Abstract
Background
Establishing the underlying cause of anaemia in general practice is a diagnostic challenge. Currently, general practitioners individually determine which laboratory tests to request (routine work-up) in order to diagnose the underlying cause. However, an extensive work-up (consisting of 14 tests) increases the proportion of patients correctly diagnosed. This study investigates the cost-effectiveness of this extensive work-up.
Methods
A decision-analytic model was developed, incorporating all societal costs from the moment a patient presents to a general practitioner with symptoms suggestive of anaemia (aged ≥ 50 years), until the patient was (correctly) diagnosed and treated in primary care, or referred to (and diagnosed in) secondary care. Model inputs were derived from an online survey among general practitioners, expert estimates and published data. The primary outcome measure was expressed as incremental cost per additional patient diagnosed with the correct underlying cause of anaemia in either work-up.
Results
The probability of general practitioners diagnosing the correct underlying cause increased from 49.6% (95% CI: 44.8% to 54.5%) in the routine work-up to 56.0% (95% CI: 51.2% to 60.8%) in the extensive work-up (i.e. +6.4% [95% CI: −0.6% to 13.1%]). Costs are expected to increase slightly from €842/patient (95% CI: €704 to €994) to €845/patient (95% CI: €711 to €994), i.e. +€3/patient (95% CI: €−35 to €40) in the extensive work-up, indicating incremental costs of €43 per additional patient correctly diagnosed.
Conclusions
The extensive laboratory work-up is more effective for diagnosing the underlying cause of anaemia by general practitioners, at a minimal increase in costs. As accompanying benefits in terms of quality of life and reduced productivity losses could not be captured in this analysis, the extensive work-up is likely cost-effective.
Background
Anaemia is a common medical problem that carries substantial costs to the healthcare system and can be a burden on the health and quality of life of many individuals.1–10 Therefore, adequate diagnosis and early initiation of correct treatment are essential. 11 However, anaemia is not a disease in itself, but is considered a sign of an underlying condition. Consequently, diagnosing the underlying cause of anaemia is often complex.12–14 More specifically, previous research estimated that in 14–33% of older persons with anaemia, the underlying cause is unknown.1,12 In addition, anaemia is often under-diagnosed 15 and under-treated, as it is often considered a consequence of aging 12 and not as a specific symptom of disease.
The three most common underlying causes are: iron deficiency anaemia (IDA), anaemia of chronic disease (ACD) and renal anaemia. 16 Anaemia may also have a variety of other causes, including bone marrow diseases or vitamin deficiencies, such as B12 and/or folic acid. Besides anamnesis and physical examination, laboratory analyses are required to identify the different underlying causes of anaemia. However, depending on the laboratory protocol used, the proportion of patients for whom no underlying cause of anaemia can be identified based on their laboratory analyses ranges from 28% to 52%. 17 In order to enhance the effectiveness of laboratory analyses, a guideline has been developed by the Dutch College of General Practitioners (DCGP). 18 Previous research evaluated all laboratory tests of patients (women > 50 years and men ≥ 18 years) with anaemia newly diagnosed by general practitioners (GPs) within a two-year time period. Unfortunately, 83.9% of those patients could not be diagnosed when applying the DCGP-guideline, because at least one of the required laboratory tests had not been performed. 11
In a recently performed study, we investigated whether an extensive laboratory work-up (consisting of a set of 14 tests) could increase the probability that patients are diagnosed with the correct underlying cause of anaemia in Dutch general practice. 19 Although this study has shown that this work-up likely improves the probability that patients are correctly diagnosed with the underlying cause of anaemia, it is unknown whether this approach is cost-effective. In addition, even though most routine laboratory tests for diagnosing anaemia are relatively inexpensive, these test results will likely impact subsequent patient management decisions. As this may involve more expensive diagnostic testing and the referral of patients to secondary care, it is crucial to quantify the impact of such an extensive laboratory work-up further downstream the patient management pathway. Therefore, the current study aims to estimate the cost-effectiveness of this extensive laboratory work-up as compared with the current situation, the routine work-up, in which GPs decide for themselves which tests to request in patients presenting with symptoms of anaemia.
Methods
Survey
The effectiveness of diagnosing the underlying cause of anaemia in general practice, using either the extensive or a routine work-up, was investigated through an online survey using LimeSurvey. 20 A full description of this survey is provided elsewhere. 19 Details on how this survey was distributed are provided in Supplemental Data 1. In the survey, all participating GPs (139 out of 836, i.e. 16.6%) received six real-world cases of patients presenting with a new anaemia in general practice. In all six cases, the participating GPs were only provided with the age and gender of the patient and were informed that the patient was suspected of anaemia. In the first three cases, GPs were able to choose freely which tests they would request based on a predefined list of 14 common tests (haemoglobin, mean corpuscular volume [MCV], C-reactive protein [CRP] and/or erythrocyte sedimentation rate [ESR], vitamin B12, creatinine, ferritin, folic acid, lactate dehydrogenase [LDH], transferrin, reticulocytes, leukocytes, thrombocytes and serum iron) and were only given the results of the tests they selected. In the second set of three cases, GPs received the results of all 14 tests. In all six cases, the GPs were asked to choose between IDA, ACD, renal anaemia and ‘other’. In cases where the GPs chose ‘other’, they were asked to specify the underlying cause of anaemia. GPs could also state that the cause was ‘unknown’, if they did not consider it possible to determine the underlying cause based on the laboratory results provided. Besides, the GPs were also asked which of the subsequent actions they would take in each case: close the consultation (i.e. do nothing), refer the patient to secondary care, prescribe medication (which involved prescribing iron, vitamin B12, folic acid or antibiotics), or see the patient again in a few weeks (follow-up). During the survey, the GPs were encouraged to use guidelines (e.g. the DCGP guideline) or other tools they use in daily practice.
Database used in the survey
A detailed explanation regarding how the cases used in the survey were obtained from this database has been described previously. 19
Health economic model
As health economic models are typically complex, a description of the main aspects regarding model structure and model inputs is provided below. A more extensive description, including the assumptions used, is provided in Supplemental Data 1.
A decision tree was developed to estimate the cost-effectiveness of an extensive laboratory work-up compared with a routine work-up in diagnosing the underlying cause of anaemia in patients presenting with anaemia, aged ≥ 50 years, in Dutch general practice. A simplified version is shown in Figure 1. The correct underlying cause of anaemia was determined according to an expert panel, composed of an internist, a GP and a clinical chemist. Incremental effect was defined as the difference in the percentage of patients for whom the underlying cause of anaemia was correctly determined using a routine laboratory work-up (i.e. current practice) as compared with the extensive work-up. According to Dutch health economic guidelines, a societal perspective was taken in the cost-effectiveness analysis.
21
This means that all costs were included from the moment such a patient presents at the GP, until the patient is (correctly) diagnosed and treated in primary care, or referred to (and diagnosed in) secondary care. As the treatment of anaemia in secondary care strongly varies depending on its underlying cause, quantifying the impact of either work-up on patients’ quality of life would require extensive individual patient-level data, which were not available. As such, this was considered outside the scope of this analysis. The time horizon in this study was therefore estimated to be, at most, 200 days. Incremental cost was defined as the difference in average costs per patient for whom the underlying cause of anaemia was determined using the extensive work-up as compared with the routine work-up. Costs were expressed in 2016 Euros. The model outcome was expressed as an incremental cost-effectiveness ratio (ICER), representing the incremental costs per additional patient diagnosed with the correct underlying cause of anaemia. To obtain further insight as to testing for which underlying causes of anaemia (i.e. IDA, ACD, renal anaemia and ‘other’) potentially has the most room for improvement in terms of cost-effectiveness, subgroup analyses were performed.
Simplified decision tree demonstrating both laboratory work-ups (routine and extensive) of patients presenting with new anaemia in general practices.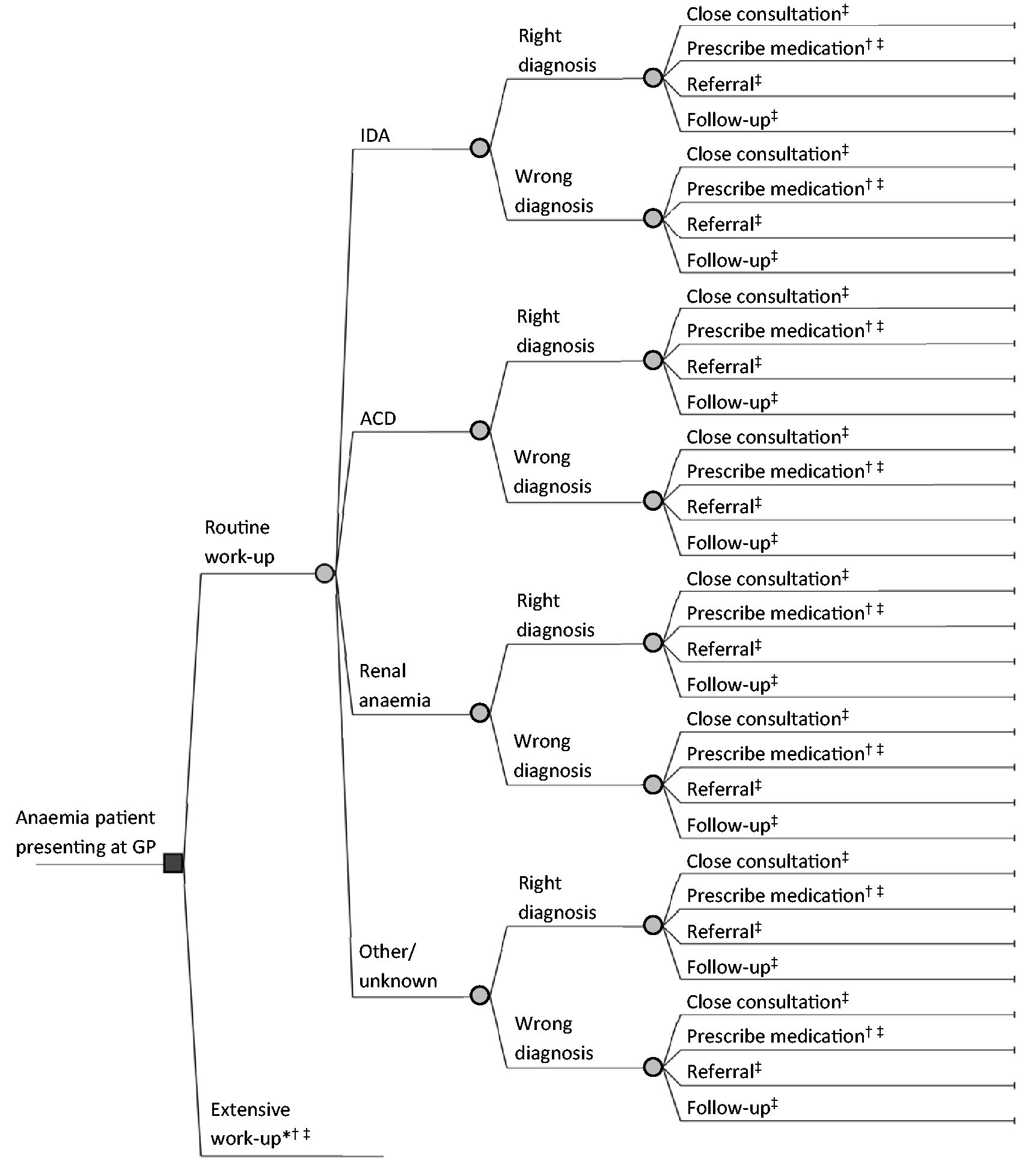
Model inputs
The incidence of the different underlying causes of anaemia in Dutch patients aged ≥ 50 years, was based on the abovementioned database. 16 The results of the survey were used to calculate the probability that the right underlying cause of anaemia (according to the expert panel) was established within each of those patient categories, for both work-ups. The likelihoods that GPs chose a certain type of treatment based on the different underlying causes of anaemia (i.e. prescribe medication (including the type of medication), refer the patient to secondary care, perform follow-up of the patient or to close the consultation without performing additional actions) were also derived from this survey. In cases where the GP decided to hold a follow-up appointment with the patient or to close the consultation, the probability of spontaneous recovery of a patient’s anaemia was taken into account for each of the different underlying causes. A detailed overview of the model structure, input parameters and the assumptions used is provided in Supplemental Data 1. Values for input parameters that could not be obtained from literature, such as the duration of oral iron supplementation, were derived from expert elicitations with two internist-haematologists (Supplemental Data 1).
Probabilistic sensitivity analyses
Random samples were simultaneously drawn for all input parameters based on predefined parameter distributions. Distributions were parameterized based on the observed parameter mean and on the observed or assumed standard error (Supplemental Data 1). To determine the effect of joint uncertainty in all input parameters on model outcomes, a probabilistic sensitivity analysis (PSA) was performed based on a Monte Carlo simulation with 10,000 samples.
One-way sensitivity analysis
To identify which individual parameters substantially influence model outcomes, a one-way deterministic sensitivity analysis was conducted. For each parameter, the impact on total costs per patient resulting from a change from the base case value to the lower and to the upper limit for the corresponding 95% confidence interval was analysed. A detailed overview of the inputs used in the one-way sensitivity analysis is provided in Supplemental Data 1.
Results
Overall cost-effectiveness
Detailed overview of costs and effects of both the routine and the extensive laboratory work-up, for the different underlying causes of anaemia.
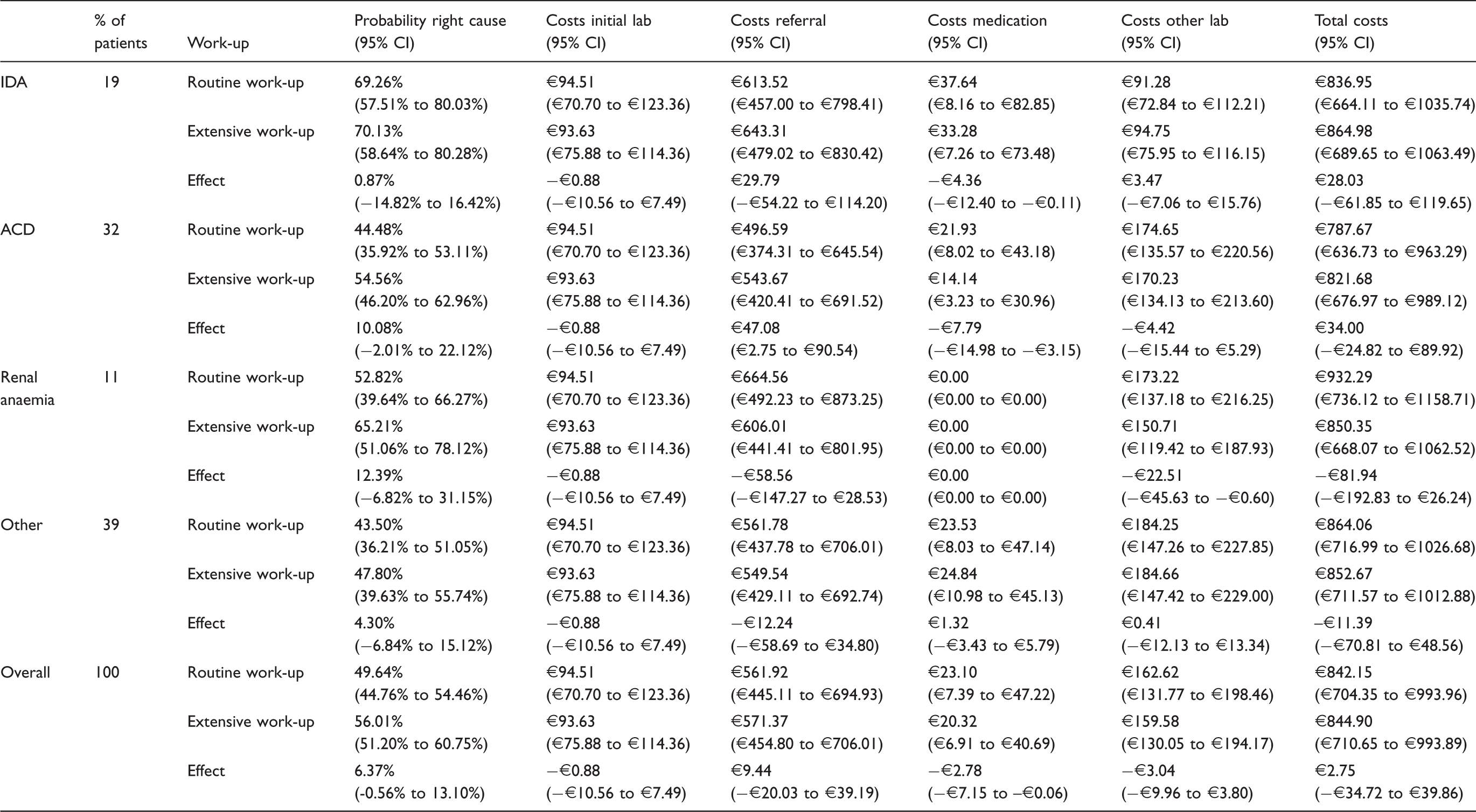
Note: In addition, costs of initial lab (at GP presentation, maximum of two times), costs of referrals, costs of medication and costs of other laboratory tests (performed during follow-up, to monitor medication or during referral) are presented. Percentages may not add up to exactly 100% due to rounding.
IDA: iron deficiency anaemia; ACD: anaemia of chronic disease.
Diagnosis-specific cost-effectiveness
The results of subgroup analyses indicated that average per patient costs were expected to increase by €28 (95% CI: €−62 to €120) among IDA patients and by €34 (95% CI: €−25 to €90) among ACD patients. In contrast, among renal anaemia patients and patients with ‘other’ underlying causes of anaemia, those costs are expected to decrease by €82 (95% CI: €−193 to €26) and by €11 (95% CI: €−71 to €49), respectively. The percentage of patients for whom the correct underlying cause is established is expected to increase by 0.9% (95% CI: −14.8% to 16.4%) among IDA patients, by 10.1% (95% CI: −2.0% to 22.1%) among ACD patients, by 12.4% (95% CI: −6.8% to 31.2%) among renal anaemia patients and by 4.3% (95% CI: −6.8% to 15.1%) among patients with ‘other’ underlying causes. Table 1 shows a detailed overview of all model outcomes, sorted by underlying cause of anaemia. The incremental cost-effectiveness plane demonstrating the overall result of 10,000 model simulations is shown in Figure 2. This figure indicates that 44.2% of the model simulations resulted in lower total costs of the extensive work-up as compared with the routine work-up. The incremental cost-effectiveness plane for the subgroups of anaemia patients (i.e. IDA, ACD, renal anaemia and ‘other’) is shown in Supplemental Data 1. The impact of separately inserting the inputs of the internist-haematologists on model outcomes as opposed to using their averaged estimates (as in the base case analysis) was considered negligible (Supplemental Data 1). In addition, 56.2% of the model simulations indicated lower costs in the extensive work-up (data not shown), when only considering costs of the initial GP consultation and accompanying phlebotomy and laboratory analyses (with a maximum of requesting laboratory tests twice in the routine work-up).
Incremental cost-effectiveness plane showing the impact of the extensive laboratory work-up as compared with the routine laboratory work-up on the difference in the percentage of correctly diagnosed underlying causes of anaemia, as well as the difference in costs per patient, for 10,000 model simulations.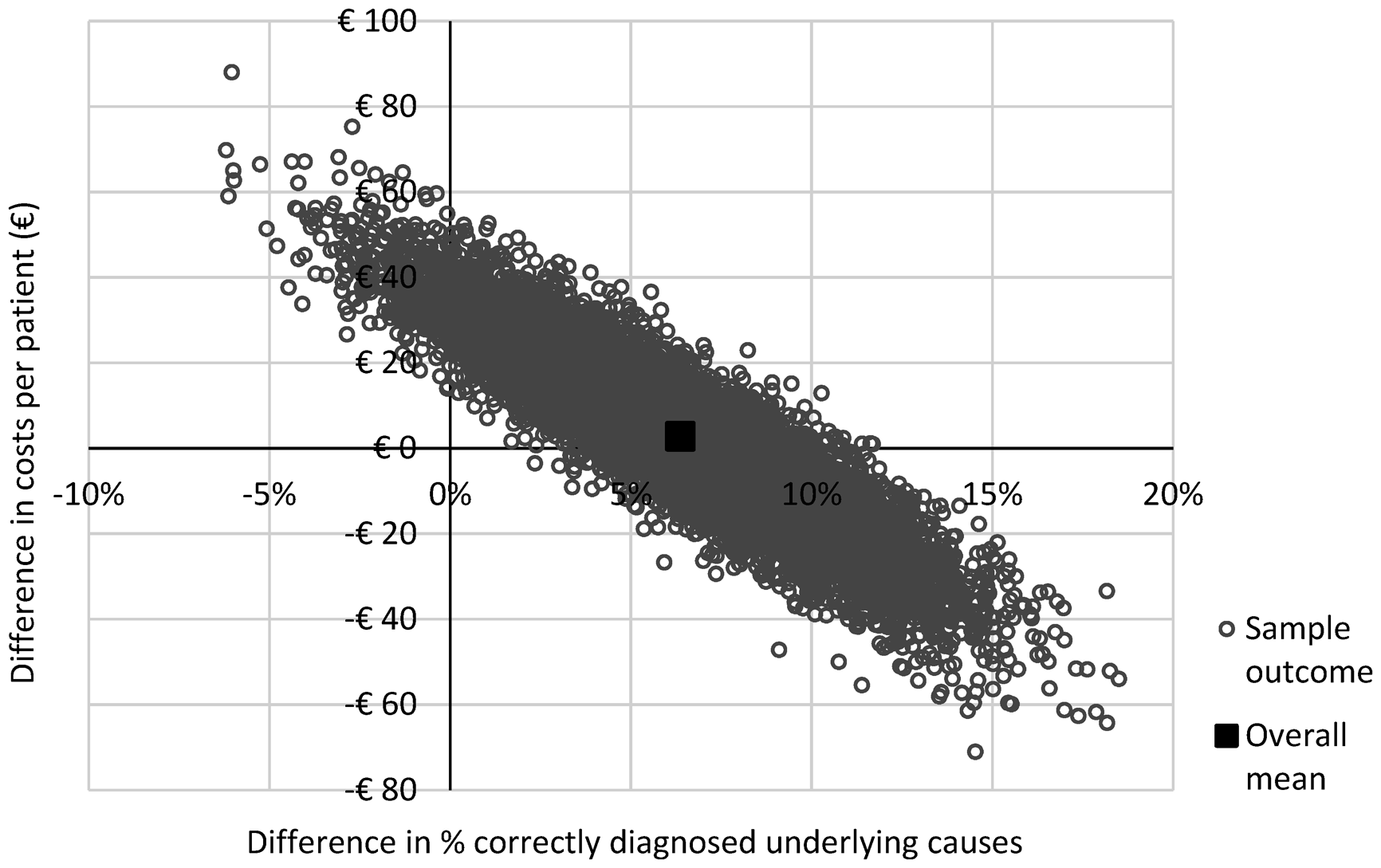
One-way sensitivity analysis
In Figure 3, the results of one-way sensitivity analyses are shown for the 10 parameters with the highest impact on the difference in costs. The difference in costs between both strategies was found to be most sensitive to changes in the frequency with which GPs correctly diagnose the underlying cause of anaemia, in both the extensive and the routine work-ups. The costs per GP consultation and the probability of spontaneous recovery of ACD have the highest impact on the difference in costs between both work-ups.
Tornado diagram showing the impact of changes in the most relevant input parameters on the difference in costs.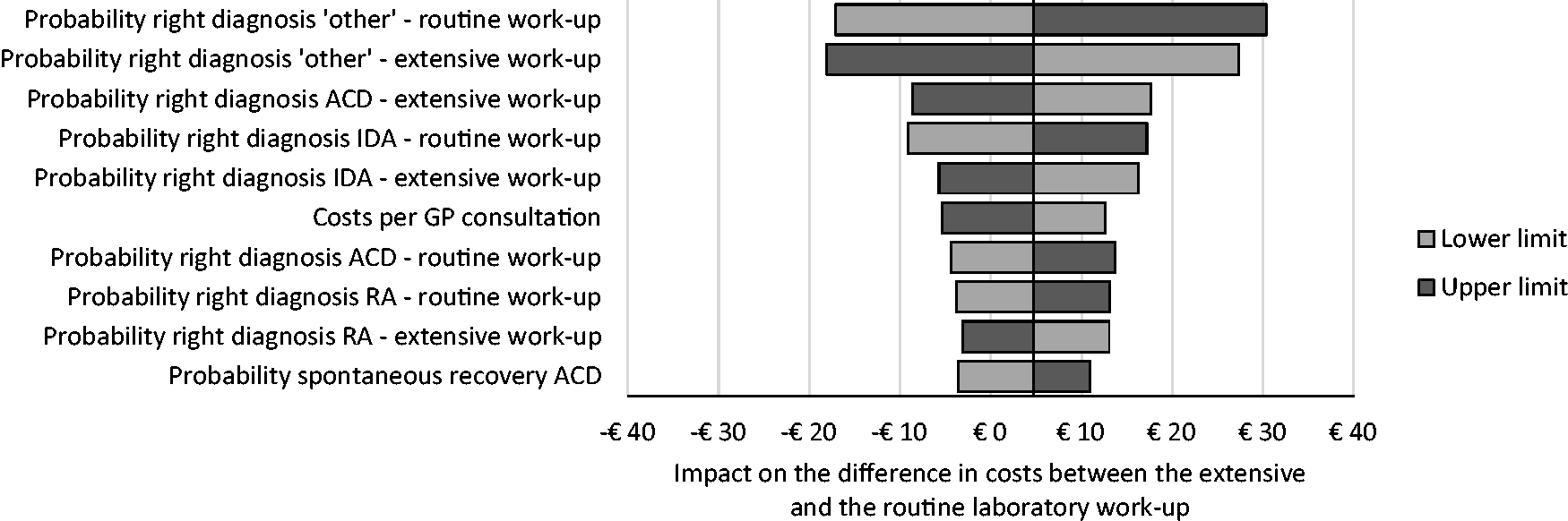
Conclusion and discussion
The use of an extensive laboratory work-up likely increases the percentage of patients diagnosed with the correct underlying cause of anaemia as compared with the routine work-up. Simultaneously, a very minor increase in costs of ∼€3/patient (+0.3%) was expected. Whereas the improvement in diagnosis was quite likely (chance of an increase in correct diagnosis equals 6.4%), the exact effect of this extensive work-up on the changes in costs was quite uncertain (chance that the extensive work-up would actually save costs equals 44.2%). Although higher costs were expected among IDA and ACD patients, most cost-savings were expected to be achieved among patients with renal and ‘other’ types of anaemia.
Despite the probable increase of 6.4% in correct diagnoses, the results varied considerably between the different underlying causes of anaemia. The largest improvements in this probability were expected in ACD and renal anaemia patients (i.e. +10.1% and +12.4%, respectively). However, costs of referrals were expected to increase by €47/patient among ACD patients, whereas those costs decreased by €59/patient among renal anaemia patients. The increase in costs among ACD patients was explained by the higher immediate referral rate to secondary care (i.e. referral following the initial GP consultation) after the extensive work-up rather than after the routine work-up (38% vs. 26%), while the probability of follow-up was lower (48% vs. 60%). As the probability of spontaneous recovery among ACD patients was estimated to be relatively high (i.e. 38%), higher referral rates lead to higher costs in the extensive work-up in this patient category. Combined with the relatively high frequency of ACD (32% of all newly diagnosed anaemia patients in general practice), this may strongly impact the overall costs. Therefore, effort should be spent on deciding which patient management strategy (i.e. referral or follow-up) results in most improvements in the patient’s quality of life, at the lowest costs.
For renal anaemia patients, the results indicate that 42% and 48% of those patients were immediately referred to secondary care in the routine versus the extensive work-up, which was considered the only appropriate management decision according to the expert panel (Supplemental Data 1). Thus, besides expected improvements in diagnosing renal anaemia due to the extensive work-up, this work-up also increased the number of patients who would immediately receive an effective management strategy, which likely decreases costs further downstream the care pathway.
In IDA patients, the probability of a correct diagnosis remained almost unchanged in the extensive as compared with the routine work-up (i.e. +0.9%), while costs increased with €28/patient. When considering the management decisions made in those patients, it was found that this increase in costs was mostly attributable to an expected 6% increase in immediate referrals (43% vs. 37%). However, the DCGP guideline recommends a colonoscopy and/or gastroscopy in IDA patients aged > 50 years, to exclude a gastrointestinal malignancy. 24 As such malignancies can be detected in 6–15% of IDA patients,25–30 this increase in immediate referrals may increase the probability that a gastrointestinal malignancy is diagnosed. Thereby, this increase in costs will likely enhance rapid initiation of adequate treatment, potentially improving treatment effectiveness. However, as such a referral decision should be based on a patient’s clinical signs and symptoms (which were unknown in the current analysis), the real-life impact of this extensive work-up on the diagnosis and treatment of gastrointestinal malignancies remains to be investigated.
Although the previously performed effectiveness analysis distinguished only four underlying causes of anaemia (IDA, ACD, renal anaemia and ‘other’), 19 the current study further divided the ‘other’ category into suspected bone marrow disease, vitamin B12 or folic acid deficiency and unknown causes. Subsequently, only the remaining patients were categorized as ‘other’. Although those additional subgroups were too small for any demonstrable results, this subdivision allowed for a more precise calculation on the costs of these diagnoses. However, it is reasonable to assume that GPs are unable to diagnose these less common causes of anaemia based solely on laboratory test results: they often require further diagnostic testing or referral to secondary care to determine these causes. Therefore, in order to allow an accurate cost estimation, the approach taken to analyse the effectiveness in the current study differed slightly from the approach taken in the previously published article. Consequently, the results from the current study indicate a slightly lower percentage of correctly diagnosed underlying causes when compared with the previous study. 19
Strengths
The results are expected to provide a good representation of the Dutch population aged ≥ 50 years with newly diagnosed anaemia in primary care, because the cases in the survey were based on real-life patient data, the incidence of the various causes of anaemia in the survey correspond with their occurrence in daily practice, 19 and because the participating GPs were representative of the GP population in the Netherlands. 19
As 96.4% of the 10,000 model simulations indicated that the extensive laboratory work-up would increase the percentage of patients correctly diagnosed, this result was robust to uncertainty in input parameters. Of those simulations, 44.2% indicated lower total costs with the use of an extensive work-up, although there was an average increase of ∼€3 per patient, overall, indicating that the exact impact on costs is likely very limited but remains uncertain. However, the average number of tests performed in the extensive work-up doubled those performed in the routine work-up (14 vs. 7). That the increase in costs remains so small can easily be explained as the costs of additional diagnostic tests are offset by performing all tests during one GP visit and one phlebotomy, thereby preventing repeated blood sampling (involving additional costs of GP visits, the order tariff for requesting laboratory tests, and lost productivity among patients). This is also confirmed by the results, as 55.4% of 10,000 model simulations indicated that the costs of diagnostic testing at the GP were actually lower in the extensive work-up.
Limitations
This study has certain limitations. First, as described previously, 19 the GPs were not provided with the patient’s anamnesis, medical history and physical examination. It is therefore likely that the accuracy of the diagnoses of the responding GPs may be higher in real-life. Although this limitation was present in all cases within the study, their potential effect on the differences between the two analysis methods is most likely limited, although it cannot be excluded that the abovementioned patient characteristics may have affected the tests that would have been requested by the GPs. Furthermore, it is uncertain to what extent GPs’ diagnostic and treatment decisions in the survey may differ from real life.
Secondly, as mentioned previously, the costs of treating anaemia in secondary care have not been included in the model because of large differences in treatments for the different underlying causes and the lack of patient-level data. However, a delayed correct diagnosis likely delays the initiation of adequate treatment, negatively affects quality of life 3 and potentially increases treatment costs owing to an increased severity of anaemia. Therefore, it was conservatively assumed that costs after establishing the correct diagnosis will not differ between patients. Consequently, current results are likely an underestimation of the potential additional benefits provided by the extensive laboratory work-up.
Third, as information for some model input parameters could not be obtained from literature, expert estimates had to be used. Although the number of experts was limited (n = 2), the results of probabilistic (Supplemental Data 1) and one-way sensitivity analyses indicated that the impact of changes in model parameters (as based on those expert elicitations) on model outcomes was limited.
Fourth, in the Netherlands, 64% of the clinical chemistry laboratories offer reflex testing. 31 In reflex testing, GPs do not decide themselves which laboratory tests to perform in suspected anaemia patients, but instead request ‘anaemia analysis’. The laboratory will then perform a predefined set of tests, and sequentially perform additional tests if the initial tests indicate the presence of anaemia. 31 In addition, 27% of these laboratories provide an interpretative comment along with the test result. 31 As this reflex testing could not be incorporated in the current study, the proportion of patients correctly diagnosed in current clinical practice (reflected by the routine work-up) may be underestimated. However, the set of tests performed in reflex testing differs strongly between laboratories 31 and often involves fewer diagnostic tests compared with the extensive work-up presented in the current study. In addition, although the DCGP -guidelines recommend reflex testing in patients with newly diagnosed anaemia, 24 it is not yet offered by all laboratories and can therefore not yet be considered common practice among GPs. Thus, although the added benefit of the extensive work-up may be slightly overestimated in the current analysis, it likely still increases the number of patients for whom the underlying cause is correctly diagnosed by the GP. Furthermore, the insights obtained from this study are likely of added value to decide upon the optimal combination of tests to be used for reflex testing, which may increase similarity in diagnostic work-ups between laboratories.
Implications for practice
As subgroup analyses revealed that the added value of the extensive laboratory work-up depends on the underlying cause of anaemia, further research into this variation is recommended. Which tests contribute most to establishing the correct underlying cause should be investigated, in order to select a laboratory work-up that can correctly diagnose the majority of patients with minimal inconvenience (i.e. multiple venipunctures) and at minimal costs.
Transferability of these results to other countries is not straightforward, as the work-up regarding the diagnosis of anaemia patients and the attributed costs may vary greatly between countries. Populating the model with country-specific data would support reliable country-specific estimations of the cost-effectiveness of both work-ups.
In conclusion, although the extensive laboratory work-up is usually more effective for diagnosing the underlying cause of anaemia by GPs, it is not always more cost-effective than the routine work-up. Nevertheless, the impact on costs was found to be minimal. Given that the extensive work-up results in additional benefits which could not be captured in the current analysis (i.e. in terms of a faster diagnosis which may improve a patient’s quality of life, and reduced productivity losses among anaemia patients and their caregivers), using an extensive laboratory work-up is expected to be cost-effective.
Footnotes
Acknowledgments
We thank all of the GPs for their participation in the survey, and the internist-haematologist for participating in the expert elicitation. In addition, we thank Dr. Rebecca Buis for critical reading of the manuscript.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethical approval
The database used in the analysis was approved by the internal ethics committee of the Albert Schweitzer Hospital.
Guarantor
RK.
Contributorship
MMAK and AS contributed equally to this article. MMAK, AS, KS, SD, MDL and RK were involved in the conception and design of the study and performed the data collection. MMAK and HK designed the health economic model and analysed the data. All authors were involved in interpreting the data. MMAK drafted the manuscript, and all author authors were major contributors in critically reviewing the manuscript. All authors read and approved the final manuscript.
Supplementary materials
All input parameters used in the health economic model have been provided in the online supplementary material.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
