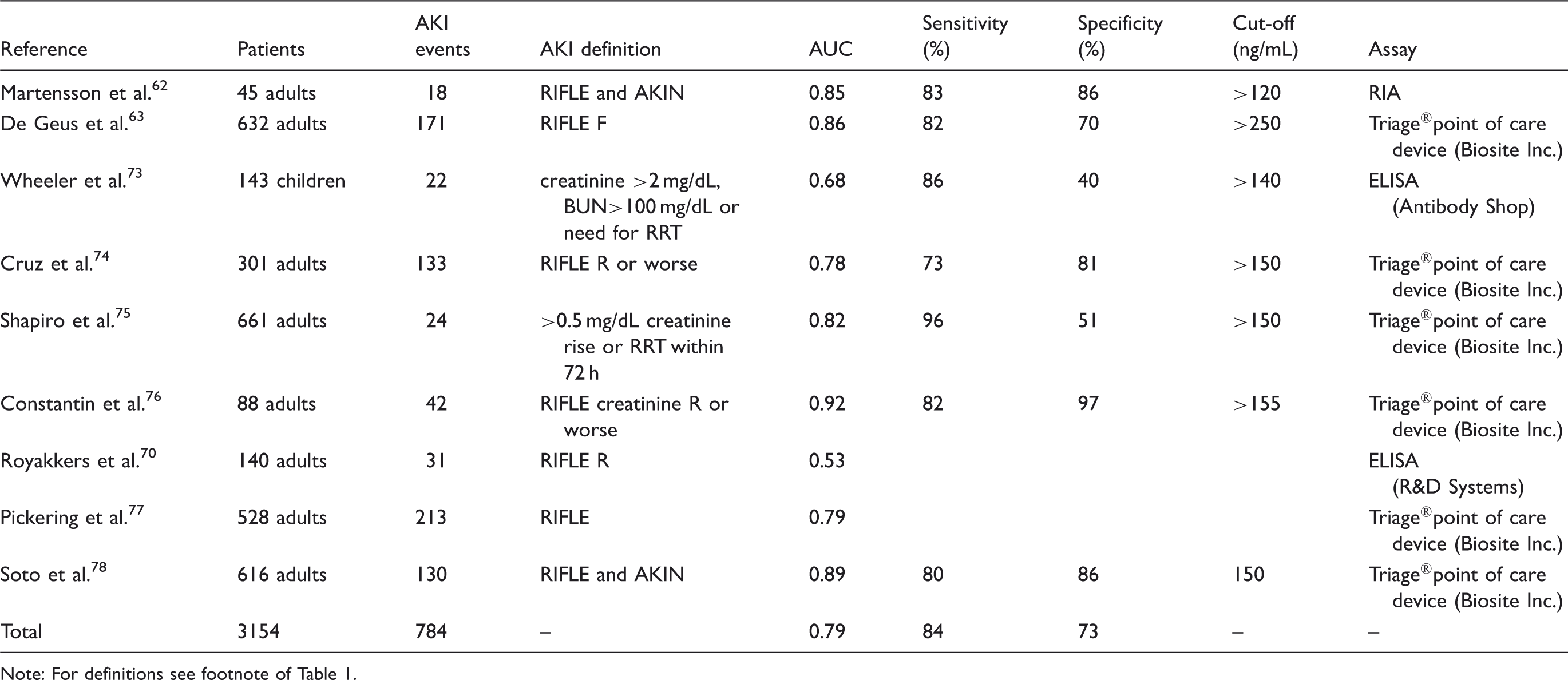
Note: For definitions see footnote of Table 1.
NGAL for the prediction of delayed graft function.
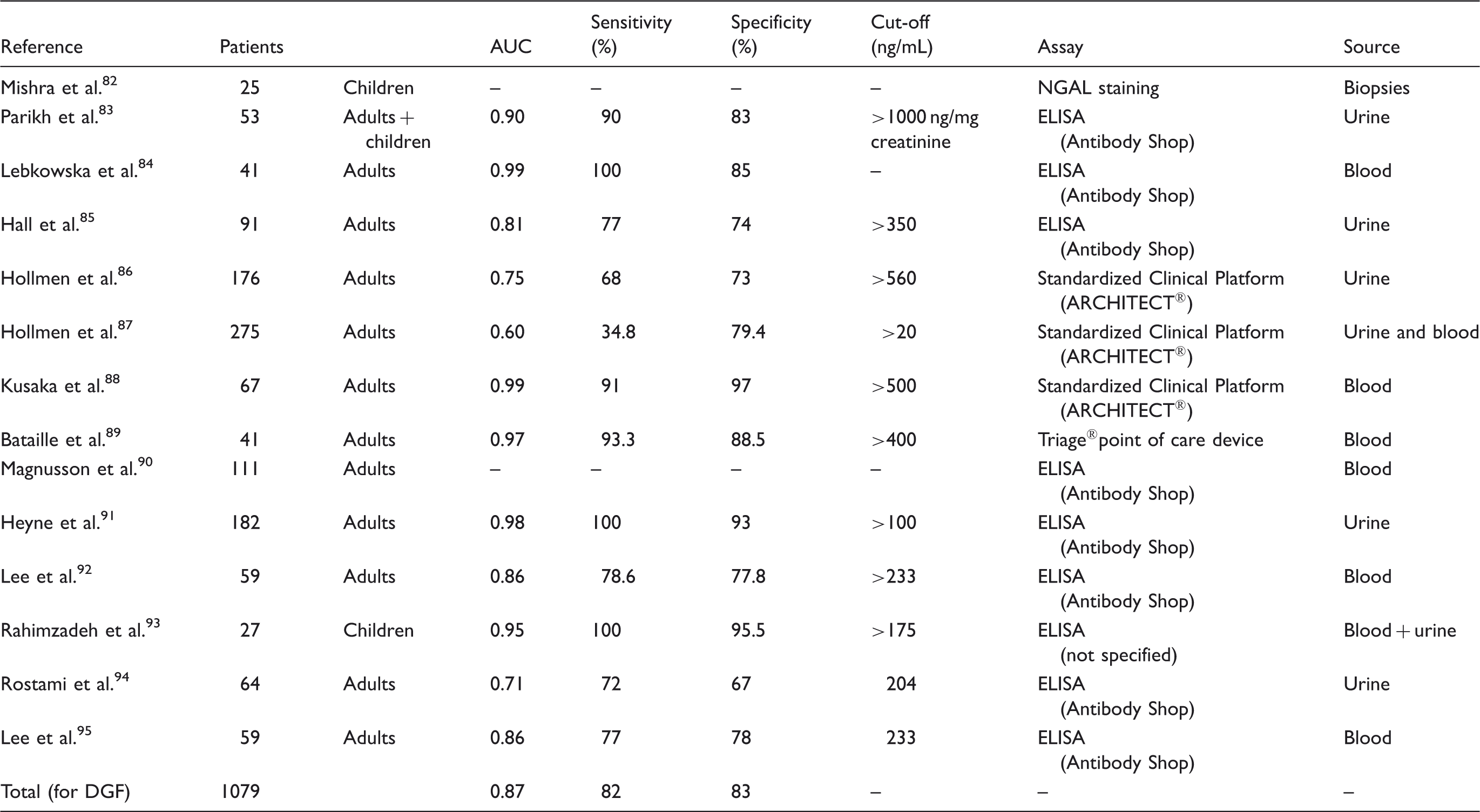
NGAL for the prediction of AKI after cardiac surgery
NGAL measured in more than 7000 patients after cardiac surgery can predict the subsequent development of AKI (AUC 0.82–0.83, Tables 1 and 2). Most recently, the multicentre TRIBE (Translational Research Involving Biomarkers and Endpoints) study prospectively assessed the value of NGAL in 1219 adults 47 and 311 children 46 undergoing cardiac surgery. In both populations, NGAL concentrations measured in urine or plasma peaked within 6 h after surgery and significantly improved risk prediction over the clinical models alone. However, compared to the excellent predictive indices reported in previous single-centre studies,32,34,43,44,55 Parikh et al. demonstrated lower values for the prediction of AKI in children, defined as doubling in creatinine or the need for acute RRT (AUC approximately 0.7) and lower cut-off values. 46 The reasons for this discrepancy may include the different patient populations (exclusion of neonatal patients), differences in assay platforms and prolonged sample storage times compared to earlier single-centre studies.32,34,43,44,55
The importance of the timing of biomarker measurement after cardiac surgery has recently been reported by Krawczeski et al. 44 These authors investigated four urinary AKI biomarkers in a cohort of 220 children without pre-existing CKD and other major comorbidities. 44 They found a temporal relationship of the predictive value of these biomarkers and their combinations. Specifically, urine NGAL was the only biomarker with predictive value as early as 2 h postoperatively, whereas the other biomarkers had no value at this early time point. The predictive value of urine NGAL was excellent (AUC >0.9) and independent of the timing of its measurement.
NGAL for the prediction of AKI in the critically Ill
NGAL has been measured in more than 8500 critically ill patients or in patients admitted in the emergency department, with an excellent overall predictive performance (AUC 0.79-0.80, Tables 3 and 4). In one of the largest recent studies, NGAL measured at ICU admission predicted the development of severe AKI similarly to serum-creatinine-derived estimated glomerular filtration rate (eGFR). 63 However, when patients with eGFR<60 mL/min/1.73 m2 at ICU admission were excluded, plasma (AUC 0.75) and urine NGAL (AUC 0.79) displayed diagnostic superiority over serum creatinine (AUC 0.65) and eGFR (AUC 0.67) for predicting AKI. 63 Thus, the accuracy of NGAL in predicting AKI is highest in patients with normal kidney function prior to the acute illness.
Martensson and colleagues studied the impact of inflammation on NGAL concentrations in plasma and urine in patients with systemic inflammatory response syndrome, severe sepsis and septic shock with and without AKI. 62 In this cohort, both plasma (AUC 0.85) and urine NGAL (AUC 0.86) were good predictors of AKI developing within the subsequent 12 h. However, the ability of plasma NGAL to predict AKI in patients with septic shock was less robust (AUC 0.67) compared to urine NGAL (AUC 0.86). In another study including 661 patients with suspected sepsis, plasma NGAL was superior to plasma creatinine in the prediction of creatinine-based AKI and mortality. 75 Plasma NGAL concentrations of greater than 150 ng/mL were 96% sensitive and 51% specific for AKI. 75 In a recent multicentre study investigating five urinary biomarkers in 1635 emergency department patients at the time of hospital admission, NGAL was the most useful biomarker (81% specificity, 68% sensitivity at a 104 ng/mL cut-off) and predictive of the severity and duration of AKI. 69
NGAL for the prediction of AKI after kidney transplantation
AKI due to delayed graft function (DGF) complicates 5–10% of live donor transplants.79,80 Recipients of deceased donor kidneys develop DGF in up to 50% of cases, implying the need for haemodialysis during the first week after kidney transplantation. 79 DGF results in increased risk of acute rejection episodes, and decreased long-term survival. 81 Clinical studies investigating NGAL as a biomarker for DGF include data from 1079 patients after renal transplantation (Table 5). NGAL measured 6–12 h after kidney transplantation showed an overall excellent performance (AUC 0.87, sensitivity 82% and specificity 82%) for the prediction of DGF (Table 5).
NGAL staining intensity obtained in deceased donor kidney biopsy samples within 1 h of vascular anastomosis predicted the degree of AKI and correlated with peak postoperative serum creatinine as well as RRT requirement which occurred 2–3 days later. 82 In another study, urine NGAL measured on the day of kidney transplantation identified patients with severe kidney injury and inferior long-term organ survival, and allowed for prediction of DGF as well as prolonged DGF, even in patients with good urine output and decreasing creatinine. 86 In 41 patients on haemodialysis prior to kidney transplant, plasma NGAL decreased significantly one day after successful kidney transplantation, prior to a fall in functional renal markers such as serum cystatin C or creatinine. 84 In a large study by Parikh and colleagues, urine NGAL measured on day 0 of deceased donor kidney transplantation predicted DGF earlier than other clinical scores and markers that are currently in use. 83 NGAL concentrations also showed excellent separation at nearly every time point between recipients with DGF, slow graft function and immediate graft function. Adding NGAL measurements significantly improved the risk prediction of the current clinical model for the diagnosis of DGF. 85
Discussion
Critical appraisal of the current status of NGAL as a biomarker of AKI
From 58 manuscripts that examined more than 16,500 patients, we identified collective support for the use of NGAL as a biomarker of AKI. Irrespective of the clinical setting, both urine and plasma NGAL were predictive of AKI and its severity, with an overall AUC of 0.79–0.87. More importantly, NGAL significantly improved the prediction of AKI risk over the clinical model alone in all three clinical settings investigated. In comparing urine and plasma, the availability of biofluid and assay platform will likely be the primary determinants of whether urine or plasma NGAL will be measured in each specific setting of AKI.
However, none of the published studies has paid adequate attention to stringent guidelines and tools for assessing the accuracy of reporting biomarker studies, such as Standards for Reporting Studies of Diagnostic Accuracy (STARD), 96 Quality Assessment of Diagnostic Accuracy Studies(QUADAS) 97 and Strengthening the Reporting of Observational studies in Epidemiology(STROBE) 98 guidelines for observational cohort studies. The objective of the STARD initiative is to improve the accuracy and completeness of reporting of studies of diagnostic accuracy and to allow readers to assess the potential for bias in the study and its generalisability. The STARD criteria are not solely a quality assessment tool but rather a prospective tool used to plan a well-designed study. By using the STARD checklist 96 during the design phase, study errors such as selection bias or overlap between the index test (e.g. biomarker) and reference test (e.g. plasma creatinine) findings can be minimized. Quality lists such as QUADAS 97 are used to retrospectively evaluate the methodological rigour of a completed study investigating the diagnostic accuracy of a test or measure. Researchers should be aware of such guidelines when planning future biomarker studies and interpreting the results of such studies. In addition, there has been some heterogeneity in the reported predictive value of NGAL in part depending on the AKI definition, 99 conditions of sample storage, 100 the time point of measurement 44 or the assay used.
The concept of outcomes in ‘biomarker-positive, creatinine-negative’ patients has recently been explored. There are two studies enrolling about 4000 cardiac surgical, critically ill or emergency department patients who were grouped according to their NGAL and serum creatinine (sCrea) status (NGAL−/sCrea−, NGAL+/sCrea−, NGAL−/sCrea+, NGAL +/sCrea+).69,101 Both studies found that measurement of the concentrations of NGAL complemented the information obtained by measurement of creatinine concentrations in establishing the diagnosis of AKI and predicting prognosis. A substantial proportion of patients (15–20%) had increased NGAL concentrations even in the absence of a rise in serum creatinine concentration. This previously undetectable condition (‘subclinical AKI’) was associated with a 2–3-fold increased risk of death or the need for RRT compared to that of patients in whom serum creatinine concentration was not increased. Notably, even in patients with significant loss of renal function, measurement of tubule damage biomarker levels still added prognostic information, as patients with increased concentrations of both NGAL and serum creatinine displayed by far the worst prognosis.
Overall, the data support the view that measurement of tubule damage markers such as NGAL results in substantial added value to serum creatinine measurements. Based on the available data, an NGAL cutoff value of approximately 100 ng/mL, measured on a standardized clinical laboratory platform, appears to be useful for ruling out AKI in those with normal baseline kidney function whereas a cut-off value of >150 ng/mL can be used as diagnostic for AKI.29,46,69,74 A potential cut-off value for the differentiation of prerenal azotaemia from intrinsic AKI is in the range of 100 ng/mL.29,69 The concept of NGAL cutoff values has recently been re-examined for patients admitted from the emergency department setting. 78 In this prospective study of 616 patients, plasma NGAL measured using a point-of-care clinical platform accurately predicted intrinsic AKI and its severity. However, a discrete plasma NGAL cut-off for dichotomizing between AKI and non-AKI could not be identified. Instead, a grey zone of plasma NGAL concentrations (>97 ng/mL to <133 ng/mL) was associated with a moderate risk for AKI. Patients whose plasma NGAL concentrations exceeded the grey zone were found to have a 10-fold greater risk of AKI. Thus, patients who are at risk for AKI based on clinical presentation would benefit from point-of-care plasma NGAL measurements. Irrespective of serum creatinine concentrations, patients with plasma NGAL concentrations that exceed the grey zone levels (>133 ng/mL) could be triaged and managed as cases of true intrinsic AKI. Patients in the grey zone of plasma NGAL concentrations who have clinical AKI risk factors (older age, CKD, cardiovascular disease) should also be considered at high risk for intrinsic AKI. Additional cut-offs for blood and urine NGAL and their bedside utility are forthcoming and will be in part determined by the clinical setting, regulatory intended use guidelines, platform standardization and interlaboratory calibration.
Given the above considerations, NGAL may complement a standardized diagnostic approach to AKI, and help clinicians improve their ability to make an early AKI diagnosis. Markers of renal function will likely remain important even after tubule damage markers are fully established. Glomerular filtration markers such as serum creatinine or cystatin C are still valuable for the diagnosis and quantification of any excretory function loss (e.g. for drug dosing) and prognosis (e.g. for development or deterioration of CKD). Urine output still represents a useful criterion for adjustments to fluid balance, and for the commencement or ending of RRT. Specifically in the case of kidney transplant recipients, the degree of injury and the likelihood of DGF, determined during the first postoperative days by NGAL concentrations, could guide protocol transplant biopsy decisions in addition to the frequency of early follow-up visits. Future research should focus on the question whether NGAL could help to distinguish between acute rejection and nephrotoxicity due to medication (e.g. calcineurin inhibitors) in patients after renal transplantation.
NGAL clinical analytical platforms, assays and assay performances
Analytical methods of NGAL measurement.

The point-of-care Triage® NGAL test performed on the Triage Meter can process a single sample. Using this device, plasma NGAL concentration was found to be an excellent early predictive biomarker of AKI, morbidity and mortality in various clinical settings.55,56,74,76 High correlation between research ELISA’s and the Triage® NGAL device has been reported. 55
The Abbott ARCHITECT analyser can process large numbers of samples and this urine NGAL assay proved to be precise and reproducible including between days and between reagent lots. 100 There was also good agreement with a commercially available ELISA assay (Bioporto) as well as good short-term sample stability at 2–8℃. However, for longer-term storage samples should be kept at −70℃ or colder. 100 Urine NGAL concentrations measured by the ARCHITECT assay were found to be highly correlated with research-based ELISA measurements, 34 and an excellent early predictive biomarker of AKI in several clinical settings.34,46,47,66,69,86,87
The NGAL Test™ runs on open channels of most biochemistry analysers, effectively giving almost any laboratory immediate access to a method to measure NGAL. However, data using this system on standardized clinical platforms are not available at the present time.
A recent study has compared the analytical performance of these assays in 68 urine samples. 102 Imprecision, parallelism, recovery selectivity, limit of quantitation, interference, hook effect and interassay agreement were determined. The ARCHITECT urine NGAL assay on i2000SR demonstrated satisfactory performance for all parameters tested. Problems were identified with limit of quantitation for the NGAL Test™ on the Siemens ADVIA® 1800 platform. 102 Thus, variability may exist between NGAL assays in terms of their performance, and this should be taken into account when interpreting results of urinary NGAL testing.
Recently, it was shown that the NGAL monomer is the predominant form secreted by injured distal tubule epithelial cells, whereas the dimer is the predominant form secreted by neutrophils.7,8 Assays that specifically detect the AKI-specific monomeric form are expected to outperform assays that detect multiple forms. 69 Cai and colleagues investigated the effect of antibody configuration on assay performance and found that the epitope specificities used in the ELISA’s paralleled differences in the antibodies to identify the different forms of urine NGAL. 7 Martensson and co-workers developed two ELISA’s which were able to distinguish monomeric from dimeric NGAL. Monomer-specific assays are expected to reduce any confounding effect of neutrophil involvement during bacteriuria or sepsis. 8 In a study by Nickolas et al., a marked correlation between urine NGAL concentrations measured by immunoblot and by the ARCHITECT platform was found. 69 The AUC for the detection of intrinsic AKI by urine NGAL determined by the standardized clinical platform was identical to that of monomeric urine NGAL determined by immunoblot (AUC 0.81), confirming that monomeric urine NGAL is the form pertinent to AKI. 69
Nickolas and colleagues have shown that urine from patients with CKD display multiple forms of NGAL. 103 While the majority of NGAL was monomeric, several larger forms were also detectable, likely representing trimeric forms and perhaps complexes with other proteins. However, only the monomeric form of NGAL correlated inversely with estimated GFR, and directly with degree of tubular atrophy and interstitial fibrosis on kidney biopsies. 103 The NGAL ARCHITECT platform detected predominantly the monomeric form, 103 and commercial NGAL ELISAs also detect predominantly the monomeric form and correlate strongly with the ARCHITECT platform, 34 rendering them both potentially useful tools for exploring the role of urine NGAL in AKI and CKD.
Other limitations of NGAL as an AKI Biomarker
The information provided by tubule damage biomarker concentrations has other limitations. Although the majority of NGAL in urine or blood derives from the injured kidney, non-renal NGAL sources have been reported that might adversely affect the diagnostic accuracy. 1 The effect of CKD on the accuracy of NGAL for AKI prediction or differentiation from prerenal states remains a confounding factor that is open to further research. The available data suggest that patients with stable CKD display low concentrations of NGAL similar to normal controls, but those with progressive CKD may exhibit higher concentrations that may approach those seen in prerenal states or even in mild forms of AKI. The lack of a gold standard AKI definition, the lack of specific cut-off values for NGAL and differences in clinical assay characteristics are additional limitations to the widespread use of NGAL in clinical practice at the present time.
We recommend that future clinical studies using NGAL should employ the STARD criteria during study planning, and should be conducted as multicentre, multimarker and multitime point studies that employ pertinent clinical end points (and not merely creatinine-based endpoints) to further verify the diagnostic and prognostic value of NGAL. Attention should also be paid to the clinicians’ best guess at the bedside for AKI prediction, and biomarkers should demonstrate added value beyond clinical risk scores. In addition, freshly collected urine or blood specimens with NGAL measurement on clinical laboratory platforms or point-of-care devices reflecting closest proximity to clinical practice should be used. Further clinical and experimental characterization of patient groups according to magnitude, duration and type of renal function loss and tubular damage is needed to better understand the syndrome of AKI. Finally, large interventional trials are needed where therapeutic decisions are made based on clinical scoring and NGAL concentrations in a randomized fashion focusing on patient-relevant outcomes. A multimodal approach of clinical and biomarker-assisted decision-making for AKI prediction and treatment is likely to be most promising.
Footnotes
Acknowledgements
This article was prepared at the invitation of the Clinical Sciences Reviews Committee of the Association for Clinical Biochemistry and Laboratory Medicine.
Declaration of conflicting interests
PD is a co-inventor on submitted patents on NGAL as a marker of kidney injury. MH received lecture fees from Abbott Diagnostics and Biosite Inc/Alere.
Funding
Work described herein that was completed in the laboratory of PD was supported by grants from the NIH (P50 DK096418).
Ethical approval
Not applicable.
Guarantor
PD.
Contributorship
All three authors conceived the study. All three authors performed an independent literature search, compiled the results, and performed a critical appraisal of the literature. All three authors wrote the manuscript.
