Abstract
Background
It is not possible to diagnose acute kidney injury (AKI) in early stages with traditional biomarkers. Kidney injury molecule-1 (KIM-1) is a novel biomarker promising the diagnosis of AKI in early stages. We studied whether urinary and serum KIM-1 (KIM-1 U and KIM-1 S ) concentrations were useful in predicting cisplatin-induced AKI in early stages.
Methods
We prospectively analysed 22 patients on cisplatin treatment. KIM-1 S and KIM-1 U concentrations were assessed in the samples of the patients on four different time periods (before treatment [BT], first [AT1], third [AT3] and fifth [AT5] day after treatment).
Results
KIM-1 U concentrations on the first day after cisplatin treatment in patients with AKI were significantly increased compared to both KIM-1 U concentrations of the same patients BT (P = 0.009) and to AT1-KIM-1 U concentrations of the patients without AKI (P = 0.008). A receiver operating characteristic analysis revealed that AT1-KIM-1 U concentrations may predict AKI with an 87.5% sensitivity and 93.3% specificity (area under the curve = 0.94). KIM-1 S concentrations were not significantly changed between BT and AT periods.
Conclusions
KIM-1 U concentrations may predict cisplatin-induced AKI in early stages with high sensitivity and specificity.
Introduction
Cisplatin is one of the most effective and most widely used chemotherapeutic agents worldwide.1,2 It is preferred as first-line agent in many solid tumours especially head–neck, gastrointestinal, urogenital and lung cancers.3,4 Major side effects of cisplatin (nephrotoxicity, neurotoxicity, autotoxicity, nausea and vomiting) complicate its clinical use. 5 Nephrotoxicity is the main side effect of cisplatin that may limit its dosage. Studies in literature reported that acute kidney injury (AKI) developed in one-third of the patients on cisplatin treatment. 2 Hydration before chemotherapy is the only effective approach to prevent cisplatin-induced acute kidney injury (CI-AKI). 6 AKI may develop despite adequate hydration and even may require termination of the chemotherapy.
Drug-induced damage that results due to apoptosis in renal tubular cells is the most important physiopathological mechanism in the development of CI-AKI. 2 The most common predictors of renal injury are serum creatinine (sCr), blood urea nitrogen (BUN) and glomerular filtration rate (GFR). However, none of these laboratory parameters is enough sensitive or specific to predict AKI in early stages.7,8 Urinary neutrophil gelatinase-associated lipocalin (NGAL) concentrations have been shown to be increased in early stages of CI-AKI in a recent study. However, these data have not been confirmed yet, and thus, NGAL concentrations are not being used in clinical practice. 9 Therefore, new bioindicators are needed in the diagnosis of CI-AKI, which indicate tubular damage especially in the early period.
Kidney injury molecule-1 (KIM-1) is a transmembrane protein initially isolated from the postischaemic rat kidneys by Ichimura et al., 10 and normal renal tissue has very small amounts of that molecule. There are accumulating evidence in nephrotoxic animal models that KIM-1 U concentrations predict CI-AKI earlier than traditional markers, elevate in the earlier stage and remain stable for a long time in urine.11,12 To our knowledge, preclinical studies that observed the role of KIM-1 U in CI-AKI have not been suggested by following clinical trials. In addition, although it has been shown that KIM-1 S concentrations have an important role in predicting acute rejection in early stage renal transplant subjects, its diagnostic value in CI-AKI remains unclear. 13 The lack of data about the changes in KIM-1 S and KIM-1 U concentrations during CI-AKI requires further studies. We intended to assess the changes of KIM-1 concentrations in patients with and without AKI after cisplatin treatment and compare with traditional markers of renal damage. We also aimed to study whether KIM-1 U and KIM-1 S concentrations were early predictors of CI-AKI.
Material and methods
Study population
We observed prospectively consecutive patients with solid tumours admitted to the Oncology department of our institution between September 2012 and June 2013. We informed the patients fulfilled the inclusion criteria but without exclusion criteria about the study protocol, and informed consent was given at this stage in the beginning of the study.
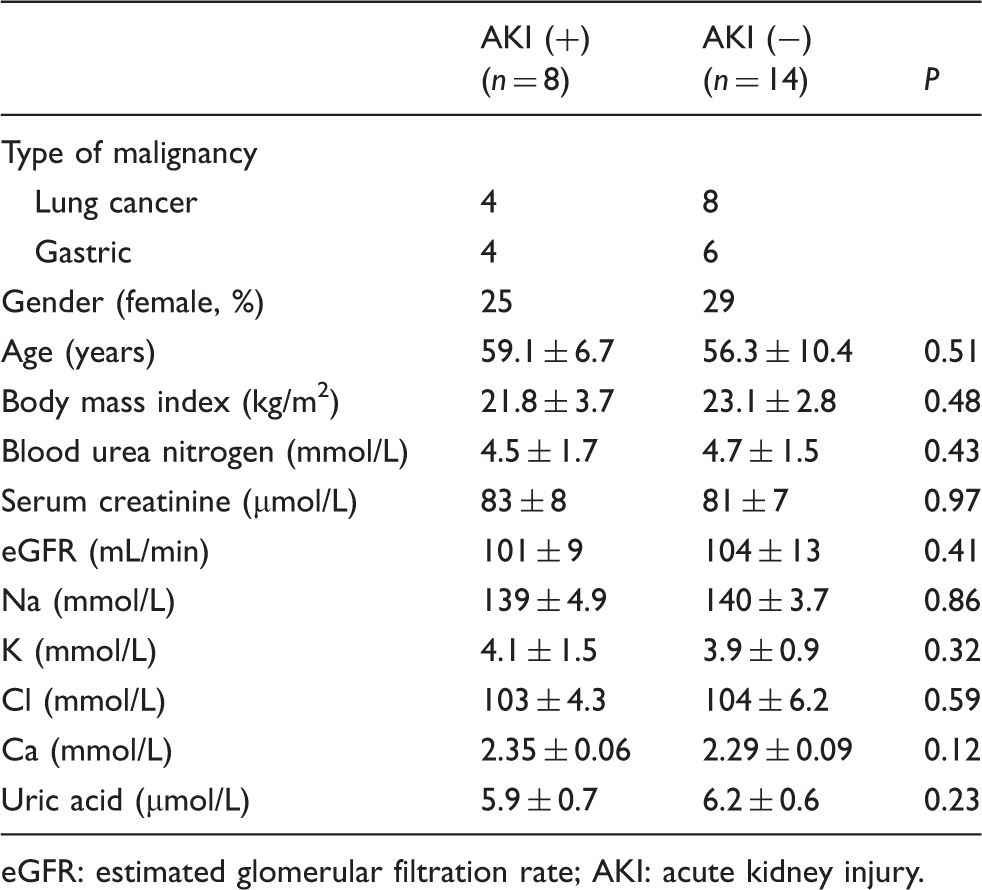
We included the patients if their renal functions were normal; sCr lower than 97 μmol/L and estimated GFR (eGFR) higher than 90 mL/min. We calculated eGFR, based on the proportion of serum/plasma creatinine. For this purpose, we used the Modification of Diet in Renal Disease formula, which is the common equation to calculate eGFR.14,15 First-line cisplatin treatment was indicated for all of these patients. Only the patients who never received chemotherapy before were included in the study for avoiding cumulative toxicity. Primary malignities of the patients were as follows: gastric cancers in 10 patients and non-small cell lung cancer in 12 patients. Serum concentrations of BUN, sCr, eGFR, Na, K, Cl and uric acid were in reference range in study population.
Exclusion criteria were as follows: a history of chemotherapy, patients younger than 18 and older than 70 years, history of renal failure and diabetes mellitus and patients with active infection, heart failure and serious endocrine diseases. We also excluded patients with a history of nephrotoxic drug or non-steroidal anti-inflammatory drug use in last 30 days, urinary operation or renal stone disease, peritonitis carcinomatosis, cerebral or hepatic metastasis and dehydrated patients because of general and neurologic impairment. Patients with elevated serum concentrations of transaminases and uric acid, anaemia (haemoglobin <100 g/L) and electrolyte disturbances were excluded from the study. Patients with possible renal dysfunction on laboratory history and renal abnormalities on imaging studies were not included to the study cohort. Contrast enhanced imaging study in the past two weeks was also considered as an exclusion criteria to avoid confounding effects of contrast nephropathy. Sixty-one patients with various cancers were assigned for this study, and 39 were not included in the study (28 patients excluded according to exclusion criteria, 6 patients were not given informed consent and 5 patients could not obey the study protocol). Study was performed with remained 22 patients. Study was approved by the local ethics committee of Abant Izzet Baysal University.
Collection of serum and urinary samples
Urinary and serum samples were obtained from all participants before cisplatin treatment and on first (AT1), third (AT3) and fifth day (AT5) after treatment. All blood samples were obtained after fasting in the morning. Midflow urinary samples were collected in disposable urine cups that did not contain preservatives. All blood and urinary samples were centrifuged for 10 min at 1200 g. Serum BUN, sCr, Na, Cl, K and uric acid concentrations were studied as a routine protocol of cisplatin treatment. Serum and urinary supernatants were stored at −70℃ until laboratory analysis for KIM-1. Storage duration for the samples was less than 10 months. Equal procedures were used in collection, handling, transport and storage of all samples to standardize preanalytical factors which could affect laboratory assessment. 16 KIM-1 analyses of samples were performed simultaneously.
Measurement of KIM-1S and KIM-1U
KIM-1 S concentrations were measured by enzyme-linked immunosorbent assay (ELISA) according to the manufacturer’s instructions (Cusabio, Wuhan, China). Detection range of the kit was 0.312–20 ng/mL. Intra-assay and inter-assay precision were %CV: <8% and <10%, respectively. Minimum detectable dose of KIM-1 was 0.078 ng/mL.
KIM-1 U concentrations were measured by ELISA according to the manufacturer’s instructions (Aviscera Bioscience, Santa Clara, USA). Detection range of the kit was 31–2000 pg/mL. Intra-assay and inter-assay precision were %CV: 4–8% and 6–8%, respectively. Minimum detectable dose of KIM-1 was 3 pg/mL.
The kits we used were intended to determine extracellular domain of KIM-1. These assays were specific for human KIM-1 which is known to have no cross-reactivity or interference.
Serum BUN, sCr, Na, K, Cl, uric acid and urinary creatinine (uCr) analyses were performed with autoanalysers (Architect c 8000, Abbot Laboratories, USA).
Prevention and detection of acute kidney damage
Acute Kidney Injury Network (AKIN) criteria were used for the detection and staging of acute renal damage.17,18 Patients were classified with stage 1 AKI based on elevation in sCr ≥1.5–2 folds (≥26.4 μmol/L) compared to baseline sCr in 48 h. Stages 2 and 3 AKI were defined as 2–3 folds increase in sCr and more than 3-fold increase in sCr compared to baseline sCr, respectively. Because none of the patients was diagnosed with AKI on the first day, eight patients were classified as AKI (+) and 14 as AKI (−) according to the sCr measurements at AT3.
Intravenous hydration with 0.9% NaCl solution was infused at a rate of 1000 cc before chemotherapy. A 75 mg/m2/day dose of cisplatin was infused in 1000 cc of saline solution during 4 h.19,20 Gemcitabine (in patients with non-small cell lung carcinoma) or Capecitabine (in patients with gastric carcinoma) added to Cisplatine, which are both antimetabolite agents excreted primarily from the liver. Patients in the study have not received any nephrotoxic agents including antibiotics. BT sCr and eGFR were compared to AT values to detect the stage of renal damage. Patients diagnosed with CI-AKI according to AKIN criteria were treated appropriately. 21
We standardized laboratory results by proportioning KIM-1 U to uCr and KIM-1 s to sCr to avoid of possible effects of hydration status on laboratory analysis.
Statistical analysis
SPSS 15.0 was used for statistical analysis. Continuous variables were presented as mean ± standard deviation, while categorical variables were presented as a percentage. Numerical variables of the groups were distributed normally, and variances were equal. Since the baseline demographics and laboratory values were normally distributed, the independent samples t-test was performed to compare these parameters between the patients with and without AKI. The capacity of urine KIM-1 values in predicting the presence of CI-AKI was analysed using receiver operating characteristic (ROC) curve analysis. When a significant cut-off value was observed, the sensitivity, specificity, positive and negative predictive values were presented. While evaluating the area under the curve (AUC), a 5% type-1 error level was used to accept a statistically significant predictive value of the test variables. A value of P < 0.05 was considered statistically significant.
Results
Study population and baseline characteristics
Baseline demographic features and laboratory data of the study population [AKI (+) and AKI (−) groups].
eGFR: estimated glomerular filtration rate; AKI: acute kidney injury.
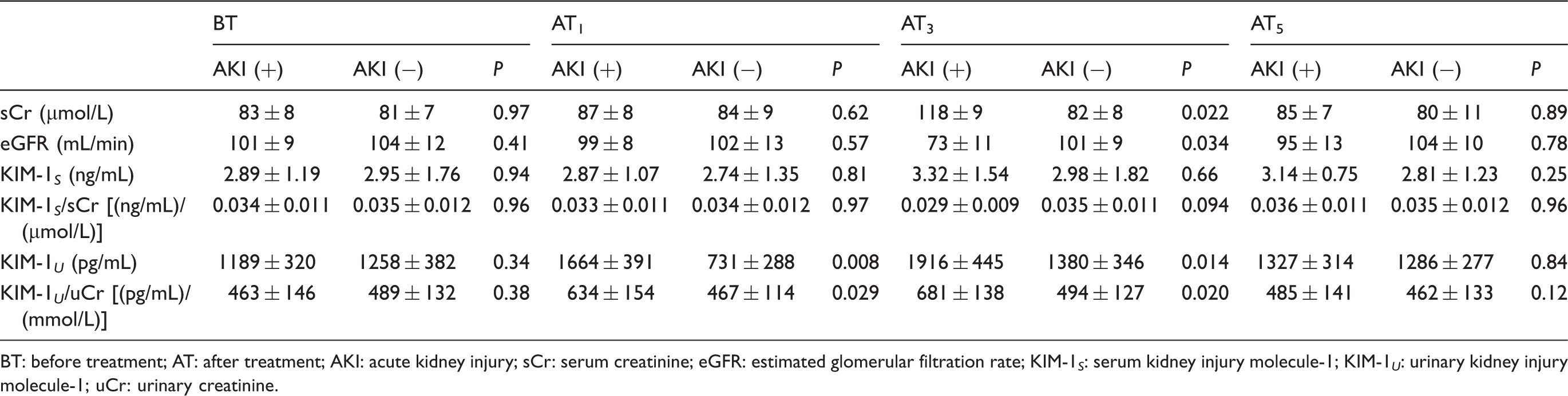
Comparison of sCr, eGFR, KIM-1 U , KIM-1 S KIM-1 S /sCr and KIM-1 U /uCr concentrations of study groups on before and after treatment periods (first, third and fifth days).
BT: before treatment; AT: after treatment; AKI: acute kidney injury; sCr: serum creatinine; eGFR: estimated glomerular filtration rate; KIM-1 S : serum kidney injury molecule-1; KIM-1 U : urinary kidney injury molecule-1; uCr: urinary creatinine.
First day after cisplatin treatment
None of 22 patients was diagnosed with AKI according to comparison of AT1-sCr and BT-sCr concentrations. AT1-KIM-1 U concentrations of AKI (+) group were significantly increased compared to both BT-KIM-1 U concentrations of the same group (P = 0.009) and AT1-KIM-1 U concentrations of AKI (−) group (P = 0.008). AT1-KIM-1 U was significantly decreased compared to BT-KIM-1 U in AKI (−) group (P = 0.021); however, there was no statistically significant difference according to KIM-1 U /uCr (P > 0.05). AT1/BT fold-change of KIM-1 U was significantly increased in AKI (+) compared to AKI (−) group (2.26 ± 0.45, 0.67 ± 0.19; P = 0.004)
The ROC curves were constructed in order to test the ability of AT1-KIM-1
U
concentration, KIM-1
U
/uCr and AT1/BT fold-change of KIM-1
U
to predict AKI. The area under ROC curve of AT1-KIM-1
U
concentration was 0.94 [95% confidence interval (CI) = 0.75–0.99; P < 0.0001]. KIM-1
U
cut-off value was 1412 pg/mL on AT1, which separated AKI (+) from AKI (−) with a sensitivity of 87.5% and a specificity of 93.3%, and the positive and negative predictive values were 87.5% and 93.3%, respectively. The AUC of KIM-1
U
/uCr was 0.83 (95% CI = 0.61–0.95; P = 0.0002). KIM-1
U
/uCr cut-off value was 510 on AT1, which separated AKI (+) from AKI (−) with a sensitivity of 87.5% and a specificity of 64.3%, and the positive and negative predictive values were 58.3% and 90%, respectively. The AUC of AT1/BT fold-change was 0.92 [95% CI = 0.74–0.99; P < 0.0001]. AT1/BT fold-change of KIM-1
U
cut-off value was 0.93 on AT1, which separated AKI (+) from AKI (−) with a sensitivity of 99% and a specificity of 80%, and the positive and negative predictive values were 72.7% and 98%, respectively. There were no significant differences between these AUCs (all P > 0.05) (Figure 1).
The receiver operating characteristic curves for AT1-KIM-1
U
concentration, AT1-KIM-1
U
/uCr and AT1/BT fold-change of KIM-1
U
. The area under the curve (AUC) was 0.94 for AT1-KIM-1
U
concentration [95% confidence interval (CI) = 0.75–0.99]. AUCs of AT1-KIM-1
U
/uCr were 0.83 [95% CI = 0.61–0.95]. AUCs of AT1/BT fold-change of KIM-1
U
were 0.92 [95% CI = 0.74–0.99]. AUCs of AT1-KIM-1
U
concentration and AT1/BT fold-change of KIM-1
U
were significantly higher than the diagonal reference line (AUC = 0.5) (all P < 0.0001). Similarly, AUCs of AT1-KIM-1
U
/uCr were significantly higher than the diagonal reference line (P = 0.0002).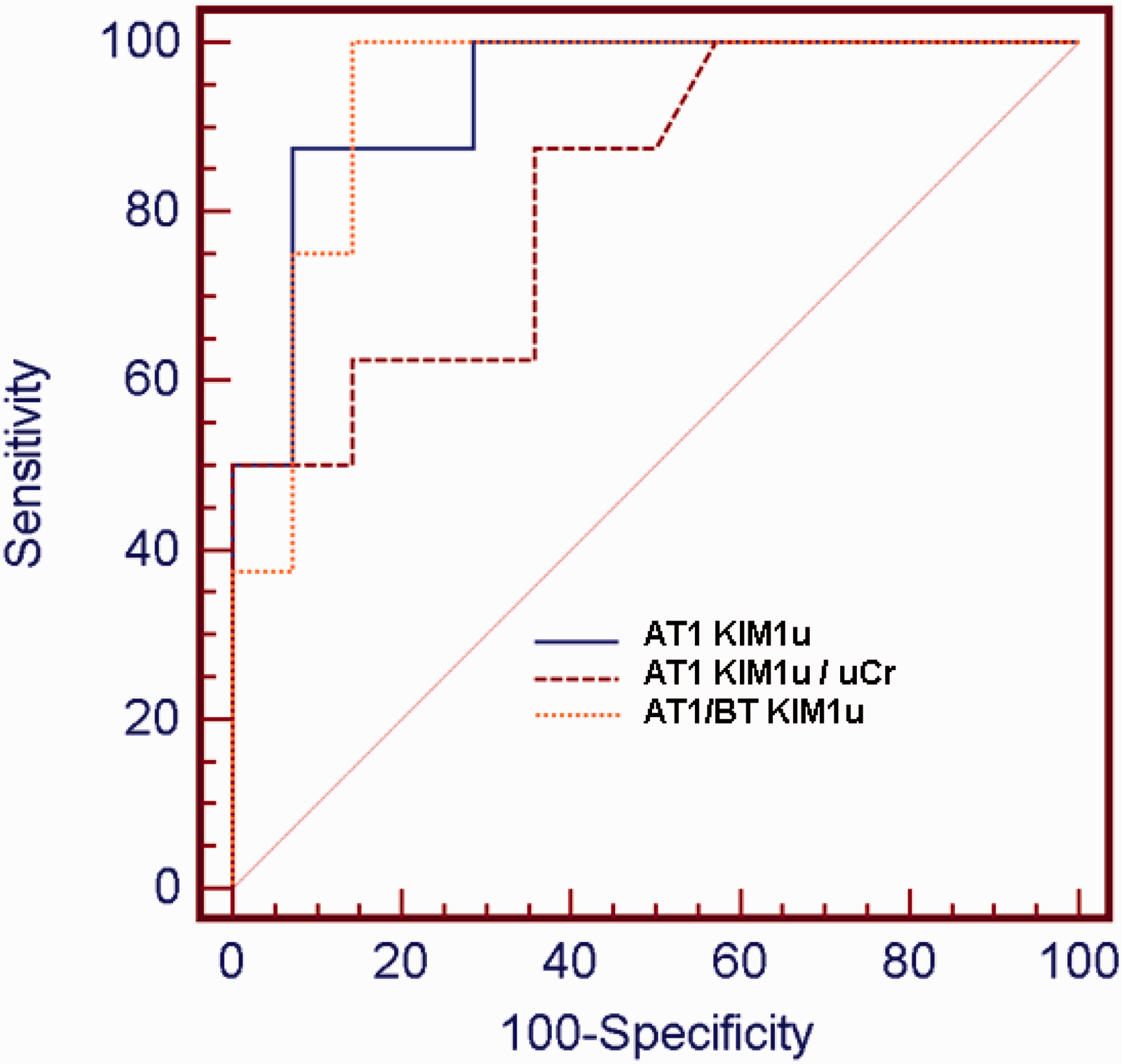
Third day after cisplatin treatment
Eight of 22 patients were classified as AKI stage 1 [AKI (+)] according to comparison of AT3-sCr and AT1-sCr concentrations. AT3-sCr (P = 0.022), AT3-KIM-1 U (P = 0.014) and KIM-1 U /uCr (P = 0.020) concentrations in AKI (+) group were significantly increased compared to AKI (−) group. AT3-sCr (P = 0.015) and AT3-KIM-1 U concentrations (P = 0.0011) were also significantly increased compared to BT values in AKI (+) group. AT3/BT fold-change of KIM-1 U concentrations in AKI (+) group were significantly increased compared to AKI (−) subjects (2.04 ± 0.55, 1.21 ± 0.39; P = 0.043).
Fifth day after cisplatin treatment
None of 22 patients was diagnosed with AKI according to comparison of AT5-sCr and AT3-sCr concentrations. There was no significant difference between AKI (+) and AKI (−) groups in terms of AT5-sCr, eGFR, KIM-1 S , KIM-1 U values and KIM-1 U /uCr (all P > 0.05). There was no significant difference between AKI (+) and AKI (−) groups in terms of AT5/BT fold-change of KIM-1 U concentrations (1.21 ± 0.22, 1.06 ± 0.59; P = 0.18).
Discussion
We studied whether serum and KIM-1 U concentrations should be considered as predictors of CI-AKI. Results of the present study showed that AT1-KIM-1 U concentrations predict AKI in early stage with a high sensitivity and specificity. Furthermore, AT1/BT fold-change of KIM-1 U and AT1-KIM-1 U /uCr was as useful as AT1-KIM-1 U concentrations in predicting CI-AKI.
Recent studies proved that even little elevation in sCr significantly increases mortality in patients with AKI. 18 Therefore, AKIN staging system was set on by modifying earlier risk injury failure loss end-stage kidney disease (RIFLE) scoring system. These new criteria allow to diagnose AKI in early periods by determining even little changes in sCr. 17 For more sensitive identification, we used AKIN criteria in diagnosis of AKI. In addition, none of the actual markers of kidney damage has enough sensitivity and specificity to predict AKI in early stage. 22 Therefore, there are accumulating studies to determine biomolecules that predict AKI in early stages.
Predictive value of KIM-1 U and KIM-1 S was studied in human especially in a selected population. In histochemical assessment, expression of KIM-1 in kidney tissue increased in the patients with acute tubular necrosis. 23 The authors also reported that KIM-1 U concentrations of the patients with acute and chronic renal disease were increased compared to controls. Renal transplantation is obviously the best in vivo model of acute renal injury. It has been reported that KIM-1 U predicts proximal tubular injury with a high sensitivity and specificity after renal transplantation. 24 KIM-1 S concentration on the first day after transplantation was found to be strong and independent predictor of acute rejection. 13 The authors also reported that KIM-1 U concentration predicted renal tubular regeneration better than other conventional markers. 25
To our knowledge, there is no report in the literature that studied whether KIM-1 U and KIM-1 S were predictors of CI-AKI. KIM-1 U concentrations were intended to increase as early as first day in patients diagnosed with AKI; however, increase in sCr concentrations and decrease in GFR occur on third day after cisplatin treatment. Our results confirming that KIM-1 U predicts renal injury earlier than sCr concentrations suggest the clinical and experimental studies in the literature.
The prediction of KIM-1 U concentrations in AKI has been evaluated differently in various studies. Some authors proportioned KIM-1 U concentrations to uCr to avoid changes of KIM-1 U concentrations according to urinary volume.26,27 Similarly, we evaluated these proportions to avoid the possible effects of standard hydration and variability in urine volume on our laboratory results. Decrease in KIM-1 U concentrations in AKI (−) patients on the first day after cisplatin treatment seems to be paradoxical. Besides an elevation, a decrease was not expected in patients without kidney injury in post-treatment compared to pre-treatment period. Standard oral/parenteral hydration may be the explanation of this paradox. Dilution of the urine caused by hydration may be responsible for relatively lower concentrations of KIM-1 U in AKI (−) patients. This is suggested by disappearance of a significant relation after normalization of AT1-KIM-1 U according to uCr in AKI (−) patients. However, this relation was still significant in AKI (+) patients after normalization of KIM-1 U according to uCr. Therefore, we suggest that both KIM-1 U and KIM-1 U /uCr ratio may be useful in early detection of CI-AKI with high sensitivity and specificity.
The prediction of AKI by KIM-1 poses a clinical advantage? The most important side effect of cisplatin, which is considered as an effective chemotherapeutic agent, is nephrotoxicity. 5 The lack of early treatment abilities results in dose reduction or cessation of the drug. 6 In addition, second- and third-line agents are not as effective as cisplatin. 4 The delay in the diagnosis of renal failure may face patients the complications of haemodialysis and cause an increase in mortality. 2 Therefore, early prediction of renal injury by KIM-1 may have potential clinical advantages.
AT5 sCr decreased to the concentrations of BT period as a consequence of renal recovery after CI-AKI. Similarly, AT5 KIM-1 U decreased to the concentrations of BT period and suggests that KIM-1 U should have additional benefits in determining renal recovery from CI-AKI. But it cannot be concluded with our results that whether decrease in KIM-1 U was an early predictor of recovery from CI-AKI.
There are two possible explanations for that we could not figure out the difference in KIM-1 S concentrations in BT and AT periods. First, urinary concentration of KIM-1 is more dominant than its serum concentration because it is a transmembrane protein in the apical surface of renal tubular cells (not basolateral surface).23,28,29 Second, because KIM-1 is a 90-kDa molecule, tubuli could not be able to reabsorb it after injury.
Study limitation
The major limitations of the present study are its unicentre design and rather small study population. Another limitation is that we could only study patients with AKI stage 1. Therefore, our results do not reflect the situation of patients with advanced AKI stages. And finally, our study does not contain long-term (>5 days) results of the patients on cisplatin treatment. So, multicentre studies with larger cohort observing AKI with advanced stages and long-term outcomes are needed.
Footnotes
Declaration of conflicting interests
None declared.
Funding
This research was funded by Scientific Research Supporting Fund of Abant Izzet Baysal University (Reference No: 2012.08.01.541).
Ethical approval
This work has been approved by local ethics committee of Abant Izzet Baysal University (Approval number: 2012/157).
Guarantor
BKT.
Contributorship
BKT, UU and HT designed the study and prepared study protocol. BKT obtained literature data. HT and UU selected the patients and performed clinical follow-up. UU and AA analysed the data. BKT, HT and GA prepared the first draft of the manuscript. All authors read and approved the final version of the study.
