Abstract
Clinical laboratory medicine has seen the introduction and evolution of liquid chromatography tandem mass spectrometry in routine clinical laboratories over the last 10–15 years. There still exists a wide diversity of assays from very esoteric and highly specialist manual assays to more simplified kit-based assays. The technology is not static as manufacturers are continually making improvements. Mass spectrometry is now commonly used in several areas of diagnostics including therapeutic drug monitoring, toxicology, endocrinology, paediatrics and microbiology. Some of the most high throughput analyses or common analytes include vitamin D, immunosuppressant monitoring, androgen measurement and newborn screening. It also offers flexibility for the measurement of analytes in a variety of different matrices which would prove difficult with immunoassays. Unlike immunoassays or high-pressure liquid chromatography assays using ultraviolet or fluorescence detection, mass spectrometry offers better specificity and reduced interferences if attention is paid to potential isobaric compounds. Furthermore, multiplexing, which enables multiple analytes to be measured with the same volume of serum is advantageous, and the requirement for large sample volumes is decreasing as instrument sensitivity increases. There are many emerging applications in the literature. Using mass spectrometry to identify novel isoforms or modified peptides is possible as is quantification of proteins and peptides, with or without protein digests. Future developments by the manufacturers may also include mechanisms to improve the throughput of samples and strategies to decrease the level of skill required by the operators.
Introduction
Mass spectrometry (MS) has grown in popularity in clinical laboratories over the last 10–15 years. The Association for Clinical Biochemistry and Laboratory Medicine (ACB) has recently supported members to set up a Specialist Interest Group for MS to reflect this increase in interest. A survey conducted in 2012 inviting labs to share their methodology and applications gave responses from 25 labs in the UK and Ireland reporting over 70 different applications (unpublished data).
The increase in popularity for quantitative assays is likely to be due largely to its perceived increase in specificity when compared to immunoassay and the scope for combining multiple analytes into one analytical run, also called ‘multiplexing’. However, this may raise some ethical considerations when measuring analytes that may not have been requested by the clinician. Another likely reason for the increase in interest in liquid chromatography tandem mass spectrometry (LC-MS/MS) assays over the last 10 years has been due to unexpected withdrawal of immunoassay equipment, kits or assays, leaving laboratories struggling to provide a service. An initial shift, particularly for vitamin D was seen in 2006 due to the withdrawal of the Nichols Advantage. More recently, customers of Siemens Healthcare Diagnostics have seen a withdrawal of an androstenedione assay. Often lot-to-lot variability of commercially available immunoassays can show significant shift, whereas in-house assays are more in the control of the user.
Despite these obvious advantages, its increased application has not been without problems. The high level of technical expertise required has been acknowledged by manufacturers resulting in modifications to software and the production of analytical kits.
Elements of LC-MS/MS operation
Ion production
Electrospray ionization (ESI)
1
and atmospheric pressure chemical ionization (APCI)
2
are the two most common ionization techniques used in clinical laboratories. In ESI, the eluent from the column passes through a stainless steel capillary to which a charge is applied. A heated gas, usually nitrogen, passes the outside of the capillary resulting in the solvent being nebulized at the end of the capillary (Figure 1). The mechanism for ion formation is not fully understood, and there are two proposed models. In the first model, the ion evaporation model,
3
the droplet forms an ideal state to assist in desorption of solvated ions. The second is the charged residue model.
4
In this, the charge is carried by the solvent and as the solvent evaporates, the charge remains on the gaseous analytes. The conditions and solvents can be optimized for the formation of positive or negative ions. APCI utilizes a plasma region for desolvation and is considered to be a harsher ionization technique; however, it is felt that this is more efficient at removing matrix components.
Ion formation. The analytes of interest are present in the mobile phase and are nebulized using heated nitrogen gas to form a spray at the capillary tip to give charged droplets. The charged droplets undergo coulomb fission and result in smaller droplets. The final ion formation mechanism is unclear; however, the two main theories are depicted: the ion evaporation model and the charged residue model.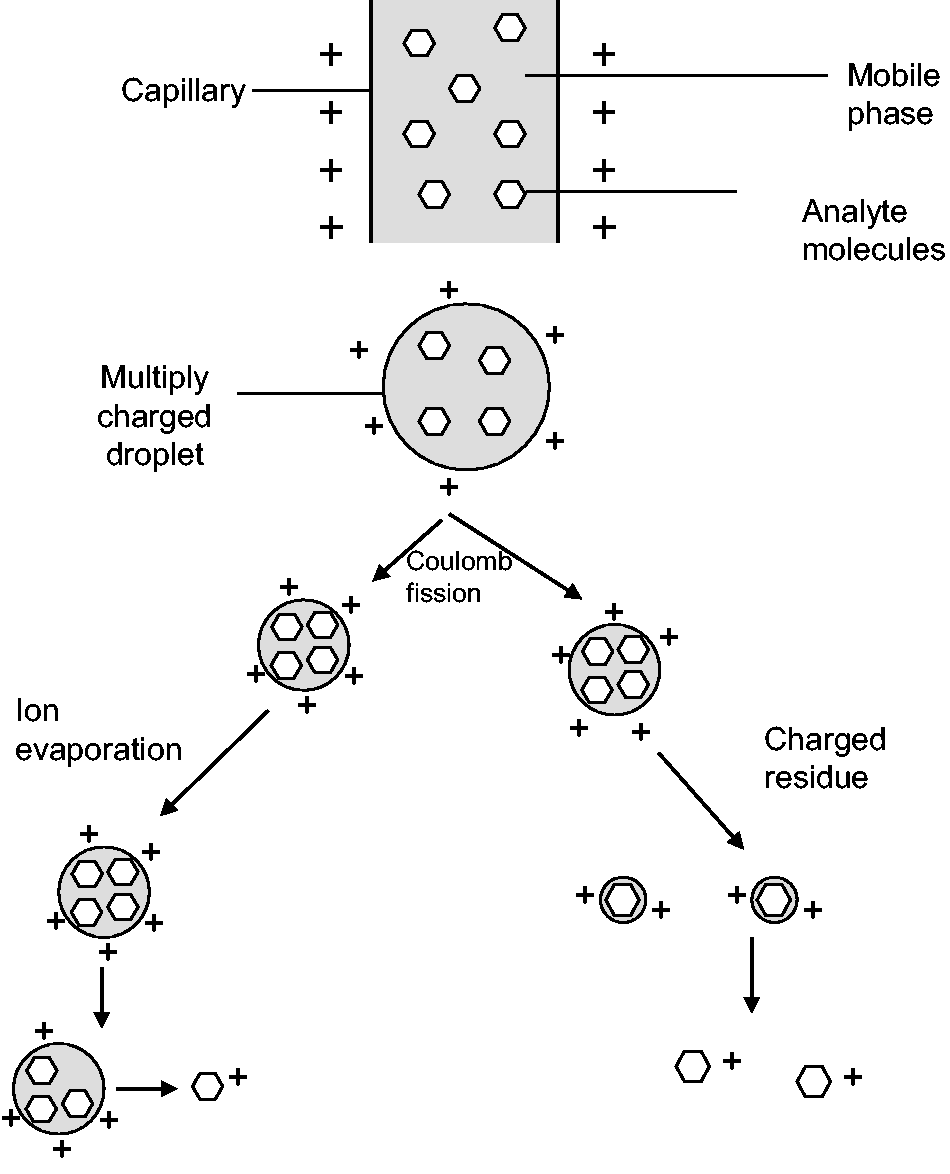
Fragmentation
After the parent ion has been selected by the first quadrupole, the ions enter a collision cell for fragmentation (Figure 2). The collision cell design varies between manufacturers, but all include the presence of a collision gas, usually argon or nitrogen. Kinetic energy is applied to the collision gas, and collision of the analyte with the gas molecules results in fragmentation.
Selected reaction monitoring. At the first quadrupole (Q1), only the analyte of interest has the correct mass-to-charge ratio and hence oscillation to progress to the collision cell. In the collision cell, the analyte collides with a gas such as argon or nitrogen to induce fragmentation. The subsequent fragments are then subjected to selection by a second quadrupole with the selected mass to charge fragment reaching the detector.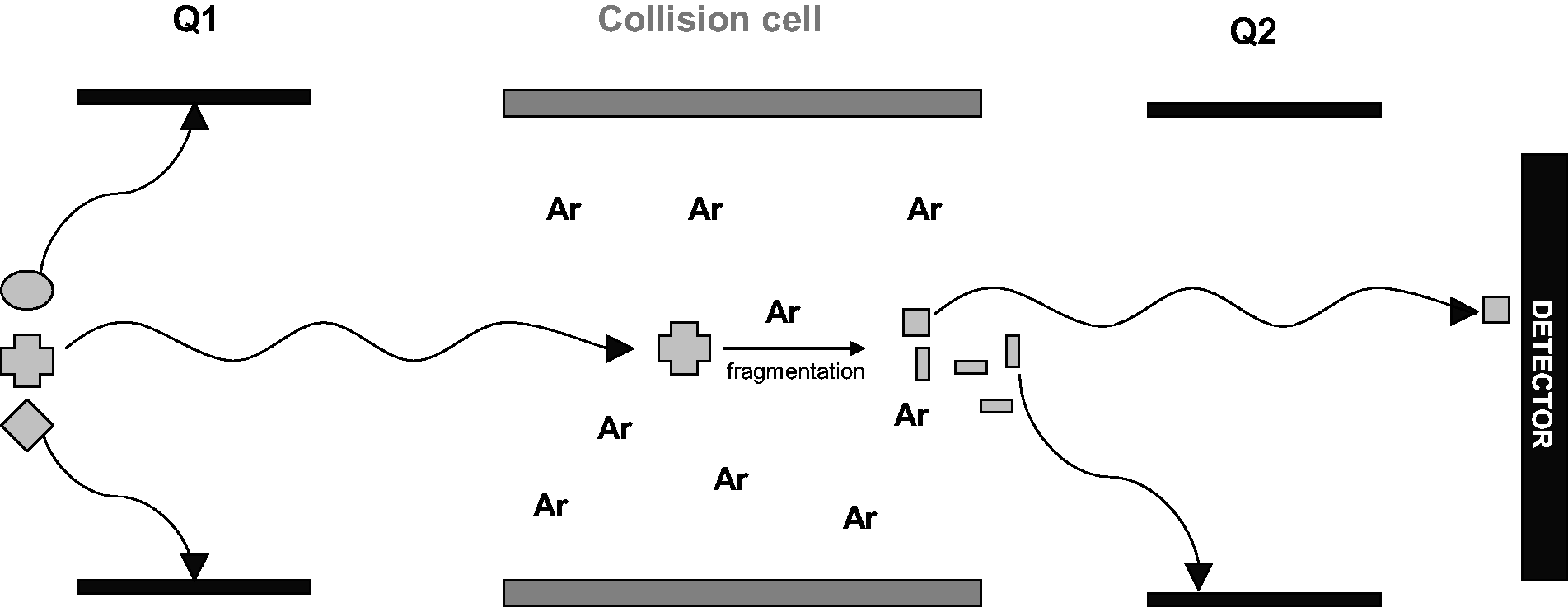
Transitions and scans
Quadrupoles operate on the basis of mass-to-charge ratio, referred to as
Mass spectrometers have the ability to monitor multiple transitions rapidly in sequence. These may include an analyte and its internal standard or also include other analytes and their internal standards. This is called ‘multiple reaction monitoring’ (MRM) and forms the basis of most quantitative assays.
LC-MS/MS may also be used in scan mode. 5 Most manufacturers can combine scans with quantitative LC-MS/MS for confirmation. Some manufacturers now offer equipment specifically designed to combine quantitation and confirmation with the introduction of an ion trap. Scans that can be performed on most LC-MS/MS include precursor scans, product ion scans or neutral loss scans. 5
Sample preparation
A wide variety of sample preparation techniques are used for the pretreatment of samples prior to LC-MS/MS analysis. A simple protein precipitation is used for many assays such as immunosuppressants.
This typically consists of the lysis of whole blood with zinc sulphate6,7 or by the addition of water followed by zinc sulphate. 8 It is thought that the addition of water prevents clumping of red blood cells, thus causing an improved lysis and a cleaner supernatant. 8 Solvent precipitation then follows by the addition of acetonitrile6,7 or methanol.9–11 This level of sample preparation is not dissimilar to the preparation required for semi-automated immunoassay methods for immunosuppressants that were used in the majority of laboratories before the widespread adoption of LC-MS/MS.
These techniques or similar are also adaptable to serum and plasma analytes and provides quick and simple sample preparation when sample dilution is acceptable. As more sensitivity or cleaner extracts are required, other sample preparation techniques may be utilized that allow for sample extraction and/or concentration, e.g. solid-phase extraction (SPE),12,13 supported liquid extraction 14 (SLE) and liquid-liquid extraction (LLE).15–17
Derivitization is commonly associated with gas chromatography techniques but is still applicable for increasing sensitivity in LC-MS/MS analysis. Although derivitization is used rarely among routine clinical LC-MS/MS assay, this and other more involved sample preparation still has a role for some analytes that are more difficult to measure.18,19 A recently published review demonstrated the importance and diversity of sample preparation. 20
Kits and calibration
A limitation of quantitative MS is the restricted availability of commercial calibrators, and these still need to be prepared in-house for many assays. There are several suppliers of immunosuppressant and vitamin D calibrators. In the case of vitamin D, harmonization of the calibrators used has shown to decrease significantly the assay-to-assay variation seen between LC-MS/MS users,21,22 highlighting the difficulties of preparing them in-house. One source of variation for endogenous compounds may be the chosen matrix as matrix matching proves difficult. Several approaches have been used, e.g. methanolic, 23 phosphate-buffered saline based,17,24 human serum or plasma,25,26 charcoal stripped serum 27 or animal serum. 28 One study has shown that if sufficient attention is paid to the removal of matrix effects and ion suppression caused by matrix components, that matrix matching of calibrators is not necessary. 29
Manufacturers have begun to offer kits for the analysis of panels of tests such as immunosuppressants,30,31 steroids 32 and vitamin D metabolites. 33 These offer a complete ‘out of the box’ solution that are usually supported by the manufacturer, thus reducing the expertise required for developing and maintaining an in-house assay. The disadvantages may include wide lot-to-lot variation as seen in immunoassay, and these kits would typically be considerably more expensive than an in-house assay.
Column choice and ion suppression
The choice of chromatography column is arguably the most critical part of LC-MS/MS method development. The selected analytical column needs to have the appropriate selectivity to separate the analyte of interest from isobaric interferences or from any non-specific interferences that can be visualized on the baseline.
34
As well as these requirements, the column must also separate the analyte of interest away from any compounds that may cause suppression of ionization not visible as interferences on the baseline. Specific experiments must be conducted to identify where these compounds elute35,36 (Figure 3). Compounds that may cause ion suppression include matrix components such as salts and phospholipids35,37,38 or compounds that may be introduced into the assay such as slipping agents, e.g. oleamide from the plastics used in blood and serum collection and storage.
38
Internal standards are used during the sample preparation and may compensate for some matrix effects seen. The choice of internal standard is important as even if deuterated stable isotope is used matrix effects, referred to as differential matrix effects may be observed.
39
Sample clean-up techniques such as protein precipitation, SPE, LLE, SLE, phospholipid removal plates or turbulent flow are often used to try to remove compounds that cause ion suppression.
40
Despite this, their removal is usually not complete by these processes, and chromatography plays a major role. Combining the separation required without creating excessive run-times is a challenge.
An example of an ion suppression infusion experiment chromatogram for prednisolone analysis. In (b), a constant infusion of prednisolone is entering the mass spectrometer as an extracted sample is being injected. An illustrative prednisolone peak is given in (a) for comparison. Ion suppression can be observed as reduction in the baseline in (b), as is seen at 0.5 min, highlighting that other compounds are elution that are competing for the limited ion formation capacity.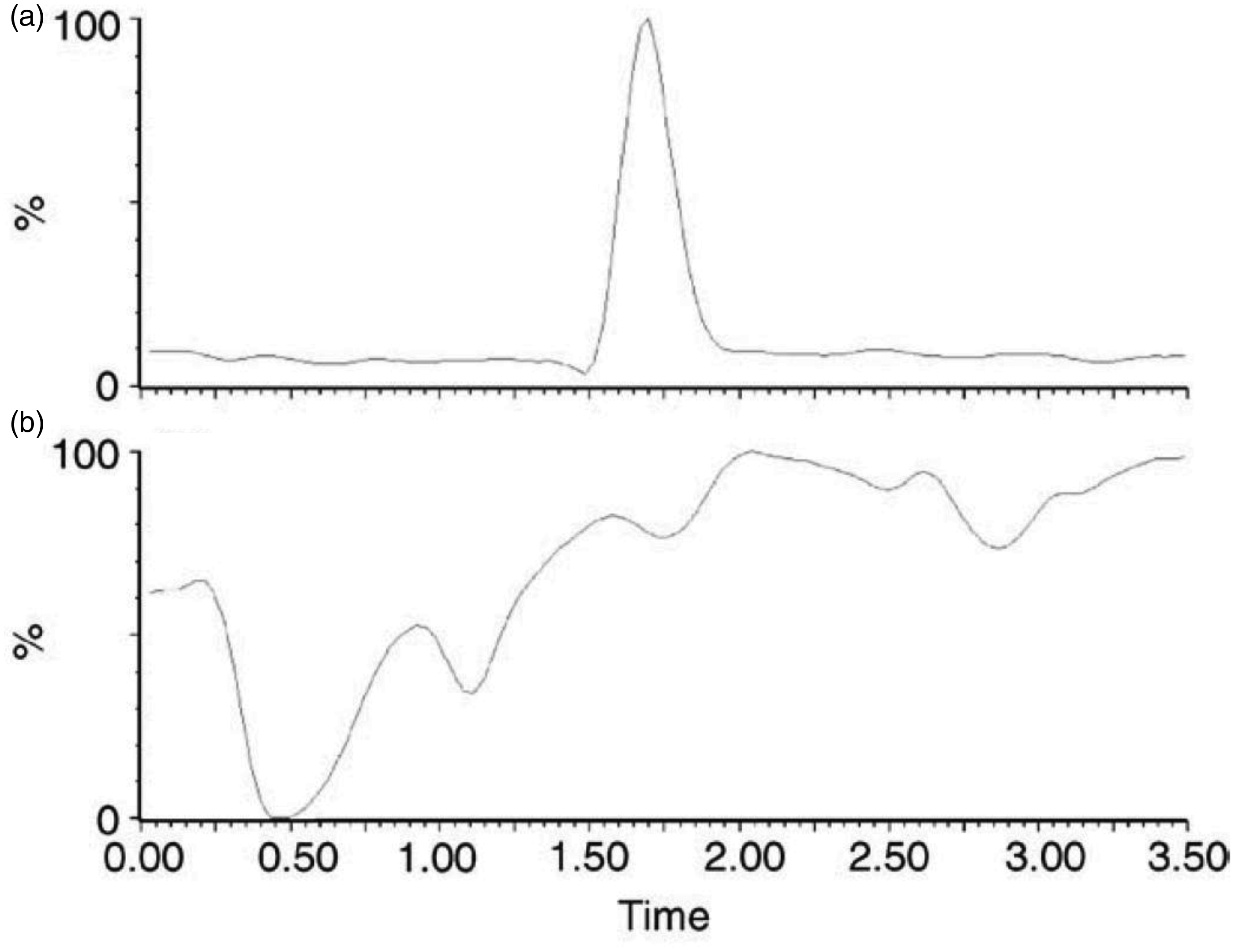
There is a large variety of chromatography columns available from each manufacturer, and this range is constantly evolving. In our experience, there are often differences between manufacturer’s products even with the same packing material. For example, two manufacturer’s silica-based columns with C18 coverage can show vastly different retention capacities and selectivity; one contributing factor is carbon loading. There have also been new developments in column technology in recent years, such as the emergence of fused core column technologies which propose faster re-equilibration times and therefore reduced run-times. 41
Standardization and reference method services
There is considerable heterogeneity between LC-MS/MS laboratories when measuring the same analyte. For 25-hydroxyvitamin D analysis, there can be different equipment, calibrators, internal standards, sample preparation, chromatography and transitions. Any of these factors can contribute to interlaboratory variation with differing extents. 42 For example, the use of common calibrators has been shown to significantly reduce the interlaboratory variation between LC-MS/MS users, suggesting that it was the preparation of calibrators that had the greatest effect. 21 However, for testosterone, the use of a common calibrator did not show significant reduction in interlaboratory coefficients of variation (CVs), and it was proposed that it was methodology differences that were responsible. 43
Standardization efforts are currently limited in the area of quantitative LC-MS/MS due to the wide variety of assays and matrices utilized. Even when a reference method exists, it is difficult for National Health Service laboratories to take part in method comparisons with reference laboratories or utilize matrixed reference panels due to expense. An ideal and affordable way for labs to compare their assays to reference methods is the use of external quality assurance (EQA) material with assigned reference values such as the NIST assigned values to vitamin D EQA since 2013. 44 The Wales External Quality Assurance Scheme also offers reference method services. The onus is now on EQA providers to make this a more widespread activity.
Isotope dilution liquid chromatography tandem mass spectrometry (ID-LC-MS/MS) is a technique that has recently grown in popularity for those developing reference methods but has been discussed in the reference method literature for at least 20 years 45 and was first proposed as an analytical tool in 1954. 46 As well as reference methods being available for analytes that are routinely measured by LC-MS/MS such as steroid hormones17,27,47,48 and amino acids,49–51 there are also methods for analytes for use as a reference method alone. These analytes include glycosylated haemoglobin (HbA1c),52,53 cholesterol54,55 and free thyroid hormones56–62 among others. It should be noted that the isotope dilution technique is quite distinct from the routine use of stable isotope internal standards, although the term is often used interchangeably.
Internal standard choice
One of the areas that has changed since the methods were first reported is the area of internal standards. Original assays developed often used analogues, e.g. cyclosporin D for cyclosporin A7,8,13,63,64 analysis and ascomycin for tacrolimus.6,8,10 Typically, analogues are not as good as stable isotopes although ascomycin is still regarded as a good internal standard in tacrolimus measurement. These were later superseded by the availability of stable isotope internal standards.31,65,66 Internal standards should be added to the sample early in the sample preparation protocol. They are present to account for any loss of sample during the extraction process and for any variation in ionization at the mass spectrometer source. To do this effectively, they must elute at the same time as the analyte of interest which was not always possible for analogues. Isotopes are more likely to behave similarly to the compound of interest during the extraction and ionization processed than analogues and should therefore best represent any losses.67,68 They will also fragment in the collision cell in the same manner as the compound of interest.
The stable isotope most commonly used is deuterium as this is easy to incorporate and inexpensive. However, loss of label can occur due to deuterium exchange with hydrogen.69,70 It is also possible to chromatograph a heavily labelled deuterium internal standard away from the compound of interest. 37 Carbon 13 isotopes give highly pure internal standards that are chemically stable with the same elution as the compound of interest, 37 e.g. 13C3 testosterone-2, 3, 4. Nitrogen 15 internal standards have similar advantages as carbon 13; however, both require a more complicated synthesis and are usually more expensive. An example of the use of a stable nitrogen isotope would be 13C-15N-Angiotensin I in the LC-MS/MS measurement of renin activity.71,72
There are stable isotope internal standards available for most common applications, and the user may even have a choice, e.g. D2 testosterone-1,2 or D3 testosterone-16, 16, 17 or D5 testosterone-2, 2, 4, 6, 6 or 13C3 testosterone-2, 3, 4. If no carbon 13 isotope is available and there is a selection of deuterium isotopes available, the location of the deuterium atoms on the molecule will determine how likely they are to exchange, so users should discuss this with the manufacturers prior to selection. Generally speaking, carbon 13 or nitrogen 15 isotopes should be the internal standard of choice compared to D2 isotopes when setting up a new assay. Care should also be taken if considering a D2 isotope of a carbon-rich molecule such as testosterone due to the effects of natural isotopes. 73
Without the use of a suitable internal standard, measurements may be prone to unpredictable ion suppression matrix effects from samples or plasticizers introduced during sample preparation stages. 35
Matrices
MS gives the user the flexibility to measure analytes in alternative matrices for which it would prove difficult to use a kit method which may not be fully validated. Drug analysis is commonly carried out on hair samples as drugs can be detected in hair for much longer time periods than in other matrices and hair analysis can also give information on previous drug abuse.74–77 Saliva sampling is viewed as less invasive than blood sampling and can be very useful for getting multiple samples over the course of time. Examples include assessing the adequacy of hydrocortisone78,79 or testosterone 16 replacement, or for obtaining late night or early morning samples for analytes that show circadian variation.80,81 Dried blood spot analysis has been used for many years for newborn screening82–84 and is now used for many different LC-MS/MS assays due to the increased stability of many analytes in dried blood spots compared to whole blood or serum. 85 Other matrices for which MS assays are reported include fibroblasts, 86 bile, 87 tears 88 and cerebral spinal fluid.89,90
Clinical MS is constantly evolving and is applicable to lots of different areas of laboratory medicine.
Current clinical uses of MS
Therapeutic drug monitoring
One of the first uses of quantitative LC-MS/MS was for the therapeutic drug monitoring (TDM) of immunosuppressants. Cyclosporin and tacrolimus were the first assays to be developed, and many of the methods in use have changed little since they were first reported.
There are still at least seven immunoassays in use as reflected in EQA schemes. Some show distinct differences to MS and bias when compared to the target concentrations. This may be due to differences in specificity and/or calibration. Some immunoassays also demonstrate high between-laboratory CVs, up to around 15%, whereas MS is approximately half of this at around 8% even though different instruments, chromatography and calibration techniques will be in use for cyclosporin (Analytical Services International Proficiency Testing Scheme, Distribution 360). At least four other immunoassays are available for tacrolimus and some demonstrate poorer interlaboratory CVs at almost 26%; however, numbers are fewer. MS users on a recent distribution have a CV of 12.1% for a sample at the lower end of the therapeutic range (Analytical Services International Proficiency testing scheme distribution 226). Recent studies have shown that this interlaboratory CV can be improved further for MS if common platforms and calibrators are used. 91
One major advantage of LC-MS/MS over immunoassay in the measurement of immunosuppressants is the ability to multiplex analysis of multiple analytes in one analytical run. Cyclosporin and tacrolimus are commonly combined into one assay, 63 and sirolimus and everolimus and/or mycophenolic acid may also be included.10,92–94 MS multiplexing also gives the ability to measure related drugs such as everolimus for which there is no immunoassay readily available for the major immunoassay platforms.
LC-MS/MS offers users the flexibility to select different sampling strategies due to its low sample volume requirement. Fingerprick sampling has been used reliably for many years7,95–98 and makes a significant difference to patient care as they can collect the samples themselves at home and post to the laboratory. This prevents regular journeys to transplant centres for long-term follow-up. Later studies have demonstrated the use of dried blood spot collection for long-term sample stability and avoidance of clotting.99–104 One further reason for the popularity of immunosuppressant assays among LC-MS/MS users is the availability of commercially produced calibrators. Early assays involved calibrator production in-house using whole blood with added drug to give target concentrations64,105 which has obvious ethical and health and safety implications. Commercial calibrator sets became readily available quickly negating the need for this process, and more recently complete kits have become available.30–33
Immunosuppressants are not the only drug group for which LC-MS/MS is used for TDM; however, they are the largest group. Other uses include the measurement of antiepileptic drugs,106–110 antibiotics79,111,112 and antifungal drugs,113–116 all of which may be as single assays or as part of a larger multiplexed panel. The details of these assays have been covered by a recent review. 117 There are some assays also reported for more esoteric analytes such as ribavirin82,118–120, clozapine,121–125 imatinib,126–131 busulfan,132,133 caffeine134,135 and amiodarone.136,137
Toxicology
For many years, gas chromatography mass spectrometry (GC-MS) was the technique of choice for toxicological analysis due to its ability to screen for unknown drugs as well as to perform targeted screening for specific drugs. Immunoassay is commonly used in many laboratories as a first-line screening test, but any positive results must be confirmed by a second method, such as gas chromatography (GC) or LC-MS/MS. Immunoassays are also notorious for producing false-negative as well as false-positive results. 138 Although GC-MS is still commonly in use in many toxicology laboratories, the complexity of sample preparation, which usually involves time-consuming derivitization, has prompted many laboratories to move over to LC-MS/MS analysis. 76
LC-MS/MS can be used to screen for the presence of specific drugs in matrices such as urine, hair or saliva, by using MRM or SRM mode. Such an approach can be used to screen for multiple targeted analytes in one mass spectrometric assay.
139
A limitation of this approach is that it is unable to detect other substances that are present which are not part of the panel. Quantification of drugs can also be carried out using this method.
140
For a more systematic approach to identify the presence of drugs in a sample, many laboratories carry out a survey scan using MRM, followed by an enhanced product ion scan as a dependent scan.141–144 Drug identification is then carried out by spectral comparison with a product ion spectral library, and such methodology can identify hundreds of drugs in a single LC-MS/MS run.
141
This has become increasingly important in toxicology laboratories due to the emergence of unregulated psychoactive drugs, also known as legal highs.
145
High-resolution MS has been shown to be a valuable tool for this approach, the accurate mass and corresponding empirical formula of the unknown compound can be determined. This gives greater confidence in drug identification compared to lower resolution instruments.
146
A drawback of this approach is that isomers such as morphine and hydromorphone cannot be distinguished from one another. This problem may be overcome by producing collision-induced dissociation (CID) spectra of the compound of interest,
147
in combination with accurate mass and relative retention time to confirm the identity of the compound. An alternative approach, ion mobility, may be used to further distinguish these difficult analytes (Figure 4). Ion mobility is the separation of ions in the gas phase and is often coupled to time of flight instrument to provide separation of analytes prior to MS detection.
Ion mobility. Ions enter a drift tube where they are exposed to an electric field. The drift tube also contains a drift gas that travels in the direction opposite to the ions. These conditions cause the ions to separate according to size as the larger the ion, the more the drift gas impedes its progress through the drift tube.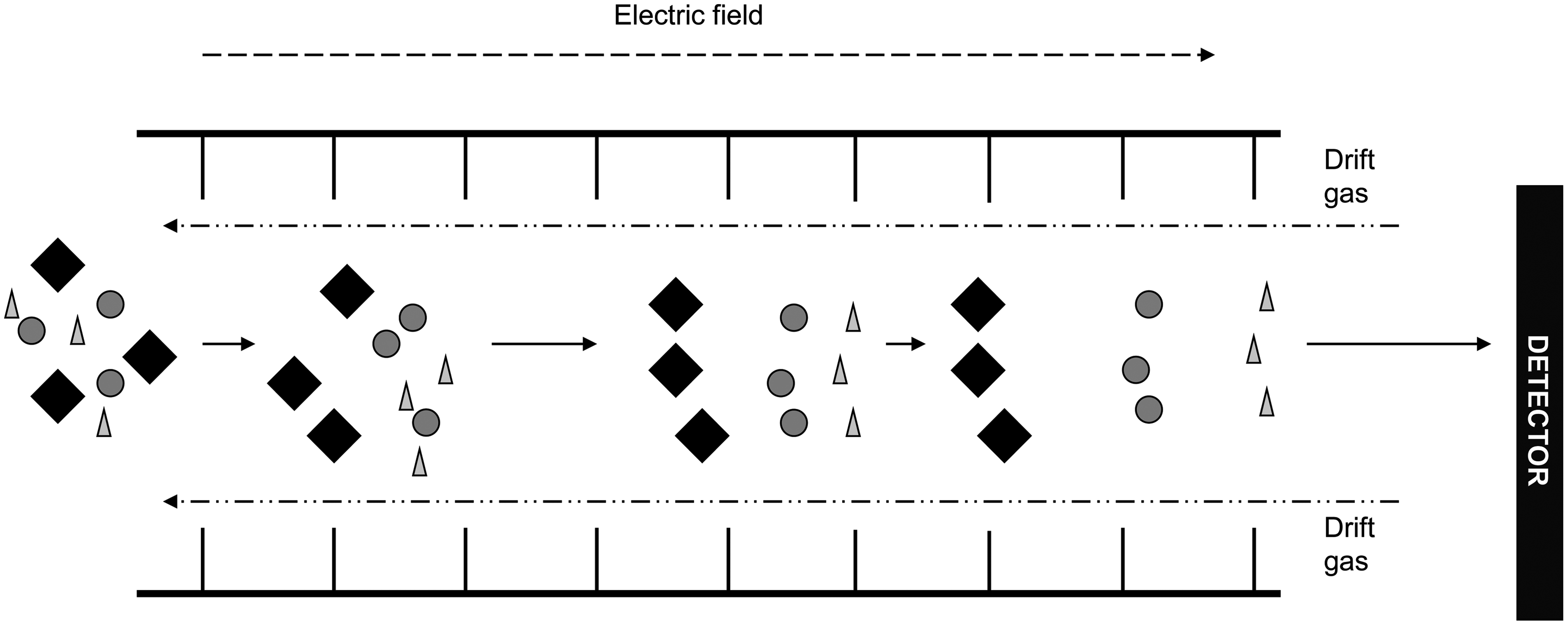
Endocrinology
LC-MS/MS is currently playing a major role in the area of endocrinology, particularly steroid hormone measurement and is useful as panels or related steroids may be measured simultaneously. Assays exist for testosterone,17,27,47,148,149 androstenedione,17,47,48,149 dehydroepiandrosterone (DHEAS),149,150 vitamin D,151–154 oestrogens,155–159 17 hydroxyprogesterone,48,149,160 serum24,161,162 and urinary cortisol161,163–166 among many more. The structural similarity between steroid hormones has been known for a long time to cause non-specificity issues for immunoassays, a well-documented example being DHEAS interference in testosterone immunoassays.
167
This can be observed as marked differences in method comparisons between immunoassay and LC-MS/MS or when reviewing EQA data, as method means for some immunoassays are 40% or greater than MS. There may be a case for measuring male testosterone by well validated immunoassays, but LC-MS/MS has also demonstrated superior performance at low concentrations for many analytes, e.g. female testosterone and oestradiol.159,168 LC-MS/MS may also give more clinical information over immunoassay in certain situations such as serum cortisol measurement in patients taking metyrapone
162
or prednisolone or the simultaneous measurement of dexamethasone and cortisol during suppression testing.
169
The use of LC-MS/MS in steroid hormone measurement has achieved recognition by clinical peer-reviewed journals such as the
Despite its generally accepted advantages, the measurement of steroid hormones by LC-MS/MS is not without problems. Some steroids have the same mass and fragment to give the same product ions and could therefore cause an isobaric interference. An example of this is 11-deoxycortisol, 21-deoxycortisol and corticosterone. 172 In these cases, careful attention to chromatography should be applied to ensure adequate resolution of these compounds. The carbon-rich nature of steroids gives rise to another type of isobaric interference due to the so called ‘mass (M)+2’ effect. 159 This refers to natural isotopes of steroids containing two carbon 13 atoms having the potential to interfere in the measurement of a compound which has a mass 2 Daltons higher. An example of this would be oestradiol and oestrone. Oestradiol has a mass of 272.4 Daltons and oestrone 270.4 Daltons, both fragment to give the same product ion. A natural isotope of oestrone containing two carbon 13 atoms would have the same mass as oestradiol. Although this interference would be small, as oestrone concentrations in serum are greater, it should still be avoided by adequate chromatographic separation of the two.
Specific binding proteins for steroids have the potential to cause variability in their measurement. For example, immunoassays may have differing abilities to remove cortisol from cortisol-binding globulin, as demonstrated by under recovery in female subjects taking the oral contraceptive pill (OCP) compared to non-OCP users. 173 On the other hand, as MS methods usually involved protein precipitation and/or extraction, they can therefore avoid significant binding protein-related differences. 24 Solvents used in the extraction can be tailored for optimal extraction efficiency.
One difficulty in the use of LC-MS/MS in the measurement of steroids arises when their concentrations are in very low picomolar concentrations, such as oestradiol and aldosterone. Radioimmunoassays have the ability to amplify signal to enable the measurement of these two steroids.174–177 Both of these steroids are additionally difficult to measure by LC-MS/MS because of their preference to ionize in negative ion mode which is typically less sensitive than positive ion mode. Nevertheless, they can still be measured using high-grade mass spectrometer instruments and careful attention to sample preparation. The use of LC-MS/MS in the field of endocrinology is not restricted to the measurement of steroids and has been applied to other analytes which have proved difficult to measure by alternative means such as urine178–180 and plasma181–185 metadrenalines, including measurement of 3-methoxytyramine (3-MT). 3-MT is not measured by immunoassay but is useful for detecting the likelihood of metastasis in paragangliomas. 186 Other applications include plasma renin activity187,188 and in a reference method capacity for, e.g. thyroxine.56,58,62
Paediatrics
MS has been a feature in paediatric laboratories for many years. It is used for the measurement of dried blood spot acylcarnitines for newborn medium chain acyl coA dehydrogenase deficiency (MCADD)189,190 and phenylalanine and tyrosine for phenylketonuria screening. 51 An extended newborn screening pilot has recently been conducted which would include tests for five additional metabolic conditions in the same analysis as the existing screening process. These five additional conditions are maple syrup urine disease, homocystinuria, glutaric acidaemia type 1, long-chain acyl coA dehydrogenase deficiency and isovaleric acidaemia. 191
The use of MS for the investigation of inborn errors of metabolism typically uses an alternative approach to quantitative assays. For example, acyl carnitines are derivatized to form their butyl esters.
190
Once derivatized, all acyl carnitines give a product ion with a mass-to-charge ratio (
Neutral loss scans of derivatized amino acids can be performed in the investigation of amino acid disorders. In this case, all parent and daughter ions are scanned with a specific loss.49,51,193 The loss is
Quantitative LC-MS/MS also plays a role in paediatrics. After the diagnosis of conditions by screening techniques, quantitative assays are used for disease monitoring. 195 Specific quantitative assays may also be used in the investigation of disorders such as those of bile acid synthesis. 196 Hexanoylglycine may also be measured in the investigation of fatty acid disorders. 197
Steroid assays are applicable to the paediatric population due to multiplexing resulting in reduced overall sample volume and the ability to measure uncommon steroids. 198 There are methods in the literature for urine steroid measurement by LC-MS/MS for the diagnosis of congenital adrenal hyperplasia (CAH) with a reduced false-positive rate; 199 however, this still typically remains a GC-MS assay. Second-tier testing for CAH is possible by the measurement of 17-hydroxyprogesterone or 11-deoxycortisol in plasma.48,160,199,200
Small molecule quantitation
The availability of LC-MS/MS in the clinical laboratory for large volume tests such as immunosuppressants, vitamin D or newborn screening gives the user the flexibility to consolidate a range of esoteric or very specialist assays for very little additional cost. Where separate assays exist such as high-pressure liquid chromatography (HPLC), enzyme-linked immunosorbent assay or colourimetric methods, additional equipment is required. These may include HPLC equipment and detectors, plate washer and plate readers and spectrophotometers and perhaps also reagent kits. Usually when these assays are converted to LC-MS/MS, additional cost is minimal, the main outlay being the analytical column and internal standard. This may be the reason why there are also niche small molecule assays reported in the literature that are performed by LC-MS/MS such as urine oxalate,73,75,201 citrate 202 and cystine69,78 which are used in renal stone clinics. Urine37,77,81 and/or plasma16,74,203 5-hydroxyindolacetic acid may be measured for the investigation or monitoring of neuroendocrine tumours. Markers of nicotine use such as cotinine204–208 may be used in patients with lung disease or transplants to monitor abstinence.
Microbiology
Quantitative LC-MS/MS has an established role in the measurement of antibiotic79,111,112,209 or antifungal113–116 drugs in Biochemistry, Microbiology or Mycology Departments. However, qualitative MS has an emerging role in the routine Microbiology Department in the form of matrix-assisted laser desorption ionization time of flight (MALDI-TOF) MS. Using this technique, an isolated colony is smeared onto the sampling slide before presentation to the MALDI-TOF instrument. After application of a 337-nm ultraviolet nitrogen laser, a collection of ionized peptides, specific to the organism, enter the flight tube. A spectrum is produced which can be compared to a library to identify the organism responsible for the colony. MALDI-TOF is reported to be up to 24 h faster at species diagnosis than typical microbiological or molecular techniques70,80,210,211 and has a high sample throughput. However, this technique has been shown not to be able to differentiate between closely related bacterial species such as
Current analytical considerations
Validation
As with all in-house assays, LC-MS/MS assays must be subject to thorough method validation as described in more detail by Honour 214 in 2011. Other guidelines for method validation have been quoted in the literature, e.g. the ACB guidelines, 215 clinical laboratory improvement amendments, 216 European medicines agency217,218 and the food and drug administration. 219 Local guidelines that laboratories use may soon require a revision if they are to meet ISO 15189 criteria. A particular area for consideration for in-house LC-MS/MS assays will be the monitoring and assessment of lot numbers and batches of reagents as these may be made frequently.
Batch versus random access
Typically, quantitative MS assays are performed in the clinical laboratory as batches rather than random access as in automated immunoassays. This type of analysis limits MS to those analytes for which a rapid turnaround time is not required. Automated sample handling coupled to mass spectrometers may go some way to reduce variability and thus the possibility of a more flexible approach, and such systems have been described for use in the microbiology laboratory 210 and for urine drug analysis. 202 Although such systems may not necessarily decrease the time required for sample preparation and analysis, they do decrease the amount of hands-on time required, which is a major advantage in today’s busy clinical laboratory.
A novel technique termed isotopic internal calibration (ICAL) utilizes differing concentrations of various stable isotopes that are added to samples to produce a calibration within the sample therefore not requiring additional time to analyse a multipoint calibration curve. 201 This could offer the ability to prepare and analyse samples in a random access fashion if required. This technique may still require significant staff time to prepare the sample and ensure the correct mobile phases and analytical column is in place if this differs from what had run previously. However, this is an advance towards random access MS.
Emerging and future applications
Automated sample handling
A major limitation to the use of quantitative MS in the routine clinical laboratory is limited throughput due to time-consuming manual sample preparation which requires skilled operator time.220,221 Many laboratories have invested in automated sample handling techniques to reduce the time needed.92,105,115,202,222–224 Methods described using automated sample handling robots often describe tube identification using barcodes, sample and reagent pipetting and may even incorporate sample extraction such as SPE.105,202 These methods often still require operator input such as mixing, drying, transferring and/or centrifugation steps. 105 Extractions involving solvents are also possible; 141 however, it is often difficult to accommodate these instruments within extraction units.
An attractive innovation would be an extraction technique that can be automated without requiring operator input from when the samples and reagents are loaded until the extracts are prepared. The direct extraction-elution method is an example of sample extraction that does not require precipitation, filtration, centrifugation or solvent evaporation 225 but is limited to neutral molecules such as vitamin D.
An alternative method of simplified, automated sample extraction utilizes magnetic microparticles with a functionalized surface to attract the analyte of interest while allowing the sample matrix to be removed from the analytical system.226,227 This approach has been successfully applied to a mycophenolic acid assay. 227
Pay-per-sample
The introduction of MS into the laboratory usually follows a capital purchase or a fixed term leasing arrangement. A novel approach provided by a collaboration of Anatune (Friiscan) and Agilent Technologies provides mass spectrometers with integrated sample preparation and preprepared methods that are run on a cost-per sample basis. This approach does not require the expertise in method development and maintenance as methods are supplied and supported by the manufacturer. Methods available include vitamin D, 228 immunosuppressants 229 and some steroids. 230 However, the use of the equipment is restricted to these applications, and they may be more expensive than in-house alternatives.
Peptide and protein quantitation
MS has been used as a tool in the proteomics field for many years particularly for protein identification using MALDI-TOF.231–233 Protein and peptide quantitation using tandem MS is a more recent advance.234–237 Tandem mass spectrometers have a limit to the size of molecule that can be quantified, often around a mass to charge ratio of 2000 Daltons. Peptides with a mass greater than this upper limit can still be quantified if they carry multiple charges; 238 however, larger peptides and proteins must be digested prior to the measurement of smaller peptides.239–241 Analogue proteins 242 or isotope-labelled proteins 243 as internal standards may be added to the sample prior to digestion or labelled peptides can be added later in the process.239,240,244,245
Using MS for protein measurement may address issues that affect the current methodology such as immunoassays for thyroglobulin. Thyroglobulin immunoassays are renowned for having interferences from heterophilic antibodies, and thyroglobulin measurement is a challenge due to its low abundance in serum.239,240 One reported assay combines immunoaffinity peptide enrichment and tryptic digest for the preparation of peptides. 246 Trypsin also was noted to digest the immunoglobulins that potentially interfere in the immunoassays, and MS allows three peptides to be monitored simultaneously. 246
Quantitation of growth hormone has also been of interest. It consists of several isoforms and variants due to post-translational modifications and complex formation. In these assays, key peptides are selected which are believed to be representative of the measurand; 247 however, such assays may not have sufficient sensitivity for all applications, such as detecting growth hormone deficiency.
A novel analytical technique, mass spectrometric immunoassay (MSIA) has been described for serum and plasma protein analysis248–250 (Figure 5). These assays utilize antibody-coated pipette tips to prepare samples and enrich proteins and peptides prior to analysis. These assays are reported to be rapid and high throughput for the analysis of e.g. parathyroid hormone (PTH)
249
and prostate-specific antigen.
250
MSIA. Antibodies are bound to a tip which is loaded with sample. The analyte of interest will bind the antibody. Wash steps are performed to remove unbound compounds followed by buffer elution to remove the analyte of interest into a collection plate for further sample preparation.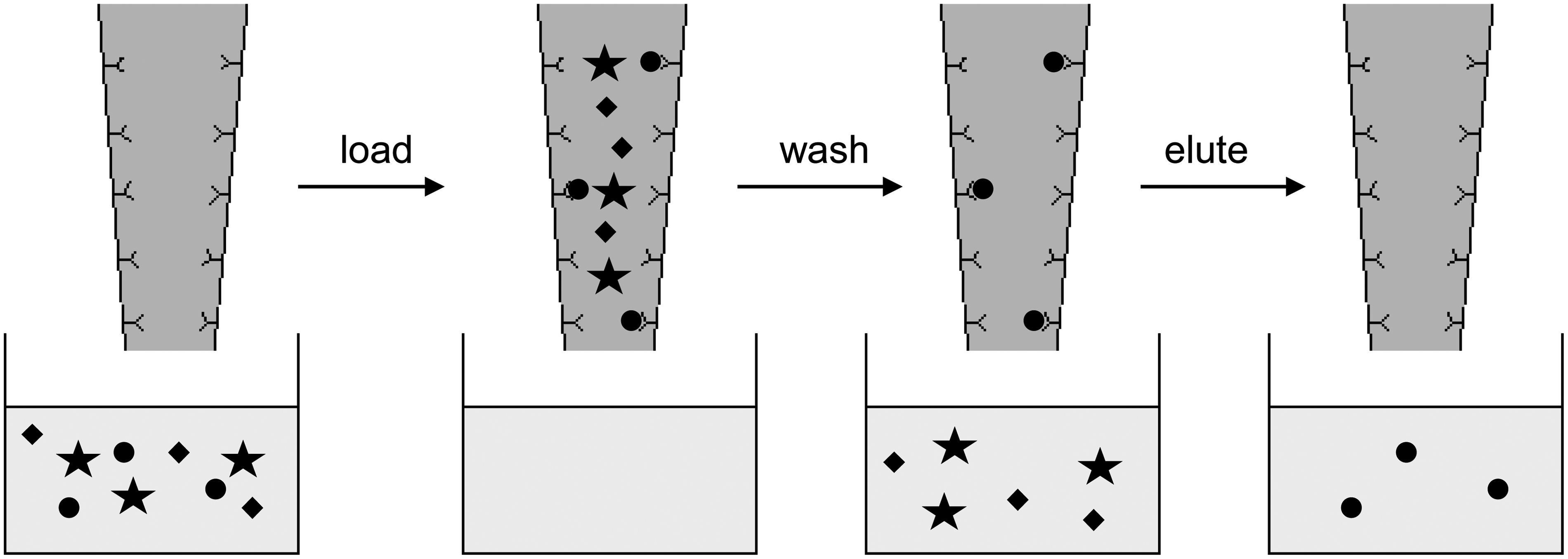
Metabolomics
Metabolomics is a key area of biomarker discovery where the global metabolite profile of test groups is compared to control groups. 251 GC-MS has typically been used in metabolomic laboratories252,253 but is limited to volatile and derivatizable compounds. LC-MS is increasingly being used as a complementary technique due to its ability to analyse a wider range of chemical species;254–257 however, top-end instruments including TOF capability are often required for biomarker identification. 254 Once biomarkers are identified, LC-MS/MS can be employed due to the rapid scan times of newer instruments to quantify large panels of metabolites simultaneously. 258
Point of care testing
MS has recently hit the headlines for its use in the operating theatre as a point of care testing device at Imperial College London. In this case, a technique called rapid evaporative ionization MS is used.259–262 The surgeon’s knife is connected to a mass spectrometer, and as the surgeon cuts through the tissue, some is vaporized and passed to the MS to produce spectra which can differentiate between healthy and cancerous tissue by comparison to a reference library. An initial study of 91 patients undergoing surgery has had a 100% success rate of determining whether the samples were cancerous or not which in the future may remove the need for intraoperative histology. 259 This advance shows that MS has an emerging role outside the laboratory and in direct clinical areas.
Total laboratory automation and random access
A combination of factors already described such as ICAL, 201 automated and/or simplified sample preparation,92,105,115,202,222–227 manufacturer prepared calibrators and reagents30–33 has the potential, if combined, to offer a completely random access solution to MS. If this is further combined with laboratory preanalytics such as centrifuges, decappers and tracking systems, a total laboratory automation solution could be a reality.
Conclusion
MS has many applications in the clinical laboratory, and the above is by no means an exhaustive list. Despite being considered to be a technically demanding process, manufacturers are making efforts to simplify sample handling, and many laboratories have adopted MS on the back of kit methods that are increasingly available. One of the main disadvantages is that assays are typically batched, and random access has not been universally developed; however, we envisage that in the coming years, further significant advancements in this technology will enable MS to be as easily used as main clinical chemistry analysers.
Footnotes
Acknowledgements
This article was prepared at the invitation of the Clinical Sciences Reviews Committee of the Association for Clinical Biochemistry and Laboratory Medicine.
Declaration of conflicting interests
None declared.
Funding
This review received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
Ethical approval
Not applicable.
Guarantor
LJO.
Contributorship
LJO and JEA researched, wrote and edited the manuscript. BGK reviewed and edited the manuscript.
