Abstract
Background
Phaeochromocytomas and paragangliomas (PPGL) are catecholamine secreting tumours associated with significant morbidity and mortality. Timely diagnosis and management are essential. A range of laboratory tests can be utilised in the investigation of PPGL. There is scope for significant variation in practice between centres. We aimed to investigate how the laboratory investigation of PPGL is performed in laboratories across the United Kingdom.
Methods
A questionnaire consisting of 21 questions was circulated to Clinical Biochemistry laboratories in the United Kingdom via the Association for Clinical Biochemistry and Laboratory Medicine office. The survey was designed to allow audit against Endocrine Society Guidelines on the Investigation and Management of PPGL and to obtain information on other important aspects not included in these guidelines.
Results
Responses were received from 58 laboratories and the data were compiled. The majority of laboratories use either urine or plasma metanephrines in first-line testing for PPGL, although a number of different combinations of biochemistry tests are utilised in different centres. All laboratories measuring metanephrines or catecholamines in-house use LC or LC-MS/MS methods. There are some marked differences between laboratories in urine metanephrines reference ranges used and sample requirements.
Conclusions
There is evidence of good practice in UK laboratories (as assessed against Endocrine Society Guidelines) such as widespread use of urine/plasma metanephrines and appropriate analytical methodologies used. However, there is also evidence of variations in practice in some areas that should be addressed.
Introduction
Phaeochromocytomas (PC) are tumours arising from the chromaffin cells of the adrenal medulla while paraganglioma (PGL) tumours are extra-adrenal tumours of the sympathetic or parasympathetic nervous system.1,2 PC and sympathetic PGL usually secrete excessive amounts of catecholamines (one or more of epinephrine, norepinephrine and dopamine). Parasympathetic PGL, typically occurring in the head and neck, do not secrete catecholamines. Excessive catecholamine secretion is responsible for most of the symptoms and signs associated with PC or PGL (PPGL) including hypertension, anxiety, sweating, tremor and headaches, which are often paroxysmal. There is a risk of catecholaminergic crises where extreme hypertension can lead to end-organ damage and potentially death. 3 Around 10% of PPGLs are malignant, with evidence of metastasis upon diagnosis (this is more common for extra-adrenal tumours) 1 and around 40% are associated with an inherited predisposition syndrome. 4 Due to the significant morbidity and mortality associated with untreated PPGL, timely and accurate diagnosis is important.
PPGLs are very rare conditions with an estimated prevalence of only 2–8 new cases per million individuals per year. 4 However, the symptoms and signs associated with PPGL are also found in much more common disorders such as primary hypertension and anxiety disorders. Incidentally discovered adrenal lesions (adrenal incidentalomas) are increasingly common (1.5 to 3% of hospitalised patients may harbour a benign adrenal lesion 5 ). These incidentalomas are often benign adenomas, but exclusion of clinically significant lesions such as PC is often sought. Therefore, although PPGLs are rare, cases where we wish to exclude the presence of these tumours are relatively common. First-line biochemistry tests that offer a high degree of diagnostic performance are required. The most important characteristic of a biochemical test for PPGL is high diagnostic sensitivity so that the disorder can be ruled-out with confidence. High diagnostic specificity is also important as this limits the number of false positive cases and the associated unnecessary further investigation (as PPGL are rare, sub-optimal specificity leads to a test with poor positive predictive value).
A number of biochemical tests have been applied to the diagnosis of PPGL and there is scope for significant variation in practice between centres. These tests include urinary catecholamines and urinary catecholamine metabolites such as vanillylmandelic acid (VMA) and the metanephrines (metanephrine, normetanephrine and 3-methoxytyramine). The catecholamines and metanephrines may also be measured in plasma. The biochemical tests all perform differently in terms of their diagnostic performance, with significant variations in the diagnostic specificity and sensitivity between these tests. 6 There is a range of methodologies that have been used to perform these tests including spectrophotometry7,8 immunoassay9,10 high performance liquid chromatography with electrochemical detection (LC-ECD)11,12 and liquid chromatography-tandem mass spectrometry (LC-MS/MS).13,14 These different methodologies have different analytical characteristics, which may influence diagnostic performance.6,10,15 In addition to variation in the biomarkers measured and methodology used, urine and plasma concentrations of these markers can be heavily influenced by patient preparation 16 and certain medications. 17 All these factors potentially contribute to variations in practice related to biochemical testing for PPGL.
Recommendations from Endocrine Society Clinical Practice Guideline [20] related to the laboratory investigation of PPGL. 1 = strong recommendation, 2 = weak recommendation, ⊕ = lowest quality evidence and ⊕⊕⊕⊕ = highest quality evidence.

1 = strong recommendation, 2 = weak recommendation, ⊕ = lowest quality evidence and ⊕⊕⊕⊕ = highest quality evidence.
Methods
A survey questionnaire was designed with input from members of the ACB NCBAG and ACB All Wales Clinical Biochemistry Audit Group. This survey consisted of 21 questions regarding tests used in laboratory testing for PPGL. Some of these questions were designed to assess concordance with the Endocrine Society Guideline recommendations in Table 1, while others were included to acquire information on other important aspects not included in this guideline. A copy of the questionnaire can be found in Supplementary Data (along with some data from the audit not included in this article). The questionnaire was distributed using Survey Monkey® (www.surveymonkey.com) to senior staff in all NHS clinical biochemistry laboratories as identified using ACB records. Data analysis was performed using Microsoft Excel® (Microsoft Corporation, Albuquerque, New Mexico, USA). Duplicate responses from the same laboratory or laboratory network were removed, with the most complete response included in the analysis.
Results
Response
There were 58 responses from the laboratories surveyed (around 150 UK biochemistry departments). Of the responding laboratories, 52% were based in a district general hospital, 21% in a teaching hospital and 27% in a hospital with a tertiary referral centre for endocrinology.
Tests used in the investigation of PPGL
Responding laboratories were asked which biochemical tests were available for the investigation of PPGL either as first-line tests or as follow-up tests (Figure 1). Urine metanephrines (UMET) was most widely available with 93.1% (54/58) of laboratories providing the test. This was performed in-house by 50% of these laboratories (27/54) with the other 50% referring the test to another centre. Plasma metanephrines (PMET) was available from 84.5% (49/58) laboratories with 12.2% (6/49) performing the test in-house. Urine catecholamines (UCAT) was available from 29.3% (17/58) of laboratories, with 47% (8/17) performing the test in-house. Urine VMA was available from 15.5% (9/58) of laboratories (none of these nine labs performed the test in-house) and plasma catecholamines (PCAT) was available from 8.6% (5/58) of laboratories (again, no responding labs performed the test in-house). The number of tests available in different centres varied, with some centres only offering a single test and others offering up to four of these tests. There were a total of 12 different combinations of UMET, PMET, UCAT, VMA and PCAT in the repertoire of the responding laboratories. Proportion of laboratories offering various biochemical tests as part of the investigation of PPGL. Darker shaded bars indicate laboratories offering the tests in-house.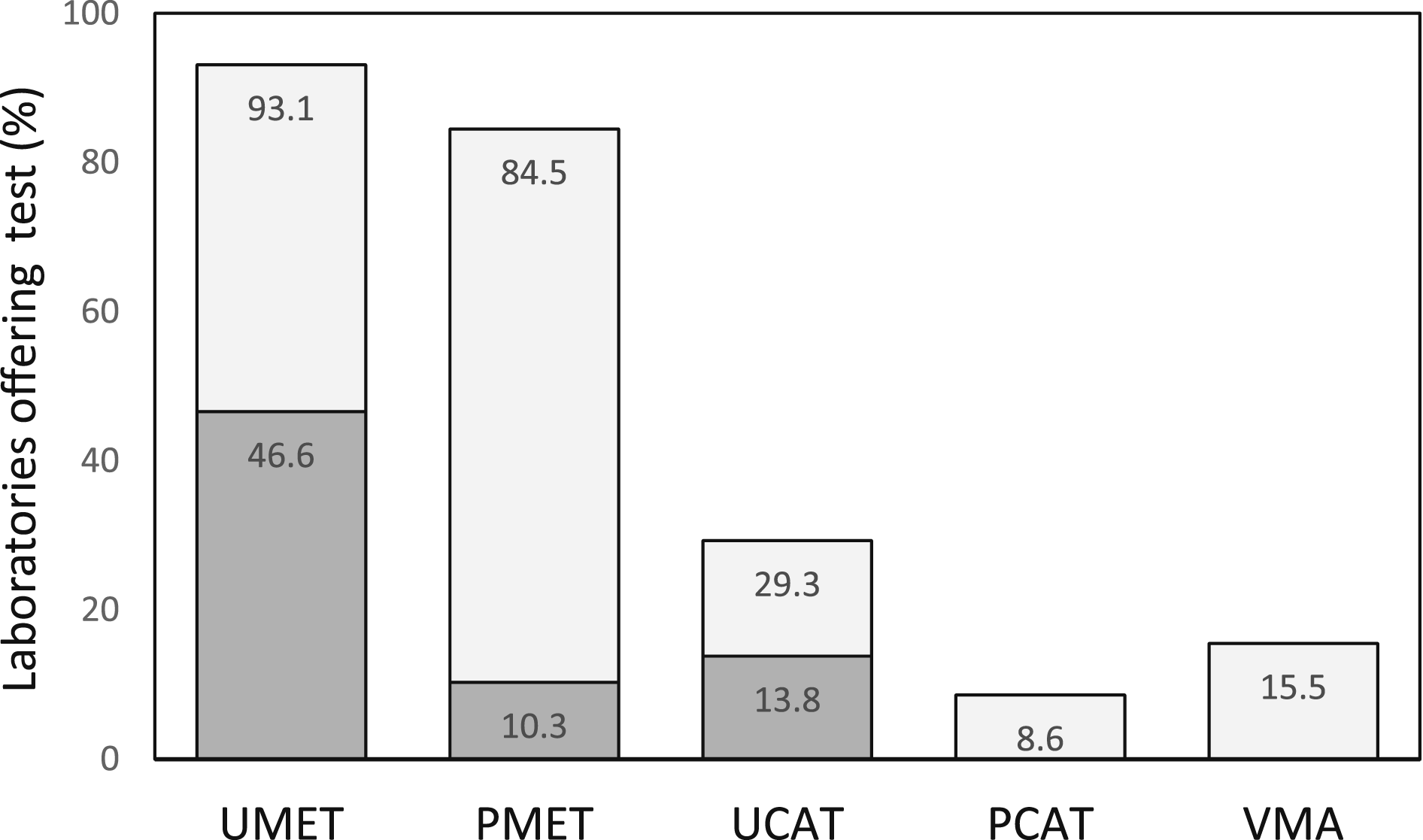
The survey also included questions about which biochemical tests were used specifically for first-line testing rather than for confirmatory purposes. UMET was used by 84.5% (49/58) of laboratories as a first-line test, while PMET was used by 50.0% (29/58). UCAT was used by 19.0% (11/58) of labs in first-line testing and VMA in 5.2% (3/58). Many centres used more than one test in initial testing for PPGL (Figure 2), but in total 94.8% (55/58) included either UMET and/or PMET in first-line testing. The 5.2% (3/58) of laboratories that did not include UMET or PMET used UCAT only. Some laboratories only used certain biochemical tests as second-line tests in the follow-up of abnormal first-line tests. PMET was used as a follow-up test only in 29.3% (17/58) of laboratories (all but one using UMET as the initial test), while 3.4% (2/58) used UMET as a follow-up test only. Proportion of laboratories including various biochemical tests as part of first-line testing. Some laboratories offer more than one test for first-line testing. In total, 95% of laboratories include either UMET or PMET in first-line testing.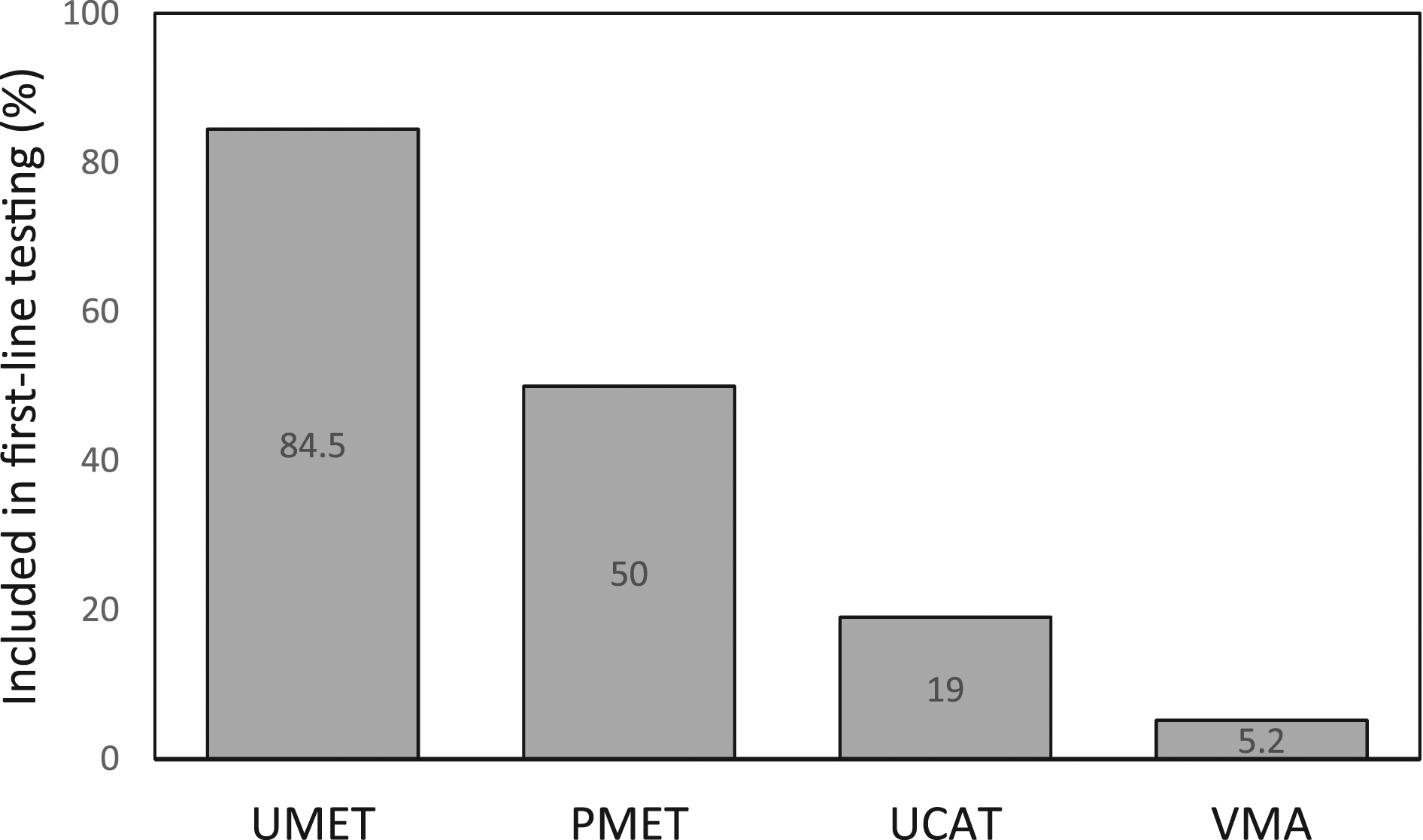
The clonidine suppression test and chromogranin A (CgA) also have a role in the investigation of PPGL in some centres. CgA is used in 43.1% (28/58) of responding laboratories, usually only in certain situations with some centres using CgA as a second-line test and some only in follow-up after treatment, for example. The clonidine suppression test is used by 15.5% (9/58) of laboratories to exclude false positive results in PMET and/or UMET with some of these labs commenting that this test is used extremely rarely. While it is unclear whether plasma norepinephrine or normetanephrine is measured during the clonidine suppression test, only one of these nine laboratories reported that they include plasma catecholamines in their repertoire.
Methodology used for biochemical tests for PPGL
UMET was performed in-house by 27 responding laboratories. HPLC with electrochemical detection (HPLC-ECD) was used by 70.4% (19/27) of these labs and LC-MS/MS used by the remaining 29.6% (8/27). 70.4% (19/27) of laboratories measuring UMET included 3-methoxytyramine (3MT) in their UMET panel. PMET was performed in-house by six responding laboratories, with all six using LC-MS/MS and all six including measurement of plasma 3MT. Of the eight laboratories performing UCAT in-house, 75% (6/8) used LC-ECD, while the remainder (2/8) used LC-MS/MS.
Urine metanephrines reference ranges
A variety of UMET reference ranges are applied in different laboratories (Figure 3). The upper limit of the reference range quoted for urine normetanephrine varied from 2.88 to 5.3 μmol/24 h. In the case of urine metanephrine, the upper limit of the reference range varied from 1.2 to 2.0 μmol/24 h and in the case of urine 3MT from 1.3 to 2.8 μmol/24 h. The laboratories that supplied their reference ranges cited different sources for these ranges. 29.6% (8/24) using data produced in-house, 18.5% (5/24) take reference range data from commercial HPLC or LC-MS/MS kit inserts, 7.4% (2/24) use published reference data and 37% (10/24) did not state the source of their reference ranges (2/24 labs measuring UMET in-house did not provide their reference ranges). Chart showing the upper limit of the reference range for the urine metanephrines in 24 laboratories (A to X). Laboratories A, J and R have gender and/or age specific ranges. Some laboratories do not include 3MT in their method and so do not quote a reference range.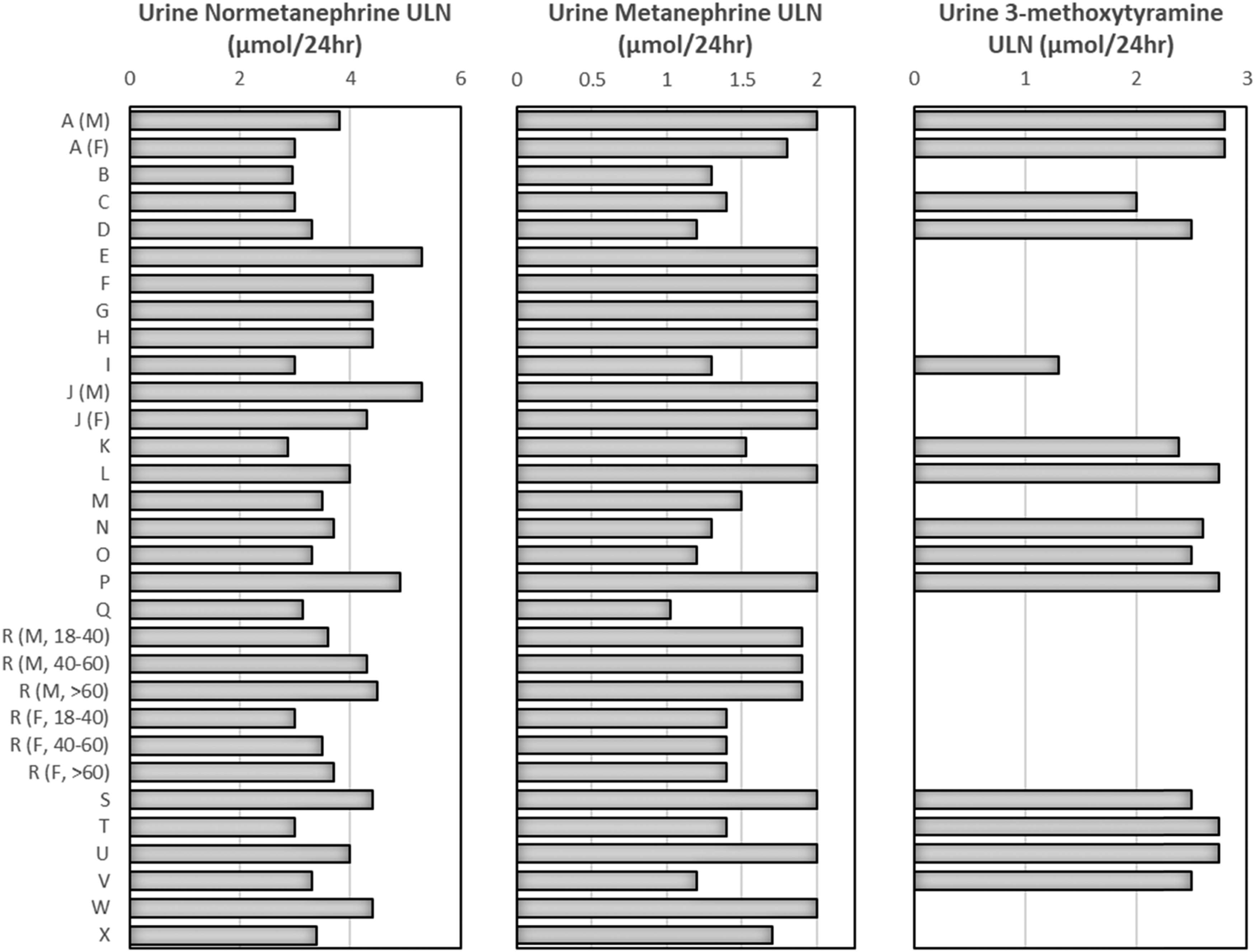
Plasma metanephrines reference ranges and patient preparation
Of the six laboratories performing PMET analysis in-house, five provided details of the reference ranges used. Reference ranges based on both seated and supine sampling are provided by four laboratories, while the fifth laboratory provides only supine reference ranges. In the case of the four laboratories providing both seated and supine reference ranges, the seated reference ranges are based on data published by Peaston et al. 14 and the supine reference ranges are based on data published by Peitzsch et al. 20 (1 laboratory used age-related supine normetanephrine ranges, while the other three used a single normetanephrine range). The laboratory that provides only supine reference ranges, used ranges based on data in Peaston et al., 14 which they validated in-house using a supine population.
40 laboratories using PMET in testing for PPGL provided details on patient preparation when taking blood samples for PMET analysis. A seated posture was used in 42.5% (17/40) of these centres, a supine posture in 30.0% (12/40) and both seated and supine postures (each posture used in different situations) in the remaining 27.5% (11/40) centres. Some of the centres using both seated and supine sampling provided more detail on the situations where the different postures were used. Six laboratories stated that seated sampling was routine with supine sampling used for follow-up of borderline seated PMET results. Four labs stated that seated sampling was used for the majority of out-patient requests but that supine sampling was used for in-patients/day cases in endocrinology units where inconclusive UMET/seated PMET results were followed up or where surveillance of susceptibility syndromes was being carried out.
The conditions for seated sampling for PMET varied slightly between centres. 17.9% (5/28) of labs recommend that samples are taken after an overnight fast and 35.7% (10/28) labs recommend that patients remain at rest in the seated position for a certain length of time before samples are taken (6/28 labs recommend 10 min, 2/28 labs 20 min and 2/28 labs 30 min). In the case of supine sampling for PMET, 39.1% (9/23) laboratories recommend an overnight fast and 78.3% (18/23) recommended a defined period of supine rest before sampling (2/23 labs recommend 10 min, 3/23 labs 20 min and 13/23 labs 30 min).
Sample requirements
The sample requirement for 24-h UMET was an acidified collection in 66.7% (36/54) laboratories, a plain collection in 16.7% (9/54) labs and 11.1% (6/54) labs accepted acidified or plain collections (3 respondents did not provide these details). The minimum acceptable pH for urine collections varied between laboratories from a pH of <2 to pH <6.5. In addition to 24-h collections for UMET, two laboratories accepted overnight urine collections and eight laboratories accepted random samples. Some of these laboratories state that random samples are only accepted in certain circumstances, such as in paediatric patients where obtaining a 24-h collection is difficult.
The sample type required for PMET was EDTA plasma for all laboratories and all required that samples be centrifuged within a specific timeframe before freezing the plasma for storage. The maximum permitted delay between sample collection and centrifugation varied from 15 min to 6 h (7.1% (3/42) within 15 min, 28.6% (12/42) 20–30 min, 28.6% (12/42) 1 h, 4.8% (2/42) 2 h, 2.4% (1/42) 6 h and 26.2% (11/42) ‘ASAP’).
The sample requirement for 24-h UCAT was an acidified collection in 88.9% (16/18) of laboratories, while 11.1% (2/18) of labs accepted plain collections. For PCAT, 2/4 of laboratories required lithium heparin plasma with sodium metabisulphite crystals on ice, 1/4 required a regular lithium heparin plasma on ice and 1/4 EDTA plasma on ice.
Provision of interpretative/clinical advice
Written advice on medications that can affect metanephrines and/or catecholamines results is provided by 62.1% (36/58) laboratories. 91.7% (33/36) of these laboratories provided more detail on the nature of this advice. A list of medications was available in the laboratory handbook or on the departmental website in 36.4% (12/33) of labs, included in information documents provided to patients in 27.2% (9/33), included in report comments in 24.2% (8/33) and listed in a clinical protocol/guideline in 12.1% (4/33).
A Trust clinical protocol or guideline for the investigation of PPGL was available in 27.5% (16/58) responding centres. 12/16 laboratories provided more detail on these protocols. In 5 centres a laboratory produced guide was available, in five centres endocrinology had produced a protocol, in one centre the protocol for investigation of PPGL was included in an adrenal incidentaloma guideline and one centre had both a laboratory guideline and a specific protocol for patients with PPGL susceptibility syndromes.
Discussion
Tests used in the investigation of PPGL
Responses to the survey showed differences in the availability of various biochemical tests for PPGL between centres. Measurement of metanephrines was more widely available than catecholamines, with 93.1% of laboratories using UMET and 84.5% using PMET. Only 29.3% of labs use UCAT with even fewer using PCAT (8.6%) or VMA (15.5%). This suggests that there has been a major shift in the choice of tests used to investigate PPGL in the United Kingdom since 2008, when more than 50% of laboratories were using urine catecholamines alone, rather than urine metanephrines. 18 The factors driving this change in practice are likely to include the growing literature providing evidence that urine and plasma metanephrines achieve significantly better diagnostic performance than UCAT, PCAT or urine VMA when testing for PPGL.6,19 Although urine and plasma metanephrines have become the most widely used tests, there is obviously still variation in testing strategies being used in different centres. In total, 12 different combinations of tests are in use in the 58 laboratories that responded to the survey. The large number of different test combinations suggests that not all laboratories are taking a similar, evidence-based approach to biochemical testing for PPGL. This also highlights a potential issue of cost effectiveness, where unnecessary testing may be being performed.
When questioned solely on tests used in first-line testing rather than as follow-up tests, 95% of laboratories stated that they include plasma and/or urine metanephrines, which is consistent with recommendations made in the Endocrine Society Guidelines 19 (Table 1). However, 5% of laboratories use urine catecholamines alone as a first-line test, which is likely to result in significantly reduced diagnostic sensitivity and specificity for PPGL.6,21,22 In addition, many centres include other tests alongside plasma/urine metanephrines as part of first-line screening or as second-line tests. (Figures 1 and 2). Evidence in the literature clearly shows that UCAT, PCAT and VMA exhibit lower diagnostic specificity and lower sensitivity than PMET and UMET. Therefore, although the Endocrine Society Guidelines do not specifically recommend against using these other tests in addition to PMET/UMET, it is difficult to see how adding tests with poorer diagnostic performance can improve either diagnostic specificity without impairing sensitivity or improve diagnostic sensitivity without impairing specificity. The discontinuation of VMA, PCAT and UCAT in testing for PPGL should be considered in view of the large number of studies indicating the inferior performance of these tests compared to UMET and PMET.
CgA and the clonidine suppression test are additional tests used in some centres. Although 43% of responding laboratories stated that CgA is used in the investigation of PPGL, the additional comments provided suggest that the test is used as a complementary test in certain cases rather than as a first-line or diagnostic test. The evidence supporting the utility of CgA in routine testing for PPGL is relatively poor. Most of the published studies investigating the diagnostic performance of CgA are relatively small and those that offer a direct comparison with PMET or UMET suggest that CgA has lower diagnostic sensitivity.23–26 The specificity of CgA is also limited. CgA can be markedly elevated in patients taking proton pump inhibitors and patients with renal failure. 27 Some authors have suggested that CgA could be used as a confirmatory test to identify false-positive PMET or UMET results. However, while following positive PMET results with measurement of CgA may reduce the number of false positives, this would come at the expense of diagnostic sensitivity. 24 The only situation where CgA has been clearly recommended in the investigation of PPGL is as a potential biomarker for PPGL that do not produce catecholamines. European Society of Endocrinology guidelines recommend that CgA is measured in PPGL with negative metanephrines pre-operatively. In cases where CgA is positive, it may be monitored post-operatively to aid detection of recurrence. 28 A consideration when assessing the diagnostic utility of CgA is that the assays are not standardised and have differing specificities for various CgA related peptides. This may lead to variations in diagnostic performance between CgA assays. 29
The clonidine suppression test is used by only a minority of centres (15.5%). Where this test is performed, it is used to investigate potentially false positive UMET/PMET results. This dynamic function test exploits the pharmacological action of clonidine in suppressing production of norepinephrine by the sympathetic nervous system. The published evidence for the diagnostic performance of this test appears to be limited, but one study found that the diagnostic sensitivity and specificity for clonidine suppression were 96% and 100%, respectively, using measurement of plasma normetanephrine before and after clonidine administration. Sensitivity was only 67% if measurement of plasma norepinephrine was used. 17 While these data suggest that the clonidine suppression test could perform well as a test to investigate equivocal UMET/PMET results, limited application of the test in practice could be due to factors such as the lack of comprehensive studies and that the test is relatively cumbersome. A number of commonly prescribed medications are reported to interfere with clonidine suppression30,31 and the patient is required to stay in a supine posture throughout the 3 h between baseline and post-clonidine plasma samples.
Methodology used for biochemical tests for PPGL
All laboratories performing UMET, PMET, UCAT, PCAT or VMA in-house use either LC with electrochemical detection or LC-MS/MS, in concordance with recommendations in the Endocrine Society Guidelines 19 (Table 1). There is considerable evidence that measurement of UMET/PMET using these techniques performs significantly better than spectrophotometric or immunoassay methods in terms of both analytical and diagnostic performance.6,10,15 Laboratories should continue to use LC or LC-MS/MS methods to maintain satisfactory performance.
The survey found that not all laboratories are measuring the same panel of analytes in their UMET methods. Although the majority of laboratories included 3MT in the UMET panel, 29.6% (8/27) did not. A minority of PPGL tumours only secrete excessive amounts of dopamine and not epinephrine or norepinephrine. This represents a very small proportion of PPGL (perhaps only one or 2%) but this is more common in patients with mutations in certain genes associated with PPGL susceptibility such as SDHB and SDHD. 4 There is a small increase in overall diagnostic sensitivity when 3MT is measured alongside normetanephrine and metanephrines 32 and raised 3MT is also predictive of the presence of metastases. 20 Therefore, inclusion of 3MT should be considered in all UMET and PMET methods.
Urine metanephrines reference ranges
There are marked differences in the reference ranges used for urine normetanephrine and urine metanephrine between some laboratories. In the case of urine normetanephrine, the lowest upper reference range limit applied is 2.88 μmol/24 h and the highest is 5.3 μmol/24 h. This is an almost 2-fold difference. The variation for urine metanephrine is on a similar scale with the upper limit of the reference range varying between 1.2 and 2.0 μmol/24 h. It seems unlikely that these reference ranges reflect genuine differences in the systematic bias between different assays. The major cause of differences in UMET reference ranges is likely to be the use of different sources for the reference data. Responses to the survey indicate that some laboratories have reference ranges based on in-house data, some on data provided by LC kit manufacturers and others on data in the literature. The differences in reference ranges between centres could lead to significant variation in the diagnostic performance of the test. If UMET reference ranges are inappropriately high, diagnostic sensitivity may be impaired leading to an increased false negative rate. On the other hand, if UMET reference ranges are inappropriately low diagnostic specificity could be impaired leading to an increased number of false positives requiring further investigation. While it may not be appropriate to establish a common reference range for all urine metanephrines methods, the degree of variation discovered seems unjustified. A recently published international study that investigated the degree of harmonisation between laboratories measuring PMET found that while there was good analytical agreement between methods (at least for normetanephrine and metanephrine), inter-laboratory agreement in interpretation was improved when harmonised reference intervals were applied. 33 A similar exercise for UMET methods could be useful in addressing the significant amount of variation found in UMET reference ranges used in the United Kingdom.
Plasma metanephrines reference ranges and patient preparation
A number of studies have been published that demonstrate the potential impact of patient preparation and posture on plasma metanephrines concentrations. Plasma samples taken from patients in a supine position, after a period of supine rest have lower metanephrines concentrations (particularly normetanephrine) on average compared to samples taken in a seated position. 16 This is due to the increased background activation of the sympathetic nervous system when seated compared to supine. This means that in order to achieve optimal diagnostic performance, reference ranges based on subjects in a supine position rather than seated are required when interpreting results from supine patients (supine ranges for normetanephrine are significantly lower than seated). There is evidence that diagnostic performance is improved when samples are taken in the supine position (and a supine reference range is used) compared to seated.16,34 It is thought that diagnostic sensitivity is improved with supine sampling due to a smaller proportion of the measured plasma metanephrines originating from background production by the sympathetic nervous system, making it easier to distinguish subtle changes in metanephrines secretion observed for smaller PPGL tumours. Diagnostic specificity may also be improved for supine sampling, again due to decreased background activation of the sympathetic nervous system and fewer false positive results. The Endocrine Society Guidelines recommend that patients have samples for PMET taken in a supine posture with the results interpreted using a supine reference range. Collecting samples for PMET in a supine posture and using supine reference ranges may be particularly important when testing for PPGL in populations with a high prevalence such as those undergoing surveillance for a susceptibility syndrome or following a previous PPGL diagnosis. 35
The majority of labs surveyed that perform PMET analysis (4 of 5 that responded with details) provide both seated and supine ranges, which likely reflects recognition that supine sampling with supine reference ranges is best practice but that in many NHS hospitals, seated sampling is widely used. While some authors have argued that lower supine reference ranges should be applied even where seated sampling is used to maintain optimal diagnostic sensitivity, 16 using a lower supine diagnostic cut-off for seated patients inevitably impairs diagnostic specificity.16,36 Many NHS departments requesting PMET will face difficulties implementing supine sampling for all of their patients 37 (70% of centres responding to this survey used seated sampling at least some of the time), so requesting clinicians and laboratories providing PMET analyses should have a clear understanding of the issues associated with seated versus supine sampling.
Sample requirements
There is significant variation in practice between laboratories in the requirement for urine collections for UMET/UCAT to be acidified. There is evidence that UCAT are more stable in acidified urine38,39 but the advice is mixed in the case of UMET. Many of the laboratories surveyed request acidified urine for UMET analysis but there is some evidence that metanephrines are sufficiently stable in urine without preservative. Two studies have found that metanephrines were stable in urine for at least a week, while catecholamines degraded significantly in some samples.38,40 The requirement for acidified urine samples for UMET is a matter that laboratories may wish to review given the patient safety issues and laboratory staff time spent preparing acidified urine collection containers. The survey also found that 10 laboratories accept either overnight or random urine samples for UMET, at least in certain circumstances. The Endocrine Society Guideline states that a 24-h urine collection is preferred, although there is a some evidence that overnight or random UMET may perform well as a diagnostic test compared to 24-h collection41–43
While all centres using PMET requested EDTA plasma, the acceptance criteria were quite variable with the maximum permissible delay between sample collection and centrifugation varying between 15 min and 6 h. Having a defined policy in place for the acceptance of PMET samples is important as published data show that while PMET may be stable pre-centrifugation for up to 3 h when kept at 4°C, 44 concentrations change quite rapidly at room temperature with concentrations potentially initially increasing (due to residual activity of the enzyme catechol-O-methyltransferase) and then undergoing degradation.44,45 Post-centrifugation, metanephrines are not stable at room temperature with significant degradation observed within 24 h. Plasma kept at 4°C appears to be stable for up to 3 days without significant degradation and frozen plasma is stable for at least 1 month. 45 The wide variation in sample requirements and acceptance criteria for UMET and PMET should be addressed. Evidence-based national guidance on this issue could be useful to laboratories providing these tests.
Provision of interpretative/clinical advice
Advice on medications affecting the interpretation of metanephrines/catecholamines results is available from 62% of responding laboratories and 28% had departmental or Trust protocols/guidance on the biochemical investigation of PPGL. As there is a wide range of medications that can affect metanephrines and catecholamines either due to the pharmacological actions of the medications17,46 or analytical interference47–49 it is important that users of these tests have access to advice on the impact of these medications in order to avoid potential misinterpretation. The availability of advice on interpretation and suitable testing protocols is also important due to the complexities involved in interpreting these tests such as responding to the common problem of borderline or equivocal results and taking into account the pre-test probability of a diagnosis of PPGL in different groups of patients. The reported proportion of centres with local departmental or clinical protocols/guidelines seems relatively low. This may partly be due to some laboratories being unaware of protocols developed by other departments in their centre.
Summary
This audit has provided a useful insight into current practice in the United Kingdom with regard to the laboratory investigation of PPGL. A cautionary note is that not all centres in the United Kingdom responded with 58 centres submitting a response. There is a possibility that the survey responses received are biased towards centres that have an active interest in testing for PPGL given that completing the questionnaire required a significant amount of work. However, the responding laboratories were based in a mixture of types of hospital with around half coming from District General Hospitals and half from Teaching Hospitals.
The results presented here have highlighted some areas of good practice, as assessed against the Endocrine Society Clinical Practice Guideline (Table 2): 1. The majority of responding laboratories (95%) include either UMET or PMET in initial biochemical testing. 2. All laboratories measuring UMET/PMET used the recommended methodology (LC-ECD or LC-MS/MS). Summary of concordance of UK practice with Endocrine Society Guideline [20] recommendations relevant to biochemical testing.
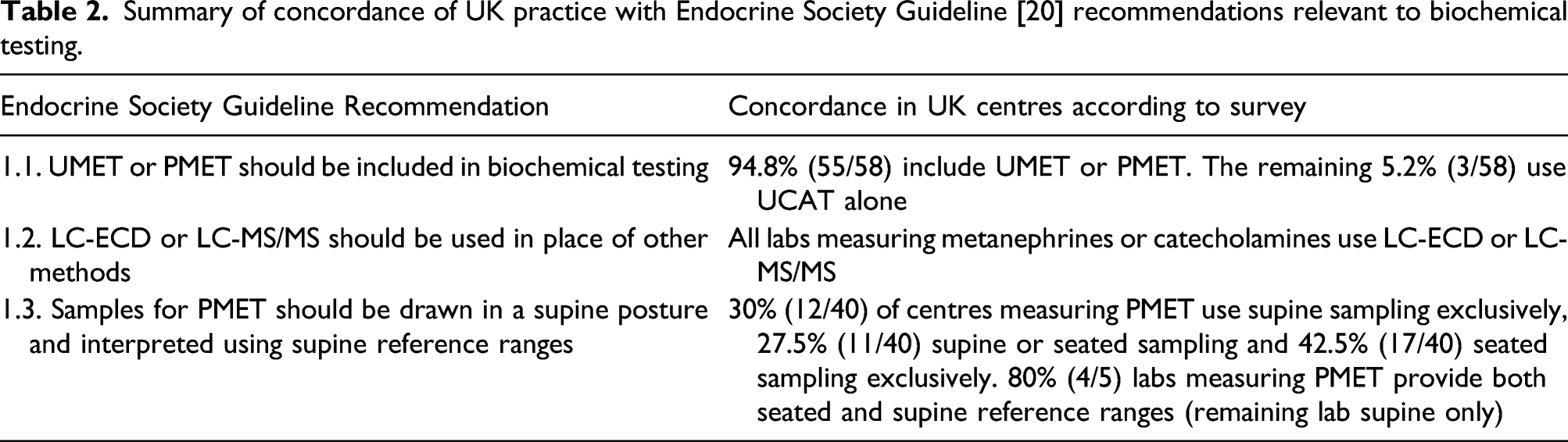
There are also areas where there is variation in practice and further review may be beneficial: 1. There are a small number of centres (5%) using UCAT alone as a first-line test for PPGL. Laboratories using UCAT or PCAT as their first-line diagnostic tests for PPGL should consider adopting UMET or PMET as these tests offer significantly better diagnostic performance. 2. A significant number of the centres including UMET or PMET as tests for PPGL also include UCAT/PCAT/VMA. While the Endocrine Society Guidelines do not expressly recommend against using these additional tests it is not clear that including these tests offering poorer diagnostic performance can improve testing protocols. Laboratories offering a range of tests in addition to UMET/PMET should consider whether this improves the diagnostic performance of testing protocols for PPGL and whether this is a cost-effective approach. 3. There is significant variation in UMET reference ranges. Further investigation and review of the reference ranges in use may help to determine if they are fit for purpose and if there is scope for harmonisation. 4. In the case of PMET, most laboratories performing the test offered both supine and seated reference ranges and there is variation in patient posture during sampling in different centres. While adopting the best practice of sampling in a supine position should be implemented where possible, this may be limited by the facilities available in NHS out-patient departments. 5. The utility of CgA and the clonidine suppression test in the diagnosis of PPGL is not clear as the evidence available is lacking. Further research may be useful.
Supplemental Material
sj-pdf-1-acb-10.1177_00045632211046759 – Supplemental Material for A UK national audit of the laboratory investigation of phaeochromocytoma and paraganglioma
Supplemental Material, sj-pdf-1-acb-10.1177_00045632211046759 for A UK national audit of the laboratory investigation of phaeochromocytoma and paraganglioma by Christopher Boot, Barry Toole, Sharman Harris, Lisa Tetlow and Wassif S Wassif in Annals of Clinical Biochemistry
Supplemental Material
sj-pdf-2-acb-10.1177_00045632211046759 – Supplemental Material for A UK national audit of the laboratory investigation of phaeochromocytoma and paraganglioma
Supplemental Material, sj-pdf-2-acb-10.1177_00045632211046759 for A UK national audit of the laboratory investigation of phaeochromocytoma and paraganglioma by Christopher Boot, Barry Toole, Sharman Harris, Lisa Tetlow and Wassif S Wassif in Annals of Clinical Biochemistry
Footnotes
Acknowledgements
We would like to thank Mike Lester at the ACB office for his assistance in distributing the survey, the Association of Clinical Biochemistry National Audit Group (particularly Louise Ward) for their help in designing the survey and all of the laboratories that submitted a response.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Guarantor
CB
Contributorship
CB, BT, SH and LT designed the survey with input from the ACB National Audit Group (WW). CB collated the survey responses and drafted the article. All authors had input into the content of the final article.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
