Abstract
Lipid-lowering therapies, particularly non-statin regimens, are underutilized as ~2/3 of patients with atherosclerotic cardiovascular (CV) disease (CVD) are not optimally managed, and do not attain target low-density lipoprotein cholesterol (LDL-C) concentrations, despite statin treatment. Statins have been the mainstay of hypolipidemic therapies; however, they are plagued by adverse effects, which have partly hindered their more widespread use. Ezetimibe is often the first added mode of treatment to attain LDL-C goals as it is efficacious and also allows the use of a smaller dose of statin, while the need for more expensive therapies is obviated. We herein provide a comprehensive review of the effects of ezetimibe in lipid lowering and reducing CV events and improving outcomes. Of the hypolipidemic therapies, oral ezetimibe, in contrast to newer agents, is the most convenient and/or affordable regimen to be utilized as mono- or combined therapy supported by data from CV outcomes studies attesting to its efficacy in reducing CVD risk and events. When combined with a statin, the statin dose could be lower, thus curtailing side-effects, while the hypolipidemic effect is enhanced (by ~20%) as the percentage of patients with target level LDL-C (<70 mg/dL) is higher with combined treatment versus a high-intensity statin. Ezetimibe could also serve as an alternative treatment in cases of statin intolerance. In conclusion, ezetimibe has an excellent safety/tolerability profile; it is the first added treatment to a statin that can attain LDL-C targets. In the combined therapy, the hypolipidemic effect is enhanced while the dose of statin could be lower, thus limiting the occurrence of side-effects. Ezetimibe could also serve as an alternative mode of treatment in cases of statin intolerance.
Keywords
Introduction
The usage of lipid-lowering treatment has been less than optimal, as about two-thirds of patients with atherosclerotic cardiovascular (CV) disease (CVD) do not attain optimal low-density lipoprotein cholesterol (LDL-C) levels, despite statin treatment. 1 The percentage of lipid goal attainment has been reported to be much lower in certain ethnic subgroups. 2 A recent (2022) retrospective United Kingdom study characterizing the adult primary care patient population (n = 279 221) with primary hypercholesterolemia (PH)/mixed dyslipidemia (MD), indicated that over a mean of 8.6 years, the basal prevalence (proportion of the population) of PH/MD almost doubled from 13.5% in 2009 to 23.5% by 2019. 3 The incidence (number of new cases) was reduced from 176 to 49 per 100 000 population. Average age of the cohort was 58 years, baseline LDL-C was 4.32 mmol/L (167 mg/dL), approximately 20% had atherosclerotic CVD, 30% diabetes mellitus (DM), and 8.5% heterozygous familial hypercholesterolemia. Respective LDL-C decreases of 40% and 50% were attained in 2.6% and 2.3% of patients. Most were administered moderate-intensity statins as single therapy (62%); high-intensity statins were employed less often (24% as commencing therapy), while <10% of patients had ezetimibe together with statins of various intensities. Thus, a high percentage of patients with PH/MD who are at increased CV risk, are less than optimally managed regarding lipid reducing, and may have CV events with attendant important clinical and financial consequences. Therefore, despite recommendations for intensive LDL-C reduction, these are not applied in the clinical arena in the broader population.
Statins have been at the center stage of hypolipidemic therapies. Unfortunately, statins have been plagued by the occurrence, or fear of the occurrence, of several side-effects or adverse actions, which have partly hindered their more widespread use. 4 Besides statins, 5 new lipid-lowering therapeutic modalities have been documented to further lower CVD risk comprising ezetimibe, proprotein convertase subtilisin/kexin type 9 inhibitors (PCSK9i), icosapent ethyl (found effective in reducing triglycerides rather than cholesterol),5,6 bempedoic acid, an ATP citrate lyase inhibitor (reduces LDL-C), 7 and inclisiran, a new small interfering RNA treatment that blocks the generation of the PCSK9 protein. 8 Of these, ezetimibe is an oral agent, in contrast to parenteral PCSK9i and inclisiran, and has been the most convenient and/or affordable regimen, with PCSK9i being the most expensive class of drugs and inclisiran the next most expensive agent. 9 Importantly, the latter still remains untested; the outcome of a broad-scale trial of the influence of inclisiran on CV course is anticipated in 2026. 9 Robust data also attest to the success of ezetimibe in decreasing CVD risk and events.10,11 The recently added, bempedoic acid, an ATP citrate lyase inhibitor that reduces LDL-C concentrations with reported low incidence of muscle-related adverse events among statin-intolerant persons, has been recently found to decrease the occurrence of major adverse CV events (MACE). 12 However, this agent was also found, according to a recent meta-analysis, to increase the occurrence of gout, cholelithiasis, and renal impairment. 13 Furthermore, the drug is still more expensive than statins, has no generic alternative and a 30-day supply can cost >$400. 9
A recent study assessed the utility of hypolipidemic drugs in 728 423 persons with atherosclerotic CVD from 89 US health systems (2018-2021) employing electronic health records. 1 As of 2021, only 6% of atherosclerotic CVD individuals were receiving ezetimibe, 1.6% were on a PCSK9i, and 1.3% on icosapent ethyl, with use only slightly rising during the trial duration. The authors concluded that dealing with the underusage of non-statin lipid-reducing treatment for secondary prevention is a crucial point in closing the management hiatus of patients with remaining risk of atherosclerotic CVD. Non-statin lipid-lowering therapies do reduce residual atherosclerotic CVD risk. Thus, increasing use of these therapies is critical to improving this treatment gap. Furthermore, the benefits of LDL cholesterol lowering, even beyond current recommendations, are also apparent in patients at lower CV risk and younger age, indicating a need for earlier and effective intervention. 14
For this narrative review, a comprehensive review of the literature was conducted and the outcome of trials examining the usage of ezetimibe, as mono- or combined treatment in patients with hyperlipidemia and/or CVD, including several subgroups, are discussed. Also, safety and cost-efficacy issues and current guidelines are detailed. A summary of the results of meta-analyses of studies employing ezetimibe is presented in Table 1.15 -27 The mechanisms of action of ezetimibe vis-a-vis those of statins are pictorially illustrated (Figure 1). Finally, a holistic lipid approach strategy is suggested.
Large meta-analyses on use of ezetimibe.
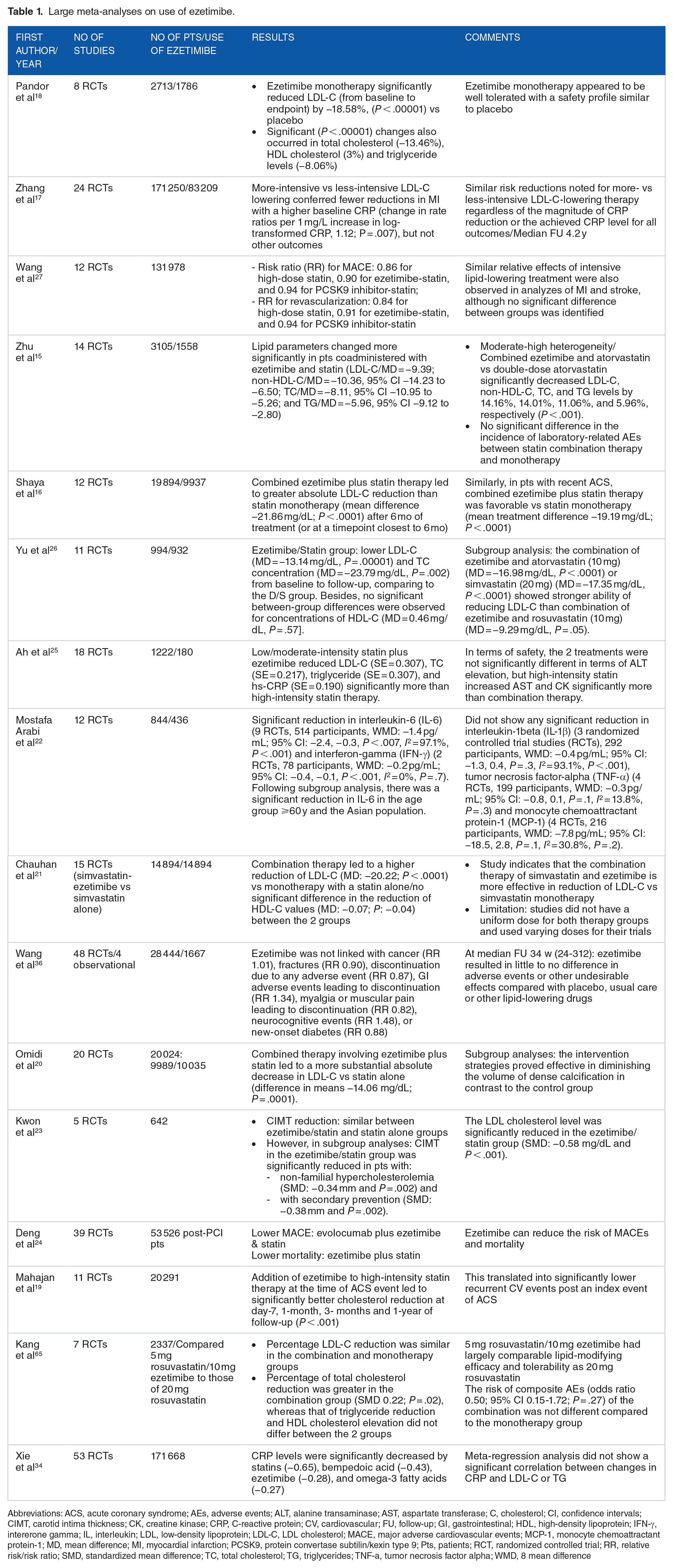
Abbreviations: ACS, acute coronary syndrome; AEs, adverse events; ALT, alanine transaminase; AST, aspartate transferase; C, cholesterol; CI, confidence intervals; CIMT, carotid intima thickness; CK, creatine kinase; CRP, C-reactive protein; CV, cardiovascular; FU, follow-up; GI, gastrointestinal; HDL, high-density lipoprotein; IFN-γ, intererone gamma; IL, interleukin; LDL, low-density lipoprotein; LDL-C, LDL cholesterol; MACE, major adverse cardiovascular events; MCP-1, monocyte chemoattractant protein-1; MD, mean difference; MI, myocardial infarction; PCSK9, protein convertase subtilin/kexin type 9; Pts, patients; RCT, randomized controlled trial; RR, relative risk/risk ratio; SMD, standardized mean difference; TC, total cholesterol; TG, triglycerides; TNF-a, tumor necrosis factor alpha; WMD, 8 mean difference

The schema demonstrates the mechanisms of action of ezetimibe vis-a-vis those of statins. The main action relates to inhibition of cholesterol absorption and reduction of its overall delivery to the liver, thus promoting the synthesis of LDL receptors with consequent reduction of serum LDL-cholesterol. The primary target of action seems to be the cholesterol transport system Nieman Pick C1 like protein (NPC1L1). Ezetimibe also demonstrates anti-inflammatory, immunomodulatory and antioxidative properties.
Mechanism of Action
Ezetimibe is a selective inhibitor of the Niemann-Pick C 1-Like 1 protein (NPC1L1P) transporter on enterocytes, leading to a reduction of the intestinal cholesterol absorption (Figure 1).28,29 A meta-analysis of 8 short-term RCTs demonstrated that after 12 weeks of ezetimibe monotherapy, a significant LDL-C lowering of 15% to 22% was noted; the drug was well tolerated with a safety profile similar to placebo. 18 Importantly, ezetimibe has low pharmacokinetic interactions with medications metabolized by cytochrome P450 with low incidence of muscle side-effects.
Ezetimibe also exhibits anti-inflammatory and immunomodulatory actions and affects the expression of specific antigens.30 -33 A recent meta-analysis indicated that ezetimibe significantly decreased C-reactive protein (CRP) levels by ~30%, independently of cholesterol reductions. 34 It is quickly absorbed from the gastrointestinal tract, then glucuronidated to produce the active metabolite; it also goes through substantial enterohepatic circulation. 30 Several genetic polymorphisms appear to affect the pharmacokinetics of ezetimibe. This agent also has an intricate influence on cytochrome P450 enzymes, as it is a metabolism-dependent inhibitor of cytochrome CYP3A4. 30 Ezetimibe does not have any clinically important interactions with fibrates, statins, or mipomersen. 30
Adverse Events
There have been some concerns about a potential increase of intestine cancer and/or breast cancer by ezetimibe. 35 However, a recent meta-analysis confirmed ezetimibe’s safety in 28 444 participants over a median of 5 months (48 randomized controlled trials—RCTs) and in 1667 participants over a median of 40 months (4 observational studies), where the drug was not associated with cancer (relative risk—RR 1.01), fractures (RR 0.90), discontinuation due to adverse event (RR 0.87), gastrointestinal event (RR 1.34), myalgias (RR 0.82), neurocognitive events (RR 1.48), or new-onset DM (RR 0.88). 36 The authors concluded that ezetimibe does not differ much from placebo, standard care or other lipid-reducing agents in untoward effects or other adverse events.
Monotherapy
As mentioned, non-statin lipid-lowering drugs are efficacious in lowering atherosclerotic CVD risk. 6 However, usage of non-statin therapies is low for secondary prevention. Furthermore, there has been limited uptake of non-statin therapies over the last several years. Increasing use of these therapies is important to improve the treatment gap, as almost two-thirds of patients with atherosclerotic CVD do not attain target LDL-C despite statin treatment.
As also mentioned, a recent report evaluating the employment of hypolipidemic agents in 728 423 persons with atherosclerotic CVD in the US indicated that as of 2021, only 6% of atherosclerotic CVD patients were receiving ezetimibe, 1.6% were receiving a PCSK9i, and 1.3% were on icosapent ethyl, pointing to the suboptimal usage of non-statin lipid-reducing treatment. 1
Early data on use of ezetimibe with meta-analysis including only short-term studies in hypercholesterolemia, showed considerable potentially positive alterations in lipid and lipoprotein concentrations compared to baseline following ezetimibe single therapy. 18 Ezetimibe alone and also combined with simvastatin has been demonstrated to lower the atherogenic small, dense LDL level in patients with DM. 37 Ezetimibe can also positively influence the spread of LDL subfractions, particularly in patients with high triglyceride levels. 38 Furthermore, combined therapies, such as those commonly employed in patients with acute myocardial infarction (MI), influence the quality of LDL particles. 39 Indeed, a link was shown of simvastatin/ezetimibe plus either clopidogrel or ticagrelor with less oxidized LDL, while simvastatin/ezetimibe with ticagrelor lowered the amount of cholesterol in the atherogenic subfractions of LDL, whereas rosuvastatin with ticagrelor was the only combined regimen linked with higher LDL size. 39
In clinical trials, single therapy with ezetimibe (10 mg/d) has been demonstrated to lower LDL-C in hypercholesterolemic patients by 15% to 22% with relatively increased interindividual fluctuation.40,41 A meta-analysis of 8 RCTs (all 12 weeks duration, n = 2722) indicated that ezetimibe monotherapy conferred an important mean reduction in LDL-C (from baseline to endpoint) of −18.6%, (P < .00001) versus placebo. 18 In addition, considerable (P < .00001) alterations were also detected in total cholesterol (−13.5%), high density lipoprotein cholesterol (HDL-C) (3%) and triglyceride concentrations (−8.1%). Single treatment with ezetimibe seemed to be tolerated well with a profile of safety which was akin to placebo. Of course, another setting where ezetimibe is used as monotherapy includes patients with intolerance to statins.42,43
However, ezetimibe has been mostly employed as an adjunctive therapy added to standard statin treatment. Nevertheless, there may also be an occasional patient with familial hypercholesterolemia who has an excellent response to this agent. 44 As mentioned, NPC1L1 is the molecular goal of ezetimibe. Detailed analysis of the NPC1L1 gene and proteome from a super responder to ezetimibe regarding the role of NPC1L1 mutations pointed to an intricate series of events in which the joint mutations were demonstrated to influence cholesterol uptake when ezetimibe was present. 44 Proteomic analysis suggested that the super response may also be explained by the type of interactions with the proteins of the cytosol.
Ezetimibe as Part of Combined Therapy/Adjunct to Statin Therapy
For patients needing a more aggressive lowering in cholesterol to aid in averting CVD, a statin can be combined with ezetimibe to enhance its efficacy. 45 Indeed, ezetimibe is often the initially added therapy to reach LDL-C goals. It decreases LDL-C by approximately an extra 20% and has a superb safety and tolerability profile. CV outcomes data indicate that such a combination therapy is a strong and by and large well-tolerated combination of drugs that increases the spectrum of choices at hand for the pharmacological treatment of hypercholesterolemia in adults, permitting more patients to attain their LDL-C target.46 -48 Combined therapy is also a choice to manage very-high-risk patients who cannot attain LDL-C goals with statin single therapy. 49
Data from RCTs indicate that all fixed-dose combinations of ezetimibe/statin considerably improve lipid patterns in patients with hypercholesterolemia versus statin monotherapy. 50 In all groups of patients managed with different doses of statin combined with a standard 10 mg-dose of ezetimibe, a reduction in mean LDL-C concentration of >50% has been noted. The safety and tolerability of ezetimibe/statin treatment seem to be equivalent with those of statin single therapy.
Among patients with atherosclerotic CVD, moderate-intensity statin with ezetimibe combination treatment was non-inferior to high-intensity statin single therapy for the 3-year composite results with a greater percentage of patients with LDL-C concentrations of <70 mg/dL and less intolerance-associated drug withdrawal or dose lowering.
51
In this context, a multicenter Korean RCT randomized 3780 patients to take either moderate-intensity statin dose (rosuvastatin 10 mg) with ezetimibe (10 mg) combination treatment (n = 1894) or high-intensity statin single-therapy (rosuvastatin 20 mg; n = 1886).
51
Over 2 years,
In patients with acute coronary syndrome (ACS) (n = 18 144), ezetimibe, when added to statin treatment, produced an incremental reduction of LDL-C concentrations and ameliorated CV outcomes. 52 Moreover, lowering LDL-C to concentrations below prior goals furnished extra advantage. Specifically, over a median of 6 years, the median time-weighted average LDL-C concentration during the trial was 53.7 mg/dL in the simvastatin-ezetimibe subset versus 69.5 mg/dL in the simvastatin-single therapy subset (P < .001). The Kaplan-Meier event rate for the primary end-point (composite of CV mortality, nonfatal MI, unstable angina needing rehospitalization, coronary revascularization at ⩾30 days after random allocation, or nonfatal cerebrovascular accident—CVA) at 7 years was 32.7% in the simvastatin-ezetimibe subset, versus 34.7% in the simvastatin-monotherapy subset (P = .016). Percentages of prespecified gallbladder, hepatic and muscle side-effects and cancer did not differ in the 2 subsets.
A post-hoc analysis of a subgroup of patients of the RACING (RAndomized Comparison of Efficacy and Safety of Lipid-lowerING With Statin Monotherapy vs Statin/Ezetimibe Combination for High-risk Cardiovascular Diseases) study comprising those who underwent percutaneous coronary intervention (PCI) (2497 patients, 67%, median 64 years, 79% men), who had increased rates of the primary endpoint (3-year composite of CV mortality, major CV events, and nonfatal cerebrovascular accident) (HR, 1.34; P = .014), indicated that moderate-intensity statin treatment combined with ezetimibe versus high-intensity statin treatment did not raise the occurrence of the primary endpoint (HR, 0.95; P = .781). 53 The proportion of patients with LDL-C <70 mg/dL at 1, 2, and 3 years was 74%, 76%, and 73%, respectively, in the combined treatment population, and was considerably greater than that in the high-intensity statin single-therapy subset (57%, 62%, and 59%, respectively, all P < .001). Withdrawal of lipid-reducing agents was noted less often in the combined therapy subset (4.2 vs 7.6%, P = .001). The authors concluded that the actions of ezetimibe combination treatment noted in the RACING trial were consistently maintained in patients with atherosclerotic CVD after PCI.
A meta-analysis was performed of 26 RCTs (n = 23 499) that compared ezetimibe versus placebo or ezetimibe plus other lipid-altering agents versus other lipid-altering agents alone in adults, with or without CVD, and who were followed-up for ⩾1 year. 54 The findings were driven by the largest study (IMProved Reduction of Outcomes: Vytorin Efficacy International Trial; IMPROVE-IT), which had weights ranging from 41.5% to 98.4% in the different meta-analyses. The authors concluded that ezetimibe with statins most likely mitigates the occurrence of major adverse CV events (MACE) versus statins alone (risk ratio—RR 0.94, 10 studies; moderate-quality evidence). Trials reporting on all-cause mortality with use of ezetimibe with statin or fenofibrate, found that these agents have little or no effect on this outcome (RR 0.98). 54 Combining ezetimibe with statins most likely lowers the occurrence of non-fatal myocardial infarction (MI) (RR 0.88, 21 145 participants; 6 studies; moderate-quality evidence) and non-fatal CVA (RR 0.83, 21 205 participants; 6 studies; moderate-quality evidence). 54 Trials reporting CV mortality with ezetimibe added to statin or fenofibrate, concluded that such regimen had little or no effect on this outcome (RR 1.00, 19 457 participants; 6 studies; moderate-quality evidence). 54 The need for coronary revascularisation might be decreased by combining ezetimibe with a statin (RR 0.94, 21 323 participating individuals; 7 studies); however, with no difference in coronary revascularisation rate noted when a sensitivity analysis was restricted to trials with a reduced risk of bias. 54 Regarding safety, ezetimibe added to statins may make little or no difference in the occurrence of liver disease (RR 1.14), while it is doubtful whether ezetimibe influences the incidence of myopathy (RR 1.31, 95% CI 0.72-2.38; 20 581 participants; 3 studies; very low-quality evidence) and rhabdomyolysis, granted the broad confidence intervals (CIs), and low event rate. 54 The authors concluded that moderate- to high-quality proof indicates that ezetimibe has modest advantageous influence on the occurrence of CVD endpoints, mainly led by a decrease in non-fatal MI and non-fatal CVA, but it has little or no influence on clinical fatal endpoints. The lowering of LDL-C, total cholesterol and triglycerides might account for the CV benefit of ezetimibe. The evidence is not sufficient to determine whether ezetimibe raises the risk of harmful events due to the low and very low quality of the evidence. The evidence for benefit was mainly acquired from patients with established atherosclerotic CVD (predominantly ACS) who were prescribed ezetimibe plus statins. However, the evidence is insufficient about the role of ezetimibe in primary prevention and the results of ezetimibe monotherapy in the prevention of CVD, and these topics thus need further investigation.
In summary, in patients with CVD, ezetimibe with a statin lowers MACE but has no influence on total and CV death rate, compared with a statin alone (strength of recommendation, A). 54 In adults with CVD, ezetimibe combined with a moderate-intensity statin (rosuvastatin 10 mg) was noninferior at lowering CV death, MACE, and nonfatal CVA, but was tolerated better, versus a high-intensity statin (rosuvastatin 20 mg) alone (strength of recommendation, B; 1 RCT). 54
Another meta-analysis of 8 RCTs (N = 19 558) of ezetimibe plus statin versus statin alone that monitored patients for a minimum of 6 months and provided information on ⩾1 end-points of total death rate, CV mortality, non-fatal MI, and non-fatal CVAs, indicated that ezetimibe added to moderate-dose statins can possibly lead to 17 fewer MIs and likely 6 fewer CVAs/1000 managed over 0.5 to 6 years but is not likely to lower total death rate or CV death. 55 The authors suggested that patients who value a small absolute decrease in MI and do not object to using an extra drug over a long period may choose ezetimibe besides statin therapy. In this analysis there were no increased specific harms conferred by ezetimibe added to statins. Importantly, in this review, the data from the IMPROVE-IT trial constituted the dominating evidence regarding sample size and number of events (responsible for >90%) among all pertinent trials comparing ezetimibe plus statin versus statin alone.
The extra action of ezetimibe to a high-potency statin regimen (eg, rosuvastatin) helps to attain cholesterol targets in a bigger number of high-risk patients, 56 while alleviating some safety concerns regarding high doses of statin treatment. 57 In addition, ezetimibe enhances rosuvastatin triglyceride-reducing and anti-inflammatory actions. 57
Finally, a recent meta-analysis of 17 articles compared the effects of statin-ezetimibe combined treatment and statin single therapy on lipid and glucose indices in patients with DM. 58 In the efficacy evaluation, the combined therapy led to a significantly larger decrease in LDL-C than did statin single therapy (standard difference in means 0.69). A larger ameliorating action was noted in the concentrations of total cholesterol, HDL-C, triglyceride, and apolipoprotein B, but not apolipoprotein A1, with combined treatment than with statin monotherapy. Furthermore, combined treatment decreased fasting blood glucose concentrations more so than did statin monotherapy. Regarding safety, there were no considerable differences in therapy-linked side-effects between the 2 therapies.
Taking into consideration the superb safety pattern and absence of clinically pertinent drug-drug interactions, the statin/ezetimibe combination is a useful alternate therapy to the up-titration of a statin dose.
Ezetimibe has also been used in combination with orlistat, an anti-obesity drug, which prevents the absorption of dietary fats by acting as a lipase inhibitor; the combined orlistat/ezetimibe regimen had a more advantageous influence on LDL-C and small dense LDL-C concentrations in overweight and obese patients with hypercholesterolemia than either agent alone. 59
Finally, ezetimibe has been suggested to be combined with bempedoic acid in an attempt to increase the proportion of patients attaining LDL cholesterol goals. 60 indeed, bempedoic acid has been heralded as a potent therapeutic ally, capable of significantly lowering LDL-C levels and curtailing CV events. 61 Its favorable safety profile attests to its suitability, particularly among persons with statin intolerance or those belonging to a high-risk vascular group, pointing to a paradigm shift in our lipid management approaches.
Moderate-Intensity Statin With Ezetimibe versus High-Intensity Statin
A post-hoc analysis of the RACING study suggested that moderate-intensity statin combined with ezetimibe demonstrated similar CV advantages with those of high-intensity statin single therapy with reduced intolerance-linked drug withdrawal or dose lowering in elderly patients with atherosclerotic CVD who have an increased risk of intolerance, noncompliance, and withdrawal with high-intensity statin treatment. 62 Thus, one can avoid the poor compliance with and worries concerning high-intensity statin treatment by using combined moderate-intensity statin and ezetimibe, at least for the elderly subpopulation. High-intensity statin therapy incurs a higher risk of intolerance, noncompliance, and drug withdrawal. 63
A recent meta-analysis of 11 trials (N = 20 291) indicated that the addition of ezetimibe to high-intensity statin therapy at the time of an ACS event leads to significantly enhanced cholesterol reduction at 1-week, 1-month, 3- months and 1-year of follow-up, which translates into a significantly lower recurrent CV events after an index event of ACS. 19 Interestingly, a recent propensity-matched nationwide cohort study indicated that among individuals without pre-existing CVD, combined moderate-intensity statin and ezetimibe were superior to high-intensity statin monotherapy in preventing composite outcomes as well as each of MI and stroke, whereas low-intensity statin with ezetimibe combination reduced the risk of composite but not individual outcomes. 64
A most recent meta-analysis of 7 trials (N = 2337) employing rosuvastatin/ezetimibe (5/10 mg) versus rosuvastatin alone (20 mg) indicated that these 2 regimens had comparable lipid-lowering efficacy and tolerability. 65 The percentage of total cholesterol reduction was greater in the combination group (SMD 0.22; P = .02), whereas that of triglyceride reduction and HDL cholesterol elevation was similar between the 2 groups.
Adding Ezetimibe Versus a PCSK9 Inhibitor to a Statin Regimen
A systematic review and network meta-analysis comprised 14 trials evaluating ezetimibe and PCSK9 inhibitors among 83 660 adult patients receiving statins. 66 The addition of ezetimibe to statins decreased MI (RR 0.87) and stroke (RR 0.82) but not all-cause death rate (RR 0.99) or CV mortality (RR 0.97). Likewise, adding a PCSK9 inhibitor to statins decreased MI (RR 0.81) and stroke (RR 0.74) but not all-cause (RR 0.95) or CV death rate (RR 0.95). Among adult patients with very high CV risk, the addition of a PCSK9 inhibitor was possibly able to decrease MI (16 per 1000) and stroke (21 per 1000) (moderate to high certainty); while with the addition of ezetimibe it was possible to decrease stroke (14 per 1000), but the decrease of MI (11 per 1000) (moderate certainty) did not succeed to attain the minimal important difference (MID) of 12 per 1000. The addition of ezetimibe to PCSK9 inhibitor and statin may decrease stroke (11 per 1000), but the lowering of MI (9 per 1000) (low certainty) did not attain MID. The addition of PCSK9 inhibitors to statins and ezetimibe may decrease MI (14 per 1000) and stroke (17 per 1000) (low certainty). Among adults with elevated CV risk, the addition of a PCSK9 inhibitor most likely lowered MI (12 per 1000) and stroke (16 per 1000) (moderate certainty); the addition of ezetimibe likely decreased stroke (11 per 1000), but the decrease in MI did not reach MID (8 per 1000) (moderate certainty). The addition of ezetimibe to PCSK9 inhibitor and statins did not influence the results beyond MID, while the addition of a PCSK9 inhibitor to ezetimibe and statins may decrease stroke (13 per 1000). These influences were consistent in patients who could not tolerate statins. Among the groups of moderate and low CV risk, the addition of a PCSK9 inhibitor or ezetimibe to statins had little or no advantage for MI and stroke. The authors concluded that ezetimibe or PCSK9 inhibitors may lower non-fatal MI and stroke in adult patients who are at very high or high CV risk and are having maximally tolerated statin treatment or who do not tolerate statins, but not in those with moderate and low CV risk. Hence, one can consider adding first ezetimibe to statin, before thinking of resorting to more expensive therapies involving injections.
In keeping with the above results, an international panel recently made some recommendations, mostly weak recommendations, meaning that one has to trust and be guided by shared decision making when following these guidelines. 67 For adults already employing statins, the panel proposes the addition of a second lipid-lowering agent in individuals at very high and high CV risk but advises against adding it in individuals at low CV risk. For adults who do not tolerate statins, the panel suggests employing a lipid-reducing agent in people at very high and high CV risk but advises against its addition in those at reduced CV risk. When opting to include another lipid-reducing agent, the panel recommends ezetimibe in lieu of PCSK9 inhibitors. The panel proposes further the additional use of a PCSK9 inhibitor to ezetimibe for adults already receiving statins and who are at very high risk and also for those at very high and high risk who do not tolerate statins. The panel considered a strong recommendation for the addition of either ezetimibe or PCSK9 inhibitors in persons at high and very high CV risk as reflecting a definite advantage. The panel emphasized shared decision making and recognized that drug availability and costs will affect decisions when healthcare systems, clinicians, or persons decide on adding ezetimibe or PCSK9 inhibitors.
Patient Subgroups
Stroke
An RCT (Treat Stroke to Target—TST) examined patients with ischemic atherosclerotic CVA in the preceding 3 months or transient ischemic attack within the preceding 15 days who were randomly assigned to an LDL-C goal of <70 or 100 ± 10 mg/dL, employing statin and/or ezetimibe as necessary. 68 Among 2860 participating patients, those who were on double treatment during the study in the lower goal group had a higher baseline LDL-C versus patients on statin single therapy (141 ± 38 vs 131 ± 36, respectively, P < .001). In patients on double treatment and on statin single therapy, the attained LDL-C was 66.2 and 64.1 mg/dL respectively, and the primary outcome was lowered by double treatment versus the higher goal group (HR, 0.60; P = .016) but not by statin single therapy (HR, 0.92; P = .52), with no considerable rise in intracranial hemorrhage. The authors concluded that aiming for a target of an LDL-C of <70 mg/dL with combined statin and ezetimibe versus 100 ± 10 mg/dL consistently lowered the risk of next CVA.
A recent RCT assigned 584 patients with recent ischemic CVA <90 days to rosuvastatin/ezetimibe 10/10 mg qd (ROS10/EZT10) or to rosuvastatin 20 mg qd (ROS20) (the modified intention-to-treat analysis comprised 530 patients). 69 The baseline LDL-C concentration was 130.2 ± 34.7 mg/dL in the ROS10/EZT10 subset and 131.0 ± 33.9 mg/dL in the ROS20 subset. The primary endpoint (LDL-C lowering ⩾50% from baseline at 90 days) was attained in 198 patients (72.5%) in the ROS10/EZT10 subset and 148 (57.6%) in the ROS20 subset (odds ratio—OR 1.944; P = .0003). LDL-C concentration <70 mg/dL was attained in 80.2% and 65.4% in the ROS10/EZT10 and ROS20 subsets, respectively (P = .0001). Multiple lipid target attainment rate was 71.1% and 53.7% in the ROS10/EZT10 and ROS20 subsets (P < .0001). Major vascular events were recorded in 1 patient in the ROS10/EZT10 subset and 9 in the ROS20 subset (P = .0091). The untoward event rates were similar in the 2 groups. The authors reached the conclusion that moderate-intensity rosuvastatin plus ezetimibe superseded high-intensity rosuvastatin alone for intensive LDL-C lowering in patients with recent ischemic CVA. With the combined treatment, >70% of patients attained LDL-C lowering ⩾50% and 80% had an LDL-C <70 mg/dL at 3 months.
Ezetimibe, added to a statin, improves surrogate markers of vascular risk and also significantly decreases vascular events, possibly by decreasing vascular inflammation. 70 It is the lowering of LDL-C, which is more effective with added ezetimibe, which leads to a decrease or inhibited progression of the carotid intima-media thickness (cIMT), when the drop in LDL-C is considerable and the pre-therapy measurement of the cIMT is adequate to permit a sizeable reduction to take place. 71
Diabetes
The hypolipidemic treatment goal has been stricter in patients with DM, whereby an LDL-C reduction to <70 mg/dL is recommended for patients with DM at high risk and to <55 mg/dL for patients at very high risk or at least an LDL-C lowering of ⩾50% from baseline. 41 Combined therapy with statin and ezetimibe is advised if the goal is not reached with statin alone.
As mentioned, the additive influence of ezetimibe to a high-potency statin regimen (eg, rosuvastatin) helps to attain cholesterol targets in a big number of high-risk patients, such as patients with DM, while mitigating some safety problems concerning high dosages of statin treatment. 57 Importantly, patients with DM have extra advantages from ezetimibe therapy as they appear to absorb cholesterol more efficiently than non-diabetic ones, due to higher NPC1L1 gene expression. 57 Indeed, the expression of intestinal NPC1L1 cholesterol transporter has been demonstrated to be higher in patients with disorders linked with hypercholesterolemia such as DM. 72 Experimental studies have indicated that elevated levels of glucose directly enhance the expression of NPC1L1 in intestinal epithelial cells; glucose seems to directly regulate NPC1L1 expression via transcriptional pathways and the engagement of phosphatase-dependent mechanisms. 72
As mentioned, a meta-analysis of 17 articles compared the impact of statin-ezetimibe combined treatment and statin single therapy on lipid and glucose indices in patients with DM. 58 In the efficacy assessment, combined treatment led to a considerably larger decrease in LDL-C than did statin single therapy, and also improved concentrations of total cholesterol, HDL-C, triglyceride, and apolipoprotein B, but not apolipoprotein A1. Furthermore, combined treatment led to a larger decrease in fasting blood glucose concentrations than did statin monotherapy without important differences in therapy-related side-effects between the 2 therapies.
Statins have been linked with an elevated risk for DM. 73 A study Shah et al 74 assessed the occurrence of new-onset DM with ezetimibe in combination with simvastatin versus placebo plus simvastatin in 9500 patients completing the study out of 18 144 patients enrolled in IMPROVE-IT (Improved Reduction of Outcomes: Vytorin Efficacy International Trial). 52 After random allocation, 1414 patients (14.9%) developed new-onset DM with an annual occurrence of ~2.7%/year. 74 For those who had new-onset DM, baseline features did not differ between those who took simvastatin/ezetimibe (n = 720) and those who had simvastatin/placebo (n = 694). When patients who did not manifest DM (n = 8086) were compared with those with DM, the latter had a higher likelihood at baseline to have a greater body mass index (BMI), higher systolic blood pressure, higher triglycerides, and ⩾3 elements of the metabolic syndrome. There were no important differences in baseline medical treatments or LDL-C between any of the subsets. Compared with the placebo/simvastatin group, there was no difference in the occurrence of new-onset DM when ezetimibe was added to simvastatin (HR, 1.03). This association remained qualitatively the same across sensitivity analyses using different new-onset DM definitions. Adding ezetimibe did not influence the likelihood of new-onset DM in either statin-naïve or statin-experienced (having statin in the past) patients. Furthermore, adding ezetimibe did not influence the risk in those with or without metabolic syndrome. Thus, in this assessment of patients participating in IMPROVE-IT, there was no elevation in the likelihood of new-onset DM with ezetimibe added to statin treatment. These data are in keeping with the outcome of a recent trial which reported a reduced risk of new-onset DM with reduced dose of rosuvastatin and ezetimibe versus increased dose of rosuvastatin. 51 Similar absence of new onset DM has been reported with PCSK9 inhibitors. 75 These results back the employment and lend support to the safety of ezetimibe as an addition to statins for more LDL-C lowering without the concern of aggravating the glycemic status.
To further ease the concern of a statin implicated in the risk of DM, a pre-specified, stratified subset analysis of the DM group in the RACING trial supports the use of moderate- rather than high-intensity statin and ezetimibe. 76 The primary outcome was a 3-year composite of CV death, MACE or non-fatal CVA. Among all patients, 1398 (37%) had DM at baseline. The occurrence of the primary outcome was 10% and 11.3% among patients with DM allocated to ezetimibe combined treatment versus single therapy with high-intensity statin (HR: 0.89; P = NS). Drug withdrawal or dose lowering of the trial agent related to intolerance was reported in 5.2% and 8.7% of patients in each subset, respectively (P = .014). LDL-C concentrations <70 mg/dL at 1, 2, and 3 years were noted in 81%, 83%, and 80% of patients in the ezetimibe combined treatment subset, and 64%, 70%, and 67% of patients in the high-intensity statin single-therapy subset (all P < .001). In the whole study group, no important interactions were encountered between DM state and treatment with regards to primary outcome, drug withdrawal or dose lowering related to intolerance, and the percentage of patients with LDL-C values <70 mg/dL. The authors concluded that the results of the ezetimibe/statin combination treatment noted in the RACING trial patients are maintained among patients with DM. These results support combined therapy with moderate-intensity statin and ezetimibe as an alternative to high-intensity statins if patients cannot tolerate these agents, or additional lowering in LDL-C is needed among patients with DM and atherosclerotic CVD.
Chronic kidney disease (CKD)
An early (2011) RCT (Study of Heart and Renal Protection—SHARP) 77 examined the influence of lipid reducing regimen with combined simvastatin and ezetimibe in 9270 patients (mean age 61 years; 67% men, 20% with DM, one-sixth with vascular disease) with late-stage CKD (creatinine ⩾1.7 mg/dL in males or ⩾1.5 mg/dL in females) with no known history of MI or coronary revascularization, of whom 3023 were on dialysis, randomly allocated to simvastatin 20 mg plus ezetimibe 10 mg daily (n = 4650) versus matching placebo (n = 4620). Allocation to simvastatin plus ezetimibe led to an average LDL cholesterol difference of 15.3 mg/dL during a median of 4.9 years and to a 17% relevant decrease in major atherosclerotic events (11.3% vs 13·4% placebo; rate ratio—RR 0.83; log-rank P = ·0021). 78 Fewer patients (albeit non-significantly) assigned to simvastatin plus ezetimibe had a non-fatal MI or succumbed to CAD (4.6% vs 5%; RR 0.92, P = .37); there were significantly lower rates of non-hemorrhagic CVA (2.8% vs 3.8%; RR 0.75; P = .01) and arterial revascularisation procedures (6.1% vs 7.6%; RR 0·.9; P = .0036). The extra risk of myopathy was only 2 per 10 000 patients per year of combined therapy (0.2% vs 0.1%). There were no data of excessive risk of hepatitis, cholelithiasis, or cancer and there was no considerable increase in non-vascular death. The authors concluded that lowering of LDL cholesterol with simvastatin 20 mg plus ezetimibe 10 mg daily safely decreased the occurrence of major atherosclerotic events in a broad range of patients with late-stage CKD. A subsequent supplementary analysis of the SHARP trial reported on the influence of treatment on total non-vascular serious adverse events, by system of disease, by baseline features, and by length of follow-up. 79 Over a median of 4.9 years, equal numbers of patients in the 2 groups had at least 1 non-vascular serious adverse event (76.4% vs 76.6%; risk ratio—RR 0.99). The relative risk of any nonvascular serious adverse event did not differ considerably among specific prognostic subsets or by length of follow-up.
A recent nationwide retrospective population-based cohort trial analyzing data from the Korean National Health Insurance Service comprised 17 242 adult patients (aged ⩾18 years) having chronic dialysis who had a first atherosclerotic CVD event in the time period of 2013 to 2018, of whom 9611 (55.7%) patients were using statin. 80 The total prevalence of statin therapy rose from 53% in 2013 to 58% in 2018; most (77%) of dialysis patients were given moderate-intensity statins. The percentages of low- or moderate-intensity statin use were equivalent, but use of high-intensity statin rose from 5.7% in 2013 to 10.5% in 2018. The use of combined statin/ezetimibe regimen progressively rose since 2016. Statin use independently conferred a reduction in 1-year total death rate after correcting for confounders (HR 0.89, P = .004). Interestingly, the use of high-intensity statins and combined statin/ezetimibe regimens steadily rose in dialysis patients with CVD. Furthermore, the combined statin/ezetimibe regimen was shown to be more efficacious than statin single therapy in these patients, which has likely led to a rise in the usage of high-intensity statins and combined statin/ezetimibe regimens in dialysis patients with atherosclerotic CVD.
A recent meta-analysis of 7 trials (8 articles, 14 016 patients) reviewed the efficacy and safety of statin plus ezetimibe treatment in patients with CKD. 81 The combined statin/ezetimibe regimen conferred beneficial influences on plasma total cholesterol (weighted mean difference—WMD—20.31 mg/dL, P < .001), LDL-C (WMD—17.22 mg/dL, P < .001), and triglycerides (WMD—15.08 mg/dL, P < .001) versus statin single therapy. Combined statin and ezetimibe treatment significantly lowered total death rate and MACE (risk ratio 0.86, P = .01). The occurrence of adverse events was similarly low in the 2 therapy groups. The authors concluded that the combined statin/ezetimibe therapy considerably ameliorated plasma lipid levels and decreased risks of total mortality and MACE versus the control subset in patients with CKD.
Pediatric patients
Bile acid sequestrants (cholestyramine) had been the only possible pharmacological therapy for hypercholesterolemia in childhood for several years. 82 However, nowadays, statin treatment is the principal pharmacological management in this group as well. 83 In the United States, pravastatin can be employed from 8 years of age, while all other statins can be considered from 10 years of age. 82 In Europe, rosuvastatin has been granted approval in children from 6 years of age. 83 Statin treatment in childhood has been demonstrated to be safe. A recent trial has shown that the sooner statin treatment is initiated, the higher is the decrease of coronary artery disease (CAD) risk in adulthood. 84 Statin treatment should be commenced with the smallest advised dose, and increased as needed. LDL-C levels ⩽130 mg/dL from 10 years of age or a decrease of 50% of pre-therapy cholesterol concentrations in children aged 8 to 10 years are advised as a goal during statin therapy. 83 To reach this goal, in some cases ezetimibe will need to be added which has been granted approval in childhood commencing from 10 years of age in the USA and Europe, and is usually well tolerated with few untoward effects. 83 Of course, there is need to involve the family in this shared decision-making process; yet, there are particular adherence issues in this group.
A prospective study assessed LDL-C reducing effectiveness and tolerability of ezetimibe as single treatment (10 mg/d) in children and adolescents (5-15 years old) with polygenic hypercholesterolemia (PH) (n = 6) or familial hypercholesterolemia (FH) (n = 11) over a mean of 11 to 16 months. 85 Ezetimibe considerably decreased total cholesterol (TC) and LDL-C in patients with PH and FH: TC from 260.5 ± 12.4 to 180.0 ± 21.6 mg/dL (P = .02) and from 315.3 ± 41.8 to 233.3 ± 36.8 mg/dL (P = .003), respectively, and LDL-C from 177.1 ± 17.7 to 102.6 ± 16.7 mg/dL (P = .02) and from 243.0 ± 41.8 to 170.0 ± 29.8 mg/dL (P = .003), respectively. HDL-C only dropped considerably (from 58.1 ± 10.0 to 49.3 ± 9.1 mg/dL) (P < .01) in patients with FH and stayed the same in patients with PH. Triglyceride concentrations were not altered in both groups. No adverse effects were noted.
In a small retrospective series of 36 children and adolescents with hypercholesterolemia (26 with familial hypercholesterolemia, and 10 with familial combined hyperlipidemia), ezetimibe monotherapy was safe and efficacious in decreasing LDL-C concentrations, with no significant alteration in triglyceride or HDL-C concentrations. 86 Patients were kept on ezetimibe with no adverse actions ascribable to the drug for as long as 3.5 years.
Older patients
A multicenter RCT performed at 363 medical centers in Japan assessed the preventive ability of ezetimibe (10 mg qd) versus usual care with dietary counseling for 3796 patients older than 75 years, with high LDL-C with no history of CAD, over a median follow-up of 4.1 years. 87 After excluding 182 ezetimibe patients and 203 controls for various reasons, 1716 (90.4%) and 1695 (89.3%) patients underwent primary analysis, respectively. Ezetimibe lowered the occurrence of the primary outcome (composite of sudden cardiac death, MI, coronary revascularization, or CVA; hazard ratio—HR, 0.66; P = .002). With regards to the secondary outcomes, the occurrences of composite CV events (HR, 0.60; P = .039) and coronary revascularization (HR, 0.38; P = .007) were decreased in the ezetimibe subset than in the control subset; however, the occurrence of CVA, total death rate, or untoward events was equal between study groups. The authors concluded that LDL-C-reducing treatment with ezetimibe averted CV events, indicating the importance of LDL-C reduction for primary prevention in persons aged ⩾75 years with high LDL-C.
A post-hoc analysis of the RACING trial comprising 574 (15.2%) patients aged ⩾75 years indicated that the rates of the primary endpoint (a 3-year composite of CV death, MACE, or nonfatal CVA) did not differ between the moderate-intensity statin with ezetimibe combined treatment subset and the high-intensity statin single-therapy subset in older >75 years (10.6% vs 12.3%; HR: 0.87; P = .581) and younger <75 years patients (8.8% vs 9.4%; HR: 0.94; P = .570) (P for interaction = .797). 62 Moderate-intensity statin combined with ezetimibe conferred reduced rates of intolerance-related drug withdrawal or dose lowering among patients aged ⩾75 years (HR 2.3 vs 7.2%; P = .010) and those <75 years (HR 5.2 vs 8.4%; P < .001) (P for interaction = .159).
Patients with hyperuricemia
Uric acid is deemed a risk factor for CVD. 88 The influence of statins and ezetimibe on serum uric acid concentrations was examined in a prospective RCT where after 3-month dietary intervention, 153 patients (56 males) with hypercholesterolemia were placed on simvastatin/ezetimibe 10/10 mg or simvastatin 40 mg or rosuvastatin 10 mg. 89 At week 12, a significant decrease in serum uric acid concentrations was observed in all therapy subsets (simvastatin/ezetimibe 10/10 mg: −3.8%, simvastatin 40 mg: −5.7%, and rosuvastatin 10 mg: −3.8%; P < .05 compared with baseline; p = NS for comparison between subsets). Fractional excretion of uric acid was non-significantly elevated in all subsets. The decrease in serum uric acid concentrations correlated with the rise in fractional excretion of uric acid and baseline uric acid concentrations. Renal function indices and blood concentrations and fractional excretions of electrolytes stayed unaltered in all subsets. Alterations in plasma lipids were equivalent across various subsets. The authors concluded that simvastatin/ezetimibe 10/10 mg, simvastatin 40 mg, and rosuvastatin 10 mg exert a similar lowering effect in uric acid levels.
Other patient subgroups
Additional substudies of the IMPROVE-IT trial, both prespecified and post-hoc analyses, were conducted regarding particular very-high-risk subsets of patients, such as patients with prior acute events and/or coronary revascularization, patients aged >75 years, and patients with DM, CKD or non-alcoholic fatty liver disease, etc; all showed positive results for ezetimibe, as described elsewhere. 56 Thus, the data from IMPROVE-IT are reassuring with regards to longer-term safety and efficacy of the intensified lipid-lowering treatment in very-high-risk patients leading to very low LDL-C concentrations. 56 In keeping with the above, a secondary analysis of the IMPROVE-IT trial indicated that the greatest absolute risk reduction in older (>75 years) patients hospitalized for ACS was derived with use of simvastatin-ezetimibe compared with simvastatin monotherapy. Importantly, addition of ezetimibe to simvastatin did not confer any significant increase in safety issues among older patients. 90
Pleiotropic Effects/Markers of Inflammation/Atherosclerotic Plaque Composition
A recent systematic literature search examining the influence of ezetimibe on inflammatory markers, such as C-reactive protein (CRP), tumor necrosis factor alpha (TNFα) and Interleukin 6 (L-6), when administered in addition to statin therapy, indicated that overall ezetimibe curtails inflammation further to statin therapy. However, evidence was scant for CRP and also for TNFα and IL-6. 91 A study evaluating the effectiveness of ezetimibe to influence coronary atherosclerotic plaque composition, indicated that compared with the control group, ezetimibe significantly reduced fibro-fatty plaque volume, but it had no significant effect on reduction of fibrous plaque, necrotic core, or change in dense calcification. 92
Importantly, ezetimibe enhances statin triglyceride-lowering and anti-inflammatory actions. 57 Furthermore, ezetimibe was shown to have a small (7%) but statistically significant decrease in the serum concentrations of lipoprotein Lp(a), an underrecognized causal risk factor for CVD, 93 in patients with primary hypercholesterolemia, albeit with unknown clinical significance. 94
Ezetimibe has also been found to effectively decrease phytosterol, besides cholesterol, absorption. 95 Phytosterol is a plant sterol, a molecule structurally akin to cholesterol, found only in dietary sources (eg, fruits, vegetables, cereals, nuts) since it cannot be generated in humans. 95 Sterol-enriched diets (⩾2 g/day) may lower total and LDL-C levels by 5% to 10%, either alone or when combined with statins, as they antagonize the absorption of dietary cholesterol in the gut. Importantly high serum phytosterol levels, such as those linked with sitosterolemia (a scarce genetic defect), 96 may confer a high atherosclerotic CV risk, albeit a threshold for such an action has not been determined. 97 Importantly, drugs such as ezetimibe may efficiently lower cholesterol and phytosterol absorption. It remains to be shown whether such therapy leading to reduced phytosterol absorption also results in CVD risk reduction.
More Data on Safety and Efficacy of Ezetimibe
A meta-analysis of 7 studies registering 31 048 patients (median follow-up 34.1 ± 26.3 months; 70% females; mean age 61 ± 8 years) indicated that compared with control treatment, ezetimibe considerably lowered the risk of MI by 13.5% (RR: 0.865, P < .001) and the risk of any CVA by 16% (RR: 0.840, P = .005), without any influence on total and CV death rate (RR: 1.003, P = .908; RR: 0.958, P = .330) and risk of new cancer (RR: 1.040, P = .303). 98 The authors concluded that ezetimibe significantly decreases the risk of MI and CVA without any influence on total and CV death rate and risk of cancer.
Ezetimibe, administered at a dose of 10 mg/d in the morning or the evening before or after food ingestion, is absorbed swiftly and metabolized extensively to pharmacologically active ezetimibe glucuronide. 41 There is no clinically important influence of age, gender, or race on ezetimibe pharmacokinetics; no dose modification is required in patients with mild hepatic insufficiency or mild-to-severe renal function worsening. Ezetimibe added to statin does not seem to aggravate muscle toxicity (myositis) and creatine kinase (CK) concentrations beyond what is observed with statin therapy alone.
With regards to carcinogenicity of ezetimibe, a prospective analysis from IMPROVE-IT indicated that among 17 708 patients having simvastatin 40 mg daily, those allocated to ezetimibe 10 mg daily had a similar occurrence of malignancy and deaths due to malignancy versus those having placebo over a median of 6 years. 99 This study refuted the results of the SEAS (Simvastatin and Ezetimibe in Aortic Stenosis) trial which raised concerns about a possible elevated risk of malignancy incurred by ezetimibe (11.1%. versus 7.5%). 100
A recent review and meta-analysis of 48 RCTs (n = 28 444; median follow-up 34 weeks) and 4 observational trials (n = 1667; median follow-up 288 weeks) regarding the safety of ezetimibe, indicated moderate to high certainty that ezetimibe was not linked with cancer (relative risk—RR 1.01), fractures (RR 0.90), drug withdrawal due to any adverse event (RR 0.87), gastrointestinal side-effects leading to withdrawal (RR 1.34), myalgia or muscular pain necessitating withdrawal (RR 0.82), neurocognitive events (RR 0.48), or new-onset diabetes (RR 0.88). 36 Other reviews also indicate that ezetimibe does not elevate the risk of cancers; rather this agent could even tard the emergence and progression of cancer via various mechanisms (stem cell suppression, anti-angiogenesis, anti-inflammatory action, anti-proliferation, immune-enhancement). 101 Nevertheless, more recent data suggest that ezetimibe may elevate the risk of intestinal cancer and has a trend of raising the risk of breast cancer, with no evidence about affecting the risk of other types of cancer. 35
As mentioned, a subanalysis of patients participating in the IMPROVE-IT study, detected no elevation in the risk of new-onset DM with the addition of ezetimibe to statin treatment. 74 Rather, ezetimibe therapy may be linked with ameliorated insulin sensitivity, ascribed, at least partly, to actions on adipocyte size in visceral fat, adipocyte inflammation, free fatty acid concentrations, and fatty acid oxidation. 102 However, further trials are needed to establish these effects. Furthermore, the combination therapy has been established safe, and drug discontinuation rates have not been higher by adding ezetimibe. 56
Finally, concerns about cholelithiasis among patients treated with ezetimibe 103 may probably be put to rest as the data do not seem to support them. 104 Indeed, long-term therapy with ezetimibe ameliorates lipid metabolism without much affecting the tendency for gall bladder stones. 105 It has even been suggested that the specific inhibitory influence of ezetimibe on the intestinal NPC1L1 protein designates this drug as potential agent for averting gallstone production by decreasing bioavailability of intestine-derived cholesterol to the liver for biliary secretion and desaturating bile via the inhibition of intestinal absorption of cholesterol. 106
Cost-efficacy
A cost-efficacy study conducted in China indicated that in patients receiving high-dose statins for the secondary prevention of CVDs, adding ezetimibe is cost-effective compared to adding PCSK9 inhibitors and adding both drugs. 107 A recent British study indicated that the most effective intervention is low/moderate-intensity statins and ezetimibe, and was projected to lead to a gain in quality-adjusted life-years of 0.067 per person initiated at 30 and 0.026 at age 60 years. 108 Finally, a systematic review and meta-analysis of cost-utility studies regarding cost-efficacy of ezetimibe plus statin lipid-lowering therapy indicated that ezetimibe plus statin therapy was significantly cost-effective versus other lipid-lowering therapeutic agents or placebo. 109 The pooled incremental net benefit (INB) (95% CI) was $4274, but there was large heterogeneity (I 2 = 84.21). On subgroup analysis, ezetimibe plus statin therapy was significantly cost-effective in high-income countries ($4356), for primary prevention ($4814), and for payers’ perspective ($3255), and from lifetime horizon ($4571). The evidence from lower-middle-income countries and the societal perspective remains inadequate.
Guideline-Directed Cholesterol-Lowering Therapies
Current American and European guidelines on cholesterol advised the addition of non-statins to statin treatment for high-risk secondary prevention patients above an LDL-C threshold of ⩾70 mg/dL (1.8 mmol/L) (Table 2).41,63 A systematic review and meta-analysis of 11 RCTs (130 070 patients) (selected by follow-up ⩾6 months and sample size ⩾1000 patients) compared efficacy and safety of therapy to attain lower (<70) versus higher (⩾70 mg/dL) LDL-C among patients having intensive lipid-reducing treatment (statins alone or plus ezetimibe or PCSK-9i). 66 The median LDL-C concentrations reached in lower versus higher cholesterol subsets were 62 and 103 mg/dL, respectively. At median follow-up of 2 years, the lower versus higher LDL-C subset had significant diminution in all-cause death rate (absolute risk difference—ARD −1.56, RR 0.94) and in CV death rate (ARD −1.49; RR 0.90), and lower risk of MI, revascularization, CVAs, and MACE. These advantageous results were attained without raising the risk of cancer, DM, or bleeding CVA. The benefit of total death rate in the lower LDL-C subset was restricted to statin treatment and those with increased baseline LDL-C (⩾100 mg/dL). However, the RR decrease in ischemic and safety endpoints was not dependent on baseline LDL-C or drug treatment. The authors concluded that therapy to reach LDL-C concentrations <70 mg/dL employing intensive lipid-lowering treatment with current hypolipidemic therapies can safely lower the risk of death and MACE. Such guideline-directed reductions in LDL-C appear feasible with use of combined statin/ezetimibe regimens without the need to resort to more expensive or inconvenient (parenteral) regimens.
Recommendations from current guidelines on use of ezetimibe.

Following guideline-directed cholesterol-lowering treatment could avert a considerable number of recurrent CVD episodes. Simulations using data from 279 395 US patients with an MI hospitalization (2018-2019) (mean age 75 years, mean LDL-C 92 mg/dL), indicated that 27% were having guideline-directed cholesterol-lowering treatment. 110 With current use of cholesterol-lowering treatment, 25% of patients had an atherosclerotic CVD episode over 3 years. If all patients were to avail themselves of guideline-directed treatment, ~20% were deemed to have a CVD episode over 3 years, representing a 21.6% relative risk reduction.
Current evidence indicates that attaining the guideline-directed LDL-C target rate is still suboptimal despite employing high-intensity statin treatment in a real-world scenario. 111 Hence, the need to add ezetimibe before one resorts to other more expensive and/or less convenient therapies. Unfortunately, the problem remains as the majority of adults with atherosclerotic CVD are not currently receiving guideline-recommended add-on lipid-lowering therapy. 112
Indeed, a recent retrospective observational population study examined the rate of attainment of guideline-directed lipid levels employing associated health data in 10 071 patients submitted to PCI (2012-2017), of whom 48% had LDL-C <1.8 mmol/L (2016 goal) and (23%) <1.4 mmol/L (2019 goal). 113 A total of 5340 patients had non-HDL cholesterol (non-HDL-C) measured with 57% <2.6 mmol/L (2016) and 37% <2.2 mmol/L (2019). In patients with vascular event recurrences, <6% of the patients attained the 2019 LDL-C goal of <1.0 mmol/L. Totally, 10 592 patients had triglyceride concentrations measured, of whom 14% were ⩾2.3 mmol/L and 41% ⩾1.5 mmol/L (2019). High-intensity statins were administered in 56.4% of the population, only 3% were given ezetimibe, fibrates or prescription-grade N-3 fatty acids. Females were more likely to have lipid concentrations above target. The authors concluded that there was a low rate of attainment of the new European Society of Cardiology (ESC)/European Atherosclerosis Society (EAS) lipid goals in this large post-PCI cohort and relatively low rates of intensive lipid-lowering drug prescriptions in those with elevated lipids. Thus, there is great potential to optimize lipid-reducing treatment further and 1 such way is by adding ezetimibe to statin therapy, especially in females.
A practical (real-world) guidance (2021) from the EAS Task Force indicates that statin-ezetimibe combined therapy is the first choice for dealing with high LDL-C and should be administered upfront in very-high-risk patients with high LDL-C less likely to attain target with a statin, and in primary prevention familial hypercholesterolemia patients. 114 A PCSK9 inhibitor may be also prescribed if LDL-C concentrations are still elevated. In high and very-high-risk patients with mild to moderately high triglyceride concentrations (>2.3 and <5.6 mmol/L [>200 and <500 mg/dL) on a statin, management with either a fibrate or high-dose omega-3 fatty acids (icosapent ethyl) may be contemplated, balancing the advantages versus risks. Combined therapy with fenofibrate may be provided for both macro- and microvascular gains in patients with type 2 DM.
Subset of Patients Deriving the Most Benefits From Reduction of LDL-C Need to be Defined
A recent meta-analysis confirmed that LDL-C-reducing therapies significantly mitigate death rate. 115 However, the meta-regressions and meta-analyses by subsets indicated that LDL-C reduction may not be advantageous for total and CV death rate end-points in trials with >50% LDL-C lowering and in studies with low baseline LDL-C concentrations. Nevertheless, the decrease in MI risk was consistent across all analyses. Importantly, annual numbers-needed-to-treat were overall relatively increased, and studies including patients with high baseline LDL-C concentrations recorded the largest gain from LDL-C—reducing treatment particularly for MI. Attaining lower LDL-C targets did not further enhance risk reduction consistently. Defining subsets of patients who obtain the greatest gains from LDL-C level lowering is clinically important and necessary. Ezetimibe can be effective as monotherapy in certain groups, but is most apparently needed in combined therapies to achieve these goals.
Fixed Combinations of Statin/Ezetimibe
Fixed combinations of a statin with ezetimibe have been available as a robust therapy of hypercholesterolemia which is more effective than statin therapy alone, even when the statin dose in the combination is low or moderate compared with a high-dose statin monotherapy regimen. 46 These combinations enable higher numbers of patients to meet the needs in LDL-C lowering 116 and attain guideline-recommended LDL-C levels. 45 The safety profile of these combinations is preserved or enhanced by allowing the prescription of a decreased dose of statin which also mitigates or averts the occurrence of statin-intolerance. 117 Furthermore, patient compliance seems to increase with use of single-pill regimens. 118 Also, this approach has been reported to unexpectedly enhance triglyceride control. 119 Additional CV outcomes data are needed to further confirm and establish their efficacy.
In this respect, a multicenter randomized, double-blind trial examined the safety and efficacy of a fixed-dose combined regimen of rosuvastatin 2.5 mg and ezetimibe 10 mg versus those of ezetimibe 10 mg single therapy, rosuvastatin 2.5 mg, and rosuvastatin 5 mg single therapy in 279 patients with hypercholesterolemia. 120 At the 8-week follow-up, a larger reduction in the LDL-C concentrations (primary endpoint) was demonstrated in the statin 2.5 mg plus ezetimibe 10 mg group (−45.7% ± 18.6%) than in the ezetimibe 10 mg group (−16.7% ± 14.7%, P < .0001), statin 2.5 mg group (−32.6% ± 15.1%, P < .0001), and statin 5 mg group (−38.9% ± 13.9%, P = .0003). Comparable results were noted with the lowering of total cholesterol, non-HDL-cholesterol, and apolipoprotein B protein. In patients with low and moderate risk, all patients attained the goal LDL-C concentrations in the statin 2.5 mg plus ezetimibe 10 group (100%) compared with 13% in the ezetimibe 10 mg group, 47.6% in the statin 2.5 mg group, and 65% in the statin 5 mg group. Untoward events were scarce and comparable in the 4 groups. The authors concluded that fixed-dose combined treatment with low-intensity rosuvastatin and ezetimibe was more efficacious in decreasing LDL-C and attaining LDL-C targets than moderate-intensity rosuvastatin single therapy.
Cost-Efficacy of Ezetimibe or of Combined Ezetimibe Plus Statin Lipid Lowering Therapy
A recent cost-effectiveness analysis using a Markov model indicated that ezetimibe increased quality-adjusted life years (QALYs) by 0.60 at cost lowering of −£2529 (incremental cost-effectiveness ratio—ICER = −£4231/QALY). 121
Another recent cost-efficacy study spotted economic assessment studies reporting outcomes of ezetimibe plus statin treatment versus other lipid-reducing drugs or placebo. 109 The pooled incremental net benefit (INB) from 21 eligible trials indicated that the ezetimibe plus statin regimen was significantly cost-effective versus the other lipid-reducing drugs (statin monotherapy/PCSK9i plus ezetimibe and statins/PCSK9i with statin therapy), or placebo. The pooled INB (95% CI) was $4274 (621-7927), but there was high heterogeneity (I 2 = 84.21). On subgroup analysis ezetimibe plus statin treatment was significantly cost-effective in high-income countries [$4356 (621-8092)], for primary prevention [$4814 (2523-7106)], and for payers’ viewpoint [$3255 (571-5939)], and from lifetime horizon (survival extending over the entire lifespan) [$4571 (746-8395)]. There were limited data from lower-middle-income countries and the societal standpoint.
Finally, a more recent cost-efficacy analysis further confirmed ezetimibe plus statin (6 studies) to be a cost-effective strategy compared with statin monotherapy. 122
Statin Intolerance
Statin intolerance deprives many patients from the standard hypolipidemic therapy. Current choices for such patients comprise the usage of a reduced but tolerated dose of a statin and adding or changing to ezetimibe or other non-statin regimens. Of course, PCSKi produce larger LDL-C decreases than ezetimibe in such patients, with fewer skeletal-muscle deleterious occurrences versus a statin. 122 However, such therapy is costly and involves injections.
Another effective hypolipidemic approach to patients who cannot tolerate daily statin single treatment that has been suggested entails the combination of daily ezetimibe plus low-dose statin twice a week might be an alternative regimen for high-risk patients who are intolerant to daily statin regimen. 123
One important caveat regarding statin intolerance relates to the possibility that the prevalence of total statin intolerance might be frequently overestimated. 124 In this regard, a recent large meta-analysis of 176 trials (112 RCTs, 64 cohort studies) of >4 million (4 143 517) patients indicated that the prevalence of statin intolerance was low at 9.1% (95% confidence interval 8.0%-10%) when diagnosed by applying the international definitions. 124 The prevalence was not different when defined by employing criteria from various societies (5.9%-7%). The prevalence of statin intolerance in RCTs was considerably lower versus cohort trials (4.9% vs 17%). The prevalence of statin intolerance was much greater when primary or secondary prevention patients were examined separately rather than when grouped together (18%, 8.2%, 9.1%, respectively). Lipid solubility did not influence statin intolerance (4% vs 5%), but several other factors did, such as age, odds ratio—OR 1.33, P = .04; female sex, OR 1.47, P = .007; Asian and Black race, P < .05 for both; obesity, OR 1.30, P = .02; DM, OR 1.26, P = .02; hypothyroidism, OR 1.37, P = .01; chronic liver, and renal failure, P < .05 for both. Antiarrhythmic agents, calcium channel blockers, alcohol use, and increased statin dose also conferred an elevated risk of statin intolerance. 124
A Holistic Lipid Approach Strategy/Patients With High Triglycerides/the Role of Icosapent Ethyl
The usage of icosapent ethyl has been demonstrated to significantly lower triglyceride concentrations and decrease CV risks in patients having optimal statin or combined statin/ezetimibe therapy.125,126 Icosapent ethyl is a highly purified eicosapentaenoic acid ethyl ester that decreases triglyceride concentrations without increasing LDL-C. 127 Icosapent ethyl was the first fish oil product which was granted approval by the US FDA to decrease the risk of atherosclerotic CVD in adults. 125 The results of the Reduction of Cardiovascular Events with Icosapent Ethyl-Intervention Trial (REDUCE-IT) trial where 8179 patients with proven CVD or with DM and other risk factors, who had been on statin treatment and who had a fasting triglyceride concentration of 135 to 499 mg/dL and a LDL-C concentration of 41 to 100 mg/dL, and were randomized to 2 g of icosapent ethyl bid (total daily dose, 4 g) or placebo, indicated that the likelihood of ischemic events, comprising CV death, was considerably decreased with icosapent ethyl versus placebo over a median of 4.9 years. 6 The primary end point (CV death, nonfatal MI, nonfatal CVA, coronary revascularization, or unstable angina) was noted in 17% in the icosapent ethyl group versus 22% in the placebo group (hazard ratio—HR, 0.75; P < .001); the corresponding rates of the key secondary end point (composite of CV mortality, nonfatal stroke or MI) were 11.2% and 14.8% (HR, 0.74; P < .001). The rates of further ischemic end-points, as evaluated per a prespecified hierarchical protocol, were considerably reduced in the icosapent ethyl subset than in the placebo subset, including CV mortality (4.3% vs 5.2%; HR, 0.80; P = .03). More patients in the icosapent ethyl subset than in the placebo subset were hospitalized for atrial fibrillation or flutter (3.1% vs 2.1%, P = .004). Serious hemorrhagic episodes were noted in 2.7% of the patients in the icosapent ethyl subset and in 2.1% in the placebo subset (P = .06). Further analysis of this trial indicated that among statin-receiving patients with high triglycerides and CVD or DM, icosapent ethyl considerably decreases the load of first, subsequent, and total ischemic episodes. 128
Besides the REDUCE-IT trial and several subanalyses of this trial (in US patients, patients with a history of coronary artery bypass grafting, patients with CKD) where a consistent CV benefit was shown with use of icosapent ethyl, 125 additional trials have advocated the efficacy of icosapent ethyl in hypertriglyceridemia and CV risk lowering in patients receiving optimal hypolipidemic therapy with use of a statin and/or ezetimibe. 129
The position of the National Lipid Association (NLA) is that for patients 45 years of age or older with clinical atherosclerotic CVD, or 50 years or older with DM needing medical treatment plus ⩾ 1 extra risk factor, with fasting triglycerides 135 to 499 mg/dL on high-intensity or maximally tolerated statin treatment (±ezetimibe), addition of icosapent ethyl is advised for lowering the atherosclerotic CVD risk (evidence rating: class I; evidence level: B-R). 130
Finally, over a decade ago, an Expert Panel group put forth recommendations regarding triglycerides indicating that high non-fasting triglycerides is a risk factor for CVD; hence, they should be managed by aggressive lifestyle changes and/or triglyceride lowering agents, like statins, and fibrates. 131 Importantly, as mentioned, ezetimibe enhances statin triglyceride-lowering and anti-inflammatory actions. 57
Limitations
There are some limitations to this study. Women are under-represented in CV outcome trials and particularly in trials using ezetimibe. 132 Importantly, women, older persons, and patients with noncardiac atherosclerotic CVD are specifically undertreated. 133 Available data are limited regarding the effect of ezetimibe on inflammatory markers. Cost efficacy data are limited to certain countries. High- and very high-risk patients are not achieving LDL-cholesterol level goal at high percentages despite use of high-intensity statin therapy for various reasons, 1 of which may be related to underuse of ezetimibe. 111 Studies of lipid management in the real-world setting indicate that LDL-cholesterol levels remains suboptimal, as currently available therapies are underutilized in high/very-high risk patients leading to suboptimal management of CV risk. 134 There are also ethnic group discrepancies that need to be addressed. 135 Importantly, prescription of the 1-pill combination of statin-ezetimibe exhibits better patient adherence. 118
Perspective
According with the National Health and Nutrition Examination Survey data from 2017 to 2020, eligibility and usage of statins, ezetimibe, PCSK9i, and icosapent ethyl among US adults were assessed per the American College of Cardiology/American Heart Association guidelines. 136 In this nationally representative US study, <50% fewer persons who had guideline-directed indications for lipid lowering with use of statins were actually receiving these drugs, with particular underuse among Black adults and younger adults 40 to 64 years of age. In addition, a considerable proportion of persons who appear to be candidates for PCSK9i and icosapent ethyl are on suboptimal treatment with statins and/or ezetimibe, and if optimally treated with these agents, significantly fewer persons would merit consideration for more expensive and injection-based hypolipidemic therapies. Fear of side-effects and cost of treatment seem to be a major barrier to optimal use of hypolipidemic therapies explaining such a degree of undertreatment with such therapies. Nevertheless, with maximal escalation of statins and even when ezetimibe has been added, 4.1% of adults or 6.1 million would still derive a gain from PCSK9i and 6.8% or 10.2 million from icosapent ethyl. 136
Conclusion
Statins remain the cornerstone of hypolipidemic therapy. Unfortunately, these agents have several side-effects, which have partly hindered their more widespread use. Ezetimibe is commonly the first added treatment to attain LDL-C targets. It reduces LDL-C by approximately an extra 20% and has an excellent safety and tolerability profile. When a statin is combined with ezetimibe, the dose of statin could be lower, thus averting or limiting the occurrence of adverse effects. Of course, such a combination does enhance the hypolipidemic effect. Ezetimibe could also serve as an alternative mode of treatment in certain cases of statin intolerance. Importantly, besides statins, among the lipid-lowering therapies, ezetimibe has also been proven by prospective studies and RCTs to further reduce CVD risk. Regarding possible pleiotropic effects which are inherent in statin therapies, there is a report of an anti-inflammatory effect of ezetimibe as well, which has been noted to occur on top of statin treatment. The percentage of patients with target level LDL-C (<70 mg/dL) can be much higher in a combined treatment group versus a high-intensity statin monotherapy group. This holds true also in patients with recent ischemic CVA, where moderate-intensity statin plus ezetimibe was shown to be superior to high-intensity statin alone for intensive LDL-C lowering. Also, withdrawal of lipid-lowering agents occurs less often in the combined therapy group. Importantly, in the pursuit of an escalation of hypolipidemic therapies, the use of non-statin therapies alone or in combination with statins, therapies that could also be affordable for the general public, the wider use of ezetimibe might help attain these goals. 137 In keeping with this view, a recent RCT comprising 150 patients with acute ischemic cerebrovascular disease randomly allocated to moderate-intensity statin with ezetimibe or high-intensity statin for 3 months indicated that the latter regimen further improved the attainment rate of LDL-C in these patients, with a higher reduction magnitude in LDL-C without compromising safety. 138 Finally, in a holistic lipid approach and strategy, adding icosapent ethyl might also decrease CV morbidity in patients who have reached LDL-C targets but have persistently high triglyceride concentrations considered an additional factor often contributing to residual CV risk in patients who are already at high risk. Indeed, most recent data from an RCT comprising 840 patients with a recent ACS (<1 year) indicated that this agent dramatically reduced the risk of ischemic events in high-risk statin-treated patients with no excess bleeding, suggesting the need for prompt use of this agent after ACS. 139
Footnotes
Acknowledgements
None
Declarations
Ethics Approval and Consent to Participate
Not applicable/this study is a review based exclusively on published literature
Consent for Publication
All Authors consented
Author Contributions
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Competing interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: AAM, TAM, and ASM have nothing to declare; DPM has given talks and attended conferences sponsored by Amgen, Novo Nordisk, and Libytec.
Availability of Data and Material
Data sharing is not applicable to this article as no new data were created or analyzed in this study.
