Abstract
Background
We occasionally encounter increases in direct bilirubin value on reanalysis of the surplus serum collected in the past from a neonate with indirect hyperbilirubinemia. But the details of this phenomenon are unclear. We evaluated the change of direct bilirubin and the relation of bilirubin photoisomer of the serum exposed to room light.
Methods
Surplus serum samples from neonates with indirect hyperbilirubinemia were exposed to room light for 24 h. The bilirubin fraction assay of samples was performed by the bilirubin oxidase method (Nescauto and Aqua-auto Kainos reagent) and high-performance liquid chromatography.
Results
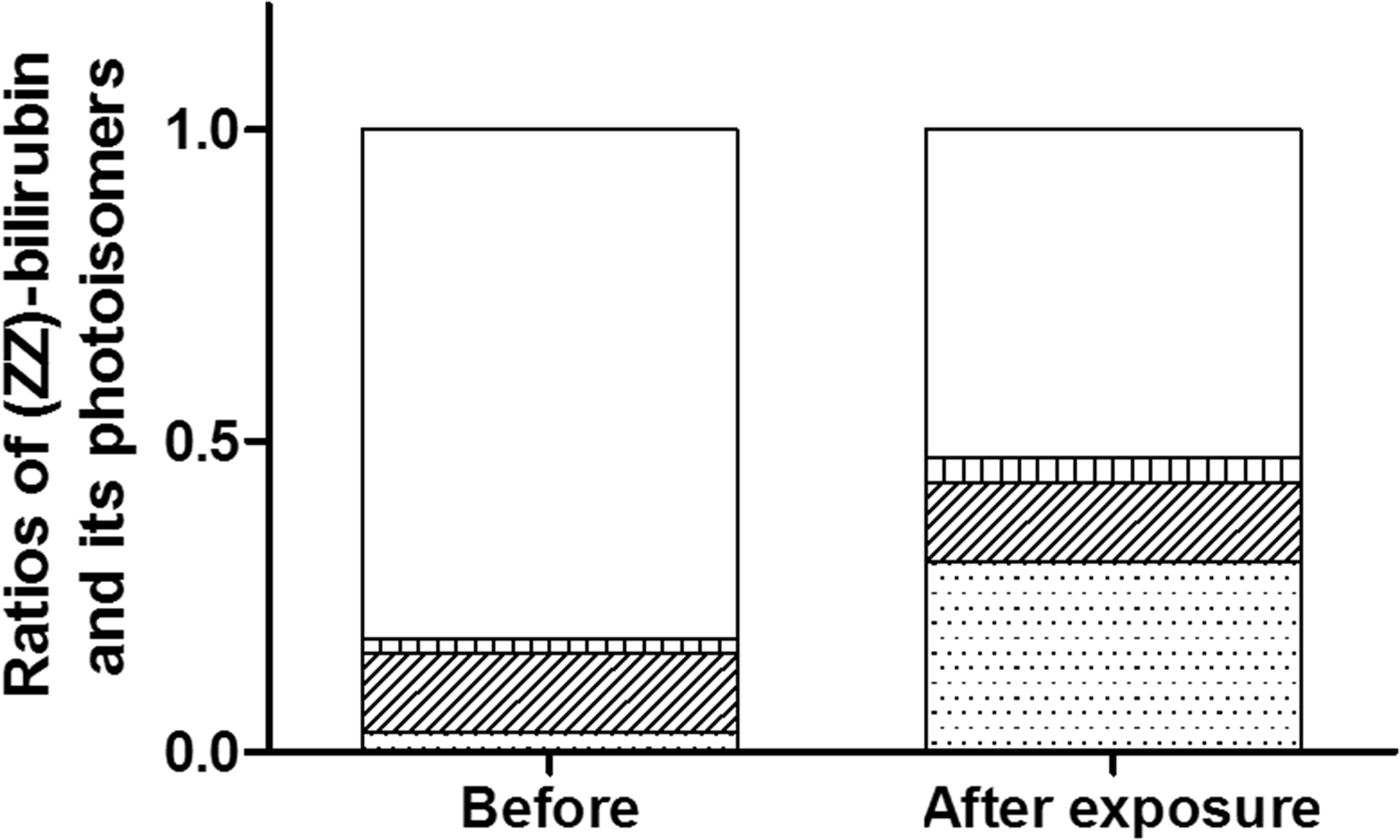
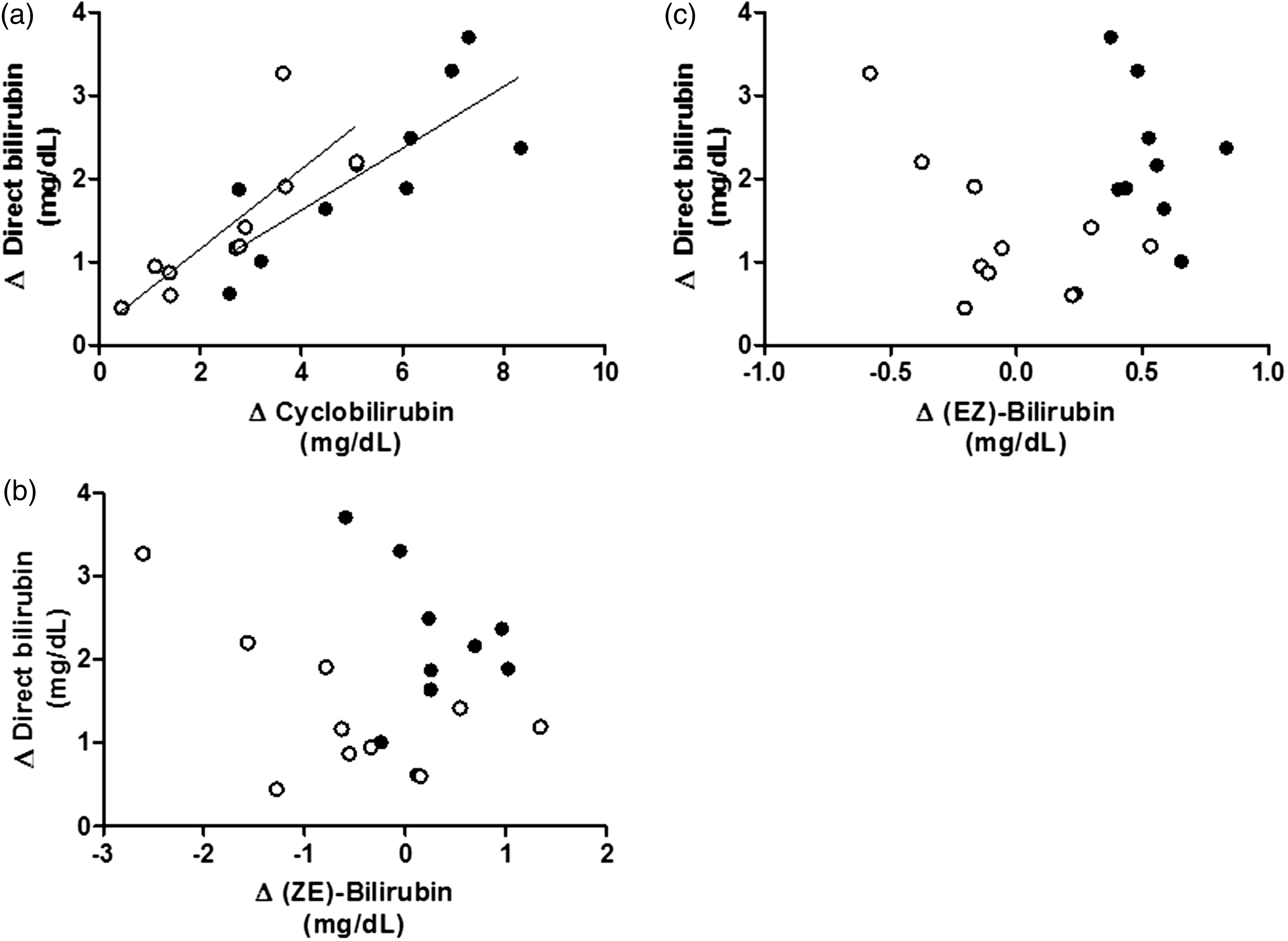
Direct bilirubin increased significantly from 0.61 to 2.36 mg/dL. The respective ratios of bilirubin photoisomers before and after exposure were as follows: cyclobilirubin (0.007 to 0.29) and (EZ)-bilirubin (0.018 to 0.041) increased significantly, (ZZ)-bilirubin decreased 0.84 to 0.55 significantly. The difference of the cyclobilirubin concentration was most closely associated with those of the direct bilirubin concentration.
Conclusion
Direct bilirubin value was increased after exposure to the room light, and increase in direct bilirubin was significantly correlated by cyclobilirubin increase in the serum samples from neonates with indirect hyperbilirubinemia.
Introduction
In routine laboratory practice, the serum bilirubin concentration is determined by various methods, including the bilirubin fraction assay. In Japanese central clinical laboratories, the serum bilirubin concentrations including the direct bilirubin (DB) concentration are currently measured primarily by the enzymatic method (bilirubin oxidase (BO) method1–3 and chemical oxidation method. Bilirubin is oxidized to biliverdin by reacting with BO, which is an enzyme that reacts specifically with bilirubin. By the BO method, DB is measured according to the decrease in absorbance at 450 nm under an acidic condition, which allows conjugated bilirubin to react with the enzyme.1–3 Bilirubin photoisomers, which are unconjugated bilirubin, have been reported to act as substrates in this DB assay method.
4
Especially (4Z,15E)-bilirubin((ZE)-bilirubin) is produced rapidly by exposing to light, and increase in (ZE)-bilirubin was correlated to increase in DB
Clinically, when some kind of abnormality is found in the clinical test, it is sometimes examined whether there was no abnormality by using the past surplus serum in clinical practice. We occasionally encounter increases in DB on reanalysis of the surplus serum collected in the past from a neonate with indirect hyperbilirubinemia. Although (ZE)-bilirubin is rapidly produced, neonatal serum has already contained (ZE)-bilirubin, and amount of (ZE)-bilirubin production by room light exposure is not large. The details of this phenomenon are unclear. In this study, we evaluated that whether DB is increased in the neonatal serum exposed to light or not, and what kind of bilirubin photoisomer is causing the increase in DB.
Methods
Samples
We selected surplus serum samples that had indirect hyperbilirubinemia in the neonate and remained more than 100 µL among the specimens examined in the day. Ten unlinkable anonymized surplus serum samples of these in each facility were use in this study. Due to the method of sample collection at the clinical practice, surplus serum samples are already exposed to environmental light, and the time of exposure to environmental light varies. Time point at ‘Before’ is the time at the start of the experiment not at the blood sampling. Time point at ‘After exposure’ is the time at after 24 h from ‘Before'. Samples were put only on the desk used for keeping blood sample as routine for 24 h under room light in the central laboratory of each facility. They were stored by freezing in the dark and assayed. Room temperature is controlled to by air conditioning. There was no problem in quality control of DB measurement during the study period. In the sample of before and after exposure, total bilirubin (TB) and DB were measured by the BO method, which is widely employed by the central clinical laboratories of various facilities, and bilirubin and its photoisomers were measured by HPLC.
At Facility A, TB and DB were assayed using Nescauto T-BIL-VE and D-BIL-VE reagents (Alfresa, Osaka, Japan). The room lights in the laboratories were FLR40S EXN-/M-A 40 W (HITACHI, Tokyo, Japan), and the light energy at the site where the samples were placed was 0.231
Bilirubin photoisomers were assayed by the method of Itoh et al.
6
using HPLC. HPLC was performed using Shimadzu LC-9 A (Shimadzu Co., Kyoto, Japan) and a 5 mm × 10-cm Nova-Pack C18 radial compression cartridge column (Nihon Waters KK, Tokyo, Japan). Using a mixture of acetonitrile, 0.01 M phosphate buffer (pH 5.5), and dimethylformamide at 5:30:65 (v/v/v) as Liquid A and their mixture at 20:15:65 (v/v/v) as Liquid B, the identification and quantification of bilirubin were performed by flowing Liquid A at 1 mL/min with gradients of 0–34% over 17 min and 34–100% over the next 8 min. Measurements were performed using the SPD-6AV detector (Shimadzu Co., Kyoto, Japan) at a wavelength of 455 nm. The samples were deproteinized by mixing them with DMSO and acetonitrile at the same volume ratio and centrifuging the mixture, and 25
Statistical procedures
Comparison of data between two groups was performed by the paired
Results
After exposure to room light, TB decreased significantly ( Changes in total and direct bilirubin due to exposure to room light. Changes in the ratios of bilirubin fractions after exposure to room light. The ratio of each fraction is shown by assuming the sum of (ZZ)-bilirubin and bilirubin photoisomers as 1.0. Cyclobilirubin is defined as the sum of (EE)-cyclobilirubin and (EZ)-cyclobilirubin. The open, vertically striped, diagonally striped, and dotted areas represent (ZZ)-bilirubin, (EZ)-bilirubin, (ZE)-bilirubin, and cyclobilirubin, respectively. Relationships between the difference of bilirubin photoisomers and that of direct bilirubin according to the reagent used. (a) Relationship between Δcyclobilirubin and ΔDB. (b) Relationship between Δ(ZE)-bilirubin and ΔDB. (c) Relationship between Δ(EZ)-bilirubin and ΔDB. The difference between the measured values before and after exposure to room light is indicated by Δ. A positive correlation was observed only between Δcyclobilirubin and ΔDB (R2 = 0.63, 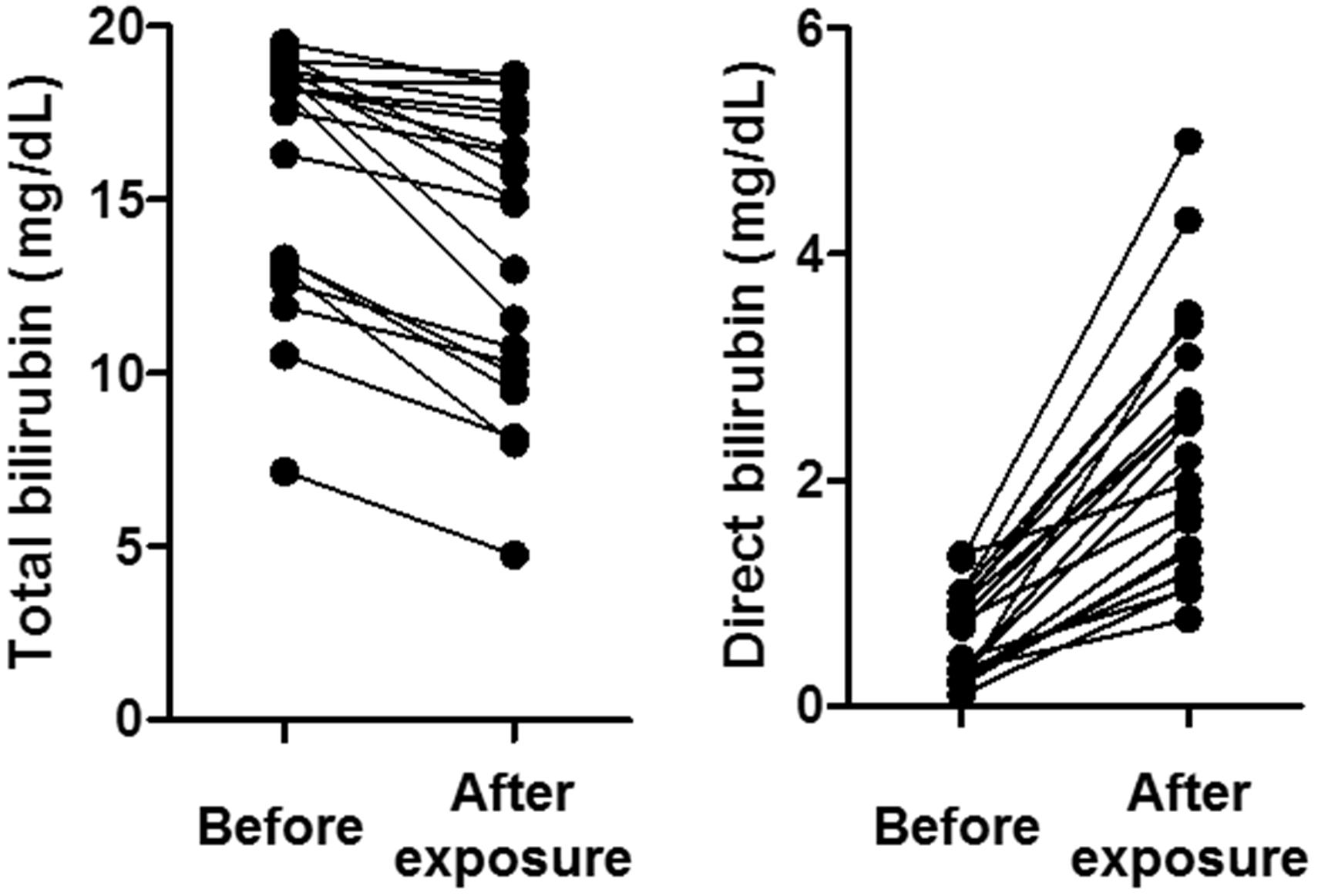
Discussion
In this study, we found out that DB is increased when serum of neonates with unconjugated hyperbilirubinemia is exposed to room light in clinical practice, and the increase in DB is significantly correlated with increase in cyclobilirubin, which is bilirubin photoisomer.
Bilirubin photoisomers are rapidly produced from (ZZ)-bilirubin when it is exposed to light at a particular wavelength. 7 Bilirubin photoisomers include (EZ)-bilirubin, (ZE)-bilirubin, and (EE)-bilirubin, which are configurational isomers, and (EZ)-cyclobilirubin and (EE)-cyclobilirubin, which are structural isomers. In physiologic human serum, (ZE)-bilirubin as a photoisomer is present in the largest amount next to (ZZ)-bilirubin, and (ZE)-bilirubin/(ZZ)-bilirubin ratio is about 0.1 in serum of neonates with unconjugated hyperbilirubinemia. 8 Therefor, DB value is indicated including bilirubin photoisomers reacted to BO. For short-term exposure, such as some seconds to minutes, in serum immediately after collection, since cyclobilirubin production is small, change of DB is most likely affected by change of (ZE)-bilirubin. (ZE)-Bilirubin and (ZZ)-bilirubin are in a photostationary state, and (ZE)-bilirubin/(ZZ)-bilirubin ratio is determined by the light source characteristics (the (ZE)-bilirubin/(ZZ)-bilirubin ratio could be the same if the ratio of light energy at each wavelength contained in the light source were the same). 9 In this study, fluorescent lamps which were used in facilities are commercially marketed in Japan. (ZE)-Bilirubin/(ZZ)-bilirubin ratio is about 0.2 in the serum after exposure to the room light. Since the reactivity of (ZE)-bilirubin with BO decreases in acidic condition , 10 a part of increase in (ZE)-bilirubin influences DB value.
(EZ)-cyclobilirubin is produced from (ZZ)-bilirubin through (EZ)-bilirubin11–13 and is the most important photoisomer in the action mechanism of phototherapy for humans.8,14 In bilirubin-human serum albumin complex solution, production of (EZ)-cyclobilirubin has a dose-response relationship with the irradiation time, and also the light energy.
15
(EE)-Cyclobilirubin and (EZ)-cyclobilirubin maintain a relationship of a photostationary state.6,16 Unlike configurational isomers, structural isomers do not return to (ZZ)-bilirubin. Therefore, under
The ‘direct’ of DB derives from the reactivity of bilirubin to diazo reagent. However, the classification based on the structure of bilirubin is not in complete agreement with that based on the reactivity to diazo reagent. Since bilirubin that reacts under an acidic condition is regarded as DB by the BO method,1–3 the results of DB measurement by the BO method are also partially inconsistent with the structural classification of bilirubin. In Nescaut reagent, BO is reacted in a citrate-lactate buffer at pH 3.7, and δ-bilirubin and bilirubin photoisomers 4 are reacted as DB. In Aqua-auto Kainos reagent, BO is reacted at pH 4.5–5.0 although the buffer is not specified, and δ-bilirubin is not reacted as DB, 3 it has not been reported, however, whether the bilirubin photoisomer is reacted nor not. Concerning the reactivity of BO with bilirubin photoisomers, while (EZ)-cyclobilirubin shows 100% reactivity with BO at pH 3.5–7.4, the reactivity of (ZE)-bilirubin decreases at pH 4.5–7.4. 10 As the absorption spectrum of (ZZ)-bilirubin varies with the buffer type and pH, 10 the absorption spectra of its photoisomers are also considered to vary with the buffer type and pH. The reaction conditions differ between the two assay methods, and since there was the possibility that photoisomers exert different effects on the DB assay, analysis was performed separately with each method. Although the amount of bilirubin photoisomers differed between the two facilities due to the difference in room light characteristics, the relation between ΔDB and Δbilirubin photoisomers was not different in two assay methods.
In the measurement of TB by the BO method, all bilirubin including conjugated and unconjugated bilirubin are considered to react. Since the total amount of unconjugated bilirubin does not decrease despite the generation of bilirubin photoisomers, TB is not considered to decrease even if DB increases. Actually, however, a decrease in TB was observed. One cause of this decrease in TB may be that this assay method is based on the change in absorbance: The absorbance near 450 nm decreased when photoisomers was produced
6
with a consequent decrease in the absorbance already before the addition of the BO reagent in exposed samples. Another cause may be bronze baby syndrome, which is considered to result from the polymerization of cyclobilirubin due to its accumulation in the body,
17
because the samples exposed to environmental light appeared somewhat browned, and this appearance resembled that of serum in bronze baby syndrome, which is an adverse effect of phototherapy in neonates. Cyclobilirubin generated
Conclusion
In this study, we found out that in the assay of DB by the BO method, DB is increased when serum of neonates with indirect hyperbilirubinemia is exposed to room light in clinical practice, and the increase in DB is significantly correlated with increase in cyclobilirubin.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
This article was supported by JSPS KAKENHI Grant Numbers 16K10092, 16K10093.
Ethical approval
The study was approved by the institutional ethics committee (Kagawa University Heisei 27-025, Kyoto Medical Center #10-145).
Guarantor
KT.
Contributorship
OH, KK, IS, KT researched literature and conceived the study and was involved in protocol development. KK, IT, KK, YS collected samples. OH, OM, KI, AT measured bilirubin fractions. OH wrote the first draft of the manuscript. OH, KT, MK, KK was gaining ethical approval. All authors reviewed and edited the manuscript and approved the final manuscript.
