Abstract
Background
The Dutch guideline algorithm for the analysis of anaemia in patients of general practitioners (GPs) was programmed in a Clinical Decision Support system (CDS-anaemia) to support the process of diagnosing the cause of anaemia in the laboratory. This study investigates the diagnostic yield of the automated anaemia algorithm compared to that of the manual work up by the GP.
Methods
This retrospective population-based study consisted of 2697 people ≥18 years. Anaemia was defined according to the Dutch College of General Practitioners (DCGP) guideline. Causes of anaemia were based on the DCGP guidelines with the corresponding blood tests. The number of blood tests and causes of anaemia were measured in two separate periods in both the (CDS-anaemia) pilot group and a control group in which routine care was provided.
Results
Patients from GPs supported by CDS-anaemia had higher chances of having more anaemia-related blood tests being performed. This resulted in finding significantly more causes of anaemia in the pilot group compared to the control group with respect to iron deficiency (resp. 31.3% vs 14.5%), possible iron deficiency (resp. 11.4% vs 2.8%), iron deficiency in acute phase (2.6% vs 0.5%), chronic disease/infection/inflammation (23.5% vs 1.9%), vitamin B12 deficiency (4.5% vs 1.9%), possible vitamin B12 deficiency (16.8% vs 8.7%), folate deficiency (3.3% vs 0.9%) and possible bone marrow disorder (3.8% vs 0.0%); p < 0.05.
Conclusions
This study suggests that an automated-algorithm support can effectively aid in the diagnostic work-up of anaemia in primary care to find more causes of anaemia.
Introduction
Anaemia is a highly prevalent disorder worldwide affecting 24.8% of the population. 1 Children (up to 40%), women (29% non-pregnant and 38% during pregnancy) and older individuals (ranging from 20 to 37%) are most often affected.1–4 Anaemia is not a diagnosis but often a presentation of an underlying condition.5,6 It is therefore relevant to diagnose the cause of anaemia to treat the patient as adequately as possible. Remarkably, this aspect has not been studied well, although some reports state that the cause of anaemia is often underdiagnosed.7,8
General practitioners (GPs) need laboratory tests alongside anamnesis and physical examination in order to infer accurate conclusions about the cause of anaemia. The Dutch College of General Practitioners (DCGP) in the Netherlands provides a guideline for GPs to support medical decision making for anaemia. 9 This guideline provides an algorithm which includes laboratory tests that need to be chosen under different circumstances. These tests consist of haemoglobin (Hb), mean corpuscular volume (MCV), ferritin, vitamin B12, folate, lactate dehydrogenase (LDH), leukocytes, thrombocytes, erythrocytes, reticulocytes, iron (Fe), transferrin, creatinine/estimated glomerular filtration rate (eGFR) and erythrocyte sedimentation rate (ESR). 9
Despite the existing guidelines, a study among 1342 anaemic patients from GPs has shown that in 86% of the patients one or more laboratory tests were missing for explaining the cause of anaemia. 7 We recently also targeted cause finding in anaemia by GPs. This study showed that a laboratory diagnosis is found only in 22,5% of the male patients aged 18–44, while a diagnosis is most often found in older females (49,1% of anaemic patients). 8 Since anaemia is much more prevalent in older individuals, finding anaemia in young males is especially abnormal. 3 It is important that laboratory tests are requested at the right time, that is, pursuing cause finding of anaemia only when anaemia is present. Otherwise overutilization of diagnostic blood tests might occur, but also underutilization in anaemic patients with the risk of incomplete or missing diagnosis and potentially leading to increased healthcare costs.8–11
General practitioners may therefore benefit from added guidance from laboratory diagnostics to reveal the cause of anaemia.10,12 To overcome missing causes of anaemia and supporting correct testing strategy, an automated algorithm was developed by Atalmedial in 2020 that supports the GP in finding the cause of anaemia according to the DCGP guidelines. The subsequent steps of the DCGP anaemia guideline were programmed in a Clinical Decision Support (CDS) software further called CDS-anaemia. This is an automated process, which is set in motion after blood samples are analysed for the presence of anaemia. Clinical decision support-anaemia automatically adds extra tests in a stepwise manner when anaemia is found. However, the effect of CDS-anaemia in supporting the GP with the diagnostic process and the cause finding of anaemia in primary care is unknown. Therefore, in the current study we investigated to which extent CDS-anaemia is effective in finding the possible cause(s) of anaemia in undiagnosed patients from GPs, compared to GPs who were not supported by CDS-anaemia (i.e. manual diagnostic work-up).
Methods
Study design
The data used for this analysis were obtained from the Atalmedial Medical Diagnostics laboratory request and results database. Data consisted of primary care patients, including sex, age and dates of measurement. Data also included the relevant blood parameters: Hb, erythrocytes, MCV, red cell distribution width (RDW), ferritin, iron, transferrin, vitamin B12, folate, LDH, reticulocytes, leukocytes, thrombocytes, creatinine, ESR and C-reactive protein (CRP). The GPs were located in the West of the Netherlands and included in total 53 GPs.
Pilot and control group
This study used two groups in order to establish the effectiveness of the intervention: a pilot group and a control group. The pilot group consisted of patients from GPs who were supported by Atalmedial Medical Diagnostics by providing automated-anaemia algorithm support when doing their blood diagnostics. The automated anaemia algorithm is performed by a software tool called AlinIQ (Abbott Diagnostics), hence referred to as CDS-anaemia. A distinction was made between data that originated from before the use and during the use of CDS-anaemia. Data derived from individuals during CDS-anaemia implementation, effective from 11–11–2020 till 1–3–2022 (second time period), was used to compare with the data from individuals pre-CDS-anaemia implementation from 11–11–2018 till 1–3–2020 (first time period). This resulted in a 15 months’ time period for both time frames. In addition, a control group was added, consisting of patients from GPs who did not receive CDS-anaemia assistance in their blood diagnostics, that is, did their work-up manually. This control group was added to control for the effects of the COVID-19 pandemic and the possible change in requesting behaviour by the GP over time. The same timeline was applied to each group to control for possible seasonal effects that could potentially influence results.
Patient characteristics
Four age groups were defined: 18–44, 45–64, 65–79 and >79 years. In order to measure effectiveness of the intervention in undiagnosed patients, it was assumed that only newly admitted patients within the Atalmedial system should be included. This was established by including patients in the time window as described above, who had Hb and indices (MCV, RDW and erythrocytes) measurements, but without an Hb measurement 6 months prior to the study Hb. Patients were excluded if they were <18 years. Since the manual follow up of anaemia by a GP according to the DCGP anaemia guideline will take time, we also included blood test results in the analysis that were requested within 6 weeks of the study Hb (second request).
Definitions and anaemia algorithm
Anaemia and the anaemia algorithm were defined according to the DCGP criteria.
9
ESR in the DCGP guideline was replaced by CRP, as the test specifications favour use of this parameter in the algorithm. The DCGP criteria were programmed in CDS. The CDS algorithm is implemented at the laboratory site. Clinical decision support-anaemia was only activated for patients from GPs that participated in the pilot group. Clinical decision support-anaemia became activated in this group when Hb and indices were requested. Subsequently, CDS-anaemia added ferritin tests in all patients with anaemia. Depending on the results of ferritin and MCV, RDW and number of erythrocytes, a conclusion was generated or subsequent tests were added. The CDS-anaemia workflow is depicted in Figure 1. The combination of tests added is based upon the DCGP guideline anaemia.
9
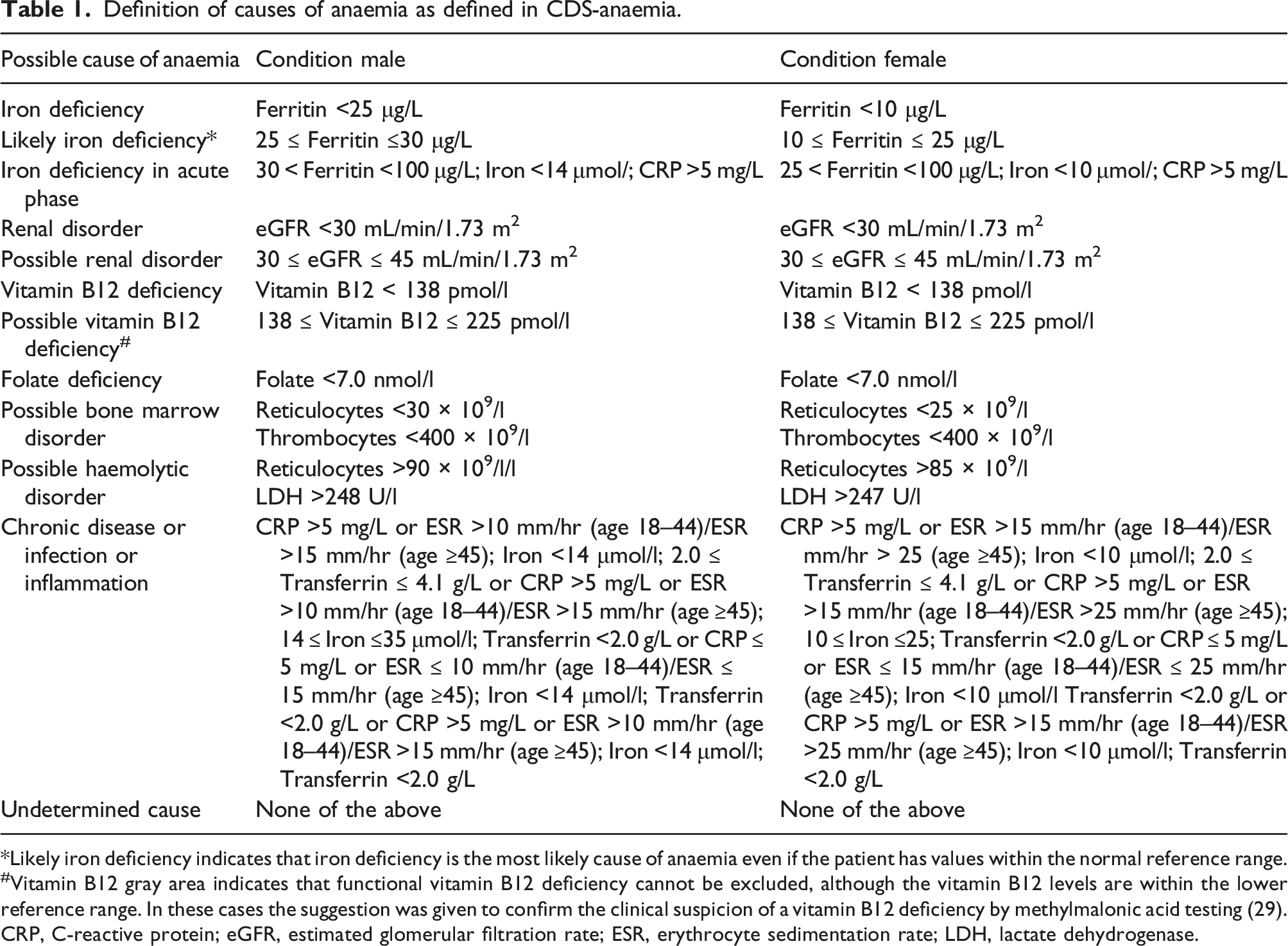
The results were approved by a specialist laboratory medicine, tests results and the conclusion were reported to the GP. The causes of anaemia were defined according to the conditions specified in Table 1. Flowchart clinical decision support-anaemia algorithm. Definition of causes of anaemia as defined in CDS-anaemia. *Likely iron deficiency indicates that iron deficiency is the most likely cause of anaemia even if the patient has values within the normal reference range. #Vitamin B12 gray area indicates that functional vitamin B12 deficiency cannot be excluded, although the vitamin B12 levels are within the lower reference range. In these cases the suggestion was given to confirm the clinical suspicion of a vitamin B12 deficiency by methylmalonic acid testing (29). CRP, C-reactive protein; eGFR, estimated glomerular filtration rate; ESR, erythrocyte sedimentation rate; LDH, lactate dehydrogenase.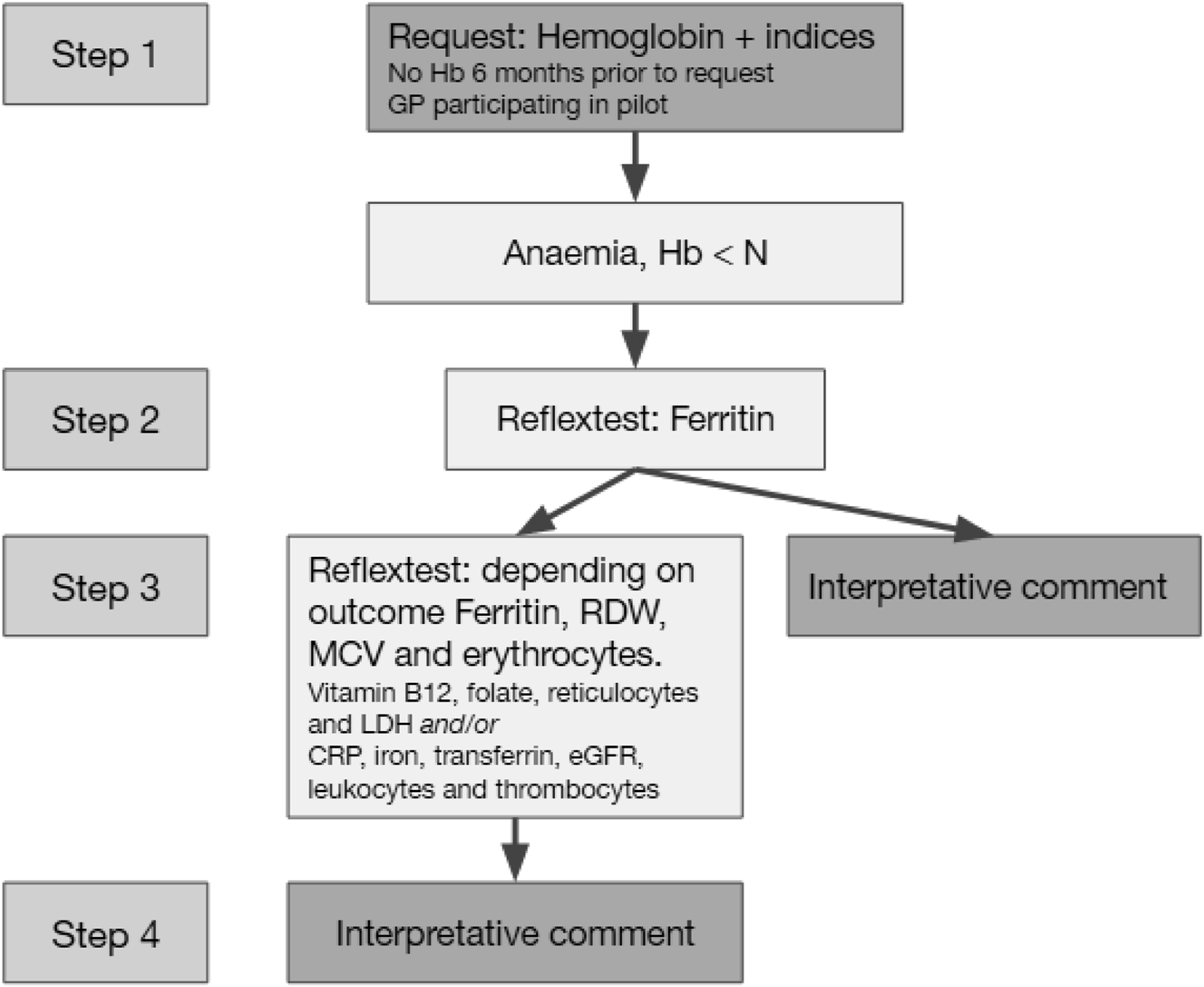
Statistical analysis
Differences in patient populations between the groups were compared using the Mann–Whitney U test (for the continuous variable Hb) and the Chi-squared test (for categorical variables). Differences in test frequencies and found causes of anaemia were compared using the Chi-squared test. Data were analysed using SPSS version 28. A p-value <0.05 was considered statistically significant.
Results
Study population
Patient characteristics, depending on study group.
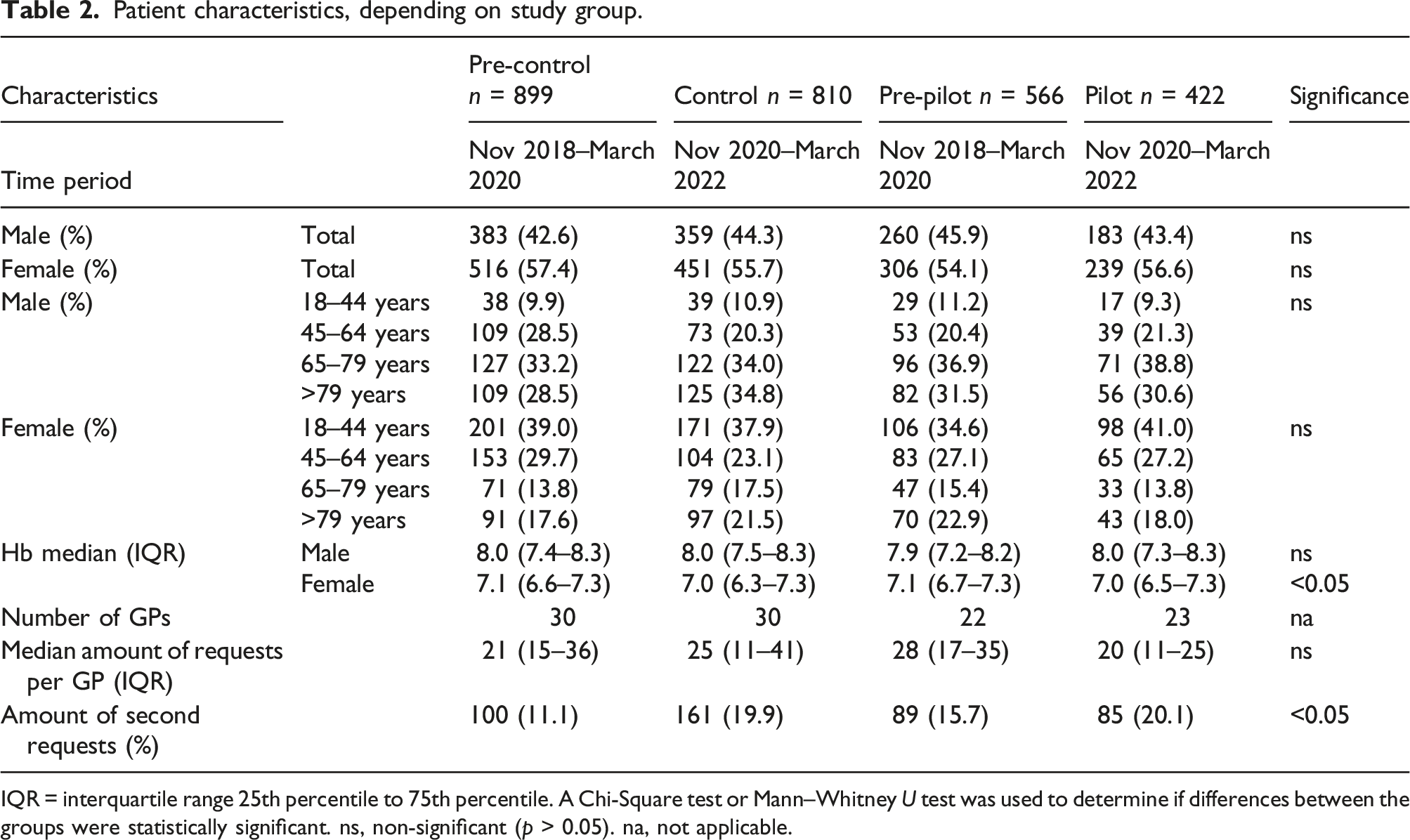
IQR = interquartile range 25th percentile to 75th percentile. A Chi-Square test or Mann–Whitney U test was used to determine if differences between the groups were statistically significant. ns, non-significant (p > 0.05). na, not applicable.
Test frequencies
Test frequencies of determined tests per group.
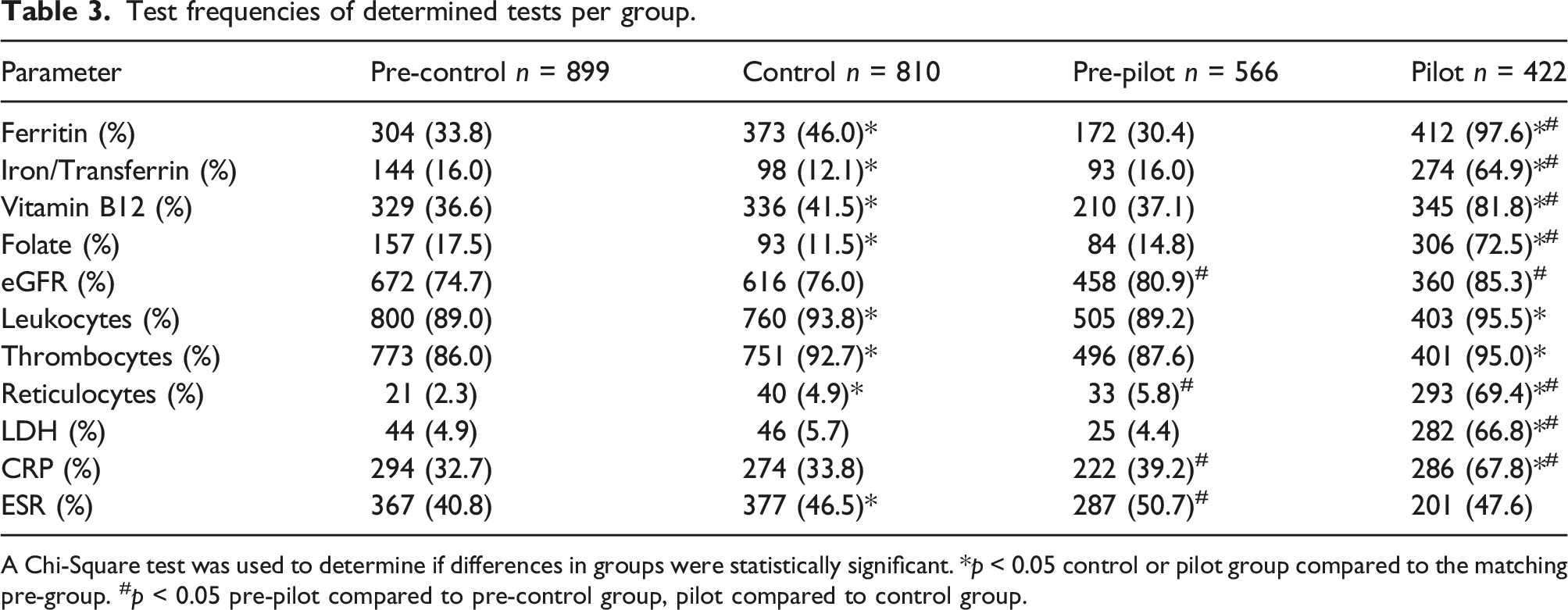
A Chi-Square test was used to determine if differences in groups were statistically significant. *p < 0.05 control or pilot group compared to the matching pre-group. #p < 0.05 pre-pilot compared to pre-control group, pilot compared to control group.
Causes of anaemia
Detected possible causes of anaemia per group.
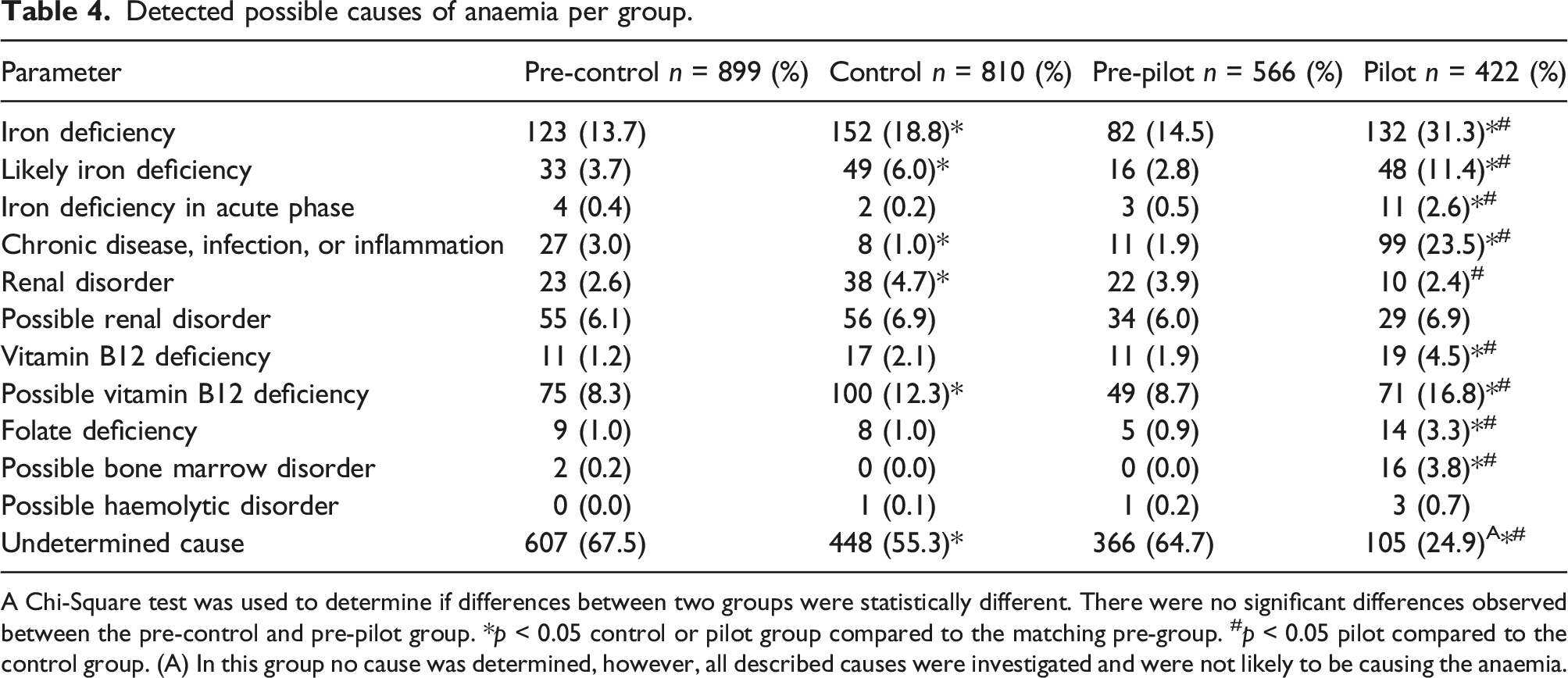
A Chi-Square test was used to determine if differences between two groups were statistically different. There were no significant differences observed between the pre-control and pre-pilot group. *p < 0.05 control or pilot group compared to the matching pre-group. #p < 0.05 pilot compared to the control group. (A) In this group no cause was determined, however, all described causes were investigated and were not likely to be causing the anaemia.
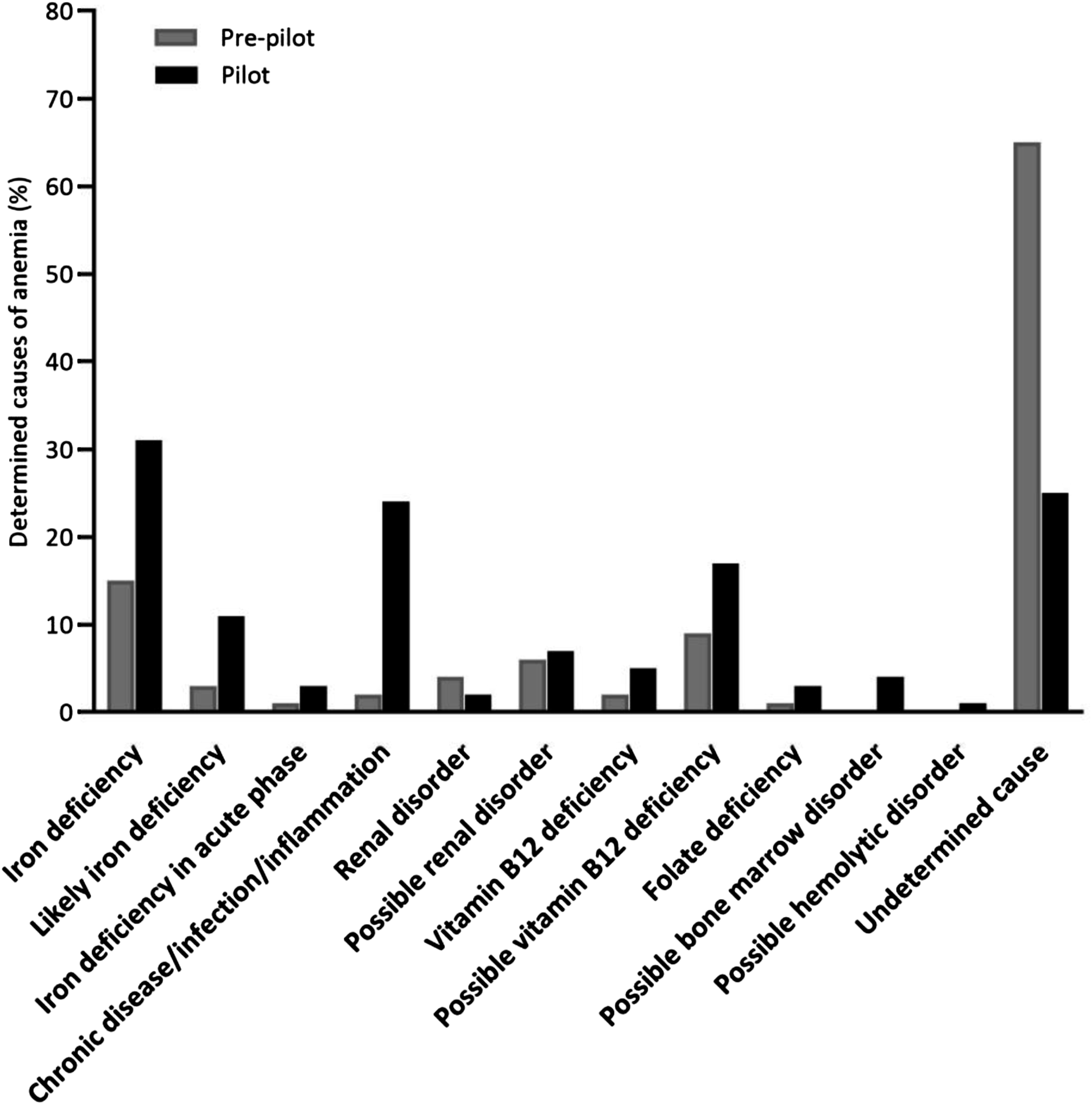
Detected possible causes of anaemia in the pre-pilot and pilot group. The numbers and significance are shown in Table 4.
Discussion
This study investigated the diagnostic yield of the cause of anaemia by comparing an automated algorithm performing guideline anaemia work-up versus manual anaemia work-up by GPs.
Although most countries, including the Netherlands, have guidelines for the work-up of anaemic patients from GPs, the actual implementation of guidelines is disputable and in general there seems to be a noticeable difference between actual guideline adherence and perceived guideline adherence. 13 For several reasons, overuse of medical services is a general phenomenon. 14 Indeed many GPs choose to add additional tests to the initial laboratory request when having Hb analysed (manuscript in preparation). However, the majority of patients do not have anaemia, therefore over-utilization of laboratory testing is common practice. On the other hand, when anaemia is present only a part of the patients is scrutinized into further detail.7,8 According to the guidelines of the Dutch Society for Clinical Chemistry and Laboratory Medicine (NVKC), medical diagnostic laboratories should offer a standardized protocol for anaemia work-up. 15 Atalmedial therefore implemented clinical decision support (CDS-anaemia) software to comply with the NVKC guidelines, while following the DCGP guideline.9,15 Since very little is published about the effectiveness of such automated diagnostic algorithms, we studied the effectiveness of the automated anaemia algorithm.
Most importantly, this study clearly showed that within patients of the pilot (CDS-anaemia) group much higher proportions of patients were diagnosed with an underlying cause of anaemia. On the other hand, the proportion of patients with an undetermined cause dropped from 67.5% in the pre-control group, to 24.9% in the pilot group. Importantly, in the pilot group in the group of patients in which no cause of anaemia could be found, nutrient deficiency, chronic disease, infection and renal failure as a cause of the observed anaemia were excluded by the added tests, whereas in the pre-pilot group these possible causes of anaemia were not consequently investigated. Many more patients were diagnosed with proven, likely or probable nutrient deficiency in the CDS-anaemia group as compared to the control group. This is a very relevant finding since in general nutrient deficiency can be reversed by targeted nutrient supplements or dietary advice, or can direct at somatic disease such as blood loss/gastrointestinal cancer, inflammatory bowel disease or pernicious anaemia. 16
The biggest difference in observed cause of anaemia was found for chronic disease or infection or inflammation (23.5% vs 1.9%). Since improvement of Hb is highly dependent on the type of disease, the laboratory report clearly pinpoints the probable cause and therefore supports the GP in the diagnosis. Despite the low incidence, CDS-anaemia did also aid in the detection of possible bone marrow disorders (3.8% vs 0.0%). Although the follow up of these patients was not part of this work, bone marrow disorders, especially myelodysplastic syndromes are more common in the elderly and therefore future research should address this finding into more detail. 17 Chronic kidney disease as a cause of anaemia was not found to be increased when using CDS-anaemia. This can easily be explained by the notion that kidney disease is highly correlated with age and GPs are used to measure creatinine to determine renal clearance in the elderly. 18 Therefore CDS-anaemia does not seem to aid in the detection of more kidney related anaemia.
To be able to increase the number of causes found, CDS-anaemia consequently adds laboratory tests, when Hb is below reference value. The most striking increase for nutrients deficiency was disclosed by ferritin testing. The proportions for testing frequencies went up from 30.3% pre-CDS-anaemia to 97.2% during implementation. This meant that CDS-anaemia was very effective at adding ferritin and pointing to iron deficiency as the most likely cause of anaemia, according to the anaemia guideline of the DCGP. Although the DCGP anaemia guideline does not include ferritin testing for women in their reproductive age, we found that many GPs particularly favoured ferritin testing for this group. 8 The small number without ferritin was caused by the lack of the necessary testing material. The clinical relevance of increased test frequencies for blood parameters is important to consider. Literature indicates that the ability of GPs to diagnose a specific cause for anaemia increases with more blood diagnostics.19,20 The use of an automated-anaemia algorithm supports this implication, so more causes of anaemia are found in the CDS-anaemia group. This may eventually lead to a more favourable outcome for the patient, since specific utilization of diagnostics can accelerate targeting the disease and shorten the time in anaemia or distress.
The unexplained anaemia results from our pre-control (67.5%), control (55.3%) and pre-pilot (64.7%) groups are similar to the results of Droogendijk et al. but exceed current literature that unexplained anaemia ranges around 28–30%.7,21,22 The simplest explanation is lack of follow-up testing in the anaemic individuals. 8 CDS-anaemia reduced this number of unexplained anaemia to 24.9%, in line with the findings of unexplained anaemia in the elderly.
The anaemia algorithm implemented was based upon the DCGP guideline anaemia. 9 However, some aspects of the implemented algorithm can be criticized and some improvements could potentially be made. Firstly, do we really need LDH with each vitamin B12 and folate measurement and in which cases do we need reticulocytes? Also the interpretation is quite complex for certain less common causes of anaemia such as haemolytic disease. Secondly, the guideline and algorithm use a one size fits all approach. We, however, know that in different patients groups different conditions cause anaemia.8,9 Therefore, an algorithm based upon the differential diagnosis per patient group would be more cost-effective. In addition, a step-by-step approach could be an option, although potentially some patients may have multiple causes of anaemia, which might be missed as the analysis stops after one cause of anaemia is found. Both subjects are an important subject of further research and could reduce the amount of unnecessary added tests. 23 Lastly, it is our experience that in a significant number of patients anaemia related tests are requested together with Hb. Since the majority of the Hb tests appear to be non-anemic, 24 the algorithm can be helpful by reducing unnecessary laboratory testing and ultimately improve diagnostics and reduce costs at the same time.
Future research should focus on what value CDS-anaemia really adds to anaemia care. Since it is unknown if GPs actually diagnose more causes of anaemia with CDS-anaemia assistance, future research should evaluate that topic. Even more importantly, it is also unknown whether or not patients receive more adequate treatment with CDS-anaemia assistance. This requires more intensive research, since treatment originating from the GP needs to be quantified. And lastly it is not yet investigated whether CDS-anaemia leads to a better clinical outcome of anaemic patients.
Another topic of investigation should include the financial impact blood tests have on diagnosing anaemia. Kip et al. showed by an online survey that extensive testing by GPs leads to increased cause finding of anaemia and consequently increase in costs. 18 Although the increase in costs was reported to be limited, it may be advantageous to investigate whether the (DCGP) anaemia algorithm can be tuned to optimize effective blood testing. Alternative approaches have been suggested by Oosterhuis et al. 23 but state of the art data mining and logistic solutions may help to proceed in this field.
Conclusion
The results of this study show that the cause of anaemia in anaemic patients of GPs can be determined in a larger proportion of patients when GPs are supported by an automated algorithm performing guideline anaemia testing than in anaemic patients of GPs who have to do the work-up manually.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethical approval
N/A.
Guarantor
BdB.
Contributorship
All authors reviewed and edited the manuscript and approved the final version of the manuscript.
