Abstract
Background
Liquid chromatography tandem mass spectrometry (LC-MS/MS) enables specific and sensitive quantification of steroids with a high throughput. Saliva sampling is advantageous for multisample profiling over longer periods of time, as it is non-invasive, cheap, can be carried out at home and does not require the attendance of clinical personnel. We developed a rapid LC-MS/MS for the measurement of salivary progesterone, frequently assessed as ovulation marker in patients desiring fertility.
Methods
Samples (300 μL) were prepared by supported liquid extraction using dichloromethane and were reconstituted in 40% methanol. Chromatography was performed using a C8 column with a water/methanol gradient containing 0.1% formic acid and 2 mmol/L ammonium acetate. Quantification was performed with a Waters TQ-S mass spectrometer.
Results
Total run time was 5.5 min. The lower limit of quantification was 20 pmol/L (1.2 fmol on column). Inter- and intra-assay comparison showed coefficients of variation and bias between measured and nominal concentrations of less than 11%. Mean recovery was 91%. Interference with a large set of natural and synthetic steroids was excluded. The assay was successfully applied to measure progesterone variation during the menstrual cycle (n = 9) and diurnal variations during luteal phase (n = 7) in regularly cycling women.
Discussion
We present a novel LC-MS/MS assay for the determination of salivary progesterone with high-throughput potential. The applicability of the assay for progesterone profiling during the menstrual cycle is demonstrated.
Introduction
Progesterone plays a crucial role in female fertility and reproduction. During the menstrual cycle, progesterone concentrations increase in the luteal phase and the peak in progesterone concentration is a widely accepted indicator of ovulation. 1 In female infertility, monitoring of progesterone concentrations can contribute beneficially to the success of assisted and non-assisted reproductive therapies.2,3 Additionally, increased production and maintenance of progesterone concentrations, first by the corpus luteum and then by the placenta, is essential for the maintenance of pregnancy, and therefore progesterone administration in early pregnancy has been investigated as a potential tool to reduce preterm birth risk.4,5
In clinical practice, monitoring of progesterone production for the surveillance of luteal and placental function requires the collection of multiple samples, repeated over periods of weeks to months, and therefore, a simple and cheap sampling technique would be most advantageous to use. The collection of saliva, in contrast to the standard medium of blood, is non-invasive and can be performed by the patients on their own without the need for attendance of clinical personnel. 6 Additionally, the concentration in saliva represents the free, unbound fraction of the hormone and is thus more likely to reflect the bioactive concentrations to which a target tissue is exposed. 7 Significant correlations between total and free progesterone concentrations in plasma and salivary concentrations have been described during pregnancy. 8 Daily saliva progesterone profiling has proven useful for the diagnosis of luteal phase defects, 9 and salivary progesterone has been suggested as biomarker for preterm birth.10,11 Saliva would therefore represent the preferred matrix for multisample analysis of progesterone, if a reliable, high-throughput assay were available.
For the measurement of salivary progesterone, a variety of immunoassays have been established since the 1980s12–20 and several of those are commercially available. However, immunoassays have the disadvantage of limited specificity due to cross-reactivity with compounds structurally related to the target analyte, which is particularly relevant for steroid analysis considering their large natural variety. 21 This limitation can be overcome by the application of liquid chromatography tandem mass spectrometry (LC-MS/MS), which exhibits outstanding specificity and also allows high throughput, if chromatography conditions are chosen carefully.21,22 Additionally, LC-MS/MS has the sensitivity to reliably quantify the low concentrations of steroid hormones present in saliva. Previously described LC-MS/MS assays for salivary progesterone had limited potential for the applicability in the routine clinical laboratory due to time-consuming sample preparation and lengthy run time, 23 or were using a Salivette devices for sample collection, 24 which can falsify the measurement by unspecific binding of salivary hormones to the Salivette swab.25–27 Our aim was to develop a direct, rapid LC-MS/MS assay combined with a simple, one-step sample preparation for the measurement of progesterone in unstimulated whole saliva, which is suitable for routine application. We strictly validated its analytical performance and demonstrated its applicability to monitor alteration of progesterone concentrations across the menstrual cycle.
Materials and methods
Collection of saliva samples
Unstimulated whole saliva was collected by the passive drool technique previously described.25,28 Blood contamination of saliva samples may lead to a false increase in saliva steroid concentrations due to the significantly higher concentrations in blood compared with saliva and needs to be avoided. Participants were therefore asked to avoid brushing teeth 2 h before the sample collection, to avoid eating 1 h before the sample collection and to rinse their mouth with water 10–15 min prior to the collection. Upon arrival at the laboratory, blood contamination was excluded visually and samples were frozen at −20°C. Samples were thawed, centrifuged at 3000 × g for 5–10 min and the clear supernatant was aliquoted and stored at −80°C for analysis. For the measurement of diurnal and menstrual variations of salivary progesterone, volunteers were healthy women (n = 9, age range: 26–39 years) with regular menstrual cycles taking no regular medication or oral contraceptives. All participants were asked to collect daily morning samples (between awakening and 10:00) on days 1–8 (follicular phase) and days 18–24 (luteal phase) of their menstrual cycle. Day 1 was defined as the start of the menstrual bleed. The number of samples provided per participants was four to seven for each of the luteal and the follicular phase morning sample collection (see Figure 2(a)). The time of collection ranged between 05:30 and 10:00. Additionally, participants were asked to collect a seven-time point diurnal profile during the luteal phase of the same cycle. Requested time points were 07:00, 09:00, 11:00, 13:00, 15:00, 19:00, 23:00. Samples collected within plus and minus 30 min of the requested time were accepted. Only samples from women with a surge of salivary progesterone during luteal phase, as determined by the analysis of the daily morning samples, were considered for the measurement of the diurnal profile (n = 7). All participants provided samples for at least five of the time points (see Figure 2(b)), and all samples have been collected between days 19 and 22. In total, the number of samples requested per participant over one cycle was 20.
The sample collection has been ethically approved by the Science, Technology, Engineering and Mathematics Ethical Review Committee of the University of Birmingham (ERN_17–0494); all volunteers gave written informed consent prior to sample collection; samples were anonymized and no personal identifying information was available at the laboratory.
Sample and standard preparation
Stock solutions were prepared by dissolving progesterone (Sigma Aldrich, Poole, UK) in ultrapure methanol (Fluka, Poole, UK) and were stored at −20°C. Two separately weighed stock solutions of progesterone were used to prepare standards (0–10,000 pmol/L) and quality controls (QCs; 100, 750, 1500 and 3000 pmol/L) by dilution in phosphate-buffered saline pH 7.4 (Sigma, Poole, UK), supplemented with 0.1% (w/v) bovine serum albumin (Sigma, Poole, UK) (PBS/BSA). The standards in PBS/BSA were used to prepare a calibration curve. Samples, standards and QCs were prepared in parallel by supported liquid extraction (SLE). Therefore, the 300 μL sample, standard and QC were pipetted into the wells of a 2-mL square well 96-well plate (Porvair, Leatherhead, UK) and 10 μL of approximately 40 nmol/L progesterone-2,2,4,6,6,17alpha-21,21,21-d9 (CDN isotopes, Quebec, Canada) in methanol was added as internal standard (IS) using a multipette. The plate was mixed for 1 min and samples were transferred onto the wells of an SLE plate (SLE+, Biotage, Uppsala, Sweden). A vacuum was applied to draw the entire sample into the matrix of the SLE plate. A fresh 96-well plate was placed under the SLE plate. Following an incubation of the plate at room temperature for 20 min, 1 mL dichloromethane (Chromasolv Plus, Sigma Aldrich, Poole, UK) was pipetted onto each well of the SLE plate and allowed to pass through to the collection plate until no residual dichloromethane was visible on the wells. A vacuum was subsequently applied for 30 s to transfer the remaining solvent into the collection plate. The dichloromethane was evaporated to dryness using a Vacmaster 96 (Biotage, Uppsala, Sweden), and samples were reconstituted in 40% (v/v) ultrapure methanol in ultrapure Milli-Q water (Merck-Millipore, Hertfordshire, UK).
Liquid chromatography
Chromatography was performed on an Acquity ultra high pressure liquid chromatography system (UPLC; Waters, Manchester, UK) using a 100 × 3 mm 2.6 μm Kinetex® C8 (Phenomenex, Torrance, USA) column at 45°C. Twenty microliters of sample were injected. Mobile phase A consisted of distilled Milli-Q water with 0.1% (v/v) formic acid and 2 mmol/L ammonium acetate, mobile phase B was ultrapure methanol with 0.1% formic acid and 2 mmol/L ammonium acetate. A linear gradient from 85% to 95% of mobile phase B over 3 min at a flow rate of 0.45 mL/min was applied followed by 1 min at 100% B with a flow rate of 0.6 mL/min before returning to starting conditions for a further minute.
Mass spectrometry
The LC eluate was injected into a XEVO™ TQ-S tandem mass spectrometer (Waters, Manchester, UK), which was operated in positive ion mode. The capillary was maintained at 0.5 kV, and the source temperature was 150°C. Desolvation temperature and gas flow were 600°C and 800 L/h. The source offset was maintained at 30 V. Cone and collisions energies were 25 V and 18 eV. The transitions for progesterone were m/z 314.8 > 97.1 and 314.8 > 109.1 (quantifier and qualifier transitions, respectively). The IS D9-progesterone was monitored at the transition m/z 323.8 > 100.1. Multiple reaction monitoring mode was used with a dwell time of 0.059 s. MassLynx NT 4.1 software was used for system control and the MassLynx TargetLynx program for data processing. Peak area ratios of analyte and IS, 1/x weighting and linear least square regression were used to produce the standard curve.
Validation
We undertook method validation according to accepted guidelines. 29
Ion suppression
Six different saliva samples or distilled water were extracted by SLE as described above. Dried extracts were reconstituted in 100 μL of 40% methanol and spiked with 10 μL of progesterone in methanol to final concentrations of 800 and 3200 pmol/L. As a background control to determine the signal from the non-spiked sample, a corresponding volume of water was added to the reconstituted extract of saliva samples or water. Ion suppression was calculated from the peak area of progesterone determined in extracts of saliva compared with peak area determined in the extract of water after deducting the respective background signal: 100−[(areaH2O−areasaliva)/areaH2O]×100.
Recovery
A total of 490 μL of saliva or 50% (v/v) methanol were spiked with 10 μL of a progesterone standard in methanol yielding final concentrations of 80, 800 and 3200 pmol/L progesterone and a final methanol concentration of 2% in the sample. Five different saliva samples were tested. As a background control, 10 μL of water was added to 490 μL of saliva samples or 50% (v/v) methanol. The spiked samples (300 μL) were subsequently extracted and analysed as described above. The recovery was calculated from the concentrations measured in saliva compared with the concentrations measured in 50% (v/v) methanol deducting the respective background control. The presented values for recovery thus include extraction efficiency and matrix effects.
Specificity
Solutions of common endogenous and synthetic steroids were prepared in 50% (v/v) methanol in distilled water at supraphysiological concentrations and analysed for interference in the progesterone and D9-progesterone channels at the relevant retention time. 21-deoxycortisol, aldosterone, androstenedione, corticosterone, cortisol, cortisone, DHEA, dexamethasone, testosterone and pregnenolone were tested to a final concentration of 100 nmol/L. DHEAS (10 μmol/L), budesonide (232 nmol/L), fludrocortisone (263 nmol/L), fludrocortisone acetate (234 nmol/L) fluocinolone (243 nmol/L), triamcinolone (254 nmol/L), triamcinolone acetonide (230 nmol/L), prednisolone (278 nmol/L) and prednisone (279 nmol/L), 17-hydroxyprogesterone (1500 pmol/L), 21-hydroxyprogesterone (1500 pmol/L), epitestosterone (0.1 nmol/L), beclomethasone (96 nmol/L), 11-deoxycortisol (172 pmol/L), dihydrotestosterone (40 pmol/L), cyproterone acetate (48 nmol/L), levonorgestrel (9.6 μmol/L), desogestrel (24 μmol/L), norethisterone (1.2 mmol/L) and ethinylestradiol (34 μmol/L) were also tested. These concentrations are all in excess of those that would be expected to be seen in saliva.
Dilution linearity
Three different saliva samples were spiked with 10,000 pmol/L progesterone and serially diluted in six steps with PBS/BSA to 156.25 pmol/L. Samples were collected from men and from women on oral contraceptives and thus had negligible progesterone concentrations. Measured concentrations were plotted against calculated theoretical concentrations, and curves were judged linear if the correlation coefficient (R2) after linear regression was higher than 0.9900.
Imprecision and bias
Imprecision was assessed for different concentrations in PBS/BSA- or saliva-based samples. The same PBS/BSA QC samples (100, 750, 1500 and 3000 pmol/L) were analysed in 10 different runs to assess inter-assay imprecision as well as 10 times within the same run to determine intra-assay imprecision. Additionally, saliva samples spiked with three different concentrations (100, 1500, 3000 pmol/L) were tested for intra-assay imprecision; saliva samples for this experiment were collected from men and women taking an oral contraceptive, who have negligible progesterone concentrations. The bias was calculated as percentage deviation between the mean observed and nominal concentrations. The percentage coefficient of variation (CV) was calculated as ratio of standard deviation and mean.
Limit of quantification
Ten replicates of low concentrations of progesterone in PBS/BSA were analysed in one assay. The lower limit of quantification (LLOQ) was defined as the concentration for which a CV and bias of less than 20% were determined.
Post-extraction stability
Saliva samples (n = 24) were extracted and reconstituted for analysis. The plate was measured immediately after extraction, subsequently stored at 4°C for 72 h and the measurement was repeated. Percentage bias between the results of the two measurements was calculated.
Carry-over
A PBS/BSA sample containing 100,000 pmol/L progesterone was analysed followed by sample containing only PBS/BSA. Percentage carry-over of progesterone into the following sample was calculated from the progesterone peak area determined for the two samples.
Results
Analytical performance
Progesterone and D9-progesterone had retention times of 1.32 and 1.31 min, respectively, producing distinct, identifiable peaks. Extracts of saliva samples showed no interfering peaks when using the progesterone quantifier transition of m/z 314.8 > 97.1 (qualifier transition 314.8 > 109.1) (Figure 1). Total run time was 5.5 min from injection to injection. All validation criteria are summarized in Table 1. The mean peak area of progesterone spiked into a reconstituted extract of saliva compared with the spiked extract of water was 93.2 ± 8.1% (n = 6 for two concentrations), indicating no significant ion suppression by matrix compounds. Therefore, the percentage signal for different saliva samples ranged between 79.6% and 110% for 800 pmol/L and between 80.3% and 98.6% for 3200 pmol/L, demonstrating no significant differences between saliva samples from different individuals. The mean recoveries were 92.4 ± 3.1%, 89.9 ± 2.7% and 90.6 ± 2.8% after the addition of 80, 800 and 3200 pmol/L progesterone to saliva (n = 5), respectively. No interference with a variety of natural and synthetic steroids with structural similarity to progesterone was observed. A standard curve was obtained by plotting the progesterone/D9-progesterone peak area ratio against the nominal progesterone concentrations. The curve was linear over the standard range (0–10,000 pmol/L), reproducible between assays and showed correlation with the assigned concentrations with R2 values >0.99. Serial dilutions of saliva samples after the addition of 10,000 pmol/L progesterone with PBS/BSA showed linearity with a mean R2 of 0.997 ± 0.001 (n = 3), when the measured concentrations were plotted against the calculated, theoretical concentrations. However, a bias between measured and theoretical concentrations of <40% was observed.
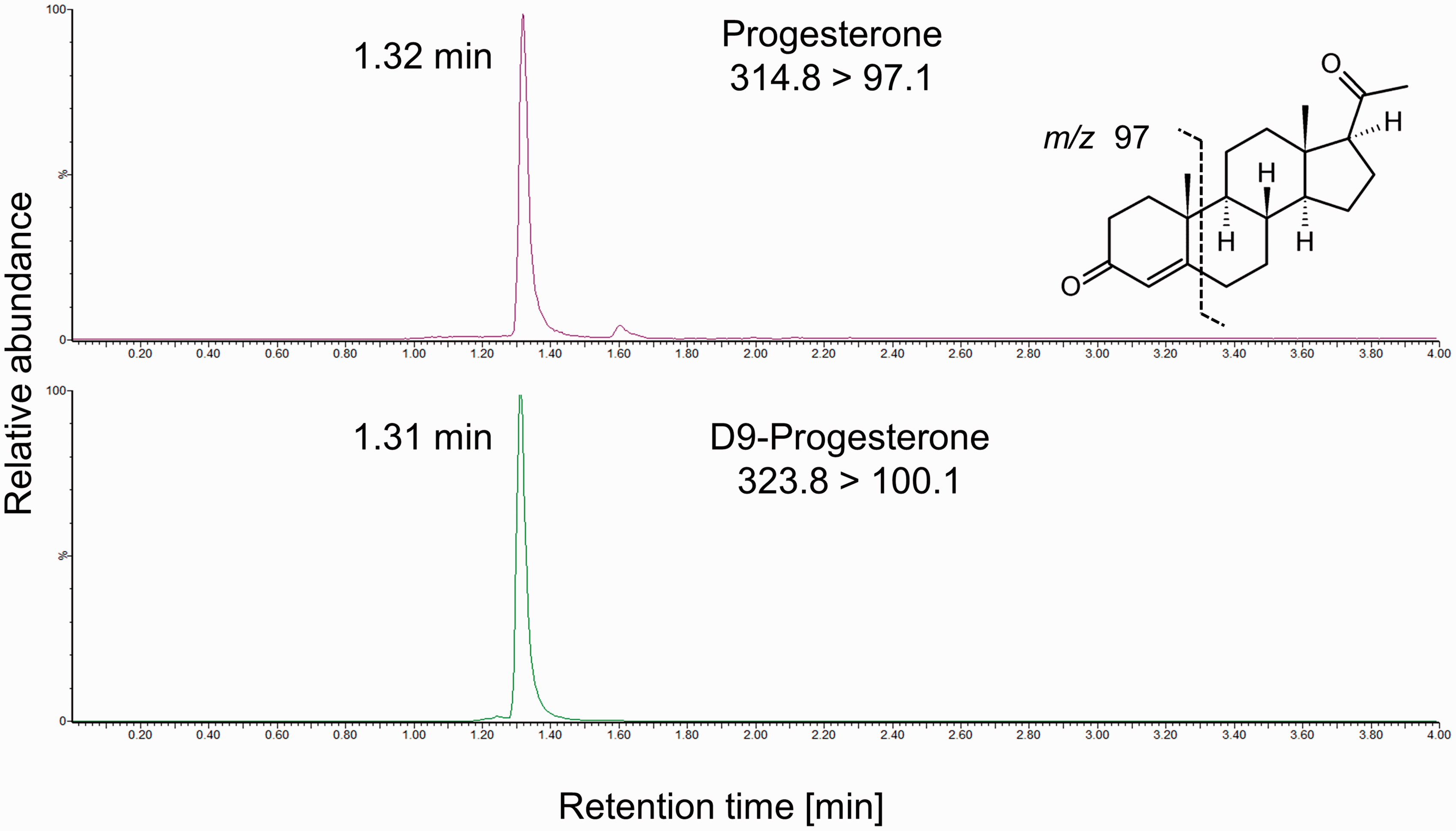
Chromatogram of a saliva sample containing progesterone. Progesterone (upper panel) and the internal standard D9-progesterone (lower panel) co-elute as distinct peaks with no interfering signals. Quantifier transitions are shown and the fragmentation is indicated.
Summary of the analytical performance validation.
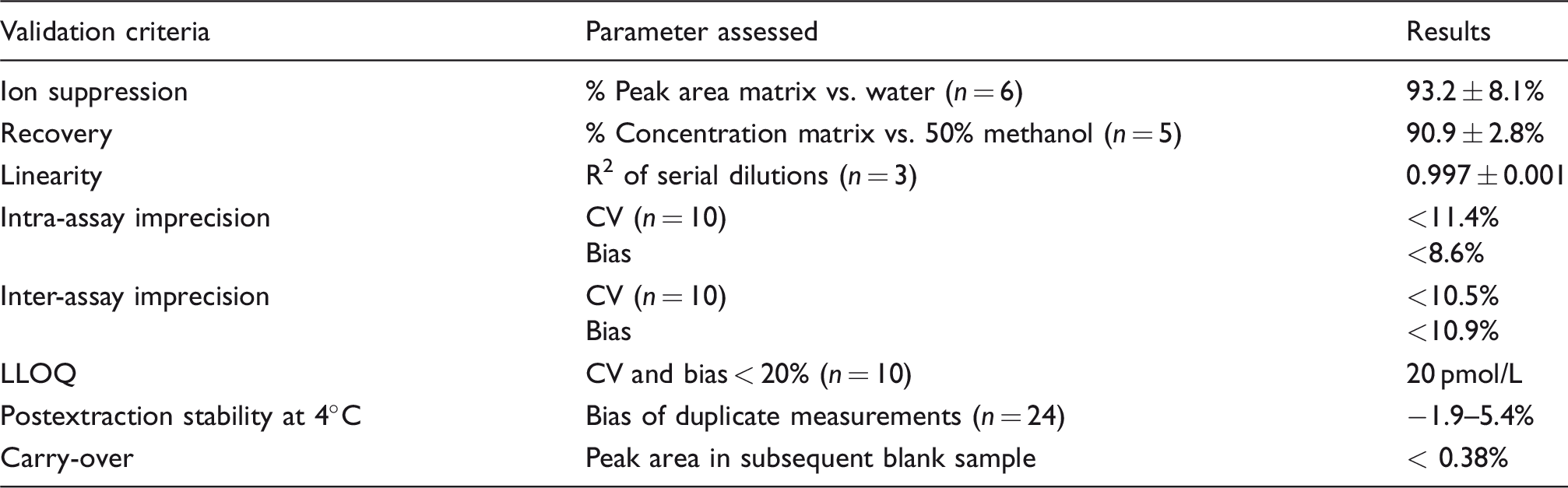
Note: Criteria were assessed at three different concentrations within the standard curve (80, 800, 3200 pmol/L).
LLOQ: lower limit of quantification.
Inter-assay imprecision assessment for PBS/BSA-based QCs at concentrations of 100, 750, 1500 and 3000 pmol/L gave a bias of 10.9, −0.8, 5.7 and 5.0%, respectively and a CV of 10.5, 3.2, 9.9, 3.2%, respectively (n = 10). In an intra-assay experiment for the PBS/BSA-based QCs at the same concentrations, a respective bias of 3.4, 3.6, 6.9 and 8.6% and respective CVs of 11.4, 3.4, 2.6 and 5.1% were determined (n = 10 for 100 and 1500 pmol/L; n = 9 for 3000 pmol/L). The analysis of saliva spiked with 100, 1500 and 3000 pmol/L progesterone showed an intra-assay CV of 9.3, 1.7 and 9.3%, respectively. The LLOQ was determined as 20 pmol/L, which corresponds to 0.4 pg or 1.2 fmol on the column, giving a bias of 11.5% and a CV of 6.8% (n = 10), which is below the defined cut-off of 20%. After reconstitution in 40% (v/v) methanol, the extract was stable for at least 72 h at 4°C. The percentage bias between the results before and after the storage was between −1.9 and 5.4%, which is below the inter-assay variation. The percentage carry-over of progesterone from a sample containing 100,000 pmol/L to the following blank sample was less than 0.38%.
Salivary progesterone concentrations during the menstrual cycle
We have applied the assay for preliminary investigations of variations of morning salivary progesterone concentrations over the menstrual cycle (n = 9) and of diurnal variations during luteal phase (n = 7). Participating women were aged 26–39 and had a regular menstrual cycle. Saliva samples were collected each morning before 10:00 on days 1–7 and days 18–24 (luteal phase) of the cycle. During days 1–7, for all except four samples (collected by three different participants) concentrations were below the LLOQ (Figure 2(a)). Seven out of nine participants subsequently showed an increase of salivary progesterone from day 18 onwards and concentrations remained elevated for several days. Maximum concentration reached by the individual participants ranged between 155 and 526 pmol/L. In four out of seven women with an increase of salivary progesterone during luteal phase, the concentrations were going down towards baseline again on day 24, with two of them reaching concentrations below the LLOQ. For two participants, concentrations of morning salivary progesterone remained below the LLOQ for the entire time monitored (Figure 2(a)).
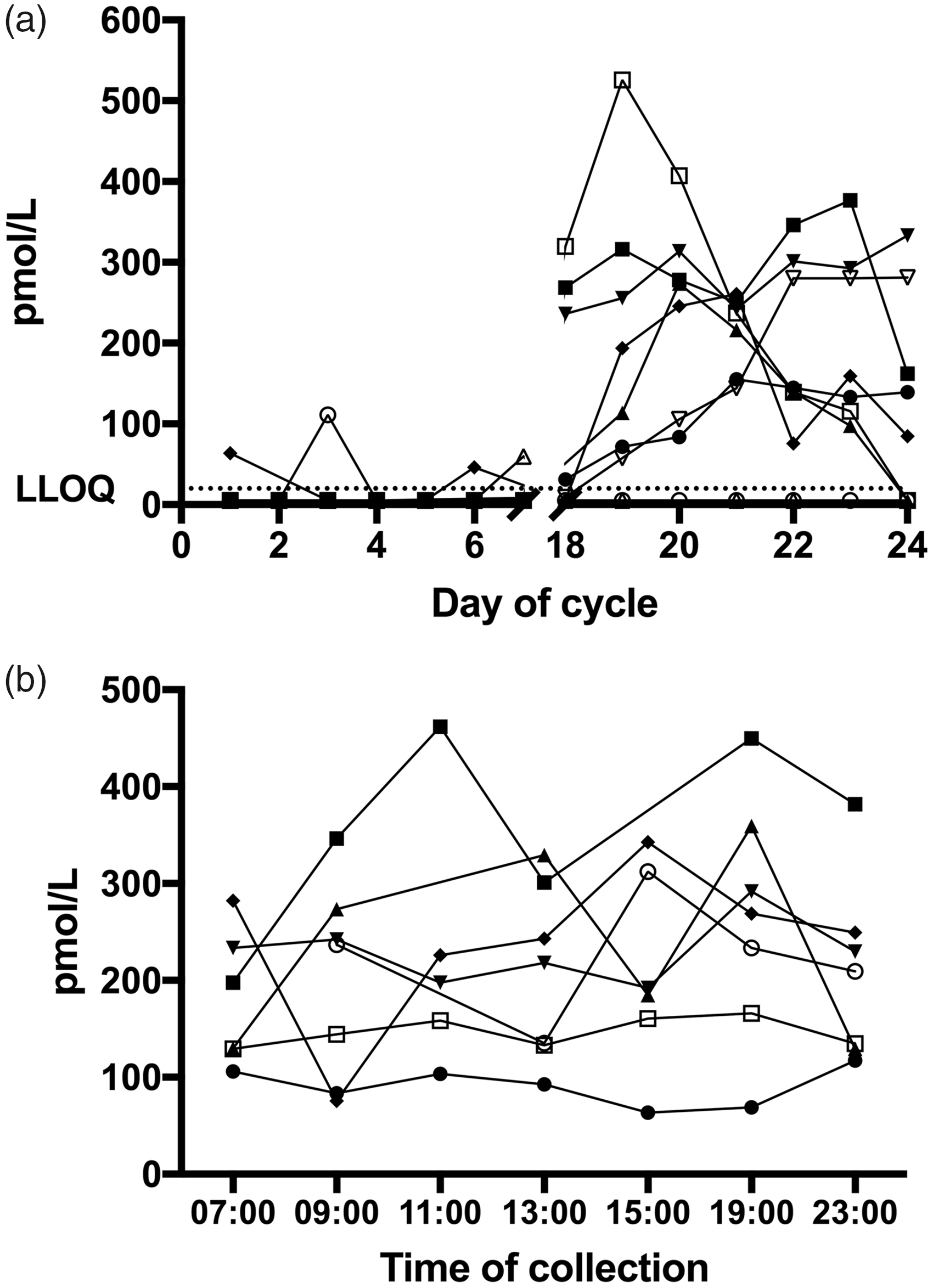
Menstrual and diurnal variations of salivary progesterone in women with a regular menstrual cycle. (a) Variations of morning salivary progesterone over the menstrual cycle (n = 9). Samples were collected daily on days 1–7 and 18–24 between awakening and 10:00. (b) Diurnal variation of salivary progesterone during luteal phase (days 19–22) in women with a luteal progesterone surge (n = 7). Samples were collected within ±30 min of the indicated time points.
Additionally, diurnal profiles composed of seven time points (Figure 2(b)) during luteal phase were established for seven women who showed the characteristic luteal phase surge of progesterone. A tendency for an increase over the morning hours after awakening followed by a decrease in the afternoon can be observed.
Discussion
We have developed a novel direct assay for rapid high-throughput measurement of salivary progesterone by LC-MS/MS. The single-step extraction using SLE in combination with the short chromatography renders the assay suitable for routine applications with appropriate throughput as opposed to previously published assays. 23 This is essential for multisample measurements that are required to monitor progesterone concentrations during pregnancy and patients undergoing diagnostic assessment or therapy for desired fertility. The use of saliva as matrix for hormone quantification can be limited by the low hormone concentrations present in saliva and by the limited possibility to standardize sample collection, as the collection is carried out by patients on their own. However, LC-MS/MS provides the sensitivity and specificity to overcome these limitations.21,22
We performed a strict validation of the developed assay that guarantees its accurate, precise and reproducible performance. The assay was demonstrated to be highly selective and no interference with other common natural steroids and steroidal drugs was detected. There is no significant ion suppression and the assay demonstrates excellent recovery, intra- and inter-assay precision and linearity. The high bias between measured and theoretical concentrations when diluting samples suggests that dilution of a sample with PBS/BSA prior to extraction cannot be applied. However, given that the highest concentrations measured in women during the luteal phase of their menstrual cycle (Figure 2) are within the linear range of our standard curve, dilutions are generally not required. The sample remains stable after extraction and reconstitution, which facilitates its implementation in the workflows of a clinical laboratory.
We have successfully applied the LC-MS/MS assay to measure salivary progesterone concentrations in nine healthy female volunteers during their physiological menstrual cycle (Figure 2). Seven out of nine participants showed the expected increase in progesterone concentrations during the luteal phase indicative of ovulation at mid-cycle. 1 The determined maximum concentrations of up to over 500 pmol/L confirmed the correct choice of the working standard concentrations. The assay allows for the monitoring of physiological alterations of progesterone concentrations using a non-invasive sampling technique and high-throughput analysis demonstrating clinical applicability for long-term monitoring. The tendency for an increase of salivary progesterone in the morning hours (Figure 2(b)) is consistent with the awakening response for progesterone established for a large cohort of women using a radioimmunoassay for the measurement of salivary progesterone. 30 However, we would like to emphasize that, due to the small number of participants, the presented data of salivary progesterone concentrations for healthy women do not allow conclusions about reference ranges and natural variations.
The presented LC-MS/MS method allows the robust quantification of salivary progesterone without the risk of interference with related compounds, as opposed to immunoassays. In combination with the self-administered sampling of saliva and the high throughput of the assay, this gives the opportunity to establish reliable reference ranges from large populations to assess luteal and placental functions. In addition, the availability of this high-throughput salivary progesterone assay will help to explore the role of progesterone in disorders of steroidogenesis with increased progesterone concentrations, specifically the monitoring of steroid replacement therapy in congenital adrenal hyperplasia due to 21-hydroxylase deficiency, in whom increased serum progesterone indicates insufficient replacement,31,32 with increased progesterone often adversely impacting the ability of these patients to conceive. 33
Footnotes
Acknowledgements
The authors would like to thank the participants for sample collection.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Ethical approval
The study had full ethical approval from the Science, Technology, Engineering and Mathematics Ethical Review Committee of the University of Birmingham (ERN_17–0494).
Guarantor
BGK.
Contributorship
LS and JEA contributed equally to this work. The development and validation of the method described was performed by LS, JEA and BGK. The collection of saliva samples from healthy volunteers was done by ESB, LS and WA. All authors contributed to writing the manuscript.
