Abstract
Background
Direct-reacting bilirubin concentrations measured using vanadate chemical oxidation method do not exactly match the conjugated bilirubin concentration. One of the causes is the effect of bilirubin photoisomers. However, the quantitative evaluation of the effects of these photoisomers has not been sufficiently conducted. In particular, the influence of bilirubin configurational isomers on direct bilirubin is the most critical factor.
Methods
Sixteen residual serum samples were used. For quantitative analysis based on the change in direct bilirubin and bilirubin configurational isomer, samples were irradiated via blue light-emitting diodes to suppress the production of bilirubin structural isomers. Total bilirubin and direct bilirubin concentrations were measured using the vanadate chemical oxidation method. Concentrations of 4Z,15Z-bilirubin IXα and its photoisomers were measured using high-performance liquid chromatography. The sum of 4Z,15E-bilirubin IXα and 4E,15Z-bilirubin IXα was notated as bilirubin configurational isomer, and the differences between the measured values of the irradiated and non-irradiated samples were calculated and notated as ΔDB and ΔBCI.
Results
In non-irradiated and irradiated samples, total bilirubin and direct bilirubin concentrations were 10.73 mg/dL with significant a decrease to 10.60 mg/dL and 0.69 mg/dL with a significant increase to 0.78 mg/dL, while bilirubin configurational isomer values were 1.00 mg/dL and 1.52 mg/dL, respectively. The linear regression equation revealed a significant positive correlation of Y = 0.187X−0.006 between ΔDB (Y) and ΔBCI (X).
Conclusion
Applying the vanadate chemical oxidation method affected approximately 19% of the bilirubin configurational isomer concentration for direct bilirubin. Extreme caution is necessary when interpreting the measured values of samples indicative of unconjugated hyperbilirubinaemia.
Introduction
Direct-reacting bilirubin (DB) is a representative substance measured by clinical testing using the diazo method, 1 and is also detected in subsequent measurement of the bilirubin fraction via enzymatic methods 2 , 3 or the chemical oxidation method. 4 In clinical settings, DB is considered conjugated bilirubin; however, the amount of DB measured using these methods does not precisely match the concentration of conjugated bilirubin owing to the presence of delta bilirubin 5 and bilirubin photoisomers. 6
Bilirubin photoisomers are generated through the irradiation of 4Z,15Z-bilirubin IXα ((
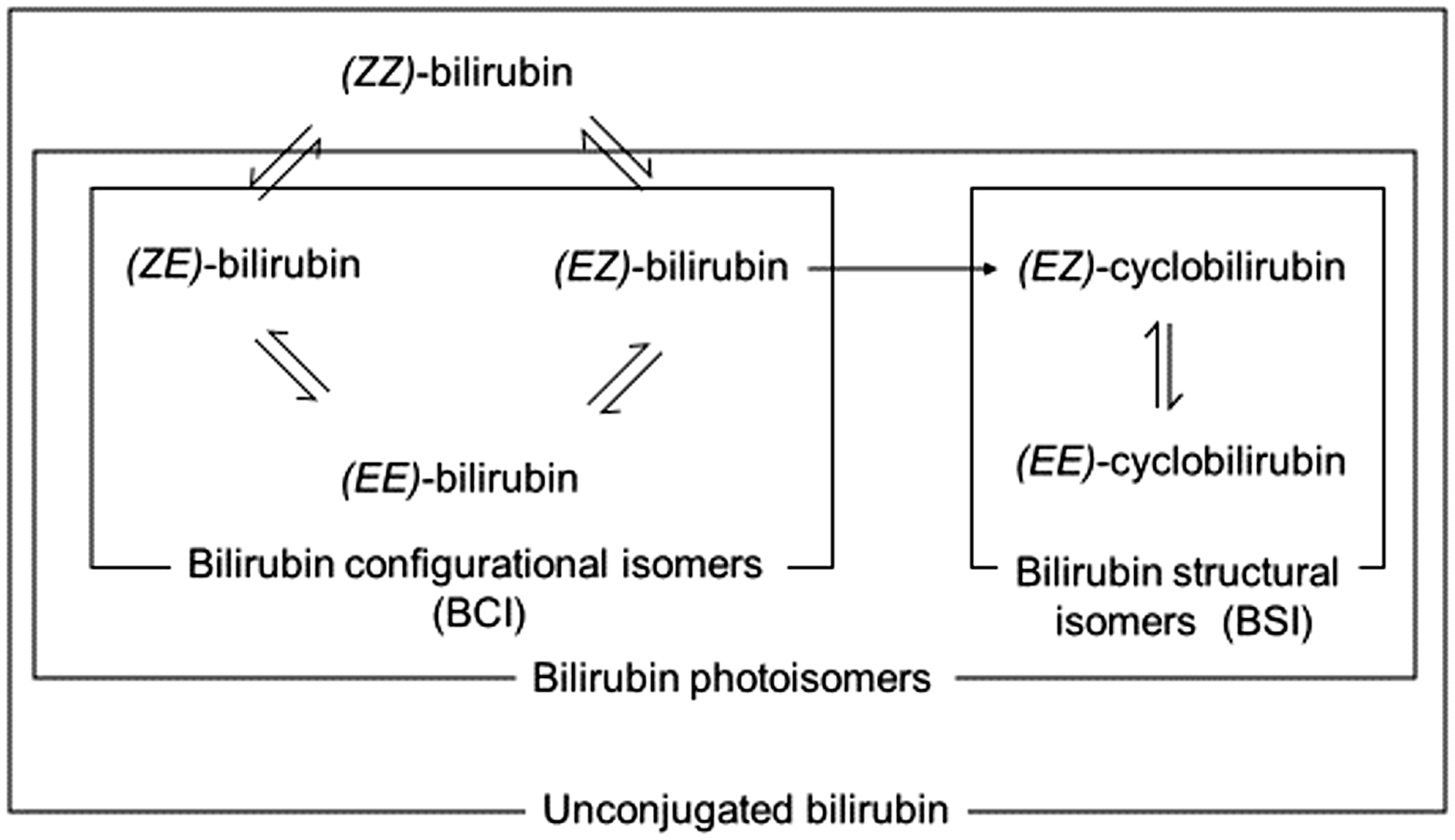
Illustration of the bilirubin photochemical reaction. (
Because humans are exposed to environmental light, these bilirubin photoisomers are always present in the human blood serum. BCI constitute approximately 10–20% of (
Vanadate chemical oxidation, a DB measurement method used in clinical laboratories, requires sodium metavanadate as an oxidizing agent. 4 Currently, this method is used in over half of all the testing facilities in Japan. 15 We previously reported that all bilirubin photoisomers in the human serum albumin-bilirubin solutions act as substrates upon vanadate addition, 16 resulting in an increased DB concentration measurement using this method. However, the proportion of each reacting bilirubin photoisomer that contributes to the altered DB amount is not the same because each isomer exhibits a different molar absorptivity. Therefore, a quantitative impact assessment is still needed.
Accordingly, in this study, we report our findings regarding the quantitative effects of BCI on DB by employing the chemical oxidation method using vanadic acid.
Materials and methods
Principles of analysis and irradiation conditions
Quantitative analysis of the effects of BCI on DB concentration was performed by comparing the increase in BCI and changes in DB concentration in light-irradiated samples. The change in the DB concentration was calculated from the decrease in the absorbance of BCI and BSI that reacted with vanadate. Because both BCI and BSI are produced when bilirubin is exposed to light, it was necessary to minimize the increase in BSI. The change in the DB concentration due to the reaction of BSI with vanadate did not exceed that in BSI alone because (
The DB concentration used in the clinical setting is expressed in mg/dL up to two decimal places, and clinical judgments are made with a value up to one decimal place or more. Therefore, we set the change in BCI concentration to 0.2 mg/dL or more and the change in BSI concentration to below 0.05 mg/dL for quantitative analysis. The criteria for BCI production included production of a constant ratio to (
Sample collection and preparation
Residual serum samples obtained from 16 newborn infants born at Kagawa University Hospital, in conjunction with blood tests performed as part of routine medical treatment, were used as samples, and were collected between April 2018 and March 2020. The study was approved by the Institutional Ethics Committee of Kagawa University Heisei (approval number 30–188). Samples were protected from light and were frozen. Before use, samples were thawed in the dark and divided into 200-μL aliquots; nonirradiated samples were designated as Group A and irradiated samples as Group B.
Bilirubin measurement
Total bilirubin (TB), DB, (
The following bilirubin fractions were evaluated: the sum of (
Measurement of DB using the vanadate chemical oxidation method
After mixing 10 µL of the sample with 280 μL tartrate buffer (pH 2.9) and incubating, absorbance was measured at 450 nm. Next, 70 μL sodium metavanadate solution was dissolved in a phosphate buffer (pH 7.0). After the reaction, the absorbance was measured at 450 nm, and DB concentrations were calculated based on the decrease in absorbance.
Statistical analysis
Paired
Results
The bilirubin fractions in Groups A and B, and changes in the concentrations of various bilirubin photoisomers are shown in Table 1.
Changes in TB, DB and bilirubin photoisomer concentrations before and after light irradiation.
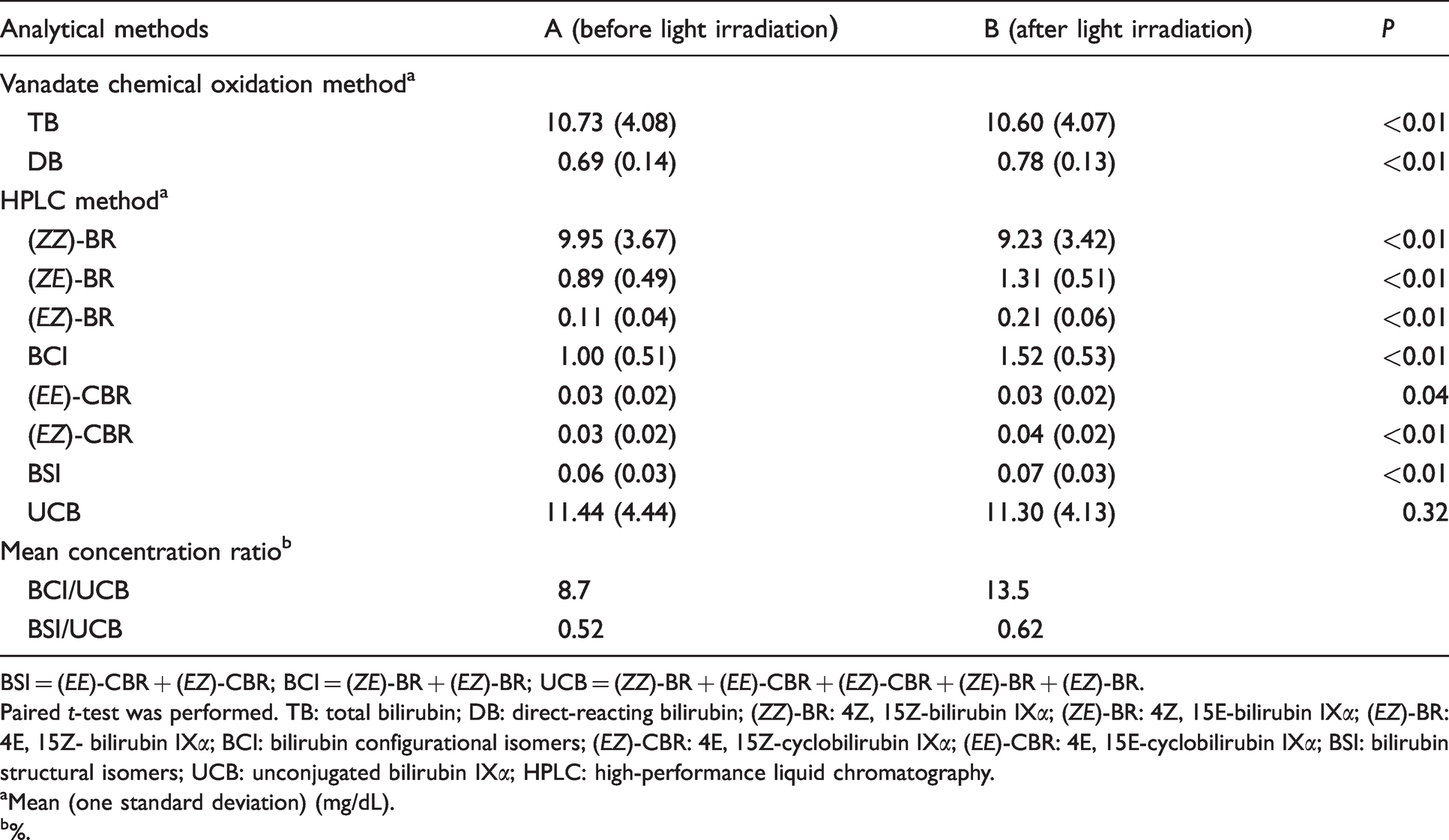
BSI = (
Paired
aMean (one standard deviation) (mg/dL).
b%.
The TB concentration (mean (1 standard deviation)) was significantly higher in Group A (10.73 (4.08) mg/dL) than in Group B (10.60 (4.07) mg/dL;
The mean (range) differences in the bilirubin photoisomer groups were as follows: (
The mean (range) differences in the bilirubin photoisomer groups.
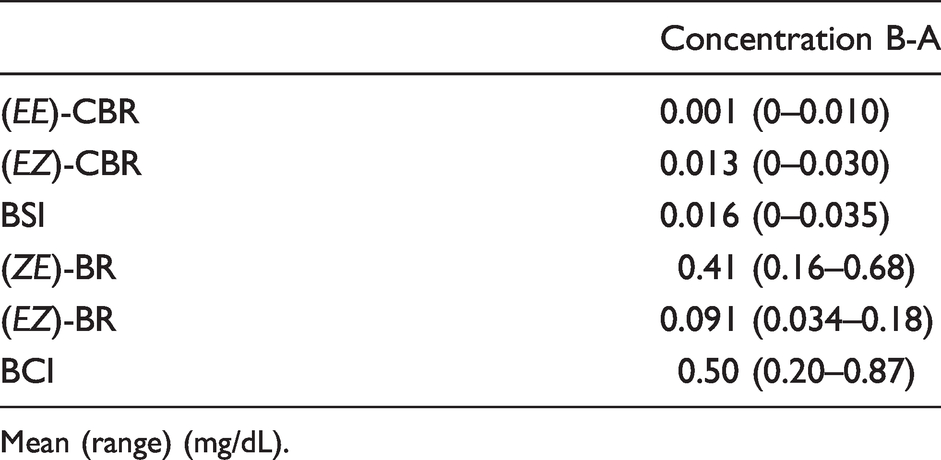
Mean (range) (mg/dL).
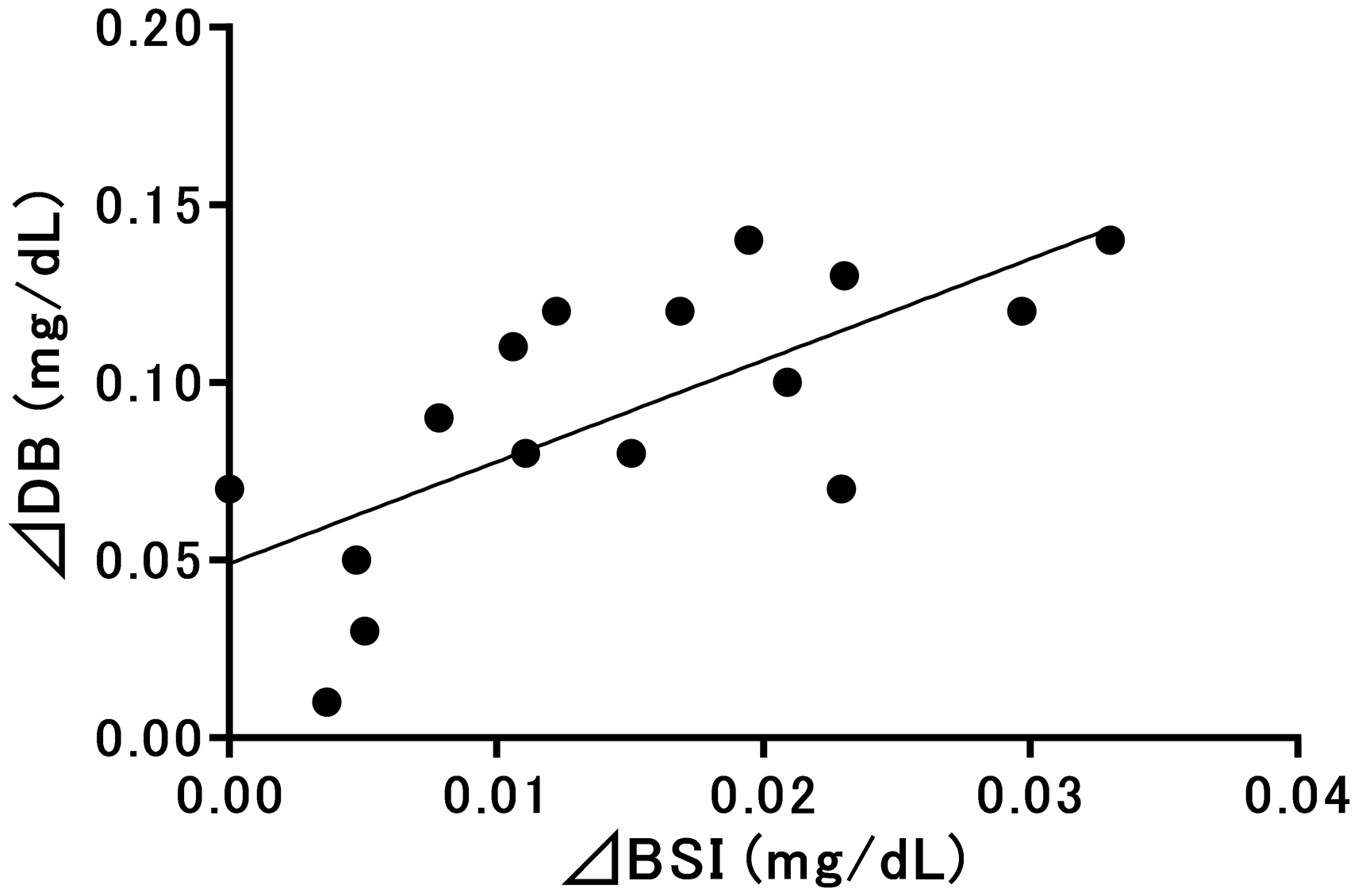
Figure 2 shows the correlations between ΔDB and ΔBCI. The linear regression equation showed a significant positive correlation of Y = 0.187X–0.006 between ΔDB and ΔBCI (R2 = 0.76,
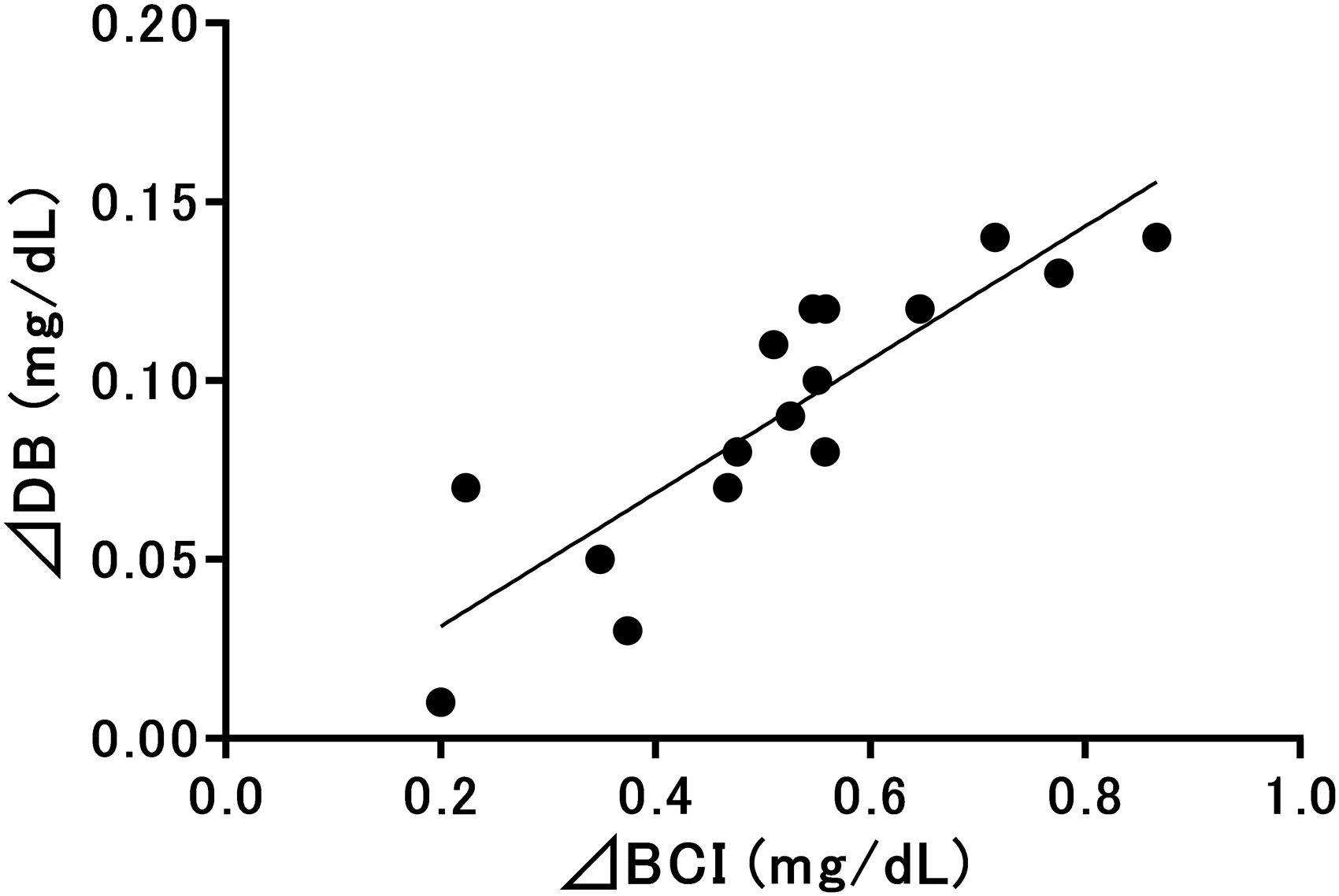
Relationships between direct-reacting bilirubin and bilirubin configurational isomers evaluated using 16 specimens. DB: direct-reacting bilirubin; BCI: bilirubin configurational isomer; ΔDB (Y) and ΔBCI (X). Y = 0.187X−0.006 (R2 = 0.76,
Relationships between direct-reacting bilirubin and bilirubin structural isomers evaluated using 16 specimens. DB: direct-reacting bilirubin; BSI: bilirubin structural isomer; ΔDB (Y) and ΔBSI (X). Y = 2.858X + 0.049 (R2 = 0.50,
Discussion
DB measurement is a clinical test that remains difficult to standardize. Therefore, it is important to elucidate the factors that influence this measurement. In particular, it is necessary to investigate the effects of BCI, which are abundant
In this study, we investigated the effects of vanadate chemical oxidation on TB and DB concentrations by selectively increasing the proportion of BCI in bilirubin photoisomers using brief light irradiation. TB values were significantly decreased after irradiation. In contrast, UCB concentrations did not exhibit a significant change. The relative molar extinction coefficients (455 nm) of (
DB concentrations significantly increased in specimens following light irradiation. Conjugated bilirubin is present in trace quantities in the serum of healthy newborn infants 12 and is not significantly changed by light exposure. In addition, because no substances other than bilirubin exhibit absorbance at 450 nm or show substantial changes in concentration owing to light irradiation, these changes were attributed to the increased BCI concentrations in the samples.
In this study, BCI and BSI concentrations significantly increased upon light irradiation. BCI and BSI act as substrates in vanadate chemical oxidation. 16 The mean BCI to UCB ratio increased by 4.8% after light irradiation, and the mean concentration of BCI increased by 0.52 mg/dL. At the same time, a 0.09-mg/dL increase in DB concentrations was observed. Linear regression analysis revealed a positive correlation between ΔDB and ΔBCI, and as the intercept was close to 0 and the slope was 0.187 in the approximate linear equation, DB concentrations were determined to have increased by approximately 0.19 mg/dL as BCI concentrations increased by 1 mg/dL. As this relationship was consistent across various samples, including samples from Group A, DB concentrations accounted for approximately 19% of the BCI concentration in the sample. Regarding the effects of BSI on DB in the current study, the mean BSI to UCB ratio increased by 0.10%, and the mean concentration of BSI increased by 0.01 mg/dL. No reports have described the quantitative effects of BSI on DB values using this method; however, the mean DB value representing BSI was thought to be less than 0.01 mg/dL owing to the relationship between the molar absorptivities of the calibrator and BSI.
Among bilirubin photoisomers, (
In the diazo method, as well as the bilirubin oxidase method, DB is affected by BCI; however, few studies have described the quantitative effects of BCI on these methods, 6 and it is therefore not possible to discuss the differences between reagents. However, in all methods, DB values in serum containing large amounts of BCI, such as the neonatal serum, may be overestimated.
Analysis of the effects of BSI on DB values revealed a correlation between elevated BSI and DB concentrations in serum samples exposed to environmental light over a 24-h period. In contrast, in the diazo method, BSI did not affect DB measurement. 6 BSI are produced and accumulate because of long-term exposure to room lighting after serum separation. 13 Thus, this method also had some problems regarding the effects of BSI, and detailed quantitative studies are needed in the future.
Conclusion
In this study, we demonstrated that the presence of BCI could affect the measurement of DB using vanadate chemical oxidation. The vanadate chemical oxidation method is an excellent method to measure bilirubin. However, caution is necessary when interpreting the measured values in samples from individuals with unconjugated hyperbilirubinaemia.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research was funded by KAKENHI (16K10093, 17K10179).
Ethical approval
Ethics Committee Kagawa University Heisei 30–188.
Guarantor
TK.
Contributorship
SK, KK, HO, SI, TK researched literature and conceived the study, and were involved in protocol development. SK, KK collected samples. SK, KK, HO, MO, TA measured bilirubin fractions. SK wrote the first draft of the manuscript. TI, HO, TK, KM worked to obtain ethical approval. All authors reviewed and edited the manuscript and approved the final manuscript.
