Abstract
Background
Hypercholesterolemia (plasma cholesterol concentration ≥5.2 mmol/L) is a risk factor for cardiovascular disease and stroke. Many different cholesterol self-tests are readily available at general stores, pharmacies and web shops. However, there is limited information on their analytical and diagnostic performance.
Methods
We included 62 adult patients who required a lipid panel measurement (cholesterol, high-density lipoprotein (HDL), triglycerides and LDLcalc) for routine care. The performance of five different cholesterol self-tests, three quantitative meters (Roche Accutrend Plus, Mission 3-in-1 and Qucare) and two semi-quantitative strip tests (Veroval and Mylan MyTest), was assessed according to the manufacturers’ protocol.
Results
The average plasma cholesterol concentration was 5.2 ± 1.2 mmol/L. The mean absolute relative difference (MARD) of the five cholesterol self-tests ranged from 6 ± 5% (Accutrend Plus) to 20 ± 12% (Mylan Mytest). The Accutrend Plus cholesterol meter showed the best diagnostic performance with a 92% sensitivity and 89% specificity. The Qucare and Mission 3-in-1 are able to measure HDL concentrations and can thus provide a cholesterol:HDL ratio. The Passing-Bablok regression analyses for the ratio showed poor performance in both self-tests (Mission 3-in-1: y = 1.62x–1.20; Qucare: y = 0.61x + 1.75). The Accutrend Plus is unable to measure the plasma high-density lipoprotein concentration.
Conclusions/interpretation: The Accutrend Plus cholesterol meter (Roche) had excellent diagnostic and analytic performance. However, several of the commercially-available self-tests had considerably poor accuracy and diagnostic performance and therefore do not meet the required qualifications, potentially leading to erroneous results. Better regulation, standardization and harmonization of cholesterol self-tests is warranted.
Introduction
Hypercholesterolemia (plasma cholesterol concentration ≥5.2 mmol/L or 200 mg/dL) is a major modifiable risk factor for cardiovascular disease and stroke.1–3 Numerous cholesterol self-tests are commercially available at general stores, pharmacies and web shops. Self-tests can be used on an individual basis, but also as population-wide screening tools. However, in particular, when using self-tests as screening tools, the performance of these tests is of great importance.
While some cholesterol self-tests measure solely the total cholesterol value, others additionally provide the concentration of triglycerides, high-density lipoprotein (HDL) and calculated low-density lipoprotein (LDLcalc). However, there is limited information available on the validity, accuracy, precision and diagnostic performance of these commercially-available cholesterol self-tests. As there is an increasing public awareness of the importance of early detection of lipid disorders, self-testing may become part of the standard home-care devices in peoples’ homes. However, it is important that these devices are safe to be used in the hands of non-professionals. Besides user friendliness of the equipment and unambiguous user manuals, test results should be accurate and easy to interpret. Inaccurate results could lead to unnecessary visits to the hospital or general practitioner or to false reassurance and withholding of adequate treatment.
In this study, the performance of five cholesterol self-tests, two semi-quantitative strip-based tests (Mytest cholesterol and Veroval) and three quantitative meters (Mission 3-in-1, Quare and Accutrend Plus), was compared with routine chemistry analyses. Plasma lipid concentrations obtained from the routine analyses at the Jeroen Bosch Hospital (JBZ) were validated by three independent laboratories, using different routine chemistry analysers.
Methods
Patient population
In this study, 62 adult patients were included who required a lipid profile (cholesterol, triglyceride, HDL and LDL) measurement at the JBZ for routine care. Patients were included from 17 till 29 April 2019. For one patient, the routine lipid profile was not measured and, therefore, this patient was excluded from future analyses, leaving 61 patients for data analysis. The number of included patients is adequate, based on the ‘evaluation protocol 9’ (EP9) protocol guidelines. 4 The study was performed according to the ‘Declaration of Helsinki’. The local ethics board approved the study, and all patients provided their written informed consent.
Study protocol
Venous blood was collected via venipuncture in lithium-heparin tubes (LH PST II, Thermo Fisher Scientific) for routine analysis. The concentration of cholesterol, HDL, triglycerides and LDLcalc was determined using routine protocols on the chemistry analyser (Advia Chemistry XPT system, Siemens, Germany) at the JBZ clinical chemistry laboratory. Residual plasma was aliquoted into four separate tubes and stored at –70°C for future analyses. Subsequent to the venipuncture, the patients underwent five different cholesterol self-tests. The tests were carried out according to the information leaflet provided with the self-test and were performed by experienced and trained staff. For the ‘Mission’ self-test, two patients were excluded due to an insufficient number of test strips.
Cholesterol self-tests
Five commercially available cholesterol self-tests were included in this study, three strip-based quantitative meters and two single-use semi-quantitative colour-based test strips. The three quantitative meters included the ‘Mission 3-in-1’ (ACON Laboratories, Inc., San Diego, USA), the ‘Qucare’ (DFI Co., Ltd, Gimhae, South Korea) and the ‘Accutrend Plus’ (Roche, Basel, Switzerland). The single-use test-strips that were included in this study were the ‘Mytest Cholesterol’ (Mylan, Canonsburg, USA) and the ‘Veroval’ (Hartmann, Nijmegen, the Netherlands). General information regarding these self-tests can be found in Table 1. The results obtained by the self-tests were recorded manually and subsequently immediately checked by a second laboratory technician. Hereafter, the results were digitalized, a process that was also validated by a second employee.
Basic characteristics of the five included cholesterol self-tests.
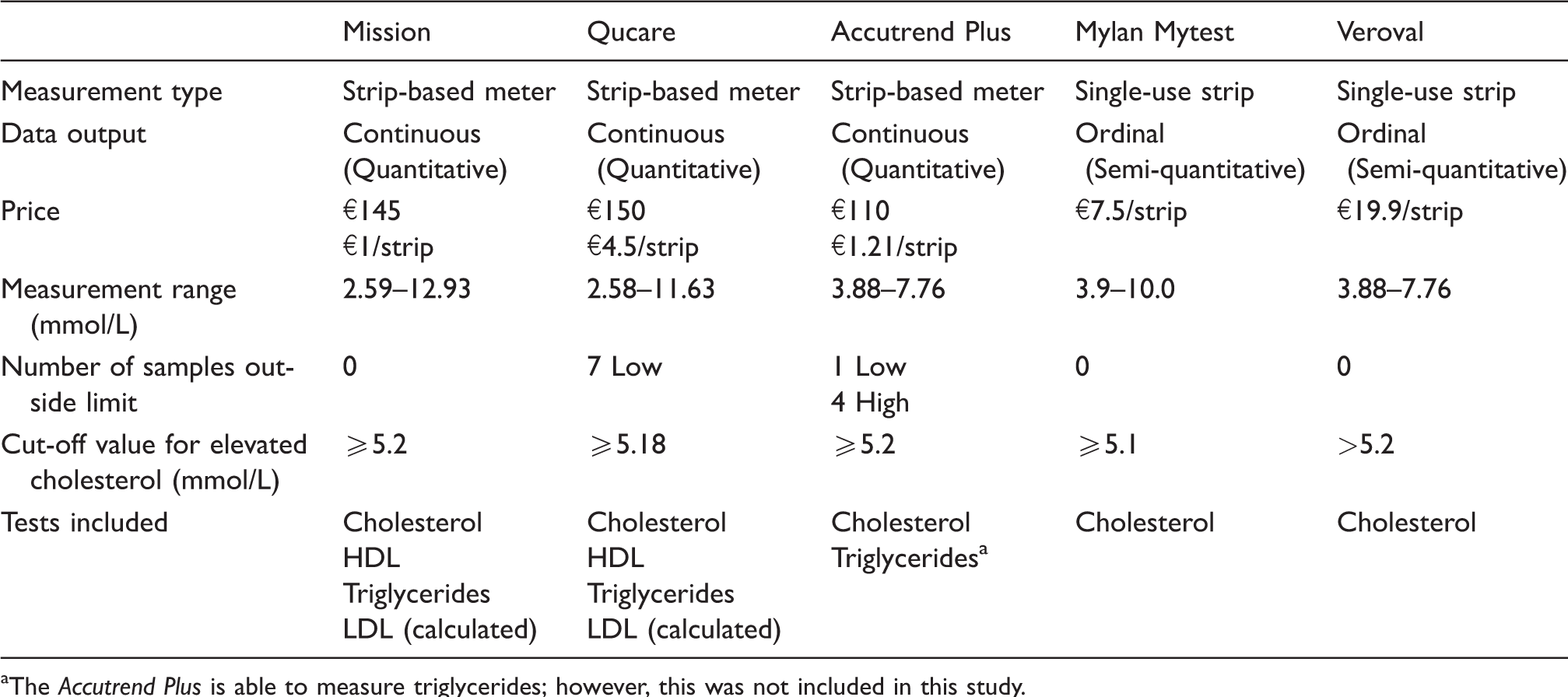
aThe Accutrend Plus is able to measure triglycerides; however, this was not included in this study.
External verification of the results from the routine chemistry analyser
Stored plasma samples (one year at –70°C) were sent on dry ice to three separate clinical chemistry laboratories, which use different routine chemistry analysers: Meander Medical Center (Architect c16000, Abbott Diagnostics, the Netherlands), Medlon (COBAS 702, Roche diagnostics, the Netherlands) and Saltro (Beckman AU 700). The lipid profile was measured and the values were compared with the results from the JBZ (Siemens, Advia Chemistry XPT system). All cholesterol measurements of the routine chemistry analysers and self-tests are based on the enzymatic reactions performed by sequentially cholesterol esterase and cholesterol oxidase. LDL was calculated using the Friedewald formula (LDL = cholesterol–HDL–[triglyceride/2.2]). 5 All laboratories that took part in this study participate in the external quality control scheme of the Dutch Foundation for Quality Assessment in Medical Laboratories (SKML), with an average coefficient of variation for cholesterol of 1.3%.
Statistical analyses
Data were analysed using the Microsoft Office Excel (2010) plugin ‘Analyse-IT v5.11’ (Analyse-it Software, Ltd, UK) and SPSS statistics v22 (IBM, USA). Out-of-range values were given the same value as the lowest or highest measurement limit. For the calculation of the mean relative difference (MRD), mean absolute relative difference (MARD) and the Passing-Bablok analyses, the out-of-range values were excluded in the analyses.
Results
Validation of results
In 61 patients who required a routine lipid panel measurement, the blood cholesterol concentration was determined by five separate commercially available cholesterol self-test; three strip-based quantitative meters (‘Mission 3-in-1’, ‘Qucare’ and ‘Accutrend Plus’) and two single-use semi-quantitative colour-based test strips (‘Mylan Mytest’ and ‘Veroval’). An overview of the basic characteristics of these cholesterol self-tests is shown in Table 1. Results from the self-test measurements were compared with the venous blood cholesterol concentrations measured by routine chemistry analyser (Advia Chemistry XPT, Siemens) at the JBZ. The values from this analyser were commensurate to the concentrations measured by other routine chemistry analysers from different firms (Roche, Abbott and Beckman), as shown by excellent Passing-Bablok analyses and biases below 5% (Supplemental Figure 1 and Supplemental Tables 1 and 2).
Accuracy analysis
The MRD of cholesterol concentrations obtained from the Accutrend Plus compared with the routine analyser was the lowest with 4 ± 7%, followed by the Mission with 11 ± 7% and the Qucare with –19 ± 13% (Figure 1(a) to (c) and (f)). Of the two single-use strips, the MRD of the Mylan Mytest was –16 ± 17% and the MRD of the Veroval was 5 ± 12% (Figure 1(d) to (f)). The MARD ranged from 6 ± 5% (Accutrend Plus) to 20 ± 12% (Mylan Mytest) (Figure 1(g)).
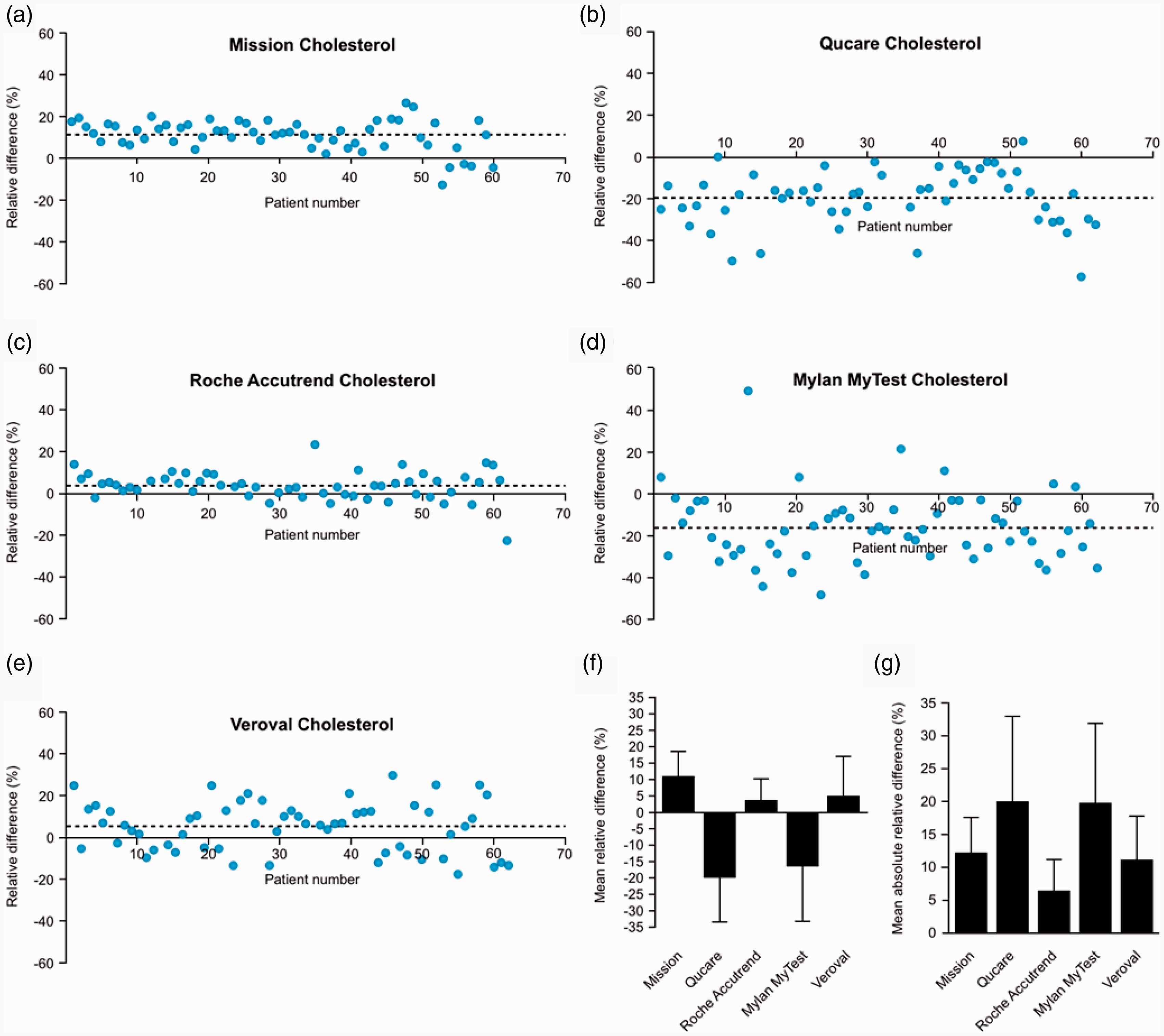
Accuracy analyses. (a–e) The relative difference between the blood cholesterol concentration of each individual patient (blue dot) measured by the self-tests (a) Mission, (b) Qucare, (c) Accutrend Plus, (d) Mylan MyTest and (e) Veroval, compared with the routine chemistry analyser from Siemens (Advia XPT). The dotted lines indicate the mean relative difference (MRD). (f) The MRD of each self-test, data taken from A-E. (g) The mean absolute relative difference of each self-test.
Method comparison
The accuracy analysis results were confirmed by using Passing and Bablok (PB) method comparison analysis. The Mission measured a higher cholesterol concentration, which deviated more at higher cholesterol concentrations (Figure 2(a) and Table 2. PB regression: y = 1.15x–0.15. Pearson’s r = 0.96). The PB of the Mission had a 95% confidence interval (CI) for the intercept that contains zero, indicating the absence of a systematic difference, but had a slope above 1, showing a modest proportional error. The Qucare showed significantly lower results for cholesterol and with considerable variation (PB regression: y = 1.00x–0.90. Pearson’s r = 0.69; Figure 2(b) and Table 2). However, the 95% CI of the Qucare had an intercept containing zero and a slope containing one. The blood cholesterol concentration measured by the Accutrend Plus was comparable to the concentration determined by the routine analyser with very low variation (PB regression: y = 1.08x–0.21. Pearson’s r = 0.94; Figure 2(c) and Table 2). As the semi-quantitative single-use test strips produce ordinal data, no PB analysis was performed for the Mylan Mytest and the Veroval.
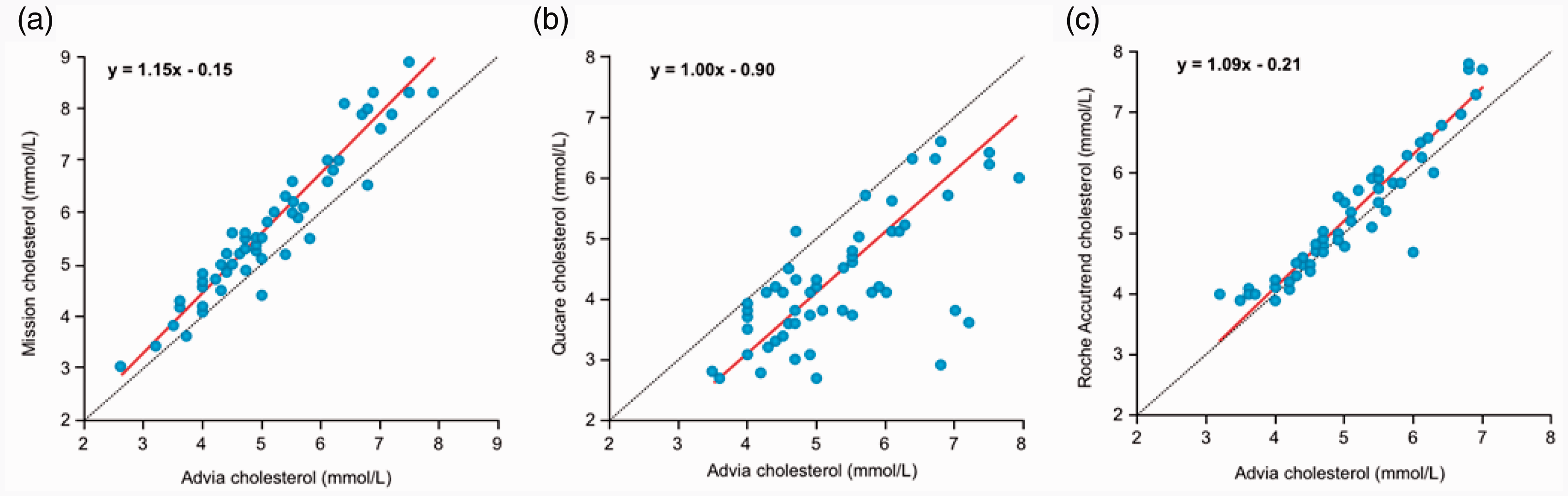
Method comparison analyses. (a–c) Passing Bablok analyses comparing the self-tests to the routine chemistry analyser from Siemens (Advia XPT). (a) Mission (intercept –0.15 [95% CI –0.64–0.35], slope 1.15 [95% CI 1.05–1.25]), (b) Qucare (intercept –0.90 [95% CI –1.79–0.11], slope 1.00 [95% CI 0.80–1.18]) and (c) Accutrend Plus (intercept –0.21 [95% CI –0.76–0.25], slope 1.08 [95% CI 1.00–1.19]).
The diagnostic properties of the cholesterol self-tests based on the values from the routine chemistry analyser (Siemens Advia XPT) as reference, with a cut-off of 5.2 mmol/L.
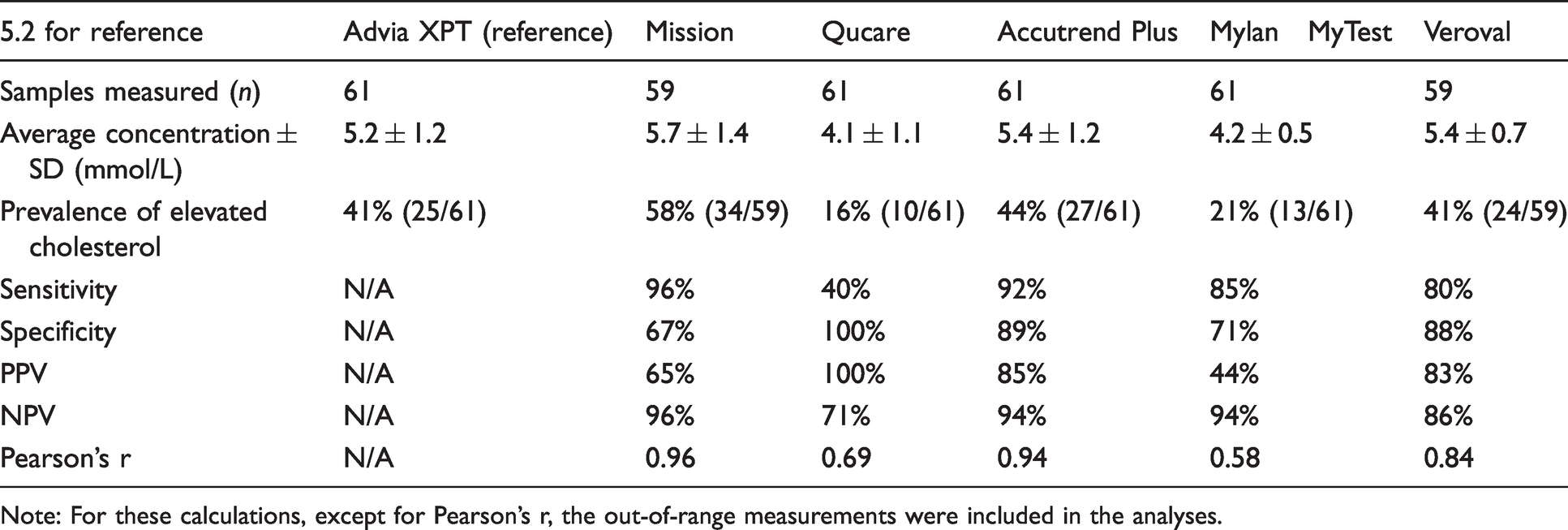
Note: For these calculations, except for Pearson’s r, the out-of-range measurements were included in the analyses.
Diagnostic performance
An elevated blood cholesterol is defined as a concentration >200 mg/dL (>5.2 mmol/L) in the U.S. guidelines. 6 For the self-tests, the cut-off concentration for elevated cholesterol described in the medication package insert was used, which was between 5.1 and 5.2 mmol/L for all self-tests (Table 1). Based on the results from the routine chemistry analyser, a total of 25 patients (41%) had an elevated blood cholesterol concentration (Table 2). The values from the laboratory were used as reference value to calculate the diagnostic properties of the self-tests.
The Mission self-test had a high sensitivity (96%) and negative predictive value (NPV, 96%), but poor specificity (67%) and positive predictive value (PPV, 65%; Table 2). Because the Qucare measured a much lower average cholesterol concentration than the routine chemistry analyser, it had a sensitivity of 40% but a 100% specificity (Table 2). For the Accutrend Plus, all parameters related to diagnostic performance were between 85 and 94% (Table 2). The semi-quantitative colour-based strip MylanMyTest had a sensitivity of 85% and a NPV of 94%, but a poor specificity (71%) and PPV (44%; Table 2). For the semi-quantitative colour-based strip Veroval, all parameters related to diagnostic performance were between 80 and 86% (Table 2). The diagnostic ability of the self-tests based on the European reference value (>5.0 mmol/L) is shown in Supplemental Table 3.
Lipid panel analysis
The Mission and Qucare self-tests are able to additionally measure the blood triglyceride (TG) and HDL concentration, and produce a calculated LDL value (LDLcalc) based on the Friedewald formula. The Mission measured the blood TG concentration 22 ± 25% above the routine laboratory measurement, whereas the Qucare measured 7 ± 44% lower (Table 3, Figure 3(a) and (b)). On the other hand, the HDL concentration was measured 13 ± 10% and 26 ± 16% lower by the Mission and the Qucare, respectively (Table 3, Figure 3(c) and (d)). For the Qucare, the error in the HDL measurement increased considerably at higher HDL concentrations (PB regression y = 0.40x + 0.46; Figure 3(d)). For both the Mission and the Qucare, this led to erroneously calculated LDL concentrations (Table 3, Figure 3(e) and (f)). Moreover, the Qucare reported the LDLcalc value even when the total cholesterol or the HDL concentration was out of range. The cholesterol:HDL ratio is an important parameter in estimating an individual’s risk for developing heart disease. 7 As the Mission measured the concentration of cholesterol higher and the HDL lower than the reference value, the error in the cholesterol:HDL ratio was amplified (PB regression y = 1.62x–1.20; Figure 3(g)). The cholesterol:HDL ratio calculated from the values measured by the Qucare also deviated strongly from the ratio calculated from the laboratory measurements (PB regression y = 0.61x + 1.75, Figure 3(h)).
The accuracy analyses for the blood triglyceride and HDL concentrations, and calculated LDL (LDLcalc) value of the Mission and the Qucare self-tests with the Advia XPT routine chemistry analyser from Siemens as reference.

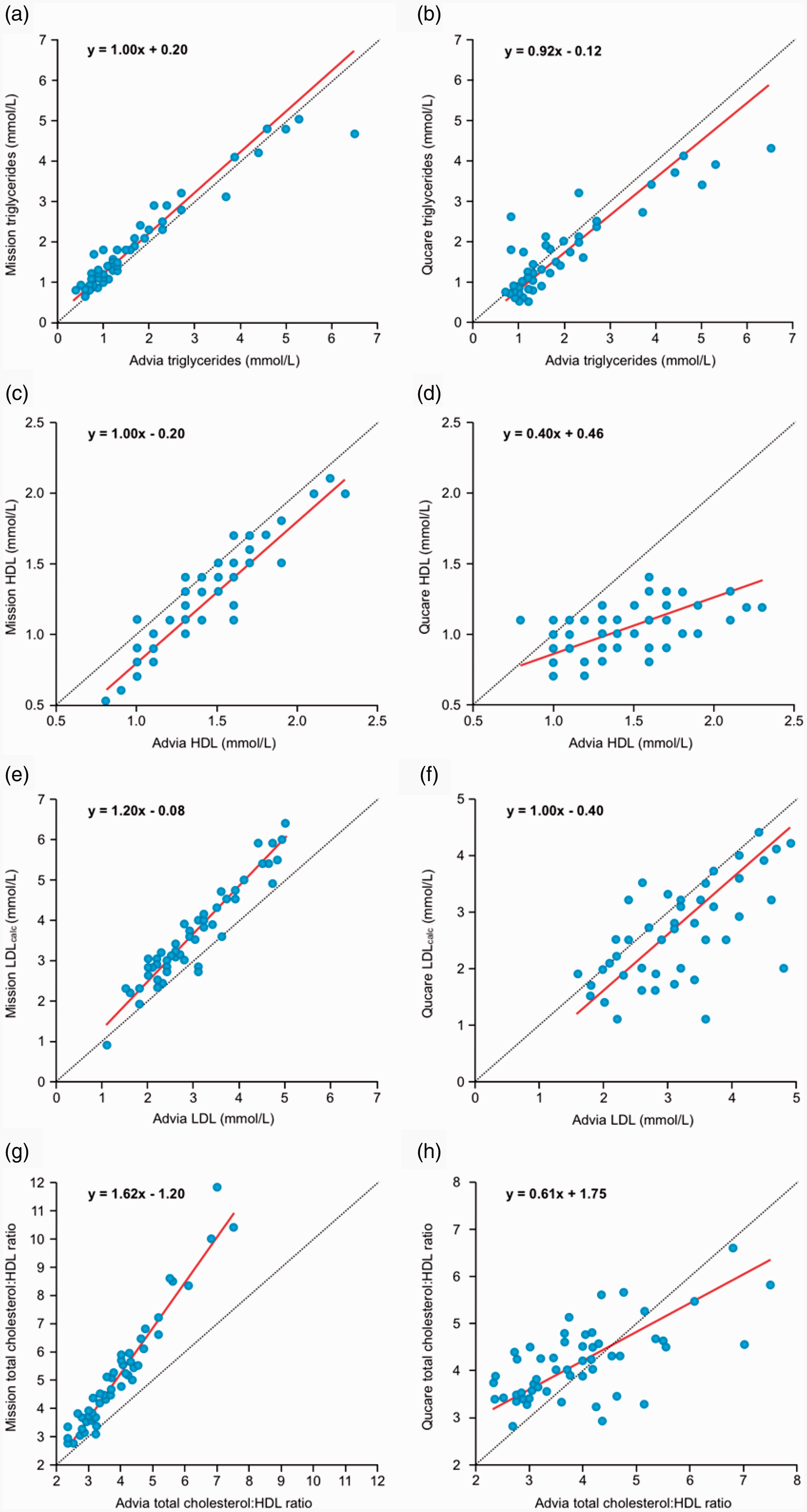
Method comparison for the lipid panel. Passing Bablok (PB) analyses comparing the triglyceride and HDL concentrations, and the calculated LDL and cholesterol:HDL ratio of the self-tests to the values from the routine chemistry analyser from Siemens (Advia XPT). (a) PB Mission triglycerides (intercept 0.20 [95% CI 0.15–0.38], slope 1.00 [95% CI 0.90–1.06]) and (b) PB Qucare triglycerides (intercept –0.12 [95% CI –0.33–0.13], slope 0.92 [95% CI 0.76–1.12]). (c) PB Mission HDL (intercept –0.20 [95% CI –0.42 to –0.10], slope 1.00 [95% CI 1.00–1.17]). (d) PB Qucare HDL (intercept 0.46 [95% CI 0.25–0.66], slope 0.40 [95% CI 0.25–0.5]). (e) PB Mission calculated LDL (LDLcalc) (intercept 0.08 (95% CI -0.37 – 0.43), slope 1.2 (95% CI 1.09 – 1.33)). (f) PB Qucare LDLcalc (intercept –0.40 [95% CI –1.16–0.29], slope 1.00 [95% CI 0.79–1.23]). (g) PB Mission cholesterol:HDL ratio (intercept –1.20 [95% CI –1.82 to –0.74], slope 1.62 [95% CI 1.48–1.80]). (h) PB Qucare total cholesterol:HDL ratio (intercept 1.75 [95% CI 1.07–2.25), slope 0.61 [95% CI 0.46–0.82]).
Discussion
In this study, conducted independently and without support from any manufacturer, we investigated the performance and diagnostic ability of five commercially-available cholesterol self-tests. This is the first study investigating the analytical and diagnostic performance of commercially-available cholesterol self-tests. The performance of the self-tests differs greatly between suppliers, with some performing satisfactory, such as the Accutrend Plus from Roche, whereas others perform poorly on both accuracy and diagnostic ability.
With an MARD of 6%, the Accutrend Plus from Roche is the most accurate of the cholesterol self-tests. Moreover, it has the best diagnostic performance, with a sensitivity of 92% and a specificity of 89%. The Accutrend Plus is unable to measure the concentration of triglycerides, HDL and LDLcalc. This makes it impossible for the Accutrend Plus, in contrast to the Mission and Qucare, to determine the cholesterol:HDL ratio, an important parameter to estimate the risk of coronary heart disease and acute myocardial infarction.8–11 However, both the Mission and Qucare perform poorly when calculating the cholesterol:HDL ratio and should not be used for this purpose.
For the single-use semi-quantitative strips, the diagnostic ability of the Veroval (80% sensitivity and 88% specificity) was superior to the Mylan MyTest. Moreover, the Veroval had the more optimal accuracy (MARD 11%), which is superior to the quantitative meters Qucare and Mission.
This study has several key strengths. The self-tests were handled according to the manufacturers’ guidelines. Many studies comparing point-of-care equipment or self-tests assess their performance on residual venous plasma samples or venous whole blood, instead of using capillary blood directly taken from a finger prick.12,13 Moreover, the plasma cholesterol concentrations measured by our routine chemistry analysers were validated by three independent laboratories using chemistry analysers from different manufacturers, ensuring the validity of the measured concentrations. Lastly, the tests were performed by experienced and trained personnel, maximizing their performance compared with a layman operating these devices.
Some limitations have to be considered. There was no information on possible interfering substances (such as medication or underlying illness). However, since all self-tests and chemistry analysers measured the same samples and use the same enzymatic reaction (e.g. cholesterol oxidase), major interferences from such sources are unlikely. Secondly, the two semi-quantitative self-tests rely on colour change and the subsequent subjective personal judgment of the corresponding colour. This can lead to an erroneous interpretation of the result. To minimize the impact of subjective interpretation, a second employee immediately validated the colour interpretation. Lastly, in this study, we only investigated the analytical performance of the self-tests. The devices were handled by professionals instead of lay people, which could lead to an overestimation of the performance. Moreover, an important aspect of the usability is the user friendliness of the devices, which was not investigated in this study. When reading the manuals, there were obvious differences in clarity. Importantly, incorrect use of self-tests can impact the validity of the results.
Cholesterol self-tests are readily commercially available. However, there are no data on their performance. In this study, we have demonstrated that the Accutrend Plus shows the best performance compared with the routine chemistry analyser. However, the Accutrend Plus is not able to measure the HDL concentration, limiting its practical use as a screening tool. Both the accuracy and diagnostic performance of the Qucare are poor, and hence, this device led to erroneous interpretations. While the Qucare and Mission cholesterol meters are able to measure HDL, both meters have a poor performance when calculating the cholesterol:HDL ratio. In view of our data, several commercially-available self-tests do not meet the required ISO standards, possibly leading to erroneous diagnoses. Manufacturers of diagnostic self-tests, and professionals intending to use them, should take into consideration the upcoming in vitro diagnostic regulation going into effect in 2022. 14 When using these meters as screening tools, analytical bias and inaccuracy should be taken into account. In the case of abnormal values, the results should always be validated with a subsequent venous blood sampling and measurements at the central laboratory. Moreover, better regulation, standardization and harmonization of cholesterol self-tests is warranted.
Supplemental Material
sj-pdf-1-acb-10.1177_0004563221992393 - Supplemental material for Performance of commercially-available cholesterol self-tests
Supplemental material, sj-pdf-1-acb-10.1177_0004563221992393 for Performance of commercially-available cholesterol self-tests by Steef Kurstjens, Eugenie Gemen, Selina Walk, Tjin Njo, Johannes Krabbe, Karlijn Gijzen, Marc GLM Elisen and Ron Kusters in Annals of Clinical Biochemistry
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The costs for the self-tests and the strips were covered by the Dutch Heart Foundation.
Ethical approval
Medisch Ethische Toetingscommissie Brabant (METC Brabant), Ref: NW2019-28.
Guarantor
RK.
Contributorship
SK analysed the data and wrote the article. EG designed and coordinated the study, and critically read the article. RK initiated, designed and coordinated the study and critically read the article. SW, TN, JK, KG, MGLME performed analyses, designed the study and critically read the article.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
