Abstract
Background
The use of supine reference intervals instead of the corresponding seated reference intervals for seated plasma-free metanephrines (MNs) in pheochromocytoma/paraganglioma (PPGL) screening has been controversial in recent years. Each clinical laboratory should choose the optimal sampling posture and diagnostic strategy according to local conditions.
Methods
The reference population consisted of 736 cases aged 14–92 years old and the validation population consisted of 1068 patients aged 8–87 years old. Seated MNs were measured by liquid chromatography-tandem mass spectrometry (LC-MS/MS), and the reference intervals and diagnostic cut-off values were established and the diagnostic performance compared with reference intervals established in a supine position.
Results
There was no correlation between seated plasma MNs and age (p > 0.05) and there were differences in MNs among the various disease groups (p < 0.05). MNs were different in gender (p < 0.0001). The upper reference limit (URL) established in this study had the same sensitivity (100%) and better specificity (94.6% vs 83.5%) compared with the published age-adjusted supine reference intervals. The proportion of suspected patients with MNs within the URL-2×URL range was lower using seated reference intervals compared to supine intervals (5.3% vs 15.7%).
Conclusion
Using the corresponding seated reference intervals for seated plasma MNs can reduce the unnecessary re-examinations of suspected patients with slightly elevated MNs. The cut-off value established by seated plasma MNs has good diagnostic performance in PPGL. Use of seated sampling is an acceptable practice and is more convenient and economical than supine sampling.
Keywords
Introduction
Pheochromocytoma/paraganglioma (PPGL) is a neuroendocrine tumour that originates from the adrenal medulla or extra-adrenal sympathetic chain and has a hormone secretion function. A series of clinical syndromes such as metabolic changes have caused serious complications of the heart, brain, kidney, blood vessels, and other organs, and even become the main cause of death in patients.1,2 Therefore, early diagnosis is of great significance for the early detection of lesions as well as timely and adequate preoperative and intraoperative preparations to reduce the risk of surgical accidents. Compared with catecholamines, its metabolites metanephrine (MN) and normetanephrine (NMN), collectively defined as metanephrines (MNs), have the characteristics of long half-life with good stability and specificity and have become important biomarkers in the diagnosis of PPGL.3,4 Liquid chromatography-tandem mass spectrometry (LC-MS/MS) has become the preferred method for the measurement of MNs in the PPGL diagnostic guidelines due to its advantages of high selectivity, high specificity, and high sensitivity.5,6
Since the upright position can further activate the sympathetic nervous system, 7 the supine position sampling of plasma MNs has a lower false-positive rate than the seated position. 8 Therefore, sampling blood after 30 min of rest in the supine position is recommended by many scholars.9,10 However, in practical clinical settings, plasma MNs sampling in a seated position is more convenient and economical than sampling in a supine position or collecting samples for 24-h urine MNs and has high patient acceptability. So far, it is still the most widely used screening method position for PPGL in many clinical laboratories, including our laboratory. Therefore, in recent years, the choice of MNs sampling position has been controversial.11–13 Pheochromocytoma is a dangerous rare disease, and it is very important to ensure the high sensitivity of the screening test. Some research data show that9,14 the use of the supine reference intervals under the condition of seated sampling has higher sensitivity than the use of the seated reference intervals, so it is recommended that when using seated sampling, the age-adjusted supine reference intervals should be used instead of the corresponding seated reference intervals. However, some other studies have shown15–17 that seated sampling provides sufficient diagnostic performance, especially a high level of diagnostic sensitivity, and it is feasible to use the seated reference intervals for the seated population.
Our laboratory established an MNs assay using LC-MS/MS at an early stage. In view of the recommendations of previous reports and local clinical practice, seated sampling for MNs interpreted according to supine reference intervals is used in screening for PPGL. However, a considerable number of false-positive samples are found in current routine work, which causes confusion and inconvenience to patients and clinicians. Therefore, this study selected a Chinese population who visited our hospital to establish the reference intervals of seated plasma MNs and compared the diagnostic performance with the use of supine reference intervals 18 to explore the above problems further.
Methods
Subjects
Baseline characteristics of the reference and validation population.
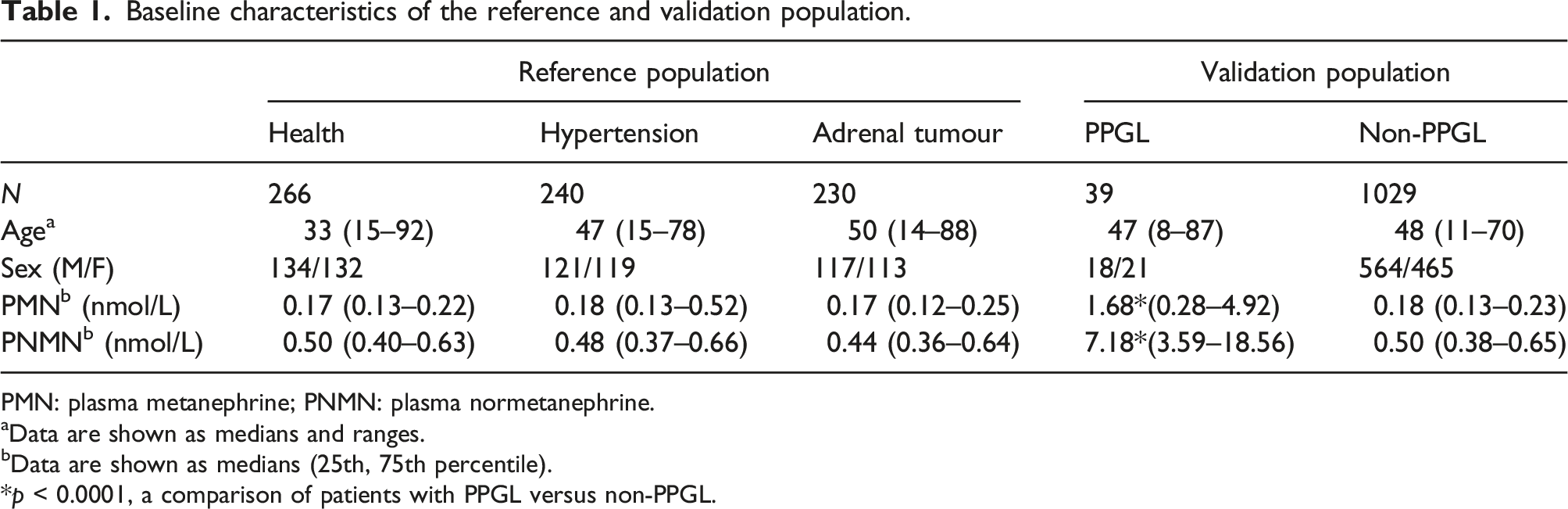
PMN: plasma metanephrine; PNMN: plasma normetanephrine.
aData are shown as medians and ranges.
bData are shown as medians (25th, 75th percentile).
*p < 0.0001, a comparison of patients with PPGL versus non-PPGL.
Collection of samples
All participants should not take any drugs that affect the metabolism of catecholamines within 48 h before blood collection, and injection of epinephrine, norepinephrine, and dopamine is also prohibited. Avoid smoking, caffeine, alcoholic beverages, or food containing tyramine for at least 8 h before blood collection. Blood samples were collected after sitting for at least 30 min. EDTA-K2 anticoagulant vacuum blood collection tubes were used to collect the venous blood. Samples were delivered to the laboratory within 2 h, centrifuged at 4°C 3000 g for 5 min, and tested as soon as possible. Plasma aliquots were stored at −80°C and tested within 1 week if they couldn’t be tested immediately.
Laboratory tests
MNs were measured by LC-MS/MS, which consisted of Shimadzu LC-20AD (Shimadzu Corporation, Japan) high-performance liquid chromatograph and 4500 MD mass spectrometer (AB SCIEX Corporation, USA). Calibrants were MN hydrochloride and NMN hydrochloride, and internal standards were MN-d3 hydrochloride and NMN-d3 hydrochloride (Toronto Research Chemicals, CAN). Quality controls (Qlife Corporation, CHN). Methanol and acetonitrile (Fisher Scientific, USA). The samples were pre-treated by solid-phase extraction (SPE), and the extraction column was equilibrated with 200 μL of pure methanol and ultrapure water in turn, and 400 μL of the mixture was loaded (200 μL of the plasma was added with 100 μL of ultrapure water and 100 μL of internal standard composition mixture). Each well was rinsed with 200 μL ultrapure water and acetonitrile and isopropanol mixture in turn. The eluate (100 μL 1% ascorbic acid methanol solution) was collected and evaporated to near dryness with nitrogen. 50 μL containing 0.1% ascorbic acid ultrapure water was added into each well and samples were then analysed by the LC-MS/MS. Chromatographic conditions: Agilent Technologies Pursuit 3 PFP (150 mm × 2.0 mm, 3 μm), column temperature 40°C, flow rate 0.4 mL/min. Mobile phase A was 0.1% formic acid aqueous solution, mobile phase B was 0.1% formic acid acetonitrile solution, and gradient elution was performed. Mass spectrometry conditions: an electrospray ion source, positive ion scanning mode, spray voltage 5.5 kV, temperature 500°C, curtain gas 20 lbf/in2, spray gas 50 lbf/in2, auxiliary heating gas 60 lbf/in2, multiple reaction monitoring modes (MRM). The lower limit of quantitation was 0.05 nmol/L for both. The linear range was 0.05–10.00 nmol/L and the reportable range was 0.05–200.00 nmol/L for both. The intra-assay and inter-assay precision were within 5%. The recovery effects of MN and NMN were 91.0–103.6% and 98.7–104.5% with the CV<20%. To ensure the accuracy of the method results, we regularly participated in the National External Quality Assurance Scheme organized by the National Health Commission of the PRC and passed with full marks.
Data analyses
LC-MS/MS data were analysed by Analyst 1.6.3 (AB SCIEX, USA), SPSS 23.0 (IBM, USA) and GraphPad Prism 9.0 (LaJolla, USA) were used to analyse and graph the data. Since plasma MNs were not normally distributed, the results of each group analysis and the reference intervals were presented as medians and percentiles (2.5 and 97.5). Spearman’s test was used for correlation analysis. The non-normal distribution data were compared between groups using Mann–Whitney U test. Enumeration data were expressed as %, and the Fisher’s exact test and chi-square test were used for comparison between groups. Logistic regression and receiver-operating characteristic (ROC) curve were used to analyse the diagnostic performance of combination mode and evaluate the cut-off values for the best sensitivity and specificity. Differences were considered statistically significant at p < 0.05.
Results
The influence of age, disease and gender
In the reference population, there was no correlation between plasma MN and age (r = 0.040, p = 0.274) or plasma NMN and age (r = 0.042, p = 0.260).
There was a significant difference in plasma NMN between the healthy group and the adrenal tumour group in the reference population (Z = −2.345, p = 0.019). Plasma MN differed between the healthy group and hypertensive group in the reference population (Z = −2.052, p = 0.040), but not among the remaining groups (Figure 1(a)). Boxes plots showing the median concentrations and 1.5 interquartile ranges of plasma MNs in the seated position for subjects of the reference population. (a) Comparison of plasma MNs in seated position between different groups. (b) Comparison of plasma MNs in seated position between males and females. *p < 0.05, ***p < 0.0001.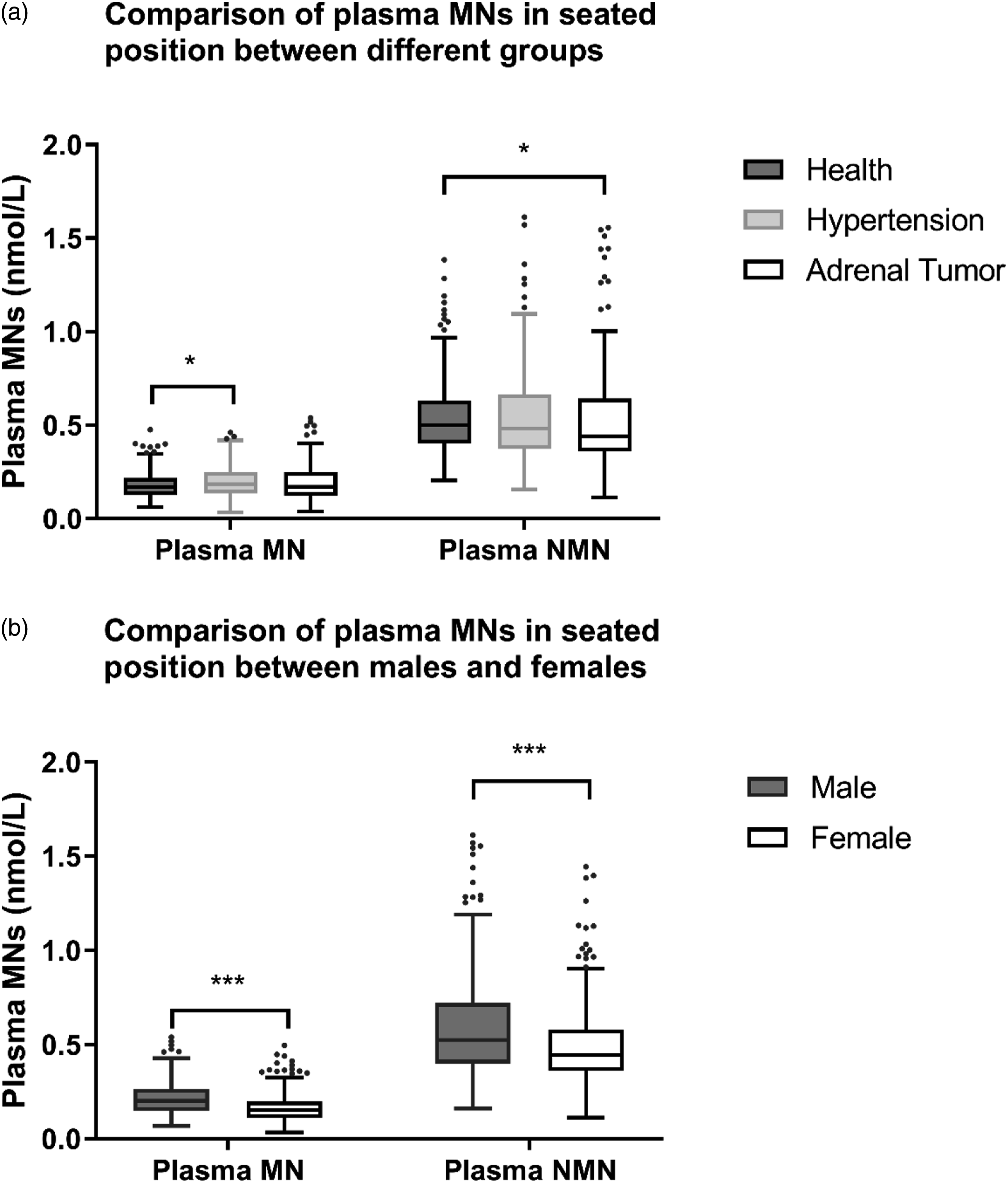
Plasma MN and NMN were different in gender (Z = −4.500, p < 0.0001, Z = −8.285, p < 0.0001). The reference population had comparable median ages for men and women (41.0y vs. 44.5y), whereas men had higher medians for NMN (0.52 nmol/L vs. 0.44 nmol/L) and MN (0.20 nmol/L vs. 0.15 nmol/L) than women (Figure 1(b)).
Establishment and evaluation of reference intervals and diagnostic cut-off values
The reference population was non-normally distributed, and the reference intervals for seated plasma MN and NMN were established at the 2.5 and 97.5 percentiles: MN 0.07–0.40 nmol/L, NMN 0.22–1.17 nmol/L.
Diagnostic performance of MNs for detection of pheochromocytoma/paraganglioma.
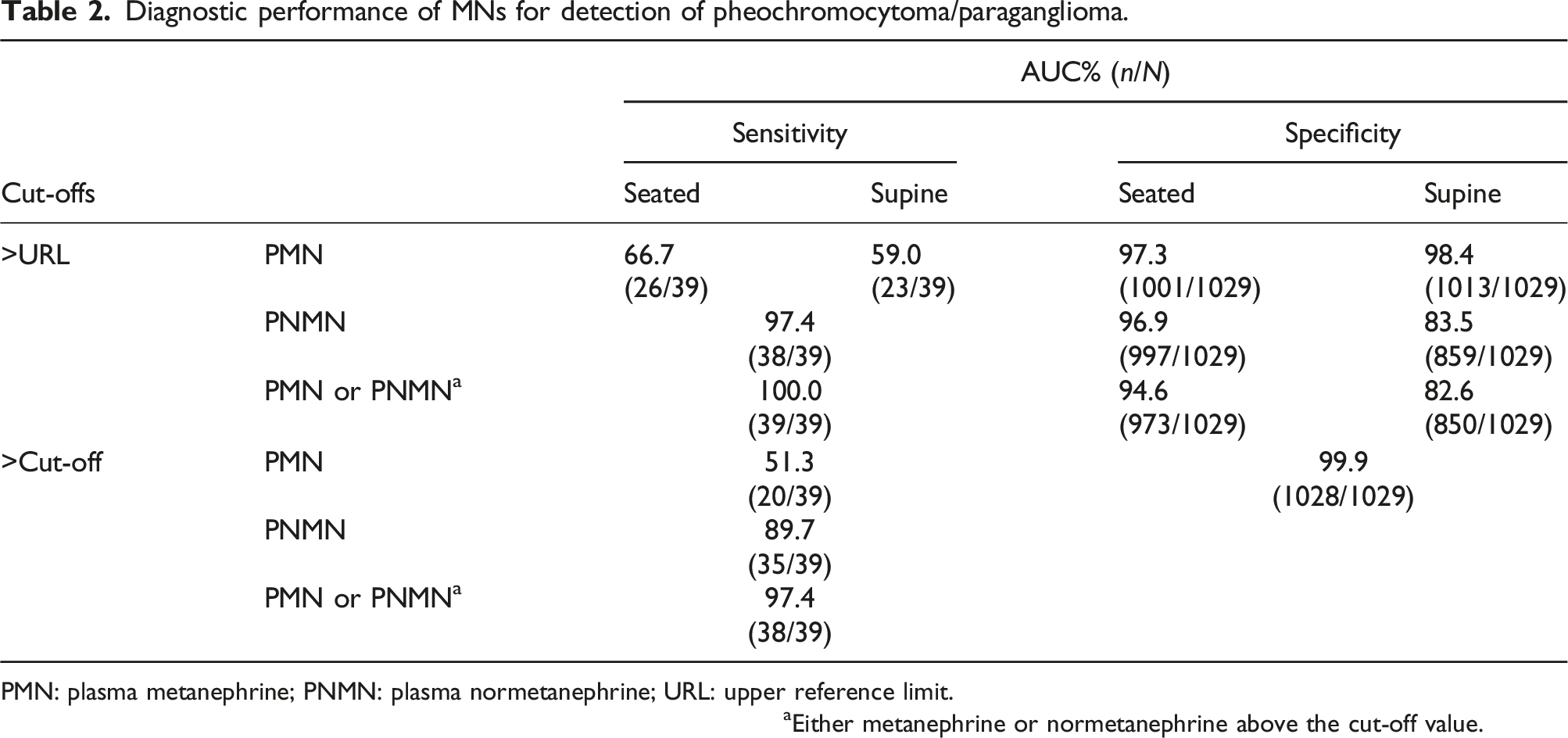
PMN: plasma metanephrine; PNMN: plasma normetanephrine; URL: upper reference limit.
aEither metanephrine or normetanephrine above the cut-off value.
According to the ROC curve, combining the requirements for sensitivity and specificity (the maximum value of the Youden index), the cut-off value was 0.33 nmol/L for MN (sensitivity 71.8%, specificity 93.9%), and 0.98 nmol/L for NMN (sensitivity 100%, specificity 93.8%). Taking the point with 99.9% specificity and the best sensitivity as the diagnostic cut-off point of PPGL, the cut-off values were: MN 1.63 nmol/L, NMN 2.17 nmol/L. They were about 4 times and 2 times the upper limit of the reference intervals of this study, respectively. When MN and NMN were used as joint test indicators, the diagnostic performance was optimal (AUC = 0.999) (Figure 2). Receiver-operating characteristic (ROC) curves for comparisons. Areas under the curve (AUC) of ROC for plasma MNs tests are shown.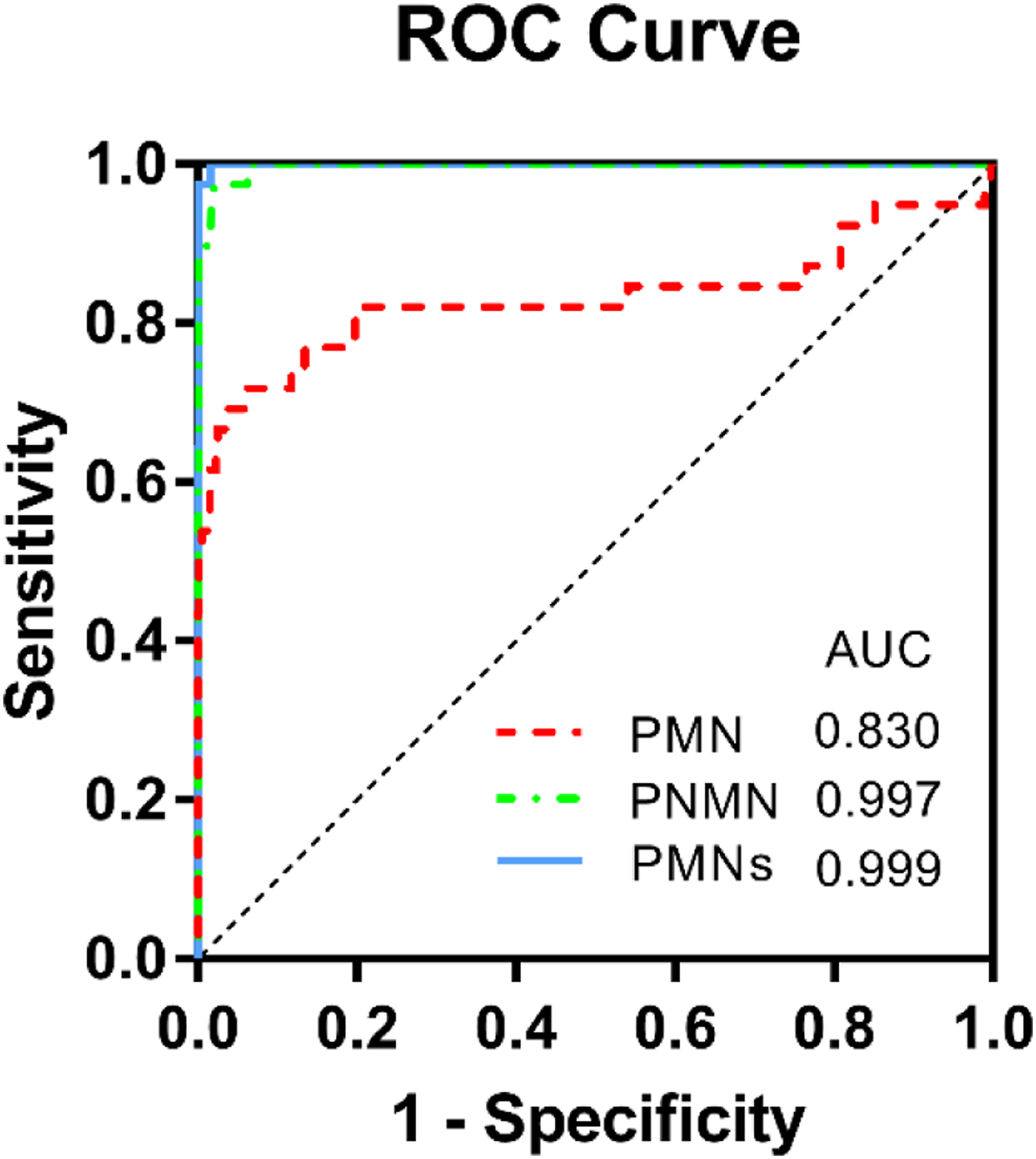
Comparison of two reference intervals applications
Comparison of proportion in seated plasma MNs according to age, using spine age-adjusted upper reference limits and seated fixed upper reference limits.
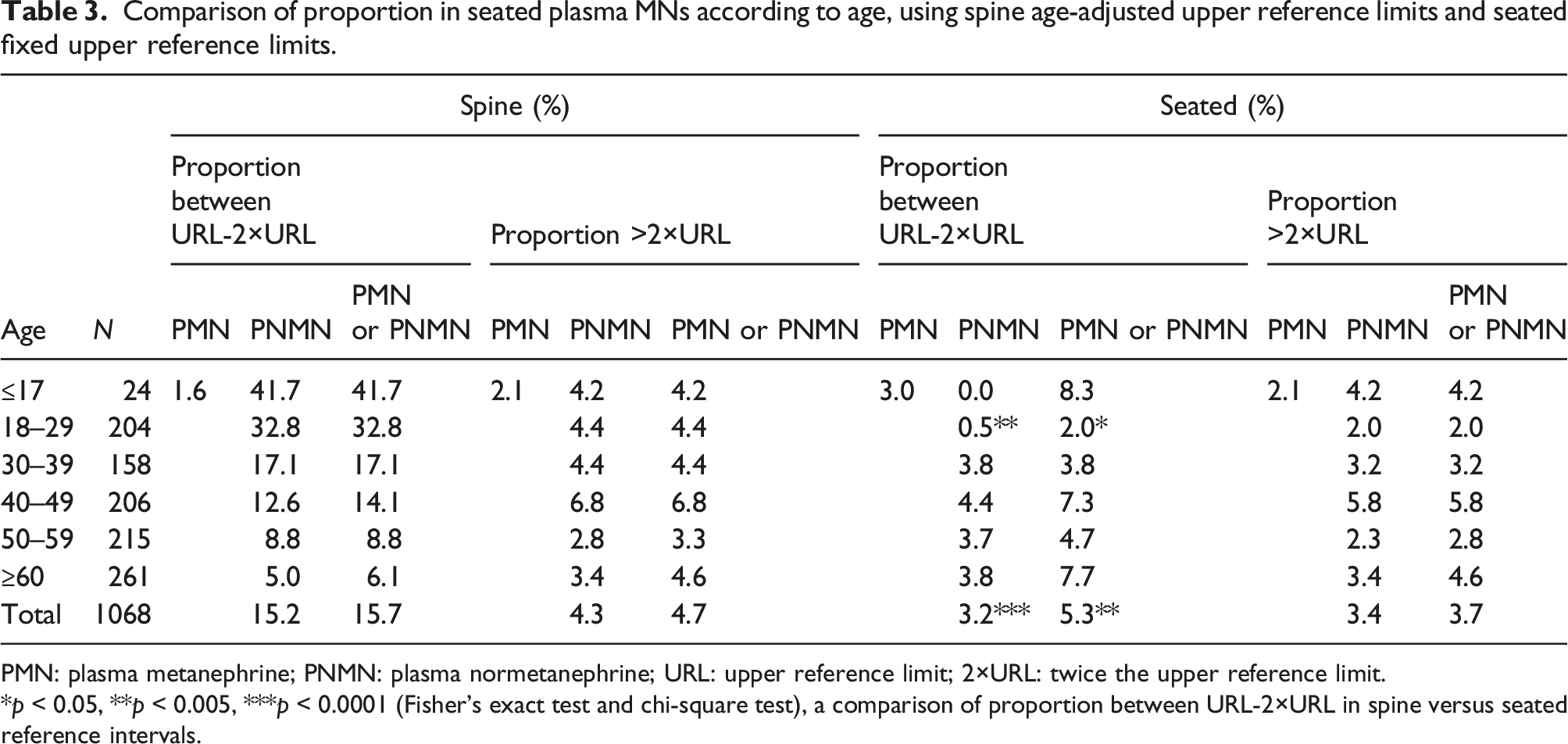
PMN: plasma metanephrine; PNMN: plasma normetanephrine; URL: upper reference limit; 2×URL: twice the upper reference limit.
*p < 0.05, **p < 0.005, ***p < 0.0001 (Fisher’s exact test and chi-square test), a comparison of proportion between URL-2×URL in spine versus seated reference intervals.
Discussion
It has been reported that there is a strong correlation between supine plasma NMN and age, so the reference intervals should be grouped by age.18–20 The reference population data in this study shows that there was no correlation between seated plasma NMN and age. MN did not correlate with age, which is consistent with previous reports.16,18,20 Therefore, instead of dividing the reference intervals of NMN by age group in the supine position, we used unified reference intervals for all age groups in the seated position. It is generally accepted that healthy populations should be selected for the establishment of reference intervals. However, in actual clinical work, among the patients who came for PPGL screening, there are a considerable number of hypertensive patients and patients with adrenal tumours found on imaging. Because PPGL patients often come to specialists with hypertension and headache as their first symptoms, and patients with occult PPGL often find tumours incidentally in imaging during physical examination or treatment of other diseases. Therefore, we included hypertensive patients and patients newly diagnosed with adrenal tumours in the reference population for analysis. Plasma NMN only differs between the healthy group and adrenal tumour group in the reference population, but did not differ among the remaining group, which is different from the supine position.18,19 We speculate that the increased catecholamine release is due to further activation of sympathetic nerves in the seated position, and the magnitude of the increase in NMN masks the differences in NMN due to non-PPGL disease. Although there were differences in plasma MN between the healthy group and the hypertensive group, considering that the difference itself was small, and we often judge the disease together with NMN, this difference would not have a decisive impact on the diagnosis, so we did not establish the reference intervals for these two groups, respectively. There was a gender difference in plasma NMN; males were higher than females, which is consistent with previous reports.18,19,22 However, our data shows that there was also a gender difference in MN between males and females, which is different from the previous reports. Of course, this difference does not affect the use of our established reference intervals.
In this study, the reference intervals for seated plasma MN and NMN were established as 0.07–0.40 nmol/L and 0.22–1.17 nmol/L, respectively, which are similar to the findings of Lagerstedt et al. 3 and Peaston et al. 15 Compared with the 0.15–0.9 nmol/L reported by Kim HJ et al., 16 the NMN is slightly higher. At present, most seated reference range data has been published by labs in western countries. In China and even in East Asia, only individual single-centre seated 16 or supine reference intervals have been reported,21,22 and some studies just used HPLC or LC-ECD methods, and no large-sample or multi-centre seated plasma reference has been established. Therefore, the reference intervals established in this study have certain reference values, especially in non-western countries. Based on the ROC curve, we combined the sensitivity and specificity requirements, and the cut-off values obtained were 0.33 nmol/L of MN and 0.98 nmol/L of NMN. Compared with the results of Lagerstedt et al., NMN is similar (0.90 nmol/L) and MN is slightly lower (0.50 nmol/L), and compared with Kim HJ et al., MN is similar (0.33 nmol/L) and NMN is higher (0.61 nmol/L). We conjecture that these differences are more likely due to the type of patients included, the status of patients before blood collection, and the preparation of specimens, rather than ethnic differences. We took the point with 99.9% specificity and the best sensitivity as the diagnostic cut-off point of PPGL, then the cut-off values were 1.63 nmol/L of MN and 2.17 nmol/L of NMN. In this study, they were about 4 times and 2 times the URL respectively. When MN and NMN were used as joint inspection indicators, the sensitivity was as high as 97.4%. Therefore, when any one of the MN and NMN indicators of a suspected patient exceeds 2 times or more of the upper limit of the reference intervals in this study (i.e. >2×URL), it is strongly suggested that the patient has PPGL.
However, it is more troublesome for patients whose MNs value (especially NMN) is in the URL-2×URL, the “Grey Area”. At present, the latest guidelines4–6 and studies suggest that8,14 for such patients, at least one (preferably multiple) re-examination in the supine position should be performed to help confirm the diagnosis. For PPGL, a rare disease with potentially serious consequences, ensuring screening sensitivity is paramount. As mentioned above, previous studies have found that for seated sampling, the application of the reference intervals in the supine position is more sensitive, so it is recommended to use the supine intervals instead of the corresponding seated reference intervals in the seated position. When we compared the diagnostic performance of the age-adjusted supine reference intervals with the self-established seated reference intervals by the upper limit, we found that the sensitivity of the seated intervals was similar to that of the supine ones and the specificity was improved, especially in the URL-2×URL interval (Table 3, Figure 3). Diagnostic sensitivity was comparable between the seated and the supine intervals. However, 15.7% of patients without PPGL had false-positive MNs using the supine reference intervals (especially in the juvenile and young population; 41.7% and 32.8%), while only 5.3% had false-positive results using the seated range (the higher false-positive rate among young people was only 8.3%). Using the seated reference intervals can greatly reduce the proportion of false positive of MNs in the URL-2×URL range. In addition, it can also be seen from Figure 3 that no matter whether the supine or seated reference intervals is used, the false-negative patients with NMN in the URL-2×URL interval cannot be distinguished. Although in our study, the MN of these patients was higher than 2×URL, according to the aforementioned judgement strategy, they could be classified as strongly suspected of PPGL. However, for the sake of safety, for suspected patients whose MN and NMN results are in the URL-2×URL interval, they should be resampled for re-examination. Distribution of seated PNMN results in 1068 subjects according to their age and with seated fixed upper reference limits and spine age-adjusted upper reference limits.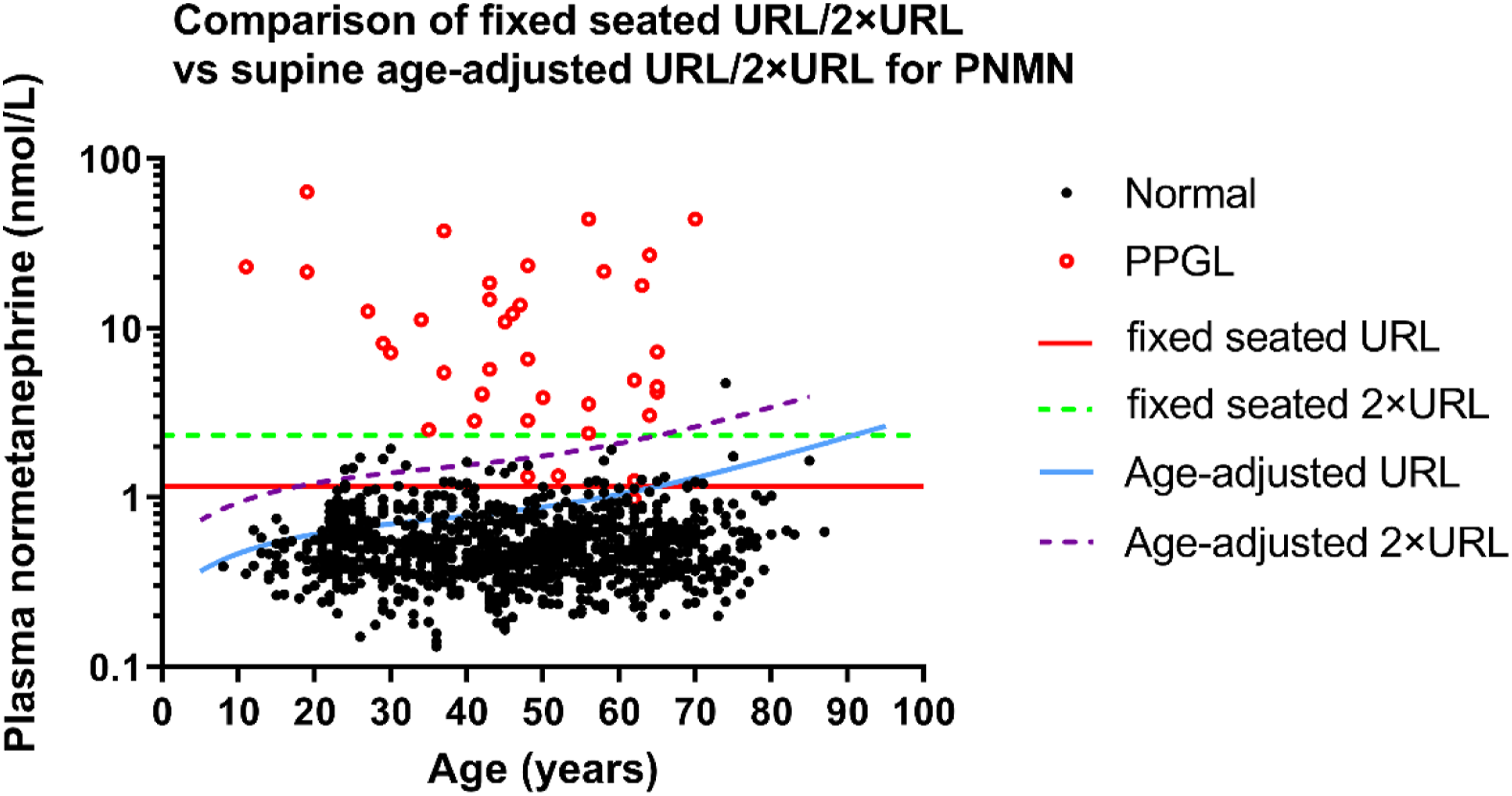
Higher specificity means a lower false-positive rate, which is also important for the diagnosis of PPGL as well as sensitivity, especially for patients whose MNs are only slightly above the URL, which will greatly reduce their economic and mental burden. But at present, in most hospitals in many different areas, it is difficult to carry out a strict 30-min supine position sampling for every patient. In our hospital, blood drawing in the supine position is only performed in inpatients, and all patients in outpatient are sampled in the seated position. On the one hand, the patient’s subjective acceptance is low, and the main reason is that blood drawing in the supine position requires additional economic costs for both the patient and the hospital. For example, most hospitals currently have limited space for outpatient clinics and physical examination blood collection rooms, so there are no special quiet rooms and beds. If a patient needs to have blood drawn in a supine position, this can increase the budget for admission and phlebotomy. For the hospital, it is also impractical to allocate dedicated sites and personnel for universal supine sampling. 12 Therefore, under deliberation, seated blood collection is still an economical and practical solution at present. The seated reference intervals we established have excellent diagnostic performance and may provide a reference for other laboratories. The shortcomings of this study are as follows. First, it is a single-centre study, and the number of collected specimens is limited, especially that of minors (<18 years old) is insufficient. Peitzsch et al. 23 established reference intervals for the supine position of MNs from infants 2 days old to 17-year-old minors in Germany, which is quite different from that of adults. This needs to be further explored by collecting more samples from different populations in the future. Second, some patients in the validation population lack imaging. 209 hypertensive patients lacked imaging diagnosis data and were excluded as PPGL based on symptoms, signs, and laboratory indicators. Considering the very low incidence of pheochromocytoma (approximately 1:6500–1:2500 in the US population and 0.1–0.6% in hypertensive patients), 24 the impact of these deletions in this study can be ignored. Moreover, it has been reported 25 that false negatives are more likely in patients under surveillance (previous PPGLs history or hereditary risk). There may be a difference in the diagnostic sensitivity of using seated versus supine ranges in these surveillance populations. However, there are only 3 patients belonging to surveillance populations of PPGL in this study, so it is difficult for us to make the above comparison, which needs further research in the future.
In conclusion, this study established reference intervals for seated plasma MNs from a large single-centre Chinese population sample. The data shows that using the corresponding seated reference intervals for seated plasma MNs has the same sensitivity as using the age-adjusted supine reference intervals, while the specificity is superior, which can reduce the unnecessary re-examinations of suspected patients with plasma MNs slightly elevated. Moreover, the cut-off value established by seated plasma MNs according to the ROC curve has good sensitivity and specificity for the diagnosis of PPGL. Although collection in a supine position is currently recognized as the best standard for plasma MNs sampling, it is difficult to establish universally due to the requirements for site and personnel. Seated sampling is more convenient and economical, and can be used in practical clinical work as an important method for MNs sampling.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethical approval
This research was approved by the Ethics Committee of Fujian Medical University Union Hospital (Ethics approval number: 2022KY039).
Guarantor
Cao YP.
Contributorship
Lin YX designed and performed the experiment, collected and analysed the data, and wrote the initial manuscript. Wang MH helped perform the analysis with constructive discussions and revised the manuscript. Cao YP contributed to the conception and design of the research and revised the manuscript. All authors reviewed and edited the manuscript and approved the final version.
