Abstract
Objective
Investigation of the reactivity of fractions of bilirubin photoisomers with the vanadic acid oxidation method.
Methods
Bilirubin photoisomers were prepared by irradiating a bilirubin/human serum albumin solution with blue light emitting diode. Direct bilirubin and bilirubin fractions were measured using the vanadic acid oxidation method and high-performance liquid chromatography in the sample before and after irradiation.
Results
Direct bilirubin was increased in the solution containing bilirubin photoisomers. (EE)-/(EZ)-cyclobilirubin-IXα and (ZE)-/(EZ)-bilirubin-IXα completely disappeared after the addition of vanadic acid.
Conclusion
Bilirubin photoisomers reacted as direct bilirubin in the vanadic acid oxidation method.
Introduction
The vanadic acid oxidation method for the evaluation of serum direct bilirubin (DB) has recently been developed as a substitute for the diazo method 1 and is now widely used clinically in Japan. Since it is difficult to standardize DB, it is important to find substances that affect each measurement methods. The reactivity of δ-bilirubin as DB is comparable to that of diazo method, 1 but the effect of bilirubin photoisomers is currently unclear in the vanadic acid oxidation method.
Bilirubin photoisomers are generated from 4Z,15Z-biliruibin-IXα ((ZZ)-bilirubin-IXα) by light exposure. They include (ZE)-/(EZ)-bilirubin-IXα, (EZ)-/(EE)-cyclobilirubin-IXα; the relative molar extinction coefficients for (ZZ)-bilirubin-IXα at 455 nm are 0.81, 0.54, 0.47 and 0.39, respectively. 2 In the serum of unconjugated hyperbilirubinaemia, the percentage of (ZE)-bilirubin-IXα is approximately 10% of (ZZ)-bilirubin-IXα, and (EZ)-cyclobilirubin-IXα is under 1% even if during phototherapy in hyperbilirubinimic neonate. 3 In the present study, we investigated the reactivity of bilirubin photoisomers with the vanadic acid oxidation method.
Methods
Bilirubin (Tokyo Chemical Industry Co., Ltd, Tokyo, Japan), human serum albumin (HSA) (Sigma Aldrich Inc., St Louis, USA), HPLC-grade distilled water, and HPLC-grade acetonitrile were used as reagents.
Bilirubin solution dissolved in 0.1 N NaOH solution was mixed with HSA solution adjusted to 3 g/dL with 0.1 M phosphate buffer (pH 7.4) in order to prepare bilirubin/HSA complex solution with a bilirubin concentration of 10 mg/dL. This solution was irradiated with blue light emitting diode (Atom Bili-Therapy Pad type, ATOM Medical, Tokyo, Japan) for 60 min in order to prepare a sample containing bilirubin photoisomers. The amount of light energy was measured using Minolta Photometer M451 (Minolta Air-Shields) and was 44.3 μW/cm2/nm.
Direct Bilirubin E-HA Test Wako (Wako Co., Ltd, Osaka, Japan) was used as the DB measurement kit. The buffer attached to the measurement kit was tartrate buffer (pH 2.9), and vanadic acid solution was a mixture solution of phosphate buffer (pH 7.0) and sodium metavanadate. Ten microlitres of the sample and 280 μL of the buffer attached to the measurement kit were mixed, incubated at 37℃ for 5 min, and absorbance was measured as a sample blank (point A). Seventy microlitres of vanadic acid solution was then added, and the absorbance was measured after the reaction for 5 min at a main wavelength of 450 nm and subwavelength of 546 nm (point B). Using these values, the DB concentrations were assessed from a calibration curve prepared using the absorbance of samples for calibration. DB was measured by the central laboratories in our hospital.
Bilirubin fractions were measured using high-performance liquid chromatography (HPLC). Shimadzu LC-9A (Shimadzu Co., Kyoto, Japan) was used for HPLC. Nova-Pack C18 and a radial compression cartridge 5 mm × 10 cm (Nihon Waters KK, Tokyo Japan) were used as the column. A SPD-6AV detector (Shimadzu Co., Kyoto, Japan) was used as the measurement device. The measurement wavelength was 455 nm. Measurements were performed based on the HPLC method reported by Itoh et al.: 2 after designating mixtures of acetonitrile: 0.01 M phosphate buffer (pH 5.5): dimethyl formaldehyde = 5:30:65 (v/v/v) and 20:15:65 (v/v/v) as the first and second solutions, respectively, the solution was flowed at 1 mL/min employing the gradient method, changing the ratio of the second solution from 0 to 34% over 17 min and to 100% for the subsequent 8 min. Each bilirubin photoisomer was isolated and quantitated after corrections with the molecular absorbance ratio. The sample, DMSO and acetonitrile were mixed at the same volume ratio, centrifuged and deproteinized, and 25 µL of the supernatant was injected into HPLC.
DB and bilirubin fractions from bilirubin/HSA solution before and after irradiation were measured, and the measurement was repeated for 10 times.
Results
The DB level (mean (standard deviation)) in the sample increased by 1.12 mg/dL (0.15) after irradiation from that before irradiation.
In HPLC, only (ZZ)-bilirubin-XIIIα, -IXα and -IIIα were eluted in sample before irradiation at points A and B. Figure 1 shows the chromatogram obtained at points A and B in sample after irradiation. Bilirubin fractions and the mean (standard deviation) percentage of total area under the curve were eluted after the addition of buffer in the order of (EE)-cyclobilirubin-IXα, 2.2 (1.0)%, (EZ)-cyclobilirubin-XIIIα, 6.2 (0.5)%, (EZ)-cyclobilirubin-IXα, 21.1 (8.0)%, (ZE)-bilirubin-IXα, 10.4 (2.2)%, (EZ)-bilirubin-IXα, 2.4 (0.6)%, (ZZ)-bilirubin-IXα, 47.0 (9.7)%, and (ZZ)-bilirubin-IIIα, 10.5 (0.5)%, at point A. Peaks other than those of (ZZ)-bilirubin-IXα and (ZZ)-bilirubin-IIIα completely disappeared at point B.
Changes in bilirubin fractions in the direct bilirubin measurement system using the vanadic acid oxidation method. (a) The chromatogram obtained after 5 min incubation mixing with 280 μL of buffer (sample is diluted 1/29), and (b) the chromatogram obtained after 5 min incubation with the addition of 70 μL of vanadic acid (sample is diluted 1/36). Direct bilirubin levels were assessed from changes in absorbance from (a) to (b). Numbers and names of peaks: 1. 4E, 15E-cyclobilirubin-IXα ((EE)-cyclobilirubin-IXα), 2. (EZ)-cyclobilirubin-XIIIα, 3. (EZ)-cyclobilirubin-IXα, 4. (ZE)-bilirubin-IXα, 5. (EZ)-bilirubin-IXα, 6. (ZZ)-bilirubin-IXα, 7. (ZZ)-bilirubin-IIIα.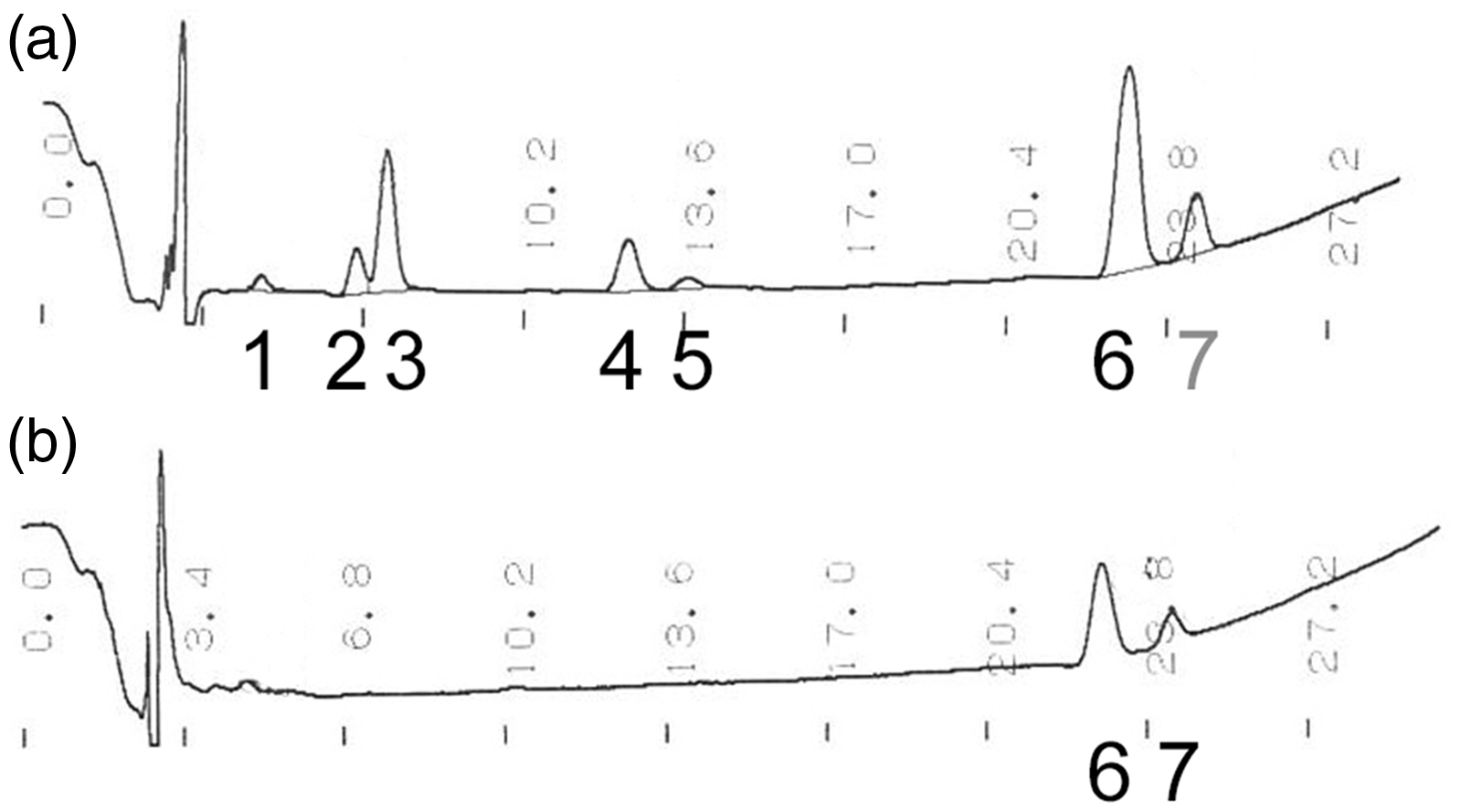
Discussion
The name direct bilirubin was decided because it directly reacts with diazo reagent; however, this does not mean that only conjugated bilirubin is involved in the reaction. Bilirubin photoisomers are non-conjugated, and (EE)-/(EZ)-cyclobilirubin-IXα does not react as DB in DB measurements using the diazo method, while (ZE)-/(EZ)-bilirubin-IXα reacts as DB. 4 In the bilirubin oxidase method, (EZ)-cyclobilirubin-IXα and (ZE)-/(EZ)-bilirubin-IXα react as DB. 5 The present study demonstrated that (EE)-/(EZ)-cyclobilirubin-IXα and (ZE)-/(EZ)-bilirubin-IXα reacted as DB.
DB is measured with change in absorption at 450 nm in vanadic acid oxidation method. The peak detection is measured by absorption at 455 nm in HPLC. The oxidized substance of bilirubin photoisomer is more hydrophilic than (ZZ)-bilirubin, therefore the retention time of oxidized substance is most likely shorter than that of (ZZ)-bilirubin on the chromatogram of HPLC. Peaks other than those of (ZZ)-bilirubin were not detected. Although the presence of oxidized substances of bilirubin photoisomers is not identified, it has no impact on DB value. After irradiation, the substances with absorption at 450 nm of bilirubin/HSA solution are bilirubin photoisomers and (ZZ)-bilirubin. Influence of (ZZ)-bilirubin is very small in this method. 1 We consider that increase in DB is caused by the disappearance of bilirubin photoisomers which causes the decrease in absorption at 455 nm. Since this was an in vitro study, (ZZ)-bilirubin-XIIIα, -IIIα and the photoisomers of (ZZ)-bilirubin-XIIIα were included even though these were not detected in the human serum. The most abundant photoisomer in clinical serum samples, (ZE)-bilirubin-IXα, changes to (ZZ)-bilirubin-IXα under natural environmental condition, during phototherapy, or acidic conditions. 5 This influence may be slightly reduced because this measurement method reacts under acidic conditions.
Conclusion
Bilirubin photoisomers react as DB in the vanadic acid oxidation method.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This article was supported by JSPA KAKENHI Grant Numbers 16K10092, 16K10093 and 17K10179, and Japan Agency for Medical Research and Development Grant Number 17824886.
Ethical approval
Not applicable.
Guarantor
KT.
Contributorship
OH, IS researched literature and conceived the study. OH, KS, OM measured bilirubin fractions. OH wrote the first draft of the manuscript. All authors reviewed, edited and approved the final version of the manuscript.
